Abstract
Objective:
We hypothesize offset theta-tACS (transcranial alternating current stimulation) improves emotion regulation (ER) and psychopathology more than transcranial direct current stimulation (tDCS) in participants with internalizing psychopathologies (IPs).
Methods:
This pilot study utilized a double-blind, pseudo-counterbalanced, sham-controlled design with participants with IPs. Participants were assigned to receive tDCS or tACS, underwent four stimulation sessions (two sham), and completed an emotion regulation task (ERT) during or after stimulation. Participants completed the Beck Depression Inventory before/after the study, the Spielberger State and Trait Anxiety Index after each ERT, and rated their arousal, valence, and perceived reappraisal ability during the ERT.
Results:
Participants receiving either stimulation type showed a reduction in anxiety, depression, and valence and arousal ratings. We additionally discovered an effect demonstrating those who received sham stimulation first displayed little-to-no change in any score across the study, but tACS participants who received verum stimulation first showed significant improvements in each metric.
Conclusions:
Improving ER capabilities via theta tACS has the potential to yield beneficial clinical effects.
Significance:
This study adds validity to the use of non-invasive neuromodulatory methods, especially tACS, to alleviate IPs. Additional research is needed to better understand the effects of sham stimulation. Careful consideration of sham incorporation should be made in future studies.
Keywords: Transcranial Alternating Current Stimulation, Transcranial Direct Current Stimulation, Emotion Regulation, Theta Synchrony, Internalizing Psychopathologies, Sham Stimulation
1. Introduction
Internalizing psychopathologies (IPs) including depression and anxiety affect nearly 30% of the United States population (Kessler, 2012; Konnopka et al., 2009). These psychopathologies are characterized by disturbances in emotion regulation (ER) (Aldao et al., 2016; Bar-Haim et al., 2007; Price et al., 2011), the processes by which individuals modify their emotions to achieve goals in response to contextual demands (Aldao et al., 2016; Berking et al., 2013), such that these individuals have difficulty identifying and tolerating negative emotions and effectively modifying to adapt to their situation(Campbell-Sills et al., 2006; Honkalampi et al., 2001; Kassel et al., 2007). In a “real world” setting, ER is involved in situations such as trying to get through a workday after the loss of a loved one or trying to maintain composure after receiving an email from your boss asking you to come to their office to discuss an issue. Relevant to our current study are ER paradigms that utilize a cognitive approach (i.e., ‘reappraisal’) when viewing negative images to reduce the negative affect that would otherwise occur when simply viewing the negative image(Etkin et al., 2015; Fitzgerald et al., 2016; Hajcak and Nieuwenhuis, 2006; Kinney et al., 2019).
Transcranial direct current stimulation (tDCS) has been a dominant form of transcranial electrical stimulation (tES) over the last several decades. tDCS acts by applying a constant electrical stimulation to the brain to either raise or lower the resting membrane potentials of the neurons lying below the electrode, in effect making them more or less likely to fire, respectively. This technique has been utilized in the context of ER previously, targeting the dlPFC (Conson et al., 2015; Feeser et al., 2014; Peña-Gómez et al., 2011) and/or the medial prefrontal cortex (Abend et al., 2019) – both key regions in emotion regulation – in healthy populations. Results from these studies indicate that the use of tDCS to target ER-related regions has the potential to modulate and facilitate the regulation and processing of negative emotions.
Over the last decade and a half, another type of tES has been gaining popularity due to a shift in the field towards focusing on intrinsic neural rhythms. Transcranial alternating current stimulation (tACS) is a non-invasive stimulation technique that applies a frequency-specific sinusoidal current to the scalp. Though the underlying neuronal mechanisms are not yet fully understood, tACS is thought to entrain endogenous brain rhythms by altering underlying membrane potentials (Chan and Nicholson, 1986) and has been shown to affect both frequency (Helfrich et al., 2014) and synchrony (Polanía et al., 2012). On-set tACS – stimulation delivered at each electrode with a zero-degree phase difference – has been shown to enhance synchrony at the stimulation frequency, whereas off-set tACS – stimulation delivered with a 180-degree phase difference between electrodes – has been shown to decrease underlying synchrony at the stimulation frequency(Polanía et al., 2012). Specifically, results from Polanía (Polanía et al., 2012), Strüber (Strüber et al., 2014), and Tseng and colleagues (Tseng et al., 2018) indicates that offset tACS induces oppositional effects on the stimulated frequency band as well as on the relevant synchrony-related constructs. tACS has previously been used in research settings to modulate higher order functions such as working memory and executive processing via the dlPFC and the left parietal region (Jaušovec and Jaušovec, 2014), fluid intelligence via the left parietal region (Pahor and Jaušovec, 2014), and risk taking behavior via the dlPFC(Sela et al., 2012). To the best of our knowledge, however, tACS has not yet been utilized to target ER and has only been used minimally to study IPs. Because the theta band in particular has been shown to have higher baseline levels of global theta synchrony in a subset of IP individuals (Xing et al., 2017) and is aberrant during emotion regulation in these same individuals (Xing et al., 2019), we expect that disrupting theta synchrony in an offset manner would improve ER capability.
The goal of our study is to utilize these forms of tES to improve participants’ ER ability, and we believe this will lead to an improvement in clinical symptoms of anxiety and depression. This pilot study provides a preliminary side-by-side analysis of the two stimulation methods in the context of emotion regulation in individuals with IPs., Though tDCS has previously been demonstrated to effectively improve ER, we hypothesize that offset tACS will also yield improvements in participants’ratings of negative affect, anxiety scores, and depression scores.
2. Materials and Methods
2.1. Participants
Thirty-three volunteers with depression and/or anxiety were recruited from a clinical population (Table 1). Inclusion required a psychiatric evaluation by a board-certified psychiatrist, a score greater than 23 on the Depression Anxiety Stress Scale (DASS-21), ability and willingness to give written consent, and no major active neurologic or medical problems determined by the Cumulative Illness Rating Scale. Participants were excluded if they exhibited active suicidal ideation at any point in the protocol determined by the psychiatrist, past or current substance dependence, or pregnancy at the time of study. The study protocol was approved by the Institutional Review Board of the University of Illinois at Chicago and participants were monetarily compensated for their time.
Table 1. Participant Demographics.
Averages +/− one standard deviation for transcranial alternating current stimulation (tACS) and transcranial direct current stimulation (tDCS) participants for the Depression Anxiety Stress Scale (DASS) metrics and age. Categorical demographics were coded and analyses were conducted on the coded variables. Two tACS participants did not disclose their demographic information.
| tACS | tDCS | t | p | SE | |
|---|---|---|---|---|---|
| N | 16 | 17 | |||
| DASS Depression | 22.0 +/− 9.2 | 23.8 +/−9.3 | 0.548 | 0.587 | 3.219 |
| DASS Anxiety | 21.1 +/− 8.2 | 23.2 +/− 10.7 | 0.617 | 0.542 | 3.325 |
| DASS Total | 34.6 +/− 9.6 | 37.3 +/−10.5 | 0.779 | 0.442 | 3.509 |
| Age | 30.0 +/− 12.1 years | 34.4 +/− 12.0 years | 1.016 | 0.318 | 4.344 |
| Sex | n | n | 0.202 | 0.842 | 0.125 |
| Female | 12 | 15 | |||
| Male | 2 | 2 | |||
| Education | n | n | 0.07 | 0.945 | 0.422 |
| Less than high school or GED | 0 | 1 | |||
| High School or GED | 5 | 2 | |||
| Associates | 1 | 4 | |||
| Bachelor's | 4 | 8 | |||
| Masters | 4 | 2 | |||
| Ethnicity | n | n | 0.202 | 0.842 | 0.125 |
| Hispanic/Latino | 2 | 2 | |||
| Not Hispanic/Latino | 12 | 15 | |||
| Race | n | n | 0.363 | 0.719 | 0.238 |
| American Indian | 1 | 0 | |||
| Asian | 1 | 1 | |||
| Black or African American | 4 | 4 | |||
| White | 8 | 11 | |||
| Other | 0 | 1 |
2.2. Transcranial Stimulation
All transcranial stimulation was conducted with the XCITE device (Pulvinar Neuro, LLC, Chapel Hill, NC). tDCS and tACS stimulation parameters were chosen and implemented based on previous tES studies with emotion-related tasks(Feeser et al., 2014; Polanía et al., 2012; Sanchez et al., 2016). For tDCS stimulation, a 32-channel EEG cap was placed on participants’ heads and a mark for the anode was made at the F3 location to approximate the left dlPFC and a mark for the cathode was made at location Fp2 using a china marker. The EEG cap was then removed. Gel was applied to the stimulation electrodes and placed on the scalp at the markings. tDCS sham stimulation consisted of a 30 second ramp-up to 1.5 mA, then cessation of stimulation. Verum stimulation was administered at 2.0 mA.
We chose to use theta-tACS in an offset manner due to previous work noting increased theta synchrony in a subset of individuals with IPs at baseline (Xing et al., 2017) as well as an additional study indicating an aberrant theta band in these individuals during an emotion regulation task (ERT); (Xing et al., 2019). tACS electrode sites varied by participant, determined by a baseline EEG recording. The baseline recording was immediately analyzed and the electrodes between which theta synchrony was the highest was determined using a weighted phase lag index analysis with MATLAB toolbox Fieldtrip (See Figure 1; Donders Centre for Cognitive Neuroimaging, Nijmegen, Netherlands). Marks were made through the electrode cap using a china marker, caps were removed, and stimulation electrodes were placed following the same procedure as tDCS. After putting the EEG caps on over the tACS electrodes, gel was reapplied using a blunted tip syringe for each electrode. Verum stimulation was delivered at 6 Hz with a 180-degree phase difference between electrodes. For sham condition, stimulation was turned off following a 30 second ramp-up period.
Figure 1. Transcranial Alternating Current Stimulation Electrode Pairings.
Transcranial alternating current stimulation (tACS) electrodes were placed at the two locations between which baseline theta synchrony was recorded to be the highest. Each number on the figure represents the number of participants with that specific electrode pairing. Any electrodes not displayed above were not utilized for tACS administration.
2.3. Emotion regulation task
Participants completed several iterations of a validated ERT(Fitzgerald et al., 2016; Kinney et al., 2019), previously used while studying the theta band during emotion regulation (Xing et al., 2019). Each iteration consisted of 6 blocks of 20 novel stimuli presented in pseudorandomized order, during which participants were instructed to look at neutral images (‘look’), maintain negative affect while viewing negative images (‘maintain’), or reappraise negative images to reduce negative affect (‘reappraise’). Each iteration of the ERT included each instruction twice, once for blocks including images of only faces and once for blocks including only images of scenes. For this study, only data from stimuli blocks of scenes were analyzed. Images were from the International Affective Picture System (Lang et al., 1997) and were not repeated. Each image remained on screen for 8 seconds. Following each block of stimuli, participants were asked to rate on a 1-5 Likert scale how emotionally aroused they felt (‘arousal rate’; 1=not at all aroused, 5=very aroused), how negatively they felt (‘valence rate’; 1= not at all negative, 5=very negative), as well as how well they believed they were able to reappraise following each ‘reappraise’ block (‘reappraisal rate’; 1=low belief in reappraisal ability, 5=high belief in reappraisal ability). Ratings questions remained on the screen until answered by the participant.
2.4. Procedure
Participants came to the lab for an initial interview and screening with a board-certified psychiatrist who administered a psychiatric evaluation, the DASS-21 questionnaire, a demographics form, and the cumulative illness rating scale to assess general health and to determine eligibility.
Participants returned to the lab within two weeks of the initial interview and were randomly assigned to receive either tACS or tDCS. tDCS participants had stimulation electrodes placed and then all participants were fitted with a 32-electrode cap and sat at rest for an 8-minute baseline period. tACS participants had their caps removed following their baseline recording, baseline data analyzed for theta synchrony, and then stimulating electrodes and EEG caps were placed according to the above protocol (for details regarding EEG recording and preprocessing, see Supplementary Information). Electrodes and EEG caps were worn for the duration of the procedure. Participants then received their first session of stimulation while completing their first ERT (approximately 20 minutes), followed by an 8-minute rest without stimulation. They then received their second stimulation session during their second ERT, followed by an additional 8-minute rest. Participants then received 20 minutes of stimulation while resting and then performed their third ERT without stimulation, followed by an additional 20 minutes of stimulation during rest and then their final ERT iteration (Figure 2). Sham/Verum stimulations were pseudo-counterbalanced, such that some participants completed sham-verum-verum-sham and others ordered verum-sham-sham-verum for their sessions. Following each rest+ERT pair, participants completed the Spielberger State and Trait Anxiety Index (STAI) focusing on state-based items as well as a TCS safety questionnaire to assess any potential discomfort from stimulation. Participants completed the Beck Depression Index (BDI) before their baseline recording and after completion of the final ERT.
Figure 2. Procedure.
Participants completed alternating iterations of the emotion regulation task (ERT) and rest. Stimulation was delivered during the first two ERTs and last two rests. Stimulation order was either sham-verum-verum-sham or verum-sham-sham-verum. ERTs were approximately 20 minutes in length. State Trait Anxiety Index (STAI) was completed before baseline and following each ERT+rest pair and the Beck Depression Inventory (BDI) was completed before baseline and following the final ERT.
2.5. Statistical analysis
All analyses were performed using the Statistical Package for the Social Sciences Statistics (SPSS) (Version 27.0), were two-tailed, with an alpha level of 0.05. Independent samples t-tests indicated that participants randomized to receive tACS or tDCS did not differ based on age, sex, level of education, ethnicity, race, DASS-21 depression scores, DASS-21 anxiety scores, or DASS-21 total scores (Table 1).
BDI and STAI were chosen to detect state changes throughout the study. BDI analyses were paired samples t-tests comparing scores from the beginning of the study to scores following neuromodulation. STAI analyses included repeated measures ANOVAs with five conditions (baseline, sham, verum, after sham, after verum) as well as t-tests to compare baseline scores to final scores. For valence ratings and arousal ratings, analyses were 4 conditions (sham, verum, after sham, after verum) by 3 tasks (look, maintain, reappraise) repeated measures ANOVAs followed by t-tests to compare initial ‘reappraise’ scores with final ‘reappraise’ scores. Reappraisal scores – how well participants felt they were able to reappraise during ‘reappraise’ stimuli blocks – were analyzed using a repeated measures ANOVA with 4 conditions (sham, verum, after sham, after verum). Finally, correlational analyses were conducted to compare changes seen in clinical scores with changes seen in ‘reappraise’ valence and arousal ratings.
Due to the small sample size of this pilot study, the study design was not powered to be able to detect differences between the tACS and tDCS conditions and thusly no analyses were conducted to include current type as a between-subjects variable.
3. Results
3.1. Beck Depression Index
Participants who received tACS showed a significant reduction in depression scores from the beginning of the study () to immediately following the study () (F(1,15)=5.360, p=.035, η2p=.263). tDCS participants showed a slight decrease as well, but did not quite reach significance (, , F(1,16)=4.102, p=.060, η2p=.204)(Figure 3A). By stratifying the data by the order of stimulation, however, we uncovered separate trends. tACS participants who received verum (n=8) stimulation for their first session showed an average decrease in depression scores of 4.825 points (F(1,7)=21.862, p=.002, η2p=.757). In contrast, tACS participants who received sham first (n=8) showed no change in their BDI scores (, , F(1,7)=.048, p=.833, η2p=.007). A similar pattern was seen in the tDCS participants; those receiving verum stimulation first (n=10) showed a significant decrease in BDI from the start to the end of the study (, , F(1,9)=6.183, p=.035, η2p=.407) while no significant change was seen in those receiving sham first (n=7; , , F(1,6)=.199, p=.671, η2p=.032)(Figure 3B).
Figure 3. Beck Depression Index Scores.
Pre and post scores for transcranial alternating current stimulation (tACS) and transcranial direct current stimulation (tDCS) participants (A) and the stratified results (B). Significant differences from baseline are indicated on graphs. Significances in stratified data (B) were only displayed by verum-first participants.
3.2. Spielberger State and Trait Anxiety Index
tACS participants did not show a main effect of stimulation type on anxiety over their five iterations of STAI (baseline, during sham, during verum, after sham, and after verum) (F(1,15)=.983, p=.337, η2p=.062) and demonstrated a nonsignificant decrease in STAI scores from the start to the end of the study (, , t(15)=1.179, p=.257, d=0.295, 95% CI [−.211, .791]). tDCS participants, however, showed a main effect of STAI through a general decrease over the course of their STAI iterations (F(1,16)=7.128, p=.017, η2p=.308), as well as a significant decrease in STAI scores from the beginning to the end of the study (, , t(16)=2.945, p=.010, d=.714, 95% CI [.170, 1.240])(Figure 4A).
Figure 4. Spielberger State and Trait Anxiety Index Results.
Average anxiety scores (STAI) for transcranial alternating current stimulation (tACS) and transcranial direct current stimulation (tDCS) participants by stimulation type (A) and stratified data for sham-first and verum-first participants ordered by time (B). Significant post-hoc comparisons to baselines are indicated on graphs. All significances in stratified data (B) were displayed by verum-first participants.
The data stratified by stimulation order revealed a more complex pattern. tACS participants who received sham first again showed no main effect of stimulation type over their iterations (F(1,7)=.159, p=.702, η2p=.022) and no significant difference from the beginning to the end of the study (, , t(7)=−.136, p=.896, d=−.048, 95% CI [−.740, .647]). In contrast, when tACS participants received verum stimulation first they had a significant linear main effect of stimulation (F(1,7)=7.589, p=.028, η2p=.520) and a significant decrease of nearly 10 points from the beginning to the end of the study (, , t(7)=2.592, p=.036, d=.917, 95% CI [.057, 1.733]). Interestingly, tDCS participants who received sham first had a significant main effect of stimulation type (F(1,6)=11.929, p=.014, η2p=.665) but those who received verum first did not (F(1,9)=2.495, p=.149, η2p=.217)(Figure 4B). Neither group of tDCS participants (sham or verum first) showed a significant decrease in STAI pre-to-post study, though both were trending towards significance (sham first: , , t(6)=2.233, p=.067, d=.844, 95% CI [−.056, 1.695]; verum first: , , t(9)=1.985, p=.078, d=.628, 95% CI [−.069, 1.296])(Table 2).
Table 2. Pre and Post Scores.
Results from t-tests comparing transcranial alternating current stimulation (tACS) and transcranial direct current stimulation (tDCS) average post scores to average pre scores for Beck Depression Index (BD), State and Trait Anxiety Index (STAI), ‘Reappraise’ Valence Ratings, and ‘Reappraise’ Arousal ratings.
| tACS | tDCS | ||||||
|---|---|---|---|---|---|---|---|
| Total | Sham-First | Verum-First | Total | Sham-First | Verum-First | ||
| BDI | Pre | 25.188 | 25.250 | 25.130 | 25.235 | 23.857 | 26.200 |
| Post | 22.900 | 25.500 | 20.250 | 23.412 | 23.142 | 23.600 | |
| p-value | .035* | .833 | .002* | .060 | .671 | .035* | |
| STAI | Pre | 45.000 | 42.500 | 47.500 | 51.333 | 50.952 | 51.333 |
| Post | 40.625 | 43.330 | 37.917 | 42.550 | 42.381 | 42.667 | |
| p-value | .257 | .896 | .036* | .010* | .067 | .078 | |
| Reappraise Valence | First | 2.440 | 2.500 | 2.380 | 1.290 | 1.000 | 1.500 |
| Last | 1.060 | 1.250 | 0.880 | 0.820 | 1.140 | 0.600 | |
| p-value | < .001* | .060 | .009* | .239 | .853 | .041* | |
| Reappraise Arousal | First | 1.190 | 0.750 | 1.630 | 0.710 | 0.710 | 0.700 |
| Last | 0.380 | 0.130 | 0.630 | 0.240 | −0.140 | 0.500 | |
| p-value | .018* | .305 | .007* | .134 | .200 | .509 | |
3.3. Valence Ratings
All valence ratings were normalized, such that participants’ ratings for neutral stimuli were subtracted from their neutral, maintain, and reappraise scores. Following normalization, “look” scores for neutral stimuli became zero, maintain scores represented the difference between maintain scores and neutral scores, and reappraise is the difference between reappraisal scores and the neutral condition.
Both groups showed a significant main effect of stimulation type (tACS: F(1,15)=8.457, p=.011, η2p=.361; tDCS: F(1,16)=4.561, p=.049, η2p=.222) such that they displayed a general decrease in valence for both maintain and reappraise from the first half of the study to the second half. Additionally, tACS participants showed a significant interaction between stimulation type and task (F(1,15)=12.736, p=.003, η2p=.459); reappraise valence scores decreased more drastically during the second half of the study than did the maintain valence scores. This pattern was not seen in the tDCS participants (Figure 5A).
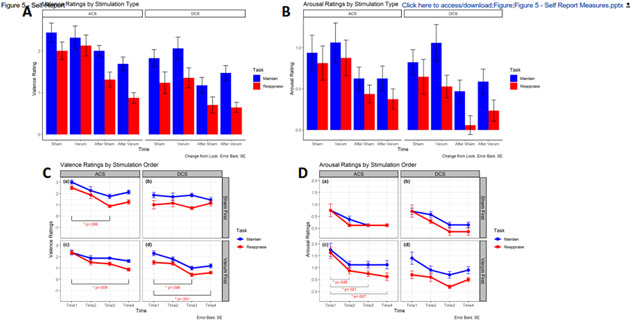
Figure 5. Valence and Arousal Scores.
Valence ratings (A and C) and arousal ratings (B and D) for transcranial alternating current stimulation (tACS) and transcranial direct current stimulation (tDCS) participants by stimulation type (A and B) and stratified by stimulation order and ordered by time (C and D). Significant post-hoc comparisons of ‘reappraise’ ratings to their baseline are displayed on graphs.
When stratified by stimulation order, both tACS groups had significant main effects of stimulation type, though those receiving sham first showed a linear main effect (F(1,7)=6.875, p=.034, η2p=.496) and those who received verum first had a quadratic main effect (F(1,7)=5.898, p=.046, η2p=.457) such that both groups displayed a general decrease in valence scores following their first session of stimulation. Of the tDCS participants, only those who received verum first showed a main effect of stimulation type (F(1,9)=7.149, p=.025, η2p=.443) such that valence scores decreased following their first session of stimulation. None of the four groups had a significant interaction between stimulation type and task (Figure 5C).
From the first to the final ERT iteration, tACS participants displayed a significant difference in the valence ratings following ‘reappraise’ stimulus blocks (, , t(15)=4.044, p=.001, d=1.011, 95% CI [.393, 1.607]), whereas the difference in tDCS participants was not significant (, , t(16)=1.224, p=.239, d=.297, 95% CI [−.194, .779]). Verum-first tACS participants drove this decrease (, , t(7)=3.550, p=.009, d=1.255, 95% CI [.286, 2.178]), though sham-first tACS participants trended towards significance (, , t(7)=2.236, p=.060, d=.791, 95% CI [−.033, 1.573]). A significant decrease in ‘reappraise’ valence scores was also seen in tDCS verum-first participants (, , t(9)=2.377, p=.041, d=.752, 95% CI [.028, 1.444]), but sham-first tDCS participants showed no change (, , t(6)=−.194, p=.853, d=−.073, 95% CI [−.812, .672])(Table 2).
3.4. Arousal Ratings
Arousal ratings were normalized similarly to valence ratings, such that ratings for neutral stimuli were subtracted from maintain and reappraise ratings.
Neither tACS nor tDCS groups showed large changes in arousal ratings across stimulation types (Figure 5B). This remained consistent even after data was stratified (Figure 5D). tACS participants did, however, display a significant interaction between stimulation type and task (F(1,15)=4.804, p=.045, η2p=.243) due to the gap between maintain and reappraise growing over the course of the study. This interaction was again driven by the verum-first tACS participants. These participants also had a significant interaction between task and stimulation type (F(1,7)=6.236, p=.041, η2p=.471), but sham-first tACS participants displayed no difference between maintain and reappraise arousal scores. These same patterns were not seen in the tDCS participants.
tACS participants displayed a significant decrease in their arousal ratings following ‘reappraise’ stimulus blocks from their first to final ERT iterations (, , t(15)=2.657, p=.018, d=.664, 95% CI [.112, 1.199]), though tDCS participants did not (, , t(16)=1.577, p=.134, d=.382, 95% CI [−.116, .870], In the stratified data, only verum-first tACS participants displayed a significant decrease in their ‘reappraise’ arousal scores (, , t(7)=3.742, p=.007, d=1.323, 95% CI [.330, 2.270]). Sham-first tACS participants (, , t(7)=1.106, p=.305, d=.391, 95% CI [−.343, 1.100]), sham-first tDCS participants (, , t(6)=1.441, p=.200, d=.545, 95% CI [−.274, 1.326]), nor verum-first tDCS participants (, , t(9)=.688, p=.509, d=.218, 95% CI [−.416, .840]) displayed significant trends (Table 2).
3.5. Reappraisal Ratings
tACS participants displayed a significant linear main effect of stimulation on their rating of reappraisal ability (F(1,15)=9.509, p=.008, η2p=.388). Both tACS sub-groups displayed a similar main effect, though that effect was linear for the sham-first group (F(1,7)=9.776, p=.017, η2p=.583) and quadratic for the verum-first group (F(1,7)=10.722, p=.014, η2p=.605), such that their feelings of reappraisal ability increased following their first session of verum or sham stimulation. Because tDCS participants’ reappraisal scores did not differ much across the task, it is unlikely that the effect seen in the tACS participants is due solely to a change over time.
3.6. Correlations
Before comparing changes in clinical scores with changes in ‘reappraise’ valence and arousal, BDI, STAI, ‘reappraise’ valence, and ‘reappraise’ arousal scores were transformed into percentages of change (delta score) within their respective scales. Among tACS participants, changes in BDI and STAI scores from the start to finish of the study (delta scores) correlated most at the trend level, with changes in valence ratings when participants were asked to reappraise (BDI: r(15)=.491, p=.053; STAI: r(15)=.452, p=.079). Neither BDI nor STAI delta scores correlated with changes in ‘reappraise’ arousal ratings (p>.1). tACS participants’ normalized ‘maintain’ scores in the ERT following verum stimulation also correlated with their global theta synchrony at that time (r(13)=.579, p=.038). tDCS participants’ STAI or BDI delta scores did not correlate with arousal or valence delta scores for ‘reappraise’ (p>.1).
In the stratified data, sham-first participants receiving either tACS or tDCS displayed no significant correlations between BDI or STAI with valence or arousal ratings (p>.1). Verum-first tACS participants, however, again demonstrated a trend between STAI delta scores and ‘reappraise’ valence delta scores (r(7)=.629, p=.095). All other correlations were not significant (p>.1).
4. Discussion
The results of this pilot study support the hypothesis that tES targeting ER has a beneficial effect on internalizing symptoms. Participants displayed improvements in ER, indicated by the lower ‘reappraise’ valence and arousal scores at the end of the study compared to the beginning, as well as improvements in clinical scores for depression and anxiety. Though correlations between clinical and regulatory scores were not significant, the trends seen are a promising indicator that there may be a link between the two. Further, in line with our hypotheses, participants who received tACS stimulation in particular showed significant improvements. Lastly, we found the intriguing lack-of-effect when participants received sham stimulation before verum stimulation, most notably in tACS participants.
The results from the analyses of pre-post/first-last changes in clinical scores as well as ‘reappraise’ valence and arousal ratings indicate that improvements in ER capability may be beneficial to internalizing psychopathology symptoms. Emotion regulation has been gaining increasing attention as a target within the transdiagnostic approach to studying and treating IPs and growing evidence indicates that treatments that provide ER training have merit in alleviating symptoms (Aldao et al., 2016; Barlow et al., 2016; Ehrenreich-May and Bilek, 2012; Goldman et al., 2006; Grosse Holtforth et al., 2019). In fact, a study by Berking and colleagues (Berking et al., 2013) found that patients who receive cognitive behavioral therapy with ER training achieved a 14% higher remission rate than participants who received therapy without ER training. The results of our study add weight to the argument that targeting ER may improve depression and anxiety symptoms.
Of further interest, using theta-targeting offset tACS appears to have had a stronger effect on ER, anxiety, and depression than dlPFC-targeting tDCS indicated by a greater number of significant changes. tDCS has been utilized more commonly in previous studies of ER (Conson et al., 2015; Feeser et al., 2014; Peña-Gómez et al., 2011) and depression (Berlim et al., 2013; Nitsche et al., 2009), but our results indicate that tACS, particularly offset tACS, is an under-researched methodology of tES for IPs. Here, tACS may be a superior form of tES because it can target specific frequency irregularities and modulate the EEG rhythms theorized to be involved in emotion dysregulation. tACS provides researchers with the ability to target irregularities within specific frequency bands (Helfrich et al., 2014) as opposed to having a general increasing or decreasing effect on underlying neuronal activity as with tDCS (Nitsche et al., 2009). These preliminary findings indicate that a frequency-specific approach to targeting ER and internalizing symptomology may achieve more dramatic benefits than a general increase in underlying neuronal activity.
Perhaps the most intriguing result from this study is the lack of effect when participants received sham first, particularly for anxiety and depression scores. Sham stimulation is designed to act as a placebo control, providing only enough stimulation to give participants the initial tingling sensation at the start of administration. The lack of effects when participants receive sham first begs the question, though, if sham stimulation is having an unintentional and unanticipated neurobiological effect. There has been limited mentioning of sham effects in the literature, but Kortuem et al.(Kortuem et al., 2019) and Splittgerber(Splittgerber et al., 2020) noted differences in participants’ responses to sham stimulation, such that they classified participants as “responders” and “non-responders” to sham and found that sham “responders” did not respond to verum stimulation. Though their stimulation parameters differ slightly from the current study, it is possible that this sham effect is occurring for similar reasons. One hypothesized mechanism of these sham effects can be explained by the Bienenstock-Cooper-Munro theory (Bienenstock et al., 1982) which posits that high levels of prior activity favor long-term depression and, subsequently, the 30 seconds of ramped up stimulation, our sham procedure, actually blocks the long-term potentiation which may occur from later verum stimulation. Another possible explanation, however, is that even though participants and researchers were blinded to stimulation condition, participants were 36% accurate in guessing sham stimulation and 76% accurate in guessing verum stimulation, and therefore sham may not have acted as a true placebo as intended. This potential sham effect calls into question the current standards for tES suggested by the National Institute of Mental Health (Bikson et al., 2018) which support counterbalanced tES study designs with sham inclusion. Such designs may be masking potential effects of stimulation and blunting others, which should be considered in future research.
Though the current study had several strengths including the double-blind design and a side-by-side analysis of stimulation methods, there were several limitations. First, without the inclusion of a baseline run of the emotion regulation task, it is impossible to determine overall improvement in ER capabilities, especially due to the pseudo-counterbalanced nature of the study design. Some participants’ initial run of the task was during verum stimulation and others’ was during sham, eliminating a true baseline for comparison. Second, because the two stimulation techniques had unique targets and effects, it is difficult to determine whether tACS outperformed tDCS or if instead theta was a better target than the dlPFC. Third, because all four sessions of stimulation were administered in one day, we suspect there were carry-over effects as outlined in the current literature(Heise et al., 2019; Klink et al., 2020; Mansouri et al., 2019; Splittgerber et al., 2020), meaning that the after-sham ERT was also after a previous verum stimulation. Additionally, because of the pseudo-counterbalanced design, and considering the discovered sham effects, there may have been a confounding, blunting effect from the sham stimulations on the verum-stimulation metrics. Fourth, though the study was double-blind, participants’ stimulation guess accuracies differed greatly from the 50% accuracy that would have been achieved due to simply guessing. Finally, the small sample sizes in this pilot study make it nearly impossible to draw strong conclusions. The effect sizes coupled with significant preliminary results, however, are promising and justify further exploration in future studies.
Despite these limitations, our results indicate that targeting EEG rhythms involved in ER may have beneficial effects on symptoms of depression and anxiety. Our results further indicate that offset theta-targeting tACS may be a promising intervention for future research and treatment of IPs. Lastly, the field of neuromodulation may need to re-evaluate how to incorporate sham stimulation as an effective placebo condition. Additional research is needed to validate the efficacy of theta-targeting offset tACS as an intervention for IPs, to understand the effect of current sham protocols on underlying neural activity, and to extend our understanding of the relationship between theta and ER in IPs by looking beyond theta coherence to theta dynamics through time-frequency and thought chart analyses(Xing et al., 2019).
Supplementary Material
Highlights.
Offset theta-tACS improves emotion regulation capabilities and depression scores.
Sham stimulation appears to have a blunting effect.
Verum-first participants show improvement in emotion regulation and clinical scores.
Acknowledgements
This work was supported by the National Institute of Health and the University of Illinois Chicago, IL, USA [grant number UL1TR002003]. The authors would like to thank Dr. Flavio Frohlich with Pulvinar Neuro for providing us with the transcranial stimulation device.
Abbreviations
- dlPFC
dorsolateral prefrontal cortex
- tDCS
transcranial direct current stimulation
- tACS
transcranial alternating current stimulation
- tES
transcranial electrical stimulation
- ER
emotion regulation
- ERT
emotion regulation task
- IPs
internalizing psychopathologies
- STAI
Spielberger State and Trait Anxiety Index
- BDI
Beck Depression Index
Footnotes
Declaration of Conflicts of Interest
We report that O.A. maintains an employment relationship with KeyWise AI, board membership with Embodied Labs and Blueprint, as well as consulting/advisory roles with SAGE Therapeutics Inc and Milken.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Abend R, Sar-el R, Gonen T, Jalon I, Vaisvaser S, Bar-Haim Y, et al. Modulating Emotional Experience Using Electrical Stimulation of the Medial-Prefrontal Cortex: A Preliminary tDCS-fMRI Study. Neuromodulation 2019;22:884–93. 10.1111/ner.12787. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aldao A, Gee DG, Reyes ADL, Seager I. Emotion regulation as a transdiagnostic factor in the development of internalizing and externalizing psychopathology: Current and future directions. Dev Psychopathol 2016;28:927–46. 10.1017/S0954579416000638. [DOI] [PubMed] [Google Scholar]
- Bar-Haim Y, Lamy D, Pergamin L, Bakermans-Kranenburg MJ, van IJzendoorn MH. Threat-related attentional bias in anxious and nonanxious individuals: A meta-analytic study. Psychol Bull 2007;133:1–24. 10.1037/0033-2909.133.1.1. [DOI] [PubMed] [Google Scholar]
- Barlow DH, Allen LB, Choate ML. Toward a Unified Treatment for Emotional Disorders – Republished Article. Behav Ther 2016;47:838–53. 10.1016/j.beth.2016.11.005. [DOI] [PubMed] [Google Scholar]
- Berking M, Ebert D, Cuijpers P, Hofmann SG. Emotion Regulation Skills Training Enhances the Efficacy of Inpatient Cognitive Behavioral Therapy for Major Depressive Disorder: A Randomized Controlled Trial. Psychother Psychosom 2013;82:234–45. 10.1159/000348448. [DOI] [PubMed] [Google Scholar]
- Berlim MT, Van den Eynde F, Daskalakis ZJ. Clinical utility of transcranial direct current stimulation (tDCS) for treating major depression: A systematic review and meta-analysis of randomized, double-blind and sham-controlled trials. J Psychiatr Res 2013;47:1–7. 10.1016/j.jpsychires.2012.09.025. [DOI] [PubMed] [Google Scholar]
- Bienenstock L, Cooper N, Munro W. Theory for the development of neuron selectivity: orientation specificity and binocular interaction in visual cortex. J Neurosci 1982;2:17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bikson M, Brunoni AR, Charvet LE, Clark VP, Cohen LG, Deng Z-D, et al. Rigor and reproducibility in research with transcranial electrical stimulation: An NIMH-sponsored workshop. Brain Stimulat 2018;11:465–80. 10.1016/j.brs.2017.12.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Campbell-Sills L, Barlow DH, Brown TA, Hofmann SG. Effects of suppression and acceptance on emotional responses of individuals with anxiety and mood disorders. Behav Res Ther 2006;44:1251–63. 10.1016/j.brat.2005.10.001. [DOI] [PubMed] [Google Scholar]
- Chan CY, Nicholson C. Modulation by applied electric fields of Purkinje and stellate cell activity in the isolated turtle cerebellum. J Physiol 1986;371:89–114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Conson M, Errico D, Mazzarella E, Giordano M, Grossi D, Trojano L. Transcranial Electrical Stimulation over Dorsolateral Prefrontal Cortex Modulates Processing of Social Cognitive and Affective Information. PLoS One 2015;10:e0126448. 10.1371/journal.pone.0126448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ehrenreich-May J, Bilek EL. The Development of a Transdiagnostic, Cognitive Behavioral Group Intervention for Childhood Anxiety Disorders and Co-Occurring Depression Symptoms. Cogn Behav Pract 2012;19:41–55. 10.1016/j.cbpra.2011.02.003. [DOI] [Google Scholar]
- Etkin A, Büchel C, Gross JJ. The neural bases of emotion regulation. Nat Rev Neurosci 2015;16:693–700. 10.1038/nrn4044. [DOI] [PubMed] [Google Scholar]
- Feeser M, Prehn K, Kazzer P, Mungee A, Bajbouj M. Transcranial Direct Current Stimulation Enhances Cognitive Control During Emotion Regulation. Brain Stimulat 2014;7:105–12. 10.1016/j.brs.2013.08.006. [DOI] [PubMed] [Google Scholar]
- Fitzgerald JM, MacNamara A, DiGangi JA, Kennedy AE, Rabinak CA, Patwell R, et al. An electrocortical investigation of voluntary emotion regulation in combat-related posttraumatic stress disorder. Psychiatry Res Neuroimaging 2016;249:113–21. 10.1016/j.pscychresns.2015.12.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goldman RhondaN, Greenberg LeslieS, Angus L.. The effects of adding emotion-focused interventions to the client-centered relationship conditions in the treatment of depressionResults from this paper were presented at the Society for Psychotherapy Research, Chicago, Illinois, June 2000. Psychother Res 2006;16:537–49. 10.1080/10503300600589456. [DOI] [Google Scholar]
- Grosse Holtforth M, Krieger T, Zimmermann J, Altenstein-Yamanaka D, Dörig N, Meisch L, et al. A randomized-controlled trial of cognitive-behavioral therapy for depression with integrated techniques from emotion-focused and exposure therapies. Psychother Res 2019;29:30–44. 10.1080/10503307.2017.1397796. [DOI] [PubMed] [Google Scholar]
- Hajcak G, Nieuwenhuis S. Reappraisal modulates the electrocortical response to unpleasant pictures. Cogn Affect Behav Neurosci 2006;6:291–7. 10.3758/CABN.6.4.291. [DOI] [PubMed] [Google Scholar]
- Heise K-F, Monteiro TS, Leunissen I, Mantini D, Swinnen SP. Distinct online and offline effects of alpha and beta transcranial alternating current stimulation (tACS) on continuous bimanual performance and task-set switching. Sci Rep 2019;9:3144. 10.1038/s41598-019-39900-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Helfrich RF, Schneider TR, Rach S, Trautmann-Lengsfeld SA, Engel AK, Herrmann CS. Entrainment of Brain Oscillations by Transcranial Alternating Current Stimulation. Curr Biol 2014;24:333–9. 10.1016/j.cub.2013.12.041. [DOI] [PubMed] [Google Scholar]
- Honkalampi K, Hintikka J, Laukkanen E, Viinamäki JLH. Alexithymia and Depression: A Prospective Study of Patients With Major Depressive Disorder. Psychosomatics 2001;42:229–34. 10.1176/appi.psy.42.3.229. [DOI] [PubMed] [Google Scholar]
- Jaušovec N, Jaušovec K. Increasing working memory capacity with theta transcranial alternating current stimulation (tACS). Biol Psychol 2014;96:42–7. 10.1016/j.biopsycho.2013.11.006. [DOI] [PubMed] [Google Scholar]
- Kassel JD, Bornovalova M, Mehta N. Generalized expectancies for negative mood regulation predict change in anxiety and depression among college students. Behav Res Ther 2007;45:939–50. 10.1016/j.brat.2006.07.014. [DOI] [PubMed] [Google Scholar]
- Kessler RC. The Costs of Depression. Psychiatr Clin North Am 2012;35:1–14. 10.1016/j.psc.2011.11.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kinney KL, Burkhouse KL, Klumpp H. Self-report and neurophysiological indicators of emotion processing and regulation in social anxiety disorder. Biol Psychol 2019;142:126–31. 10.1016/j.biopsycho.2019.01.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klink K, Peter J, Wyss P, Klöppel S. Transcranial Electric Current Stimulation During Associative Memory Encoding: Comparing tACS and tDCS Effects in Healthy Aging. Front Aging Neurosci 2020;12:66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Konnopka A, Leichsenring F, Leibing E, König H-H. Cost-of-illness studies and cost-effectiveness analyses in anxiety disorders: A systematic review. J Affect Disord 2009;114:14–31. 10.1016/j.jad.2008.07.014. [DOI] [PubMed] [Google Scholar]
- Kortuem V, Kadish NE, Siniatchkin M, Moliadze V. Efficacy of tRNS and 140 Hz tACS on motor cortex excitability seemingly dependent on sensitivity to sham stimulation. Exp Brain Res 2019;237:2885–95. 10.1007/s00221-019-05640-w. [DOI] [PubMed] [Google Scholar]
- Lang PJ, Bradley MM, Cuthbert BN. International Affective Picture System (IAPS): Technical Manual and Affective Ratings. NIMH Center for the Study of Emotion and Attention, Gainesville, 1997, pp 39–58. [Google Scholar]
- Mansouri F, Shanbour A, Mazza F, Fettes P, Zariffa J, Downar J. Effect of Theta Transcranial Alternating Current Stimulation and Phase-Locked Transcranial Pulsed Current Stimulation on Learning and Cognitive Control. Front Neurosci 2019;13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nitsche MA, Boggio PS, Fregni F, Pascual-Leone A. Treatment of depression with transcranial direct current stimulation (tDCS): A Review. Exp Neurol 2009;219:14–9. 10.1016/j.expneurol.2009.03.038. [DOI] [PubMed] [Google Scholar]
- Pahor A, Jaušovec N. The effects of theta transcranial alternating current stimulation (tACS) on fluid intelligence. Int J Psychophysiol 2014;93:322–31. 10.1016/j.ijpsycho.2014.06.015. [DOI] [PubMed] [Google Scholar]
- Peña-Gómez C, Vidal-Piñeiro D, Clemente IC, Pascual-Leone Á, Bartrés-Faz D. Down-Regulation of Negative Emotional Processing by Transcranial Direct Current Stimulation: Effects of Personality Characteristics. PLoS One 2011;6:e22812. 10.1371/journal.pone.0022812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Polanía R, Nitsche MA, Korman C, Batsikadze G, Paulus W. The Importance of Timing in Segregated Theta Phase-Coupling for Cognitive Performance. Curr Biol 2012;22:1314–8. 10.1016/j.cub.2012.05.021. [DOI] [PubMed] [Google Scholar]
- Price RB, Eldreth DA, Mohlman J. Deficient prefrontal attentional control in late-life generalized anxiety disorder: an fMRI investigation. Transl Psychiatry 2011;1:e46–e46. 10.1038/tp.2011.46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sanchez A, Vanderhasselt M-A, Baeken C, De Raedt R. Effects of tDCS over the right DLPFC on attentional disengagement from positive and negative faces: An eye-tracking study. Cogn Affect Behav Neurosci 2016;16:1027–38. 10.3758/s13415-016-0450-3. [DOI] [PubMed] [Google Scholar]
- Sela T, Kilim A, Lavidor M. Transcranial alternating current stimulation increases risk-taking behavior in the balloon analog risk task. Front Neurosci 2012;6:22–22. 10.3389/fnins.2012.00022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Splittgerber M, Suwelack JH, Kadish NE, Moliadze V. The Effects of 1 mA tACS and tRNS on Children/Adolescents and Adults: Investigating Age and Sensitivity to Sham Stimulation. Neural Plast 2020;2020:1–14. 10.1155/2020/8896423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Strüber D, Rach S, Trautmann-Lengsfeld SA, Engel AK, Herrmann CS. Antiphasic 40 Hz Oscillatory Current Stimulation Affects Bistable Motion Perception. Brain Topogr 2014;27:158–71. 10.1007/s10548-013-0294-x. [DOI] [PubMed] [Google Scholar]
- Tseng P, Iu K-C, Juan C-H. The critical role of phase difference in theta oscillation between bilateral parietal cortices for visuospatial working memory. Sci Rep 2018;8:349. 10.1038/s41598-017-18449-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xing M, Lee H, Morrissey Z, Chung MK, Phan KL, Klumpp H, et al. Altered dynamic electroencephalography connectome phase-space features of emotion regulation in social anxiety. Neuroimage 2019;186:338–49. 10.1016/j.neuroimage.2018.10.073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xing M, Tadayonnejad R, MacNamara A, Ajilore O, DiGangi J, Phan KL, et al. Resting-state theta band connectivity and graph analysis in generalized social anxiety disorder. Neuroimage Clin 2017;13:24–32. 10.1016/j.nicl.2016.11.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.