Summary
Management of the patient with sepsis comprises three key branches: control of the underlying infection, haemodynamic stabilization, and modulation of the host response. Each aspect should be considered in all patients and, when relevant, managed at the same time. Infection control is applicable to all patients with sepsis and will include antibiotic therapy and often surgical intervention to remove an infectious source. Haemodynamic support involves fluid administration in all patients and vasoactive agents in patients with associated circulatory shock. Noradrenaline is the first choice vasopressor agent; inotropic agents, usually dobutamine, may be added in case of myocardial depression. No interventions directed at individual components of the host response to sepsis have yet been shown to improve outcomes, but glucocorticoids and vasopressin have a global impact on the response and can thus be considered in this category. A move toward more personalized treatment is needed across all three arms of sepsis management.
Keywords: Dobutamine, Lactate, Haemodynamic monitoring, Personalization, Organ failure
Introduction
Sepsis is a severe, systemic infection. It is therefore associated with some degree of organ dysfunction1 and requires urgent action. Without prompt and effective intervention, mortality rates rapidly exceed 30–35%.2
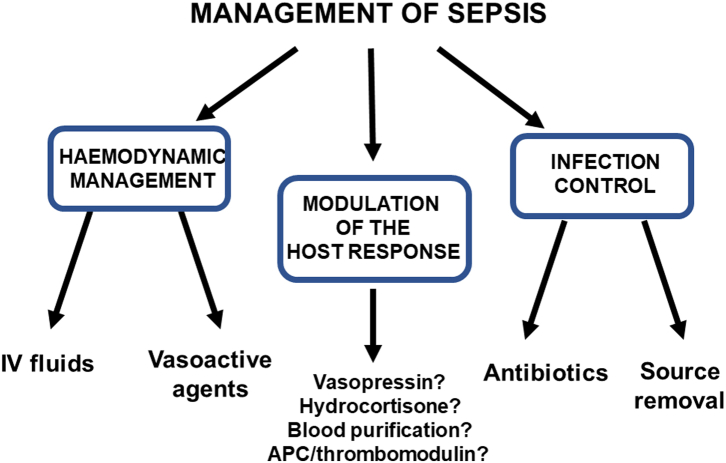
The management of sepsis includes three major aspects, which are summarized in Fig. 1. The first element, infection control, is applicable to all cases of sepsis. Haemodynamic stabilization is relevant essentially when there is associated circulatory shock. Modulation of the host response, with the interventions currently available, is also primarily applicable in cases of septic shock. Organ support, for example mechanical ventilation or renal replacement therapy, will be needed in some patients.
Fig. 1.
The three arms of sepsis management. APC, activated protein C.
Management of sepsis
Infection control
Infection is the underlying cause of sepsis and responsible for the initiation and perpetuation of immune dysregulation. As such, every attempt must be made to eradicate the infection and its source of origin. Antibiotics should be given in all cases, even when infection cannot be definitively proven and no specific microorganism is isolated, which can occur in more than 30% of sepsis patients.3,4 Antibiotics should be administered very early, especially when sepsis is severe, i.e., in septic shock. However, the concept that antibiotics must be administered within 1 h of identification of sepsis5 is not supported by evidence.6, 7, 8 Importantly, it is not only the time when the antibiotics are ordered that counts, but also the time when they are actually given to the patient as there may be a delay between prescription and administration.9
Whenever possible, cultures from blood and all relevant, potentially infected sites should be obtained before antibiotics are started. The initial antibiotic spectrum should cover all likely organisms but not more. Expected pathogens and likely antimicrobial sensitivities will vary according to the origin of the infection, recent use of antimicrobial agents, and local microbial patterns and antimicrobial resistance, so these aspects must be taken into account when making empirical antibiotic choices. There is some evidence that using too large a spectrum may contribute to the emergence of resistant organisms in recurrent infections with all the potentially negative effects this can have on outcomes.10
Source control may necessitate surgical intervention with drainage or debridement of infected loci, or removal of (potentially) infected catheters or devices. As with antibiotic administration, these procedures should be performed without delay.
Haemodynamic management
Haemodynamic management includes fluids in all cases, with vasoactive agents administered primarily in the presence of shock. Fluid administration is always required because of the external fluid losses (including perspiration) often present in sepsis as well as internal fluid loss (including the extravascular passage of fluid into the interstitium as a consequence of the inevitable alteration in microvascular permeability). Patients with sepsis may also be dehydrated because of reduced fluid intake and sepsis is typically associated with vasodilation, which increases blood volume.
It is not possible to guess or determine in advance how much fluid each patient should be given as there are too many factors influencing fluid requirements, including the degree of fluid loss from the factors identified above, the patient's cardiac function, and how long since the onset and/or recognition of sepsis. Therefore, the amount of fluid administered must be individualized and ongoing fluid needs regularly reassessed to limit the risks of hypervolemia, which can negatively affect gas exchange, renal function, wound healing, and cognition, among others.11 The traditional approach to evaluate fluid requirements is the fluid challenge technique, in which a small amount of fluid (usually around 200 mL of a crystalloid solution) is given over a short period (usually 10 min) and the patient's response is evaluated using pre-established objectives (e.g., 10% increase in cardiac output) and safety markers (e.g., central venous pressure).12 This strategy can be repeated as necessary.
Importantly, the blood pressure response to fluid administration is not always immediate, because the associated decrease in vascular tone can increase blood flow without increasing arterial pressure. It is therefore preferable to evaluate the cardiac output (or stroke volume) response. This can be done using any technique (e.g., thermodilution, pulse contour analysis, echocardiography, bioreactance, …). If flow does not increase in response to the fluid, fluid administration should be stopped. A smart evaluation of fluid responsiveness without giving any fluid can be done by a passive leg raising test13 but this is relatively complicated in non-sedated patients who may have a stress response to the change in position. Also, if present, the increase in stroke volume will be mild and transient and this is not easy to detect. Other techniques based on heart/lung interactions can be considered in mechanically ventilated, profoundly sedated patients. However, the use of sedative agents has decreased considerably in recent years as septic patients are typically obtunded so sedation is less necessary; moreover, sedative agents can negatively affect haemodynamic status by decreasing myocardial contractility and vascular tone.14
As with quantity of fluid, the type of fluid should also be individualized with no one type recommended for all patients. Crystalloids are usually recommended first.15 Albumin, a natural colloid, could be added when relatively large amounts of crystalloids have been given. The use of large amounts of 0.9% (normal) saline solution can result in hyperchloraemic acidosis that may have untoward renal effects by altering renal haemodynamics.16 If blood chloride levels increase, balanced crystalloids, with various electrolyte compositions may be preferable.
Fluids alone may not be sufficient to restore an adequate mean arterial pressure (MAP) so that addition of a vasopressor agent may be required. Noradrenaline is the vasopressor agent of first choice, as it is a natural molecule, has a short half-life, and is not a pure vasopressor: it predominantly has alpha-adrenergic properties but also some beta-adrenergic properties, resulting in vasoconstriction but little effect on the heart rate. Noradrenaline administration can be started early to avoid any episode of profound hypotension, which always has deleterious effects on the organs. Although an initial MAP of 65 mmHg has been recommended,15 the blood pressure target should be individualized14; in particular, patients with a history of hypertension may require a higher blood pressure than other patients. In each case, the clinical response to an increase in the dose of a vasopressor agent should be assessed, including effects on cutaneous perfusion, urine output, and mentation. Careful attention should be paid to making sure that blood flow is preserved, because vasopressors can cause excessive vasoconstriction thus impairing blood flow and hence reducing organ and tissue perfusion.
If there is any suspicion of myocardial depression, manifest by intolerance to fluid and signs of decreased contractility on echography, then prudent administration of an inotropic agent could be attempted; dobutamine, initially at a dose of only a few µg/kg/min, is the first line inotropic agent.
Modulation of the host response
No strategies targeting individual elements of the immune response have so far been shown to consistently improve outcomes in randomized clinical trials or are commercially available. Currently, this category of therapeutic interventions thus includes strategies that affect more general aspects of the immune response. The most widely used adjunct intervention in this context is the administration of glucocorticoids, which are known to have various effects on inflammation including via nuclear factor kappa B.17 Corticosteroid administration is reserved for patients with severe septic shock, which is usually defined by the need for relatively high doses of noradrenaline to maintain arterial pressure, approaching or exceeding 1 μg/kg/min. A randomized controlled trial of 3800 patients conducted in Australasia18 showed no improvement in survival with moderate doses of hydrocortisone, but the patients were not very ill (placebo mortality rate 29%18); blood lactate levels were not reported. By contrast, in a multicentre trial in France in which the placebo patients had a mortality rate close to 50% and a mean lactate level greater than 4 mmol/l there were significant reductions in ICU, hospital, and 6-month mortality rates in hydrocortisone treated patients.19 Hence, a dose of 200 mg of hydrocortisone per day is recommended in patients with severe septic shock.15
Vasopressin can also be used in the context of host response modulation. In addition to its vasopressor properties, vasopressin is a hormone that influences kidney function and water metabolism. The underlying concept is that exogenous vasopressin administration in patients with sepsis can substitute inadequate vasopressin levels, with a fixed dose of 0.03–0.05 units/min. Vasopressin is mostly used as an additional second-line vasopressor for patients who do not respond to noradrenaline, but there is some evidence that early administration of vasopressin in patients with septic shock may improve kidney function, increase urine output, decrease fluid requirements, and decrease oedema formation.20
Extracorporeal blood purification using different systems to remove excess mediators and/or endotoxin has also been proposed and with good rationale, but none of the modalities available has been consistently shown to reduce mortality.15 The early use of renal replacement therapy in the absence of renal failure is not recommended.15
Vitamin C may also have anti-inflammatory effects, notably at the endothelial level,21 but has shown limited efficacy in large, randomized clinical trials and administration of high doses of vitamin C in patients with sepsis may be harmful.22
Outstanding questions
One of the main problems when trying to translate preclinical or phase I clinical trial data to the larger clinical arena has been the heterogeneity of the patient populations studied. Sepsis is an ‘umbrella’ term that covers patients with many diverse characteristics related to their underlying infection, the degree and phase of immune response, as well as intrinsic differences in age, sex, genomics, comorbid conditions, and chronic treatments. Thus, despite promising results of some sepsis interventions in the relatively controlled, carefully selected conditions of early phase clinical trials, none has been shown to have a convincing beneficial effect on survival in large scale RCTs, which have all included heterogeneous groups of patients with “sepsis”. Drotrecogin alpha (activated) was a possible exception, with treated patients having reduced mortality in a landmark study,23 but a subsequent study was entirely negative so that the drug was removed from the market.24
So, how can the translation of research data be improved in sepsis? We are increasingly moving towards applying a more personalized approach to patient management across most medical spheres, in which treatments are adapted to individual patients based on more accurate assessment of their likelihood to respond. Phenotypic grouping of patients with sepsis based on the presence of visible characteristics is gradually being replaced by a pathogenetic-based endotype-driven approach (defined by a distinct pathobiological mechanism). Using such endotypes to select patients more appropriately for clinical trials in sepsis may be a key step toward improving translation of research data to the clinical arena. For example, a retrospective analysis of data from a trial of thrombomodulin administration showed encouraging results in patients with sepsis-associated coagulopathy and raised baseline coagulation markers (prothrombin fragment 1.2 F1.2, thrombin-antithrombin complexes, and D-dimer),25 suggesting this characteristic could be used to select patients most likely to respond. The impact different patient endotypes can have on response to treatment is also becoming more apparent. Post hoc or secondary trial data analysis has suggested differential effects of treatments, including IL-1 receptor antagonist26 and corticosteroids,27,28 according to the presence of different endotypes, determined based on the presence of specific biomarkers or a transcriptomic profile. ‘Omics techniques involve the measurement of an entire class of compounds (e.g., DNA for genomics, RNA for transcriptomics, proteins for proteomics, metabolites for metabolomics, etc), and enable characterization of multiple patient factors rather than just a few molecules.29 With improved computing power and technology, enabling increased analysis of the vast amounts of data that are now being collected from patients, and the advances in polyomics approaches, patient profiling will become increasingly precise. Use of such methods to enrich clinical trials needs further testing and validation but holds huge promise for the future of sepsis therapeutics.
Conclusions
The management of sepsis involves the three elements presented herein—infection control, haemodynamic stabilization, and modulation of the host response—which should be kept in consideration at all times during a patient's trajectory. Improved characterization of patients with sepsis should enable more appropriate, personalized selection of therapies in the future and thus improved outcomes.
Search strategy and selection criteria
I searched PubMed for original research papers, reviews, editorials, and commentaries published in English using the following terms: sepsis and septic shock; combined with randomized controlled trial; treatment; therapy. I predominantly selected publications from the past 5 years, but also included commonly referenced and highly regarded older publications. I also checked the reference lists of articles identified by this search strategy for any relevant articles. Clinical trial databases were searched for new and currently ongoing studies in this field.
Contributors
JLV wrote the manuscript.
Declaration of interests
JLV has no conflicts of interest related to this article.
Acknowledgement
No funding was received for the manuscript.
References
- 1.Singer M., Deutschman C.S., Seymour C.W., et al. The third international consensus definitions for sepsis and septic shock (Sepsis-3) JAMA. 2016;315:801–810. doi: 10.1001/jama.2016.0287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Vincent J.L., Jones G., David S., Olariu E., Cadwell K.K. Frequency and mortality of septic shock in Europe and North America: a systematic review and meta-analysis. Crit Care. 2019;23:196. doi: 10.1186/s13054-019-2478-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Lakbar I., Munoz M., Pauly V., et al. Septic shock: incidence, mortality and hospital readmission rates in French intensive care units from 2014 to 2018. Anaesth Crit Care Pain Med. 2022;41 doi: 10.1016/j.accpm.2022.101082. [DOI] [PubMed] [Google Scholar]
- 4.Sakr Y., Jaschinski U., Wittebole X., et al. Sepsis in intensive care unit patients: worldwide data from the intensive care over nations audit. Open Forum Infect Dis. 2018;5:ofy313. doi: 10.1093/ofid/ofy313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Levy M.M., Evans L.E., Rhodes A. The surviving sepsis campaign bundle: 2018 update. Crit Care Med. 2018;46:997–1000. doi: 10.1097/CCM.0000000000003119. [DOI] [PubMed] [Google Scholar]
- 6.Sterling S.A., Miller W.R., Pryor J., Puskarich M.A., Jones A.E. The impact of timing of antibiotics on outcomes in severe sepsis and septic shock: a systematic review and meta-analysis. Crit Care Med. 2015;43:1907–1915. doi: 10.1097/CCM.0000000000001142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Im Y., Kang D., Ko R.E., et al. Time-to-antibiotics and clinical outcomes in patients with sepsis and septic shock: a prospective nationwide multicenter cohort study. Crit Care. 2022;26:19. doi: 10.1186/s13054-021-03883-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ruddel H., Thomas-Ruddel D.O., Reinhart K., et al. Adverse effects of delayed antimicrobial treatment and surgical source control in adults with sepsis: results of a planned secondary analysis of a cluster-randomized controlled trial. Crit Care. 2022;26:51. doi: 10.1186/s13054-022-03901-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Taylor S.P., Anderson W.E., Beam K., Taylor B., Ellerman J., Kowalkowski M.A. The association between antibiotic delay intervals and hospital mortality among patients treated in the emergency department for suspected sepsis. Crit Care Med. 2021;49:741–747. doi: 10.1097/CCM.0000000000004863. [DOI] [PubMed] [Google Scholar]
- 10.Rhee C., Kadri S.S., Dekker J.P., et al. Prevalence of antibiotic-resistant pathogens in culture-proven sepsis and outcomes associated with inadequate and broad-spectrum empiric antibiotic use. JAMA Netw Open. 2020;3 doi: 10.1001/jamanetworkopen.2020.2899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Vincent J.L. Fluid management in the critically ill. Kidney Int. 2019;96:52–57. doi: 10.1016/j.kint.2018.11.047. [DOI] [PubMed] [Google Scholar]
- 12.Vincent J.L., Cecconi M., De Backer D. The fluid challenge. Crit Care. 2020;24:703. doi: 10.1186/s13054-020-03443-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Monnet X., Teboul J.L. Passive leg raising: five rules, not a drop of fluid! Crit Care. 2015;19:18. doi: 10.1186/s13054-014-0708-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Vincent J.L., Singer M., Einav S., et al. Equilibrating SSC guidelines with individualized care. Crit Care. 2021;25:397. doi: 10.1186/s13054-021-03813-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Evans L., Rhodes A., Alhazzani W., et al. Surviving sepsis campaign: international guidelines for management of sepsis and septic shock 2021. Intensive Care Med. 2021;47:1181–1247. doi: 10.1007/s00134-021-06506-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Chowdhury A.H., Cox E.F., Francis S.T., Lobo D.N. A randomized, controlled, double-blind crossover study on the effects of 2-L infusions of 0.9% saline and plasma-lyte(R) 148 on renal blood flow velocity and renal cortical tissue perfusion in healthy volunteers. Ann Surg. 2012;256:18–24. doi: 10.1097/SLA.0b013e318256be72. [DOI] [PubMed] [Google Scholar]
- 17.Heming N., Sivanandamoorthy S., Meng P., Bounab R., Annane D. Immune effects of corticosteroids in sepsis. Front Immunol. 2018;9:1736. doi: 10.3389/fimmu.2018.01736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Venkatesh B., Finfer S., Cohen J., et al. Adjunctive glucocorticoid therapy in patients with septic shock. N Engl J Med. 2018;378:797–808. doi: 10.1056/NEJMoa1705835. [DOI] [PubMed] [Google Scholar]
- 19.Annane D., Renault A., Brun-Buisson C., et al. Hydrocortisone plus fludrocortisone for adults with septic shock. N Engl J Med. 2018;378:809–818. doi: 10.1056/NEJMoa1705716. [DOI] [PubMed] [Google Scholar]
- 20.Vincent J.L., Post E.H. Sepsis: vasopressin: a first-line agent for septic shock? Nat Rev Nephrol. 2016;12:718–719. doi: 10.1038/nrneph.2016.144. [DOI] [PubMed] [Google Scholar]
- 21.Vincent J.L., Ince C., Pickkers P. Endothelial dysfunction: a therapeutic target in bacterial sepsis? Expert Opin Ther Targets. 2021;25:733–748. doi: 10.1080/14728222.2021.1988928. [DOI] [PubMed] [Google Scholar]
- 22.Lamontagne F., Masse M.H., Menard J., et al. Intravenous vitamin C in adults with sepsis in the intensive care unit. N Engl J Med. 2022;386:2387–2398. doi: 10.1056/NEJMoa2200644. [DOI] [PubMed] [Google Scholar]
- 23.Bernard G.R., Vincent J.L., Laterre P.F., et al. Efficacy and safety of recombinant human activated protein C for severe sepsis. N Engl J Med. 2001;344:699–709. doi: 10.1056/NEJM200103083441001. [DOI] [PubMed] [Google Scholar]
- 24.Ranieri V.M., Thompson B.T., Barie P.S., et al. Drotrecogin alfa (activated) in adults with septic shock. N Engl J Med. 2012;366:2055–2064. doi: 10.1056/NEJMoa1202290. [DOI] [PubMed] [Google Scholar]
- 25.Levi M., Vincent J.L., Tanaka K., et al. Effect of a recombinant human soluble thrombomodulin on baseline coagulation biomarker levels and mortality outcome in patients with sepsis-associated coagulopathy. Crit Care Med. 2020;48:1140–1147. doi: 10.1097/CCM.0000000000004426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Shakoory B., Carcillo J.A., Chatham W.W., et al. Interleukin-1 receptor blockade is associated with reduced mortality in sepsis patients with features of macrophage activation syndrome: reanalysis of a prior phase III trial. Crit Care Med. 2016;44:275–281. doi: 10.1097/CCM.0000000000001402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Antcliffe D.B., Burnham K.L., Al-Beidh F., et al. Transcriptomic signatures in sepsis and a differential response to steroids: from the VANISH randomized trial. Am J Respir Crit Care Med. 2018;199:980–986. doi: 10.1164/rccm.201807-1419OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Wong H.R., Hart K.W., Lindsell C.J., Sweeney T.E. External corroboration that corticosteroids may be harmful to septic shock endotype A patients. Crit Care Med. 2021;49:e98–e101. doi: 10.1097/CCM.0000000000004709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Schuurman A.R., Reijnders T.D.Y., Kullberg R.F.J., Butler J.M., van der Poll T., Wiersinga W.J. Sepsis: deriving biological meaning and clinical applications from high-dimensional data. Intensive Care Med Exp. 2021;9:27. doi: 10.1186/s40635-021-00383-x. [DOI] [PMC free article] [PubMed] [Google Scholar]