Abstract
Background
Respiratory distress syndrome (RDS) is one of the commonest complication preterm neonates suffer and accounts for a significant morbidity and mortality in low and middle income countries (LMICs). Addressing RDS is therefore crucial in reducing the under 5 mortality in LMICs. This study aimed at describing early outcomes (death/survival) of preterm neonates with RDS and identify factors associated with the outcomes among neonates admitted at Muhimbili national hospital, Tanzania.
Methods
Between October 2019 and January 2020 we conducted a prospective study on 246 preterm neonates with RDS at Muhimbili National Hospital. These were followed up for 7 days. We generated Kaplan–Meier survival curve to demonstrate time to death. We performed a cox regression analysis to ascertain factors associated with outcomes. The risk of mortality was analyzed and presented with hazard ratio. Confidence interval of 95% and P-value less than 0.05 were considered as significant.
Results
Of the 246 study participants 51.6% were male. The median birth weight and gestational age of participants (Inter-Quartile range) was 1.3 kg (1.0, 1.7) and 31 weeks (29, 32) respectively. Majority (60%) of study participants were inborn. Only 11.4% of mothers of study participants received steroids. Of the study participants 49 (20%) received surfactant. By day 7 of age 77/246 (31.3%) study participants had died while the majority of those alive 109/169 (64.5%) continued to need some respiratory support. Factors independently associated with mortality by day 7 included birth weight of < 1500 g (AHR = 2.11 (1.16–3.85), CI95%; p = 0.015), lack of antenatal steroids (AHR = 4.59 (1.11–18.9), CI95%; p = 0.035), 5th minute APGAR score of < 7 (AHR = 2.18 (1.33–3.56), CI95%; p = 0.002) and oxygen saturation < 90% at 6 hours post admission (AHR = 4.45 (1.68–11.7), CI95%; p = 0.003).
Conclusion
Our study reports that there was high mortality among preterm neonates admitted with RDS mainly occurring within the first week of life. Preterm neonates with very low birth weight (VLBW), whose mother did not receive antenatal steroid, who scored < 7 at 5th minute and whose saturation was < 90% at 6 hours were at higher risk of dying. There is need to scale up antenatal corticosteroids, neonatal resuscitation training and saturation monitoring among preterm neonates with RDS.
Keywords: Prematurity, Respiratory distress syndrome, Mortality
Background
Respiratory distress syndrome (RDS) is among the major complications of preterm birth [1]. The risk of RDS is related to production of surfactant in the lungs. RDS increases with degree of prematurity and its frequency decreases significantly after 37 weeks of gestation [2].
The clinical signs of RDS manifest within the first minutes or hours after birth and progress over the first 48 to 72 hours thereafter, begins to resolve. The subsequent improvement coincides with increased production of endogenous surfactant resulting in resolution of symptoms by one week of age [3].
The diagnosis of RDS is based on clinical findings of a preterm infant with onset of progressive respiratory distress shortly after birth and a characteristic chest radiograph, which demonstrates low lung volume and diffuse reticulogranular ground glass appearance with air bronchograms [4].
The management of RDS has evolved over the past few decades in the developed countries [5, 6]. Despite the increase in RDS cases, in developed countries the infant mortality rate from RDS has significantly reduced but in resource restricted setting RDS is still among the commonest cause of mortality among preterm neonates [5, 7].
The findings of previous studies in the globe revealed that several factors are associated with mortality in preterm neonates with RDS. These include: gestational age [8–12], birth weight [9–12], sex [13, 14], APGAR score [15, 16], hypothermia [17], Silverman Andersen score (SAS) [10], place of delivery [12, 18], Antenatal Corticosteroids (ACS) [8, 11, 19–21], mode of delivery [11, 12], Continuous positive airway pressure (CPAP) [22–25], and surfactant [23].
Despite interventions recommended by WHO to improve outcome of preterm neonates, mortality of preterm neonates in low- and middle-income countries remains significant. Tanzania is among countries with high preterm births and hence of RDS. Neonatal mortality contributes a significant proportion to under 5 deaths. We conducted this study to ascertain outcome of preterm neonates admitted with RDS and identifying factors associated with the outcomes. This knowledge is important in identifying area needing improvement to reduce preterm neonatal deaths and hence under 5 deaths.
Methods
Setting
A prospective study was conducted at the newborn intensive care unit (NICU) of Muhimbili National Hospital (MNH), Dar-es-Salaam, Tanzania. MNH is a national tertiary level referral hospital for Tanzania and a teaching hospital for Muhimbili University of Health and Allied Sciences. Neonatal unit is located at the maternity block, second floor, it has two wards one for preterm (ward 37) and another for term babies (ward 36). In preterm ward there are 4 cubes, with a total bed capacity of 60 beds. At the time of the study the unit has wall mounted oxygen, 3 mechanical ventilators, 5 radiant warmers, 5 wall cardiac monitors, 5 diamedic continuous positive airway pressure (CPAP), 10 high flow CPAP and 20 infusion pumps. It has well trained staffs for premature care, 7 specialist, 3 registered medical officers, 3–5 Paediatric resident’s, minimum of 4 intern medical doctors and well-trained nurses. MNH has approximately 10,000 neonates born per year both term and preterm. Admissions in preterm unit is approximately 4–10 babies per day with monthly admission rate of 120–280 neonates and an annual admission rate of 1440–3360 neonates, of which RDS is one of the admission diagnoses. MNH neonatal unit therefore was selected purposively because most preterm infants with RDS are admitted here.
Inborn preterm neonates are received by midwives in the delivery rooms and transferred immediately to NICU. Delayed cord clamping is hardly observed for preterm infants and no CPAP is provided in the delivery rooms.
Preterm neonates receive intravenous fluids, antibiotics, and aminophylline. They are cared for on open radiant warmers with on many occasions one warmer hosting 2-3infants. Preterm neonates with SAS score of > 3 are commenced on CPAP with blended oxygen but not with heated air. Infants needing < 30% oxygen with SAS score < 3 are transitioned to nasal prong oxygen 2 l/min and gradually weaned off. Surfactant is given to infants with severe distress whose parents can purchase.
Tanzania is a lower-middle-income country, it has an estimated population of about 60 million people. Health services in the United Republic of Tanzania are delivered through a decentralized system. Access to health care in Tanzania is still a challenge especially in women. Health insurance coverage is still low approximately 32%.
Study population
All preterm neonates < 24 hours old with clinical signs of RDS. Clinical signs of RDS which starts in < 6 hours after birth were tachypnea, nasal flaring, expiratory grunting, intercostal, subcostal, and subxiphoid retractions, and cyanosis.
Variables of study
Outcome variables
The outcome for this study was Death or Survival at 7 days of life.
Independent variables
In this study, maternal and Preterm neonates variables were considered. Antenatal steroid, Place of delivery, Mode of delivery, pPROM> 18 hours, maternal fever. Gestational age, Birth weight, Sex, APGAR score at 5th minute, Age at admission, SAS at admission and 6 hours, Oxygen saturation at commence of care and at 6 hours, Body temperature, random blood sugar, Surfactant.
Procedure
Between October 2019 and January 2020, all preterm neonates < 24 hours of age whose respiratory distress commenced < 6 hours after birth were enrolled. New Ballard score [26] was used to determine gestational age and then preterm neonates were categorized in two groups for analysis (< 32 weeks and > 32 weeks), while Silverman Andersen score (SAS) [27] was used to grade the severity of RDS. Neonates with major anomalies and those with severe birth asphyxia (APGAR score < 4 at 5 min) were excluded.
All study variables were obtained from maternal history, antenatal cards and NICU records. These were entered in the data sheet.
Daily follow-up was done, vital signs and oxygen saturations were observed daily or obtained from the nursing charts.
Data management and analysis
The data was analysed using SPSS software packages version 23.0. Frequency distribution and Kaplan–Meier survival curves were used to show pattern of death in 7 days for birth weight and gestational age. Independent and adjusted relationships of different predictors with preterm neonates’ survival were assessed with Cox regression model. Factors associated with mortality in crude model with a p value < 0.05 were entered in the multivariable Cox regression model to identify and quantify predictors of deaths while controlling for potential confounder. The risk of mortality was explored and presented with hazard ratio and 95% confidence interval. P-value less than 0.05 was considered as significant.
Results
The median birth weight of participants (Inter-Quartile range) was 1.3 (1.0, 1.7) kilograms. Of the study participants 79/246 (32%) were small for gestational age (SGA). The median gestational age (Inter-Quartile range) was 31 (29, 32) weeks. The median age of participants (Inter-Quartile range) was 1.3 (0.7, 5.5) hours at admission and median age at start of respiratory support (Inter-Quartile range) was 1.66 (0.75, 6.41) hours.
Most 240 (97.6%) preterm neonates presented with respiratory rate > 60breath/minute and nasal flaring at recruitment. Other clinical presentation were subcostal retractions 202 (82.1%) and grunting 131 (53.3%). (Table 1).
Table 1.
Demographic and baseline characteristics of study participants
| Characteristics | N (%) |
|---|---|
| Sex | |
| Male | 127 (51.6) |
| Female | 119 (48.4) |
| Gestational Age (weeks) | |
| 26 to < 28 | 9 (3.7) |
| 28 to ≤32 | 132 (53.7) |
| > 32 to 37 | 105 (42.7) |
| SGA | |
| Yes | 79 (32.1) |
| No | 167 (67.9) |
| Birth weight (grams) | |
| < 1500 | 152 (61.8) |
| ≥ 1500 | 94 (38.2) |
| 5th minute APGAR Score | |
| > 7 | 156 (63.4) |
| 4–7 | 80 (32.8) |
| Not known | 10 (3.8) |
| Age at admission | |
| < 1 hour | 89 (36.2) |
| ≥ 1 hour-24 hours | 157 (63.8) |
| Age at start of respiratory support | |
| < 1 hour | 77 (31.3) |
| ≥ 1 hour | 169 (68.7) |
| Respiratory support initiated | |
| CPAP | 158 (64.2) |
| Oxygen by Nasal prongs | 82 (33.3) |
| Mechanical ventilation | 6 (2.4) |
| Surfactant | |
| Yes | 49 (19.9) |
| No | 197 (80.1) |
| Maternal Age | |
| < 20 years | 21 (8.5) |
| 20–34 years | 175 (71.2) |
| > 35 years | 50 (20.3) |
| Parity | |
| Primipara | 79 (32.1) |
| Multipara | 167 (67.9) |
| Antenatal steroid (Dexamethasone) | |
| Received | 28 (11.4) |
| Not received | 218 (88.6) |
| Prolonged rapture of membranes > 18 hours (PROM) | |
| Yes | 82 (33.3) |
| No | 164 (66.7) |
| Maternal Fever | |
| Yes | 33 (13.4) |
| No | 213 (86.6) |
| Maternal Diabetes | |
| Yes | 2 (0.8) |
| No | 244 (99.2) |
At 6 hours of admission the majority 56.1% had moderate to severe respiratory distress, a third 32.1% were hypothermic and 11.4% were hypoglycemic (Table 2).
Table 2.
Vitals at admission and at 6 hours after admission
| Characteristics | At admission N (%) | At 6 hours N (%) |
|---|---|---|
| Silverman Andersen score | ||
| ≥ 6 (severe) | 66 (26.8) | 50 (20.3) |
| 4 to 5 (moderate) | 83 (33.7) | 88 (35.8) |
| 1 to 3 (mild) | 97 (39.4) | 108 (43.9) |
| Oxygen Saturation | ||
| ≥ 90% | 156 (63.4) | 238 (96.7) |
| < 90% | 90 (36.6) | 8 (3.3) |
| Temperature (°c) | ||
| ≥ 36.5 | 109 (44.3) | 167 (67.9) |
| < 36.5 | 137 (55.7) | 79 (32.1) |
| Random blood sugar (mmol/L) | ||
| ≥ 2.6 | 172 (69.9) | 218 (88.6) |
| < 2.6 | 74 (30.1) | 28 (11.4) |
The overall mortality was 31.3% there as 77/246, 35 (45.5%) deaths occurred in the first 72 hours. Mortality among preterm neonates with birth weight < 1500 g was 40% and those with birth weight ≥ 1500 g was 17%. Among preterm neonates who were alive in the first week of life, 109 (64.5%) were still in need of respiratory support.
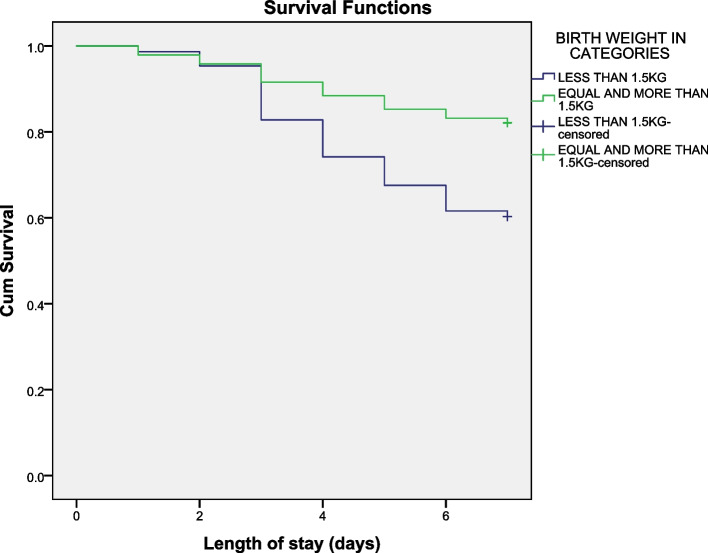
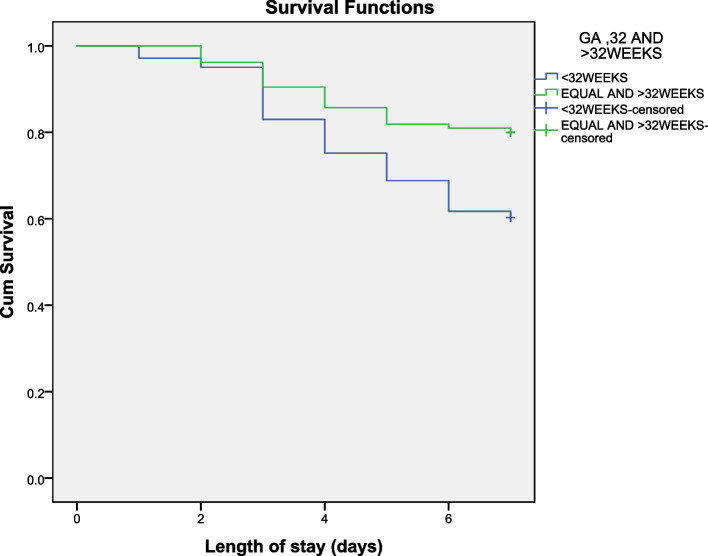
From the log rank test, survival distribution of different birth weight categories was statistically significant (P = < 0.001) indicating that those with birth weight ≥ 1500 g were more likely to survive (Fig. 1). From the log rank test, survival distribution of different GA categories was statistically significant (P = 0.002) indicating that having a GA above 32 weeks was significantly associated with better survival (Fig. 2). On day 7, the probability of survival of preterm neonate who had GA above 32 weeks was 80% and for a preterm neonate who had GA < 32 weeks was 60% (Fig. 3).
Fig. 1.
Kaplan-Meier survival pattern of Preterm Neonates and Birth weight
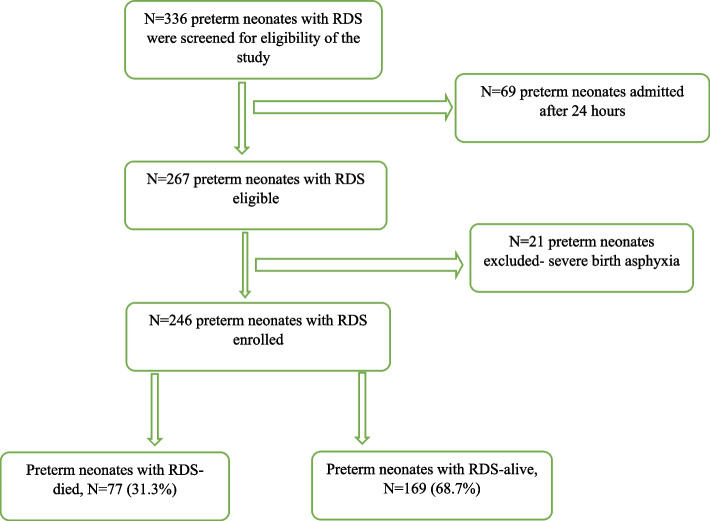
Fig. 2.
Patient Recruitment
Fig. 3.
Kaplan-Meier survival pattern of Preterm Neonates and Gestational Age
Cox proportional hazards regression models
Variables with P-value less than 0.05 in crude model were included in the Cox proportional hazards regression model. Gestational age, birth weight, Apgar score at 5th minute, age at admission, temperature at 6 hours of admission, antenatal steroids, oxygen saturation at admission and at 6 hours post admission were variables included in multivariate Cox regression model. After adjusting for confounders, we found that hazards of preterm mortality by day 7 of age was high for preterm neonates with birth weight < 1500 g (AHR = 2.11 (1.16–3.85), CI95%; p = 0.015), preterms whose mother’s lack antenatal steroids (AHR = 4.59 (1.11–18.9), CI95%; p = 0.035), preterm neonates with APGAR score at 5th minute of 4–7 (AHR = 2.18 (1.33–3.56), CI95%; p = 0.002) and preterm neonates with oxygen saturation < 90% at 6 hours post admission (AHR = 4.45 (1.68–11.7), CI95%; p = 0.003), (Table 3).
Table 3.
Determinants of Survival in Preterm neonates with Respiratory distress syndrome
| Factors | Preterm Survival Status | |||||
|---|---|---|---|---|---|---|
| Censored N (%) | Died N (%) | CHR 95% CI | P-value | AHR 95% CI | P-value | |
| Sex | ||||||
| Male | 93 (73.2) | 34 (26.8) | 1.45 (0.92–2.27) | 0.11 | ||
| Female | 76 (63.9) | 43 (36.1) | 1 | – | – | |
| Gestational Age | ||||||
| < 32 weeks | 85 (60.3) | 56 (39.7) | 2.16 (1.31–3.56) | 0.003* | 1.61 (0.91–2.85) | 0.10 |
| ≥ 32–37 weeks | 84 (80) | 21 (20) | 1 | 1 | ||
| Birth weight (grams) | ||||||
| < 1500 | 91 (59.9) | 61 (40.1) | 2.45 (1.43–4.20) | 0.001* | 2.11 (1.16–3.85) | 0.015* |
| ≥ 1500 | 78 (83) | 16 (17) | 1 | 1 | ||
| 5th minute APGAR Score | ||||||
| > 7 | 116 (74.4) | 40 (25.6) | 1 | 1 | ||
| 4–7 | 48 (60) | 32 (40) | 1.71 (1.07–2.72) | 0.024* | 2.18 (1.33–3.56) | 0.002* |
| Not known | 5 (50) | 5 (50) | 2.22 (0.87–5.61) | 0.09 | 1.97 (0.74–5.21) | 0.17 |
| Age at admission | ||||||
| < 1 hour | 69 (77.5) | 20 (22.5) | 1 | 1 | ||
| ≥ 1 hour | 100 (63.7) | 57 (36.3) | 1.73 (1.04–2.88) | 0.036* | 1.56 (0.91–2.67) | 0.10 |
| Admission saturation | ||||||
| ≥ 90% | 115 (73.7) | 41 (26.3) | 1 | 1 | ||
| < 90% | 54 (60) | 36 (40) | 1.69 (1.08–2.64) | 0.022* | 1.50 (0.95–2.38) | 0.09 |
| Admission SAS | ||||||
| ≥ 6 (severe) | 50 (75.8) | 16 (24.2) | 0.79 (0.43–1.46) | 0.46 | ||
| 4 to 5 (moderate) | 51 (61.4) | 32 (38.6) | 1.36 (0.82–2.25) | 0.23 | ||
| 1 to 3 (mild) | 68 (70.1) | 29 (29.9) | 1 | – | – | |
| Admission temp (°C) | ||||||
| ≥ 36.5 | 81 (74.3) | 28 (25.7) | 1 | – | – | |
| < 36.5 | 88 (64.2) | 49 (35.8) | 1.46 (0.92–2.32) | 0.11 | ||
| Admission RBS | ||||||
| ≥ 2.6 mmol/l | 124 (72) | 48 (28) | 1 | – | – | |
| < 2.6 mmol/l | 45 (61) | 29 (39) | 1.54 (0.97–2.43) | 0.07 | ||
| 6th hour temp (°C) | ||||||
| ≥ 36.5 | 123 (73.7) | 44 (26.3) | 1 | 1 | ||
| < 36.5 | 46 (58.2) | 33 (41.8) | 1.67 (1.07–2.63) | 0.025* | 1.02 (0.63–1.66) | 0.92 |
| 6Th hour RBS | ||||||
| ≥ 2.6 mmol/l | 151 (69.3) | 67 (30.7) | 1 | – | – | |
| < 2.6 mmol/l | 18 (64.3) | 10 (35.7) | 1.23 (0.63–2.38) | 0.55 | ||
| 6th hour saturation | ||||||
| ≥ 90% | 166 (69.7) | 72 (30.3) | 1 | 1 | – | |
| < 90% | 3 (37.5) | 5 (62.5) | 3.03 (1.22–7.52) | 0.017* | 4.45 (1.68–11.7) | 0.003* |
| 6th hour SAS | ||||||
| ≥ 6 (severe) | 37 (74) | 13 (26) | 0.87 (0.46–1.67) | 0.68 | ||
| 4–5 (moderate) | 56 (63.6) | 32 (36.4) | 1.25 (0.76–2.04) | 0.38 | ||
| 1–3 (mild) | 76 (70.4) | 32 (29.6) | 1 | – | – | |
| Surfactant | ||||||
| Yes | 38 (77.6) | 11 (22.4) | 1 | – | – | |
| No | 131 (66.5) | 66 (33.5) | 1.61 (0.85–3.0) | 0.15 | ||
| Place of Delivery | ||||||
| MNH | 109 (73.2) | 40 (26.8) | 1 | – | – | |
| Others | 60 (62) | 37 (38) | 1.47 (0.94–2.31) | 0.09 | ||
| Mode of Delivery | ||||||
| Vaginal | 108 (66.7) | 54 (33.3) | 1 | – | – | |
| Caesarian Section | 61 (2.6) | 23 (27.4) | 0.82 (0.50–1.34) | 0.43 | ||
| Antenatal steroid | ||||||
| Received | 26 (92.9) | 2 (7.1) | 1 | 1 | ||
| Not received | 143 (65.6) | 75 (34.4) | 5.35 (1.31–21.8) | 0.019* | 4.59 (1.11–18.9) | 0.035* |
| PROM > 18 hours | ||||||
| Yes | 51 (62.2) | 31 (37.8) | 1.38 (0.88–2.18) | 0.17 | ||
| No | 118 (72) | 46 (28) | 1 | – | – | |
| Maternal Fever | ||||||
| Yes | 20 (60.6) | 13 (39.4) | 1.42 (0.78–2.58) | 0.25 | ||
| No | 149 (70) | 64 (30) | 1 | – | – | |
* = significant at P < 0.05, CHR Crude Hazard ratio, AHR Adjusted Hazard ratio, 95% CI 95% Confidence Interval
Discussion
This study aimed to determine the early outcomes and associated factors among preterm neonates with RDS admitted at MNH. Results from this study shows that the mortality rate during the first week of life was 31.3% and Preterm neonates with very low birth weight (VLBW), whose mother did not receive antenatal steroid, who scored APGAR < 7 at 5th minute and whose oxygen saturation was < 90% at 6 hours were at higher risk of dying.
The mortality rate of preterm neonates with RDS have a wide variation ranging from 6.5 to 88% [22, 28–30]. Our findings are consistent with several studies in Low Income Countries [30, 31]. A previous study in the same unit showed higher rates of mortality in premature babies due to RDS mostly occurring in the first week of life 88% and another study in a different region was 81.1% [28, 32]. Comparing with this current study the mortality has reduced. This is possibly because of the introduction of CPAP [33] as other supporting care has been the same except earlier on there was no easy availability of CPAP.
In this study birth weight was found to be independently associated with outcome death. Mortality was higher among neonates with weight < 1500 g (40%) compared to those > 1500 g (17%), this might be because those < 1500 g were more premature and had more severe disease than those > 1500 g [2].
We observed that preterm neonates with low oxygen saturation < 90% were at higher risk for mortality (AHR = 4.45 (1.68–11.7), CI95%; p = 0.003). This is similar to one study done by Sathenahalli et al. [12] Infants with RDS who fail to maintain saturation were likely in respiratory failure. Low oxygen saturation target has previously been demonstrated to be associated with high mortality [34].
Infants with RDS and Apgar score at 5th minute of 4–7 had higher mortality rate of 40% compared to those with Apgar score at 5th minute of more than 7 which was 25.6%. Thus, low Apgar score at 5th minute was an independent predictor of mortality. This finding is comparable to other two studies which showed increase in RDS and mortality in preterm babies with low Apgar score [15, 16]. This may be because of the following two reasons, one is acute lung injury caused by severe birth asphyxia decreases the synthesis and secretion of pulmonary surfactant and two hypoxia which inhibits the activity of surfactant and even leads to its inactivation. Moreover, severe asphyxia can increase mortality and thus, it is important for us to tailor high impact maternal and preterm care interventions to reduce mortality [35].
Only 2/28 (7.1%) study participants whose mother received steroids died and 75/200 (34.4%) of those whose mothers did not receive steroids died. These findings were comparable with a study done in India by Palod P. H et al. [11]. Similar observation from other studies have also shown that lack of antenatal steroids were associated with higher mortality rates in premature babies with RDS [8, 21, 36, 37]. This can be explained as steroids help in lung maturation especially in extreme to very preterm babies, and thus improving lung function. Also contributes to less severe disease as it has been shown that use of antenatal steroids reduces the incidence of RDS and mortality in preterm neonates [38]. Again, Initiative of implementing the care bundle which was done in Tanzania in four hospitals, including MNH for preterm infants was associated with significant reduction in neonatal mortality presumably by reducing respiratory morbidity with antenatal steroids and minimizing infection with antibiotics [21]. In this current study only 11.4% received antenatal steroids this might be due to gaps in implementation of evidence-based guideline at various levels, poor identification and follow up of women at risk of preterm delivery and high level of spontaneous preterm deliveries in otherwise normal pregnancies.
Preterm neonates who received surfactant had less mortality; however, it was not an independent predictor of mortality. These finding are similar to the study done by Niknafs et al. [25] which showed no difference in outcome between two groups of surfactants. In contrast to other study done by Hala et al. [23] showed a significant reduction in mortality by 50% with administration of timely surfactant. In current study there was slight decrease in mortality among the 20% preterm neonates who received surfactant this might be due to majority received surfactant late but also cost implication which required parental consent.
We still observe high mortality in our setting (LMIC) as compared to high income countries. Our setting is equipped but overwhelmed by number of premature babies delivered with complications, and thus compromising quality of care and availability of resources. Low uptake of ACS as demonstrated from our findings it talks about our poor health care access as well as lack of surfactant due to cost implication which parents need to cover while most of our mothers can’t afford.
Study limitations
Lack of chest radiography to confirm the diagnosis, severity, and possible complications such as air leaks was lacking, and this would affect the interpretation of some findings.
All preterm neonates are managed empirically with antibiotics for presumed sepsis without appropriate investigations hence difficulty to delineate between sepsis and RDS.
Lack of routine cranial scan on these vulnerable infants made it difficult to ascertain other contributors to poor outcome including death.
Conclusion
Mortality of premature neonates with respiratory distress syndrome (RDS) admitted at MNH was 31.3% in the first week of life. Preterm neonates with very low birth weight (VLBW), whose mother did not receive antenatal steroid, who scored < 7 at 5 min and whose saturation was < 90% at 6 hours were at higher risk of dying.
Tailored interventions addressing risk factors should be devised to improve the preterm neonate’s survival in the study area. Close attention to early stabilization and improve preterm care may further reduce the mortality associated with this disorder especially to those with low birth weight and lower gestational age.
Acknowledgements
We would like to thank Muhimbili National hospital where this study was conducted and extend our gratitude to research office and department of Paediatrics at Muhimbili university of health and allied sciences. We also thank the parents who participated in the study.
Abbreviations
- AHR
Adjusted Hazard Ratio
- CPAP
Continuous Positive Airway Pressure
- CHR
Crude Hazard Ratio
- GA
Gestational Age
- GCP
Good clinical practice
- LMIC
Low middle income countries
- MNH
Muhimbili National Hospital
- MUHAS
Muhimbili University of Health and Allied Sciences
- NICU
Neonatal Intensive Care Unit
- PROM
Premature Rupture of Membrane
- RBS
Random Blood Sugar
- RDS
Respiratory Distress Syndrome
- SAS
Silverman Andersen score
- SGA
Small for Gestational Age
- VLBW
Very low birth weight
Authors’ contributions
MB: Synthesized the protocol, undertook data collection, data management, analysis, wrote the draft manuscript, revised, and finalized the manuscript. (Corresponding author). KM: Supervised the conduct of the research, revised the proposal, participated in writing the manuscript, reviewed and accepted the final manuscript. YA: Supervised the conduct of the research, revised the proposal, participated in writing the manuscript, reviewed and accepted the final manuscript. JC: Supervised the conduct of the research, revised the proposal as well as the first draft of manuscript. AM: Before his demise has contributed significantly to the development of proposal, supervised data collection, reporting, and analysis, as well as the first draft of manuscript. He was unfortunately demised before the final manuscript. The author(s) read and approved the final manuscript.
Funding
No funding.
Availability of data and materials
The data from this study can be obtained upon request from the first author, Dr. Maria Bulimba email: mbulimba@yahoo.com.
Declarations
Ethics approval and consent to participate
All methods were carried out in accordance with relevant guidelines and regulations and the study was conducted in accordance with the Declaration of Helsinki. Ethical approval was obtained from the Ethics Review Committee of Muhimbili University of Health and Allied Sciences (MUHAS) and the Directorate of Research, Training and Consultancy at MNH. The ethical clearance approval reference number was DA.287/298/01A and permission number was MNH/TRCU/permission/2019/210. Written informed consent was sought from the mother/care giver. All the GCP principles were observed.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Augustine Massawe is deceased.
Contributor Information
Maria Bulimba, Email: mbulimba@yahoo.com.
Judith Cosmas, Email: judysise@gmail.com.
Yaser Abdallah, Email: yasam786@hotmail.com.
Karim Manji, Email: kpmanji@gmail.com.
References
- 1.Bhutta ZA, Yusuf K. Profile and outcome of the respiratory distress syndrome among newborns in Karachi: risk factors for mortality. J Trop Pediatr. 1997;43(3):143–148. doi: 10.1093/tropej/43.3.143. [DOI] [PubMed] [Google Scholar]
- 2.General SJ, Lanka S. Respiratory distress syndrome of the newborn. Sri Lanka J Child Heal. 2018;47(4):295–300. doi: 10.4038/sljch.v47i4.8588. [DOI] [Google Scholar]
- 3.Olme N. The pathophysiology of respiratory distress syndrome in neonates. Paediatr Child Health (Oxford) 2012;22(12):556. [Google Scholar]
- 4.Respiration A, Age G, Hemorrhage C, Positive C, Pressure A, Dysplasia L. Berman’s Pediatric Decision Making (Fifth Edition) 2016. Respiratory Distress Syndrome. [Google Scholar]
- 5.Kamath BD, MacGuire ER, McClure EM, Goldenberg RL, Jobe AH. Neonatal mortality from respiratory distress syndrome: lessons for low-resource countries. Pediatrics. 2011;127(6):1139–1146. doi: 10.1542/peds.2010-3212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Sweet DG, Carnielli V, Greisen G, Hallman M, Ozek E, Te Pas A, et al. European consensus guidelines on the Management of Respiratory Distress Syndrome - 2019 update. Neonatology. 2019;115(4):432–450. doi: 10.1159/000499361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Dyer J. Neonatal Respiratory Distress Syndrome: Tackling A Worldwide Problem. P T. 2019;44(1):12–14. [PMC free article] [PubMed] [Google Scholar]
- 8.Saboute M, Kashaki M, Bordbar A, Khalessi N, Farahani Z, Saboute M. The incidence of respiratory distress syndrome among preterm infants admitted to neonatal intensive care unit: a retrospective study. Open J Pediatr. 2011;5(5):285–289. [Google Scholar]
- 9.Parkash A, Haider N, Khoso ZA, Shaikh AS. Frequency , causes and outcome of neonates with respiratory distress admitted to Neonatal Intensive Care Unit. In: . Karachi: National Institute of Child Health. p. 771–5. [PubMed]
- 10.Bajad M, Goyal S, Jain B. Clinical profile of neonates with respiratory distress. Int J Contemp Pediatr. 2016;3(3):1009–1013. doi: 10.18203/2349-3291.ijcp20162382. [DOI] [Google Scholar]
- 11.Palod PH, Lawate BB, Sonar MN, Bajaj SP. A study of clinical profile of neonates with respiratory distress and predictors of their survival admitted in neonatal intensive care unit of tertiary care hospital. Int J Contemp Pediatr. 2017;4(6):2027–2031. doi: 10.18203/2349-3291.ijcp20174724. [DOI] [Google Scholar]
- 12.Sathenahalli VB, Dwivedi D, Bajaj N, Singh HP. Predictors of poor outcome in neonates with respiratory distress. Int J Contemp Pediatr. 2016;3(1):76–79. doi: 10.18203/2349-3291.ijcp20160092. [DOI] [Google Scholar]
- 13.Stevenson DK, Verter J, Fanaro AA V, Oh W, Ehrenkranz RA, Shankaran S, et al. Sex di V erences in outcomes of very low birthweight infants: the newborn male disadvantage. Arch Dis Child Fetal Neonatal. 2000;83(3)(table 3):182–5. [DOI] [PMC free article] [PubMed]
- 14.Townsel CD, Emmer SF, Campbell WA, Dempsey EM, Townsel CD. Gender differences in respiratory morbidity and mortality of preterm neonates. Front Paediatr. 2017;5(January):1–6. doi: 10.3389/fped.2017.00006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Chambliss LR, Curtis BR. The predictive value of a 5-minute Apgar for developing respiratory distress syndrome. Obstet Gynecol. 2005;105(4):1–5. [Google Scholar]
- 16.Bopape-Chinyanga TC, Thomas R, Velaphi S. Outcome of very-low-birth-weight babies managed with nasal continuous positive airway pressure, with or without surfactant, in a high-care nursery. South African J Child Heal. 2016;10(4):199. doi: 10.7196/SAJCH.2016.v10i4.1096. [DOI] [Google Scholar]
- 17.Phoya F, Langton J, Dube Q, Tam PI. Association of Neonatal Hypothermia with morbidity and mortality in a tertiary Hospital in Malawi. Bmj. 2020;66(5):1–9. [DOI] [PMC free article] [PubMed]
- 18.Shah S, Zemichael O, Meng HD. Factors associated with mortality and length of stay in hospitalised neonates in Eritrea , Africa: a cross-sectional study. BMJ Open. 2012;2(5):1–10. [DOI] [PMC free article] [PubMed]
- 19.Manktelow BN, Lal MK, Field DJ, Sinha SK. Antenatal corticosteroids and neonatal outcomes according to gestational age: a cohort study. Arch Dis Child Fetal Neonatal Ed. 2010;95(2):95–99. doi: 10.1136/adc.2009.170340. [DOI] [PubMed] [Google Scholar]
- 20.Morris I. Minimizing the risk of respiratory distress syndrome. Paediatr Child Health (Oxford). 2012;22(12):513–517. doi: 10.1016/j.paed.2012.08.012. [DOI] [Google Scholar]
- 21.Massawe A, Kidanto HL, Moshiro R, Majaliwa E, Chacha F, Shayo A, et al. A care bundle including antenatal corticosteroids reduces preterm infant mortality in Tanzania a low resource country. PLoS One. 2018;1000:1–16. doi: 10.1371/journal.pone.0193146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Sunil B, Girish N, Bhuyan M. Outcome of preterm babies with respiratory distress syndrome on nasal CPAP. Int J Contemp Pediatr. 2017;4(4):1206–1209. doi: 10.18203/2349-3291.ijcp20172067. [DOI] [Google Scholar]
- 23.Abou-faddan HH. Respiratory distress and its outcome among neonates admitted to neonatal intensive care unit of Assiut University children hospital. Egypt Egypt J Community Med. 2018;36(2):69–78. doi: 10.21608/ejcm.2018.11052. [DOI] [Google Scholar]
- 24.Myhre J, Immaculate M, Okeyo B, Anand M, Omoding A, Myhre L, et al. Effect of treatment of premature infants with respiratory distress using low-cost bubble CPAP in a rural African hospital. J Trop Pediatr. 2016;62(5):385–389. doi: 10.1093/tropej/fmw023. [DOI] [PubMed] [Google Scholar]
- 25.Niknafs P, Faghani A, Afjeh S, Moradinazer M. Management of Neonatal Respiratory Distress Syndrome Employing ACoRN respiratory sequence protocol versus early nasal continuous Positive airway Pressure protocol. Iran J Pediatr. 2014;24(1):57–63. [PMC free article] [PubMed] [Google Scholar]
- 26.Ballard JL, Khoury JC, Wang L, Lipp R. FETAL AND New Ballard Score , expanded to include extremely premature infants Uo--. J Pediatr. 1991;119(3):417–23. [DOI] [PubMed]
- 27.Bruett A, Nancy H, Dennis EG, Maneesh EM. Performance of the Silverman Andersen respiratory severity score in predicting PCO 2 and respiratory support in newborns: a prospective cohort study. J Perinatol. 2018;38(5):505–11. [DOI] [PMC free article] [PubMed]
- 28.Mbawala GB, Fredrick F, Kamugisha E, Konje E, Hokororo A. Factors associated with mortality among premature babies admitted at Bugando medical Centre, Mwanza - Tanzania. East Afr J Public Health. 2014;11:641–645. [Google Scholar]
- 29.Nyandiko WM, Sara NE. Outcomes and associated factors among premature neonates with respiratory distress syndrome managed at Moi teaching and referal hospital, Eldoret, Kenya. East Afr Med J. 2018;95(1):1098–1107. [Google Scholar]
- 30.Hubbard RM, Choudhury KM, Lim G. Treatment patterns and clinical outcomes in neonates diagnosed with respiratory distress syndrome in a low-income country: a report from Bangladesh. Int Anesth Res Soc. 2018;XXX(Xxx):1–3. doi: 10.1213/ANE.0000000000002865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Tochie JN, Choukem SP, Langmia RN, Barla E, Ndombo PK. Neonatal respiratory distress in a reference neonatal unit in Cameroon: an analysis of prevalence, predictors, etiologies and outcomes. Pan Afr Med J. 2016;24:1–10. doi: 10.11604/pamj.2016.24.152.7066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.G S Mlay, K P Manji BR. Respiratory distress syndrome among neonates admitted at Muhimbili medical Centre, Dar Es Salaam, Tanzania. J Trop Pediatr 2000; [DOI] [PubMed]
- 33.Dewez JE, van den Broek N. Continuous positive airway pressure (CPAP) to treat respiratory distress in newborns in low- and middle-income countries. Trop Dr. 2017;47(1):19–22. doi: 10.1177/0049475516630210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Network* SSG of the EKSNNR. Target ranges of oxygen saturation in extremely preterm infants. N Engl J Med. 2010;362(21):1959–69. [DOI] [PMC free article] [PubMed]
- 35.Mu Y, Li M, Zhu J, Wang Y, Xing A, Liu Z, et al. Apgar score and neonatal mortality in China: an observational study from a national surveillance system. BMC Pregnancy Childbirth. 2021;1–12. [DOI] [PMC free article] [PubMed]
- 36.Zeitlin J, Manktelow BN, Piedvache A, Cuttini M, Boyle E, Van Heijst A, et al. Use of evidence based practices to improve survival without severe morbidity for very preterm infants: results from the EPICE population based cohort. BMJ. 2016;354:1–10. doi: 10.1136/bmj.i2976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Morris I, Adappa R. Minimizing the risk of respiratory distress syndrome. Paediatr Child Heal (United Kingdom) 2012;22(12):513–517. doi: 10.1016/j.paed.2012.08.012. [DOI] [Google Scholar]
- 38.Mcgoldrick E, Stewart F, Parker R, Sr D, Mcgoldrick E, Stewart F, et al. Antenatal corticosteroids for accelerating fetal lung maturation for women at risk of preterm birth (review). Cochrane Database Syst Rev. 2020;12(12):1–44. [DOI] [PMC free article] [PubMed]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The data from this study can be obtained upon request from the first author, Dr. Maria Bulimba email: mbulimba@yahoo.com.