Abstract
Chloroplast DNA (cpDNA) encodes up to 315 (typically, 120–130) genes1, including those for essential components in photosystems I and II and the large subunit of RuBisCo, which catalyses CO2 fixation in plants. Targeted mutagenesis in cpDNA will be broadly useful for studying the functions of these genes in molecular detail and for developing crops and other plants with desired traits. Unfortunately, CRISPR–Cas9 and CRISPR-derived base editors, which enable targeted genetic modifications in nuclear DNA, are not suitable for organellar DNA editing2, owing to the difficulty of delivering guide RNA into organelles. CRISPR-free, protein-only base editors (including DddA-derived cytosine base editors3–8 and zinc finger deaminases9), originally developed for mitochondrial DNA editing in mammalian cells, can be used for C-to-T, rather than A-to-G, editing in cpDNA10–12. Here we show that heritable homoplasmic A-to-G edits can be induced in cpDNA, leading to phenotypic changes, using transcription activator-like effector-linked deaminases13.
Subject terms: Genetic engineering, Molecular engineering in plants
Plant-optimized transcription activator-like effector-linked deaminases enable site-specific A-to-G base editing in the chloroplast genome, leading to heritable homoplasmic base conversions and phenotypic changes.
Main
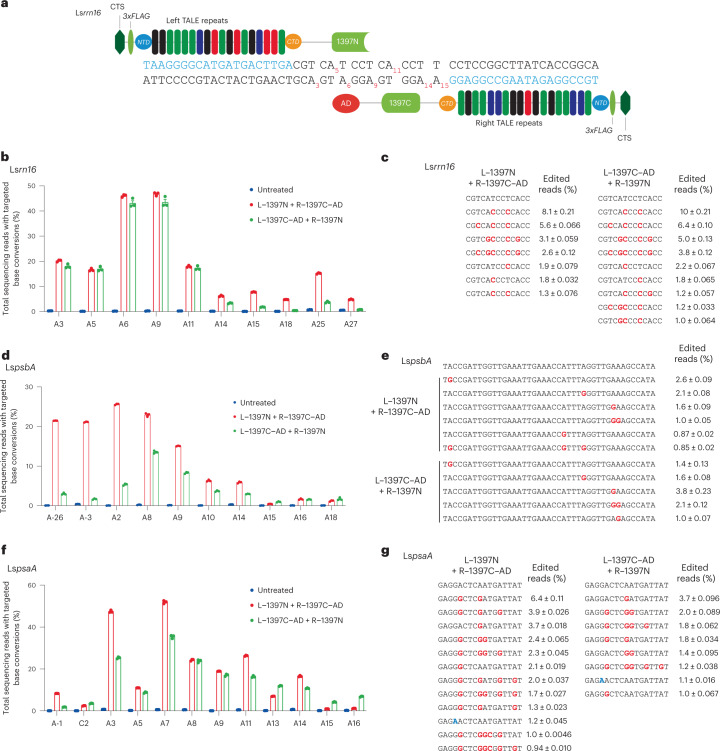
To demonstrate targeted A-to-G editing in plant organelles using transcription activator-like effector (TALE)-linked deaminases (TALEDs), which are composed of custom-designed TALE DNA-binding arrays, split DddAtox originating from Burkholderia cenocepacia and an engineered deoxyadenosine deaminase (TadA-8e) derived from the Escherichia coli TadA protein, we first chose three chloroplast genes (rrn16, psbA and psaA) in lettuce (Lactuca sativa) (Fig. 1a and Extended Data Fig. 1). Mutations in these genes give rise to resistance to antibiotics (rrn16 encoding 16S ribosomal RNA)14 or herbicide (psbA)15 and to an albino phenotype (psaA)12. We co-transfected in vitro transcripts (mRNA) encoding TALEDs with a plastid transit peptide (PTP) of the Arabidopsis RecA1 protein into lettuce protoplasts and measured base editing frequencies using targeted deep sequencing at day 7 post-transfection (Fig. 1b–g). As expected, two TALED pairs (L–1397N (left-side TALE fused to the amino-terminal half of DddAtox split at Gly1397) + R–1397C–AD (right-side TALE fused to the carboxy-terminal half of DddAtox split at Gly1397 and the TadA-8e adenine deaminase) and L–1397C–AD + R–1397N) targeted to the rrn16 site induced A-to-G conversions with editing frequencies of up to 46% without causing C-to-T conversions (Fig. 1b,c). Adenines in the spacer region between the two TALE-binding sites were edited more efficiently than those positioned outside the region. Two TALED pairs (L–1397N + R–1397C–AD and L–1397C–AD + R–1397N) targeted to the psbA site also induced A-to-G conversions with editing frequencies of up to 25%. Bystander A-to-G edits (A-26) were also induced outside of the spacer region with a frequency of 21% by the L–1397N + R–1397C–AD pair (Fig. 1d), which could have been caused by relatively poor affinity of the left-side TALE array (L) for the target DNA. At the psaA target site, two TALED pairs (L–1397N + R–1397C–AD and L–1397C–AD + R–1397N) induced A-to-G conversions in lettuce protoplasts with editing frequencies of up to 51% (Fig. 1f). Unexpectedly, C-to-T conversions (C2) were also induced with up to 3.5% efficiencies (Fig. 1f), suggesting that uracil-glycosylase-inhibitor-free split TALEDs could induce C-to-T edits in addition to A-to-G edits (albeit less efficiently) in chloroplasts, unlike in mammalian mitochondria.
Fig. 1. TALED-mediated cpDNA editing in lettuce protoplasts.
a, TALED pairs designed to target the rrn16 chloroplast gene in lettuce (Ls). The TALE-binding sites are shown in blue. CTS, chloroplasts target signal; NTD, N-terminal domain; CTD, C-terminal domain. b–g, Frequencies of base conversions and edited alleles induced by TALEDs in rrn16 (b,c), psbA (d,e) and psaA (f,g). A-to-G and C-to-T edited bases are shown in red and blue, respectively (c,e,g). The error bars show the standard error of the mean (±s.e.m.) for three biologically independent experiments.
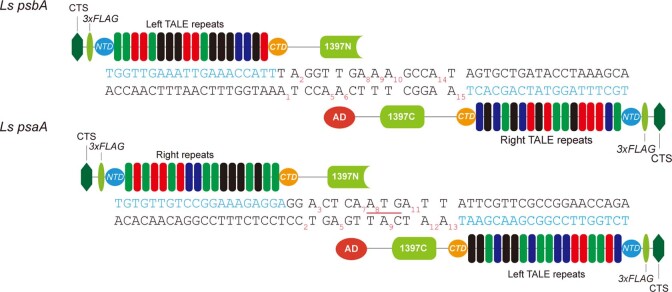
Extended Data Fig. 1. TALED pairs designed to target psbA and psaA chloroplast genes in lettuce.
The TALE-binding sites are shown in blue. CTS, Chloroplasts target signal; NTD, N-terminal domain; CTD, C-terminal domain, 1397 N and 1397 C, split DddAtox half; AD, adenine deaminase (ABE8e).
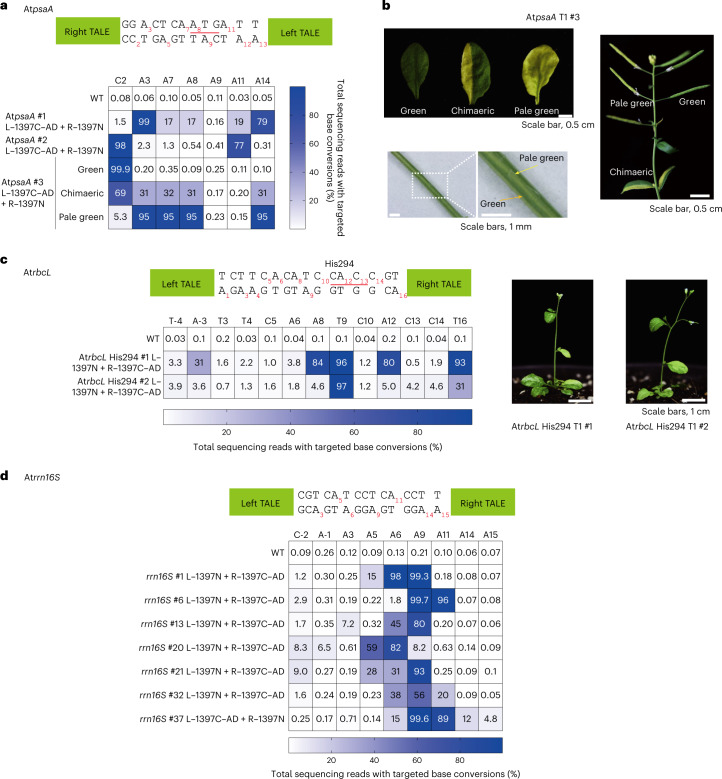
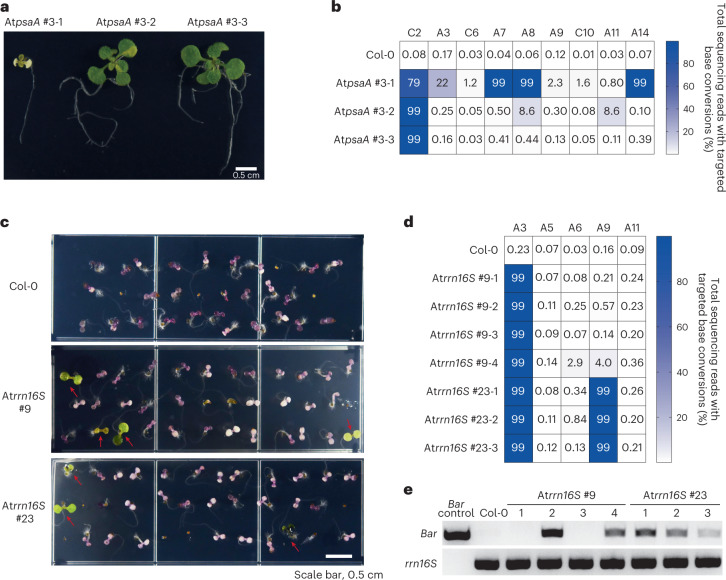
We next chose three Arabidopsis chloroplast genes (psaA, rbcL and rrn16S) to investigate whether TALED-mediated edits in chloroplast DNA (cpDNA) could be stably maintained in whole plants. We obtained T1 transformants using a transfer DNA binary vector encoding a TALED pair specific to each gene under the control of the RPS5A promoter and the 35S terminator11 (Extended Data Fig. 2). Targeted deep sequencing showed that A-to-G base editing was induced at multiple positions with frequencies of up to 99% in 16 of 20 T1 plants transformed with the psaA-targeted TALED pair (Extended Data Fig. 3a). Among the 16 edited transformants, 15 plants had mutations that disrupted the start codon (A8 and A9, underlined in Fig. 2a). As a result, seven transformants showed an albino phenotype to varying degrees. Two plantlets with almost complete albino phenotypes failed to grow (Extended Data Fig. 3b). As shown with lettuce protoplasts (Fig. 1f,g), C-to-T conversions were also observed, with editing frequencies of up to 98% (#2, C2) in these T1 plants (Extended Data Fig. 3c). Interestingly, unlike psaA T1 #1 and #2 plants showing a wild-type morphology without an albino phenotype, the psaA T1 #3 plant was mosaic, with green, chimaeric and pale-green leaves. Pale-green traits were also observed in stems and siliques (Fig. 2b). In particular, the editing frequencies of an adenine (A8) in the start codon were 0.1%, 31% and 95% in green, chimaeric and pale-green leaves, suggesting that albinism was caused by the disruption of the psaA gene via adenine base editing (Fig. 2a,b). The other five chimaeric T1 plants (#16–20) also showed A-to-G conversions at this position (A8), with frequencies that ranged from 52% to 78% (Extended Data Fig. 3a). Two T1 (#9 and #10) plantlets with the highest editing frequencies (97% and 99%) at this position died prematurely. These results demonstrate that nearly homoplasmic (~99%) adenine editing can be obtained in Arabidopsis T1 transformants using TALEDs expressed under the control of the RPS5A promoter, which is active at an early embryonic stage in meristem regions16.
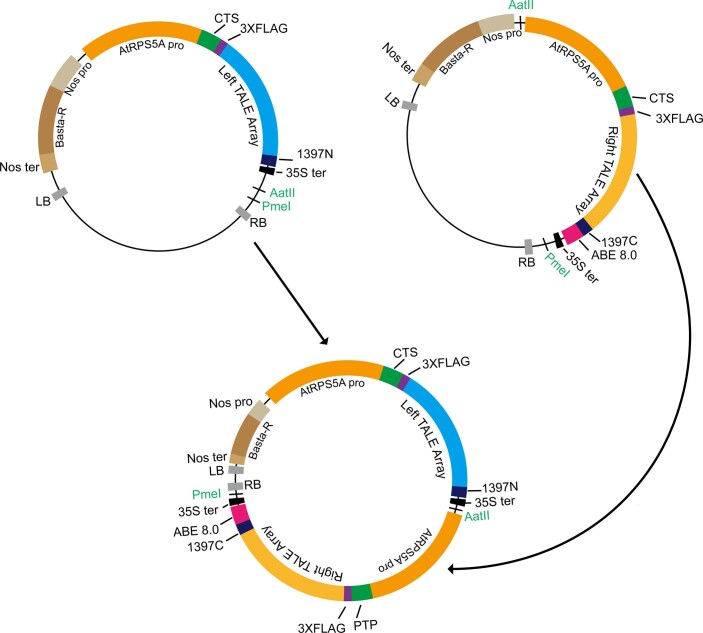
Extended Data Fig. 2. TALED of chloroplast target constructs assembly schematic.
Each TALED pairs were cloned into a different T-vector: Left TALE(L-1397N) cloned into RPS5Apro-CTS-35Ster-Aat II-Pme I T-vector and Right TALE (R-1397C-AD) cloned into Aat II-RPS5Apro- CTS-35Ster-PmeI. After digesting each vector with Aat II and Pme I, Aat II-RPS5Apro-CTS-Right TALE-1397C-AD-35Ster-PmeI was ligated to a vector with pRPS5Apro-CTS-Left TALE-1397N-35Ster-Aat II-Pme I, and the TALED pair was cloned into one vector. 1397 N and 1397C-ABE8.0 (ABE8e), which intersected in Left TALE and Right TALE, were also constructed.
Extended Data Fig. 3. Analysis of AtpsaA T1 plants.
Editing frequencies (a) and phenotypes (b) and allele frequencies (c) of psaA targeted T1 plants. The TALE binding regions are shown in green, and analyzed nucleotides are shown in numbers. The adenine of the desired target for base editing is indicated in red underline. Edited bases were represented by red (c). The base conversion rates were measured by targeted deep sequencing.
Fig. 2. TALED-mediated cpDNA editing in Arabidopsis.
a–d, Heat maps showing editing frequencies and phenotypes of Arabidopsis T1 plants expressing TALEDs targeted to psaA (a,b), rbcL (c) and rrn16S (d). The TALE-binding regions are shown in green. The start codon in psaA (a) and the codon for His294 in rbcL (c) are underlined. WT, wild type.
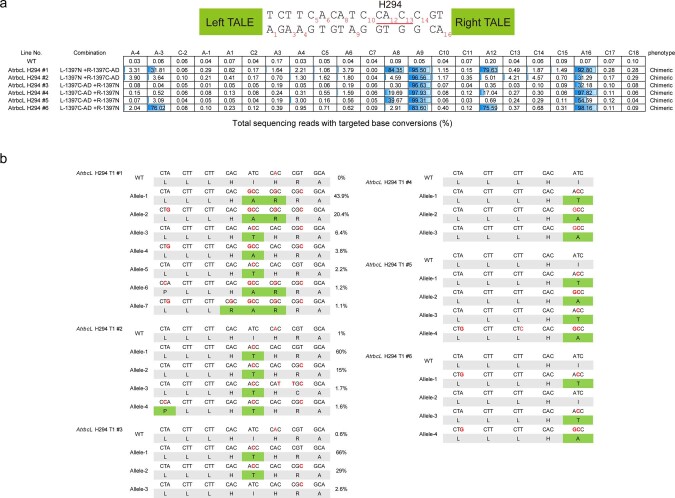
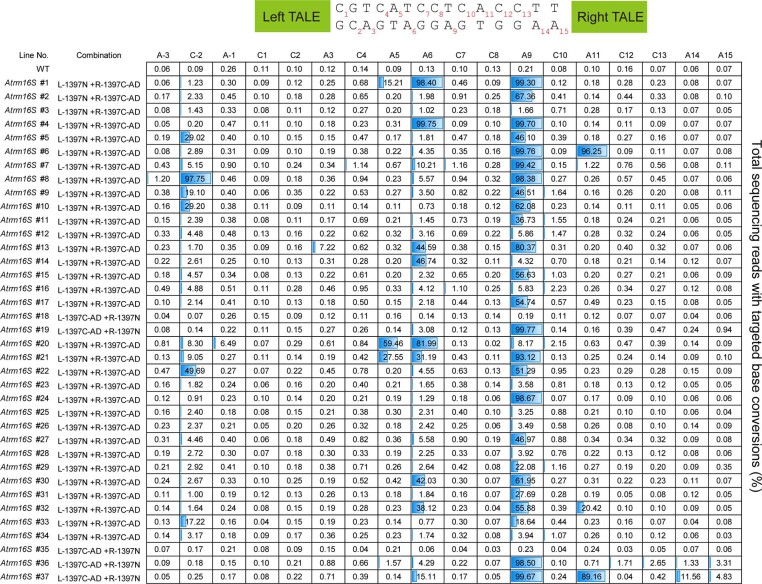
We next analysed rbcL-targeted T1 plants. The rbcL gene encodes the large, catalytic subunit of RuBisCo, the enzyme catalysing CO2 fixation in chloroplasts1. A-to-G conversions were induced at several positions in the spacer region with editing frequencies of up to 99.3% in a total of six T1 transformants with chimaeric leaves (Fig. 2c and Extended Data Fig. 4). C-to-T conversions were rarely induced at two positions in a 5′-TC-3′ context (C5 and C10) in these plants. These mutations gave rise to alterations in amino acid sequences around His294 (underlined in Fig. 2c), an amino acid residue absolutely conserved among all members of the RuBisCo superfamily. Apparently, the resulting RuBisCo proteins were poorly active, resulting in partial albinism. In addition, we obtained a total of 37 T1 plants with the rrn16S-targeted TALED. Among these, 35 plants were base edited with frequencies that ranged from 1.3% to 99.8% (Fig. 2d and Extended Data Fig. 5), demonstrating that homoplasmic mutations can be induced in whole plants using TALEDs.
Extended Data Fig. 4. Analysis of AtrbcL T1 plants.
Editing frequencies (a) and allele frequencies (b) of rbcL targeted T1 plants. The TALE binding regions are shown in green, and analyzed nucleotides are shown in numbers. The adenine of the desired target for base editing is indicated in red underline. Edited bases were represented by red (b). The base conversion rates were measured by targeted deep sequencing.
Extended Data Fig. 5. Analysis of Atrrn16S T1 plants.
Editing frequencies of rrn16S targeted T1 plants. The TALE binding regions are shown in green, and analyzed nucleotides are shown in numbers. The base conversion rates were measured by targeted deep sequencing.
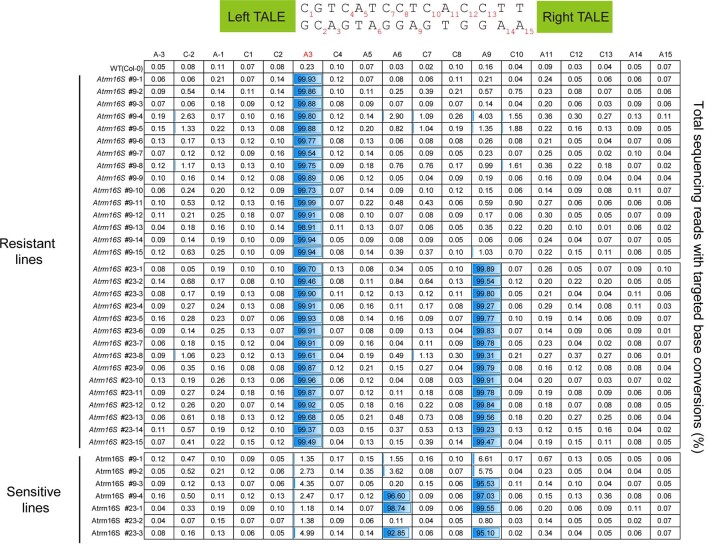
Next, we investigated whether TALED-induced edits in cpDNA were inherited in the next generation. We harvested T2 seeds from the #3 psaA-edited T1 plant and grew T2 progenies on half-strength Murashige and Skoog (MS) media under long-day (16 h of light and 8 h of dark) conditions for 14 days to confirm phenotypic alterations. Interestingly, T2 plants showed diverse phenotypes and genotypes (Fig. 3a,b). Targeted deep sequencing showed that A8 in the start codon was almost completely (99%), partially (8.6%) and poorly (0.44%) edited in albino (#3-1), chimaeric (#3-2) and wild-type-like (#3-3) plants, respectively (Fig. 3b), suggesting that the disruption of the initiation codon gave rise to albinism. Furthermore, we were able to obtain spectinomycin-resistant plants after T2 seeds of rrn16S-edited T1 lines were sown in half-strength MS medium containing spectinomycin, an antibiotic that inhibits protein synthesis (Fig. 3c). Interestingly, A3, which had been minimally edited in T1 plants with an average editing frequency of 0.47 ± 0.19%, was almost completely (99%) converted to guanine in several T2 plants resistant to spectinomycin (Fig. 3d). No other mutations, in addition to A3-to-G conversions, were induced at >1.0% frequencies in a total of 13 spectinomycin-resistant, rrn16S-edited #9 T2 lines (Extended Data Fig. 6), suggesting that A3 editing was responsible for the drug resistance. We also found that two spectinomycin-resistant T2 plants (#9-1 and 3) were transgene-free (Fig. 3e), indicating that the A3-to-G edit was induced in T1 plants and that the insertion of multiple copies of transgene is not required to induce homoplasmic editing. Note also that transgene-free, cpDNA-edited plants can be exempt from genetically modified organism regulations17,18. Taken together, these results demonstrate that base edits induced by TALEDs in cpDNA are transmitted to the next generation.
Fig. 3. Phenotypes and genotypes of Arabidopsis T2 plants.
a, Two-week-old T2 seedlings from the #3 psaA-edited T1 plant grown on half-strength MS medium containing 1% sucrose under long-day conditions. b, Base-editing frequencies of T2 progenies. c, One-week-old Col-0 and T2 seedlings from Atrrn16S-edited T1 plants (#9 and #23) grown on half-strength MS medium containing 1% sucrose and spectinomycin (10 mg l−1) under long-day conditions. The red arrows indicate spectinomycin-resistant seedlings. d, Base-editing frequencies of spectinomycin-resistant Atrrn16S-edited T2 progenies. e, PCR amplification of the Bar transgene (552 base pairs) in genomic DNA isolated from spectinomycin-resistant T2 seedlings. rrn16S (235 base pairs) was used as an internal control.
Extended Data Fig. 6. Analysis of Atrrn16S T2 plants.
Editing frequency of rrn16S-targeted T2 plants with spectinomycin (10 mg l-1) resistance and sensitivity. The TALE binding regions are shown in green, and analyzed nucleotides are shown in numbers. The base conversion rates were measured by targeted deep sequencing.
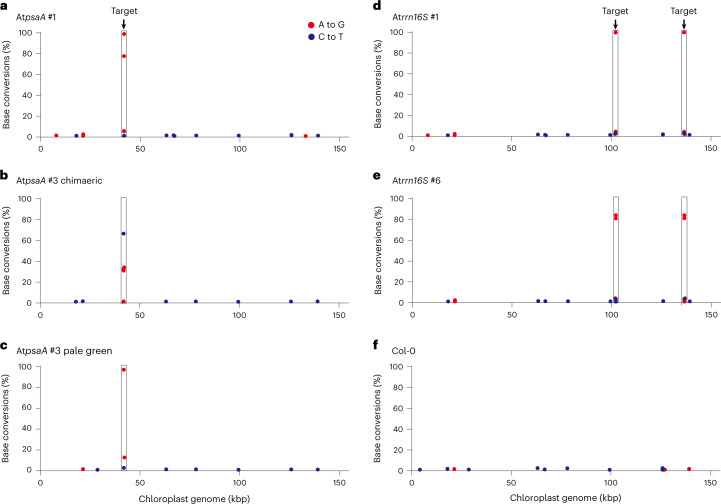
Last, we profiled the off-target activity of the TALEDs targeted to the psaA and rrn16S sites in T1 plants using whole-genome sequencing (Fig. 4). We were able to confirm on-target edits in each sample and to identify several A-to-G or C-to-T mutations with low conversion frequencies that ranged from 1.0% to 4.4% in the chloroplast genome. Most of these single-nucleotide conversions, relative to the reference chloroplast genome, were also observed in the negative control (Col-0), suggesting that they represent naturally occurring heteroplasmy rather than TALED-induced off-target edits. Note also that a few of the single-nucleotide variations with low heteroplasmic fractions found in Col-0 were not detected in T1 plants or vice versa. These results show that TALEDs induce on-target edits specifically without substantial unlinked off-target mutations in cpDNA.
Fig. 4. Analysis of TALED-induced off-target effects in the Arabidopsis plastid genome.
a–f, Chloroplast-genome-wide plots showing A-to-G or C-to-T base conversions relative to the reference genome (AP000423.1). We analysed psaA-edited (a–c) and rrn16S-edited (d,e) T1 plants in addition to Col-0 (f). Note that there are two identical target sites for the rrn16S-specific TALED. kbp, kilobases.
The TALED-mediated organellar base editing described in this study has a number of advantages over plastid transformation via homologous recombination using a targeting vector. First, organellar base editing is more broadly applicable, not limited to certain plant species amenable for plastid transformation. Second, homoplasmic editing can be achieved in a single round of Agrobacterium-mediated transformation, as shown in this study. In contrast, multiple rounds of marker selection and regeneration are required to obtain homoplasmy using plastid transformation. Third, no transgenes are integrated in plastid DNA with organellar base editing. In contrast, plastid transformation involves the targeted integration of a selectable marker gene into plastid DNA, which cannot be removed by breeding.
In summary, we developed plant-optimized TALEDs for A-to-G base editing in cpDNA in protoplasts and whole plants. In particular, targeted adenine editing in chloroplast genes using Agrobacterium-mediated transformation gave rise to heritable homoplasmic mutations, leading to phenotypic changes in Arabidopsis. Unlike in human mitochondrial DNA, uracil-glycosylase-inhibitor-free TALEDs catalysed C-to-T (in addition to A-to-G) conversions at some but not all positions in the context of a 5′-TC-3′ motif in cpDNA. It will be important to investigate whether TALEDs yield different editing outcomes in mammalian mitochondria and in plant chloroplasts, possibly because of the differences in mismatch DNA repair systems, and to develop TALEDs catalysing A-to-G conversions exclusively (without C-to-T conversions) in plants. TALEDs, together with DddA-derived cytosine base editors, can now induce targeted A-to-G and C-to-T base editing in plant organellar DNA, which could pave the way for enhancing the efficiency of photosynthesis and CO2 fixation in plants, contributing to agricultural innovations and carbon neutralization.
Methods
Plasmid construction
TALE arrays were designed to target rrn16S, psaA, psbA and rbcL following the approach used in previous reports4,5,10,13,19. PCR amplicons encoding the TALE array, DddAtox split and ABE8e, generated using PrimeSTAR GXL DNA Polymerase (TAKARA), were cloned into pRPS5A–CTS digested with SmaI and KpnI (NEB) using Gibson assembly (NEB). Specifically, the left-side and right-side TALED sequences were cloned into pRPS5A–CTS–35S terminator–AatII–PmeI vector and AatII–pRPS5A–CTS–35S terminator–PmeI, respectively. The left-side and right-side TALED constructs were transcribed in vitro, and the resulting mRNAs were used for lettuce protoplast transfection. For Arabidopsis transformation, the left and right TALED vectors were digested with AatII and PmeI (NEB) and ligated using T4 DNA ligase (NEB) (Extended Data Fig. 2). The assembled plasmids were chemically transformed into E. coli DH5ɑ, and plasmids from the surviving colonies were analysed by the Sanger sequencing method. The TALE binding sequences are listed in Supplementary Table 1. The primers for PCR amplification are listed in Supplementary Table 2. The plasmids used in this study and their annotated DNA sequences are available from Addgene (ID 189639–189652).
Lettuce protoplast isolation and transfection
Lettuce (Lactuca sativa cv. Cheongchima) seeds were sterilized by immersing them in 70% ethanol for 30 s and then in a 0.4% hypochlorite solution for 15 min prior to washing them three times in distilled water. The sterilized lettuce seeds were germinated on half-strength MS medium supplemented with 2% sucrose at 25 °C under 16 h of light and 8 h of dark. Protoplast isolation and transfection were performed as described in a previous report10.
mRNA in vitro transcription
For use as in vitro transcription templates, DNA templates were amplified using PrimeSTAR GXL DNA Polymerase (TAKARA). The mRNAs were synthesized and purified using an in vitro mRNA synthesis kit (Enzynomics). We used 40 μg of each type of transcript for protoplast transfection. The primers for DNA template PCR amplification are listed in Supplementary Table 2.
Plant transformation and transformant selection
A. thaliana Columbia-0 (Col-0) plants were transformed by floral dipping with Agrobacterium tumefaciens strain GV3101 as described in a previous report20. After transformation, Arabidopsis T1 seeds were plated on half-strength MS medium containing 1% sucrose, 20 mg l−1 phosphinothricin and 250 mg l−1 cefataxim. All transgenic plants were grown at 23 °C under long-day conditions (16 h of light and 8 h of dark).
Targeted deep sequencing
Total DNA was isolated from cultured cells at day 7 post-transfection10 and true leaves from selected plants using a DNeasy Plant Mini kit (Qiagen). On-target sites were amplified through a primary PCR, a secondary PCR and a third PCR to generate deep sequencing libraries using TruSeq HT Dual index-containing primers and PrimeSTAR GXL DNA Polymerase (TAKARA). Illumina MiniSeq paired-end sequencing systems were used to sequence the libraries. The base editing frequencies are presented as percentages of sequencing reads containing base conversions among total sequencing reads. The program used to analyse the frequency of edits is available at https://github.com/ibs-cge/maund. The PCR primer sequences are shown in Supplementary Tables 3 and 4.
Genotyping of T2 plants
Total DNAs were isolated from true leaves from T2 plants using a DNeasy Plant Mini kit (Qiagen). The regions of interest were amplified from total DNAs using PrimeSTAR GXL DNA Polymerase (TAKARA), after which the PCR amplicons were analysed on a 1% agarose gel. The PCR primer sequences are shown in Supplementary Tables 2 and 4.
Next-generation sequencing
For analysis of off-target effects, single nucleotide polymorphisms were called in the plastid genomes using sequencing data from total DNA. First, paired-end libraries were prepared from total DNA using a TruSeq DNA PCR-Free Kit (Illumina) for Atpsa #1 and Atrrn16S #1 and #6 and a TruSeq Nano DNA Kit (Illumina) for Atpsa #3 (chimaeric and pale-green leaf samples) and Col-0. Sequencing was performed with an Illumina HiSeq X Ten platform. To analyse the next-generation sequencing data from whole-chloroplast-genome sequencing, we followed the methods in a previously published report11. Paired-end reads were mapped to reference sequences (AP000423.1) using BWA (v.0.7.17) (ref. 21) in single-ended mode. We filtered out mapped reads with mapping identities ≤99%. Single nucleotide polymorphisms were then called using pysam (v.0.18.0) (ref. 22). Finally, we listed the positions of variants with A-to-G and C-to-T conversion rates ≥1% with read depths ≥5,000.
Reporting summary
Further information on research design is available in the Nature Portfilio Reporting Summary linked to this article.
Supplementary information
Supplementary Tables 1–4 and Sequences 1 and 2.
Acknowledgements
This work was supported by the Institute for Basic Science (grant no. IBS-R021-D1-a00 to J.-S.K.).
Extended data
Source data
Targeted deep sequencing reads.
Targeted deep sequencing reads.
Targeted deep sequencing reads and unprocessed gel image.
Next-generation sequencing depths.
Targeted deep sequencing reads.
Targeted deep sequencing reads data.
Targeted deep sequencing reads data.
Targeted deep sequencing reads data.
Author contributions
Y.G.M., S.H. and J.-S.K. designed the study. Y.G.M., S.H., S.-J.B. and S.-I.C. performed the experiments. Y.G.M., S.H. and J.-S.K. wrote the manuscript. J.-S.K. supervised the research.
Peer review
Peer review information
Nature Plants thanks Yiping Qi and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Data availability
The DNA sequencing data have been deposited in the National Center for Biotechnology Information (NCBI) Sequence Read Archive (SRA) database with BioProject accession code PRJNA858174. The protein sequences of the TALE arrays and TadA-8e are provided in the Supplementary Information. Any other additional data are available in the Supplementary Information. Source data are provided with this paper.
Code availability
Source code (https://github.com/ibs-cge/maund, created by BotBot Inc.) was used to calculate base editing frequencies from targeted deep sequencing data.
Competing interests
A provisional patent application was submitted based on the results reported in this paper. J.-S.K. is a founder of and shareholder in ToolGen.
Footnotes
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
These authors contributed equally: Young Geun Mok, Sunghyun Hong.
Extended data
is available for this paper at 10.1038/s41477-022-01279-8.
Supplementary information
The online version contains supplementary material available at 10.1038/s41477-022-01279-8.
References
- 1.Dobrogojski J, Adamiec M, Lucinski R. The chloroplast genome: a review. Acta Physiol. Plant. 2020;42:98. doi: 10.1007/s11738-020-03089-x. [DOI] [Google Scholar]
- 2.Gammage PA, Moraes CT, Minczuk M. Mitochondrial genome engineering: the revolution may not be CRISPR-ized. Trends Genet. 2018;34:101–110. doi: 10.1016/j.tig.2017.11.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Mok BY, et al. A bacterial cytidine deaminase toxin enables CRISPR-free mitochondrial base editing. Nature. 2020;583:631–637. doi: 10.1038/s41586-020-2477-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Mok YG, et al. Base editing in human cells with monomeric DddA–TALE fusion deaminases. Nat. Commun. 2022;13:4038. doi: 10.1038/s41467-022-31745-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Lee H, et al. Mitochondrial DNA editing in mice with DddA–TALE fusion deaminases. Nat. Commun. 2021;12:1190. doi: 10.1038/s41467-021-21464-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Guo J, et al. DdCBE mediates efficient and inheritable modifications in mouse mitochondrial genome. Mol. Ther. Nucleic Acids. 2021;27:73–80. doi: 10.1016/j.omtn.2021.11.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Silva-Pinheiro P, et al. In vivo mitochondrial base editing via adeno-associated viral delivery to mouse post-mitotic tissue. Nat. Commun. 2022;13:750. doi: 10.1038/s41467-022-28358-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Mok BY, et al. CRISPR-free base editors with enhanced activity and expanded targeting scope in mitochondrial and nuclear DNA. Nat. Biotechnol. 2022;40:1378–1387. doi: 10.1038/s41587-022-01256-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Lim K, et al. Nuclear and mitochondrial DNA editing in human cells with zinc finger deaminases. Nat. Commun. 2022;13:366. doi: 10.1038/s41467-022-27962-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kang B-C, et al. Chloroplast and mitochondrial DNA editing in plants. Nat. Plants. 2021;7:899–905. doi: 10.1038/s41477-021-00943-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Nakazato I, et al. Targeted base editing in the plastid genome of Arabidopsis thaliana. Nat. Plants. 2021;7:906–913. doi: 10.1038/s41477-021-00954-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Li R, et al. High-efficiency plastome base editing in rice with TAL cytosine deaminase. Mol. Plant. 2021;14:1412–1414. doi: 10.1016/j.molp.2021.07.007. [DOI] [PubMed] [Google Scholar]
- 13.Cho S-I, et al. Targeted A-to-G base editing in human mitochondrial DNA with programmable deaminases. Cell. 2022;185:1764–1776. doi: 10.1016/j.cell.2022.03.039. [DOI] [PubMed] [Google Scholar]
- 14.Miyazaki K, Kitahara K. Functional metagenomic approach to identify overlooked antibiotic resistance mutations in bacterial rRNA. Sci. Rep. 2018;8:5179. doi: 10.1038/s41598-018-23474-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Narusaka Y, Narusaka M, Kobayashi H, Satoh K. The herbicide-resistant species of the cyanobacterial Dl protein obtained by thorough and random in vitro mutagenesis. Plant Cell Physiol. 1998;39:620–626. doi: 10.1093/oxfordjournals.pcp.a029413. [DOI] [PubMed] [Google Scholar]
- 16.Weijers D, et al. An Arabidopsis Minute-like phenotype caused by a semi-dominant mutation in a RIBOSOMAL PROTEIN S5 gene. Development. 2001;128:4289–4299. doi: 10.1242/dev.128.21.4289. [DOI] [PubMed] [Google Scholar]
- 17.Kim J, Kim J–S. Bypassing GMO regulation with CRISPR gene editing. Nat. Biotechnol. 2016;34:1014–1015. doi: 10.1038/nbt.3680. [DOI] [PubMed] [Google Scholar]
- 18.Ishii T. Crop gene-editing: should we bypass or apply existing GMO policy? Trends Plant Sci. 2018;23:947–950. doi: 10.1016/j.tplants.2018.09.001. [DOI] [PubMed] [Google Scholar]
- 19.Kim Y, et al. A library of TAL effector nucleases spanning the human genome. Nat. Biotechnol. 2013;31:251–258. doi: 10.1038/nbt.2517. [DOI] [PubMed] [Google Scholar]
- 20.Zhang X, Henriques R, Lin SS, Niu QW, Chua NH. Agrobacterium-mediated transformation of Arabidopsis thaliana using the floral dip method. Nat. Protoc. 2006;1:641–646. doi: 10.1038/nprot.2006.97. [DOI] [PubMed] [Google Scholar]
- 21.Li H, Durbin R. Bioinformatics. 2009;25:1754–1760. doi: 10.1093/bioinformatics/btp324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Gilman P. et al. PySAM (Python Wrapper for System Advisor Model “SAM”) 10.11578/dc.20190903.1 (2019).
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary Tables 1–4 and Sequences 1 and 2.
Data Availability Statement
The DNA sequencing data have been deposited in the National Center for Biotechnology Information (NCBI) Sequence Read Archive (SRA) database with BioProject accession code PRJNA858174. The protein sequences of the TALE arrays and TadA-8e are provided in the Supplementary Information. Any other additional data are available in the Supplementary Information. Source data are provided with this paper.
Source code (https://github.com/ibs-cge/maund, created by BotBot Inc.) was used to calculate base editing frequencies from targeted deep sequencing data.