Abstract
Sex and racial disparities in the presentation, diagnosis, and management of cardiac arrhythmias are recognized. Sex-specific differences in electrophysiological parameters are well known and are predominantly related to differences in ion channel expression and the influence of sex hormones. However, the relationship between hormonal or racial influence and arrhythmia mechanisms, presentation, and management needs to be better defined. Women and racial and ethnic groups are less likely to undergo catheter ablation procedures for treatment of cardiac arrhythmias. Underrepresentation of women and racial/ethnic groups in clinical trials has resulted in significant knowledge gaps. Whether sex and racial disparities in arrhythmia management reflect barriers in access to care, physician bias, patient values, and preferences or other factors requires further study.
Keywords: Atrial fibrillation, Catheter ablation, Ethnicity, Race, Sex differences, Supraventricular tachycardia, Ventricular arrhythmia
Key Findings.
-
▪
Sex differences in cardiac electrophysiology are predominantly related to differences in ion channel expression and the influence of sex hormones, but racial differences are less well studied.
-
▪
Studies assessing the clinical presentation, diagnosis, and management of supraventricular tachycardia (SVT) show that women present with symptoms at a younger age, but there are sex-specific disparities in management because of delays in SVT diagnosis, provider preference for medications, and catheter ablation. The delay in catheter ablation results in higher health care utilization by women.
-
▪
Sex and race differences in atrial fibrillation (AF) treatment continue to exist. Women and Black patients are less likely to undergo electrical cardioversion or catheter ablation despite experiencing more symptoms and impaired quality of life attributed to AF.
-
▪
Females and diverse racial populations are underrepresented in clinical trials on ventricular tachycardia (VT) ablation. Referral bias and worse outcomes have been reported for these patients undergoing VT ablation in the setting of structural heart disease.
-
▪
Sex-specific and racial/ethnically representative studies are required to address the knowledge gaps, identify the major factors that influence referral/preference for ablation therapy, and improve ablation outcomes.
Introduction
In cardiac electrophysiology, variations in the epidemiology and presentation of arrhythmias have been associated with sex and race, which may have clinical and therapeutic implications. Despite efforts from the National Institutes of Health and the United States Food and Drug Administration to increase inclusion of women and racial/ethnic representation in clinical trials, these groups remain largely underrepresented in cardiovascular clinical trials.1, 2, 3 Exclusion of these populations has resulted in knowledge gaps, which hamper optimal management strategies. Although sex-specific differences in electrophysiological parameters are well known, the sex and racial differences that influence arrhythmia management, including ablation therapy, require further attention.4
Sex differences in cardiac electrophysiology
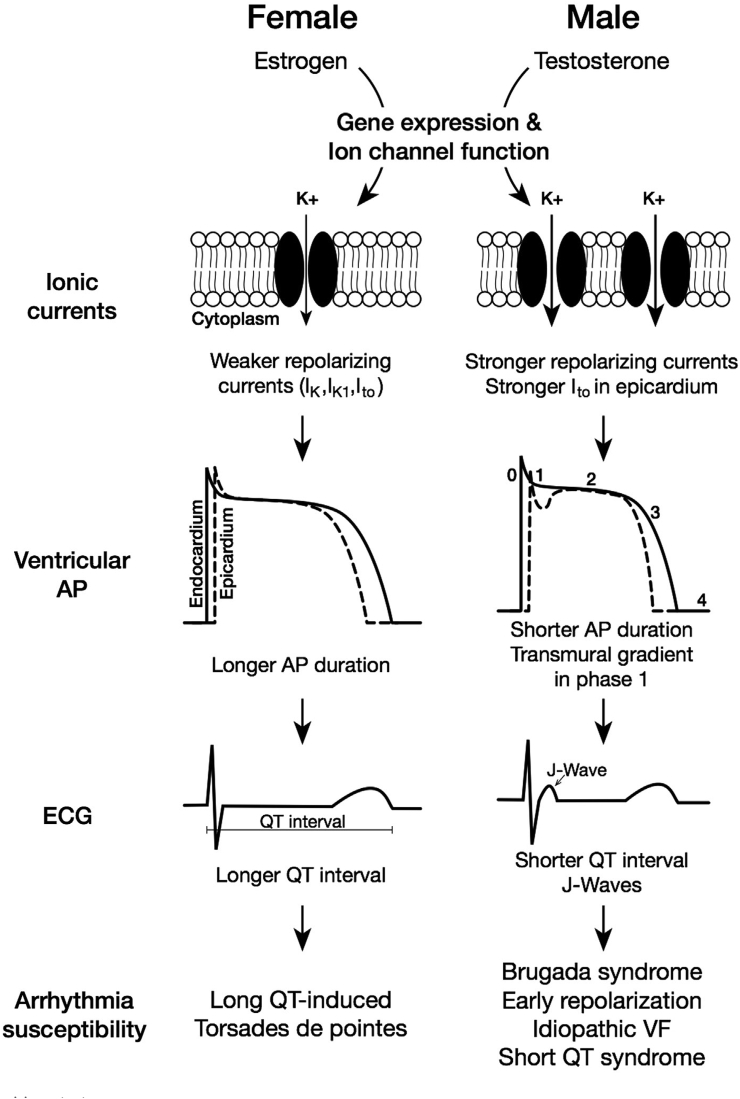
Sex differences in fundamental electrophysiology parameters are well described (Table 1). These differences are predominantly related to sex differences in ion channel expression and the influence of sex hormones.5 Sex hormones have variable effects on the up- or downregulation of ion channels and their function (Figure 1). Reported sex differences in human atrial electrophysiology are inconsistent, but human ventricular myocytes have been studied extensively.6 In female ventricular myocytes, the expression of variable potassium channel subunits is reduced compared to male ventricular myocytes, resulting in longer ventricular action potential durations, ventricular refractory periods, and QT intervals.6 Estrogen reduces IKr channel expression and hence reduces the rapid delayed rectifier current, whereas testosterone and progesterone shorten the ventricular action potential duration by enhancing IKs expression and increase the slow delayed potassium rectifier current.6,7 Testosterone has also been associated with stronger Ito currents in male ventricular epicardium, which correlates to the sex difference in J-wave syndromes, such as early repolarization.6 Testosterone has also been reported to decrease the sodium/calcium exchange current, as well as to reduce the calcium release mediated by ryanodine receptors.8,9 Because estrogen has the opposing effect, women may have a predisposition to triggered activity.8,9 Therefore, sex differences in arrhythmogenesis may be explained, in part, by differential influences of the sex hormones. For example, the longer ventricular repolarization together with the increased likelihood of triggered activity may explain the higher incidence of torsade de pointes in female patients with congenital or acquired long QT syndrome.6 Sex differences in autonomic function have also been described, which may explain the observed differences in baseline heart rate, PR interval, and inconsistent results for the AH interval.10
Table 1.
Sex differences in electrophysiological parameters
| Variable | Women vs Men |
|---|---|
| Action potential | |
| Atrial APD | No difference |
| Ventricular APD | Longer |
| INa | No difference |
| Ito | Decreased |
| IKr | Decreased |
| IKs | Decreased |
| IK1 | Decreased |
| ICa,L | Increased |
| ECG | |
| Resting heart rate | Higher |
| PR interval | Shorter |
| QRS duration | Shorter |
| Corrected QT interval | Longer |
| Invasive EP | |
| Sinus node recovery time | Shorter |
| AH interval | Equal or shorter |
| HV interval | Shorter |
| Atrial ERP | Shorter |
| AV nodal ERP | Shorter |
| Ventricular ERP | Longer |
APD = action potential duration; AV = atrioventricular; ECG = electrocardiography; EP = electrophysiology; ERP = effective refractory period.
Figure 1.
Sex differences in ventricular cardiomyocyte electrophysiology. AP = action potential; ECG = electrocardiogram; IK = delayed rectifier potassium current; IK1 = inward rectifier potassium current; Ito = transient outward current; VF = ventricular fibrillation. (Reproduced from Tadros et al6 with permission.)
Data on sex differences in atrial electrophysiology and mechanisms of atrial fibrillation (AF) are limited. Sex differences in the electrophysiology of the left atrium and pulmonary veins observed in mice might explain an increased risk of arrhythmogenesis in males.4 Compared to females, a faster spontaneous beating rate, increased burst firing, and isoproterenol-induced triggering of delayed afterdepolarizations were observed in male pulmonary veins. In addition, delayed afterdepolarizations were more frequently observed in male left atrial myocytes than in female myocytes. These differences were associated with an increase in the late sodium current, calcium transients, and sarcoplasmic reticulum calcium content. Women have been reported to have a higher incidence of nonpulmonary vein triggers for AF compared with men. Whether this finding is due to differences in atrial electrical and structural remodeling between the sexes is uncertain.
Racial differences in cardiac electrophysiology
Studies reporting racial or ethnic differences in electrophysiology variables are rare. One study reported that South Asian patients had a significantly higher resting heart rate and shorter QRS duration compared to a White control population.11 The atrioventricular (AV) nodal effective refractory period was also significantly shorter in these South Asian patients compared to controls.11 Despite significant racial differences in AF, data on differences in AF mechanisms are lacking.
Supraventricular tachycardias
Sex differences in supraventricular tachycardia diagnosis and management
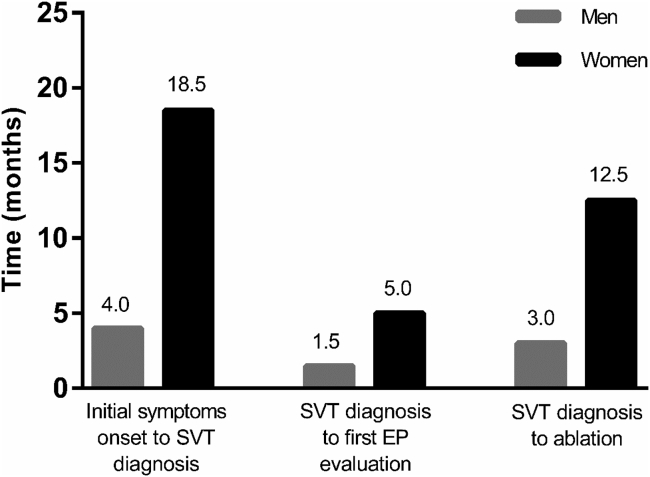
Women typically are younger at the onset of symptoms for both atrioventricular nodal reentrant tachycardia (AVNRT) and atrioventricular reentrant tachycardia AVRT.12, 13, 14 Women are more symptomatic with supraventricular tachycardia (SVT) and are more often treated with antiarrhythmic drugs.14, 15, 16 The interval between onset of symptoms and first electrophysiological evaluation is longer in women compared to men, as is the time interval between this first evaluation and catheter ablation (Figure 2).12,14 Men were more likely to undergo catheter ablation for SVT compared to women.17 In a previous study, the delay in presentation and ablation of SVT in women was found to be driven by several factors, including a delay in SVT diagnosis, a personal preference of women for medications over the potential risk associated with catheter ablation, and the major responsibilities of women as the primary caregiver within their family.14
Figure 2.
Sex differences in supraventricular tachycardia (SVT) management. EP = electrophysiology. (Reproduced from Musa et al14 with permission.)
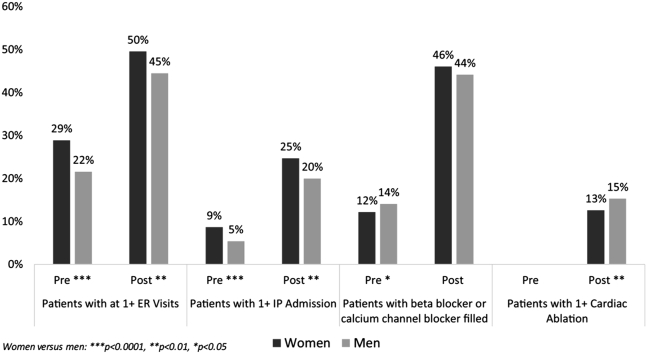
Similar findings were reported in a contemporary cohort of 5466 patients with a new diagnosis of SVT, of which 67% were female.17 In the year before their SVT diagnosis, women had overall higher health care utilization with more ambulatory clinic visits, emergency department visits, and hospital admissions for SVT (Figure 3).14,17 After the SVT diagnosis, health care utilization increased in both men and women, but significantly more in men.17 This difference was driven by the higher likelihood of catheter ablation and the associated hospital admission in men. However, nonablation-related, outpatient expenditures were significantly higher in women because of more frequent health care visits, greater medication use, and a higher proportion of women who remained symptomatic after ablation.17
Figure 3.
Health care resource use before and after supraventricular tachycardia diagnosis. ER = emergency room; IP = inpatient admission. (Reproduced from Sacks et al17 with permission.)
During pregnancy, the most common SVT is inappropriate sinus tachycardia, with an incidence of approximately 0.1%.18 AF occurs is 0.03–0.06 of pregnancies, whereas reported rates for other SVTs are between 0.02% and 0.5% of pregnancies.18,19 For about half of these women this is the first presentation, whereas women with known AF or SVT are likely to experience worsening of their symptoms and an increased number of episodes because of the altered volume status and increased sympathetic activity.19,20 The management of SVTs during pregnancy is limited to electrical cardioversion and/or pharmacologic management; however, the choice of antiarrhythmic drug should be well considered.18,19 Fluoroless catheter ablation during pregnancy has been performed successfully but requires further research, as data currently are limited.21
Sex differences in AVNRT and AVRT
Sex differences in the incidences of AVNRT and AVRT have been widely reported, with a female-to-male ratio of 2:1 for AVNRT and 1:2 for AVRT.15,22,23 Multiple studies have reported significant sex differences in invasive electrophysiological measurements in patients with AVNRT and AVRT.13,24,25 The higher incidence of AVNRT in women has been linked to shorter AV nodal refractory cycle lengths and a wider tachycardia window.12,24,25 The latter reflects the shorter anterograde AV nodal slow pathway effective refractory periods in women, with similar refractory periods for the AV nodal fast pathway compared to men.24 However, other studies have reported minimal or no difference in the tachycardia window; rather, they suggest a causal relation with a higher incidence of premature atrial complexes in women and a potential proarrhythmic effect of progesterone during the luteal phase of the menstrual cycle.12,25 The inducibility of SVT was highest at the onset of menses and during the premenstrual phase when estrogen levels are low and progesterone levels and plasma norepinephrine levels are high.6 These findings are supported by the equal distribution of AVNRT in prepubertal males and females. In AVRT, similar invasive electrophysiological differences have been reported between males and females.13 However, these differences were less pronounced than in patients with AVNRT, likely because of the younger age at presentation for patients with AVRT.
The acute procedural success rates of catheter ablation for AVNRT and AVRT ranged between 91% and 99%.22,25 No studies have reported any significant difference in acute success rates or complication rates between men and women; however, women were at a 3 times higher risk for long-term recurrences and need for repeat ablation.22,23 This finding may be related to fewer radiofrequency applications during SVT ablation in women.15,25
Sex differences in focal atrial tachycardia
The incidence of focal atrial tachycardia (AT) is higher in women compared to men, with a female-to-male ratio of 3:2.6 In European and Asian registries including patients undergoing SVT ablation, the proportion of female patients undergoing focal AT ablation is very consistent at approximately 56%.23,26,27 This suggests a minimal referral bias for AT ablation given the discrepancy between the reported incidence and the proportion of female patients in the ablation registries. Although left atrial and biatrial focal etiologies are comparable between male and female patients, a right atrial etiology was more common in women.26 The acute success rates for ablation ranged between 84% and 94%, without a significant difference between men and women.23,26,27 Only 1 registry reported a long-term success rate of 72%, but it did not report on sex differences.27 One study from Singapore reported that 4% of patients underwent repeat ablation for focal ATs, with a significant higher risk for repeat ablation in women (male sex: odds ratio 0.34; 95% confidence interval 0.21–0.56).23
Sex differences in atrial flutter
The incidence of atrial flutter is closely related to that of AF; therefore, sex differences often are reported for both AF and atrial flutter together. The proportion of female patients with atrial flutter in ablation registries depends on the selection criteria and ranged overall between 21% and 27%.27, 28, 29 The lowest rate of 21% was reported in a Danish nationwide registry, which excluded patients with heart failure, implantable cardiac devices, and cardiomyopathies.29 In a German registry, the reported rates differed significantly between subgroups with structural heart disease (26% female) and patients with structural normal heart (46% female).30 Reported acute success rates of atrial flutter ablation, including both typical and atypical flutters, was 96%, with a long-term success rate of 71%.27 Success rates were similar between men and women.28,29 However, the largest registry reported a significantly higher complication rate in female patients (3.6% vs 3.0%; P = .030) but did not provide insights on sex differences for specific complications.28
Racial and ethnic differences in SVT, AT, and atrial flutter
Although several studies have reported sex differences in SVT, data on racial differences are rare. Go et al reported comparable SVT incidence in Whites (100/00,000) and Blacks (107/100,000), whereas the incidence in Asians/Pacific Islanders was significantly lower (51/100,000).31 However, no incidence rates by type of SVT were presented. Because most registries including AVNRT and AVRT patients did not either report or show significant differences between races, procedural differences and referral bias could not be assessed.23,28 For atrial flutter, limited data from the United States are available using the Nationwide Inpatient Sample and administrative data from Florida.28,32 Whereas Patel et al28 did not report any nationwide racial differences in rates of atrial flutter ablation, Tamariz et al32 described racial disparities in access to atrial flutter ablation. Although more Blacks underwent atrial flutter ablation (odds ratio 1.08; 95% confidence interval 0.96–1.21) compared to Whites, this did not reach statistical significance. These data clearly illustrate the need for more detailed reports to determine whether racial disparities in management exist.
AF
Sex differences in clinical presentation
Sex differences in the clinical characteristics, referral patterns, and management of AF have been described. ORBIT-AF (Outcomes Registry for Better Informed Treatment of Atrial Fibrillation), an observational cohort study of over 10,000 patients, reported that women were significantly more symptomatic with AF.33 Women reported more frequent palpitations, dyspnea with exertion, exercise intolerance, light-headedness, dyspnea at rest, fatigue, and chest discomfort. In the ORBIT-AF study, women also reported worse quality of life as reflected in significantly lower Atrial Fibrillation Effects on Quality-of-Life scores. The Basel Atrial Fibrillation Cohort investigators also reported that women were more symptomatic than men (any symptoms 85% vs 68%; P <.001).34 Perhaps of more concerning, although the AF symptom burden decreased over time from diagnosis in both men and women, these sex-related differences in symptoms and health perception persisted during long-term follow-up. At 1-year follow-up, 49% of women continued to report symptoms compared to 33% of men (P <.001).
Sex differences in AF management
Although women are more symptomatic with AF, the ORBIT-AF Investigators reported that women were less likely to undergo electrical cardioversion or AF ablation and were more likely to undergo AV nodal ablation compared to men.33 They also were more likely to be treated with digoxin and less likely to receive a β-blocker for rate control. Sex differences in AF treatment have been confirmed by the Euro Observational Research Program, an observational cohort study that included over 3000 patients.35 In this cohort, women were more likely to undergo trials of pharmacologic conversion and were less likely to undergo electrical cardioversion or catheter ablation for AF. Similar findings have been reported in an analysis of Medicare beneficiaries with newly diagnosed AF in 2010–2011.36 Men were more likely to be referred to a general cardiologist or electrophysiologist and were more likely to receive rhythm-controlling medication or catheter ablation compared to women (Table 2).
Table 2.
Sex and racial differences in AF management
| Health service | Male (N = 209,788) | Female (N = 308,153) | White (N = 452,986) | Black (N = 36,425) | Hispanic (N = 28,530) |
|---|---|---|---|---|---|
| Rhythm-controlling medication | 45,947 (21.9) | 61,237 (19.9) | 96,387 (21.3) | 5527 (15.2) | 5270 (18.5) |
| Catheter ablation | 2626 (1.3) | 1700 (0.6) | 3884 (0.9) | 278 (0.8) | 164 (0.6) |
Sex differences in referral for AF ablation have been confirmed by other administrative data analyses. Data from admissions for symptomatic AF to acute care hospitals in the states of California, Florida, and New York between 2006 and 2011 identified that those patients subsequently readmitted for catheter ablation of AF were younger, more often were male, and more often were White with private health insurance and higher household incomes.37 More contemporary analysis of data from the National Inpatient Sample for the year 2018, which included over 82,000 patients admitted for AF in the United States, confirms that men are more likely to undergo electrical cardioversion (21.4% vs 16.0%; P <.001) or AF ablation (5.2% vs 3.7%) compared to women.38
Sex differences in AF ablation and outcome
Sex disparity in AF ablation has been reported in a contemporary AF registry from Kansai Japan (n = 5010).39 In this cohort, women undergoing AF ablation were older, had more paroxysmal AF, and at the time of ablation were noted to have more nonpulmonary vein foci. Over median follow-up of 2.9 years, the cumulative incidence of AF recurrence was significantly higher for women compared to men (43.3% vs 39.0%; P = .005), and these differences were observed for both paroxysmal and persistent AF.39 Furthermore, over long-term follow-up women were more likely to experience major bleeding complications, hospitalizations for heart failure, or implantation of a cardiac implantable electronic device. The German Ablation Registry reported periprocedural complications in 3652 patients (33% female) undergoing AF ablation.40 In this cohort, women were older at the time of ablation and had a higher prevalence of paroxysmal AF. The rate of major in-hospital complications was higher in women (1.9% vs 0.8%; P = .023), driven mainly by major bleeding events (1.7% vs 0.5%; P = .004). In this cohort, women experienced higher AF recurrence rates, were more likely to have persistent symptoms attributed to AF, and were more likely to continue to receive oral medication for rate and rhythm control during long-term follow-up. The higher complication rates and readmissions after AF ablation observed in women have been confirmed from analysis of a larger administrative United States Nationwide Readmissions Database of patients undergoing AF ablation between 2010 and 2014 (n = 54,597).41 Women were more likely to experience cardiac perforation/tamponade and major bleeding complications. Furthermore, women had higher 30-day readmission rates for any cause (AF, atrial flutter, other cardiovascular causes).
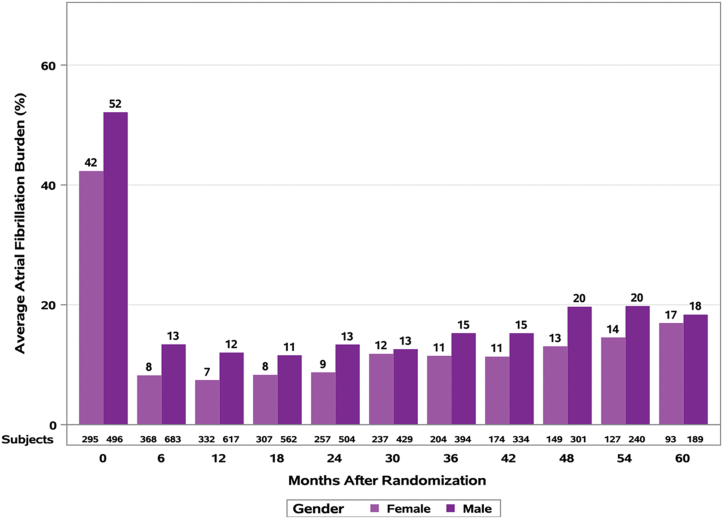
A prespecified analysis of outcomes by sex was recently reported by the CABANA (Catheter Ablation Versus Antiarrhythmic Drug Therapy for Atrial Fibrillation) Investigators.42 As in earlier reports, in CABANA, women undergoing ablation were older, had more symptoms associated with AF, and were more likely to have paroxysmal AF. They were less likely to have nonpulmonary vein trigger ablation procedures at the index ablation. The primary outcome of death, disabling stroke, serious bleeding, or cardiac arrest was similar in both women and men undergoing ablation. However, consistent with the previously reported cohort studies, in CABANA, women were more likely to experience recurrence of AF after the blanking period. At 12-month follow-up, 59% of women compared to 66% of men were free from AF recurrence after AF ablation. Women randomized in CABANA had a significantly lower AF burden at enrollment compared to men (42% vs 52% of Holter recording time; P = .006) (Figure 4). However, the incremental reduction in AF burden postablation was lower in women. In CABANA, complication rates related to ablation were similar in men and women, which may reflect the evolution of AF ablation procedures and the experience of participating centers.
Figure 4.
Sex differences in atrial fibrillation burden at baseline and after catheter ablation. (Reproduced from Russo et al42 with permission.)
The reason for higher rates of AF recurrence noted in women was explored in a small cohort of 117 patients.43 Differences in atrial remodeling have been identified, with more areas of reduced voltage, decreased conduction velocity, and fractionated atrial electrograms noted in women. These findings suggest that more adverse electrical remodeling in women compared to men may explain the higher risk of AF recurrence postablation. Sex differences in atrial remodeling may explain the need for more adjunctive ablation lesions required in women undergoing AF ablation as recently reported by the Get with the Guidelines AF Registry.44
Racial differences in AF
Epidemiologic studies have consistently reported racial differences in the incidence and prevalence of AF. Whites are at higher risk for AF despite a higher prevalence of known risk factors for AF documented among other racial and ethnic groups.45 The lifetime risk of AF increases markedly after age 50 years in European men and after age 60 years in women.46 A similar age-dependent difference in AF risk has also been reported in Black men compared to Black women.47
Racial differences in symptoms attributable to AF also have been reported. The ORBIT-AF Registry reported that Black patients were more likely to report symptoms of palpitations, dyspnea on exertion, decreased exercise tolerance, dizziness, dyspnea at rest, fatigue, and chest discomfort compared with White or Hispanic patients.48 Black patients also reported more severe or disabling symptoms (20.4%) compared with White (16.4%) and Hispanic (8.5%) patients. Black patients reported lower overall quality-of-life scores within 2 years of follow-up. In the analysis of Medicare beneficiaries with newly diagnosed AF, Blacks and Hispanics were less likely to receive rhythm-controlling antiarrhythmic drug therapy or be referred for catheter ablation for AF (Table 2).36
Race differences in AF ablation have been reported.36,49,50 Analysis of the National Cardiovascular Data Registry (NCDR®) PINNACLE (Practice Innovation and Clinical Excellence) Registry compared outcomes between Asian and White patients with AF between January 2013 and June 2018.49 Asians were less likely to be managed with rhythm control strategies (26.3% vs 30.0%; P <.001), including catheter ablation (2.2% vs 3.2%; P <.001). A separate analysis of a large, diverse, commercially insured population reported that overall rates of antiarrhythmic drug use and catheter ablation for the management of AF increased between 2016 and 2019. However, Black and Hispanic groups were less likely to receive catheter ablation. This factor was strongly associated with lower socioeconomic status.50
The CABANA Investigators studied outcomes of ablation and antiarrhythmic drug use in racial and ethnic minorities, which comprised 9.9% of the study population.51 Compared to the nonminority group, racial and ethnic minorities experienced a worse outcome with antiarrhythmic drug therapy and similar outcomes with catheter ablation. In the racial and ethnic minority groups, the burden of AF was significantly reduced after ablation compared to antiarrhythmic drug use despite a higher baseline AF burden in the subgroup randomized to ablation.
Ventricular tachycardia
Sex differences in structural normal hearts
The underlying mechanism of ventricular tachycardia (VT) in patients with a structural normal heart is predominantly enhanced automaticity or triggered activity. Whereas a right ventricular outflow tract origin is twice as common in female patients, there was no difference between men and women with regard to left ventricular outflow tract origins.52 In a large retrospective German registry, 53% of patients with structural normal heart who underwent catheter ablation for VT or ventricular premature beats were female.53 Similar proportions among patients with structural normal hearts were reported in other retrospective registries in the United States (48% female) and Japan (42% female).54,55 No sex-related differences in outcome were reported.
Verapamil-sensitive fascicular VT, most often related to reentry between the posteroseptal left ventricular myocardium and the Purkinje network, is 3 times more common in male patients.52 It has been hypothesized that this difference is related to shorter Purkinje cell action potential duration and slower His-Purkinje conduction in men; however, data on ion channel expression in human Purkinje fibers are rare.56
Although sustained VT occurs in <0.01% of pregnancies, they occur more frequently in women with a structural normal heart, the majority of which originate from the right ventricular outflow tract.18 In women with a history of idiopathic VT, recurrences were reported in 27% of pregnancies.20
Sex differences in structural abnormal hearts
In patients with structural abnormal hearts, the predominant mechanism underlying ventricular arrhythmias is reentry due to the presence of fibrosis or scar. The population at arrhythmic risk due to structural abnormal hearts can largely be divided into patients with ischemic and nonischemic cardiomyopathy. Table 3 presents an overview of large clinical trials from the past decades on VT ablation. The majority of these studies were conducted in patients with ischemic cardiomyopathy, and only 10.3% of included patients were female. Registries have reported comparable rates for female patients with ischemic cardiomyopathy undergoing VT ablation between 9% and 12%.53,54 Although this discrepancy has been related to the difference in incidence and prevalence of ischemic cardiomyopathy with a higher proportion of male patients, a selection bias in referral of women for VT ablation has also been reported.57 In a retrospective registry including 10-year of data from the United States, 6% of patients with ischemic cardiomyopathy who presented with VT underwent catheter ablation.57 When comparing the characteristics of those receiving ablation vs conservative management, only 10% of patients undergoing catheter ablation were female, whereas in the conservative management group, 17% of patients were female. Furthermore, during the 10-year inclusion period, the proportion of women undergoing VT ablation decreased from 19% in 2002 to 13% in 2011.57 Unfortunately, no contemporary data are available.
Table 3.
Overview of the inclusion criteria for the major VT catheter ablation studies
| Study | Year | Total no. | Ischemic cardiomyopathy (%) | Female [n (%)] | Ethnicity [n (%)] | Subgroup analysis by sex |
|---|---|---|---|---|---|---|
| COOLED-RF74 | 2000 | 146 | 82 | 12 (8) | NA | Not significant |
| SMASH-VT75 | 2007 | 128 | 100 | 17 (13) | NA | Not significant |
| Carbucicchio et al76 | 2008 | 95 | 76 | 10 (11) | NA | Not significant. |
| VTACH77 | 2010 | 107 | 100 | 7 (7) | NA | NA |
| CALYPSO64 | 2014 | 27 | 100 | 2 (7) | Black: 6 (22) | NA |
| VISTA78 | 2015 | 118 | 100 | 8 (7) | NA | Males had a higher risk of VT recurrence in univariate analysis, not in multivariable analysis |
| THERMOCOOL65 | 2016 | 249 | 100 | 15 (6) | Non-White: 20 (8) | NA |
| VANISH79 | 2016 | 259 | 100 | 18 (7) | NA | Not significant |
| SMS80 | 2017 | 111 | 100 | 18 (16) | NA | NA |
| BERLIN-VT81 | 2020 | 159 | 100 | 20 (13) | NA | NA |
| Fernandez-Armenta et al82 | 2020 | 412 | 66 | 36 (9) | NA | NA |
NA = not available; VT = ventricular tachycardia.
In patients with nonischemic cardiomyopathy, sex disparities in access to catheter are less pronounced, but reported rates of ablation are variable. The proportion of female patients with nonischemic cardiomyopathy undergoing VT ablation ranged between 19% and 29% in retrospective registries.53,54,58 In a nationwide cohort reporting on 10-year trends in VT ablation in patients with nonischemic cardiomyopathy in the United States, 47% was female.59 In a propensity score-matched analysis from this cohort, there was no evident referral bias because there was no significant difference in the proportion of women in the subgroup not referred for catheter ablation compared to those referred for catheter ablation (45% vs 46%, respectively). However, in the 10-year trend analysis, the proportion of women undergoing catheter ablation for nonischemic VT decreased from 55% in 2003 to 41% in 2014.59 The reason for this decline is uncertain; however, a significant gradual increase in in-hospital mortality (0.2% in 2003 to 1.6% in 2014) and complication rate (2.9% in 2003 to 10.3% in 2014) was observed. This increase in complication rates was driven by vascular and cardiac complications, but no data on sex differences in complications were reported.59
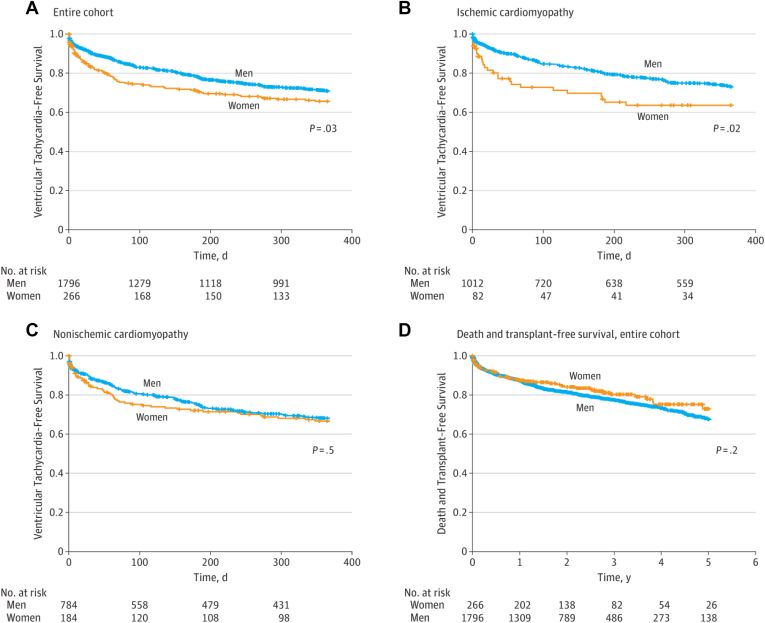
Several large retrospective registries of patients undergoing catheter ablation for VT in the setting of structural heart disease have reported significant sex differences in disease management and clinical outcomes. First, before VT ablation, women less frequently received an implantable cardioverter-defibrillator or cardiac resynchronization therapy device.58 Second, women were less frequently prescribed beta-blockers or antiarrhythmic drugs.58 More specifically, the use of amiodarone in women was lower, whereas the use of sotalol was significantly higher in female patients.58 Third, although complication rates were comparable between male and female patients, the procedural outcome differed significantly.54,58 At the end of the procedure, the clinical VT more often still was inducible in women, and there was a higher recurrence of VT during follow-up in women with ischemic cardiomyopathy (Figure 5).58,60 Interestingly, shorter total ablation time and shorter ablation time per clinical inducible VT were observed among female patients, despite a comparable complexity of cases with similar proportions of epicardial mapping and total number of mappable VTs noted between males and females.58 However, there was no difference in readmission rates or referral for repeat ablation.60,61 Lastly, female patients were reported to have comparable60 or better survival53,62 compared to male patients. Detailed analysis of cardiac magnetic resonance imaging and electroanatomic mapping has not identified a significant difference in the substrate for VT between male and female patients with nonischemic cardiomyopathy.63 Only 5 of the clinical trials summarized in Table 3 reported a subgroup analysis by sex, and none of them showed significant differences in their primary outcomes.
Figure 5.
Sex differences in ventricular tachycardia–free survival and all-cause mortality. A: Entire cohort. B: Ischemic cardiomyopathy. C: Nonischemic cardiomyopathy. D: Death and transplant-free survival, entire cohort. (Reproduced from Frankel et al58 with permission.)
Racial differences in VT
Overall, data on racial and ethnic differences with regard to VT ablation are limited. Only 2 of the 11 studies summarized in Table 3 reported patient race/ethnicity, but no subgroup analyses were performed.64,65 In the United States, 87% of patients with ischemic cardiomyopathy undergoing VT ablation between 2002 and 2011 were White, whereas 5% were Black, 4% Hispanic, and 1% Asian.57 The data suggested a referral bias for Blacks, as approximately 7.3% of patients who were not referred for catheter ablation were Black compared to 4.6% for those who were referred for catheter ablation.57 Temporal trends over 10 years suggest that the proportion of non-White patients undergoing catheter ablation may be increasing, albeit marginally.57 Among patients with nonischemic cardiomyopathy referred for VT ablation between 2003 and 2014, 59% were White, whereas 10% were Black, 6% Hispanic, and 2% Asian or Pacific Islander.59 Although the 10-year trends showed significant improvement in VT ablation rates, the data were confounded by a high proportion of missing data in the initial years of the registry (35% missing data on race in 2003–2004 vs 7% in 2013–2014).59 As such, the true trend over time cannot be accurately assessed.
Clinical perspective and future directions
While ethnic and racial diversity in North America continues to grow, the disparities between sex and race in cardiovascular clinical research and health care utilization remains striking.66 The underrepresentation of females and racial/ethnic groups in clinical trials and registries is multifactorial and is incompletely understood. Although differences in disease prevalence and clinical presentation might impact inclusion and exclusion criteria, studies reporting participation-to-prevalence ratios have clearly illustrated inclusion bias.1 Sex and race/ethnicity disparities remain prevalent in primary health care and clinical trial participation.1,66,67 Women and individuals from non-White racial or ethnic groups are less likely to opt for invasive procedures or to engage in clinical research.67,68 Studies have revealed implicit bias, systemic prejudice, and absence of trust in health care in combination with differences in health literacy and information-seeking behavior as factors influencing sex and racial disparities in care.68 In addition, differences in an individual’s values and preferences for treatment as well as socioeconomic factors may influence these disparities in care. It has been suggested that physicians should adapt their communication strategies to overcome discrepancies in symptom interpretation, referral bias, and risk-to-benefit discussions for catheter ablation therapy.69 The latter has been linked to anxiety of women toward adverse effects of radiation exposure, particularly in premenopausal women, which should be overcome given the high success rates of SVT ablation with a minimal fluoroscopy approach.
Meanwhile, several attempts to close the gap have been initiated, such as the “Go Red for Women” movement and research network, which aims to increase women’s heart health awareness and serve as a catalyst for change to improve the lives of women globally.70 The interaction of sex and ethnic and racial differences with pathophysiology and treatment responses for cardiac arrhythmias is complex. The knowledge gaps created by exclusion of specific populations in the past decades require immediate action, particularly in an era with shifts toward personalized and precision medicine.
We therefore wish to launch a call for action:
-
•
Funding agencies should prioritize research to close the gap in basic science knowledge and improve the understanding of sex and racial disparities.
-
•
Clinical trial steering committees should strive for sex and racial equality because research has shown that greater representation of women on steering committees leads to more diverse recruitment.71,72
-
•
Improvements should be made in the recruitment and retention of underrepresented groups, which requires community engagement, decentralization of study sites to enhance accessibility, and increased racial representation among investigators.73
-
•
Clinical trials and registries should report detailed results for sex and racial subgroups, as this may increase the variability and sample size for meta-analyses.
-
•
Use of social media should be maximized to create disparity awareness, not neglecting the fact that at least part of these populations is hard to reach this way.
-
•
Health illiteracy should be countered by providing free information resources, available in different languages and at a basic level.
Conclusion
Sex, ethnic, and racial disparities are evident in the presentation, diagnosis, and management of cardiac arrythmias. However, the understanding of mechanisms by which sex, ethnic, and racial differences affect cardiac arrhythmias is still at an early stage. Engagement of the research community and industry is essential to address the disparities and improve patient care on a global level. Sex- and minority-specific trials are needed to acquire a detailed understanding of arrhythmia mechanisms, determinants, and therapeutic responses, together with measures to overcome sex and minority exclusion from health care.
Acknowledgments
Funding Sources
Dr Vandenberk is supported by a research grant from the Frans Van de Werf Fund for Clinical Cardiovascular Research (Leuven, Belgium).
Disclosures
Dr Parkash reports research funding from Medtronic and Abbott. Drs Vandenberk, Chew, and Gillis have no relationships to disclose.
Authorship
All authors attest they meet the current ICMJE criteria for authorship.
References
- 1.Scott P.E., Unger E.F., Jenkins M.R., et al. Participation of women in clinical trials supporting FDA approval of cardiovascular drugs. J Am Coll Cardiol. 2018;71:1960–1969. doi: 10.1016/j.jacc.2018.02.070. [DOI] [PubMed] [Google Scholar]
- 2.Preventza O., Critsinelis A., Simpson K., et al. Sex, racial, and ethnic disparities in U.S. cardiovascular trials in more than 230,000 patients. Ann Thorac Surg. 2021;112:726–735. doi: 10.1016/j.athoracsur.2020.08.075. [DOI] [PubMed] [Google Scholar]
- 3.Freedman L.S., Simon R., Foulkes M.A., et al. Inclusion of women and minorities in clinical trials and the NIH Revitalization Act of 1993—the perspective of NIH clinical trialists. Control Clin Trials. 1995;16:277–285. doi: 10.1016/0197-2456(95)00048-8. ; discussion 286–279, 293–309. [DOI] [PubMed] [Google Scholar]
- 4.Gillis A.M. Atrial fibrillation and ventricular arrhythmias: sex differences in electrophysiology, epidemiology, clinical presentation, and clinical outcomes. Circulation. 2017;135:593–608. doi: 10.1161/CIRCULATIONAHA.116.025312. [DOI] [PubMed] [Google Scholar]
- 5.Macfarlane P.W. The influence of age and sex on the electrocardiogram. Adv Exp Med Biol. 2018;1065:93–106. doi: 10.1007/978-3-319-77932-4_6. [DOI] [PubMed] [Google Scholar]
- 6.Tadros R., Ton A.T., Fiset C., Nattel S. Sex differences in cardiac electrophysiology and clinical arrhythmias: epidemiology, therapeutics, and mechanisms. Can J Cardiol. 2014;30:783–792. doi: 10.1016/j.cjca.2014.03.032. [DOI] [PubMed] [Google Scholar]
- 7.Ravens U. Sex differences in cardiac electrophysiology. Can J Physiol Pharmacol. 2018;96:985–990. doi: 10.1139/cjpp-2018-0179. [DOI] [PubMed] [Google Scholar]
- 8.Odening K.E., Koren G. How do sex hormones modify arrhythmogenesis in long QT syndrome? Sex hormone effects on arrhythmogenic substrate and triggered activity. Heart Rhythm. 2014;11:2107–2115. doi: 10.1016/j.hrthm.2014.06.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kurokawa J., Kodama M., Clancy C.E., Furukawa T. Sex hormonal regulation of cardiac ion channels in drug-induced QT syndromes. Pharmacol Ther. 2016;168:23–28. doi: 10.1016/j.pharmthera.2016.09.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kappus R.M., Ranadive S.M., Yan H., et al. Sex differences in autonomic function following maximal exercise. Biol Sex Differ. 2015;6:28. doi: 10.1186/s13293-015-0046-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.O’Neill J., Tayebjee M.H. Electrophysiological properties of the South Asian heart. Heart Asia. 2018;10 doi: 10.1136/heartasia-2018-011079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Deneke T., Muller P., Lawo T., et al. Gender differences in onset of symptoms in AV nodal re-entrant and accessory pathway-mediated re-entrant tachycardia. Herzschrittmacherther Elektrophysiol. 2009;20:33–38. doi: 10.1007/s00399-009-0036-7. [DOI] [PubMed] [Google Scholar]
- 13.Huang S.Y., Hu Y.F., Chang S.L., et al. Gender differences of electrophysiologic characteristics in patients with accessory atrioventricular pathways. Heart Rhythm. 2011;8:571–574. doi: 10.1016/j.hrthm.2010.12.013. [DOI] [PubMed] [Google Scholar]
- 14.Musa T., Darrat Y., Etaee F., et al. Gender differences in management of patients undergoing catheter ablation of atrioventricular nodal reentry tachycardia. Pacing Clin Electrophysiol. 2019;42:937–941. doi: 10.1111/pace.13735. [DOI] [PubMed] [Google Scholar]
- 15.Dagres N., Clague J.R., Breithardt G., Borggrefe M. Significant gender-related differences in radiofrequency catheter ablation therapy. J Am Coll Cardiol. 2003;42:1103–1107. doi: 10.1016/s0735-1097(03)00925-2. [DOI] [PubMed] [Google Scholar]
- 16.Farkowski M.M., Pytkowski M., Maciag A., et al. Gender-related differences in outcomes and resource utilization in patients undergoing radiofrequency ablation of supraventricular tachycardia: results from Patients' Perspective on Radiofrequency Catheter Ablation of AVRT and AVNRT Study. Europace. 2014;16:1821–1827. doi: 10.1093/europace/euu130. [DOI] [PubMed] [Google Scholar]
- 17.Sacks N.C., Everson K., Emden M.R., et al. Disparities in the management of newly diagnosed paroxysmal supraventricular tachycardia for women versus men in the United States. J Am Heart Assoc. 2020;9 doi: 10.1161/JAHA.120.015910. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kugamoorthy P., Spears D.A. Management of tachyarrhythmias in pregnancy—a review. Obstet Med. 2020;13:159–173. doi: 10.1177/1753495X20913448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ramlakhan K.P., Kauling R.M., Schenkelaars N., et al. Supraventricular arrhythmia in pregnancy. Heart. 2022 Jan 27 doi: 10.1136/heartjnl-2021-320451. heartjnl-2021-320451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Silversides C.K., Harris L., Haberer K., et al. Recurrence rates of arrhythmias during pregnancy in women with previous tachyarrhythmia and impact on fetal and neonatal outcomes. Am J Cardiol. 2006;97:1206–1212. doi: 10.1016/j.amjcard.2005.11.041. [DOI] [PubMed] [Google Scholar]
- 21.Abdrakhmanov A., Ainabekova B., Nuralinov O., Bakytzhanuly A., Smagulova A. The efficacy and safety of non-fluoroscopic catheter ablation of arrhythmias in pregnancy after mid-long-term follow-up: a single-center experience. EP Europace. 2021;23(Suppl 3):058. euab116. [Google Scholar]
- 22.Feldman A., Voskoboinik A., Kumar S., et al. Predictors of acute and long-term success of slow pathway ablation for atrioventricular nodal reentrant tachycardia: a single center series of 1,419 consecutive patients. Pacing Clin Electrophysiol. 2011;34:927–933. doi: 10.1111/j.1540-8159.2011.03092.x. [DOI] [PubMed] [Google Scholar]
- 23.Tan E.S.J., Chan S.P., Seow S.C., et al. Outcomes of supraventricular tachycardia ablation: results from the Singapore ablation and cardiac devices registry. Pacing Clin Electrophysiol. 2022;45:50–58. doi: 10.1111/pace.14410. [DOI] [PubMed] [Google Scholar]
- 24.Liuba I., Jonsson A., Safstrom K., Walfridsson H. Gender-related differences in patients with atrioventricular nodal reentry tachycardia. Am J Cardiol. 2006;97:384–388. doi: 10.1016/j.amjcard.2005.08.042. [DOI] [PubMed] [Google Scholar]
- 25.Suenari K., Hu Y.F., Tsao H.M., et al. Gender differences in the clinical characteristics and atrioventricular nodal conduction properties in patients with atrioventricular nodal reentrant tachycardia. J Cardiovasc Electrophysiol. 2010;21:1114–1119. doi: 10.1111/j.1540-8167.2010.01779.x. [DOI] [PubMed] [Google Scholar]
- 26.Busch S., Forkmann M., Kuck K.H., et al. Acute and long-term outcome of focal atrial tachycardia ablation in the real world: results of the german ablation registry. Clin Res Cardiol. 2018;107:430–436. doi: 10.1007/s00392-018-1204-8. [DOI] [PubMed] [Google Scholar]
- 27.Brachmann J., Lewalter T., Kuck K.H., et al. Long-term symptom improvement and patient satisfaction following catheter ablation of supraventricular tachycardia: insights from the German ablation registry. Eur Heart J. 2017;38:1317–1326. doi: 10.1093/eurheartj/ehx101. [DOI] [PubMed] [Google Scholar]
- 28.Patel N.J., Deshmukh A., Pau D., et al. Contemporary utilization and safety outcomes of catheter ablation of atrial flutter in the United States: analysis of 89,638 procedures. Heart Rhythm. 2016;13:1317–1325. doi: 10.1016/j.hrthm.2016.02.005. [DOI] [PubMed] [Google Scholar]
- 29.Vadmann H., Gorst-Rasmussen A., Hjortshoj S.P., et al. Death and thrombo-embolic risk after ablation of atrial flutter compared with atrial fibrillation: a nationwide cohort study. Europace. 2017;19:838–842. doi: 10.1093/europace/euw107. [DOI] [PubMed] [Google Scholar]
- 30.Eitel C., Ince H., Brachmann J., et al. Catheter ablation of supraventricular tachycardia in patients with and without structural heart disease: insights from the German ablation registry. Clin Res Cardiol. 2022;111:522–529. doi: 10.1007/s00392-021-01878-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Go A.S., Hlatky M.A., Liu T.I., et al. Contemporary burden and correlates of symptomatic paroxysmal supraventricular tachycardia. J Am Heart Assoc. 2018;7 doi: 10.1161/JAHA.118.008759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Tamariz L., Rodriguez A., Palacio A., Li H., Myerburg R. Racial disparities in the use of catheter ablation for atrial fibrillation and flutter. Clin Cardiol. 2014;37:733–737. doi: 10.1002/clc.22330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Piccini J.P., Simon D.N., Steinberg B.A., et al. Differences in clinical and functional outcomes of atrial fibrillation in women and men. JAMA Cardiol. 2016;1:282. doi: 10.1001/jamacardio.2016.0529. [DOI] [PubMed] [Google Scholar]
- 34.Blum S., Muff C., Aeschbacher S., et al. Prospective assessment of sex-related differences in symptom status and health perception among patients with atrial fibrillation. J Am Heart Assoc. 2017;6 doi: 10.1161/JAHA.116.005401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Lip G.Y.H., Laroche C., Boriani G., et al. Sex-related differences in presentation, treatment, and outcome of patients with atrial fibrillation in Europe: a report from the Euro Observational Research Programme Pilot survey on Atrial Fibrillation. EP Europace. 2014;17:24–31. doi: 10.1093/europace/euu155. [DOI] [PubMed] [Google Scholar]
- 36.Bhave P.D., Lu X., Girotra S., Kamel H., Vaughan Sarrazin M.S. Race- and sex-related differences in care for patients newly diagnosed with atrial fibrillation. Heart Rhythm. 2015;12:1406–1412. doi: 10.1016/j.hrthm.2015.03.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Kummer B.R., Bhave P.D., Merkler A.E., et al. Demographic differences in catheter ablation after hospital presentation with symptomatic atrial fibrillation. J Am Heart Assoc. 2015;4:e002097. doi: 10.1161/JAHA.115.002097. e002097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Noubiap J.J., Thomas G., Agbaedeng T.A., et al. Sex differences in clinical profile, management, and outcomes of patients hospitalized for atrial fibrillation in the United States. Eur Heart J Qual Care Clin Outcomes. 2021 Dec 21:qcab096. doi: 10.1093/ehjqcco/qcab096. [DOI] [PubMed] [Google Scholar]
- 39.Tanaka N., Inoue K., Kobori A., et al. Sex differences in atrial fibrillation ablation outcomes: insights from a large-scale multicentre registry. EP Europace. 2020;22:1345–1357. doi: 10.1093/europace/euaa104. [DOI] [PubMed] [Google Scholar]
- 40.Zylla M.M., Brachmann J., Lewalter T., et al. Sex-related outcome of atrial fibrillation ablation: insights from the German Ablation Registry. Heart Rhythm. 2016;13:1837–1844. doi: 10.1016/j.hrthm.2016.06.005. [DOI] [PubMed] [Google Scholar]
- 41.Cheung J.W., Cheng E.P., Wu X., et al. Sex-based differences in outcomes, 30-day readmissions, and costs following catheter ablation of atrial fibrillation: the United States Nationwide Readmissions Database 2010-14. Eur Heart J. 2019;40:3035–3043. doi: 10.1093/eurheartj/ehz151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Russo A.M., Zeitler E.P., Giczewska A., et al. Association between sex and treatment outcomes of atrial fibrillation ablation versus drug therapy: results from the CABANA trial. Circulation. 2021;143:661–672. doi: 10.1161/CIRCULATIONAHA.120.051558. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Wong G.R., Nalliah C.J., Lee G., et al. Sex-related differences in atrial remodeling in patients with atrial fibrillation: relationship to ablation outcomes. Circ Arrhythm Electrophysiol. 2022;15 doi: 10.1161/CIRCEP.121.009925. [DOI] [PubMed] [Google Scholar]
- 44.Yunus F.N., Perino A.C., Holmes D.N., et al. Sex differences in ablation strategy, lesion sets, and complications of catheter ablation for atrial fibrillation: an analysis from the GWTG-AFIB Registry. Circ Arrhythm Electrophysiol. 2021;14 doi: 10.1161/CIRCEP.121.009790. [DOI] [PubMed] [Google Scholar]
- 45.Tamirisa K.P., Al-Khatib S.M., Mohanty S., et al. Racial and ethnic differences in the management of atrial fibrillation. CJC Open. 2021;3:S137–S148. doi: 10.1016/j.cjco.2021.09.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Magnussen C., Niiranen T.J., Ojeda F.M., et al. Sex differences and similarities in atrial fibrillation epidemiology, risk factors, and mortality in community cohorts: results from the BiomarCaRE Consortium (Biomarker for Cardiovascular Risk Assessment in Europe) Circulation. 2017;136:1588–1597. doi: 10.1161/CIRCULATIONAHA.117.028981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Ghelani K.P., Chen L.Y., Norby F.L., et al. Thirty-year trends in the incidence of atrial fibrillation: the ARIC study. J Am Heart Assoc. 2022;11 doi: 10.1161/JAHA.121.023583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Golwala H., Jackson L.R., Simon D.N., et al. Racial/ethnic differences in atrial fibrillation symptoms, treatment patterns, and outcomes: insights from Outcomes Registry for Better Informed Treatment for Atrial Fibrillation Registry. Am Heart J. 2016;174:29–36. doi: 10.1016/j.ahj.2015.10.028. [DOI] [PubMed] [Google Scholar]
- 49.Gu K., Mahtta D., Kaneria A., et al. Racial disparities among Asian Americans with atrial fibrillation: an analysis from the NCDR® PINNACLE Registry. Int J Cardiol. 2021;329:209–216. doi: 10.1016/j.ijcard.2020.12.064. [DOI] [PubMed] [Google Scholar]
- 50.Eberly L.A., Garg L., Yang L., et al. Racial/ethnic and socioeconomic disparities in management of incident paroxysmal atrial fibrillation. JAMA Netw Open. 2021;4 doi: 10.1001/jamanetworkopen.2021.0247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Thomas K.L., Al-Khalidi H.R., Silverstein A.P., et al. Ablation versus drug therapy for atrial fibrillation in racial and ethnic minorities. J Am Coll Cardiol. 2021;78:126–138. doi: 10.1016/j.jacc.2021.04.092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Nakagawa M., Takahashi N., Nobe S., et al. Gender differences in various types of idiopathic ventricular tachycardia. J Cardiovasc Electrophysiol. 2002;13:633–638. doi: 10.1046/j.1540-8167.2002.00633.x. [DOI] [PubMed] [Google Scholar]
- 53.Konig S., Ueberham L., Muller-Rothing R., et al. Catheter ablation of ventricular arrhythmias and in-hospital mortality: insights from the German-wide Helios hospital network of 5052 cases. Europace. 2020;22:100–108. doi: 10.1093/europace/euz260. [DOI] [PubMed] [Google Scholar]
- 54.Baldinger S.H., Kumar S., Romero J., et al. A comparison of women and men undergoing catheter ablation for sustained monomorphic ventricular tachycardia. J Cardiovasc Electrophysiol. 2017;28:201–207. doi: 10.1111/jce.13127. [DOI] [PubMed] [Google Scholar]
- 55.Kitamura T., Nakajima M., Kawamura I., et al. Patient characteristics, procedure details including catheter devices, and complications of catheter ablation for ventricular tachycardia: a nationwide observational study. J Arrhythm. 2020;36:464–470. doi: 10.1002/joa3.12356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Abi-Gerges N., Small B.G., Lawrence C.L., et al. Evidence for gender differences in electrophysiological properties of canine Purkinje fibres. Br J Pharmacol. 2004;142:1255–1264. doi: 10.1038/sj.bjp.0705880. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Palaniswamy C., Kolte D., Harikrishnan P., et al. Catheter ablation of postinfarction ventricular tachycardia: ten-year trends in utilization, in-hospital complications, and in-hospital mortality in the United States. Heart Rhythm. 2014;11:2056–2063. doi: 10.1016/j.hrthm.2014.07.012. [DOI] [PubMed] [Google Scholar]
- 58.Frankel D.S., Tung R., Santangeli P., et al. Sex and catheter ablation for ventricular tachycardia: an International Ventricular Tachycardia Ablation Center Collaborative Group study. JAMA Cardiol. 2016;1:938–944. doi: 10.1001/jamacardio.2016.2361. [DOI] [PubMed] [Google Scholar]
- 59.Briceno D.F., Gupta T., Romero J., et al. Catheter ablation of ventricular tachycardia in nonischemic cardiomyopathy: a propensity score-matched analysis of in-hospital outcomes in the United States. J Cardiovasc Electrophysiol. 2018;29:771–779. doi: 10.1111/jce.13452. [DOI] [PubMed] [Google Scholar]
- 60.Tung R., Vaseghi M., Frankel D.S., et al. Freedom from recurrent ventricular tachycardia after catheter ablation is associated with improved survival in patients with structural heart disease: an International VT Ablation Center Collaborative Group study. Heart Rhythm. 2015;12:1997–2007. doi: 10.1016/j.hrthm.2015.05.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Cheung J.W., Yeo I., Ip J.E., et al. Outcomes, costs, and 30-day readmissions after catheter ablation of myocardial infarct-associated ventricular tachycardia in the real world: nationwide readmissions database 2010 to 2015. Circ Arrhythm Electrophysiol. 2018;11 doi: 10.1161/CIRCEP.118.006754. [DOI] [PubMed] [Google Scholar]
- 62.Darma A., Bertagnolli L., Dinov B., et al. Predictors of long-term mortality after catheter ablation of ventricular tachycardia in a contemporary cohort of patients with structural heart disease. Europace. 2020;22:1672–1679. doi: 10.1093/europace/euaa189. [DOI] [PubMed] [Google Scholar]
- 63.Kuo L., Shirai Y., Muser D., et al. Comparison of the arrhythmogenic substrate between men and women with nonischemic cardiomyopathy. Heart Rhythm. 2019;16:1414–1420. doi: 10.1016/j.hrthm.2019.03.024. [DOI] [PubMed] [Google Scholar]
- 64.Al-Khatib S.M., Daubert J.P., Anstrom K.J., et al. Catheter ablation for ventricular tachycardia in patients with an implantable cardioverter defibrillator (CALYPSO) pilot trial. J Cardiovasc Electrophysiol. 2015;26:151–157. doi: 10.1111/jce.12567. [DOI] [PubMed] [Google Scholar]
- 65.Marchlinski F.E., Haffajee C.I., Beshai J.F., et al. Long-term success of irrigated radiofrequency catheter ablation of sustained ventricular tachycardia: post-approval THERMOCOOL VT trial. J Am Coll Cardiol. 2016;67:674–683. doi: 10.1016/j.jacc.2015.11.041. [DOI] [PubMed] [Google Scholar]
- 66.James C.V., Moonesinghe R., Wilson-Frederick S.M., et al. Racial/ethnic health disparities among rural adults—United States, 2012-2015. MMWR Surveill Summ. 2017;66:1–9. doi: 10.15585/mmwr.ss6623a1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Ferguson J.A., Tierney W.M., Westmoreland G.R., et al. Examination of racial differences in management of cardiovascular disease. J Am Coll Cardiol. 1997;30:1707–1713. doi: 10.1016/s0735-1097(97)00365-3. [DOI] [PubMed] [Google Scholar]
- 68.Strekalova Y.A. When trust is not enough: a serial mediation model explaining the effect of race identity, ehealth information efficacy, and information behavior on intention to participate in clinical research. Health Educ Behav. 2018;45:1036–1042. doi: 10.1177/1090198118757822. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Etaee F., Elayi C.S., Catanzarro J., et al. Gender associated disparities in atrioventricular nodal reentrant tachycardia: a review article. J Cardiovasc Electrophysiol. 2021;32:1772–1777. doi: 10.1111/jce.15078. [DOI] [PubMed] [Google Scholar]
- 70.St-Onge M.P., Aggarwal B., Allison M.A., et al. Go red for women strategically focused research network: summary of findings and network outcomes. J Am Heart Assoc. 2021;10 doi: 10.1161/JAHA.120.019519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Van Spall H.G.C., Lala A., Deering T.F., et al. Ending gender inequality in cardiovascular clinical trial leadership: JACC review topic of the week. J Am Coll Cardiol. 2021;77:2960–2972. doi: 10.1016/j.jacc.2021.04.038. [DOI] [PubMed] [Google Scholar]
- 72.Moin E.E., Reza N. Women and leadership of cardiology and oncology clinical trials-swimming against the tide. JAMA Netw Open. 2022;5 doi: 10.1001/jamanetworkopen.2022.0050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Kelsey M.D., Patrick-Lake B., Abdulai R., et al. Inclusion and diversity in clinical trials: actionable steps to drive lasting change. Contemp Clin Trials. 2022;116 doi: 10.1016/j.cct.2022.106740. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Calkins H., Epstein A., Packer D., et al. Catheter ablation of ventricular tachycardia in patients with structural heart disease using cooled radiofrequency energy: results of a prospective multicenter study. Cooled RF Multi Center Investigators Group. J Am Coll Cardiol. 2000;35:1905–1914. doi: 10.1016/s0735-1097(00)00615-x. [DOI] [PubMed] [Google Scholar]
- 75.Reddy V.Y., Reynolds M.R., Neuzil P., et al. Prophylactic catheter ablation for the prevention of defibrillator therapy. N Engl J Med. 2007;357:2657–2665. doi: 10.1056/NEJMoa065457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Carbucicchio C., Santamaria M., Trevisi N., et al. Catheter ablation for the treatment of electrical storm in patients with implantable cardioverter-defibrillators: short- and long-term outcomes in a prospective single-center study. Circulation. 2008;117:462–469. doi: 10.1161/CIRCULATIONAHA.106.686534. [DOI] [PubMed] [Google Scholar]
- 77.Kuck K.H., Schaumann A., Eckardt L., et al. Catheter ablation of stable ventricular tachycardia before defibrillator implantation in patients with coronary heart disease (VTACH): a multicentre randomised controlled trial. Lancet. 2010;375:31–40. doi: 10.1016/S0140-6736(09)61755-4. [DOI] [PubMed] [Google Scholar]
- 78.Di Biase L., Burkhardt J.D., Lakkireddy D., et al. Ablation of stable VTs versus substrate ablation in ischemic cardiomyopathy: the VISTA randomized multicenter trial. J Am Coll Cardiol. 2015;66:2872–2882. doi: 10.1016/j.jacc.2015.10.026. [DOI] [PubMed] [Google Scholar]
- 79.Sapp J.L., Wells G.A., Parkash R., et al. Ventricular tachycardia ablation versus escalation of antiarrhythmic drugs. N Engl J Med. 2016;375:111–121. doi: 10.1056/NEJMoa1513614. [DOI] [PubMed] [Google Scholar]
- 80.Kuck K.H., Tilz R.R., Deneke T., et al. Impact of substrate modification by catheter ablation on implantable cardioverter-defibrillator interventions in patients with unstable ventricular arrhythmias and coronary artery disease: results from the multicenter randomized controlled SMS (Substrate Modification Study) Circ Arrhythm Electrophysiol. 2017;10 doi: 10.1161/CIRCEP.116.004422. [DOI] [PubMed] [Google Scholar]
- 81.Willems S., Tilz R.R., Steven D., et al. Preventive or deferred ablation of ventricular tachycardia in patients with ischemic cardiomyopathy and implantable defibrillator (BERLIN VT): a multicenter randomized trial. Circulation. 2020;141:1057–1067. doi: 10.1161/CIRCULATIONAHA.119.043400. [DOI] [PubMed] [Google Scholar]
- 82.Fernandez-Armenta J., Soto-Iglesias D., Silva E., et al. Safety and outcomes of ventricular tachycardia substrate ablation during sinus rhythm: a prospective multicenter registry. JACC Clin Electrophysiol. 2020;6:1435–1448. doi: 10.1016/j.jacep.2020.07.028. [DOI] [PubMed] [Google Scholar]