Abstract
Introduction
Little is known about the role played by anticoagulants in COVID-19.
Objective
The aim of this study was to assess the impact of previous anticoagulant treatment on risk of hospitalization due to COVID-19, progression to severe COVID-19 and susceptibility to COVID-19 infection.
Methods
We conducted a multiple population-based case–control study in northwest Spain, in 2020, to assess (1) risk of hospitalization: cases were all patients admitted due to COVID-19 with PCR confirmation, and controls were a random matched sample of subjects without a positive PCR; (2) progression: cases were hospitalized COVID-19 subjects, and controls were all non-hospitalized COVID-19 patients; and (3) susceptibility: cases were patients with a positive PCR (hospitalized and non-hospitalized), and the controls were the same as for the hospitalization model. Adjusted odds ratios (ORs) and 95% confidence intervals (95% CIs) were calculated using a generalized linear mixed model.
Results
The consumption of antivitamin K and direct-acting anticoagulants apparently was not associated with the risk of progression to severe COVID-19 (OR 0.93 [95% CI 0.74–1.17] and OR 1.04 [95% CI 0.79–1.36], respectively). Antivitamin K anticoagulants were associated with a significantly lower risk of hospitalization (OR 0.77 [95% CI 0.64–0.93]), which, in part, can be explained by a decreased risk of susceptibility to infection (OR 0.83 [95% CI 0.74–0.92]). The use of direct-acting anticoagulants was not associated with the risk of hospitalization, although it also seems to decrease susceptibility (OR 0.85 [95% CI 0.74–0.98]). It has also been observed that low-molecular-weight heparins were associated with an increased risk of progression to severe COVID-19 (OR 1.25 [95% CI 1.01–1.55]).
Conclusion
The results of this study have shown that antivitamin K anticoagulants and direct-acting anticoagulants do not increase the risk of progression to more severe stages. Antivitamin K consumption was associated with a lower risk of hospitalization and susceptibility to infection.
Supplementary Information
The online version contains supplementary material available at 10.1007/s40264-022-01266-0.
Key Points
| The influence of pre-existing long-term treatment with anticoagulants on COVID-19 was unclear. |
| In our study, neither the consumption of antivitamin K nor the consumption of direct-acting anticoagulants was associated with the risk of progression to severe COVID-19. |
| In our study, consumption of antivitamin K anticoagulants was linked to better outcomes in both COVID-19 hospitalization and susceptibility to COVID-19 infection. |
Introduction
Severe Acute Respiratory Syndrome Coronavirus-2 (COVID-19) continues to pose a threat to public health worldwide, despite the availability of vaccines. It could thus be of great relevance, on the one hand, to identify new treatments or use existing treatments that might help mitigate the health impacts of COVID-19 [1], and on the other, to identify medications that could be a risk factor [2, 3].
After infecting target cells, COVID-19 is thought to target vascular endothelial cells, which cause the inflammatory response via cytokine storm and contribute to disease severity [4]. Inflammation, along with cytokine concentrations, activate clotting, followed by platelet activation and increased risk of thrombotic events and fibrin-degradation products, such as d-dimer [4–6]. Evidence indicates that high d-dimer levels are associated with coagulopathy and are an indicator of severe COVID-19 and worse prognosis [7]. Anticoagulant treatment might thus be assumed to prevent some severe complications of COVID-19, and indeed, these drugs are currently being widely administered to COVID-19 inpatients [8]. It is not clear, however, what role pre-existing long-term treatment with anticoagulants might play in disease severity or susceptibility to the virus [9–12].
Accordingly, the aims of this study were to assess the impact of pre-existing long-term treatment with anticoagulants (direct-acting anticoagulants, antivitamin K anticoagulants, low-molecular-weight heparins [LMWH]) on risk of hospitalization, risk of progression to severe COVID-19 and its influence on susceptibility to COVID-19 infection.
Methods
Setting
This study was conducted in Galicia (north-west Spain), a region with 2.7 million inhabitants (25.7% over 65 years of age), practically all of whom (98%) are covered by the Public Health System (PHS). The range of services provided to citizens includes preventive, diagnostic, therapeutic and rehabilitative care, as well as health promotion and maintenance activities. Visits to the doctor are free of charge but out-of-hospital pharmaceutical services are subject to a financial contribution (co-payment). Since patients’ contributions are income-linked, co-payment for medication ranges from 0 to 60%. In Spain, medication in the outpatient setting is mostly dispensed by community pharmacies.
The Galician PHS has a clinical record system which allows access to a unified register of all patients’ clinical information (diagnostic tests, drug prescriptions, International Classification of Primary Care codes, hospitalizations, etc.) for both primary and hospital care.
Study Design and Participants
We used a population-based case–control design [13]. This design is characterized by using data from a representative sample of all cases (in our instance, exhaustive sampling) in a precisely defined and identified population (in our instance, the population attended by the PHS in Galicia), and comparing these to data on persons (controls) randomly extracted from the same population as the cases (population-based case controls), something that could be assumed to give a valid estimate of the prevalence of exposure and covariates in the population of origin [14]. According to Rothman et al., this design can be considered the most desirable option for a case–control study [13].
To study risk of hospitalization, ‘case’ was defined as any patient admitted due to COVID-19, with PCR confirmation, to any public hospital in Galicia since the onset of the pandemic, whose clinical course ended before 1 January 2021. As controls, we selected a random sample of subjects who had no positive PCR (as a result of not having done any PCR or for being PCR negative) during the same period. With the aim of enhancing the efficiency of our analysis of risk of hospitalization [15], controls were randomly selected and matched with cases by age, sex and primary healthcare centre. We selected up to 20 controls for each case.
To assess the risk of progression to severe COVID-19, understood as the risk of requiring admission among COVID-19 positive subjects, we used the same cases as those used to assess the risk of hospitalization (all patients admitted due to COVID-19 with PCR confirmation). As controls, we used all patients with diagnosis of COVID-19, who did not require hospitalization. While the use of these controls unmatched with cases of progression reduces the efficiency of estimates for this model [16], it does not generate confounding bias because (1) the controls were drawn from the same population as the cases [13]; (2) they were selected regardless of exposure [13]; and (3) in the statistical analysis, adjustment was made for available potential confusing variables [15].
To assess susceptibility, cases were defined as all persons diagnosed with COVID-19 confirmed by a positive PCR (hospitalized and not hospitalized) across the study period in Galicia [15]. As controls, we used the same persons as those used to assess risk of hospitalization (subjects who had no positive PCR). As in the case of progression to severe COVID-19, the cases were unmatched with controls, which does not produce any type of bias, only a decrease in efficiency [13, 16].
Ethical Aspects
The study was approved by the Galician Clinical Research Ethics Committee (Comité de Ética de Investigación de Galicia), reference 2020/349, certified by the Spanish Agency of Medicines and Medical Devices (Agencia Española del Medicamento y Productos Sanitarios/AEMPS), and conducted in accordance with the Helsinki Declaration principles and current biomedical research legislation. The study protocol is registered in the EU electronic Register of Post-Authorisation Studies (EUPAS44587) and is available online at https://www.encepp.eu/encepp/viewResource.htm?id=44588.
Data extraction was automated, and the data were anonymized to ensure that the subjects concerned could not be identified.
Data Source and Collection
Automated data extraction was performed by an independent information technology services (IT) company, based on SERGAS’ Complex Data-Analysis System (Sistemas de Información y Análisis Complejos/SIAC). SIAC acts as a data warehouse which stores information for the management of different systems (dispensing of medications, diagnoses and hospitalizations, among others).
Exposure
The variables of exposure were defined as any of the following anticoagulant medications: direct-acting oral anticoagulants (apixaban, edoxaban, dabigatran and rivaroxaban); vitamin K antagonists (acenocumarol and warfarin); LMWH (enoxaparin, bemiparin, dalteparin, fraxiparine, nadroparin and tinzaparin); and other anticoagulants (fondaparinux and protein C) (see electronic supplementary material [ESM]). Consumption of one type of anticoagulant does not exclude the fact that the patient may consume another type of anticoagulant. We recorded those prescribed and dispensed to cases and controls alike across the study period, in the 6 months leading up to the index date. The index date was defined as the tenth day prior to the PCR+ date, or, for non-PCR+, the same index day as their matched case.
As study covariates, we recorded demographic and anthropometric variables, clinical COVID-19 variables (where applicable), and data on hospitalization, comorbidities (hypertension, diabetes mellitus, chronic obstructive pulmonary disease [COPD], obesity, ischaemic heart disease, cerebrovascular accident, heart failure, atrial fibrillation, chronic renal failure, cancer, asthma, current smoker) and exposure to the other medications prescribed for and dispensed to each of the subjects (antihypertensives, diuretics, non-steroidal anti-inflammatory drugs, hypolipidemic agents, other anticoagulants, antiplatelet agents and glucocorticoids). Polypharmacy (number of different medications prescribed and dispensed for chronic diseases in the last 6 months before the index day) was used as a proxy for the degree of chronicity of the patients [17]. All covariates were recorded for the 6 months prior to the index date.
Statistical Analysis
Generalized linear mixed models were subjected to statistical analysis, with the binomial link function. These models were used because of the structure of the data and because they have many advantages over conditional regression [18–20]: (1) they allow the analysis of matched and unmatched models; (2) they permit the introduction of random terms to control for heterogeneity of initial clusters and time periods; (3) strata in which cases coincide in exposures with controls continue to count as events for the calculation and for the estimates.
To construct the models, the following four levels were considered: patient; case–control strata (case and matched control); health centre; and pandemic wave. We used random effects to assess the effect of the pandemic wave, and nested random effects for patients, case–control strata, and health centre. Adjustments were made for potential confounding variables, including sex, age, comorbidities, smoking habit, and each additional pharmacological treatment. Results were expressed in adjusted odds ratio (aOR) with its 95% confidence interval (CI). Adjusted estimates were obtained for the effect of anticoagulant therapy dispensed compared with the absence of any anticoagulant drug therapy. The models were estimated using the glmer function of the lme4 R package [21] (R Software version 4.1.0).
Results
Risk of Hospitalization
For analysis purposes, the study covered a total of 3060 cases who had a PCR+ and had been hospitalized. The control group comprised 56,785 subjects without a PCR+, matched by age, sex and primary healthcare centre. As shown in Table 1, cases had a higher prevalence of several comorbidities (obesity, diabetes, heart failure, hypertension) and previous use of anticoagulants than did controls.
Table 1.
Demographic and clinical characteristics of subjects in the hospitalization and progression models.
| Cases n = 3060 (%) |
Hospitalization | Progression | |
|---|---|---|---|
| Controls n = 56,785 (%) | Controls n = 26,757 (%) |
||
| Sex | |||
| Male | 1552 (50.7) | 28,729 (50.6) | 11,122 (41.6) |
| Female | 1508 (49.3) | 28,056 (49.4) | 15,635 (58.4) |
| Age, median (IQR) | 74 (59.0–84.0) | 73 (59.0–84.0) | 47 (33–63) |
| Comorbidities | |||
| Hypertension | 1754 (57.3) | 28,020 (49.3) | 6093 (22.8) |
| Diabetes | 841 (27.5) | 10,920 (19.2) | 2460 (9.2) |
| COPD | 398 (13.0) | 4569 (8.0) | 730 (2.7) |
| Obesity | 889 (29.1) | 10,817 (19.0) | 3901 (14.6) |
| Ischaemic heart disease | 359 (11.7) | 4768 (8.4) | 832 (3.1) |
| Cerebrovascular accident | 306 (10.0) | 3874 (6.8) | 838 (3.1) |
| Heart failure | 469 (15.3) | 4030 (7.1) | 639 (2.4) |
| Atrial fibrillation | 466 (15.2) | 5769 (10.2) | 1035 (3.9) |
| Chronic renal failure | 437 (14.3) | 4316 (7.6) | 678 (2.5) |
| Cancer | 529 (17.3) | 7770 (13.7) | 1701 (6.4) |
| Asthma | 285 (9.3) | 3388 (6.0) | 2152 (8) |
| Current smoker | 809 (26.4) | 8532 (15.0) | 4036 (15.1) |
| Direct-acting anticoagulantsa | 154 (5.0) | 1832 (3.2) | 303 (1.1) |
| Antivitamin K anticoagulantsa | 242 (7.9) | 3782 (6.7) | 536 (2) |
| LMWHa | 174 (5.7) | 2023 (3.6) | 489 (1.8) |
| Other anticoagulantsa | 1 (0.0) | 3 (0.0) | 0 (0.0) |
| Platelet aggregation inhibitors excluding heparins | 608 (19.9) | 9582 (16.9) | 1598 (6) |
Others = Fondaparinux and protein c
COPD chronic obstructive pulmonary disease, IQR interquartile range, LMWH low-molecular-weight heparins
aConsumption of one type of anticoagulant does not exclude the fact that the patient may consume another type of anticoagulant. Hence, the sum of frequencies does not equal the total number of exposed subjects
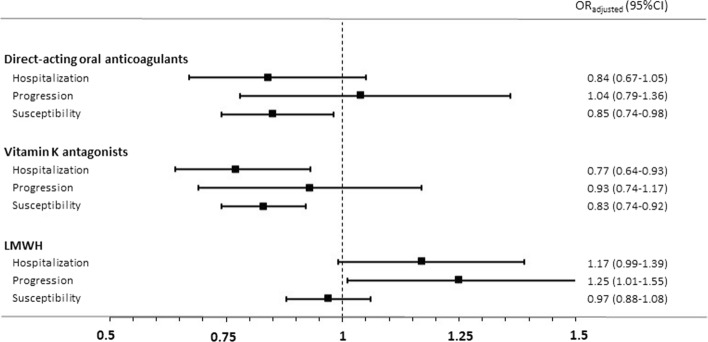
Table 2 and Fig. 1 show the adjusted estimates of risk of hospitalization due to COVID-19. Comorbidities and other drug treatments were included as potential confounding variables. After adjustment, the use of antivitamin K antagonists appeared to reduce the risk (OR 0.77 [95% CI 0.64–0.93]), while the use of LMWH suggested an increased risk (OR 1.17 [95% CI 0.99–1.39]), close to statistical significance, and the use of direct-acting anticoagulants showed no statistical risk effect (OR 0.84 [95% CI 0.67–1.05]).
Table 2.
Risk of COVID-19 requiring hospitalization and current use of anticoagulants
| Exposure | Crude odds ratio (95% CI) | Adjusted odds ratiob (95% CI) | p value |
|---|---|---|---|
| Direct actinga | 1.59 (1.34–1.88) | 0.84 (0.67–1.05) | 0.1248 |
| Antivitamin Ka | 1.20 (1.05–1.38) | 0.77 (0.64–0.93) | 0.0059 |
| LMWHa | 1.63 (1.39–1.91) | 1.17 (0.99–1.39) | 0.0699 |
| Othersa | 6.19 (0.64–59.51) | 3.00 (0.25–36.75) | 0.3891 |
Others = Fondaparinux and protein c
CI confidence interval, LMWH low-molecular-weight heparins
aConsumption of one type of anticoagulant does not exclude the fact that the patient may consume another type of anticoagulant
bAdjusted for sex, age, and comorbidities: hypertension, diabetes, COPD (chronic obstructive pulmonary disease), obesity, ischaemic heart disease, cerebrovascular accident, heart failure, atrial fibrillation, chronic renal failure, cancer, asthma, current smoker, current use of other pharmacological treatment (antihypertensives, diuretics, non-steroidal anti-inflammatory drugs, hypolipidemic agents, anticoagulants, antiplatelet agents and glucocorticoids) and polypharmacy (number of different treatments)
Fig. 1.
Odds ratios of the association between current use of direct-acting oral anticoagulants, vitamin K antagonists, low-molecular-weight heparins (LMWH) and COVID-19-related outcomes
Progression to Severe COVID-19
For this analysis, there were 3060 cases with COVID-19 who were hospitalized, and 26,747 controls, who had COVID-19 but did not require hospitalization. As shown in Table 1, cases had a higher prevalence of age, hypertension, diabetes, COPD, obesity, ischaemic heart disease, cerebrovascular accident, heart failure, chronic renal failure, cancer, asthma, current smoker, direct-acting anticoagulants, antivitamin K anticoagulants, LMWH and platelets aggregation excluding heparins. All these covariables were considered when fitting the model.
Only LMWH users had an increased risk of progression to severe COVID-19 (OR 1.25 [95% CI 1.01–1.55]; see Table 3 and Fig. 1).
Table 3.
Risk of progression to severe COVID-19 and current use of anticoagulants
| Exposure | Cases n = 3060 (%) |
Controls n = 26,757 (%) |
Crude odds ratio (95% CI) | Adjusted odds ratiob (95% CI) | p value |
|---|---|---|---|---|---|
| Direct actinga | 154 (5.0) | 303 (1.1) | 4.85 (3.93–5.99) | 1.04 (0.79–1.36) | 0.7724 |
| Antivitamin Ka | 242 (7.9) | 536 (2.0) | 4.09 (3.46–4.83) | 0.93 (0.74–1.17) | 0.5448 |
| LMWHa | 174 (5.7) | 489 (1.8) | 3.02 (2.51–3.65) | 1.25 (1.01–1.55) | 0.0426 |
| Othersa | 1 (0.0) | 0 (0.0) | 3.49 (3.13–3.88) | 1.00 (0.87–1.14) | 0.9500 |
Others = Fondaparinux and protein c
CI confidence interval, LMWH low-molecular-weight heparins
aConsumption of one type of anticoagulant does not exclude the fact that the patient may consume another type of anticoagulant
bAdjusted for sex, age, and comorbidities: hypertension, diabetes, COPD (chronic obstructive pulmonary disease), obesity, ischaemic heart disease, cerebrovascular accident, heart failure, atrial fibrillation, chronic renal failure, cancer, asthma, current smoker, current use of other pharmacological treatment (antihypertensives, diuretics, non-steroidal anti-inflammatory drugs, hypolipidemic agents, anticoagulants, antiplatelet agents and glucocorticoids) and polypharmacy (number of different treatments)
Susceptibility
For this analysis, there were 29,817 cases of patients with a PCR+ (3060 hospitalized and 26,757 not hospitalized), and 56,785 controls who did not have a PCR+. Compared with controls, cases were younger (median age [interquartile range] 49 years [34–67] vs 73 years [59–84]) and there were more women (37.9 vs 30.6%). Both of these variables were considered when fitting the model.
The use of vitamin K antagonists appeared to reduce the risk of susceptibility to infection (COVID-19+) (OR 0.83 [95% CI 0.74–0.92]), after adjusting for covariates (see Table 4 and Fig. 1). The use of direct-acting anticoagulants also showed an apparent decrease in risk (OR 0.85 [95% CI 0.74–0.98]), whereas the use of LMWH did not appear to show any risk effect (OR 0.97 [95% CI 0.88–1.08]).
Table 4.
Risk of COVID-19 susceptibility and current use of anticoagulants
| Exposure | Cases n = 29817 (%) |
Controls n = 56,785 (%) |
Crude odds ratio (95% CI) | Adjusted odds ratiob (95% CI) | p value |
|---|---|---|---|---|---|
| Direct actinga | 457 (1.5) | 1832 (3.2) | 0.44 (0.39–0.48) | 0.85 (0.74–0.98) | 0.0223 |
| Antivitamin Ka | 778 (2.6) | 3782 (6.7) | 0.40 (0.37–0.43) | 0.83 (0.74–0.92) | 0.0007 |
| LMWHa | 663 (2.2) | 2023 (3.6) | 0.65 (0.60–0.72) | 0.97 (0.88–1.08) | 0.5698 |
| Othersa | 1 (0.0) | 3 (0.0) | 0.47 (0.27–0.80) | 0.37 (0.04–3.28) | 0.3727 |
Others = Fondaparinux and protein c
CI confidence interval, LMWH low-molecular-weight heparins
aConsumption of one type of anticoagulant does not exclude the fact that the patient may consume another type of anticoagulant
bAdjusted for sex, age, and comorbidities: hypertension, diabetes, COPD (chronic obstructive pulmonary disease), obesity, ischaemic heart disease, cerebrovascular accident, heart failure, atrial fibrillation, chronic renal failure, cancer, asthma, current smoker, current use of other pharmacological treatment (antihypertensives, diuretics, non-steroidal anti-inflammatory drugs, hypolipidemic agents, anticoagulants, antiplatelet agents and glucocorticoids) and polypharmacy (number of different treatments)
Discussion
The results of this large-scale, population-based, case–control study show that neither antivitamin K anticoagulants nor direct-acting anticoagulants have an effect on the risk of progression to severe COVID-19. The results of our study also show that antivitamin K anticoagulants were associated with a significantly lower risk of hospitalization, which, in part, can be explained by a decreased risk of the susceptibility to infection. The use of direct-acting anticoagulants was not associated with the risk of hospitalization, although it also seems to decrease susceptibility. It has also been observed that LMWH are associated with an increased risk of progression to severe COVID-19. This difference in the effects found for each type of anticoagulant underscores the importance of analysing and interpreting the effect of each separately.
Our study indicates that use of antivitamin K anticoagulants (mainly acenocumarol in our study, as it is the most widely used in Spain) [22] is associated with an increased risk of hospitalization by over 20% (OR 0.77 [95% CI 0.64–0.93]), but no effect on risk of progression to severe COVID-19 was observed. We found no study that evaluated risk of hospitalization for users of antivitamin K anticoagulants versus non-users of anticoagulants. The only two studies that we managed to locate compared the use of antivitamin K anticoagulants with that of direct-acting anticoagulants, revealing inconsistencies between them (i.e., while Wong et al. [23] found a lower risk of hospitalization for antivitamin K anticoagulants, Rivera-Caravaca et al. [24] reported no differences).
With respect to susceptibility to infection (PCR COVID-19 −/+), our results suggest that the effect found on hospitalization with antivitamin K may be due to the fact that the consumption of antivitamin K anticoagulants decreases the risk by 17% (OR 0.83 [95% CI 0.74–0.92]) compared with non-consumers of anticoagulants. We found only one study [11] that allowed for comparison with our results, and this found no association between use of warfarin (antivitamin K anticoagulant) and susceptibility to the virus.
Furthermore, our data do not support the hypothesis that the use of direct-acting anticoagulants has a significant effect on the risk of hospitalization, nor on progression, findings in line with Flam et al. [25]. In contrast, the study by Rivera-Caravaca et al. [24] observed a higher risk of hospitalization after receiving direct-acting anticoagulants, possibly because this was a retrospective cohort of elderly COVID-19+ patients, in which there may have been misclassification in the COVID-19 variable.
With reference to heparins (LMWHs), our results afford evidence to show that these could increase the risk of hospitalization close to statistical significance (OR 1.17 [95% CI 0.99–1.39]; p = 0.0699), with an increased risk of progression to severe COVID-19 (OR 1.25 [95% CI 1.01–1.55]). To our knowledge, there are no published studies with which our findings can be compared.
It might be thought that the possible decreased risk of hospitalization associated with the use of antivitamin K anticoagulants would be due to non-clinical factors, such as stricter follow-up of social-distancing regulations owing to greater aversion to risk [26]. However, our data show that the susceptibility of antivitamin K anticoagulants is very similar to that of direct-acting anticoagulants (OR 0.83 [95% CI 0.74–0.92] vs OR 0.85 [95% CI 0.74–0.98]), which might suggest that social aversion does not have a major impact as initially thought, and that this decrease in susceptibility is due to their anticoagulant effect. In contrast to the effects found for antivitamin K anticoagulants, our data show no association between the risk of hospitalization and the use of direct-acting anticoagulants. This could be due to the absence of effect on desphospho-uncarboxylated matrix Gla protein (dp-ucMGP) [26], or to the fact that in Spain, direct-acting anticoagulants are frequently prescribed for polypathological, polymedicated patients, with greater drug interactions than those treated with antivitamin K anticoagulants [27].
In contrast, heparins (LMWHs) are used for prolonged treatment in more exceptional situations and in onco-haematological patients, among whom there is a high risk of venous thrombosis and haemorrhages [28]. This could account for the higher risk of progression to severe COVID-19, possibly due to residual confounding attributable to the severity of the disease and such a high risk of thromboembolic events, which may remain despite the adjustment made for diagnosis of other comorbidities.
Lastly, it should be borne in mind that there is little literature with which to compare our findings. In the first place, this is because most of the published studies on anticoagulant use tend to study outcomes—essentially mortality—at an inpatient or hospital level [27–34].
Advantages and Limitations
The great advantage of our study is its sample size. Moreover, to our knowledge, it is the first population-based, case–control design study able to provide robust data on the effects of anticoagulant use.
This study had a large number of variables of adjustment for comorbidities and use of medications, both with a high degree of validity due to the very low risk of misclassification [35]: in the former case, because these demonstrated their validity when compared against external comorbidity records, and in the latter case, because this is an administrative database free of any risk of misclassification, being the source from which the PHS pays the pharmacies that dispense the medications. There would only be the possibility of (1) presence of concomitant over-the-counter (OTC) treatments, which are not recorded in our data; (2) private healthcare prescriptions and (3) adherence to treatment other than that prescribed. Even so, we feel that this does not constitute an important study limitation, because (1) anticoagulants are not OTC medications and (2) the majority of the study population (98%) is covered by the PHS. Furthermore, unlike other prescription-based data sources, our data are based on medications actually acquired from pharmacies (dispensing) rather than prescriptions issued, which is an added advantage.
The sampling of susceptibility to infection and progression to severe COVID-19 controls does not exactly follow sampling by incidence density. However, during 2020, admissions would be proportional to COVID-19 cases. Therefore, in practice we may consider that the controls for the susceptibility to infection and progression to severe COVID-19 models could be considered as selected by incidence density. It might be thought that the lack of age matching could be a limitation (given that hospitalized patients are generally older than unhospitalized). Nevertheless, in our study there is a clear overlap between the age distribution of cases and controls, which allows statistical adjustment to control for differences in both groups. Moreover, according to many authors, the lack of matching in case–control studies does not produce bias, only a decrease in efficiency [13, 16].
Finally, and as in all observational studies with a secondary database, one cannot rule out the possibility of there being confounding variables that were not measured or might have undergone a certain degree of misclassification. In those variables that were collected (such as indications and diseases), data on the level of severity were not available, something that may pose a certain risk of residual confounding by indication.
Conclusion
The results of this study have shown that antivitamin K anticoagulants and direct-acting anticoagulants do not increase the risk of progression to more severe stages. This would imply that it would not be justified to modify these treatments in anticoagulated and COVID-19 positive patients. On the other hand, consumption of antivitamin K anticoagulants was associated with a lower risk of hospitalization and susceptibility to infection. Additional studies are needed to compare the results and increase the level of evidence in this field.
Supplementary Information
Below is the link to the electronic supplementary material.
Acknowledgments
The authors would like to thank The General Directorate of Health Care of the Galician Health Service for furnishing the data necessary to conduct this study.
Funding
Open Access funding provided thanks to the CRUE-CSIC agreement with Springer Nature.
Declarations
Funding
This study was sponsored by the Carlos III Institute of Health via the “COV20/00470” project (co-funded by the European Regional Development Fund, “A way to make Europe”). Funding for open access charge by Universidade de Santiago de Compostela/CISUG.
Conflict of interest
The authors declare they have no conflicts of interest.
Ethical approval
The study was approved by the Galician Clinical Research Ethics Committee (Comité de Ética de Investigación de Galicia), reference 2020/349.
Consent to participate and consent for publication
Consent from participants was not required as this study extracted anonymous data from an electronic health database.
Availability of data and material
The datasets generated and analysed during the current study are not publicly available due to Galician Public Health System restrictions.
Code availability
Not applicable.
Author contributions
MZC: Designed study, wrote paper; APC: designed study, wrote paper; MPR: designed study, revised paper; ECM: designed study, revised paper; MLO: designed study, revised paper; MPL: designed study, analysed data, revised paper; SC: analysed data, revised paper; ASB: designed study, analysed data, revised paper; AF: designed study, analysed data, revised paper. All authors read and approved the final version.
REFERENCES
- 1.Sen-Crowe B, McKenney M, Elkbuli A. Disparities in global COVID-19 vaccination rates and allocation of resources to countries in need. Ann Med Surg. 2021;68:102620. doi: 10.1016/j.amsu.2021.102620. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Laporte J-R. In the midst of the SARS-CoV-2 pandemia, caution is needed with commonly used drugs that increase the risk of pneumonia. In: Medications-compromising-Covid-infections. https://www.isdbweb.org/wp-content/uploads/Medications-Compromising-Covid-Infections-.pdf.
- 3.McKeigue PM, Kennedy S, Weir A, Bishop J, McGurnaghan SJ, McAllister D, et al. Relation of severe COVID-19 to polypharmacy and prescribing of psychotropic drugs: the REACT-SCOT case–control study. BMC Med. 2021;19:51. doi: 10.1186/s12916-021-01907-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Prasad M, Leon M, Lerman LO, Lerman A. Viral endothelial dysfunction: a unifying mechanism for COVID-19. Mayo Clin Proc. 2021;96:3099–3108. doi: 10.1016/j.mayocp.2021.06.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Casella IB. Physiopathology of SARS-CoV-2-infection-associated thrombosis. J Vasc Bras. 2020;19:e20200128. doi: 10.1590/1677-5449.200128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Canoğlu K, Şaylan B, Çalışkan T. COVID-19 and thrombosis: prophylaxis and management. Tuberk Toraks. 2021;69:269–278. doi: 10.5578/tt.20219818. [DOI] [PubMed] [Google Scholar]
- 7.Kohansal Vajari M, Shirin M, Pourbagheri-Sigaroodi A, Akbari ME, Abolghasemi H, Bashash D. COVID-19-related coagulopathy: a review of pathophysiology and pharmaceutical management. Cell Biol Int. 2021;45:1832–1850. doi: 10.1002/cbin.11623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Kollias A, Kyriakoulis KG, Dimakakos E, Poulakou G, Stergiou GS, Syrigos K. Thromboembolic risk and anticoagulant therapy in COVID-19 patients: emerging evidence and call for action. Br J Haematol. 2020;189:846–847. doi: 10.1111/bjh.16727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Zhou J, Lee S, Guo CL, Chang C, Liu T, Leung KSK, et al. Anticoagulant or antiplatelet use and severe COVID-19 disease: a propensity score-matched territory-wide study. Pharmacol Res. 2021;165:105473. doi: 10.1016/j.phrs.2021.105473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Togano T, Uemura Y, Asai Y, Hayakawa K, Matsunaga N, Terada M, et al. The influence of pre-admission antiplatelet and anticoagulation therapy on the illness severity in hospitalized patients with COVID-19 in Japan. J Infect Chemother. 2021;27:1498–1503. doi: 10.1016/j.jiac.2021.07.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Kavecansky J, Dusendang JR, Tavakoli J, Schmittdiel J, Ho G, Loyles J, et al. Association of anticoagulation use with SARS-CoV2 detection. Thromb Res. 2021;198:99–102. doi: 10.1016/j.thromres.2020.11.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Satué-Gracia EM, Vila-Córcoles A, de Diego-Cabanes C, Vila-Rovira A, Torrente-Fraga C, Gómez-Bertomeu F, et al. Susceptibility and risk of SARS-COV-2 infection among middle-aged and older adults in Tarragona area. Spain. Med Clin (Barc) 2021;S0025–7753(21):00240–242. doi: 10.1016/j.medcli.2021.03.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Rothman KJ, Greenland S, Lash TL. Case–control studies. In: Melnick EL, Everitt BS, editors. Encyclopedia of quantitative risk analysis and assessment. 3a. Chichester: Wiley; 2008. p. risk0599. [Google Scholar]
- 14.de Abajo FJ, Rodríguez-Martín S, Lerma V, Mejía-Abril G, Aguilar M, García-Luque A, et al. Use of renin-angiotensin-aldosterone system inhibitors and risk of COVID-19 requiring admission to hospital: a case-population study. Lancet. 2020;395:1705–1714. doi: 10.1016/S0140-6736(20)31030-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Pearce N. Analysis of matched case–control studies. BMJ. 2016;352:i969. doi: 10.1136/bmj.i969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Rose S, Laan MJ. Why match? Investigating matched case-control study designs with causal effect estimation. Int J Biostat. 2009;5(1):1. doi: 10.2202/1557-4679.1127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Huber CA, Szucs TD, Rapold R, Reich O. Identifying patients with chronic conditions using pharmacy data in Switzerland: an updated mapping approach to the classification of medications. BMC Public Health. 2013;30(13):1030. doi: 10.1186/1471-2458-13-1030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Brown H, Prescott R. Applied mixed models in medicine. 2a. New York: Wiley; 2006. [Google Scholar]
- 19.Pinheiro JC, Bates DM. Mixed-effects models in S and S-PLUS. 1a. Berlin: Springer; 2000. [Google Scholar]
- 20.Stroup WW. Generalized linear mixed models: modern concepts, methods and applications. 1a. Boca Raton: CRC Press; 2021. [Google Scholar]
- 21.Bates D, Mächler M, Bolker B, Walker S. Fitting linear mixed-effects models using lme4. J Stat Softw. 2015;67:1–48. doi: 10.18637/jss.v067.i01. [DOI] [Google Scholar]
- 22.López de Luca B, Murgadella Sancho A, Padullés Zamora A, Serrano Arjona O, Roca Massa M, March Pujol M. Acenocumarol. Offarm. 2006;25:90–95. [Google Scholar]
- 23.Wong AY, Tomlinson L, Brown JP, Elson W, Walker AJ, Schultze A, et al. Association between oral anticoagulants and COVID-19-related outcomes: a population-based cohort study. Br J Gen Pract. 2022;72:e456-e463. 10.3399/BJGP.2021.0689. [DOI] [PMC free article] [PubMed]
- 24.Rivera-Caravaca JM, Buckley BJR, Harrison SL, Fazio-Eynullayeva E, Underhill P, Marín F, et al. Direct-acting oral anticoagulants use prior to COVID-19 diagnosis and associations with 30-day clinical outcomes. Thromb Res. 2021;205:1–7. doi: 10.1016/j.thromres.2021.06.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Flam B, Wintzell V, Ludvigsson JF, Mårtensson J, Pasternak B. Direct oral anticoagulant use and risk of severe COVID-19. J Intern Med. 2021;289:411–419. doi: 10.1111/joim.13205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.OpenSAFELY Collaborative. Wong AYS, Tomlinson LA, Brown JP, Elson W, Walker AJ, et al. Association between warfarin and COVID-19-related outcomes compared with direct oral anticoagulants: population-based cohort study. J Hematol Oncol. 2021;14:172. doi: 10.1186/s13045-021-01185-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Dela Figuera M, Prieto MA, Marín N, Egocheaga I, Cinza S, en nombre de los investigadores del estudio SILVER BRONCE-AP Differences in the management of patients with atrial fibrillation according to whether primary care or the specialist initiates treatment with direct oral anticoagulants. The SILVER-AP and BRONCE-AP studies. SEMERGEN. 2018;44:323–334. doi: 10.1016/j.semerg.2017.09.005. [DOI] [PubMed] [Google Scholar]
- 28.Grupo de Trabajo SEOM de Tromboembolismo y CánceR. Recurrencia de tromboembolismo venoso en el paciente oncológico: ¿Se puede predecir? ¿Cómo manejarla? IN: SEOM: Sociedad Española de Oncología Médica. 2019. https://seom.org/formacion/formacion-on-line/revisiones-cientificas/105818-recurrencia-de-tromboembolismo-venoso-en-el-paciente-oncologico-ise-puede-predecir-icomo-manejarla. Accessed 11 Feb 2022.30.
- 29.Portillo J. Prophylaxis of venous thromboembolism disease in patients with cancer. Rev Clin Esp. 2020;2565:30133–30138. doi: 10.1200/JCO.2013.49.1118. [DOI] [PubMed] [Google Scholar]
- 30.Cippà PE, Cugnata F, Ferrari P, Brombin C, Ruinelli L, Bianchi G, et al. A data-driven approach to identify risk profiles and protective drugs in COVID-19. Proc Natl Acad Sci USA. 2021;118:e2016877118. doi: 10.1073/pnas.2016877118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Ayerbe L, Risco C, Ayis S. The association between treatment with heparin and survival in patients with Covid-19. J Thromb Thrombolysis. 2020;50:298–301. doi: 10.1007/s11239-020-02162-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Paranjpe I, Fuster V, Lala A, Russak AJ, Glicksberg BS, Levin MA, et al. Association of treatment dose anticoagulation with in-hospital survival among hospitalized patients with COVID-19. J Am Coll Cardiol. 2020;76:122–124. doi: 10.1016/j.jacc.2020.05.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Chocron R, Galand V, Cellier J, Gendron N, Pommier T, Bory O, et al. Anticoagulation before hospitalization is a potential protective factor for COVID-19: insight from a French Multicenter Cohort Study. J Am Heart Assoc. 2021;10:e018624. doi: 10.1161/JAHA.120.018624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Fröhlich GM, Jeschke E, Eichler U, Thiele H, Alhariri L, Reinthaler M, et al. Impact of oral anticoagulation on clinical outcomes of COVID-19: a nationwide cohort study of hospitalized patients in Germany. Clin Res Cardiol. 2021;110:1041–1050. doi: 10.1007/s00392-020-01783-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Prada-Ramallal G, Takkouche B, Figueiras A. Bias in pharmacoepidemiologic studies using secondary health care databases: a scoping review. BMC Med Res Methodol. 2019;19:53. doi: 10.1186/s12874-019-0695-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.