Liver cancer remains a major, and growing, global health concern, standing as the third leading cause of cancer-related morality worldwide. Primary liver cancer is an umbrella term representing a heterogenous groups of cancers of which hepatocellular carcinoma (HCC) is the most frequent form (1). The majority of HCC arises in the context of chronic liver damage resulting from viral infections of the liver (e.g. hepatitis B or C), alcohol abuse, as well as non-alcoholic fatty liver disease (NAFLD) (1). Non-alcoholic steatohepatitis (NASH), a subtype of NAFLD characterized by inflammation and liver damage in addition to fat build-up within the liver, is a rapidly growing cause of HCC in Western countries (2).
With the advancement of clinical surveillance programs, approximately 40–50% of HCC patients receive an early diagnosis at which point potentially curative therapeutic options are applicable (3). However, this still renders half of all HCC patients at the mercy of systemic therapies that for decades have been limited to broad-acting tyrosine kinase inhibitors (TKIs) (3). The first of these systemic therapies approved in 2007 as first-line treatment for advanced HCC was sorafenib, which enhanced median overall survival of patients from 8 months to 11 months (4). While 3 months may sound marginal, this was a major breakthrough in disease management for advanced HCC patients. Since that time, a plethora of TKIs, including lenvatinib, cabozantinib, regorafenib, and ramucirumab, became clinically available as first- or second-line to treat advanced HCC, although with similar survival benefits to that of sorafenib (just a few months) (3).
Of recent, advanced HCC treatments have been revolutionized by the success of immunotherapies, which harness the host’s immune system to recognize and kill cancer. In 2017 and 2018, respectively, nivolumab (anti-PD-1) (5) and pembrolizumab (anti-PD-1) (6) were granted accelerated approval as second-line therapies in the treatment of advanced HCC. This was followed in 2020 by the approval of nivolumab in combination with ipilimumab (anti-CTLA-4) as second-line therapy for HCC (7). Most recently in 2020, the combination of atezolizumab (anti-PD-L1) with bevacizumab (anti-VEGFA) demonstrated superiority over sorafenib and was granted accelerated approval as first-line therapy in the treatment of advanced HCC (8). These immunotherapies together have demonstrated durable objective response rates of approximately 20–30%; a more detailed summary of advancements in HCC immunotherapies can be found in this review (9). While these therapeutic responses to immunotherapies are unprecedented in the treatment of advanced HCC, approximately 70–80% of patients still remain unresponsive to these treatments. This underscores the need, not only for novel therapeutics and combination regimens, but for better patient selection criteria for those who may or may not receive clinical benefit from currently approved therapies.
In this issue, Murai, Kodama et al. set out to address these questions through the characterization of immunotherapy-susceptible HCC subtypes utilizing a multiomics-based approach in a cohort of 113 non-viral HCC patients who underwent surgical resection. As aforementioned, the prevalence of non-viral HCC is on the rise due to risk factors such as NAFLD/NASH and metabolic syndrome; as such, the authors focused their studies on this subset of patients. It is important to reiterate that this cohort of patients underwent surgical resection for HCC, meaning these patients had early-stage disease and findings may or may not fully phenocopy patients with advanced stage HCC - a limitation of the study recognized by the authors in their concluding remarks.
As an initial approach, Murai, Kodama et al. performed bulk transcriptional characterization from tumor-derived RNA on resected tumors from these 113 non-viral HCC patients, through which unsupervised hierarchical clustering yielded three subclasses of HCC patients based on gene expression profiles – Class I, II, and III. In 2008(10), 2009(11), and 2017(12), three studies led by Hoshida et al., Chiang et al., and Sia et al., respectively, established key classification systems for human HCC patients based on transcriptomic and genomic analysis. Murai et al. compared their three molecular subclasses to those defined within these studies. The authors noted an association of Class II with the IFN subclass defined by Chiang et al. and S1 defined by Hoshida et al. Compellingly, Class I was associated with the Proliferation subclass defined by Chiang et al. while Class III was associated with the CTNNB1 subclass identified by Chiang et al. and the S3 subclass defined by Hoshida et al. Additionally, Class I was associated with larger tumor size, portal vein invasion, enrichment of cell cycle and mitosis related gene sets, and the poorest prognosis of the three classes. On the other hand, Class III was associated with upregulation of WNT-ß-catenin signaling and downregulation of genes involved in dendritic cell recruitment into the tumor microenvironment and immune cell activation, which falls in line with previous reports suggesting that CTNNB1 activation in HCC impairs dendritic cell recruitment to the tumor microenvironment (TME) and, subsequently, results in diminished tumor-specific cytotoxic T cell presence (i.e. an “immune desert” phenotype)(13). As previously shown, Class III was associated with the best prognosis.
Murai, Kodama et al. further characterized these non-viral HCCs through cancer genome sequencing on 55 of the 113 patients using a human HCC-targeted panel that detects aberrancies in 69 genes. They found Class I to be associated with TP53 mutations and Class III with CTNNB1 mutations. Further, patients with TP53 mutation had the worst prognosis, whereas patients with the CTNNB1 mutations had the best prognosis, confirming previous studies. The authors validated these findings using a dataset from The Cancer Genome Atlas of 180 patients with non-viral HCC. Again, the authors found that TP53 mutations were associated with class I and poorer prognosis whilst CTNNB1 mutations were associated with Class III, although this was not associated with significantly better prognosis.
Deeper transcriptomic analysis classified 43 of the 113 patients into the immune class of HCC, previously described by Sia et al. and this was associated with Class II. These tumors expressed significantly higher levels of immune cell-related transcripts, suggesting increased infiltration of immune cells into the tumors. The authors next aimed to elucidate which subsets of patients, and specifically which of their clinicopathological features, comprise the immune class as this could have important therapeutic implications. To this end, Murai et al. found that steatotic HCC, which represented 23% of their non-viral cases, was specifically associated with the immune class. Steatotic HCC demonstrated significantly higher cytotoxic T cell exhaustion and stromal/TGFß activation signatures, suggesting presence but potential functional impairment of immune cells within these tumors. Expressly, these tumors displayed significantly higher levels of transcripts associated with T cell-associated immune checkpoints, cancer-associated fibroblasts (CAFs), as well as cytokines involved in M2-like macrophage polarization, which was confirmed at the protein level through PDL1, aSMA, and CD163 immunohistochemical staining. To better characterize the interactions of these cell types within the TME, the authors employed spatial transcriptomics technology using the Visium platform. In tissue areas with high levels of exhausted cytotoxic T cell (defined by CD8A+NR4A1+), there were also high levels of CD163, VIM, and TGFß1. The authors hypothesized that M2-like macrophages and CAFs might produce TGFß, promoting local exhaustion of cytotoxic T cells.
To delve into the mechanism by which intratumoral steatosis is impacting the exhausted state of immune cells within the TME, the authors performed lipidomics-based profiling of total fatty acids within the tumors. Palmitic acid (PA) levels were significantly enriched in steatotic HCC compared with non-steatotic HCC samples. To assess the role of PA in vitro, PA was added to Hep3B human HCC cell lines and the authors observed lipid accumulation in addition to upregulation of PD-L1, both at the mRNA and surface protein level. Further, these cells displayed upregulation of CSF1, CXCL8, and TGFß1, both at the mRNA and protein level, which are associated with M2-like macrophage polarization and CAF activation. Moreover, these PA-treated Hep3B cells upregulated mRNA levels of CD206 and IL10 when co-cultured with a human macrophage cell line as well as TGFß1 when co-cultured with a hepatic stellate cell line. Upregulation of these cytokines was confirmed in steatotic HCC patients. This suggests that PA accumulation in steatotic HCC tumors may promote an immunosuppressive TME, leading to the observed immune exhaustion.
Lastly, and perhaps most importantly, the authors hypothesized that steatotic HCC may be susceptible to immunotherapy (Fig. 1). This hypothesis stems from the fact that these steatotic HCC tumors appear to harbor the necessary anti-tumor immune components, though in their exhausted state, these cells require a “boost” to reinvigorate their anti-tumor killing capacity – a role served quite well by currently approved immunotherapies. However, the authors asked an important question first – is steatotic HCC a feasible biomarker to identify in the clinic for patient stratification, especially considering most advanced HCC patients do not routinely receive biopsies? To this end, the authors analyzed surgically resected HCCs from 20 patients and found a strong positive correlation between histological lipid deposition and fat fraction measured by magnetic resonance (MR) imaging, suggesting that steatosis in HCC can in fact be identified through non-invasive MR imaging in the clinic. The authors then retrospectively analyzed 30 patients with advanced HCC treated with Atezolizumab and Bevacizumab, 7 of whom had steatotic HCC, as determined by MR imaging. The steatotic patients demonstrated significantly enhanced progression free survival compared with the non-steatotic patients – in fact, none of the patients with steatotic HCC underwent disease progression during the 5.4-month observation period.
Figure 1.
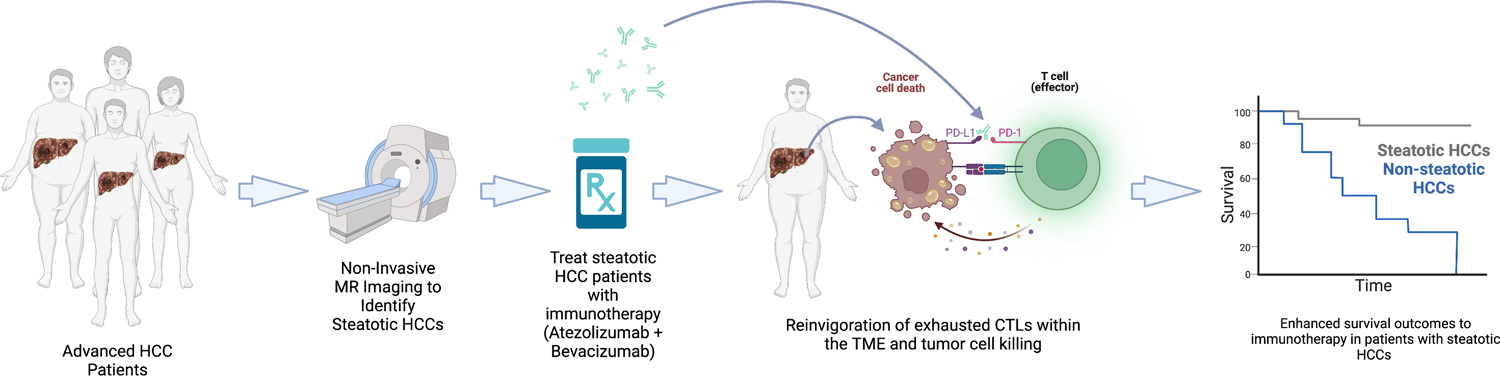
Pipeline for selection of HCC patients with steatosis and treatment with combination immunotherapy. MR, magnetic resonance; CTL, cytotoxic T cells; TME, tumor microenvironment.
In brief, Murai et al. utilized a multiomics approach to characterize non-viral early-stage human HCCs. Transcriptomically, three subclasses were identified and linked to patient prognosis, genetic alteration status, and immune status of the TME. Integration of clinicopathological data led to the association of steatotic HCC with an immune-inflamed, but exhausted, phenotype. Based on retrospective analysis of patients that received combination immunotherapy (Atezolizumab + Bevacizumab), the authors suggest that patients with steatotic HCC may be ideal candidates as recipients for this combination immunotherapy and that MR imaging for steatosis may serve as a novel biomarker for patient stratification. However, the sample size of patients treated with immunotherapy in this study is very small, so additional follow-up studies in larger cohorts of patients will be critical. Finally, it will be important to establish which is the response of steatotic patients to single-agent immunotherapy. A recent study by Pfister et al. showed that HCC patients with NASH are resistant to anti-PD-1 immunotherapy. It will be critical to confirm whether this resistance can be overcome by combination of bevazicumab and atezolizumab and whether there are response differences between NAFLD and NASH patients.
Acknowledgments
AL has received research funding from Pfizer and Genentech and consulting fees from Astra Zeneca. KEL declares no conflict of interest.
This work was funded by NIH/NCI R37CA230636 Merit Award and Damon Runyon-Rachleff Innovator Award (DR52-18) for AL.
References:
- 1.Villanueva A. Hepatocellular Carcinoma. N Engl J Med. 2019;380(15):1450–62. Epub 2019/04/11. doi: 10.1056/NEJMra1713263. [DOI] [PubMed] [Google Scholar]
- 2.Younossi Z, Stepanova M, Ong JP, Jacobson IM, Bugianesi E, Duseja A, Eguchi Y, Wong VW, Negro F, Yilmaz Y, Romero-Gomez M, George J, Ahmed A, Wong R, Younossi I, Ziayee M, Afendy A, Global Nonalcoholic Steatohepatitis C. Nonalcoholic Steatohepatitis Is the Fastest Growing Cause of Hepatocellular Carcinoma in Liver Transplant Candidates. Clin Gastroenterol Hepatol. 2019;17(4):748–55 e3. Epub 2018/06/17. doi: 10.1016/j.cgh.2018.05.057. [DOI] [PubMed] [Google Scholar]
- 3.Llovet JM, Montal R, Sia D, Finn RS. Molecular therapies and precision medicine for hepatocellular carcinoma. Nat Rev Clin Oncol. 2018;15(10):599–616. Epub 2018/08/01. doi: 10.1038/s41571-018-0073-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Llovet JM, Ricci S, Mazzaferro V, Hilgard P, Gane E, Blanc JF, de Oliveira AC, Santoro A, Raoul JL, Forner A, Schwartz M, Porta C, Zeuzem S, Bolondi L, Greten TF, Galle PR, Seitz JF, Borbath I, Haussinger D, Giannaris T, Shan M, Moscovici M, Voliotis D, Bruix J, Group SIS. Sorafenib in advanced hepatocellular carcinoma. N Engl J Med. 2008;359(4):378–90. Epub 2008/07/25. doi: 10.1056/NEJMoa0708857. [DOI] [PubMed] [Google Scholar]
- 5.El-Khoueiry AB, Sangro B, Yau T, Crocenzi TS, Kudo M, Hsu C, Kim TY, Choo SP, Trojan J, Welling THR, Meyer T, Kang YK, Yeo W, Chopra A, Anderson J, Dela Cruz C, Lang L, Neely J, Tang H, Dastani HB, Melero I. Nivolumab in patients with advanced hepatocellular carcinoma (CheckMate 040): an open-label, non-comparative, phase 1/2 dose escalation and expansion trial. Lancet. 2017;389(10088):2492–502. Epub 2017/04/25. doi: 10.1016/S0140-6736(17)31046-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Zhu AX, Finn RS, Edeline J, Cattan S, Ogasawara S, Palmer D, Verslype C, Zagonel V, Fartoux L, Vogel A, Sarker D, Verset G, Chan SL, Knox J, Daniele B, Webber AL, Ebbinghaus SW, Ma J, Siegel AB, Cheng AL, Kudo M, investigators K-. Pembrolizumab in patients with advanced hepatocellular carcinoma previously treated with sorafenib (KEYNOTE-224): a non-randomised, open-label phase 2 trial. Lancet Oncol. 2018;19(7):940–52. Epub 2018/06/08. doi: 10.1016/S1470-2045(18)30351-6. [DOI] [PubMed] [Google Scholar]
- 7.Yau T, Kang YK, Kim TY, El-Khoueiry AB, Santoro A, Sangro B, Melero I, Kudo M, Hou MM, Matilla A, Tovoli F, Knox JJ, Ruth He A, El-Rayes BF, Acosta-Rivera M, Lim HY, Neely J, Shen Y, Wisniewski T, Anderson J, Hsu C. Efficacy and Safety of Nivolumab Plus Ipilimumab in Patients With Advanced Hepatocellular Carcinoma Previously Treated With Sorafenib: The CheckMate 040 Randomized Clinical Trial. JAMA Oncol. 2020;6(11):e204564. Epub 2020/10/02. doi: 10.1001/jamaoncol.2020.4564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Finn RS, Qin S, Ikeda M, Galle PR, Ducreux M, Kim TY, Kudo M, Breder V, Merle P, Kaseb AO, Li D, Verret W, Xu DZ, Hernandez S, Liu J, Huang C, Mulla S, Wang Y, Lim HY, Zhu AX, Cheng AL, Investigators IM. Atezolizumab plus Bevacizumab in Unresectable Hepatocellular Carcinoma. N Engl J Med. 2020;382(20):1894–905. Epub 2020/05/14. doi: 10.1056/NEJMoa1915745. [DOI] [PubMed] [Google Scholar]
- 9.Sangro B, Sarobe P, Hervas-Stubbs S, Melero I. Advances in immunotherapy for hepatocellular carcinoma. Nat Rev Gastroenterol Hepatol. 2021;18(8):525–43. Epub 2021/04/15. doi: 10.1038/s41575-021-00438-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Chiang DY, Villanueva A, Hoshida Y, Peix J, Newell P, Minguez B, LeBlanc AC, Donovan DJ, Thung SN, Sole M, Tovar V, Alsinet C, Ramos AH, Barretina J, Roayaie S, Schwartz M, Waxman S, Bruix J, Mazzaferro V, Ligon AH, Najfeld V, Friedman SL, Sellers WR, Meyerson M, Llovet JM. Focal gains of VEGFA and molecular classification of hepatocellular carcinoma. Cancer Res. 2008;68(16):6779–88. Epub 2008/08/15. doi: 10.1158/0008-5472.CAN-08-0742. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hoshida Y, Nijman SM, Kobayashi M, Chan JA, Brunet JP, Chiang DY, Villanueva A, Newell P, Ikeda K, Hashimoto M, Watanabe G, Gabriel S, Friedman SL, Kumada H, Llovet JM, Golub TR. Integrative transcriptome analysis reveals common molecular subclasses of human hepatocellular carcinoma. Cancer Res. 2009;69(18):7385–92. Epub 2009/09/03. doi: 10.1158/0008-5472.CAN-09-1089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Sia D, Jiao Y, Martinez-Quetglas I, Kuchuk O, Villacorta-Martin C, Castro de Moura M, Putra J, Camprecios G, Bassaganyas L, Akers N, Losic B, Waxman S, Thung SN, Mazzaferro V, Esteller M, Friedman SL, Schwartz M, Villanueva A, Llovet JM. Identification of an Immune-specific Class of Hepatocellular Carcinoma, Based on Molecular Features. Gastroenterology. 2017;153(3):812–26. Epub 2017/06/19. doi: 10.1053/j.gastro.2017.06.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Ruiz de Galarreta M, Bresnahan E, Molina-Sanchez P, Lindblad KE, Maier B, Sia D, Puigvehi M, Miguela V, Casanova-Acebes M, Dhainaut M, Villacorta-Martin C, Singhi AD, Moghe A, von Felden J, Tal Grinspan L, Wang S, Kamphorst AO, Monga SP, Brown BD, Villanueva A, Llovet JM, Merad M, Lujambio A. beta-Catenin Activation Promotes Immune Escape and Resistance to Anti-PD-1 Therapy in Hepatocellular Carcinoma. Cancer Discov. 2019;9(8):1124–41. Epub 2019/06/13. doi: 10.1158/2159-8290.CD-19-0074. [DOI] [PMC free article] [PubMed] [Google Scholar]


