Abstract
Background and Objectives
Fingolimod, an oral therapy for MS, decreases expression of membrane S1P1 receptors on CD4+ memory cells, causing their retention and deactivation in lymph nodes. We determined fingolimod effects on the number and proportion of potentially CNS-damaging CD8+CD28+ cytolytic T lymphocyte cells (CTLs) and on MS-depleted and dysfunctional CD8+CD28− anti-inflammatory suppressor/regulatory T cells (Treg) and on CD8+ T-cell expression of the CD69 activation/lymph node retention protein in MS.
Methods
CD8, CD28, CD4, and CD69 expression on peripheral blood mononuclear cells was measured with flow cytometry. In vitro concanavalin A (ConA) activation of T cells, including CD8+CD28− cells, was used to mimic inflammation.
Results
Fifty-nine patients with MS, 35 therapy-naive (16 clinically stable; 19 exacerbating) and 24 fingolimod-treated (19 clinically stable; 5 exacerbating), and 26 matched healthy controls (HCs) were compared. In therapy-naive patients, the CD8+ Treg percent of total lymphocytes was only 1/4 of HC levels. In fingolimod-treated patients, however, CD8+ Treg percentages rose to 2.5-fold higher than in HC and 10-fold higher than in therapy-naive MS. With fingolimod therapy, in contrast, CD8+ CTL levels were less than half of levels in HCs and therapy-naive patients. In HCs and all MS, activation with ConA strongly induced CD69 expression on CD4+ cells and induced 3-fold higher CD69 levels on CD8+ CTL than on CD8+ Treg. Fingolimod and analogs in vitro did not modify lymphocyte CD69 expression. Lower levels of CD69 on CD8+ Treg than on CTL may allow easier Treg egress from lymph nodes and enhance control of peripheral inflammation. In vitro activation reduced the already low CD8+ Treg population in therapy-naive MS, but only slightly altered Treg levels in fingolimod-treated MS.
Discussion
Fingolimod therapy markedly increases the percentage of CD8+ Treg in MS, reversing the low CD8+ Treg:CTL ratio seen in untreated MS. The increase in immune regulatory cells has potential therapeutic benefit in MS. Activation in vitro depletes CD8+CD28+CTL in patients with MS; the loss is more pronounced in older patients with MS. This suggests that inflammation can disrupt the tenuous immune regulation in MS, especially in older patients.
MS is a demyelinating disease of the CNS, with immune dysregulation in peripheral blood mononuclear cells (PBMC).1 In healthy controls (HCs), CD8+ regulatory T cells (Treg) are 10% of lymphocytes but in autoimmune disorders, CD8 Treg are reduced in number and also have weak anti-inflammatory function, leading to immune dysregulation. These autoimmune disorders include murine models of inflammatory bowel disease and MS (experimental autoimmune encephalomyelitis)2,3 and human systemic lupus erythematosus and MS itself.4-6
The CD28 costimulatory molecule can differentiate CD8+ cytolytic T lymphocyte (CTL) from CD8+ Treg. In total, 50% of human CD8+ T cells are antiviral/antitumor CD8+CD28+ CTL and 50% are immunoregulatory CD8+CD28− cells (Treg, T suppressor cells).7,8 Human CD8+ Treg have powerful immune suppressive function. In HCs, suppression of proliferation by CD8+ cells is 2.2-fold more potent than by CD4+ cells and is 2.4-fold more potent than suppression by enriched CD4+CD25+ Treg.6,9
CD8+ Treg numbers are subnormal in relapsing and progressing patients with MS.5,6,10-13 By contrast, CD8+ CTL numbers are normal in MS.9,10,12 In parallel, CNS lesions of MS contain few CD8+ Treg but high numbers of potentially destructive CD8+ CTL.14,15 CD8+ Treg numbers in vivo, and their function in vitro, decrease in therapy-naive MS during exacerbations and progression, but CD8+ Treg numbers and function rebound above normal during remission.5,16
Decreased function of CD8+CD28− cells parallels their decreased numbers in MS. We and others have shown reduced function of peripheral CD8+CD28− Treg in patients with MS compared with HCs, based on concanavalin A (ConA)–activated and CNS antigen-specific suppressor assays.12,17,18
Fingolimod (FTY720) is a potent immunotherapy for relapsing forms of MS. It prevents migration of educated and activated lymphocytes out of lymph nodes and other immune organs to target in the periphery. Lymphocytes express sphingosine 1-phosphate receptors (S1PR), guiding them to traffic along S1P1 gradients from lymph node to target tissues. During FTY720 therapy, sphingosine kinase phosphorylates fingolimod. FTY-phosphate (FTY-P) then downmodulates S1P1R on lymphocytes and prevents 75% of lymphocytes from leaving the thymus, spleen, and lymph nodes.19,20 Fingolimod therapy reduces peripheral CD4+ central memory and naive T cells and total CD4+ cells by 60%–88%.21,22 Reduced thymic emigrants and lymphopenia precipitate peripheral expansion of CD4+ Treg. These Treg have less rigorous tolerance of self-antigens than Treg generated in the thymus. CD8+ cells are less affected. FTY-P inhibits migration of only 45% of CD8+ cells, increasing the CD8/CD4 T-cell ratio in blood. Inflammation and type I interferons induce CD69, a second immune cell retention protein.23 CD69 on monocytes allows these inflammatory cells to secrete S1P in lymph nodes. Secreted S1P reduces S1P1 expression on T cells and inhibits their chemotaxis out of lymph nodes.20,23-25 Effects of fingolimod on CD8+ regulatory cells and CD69 expression are not well-described.
We investigated the effects of fingolimod therapy on circulating numbers of CD8+CD28− Treg and CD8+CD28+ CTL, and expression of CD69 on these CD8+ subpopulations, through multiparameter flow cytometry. We also determined the effects of fingolimod on ConA-activated CD4+ and CD8+ T cells from HCs and stable, exacerbating, and therapy-naive patients with MS.
Methods
Study Population
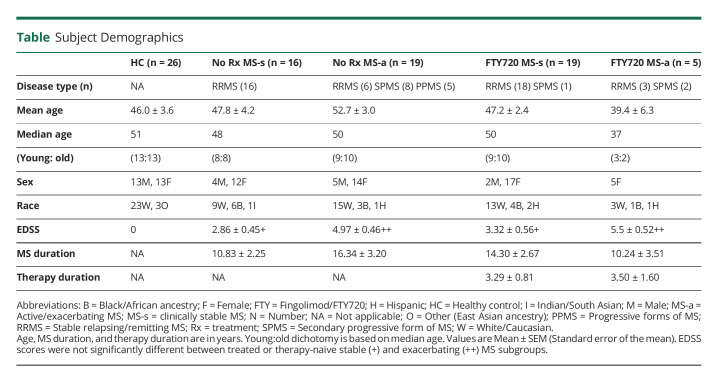
Fifty-nine patients with MS included 35 therapy-naive (16 clinically stable; 19 exacerbating) and 24 fingolimod-treated (19 clinically stable; 5 exacerbating), who were examined at the 6-month intervals in the University of Chicago Neurology Comprehensive MS Center (Table 1). The low number of treated patients with exacerbations is due to the clinical effect of daily 0.5 mg oral fingolimod—on therapy, attacks are 54% fewer and less severe.26 HCs were age and sex-matched.
Table.
Subject Demographics
Standard Protocol Approvals, Registrations, and Patient Consents
All subjects signed University of Chicago IRB-approved informed consents.
In Vitro Treatment of PBMC
Blood was drawn in the morning and then rapidly processed. PBMCs were isolated on density gradients (Lympholyte, Cerdarlane, Burlington, NC).27 For activation, PBMCs were incubated with 5 ug/mL ConA in AIM-V serum-free media for 0, 20, and 48 hours in the presence of 100 ng/mL of fingolimod, FTY-phosphate (FTY-P, S1P expression antagonist), and FTY AAL-R (2-amino-4-(4-heptyloxyphenyl)-2-methylbutanol, a S1P agonist with rapid phosphorylation and activation; positive control for FTY720, Novartis, Cambridge, MA). These concentrations were not toxic to lymphocytes. Serum-free media was used to model lymph node conditions with low S1P concentrations. Serum contains S1P, which negates the effect of in vitro fingolimod (V Brinkmann, personal communication, September 20, 2010). ConA was used to activate lymphocytes, including the CD8+CD28− subset. Anti-CD3 and anti-CD28 antibodies are commonly used to activate lymphocytes but provide only partial stimulation of the important CD8+CD28− cell subpopulation. In preliminary experiments, activation with PMA/ionomycin generated similar but more variable results than ConA and therefore was not used.
The bioactivity of fingolimod and the AAL-R analog was analyzed in Sprague-Dawley rats (250–300 g). Both compounds dose-dependently decreased immune cells in rat blood (eFigure 1, links.lww.com/NXI/A787). Lower doses of AAL-R and fingolimod (0.1 mg/kg) decreased total white blood cells (59% and 31%, respectively) and lymphocyte counts (76% and 33%), with recovery after day 1 (eFigure 1, A and B). Higher doses of AAL-R (0.3 mg/kg) and fingolimod (0.5 mg/kg) caused rapid drops in total white blood cells (60% and 77%, respectively) and lymphocyte counts (82% and 85%) that were sustained below baseline levels through day 7 (eFigure 1, C and D).
Flow Cytometry
PBMCs were FcR-blocked with 10% normal goat serum (Life Technologies, Gaithersburg, MD), then stained with antibodies CD4-APC-H7 (clone RPA-T4), CD8-APC-H7 (SK1), CD28-APC (CD28.2, all BD Biosciences, Franklin Lakes, NJ), and CD56-PE-Cy7 (HCD56, BioLegend, San Diego, CA) for lineage analysis, and CD69-PerCP-Cy5.5 (clone FN50, BD Biosciences), an activation marker. Cell death was measured with trypan blue exclusion and apoptosis with Apotracker (BioLegend).
Lymphocytes were gated using FSC and SSC using LSRFortessa 4–15 or 4–15 HTS flow cytometers (BD Biosciences) (eFigure 2, links.lww.com/NXI/A787). A total of 10,000 lymphocytes were run for each condition, so absolute numbers parallel percentages within the lymphocyte population. Lymphocyte subset frequency, median fluorescence intensity (MFI), and multiparameter compensation values were calculated with FlowJo software.28 Clinical laboratory testing quantitated total white blood cells and absolute lymphocyte count, which paralleled flow cytometry classified lymphocytes.
Statistical Analysis
Differences between groups were measured with appropriate paired and unpaired 2-tailed t tests. CD8+CD28− Treg, CD8+CD28+ CTL, and CD4+ subsets were expressed as percent of 10,000 total lymphocytes in each condition, given as mean ± SEM. Surface markers and immunologic responses did not differ between SPMS, PPMS, and RRMS; these groups were combined, and MS groups were defined as clinically stable or exacerbating.
Age was dichotomized by defining values below the median age of each group as young and values above the median age as old (Table 1). Average Extended Disability Scale Scores (EDSS) of older patients were 1–1.5 points higher than younger in therapy-naive and in fingolimod-treated conditions.
Data Availability
Anonymized data, per reasonable request, will be shared with qualified investigators by the corresponding author.
Results
Fingolimod Therapy Elevates CD8+CD28− Treg Numbers and Decreases CD8+CD28+ CTL in Peripheral Blood
CD8+CD28− Treg percentages in HCs were 8.30 ± 1.95% (percent of total lymphocytes) but in therapy-naive stable patients with MS were only 3.68 ± 0.87% (p < 0.05 vs HC) and were even lower during MS exacerbations (1.47 ± 0.35%, p < 0.01 vs HC) (Figure 1A). Fingolimod treatment in stable MS elevated Treg (17.69 ± 3.10%) to above the percentage in HCs (p < 0.01) or in therapy-naive stable MS (p < 0.0003) and maintained the increase in exacerbating MS (6.52 ± 1.3%). The percent of Treg in the lymphocyte population increased 5-fold (p < 0.002) in fingolimod-treated stable MS and 4-fold (p < 0.00004) in fingolimod-treated exacerbating MS compared with therapy-naive MS. Absolute counts, based on peripheral blood differential counts, remove the effect of the therapy-induced drop in CD4+ cells and CD8+CD28+ CTL. In a smaller cohort, the increase in absolute Treg counts within blood lymphocytes of fingolimod-treated stable patients was not statistically significant (Figure 2A). CD8+CD28− Treg in therapy-naive stable MS were 60 cells/µL of blood and in fingolimod-treated stable MS were 83 cells/µL; CD8+CD28− Treg in therapy-naive active MS were 58 cells/µL and in fingolimod-treated active MS with 25 cells/µL (both comparisons NS). The relative change in Treg compared with CD8+CD28+ CTL, and Treg to CTL ratios, were therefore investigated.
Figure 1. Fingolimod Therapy Elevates Subnormal CD8+CD28− Treg in Therapy-Naive MS to Above HC Levels.
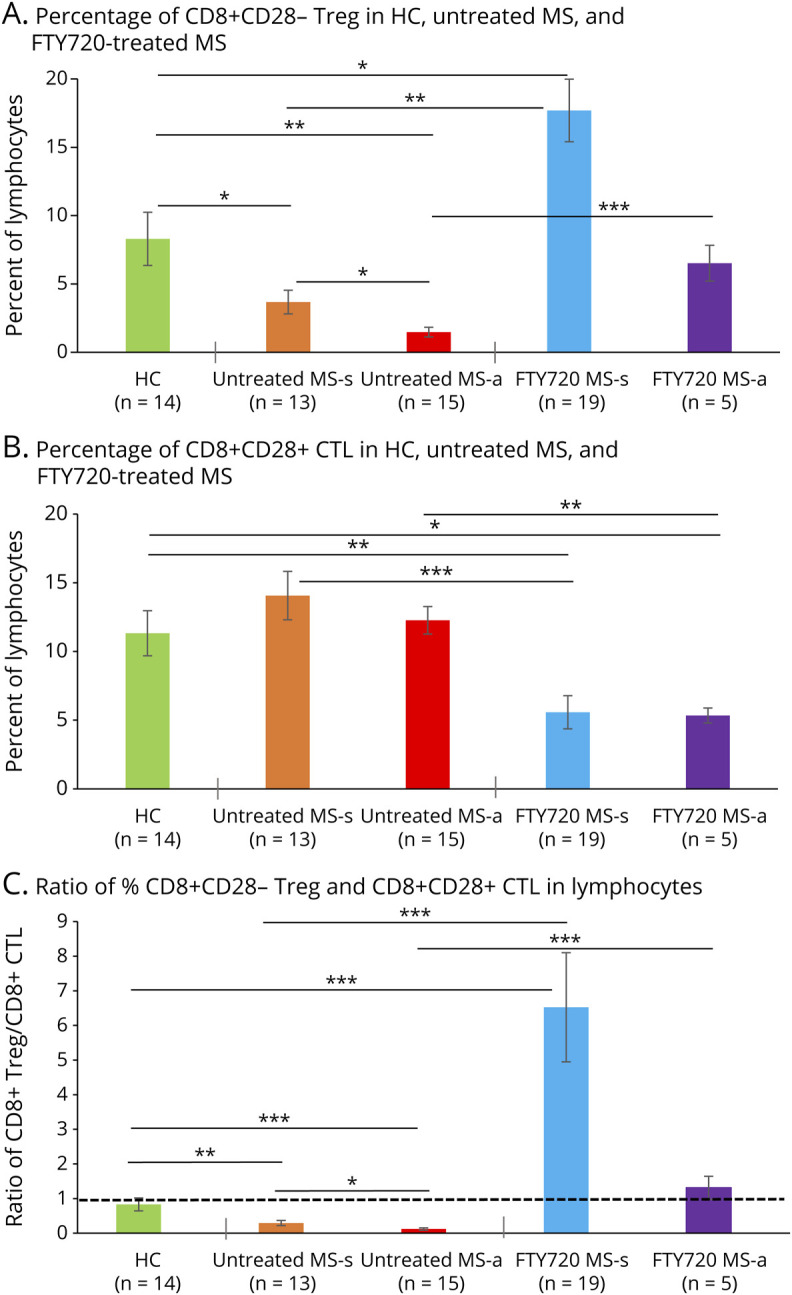
(A) CD8+CD28− Treg as percent of lymphocytes were lower in therapy-naive stable patients with MS (3.68 ± 0.87%: p = 0.04) than in HCs (8.30 ± 1.95%) and even lower in therapy-naive exacerbating MS (1.47 ± 0.35%: p = 0.001 vs HC). Fingolimod therapy increased Treg to supranormal levels in stable MS (17.69 ± 3.10%: p = 0.002 vs therapy-naive stable) and maintained Treg at normal levels during attacks (6.52 ± 1.30%: p = 0.00004). (B) CD8+CD28+ CTL as percent of lymphocytes was greater in therapy-naive stable MS (14.06 ± 1.76%) than in fingolimod-treated stable patients (5.57 ± 1.20%: p = 0.0003). Similarly, CTLs were 12.27 ± 1.00% in therapy-naive exacerbating patients and only 5.34 ± 0.54% in fingolimod-treated exacerbating patients (p = 0.001). CTLs in HCs (11.33 ± 1.64%) were within the range of the therapy-naive stable and active MS groups. (C) Compared with HCs, the CD8+ Treg:CTL ratio was low in therapy-naive stable MS (ratio = 0.29; 0.35x of HC = 0.83) and in exacerbating patients (ratio = 0.12, 0.16x of HC). The subnormal CD8+ Treg:CTL ratio was dramatically reversed by fingolimod in stable MS (ratio = 6.52, 7.86x of HC) and in exacerbating MS (ratio = 1.31, 1.58x of HC). Flow cytometry of lymphocyte subsets. * p ≤ 0.05, ** p ≤ 0.01, *** p ≤ 0.001. Abbreviations: CTL = Cytolytic T lymphocyte; HC = healthy control; Treg = regulatory T cell.
Figure 2. Fingolimod Therapy Decreases the Absolute Number of CD8+CD28+ CTL but Increases CD8+CD28− Treg In Vivo.
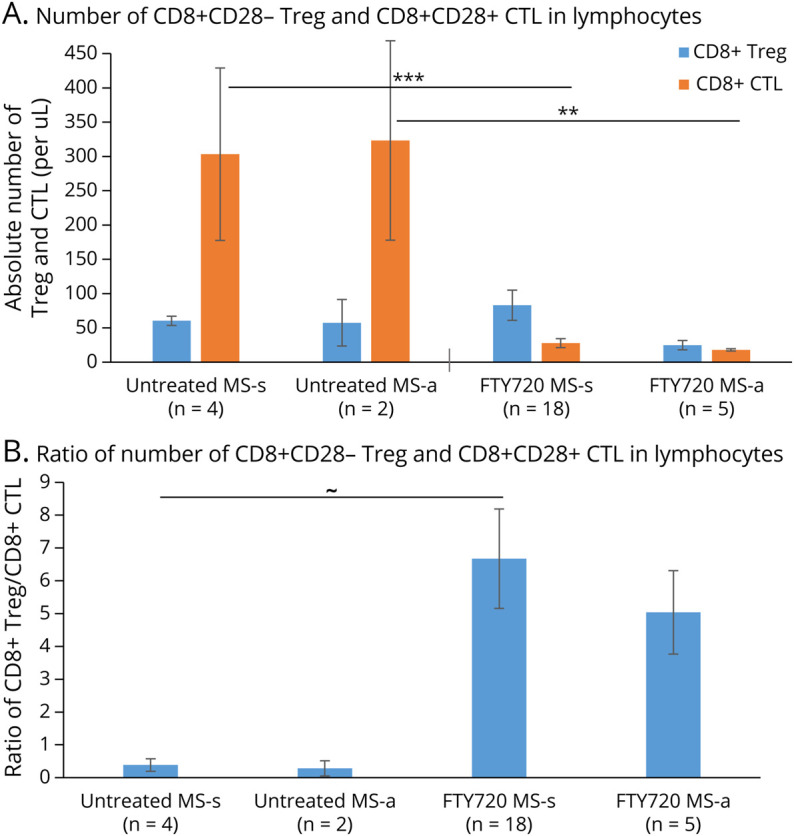
(A) Absolute Treg numbers were 60 cells per microliter (µL) of blood in therapy-naive stable patients with MS (MS-s) and 83 cells/uL in stable fingolimod-treated patients (NS), but fell to 25 cells/µL with exacerbations (MS-a). Absolute CD8+ CTL counts decreased from 303 cells/µL blood in therapy-naive stable MS to 28 cells/µL in fingolimod-treated stable patients (p = 0.00005) and from 323 cells/uL in therapy-naive exacerbating MS to 18 cells/uL in fingolimod-treated exacerbating patients (p = 0.01). Absolute numbers were obtained by measuring the Treg:CTL as a % of the lymphocytes from flow cytometry and multiplying this percent by the absolute lymphocyte counts obtained from clinic visits. (B) The ratio of CD8+ Treg to CD8+ CTL is low in therapy-naive MS (therapy-naive stable MS = 0.4 and therapy-naive active MS = 0.3) but is increased 16.8-fold in fingolimod-treated stable patients and 16.7-fold in fingolimod-treated active patients. ∼ = 0.06, * p ≤ 0.05, ** p ≤ 0.01, *** p ≤ 0.001. Abbreviations: CTL = Cytolytic T lymphocyte; Treg = regulatory T cell.
CD8+CD28+ CTL percentages in blood were higher in stable therapy-naive MS (14.06 ± 1.76%, percent of lymphocytes) than in stable fingolimod-treated patients (5.57 ± 1.20%, p < 0.0003) (Figure 1B). During exacerbations, these CTLs were 12.27 ± 1.00% in therapy-naive MS, but fell to 5.34 ± 0.54% in fingolimod-treated patients (p < 0.001). The CTL percentage differed between HCs (11.3 ± 1.64%) and fingolimod-treated MS groups (stable MS, 5.57 ± 1.20%, p < 0.0003; exacerbating MS, 5.34 ± 0.54%, p = 0.05). The absolute CD8+CD28+ CTL number also markedly decreased with fingolimod therapy (Figure 2A). Therapy-naive stable MS (303 cells/µL of blood) had more CTL than fingolimod-treated stable MS (28 cells/µL; p < 0.00005), as did therapy-naive exacerbating MS (323 cells/µL) compared with fingolimod-treated exacerbating MS (18 cells/µL, p < 0.01).
The ratio of CD8 regulatory to cytolytic cells in blood affects inflammation in MS.5,6,10-13 The CD8+CD28−/+ Treg:CTL ratio was low in therapy-naive stable MS (ratio = 0.29; 0.35x of the level in HCs, where the ratio was 0.83) (Figure 1C) and in exacerbating patients compared with HCs (ratio = 0.12, 0.16x of HC). Fingolimod therapy in therapy-naive stable MS dramatically reversed the subnormal Treg:CTL ratio (6.52, 7.86x of HC) and in exacerbating MS (1.31, 1.58x of HC). The Treg:CTL ratio derived from absolute counts followed the same pattern (Figure 2B). The increased Treg:CTL ratio with fingolimod therapy mirrors the lesser sequestration of CD4+CD25+ Treg than Th1 cells in mice receiving fingolimod.19,20 Thus, the CD8+ CD28−/+ Treg:CTL ratio, which may reflect the balance of immune control vs CNS destruction in MS, is low in therapy-naive stable MS and even lower during exacerbations. During fingolimod treatment, the CD8+ CD28−/+ Treg:CTL ratio rises to supranormal, with CD8+CD28− Treg dominating the CD8+ population.
ConA Activation Increases CD69 Expression on CD4+ T Cells and on CD8+CD28+ CTL More Than on CD8+CD28− Treg in HCs and Therapy-Naive RRMS
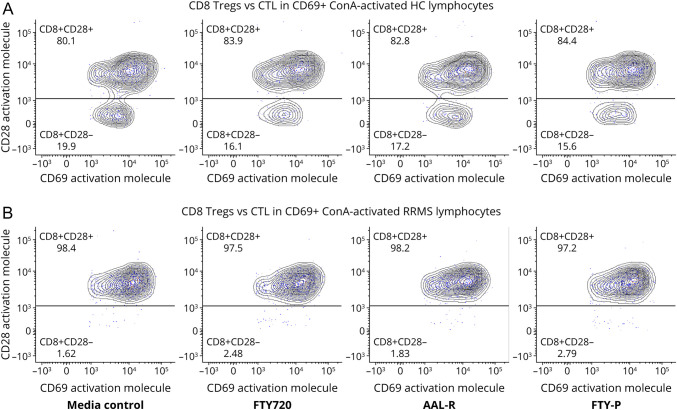
As a model for inflammation, PBMCs were activated with ConA, which targets not only CD28+ cells but also CD28− cells. ConA activates CD8+ Treg, unlike commonly used anti-CD3 plus anti-CD28 stimulation, which cannot maximally activate CD28− cells. In resting MNC, only 0.02–4% of CD8+ cells were CD69+ and CD69 expression was low (eFigure 3, links.lww.com/NXI/A787, unstimulated (A) compared with activated (B) and (C) and (D) compared with (E) and (F)). On ConA activation, approximately 60% of CD4+ cells and 10–15% of CD8+ cells become CD69+ in RRMS and in HCs. The percentage of CD4+CD69+ cells was 15- to 30-fold greater than CD4+CD69− cells, and the percentage of CD8+CD69+ cells was 2- to 10-fold greater than CD8+CD69− cells. The MFI of membrane CD69 expression was 4–5 times more on CD8+ CTL than on CD8+ Treg (Figure 3). Addition of the S1P1 modulators, fingolimod, AAL-R, and FTY-P to resting cells ± ConA did not affect the number of CD69+ cells, CD69 expression, or the CD8+CD28−/+ Treg:CTL ratio in HCs or therapy-naive stable or exacerbating MS (Figure 3). Activation-induced expression of CD69 on cultured lymphocytes, with CD69 on CD4+ cells > CD8+ CTL > CD8+ Treg, paralleled fingolimod's reduction of these circulating subsets.21,22
Figure 3. ConA Activation Increases CD69 Expression on CD8+CD28+ CTL More Than on CD8+CD28− Treg in HCs and Therapy-Naive RRMS.
(A) After ConA activation of HC PBMCs, the median fluorescence intensity of CD69 expression on CD8+CD28+ CTL was 4–5-fold more than on CD8+CD28− Treg, across all conditions: media control, fingolimod, the FTY720 agonists, AAL-R, and FTY-P. (B) After ConA activation of therapy-naive, exacerbating RRMS (MS-a) PBMCs, CD69 expression on CTL and Treg in across all conditions was similar to activated HC cells. However, ConA activation decreased the number of CD8+CD28− Treg in MS compared with HCs. In unstimulated lymphocytes, CD69 expression on CD8+CD28 ± cells was minimal across all conditions and groups (0.2–4%; data not shown). Representative figure of 17 experiments with stable and active MS, see Supplemental eFigure 2 (links.lww.com/NXI/A787) for gating procedure. All CD28 ± cells are CD8+ and CD69+. Abbreviations: ConA = concanavalin A mitogen; CTL = Cytolytic T lymphocyte; HC = healthy control; PBMC = peripheral blood mononuclear cell; FTY-p = FTY-phosphate; Treg = regulatory T cell.
Activated CD8+CD69+ cells were largely CD8+CD28+ CTL, especially in therapy-naive, exacerbating patients with MS (97.5%) compared with HCs (83%) (Figure 3). These exacerbating patients had many fewer CD8+CD28− Treg (2.5%) than HCs (17%) across all in vitro FTY720 conditions. We therefore investigated the effect of in vitro activation on CD8 Treg numbers.
In Vitro Immune Activation Depletes CD8+CD28− Treg
In unstimulated HC PBMCs, the percentage of CD8+CD28− Treg (9.12 ± 2.19%, as percent of total lymphocytes in media control) was more numerous than in therapy-naive stable MS (2.89 ± 0.98%, p < 0.04) or in exacerbating MS (1.25 ± 0.17%, p < 0.003) (Figure 4A). By contrast, there were more Treg in unstimulated PBMCs from fingolimod-treated stable patients (17.69 ± 3.10%) than in therapy-naive stable MS (2.89 ± 0.98%, p < 0.01) and more Treg in fingolimod-treated active patients (6.52 ± 1.31%) than in therapy-naive exacerbating MS (1.25 ± 0.17%, p < 0.01) (Figure 1A). Treg in all fingolimod-treated MS (14.78 ± 3.72%) were at least as numerous as in HCs (9.12 ± 2.19%) (Figure 4A). The percentage of CD8+CD28+ CTL was equivalent between HCs (10.16 ± 1.11%) and stable (11.97 ± 1.7%) and active (12.3 ± 1.19%) therapy-naive MS (Figure 4B). However, CTL percentages tended to decrease in fingolimod-treated patients (5.57 ± 2.15%) compared with HCs (p = 0.06).
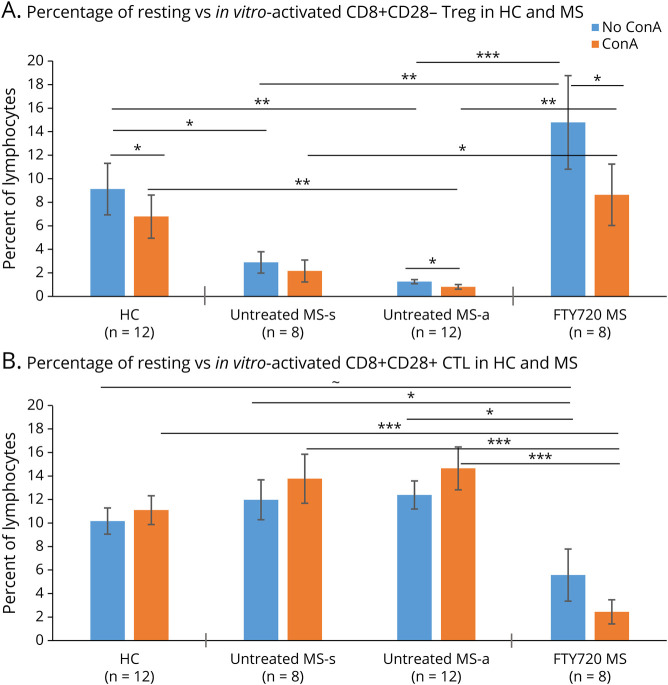
Figure 4. In Vitro Immune Activation Depletes CD8+CD28− Treg.
(A) The percentage of CD8+CD28− Treg in resting PBMCs was lower in therapy-naive patients with MS than in HCs (HC vs stable MS [MS-s], p = 0.04; HC vs exacerbating MS [MS-a], p = 0.003, unpaired t tests). Fingolimod-treated patients had higher Treg levels than therapy-naive MS (MS-s, p = 0.01; MS-a, p = 0.001), equivalent to levels in HCs. ConA activation decreased the percent of Treg in HCs (p = 0.05 vs resting/media controls; paired t tests). Activation tended to reduce the already low Treg population in therapy-naive stable (p < 0.11) and significantly reduced the percentage of Treg in exacerbating MS (p = 0.05). After activation, fingolimod-treated MS Treg levels drop (p = 0.04) to levels of HCs. (B) The percentage of CD8+CD28+ CTL in resting PBMCs tended to be lower in fingolimod-treated patients than in HCs (p = 0.06). ConA activation in fingolimod-treated patients further decreased CTL levels (p = 0.00005 vs activated HCs cells). ConA-activated PBMCs from therapy-naive stable MS and active MS had greater CTL levels than fingolimod-treated patients (p = 0.001 stable and p = 0.00003 active patients). The trend for a fall in CTLs after ConA activation in fingolimod-treated MS was nonsignificant. ∼ = 0.06, * p ≤ 0.05, ** p ≤ 0.01, *** p ≤ 0.001. Abbreviations: ConA = concanavalin A mitogen; CTL = Cytolytic T lymphocyte; HC = healthy control; PBMC = peripheral blood mononuclear cell; Treg = regulatory T cell.
ConA activation of HC lymphocytes slightly decreased the percent of CD8+CD28− Treg by 25.7%, from 9.12 ± 2.19% in unstimulated cells to 6.78 ± 1.84% (p < 0.05) (Figure 4A). Activation in vitro in therapy-naive exacerbating MS reduced the already low Treg population by 35.2%, from 1.25 ± 0.17% to 0.81 ± 0.20% (p = 0.05), and reduced CD8+ Treg in therapy-naive stable MS by 25.3%, from 2.89 ± 0.98% to 2.16 ± 0.93%. Activation of PBMCs from fingolimod-treated patients reduced Treg by 41.9%, from 14.78 ± 3.72% unstimulated down to 8.64 ± 2.29% after ConA (p < 0.05), but above levels in therapy-naive MS (Figure 4A). Activation-induced cell death was due to apoptosis across all groups (APO tracker flow cytometry, DNS).
In vitro activation did not significantly reduce the percentage of CD8+CD28+ CTL (Figure 4B, blue vs orange). The absolute number of CD8+CD28− Treg and CD8+CD28+ CTL paralleled the change in percentages (eFigures 4A and B, links.lww.com/NXI/A787). The increase in Treg and the decline in number of CTL after fingolimod therapy are reflected in the high Treg:CTL ratio (Figure e4C). Thus, the activation in vitro depletes CD8+CD28+ CTL in all patients with MS; the loss is more pronounced in older patients with MS.
Age and Multiple Sclerosis Affect the Increase in CD8+CD28− Treg by Fingolimod Therapy
Aging affects immunity and could affect immune subsets in MS. In healthy older subjects (older than 51 years, based on median age), the percentage of CD8+CD28− Treg in the lymphocyte population was 2.5-fold higher (11.2 ± 2.8%) than in younger HCs (4.47 ± 1.67%, p = 0.08) (Figure 5A). In older HCs, however, the percentage of CD8+CD28+ CTL (9.05 ± 1.06%) was 0.64-fold lower than in younger HCs (14.4 ± 3.3%; p = 0.10) (Figure 5B). Young HCs had a 1:3 CD8+ Treg:CTL ratio. In older HCs, however, the Treg:CTL ratio reversed and rose to 1.2:1. This parallels normal age-dependent increases in CD8+CD28− Treg and decreases in CD8+CD28+ CTL.29,30
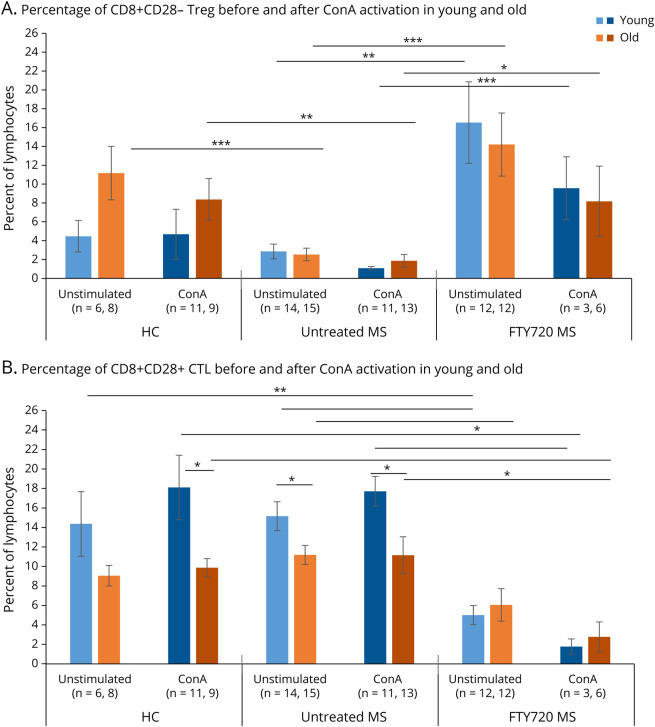
Figure 5. Age and MS Affect the Increase of CD8+CD28− Treg by Fingolimod Therapy.
(A) The percentage of CD8+CD28− Treg in the resting lymphocyte population trended 2-fold higher in healthy older than in younger subjects (p = 0.09). The percentage of Treg in unstimulated older therapy-naive MS was lower than in unstimulated older HCs (p = 0.001) and did not increase with aging. The percentage of Treg in unstimulated fingolimod-treated younger patients was 5.8-fold higher than in unstimulated younger therapy-naive MS (p = 0.005) and 5.6-fold higher in unstimulated fingolimod-treated older patients than in older unstimulated therapy-naive MS (p = 0.001). On activation with ConA, Treg percentages did not significantly change in young and old HCs or in therapy-naive patients and tended to lose some of the Treg increase because of fingolimod therapy, although levels were still greater than in therapy-naive MS (p = 0.0002 for young and p = 0.03 for old). Stable and active patients with MS had similar profiles and were combined for analysis. (B) The percentage of CD8+CD28+ CTL within total lymphocytes in older HCs and in older therapy-naive MS was lower than that of younger subjects and did not change with ConA activation. However, fingolimod therapy decreased the percentage of CTL in unstimulated young (p = 0.00004) and old MS (p = 0.01) and in ConA-activated young (p = 0.02) and old MS (p = 0.01). Hashing indicates ConA activation. * p ≤ 0.05, ** p ≤ 0.01, *** p ≤ 0.001. Abbreviations: ConA = concanavalin A mitogen; CTL = Cytolytic T lymphocyte; HC = healthy control; Treg = regulatory T cell.
In therapy-naive MS, CD8+CD28− Treg did not increase with age, unlike in HCs (young MS, younger than 50 years = 2.85 ± 0.78%; older MS = 2.53 ± 0.67%, NS) (Figure 5A). With fingolimod treatment, however, Treg levels rose markedly in young and in old. Young fingolimod-treated patients (younger than 50 years) had a 5.8-fold increase in Treg compared with therapy-naive MS (16.5 ± 4.3%), and older fingolimod-treated patients had a 5.6-fold increase (14.2 ± 3.3%). Young therapy-naive patients with MS had more CD8+CD28+ CTL (15.2 ± 1.5% of total lymphocytes) than CD8+ Treg (2.85 ± 0.78%, p < 0.01), with a Treg:CTL ratio of 1:5.3 (Figure 5B). The Treg:CTL ratio was 3.3:1 in young and 2.35:1 in older fingolimod-treated patients with MS. Thus, fingolimod increased the low percent of Treg in MS and prevented the age-associated loss of CD8+ Treg otherwise seen in older patients with MS.
Activation had minimal effect on the percentage of CD8+CD28− Treg in HCs, but activation depleted Treg in young and old, therapy-naive, and fingolimod-treated MS. High levels of Treg during fingolimod treatment were halved by in vitro activation compared with unstimulated media controls, but remained higher than in activated therapy-naive MS lymphocytes. After activation with ConA, CD8+CD28+ CTL levels did not change in HCs or therapy-naive MS (Figure 5B). However, during fingolimod therapy, in vitro activation tended to decrease CD8+ CTL in young and old patients, indicating greater susceptibility to inflammation-induced death in CTL than in Treg during fingolimod therapy. The absolute number of Treg and CTL paralleled the change in percentages (eFigure 5, A and B, links.lww.com/NXI/A787). The Treg/CTL ratio rose after in vitro activation and was more pronounced in young than in old fingolimod-treated MS (eFigure 5A).
Discussion
Fingolimod therapy increased the percentage of circulating CD8+CD28− Treg cells from 1/3 of HC levels in therapy-naive stable MS and only 1/6 of HC levels in exacerbating MS to twice normal levels after fingolimod therapy. CD8+CD28− Treg were 50% of CD8+ lymphocytes in HCs (Figures 1 and 4) but were only 17% in therapy-naive stable MS and 15% in active MS (Figure 2B). In fingolimod-treated patients, Treg levels within in CD8+ lymphocytes rose above levels in HCs to 77% in stable MS and to 58% in active MS (Figures 1 and 2). Fingolimod therapy concordantly reduced the percentage of CD8+CD28+ CTL, negating the possibility that all CD8+ subsets rise in relation to the diminished numbers of CD4+ cells.
The increase in the percentage of CD8+CD28− Treg by fingolimod therapy may be linked to clinical benefit in MS. There are 11 classes of approved MS therapies, based on mechanism of action. At least 4 of them increase the number and sometimes the function of CD8+ Treg, including interferon-β,31 glatiramer acetate,32 anti-CD20 therapy,28 and fingolimod (present data). The mechanism for the therapy-induced increase in CD8+ Treg is unknown but could include diminished liberation of CNS antigens that drive inflammation or induction of multiple regulatory pathways.27,29,30,32,33 The mechanisms controlling CD4 and CD8 subset homeostasis during fingolimod therapy await definition.
CD8 Treg in humans are 10-fold more numerous than CD4 Treg. CD8 Treg are more potent than CD4 Treg on a per-cell basis, and defective CD8 Treg function is more subnormal than CD4 Treg function in MS.6,9,12,34 CD8+CD28− Treg cells are more suppressive than the total CD8 population and the enriched CD4 Treg population.8,12 CD8 Treg function assays typically study inhibition of mitogen or myelin antigen-specific activation,5,12,13 response to allogeneic cells, or downregulation of CD80 and CD86 costimulatory/activation molecule expression.12,35 Reducing CD80/CD86 is of potential benefit in MS, where high levels of these molecules on B cells and monocytes could promote inflammation.36
CD8 Treg are very different between mice and men. In mice, CD8+CD28− cells are rare (1–5%), and CD4 Treg have a potent regulatory role.34 In healthy humans, however, CD8+CD28+ CTL and CD8−CD28− Treg are in a 50:50 ratio10 (Figures 1 and 4). Surface markers also differ between species. In mice, CD8 Treg are CD25+ and CD122+ (IL-15R), PD-1+, and Ly49d+ 0.37 In humans, CD8 Treg are CD25+, CD27±, CD28−, CD57+, CD62+, CD127- (IL-7Rα), CCR7+, CD45RA+, CTLA4+, GITR+, ILT3+, and NKG2D+. In man, CD8 Treg markers ranked in functional relevance are CD28 > CD27 = NKG2D > CD45RO.12,13
Surface markers that reflect regulatory function are often similar to markers of immune cell aging or chronic antigen exposure linked to immune senescence, but regulatory function, aging effects, induction of memory cells, and senescence-induced exhaustion are measured in different ways.38 Senescent antigen-induced CD4+CD27−CD28− cells have Th1 and cytolytic function and do not differ between HCs and MS or between different forms of MS.38 CD8 cells after chronic antigen exposure, in contrast, differentiate from CD27+CD28+ to CD27−CD28+ and to CD27−CD28− cells.39 Senescent cells as defined in these assays may differ from MS CD8+CD28− Treg. With multiplex flow cytometry, CD8+CD27−CD28−CD57+ cell levels do not change, but the larger population of CD8+CD28− cells increase (Figures 1 and 2).40 Exquisitely defined immune subsets seem more specific but may not encompass most Treg function.
We and others find that subnormal function of CD8+ Treg in MS parallels low numbers of CD8+ Treg and that there is a decline in CD8+ Treg function and numbers during disease activity (Figure 2).5,10-13,18 This functional deficit is present with mitogen activation and with CNS-specific antigen responses.17,18 On a per-cell basis, the reduction in regulatory function in MS is more pronounced in CD8+CD28− Treg than in CD4+ Treg or in NKreg.6,9,33 CD8+ and CD28− markers were used herein because they reflect an important regulatory defect in MS. However, the function of these subsets has not been rigorously evaluated during fingolimod therapy.
Thymic education optimizes helper and regulatory responses to foreign and self-antigens, and then T cells from the thymus populate the peripheral immune system. Robust thymic emigration declines after childhood but is present at low levels throughout life. Recent thymic emigrants (RTE) containing T-cell receptor excision circles (Trec) markedly diminish in MS, corresponding to a 25–30 years advance in immune age compared with normal.41 To compensate for the decline in RTE, peripheral T cells must expand in the periphery without rigorous thymic immune control, allowing autoimmune excess. MS therapies such as fingolimod that prevent thymic egress, and other therapies that deplete peripheral immune cells, may further modify other aspects of immune regulation in MS.
In lymph nodes, CD69 allows inflammatory monocytes to secrete S1P, which causes internalization and degradation of S1PR in lymphocytes,23 similar to the effect of fingolimod. Reduced S1P1R and enhanced CD69 expression will increase immune cell dwell time in lymph nodes during inflammation.20,21,26 Here, fingolimod and analogues did not modify the expression of CD69 on resting or activated lymphocytes in vitro, suggesting that change in CD69 levels is not a mechanism of action of fingolimod therapy. Nonetheless, after in vitro activation, CD69 was 3-fold higher on CD8+ CTL than on CD8+ Treg in MS and control cells, suggesting that CD69-facilitated lymph node retention would be stronger for antigen-activated CTL than for Treg in vivo. Although other molecules also affect retention, CD69 is a potential target for therapies that could induce lymph node retention of CTL or effector CD4 cells.
To model peripheral immune activation, CD4+ Treg are often activated with antibodies recognizing CD3, a protein paired with the T-cell receptor, plus antibodies to the CD28 costimulatory molecule.42 However, anti-CD28 antibodies do not activate CD8+CD28− Treg. Activation with ConA, a mitogen that binds mannose-containing proteins on immune cells, overcomes this limitation and models inflammation. ConA activation diminished CD8+CD28− Treg in therapy-naive MS compared with HCs, suggesting that these cells are more likely to die during inflammation. During fingolimod therapy, CD8+CD28− Treg were partially resistant to activation-induced cell death and remained 4-fold above levels seen in therapy-naive patients (Figure 5A). We therefore studied the effect of age on cell death.
With increasing age, CD8+CD28+ CTL levels decrease in HCs and in MS (Figure 5 and S5, links.lww.com/NXI/A787), in parallel with weak cell-mediated immunity, waning responses to viruses, and more cancer with aging.43 Aging is also associated with more autoimmune disorders—prevented by Treg,43 which are highly susceptible to activation-induced death in older patients. Healthy older people have fewer CD8+ CTL and more CD8+ Treg than young HCs, and aged HCs and aged therapy-naive MS had similar low levels of CTL (Figure 5). Activation in vitro did not change the number of CTL in young and old HCs or in therapy-naive MS PBMCs. In older fingolimod-treated patients with MS, however, activation reduced the number of CTL. Loss of CTL during fingolimod therapy is linked not only to less CNS damage in MS but also to a slight increase in opportunistic infections in this age group.44
CD8+ Treg levels rise with age in HCs. CD8+CD28− Treg did not increase with age in MS and remained at subnormal numbers compared with HCs. CD8+CD28− cell function is subnormal in MS,5,10-13,18 and aging may further diminishes function because of reduced thymic emigrants in MS.45 In young and old patients with MS patients, activation decreased already low CD8+CD28− Treg by 25% in unstimulated therapy-naive MS PBMCs. By contrast, fingolimod therapy elevated Treg in young and old patients with MS to levels seen in HCs. Activation reduced CD8+CD28− Treg by 40% in fingolimod-treated MS, but already high Treg remained above the levels seen in therapy-naive MS. Thus, during inflammation, levels of Treg fall in therapy-naive MS but with fingolimod therapy potentially therapeutic Treg are maintained during exacerbations.
CD28− cells may be sensitive to damage during inflammation. The CD28 molecule prevents cell death, and CD28-negative cells are more susceptible to activation-induced cell death39,42,46 because CD28 expression on T cells varies between subsets (CD4+CD28+ > CD8+CD28+ CTL > CD8+CD28-Treg). Senescent CD8+CD28− cells have shorter telomeres and shorter lifespan and are more susceptible to activation-induced apoptosis, and higher levels of these cells predict geriatric physical frailty.29,30
Fingolimod therapy elevates the relative number of circulating CD8+CD28− Treg compared with CD8+CD28+ CTL, from subnormal to supranormal levels. These changes in circulating immune cells are not from destruction of lymphocytes but may nonetheless affect peripheral immune regulation. Compared with therapy-naive MS, fingolimod therapy prevents the in vitro activation-induced loss of these Treg. Effects on CD8 cells add to the substantial literature on how S1P modulators affect CD4 cells. Fingolimod reduced CD8+CD28+ CTL, most prominently in older patients, paralleling the age-related increase in rare opportunistic infections, but also possibly linked to its therapeutic benefit in MS.
Acknowledgment
Gratitude for helpful discussions from Jack Antel Montreal Neurologic Institute, Amit Bar-Or, University of Pennsylvania, help from the University of Chicago Flow Cytometry Core, and data analysis performed by Kayla Haile.
Glossary
- B
Black/African ancestry
- ConA
Concanavalin A mitogen
- CTL
Cytolytic T lymphocyte
- DNS
Data not shown
- EDSS
Extended disability scale score of Kurtzke
- F
Female
- FTY
Fingolimod/FTY720
- FTY-p
FTY-phosphate
- H
Hispanic
- HC
healthy control
- I
Indian/South Asian
- M
Male
- MFI
Median fluorescence intensity
- MS-a
Active/exacerbating MS
- MS-s
Clinically stable MS
- N
Number
- NA
Not applicable
- NS
Not significant
- O
Other (East Asian ancestry)
- PBMC
Peripheral blood mononuclear cell
- PPMS
Progressive forms of MS
- RRMS
Stable relapsing/remitting MS
- Rx
Treatment
- S1PR
Sphingosine 1-phosphate receptor
- SEM
Standard error of the mean
- SPMS
Secondary Progressive form of MS
- Treg
Regulatory T cell
- W
White/Caucasian
Appendix. Authors
Study Funding
This research was funded by an unrestricted research grant from Novartis (Award Number: IIRP-1340), the National MS Society (Award Number: RG#4509A), private donations from Louis and Christine Friedrich (FDT), and the Metcalf Fund (TWH).
Disclosure
T.W. Houston, F. Yao, and Q. Howlett-Prieto report nothing to disclose. X.F. has received unrestricted research support from Bayer, Biogen, BMS, Mallinckrodt, Merck-Serono, and Novartis, which produce drugs for the treatment of MS, some of which are anti-CD20 therapies, and Roche-Genentech, which produces rituximab and ocrelizumab studied in this paper. A.T. Reder has received unrestricted research support from Bayer, Biogen, BMS, Roche-Genentech, Mallinckrodt, Merck-Serono, and Novartis and is a consultant for Bayer, Biogen, Merck-Serono, Novartis, and Roche-Genentech. F.D. Testai reports private donations from Louis and Christine Friedrich (FDT).Go to Neurology.org/NN for full disclosures.
References
- 1.Dendrou CA, Fugger L, Friese MA. Immunopathology of multiple sclerosis. Nat Rev Immunol. 2015;15(9):545-558. [DOI] [PubMed] [Google Scholar]
- 2.Ménager-Marcq I, Pomié C, Romagnoli P, van Meerwijk JP. CD8+CD28- regulatory T lymphocytes prevent experimental inflammatory bowel disease in mice. Gastroenterology. 2006;131(6):1775-1785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Najafian N, Chitnis T, Salama AD, et al. Regulatory functions of CD8+CD28- T cells in an autoimmune disease model. J Clin Invest. 2003;112(7):1037-1048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Tulunay A, Yavuz S, Direskeneli H, Eksioglu-Demiralp E. CD8+CD28-suppressive T cells in systemic lupus erythematosus. Lupus. 2008;17(7):630-637. [DOI] [PubMed] [Google Scholar]
- 5.Antel JP, Arnason BG, Medof ME. Suppressor cell function in multiple sclerosis: correlation with clinical disease activity. Ann Neurol. 1979;5(4):338-342. [DOI] [PubMed] [Google Scholar]
- 6.Baughman EJ, Mendoza JP, Ortega SB, et al. Neuroantigen-specific CD8+ regulatory T-cell function is deficient during acute exacerbation of multiple sclerosis. J Autoimmun. 2011;36(2):115-124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Koide J, Engleman EG. Differences in surface phenotype and mechanism of action between alloantigen-specific CD8+ cytotoxic and suppressor T cell clones. J Immunol. 1990;144(1):32-40. [PubMed] [Google Scholar]
- 8.Cortesini R, LeMaoult J, Ciubotariu R, Cortesini NS. CD8+CD28- T suppressor cells and the induction of antigen-specific, antigen-presenting cell-mediated suppression of Th reactivity. Immunol Rev. 2001;182:201-206. [DOI] [PubMed] [Google Scholar]
- 9.Antel JP, Bania MB, Reder A, et al. Activated suppressor cell dysfunction in progressive multiple sclerosis. J Immunol. 1986;137(1):137-141. [PubMed] [Google Scholar]
- 10.Crucian B, Dunne P, Friedman H, et al. Alterations in levels of CD28-/CD8+ suppressor cell precursor and CD45RO+/CD4+ memory T lymphocytes in the peripheral blood of multiple sclerosis patients. Clin Diagn Lab Immunol. 1995;2(2):249-252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Karaszewski JW, Reder AT, Anlar B, et al. Increased lymphocyte beta-adrenergic receptor density in progressive multiple sclerosis is specific for the CD8+, CD28- suppressor cell. Ann Neurol. 1991;30(1):42-47. [DOI] [PubMed] [Google Scholar]
- 12.Correale J, Villa A. Role of CD8+ CD25+ Foxp3+ regulatory T cells in multiple sclerosis. Ann Neurol. 2010;67(5):625-638. [DOI] [PubMed] [Google Scholar]
- 13.Cunnusamy K, Baughman EJ, Franco J, et al. Disease exacerbation of multiple sclerosis is characterized by loss of terminally differentiated autoregulatory CD8+ T cells. Clin Immunol. 2014;152(1-2):115-126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.van Nierop GP, van Luijn MM, Michels SS, et al. Phenotypic and functional characterization of T cells in white matter lesions of multiple sclerosis patients. Acta Neuropathol. 2017;134(3):383-401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Mikulkova Z, Praksova P, Stourac P, et al. Numerical defects in CD8+CD28- T-suppressor lymphocyte population in patients with type 1 diabetes mellitus and multiple sclerosis. Cell Immunol. 2010;262(2):75-79. [DOI] [PubMed] [Google Scholar]
- 16.Arnason BG, Antel JP, Reder AT. Immunoregulation in multiple sclerosis. Ann N Y Acad Sci. 1984;436:133-139. [DOI] [PubMed] [Google Scholar]
- 17.Tyler AF, Mendoza JP, Firan M, Karandikar NJ. CD8(+) T cells are required for glatiramer acetate therapy in autoimmune demyelinating disease. PLoS One. 2013;8(6):e66772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Mikulkova Z, Praksova P, Stourac P, et al. Numerical defects in CD8+CD28- T-suppressor lymphocyte population in patients with type 1 diabetes mellitus and multiple sclerosis. Cell Immunol. 2010;262(2):75-79. [DOI] [PubMed] [Google Scholar]
- 19.Yagi H, Kamba R, Chiba K, et al. Immunosuppressant fingolimod inhibits thymocyte emigration. Eur J Immunol. 2000;30(5):1435-1444. [DOI] [PubMed] [Google Scholar]
- 20.Cyster JG, Schwab SR. Sphingosine-1-phosphate and lymphocyte egress from lymphoid organs. Annu Rev Immunol. 2012;30:69-94. [DOI] [PubMed] [Google Scholar]
- 21.Mehling M, Brinkmann V, Antel J, et al. Fingolimod therapy exerts differential effects on T cell subsets in multiple sclerosis. Neurology. 2008;71(16):1261-1267. [DOI] [PubMed] [Google Scholar]
- 22.Johnson TA, Lapierre Y, Bar-Or A, Antel JP. Distinct properties of circulating CD8+ T cells in fingolimod-treated patients with multiple sclerosis. Arch Neurol. 2010;67(12):1449-1555. [DOI] [PubMed] [Google Scholar]
- 23.Baeyens A, Bracero S, Chaluvadi VS, et al. Monocyte- derived S1P in the lymph node regulates immune responses. Nature. 2021;592(7853):290-295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Shiow LR, Rosen DB, Brdicková N, et al. CD69 acts downstream of interferon-α/β to inhibit S1P1 and lymphocyte egress from lymphoid organs. Nature. 2006;440(7083):540-544. [DOI] [PubMed] [Google Scholar]
- 25.Mackay LK, Braun A, Macleod BL, et al. Cutting edge: CD69 interference with sphingosine-1-phosphate receptor function regulates peripheral T cell retention. J Immunol. 2015;194(5):2059-2063. [DOI] [PubMed] [Google Scholar]
- 26.Kappos L, Radue EW, O'Connor P, et al. ; FREEDOMSStudy Group. A placebo-controlled trial of oral fingolimod in relapsing multiple sclerosis. N Engl J Med. 2010;362(5):387-401. [DOI] [PubMed] [Google Scholar]
- 27.Feng X, Bao R, Li L, et al. Interferon-β corrects massive gene dysregulation in multiple sclerosis: short-term and long-term effects on immune regulation and neuroprotection. Ebiomedicine. 2019;49:269-283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Howlett-Prieto Q, Feng X, Kramer JF, Kramer KJ, Houston TW, Reder AT. Anti-CD20 therapy corrects a CD8 regulatory T cell deficit in multiple sclerosis. Mult Scler. 2021;27(14):2170-2179. [DOI] [PubMed] [Google Scholar]
- 29.Borthwick NJ, Lowdell M, Salmon M, Akbar AN. Loss of CD28 expression on CD8(+) T cells is induced by IL-2 receptor gamma chain signalling cytokines and type I IFN, and increases susceptibility to activation-induced apoptosis. Int Immunol. 2000;12(7):1005-1013. [DOI] [PubMed] [Google Scholar]
- 30.Valenzuela HF, Effros RB. Divergent telomerase and CD28 expression patterns in human CD4 and CD8 T cells following repeated encounters with the same antigenic stimulus. Clin Immunol. 2002;105(2):117-125. [DOI] [PubMed] [Google Scholar]
- 31.Noronha A, Toscas A, Jensen MA. Contrasting effects of alpha, beta, and gamma interferons on nonspecific suppressor function in multiple sclerosis. Ann Neurol. 1992;31(1):103-106. [DOI] [PubMed] [Google Scholar]
- 32.Allie R, Hu L, Mullen KM, Dhib-Jalbut S, Calabresi PA. Bystander modulation of chemokine receptor expression on peripheral blood T lymphocytes mediated by glatiramer therapy. Arch Neurol. 2005(6):889-894. [DOI] [PubMed] [Google Scholar]
- 33.Freedman MS, Ruijs TC, Blain M, et al. Phenotypic and functional characteristics of activated CD8+ cells: a CD11b-CD28- subset mediates noncytolytic functional suppression. Clin Immunol Immunopathol. 1991;60(2):254-267. [DOI] [PubMed] [Google Scholar]
- 34.Viglietta V, Baecher-Allan C, Weiner HL, Hafler DA. Loss of functional suppression by CD4+CD25+ regulatory T cells in patients with multiple sclerosis. J Exp Med. 2004;199(7):971-979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Mishra S, Srinivasan S, Ma C, Zhang N. CD8 + regulatory T cell—a mystery to be revealed. Front Immunol. 2021;12:708874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Genç K, Dona DL, Reder AT. Increased CD80(+) B cells in active multiple sclerosis and reversal by interferon beta-1b therapy. J Clin Invest. 1997;99(11):2664-2671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Vieyra-Lobato MR, Vela-Ojeda J, Montiel-Cervantes L, López-Santiago R, Moreno-Lafont MC. Description of CD8+ regulatory T lymphocytes and their specific intervention in graft-versus-host and infectious diseases, autoimmunity, and cancer. J Immunol Res. 2018;2018:3758713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Tomas-Ojer P, Puthenparampil M, Cruciani C, et al. Characterization of antigen-induced CD4+ T-cell senescence in multiple sclerosis. Front Neurol. 2022;13:790884. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Sawicka E, Dubois G, Jarai G, et al. The sphingosine 1-phosphate receptor agonist FTY720 differentially affects the sequestration of CD4+/CD25+ T-regulatory cells and enhances their functional activity. J Immunol. 2005;175(12):7973-7980. [DOI] [PubMed] [Google Scholar]
- 40.Ghadiri M, Rezk A, Li R, et al. Pre-treatment T-cell subsets associate with fingolimod treatment responsiveness in multiple sclerosis. Sci Rep 2020;10(1):356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Duszczyszyn DA, Williams JL, Mason H, et al. Thymic involution and proliferative T-cell responses in multiple sclerosis. J Immunol. 2010;221(1-2):73-80. [DOI] [PubMed] [Google Scholar]
- 42.La Rocca G, Olejniczak SH, González AJ, et al. In vivo, Argonaute-bound microRNAs exist predominantly in a reservoir of low molecular weight complexes not associated with mRNA. Proc Natl Acad Sci USA. 2015. ;112(3):767-772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Weng NP, Akbar AN, Goronzy J. CD28(-) T cells: their role in the age-associated decline of immune function. Trends Immunol. 2009;30(7):306-312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Grebenciucova E, Pruitt A. Infections in patients receiving multiple sclerosis disease-modifying therapies. Curr Neurol Neurosci Rep. 2017;17(11):88. [DOI] [PubMed] [Google Scholar]
- 45.Duszczyszyn DA, Williams JL, Mason H, et al. Thymic involution and proliferative T-cell responses in multiple sclerosis. J Immunol. 2010;221(1-2):73-80. [DOI] [PubMed] [Google Scholar]
- 46.Panda AK, Kim Y-H, Shevach EM. Control of memory phenotype T lymphocyte homeostasis: role of costimulation. J Immunol. 2022;208(4):851-860. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Anonymized data, per reasonable request, will be shared with qualified investigators by the corresponding author.