Abstract
Anxiety disorders are the most common mental health disorder. Therefore, elucidating brain mechanisms implicated in anxiety disorders is important avenue for developing novel treatments and improving care. The dorsolateral prefrontal cortex (dlPFC) is thought to be critically involved in working memory processes (i.e. maintenance, manipulation, suppression, etc.). In addition, there is evidence that this region is involved in anxiety regulation. However, it is unclear how working memory related dlPFC processes contribute to anxiety regulation. Furthermore, we know that laterality plays an important role in working memory related dlPFC processing, however there is no current model of dlPFC mediated anxiety regulation that accounts for potential laterality effects. To address this gap, we propose a potential framework where the dlPFC contributes to emotion regulation via working memory processing. According to this framework, working memory is a fundamental process executed by the dlPFC. However, the domain of content differs across the left and right dlPFC, with the left dlPFC sensitive to primarily verbal content, and the right dlPFC sensitive to primarily non-verbal (affective content). Critically, working memory processes allow for both the retention and suppression of affective information in working memory and the overall net effect of processing on mood will depend on the balance of retention and suppression, the valence of the information being processed (positive vs. negative), and the domain of the information (verbal vs. non-verbal). If accurate, the proposed framework predicts that effects of neuromodulation targeting the dlPFC may be dependent upon the context during which the stimulation is presented.
Keywords: dlPFC, anxiety, threat, working memory, neuromodulation
Introduction
Anxiety disorders are the most common mental health disorder1. The frontline treatments for anxiety disorders are cognitive behavior therapy and psychoactive medications (e.g., serotonin reuptake inhibitors; SRRIs). While these treatments are effective, the rate of treatment resistance is high and prevention efforts are lacking2. Elucidating brain mechanisms implicated in anxiety disorders is an important avenue for developing novel treatments and improving the current standard of care3. There are a host of brain regions that have been implicated in anxiety (e.g., amygdala, dorsal prefrontal cortex, anterior cingulate cortex) and connectivity between these regions appears to be particularly important in anxiety4. The dorsolateral prefrontal cortex (dlPFC), a region of the lateral prefrontal cortex in the middle frontal gyrus, generally thought to overlap especially Brodmann areas 9 and 46, appears to be integrally involved in anxiety and is a likely target for treatment and prevention efforts. The function and organization of the dlPFC can be best understood with reference to its connections with other structures, as it is a highly connected node in the frontoparietal attention network with connections to temporal, parietal, and limbic regions. The dlPFC is critically important for working memory, executive control, and regulation of emotions, particularly anxiety.
While most models of anxiety treat the dlPFC as a unitary structure, there is evidence that the left and right dlPFC contribute uniquely to the expression and regulation of anxiety. Therefore, the purpose of this review is to propose a potential mechanistic framework describing anxiety regulation that includes distinct roles for the left and right dlPFC, based on the content of items in working memory. Importantly, the goal of this review is not to summarize emotion regulation and the neural architecture underlying emotion regulation, as these topics have been covered previously5,6. The current review will 1) Summarize the literature demonstrating a role for the dlPFC in working memory and anxiety regulation 2) Summarize the dlPFC laterality literature, with respect to anxiety regulation 3) Describe our content domain framework of dlPFC function and 4) Highlight the testable predictions and treatment implications of this framework in anxiety research.
Methods
Although this is not a systematic review, we adopted a systematic approach to retrieve and sort articles for the current work. We used a custom python script (https://github.com/balders2/pubmed_search) that queried pubmed and fetched the publication date, pubmed ID, first author, last author, title, number of citations, and article pdf (if available) of the articles retuned from the query. We queried pubmed during early to mid 2022 using the following search terms: “dlPFC anxiety”, “dlPFC connectivity”, “dlPFC depression”, “dlPFC DMN”, “dlPFC GAD”, “dlPFC inhibition”, “dlPFC manipulation”, “dlPFC MDD”, “dlPFC n-back”, “dlPFC regulation”, “dlPFC spatial”, “dlPFC Sternberg”, “frontal alpha asymmetry”. These searches returned 4451 unique results. We then screened these articles and excluded those that were either uncited or off topic (i.e., included terms like: schizophrenia, parkinsons, anorexia, addiction, autism, adhd, stroke, dementia, etc.). From there we were left with 379 articles. These articles were then reviewed by the authors (NLB and LKW) and included in the review if they contributed to the pre-planned sections of the article. Although care was taken not to exclude articles that ran counter to our proposed framework, we did not systematically define a set of inclusion/exclusion criteria for these remaining articles. Accordingly, we believe this article does not meet the criteria of a systematic review and acknowledge that the topics contained within may be biased by the perspectives of the authors.
dlPFC and Working Memory
A classic body of literature supports the role of the dlPFC in cognitive control in goal-directed behavior, including a host of cognitive processes, such as directing attention, response selection, inhibitory control, working memory, and long-term memory7–11. The dlPFC plays a critical role in the representation, updating, and maintenance of context information that can be used to guide behavior12; such context information can be task instructions, internal goal states, or the results of processing previous stimuli.
In imaging work by our group and others, we show the dlPFC plays a particularly important role in working memory13,14. Working memory – the flexible online maintenance and manipulation of information in conscious awareness – can involve multiple processes like rehearsal, item shifting, spatial rotation, information suppression, and filtering depending on task demands. For the purposes of this review, we primarily distinguish between maintenance-like processes (those that keep specific items active in working memory) and suppression-like processes (those that keep specific items from becoming active in working memory). It is known that the dlPFC engages in both, and that maintenance and suppression processes can occur concurrently in the dlPFC.
Multiple brain imaging methods support the role of dlPFC is working memory maintenance. For instance, noninvasive brain stimulation such as transcranial direct current stimulation (tDCS) and transcranial magnetic stimulation (TMS) to the dlPFC is associated with improved working memory maintenance7. In lesion studies, patients with dlPFC damage showed specific impairment in manipulations and maintenance of information in working memory15, a finding also supported by imaging work16. Finally, connectivity between the dlPFC and dorsal anterior cingulate cortex/pre supplemental motor area (dACC/pSMA) were related to working memory demands17. Likewise, imaging work finds the dlPFC also plays a crucial role in suppressing distractors from working memory18, possibly through top-down attentional filtering or goal-oriented processes16. Working memory suppression processes may be mediated through connectivity between the dlPFC and the inferior frontal gyrus (IFG) and may be related to cognitive control demands17.
Interestingly, not all types of information are processed similarly by the dlPFC. More specifically, the left and right dlPFC seem to be specialized for distinct domains of content. The left dlPFC appears to be primarily sensitive to verbal content; the right dlPFC appears to be primarily sensitive to non-verbal content. For instance, verbal n-back tasks evoke more BOLD activity in the left dlPFC compared to the right. In contrast, spatial n-back tasks evoke more BOLD activity in the right dlPFC compared to the left19. Accordingly, although the overall processes engaged by the left and right dlPFC are similar, their functional profile is largely dependent upon the stimulus type.
Repetitive TMS (rTMS) targeting the dlPFC during WM tasks also shows laterality effects based on stimulus content. This was found in a study in healthy subjects who received 10 Hz rTMS during the retention interval of a 2-back WM task that required maintaining either verbal or spatial information about the task stimuli. rTMS to the right dlPFC interfered with performance in the verbal 2-back condition; rTMS to the left dlPFC interfered with performance in the spatial 2-back condition. The stimuli and task were identical across domain conditions, suggesting that excitatory rTMS interferes with WM performance in a domain-specific manner, biasing WM processes toward the dominant domain of the stimulation site, such that this interference depends on the laterality of the stimulation20. In another example, patients with left dlPFC damage show impairments in a WM task that required subjects to name pictures based on semantic associations, suggesting an impairment in the maintenance of verbal information. In contrast, patients with right dlPFC damage showed impairments in a procedural executive control task that required matching colors and labels, but that did not require maintenance of semantic information. Critically, both tasks required the use of task rules across trials but differed in the domain of the rules to be maintained (semantic vs non-semantic). Thus, the results demonstrated a double dissociation in the domain of information maintenance across hemispheres, even when similar working memory processes were engaged21.
Together the work in this section suggests that the dlPFC plays a critical role in working memory and that the laterality of processing largely depends on the content of the information to be processed.
dlPFC and Emotion Regulation
Despite being characterized as a cognitive region of the brain, the dlPFC is known to play a crucial role in emotion regulation. Emotion regulation is broadly defined as the effortful or automatic strategies employed to reduce, induce, manipulate, or modify an emotion22. Functional imaging studies have found the dlPFC activates when emotional processing needs to be inhibited or integrated with ongoing cognitive processing23–25. Lesion works shows that patients with dlPFC damage were not able to downregulate their subjective fear to a conditioned stimulus26. Work with tDCS shows that stimulation to the dlPFC is associated with increased cognitive control during emotion regulation, facilitating subjects’ ability to reduce skin conductance responses to affective pictures27. The dlPFC is largely thought to regulate emotional processing in a top-down manner through its inhibitory connections to other brain regions, such as the anterior cingulate cortex, dorsomedial prefrontal cortex (dmPFC), and amygdala28. The negative coupling between the dlPFC and these other regions has been well replicated29,30, with a particularly robust negative correlation emerging between the dlPFC and amygdala31–33.
Support for dlPFC involvement in emotion regulation also comes from observed dlPFC activation on executive control tasks that use emotion stimuli34 and from disruption to emotion regulation when cognitive control and working memory demands are increased35. Such top-down control is indexed by increased dlPFC recruitment when emotion-related distractors need to be inhibited (i.e., suppressed)36, where the dlPFC is thought to down-regulate activity in the amygdala and other limbic regions37,38. Interestingly, anxiety has been related to both hypo-39 and hyper-activation of the dlPFC during the suppression of threat distractors40. We have shown that increasing task difficulty during periods of unpredictable threat activates the right dlPFC more than during neutral conditions and this activation was positively correlated with task performance41. dlPFC activity is also negatively correlated with subgenual Anterior Cingulate Cortex (sgACC) activity, another emotion regulation hub of the brain, during the suppression of threat distracrtors42. dlPFC activity is also negatively correlated with parietal activity evoked by task-irrelevant affective face stimuli in a face-stroop task43. dlPFC activation is also observed during neutral conditions of cognitive reappraisal tasks44,45, suggesting the dlPFC may underlie more general cognitive demands of the emotion regulation process, irrespective of emotional content.
Our work using the No-shock, Predictable Shock, Unpredictable shock (i.e., NPU) task, a paradigm designed to study anxiety in humans, has shown that the right dlPFC is activated by cues presented during periods of unpredictable threat, and this increased dlPFC activation is associated with reduced anxiety46. In other work with the NPU task, we have shown that increased activation of the dlPFC is associated with a smaller anxiety potentiated startle response47,48. Importantly, reduction in startle potentiation is directly related to the cognitive load of the task49,50. These results suggest that cognitive and affective information interacts in the dlPFC, possibly competing for limited resources in working memory.
Neuroimaging work showing dlPFC abnormalities during cognitive and emotional tasks also supports dlPFC-mediated emotion regulation. Greater dlPFC activity during an emotional task condition compared to a neutral condition was associated with PTSD symptoms in trauma exposed individuals with subthreshold PTSD51. Greater dlPFC activity and less amygdala activity prior to treatment is also associated with better treatment outcomes in PTSD patients52, suggesting better top-down regulation mechanisms may assist in treatment response. Emotion-regulation dlPFC related findings have been mixed in studies using anxious populations. For instance, GAD patients showed reduced dlPFC activation in a working memory task when the task required suppression of an emotional distractor53. In contrast, GAD patients show increased dlPFC activity during the presentation of negative picture compared to SAD and control subjects, suggesting the GAD patients are possibly attempting to implement more emotion regulation while viewing negative images54.
Together the work in this section suggests that the dlPFC plays a critical role in general emotion regulation and the regulation of anxious states; however, more research is needed to fully understand the role of dlPFC in anxious pathology.
dlPFC and Effort
Despite clear evidence that the dlPFC contributes to emotion regulation processes, it is unclear how emotion regulation fits in with the broader profile of dlPFC working memory function, especially in the context of anxiety. One possibility is that the dlPFC contributes to emotion regulation by supporting active effortful cognitive strategies. In this case, it may be argued that emotion regulation is mediated by maintenance processes of working memory, those processes required to keep certain information maintained in working memory. For instance, working memory dlPFC-related dysfunction, even under neutral conditions, is common in individuals with mood and anxiety disorders55,56 Thus, for anxious individuals, perturbations in ability to maintain information in working memory may contribute to their dysregulated anxiety. We found hyperactivation in dlPFC during a neutral working memory task was related to anxiety14,57, a finding replicated in other work58. However, several studies have found anxiety is related to dlPFC hypoactivation during working memory tasks59,60; GAD patients show reduced loadrelated dlPFC activation during the N-back WM task61. Behaviorally, we have also shown that better working memory performance was related to greater anxiety-related symptoms62; however, anxiety-related WM deficits are also reported49. Further work is needed to understand these contradictory dlPFC dysfunction findings in the context of working memory maintenance and anxiety; dlPFC laterality, different cognitive demands, or task parameters may underlie these differences.
On the other hand, it might be that the dlPFC contributes to emotion regulation by supporting more automatic emotion processes, like the down regulation of activation in emotional processing regions. In this case, it may be argued that emotion regulation is mediated by a suppression-like working memory process, those processes that inhibit certain information from entering working memory space; the dlPFC works to filter out or suppress negative emotional responses. For instance, the level of functional connectivity between the dlPFC and amygdala are related to outcomes following exposure therapy63. In addition, mindfulness training, thought to strengthen top-down control of emotion, increases connectivity between the dlPFC and brain regions64. Finally, neurofeedback aimed at increasing dlPFC activity has been found to reduce anxiety in phobic patients65. On the other hand, stress induction can lead to a decoupling of the amygdala and dlPFC, which affects one’s ability to filter out (i.e., suppress) emotional distractors during ongoing cognitive tasks66. Additionally, trauma exposure, independent of PTSD diagnosis, can lead to decreases in top-down connectivity between the dlPFC and the amygdala67. Thus, poor connectivity between dlPFC and other emotion hubs, can lead to the inability to suppress or inhibit negative emotions or emotional information, thereby increasing or maintaining anxiety.
Together the work in this section suggests that the dlPFC potentially contributes to emotion regulation through both effortful and automatic processes, relying on multiple working memory processes.
dlPFC and Laterality
In addition to ambiguity related to the working memory process engaged during the regulation of anxiety, there is also ambiguity related to how the unique roles of the left and right dlPFC influence emotion regulation. Specifically, it is unclear whether the left and right dlPFC contribute to emotion regulation in similar or distinct ways. Indeed, there is mounting evidence that suggests that left and right dlPFC contribute to emotion regulation through distinct processes. For instance, early EEG research has historically offered support for a lateralized view of the dlPFC68. Although more recent work has questioned the clinical utility of frontal alpha as a biomarker for mood disorders69,70, there is evidence to suggest that mood states differentially affect alpha power in the left and right hemisphere. Specifically, lower left alpha power relative to right alpha power was associated with positive affect or approach (positive emotional reactivity); higher left alpha power relative to right was associated with negative affect or avoidance (i.e., negative emotional reactivity)71. Given that alpha power is generally thought to reflect cortical inhibition72, it is thought that greater activity or excitability in the left dlPFC (i.e., left lateralized frontal alpha asymmetry [FAA]) is associated with positive mood and approach behaviors while greater activity or excitability in the right dlPFC (i.e., right lateralized FAA) is associated with a more negative mood and avoidance or anxiety-related behaviors73. Left-lateralized FAA is correlated with Behavioral Approach System (BAS)74 scores as well as reward bias in an incentivized verbal WM paradigm75. Individuals who have left-lateralized FAA also exhibit a positive affective bias when evaluating affectively neutral stimulus relationships76. Similarly, FAA is left-lateralized during presentations of positive facial expressions, while right-lateralized during presentations of negative expressions77. Several studies have shown increased right-lateralized FAA in depression78, anxiety79, and PTSD patients80. PTSD patients also show increased right-lateralized parietal alpha asymmetry81,82. More recent imaging work beyond FFA83–85 has shown that depressed patients have lower activity in the left prefrontal cortex and relatively higher activity in the right prefrontal cortex, suggesting an imbalance between left and right prefrontal activity may be associated with the pathophysiology of depression. It remains unclear if similar findings hold true in anxiety.
The lateralization FAA work formed much of the basis for the emergence of lateralized dlPFC neuromodulation protocols for the treatment of depression and anxiety. One such technique, TMS, uses either repetitive (rTMS) or patterned (theta burst stimulation; [TBS]) stimulation to modify excitability in localized cortical circuits. Work in motor cortex suggest that some frequencies (> 5 Hz) and patterns (intermittent TBS) of stimulation are often found to increase excitability, while other frequencies (< 5 Hz) and patterns (continuous TBS) reduce excitability86–92. Researchers designed clinical trials aimed at using rTMS to alter excitability levels in the dlPFC, most commonly with high frequency (excitatory; 10 Hz) stimulation to the left dlPFC and low frequency (inhibitory; 1 Hz) stimulation to the right dlPFC93. A network meta-analysis (81 clinical trials, N = 4,233 patients) compared the effects of eight rTMS interventions for the treatment of depression and anxiety, including the high frequency left and low frequency right with sham stimulation. The study found that compared with sham, both high frequency left and low frequency right rTMS were significantly more effective with respect to remission of depression symptoms94. Some studies have combined high frequency left and low frequency right in a sequential bilateral rTMS treatment and, while this was shown to have clear superiority over sham,94 bilateral treatment was not superior to high frequency left treatment. There is also evidence that these asymmetric rTMS treatment protocols may be effective for comorbid anxiety symptoms as well. For instance, left HF-rTMS95, right LF-rTMS96, and sequential bilateral rTMS97 can reduce anxiety symptom in depressed individuals. However, several meta-analyses have failed to find efficacy in using rTMS to treat primary GAD patients98–101. Thus, further work is needed to understand the role of dlPFC laterality in anxiety and the current stimulation approaches need to be optimized for anxiety.
Together the work in this section suggests lateralization of dlPFC function is critical for understanding how the dlPFC contributes to emotion regulation, specifically anxiety.
Proposed Framework
Although there is evidence to suggest left and right dlPFC support distinct functions in emotion regulation, this laterality is not often addressed in anxiety research. To date, there is not a basic mechanistic framework of dlPFC function that accounts for such laterality as it relates to anxiety regulation. We believe that having such a mechanistic framework will help bridge seemingly disparate findings in the literature, elucidate the pathways leading to anxiety, and inform treatment protocols. Here we put forth a framework based on three guiding principles (See Figure 1; See also Table 1 for a list of papers supporting each of these claims). First, the dlPFC’s role in the executive function network5,6, specifically the role of working memory in maintaining and manipulating (e.g. shifting, sorting, suppressing) information6,102, is critical to emotion regulation (Table 1: emotion regulation). Second, although both the left and right dlPFC are engaged in common working memory processes, the different hemispheres mostly contribute to these processes within distinct content domains (i.e., verbal vs non-verbal; Table 1: content domains). Third, verbal and non-verbal emotion-related content is subject to these working memory processes and the degree to which the right and left dlPFC contribute to emotion expression and regulation is dependent upon the working memory process currently engaged (i.e. maintaining, shifting, sorting, suppressing; Table 1: maintenance vs. suppression).
Figure 1. Content domain framework.
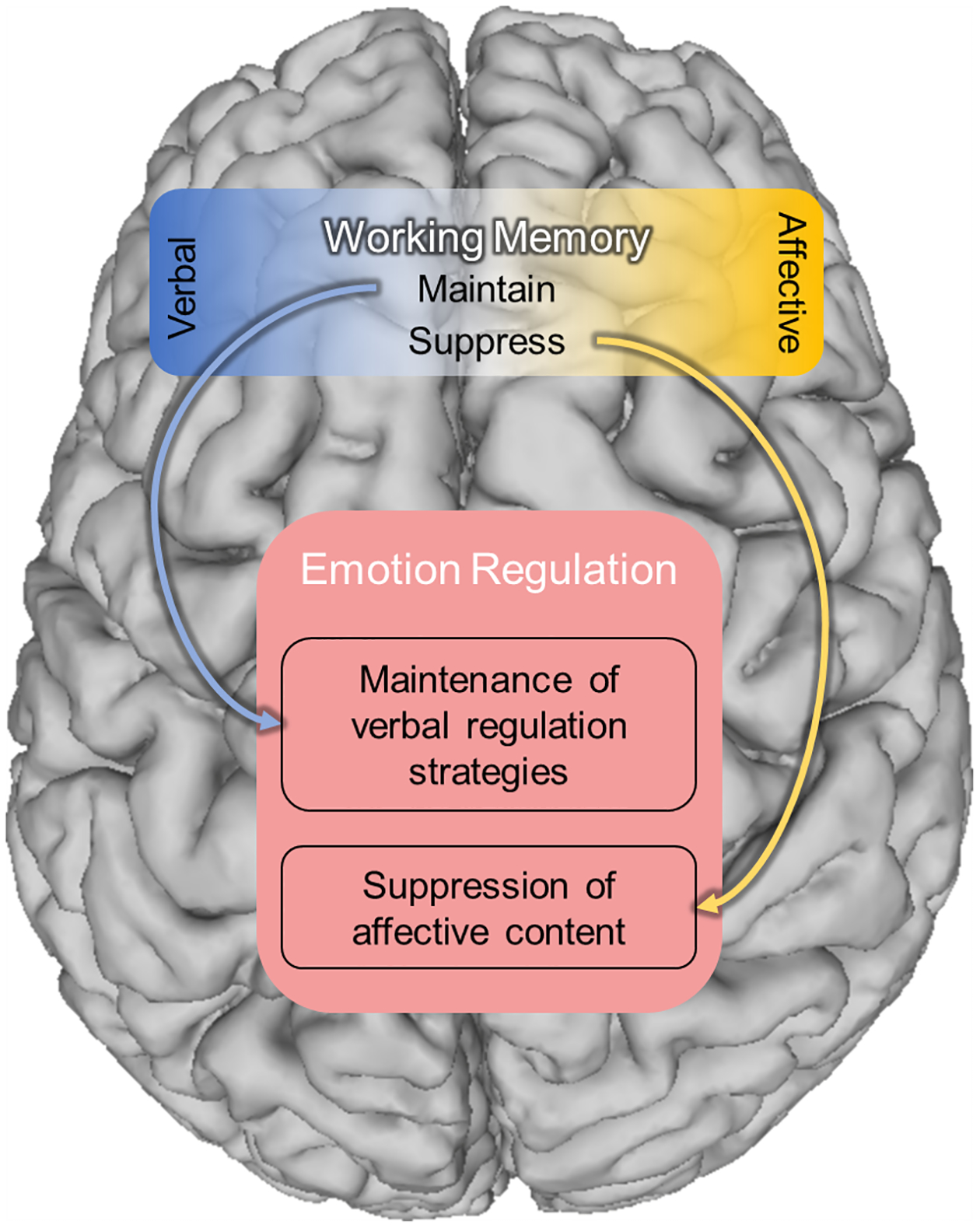
According to the content domain framework, the dlPFC is primarily engaged in working memory related processes. However, the domain of information being processed differs across hemispheres. We posit that the primary domain of information processed by the left dlPFC is verbal, while the primary domain of information processed by the right dlPFC is non-verbal, with affect-related content making up a key sub-domain. Accordingly, both the left and right dlPFC can contribute to emotion regulation, depending on which working memory process is engaged, but their contributions will differ based on the domain of information being processed.
Table 1.
Papers related to the development of the proposed framework.
| Citation | Reference Number | Principle | Primary Method | Main Finding |
|---|---|---|---|---|
| Nagel et al., 2013 | 19 | content domains | fMRI | Left lateralized activity for verbal WM, right lateralized activity for spatial WM. |
| Sandrini et al., 2008 | 20 | content domains | rTMS | Excitatory rTMS to right dlPFC slows verbal WM performance, while excitatory rTMS to the left dlPFC slows spatial. |
| Davidson et al., 1990 | 68 | content domains | EEG | Verbal and spatial tasks differentially affect left and right EEG alpha power. |
| Sutton & Davidson, 2000 | 76 | content domains | EEG | Left frontal alpha activity is associated with processing of verbal information in an affective task. |
| Metzger et al., 2004 | 82 | content domains | EEG | Right parietal excitability is associated with severity of PTSD arousal symptoms. |
| O’Reardon et al., 2007 | 95 | content domains | TMS | High frequency rTMS to the left dlPFC decreases depression symptoms. |
| Mantovani et al., 2013 | 96 | content domains | TMS | Low frequency rTMS to the right dlPFC improves panic symptoms. |
| White & Tavakoli, 2015 | 97 | content domains | TMS | Low frequency rTMS to the right dlPFC improves anxiety symptoms in comorbid GAD/MDD patients. |
| Gray et al., 2002 | 23 | emotion regulation | fMRI | dlPFC is activated by emotional content when task demands are high. |
| Liberzon et al., 2000 | 24 | emotion regulation | PET | Emotion regulation during affective picture viewing increases metabolic activity in the dlPFC. |
| Beauregard et al., 2001 | 25 | emotion regulation | fMRI | Suppression of emotional responses during erotic picture viewing is associated with right dlPFC activity. |
| Kroes et al., 2019 | 26 | emotion regulation | lesion | Patients with dlPFC are unable to use active strategies to down regulate arousal responses during fear conditioning. |
| Feeser et al., 2014 | 27 | emotion regulation | tDCS | Anodal tDCS over right dlPFC decreases affective responding when subjects are instructed to down-reglate their emotions to affective images. |
| Simpson et al., 2000 | 30 | emotion regulation | fMRI | Increased activation in the dlPFC and limbic regions were detected in response to negative vs neutral images |
| Hartling et al., 2021 | 34 | emotion regulation | fMRI | Increased dlPFC activation was detected during an N-back task using emotional/affective stimuli |
| Adamczyk et al., 2022 | 35 | emotion regulation | EEG/ERP | Increased cognitive load was associated with a decreased emotion regulation-realted ERP component |
| Ironside et al., 2018 | 37 | emotion regulation | tDCS + fMRI | Stimulation of the dlPFC decreased amygdala response to threats and increased activation in cortical regions associated with attentional control during a threat attention task |
| Loos et al., 2020 | 38 | emotion regulation | fMRI | Anxiety Patients showed decreased amygdala response to neutral and threat stimuli when cognitive load; this was mediated by cognitive load related increases in the dlPFC |
| Bishop et al., 2004 | 39 | emotion regulation | fMRI | High anxious participants showed larger decreased left dlPFC on a threat-attention task when threat distractors were frequent |
| Fu et al., 2017 | 40 | emotion regulation | fMRI | Behaviorally inhibited individuals (anxiety risk) showed increased right dlPFC during an attention-threat task |
| Balderston, Hsiung, et al., 2017 | 41 | emotion regulation | fMRI | Increased dlPFC activation was seen under threat conditions compared to neutral conditions during a memory task |
| Mitchell et al., 2007 | 42 | emotion regulation | fMRI | Negative coupling between dlPFC and ventromedial PFC/subgenual Anterior Cingulate Cortex (sgACC) was detected on tasks requiring the suppression of emotional stimuli |
| Banich et al., 2019 | 43 | emotion regulation | fMRI | Increased dlPFC activity was associated with less processing of task irrelevant emotional information |
| Morawetz et al., 2020 | 44 | emotion regulation | fMRI | Increased dlPFC activity and dlPFC-amygdala connectivity was associated with reappraisal; level of connectivity was associated with the success of reappraisal |
| Golkar et al., 2012 | 45 | emotion regulation | fMRI | dlPFC activity was associated with reappraisal of both negative and neutral stimuli |
| Balderston, Liu, et al., 2017 | 46 | emotion regulation | fMRI | During an anxiety manipulation, dlPFC activity was associated with increased subjective and objective anxiety responses |
| Vytal et al., 2013 | 49 | emotion regulation | behvaior | High WM load was associated with decreased effects of an anxiety manipulation compared to medium and low WM loads on a verbal WM task; decreased anxiety disruptions on WM were seen across all WM loads on a spatial WM task |
| Vytal et al., 2012 | 50 | emotion regulation | behavior | High WM load was associated with decreased effects of an anxiety manipulation compared to low WM load; increased WM load was also associated with decreased anxiety |
| Fonzo et al., 2017 | 52 | emotion regulation | fMRI/TMS | In PTSD patients, increased dlPFC activation during an emotion task was associated with greater treatment response. TMS to right dlPFC was associated with decreased amygdala activity on the task |
| Moon & Jeong, 2015 | 53 | emotion regulation | fMR | Anxiety patients showed lower dlPFC activity in the presence of anxiety-related distractors during a WM task |
| Buff et al., 2016 | 54 | emotion regulation | fMRI | Anxiety patients showed increased dlPFC-amygdala connectivity when viewing threat vs neutral images |
| Balderston et al., 2016 | 61 | emotion regulation | fMRI | Anxiety patients showed decreased dlPFC activity as a function of cognitive load across threat and neutral conditions on a spatial WM task |
| Balderston et al., 2020 | 14 | retention vs. suppression | fMRI | Anxiety patients showed decreased dlPFC during an anxiety manipulation during a letter WM task; patients did not show hemispheric differences, but control participants showed decreased activity in right vs left dlPFC |
| Vasic et al., 2009 | 55 | retention vs. suppression | fMRI | Depressed patients showed increased left dlPFC connectivity during a letter WM task |
| Fernández-Corcuera et al., 2013 | 56 | retention vs. suppression | fMRI | Bipolar patients showed decreased bilateral dlPFC during a letter WM task |
| Basten et al., 2012 | 57 | retention vs. suppression | fMRI | Trait anxiety was associated with increased right dlPFC and stronger right dlPFC connectivity during the manipulation phase of a letter working memory task compared to maintenance phase |
| Fales et al., 2008 | 58 | retention vs. suppression | fMRI | High anxious participants showed increased transient WM-related bilateral dlPFC activation but reduced sustained right dlPFC activation during a verbal WM task |
| Moon et al., 2016 | 59 | retention vs. suppression | fMRI | Anxiety patients showed decreased right dlPFC activation in response to emotional distractors on a face WM task |
| Moon & Jeong, 2017 | 60 | retention vs. suppression | fMRI | Anxiety patients showed decreased left dlPFC to emotional distractors during a face WM task |
| Balderston et al., 2016 | 61 | retention vs. suppression | fMRI | Anxiety patients showed decreased right dlPFC activation as a function of cognitive load during a spatial WM task across threat and neutral conditions |
| Luo et al., 2018 | 66 | retention vs. suppression | fMRI | During a stress induction, decreased left dlPFC-amygdala connectivity was detected during encoding phase on a letter search task, this was especially true under conditions of low cognitive load and emotion distractors |
| Chen et al., 2018 | 67 | retention vs. suppression | fMRI | PTSD patients and trauma exposed controls showed decreased dlPFC to amygdala connecting during resting state activity |
Discussion
We believe that the perspective encompassed by the proposed dlPFC processing framework fits into a larger trend of recognizing commonalities between cognition and emotion. Indeed, there is ample information to suggest that cognition and emotion mutually interact and affect one another103. According to the framework presented here, there should be little distinction between affective and non-affective information in the dlPFC as both are subject to the generalized processes of working memory carried out by this region. Critically, these working memory/executive processes allow for both the maintenance and suppression of affective information in working memory102,104, which has implications for understanding emotion regulation strategies. For instance, maintenance-like processes engaged by the left dlPFC may regulate emotion by supporting the active, effortful engagement of verbal emotion regulation strategies, like reappraisal. In contrast, suppression-like processes engaged by the right dlPFC may regulate emotion by supporting the automatic top-down filtering of negative affective information, resulting in reduced activity in emotional expression regions. However, in real-life situations, the dlPFC is likely constantly engaged in some combination of maintenance and suppression processes across a wide variety of content types. Additionally, with the limited resources of working memory, the active maintenance of one type of information will likely lead to the suppression of other types of information, resulting in competition for resources105. Accordingly, as discussed above, active TMS to the left dlPFC during spatial WM memory may bias the system toward the maintenance of verbal information and as a result lead to the suppression of non-verbal information, resulting in a site-specific decrease in performance20. According to the proposed framework, the overall net effect of this WM-related dlPFC processing on mood will depend on the balance of maintenance and suppression, the valence of the information being processed (positive vs. negative), and the domain of the information (verbal vs. non-verbal).
Model Predictions
The proposed framework predicts that the dlPFC can both up and down-regulate negative emotional information depending on context. If this is the case, it should be possible to show increases in dlPFC (specifically right hemisphere) activity are associated with the active maintenance of positive mood as well as the active suppression of negative mood and these directly influence anxiety levels. Such a result would be inconsistent with the frontal asymmetry model mentioned above68,71.These results have implications for how neuromodulation may affect mood and, importantly, anxiety.
The standard clinical practice for treating depression and anxiety symptoms with TMS is to apply excitatory TMS to the left dlPFC and inhibitory TMS to the right dlPFC93,94. In the proposed framework, the left dlPFC primarily operates on verbal content domains, while the right dlPFC primarily operates on non-verbal content domains. Accordingly, high frequency (excitatory) stimulation of the left dlPFC should facilitate effortful, verbal cognitive emotion regulation strategies. In contrast, this framework suggests low frequency (inhibitory) rTMS to the right dlPFC would affect anxiety primarily by weakening the maintenance processes that stabilize negative affect, leading to a net decrease in negative affect (e.g., decreased anxiety). However, TMS stimulation can have a state-dependent effect, impairing or facilitating specific functions that are active at the time of stimulation. (i.e., the underlying activity/connectivity patterns corresponding to the current mood state). Thus, in healthy subjects who typically have a net positive affect, the proposed framework predicts opposing effects on mood; low frequency (inhibitory) rTMS to the right dlPFC might weaken the maintenance of positive affect, leading to a net decrease in positive affect. More interestingly, the proposed framework predicts that manipulation of mood at the time of stimulation may be leveraged to modulate the lasting effects of the stimulation. Take these examples. First, positive mood induction could be paired with excitatory stimulation to the right dlPFC to strengthen the retention processes important for maintaining positive affective tone. Alternatively, negative mood induction could be paired with inhibitory stimulation of the right dlPFC to weaken the retention processes important for maintaining negative affective tone. In both cases, the result would be a net positive effect on mood.
Indeed, we have seen similar effects when comparing the effects of within-session vs. between session stimulation of the right dlPFC on anxiety during threat of unpredictable shock. When we use 10 Hz (presumably excitatory) stimulation to modulate right dlPFC activity within-session, we get increases in anxiety potentiated startle (APS). This suggests a potential strengthening of the anxiety maintenance circuits activated during the anxiety manipulation106. Accordingly, we predicted that an inhibitory stimulation pattern to the right dlPFC would lead to a reduction in APS. However, when we use cTBS (presumably inhibitory) stimulation to modulate right dlPFC activity between sessions (i.e., not during an anxiety manipulation) this inhibitory stimulation also leads to an increase in anxiety potentiated startle107. According to the proposed framework, this increase in startle, an anxiety-related response, following offline cTBS may be due to a weakening of the circuits underling the maintenance of a non-anxious affective tone.
Importantly, the proposed framework and associated evidence suggests a path forward for treatment targets for anxiety. Neuromodulation has to date been less successful for treatment of anxiety disorders than it has been for depression98–101. The current approach highlights the nuances involved with cognition-emotion interactions across the two hemispheres; understanding these nuances is especially important for informing both mechanistic and treatment studies. As highlighted in the introduction, however, important differences may emerge across different disorders and across different individuals, requiring a precision medicine approach.
Limitations
Although emerging evidence is consistent with the proposed framework, it should be noted that this explanation for our conflicting findings is post hoc and should be replicated. This is particularly true for the neuroimaging data presented above. While hemisphere-specific neuroimaging findings support the proposed framework, imaging studies often lack the power to determine whether a lack of dlPFC responding in one hemisphere represents a true null result. It is therefore critical that causal methods like TMS continue to be employed to understand how the left and right dlPFC contribute to emotion regulation. Care must also be taken when interpreting the results of TMS studies as well. For instance, although there is evidence in motor cortex for the excitatory/inhibitory rTMS patterns described above86–92, we understand that it is premature to generalize these patterns to prefrontal cortex. It will be critical to expand these findings to other patterns of stimulation like low-frequency stimulation and iTBS, delivered during both online and offline sessions. Finally, future work should also test whether explicit positive mood induction moderates the effect of right dlPFC rTMS on anxiety. Indeed, contextual modulation of rTMS delivery is an active area of research aimed at improving the precision of these neuromodulatory techniques108.
Conclusions
Here we summarize literature showing that the dlPFC contributes to both working memory processes and emotion regulation. Our primary interest is understanding how these two processes relate in the context of anxiety. Work showing that cognitive and affective processes interact, suggest that they are part of the same underlying process in the dlPFC. However, work showing that the left and right dlPFC can have opposing effects on affective tone suggest that affect can be modulated up or down, independent of ongoing cognitive processes. Until now, there was not a framework to explain these two discrepant findings. By reframing emotion regulation in terms of the type of working memory process employed (maintenance vs suppression), the proposed framework can account the discrepant dlPFC findings. Additionally, the proposed framework offers a mechanistic understanding for how dlPFC neuromodulation affects mood and anxiety and has implications for how to improve and personalize neuromodulatory treatments.
Highlights.
We propose a model relating dlPFC-mediated emotion regulation to working memory.
We argue that working memory is the fundamental process executed by the dlPFC.
We argue that only the content domain differs across the left and right dlPFC.
Left dlPFC can maintain or suppress verbal content in working memory.
Right dlPFC can maintain or suppress non-verbal content in working memory.
Acknowledgments:
We would like to thank the following individuals who contributed to Dr. Balderston’s K01 project: Dr. Kerry Ressler, Dr. Michael Thase, and Dr. Kristin Linn. The authors would like to thank Maria Prociuk for her expertise and assistance in submitting the paper.
Financial Support:
This project was supported in part by a NARSAD Young Investigator Grants from the Brain & Behavior Research Foundation (NLB: 2021), and a career development grant from the National Institutes of Health: K01MH121777 (NLB).
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Author contributions: CRediT author statement according to: https://www.elsevier.com/authors/policies-and-guidelines/credit-author-statement.
Conceptualization: LKW, NLB; Writing - Original Draft: LKW, MT, YS, NLB; Writing - Review & Editing: LKW, MT, WM, YS, NLB; Visualization: MT, NLB; Supervision: YS, NLB; Funding acquisition: NLB.
Conflicts of Interest: The authors report no biomedical financial interests or potential conflicts of interest.
References
- 1.Kessler RC, Chiu WT. Prevalence, severity, and comorbidity of 12-month DSM-IV disorders in the National Comorbidity Survey Replication. Arch Gen … 2005; 62: 617–627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Penninx BWJH, Pine DS, Holmes EA, Reif A. Anxiety disorders. Lancet 2021; 397: 914–927. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.White LK, Sequeira S, Britton JC, Brotman MA, Gold AL, Berman E et al. Complementary Features of Attention Bias Modification Therapy and Cognitive-Behavioral Therapy in Pediatric Anxiety Disorders. Am J Psychiatry 2017; 174: 775–784. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Pagliaccio D, Luby JL, Bogdan R, Agrawal A, Gaffrey MS, Belden AC et al. Amygdala functional connectivity, HPA axis genetic variation, and life stress in children and relations to anxiety and emotion regulation. J. Abnorm. Psychol 2015; 124: 817–833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Van der Plas M, Braun V, Stauch BJ, Hanslmayr S. Stimulation of the left dorsolateral prefrontal cortex with slow rTMS enhances verbal memory formation. PLoS Biol 2021; 19. doi: 10.1371/journal.pbio.3001363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Diamond A Executive Functions. Annu Rev Psychol 2013; 64: 135–168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Mancuso LE, Ilieva IP, Hamilton RH, Farah MJ. Does transcranial direct current stimulation improve healthy working memory?: A meta-analytic review. J. Cogn. Neurosci 2016; 28: 1063–1089. [DOI] [PubMed] [Google Scholar]
- 8.Brevet-Aeby C, Brunelin J, Iceta S, Padovan C, Poulet E. Prefrontal cortex and impulsivity: Interest of noninvasive brain stimulation. Neurosci. Biobehav. Rev 2016; 71: 112–134. [DOI] [PubMed] [Google Scholar]
- 9.Diamond A. Abilities and neural mechanisms underlying AB performance. Child Dev 1988; 59: 523–527. [PubMed] [Google Scholar]
- 10.Goldman-Rakic PS. Topography of Cognition: Parallel Distributed Networks in Primate Association Cortex. Annu Rev Neurosci 1988; 11: 137–156. [DOI] [PubMed] [Google Scholar]
- 11.Stuss DT, Benson DF. Neuropsychological studies of the frontal lobes. Psychol. Bull 1984; 95: 3–28. [PubMed] [Google Scholar]
- 12.Wager TD, Smith EE. Neuroimaging studies of working memory: a meta-analysis. Cogn Affect Behav Neurosci 2003; 3: 255–274. [DOI] [PubMed] [Google Scholar]
- 13.Balderston NL, Vytal KE, O’Connell K, Torrisi S, Letkiewicz A, Ernst M et al. Anxiety Patients Show Reduced Working Memory Related dlPFC Activation During Safety and Threat. Depress Anxiety 2017; 34: 25–36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Balderston NL, Flook E, Hsiung A, Liu J, Thongarong A, Stahl S et al. Patients with anxiety disorders rely on bilateral dlPFC activation during verbal working memory. Soc. Cogn. Affect. Neurosci 2020; 15: 1288–1298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Barbey AK, Koenigs M, Grafman J. Dorsolateral prefrontal contributions to human working memory. Cortex 2013; 49: 1195–1205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Cole MW, Schneider W. The cognitive control network: Integrated cortical regions with dissociable functions. Neuroimage 2007; 37: 343–360. [DOI] [PubMed] [Google Scholar]
- 17.Harding IH, Yücel M, Harrison BJ, Pantelis C, Breakspear M. Effective connectivity within the frontoparietal control network differentiates cognitive control and working memory. Neuroimage 2015; 106: 144–153. [DOI] [PubMed] [Google Scholar]
- 18.Sakai K, Rowe JB, Passingham RE. Active maintenance in prefrontal area 46 creates distractor-resistant memory. Nat. Neurosci 2002; 5: 479–484. [DOI] [PubMed] [Google Scholar]
- 19.Nagel BJ, Herting MM, Maxwell EC, Bruno R, Fair D. Hemispheric lateralization of verbal and spatial working memory during adolescence. Brain Cogn 2013; 82: 58–68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Sandrini M, Rossini PM, Miniussi C. Lateralized contribution of prefrontal cortex in controlling task-irrelevant information during verbal and spatial working memory tasks: rTMS evidence. Neuropsychologia 2008; 46: 2056–2063. [DOI] [PubMed] [Google Scholar]
- 21.Ries SK, Greenhouse I, Dronkers NF, Haaland KY, Knight RT. Double dissociation of the roles of the left and right prefrontal cortices in anticipatory regulation of action. Neuropsychologia 2014; 63: 215–225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Etkin A, Büchel C, Gross JJ. The neural bases of emotion regulation. Nat. Rev. Neurosci 2015; 16: 693–700. [DOI] [PubMed] [Google Scholar]
- 23.Gray JR, Braver TS, Raichle ME. Integration of emotion and cognition in the lateral prefrontal cortex. Proc Natl Acad Sci U S A 2002; 99: 4115–4120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Liberzon I, Taylor SF, Fig LM, Decker LR, Koeppe RA, Minoshima S. Limbic Activation and Psychophysiologic Responses to Aversive Visual Stimuli: Interaction with Cognitive Task. Neuropsychopharmacology 2000; 23: 508–516. [DOI] [PubMed] [Google Scholar]
- 25.Beauregard M, Lévesque J, Bourgouin P. Neural Correlates of Conscious Self-Regulation of Emotion. J Neurosci 2001; 21: RC165 LP-RC165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Kroes MCW, Dunsmoor JE, Hakimi M, Oosterwaal S, Meager MR, Phelps EA. Patients with dorsolateral prefrontal cortex lesions are capable of discriminatory threat learning but appear impaired in cognitive regulation of subjective fear. Soc. Cogn. Affect. Neurosci 2019; 14: 601–612. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Feeser M, Prehn K, Kazzer P, Mungee A, Bajbouj M. Transcranial direct current stimulation enhances cognitive control during emotion regulation. Brain Stimul 2014; 7: 105–112. [DOI] [PubMed] [Google Scholar]
- 28.Drevets WC, Raichle ME. Neuroanatomical circuits in depression: implications for treatment mechanisms. Psychopharmacol Bull 1992. [PubMed] [Google Scholar]
- 29.GD A, Erbil A, SG L, RM E. Medial prefrontal cortex and self-referential mental activity: Relation to a default mode of brain function. Proc Natl Acad Sci 2001; 98: 4259–4264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Simpson JR, Öngür D, Akbudak E, Conturo TE, Ollinger JM, Snyder AZ et al. The Emotional Modulation of Cognitive Processing: An fMRI Study. J Cogn Neurosci 2000; 12: 157–170. [DOI] [PubMed] [Google Scholar]
- 31.Mayberg HS, Liotti M, Brannan SK, McGinnis S, Mahurin RK, Jerabek PA et al. Reciprocal Limbic-Cortical Function and Negative Mood: Converging PET Findings in Depression and Normal Sadness. Am J Psychiatry 1999; 156: 675–682. [DOI] [PubMed] [Google Scholar]
- 32.Damasio AR, Grabowski TJ, Bechara A, Damasio H, Ponto LLB, Parvizi J et al. Subcortical and cortical brain activity during the feeling of self-generated emotions. Nat Neurosci 2000; 3: 1049–1056. [DOI] [PubMed] [Google Scholar]
- 33.Davidson RJ, Irwin W. The functional neuroanatomy of emotion and affective style. Trends Cogn Sci 1999; 3: 11–21. [DOI] [PubMed] [Google Scholar]
- 34.Hartling C, Metz S, Pehrs C, Scheidegger M, Gruzman R, Keicher C et al. Comparison of four FMRI paradigms probing emotion processing. Brain Sci 2021; 11. doi: 10.3390/brainsci11050525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Adamczyk AK, Wyczesany M, van Peer JM. High working memory load impairs reappraisal but facilitates distraction – An event-related potential investigation. Biol. Psychol 2022; 171. doi: 10.1016/j.biopsycho.2022.108327. [DOI] [PubMed] [Google Scholar]
- 36.Bishop SJ. Neural mechanisms underlying selective attention to threat. Ann. N. Y. Acad. Sci 2008; 1129: 141–152. [DOI] [PubMed] [Google Scholar]
- 37.Ironside M, Browning M, Ansari TL, Harvey CJ, Sekyi-Djan MN, Bishop SJ et al. Effect of Prefrontal Cortex Stimulation on Regulation of Amygdala Response to Threat in Individuals With Trait Anxiety A Randomized Clinical Trial Supplemental content. JAMA Psychiatry 2018; 02478: 1–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Loos E, Schicktanz N, Fastenrath M, Coynel D, Milnik A, Fehlmann B et al. Reducing Amygdala activity and phobic fear through cognitive top-down regulation. J. Cogn. Neurosci 2020; 32: 1117–1129. [DOI] [PubMed] [Google Scholar]
- 39.Bishop SJ, Duncan J, Brett M, Lawrence AD. Prefrontal cortical function and anxiety: Controlling attention to threat-related stimuli. Nat Neurosci 2004; 7: 184–188. [DOI] [PubMed] [Google Scholar]
- 40.Fu X, Taber-Thomas BC, Pérez-Edgar K. Frontolimbic functioning during threat-related attention: Relations to early behavioral inhibition and anxiety in children. Biol. Psychol 2017; 122: 98–109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Balderston NL, Hsiung A, Ernst M, Grillon C. Effect of threat on right dlPFC activity during behavioral pattern separation 2017. [DOI] [PMC free article] [PubMed]
- 42.Mitchell DGV, Nakic M, Fridberg D, Kamel N, Pine DS, Blair RJR. The impact of processing load on emotion. Neuroimage 2007; 34: 1299–1309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Banich MT, Smolker HR, Snyder HR, Lewis-Peacock JA, Godinez DA, Wager TD et al. Turning down the heat: Neural mechanisms of cognitive control for inhibiting task-irrelevant emotional information during adolescence. Neuropsychologia 2019; 125: 93–108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Morawetz C, Riedel MC, Salo T, Berboth S, Eickhoff SB, Laird AR et al. Multiple largescale neural networks underlying emotion regulation. Neurosci. Biobehav. Rev 2020; 116: 382–395. [DOI] [PubMed] [Google Scholar]
- 45.Golkar A, Lonsdorf TB, Olsson A, Lindstrom KM, Berrebi J, Fransson P et al. Distinct Contributions of the Dorsolateral Prefrontal and Orbitofrontal Cortex during Emotion Regulation. PLoS One 2012; 7. doi: 10.1371/journal.pone.0048107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Balderston NL, Liu J, Roberson-Nay R, Ernst M, Grillon C. The relationship between dlPFC activity during unpredictable threat and CO2-induced panic symptoms. Transl Psychiatry 2017; 7: 1266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Balderston NL, Hsiung A, Liu J, Ernst M, Grillon C. Reducing state anxiety using working memory maintenance. J Vis Exp 2017. doi: 10.3791/55727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Balderston NL, Quispe-Escudero D, Hale E, Davis A, O’Connell K, Ernst M et al. Working memory maintenance is sufficient to reduce state anxiety. Psychophysiology 2016. doi: 10.1111/psyp.12726. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Vytal KE, Cornwell BR, Letkiewicz AM, Arkin NE, Grillon C. The complex interaction between anxiety and cognition: insight from spatial and verbal working memory. Front Hum Neurosci 2013; 7: 93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Vytal KE, Cornwell BR, Arkin N, Grillon C. Describing the interplay between anxiety and cognition: From impaired performance under low cognitive load to reduced anxiety under high load. Psychophysiology 2012; 49: 842–852. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.White SF, Costanzo ME, Blair JR, Roy MJ. PTSD symptom severity is associated with increased recruitment of top-down attentional control in a trauma-exposed sample. NeuroImage Clin 2015; 7: 19–27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Fonzo GA, Goodkind MS, Oathes DJ, Zaiko YV, Harvey M, Peng KK et al. PTSD psychotherapy outcome predicted by brain activation during emotional reactivity and regulation. Am J Psychiatry 2017; 174: 1163–1174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Moon CM, Jeong GW. Functional neuroanatomy on the working memory under emotional distraction in patients with generalized anxiety disorder. Psychiatry Clin Neurosci 2015; 69: 609–619. [DOI] [PubMed] [Google Scholar]
- 54.Buff C, Brinkmann L, Neumeister P, Feldker K, Heitmann C, Gathmann B et al. Specifically altered brain responses to threat in generalized anxiety disorder relative to social anxiety disorder and panic disorder. NeuroImage Clin 2016; 12: 698–706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Vasic N, Walter H, Sambataro F, Wolf RC. Aberrant functional connectivity of dorsolateral prefrontal and cingulate networks in patients with major depression during working memory processing. Psychol. Med 2009; 39: 977–987. [DOI] [PubMed] [Google Scholar]
- 56.Fernández-Corcuera P, Salvador R, Monté GC, Salvador Sarró S, Goikolea JM, Amann B et al. Bipolar depressed patients show both failure to activate and failure to de-activate during performance of a working memory task. J. Affect. Disord 2013; 148: 170–178. [DOI] [PubMed] [Google Scholar]
- 57.Basten U, Stelzel C, Fiebach CJ. Trait anxiety and the neural efficiency of manipulation in working memory. Cogn Affect Behav Neurosci 2012; 12: 571–588. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Fales CL, Barch DM, Burgess GC, Schaefer A, Mennin DS, Gray JR et al. Anxiety and cognitive efficiency: Differential modulation of transient and sustained neural activity during a working memory task. Cogn Affect Behav Neurosci 2008; 8: 239–253. [DOI] [PubMed] [Google Scholar]
- 59.Moon CM, Sundaram T, Choi NG, Jeong GW. Working memory dysfunction associated with brain functional deficits and cellular metabolic changes in patients with generalized anxiety disorder. Psychiatry Res - Neuroimaging 2016; 254: 137–144. [DOI] [PubMed] [Google Scholar]
- 60.Moon CM, Jeong GW. Functional and morphological alterations associated with working memory dysfunction in patients with generalized anxiety disorder. Acta radiol 2017; 58: 344–352. [DOI] [PubMed] [Google Scholar]
- 61.Balderston NL, Vytal KE, O’Connell K, Torrisi S, Letkiewicz A, Ernst M et al. Anxiety Patients Show Reduced Working Memory Related Dlpfc Activation During Safety and Threat. Depress Anxiety 2016; 12: 1–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.White LK, Moore TM, Calkins ME, Wolf DH, Satterthwaite TD, Leibenluft E et al. An Evaluation of the Specificity of Executive Function Impairment in Developmental Psychopathology. J Am Acad Child Adolesc Psychiatry 2017; 56: 975–982.e3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Duval ER, Sheynin J, King AP, Phan KL, Simon NM, Martis B et al. Neural function during emotion processing and modulation associated with treatment response in a randomized clinical trial for posttraumatic stress disorder. Depress Anxiety 2020; 37: 670–681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.King AP, Block SR, Sripada RK, Rauch S, Giardino N, Favorite T et al. Altered default mode network (DMN) resting state functional connectivity following a mindfulness-based exposure therapy for posttraumatic stress disorder (PTSD) in combat veterans of Afghanistan and Iraq. Depress. Anxiety 2016; 33: 289–299. [DOI] [PubMed] [Google Scholar]
- 65.Zilverstand A, Sorger B, Sarkheil P, Goebel R. fMRI neurofeedback facilitates anxiety regulation in females with spider phobia. Front. Behav. Neurosci 2015; 9. doi: 10.3389/fnbeh.2015.00148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Luo Y, Fernández G, Hermans E, Vogel S, Zhang Y, Li H et al. How acute stress may enhance subsequent memory for threat stimuli outside the focus of attention: DLPFC-amygdala decoupling. Neuroimage 2018; 171: 311–322. [DOI] [PubMed] [Google Scholar]
- 67.Chen F, Ke J, Qi R, Xu Q, Zhong Y, Liu T et al. Increased Inhibition of the Amygdala by the mPFC may Reflect a Resilience Factor in Post-traumatic Stress Disorder: A Resting-State fMRI Granger Causality Analysis. Front. Psychiatry 2018; 9. doi: 10.3389/fpsyt.2018.00516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Davidson RJ, Chapman JP, Chapman LJ, Henriques JB. Asymmetrical brain electrical activity discriminates between psychometrically-matched verbal and spatial cognitive tasks. Psychophysiology 1990; 27: 528–543. [DOI] [PubMed] [Google Scholar]
- 69.van der Vinne N, Vollebregt MA, van Putten MJAM, Arns M. Frontal alpha asymmetry as a diagnostic marker in depression: Fact or fiction? A meta-analysis. NeuroImage Clin 2017; 16: 79–87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Jesulola E, Sharpley CF, Agnew LL. The effects of gender and depression severity on the association between alpha asymmetry and depression across four brain regions. Behav Brain Res 2017; 321: 232–239. [DOI] [PubMed] [Google Scholar]
- 71.Coan JA, Allen JJB. Frontal EEG asymmetry as a moderator and mediator of emotion. Biol Psychol 2004; 67: 7–50. [DOI] [PubMed] [Google Scholar]
- 72.Klimesch W, Sauseng P, Hanslmayr S. EEG alpha oscillations: The inhibition-timing hypothesis. Brain Res. Rev 2007. doi: 10.1016/j.brainresrev.2006.06.003. [DOI] [PubMed] [Google Scholar]
- 73.Reznik SJ, Allen JJB. Frontal asymmetry as a mediator and moderator of emotion: An updated review. Psychophysiology 2018; 55. doi: 10.1111/psyp.12965. [DOI] [PubMed] [Google Scholar]
- 74.Coan JA, Allen JJB. Frontal EEG asymmetry and the behavioral activation and inhibition systems. Psychophysiology 2003; 40: 106–114. [DOI] [PubMed] [Google Scholar]
- 75.Pizzagalli DA, Sherwood RJ, Henriques JB, Davidson RJ. Frontal brain asymmetry and reward responsiveness: A source-localization study. Psychol. Sci 2005; 16: 805–813. [DOI] [PubMed] [Google Scholar]
- 76.Sutton SK, Davidson RJ. Prefrontal brain electrical asymmetry predicts the evaluation of affective stimuli. Neuropsychologia 2000; 38: 1723–1733. [DOI] [PubMed] [Google Scholar]
- 77.Balconi M, Mazza G. Lateralisation effect in comprehension of emotional facial expression: A comparison between EEG alpha band power and behavioural inhibition (BIS) and activation (BAS) systems. Laterality 2010; 15: 361–384. [DOI] [PubMed] [Google Scholar]
- 78.Debener S, Beauducel A, Nessler D, Brocke B, Heilemann H, Kayser J. Is resting anterior EEG alpha asymmetry a trait marker for depression? Findings for healthy adults and clinically depressed patients. Neuropsychobiology 2000; 41: 31–37. [DOI] [PubMed] [Google Scholar]
- 79.Mathersul D, Williams LM, Hopkinson PJ, Kemp AH. Investigating Models of Affect: Relationships Among EEG Alpha Asymmetry, Depression, and Anxiety. Emotion 2008; 8: 560–572. [DOI] [PubMed] [Google Scholar]
- 80.Kemp AH, Griffiths K, Felmingham KL, Shankman SA, Drinkenburg W, Arns M et al. Disorder specificity despite comorbidity: Resting EEG alpha asymmetry in major depressive disorder and post-traumatic stress disorder. Biol. Psychol 2010; 85: 350–354. [DOI] [PubMed] [Google Scholar]
- 81.Meyer T, Smeets T, Giesbrecht T, Quaedflieg CWEM, Smulders FTY, Meijer EH et al. The role of frontal EEG asymmetry in post-traumatic stress disorder. Biol. Psychol 2015; 108: 62–77. [DOI] [PubMed] [Google Scholar]
- 82.Metzger LJ, Carson MA, Paulus LA, Paige SR, Lasko NB, Pitman RK et al. PTSD Arousal and Depression Symptoms Associated with Increased Right-Sided Parietal EEG Asymmetry. J. Abnorm. Psychol 2004; 113: 324–329. [DOI] [PubMed] [Google Scholar]
- 83.Grimm S, Beck J, Schuepbach D, Hell D, Boesiger P, Bermpohl F et al. Imbalance between Left and Right Dorsolateral Prefrontal Cortex in Major Depression Is Linked to Negative Emotional Judgment: An fMRI Study in Severe Major Depressive Disorder. Biol Psychiatry 2008; 63: 369–376. [DOI] [PubMed] [Google Scholar]
- 84.Hecht D. Depression and the hyperactive right-hemisphere. Neurosci Res 2010; 68: 77–87. [DOI] [PubMed] [Google Scholar]
- 85.Bajwa S, Bermpohl F, Rigonatti SP, Pascual-Leone A, Boggio PS, Fregni F. Impaired Interhemispheric Interactions in Patients With Major Depression. J Nerv Ment Dis 2008; 196. https://journals.lww.com/jonmd/Fulltext/2008/09000/Impaired_Interhemispheric_Interactions_in_Patients.3.aspx. [DOI] [PubMed] [Google Scholar]
- 86.Di Lazzaro V, Dileone M, Pilato F, Capone F, Musumeci G, Ranieri F et al. Modulation of motor cortex neuronal networks by rTMS: comparison of local and remote effects of six different protocols of stimulation. J Neurophysiol 2011; 105: 2150–2156. [DOI] [PubMed] [Google Scholar]
- 87.Huang YZ, Rothwell JC, Chen RS, Lu CS, Chuang WL. The theoretical model of theta burst form of repetitive transcranial magnetic stimulation. Clin Neurophysiol 2011; 122: 1011–1018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Huang Y-ZZ, Edwards MJ, Rounis E, Bhatia KP, Rothwell JC. Theta burst stimulation of the human motor cortex. Neuron 2005; 45: 201–206. [DOI] [PubMed] [Google Scholar]
- 89.Suppa A, Huang YZ, Funke K, Ridding MC, Cheeran B, Di Lazzaro V et al. Ten Years of Theta Burst Stimulation in Humans: Established Knowledge, Unknowns and Prospects. Brain Stimul 2016; 9: 323–335. [DOI] [PubMed] [Google Scholar]
- 90.Wischnewski M, Schutter DJLG. Efficacy and time course of theta burst stimulation in healthy humans. Brain Stimul 2015; 8: 685–692. [DOI] [PubMed] [Google Scholar]
- 91.Goldsworthy MR, Müller-Dahlhaus F, Ridding MC, Ziemann U. Inter-subject variability of LTD-like plasticity in human motor cortex: A matter of preceding motor activation. Brain Stimul 2014; 7: 864–870. [DOI] [PubMed] [Google Scholar]
- 92.Huang YZ, Rothwell JC, Lu CS, Wang JJ, Weng YH, Lai SC et al. The effect of continuous theta burst stimulation over premotor cortex on circuits in primary motor cortex and spinal cord. Clin Neurophysiol 2009; 120: 796–801. [DOI] [PubMed] [Google Scholar]
- 93.Trevizol AP, Blumberger DM. An Update on Repetitive Transcranial Magnetic Stimulation for the Treatment of Major Depressive Disorder. Clin Pharmacol Ther 2019; 106: 747–762. [DOI] [PubMed] [Google Scholar]
- 94.Brunoni AR, Chaimani A, Moffa AH, Razza LB, Gattaz WF, Daskalakis ZJ et al. Repetitive transcranial magnetic stimulation for the acute treatment of major depressive episodes a systematic review with network meta-analysis. JAMA Psychiatry 2017; 74: 143–152. [DOI] [PubMed] [Google Scholar]
- 95.O’Reardon JP, Solvason HB, Janicak PG, Sampson S, Isenberg KE, Nahas Z et al. Efficacy and Safety of Transcranial Magnetic Stimulation in the Acute Treatment of Major Depression: A Multisite Randomized Controlled Trial. Biol Psychiatry 2007; 62: 1208–1216. [DOI] [PubMed] [Google Scholar]
- 96.Mantovani A, Aly M, Dagan Y, Allart A, Lisanby SH. Randomized sham controlled trial of repetitive transcranial magnetic stimulation to the dorsolateral prefrontal cortex for the treatment of panic disorder with comorbid major depression. J Affect Disord 2013; 144: 153–159. [DOI] [PubMed] [Google Scholar]
- 97.White D, Tavakoli S. Repetitive transcranial magnetic stimulation for treatment of major depressive disorder with comorbid generalized anxiety disorder. Ann Clin Psychiatry 2015; 27: 192–196. [PubMed] [Google Scholar]
- 98.Pallanti S, Bernardi S. Neurobiology of repeated transcranial magnetic stimulation in the treatment of anxiety: a critical review. Int Clin Psychopharmacol 2009; 24: 163–173. [DOI] [PubMed] [Google Scholar]
- 99.Zwanzger P, Fallgatter AJ, Zavorotnyy M, Padberg F. Anxiolytic effects of transcranial magnetic stimulation-an alternative treatment option in anxiety disorders? J Neural Transm 2009; 116: 767–775. [DOI] [PubMed] [Google Scholar]
- 100.Lefaucheur JP, Andre-Obadia N, Antal A, Ayache SS, Baeken C, Benninger DH et al. Evidence-based guidelines on the therapeutic use of repetitive transcranial magnetic stimulation (rTMS). Clin Neurophysiol 2014; 125: 2150–2206. [DOI] [PubMed] [Google Scholar]
- 101.Pigot M, Loo C, Sachdev P. Repetitive transcranial magnetic stimulation as treatment for anxiety disorders. Expert Rev Neurother 2008; 8: 1449–1455. [DOI] [PubMed] [Google Scholar]
- 102.Owen AM, McMillan KM, Laird AR, Bullmore E. N-back working memory paradigm: A meta-analysis of normative functional neuroimaging studies. Hum Brain Mapp 2005; 25: 46–59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Robinson O, Vytal KE, Cornwell BR, Grillon C. The impact of anxiety upon cognition: perspectives from human threat of shock studies. Front Hum Neurosci 2013; 7: 203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Anderson MC, Bunce JG, Barbas H. Prefrontal–hippocampal pathways underlying inhibitory control over memory. Neurobiol. Learn. Mem 2016; 134: 145–161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Baddeley AD. Working memory. Science (80-) 1992; 255: 556–559. [DOI] [PubMed] [Google Scholar]
- 106.Balderston NL, Beydler EMEM, Roberts C, De Deng Z-D, Radman T, Lago T et al. Mechanistic link between right prefrontal cortical activity and anxious arousal revealed using transcranial magnetic stimulation in healthy subjects. Neuropsychopharmacology 2020; 45: 694–702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Teferi M, Makhoul W, Deng Z-D, Oathes DJ, Sheline Y, Balderston NL. Continuous Theta Burst Stimulation to the Right Dorsolateral Prefrontal Cortex may increase Potentiated Startle in healthy individuals. Biol Psychiatry Glob Open Sci 2022. doi: 10.1016/j.bpsgos.2022.04.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Deng Z-D, Luber BM, Balderston NL, Velez Afanador M, Noh MMM, Thomas J et al. Device-Based Modulation of Neurocircuits as a Therapeutic for Psychiatric Disorders. Annu Rev Pharmacol Toxicol 2020; 60: 591–614. [DOI] [PMC free article] [PubMed] [Google Scholar]


