Abstract
In this study we investigated the interactions between capsular and acapsular strains of Cryptococcus neoformans and blood platelets. In vivo microscopic observation of blood samples from mice inoculated with C. neoformans yeast cells demonstrated that encapsulated and nonencapsulated yeast cells disappeared quickly from the bloodstream and that platelets were attached solely to yeast cells of the nonencapsulated strains. In vitro we observed that only the acapsular strains were susceptible to the fungicidal activity of thrombin-induced platelet microbicidal proteins.
Cryptococcus neoformans is an encapsulated fungal organism, one of the most common life-threatening fungal pathogens that infect patients with AIDS (27). The established virulence factors of C. neoformans are the polysaccharidic capsule, the possibility for growth at 37°C in mammalian hosts, and the ability to synthesize melanin pigments (for reviews see references 4, 5, and 27).
It is generally accepted that the organism enters the host by the respiratory route in the form of either yeast cells or basidiospores (18, 27). Alveolar macrophages constitute the first line of defense against C. neoformans (12). The inflammatory response to C. neoformans, with an influx of neutrophils and monocytes, affords a second line of defense (5, 28). Since the yeast has a tropism for the brain, infected individuals usually develop meningoencephalitis (18). The organism hematogenously spreads to other tissues (1), and fungemia is detected during clinical (8, 15, 24, 32) and experimental (25) infections. During this stage in the blood stream, the fungus can interact with blood cells such as leukocytes, e.g., neutrophils, monocytes (23, 25, 26), and eosinophils (11), and with plasma proteins such as complement (17, 19). Among these components, we were interested in studying the interaction of C. neoformans cells with platelets. Platelets, which are known to be the critical mediators of homeostasis and blood coagulation, have recently been implicated in the metastatic process of tumor cells (2, 14) and in the pathogenesis of infectious diseases (13, 34). Previous studies have demonstrated the role of platelets in antimicrobial host defenses (34). These cells interact directly or indirectly with various microorganisms, including viruses (3, 10), bacterial pathogens (9), protozoa (38), and pathogenic fungi such as Candida albicans (29, 31) and Aspergillus fumigatus (7). Most studies of interactions between platelets and pathogenic fungi have been carried out in vitro (33), and interactions between C. neoformans and platelets have been only partially studied (35). In a recent study, it was demonstrated that platelets adhere to several Candida species in vivo, using different mechanisms according to the species of Candida (30). In the present study, we describe for the first time an in vivo procedure for the study of adherence of mouse platelets to C. neoformans cells.
By using capsular and acapsular C. neoformans, we demonstrated that the platelets were able to bind only to the acapsular yeast cells. In addition, we demonstrated in vitro that thrombin-induced platelet microbicidal proteins (tPMPs) released from platelets in response to thrombin stimulation possessed a fungicidal activity against the nonencapsulated strains of C. neoformans but were not active against the encapsulated strain of C. neoformans.
Two clinical isolates from human immunodeficiency virus patients, C. neoformans 111C (encapsulated) and 222A (nonencapsulated) (the capsule disappeared after 50 routine subcultures on Sabouraud dextrose agar slants [Merck, Darmstadt, Germany]), were obtained from the Mycological Laboratory of the Faculty of Medicine, Angers, France.
C. neoformans serotype D strain NIH B-3501 (IP 1222.80) and two acapsular isogenic mutants, strain CAP67 (ATCC 52817) and strain CAP59, which were kindly donated by E. S. Jacobson (Medical College of Virginia, Richmond), were also used. Organisms were maintained by subculture on Sabouraud dextrose agar slants at 37°C twice a month. For assay purposes, yeast cells obtained from a 48-h subculture were inoculated at a concentration of 2 × 108 per ml in 1.7‰ yeast nitrogen base broth (Difco Laboratories, Detroit, Mich.) containing 2% glucose and 5% ammonium sulfate, to which 2% maltose was added, and then were grown to stationary growth phase for 48 h at 37°C, pH 4.5. Cells were harvested by centrifugation (10 min, 500 × g), washed three times in 0.15 M NaCl, and finally resuspended in NaCl at the required concentration after a hemacytometer count.
To study interactions in vivo of C. neoformans cells with platelets, female outbred Swiss mice, 6 to 8 weeks old, weighing 18 to 20 g (Centre d'élevage Déprés-France), and maintained under conventional conditions before experimentation, were injected intravenously via the lateral caudal vein with 6 × 107 to 8 × 107 C. neoformans yeast cells of each strain suspended in 0.1 ml of 0.15 M NaCl. Concerning the statistical analyses, for each experiment at least three mice were inoculated, and the blood from each bloodletting was divided into three tubes. Statistical significance was determined by using the paired t test. All comparisons were two sided, and a P value of <0.05 was considered significant.
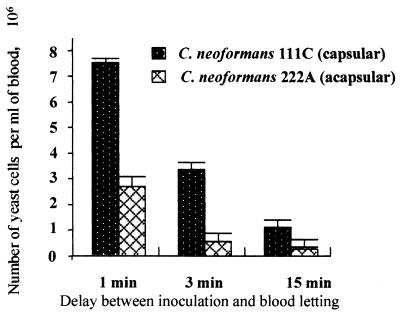
To study the clearance of C. neoformans yeast cells from the bloodstream, 7.5 × 107 yeast cells of C. neoformans 111C or 222A in 0.15 M NaCl were injected into the lateral tail veins of the mice. At 1, 3, 15, and 30 min after injection, the mice were bled by periorbital puncture and 20 μl of blood was immediately mixed with 250 μl of distilled water. After 5 min the erythrocytes were lysed and yeast cells were labeled with fluorescent brightener (Calcofluor white; Sigma, St. Louis, Mo.) as previously described (30). The enumeration of yeast cells was realized by a direct hemacytometer count under fluorescence microscopy using a Nikon incident light fluorescence microscope with an excitation filter with peak transmission at 365 nm and a barrier filter that transmitted light at wavelengths of >420 nm. Yeast cells of both C. neoformans strains disappeared quickly from the bloodstream. For examples, 1 min after inoculation and as the volume of blood was approximately 3 ml, the proportions of the remaining circulating yeast cells of C. neoformans 111C and 222A were estimated at 17 and 4.5%, respectively, of the yeast cells initially injected (P < 0.001). Three minutes after inoculation, the decrease in circulating yeasts became more pronounced, leading to the disappearance of 94 and 99% of the initially injected yeast cells of C. neoformans 111C and 222A, respectively (P < 0.001). However, for a longer period (15 min), the differences between strains 111C and 222A in the concentrations of remaining yeast cells in blood were not significant (P > 0.2) (Fig. 1).
FIG. 1.
Comparison between rates of C. neoformans 111C (capsular) and C. neoformans 222A (acapsular) yeast cell clearance from the bloodstream after intravenous injection of 7.5 × 107 cells into the lateral tail veins of the mice. Cells recovered by periorbital puncture were counted 1, 3, and 15 min after inoculation. Data are means ± standard deviations for three mice, with triplicate samples of each.
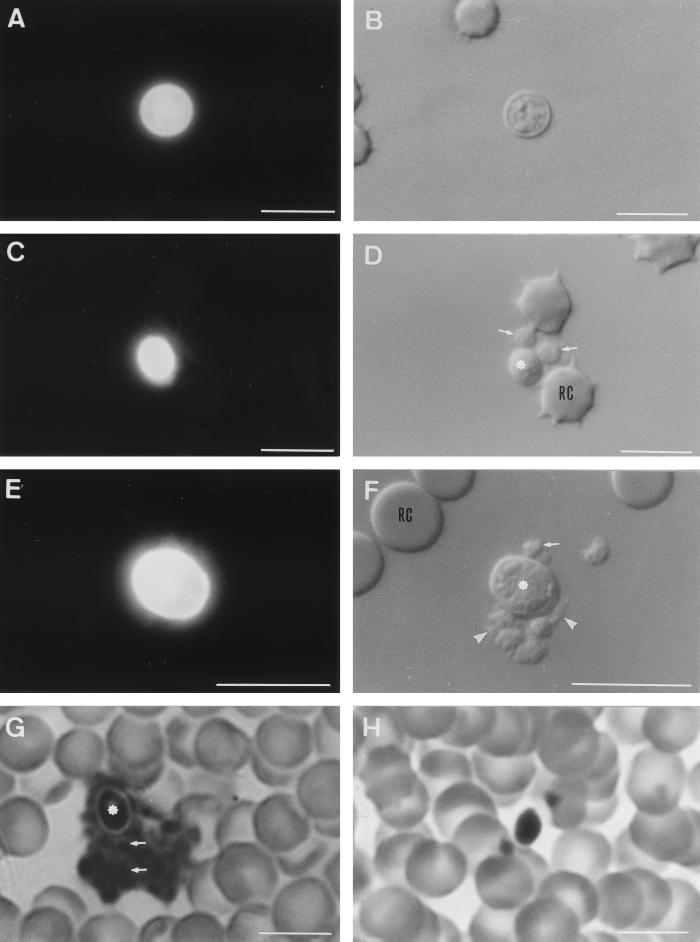
Using guidelines previously described in a study of platelet adherence to Candida species in vivo (30), we chose 3 min after injection as the time to bleed mice for the visualization of the in vivo interaction between C. neoformans strains and platelets. Blood was immediately mixed with 1 ml of phosphate-buffered saline (0.15 M, pH 7.2) containing 3.3 mM formaldehyde to prevent in vitro platelet activation. For a negative control, 20 μl of blood from an uninfected mouse was mixed with the above buffer supplemented with 5 × 105 yeast cells. The blood samples were examined by photonic fluorescence microscopy following yeast labeling with Calcofluor white as described previously (30). When blood from mice inoculated with yeast cells of C. neoformans 111C, 222A, NIH B-3501, CAP59, or CAP67 was observed in the presence of Calcofluor white with a UV microscope, it was very easy to locate yeast cells (Fig. 2A, C, and E). Observation by phase-contrast microscopy showed that platelets were attached to the acapsular yeast cells (Fig. 2D and F). The percentage of remaining circulating yeast cells bound to platelets was 96 to 98% with the nonencapsulated strains (Table 1). In contrast, with the encapsulated strains there was a very low percentage of yeast cells (1.3 to 2%) bound to platelets (Fig. 2B and Table 1). After being fixed on the surface of acapsular yeast, some platelets produced pseudopodia and were associated with small platelet aggregates (Fig. 2F). Observations of blood drawn 15 min after inoculation showed that some yeast cells of acapsular C. neoformans were associated with large aggregates of platelets (data not shown). These findings suggest an activation of the platelets. The attachment of platelets to fungal elements was confirmed when blood smears stained with May Grünwald Giemsa (MGG) (Ral, Paris, France) were examined by photonic microscopy (Fig. 2G and H). Platelets or aggregates of platelets were attached to yeast cells of the nonencapsulated strains 222A (Fig. 2G), CAP59, and CAP67 (data not shown). Conversely, free yeast cells were observed on the blood smear with the encapsulated strains 111C (Fig. 2H) and NIH B-3501 (data not shown).
FIG. 2.
Fluorescence (A, C, and E) and phase-contrast (B, D, F, G, and H) photomicrographs of platelet adherence to encapsulated (A, B, and H) and nonencapsulated (C to G) C. neoformans. Blood samples were recovered 3 min after inoculation, and yeast cells were visualized by Calcofluor white (A, C, and E) or blood smears were stained with MGG (G and H). For panels D and F, note that platelets (indicated by arrows) adhered to yeast cells (indicated by asterisks), and some platelets produced pseudopodia (indicated by arrowheads). For panel G, note the aggregates of platelets (indicated by arrows) in close association with yeast cells (indicated by an asterisk). RC, red blood cells. Bars, 10 μm.
TABLE 1.
Interaction between yeast cells of C. neoformans and plateletsa
| C. neoformans organism | % of remaining yeast cells bound to platelets in blood from:
|
|
|---|---|---|
| Inoculated mice | Uninfected miceb | |
| 111C (capsular) | 2.0 ± 0.2 | 0 |
| 222A (acapsular) | 96.0 ± 1.3 | 0 |
| NIH B-3501 (capsular) | 1.3 ± 0.1 | 0 |
| CAP59 (acapsular) | 98.6 ± 0.3 | 0 |
| CAP67 (acapsular) | 98.2 ± 0.2 | 0.2 ± 0.1 |
Mice were inoculated as described in the text, and blood recovered by periorbital puncture was diluted in phosphate-buffered saline–formaldehyde (3.3 mM). Data are means ± standard deviations of three observations of six independent experiments.
Negative controls were performed by incubating blood from uninfected mice under the same conditions and with the same buffer as for blood from infected mice supplemented with cells of homologous strains of C. neoformans.
The study of the activity of tPMPs with C. neoformans was carried out as described by Yeaman et al. (36, 37). Briefly, blood from New Zealand White rabbits was freshly collected to produce a platelet-rich plasma and to obtain washed platelets suspended in Eagle's minimal essential medium (Gibco Laboratories, Grand Island, N.Y.). Preparations rich in tPMPs were produced by stimulation of platelets with bovine-derived thrombin (Sigma). Following this activation, residual platelets were removed by centrifugation and the tPMP-rich supernatant was recovered, dialyzed against sterile deionized water, and stored at −20°C prior to use. Bioassay controls of tPMPs were performed with Bacillus subtilis ATCC 5262, and the specific activity of the tPMP preparation used in this study was 11 U of protein per mg, according to the guidelines of Yeaman et al. (36, 37). The susceptibilities of strains of C. neoformans to tPMPs were assayed by exposing the organisms, 200 μl (104 CFU/ml), to tPMPs at a final concentration of 100 U/ml. tPMP-exposed cells were incubated at 37°C for 180 min. At this time point, 20-μl samples were withdrawn and plated onto Sabouraud dextrose agar slants. Plates were then incubated at 37°C for 48 h and the surviving colonies were enumerated. Control cultures with organisms suspended in Eagle's minimum essential medium were performed in parallel and were processed identically. Results comparing organism survival in control cultures versus tPMP-exposed cultures were calculated and expressed as percent survival ± standard deviations. For the two nonencapsulated mutants, we observed a significant reduction in the percentage of survival after exposure to the tPMP preparation (P < 0.02), whereas survival was not altered by contact with tPMPs for the encapsulated strain (Table 2).
TABLE 2.
tPMP susceptibilities of C. neoformans organismsa
| C. neoformans strain | Mean % survival ± SD | P value |
|---|---|---|
| NIH B-3501 (capsular) | 104.4 ± 5.2 | NSb |
| CAP59 (acapsular) | 73.7 ± 2.3 | 0.02 |
| CAP67 (acapsular) | 73.7 ± 3.1 | 0.02 |
Susceptibilities were determined by using an organism inoculum (104 CFU/ml) in 100 U of tPMPs per ml for 180 min. Each strain was assayed independently a minimum of three times.
NS, not significantly different from control.
Under normal conditions, platelets circulate in the bloodstream as resting cells that do not interact with vascular endothelium. Upon activation, platelets become amoeboid cells that exhibit inducible receptors mediating an increased adhesion to a variety of tissues. Apart from having a vital function in hemostasis, blood platelets may have a role in the pathogenesis of tumoral metastasis (14) and in the dissemination of bacteria (9) or parasites (38). To date, little is known concerning the interaction of C. neoformans with platelets.
In the present study we compared encapsulated C. neoformans organisms (111C and serotype D NIH B-3501) and nonencapsulated organisms (222A and isogenic mutant strains from NIH B-3501, CAP59 and CAP67) for their susceptibility to platelet binding in vivo and to the fungicidal activity of tPMPs in vitro. The data presented here indicate that after injection of yeast cells into mice, the rate of clearance of nonencapsulated C. neoformans from the bloodstream was high, leading to the disappearance of 95% of the inoculated cells within the first minute of inoculation. In contrast, the rate of clearance observed with the C. neoformans encapsulated strain was lower. Microscopic observation of blood samples stained by Calcofluor white or MGG showed binding of platelets to yeast cells exclusively with acapsular C. neoformans strains. The in vivo interaction of platelets with cells of various Candida species, which are acapsular yeasts, investigated in a murine model gave similar results (30).
Yeaman et al. (36) showed that tPMPs were bactericidal against Staphylococcus aureus. The antifungal activity of tPMPs was also investigated by these and other authors, and they demonstrated that tPMPs possessed variable fungicidal activity against several species of Candida but not against C. neoformans (35). We confirmed in this study that tPMPs had no fungicidal activity against capsular yeast cells of C. neoformans, although acapsular strains were significantly susceptible to tPMPs.
While the capsule is recognized as a major virulence factor of C. neoformans, the mechanisms whereby the capsule helps the organism elude host defenses are incompletely defined. Previous studies have demonstrated that binding and internalization of encapsulated organisms by phagocytes are inhibited compared to those of mutant isolates lacking capsules (16, 20). Moreover, encapsulated C. neoformans organisms are inhibited or killed by phagocyte populations less readily than the nonencapsulated organisms (20, 21, 22).
In conclusion, although numerous studies indicate that the capsule of C. neoformans is an important virulence factor (6, 18), little is known about its role in the accomplishment of antifungal activity by blood-circulating components, platelets in particular. However, the results of this study showed that the acapsular yeasts are more susceptible than the encapsulated fungi to platelet binding and are more susceptible to the fungicidal action of tPMPs. These findings suggest that the capsule prevents the adherence of platelets and then the release of microbicidal peptides. Conversely, nonencapsulated C. neoformans binds platelets, which could be activated. Further in vivo studies using encapsulated and nonencapsulated C. neoformans strains should be carried out to determine the platelet microbicidal activity.
REFERENCES
- 1.Aberg J A, Powderly W G. Cryptococcosis. Adv Pharmacol. 1997;37:215–251. doi: 10.1016/s1054-3589(08)60951-5. [DOI] [PubMed] [Google Scholar]
- 2.Biggerstaff J P, Seth N B, Meyer T V, Amirkhosravi A, Francis J L. Fibrin monomer increases platelet adherence to tumor cells in a flowing system: a possible role in metastasis? Thromb Res. 1998;92:53–58. doi: 10.1016/s0049-3848(98)00161-3. [DOI] [PubMed] [Google Scholar]
- 3.Bik T, Sarov I, Livne A. Interaction between vaccinia virus and human blood platelets. Blood. 1982;59:482–487. [PubMed] [Google Scholar]
- 4.Brumer E. Human defenses against Cryptococcus neoformans: an update. Mycopathologia. 1999;143:121–125. doi: 10.1023/a:1006905331276. [DOI] [PubMed] [Google Scholar]
- 5.Buchanan K L, Murphy J W. What makes Cryptococcus neoformans a pathogen? Emerg Infect Dis. 1998;4:71–83. doi: 10.3201/eid0401.980109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Cherniak R, Sundstrom J B. Polysaccharide antigens of the capsule of Cryptococcus neoformans. Infect Immun. 1994;62:1507–1512. doi: 10.1128/iai.62.5.1507-1512.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Christin L, Wysong D R, Meshulam T, Hastey R, Simons E, Diamond R D. Human platelets damage Aspergillus fumigatus hyphae and may supplement killing by neutrophils. Infect Immun. 1998;66:1181–1189. doi: 10.1128/iai.66.3.1181-1189.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Chuck S L, Sande M A. Infections with Cryptococcus neoformans in the acquired immunodeficiency syndrome. N Engl J Med. 1989;321:794–799. doi: 10.1056/NEJM198909213211205. [DOI] [PubMed] [Google Scholar]
- 9.Clawson C C, White J G. Platelet interaction with bacteria. I. Reaction phases and effects of inhibitors. Am J Pathol. 1971;65:367–380. [PMC free article] [PubMed] [Google Scholar]
- 10.Davis B R, Zauli G. Effect of human immunodeficiency virus infection on haematopoiesis. Bailliere's Clin Haematol. 1995;8:113–130. doi: 10.1016/s0950-3536(05)80234-3. [DOI] [PubMed] [Google Scholar]
- 11.Feldmesser M, Casadevall A, Kress Y, Spira G, Orlofsky A. Eosinophil-Cryptococcus neoformans interactions in vivo and in vitro. Infect Immun. 1997;65:1899–1907. doi: 10.1128/iai.65.5.1899-1907.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Gross N T, Nessa K, Cammer P, Chinchilla M, Jarstrand C. Interaction between Cryptococcus neoformans and alveolar macrophages. J Med Vet Mycol. 1997;35:263–269. [PubMed] [Google Scholar]
- 13.Henderson W R. The role of the platelet in the pathogenesis of infectious disease. Curr Opin Infect Dis. 1992;5:375–380. [Google Scholar]
- 14.Honn K V, Tang D G, Crissman J D. Platelets and cancer metastasis: a causal relationship? Cancer Metastasis Rev. 1992;11:325–351. doi: 10.1007/BF01307186. [DOI] [PubMed] [Google Scholar]
- 15.Kovacs J A, Kovacs A A, Polis M, Wright W C C, Gill V J, Tuazon C U, Gelmann E P, Lane H C, Longfield R, Overturf G, Macher A M, Fauci A S, Parillo J E, Bennett J E, Masur H. Cryptococcosis in the acquired immunodeficiency syndrome. Ann Intern Med. 1985;103:533–538. doi: 10.7326/0003-4819-103-4-533. [DOI] [PubMed] [Google Scholar]
- 16.Kozel T R, Mastroianni R P. Inhibition of phagocytosis by cryptococcal polysaccharide: dissociation of the attachment and ingestion phases of phagocytosis. Infect Immun. 1976;14:62–67. doi: 10.1128/iai.14.1.62-67.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Kozel T R. Activation of the complement system by the capsule of Cryptococcus neoformans. Curr Top Med Mycol. 1993;5:1–26. [PubMed] [Google Scholar]
- 18.Kwon-Chung K J, Bennett J E. Cryptococcosis. In: Kwon-Chung K J, Bennett J E, editors. Medical mycology. Philadelphia, Pa: Lea and Febiger; 1992. pp. 397–446. [Google Scholar]
- 19.Laxalt K A, Kozel T R. Chemotaxigenesis and activation of the alternative complement pathway by encapsulated and nonencapsulated Cryptococcus neoformans. Infect Immun. 1979;26:435–440. doi: 10.1128/iai.26.2.435-440.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Levitz S M, DiBenedetto D J. Differential stimulation of murine resident peritoneal cells by selectively opsonized encapsulated and acapsular Cryptococcus neoformans. Infect Immun. 1988;56:2544–2551. doi: 10.1128/iai.56.10.2544-2551.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Levitz S M, DiBenedetto D J. Paradoxical role of capsule in murine bronchoalveolar macrophage-mediated killing of Cryptococcus neoformans. J Immunol. 1989;142:659–665. [PubMed] [Google Scholar]
- 22.Levitz S M, Farrel T P. Growth inhibition of Cryptococcus neoformans by cultured human monocytes: role of the capsule, opsonins, the cultured surface, and cytokines. Infect Immun. 1990;58:1201–1209. doi: 10.1128/iai.58.5.1201-1209.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Levitz S M. Macrophage-Cryptococcus interactions. Immunol Ser. 1994;60:533–544. [PubMed] [Google Scholar]
- 24.Lewis J L, Rabinovich S. The wide spectrum of cryptococcal infections. Am J Med. 1972;53:315–322. doi: 10.1016/0002-9343(72)90174-x. [DOI] [PubMed] [Google Scholar]
- 25.Lortholary O, Improvisi L, Nicolas M, Provost F, Dupont B, Dromer F. Fungemia during murine cryptococcosis sheds some light on pathophysiology. Med Mycol. 1999;37:167–174. [PubMed] [Google Scholar]
- 26.Miller M F, Mitchell T G. Killing of Cryptococcus neoformans strains by human neutrophils and monocytes. Infect Immun. 1991;59:24–28. doi: 10.1128/iai.59.1.24-28.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Mitchell T G, Perfect J R. Cryptococcosis in the era of AIDS— 100 years after the discovery of Cryptococcus neoformans. Clin Microbiol Rev. 1995;8:515–548. doi: 10.1128/cmr.8.4.515. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Nessa K, Gross N T, Jarstrand C, Johansson A, Cammer P. In vivo interaction between alveolar macrophages and Cryptococcus neoformans. Mycopathologia. 1997;139:1–7. doi: 10.1023/a:1006843202124. [DOI] [PubMed] [Google Scholar]
- 29.Robert R, Mahaza C, Miegeville M, Ponton J, Marot-Leblond A, Senet J M. Binding of resting platelets to Candida albicans germ tubes. Infect Immun. 1996;64:3752–3757. doi: 10.1128/iai.64.9.3752-3757.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Robert R, Nail S, Marot-Leblond A, Cottin J, Miegeville M, Quenouillère S, Mahaza C, Senet J M. Adherence of platelets to Candida species in vivo. Infect Immun. 2000;68:570–576. doi: 10.1128/iai.68.2.570-576.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Robert R, Senet J M, Mahaza C, Annaix V, Miegeville M, Bouchara J P, Tronchin G, Marot-Leblond A. Molecular basis of the interactions between Candida albicans, fibrinogen and platelets. J Mycol Med. 1992;2:19–25. [Google Scholar]
- 32.Rozenbaum R, Rios Gonçalves A J. Clinical epidemiology study of 171 cases of cryptococcosis. Clin Infect Dis. 1994;18:369–380. doi: 10.1093/clinids/18.3.369. [DOI] [PubMed] [Google Scholar]
- 33.Willcox M D P, Webb B C, Thakur A, Harty D W S. Interactions between Candida species and platelets. J Med Microbiol. 1998;47:103–110. doi: 10.1099/00222615-47-2-103. [DOI] [PubMed] [Google Scholar]
- 34.Yeaman M R. The role of platelets in antimicrobial host defense. Clin Infect Dis. 1997;25:951–970. doi: 10.1086/516120. [DOI] [PubMed] [Google Scholar]
- 35.Yeaman M R, Ibrahim A S, Edwards J E, Jr, Bayer A S, Ghannoum M A. Thrombin-induced rabbit platelet microbicidal protein is fungicidal in vitro. Antimicrob Agents Chemother. 1993;37:546–553. doi: 10.1128/aac.37.3.546. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Yeaman M R, Puentes S M, Norman D C, Bayer A S. Partial characterization and staphylocidal activity of thrombin-induced platelet microbicidal protein. Infect Immun. 1992;60:1202–1209. doi: 10.1128/iai.60.3.1202-1209.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Yeaman M R, Tang Y Q, Shen A J, Bayer A S, Selsted M E. Purification and in vitro activities of rabbit platelet microbicidal proteins. Infect Immun. 1997;65:1023–1031. doi: 10.1128/iai.65.3.1023-1031.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Yong E C, Chi E Y, Fritsche T R, Henderson W R. Human platelet-mediated cytotoxicity against Toxoplasma gondii: role of thromboxane A2. J Exp Med. 1991;173:65–78. doi: 10.1084/jem.173.1.65. [DOI] [PMC free article] [PubMed] [Google Scholar]