Over the past 7 years, many clinical studies have demonstrated that SGLT2 inhibitors reduce major cardiovascular events, progressive kidney failure, and kidney and cardiovascular death in individuals with diabetes.1–3 SGLT2 inhibitors act directly, and apparently exclusively, on kidney proximal tubule (PT) cells by decreasing the activity of the apical sodium/glucose cotransporter and thereby inhibiting glucose reabsorption by the kidney—resulting in a reduction of blood glucose levels—and also reducing glucose metabolism by the PT. Given their profound cardiovascular and kidney effects, there are ongoing efforts to identify the specific mechanisms for their protective properties, with the hope that new and even more potent treatments for diabetic kidney disease (DKD) and other chronic kidney diseases could be developed and that these diseases could be prevented entirely in many patients. One focus of interest has been the role of mTORC1, a key metabolic regulator and a determinant of PT responses in diabetes. This ubiquitous metabolic controller sits at the center of cellular energy production and consumption, and regulates protein translation in specific ways to either increase or decrease metabolic activities in response to cues from the cellular environment and systemic energy supply. Responding to the energy state of the cell, mTORC1 also regulates cell proliferation and critical processes such as autophagy and apoptosis.
The role of mTORC1 in the development of experimental DKD is well established. Pathologic overactivation of mTORC1 in podocytes leads to podocyte injury and loss, while its activation in the PT is associated with increased tubulointerstitial fibrosis.4–6 There is substantial evidence that SGLT2 inhibitors reduce mTORC1 activity in both experimental animal models and humans with diabetes.6 Conversely, nonphysiologic reduction of mTORC1 activity can lead to proteinuria and glomerulosclerosis, indicating that precise regulation of mTORC1 activity is critical for kidney cell function and integrity.5
What Does This Important Study Show?
As a potential regulator of mTORC1 and other important cellular processes, there has been increasing interest in the role of endocannabinoid (eCB) signaling in DKD.7 In mouse kidneys, cannabinoid receptor 1 (CB1R) is expressed in podocytes, mesangial cells, and PT cells.8,9 Accumulating evidence, derived mainly from mouse models of diabetes, has suggested that activation of eCB/CB1R signaling contributes to DKD development. Conversely, pharmacological inhibition of CB1R has been shown to ameliorate diabetes-induced kidney dysfunction, inflammation, and fibrosis in these models.10–13
The article from one of the main groups studying eCB/CB1R signaling recently published in Nature Communications extends our understanding of this signaling system in PT cells in mice.14 The authors of this study bred genetically diabetic mice that lacked CB1R gene expression only in PT cells. They then compared these PT-specific CB1R knockout animals to mice that maintained PT CB1R expression but had the same background of genetic diabetes. The authors demonstrated that enhanced CB1R signaling in the PT augmented DKD, as the knockout animals had significantly lower levels of proteinuria, glomerular hypertrophy, markers of kidney injury, inflammation, and fibrosis—as well as preservation of more normal histology—compared to those with normal expression of the gene. CB1R gene deletion also prevented mTORC1 activation in diabetes.
The authors also explored the effects of elevated glucose levels on eCB signaling in cultured PT cells. On acute exposure to high glucose there was an increase in CB1R signaling. Although the effects of CB1R expression on SGLT2 expression were not reported, CB1R did enhance expression of GLUT2, the facilitative glucose transporter on the basolateral membrane of PT cells that is responsible for transporting glucose out of these cells into the extracellular fluid and back into the bloodstream (Fig 1A).15 Along with the increase in GLUT2 expression, the increased CB1R signaling activated the mTORC1 pathway in a manner dependent on PI3K (since the PI3K inhibitor wortmannin abolished mTORC1 activation). A strong relationship between mTORC1 activity and GLUT2 was also demonstrated by upregulating or downregulating mTORC1 activity. As expected, with mTORC1 overactivation, there was increased GLUT2 expression and the development of a DKD phenotype; with reduced mTORC1 activation, there was lower expression of GLUT2 and a less profound DKD phenotype. Using a different cultured cell system, this increase in mTORC1-stimulated GLUT2 expression was shown to be transcriptionally driven via a number of intermediary transcription factors.
Figure 1.
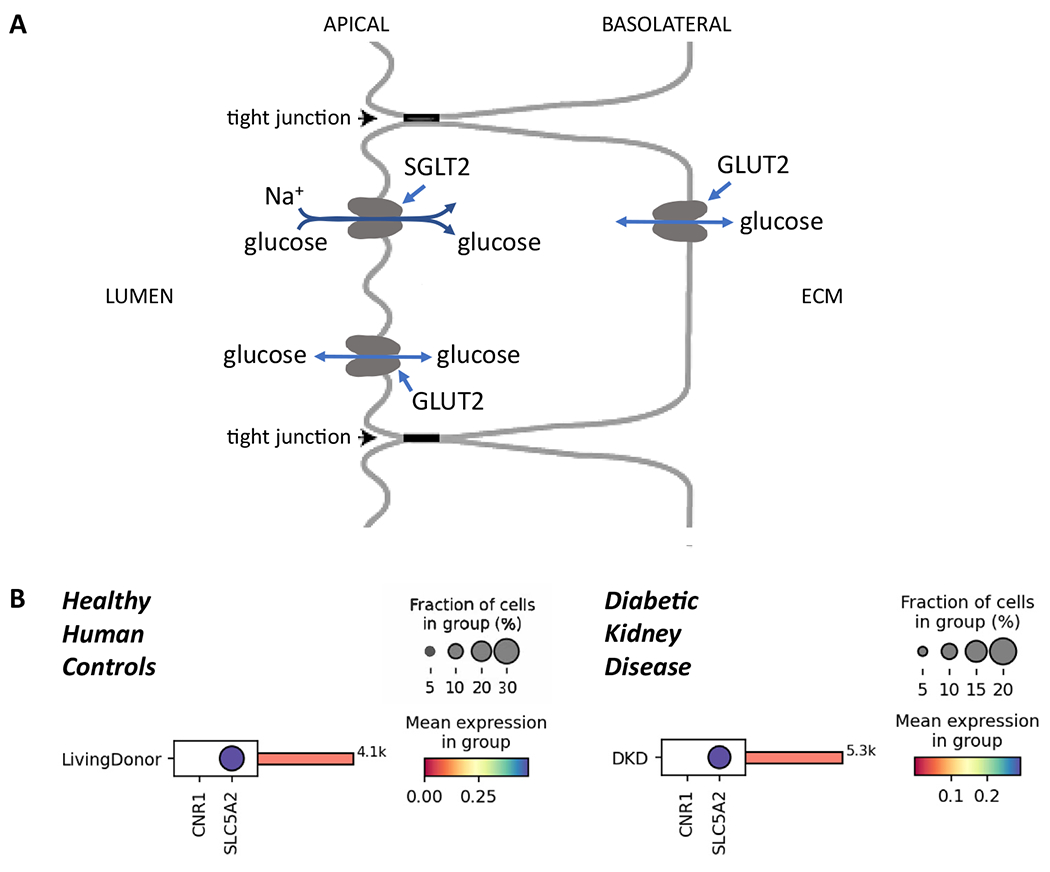
Proximal tubule (PT) cells and diabetic kidney disease (DKD). (A) Schematic of glucose transporters in the initial segments of the kidney PT. SGLT2 transports glucose from the PT lumen into the cytoplasm against its concentration gradient owing to the energetically favorable electrochemical gradient for sodium. The reabsorbed glucose is largely transported out of the cell down its concentration gradient by GLUT2, a high-capacity facilitative glucose transporter found on the basolateral surface. Relatively small numbers of GLUT2 molecules can be found on the luminal (apical) surface of the PT cells, where they transport glucose down its concentration gradient either in or out of the cell. Under usual physiologic conditions such transport would be out of the cell, since SGLT2 action increases glucose concentrations inside the cell relative to the concentration in the lumen, which falls substantially along the length of the proximal tubule. (B) Comparison between the cell-specific transcript expression of CNR1 (transcript coding for CB1R) and SLC5A2 (SGLT2) in PT cells from healthy controls (n = 18; 4,100 cells; left panel) or patients with DKD (n = 12; 5,300 cells; right panel). Data for healthy controls were adapted from Menon et al23 while data for the patients with DKD were from the Kidney Precision Medicine Project and obtained from Lake et al.24 At the protein level, CB1R is not detectable in glomeruli and tubules in human kidneys (sample image from Human Protein Atlas, available at www.proteinatlas.org/ENSG00000118432-CNR1/tissue/kidney#img).
Using similar genetic strategies as used for CB1R, the authors developed a diabetic mouse model with a gene deletion causing GLUT2 levels to be partially reduced in PT cells. Features of DKD were mostly prevented in these mice when compared to diabetic mice with intact GLUT2 expression. The authors suggest that these effects may be due to the reduction in glucose uptake by the small percentage of GLUT2 transporters that are translocated to the apical membrane of PT cells. SGLT2 levels were also markedly reduced in the GLUT2 knockout mouse kidneys and therefore luminal uptake of glucose via SGLT2 was almost certainly lowered as well. Together, these results suggest that directly reducing GLUT2-mediated glucose reabsorption by the kidneys could result in protection from DKD. This study adds to the literature suggesting a potential protective mechanism of action for SGLT2 inhibitors in DKD and other chronic kidney diseases, namely, that the inhibition of PT-mediated glucose and sodium reabsorption leads to lower ATP consumption by the basolateral adenosine triphosphatase sodium/potassium pump and a reduction in PT energy production and consumption.
How Does This Study Compare With Prior Studies?
The findings from Hinden et al,14 along with those in prior publications by this group15 and others,8 demonstrate that CB1R enhances mTORC1 activity and GLUT2 expression in diabetic mouse models and that increased CB1R levels and activity result in worse kidney disease in these models. These data are consistent with the hypothesis that diabetes induces CB1R signaling, which, in turn, activates mTORC1, enhancing GLUT2 expression and GLUT2-mediated glucose transport in PT cells, leading to DKD. These findings suggest that CB1R and GLUT2 are potential targets for treating and preventing DKD.
While mechanistically interesting, the relevance of these findings to human DKD remains unclear for a number of reasons. GLUT2 gene deletion in PT cells prevented the development of a DKD phenotype. However, as noted above, the reduction of GLUT2 on the basolateral membrane resulted in a substantial reduction of active SGLT2 transporters on the luminal surface, which is important, since SGLT2 activity is rate-limiting for PT glucose reabsorption.16 This is why SGLT2 inhibition leads directly to the favorable clinical phenotype. Indeed, complete GLUT2 knockout in mouse kidney has been shown to lead to almost-complete elimination of SGLT2 expression.17 Thus, the effects seen in the GLUT2 knockout model are likely to be due to reduced SGLT2 activity. In addition, it seems unlikely that regulation of GLUT2 expression or apical translocation of GLUT2 contributes greatly to either intracellular glucotoxicity or net glucose reabsorption. Unlike SGLT2, GLUT2 transports glucose down its concentration gradient either in or out of cells. It cannot “pull” glucose into PT cells against its concentration gradient, as does SGLT2, which, like all sodium-coupled secondary active transporters, uses the sodium gradient to drive the transport of other molecules against their electrochemical potential into or out of PT cells (Fig 1A). Depending on the glucose concentrations in the PT cytoplasm and in the tubular lumen, apical GLUT2 transports glucose either into PT cells or in the opposite direction into the tubular lumen. Since SGLT2 increases intracellular glucose in the PT to levels higher than those in the tubular lumen, any active apical GLUT2 molecules would transport glucose from the cell into the lumen, reducing net glucose reabsorption (Fig 1A). GLUT2 can indirectly contribute to apical sodium reabsorption but is unlikely to add to net glucose reabsorption. Since inhibition of SGLT2 and SGLT1 completely abrogates PT glucose reabsorption, it seems that active glucose reabsorption by apical GLUT2 would be minor even in favorable conditions.16 Given that glucose transport mediated by the SGLTs is the rate-limiting step in this process, increasing GLUT2 levels is unlikely to substantially alter net glucose reabsorption across either apical or basolateral membranes. Moreover, although GLUT2 levels rise in rodent models of diabetes, such changes in GLUT2 expression in humans with diabetes are modest and inconsistent.16 In addition, given the critical role that GLUT2 plays in liver, brain, intestine, and islet cells in the pancreas,18 it is unlikely that GLUT2 inhibitors would be practical alternatives to SGLT2 inhibitors, which are selective for PT cells.
Another limitation in translating the results of this study to humans with diabetes is that the mouse models had relatively mild phenotypes because their genetic background is the nephropathy-resistant C57BL/6 strain. Thus, the diabetic animals had relatively limited albuminuria, mesangial expansion, and podocytopathy. This well-known resistance to diabetic changes19,20 makes this particular mouse background strain less applicable to studying treatment effects, since the initial disease is so mild. Indeed, many treatments shown to reverse diabetic changes in this strain of mice have not been confirmed in humans or in mouse models with more human-like features.
Perhaps most important, while the transcript encoding CB1R is present in many human tissues, there is no detectable expression in PT cells from healthy humans or from patients with established DKD (Fig 1B). Similarly, there is no detectable CB1R protein in the PT or kidney cortex of any of the samples (with or without disease) assessed by the Human Protein Atlas.21,22 These observations raise questions about the relevance of eCB/CB1R signaling to human DKD and merit further detailed evaluation of cannabinoid signaling in human kidney samples and disease processes.
What Are the Implications for Nephrologists?
Although the intricacies of cannabinoid receptor signaling in the kidney are of interest and clearly play a role in regulating mTORC1, GLUT2, and other pathways that are crucial in metabolic diseases, it remains uncertain whether these findings apply to humans with DKD or whether the findings from Hinden et al are relevant to DKD pathogenesis. Animal disease models can be quite helpful, but pathways operative in mice are not always relevant to human kidney diseases.
Acknowledgments:
We thank Philip McCown, PhD, Viji Nair, MS, Felix Eichinger, MS, and Sean Eddy, PhD, for providing valuable input and data.
Support:
Dr Naik acknowledges ongoing support from the University of Michigan O’Brien Kidney Translational Core Center (P30 DK08193) and National Institute of Health (NIH) grant K23 DK 125529. Dr Brosius acknowledges support from the University of Michigan O’Brien Kidney Translational Core Center (P30 DK08193) and NIH grant R25 DK128859.
Financial Disclosure:
Dr Naik declares that he has no relevant financial interests. Dr Brosius reports having received fees from Gilead Scientific Inc for serving on a scientific advisory board in 2021 but has no other relevant financial interests.
References
- 1.Heerspink HJ, Stefánsson BV, Correa-Rotter R, et al. Dapagliflozin in patients with chronic kidney disease. N Engl J Med. 2020;383(15):1436–1446. [DOI] [PubMed] [Google Scholar]
- 2.Zinman B, Wanner C, Lachin JM, et al. EMPA-REG OUTCOME Investigators. Empagliflozin, cardiovascular outcomes, and mortality in type 2 diabetes. N Engl J Med. 2015;373(22):2117–2128. [DOI] [PubMed] [Google Scholar]
- 3.Perkovic V, Jardine MJ, Neal B, et al. Canagliflozin and renal outcomes in type 2 diabetes and nephropathy. N Engl J Med. 2019;380(24):2295–2306. [DOI] [PubMed] [Google Scholar]
- 4.Inoki K, Mori H, Wang J, et al. mTORC1 activation in podocytes is a critical step in the development of diabetic nephropathy in mice. J Clin Invest. 2011;121(6):2181–2196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Gödel M, Hartleben B, Herbach N, et al. Role of mTOR in podocyte function and diabetic nephropathy in humans and mice. J Clin Invest. 2011;121(6):2197–2209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Kogot-Levin A, Hinden L, Riahi Y, et al. Proximal tubule mTORC1 is a central player in the pathophysiology of diabetic nephropathy and its correction by SGLT2 inhibitors. Cell Rep. 2020;32(4):107954. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Barutta F, Bruno G, Mastrocola R, Bellini S, Gruden G. The role of cannabinoid signaling in acute and chronic kidney diseases. Kidney Int. 2018;94(2):252–258. [DOI] [PubMed] [Google Scholar]
- 8.Barutta F, Corbelli A, Mastrocola R, et al. Cannabinoid receptor 1 blockade ameliorates albuminuria in experimental diabetic nephropathy. Diabetes. 2010;59(4):1046–1054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Jenkin KA, McAinch AJ, Zhang Y, Kelly DJ, Hryciw DH. Elevated cannabinoid receptor 1 and G protein-coupled receptor 55 expression in proximal tubule cells and whole kidney exposed to diabetic conditions. Clin Exp Pharmacol Physiol. 2015;42(3):256–262. [DOI] [PubMed] [Google Scholar]
- 10.Jourdan T, Park JK, Varga ZV, et al. Cannabinoid-1 receptor deletion in podocytes mitigates both glomerular and tubular dysfunction in a mouse model of diabetic nephropathy. Diabetes Obes Metab. 2018;20(3):698–708. [DOI] [PubMed] [Google Scholar]
- 11.Lin C-L, Hsu Y-C, Lee P-H, et al. Cannabinoid receptor 1 disturbance of PPARγ2 augments hyperglycemia induction of mesangial inflammation and fibrosis in renal glomeruli. J Mol Med. 2014;92(7):779–792. [DOI] [PubMed] [Google Scholar]
- 12.Jourdan T, Szanda G, Rosenberg AZ, et al. Overactive cannabinoid 1 receptor in podocytes drives type 2 diabetic nephropathy. Proc Natl Acad Sci U S A. 2014;111(50):E5420–E5428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Nam D, Lee M, Kim J, et al. Blockade of cannabinoid receptor 1 improves insulin resistance, lipid metabolism, and diabetic nephropathy in db/db mice. Endocrinology. 2012;153(3):1387–1396. [DOI] [PubMed] [Google Scholar]
- 14.Hinden L, Ahmad M, Hamad S, et al. Opposite physiological and pathological mTORC1-mediated roles of the CB1 receptor in regulating renal tubular function. Nat Commun. 2022;13(1):1–18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Hinden L, Udi S, Drori A, et al. Modulation of renal GLUT2 by the cannabinoid-1 receptor: implications for the treatment of diabetic nephropathy. J Am Soc Nephrol. 2018;29(2):434–448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Vallon V. Glucose transporters in the kidney in health and disease. Pflugers Arch. 2020;472(9):1345–1370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.de Souza Cordeiro LM, Bainbridge L, Devisetty N, McDougal DH, Peters DJ, Chhabra KH. Loss of function of renal Glut2 reverses hyperglycaemia and normalises body weight in mouse models of diabetes and obesity. Diabetologia. 2022;65(6):1032–1047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Leturque A, Brot-Laroche E, Le Gall M. GLUT2 mutations, translocation, and receptor function in diet sugar managing. Am J Physiol Endocrinol Metab. 2009;296(5):E985–E992. [DOI] [PubMed] [Google Scholar]
- 19.Becker GJ, Hewitson TD. Animal models of chronic kidney disease: useful but not perfect. Nephrol Dial Transplant. 2013;28(10):2432–2438. [DOI] [PubMed] [Google Scholar]
- 20.Brosius FC, Alpers CE, Bottinger EP, et al. Mouse models of diabetic nephropathy. J Am Soc Nephrol. 2009;20(12):2503–2512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Uhlén M, Fagerberg L, Hallström BM, et al. Tissue-based map of the human proteome. Science. 2015;347(6220):1260419. [DOI] [PubMed] [Google Scholar]
- 22.CNR1. The Human Protein Atlas. Accessed July 11, 2022. https://www.proteinatlas.org/ENSG00000118432-CNR1/tissue
- 23.Menon R, Otto EA, Berthier CC, et al. Glomerular endothelial cell-podocyte stresses and crosstalk in structurally normal kidney transplants. Kidney Int. 2022;101(4):779–792. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Lake BB, Menon R, Winfree S, et al. An atlas of healthy and injured cell states and niches in the human kidney. bioRxiv. Preprint posted online July 29, 2021. 10.1101/2021.07.28.454201 [DOI] [PMC free article] [PubMed] [Google Scholar]


