Abstract
A large subunit of calpain, a calcium-activated neutral proteinase, from Schistosoma japonicum was cloned and expressed in Escherichia coli. When BALB/c mice were immunized with purified recombinant calpain (r-calpain) emulsified in complete Freund's adjuvant, a significant reduction in the number of recovered worms and also in egg production per female worm was observed (P < 0.01). Spleen cells of the immunized mice showed enhanced production of gamma interferon (IFN-γ) by activated CD4+ T cells. Considering our observation of elevated expression of inducible nitric oxide synthase mRNA in immunized mice, r-calpain-induced IFN-γ seemed to upregulate the production of nitric oxide by macrophages and subsequently mediated the killing of schistosomulae in the lung. On the other hand, spleen cells of immunized mice showed only faint interleukin-4 production in response to r-calpain in vitro, suggesting that immunization with r-calpain alters the Th1-Th2 balance in murine hosts even during a Th2-promoting S. japonicum infection. Furthermore, histopathological study of the livers of immunized mice showed that granulomas formed around eggs were diminished in both size and number. Egg production by female worms was clearly decreased in immunized mice, suggesting that r-calpain also has antifecundity effects. Taken together, these results point to S. japonicum calpain as a potential vaccine candidate for both worm killing and disease prevention, possibly through the induction of a strong Th1-dominant environment in immunized mice.
More than 200 million people have schistosomiasis, and almost 600 million people are exposed to the risk of schistosomiasis (36). Chemotherapy is currently the choice for control; however, vaccine development remains an important long-term goal for the integrated control of schistosomiasis because of high reinfection rates in areas where the disease is endemic. Extensive work has been carried out to identify schistosome molecules that confer partial but significant protection in different animal models. These include the Schistosoma mansoni 28-kDa and the S. japonicum 26-kDa glutathione S-transferase (GST) (5, 32), the S. mansoni and S. japonicum 97-kDa paramyosin (13, 25), the S. mansoni 28-kDa triose phosphate isomerase (29), the S. mansoni 23-kDa integral membrane antigen (24), and so forth. These vaccine candidates were selected by the World Health Organization for a series of independent trials to test their protective efficacy in laboratory animals (2). Unfortunately, the stated goal of consistent induction of 40% or better protection was not reached with any of these antigen formulations in trials with large domestic animals (35). Since S. japonicum infection is zoonotic, several vaccine candidates, such as the S. japonicum 26-kDa GST or 97-kDa paramyosin, have been tested in domestic animals. Significant and promising results were obtained in some trials; however, detailed analyses are still under way. Most of the vaccine candidates were first identified in S. mansoni, but controversy remains about whether equivalent antigens of S. japonicum could have comparable effects because there are qualitative and/or quantitative differences between the host immune responses to the two parasitic infections (25).
S. japonicum is a major schistosome species in Asia, infecting not only humans but also wild or domestic animals. Despite the availability of very successful control programs, schistosomiasis japonica remains a serious public health problem in China and the Philippines. Several types of economically important livestock, such as water buffaloes and domestic pigs; act as reservoir hosts of S. japonicum, and feces of livestock containing S. japonicum eggs are of prime importance for continued transmission of this parasite to humans. Control of schistosomiasis japonica depends substantially on the successful reduction of its prevalence in domestic livestock. Identification of an effective vaccine is an emergent task for reducing the transmission of S. japonicum from animals to humans in this region. However, relatively limited numbers of antigens from S. japonicum were identified as vaccine candidates, in comparison with S. mansoni infection (3).
Calpain from S. mansoni was shown to induce protective immunity during murine experimental schistosomiasis mansoni (11), and molecular cloning of calpain from S. japonicum has since started in several laboratories, including our own (28, 38). Although calpain is believed to be an intracellular protease, the location of this molecule seems not to be fixed and in some cases it is moved outside of the cell membrane (26). This suggests that calpain could have enough immunogenicity for both humoral and cellular responses. A previous experiment performed in our laboratory indicated that human sera from S. japonicum-infected individuals recognized r-calpain, and sera obtained from mice immunized with r-calpain showed enhanced binding to cercarial antigens (38). Together with these findings, the principal objective of our present study was to develop a livestock vaccine that can be used to prevent Asian schistosomiasis. We present here a study of the efficacy of r-calpain as a vaccine molecule against a challenge infection with S. japonicum in BALB/c mice and discuss the possible underlying mechanism of protective immunity in immunized host animals.
MATERIALS AND METHODS
Host animals and parasites.
The life cycle of S. japonicum isolated in Yamanashi Prefecture, Japan, has been maintained in our laboratory by using Onchomelania hupensis nosophora with the same geographical distribution. Six-week-old female BALB/c mice (SLC, Hamamatsu, Japan) were used for immunization and infection experiments.
Recombinant calpain (r-calpain) from S. japonicum.
A recombinant molecule of the large subunit of calpain from S. japonicum was prepared as described previously (38). In brief, cDNA encoding amino acid residues 219 to 376 of S. japonicum calpain was amplified by reverse transcription (RT)-PCR because a comparable portion was shown to be highly immunogenic in murine schistosomiasis mansoni (17). The product was digested by BamHI and EcoRI and then ligated into the GST fusion vector pGEX-2TK (Pharmacia, Uppsala, Sweden). This vector was transfected into Escherichia coli DH5α cells (Pharmacia). GST fusion protein was induced in DH5α cells, and thrombin (Pharmacia) was used to isolate the r-calpain molecule from glutathione Sepharose 4B columns (Pharmacia).
Western blot assays.
Western blotting was carried out as described elsewhere (20). Five to 10 μg of r-calpain was separated by sodium dodecyl sulfate–14% polyacrylamide gel electrophoresis and transferred to a polyvinylidene difluoride membrane (Millipore Corporation, Bedford, Mass.). Mouse anti-r-calpain serum was used as the primary antibody, and the secondary antibody used was goat anti-mouse IgG labeled with peroxidase (Kirkegaard & Perry Laboratories, Gaithersburg, Md.) at a final dilution of 1:3,000. The substrate used was 4-chloro-1-naphthol.
Immunization schedule.
Mice were divided into two groups in the first experiment and three groups in the second experiment. An immune-challenge group of 18 mice was injected subcutaneously (s.c) with 25 μg of r-calpain dissolved in phosphate-buffered saline (PBS) with complete Freund's adjuvant (Gibco, Grand Island, N.Y.). The mice were boosted s.c. with 25 μg of r-calpain dissolved in PBS with incomplete Freund's adjuvant (Gibco) 2 weeks later and were further boosted intravenously 2 weeks later with 25 μg of r-calpain dissolved in PBS. An adjuvant-treated control group comprising 18 mice was subjected to the same immunization schedule as the immune-challenge group, but PBS replaced r-calpain. Ten mice with no treatment were used as a challenge control group. In our second experiment, we prepared an additional group of 12 mice immunized s.c. with 100 irradiated cercariae (48 × 103 rads), and this group was boosted twice every 2 weeks. Two weeks after the final boosting, all groups were infected percutaneously with 30 cercariae of S. japonicum by the cover glass method.
RT-PCR for iNos, eNos, and Nos mRNAs.
Four days after the challenge infection, whole lungs of four mice were removed from r-calpain-immunized or adjuvant-treated control group mice and immediately frozen in liquid nitrogen in order to detect expression of inducible, neuronal, and endothelial nitric oxide synthase (iNos, nNos, and eNos, respectively) mRNAs. Total RNA from the lungs of the mice was isolated using PUREscript reagents (Gibco) in accordance with the manufacturer's instructions. RNA samples were stored at −80°C until use. First-strand DNA synthesis was carried out with 1 μg of RNA in 20-μl of reaction solution using an RNA-PCR kit (Perkin-Elmer, Branchburg, Calif.). The primer sequence and PCR conditions used for amplification of nNos and eNos were reported previously (10). β-Actin was used as a control. The iNos primers used were as follows: sense 5′ ctggaggagctcctgcctcatg 3′; antisense; 5′ gcagcatcccctctgatggtg 3′. These primers amplify a 449-bp fragment of iNos mRNA. The PCR solution contained 2 μl of cDNA, 50 mM KCl (pH 8.4), 20 mM Tris-HCl, 1.5 mM MgCl2, 200 mM deoxynucleoside triphosphates, 2 U of Taq gold DNA polymerase (all from Perkin-Elmer); and 1.2 mM primers in a 50-μl volume. The PCR mixture was subjected to 35 cycles, and the annealing temperature was 60°C. PCR products were evaluated on a 2% agarose gel with ethidium bromide by using a densitometry analyzer.
Worm recovery and tissue sampling.
Six weeks after a challenge infection, all vaccinated and control mice were sacrificed and perfusion was undertaken to recover the worms. The worm reduction rate, percent protection, was calculated according to the following formula: (1 − mean number of worms in immunized mice/mean number of worms in adjuvant or challenge control mice) × 100. Before the perfusion, spleens from each group were removed to test cytokine production in vitro. At the time of perfusion, the livers and intestines of mice from all groups were weighed. The livers and intestines were digested with 4% KOH overnight, and the number of eggs was determined by microscopic examination. We determined egg counts for each gram of liver or intestine, and the mean number of eggs per female adult schistosome was also calculated.
Cytokine production analysis.
Two weeks after the final boosting, spleen cells from each group of mice were cultured in RPMI 1640 medium supplemented with 10% fetal calf serum in the presence of concanavalin A (ConA, Sigma) or r-calpain at 10 μg/ml at 37°C with 5% CO2 (prechallenge). The same testing was done at 6 weeks after the challenge infection (postchallenge). For testing of interleukin-4 (IL-4) and gamma interferon (IFN-γ) production, 107 cells were cultured for 48 h in 2 ml of medium in 24-well plates (Nunc). The cell-free culture supernatants were used for measurement of the two cytokines by quantitative sandwich enzyme immunoassay kits (Genzyme, Minneapolis, Minn.).
Histopathological examination.
After portal perfusion, the liver was dissected and immediately fixed in 10% buffered formalin for morphometric analysis. Liver sections were embedded in paraffin and stained with hematoxylin and eosin for microscopic examination. Hematoxylin-eosin-stained sections of the liver were then searched for granulomas. We assessed the sizes of nonconfluent granulomas formed around a single egg containing a mature miracidium by using a video micrometer (VM-30; Olympus, Tokyo, Japan) in accordance with the manufacturer's instructions. We also evaluated the mean percentage of granulomatous areas in 1-mm2 liver sections.
Statistical analysis.
For statistical evaluation of data, we used a two-sided Student t test.
RESULTS
Preparation of r-calpain and production of anti-r-calpain antibody in mice.
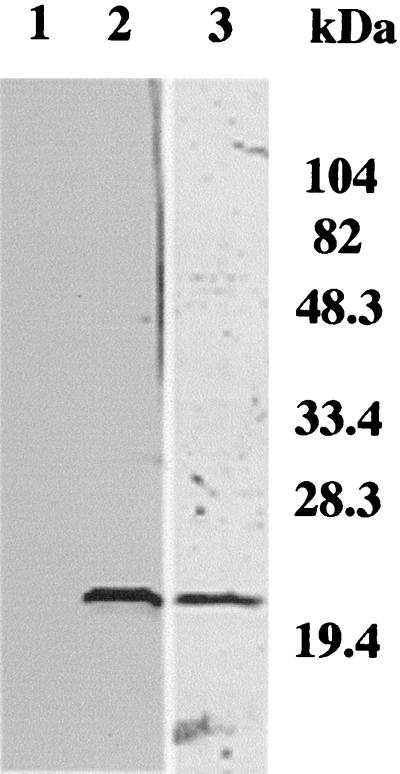
We prepared an r-calpain molecule, and the purified protein was confirmed by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (Fig. 1). Mice immunized with r-calpain produced a high level of IgG antibodies specific to the immunizing antigen. Six weeks after the immunization and prior to a challenge infection, optical density values (mean ± standard deviation) of anti-r-calpain antibody in the immunized mice and adjuvant controls were 1.037 ± 0.006 and 0.163 ± 0.009, respectively. The mean optical density value anti-r-calpain antibody in immunized mice was significantly higher than that in adjuvant controls (P < 0.01). Immunoblotting analysis showed murine anti-r-calpain serum binding specifically to r-calpain (Fig. 1), indicating that the protocol for immunization of mice with r-calpain functioned properly in our present study.
FIG. 1.
Immunoblot analysis of r-calpain of S. japonicum. R-calpain was fractionated and transferred to a polyvinylidene difluoride membrane (lane 3) and then subjected to immunostaining (lanes 1 and 2). Lane 3 was stained with Coomassie brilliant blue. IgG antibody specific to r-calpain is detected on immunoblotting membrane using sera of immunized mice (lane 2). Adjuvant-treated control mouse serum (negative control) is shown in lane 1.
Protective immunity induced by immunization with r-calpain.
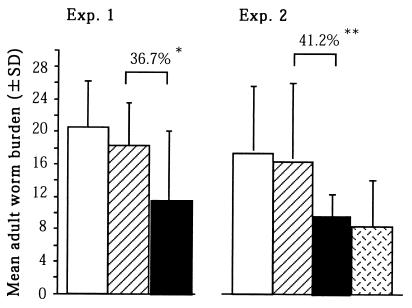
Figure 2 shows the number of worms recovered mice with or without immunization. In the repeated experiments, the worm reduction rate in immunized mice was 36.7% for experiment 1 and 41.2% for experiment 2 in comparisons with that in adjuvant-treated controls (P < 0.05). In experiment 2, 48.3% protection was provided by immunization with radiation-attenuated cercariae compared with adjuvant-treated controls. There was no significant difference in worm recovery between immunization with r-calpain and immunization with attenuated cercariae. We observed r-calpain-mediated effects on egg counts in the liver and intestine and total lymphocyte counts in the spleen (Table 1). There was a substantial reduction in the total number of eggs present in the liver and intestine in mice immunized with r-calpain or with irradiated cercariae in comparison with adjuvant-treated controls (P < 0.01). Furthermore, mice immunized with r-calpain showed a significant reduction in egg laying per female worm while there was no difference in this parameter in the case of immunization with radiation-attenuated cercariae (P < 0.01). As for the mean lymphocyte number in the spleen, mice immunized with r-calpain showed a decreased lymphocyte count compared with that in adjuvant-treated controls. There was no difference in liver and intestine weights among any of the groups (data not shown).
FIG. 2.
Adult-worm burden were assessed in two different experiments (Exp. 1 and Exp. 2). Columns: open; challenge control mice; hatched, adjuvant-treated control mice; solid, calpain-immunized mice; dotted, mice immunized with irradiated cercariae. A significant worm burden difference was found between calpain-immunized mice and adjuvant-treated controls (36.7% reduction in experiment 1 and 41.2% reduction in experiment 2). ∗, P < 0.05; ∗∗, P < 0.01. SD, standard deviation.
TABLE 1.
Results of vaccination of BALB/c mice against S. japonicum using r-calpain
| Parameter | Expt 1
|
Expt 2
|
|||||
|---|---|---|---|---|---|---|---|
| Challenged controls (n = 9) | Adjuvant-treated controls (n = 9) | Calpain-immunized group (n = 9) | Challenged controls (n = 8) | Adjuvant-treated controls (n = 8) | Calpain-immunized group (n = 8) | Group immunized with irradiated cercariae (n = 6) | |
| No. of eggs/g of liver | 36,204 ± 2,109 | 30,366 ± 9,729 | 17,486 ± 5,016a | 33,653 ± 3,971 | 34,150 ± 14,719 | 13,946 ± 6,507a | 18,423 ± 8,617 |
| No. of eggs/g of intestine | NDb | ND | ND | 43,646 ± 833 | 47,478 ± 3,056 | 15,743 ± 757a | 24,516 ± 2,071 |
| No. of eggs/female worm | 6,057 ± 812d | 5,829 ± 1,177d | 3,762 ± 2,484ad | 24,807 ± 2,034 | 23,434 ± 7,418 | 13,481 ± 2,337a | 22,059 ± 2,724c |
| Total lymphocyte count in spleen | ND | ND | ND | 1.996 × 108 | 1.875 × 108 | 1.359 × 108a | 1.33 × 108 |
Significant difference between calpain-immunized group and adjuvant-treated control group.
ND, not done.
Significant difference between calpain-immunized group and group immunized with irradiated cercariae.
Only in liver.
mRNA expression of each NOS isoform.
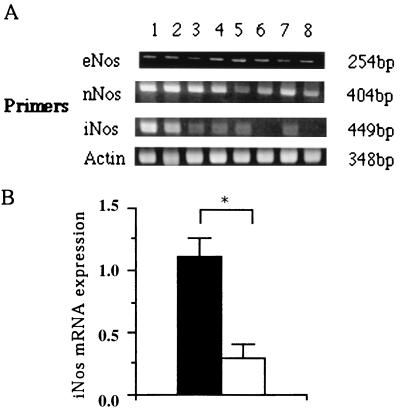
We compared expression of mRNA for three NOS isoforms in lungs removed from either r-calpain-immunized mice or adjuvant-treated control mice (Fig. 3). We observed significant increases in iNos mRNA expression in r-calpain-immunized mice compared with that in adjuvant-treated controls (P < 0.02). On the other hand, there was no difference in eNos and nNos mRNA expression between the two groups, although the latter showed a weak elevation.
FIG. 3.
RT-PCR detection of mRNA for NOS isoforms in r-calpain-immunized and adjuvant-treated control mice. (A) Primers detection of unique sequences of each isoform and produced fragments (254, 404, and 449 bp for eNos, nNos, and iNos transcripts, respectively). A 348-bp fragment of actin was used as a control. Lanes: 1 to 4, r-calpain-immunized mice; 5 to 8, adjuvant-treated controls. (B) Significant difference in iNos mRNA expression between r-calpain-immunized and adjuvant-treated control mice. Bars: solid, r-calpain-immunized group; open, adjuvant-treated control. ∗, P < 0.02.
Cytokine production.
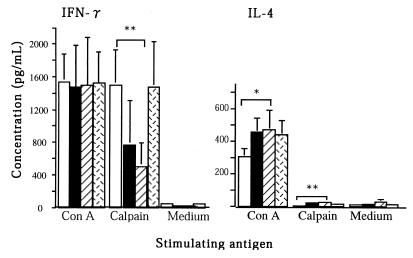
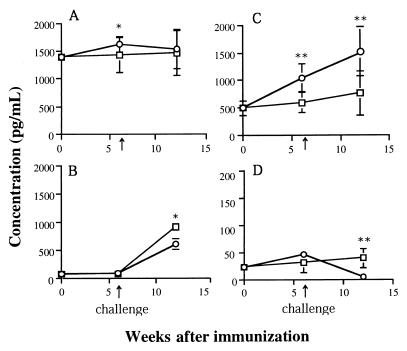
We measured the cytokine production of spleen cells in response to ConA or r-calpain on day 42 after a challenge infection (Fig. 4). Although schistosome infection is generally thought to be a strong Th2 inducer, spleen cells of control mice, both adjuvant-alone and infection control mice, did not show a typical type 2 pattern in r-calpain-driven cytokine production. Even in this case, we observed a significant reduction of IL-4 production and enhanced production of IFN-γ in response to r-calpain in mice immunized with r-calpain. A high level of r-calpain-driven IFN-γ production was also detected in mice treated with radiation-attenuated cercariae. IFN-γ production by spleen cells from mice immunized with r-calpain and with radiation-attenuated cercariae was significantly higher than in control mice (P < 0.01). However, spleen cells stimulated by ConA showed no difference in IFN-γ production among all four of the groups tested. The kinetics of r-calpain-driven IL-4 and IFN-γ production was studied: mice immunized with r-calpain showed strong IFN-γ production after vaccination and prior to challenge infection, whereas the IL-4 response was suppressed only after a challenge infection (Fig. 5C and D). Upregulation of the Th1 response during challenge infection was, thus, an apparent finding in mice immunized with r-calpain.
FIG. 4.
IFN-γ and IL-4 production by murine spleen cells (six in each group) stimulated with a mitogen or calpain in vitro. Cytokine production was evaluated in culture supernatants. Data correspond to the mean values from duplicate analyses. Bars: open, calpain-immunized mice; closed, adjuvant-treated controls; hatched, infection controls; dotted, mice immunized with irradiated cercariae. ∗, P < 0.05; ∗∗, P < 0.01.
FIG. 5.
Kinetics of cytokine production in murine spleen cells stimulated by ConA (A and B) or r-calpain (C and D) in r-calpain-immunized and adjuvant-treated control mice. Cytokines were detected at three different time points. A and C refer to IFN-γ production, and B and D refer to IL-4 production. Cytokine profiles of calpain-immunized mice are shown as circles, and those of adjuvant-treated control mice are shown as open squares. A marked increase in calpain-driven IFN-γ production and a decrease in IL-4 production were observed for the calpain-immunized group compared with the adjuvant-treated control group. ∗, P < 0.05; ∗∗, P < 0.01.
Histopathology of egg granulomas.
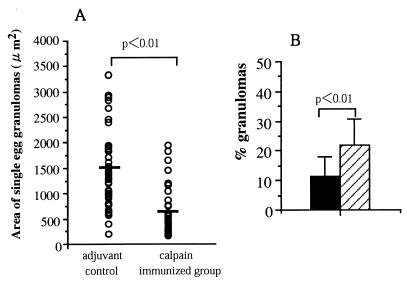
Egg granulomas formed after a challenge infection were quite a bit smaller in size and lower in number in the livers of r-calpain-immunized mice compared with those of the adjuvant-treated control group. The mean area of nonconfluent granulomas in the liver was significantly smaller in r-calpain-immunized mice (578.13 ± 257 μm2) than that in adjuvant controls (1,522.12 ± 516 μm2) (P < 0.001) (Fig. 6A). Furthermore, there was a more widespread granulomatous lesion in adjuvant-treated control mice than in the r-calpain-immunized group. According to our observation of liver sections, the mean percentage of the area occupied by granulomas was significantly different in r-calpain-immunized mice (11.6%) and adjuvant-treated control mice (22.1%) (P < 0.001) (Fig. 6B).
FIG. 6.
Comparison of granuloma size (A) and density (B) in the livers of adjuvant-treated control and immunized mice. Bars: solid, calpain-immunized group; hatched, adjuvant-treated control mice. The mean areas of single-egg granulomas and the mean areas occupied by granulomas in liver sections were compared between groups.
DISCUSSION
In the present study, we showed the efficacy of calpain as a vaccine against murine schistosomiasis japonica. Calpains are ubiquitously expressed in a wide variety of eukaryotic cell types in vivo, and it is likely that calpain modifies various regulatory and structural proteins, including the cytoskeleton, as well as inducing programmed cell death (8, 27). The biological roles of calpain in schistosome parasites are not fully understood. Calpain was shown to be an immunogenic protein that exists on or near the surface of the schistosome and is important in parasite membrane shedding and renewal (1, 19, 31). In recent vaccine development using calpain from schistosomes, Hota-Michell and coworkers reported that r-calpain afforded 39% protection against a challenge infection with S. mansoni (11), and DNA vaccine containing cDNA encoding S. mansoni calpain provided 60% protection (12). Our results also showed that S. japonicum r-calpain afforded more than 40% protection against a challenge infection of BALB/c mice with S. japonicum. The present study of S. japonicum showed that calpain induced not only a reduction in worm burden but also a reduction in egg laying per female worm in immunized mice. To our knowledge, this is the first report showing antifecundity effects of calpain on S. japonicum infecting mice. Calpain from S. japonicum seems to be a good candidate for a disease-controlling vaccine, an important step in overcoming S. japonicum infection, which causes more-severe hepatic damage through egg deposition in host animals than does an S. mansoni infection (23).
The mechanism by which r-calpain-immunized mice can induce reductions in both the worm burden and the egg production of female worms is still unclear. It has been previously reported that a helper T-cell clone recognizing the large subunit of S. mansoni calpain carried protective effects in C57BL/6 mice against cercarial challenge (11). The helper T-cell clone produced large amounts of IFN-γ in response to a truncated calpain-GST fusion protein, indicating that the protective T cells were of the Th1 phenotype (17). There is a consensus that Th1 cells are important in protective immunity against schistosome infection of mice. Previous publications have shown that mice immunized with radiation-attenuated cercariae eliminated 60 to 80% of the worms (14). Several lines of evidence suggest that responses involving IFN-γ-activated effector cells are the major mechanism of protection mediated by attenuated cercariac (15, 21, 33). In the present study, immunization with r-calpain again up-regulated IFN-γ production during a challenge infection of mice with S. japonicum.
The precise mechanisms of IFN-γ-mediated parasite elimination remain to be uncovered. James and coworkers reported killing mechanisms against lung stage schistosomulae by a NO-mediated killing mechanism (9, 16). This is not inconsistent with a recent report on a study in which iNOS mRNA was up-regulated at the time of parasite elimination (37), although other groups have disagreed with these findings (33). Other groups have shown that IFN-γ up-regulates iNos expression followed by NO production (18). Our results also suggest NO-mediated killing of lung stage schistosomulae, because there was a significant increase in the expression of iNOS mRNA in the lungs of r-calpain-immunized mice 4 days after a challenge infection. This was the time point when migrating larvae reached the lungs of host animals. Considering recent reports along with our current findings, NO seems to be involved in the protective mechanism mediated by immunization with r-calpain in schistosomiasis japonica. Further analysis is, however, still needed, as others have reported iNOS knockout mice with vaccine-mediated protection against S. mansoni (7).
Schistosomiasis caused a host response to trapped parasite eggs in the tissue resulting in the formation of granulomatous lesions in the liver and intestine. A reduction in egg number could be tightly related to lowered pathological severity in the immunized mice. Brunet et al. reported that mice treated with aminoguanidine, a selective inhibitor of iNos expression, developed severe morbidity and increased hepatic damage (4). In the present study, hepatic damage caused by egg deposition was reduced in the r-calpain-immunized group. Although we did not measure the expression of iNos mRNA in the liver, r-calpain could protect the immunized hosts from developing severe hepatic damage by expression of iNos mRNA through a systemic shift to a Th1-dominant situation. The number of deposited eggs in r-calpain-immunized mice was markedly reduced not only by a reduction in the number of adult worms but also by lowered fecundity of female worms. Attenuated cercariae did not induce such an antifecundity effect even though both immunizations stimulated strong Th1 responses. This might suggest that the antifecundity effect is mediated by a factor different from the function of IFN-γ. It is interesting to analyze whether calpain is critically involved in egg production by female schistosomes, as this might point to calpain as a possible target molecule for developing a prophylactic reagent(s).
Because an attenuated cercarial vaccine would not be practical for use in humans and livestock, investigators have attempted to use protective mechanisms defined in the model to develop a nonliving vaccine. In our experiments, we compared protective immune effects between radiation-attenuated cercariae and r-calpain. Although radiation-attenuated cercariae could induce greater protection, as was also observed by Chen et al. (6), it did not reduce egg production per female worm. Furthermore, it was interesting that r-calpain promoted only high IFN-γ production while radiation-attenuated cercariae induced high production of both IL-4 and IFN-γ after challenge infection. This suggests that there are somehow fundamental differences between the immune mechanisms of the protective effects of these two vaccine models.
In conclusion, our present study showed that r-calpain of S. japonicum could induce more than 40% protection against a challenge infection, reduce egg production by female worms, and also reduce hepatic damage in BALB/c mice. These results are highly suggestive for a future trial of calpain of S. japonicum as a vaccine candidate for livestock in the hope of controlling human schistosomiasis in Asian countries.
ACKNOWLEDGMENTS
T. Amano, Department of Parasitology, Yokohama City University Medical School, kindly provided snails used in the present study. We thank T. Kadosaka and X. G. Qiu, Aichi Medical University, for scientific and technical assistance in egg granuloma histopathological analysis.
This study was supported in part by a Grant-in-Aid for Scientific Research from the Ministry of Education, Culture and Sports, Japan (12576009); a grant for Emerging and Re-emerging Infectious Diseases from the Ministry of Health and Welfare (12101801); and a grant from the Japan-US Cooperative Medical Science Program (1999-2000).
REFERENCES
- 1.Andresen K, Tom T D, Strand M. Characterization of cDNA clones encoding a novel calcium-activated neutral proteinase from Schistosoma mansoni. J Biol Chem. 1991;266:15085–15090. [PubMed] [Google Scholar]
- 2.Bergquist R. Controlling schistosomiasis by vaccination: a realistic option? Parasitol Today. 1995;11:191–194. [Google Scholar]
- 3.Bergquist R, Colley D G. Schistosome vaccine: research to development. The search for a schistosomiasis vaccine. Parasitol Today. 1998;14:97–118. doi: 10.1016/s0169-4758(97)01207-6. [DOI] [PubMed] [Google Scholar]
- 4.Brunet L R, Beall M, Dunne D W, Pearce E J. Nitric oxide and the Th2 response combine to prevent severe hepatic damage during Schistosoma mansoni infection. J Immunol. 1999;163:4976–4984. [PubMed] [Google Scholar]
- 5.Capron A, Dessaint J P, Capron M, Pierce R J. Vaccine strategies against schistosomiasis. Immunobiology. 1992;184:282–292. doi: 10.1016/s0171-2985(11)80480-8. [DOI] [PubMed] [Google Scholar]
- 6.Chen H, Nara T, Zheng X, Satoh M, Wu G, Jiang W, Yi F, Kojima S, Zhang K S, Hirayama K. Vaccination of domestic pig with recombinant paramyosin against Schistosoma japonicum in China. Vaccine. 2000;18:2142–2146. doi: 10.1016/s0264-410x(99)00541-1. [DOI] [PubMed] [Google Scholar]
- 7.Coulson P S, Smythies L E, Betts C, Mabbott N A, Sternberg J M, Wei X-G, Liew F Y, Wilson R A. Nitric oxide produced in the lungs of mice immunized with the radiation-attenuated schistosome vaccine is not the major agent causing challenge parasite elimination. Immunology. 1998;93:55–63. doi: 10.1046/j.1365-2567.1998.00405.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Croall D E, Demartino G N. Calcium-activated neutral protease (calpain) system: structure, function, and regulation. Physiol Rev. 1991;71:813–847. doi: 10.1152/physrev.1991.71.3.813. [DOI] [PubMed] [Google Scholar]
- 9.Green S J, Nacy C A. Antimicrobial and immunopathologic effect of cytokine-induced nitric oxide synthesis. Curr Opin Infect Dis. 1993;6:384–391. [Google Scholar]
- 10.Hara H, Ayata C, Huang P L, Waeber C, Ayata G, Fujii M, Mokowitz M A. [3H]L-NG-nitroarginine binding after transient focal ischemia and NMDA-induced excitotoxicity in type I and type III nitric oxide synthase null mice. J Cereb Blood Flow Metab. 1997;17:515–526. doi: 10.1097/00004647-199705000-00005. [DOI] [PubMed] [Google Scholar]
- 11.Hota-Michell S, Siddiqui A A, Dekabab G A, Smith J, Tognon C, Podesta R B. Protection against Schistosoma mansoni infection with recombinant baculovirus-expressed subunit of calpain. Vaccine. 1997;15:1631–1640. doi: 10.1016/s0264-410x(97)00081-9. [DOI] [PubMed] [Google Scholar]
- 12.Hota-Michell S, Michael W, Clarke R B, Podesta R B, Dekaban G A. Recombinant vaccine and gene gun vectors expressing the large subunit immunization-challenge model. Vaccine. 1999;17:1338–1354. doi: 10.1016/s0264-410x(98)00391-0. [DOI] [PubMed] [Google Scholar]
- 13.James S L. Schistosoma spp.: progress towards a defined vaccine. Exp Parasitol. 1987;63:247–252. doi: 10.1016/0014-4894(87)90170-6. [DOI] [PubMed] [Google Scholar]
- 14.James S L, Labine M, Sher A. Mechanism of protection immunity against Schistosoma mansoni infection in mice vaccinated with irradiated cercariae. Analysis of antibody and T-lymphocyte responses in mouse strains developing different levels of immunity. Cell Immunol. 1981;65:75–83. doi: 10.1016/0008-8749(81)90053-8. [DOI] [PubMed] [Google Scholar]
- 15.James S L, Natovitz P C, Farrar W L, Leonard E J. Macrophages as effector cells of protective immunity in murine schistosomiasis: macrophage activation in mice vaccinated with radiation-attenuated cercariae. Infect Immun. 1984;44:569–575. doi: 10.1128/iai.44.3.569-575.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.James S L, Glaven J. Macrophage cytotoxicity against schistosomula of Schistosoma mansoni involves arginine-dependent protection of reactive nitrogen intermediates. J Immunol. 1989;143:4208–4212. [PubMed] [Google Scholar]
- 17.Jankovic D, Aslund L, Oswald I P, Caspar P, Pearce C, Coligan J E, Strand M, Sher A, James S L. Calpain is a target antigen of a Th1 clone that transfers protective immunity against Schistosoma mansoni. J Immunol. 1996;157:806–814. [PubMed] [Google Scholar]
- 18.Kamijo R, Shapiro D, Li J, Huang S, Aguet M, Vileck J. Generation of nitric oxide and induction of major histo-compatibility class II antigen in macrophages from mice lacking the interferon-γ receptor. Proc Natl Acad Sci USA. 1993;90:6626–6630. doi: 10.1073/pnas.90.14.6626. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Karcz S R, Podesta R B, Siddiqui A A, Dekaban G A, Strejan G H, Clarke M W. Molecular cloning and sequence analysis of a calcium activated neutral protease (calpain) from Schistosoma mansoni. Mol Biochem Parasitol. 1991;49:333–336. doi: 10.1016/0166-6851(91)90078-k. [DOI] [PubMed] [Google Scholar]
- 20.Kawaguchi H, Takayanagi T. Identification of a new fluke allergen identified by monoclonal IgE antibodies for Paragonimus miyazaki. J Parasitol. 1998;84:307–310. [PubMed] [Google Scholar]
- 21.Kelly E, Colly D G. In vitro effect of monoclonal anti-L3T4 antibody on immune responsiveness of mice infected with Schistosoma mansoni: reduction of irradiated cercariae-induced resistance. J Immunol. 1988;140:2737–2745. [PubMed] [Google Scholar]
- 22.Kojima S, Niimura M, Kanazawa T. Production and properties of a mouse monoclonal IgE antibody to Schistisoma japonicum. J Immunol. 1987;139:2044–2049. [PubMed] [Google Scholar]
- 23.Meleney H E, Sabdground J H, Moore D V, Most H, Carney B H. The histopathology of experimental schistosomiasis. II. Bisexual infections with S. mansoni, S. japonicum, and S. haematobium. Am J Trop Med Hyg. 1953;2:883–913. [PubMed] [Google Scholar]
- 24.Reynold S R, Charles B S, Donald A H. T and B cell epitope mapping of Sm23, an integral membrane protein of Schistosoma mansoni. J Immunol. 1992;149:3995–4001. [PubMed] [Google Scholar]
- 25.Ross A G P, Sleigh A C, Li Y S, Williams G M, Aligui G D L, McManus D P. Is there immunity to Schistosoma japonicum? Parasitol Today. 2000;16:159–164. doi: 10.1016/s0169-4758(99)01621-x. [DOI] [PubMed] [Google Scholar]
- 26.Saido T C, Suzuki H, Yamazaki H, Tanoue K, Suzuki K. In situ capture of μ-calpain activation in platelets. J Biol Chem. 1993;268:7422–7426. [PubMed] [Google Scholar]
- 27.Saido T C, Sorimachi H, Suzuki K. Calpain: new perspectives in molecular diversity and physiological-pathological involvement. FASEB J. 1994;8:814–822. [PubMed] [Google Scholar]
- 28.Scott J C, McManus D P. Characterization and expression of cDNA encoding the 80-kDa large subunit of Schistosoma japonicum calpain. Parasitol Int. 2000;48:205–214. doi: 10.1016/s1383-5769(99)00021-5. [DOI] [PubMed] [Google Scholar]
- 29.Shoemaker C, Gross A, Gebremichael A, Harn D A. cDNA cloning and functional expression of the Schistosoma mansoni protective antigen triose phosphate isomerase. Proc Natl Acad Sci USA. 1992;89:1842–1846. doi: 10.1073/pnas.89.5.1842. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Shuxian L, Guangchen S, Yuxian X, Yang W, Mcmanus D P. Immunization of mice with recombinant Sjc 26GST induces a pronounced anti-fecundity effect after experimental infection with Chinese Schistosoma japonicum. Vaccine. 1995;13:603–607. doi: 10.1016/0264-410x(94)00045-o. [DOI] [PubMed] [Google Scholar]
- 31.Siddiqui A A, Zhou Y, Podesta R B, Karcz S R, Tognon C E, Strejan G H, Dekaban G A. Characterization of Ca2+-dependent neutral protease (calpain) from human blood flukes, Schistosoma mansoni. Biochiem Biophys Acta. 1993;1181:37–44. doi: 10.1016/0925-4439(93)90087-h. [DOI] [PubMed] [Google Scholar]
- 32.Smith D B, Rubia M R, Simpson R J, Davern K, Tiu W U, Board P G, Mitchell G F. Expression of an enzymatically active parasite molecule in Escherichia coli: Schistosoma japonicum glutathione-S-transferase. Mol Biochem Parasitol. 1988;27:249–256. doi: 10.1016/0166-6851(88)90044-8. [DOI] [PubMed] [Google Scholar]
- 33.Smythies L E, Coulson P S, Wilson R A. Monoclonal antibody to IFN-γ modifies pulmonary inflammatory responses and abrogates immunity to Schistosoma japonicum in mice vaccinated with attenuated cercariae. J Immunol. 1992;149:3654–3658. [PubMed] [Google Scholar]
- 34.Wilson R A, Coulson P S. Why don't we have a schistosomiasis vaccine? Parasitol Today. 1998;14:97–99. doi: 10.1016/s0169-4758(97)01198-8. [DOI] [PubMed] [Google Scholar]
- 35.World Health Organization. Tropical diseases research, no. 50. Geneva, Switzerland: World Health Organization; 1996. [Google Scholar]
- 36.World Health Organization. The world health report 1997. Geneva, Switzerland: World Health Organization; 1988. [Google Scholar]
- 37.Wynn T A, Oswala I P, Eltoum I A. Elevated expression of Th1 cytokines and nitric oxide synthase in the lungs of vaccinated mice after challenge infection with Schistosoma mansoni. J Immunol. 1994;153:5200–5209. [PubMed] [Google Scholar]
- 38.Zhang R L, Suzuki T, Takahashi S, Yoshida A, Kawaguchi H, Maruyama H, Yabu Y, Fu J, Shirai T, Ohta N. Cloning and molecular characterization of calpain, a calcium-activated neutral proteinase, from different strains of Schistosoma japonicum. Parasitol Int. 2000;48:233–242. doi: 10.1016/s1383-5769(99)00024-0. [DOI] [PubMed] [Google Scholar]