Abstract
Four species of the Eimeriidae, Eimeria anatis Scholtyseck, 1955, Eimeria aythyae Farr, 1965, Eimeria krylovi Svanbaev & Rakhmatullina, 1967 and Tyzzeria perniciosa Allen, 1936, were morphologically identified from oöcysts recovered from a Pacific black duck, Anas superciliosa Gmelin. Additionally, genotypic characterization of E. anatis is provided via sequencing of the mitochondrial cytochrome c oxidase subunit 1 (cox1) and the small subunit ribosomal RNA (18S) genes. The four species are redescribed, providing additional morphological details. The validity of genera and coccidian species parasitizing birds of the order Anseriformes such as Wenyonella Hoare, 1933 and some Tyzzeria spp. are discussed. Molecular phylogenetic analyses for the cox1 and 18S rRNA genes resulted in monophylies of Eimeria spp. from Anseriformes which included the sequences obtained from E. anatis oöcysts.
Keywords: Coccidia, Eimeria, Tyzzeria, Pacific black duck, Anas superciliosa, 18S rRNA gene, cox1 gene
Graphical abstract

Highlights
-
•
Eimeria anatis, Eimeria aythyae, Eimeria krylovi, Tyzzeria perniciosa are identified from a new host, Anas superciliosa.
-
•
Coccidia are redrawn and redescribed to offer better characterization of the morphological features.
-
•
Genotypic characterization via sequencing of mitochondrial and ribosomal genes is provided for E. anatis.
-
•
Taxonomic validity of coccidia parasitic in Anseriformes, such as Wenyonella and some Tyzzeria spp. is discussed.
1. Introduction
The Pacific black duck Anas superciliosa Gmelin (Anseriformes: Anatidae) is a dabbling duck commonly seen in waterways, swamps, streams and ponds in Australia, New Zealand, Indonesia through to Papua New Guinea, Polynesia, the islands of the West Pacific and the sub-Antarctic islands (Pizzey and Knight, 2007).
Both domestic and wild ducks are commonly infected with gastrointestinal parasites including coccidia, which are obligate intracellular protozoans of the Apicomplexa (Gajadhar et al., 1983). Species of Eimeria Schneider, 1875 (Eimeriidae) are the most common coccidia found in birds, including ducks, with mixed infections being common (Leibovitz, 1968). Eimeria anatis Scholtyseck, 1955 infects the mallard Anas platyrhynchos (L.) (Duszynski et al., 2001) and Eimeria aythyae Farr, 1965 infects the lesser scaup Aythya affinis (Eyton) (Gajadhar et al., 1983; Duszynski et al., 2001). Windingstad et al. (1980) reported recurring epizootic infection in A. affinis resulting from infection with E. aythyae. The host range of Eimeria krylovi Svanbaev & Rakhmatullina, 1967 includes the green-winged teal Anas carolinensis Gmelin, the northern shoveler Spatula clypeata (L.), the European wigeon Mareca penelope (L.), the gadwall Mareca strepera (L.) and the garganey Spatula querquedula (L.) (Svanbaev & Rakhmatullina, 1967).
Coccidia of the genus Tyzzeria Allen, 1936, have also been described predominantly from ducks (Gajadhar et al., 1983). This genus is made up of coccidia whose oöcysts lack sporocysts (Duszynski et al., 1998). Cole & Friend (1999) reported that Tyzzeria spp. were less commonly seen in ducks than Eimeria spp. Tyzzeria perniciosa Allen, 1936 is an important pathogenic coccidian in ducks and is especially pathogenic for ducklings (Baker, 2007). Reported duck hosts of T. perniciosa are the northern pintail Anas acuta L., the lesser scaup A. affinis, the common shelduck Tadorna tadorna (L.), the tufted duck Aythya fuligula (L.), the mallard A. platyrhynchos and the white-headed duck Oxyura leucocephala (Scopoli) (Duszynski et al., 1998). A study from China reported outbreaks of coccidiosis due to T. perniciosa and Wenyonella philiplevinei Leibovitz, 1968, amongst farmed ducklings (Peiyun et al., 1982). A study in Iraq detected E. anatis in 17% and T. perniciosa in 11% of domesticated ducks screened (n = 80) (Abdullah, 2010) while another study on domestic ducks in Iran found a variety of protozoan parasites including Cryptosporidium spp., Tyzzeria spp., W. philiplevinei, Isospora mandari Bhatia, Chauhan, Arora & Agrawal, 1971 as well as other coccidian species (Larki et al., 2018).
The coccidia infecting ducks are similar in size and have very similar morphologies. This makes identification difficult using morphology alone (Leibovitz, 1968; Gajadhar et al., 1983). Those coccidia infecting wild ducks have not been well studied. In this study, we morphologically identified E. anatis, E. aythyae, E. krylovi and T. perniciosa from a Pacific black duck. Additionally, we provided genotypic characterization via sequencing of the mitochondrial cytochrome c oxidase subunit 1 (cox1) and the small subunit ribosomal RNA (18S) genes for E. anatis.
2. Materials and methods
2.1. Sample collection and examination
A wild, juvenile Pacific black duck was admitted to the Kanyana Wildlife Rehabilitation Centre (KWRC), Perth, Australia, in January 2021, after it was struck by a motor vehicle. Physical examination on admission revealed no external injuries; however, the duck was extremely quiet, reluctant to walk and was leaning to one side. It had a body condition score of 2/5. The duck was given supportive treatment of fluids and pain relief medication before being sent to a veterinarian for further assessment. A preliminary diagnosis of concussion and possible internal injuries was made. A faecal sample was collected on admission to KWRC. Initial direct light microscopy revealed a heavy, mixed parasitic load including large numbers of unsporulated coccidian oöcysts of various sizes as well as trophozoites of Trichomonas Donné, 1836, eggs of Capillaria Zeder, 1800 and tapeworm eggs. The duck was treated for the worm infection with praziquantel and moxidectin (20 mg/kg and 1 mg/kg of each ingredient respectively), per os (PO), once daily (OD), which was repeated after 14 days. The coccidia were treated with toltrazuril (15 mg/kg, PO, OD) for three consecutive days and then again 7 days later. Metronidazole (50 mg/kg, PO, OD) was given for 7 days for the Trichomonas infection. The duck made a full recovery and was released near the found location 4 weeks later.
A portion of faeces was placed in 2% (w/v) K2Cr2O7, mixed well and placed in a refrigerator, until transport to Murdoch University (within 48 h) for further investigation. On arrival at the Murdoch University laboratory, the faecal solution was poured into a Petri dish (to a depth of less than 1 cm). The Petri dish was stored in a dark environment and kept at room temperature (22 °C), to facilitate sporulation. The sample was checked daily for oöcyst sporulation using an Olympus DP71 digital microimaging camera. Sporulated oöcysts were observed using the 100× oil immersion objective. Images were taken using Nomarski contrast with a 100× oil immersion objective. Line drawings were edited using two software applications of CorelDRAW® (Corel Draw Graphics Suite, Version, 2020; Corel Corporation, Canada), i.e. Corel DRAW and Corel PHOTO-PAINT. All measurements are in micrometres and are given as the range followed by the mean in parentheses.
2.2. Oöcyst isolation, DNA extraction, PCR amplification, sequencing and phylogenetic analyses
Five morphologically similar oöcysts were isolated for a bulk DNA extraction with the method described by Yang et al. (2015). The DNA extraction, PCR amplification of the 18S rRNA and cox1 genes and sequencing were conducted according to the protocols described by Yang et al. (2013, 2016).
Phylogenetic trees were constructed for E. anatis using partial 18S rDNA and partial cox1 sequences aligned with additional species/isolates from GenBank using ClustalW (http://www.phylogeny.fr/one_task.cgi?task_type=clustalw). Distance analyses and phylogenies were conducted using MEGA X (Kumar et al., 2018) as described in detail by Yang et al. (2021) with the most appropriate nucleotide substitution models (TN93 + G + I for 18S and TN93 + G for the cox1 gene). Bootstrap support was estimated from 1000 pseudoreplicates.
3. Results
Based on the morphological analysis of the coccidian oöcysts in the faecal sample, four species were identified: E. anatis, E. aythyae, E. krylovi and T. perniciosa. The newly collected material is described below.
3.1. Eimeria anatis Scholtyseck, 1955
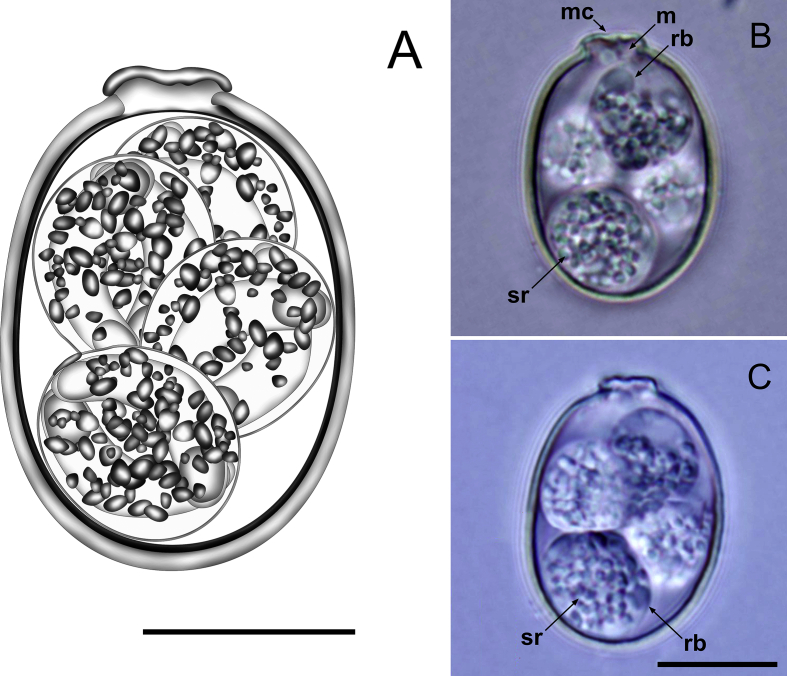
[Description based on 20 oöcysts and 40 sporocysts; Fig. 1.] Oöcysts elongate-ovoidal, 17–19 × 11–13 (17.6 × 11.9); length/width (L/W) ratio 1.4–1.6 (1.5). Oöcyst wall bi-layered, 0.9–1.3 (1.0) thick; outer layer smooth to slightly rough, c.2/3 of total thickness. Micropyle cap absent. Micropyle present, generally with invagination of inner layer. Oöcyst residuum absent, but 1–2 polar granules present. Sporocysts 4, ellipsoidal, 7–9 × 5–6 (7.9 × 5.9); L/W ratio 1.3–1.4 (1.3). Stieda body present, flattened; sub-Stieda absent or indiscernible; para-Stieda body absent. Sporocyst residuum present, composed of small, randomly dispersed granules. Sporozoites 2, with robust anterior and posterior refractile bodies and indiscernible nucleus.
Fig. 1.
Composite line drawing (A) and photomicrographs (B, C) of sporulated oöcysts of Eimeria anatis from the Pacific black duck Anas superciliosa. Note the anterior (arb) and posterior (prb) refractile bodies; micropyle (m); polar granule (pg); rough oöcyst wall (row); Stieda body (sb); and sporocyst residuum (sr). Scale-bars: 10 μm.
3.2. Eimeria aythyae Farr, 1965
[Description based on 20 oöcysts and 40 sporocysts; Fig. 2.] Oöcysts ellipsoidal, 20–23 × 14–16 (21.3 × 15.2); L/W ratio 1.3–1.5 (1.4). Oöcyst wall bi-layered, 1.0–1.4 (1.2) thick; outer layer smooth, c.2/3 of total thickness. Micropyle cap present as a translucent, delicate, curved protrusion. Micropyle present with no invagination of inner layer. Oöcyst residuum and polar granule absent. Sporocysts 4, ellipsoidal, 9–11 × 7–8 (10.5 × 7.7); L/W ratio 1.2–1.4 (1.4). Stieda body present, flattened; sub-Stieda present, but delicate or indiscernible in some sporocysts; para-Stieda body absent. Sporocyst residuum present, composed of large, randomly dispersed granules. Sporozoites 2, with robust anterior and posterior refractile bodies and centrally located nucleus.
Fig. 2.
Composite line drawing (A) and photomicrographs (B, C) of sporulated oöcysts of Eimeria aythyae from the Pacific black duck Anas superciliosa. Note the micropyle (m); micropyle cap (mc); nucleus (n); Stieda body (sb); sporocyst residuum (sr); and refractile body (rb). Scale-bars: 10 μm.
3.3. Eimeria krylovi Svanbaev & Rakhmatullina, 1967
[Description based on 25 oöcysts and 50 sporocysts; Fig. 3.] Oöcysts ellipsoidal, 20–23 × 16–17 (21.7 × 16.1); L/W ratio 1.3–1.4 (1.3). Oöcyst wall bi-layered, 1.0–1.4 (1.2) thick; outer layer smooth, c.2/3 of total thickness. Micropyle cap present as a dense cover. Micropyle present with no invagination of inner layer. Oöcyst residuum and polar granule absent. Sporocysts 4, subspheroidal to ellipsoidal, 8–10 × 7–8 (8.8 × 7.7); L/W ratio 1.1–1.2 (1.1). Stieda body flattened, barely or not discernible; sub-Stieda absent; para-Stieda body absent. Sporocyst residuum present, composed of many large and dense granules which are widely diffused within the sporocyst. Sporozoites 2, with anterior and posterior refractile bodies and indiscernible nucleus.
Fig. 3.
Composite line drawing (A) and photomicrographs (B, C) of sporulated oöcysts of Eimeria krylovi from the Pacific black duck Anas superciliosa. Note the micropyle (m); micropyle cap (mc); sporocyst residuum (sr); and refractile body (rb). Scale-bars: 10 μm.
3.4. Tyzzeria perniciosa Allen, 1936
[Description based on 25 oöcysts; Fig. 4.] Oöcysts ellipsoidal, 10–11 × 7–8 (10.7 × 7.4); L/W ratio 1.4–1.5 (1.4). Oöcyst wall bi-layered, 0.4–0.7 (0.6) thick; outer layer smooth, c.2/3 of total wall thickness. Oöcyst residuum present as granules of different sizes usually clustered at one end of oöcyst, measuring c.2.5. Sporozoites 8, curved and tapered at anterior end, 6–8 × 1–2 (6.9 × 1.8), with robust, prominent posterior refractile body and without discernible nucleus.
Fig. 4.
Composite line drawing (A) and photomicrographs (B, C) of sporulated oöcysts of Tyzzeria perniciosa from the Pacific black duck Anas superciliosa. Note the inner (il) and outer (ol) layers of the oöcyst wall; oöcyst residuum (or); refractile body (rb); and sporozoite (sz). Scale-bars: 10 μm.
3.5. Molecular identification
PCR amplification for the 18S rRNA and cox1 genes from oöcyst DNA of the four coccidian species were conducted; unfortunately, PCR amplicons were successfully obtained only for E. anatis oöcysts.
3.5.1. Phylogenetic analyses of the 18S rRNA gene
A 1209 bp 18S rDNA sequence with clean sequencing chromatography was obtained from the five morphological similar oöcysts of E. anatis isolated from the faecal samples of Anas superciliosa; this was aligned with 37 sequences for Eimeria spp., 5 for Cyclospora spp. and 2 for Isospora spp. based on the NCBI BLAST similarities. The alignment covered all available Eimeria spp. sequences. A 18S rRNA gene sequence (GenBank: L24381) of Toxoplasma gondii (Nicolle & Manceaux, 1908) was used as the outgroup. Eimeria anatis shared 97.6% and 96.6% similarity with Eimeria stigmosa Klimes, 1963 (GenBank: KP789171) and Eimeria anseris Kotlan, 1932 (GenBank: KJ000077), respectively, both of which were obtained from Anser anser (L.) in China (sequences published in GenBank only). Eimeria anatis also shared a genetic similarity of 94.6% with both Eimeria gruis Yakimoff & Matschoulsky, 1935 (GenBank: AB544336) and Eimeria reichenowi Yakimoff & Matschoulsky, 1935 (GenBank: AB544314), both identified from Grus monacha Temminck in Japan and reported by the same group (Honma et al., 2011). In addition, E. anatis shared 93.0% similarity with Eimeria paludosa (Leger & Hesse, 1922) (GenBank: KJ767187) from Gallinula tenebrosa Gould in Western Australia (Yang et al., 2014). As shown in Fig. 5A, E. anatis was placed in a separate strongly supported clade with E. stigmosa and E. anseris, closely associated with a sister clade composed of E. gruis, E. reichenowi and E. paludosa.
Fig. 5.
Evolutionary relationships of Eimeria anatis inferred by maximum likelihood analysis (ML) of 18S rDNA sequences (A, alignment length 1209 bp; B, alignment length 424 bp). Percentage support (> 70%) from 1000 pseudoreplicates from the ML analysis is indicated at the nodes.
Eimeria anatis is often related to the coccidian species W. philiplevinei; however, there is no 18S DNA sequence from W. philiplevinei available, only a 422-bp 18S sequence presented in the paper by Wu et al. (2013). The 18S sub-tree generated from a shortened alignment including both E. anatis and W. philiplevinei showed that E. anatis belongs to the same clade as that of the 18S phylogenetic tree based on the long alignment (Fig. 5A), whereas W. philiplevinei was positioned close to T. gondii, outside of the Eimeria spp. clades (Fig. 5B). The genetic similarity between E. anatis and W. philiplevinei was 86.5%.
3.5.2. Phylogenetic analyses of the cox1 gene
The cox1 gene was amplified from E. anatis oöcyst DNA and a 650-bp sequence was successfully obtained and aligned with 21 sequences for Eimeria spp. from different animal species, 4 for Isospora spp. and one for Caryospora sp. All cox1 reference sequences were selected based on the NCBI BLAST similarities and covered all Eimeria spp. in the database. A sequence for T. gondii (GenBank: HM771690) was used as the outgroup. Eimeria anatis showed the highest genetic similarity (91.9%) with an unnamed Eimeria sp. isolated from the pink-footed goose Anser brachyrhynchus Baillon (GenBank: MT833388) (Myšková et al., 2021) and grouped with this Eimeria sp. in the same clade in the phylogenetic tree (Fig. 6).
Fig. 6.
Evolutionary relationships of Eimeria anatis inferred by maximum likelihood analysis (ML) of partial cox1 gene sequences (650 bp). Percentage support (> 70%) from 1000 pseudoreplicates from the ML analysis is indicated at the nodes.
4. Discussion
Eimeria anatis, E. aythyae, E. krylovi and T. perniciosa have all been previously reported to infect ducks. The Pacific black duck in this study was found to be infected with all three of these Eimeria spp. simultaneously as well as T. perniciosa. This is not unusual as co-infections with Eimeria spp. are regularly observed in birds.
The oöcysts of the three Eimeria spp. identified in this study were morphologically compatible with their respective original descriptions (Table 1, Table 2). However, it is noteworthy that in the present study some adjustments for some characteristic features were added to the descriptions of these species. For example, a Stieda body and a sub-Stieda body were observed in E. anatis and E. aythyae, respectively, which were not identified in the original descriptions (Scholtyseck, 1955; Farr, 1965) or in later reports (Gajadhar et al., 1983). Eimeria krylovi was not originally described with a Stieda body, and indeed this structure was hardly observed and photomicrographed in this study, being reported here as “barely or not discernible”. In this context, it is important to highlight that a Stieda body is a synapomorphic characteristic of the family Eimeriidae, and its lack indicates a change in the identification to another genus and/or family, such as the genera Choleoeimeria Paperna & Landsberg, 1989, Acroemeria Paperna & Landsberg, 1989, Goussia Labbé, 1896, etc., which were originally described within Eimeria, but due to the absence of a Stieda body, among other aspects, classified in other genera (Jirků et al., 2009). In the present study, these three Eimeria spp. were redrawn and redescribed to offer better characterization of features such as micropyle, micropyle cap, Stieda body, sub-Stieda body, sporocyst residuum and refractile bodies and sporozoite nucleus, aiming to facilitate and enable reliable identification of these species in further studies.
Table 1.
Comparative morphology of the oöcysts of Eimeria spp. recorded from ducks (Anseriformes: Anatidae: Anatinae)
| Species | Host | Shape | Size (μm)a | Shape index | Polar granule | Wall (μm)a | Micropyle (μm)a | Reference |
|---|---|---|---|---|---|---|---|---|
| Eimeria abramovi Svanbaev & Rakhmatullina, 1967 | Anas platyrhynchos (L.) | Ovoidal to ellipsoidal | 21–22 × 16–17 | – | Absent | Smooth, (1.4) thick | Present, (2.4) wide, with micropyle cap | Svanbaev & Rakhmatullina, 1967 |
| Eimeria anatis Scholtyseck, 1955 | A. platyrhynchos | Ovoidal | 14–19 × 11–16 (16.8 × 14.1) | – | – | Smooth, 0.7–1.0 thick | Present, closed by a plug-like mass | Scholtyseck (1955) |
| Anas superciliosa Gmelin | Elongate-ovoidal | 17–19 × 11–13 (17.6 × 11.9) | 1.4–1.6 (1.5) | Present, 1–2 | Smooth to slightly rough, 0.9–1.3 (1.0) thick | Present, with an invagination of the inner layer, without micropyle cap | Present study | |
| Eimeria aythyae Farr, 1965 | Aythya affinis (Eyton) | Broadly ellipsoidal to a round-bottomed urn with shoulder | 15–24 × 10–18 (20.1 × 15.5) | – | Absent | Smooth or lightly sculptured, 0.6–0.8 thick | Present, (3.6) wide | Farr (1965) |
| A. superciliosa | Ellipsoidal | 20–23 × 14–16 (21.3 × 15.2) | 1.3–1.5 (1.4) | Absent | Smooth, 1.0–1.4 (1.2) thick | Present, without invagination of the inner layer; micropyle cap as a translucent and delicate curved protrusion | Present study | |
| Eimeria battakhi Dubey & Pande, 1963 | A. platyrhynchos | Subspherical to ovoidal | 19–24 × 16–21 (21.0 × 18.0) | 1.1–1.2 | Present, 1 | Smooth, 1.0–2.0 thick | Absent | Dubey & Pande (1963) |
| Eimeria boschadis Waldén, 1961 | A. platyrhynchos | Bottle-shaped | 18–27 × 12–13 (23.9 × 12.7) | – | – | Finely granulated | Present, 2–3 wide | Waldén (1961) |
| Eimeria bucephalae Christiansen & Madsen, 1948 | Bucephala clangula (L.) | Elongate-ovoidal | 25–39 × 13–20 (30.3 × 15.6) | – | – | – | Present, narrow | Christiansen & Madsen (1948) |
| Eimeria danailovi Grafner, Graubmann & Betke, 1965 | Anas platyrhynchos | Ovoidal | 19–23 × 11–15 (18.7 × 12.5) | – | – | 0.6–1.0 thick | Present | Gräfner et al. (1965) |
| Eimeria koganae Svanbaev & Rakhmatullina, 1967 | Spatula querquedula (L.) | Ovoidal or ellipsoidal | 21–25 × 13–21 (21.5 × 16.1) | – | Absent | 0.8–1.0 thick | Present, (5.0) wide | Svanbaev & Rakhmatullina, 1967 |
| Eimeria krylovi Svanbaev & Rakhmatullina, 1967 | Anas carolinensis Gmelin; Spatula clypeata (L.); S. querquedula; Mareca penelope (L.); Mareca strepera (L.) | Subspherical | 15–21 × 13–17 | – | Present, 1 | Smooth, 1.2 thick | Present at the flattened end, 4.0–6.0 wide, covered by a (4.0) wide and (2.0) high micropyle cap | Svanbaev & Rakhmatullina, 1967 |
| Anas superciliosa Gmelin | Ellipsoidal | 20–23 × 16–17 (21.7 × 16.1) | 1.3–1.4 (1.3) | Absent | Smooth, 1.0–1.4 (1.2) thick | Present, without invagination of the inner layer, with micropyle cap as a dense cover | Present study | |
| Eimeria nyroca Svanbaev & Rakhmatullina, 1967 | Aythya nyroca (Güldenstädt) | Ovoidal | 21–40 × 17–19 (25.4 × 17.7) | – | Absent | Smooth, 1.0–2.0 thick | Present, surrounded by collars, 4.0–6.0 wide, with 2.0–3.0 high micropyle cap | Svanbaev & Rakhmatullina, 1967 |
| Eimeria saitamae Inoue, 1967 | A. platyrhynchos | Ovoidal | 17–21 × 13–15 (18.6 × 13.2) | – | Present, 1 | Smooth, 0.7–0.8 thick | Present | Inoue (1967) |
| Eimeria schachdagica Musaev, Surkova, Jelchiev & Alieva, 1966 | A. platyrhynchos | Ovoidal | 16–26 × 12–20 (24.0 × 17.7) | – | Present, refractile granules | Smooth, 1.6–2.0 thick | Absent | Musaev et al. (1966) |
| Eimeria somateriae Christiansen, 1952 | Clangula hyemalis (L.) | Bottle-shaped, asymmetric | 21–41 × 11–19 (31.9 × 13.9) | – | – | Smooth, thin, colorless | Present | Christiansen (1952) |
Note: E. boschadis and E. somateriae are kidney parasites, while the remaining species are intestinal parasites.
Range (Mean).
Table 2.
Comparative morphology of the sporocysts of Eimeria spp. recorded from ducks (Anseriformes: Anatidae: Anatinae)
| Species | Host | Shape | Size (μm)a | Shape index | Stieda body | Sub-Stieda body | Sporocyst residuum | Reference |
|---|---|---|---|---|---|---|---|---|
| Eimeria abramovi Svanbaev & Rakhmatullina, 1967 | Anas platyrhynchos (L.) | – | 7–9 × 5 | – | – | – | Present, small granules | Svanbaev & Rakhmatullina, 1967 |
| Eimeria anatis Scholtyseck, 1955 | A. platyrhynchos | Ovoidal | – | – | – | – | Present, few central granules | Scholtyseck (1955) |
| Anas superciliosa Gmelin | Ellipsoidal | 7–9 × 5–6 (7.9 × 5.9) | 1.3–1.4 (1.3) | Present, flattened | Absent or indiscernible | Present, small granules randomly dispersed | Present study | |
| Eimeria aythyae Farr, 1965 | Aythya affinis (Eyton) | – | – | – | Present, small | – | Present, compact residual mass | Farr (1965) |
| A. superciliosa | Ellipsoidal | 9–11 × 7–8 (10.5 × 7.7) | 1.2–1.4 (1.4) | Present, flattened | Present, delicate or indiscernible | Present, large granules randomly dispersed | Present study | |
| Eimeria battakhi Dubey & Pande, 1963 | A. platyrhynchos | Ovoidal | 11–13 × 6–8 | – | Present, small | – | Present | Dubey & Pande (1963) |
| Eimeria boschadis Waldén, 1961 | A. platyrhynchos | – | – | – | – | – | – | Waldén (1961) |
| Eimeria bucephalae Christiansen & Madsen, 1948 | Bucephala clangula (L.) | – | – | – | – | – | – | Christiansen & Madsen (1948) |
| Eimeria danailovi Grafner, Graubmann & Betke, 1965 | Anas platyrhynchos (L.) | Ovoidal | – | – | – | – | Present | Gräfner et al. (1965) |
| Eimeria koganae Svanbaev & Rakhmatullina, 1967 | Spatula querquedula (L.) | Subspherical to ovoidal | 9–11 × 8–10 | – | – | – | Present, clear globules irregularly spaced | Svanbaev & Rakhmatullina, 1967 |
| Eimeria krylovi Svanbaev & Rakhmatullina, 1967 | Anas carolinensis Gmelin; Spatula clypeata (L.); S. querquedula; Mareca penelope (L.); Mareca strepera (L.) | Subspherical or ovoidal | 8 × 6–8 | – | – | – | Absent | Svanbaev & Rakhmatullina, 1967 |
| Anas superciliosa Gmelin | Subspheroidal to ellipsoidal | 8–10 × 7–8 (8.8 × 7.7) | 1.1–1.2 (1.1) | Barely or not discernible | Absent | Present, large, dense granules diffused | Present study | |
| Eimeria nyroca Svanbaev & Rakhmatullina, 1967 | Aythya nyroca (Güldenstädt) | Ovoidal | 11–13 × 8–11 | – | – | – | Present, granular | Svanbaev & Rakhmatullina, 1967 |
| Eimeria saitamae Inoue, 1967 | A. platyrhynchos | – | – | – | – | – | – | Inoue (1967) |
| Eimeria schachdagica Musaev, Surkova, Jelchiev & Alieva, 1966 | A. platyrhynchos | Ovoidal | (9.2 × 8.4) | – | – | – | Present, small, granular | Musaev et al. (1966) |
| Eimeria somateriae Christiansen, 1952 | Clangula hyemalis (L.) | – | (11 × 6) | – | – | – | Absent | Christiansen (1952) |
Note: E. boschadis and E. somateriae are kidney parasites, while the remaining species are intestinal parasites.
Range (Mean).
Morphologically, E. anatis is easily confused with W. philiplevinei due to the difficulty of distinguishing the sporocyst residuum and the number of sporozoites in their sporocysts. In this context, Duszynski et al. (2000) considered that the descriptions, photomicrographs and line drawings of Wenyonella spp. were inadequate; additionally, many species have been described and named from degenerate oöcysts. Thus, Duszynski et al. (2000) suggested that all species identified as Wenyonella should be viewed dubiously and considered species inquiriendae. Specifically for W. philiplevinei, Duszynski et al. (2000) considered that both the line drawing and photomicrograph suggest that the refractile bodies and/or sporozoites were all confused in the original description of Leibovitz (1968) and other studies reviewed by Gajadhar et al. (1983). In the oöcysts identified as E. anatis in the present study, two sporozoites were clearly observed with their anterior and posterior refractile bodies in each sporocyst, justifying that the material belongs to the genus Eimeria. Furthermore, the results of the phylogenetic analysis including the newly generated 18S sequence for E. anatis showed its inclusion into a clade of Eimeria spp. from ducks, while being distant from the only partial 18S sequence from oöcysts identified as W. philiplevinei by Wu et al. (2013).
Tyzzeria perniciosa is the type-species of the genus Tyzzeria, which consists of coccidia with oöcysts containing eight sporozoites without sporocysts. All consensually valid species are recorded from birds of the order Anseriformes. Descriptions in hosts of other vertebrate classes were published, although they must be misidentifications, such as Tyzzeria boae Lainson & Paperna, 1994 described from the red-tailed boa Boa constrictor L. and Tyzzeria chalcides Probert, Roberts & Wilson, 1988 described from the ocellated skink Chalcides ocellatus (Forskål), which potentially represent a species of Klossiella and a species of Choleoeimeria, respectively misidentified from oöcysts that sporulated abnormally (Duszynski et al., 1998).
The most frequently reported species in the literature are T. perniciosa from teals, mallards and other ducks (Anatinae) and Tyzzeria parvula (Kotlan, 1933) from geese (Anserinae) (Berto et al., 2007). Although the oöcysts of these species are morphologically very similar, they are specialised for parasitism at the subfamily level, i.e. T. parvula does not infect teals, mallards and ducks, just as T. perniciosa does not infect geese, even in experimental infections (Berto et al., 2007).
In addition to T. perniciosa, Tyzzeria pellerdyi Bhatia & Pande, 1966 was described and reported from Anas spp. in some studies in the 1960s, 1970s and 1980s. However, there is no morphological or biological differentiation that so far justifies and fundamentally defines T. pellerdyi. As this species was described after T. perniciosa, it is likely that T. pellerdyi is a junior synonym of T. perniciosa (Gajadhar et al., 1983; Duszynski et al., 1998) (Table 3). Similarly, two species, i.e. Tyzzeria allenae Chakravarty & Basu, 1946 and Tyzzeria chenicusae Ray & Sarkar, 1967, were described from the cotton pygmy-goose Nettapus coromandelianus (Gmelin); this host, in spite of the common name, belongs to the Anatinae. The oöcysts of these species were described with some morphometric differences that differentiate them from the original description of T. perniciosa and from the description provided here; however, these species have not been reported since their original descriptions (Table 3).
Table 3.
Comparative morphological data for Tyzzeria spp. recorded from ducks (Anseriformes: Anatidae: Anatinae).
| Species | Host | Oöcyst |
Sporozoite |
Reference | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Shape | Size (μm)a | Shape index | Residuum (μm)a | Wall (μm)a | Shape | Size (μm)a | Refractile body | Nucleus | |||
| Tyzzeria perniciosa Allen, 1936 | Anas platyrhynchos L. | Ellipsoidal | 10–13 × 9–11 | – | Present, large, composed of variously sized granules | Relatively thick | Curved, with one end more rounded and broader | (10.0 × 3.5) | – | – | Allen (1936) |
| Anas superciliosa Gmelin | Ellipsoidal | 10–11 × 7–8 (10.7 × 7.4) | 1.4–1.5 (1.4) | Present, granules of different sizes usually clustered at one end of the oöcyst, c.2.5 | Smooth, 0.4–0.7 (0.6) | Curved and tapered at anterior end | 6–8 × 1–2 (6.9 × 1.8) | Robust, prominent | Not discernible | Present study | |
| Tyzzeria alleni Chakravarty & Basu, 1946 | Nettapus coromandelianus Gmelin | Ovoidal | 14–17 × 10–12 | – | Present, coarsely granular, c.6.4 | – | Tapered at one end | 5.3–6.5 | – | Present, central | Chakravarty & Basu (1946) |
| Tyzzeria pellerdyi Bhatia & Pande, 1966 | Mareca strepera (L.); Aythya nyroca (Güldenstädt); Spatula clypeata (L.); Anas carolinensis Gmelin; A. platyrhynchos | Subspherical to ovoidal | 11–16 × 8–11 (13.0 × 10.0) | – | Present, c.4.0–5.0 | Smooth, 0.5–0.7 | Banana-shaped | (8.5 × 2.0) | Prominent | Present, central | Bhatia & Pande (1966); Bristol et al. (1981) |
| Tyzzeria chenicusae Ray & Sarkar, 1967 | N. coromandelianus | Broad and cylindrical | 20–28 × 14–20 (24.8 × 16.8) | (1.5) | Large, compact, at one pole of the oöcyst | (1.4) | Club-shaped | (13.2 × 4.2) | Present at the broader end | – | Ray & Sarkar (1967) |
Note: All species are intestinal parasites.
Range (Mean).
Our phylogenetic analyses provided strong evidence that the newly generated sequences from E. anatis in the Pacific black duck both at the 18S rRNA and cox1 loci were most close to those from domestic goose (GenBank: KP789171, KJ000077 and MT833388). As this is, to the best of our knowledge, the first study using molecular tools to the identification of duck coccidia, further similar studies on additional species of coccidia parasitic in ducks would be beneficial to the taxonomy of duck coccidia and assessment of their relationships with coccidian species parasitic in other host groups.
This study has revealed that, besides infecting the mallard A. platyrhynchos, E. anatis also infects the Pacific black duck. Hybridisation (interbreeding) between the introduced mallard and the Pacific black duck in Australia occurs at a rate of around 1.5% (Taysom, 2016), so it is likely that these two species of duck share some of their coccidian species as well.
5. Conclusion
In conclusion, the coccidia E. anatis, E. aythyae, E. krylovi and T. perniciosa are redescribed with supplementary morphological data, in order to ensure and facilitate their future identification from A. superciliosa or from other duck species. In addition, a genotypic characterization of E. anatis and taxonomic remarks on species and genera of dubious validity reported from Anseriformes are provided, aiming to contribute to the knowledge of coccidian species of ducks.
CRediT author statement
Bruno P. Berto: morphological identification of the species, preparation of line drawings, writing - review & editing. Belinda Brice: coccidian primary screening and identification, writing - original draft and paper reviewing. Gwyneth Thomas: sample collection and coccidian primary screening, writing - review & editing. Aileen Elloit: oöcyst imaging, morphological identification of the species, writing - review & editing. Alireza Zahedi: oöcyst isolation, DNA extraction, PCR, sequencing, writing - review & editing. Rongchang Yang: overseeing and coordinating this study, phylogenetic analysis, writing - review & editing. All authors read and approved the final manuscript.
Data availability
The newly generated sequences for E. anatis are deposited in the GenBank database under the accession numbers OL604501 (18S rDNA) and OL656104 (cox1). Photomicrographs and line drawings of the oöcysts are deposited and available (http://r1.ufrrj.br/labicoc/colecao.html) in the Parasitology Collection of the Laboratório de Biologia de Coccídios, at UFRRJ, under repository numbers 120/2021 (E. anatis), 121/2021 (E. aythyae), 122/2021 (E. krylovi) and 123/2021 (T. perniciosa), along with the photovouchers of the A. superciliosa specimen.
Ethical approval
Not applicable.
Funding
Official funding for this study was not available.
Declaration of competing interests
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgements
The authors would like to thank Helen Riley (KWRC Chair) and all the KWRC volunteers, as well as Daniel Oakes for help with initial microscopy screening. We are also grateful to the staff at the Wattle Grove Veterinary Hospital, Perth, for their care and treatment of wildlife treated at their hospital. Bruno P. Berto thanks fellowships from CNPq (Grant/Award Number: 303899/2019-0) and FAPERJ (Grant/Award Number: E-26/202.797/2019).
Contributor Information
Bruno P. Berto, Email: bertobp@ufrrj.br.
Rongchang Yang, Email: R.Yang@murdoch.edu.au.
References
- Abdullah D.A. Coccidiosis in domesticated ducks in Ninevah Governorate. Iraqi J. Vet. Sci. 2010;24:149–153. [Google Scholar]
- Allen E.A. Tyzzeria pernicosa gen. et sp. nov., a coccidium from the small intestine of the Pekin duck Anas domesticus L. Arch. Protistenkd. 1936;87:262–267. [Google Scholar]
- Baker D.G. Blackwell Publishing; Hoboken, USA: 2007. Flynn's Parasites of Laboratory Animals. [Google Scholar]
- Berto B.P., Teixeira M., Lopes C.W.G. Tyzzeria parvula (Kotlan, 1933) Klimes, 1963 (Apicomplexa: Eimeriidae) in the greylag goose (Anser anser Linnaeus, 1758) in southeastern Brazil. Rev. Bras. Parasitol. Vet. 2007;16:156–158. doi: 10.1590/s1984-29612007000300008. [DOI] [PubMed] [Google Scholar]
- Bhatia B.B., Pande B.P. On two new species of coccidia from wild Anatidae. Acta Vet. Acad. Sci. Hung. 1966;16:335–340. [PubMed] [Google Scholar]
- Bristol J.R., Mayberry L.F., Bueno R., Canaris A.G. Coccidia of surface-feeding ducks from the central flyway, Texas. J. Parasitol. 1981;67:276–277. [PubMed] [Google Scholar]
- Chakravarty M., Basu S.P. On a new coccidium Tyzzeria alleni n. sp. from the intestine of the bird cotton-teal. Sci. Cult. 1946;12:106. [Google Scholar]
- Christiansen M. Renal coccidiosis in wild ducklings (Anseriformes). Nord. Veterinaermed. 1952;4:1173–1191. (In Danish) [Google Scholar]
- Christiansen M., Madsen H. Eimeria bucephalae n. sp. pathogenic in goldeneye in Denmark. Dan. Rev. Game Biol. 1948;1:62–73. [Google Scholar]
- Cole R.A., Friend M. University of Nebraska; Lincoln, USA: 1999. Parasites and Parasitic Diseases (Field Manual of Wildlife Diseases)https://digitalcommons.unl.edu/zoonoticspub/15 [Google Scholar]
- Dubey J.P., Pande B.P. A preliminary note on Eimeria battakhi, n. sp. (Protozoa: Eimeriidae) from domestic duck (Anas platyrhynchos domesticus) Curr. Sci. 1963;32:329–331. [Google Scholar]
- Duszynski D.W., Couch L., Upton S.J. Kansas State University; 1998. The coccidian genus Tyzzeria.https://www.k-state.edu/parasitology/worldcoccidia/TYZZERIA [Google Scholar]
- Duszynski D.W., Couch L., Upton S.J. Kansas State University; 2000. The coccidian genus Wenyonella.https://www.k-state.edu/parasitology/worldcoccidia/WENYONELLA [Google Scholar]
- Duszynski D.W., Couch L., Upton S.J. Kansas State University; 2001. The coccidia of Anseriformes (ducks, geese and swans)https://www.k-state.edu/parasitology/worldcoccidia/ANSERIFORMES [Google Scholar]
- Farr M.M. Coccidiosis of the lesser scaup duck, Aythya affinis (Eyton, 1838), with a description of a new species, Eimeria aythyae. Proc. Helminthol. Soc. Wash. 1965;32:236–238. [Google Scholar]
- Gajadhar A.A., Wobeser G., Stockdale P.H.G. Coccidia of domestic and wild waterfowl (Anseriformes) Can. J. Zool. 1983;61:1–24. [Google Scholar]
- Gräfner G., Graubmann H.D., Betke P. Dunndarm kokzidiose bei Hausenten, verursacht durch eine neue Kokzidienart, Eimeria danailovi n. sp. Mh. Vet. Med. Nachr. 1965;20:141–143. [Google Scholar]
- Honma H., Suyama Y., Watanabe Y., Matsumoto F., Nakai Y. Accurate analysis of prevalence of coccidiosis in individually identified wild cranes in inhabiting and migrating populations in Japan. Environ. Microbiol. 2011;13:2876–2887. doi: 10.1111/j.1462-2920.2011.02563.x. [DOI] [PubMed] [Google Scholar]
- Inoue I. Eimeria saitamae n. sp.: a new cause of coccidiosis in domestic ducks (Anas platyrhyncha var. domestica) Jpn. J. Vet. Sci. 1967;29:209–215. doi: 10.1292/jvms1939.29.209. [DOI] [PubMed] [Google Scholar]
- Jirků M., Jirků M., Oborník M., Lukeš J., Modrý D. Goussia Labbé, 1896 (Apicomplexa, Eimeriorina) in Amphibia: diversity, biology, molecular phylogeny and comments on the status of the genus. Protist. 2009;160:123–136. doi: 10.1016/j.protis.2008.08.003. [DOI] [PubMed] [Google Scholar]
- Kumar S, Stecher G, Li M, Knyaz C, Tamura K. MEGA X: Molecular Evolutionary Genetics Analysis across computing platforms. Mol. Biol. Evol. 2018;35:1547–1549. doi: 10.1093/molbev/msy096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Larki S., Alborzi A., Chegini R., Amiri R. A preliminary survey of gastrointestinal parasites of domestic ducks in Ahvaz, southwest Iran. Irananian J. Parasitol. 2018;13:137–144. [PMC free article] [PubMed] [Google Scholar]
- Leibovitz L. Wenyonella philiplevinei, n. sp., a coccidial organism of the white Pekin duck. Avian Dis. 1968;12:670–681. [PubMed] [Google Scholar]
- Musaev M.A., Surkova A.M., Jelchiev J.A., Alieva F.K. New kind of coccidia from the genus Eimeria from the domestic duck (Anas domestica) Acad. Sci. Azerbaidj. S.S.R. Ser. Biol. Sci. 1966;1:34–36. [Google Scholar]
- Myšková E., Brož M., Kváč M., Sak B., Ditrich O. Intestinal parasites of dogs (Canis lupus familiaris) in Svalbard (Norway): low prevalence and limited transmission with wildlife. Can. J. Zool. 2021;99:249–255. [Google Scholar]
- Peiyun Y., Jinshu J., Kunhua L., Guiying L., Liye Q. A preliminary investigation of the coccidian species of ducks in Beijing. Acta Vet. Zootech. Sin. 1982;13:119–124. [Google Scholar]
- Pizzey G., Knight F. Harper Collins Publishers Pty Limited; Sydney: 2007. The field guide to the birds of Australia. [Google Scholar]
- Ray H.N., Sarkar A. On a new coccidium, Tyzzeria chenicusae n. sp., from cotton teal (Chenicus coromendelianus: Aves Anseriformes) J. Protozool. 1967;14:27. [Google Scholar]
- Scholtyseck E. Eimeria anatis n. sp., ein neue Coccid aus der Stockente (Anas platyrhynchos) Arch. Protistenkd. 1955;100:431–434. [Google Scholar]
- Svanbaev K., Rakhmatullina N.K. New kind of coccidia of wild ducks. Farm News Sci. 1967;10:46–52. [Google Scholar]
- Taysom A.J. Victoria University; Melbourne, Australia: 2016. The occurrence of hybridisation between the Pacific black duck (Anas superciliosa) and other dabbling ducks (Genus: Anas) in Australia.https://vuir.vu.edu.au PhD Thesis, [Google Scholar]
- Waldén H.W. Observations on renal coccidia in Swedish anseriform birds, with notes concerning two new species, Eimeria boschadis, and Eimeria christianseni (Sporozoa, Telosporidia) Arch. Zool. 1961;15:97–104. [Google Scholar]
- Windingstad R.M., McDonald M.E., Locke L.N., Kerr S.M., Sinn J.A. Epizootic of coccidiosis in free-flying lesser scaup. Avian Dis. 1980;24:1044–1049. [PubMed] [Google Scholar]
- Wu H.L., Hu P.H., Zhang J. Molecular characterization of Wenyonella philiplevinei from ducks in Hunan Province, China. Thai J. Vet. Med. 2013;43:605–608. [Google Scholar]
- Yang R., Murphy C., Song Y., Ng-Hublin J., Estcourt A., Hijjawi N., et al. Specific and quantitative detection and identification of Cryptosporidium hominis and C. parvum in clinical and environmental samples. Exp. Parasitol. 2013;135:142–147. doi: 10.1016/j.exppara.2013.06.014. [DOI] [PubMed] [Google Scholar]
- Yang R., Brice B., Ryan U. Morphological and molecular characterization of Eimeria paludosa coccidian parasite (Apicomplexa: Eimeriidae) in a dusky moorhen (Gallinula tenebrosa Gould, 1846) in Australia. Exp. Parasitol. 2014;147:16–22. doi: 10.1016/j.exppara.2014.10.010. [DOI] [PubMed] [Google Scholar]
- Yang R., Brice B., Elliot A., Lee E., Ryan U. Eimeria collieie n. sp. (Apicomplexa: Eimeriidae) from the western long-necked turtle (Chelodina colliei) Exp. Parasitol. 2015;154:75–81. doi: 10.1016/j.exppara.2015.04.019. [DOI] [PubMed] [Google Scholar]
- Yang R., Brice B., Jian F., Ryan U. Morphological and molecular characterization of Isospora manorinae n. sp. in a yellow-throated miner (Manorina flavigula wayensis) (Gould, 1840) Exp. Parasitol. 2016;163:16–23. doi: 10.1016/j.exppara.2016.01.013. [DOI] [PubMed] [Google Scholar]
- Yang R., Brice, Berto B.P., Zahedi A. Morphological and molecular characterization of a new species of Isospora Schneider, 1881 (Apicomplexa: Eimeriidae) from the western wattlebird Anthochaera lunulata Gould in western Australia. Curr. Res. Parasitol. Vector Borne Dis. 2021;1:100050. doi: 10.1016/j.crpvbd.2021.100050. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The newly generated sequences for E. anatis are deposited in the GenBank database under the accession numbers OL604501 (18S rDNA) and OL656104 (cox1). Photomicrographs and line drawings of the oöcysts are deposited and available (http://r1.ufrrj.br/labicoc/colecao.html) in the Parasitology Collection of the Laboratório de Biologia de Coccídios, at UFRRJ, under repository numbers 120/2021 (E. anatis), 121/2021 (E. aythyae), 122/2021 (E. krylovi) and 123/2021 (T. perniciosa), along with the photovouchers of the A. superciliosa specimen.