ABSTRACT
Sandy beaches are iconic interfaces that functionally link the ocean with the land via the flow of organic matter from the sea. These cross‐ecosystem fluxes often comprise uprooted seagrass and dislodged macroalgae that can form substantial accumulations of detritus, termed ‘wrack’, on sandy beaches. In addition, the tissue of the carcasses of marine animals that regularly wash up on beaches form a rich food source (‘carrion’) for a diversity of scavenging animals. Here, we provide a global review of how wrack and carrion provide spatial subsidies that shape the structure and functioning of sandy‐beach ecosystems (sandy beaches and adjacent surf zones), which typically have little in situ primary production. We also examine the spatial scaling of the influence of these processes across the broader land‐ and seascape, and identify key gaps in our knowledge to guide future research directions and priorities. Large quantities of detrital kelp and seagrass can flow into sandy‐beach ecosystems, where microbial decomposers and animals process it. The rates of wrack supply and its retention are influenced by the oceanographic processes that transport it, the geomorphology and landscape context of the recipient beaches, and the condition, life history and morphological characteristics of the macrophyte taxa that are the ultimate source of wrack. When retained in beach ecosystems, wrack often creates hotspots of microbial metabolism, secondary productivity, biodiversity, and nutrient remineralization. Nutrients are produced during wrack breakdown, and these can return to coastal waters in surface flows (swash) and aquifers discharging into the subtidal surf. Beach‐cast kelp often plays a key trophic role, being an abundant and preferred food source for mobile, semi‐aquatic invertebrates that channel imported algal matter to predatory invertebrates, fish, and birds. The role of beach‐cast marine carrion is likely to be underestimated, as it can be consumed rapidly by highly mobile scavengers (e.g. foxes, coyotes, raptors, vultures). These consumers become important vectors in transferring marine productivity inland, thereby linking marine and terrestrial ecosystems. Whilst deposits of organic matter on sandy‐beach ecosystems underpin a range of ecosystem functions and services, they can be at variance with aesthetic perceptions resulting in widespread activities, such as ‘beach cleaning and grooming’. This practice diminishes the energetic base of food webs, intertidal fauna, and biodiversity. Global declines in seagrass beds and kelp forests (linked to global warming) are predicted to cause substantial reductions in the amounts of marine organic matter reaching many beach ecosystems, likely causing flow‐on effects for food webs and biodiversity. Similarly, future sea‐level rise and increased storm frequency are likely to alter profoundly the physical attributes of beaches, which in turn can change the rates at which beaches retain and process the influxes of wrack and animal carcasses. Conservation of the multi‐faceted ecosystem services that sandy beaches provide will increasingly need to encompass a greater societal appreciation and the safeguarding of ecological functions reliant on beach‐cast organic matter on innumerable ocean shores worldwide.
Keywords: landscape ecology, seascape, spatial subsidy, ecosystem functioning, coastal ecosystems, kelp forests, seagrass beds, detritus, wrack, carrion
I. INTRODUCTION
Shorelines are formed by various coastal landforms, including sandy beaches, surf zones, different dune landscapes, subtidal and intertidal rocky reefs, and sea cliffs. Shorelines are also highly dynamic, being the interfaces between the ocean and the land (Pilkey et al., 2011). The world's coastlines have been widely transformed for human uses, providing valuable ecosystem services to society (Small & Nicholls, 2003; Bowen, Frankic & Davis, 2006). Iconic sandy beaches cover 31% of the world's ice‐free shoreline (Luijendijk et al., 2018), and represent highly valued economic, ecological, recreational and cultural assets for societies worldwide (Barbier et al., 2011). Beaches are dynamic landforms that are often heavily altered by humans; rising sea levels and storm regimes can cause massive and widespread changes to beaches and dunes (Dugan et al., 2010; Vousdoukas et al., 2020).
Beaches form an ecotone between the ocean and land, which is influenced by land and ocean inputs and processes. Beaches and adjacent surf zones along open coasts are often considered to have little in situ primary production, but their fauna can nevertheless be abundant and diverse (Brown & McLachlan, 1990). This paradox can be attributed, at least partly, to the flow of organic matter from productive to less‐productive ecosystems (i.e. ‘spatial subsidies’ sensu Polis et al., 1997). Sandy beach systems are highly permeable, lacking significant barriers amongst habitats, whilst having strong transport mechanisms driven by tides, waves and currents; these forces can bring large organic matter (e.g. macroalgae, seagrass, dead animals) ashore to become stranded on beaches as wrack and carrion (Schlacher, Strydom & Connolly, 2013a ; Hyndes et al., 2014).
Organic matter stranded on beaches is typically derived from reefs and seagrass beds that supply detached macrophytes (e.g. kelp thalli and seagrass leaves). It can also be in the form of dead animals that wash ashore to become nutritious carrion (i.e. the tissues of animal carcasses) beach‐cast on sandy shores. Many studies have focused on the production and fate of organic matter in kelp forests (e.g. Krumhansl & Scheibling, 2011; de Bettignies et al., 2013; Pedersen et al., 2020) and seagrass meadows (see Heck Jr. et al., 2008). Reviews show that these highly productive ecosystems can export large quantities of organic matter via dissolved organic nutrients or particulate organic matter to adjacent and more distant ecosystems, including beaches (Heck Jr. et al., 2008; Krumhansl & Scheibling, 2012; Hyndes et al., 2014). However, these reviews provide little detail on the fates of organic matter in beach ecosystems. Inputs of detached macrophytes (wrack) and carrion (dead animals) (Figs 1A and 2) may, however, provide critical ecosystem functions (physical, chemical and biological processes within ecosystems) on many sandy beaches. These functions include: (1) providing a food subsidy to support the high secondary production and biodiversity that underpin rich coastal food webs (e.g. Crawley et al., 2009); (2) recycling nutrients (e.g. Dugan et al., 2011); (3) enhancing key habitats, such as fish nursery grounds (surf zone) and bird nesting sites (e.g. Crawley, Hyndes & Ayvazian, 2006; Schlacher et al., 2013a ); and (4) trapping wind‐blown sand, thereby limiting the extent of coastal erosion and facilitating beach accretion and dune formation (e.g. Dugan, Hubbard & Page, 2005).
Fig. 1.

Conceptual diagrams of wrack dynamics in beach ecosystems. (A) The principal sources, transport routes, and biological fates of marine organic material cast upon sandy beaches; (B) disruption of natural processes caused by beach grooming and coastal armouring; and (C) predicted consequence of climate change for the supply, type and biological fates of marine organic matter in sandy beach ecosystems. POM, particulate organic matter. Diagrams created using IAN Image Library (http://ian.umces.edu).
Fig. 2.

Wrack, scavengers and human disturbance on sandy beaches. Wrack on beaches in (A) Cape Town, South Africa (photograph L. Harris); (B) Santa Barbara County, USA (photograph J. Dugan); (C) Salina Bay, Malta (photograph M. Mateo); and (D) Perth, Australia (photograph G. Hyndes). Dingo (E) and white‐bellied sea eagle (F) scavenging on carrion on beaches near Brisbane, Australia (photographs A. Olds), and beach cleaning on beaches in (G) Brisbane, Australia (photograph A. Olds) and (H) Carpinteria, USA (photograph J. Dugan).
The organisms and functions linked to marine matter cast upon beaches were reviewed by Colombini & Chelazzi (2003). Since then, significantly more research on these functions has been undertaken in various regions across the globe. Importantly, the effects of global warming on coastal systems that supply organic material to beaches, and on beaches themselves, are now better understood (Vitousek, Barnard & Limber, 2017; Smale et al., 2019). Indeed, marine heatwave events and subsequent poleward shifts in the distribution of tropical grazers have led to ecosystem shifts from kelp forests to barrens in parts of the world (Smale et al., 2019), which has immense ramifications for ecosystems relying on kelp as subsidies. Changes to beach structure and habitat availability through erosion, sea‐level rise, storms, and coastal development (Lee et al., 2018; Dugan et al., 2008) strongly affect the ability of drift material to flow onto and be retained in these dynamic ecosystems. In addition, management activities, such as grooming, that actively remove wrack from beaches used for tourism, impact wrack‐associated biota and processes (e.g. Schooler, Dugan & Hubbard, 2019). Furthermore, introductions of invasive species through global warming or other human‐induced mechanisms (e.g. Jiménez et al., 2015) are likely to influence the supply and form of organic inputs to beach ecosystems.
Here, we review and synthesise the existing knowledge on the ecosystem functions of wrack and carrion in sandy‐beach ecosystems. We also outline knowledge gaps and identify priorities for future research. We focus mainly on the dynamics and ecological pathways of marine matter becoming stranded on ocean sandy shores, including the supply, retention, and processing of macrophytes and carrion, and how matter can be transferred inland or back to sea with or without alteration. We reviewed the literature based on searches in Elsevier Scopus and Clarivate Web of Knowledge using the terms (‘beach*’ OR ‘shore’ OR ‘surf zone’) AND (‘wrack’ OR ‘carrion’ OR ‘allochthonous’ OR ‘beach‐cast’ OR ‘drift’ OR ‘detritus’ OR ‘kelp’ OR ‘seagrass’). Search results were supplemented with references cited in Colombini & Chelazzi (2003), and papers from the authors' personal collections. This process yielded 336 articles that report various aspects of wrack or carrion in sandy‐beach ecosystems, with 245 of those papers published after Colombini & Chelazzi (2003).
We first summarise the global distribution of the research effort and the focal themes. Secondly, we provide an overview of the main forms and amount of wrack and carrion deposited on beaches and adjacent surf zones. We then examine several topics concerning the dynamics and influence of wrack and carrion subsidies to beaches, specifically: (1) factors influencing the availability, deposition and retention of these subsidies; (2) the role of subsidies in regulating faunal abundance and biodiversity; (3) the fate of wrack and carrion, particularly their roles in beach food webs; (4) the spatial scaling of subsidy effects in sandy‐beach ecosystems and across the broader seascape and landscape; and (5) the influence of anthropogenic changes and management interventions on wrack and carrion dynamics. Finally, we identify key knowledge gaps to guide future research.
II. THE CURRENT STATE OF KNOWLEDGE
(1). Thematic areas and the geography of the global research effort
Of the 336 papers that examined various aspects of wrack or carrion in sandy‐beach ecosystems, 89% focused on the beach, with fewer studies from the surf zone or waters adjacent to sandy beaches (see Table S1). Three thematic areas dominate the research effort (Table 1, Fig. 3): (1) spatial or temporal patterns in the amount of wrack (‘wrack dynamics’, 36%); (2) spatial or temporal patterns in the abundance of fauna (‘fauna dynamics’, 33%); and (3) trophic ecology (28%). Themes with moderate research focus include population biology of fauna in wrack (16%), and chemical composition and processes or the nutrient dynamics of wrack (12%). Only 10% of studies examined distribution patterns of carcasses or carrion on beaches (‘carrion and carcass dynamics’). Similarly, only 9% of studies examined human use, such as harvesting kelp for food products or biofuel and human impacts such as the effects of beach grooming on wrack‐associated fauna. Few studies (7%) examined environmental processes and influences, including factors that affect the amount and composition of wrack on beach systems. Even fewer (only 5%) looked at decomposition and microbial processes in wrack, and a small number of studies (<2%) examined topics such as invasion biology, human health, or genetics (Table 1).
Table 1.
Themes addressed in the literature on beach‐cast organic matter (plant wrack and animal carrion). The total number of studies identified was 336, but many papers investigated more than one theme. See Table S1 for full list of publications and theme designations.
| Theme | # studies | % |
|---|---|---|
| Wrack dynamics (temporal and spatial variability) | 121 | 36.0 |
| Faunal dynamics (temporal and spatial variability) | 111 | 33.0 |
| Trophic ecology | 94 | 28.0 |
| Population biology | 54 | 16.1 |
| Chemical composition and processes* | 42 | 12.5 |
| Carrion and carcass dynamics | 32 | 9.5 |
| Human use and impacts | 31 | 9.2 |
| Environmental processes and influences | 23 | 6.8 |
| Decomposition and microbial processes** | 16 | 4.8 |
| Dispersion of sources*** | 7 | 2.1 |
| Human health | 6 | 1.8 |
| Invasion biology | 5 | 1.5 |
| Taxonomic inventories**** | 5 | 1.5 |
| Hydrodynamics | 4 | 1.2 |
| Influence on dunes | 4 | 1.2 |
| Genetics (population genetics and phylogenetics) | 3 | 0.9 |
| Methods | 3 | 0.9 |
| Restoration | 3 | 0.9 |
| Fauna physiology | 1 | 0.3 |
| Movement of fauna | 1 | 0.3 |
| Sediment transport | 1 | 0.3 |
Forms and concentrations of nutrients and nutrient cycling.
Biomass loss of wrack or carrion, and/or microbial processes leading to those losses.
Export mechanisms for the dispersal of wrack or carrion from donor systems.
Inventory of algal or faunal taxa, or new records.
Fig. 3.
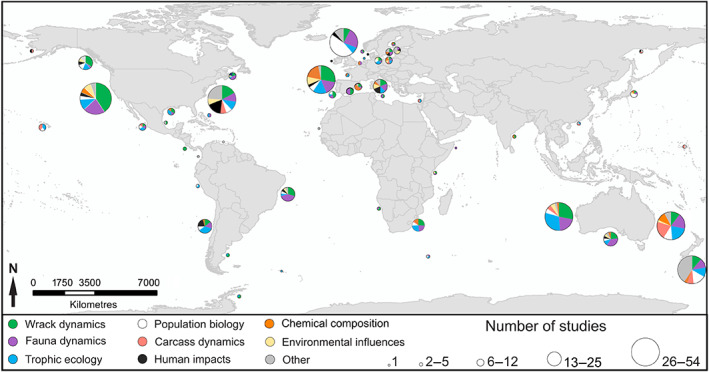
Global distribution of studies (N = 336) classified by the main theme with respect to wrack and carrion on sandy beaches and in surf zones. Pie charts illustrate the different themes of published studies, with the size indicating the total number of studies for a region. Note that more than one theme could be covered by each paper, but the number of studies in each region reflects the total number of papers regardless of theme. See Table 1 for more detail on themes.
We found few studies (N = 10) published before the 1980s. The number of published studies almost doubled from 42 during the 1980s to 76 during the 2000s, doubling again to 165 articles during the 2010s. All but four studies examined aspects of sandy beach ecosystems rather than beaches with larger sediments such as cobbles and boulders – most of these studies sampled in higher latitude regions (>40°N or S). The majority of research on wrack or carrion has been carried out in the USA (58 studies), Australia (53), Spain (30), Canada (21), and UK and New Zealand (20 each) (Fig. 3). Countries with moderate research effort (7–15 studies) include Italy, South Africa, Chile, and Brazil. Notable areas with significant sandy‐beach shorelines with a paucity of studies on wrack and carrion inputs include most of the tropics, such as the Caribbean, central and north Africa, the Arabian Peninsula, southeast Asia and South America (except Chile and Brazil) (Fig. 3). While several studies have been carried out in the polar regions, most beaches in these studies comprised coarser sediment grain, e.g. shingles or boulders (Crafford & Scholtz, 1987; Chown, 1996; Lastra et al., 2014).
(2). Global patterns in standing stock of wrack on beaches
We found over 60 papers that quantified the amount or composition of wrack on sandy beaches or in adjacent surf zones. However, the metric used to report the amount of wrack was variable, including volume (l) or wet or dry mass (g or kg) of wrack within a linear metre or square metre of beach. Because the most common unit has been kg wet weight (WW) per linear metre of beach, we use this measure to examine global patterns in standing stock. However, even for this metric, the number of studies is low. Based on papers allowing data from multiple sites or seasons to be directly extracted, or by converting extracted dry mass estimates where conversion factors were available, the average biomass of wrack on beaches varies considerably from 0.1 to 325 kg WW m−1 (Fig. 4). Exceptionally high biomass (>100 kg WW m−1) has been recorded on beaches in the Mediterranean Sea, south‐western Australia, Argentina and western Canada. However, inconsistencies in the metric used to report wrack biomass reduce the pool of comparable data and make comparisons across regions challenging. To facilitate such comparisons, we recommend that future studies provide sufficient data (e.g. beach/surf zone width length, volume to biomass conversions) to allow standing stock (wet mass) per unit area to be calculated.
Fig. 4.

Global patterns in wrack composition (N = 43) and wet mass (WW; N = 27) of wrack (kg WW wrack m−1 of coastline) on beaches and in surf zones based on published papers. Numbers in plain text to the right of each pie chart indicate the number of studies. The text in italics below the pie charts indicates the average biomass in each region, while the numbers in parentheses to the right of the text indicates the number of studies that provided the data. Note that for a study to be included, data needed to be provided for at least three sites or times. Wrack composition was based on wet and dry mass and volume data, while biomass data were based on studies where mass was either provided or data could be converted to wet mass per linear metre of coastline. Distribution of seagrass from UNEP‐WCMC seagrass maps based on Green & Short (2003), while kelp distributions are based on Filbee‐Dexter & Wernberg (2018).
The composition of wrack in sandy‐beach ecosystems varies geographically, but often is dominated by macroalgae, particularly kelp species, and seagrasses (Fig. 4). At a global scale, major factors that influence biomass and composition of wrack include the offshore benthic substrate type and the associated distribution, productivity rate and composition of coastal primary producers (seagrass, kelp, etc.) as potential sources of wrack macrophytes. For instance, kelp forests span temperate to arctic regions. Therefore, kelp is a major input of wrack along the coasts of cooler waters, particularly the western USA, Chile, southern Africa and New Zealand, and a substantial input in southern Australia (Fig. 4). Dominant taxa include Durvillea, Ecklonia, Lessonia, Macrocystis and Nereocystis. In comparison, seagrasses span boreal to tropical regions, and form a major component of wrack along coastlines in the Mediterranean and Caribbean Seas, East Africa, and Australia (Fig. 4), with Posidonia, Zostera, Cymodocea, Halodule, Halophila or Thalassia being the main genera. Indeed, seagrass is the only form of wrack on the beaches in the Mediterranean Sea, where Posidonia oceanica can form major ‘banquettes’ (wrack deposits). In the northern hemisphere (the Baltic Sea, Western Europe, Caribbean Sea and west coast of North America) and in Argentina, other forms of macroalgae (red, green and non‐kelp brown algae) also form significant components of wrack (Fig. 4).
Besides the inflow of macrophytes, carrion (dead invertebrates, fish, birds, and mammals as large as whales) can be numerous on beaches, when animals that die at sea wash ashore (Sikes & Slowik, 2010). However, few studies have focused on carrion or animal carcasses compared to beach‐cast macrophytes (Table 1, Fig. 3), possibly reflecting: (1) a lower proportion of carrion compared to wrack; (2) more episodic deposition and patchy distribution; (3) more rapid consumption and removal by scavengers; or (4) a reluctance by researchers to work on decomposing animal bodies.
(3). Factors influencing standing stocks of wrack
The standing stock of wrack on sandy‐beach systems is highly dynamic, both in space and time. Wrack biomass can range over 1–4 orders of magnitude across locations within a region (Tarr & Tarr, 1987; Dugan et al., 2003, 2011; Barreiro et al., 2011; Liebowitz et al., 2016; Holden et al., 2018a ; Reimer et al., 2018; Schooler et al., 2019). Similarly, standing stock may show temporal variation from no visible accumulation to tens or thousands of grams dry mass per square metre, within hours to days (Ruiz‐Delgado et al., 2016b ; Vieira et al., 2016), and across years (Barreiro et al., 2011; Revell, Dugan & Hubbard, 2011; Jiménez et al., 2015; Liebowitz et al., 2016; Holden et al., 2018a ). Wrack dynamics are principally influenced by the input, accumulation, and export of material. Several factors shape these dynamics, including: (1) the types and strength of physical forces that transport material; (2) the geomorphology of beaches; (3) the broader landscape context of beaches; and (4) the characteristics or traits of the donor system (Table 2).
Table 2.
Factors that either positively or negatively affect the deposition and retention of wrack on beaches.
| Factor | Description | References |
|---|---|---|
| State of donor ecosystem | ||
| Standing stock | Biomass of subsidy and availability for export | de Bettignies et al., 2013; Krumhansl & Scheibling (2011); Pedersen et al. (2020) |
| Senescence/growth | Primary productivity, biomass availability/ turnover/nutritional condition and availability for export | Rodriguez et al. (2013, 2016) |
| Macrophyte phenology | Annual versus perennial, seasonal cycles of export | Hamilton et al. (2020) |
| Management | Direct harvest, grooming, fisheries, marine protected areas, biomass available for export | Dugan & Hubbard (2010) |
| Characteristic of beach | ||
| Proximity to donor ecosystems | Rocky reef, kelp forest, seagrass bed, estuary, rocky intertidal | Orr et al. (2005); Reimer et al. (2018); Liebowitz et al. (2016) |
| Beach morphology | Width of zones and slopes affect delivery and retention | Orr et al. (2005); Revell et al. (2011); Barreiro et al. (2011); Wickham et al. (2020) |
| Beach orientation | Shore orientation relative to prevailing swell and currents affects delivery and retention | Orr et al. (2005); Gómez et al. (2013) |
| Back beach type | Retention and fate of wrack varies among dune‐, cliff‐ and seawall‐backed shores | Dugan et al. (2008); Heerhartz et al. (2014) |
| Beach management | Grooming and armouring alter retention and fate | Dugan & Hubbard (2010); Schooler et al. (2019) |
| Sediment supply/budget | Availability and resilience of beach habitat affects retention | Zoulas & Orme (2007); Orme et al. (2011); Griggs & Patsch (2018) |
| Disturbances | ||
| Storm events | Changes in wave height affect donor and recipient ecosystems through removal and erosion | Barreiro et al. (2011); Reed et al. (2011) |
| Storm season | Wave‐driven removal of substrate and subsidies | Cavanaugh et al. (2011); Reed et al. (2011) |
| Climate events | El Nino, oceanographic oscillations, marine heatwaves, etc. change primary production, supply and dynamics of recipient ecosystem | Cavanaugh et al. (2011); Revell et al. (2011); Thomsen et al. (2019); Strydom et al. (2020) |
| Climate change | Increased ocean temperature and sea‐level rise: warming impacts kelp forests/seagrass beds and sea‐level rise causes loss of beach habitat | Jordà et al. (2012); Krumhansl et al. (2016); Bell et al. (2018); Cavanaugh et al. (2019) |
| Overgrazing | Effect on standing stock and resilience, urchins and urchin barrens, biomass available for export | Rose et al. (1999); Ling et al. (2015, 2019); Rogers‐Bennett & Catton (2019) |
| Invasive species | Outcompete natives, altered life cycles and biomass production/export and food quality | Marks, Reed & Holbrook (2020); Schiel et al. (2018) |
| Ocean processes | Drivers of exchanges | |
|---|---|---|
| Tides | Daily and semi‐lunar tides affect delivery and retention on beaches | Zobell (1971); Revell et al. (2011); Orr et al. (2005) |
| Wave climate | Event‐, season‐ and climate‐driven wave dynamics affect donor (loss of biomass, whole plants) and recipient (wrack biomass dynamics and beach erosion/rotation/retention) ecosystems | Zobell (1971); Revell et al. (2011); Liebowitz et al. (2016) |
| Currents | Transport and delivery of macrophytes to beaches. Can move wrack along and on and off the beach | Orr et al. (2005); Gómez et al. (2013); Liebowitz et al. (2016) |
| Sea level rise | Erosion and long‐term loss of recipient beach habitat zones affects retention of wrack | Myers et al. (2019) |
| Wind | Surface currents and erosion processes affect both donor and recipient ecosystems, wrack burial and transport inland | Rossi & Underwood (2002); Hammann & Zimmer (2014); Liebowitz et al. (2016); Del Vecchio et al. (2017) |
Various physical forces (e.g. tides, waves, currents, wind) transport material onshore. It follows that variation in physical forces results in differences in the deposition and resuspension of wrack on sandy beaches, operating at timescales from hours to weeks and over distances of hundreds of metres to hundreds of kilometres (Table 2, Fig. 5). Rising tides tend to remove wrack from beaches, while falling tides are more conducive to intertidal deposition of buoyant material (Fig. 1A; Zobell, 1971; Orr et al., 2005). Spring high tides also remove wrack (Zobell, 1971) or shift wrack deposits up‐shore towards the base of dunes or bluffs backing the beach. Strong seasonal patterns of kelp deposition are often related to changes in wave climate (Zobell, 1971; Revell et al., 2011). For example, storms generated at monthly to annual timescales (Fig. 5) uproot and transport large amounts of macrophytes to become drift material deposited on beaches (Zobell, 1971;Revell et al., 2011; Liebowitz et al., 2016). However, the effects of storms on the supply can be modified by changes in the life‐history traits of potential wrack material in donor systems. For instance, early‐season storms can dislodge and transport large amounts of macrophytes, resulting in less material being available when storms occur later in the growing season (e.g. Zobell, 1971). However, wrack supply is further complicated by differences in the overall life histories among primary producers in the donor systems. Some kelp species are annuals (e.g. Nereocystis), while others are perennials (e.g. Macrocystis), and some macroalgae senesce as part of their life cycle (e.g. Sargassum). Supply is also influenced by ocean‐scale to global atmospheric and oceanic events, such as El Nino Southern Oscillation (ENSO) events (Seymour, 2003) or heatwave events and broader ocean warming (Wernberg et al., 2019), occurring over timescales of years to thousands of years and spatial scales of tens to tens of thousands of kilometres (Fig. 5).
Fig. 5.
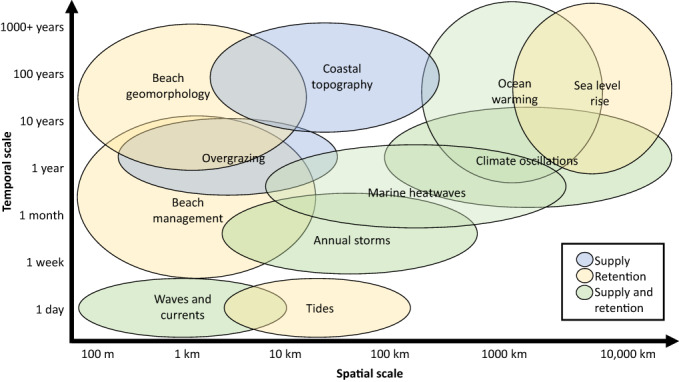
Coastal, oceanographic and atmospheric factors that influence wrack supply and retention on sandy beaches, and the spatial and temporal scales at which they operate. Sources of information are provided in Table 2. Supply = processes that influence the input of wrack to beach ecosystems; retention = processes that influence the ability of wrack to remain in beach ecosystems. The colour of the ellipse for each factor indicates whether the factor affects either supply or retention, or both supply and retention of wrack in beach systems. Beach management refers to management practices such as beach grooming and armouring that influence supply and retention of wrack, while overgrazing refers to impacts on donor systems such as kelp forests due to grazing pressure.
The geomorphology (e.g. slope, width) and aspect (orientation) of beaches influence standing stocks of wrack (Table 2), primarily by altering the capacity to retain drift material (Revell et al., 2011; Liebowitz et al., 2016). For example, Barreiro et al. (2011) showed that small, wave‐sheltered beaches retain higher amounts of wrack than more exposed beaches in Spain, while deposition rates of buoyant material can differ substantially between windward and leeward shores (Lastra et al., 2014). Wrack retention can also be influenced by the elevation, and the width and slope of the drier, upper part of beaches (Zobell, 1971; Revell et al., 2011; Liebowitz et al., 2016). Similarly, substrate type influences the retention of wrack along shorelines. Consolidated rocky shores retain far less wrack than shores comprising sand, cobble or boulders (Wickham et al., 2020). Furthermore, cobble beaches appear to retain more wrack than gravel or sandy beaches (Orr et al., 2005). In addition, coastal armouring structures (seawalls, revetments) that reduce beach width and eliminate upper beach zones have been shown to have profound effects on the retention and standing stock of wrack (see Section III.1).
The proximity of beaches to donor ecosystems, and the form and condition of those systems, can strongly influence the supply of wrack to sandy shores. For example, in northern California, USA, the deposition of macrophytes on beaches varies depending on the source of wrack: subtidal reefs, rocky intertidal shores and estuaries (Liebowitz et al., 2016). Moreover, beaches within 0.5–1 km of donor ecosystems were most strongly influenced by wrack from those sources, while the role of more distant (7 km) kelp beds was only detectable during storm events (Liebowitz et al., 2016). Similarly, Reimer et al. (2018) showed that proximity of sources, ocean upwelling, estuarine outwelling, beach geomorphology and wave climate contributed to patterns of wrack on beaches in the US Pacific. Finally, wrack supply to beaches is influenced by the changing state of the donor systems. For example, excessive grazing by urchins can shift kelp forests to urchin barrens (Ling et al., 2009), reducing the availability of wrack material over timescales of years to tens of years and spatial scales up to tens of kilometres (Fig. 5).
(4). Links between wrack and fauna in beach ecosystems
(a). Invertebrates
Sandy‐beach ecosystems have traditionally been viewed as ‘harsh’ environments, characterised by low diversity and low abundance of intertidal invertebrates (Brown & McLachlan, 1990). This historical view, however, largely ignores the pivotal role of wrack and carrion for the sandy beach fauna as well as the associated biodiversity and productivity. The effect of wrack on overall intertidal biodiversity can be substantial, with wrack‐associated macrofauna (macroinvertebrates, >1 mm) making up, on average, >45% of the species present on beaches that receive inputs of giant kelp in California, USA (Dugan et al., 2003). In fact, intertidal species richness on Californian beaches is strongly correlated with kelp wrack abundance but not with beach morphodynamics (e.g. Dugan et al., 2003; Schooler et al., 2017). Wrack supports invertebrate biodiversity (Fig. 1A) by providing a food source for populations of specialized intertidal invertebrates and a refuge from predation and environmentally stressful conditions, such as desiccation (Dugan et al., 2003; Crawley & Hyndes, 2007; Colombini et al., 2009). Interestingly, it has also been shown that wrack availability can modify the balance of biological interactions (Duarte et al., 2010a ). However, in some cases, extensive wrack deposits can negatively impact some invertebrates through physical disturbance or anoxia, including meiofauna, (invertebrates <1 mm; McGwynne, McLachlan & Furstenburg, 1988) and macrofauna (e.g. wedge clam Donax serra; Soares, Schlacher & McLachlan, 1997), but few studies have examined these disturbance processes. Relatively few studies have examined meiofauna, yet they can be abundant in wrack (McGwynne et al., 1988; Alkemade & van Rijswijk, 1993; Jȩdrzejczak, 2002a ; Urban‐Malinga et al., 2008) and appear to play a crucial role in the decomposition of detrital material (see Section II.5.a ).
Wrack contains specialized mobile intertidal macrofauna, highlighted by the greater species richness and density of macrofauna on beaches with wrack compared with those with little or no wrack both in south‐western Australia (Ince et al., 2007) and the west coast of the USA (Dugan et al., 2003). Similarly, the removal of wrack on beaches in USA, Brazil and Spain significantly reduced the diversity and density of these specialized arthropods (Dugan et al., 2003; Schooler et al., 2017, 2019; Vieira et al., 2016). By contrast, the experimental addition of wrack to the upper parts of beaches resulted in higher arthropod abundances, particularly amphipods, and increased abundances of several beetle species in southern Australia (Schlacher et al., 2017). Correlation analyses of invertebrate abundance versus wrack biomass data extracted from the literature showed that beach‐cast macrophytes positively influence invertebrate species richness and abundance (N = 15, Fig. 6). The diverse suite of invertebrates that use wrack comprises three main trophic guilds: (1) ‘detritivores’ that feed directly on the detrital macrophytes; (2) bacterivores that feed mainly on wrack‐associated bacteria; and (3) ‘predators’ and ‘scavengers’ feeding on live prey or animal carcasses of the detritivores and bacterivores.
Fig. 6.

The numbers and proportions of papers with a focus on different aspects of total invertebrate assemblages (A) and amphipod populations (B) in wrack on beaches and in surf zones, and the relationship between amphipod abundance and wrack biomass (g wet mass m−2) (C) based on data extracted from the literature. In (A) and (B), circle quarters represent summaries of correlations of wrack biomass with invertebrate assemblages and amphipod populations (i.e. abundance, biomass, species richness, diversity). In each quadrant, the number of studies is displayed in parentheses, and the percentage of studies reporting a significant effect for each variable is illustrated by the coloured region in each quadrant (e.g. 83% of invertebrate studies reported positive effects on invertebrate abundance). In C, correlations are based on generalised additive models (GAMs) that assess the relationship between amphipod abundance and wrack biomass, with GAMS limited to four knots. See Table S2 for data sources.
Detritivores, represented almost exclusively by arthropods, form the most common and diverse trophic guild of macrofauna associated with beach‐cast wrack. Amphipods dominate this guild (Fig. 6), but other groups such as isopods and several beetle species (Tenebrionidae, Histeriidae and Curculionidae) are also common detritivores on stranded wrack in many areas (Griffiths & Stenton‐Dozey, 1981; Dugan et al., 2003; Jaramillo et al., 2006). Amphipods are almost ubiquitous in wrack deposits at temperate latitudes and are typically the most abundant taxon in fresh wrack deposits around the globe (Fig. 7). Amphipods in beach‐cast wrack mainly belong to the family Talitridae, including the genera Talitrus, Megalorchestia, and Orchestoidea. Importantly, although highly mobile and building a new burrow every day (Emery et al., 2022), amphipods and several other beach crustaceans brood their young, possess no planktonic life stages, and have limited dispersal as adults. This combination of traits makes these populations dependent on local reproduction and survival (Dugan et al., 2005; Hubbard et al., 2014). Generalised additive models of amphipod abundance versus wrack biomass data extracted from the literature show that wrack biomass strongly influences the abundance and biomass of amphipods, revealing a unimodal response where peak abundance of amphipods occurs at moderate levels of wrack biomass, but decreases at very high levels of wrack biomass (Fig. 6). At very high levels of wrack standing stock, the reduced abundance of amphipods likely reflects compaction, anoxia, and other unfavourable environmental conditions created by large amounts of decomposing organic matter (e.g. McGwynne et al., 1988). Combining their high consumer densities, which can reach 10,000–100,000 individuals per m beach (e.g. Lastra et al., 2008; Lowman et al., 2019), and their high feeding rates on wrack (see Section II.5.a ), amphipods play a crucial role in linking wrack subsidies from donor ecosystems to higher trophic levels in sandy‐beach ecosystems (see Section II.6).
Fig. 7.

Abundance rank of the main invertebrate taxa found on the beach and in the surf zone based on extracted data from the literature from across the globe. Dots indicate the normalised ranks (i.e. taxa in order of abundance, e.g. 1, 2, 3, etc., converted to values between 0 and 1, where 1 indicates the most abundant) of taxa in individual studies, while the vertical lines denote the mean rank and horizontal lines the 95% confidence intervals. Sample sizes (number of studies) are provided in parentheses. See Table S3 for data sources.
Dipteran flies form one of the most abundant bacterivore groups in wrack deposits and are almost ubiquitous in wrack across the globe (Fig. 7). Even though diets show considerable variation between dipteran families, bacteria are likely the most common food source for Diptera in the beach‐cast wrack, as shown for Coelopa frigida (Cullen, Young & Day, 1987). Seaweed or kelp flies, belonging to several families within Sciomyzoidae, are the most common dipteran group in wrack (e.g. Egglishaw, 1960; Dobson, 1974b ), but taxa such as Fucellia spp. (Anthomyiidae), Ephydridae, and Sphaeroceridae are also common in wrack and are typically important bacterivores (Cole, 1969; Griffiths & Stenton‐Dozey, 1981). The larvae of these flies feed on kelp, fungi and bacteria (Cullen et al., 1987), forming an important bacterivore compartment in the food web of beach‐cast wrack. Similar to amphipods, the abundance of kelp and seaweed flies is also strongly correlated with wrack biomass on beaches in California (Dugan et al., 2003). However, several much smaller fly species from several families are often overlooked in standard surveys (Cole, 1969) and would be worthy of future investigation.
The main groups of macrofauna feeding on detritivores and bacterivores in beach‐cast wrack deposits are beetles (particularly Staphylinidae, Carabidae and Histeriidae) and spiders (Moore & Legner, 1976; Griffiths & Griffiths, 1983; Mellbrand et al., 2011). Wrack‐associated beetles include some flightless species that complete their entire life cycle in the intertidal zones of beaches. For example, the staphylinid species Thinopinus pictus and Hadrotes crassus in North America have a specialised diet, feeding throughout their life cycle on amphipods (Orchestoidea) in beach intertidal zones (Craig, 1970; Richards, 1982), while another staphylinid genus (Aleochara spp.) parasitise seaweed fly larvae (Yamazaki, 2012). In general, only a few invertebrate species have been reported to predate on amphipods, with a wider range preying on the abundant dipteran larvae (Yamazaki, 2012). However, many species, such as most spiders, have a fairly broad diet (Verschut et al., 2019).
Since wrack deposition on beaches is generally highly dynamic over time, macrofauna utilising this resource need high intertidal mobility, temporary burrows and rapid development to make the best use of an essentially ephemeral resource (Dugan, Hubbard & Quigley, 2013; Emery et al., 2022). Mobility is also critical to avoid being washed out to sea with the wrack during high tides, but some species are also reported to have very low alongshore mobility (Schooler et al., 2017). Not surprisingly, stranded macroalgal deposits are often colonized by mobile macrofauna (particularly amphipods and dipterans) within a few hours of deposition, followed by predatory staphylinid beetles (Pelletier et al., 2011; Yanenik, 1980). Studies from the UK suggest that the life cycle of kelp flies (Coelopa sp.) is completed within 1 month, depending on temperature (Dobson, 1974b ). Rapid larval development is supported by the elevated temperatures within the masses of stranded wrack, particularly in larger, deeper wrack beds, where the temperature may be >10 °C above ambient levels (Crafford & Scholtz, 1987), and moisture levels are maintained (Kompfner, 1974), but not in smaller clumps where the temperature is more similar to air temperature (Dobson, 1974b ). Development coordinated with wrack deposition has also been suggested for some Coelopa spp., where larvae develop in spring high‐tide deposits of wrack and emerge from pupae by the next spring tide (Kompfner, 1974). Development times for different kelp fly species may be linked to tidal elevation, with species at lower elevations developing faster (Kompfner, 1974). The initial burst in colonisation of freshly stranded wrack by selected macrofauna is followed by a succession of other species (Yanenik, 1980; Colombini et al., 2009), and a gradual increase in meiofauna such as nematodes, oligochaetes and mites (Jedrzejczak, 2002b ), followed by coleopteran species (Griffiths & Stenton‐Dozey, 1981; Colombini et al., 2000).
In addition to quantity, the composition of the wrack input can influence invertebrate assemblages, but this effect seems to vary among consumer species (Mews, Zimmer & Jelinski, 2006; Olabarria et al., 2010). For example, in south‐western Australia, densities of invertebrates were greater in experimental plots of stranded seagrass compared with Sargassum wrack on the beach (Mellbrand et al., 2011), while the amphipod Allorchestes compressa showed a preference for macroalgae over seagrass in the surf zone (Crawley & Hyndes, 2007). Similarly, in southern California, amphipods (Megalorchestia spp.) preferred experimental wrack patches comprising kelp rather than seagrass, and different Megalorchestia species varied in their colonisation rates in wrack patches, perhaps to avoid competition with congeners (Michaud et al., 2019). The causes of these different affinities are likely to be complex when the macrophytes making up wrack vary in species, age, decomposition levels, and in terms of physical structure, nutrient content, palatability and bacterial community (Lowman et al., 2021) (see Section II.5.a ).
(b). Fishes
Wrack in the surf zones of ocean beaches provides important feeding, sheltering, and nursery habitats for a diversity of fishes (Fig. 1A; Crawley et al., 2006; McLachlan & Defeo, 2017; Ortodossi et al., 2019). Drifting macrophytes are widely reported to influence the abundance, diversity, and species composition of surf fishes (e.g. Robertson & Lenanton, 1984; van der Merwe & McLachlan, 1987; Andrades et al., 2014), but the ecological functions of wrack as a fish habitat remain poorly understood, largely because few studies (N = 20) have examined how macrophyte accumulations affect fish populations, assemblages, or food webs (Olds et al., 2018). Studies that examine the links between wrack and surf fishes come mainly from Australia (N = 12), limiting their broader generality and highlighting the need to gain a more global understanding of the role of drifting macrophytes for fish in surf zones. Nevertheless, the biomass of drifting macrophytes in surf zones can shape the composition of fish assemblages, modify patterns in fish diversity, abundance, and biomass, and alter the structure of coastal food webs (Crawley et al., 2006; Clark, Bennett & Lamberth, 1996b ; Baring, Fairweather & Lester, 2014; Vargas‐Fonseca et al., 2016). Our analyses showed that fish abundance (N = 15 studies) and biomass (N = 2 studies) are mostly positively correlated with standing stock of wrack, whereas fish diversity is greatest at moderate levels of wrack biomass (N = 9 studies) (Fig. 8).
Fig. 8.

Proportions of papers with a focus on different aspects of fish assemblages in the wrack in surf zones (A), and the relationship between fish abundance (B) and diversity (C) with wrack volume (litres per 100 m2) based on data extracted from the literature. In (A), circle quarters represent summaries of correlations with fish assemblages (i.e. abundance, biomass, species richness, diet). In each quadrant, the number of studies is displayed in parentheses, and percentage of studies reporting a significant effect for each variable is illustrated by the coloured region in each quadrant (e.g. 67% of studies on surf fish assemblages report positive effects of wrack biomass on fish abundance). In (B) and (C), correlations are based on generalised additive models (GAMs) assessing the relationship between fish abundance and diversity against wrack volume with GAMs limited to four knots. See Table S4 for data sources.
Most research on the role of surf‐zone wrack has focused on fish assemblages, whereas data on individual taxa are sparse (Clark, Bennett & Lamberth, 1996a ; Lacerda, Barletta & Dantas, 2014; Baring, Lester & Fairweather, 2016). Numerous fish species have been reported to prey on epifaunal amphipods that accompany drifting macrophytes (Crawley et al., 2006; Baring, Lester & Fairweather, 2018b ). A greater biomass of drift macrophytes has been reported to have positive effects on the abundance of several fish species (Robertson & Lenanton, 1984; Andrades et al., 2014; Baring, Lester & Fairweather, 2019). Generalised additive models of fish diversity versus surf‐zone wrack biomass extracted from the literature showed that, at least at low to moderate levels of wrack biomass there is a positive effect on fish diversity in surf zones by providing enhanced feeding opportunities (N = 9) (Fig. 8C). However, some species show either a neutral (Robertson & Lenanton, 1984) or negative (Crawley et al., 2006) diversity response to increasing wrack biomass in surf zones, presumably due to high densities of wrack impeding the movement and foraging of fish with particular morphological and feeding traits. Variation in the type of drifting macrophytes can also affect fish abundance, which is likely to reflect primarily the different affinities of their prey to different macrophytes as habitat and food. For example, the amphipod A. compressa displays a strong preference for brown algae as food and habitat (Crawley & Hyndes, 2007) and forms the main diet of juvenile fish with strong affinities to drifting wrack (Crawley et al., 2006). The types of drifting macrophytes in surf zones are therefore likely to influence the bottom‐up control of food webs in this ecosystem.
(c). Birds, mammals and reptiles
While highly mobile birds, mammals, and reptiles can use various resources across the landscape, many species show consistent associations with wrack or carrion as food resources on sandy beaches. Birds are often the most abundant vertebrates on sandy beaches, including shorebirds, waders, gulls, and a wide variety of terrestrial birds (from vultures to flycatchers) (Dugan et al., 2003). Many waders and passerines feed on wrack‐associated invertebrate prey (Lopez‐Uriarte et al., 1997; Dugan et al., 2003), while gulls, raptors, ravens, crows and vultures feed on carrion (Table 3). Wintering waders or shorebirds can reach high abundances (>95 individuals km−1) on wrack‐strewn open coast beaches and their numbers can be strongly and positively correlated with the abundance of stranded wrack and wrack‐feeding invertebrates on those beaches (Tarr & Tarr, 1987; Dugan et al., 2003; Hubbard & Dugan, 2003), reflecting their use of wrack‐associated invertebrates as prey (Griffiths, Stenton‐Dozey & Koop, 1983; Dugan et al., 2003; Schlacher et al., 2017). Wrack is particularly important for plovers, which are visual surface feeders; this group includes IUCN red‐listed species in many parts of the world (e.g. the Western snowy plover Charadrius nivosus nivosus on the California coast; Dugan et al., 2003).
Table 3.
Examples of scavengers and the carrion they consume on ocean‐exposed sandy beaches reported in the literature.
| Scavenger(s) | Carrion type | Location | Reference |
|---|---|---|---|
| Invertebrates | |||
| Whelks (Bullia rhodostoma, B. digitalis) | Jellyfish | South Africa | Brown (1961, 1971) |
| Gastropod (Reticunassa festiva) | Fish | Hong Kong | Morton & Yuen (2000) |
| Isopods (cirolanids) | Various drift organisms | USA (California) | Dugan et al. (2003); Quilter (1987) |
| Polychaetes | Various drift organisms | USA (California) | Dugan et al. (2003) |
| Beetles | Various drift organisms | USA (California) | Dugan et al. (2003) |
| Ghost crabs (Ocypode spp.) | Diverse/mixed carrion | multiple countries and locations | Wolcott (1978); Lucrezi & Schlacher (2014); Rae et al. (2019) |
| Reptiles | |||
| Lace monitor (Varanus varius) | Fish | Australia (East Coast) | Schlacher et al. (2013a , b ); Bingham et al. (2018) |
| Cottonmouth snake (Agkistrodon piscivorus conanti) | Fish | USA (Florida, Gulf Coast Islands) | Lillywhite et al. (2008) |
| Birds | |||
| Corvidae (crows and ravens) | Fish | Australia (East & South Coast) | Huijbers et al. (2013, 2015, 2016); Schlacher et al. (2013a , b ); Brown et al. (2015); Bingham et al. (2018) |
| Gulls (several spp.) | Fish | Australia (East & South Coast) | Huijbers et al. (2013, 2015, 2016); Schlacher et al. (2013a , b ); Bingham et al. (2018); Brown et al. (2015) |
| White‐bellied sea eagle (Haliaeetus leucogaster) | Various mammals, other birds, fish, and crabs | Australia (various coastal areas) | Smith (1985); Huijbers et al. (2013, 2015, 2016); Schlacher et al. (2013a , b ); Brown et al. (2015); Bingham et al. (2018) |
| Whistling kite (Haliastur sphenurus) | Various terrestrial and marine animals (rodents, reptiles, fish) | Australia (woodland and coastal areas) | Gosper (1983); Huijbers et al. (2013, 2015, 2016); Schlacher et al. (2013a , b ); Brown et al. (2015); Bingham et al. (2018) |
| Brahminy kite (Haliastur indus) | marine carrion such as fish and crabs | Australia (various coastal areas) | Smith et al. (1978); Lutter et al. (2006); Huijbers et al. (2013, 2015, 2016); Schlacher et al. (2013a , b ); Brown et al. (2015); Bingham et al. (2018) |
| Mammals | |||
| Coyote (Canis latrans) | Seals, birds, sea turtles, fish, marine arthropods | CA, USA | Rose & Polis (1998) |
| Lion (Panthera leo) | Cape fur seal carcasses | Namibia (Skeleton Coast) | Bridgeford (1985) |
| Tasmanian devil (Sarcophilus harrisii) | Fish | Australia (Tasmania) | Moore (2002); T.A. Schlacher, personal observations |
| Brown hyena (Parahyaena brunnea) | Cape fur seal carcasses | Namibia (Skeleton Coast) | Skinner et al. (1995); Kuhn et al. (2008) |
| Black‐backed jackal (Canus mesomelas) | Mammals (seal pups), birds, and fish | Namibia (Skeleton Coast) | Oosthuizen et al. (1997); Avery et al. (1987) |
| Dingo (Canis lupus dingo) | Broad range of stranded material including dugong and whale carcasses | Australia (East Coast, Fraser Island) | Moore (2002); Behrendorff et al. (2016, 2018b ) |
| Red fox (Vulpes vulpes) | Fish | Australia (East & South) | Huijbers et al. (2013, 2015, 2016); Brown et al. (2015); Bingham et al. (2018) |
| Dogs and cats (feral and domestic) | Fish | Australia (East & South) | Huijbers et al. (2013) |
Amongst mammals, many terrestrial species occasionally forage on living macrophytes or invertebrates in the lower intertidal zone during low tides (Carlton & Hodder, 2003). Populations of the coyote Canis latrans (Rose & Polis, 1998) and foxes (Cypher et al., 2014; Bingham et al., 2018; Schlacher et al., 2020) have been linked directly to the use of either marine‐derived carrion or wrack‐associated invertebrates as food sources (Table 3). Deer species have been observed foraging directly on beach‐cast macroalgae (Conradt, 2000). The side‐blotched lizard (Uta stansburiana) consumes marine algae (Barrett et al., 2005), and the brown tree lizard (Anolis sagrei) is strongly attracted to wrack (Spiller et al., 2010).
Far less is known about the use of wrack by birds, marine mammals and reptiles in the surf zone, although many species [e.g. ducks (Neff, Page & Boehm, 2011); otters and sea lions (Somers, 2000; Osterrieder, Salgado Kent & Robinson, 2017); sea turtles, penguins and sea lions (Tershy, Breese & Croll, 1997; Witherington, Hirama & Mosier, 2011; Colombelli‐Négrel, 2019)] occur regularly in the waters adjacent to beaches. Some birds, such as geese and swans (Percival & Evans, 1997; Choney et al., 2014), are known to feed on subtidal or intertidal seagrass in meadows along the shoreline of sheltered coastal systems. However, there is a paucity of studies linking birds, mammals, and reptiles to surf‐zone wrack as a food source or habitat, except for the cormorant Phalacrocorax varius which has been observed actively foraging for fish in surf‐zone wrack in Australia (Robertson & Lenanton, 1984). Clearly, there is a need to gain knowledge on the surf zone to understand the overall influence of wrack on higher‐order consumers.
(5). Biological processing and fate of wrack
(a). Decomposition and consumption processes
Wrack on sandy beaches represents a rich source of organic matter. The principal mechanisms breaking down this organic matter pool on sandy beaches are microbial decomposition, physical processing and consumption by intertidal meiofauna and macrofauna (Fig. 9A; Jȩdrzejczak, 2002b ; Lomstein et al., 2006; Lastra et al., 2008; Lastra, López & Neves, 2015; Rodil et al., 2015c ). All these mechanisms play a key role in determining wrack residence time on the beach (e.g. Mateo, 2010).
Fig. 9.

Conceptual diagram of: (A) decomposition and nutrient cycling; (B) the grazer/detritivore food web; and (C) scavenging pathways as key processes for the fate of stranded organic material in beach ecosystems. Diagrams created using IAN Image Library (http://ian.umces.edu).
Stranded macrophytes on beaches may have undergone decomposition for hours to days before stranding (e.g. Griffiths & Stenton‐Dozey, 1981; Colombini et al., 2000; Jaramillo et al., 2006). Once stranded, bacterial concentrations increase dramatically (Koop, Newell & Lucas, 1982a ; Cullen et al., 1987; Urban‐Malinga & Burska, 2009). For example, bacterial biomass increased 12‐fold on kelp (Ecklonia maxima) stranded for 8 days (Koop et al., 1982a ). The microbial communities associated with wrack likely develop from the biofilm of the macrophytes transported onto beaches rather than from the microbial community in the surrounding water (Singh et al., 2021). Macroalgae and seagrasses are covered in diverse microbial communities that include bacteria, microalgae, fungi, and protists, and are highly specific and distinct from the seawater microbiome (Wahl et al., 2012; Tarquinio et al., 2019). For example, Bacteriodetes and Proteobacteria are dominant phyla in the biofilm of macroalgae and seagrasses (Wahl et al., 2012; Tarquinio et al., 2019). Bacteriodetes may comprise up to 25–50% of the biofilm (Berdan et al., 2021), whereas this phylum makes up only a small portion of the seawater microbiome (Sunagawa et al., 2015). Bacterial assemblages vary somewhat among species of algae in wrack (Rodil, Fernandes & Mucha, 2015a ), similar to variation in biofilms across living macroalgae species (e.g. Staufenberger et al., 2008; Trias et al., 2012). Furthermore, the microbiome is likely to shift during the decomposition process, as occurs on kelp detritus on the seafloor (Brunet et al., 2021). The analysis of these microbial communities is still in its infancy, and there is a clear knowledge gap about microbial communities and their role in wrack and the broader sandy‐beach ecosystem.
As part of the decomposition process, heterotrophic bacteria digest macrophytes, converting organic carbon and nitrogen into simpler forms of dissolved organic and inorganic nutrients, which are released into the water column or incorporated into bacterial biomass (Säwström et al., 2016) and flow into higher trophic levels in wrack accumulations (Fig. 9A). The rates of decomposition can be influenced by chemical and morphological traits of the macrophytes forming the wrack (Duggins & Eckman, 1997; Bucholc et al., 2014). For example, the kelps M. pyrifera, Saccorhiza polyschides and Undaria pinnatifida are morphologically simple algae with soft, long, strap‐like blades that stack in layers on the sand (Lastra et al., 2008; Rodil et al., 2019). These algae can decompose rapidly through the joint action of microbes and detritivores (Rodil et al., 2019). By contrast, Sargassum muticum and Cystoseira baccata are morphologically more complex, with tough thalli bearing secondary and tertiary branches, and decompose much more slowly (e.g. Olabarria et al., 2010). The structural molecules in seagrasses, which are vascular marine plants, are refractory and slow to break down (Trevathan‐Tackett et al., 2017). These reduce both decomposition and consumption rates (see below) and allow large banquettes of seagrass to accumulate in some regions, such as the Mediterranean Sea (e.g. Mateo, Sánchez‐Lizaso & Romero, 2003) (see Figs 2C and 4).
Macroinvertebrate detritivores can rapidly process large quantities of wrack in sandy‐beach ecosystems. This can result in fragmentation which can enhance decomposition of wrack (Jȩdrzejczak, 2002b ; but see Catenazzi & Donnelly, 2007a ) and drive the food web (see Section II.4.b ). Talitrid amphipods can eat >50% of macroalgal wrack standing stock, thus playing a pivotal role in wrack turnover (Colombini et al., 2000; Lastra et al., 2008). Feeding preferences due to differences in palatability influence consumption rates in wrack accumulations (e.g. Michaud et al., 2019). Based on a range of studies examining feeding preferences, kelp and other species of brown algae are generally the preferred food source for talitrid amphipods, whereas seagrass species (e.g. Phyllospadix) and green algae (e.g. Ulva spp.) are generally the least preferred (see Tables 4 and 5). Similarly, analyses of published consumption data indicate that consumption rates by amphipods are greater for kelp, particularly Durvillaea and Egregia, than other macroalgae and seagrasses (Fig. 10). Differences in preference and consumption rates can vary among regions and life stages of wrack detritivores. For example, Duarte, Jaramillo & Contreras (2008) and Duarte et al. (2010b ) showed that adults of the amphipod Orchestoidea tuberculata preferred D. antarctica over the algae M. pyrifera and L. nigrescens, whereas juveniles preferred L. nigrescens (Duarte et al., 2010b ). Interestingly, the stark contrast in consumption rates between Durvillaea and Macrocystis reported from Chile (Duarte et al., 2010b ) was not found for Bellorchestia quoyana in New Zealand, where both kelp species were consumed at similar rates (Suárez‐Jiménez et al., 2017a ). These differences may reflect varying feeding preferences among amphipod species or differences in the nutritional quality of food sources between regions, making it difficult to generalise regarding consumption and resultant turnover rates of beach wrack, at least within brown algae (Emery et al., 2021; Lowman et al., 2021). However, the far lower consumption rates of seagrass (Crawley & Hyndes, 2007; Michaud et al., 2019) indicate that this type of wrack has much lower dietary benefits despite its high biomass in wrack in sandy‐beach systems in many regions (Fig. 4).
Table 4.
Ranking of different wrack components in terms of preference, growth and survival rates of amphipods and ghost crabs. Food sources that a consumer species preferred equally are given the same rank, where 1 is the highest/most preferred rank and 4 the lowest/least preferred.
| Variable | Taxa | Consumer species | Food source | Reference | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Kelp | Other brown algae | Red algae | Green algae | Seagrass | Other | Carrion | ||||
| Food preference | Amphipod | Allorchestes compressa | 1 | 1 | 2 | 2 | 4 | Crawley & Hyndes (2007) | ||
| Amphipod | Allorchestes compressa | 1 | 2 | 3 | 3 | 3 | Robertson & Lucas (1983) | |||
| Amphipod | Bellorchestia quoyana | 1 | 2 | Suárez‐Jiménez et al. (2017a ) | ||||||
| Amphipod | Megalorchestia corniculata | 1 | 2 | Lastra et al. (2008) | ||||||
| Ghost crab | Ocypode convexa | 2 | 2 | 2 + | 1 | Rae et al. (2019) | ||||
| Growth | Amphipod | Allorchestes compressa | 1 | 3 | 4 | 2* | Robertson & Lucas (1983) | |||
| Amphipod | Megalorchestia corniculata | 1 | 2 | Lastra et al. (2008) | ||||||
| Survival | Amphipod | Allorchestes compressa | 1 | 3 | 4 | 2* | Robertson & Lucas (1983) | |||
| Amphipod | Notorchestia sp. | 1 | 2 | 3 | Poore & Gallagher (2013) | |||||
Dune vegetation.
Fine particles.
Table 5.
Median percentage contribution of different types of food sources to the diets of consumers in wrack based on mixing model outputs of stable isotopes for consumers and potential food sources extracted from the literature.
| Taxon | Consumer species | Food source (%) | Region | Reference | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Brown algae | Red algae | Green algae | Seagrass | Dune plants | POM | Carrion | ||||
| Beach | ||||||||||
| Amphipod | Talitrus saltator | 16 | — | 5 | — | 32 | 29 | 16 | Atlantic (East) | Bessa et al. (2014) |
| Amphipod | Talorchestia brito | 31 | — | 12 | — | 17 | 22 | 19 | Atlantic (East) | Bessa et al. (2014) |
| Amphipod | Tylos europaeus | 22 | — | 18 | — | 13 | 31 | 13 | Atlantic (East) | Bessa et al. (2014) |
| Amphipod | Allorchestes compressa | 5 | 12 | — | 25 | 21 | — | — | Eastern Indian Ocean | Ince et al. (2007) |
| Amphipod | Talorchestia capensis | 8 | 9 | 8 | — | — | 9 | — | Southern Arica | Porri et al. (2011) |
| Ghost crab | Ocypode convexa | 17 | 9 | — | 9 | 15 | — | 10 | Eastern Indian Ocean | Rae et al. (2019) |
| Surf zone | ||||||||||
| Amphipod | Allorchestes compressa | 32 | 14 | — | 54 | — | — | — | Eastern Indian Ocean | Crawley et al. (2009) |
POM, fine particulate organic matter in sediment or water column.
Fig. 10.

Mean + SE consumption rates by amphipods feeding on different sources of wrack in beach and surf zone habitats. WW, wet mass. Data extracted from Duarte et al. (2008, 2010b ) (Chile), Lastra et al. (2008) (Spain), Gomes Veloso et al. (2012) (Brazil), MacMillan & Quijón (2012) (Canada), Poore & Gallagher (2013) (Australia), Michaud et al. (2019) (USA) and Suárez‐Jiménez et al. (2017a ) (New Zealand).
The nutritional quality of wrack obviously plays a critical role in determining feeding choices in macroinvertebrate detritivores on beaches (Lastra et al., 2008; Duarte et al., 2010b , 2011, 2014; Lowman et al., 2021), similar to mesograzers (macrofauna grazing on living macrophytes) in subtidal ecosystems (Duffy & Hay, 1991). The protein content of algae is considered a key trait in determining their nutritional quality as food sources (Cruz‐Rivera & Hay, 2000), similar to the case for plant consumers in other systems (White, 1993), and appears selectively to influence feeding by detritivores on sandy beaches (e.g. Duarte et al., 2011, 2014). Physical structure (shape and toughness) and the presence of chemical defences (secondary metabolites) have also been shown to influence food selection in these taxa (e.g. Pennings et al., 2000; Cruz‐Rivera & Hay, 2003; Duarte et al., 2011). As expected, growth rates and survival of detritivores are strongly linked to food preferences and nutritional quality (Table 4). For example, Lastra et al. (2008) showed that the preferred macroalgae species sustained the highest growth rates in M. corniculata, and this pattern also was observed for other amphipods (Duarte et al., 2010b , 2011).
Nutritional quality and palatability are affected by environmental factors and decomposition. Moisture and solar radiation influence the nutritional quality and palatability of wrack (Lastra et al., 2015; Rodil et al., 2015c ). Even small changes in ambient ultraviolet (UV) radiation may modify the structural and nutritional status of wrack (Rodil et al., 2015c ), while ocean acidification induces changes in algal palatability and protein concentration of macrophytes (Benítez et al., 2016; Duarte et al., 2016). However, the age of the detritus (related to the level of decomposition) and algal structure seem more important for food choice by detritivores in these systems (Pennings et al., 2000; Duarte et al., 2010b , 2011, 2014). Decomposition affects the nutritional quality (Rothäusler et al., 2005; Rothäusler & Thiel, 2006) and palatability of macrophytes for consumers, but the interactions between detritus, microbes, and detritivores make it difficult to disentangle the interactive effects of microbes and detritivores in the decomposition process.
In terrestrial systems, microbes are known to improve the quality and appeal of detritus as a food source (Zimmer & Topp, 1997), but they can also be consumed directly by detritivores (Thompson, Abreu & Cavalli, 1999). For sandy beaches, we know that nematodes and many dipteran larvae feed directly on bacteria (Porri, Hill & McQuaid, 2011; Urban‐Malinga & Burska, 2009; Cullen et al., 1987). Thus, their role is likely to be similar to that seen in saltmarsh systems, where meiofauna influence the decomposition of saltmarsh detritus and fluxes of nutrients via their influence on detritus‐associated microbes (e.g. Alkemade, Wielemaker & Hemminga, 1992; Lillebø et al., 1999), although their influence on decomposition can vary across taxa (De Mesel et al., 2003). Bacteria provide a more nutritious food source than the macrophytes themselves, as they have a lower C:N ratio (Fukuda et al., 1998), and higher levels of lipids and polyunsaturated fatty acids (de Carvalho & Caramujo, 2012). Indeed, growth and reproductive rates for the amphipod A. compressa were shown to be enhanced when bacteria were abundant on the kelp Ecklonia radiata (Singh et al., 2021). Clearly, a more comprehensive understanding of these interactive effects is required.
(b). Nutrient fluxes and chemical transformation
Sandy beaches have long been considered ‘biogeochemical hotspots’, ‘reactors’, or ‘digestors’, reflecting their high levels of nutrient fluxes and transformations (e.g. Pearse, Humm & Wharton, 1942). Wrack accumulations represent peak hotspots for biogeochemical processes as indicated by high metabolic rates that release high levels of CO2 (Coupland, Duarte & Walker, 2007; Gómez et al., 2018). Indeed, CO2 production by wrack accumulations on beaches can surpass the most active soils on Earth (Gómez et al., 2018). Similarly, wrack on beaches can be three times more metabolically active than subtidal seagrass or macroalgal beds (e.g. Coupland et al., 2007; Lastra, López & Rodil, 2018; Liu et al., 2019). By contrast, methane emissions from wrack appear to be negligible, at least under dry conditions (Liu et al., 2019), despite wrack having a demonstrated potential for biogas production (Kaspersen et al., 2016; Misson et al., 2020).
During consumption and decomposition of wrack in sandy‐beach systems, complex biomolecules are transformed to simpler organic forms and mineralised to inorganic nutrients (e.g. NH4, NO3 and PO4) (Coupland et al., 2007; Dugan et al., 2011; Lowman et al., 2019). As in other systems, heterotrophic bacteria likely play a major role in this conversion, but little work has been carried out in sandy‐beach systems. Rodil et al. (2019) is one of a few studies demonstrating a strong positive relationship between bacterial diversity and dissolved inorganic nitrogen in wrack deposits.
Since nitrogen is often the limiting nutrient in coastal marine ecosystems (Howarth & Marino, 2006), much of the focus of nutrient processes in beach ecosystems has been on N cycling, including standing stocks and fluxes (Mateo, 2010; Goodridge & Melack, 2014). Nitrogen produced during mineralisation of wrack can enter several pathways: (1) incorporation into the food web, primarily via uptake by surf‐zone phytoplankton and possibly by benthic microalgae; (2) flushing back to the sea; (3) loss to the atmosphere via denitrification; (4) incorporation into dune vegetation; and/or (5) immobilisation in recalcitrant pools after burial in the long‐term sedimentary sink (Fig. 9A) (McLachlan & McGwynne, 1986; McLachlan & Romer, 1990). Nitrogen fixation in bare sands, decaying wrack, or surf zones is probably a negligible component of the N cycle of these systems, while denitrification rates on exposed beaches are low and restricted to the few top centimetres of the sand (McLachlan & Romer, 1990). Denitrification only accounted for 2% of the N supplied by kelp or 5–10% of the overall N inputs to the beach (McLachlan & McGwynne, 1986). Both nitrification (oxidation of ammonia to nitrate to nitrite) and denitrification (reduction of nitrate to nitrogen gas) are processes mainly associated with the groundwater, likely reflecting N inputs from the land rather than from the wrack itself (e.g. Santoro, Boehm & Francis, 2006).
While the role of sandy beaches in the processing of organic matter and nutrient cycling has been repeatedly demonstrated, the question of whether beaches function as sources or sinks of nutrients remains largely unanswered, and there are differences among nutrient species and locations (e.g. Cockcroft & McLachlan, 1993; Goodridge & Melack, 2014; Prasad et al., 2019). Supporting the ‘sink’ hypothesis, buried beach‐cast kelp (Fucus spp.) has been shown to enhance the growth of pioneer dune vegetation in the Netherlands (van Egmond et al., 2019). The seagrass Posidonia oceanica provides an important nutrient source for adjacent dune vegetation in the Mediterranean Sea (Cardona & García, 2008; Jiménez et al., 2017), where wrack is dominated by seagrass (Fig. 4). However, supporting the ‘source’ hypothesis, field studies show that kelp decaying on the beach can rapidly leach high concentrations of dissolved nutrients [dissolved inorganic nitrogen (DIN), dissolved organic nitrogen (DON), dissolved organic carbon (DOC), total dissolved nitrogen (TDN) and total dissolved phosphorus (TDP); see Fig. 9A] for plant uptake and export (Koop & Lucas, 1983; McGwynne et al., 1988; Dugan et al., 2011). The concept of wrack‐covered beaches functioning as nutrient sources is supported by the correlation between wrack biomass and DIN concentration in surf‐zone water reported in California (Dugan et al., 2011) and by the increased productivity of local or invasive macroalgal species and phytoplankton blooms in waters adjacent to shorelines with increased wrack biomass load (Cockcroft & McLachlan, 1993). The dominance of a source or sink role may vary over time – leaching is likely to be greater in late summer and autumn when TDN fluxes from the beach are greater (Dugan et al., 2011), or during beach erosion episodes that release stored N in intertidal porewater to the ocean.
Our capacity to reject either hypothesis is limited by the lack of comprehensive studies examining nutrient dynamics in sandy‐beach ecosystems characterised by wrack input, and their ability to return nutrients to adjacent coastal ecosystems. For example, the outflow of DON may contribute to production in adjacent coastal systems as decomposing kelp is known to leach large amounts of DON (Hyndes, Lavery & Doropoulos, 2012). Exported DON is likely to be remineralised by highly abundant heterotrophic bacteria in the biofilm of living macrophytes (Egan et al., 2013; Tarquinio et al., 2019) in adjacent ecosystems (e.g. kelp forests or seagrass beds) and provide DIN to their hosts (e.g. Tarquinio et al., 2018). In addition, nutrient budgets need to consider all sources of nutrients, including groundwater. In some regions, the discharge of groundwater derived from further inland can contribute 50–99% of the total submarine groundwater discharge (Urish & McKenna, 2004; Li et al., 2011) and supply nutrients to beach ecosystems (Santoro et al., 2006; Loveless & Oldham, 2010).
(c). Scavenging
Carrion forms a highly nutritious and widespread food resource that is exploited by a rich diversity of scavengers (Table 3; Wilson & Wolkovich, 2011). Like detrital macrophytes, carrion supply is often highly variable in time and space, and is likely to be a nutritionally and energetically important resource in many beach food webs. There is generally no physical barrier to prevent animal carcasses from becoming stranded on beaches, or for scavengers to reach those carcasses, making them an easily accessible food source for consumers on ocean‐exposed shores (Schlacher et al., 2013a ). Scavengers can be categorised into two functional groups: (1) facultative scavengers, which feed on live prey as predators and on dead animals as scavengers (e.g. foxes, birds of prey), and (2) obligate scavengers that rely exclusively on carrion as their sole source of nutrition (e.g. vultures; Ruxton, 2004). It is, however, increasingly recognised that most predators will readily scavenge, and many marine carnivores will also consume dead animal matter (Britton & Morton, 1994).
A wide variety of scavenging species is found on sandy beaches worldwide, encompassing both invertebrates (e.g. whelks, isopods, polychaetes, beetles, ghost crabs, dipterans, etc.), and vertebrates (e.g. reptiles, raptors, and carnivorous mammals) (Table 3). Some of the best‐studied invertebrate scavengers on sandy beaches are gastropods, particularly the genus Bullia in South Africa (Brown, 1961), which rapidly detect and consume a wide range of carrion (Brown & McLachlan, 1990). Crustaceans (e.g. isopods, amphipods, decapods) contain many taxa that consume carrion (Brown & McLachlan, 1990). For example, Tylos latrielle shifts its diet ontogenetically, with juveniles preferring carrion, whereas adults prefer seaweed (Kensley, 1974; Brown & McLachlan, 1990). Hippid crabs (Hippa spp.) are important scavengers on tropical island beaches, catching and consuming Portuguese man of war jellyfish (Physalia physalis) washed onto the beach (e.g. Wenner, Ricard & Dugan, 1987; Lastra et al., 2016). Ghost crabs (Ocypode spp.) are the largest invertebrates on many sandy beaches, occupying a range of trophic levels formed by a diverse diet that regularly includes dead animal flesh (Lucrezi & Schlacher, 2014). In fact, when given a choice, ghost crabs strongly prefer carrion over algae and plants (Rae, Hyndes & Schlacher, 2019). The strandline of beaches also harbours a rich fauna of insect carrion feeders, including a high diversity of Coleoptera (Rozen, Engelmoer & Smiseth, 2008; Irmler, 2012). However, despite insects being considered to be important scavengers of animal carcasses in other terrestrial systems (Redondo‐Gómez et al., 2022), little is known about their role as scavengers on sandy beaches (Blandford et al., 2019).
Reptiles, birds and mammals are functionally important scavengers in many ecosystems, and their role in sandy‐beach ecosystems is becoming more apparent. In Australia, lace monitors (Varanus spp.) are widespread consumers of birds, fish, mammals, amphibians, eggs, and insects, and carrion can at times dominate their diet (Guarino, 2001). Indeed, lace monitors regularly consume fish carcasses (Schlacher et al., 2013b ; Bingham et al., 2018). Similarly, in the USA, cottonmouth snakes (Agkistrodon piscivorus conanti) traverse areas of vegetation at the beach's edge and feed on fish that have been discarded, or regurgitated, by colonial waterbirds (Lillywhite, Sheehy III & Zaidan III, 2008). Also, birds such as raptors can dominate carrion consumption in landscapes not strongly altered by urbanisation, followed by corvids and gulls (e.g. Huijbers et al., 2016). Similarly, many mammals are attracted to, and feed on, stranded dead animals on sandy beaches, including hyenas (Parahyaena brunnea), black‐backed jackals (Canus mesomelas), coyotes, dingos, foxes (Vulpes vulpes), feral pigs, and even lions (Table 3). Strandings of cetaceans and dead seals are prominent examples of carrion providing intermittent bounties for carnivores (Behrendorff, Belonje & Allen, 2018a ).
(6). Connectivity with adjacent ecosystems
The transfer of drift macrophytes and carrion from the sea to the beach can provide a significant energy subsidy to beach ecosystems. Such ‘spatial subsidies’ across highly permeable ecosystem boundaries increase secondary productivity and biodiversity in systems that contain otherwise low in situ primary productivity. In the preceding sections, we have highlighted that seagrasses, and particularly brown algae (mainly kelp), provide the main vectors for this subsidy (sensu Hyndes et al., 2014) by supporting food webs as well as creating habitats for a diversity of microbes, invertebrates, fish, birds, reptiles and mammals in beach ecosystems (Fig. 9B,C). Biological and physical processes can recycle this imported material through consumption, fragmentation and decomposition, releasing dissolved nutrients back into the water or transferring nutrients through the food web and ultimately exporting those nutrients into other coastal ecosystems on land or in the sea (Fig. 9B,C). However, as discussed in Section II.5.b , the role of these systems as a source or sink of nutrients is equivocal, and the spatial extent and magnitude of any export of nutrients into other ecosystems is generally not well quantified for most settings.
Wrack deposits on the beach are often concentrated close to the waterline but regularly extend higher on the shore through the action of high tides, storm surges, and wind‐driven transport. Further inland transport of wrack‐ or carrion‐derived material depends on its direct or indirect consumption or transport by more mobile animals and their subsequent inland movement. For instance, Mellbrand et al. (2011) showed that seaweed flies feeding on wrack, and predators such as spiders feeding on detritivores in wrack, may move marine carbon many metres inland. However, this movement was not detectable beyond the primary dune, most likely due to the dilution of marine‐derived material as other land‐based food sources become more available, or the limited movement of those invertebrates feeding directly or indirectly on marine‐derived material, or a combination of both.
Larger and more mobile consumers are likely to provide a greater role as vectors for the inland transport of marine‐derived material. Since invertebrates associated with beach‐cast wrack provide an important food source for a variety of birds, such as plovers, swallows, and flycatchers (e.g. Dugan et al., 2003; Schlacher et al., 2017), it logically follows that this marine‐derived material may enhance the breeding success and productivity of these birds. Such bottom‐up control of populations is also likely for other mobile animals that assimilate nutrients from invertebrates associated with beach‐cast wrack, such as lizards (Barrett et al., 2005; Spiller et al., 2010), rodents, foxes, and bears (Ricci et al., 1998; Stapp & Polis, 2003; Fox et al., 2014; Davidson et al., 2021; Page et al., 2021) as well as feral pigs (M.A. Mateo & J.E. Dugan, personal observations). Also, deer can forage directly on wrack (Conradt, 2000), while domestic livestock, such as pigs, sheep and cattle, can be common on beaches in some regions (J.E. Dugan, C. Duarte & M.A. Mateo, personal observations), where they may graze on wrack. However, this mechanism has not been tested, and the magnitude of the subsidy for these mobile consumers will depend on: (1) the proportion of their food derived from the wrack; (2) the inland extent of their movement; (3) the extent to which they contribute to higher trophic levels; and (4) the release of nutrients in inland areas through their faeces or carcasses.
The consumption of carrion by scavengers on sandy beaches illustrates a broader functional role concerning the transfer of nutrients and organic matter across ecosystem boundaries, linking food webs at the landscape scale and creating meta‐population dynamics in the consumers. The mobility of birds makes them pivotal vectors for transferring nutrients and energy across ecotones, including consumption and transfer of carrion‐derived matter across surf–beach–dune landscapes (Fig. 9C; Whelan, Wenny & Marquis, 2008). Flying enables birds to search over large areas and detect patchy resources (i.e. carcases) in ways generally not possible for other consumers. The spatial extent of other mobile scavengers, such as rodents and foxes, is also likely to be substantial. One of the most seminal contributions to understanding the pivotal role of carrion subsidies on sandy shores comes from Rose & Polis (1998). They showed that coyote populations were 2–14 times higher on the coast compared to upland areas. In their study system, food supply that included carcasses washed ashore was greater on the coast.
Wrack stranded high on the shore may also influence both vegetation and the landscape structure of this dynamic zone. For example, seasonal pulses of wrack, primarily Sargassum, deposited by storms have been shown to enhance foliage growth of native shrubs and trees (Spiller et al., 2010). Such wrack deposits may provide nutrients, propagules and a favourable microhabitat for terrestrial plants, particularly the salt‐tolerant pioneering species typical of coastal strand and foredune habitats (Dugan & Hubbard, 2010). The propagules of many dune plants can be transported and delivered with wrack and other drift material (e.g. Hesp, 2002). Similar to pioneering dune plant species, wrack deposits can act as ecosystem engineers that influence the geomorphology of shorelines by trapping wind‐blown sand to form hummocks and embryo dunes, at least in the short term (Nordstrom, Jackson & Koroty, 2011a ). These features can then buffer beaches and dunes from erosion during storms.
The return of wrack‐derived nutrients back into coastal waters is likely to occur through two main processes. Firstly, the breakdown and decomposition of wrack release large quantities of dissolved nutrients (e.g. Dugan et al., 2011) and fine particulates (Soares et al., 1997) that provide vectors for the return of nutrients back to the sea. However, the spatial extent of this return of nutrients is largely unquantified. It is likely that dissolved nutrients are rapidly diluted, but they may be utilised by intertidal and shallow subtidal primary producers, such as surf‐grasses and kelps (Dugan et al., 2011). However, it does appear that organic matter derived from detrital kelp along the beaches of South Africa supports populations of the subtidal filter‐feeding bivalve Donax serra (Soares et al., 1997). Furthermore, while some studies have concluded that fine particulate kelp supports filter and suspension feeders in other coastal systems (e.g. Stuart, Field & Newell, 1982; Duggins, Simenstad & Estes, 1989), the evidence is equivocal (Miller & Page, 2012; Yorke et al., 2013). Thus, the supply rate of particulate kelp from beaches may be important for suspension feeders in coasts across the globe, a predictive hypothesis that requires testing. Secondly, surf‐zone wrack can provide an important feeding and sheltering habitat for fish, particularly for juveniles. For example, some fish feed almost exclusively on wrack‐associated amphipods in the surf zones of south‐western Australia (Crawley et al., 2006). Through the ontogenetic movement of those juveniles towards their more offshore spawning grounds, they form vectors for the return of wrack‐derived nutrients into other marine ecosystems across coastal seascapes. However, the spatial extent and magnitude of this mechanism remain unknown. The degree to which the return of nutrients from wrack on the beach and in the surf‐zone to adjacent coastal systems, regardless of the vector, occurs is likely to differ according to: (1) different types and periodicities of subsidies (e.g. kelp, seagrass, carrion) supplied to sandy‐beach ecosystems; (2) the residence time and dominant processes (e.g. decomposition, grazing, transport) acting on the wrack in those systems; (3) the type of vectors (e.g. fish, bird, reptile, mammal, invertebrate); (4) physical processes (e.g. tide and storm surges) that erode beaches and export nutrients and wrack; and (5) the seascape and landscape contexts.
III. HUMAN USE, IMPACTS AND MANAGEMENT
(1). Beach grooming and harvesting
Once stranded on beaches, humans can impact wrack deposits by grooming and harvesting (Fig. 1B). Beach grooming intentionally removes macrophyte wrack, litter and other debris from beaches, usually through raking and sieving the sand using specialised heavy equipment often on beaches in populated or urban areas (Fig. 2G,H; Dugan et al., 2003; Fanini, Cantarino & Scapini, 2005; Dugan & Hubbard, 2010). Grooming can be intensive and frequent (daily to weekly). It can have substantial adverse environmental effects, impacting the habitat quality, biodiversity, geomorphology, and functioning of beach ecosystems. Physical disturbance caused by grooming stymies dune formation and plant colonisation (Dugan & Hubbard, 2010). It also reduces the species richness, abundance, and biomass of wrack‐associated fauna, such as amphipods, isopods, beetles and flies (Dugan et al., 2003; Gilburn, 2012; Schooler et al., 2019). In southern California, impacts of widespread grooming have contributed to local and regional losses of populations of vulnerable wrack‐dependent taxa, such as isopod species occurring only on beaches (Hubbard et al., 2014). Although meiofaunal communities can recover quickly (within 24 h) from a single, short‐term grooming event (Gheskiere et al., 2006), the consequences of repeated, regular beach cleaning may be significant for these intertidal communities. These direct impacts are likely to extend through the food web and to affect mobile predators, such as shorebirds that rely on wrack‐associated prey as food. Also, since wrack can supply nutrients to the adjacent sea (see Section II.5.b ), its removal from beaches may impact the donor and other neighbouring marine ecosystems by depriving them of a substantial part of their recycled production (e.g. Mateo et al., 2003).
The practice of beach grooming or raking is widespread globally and is often a component of well‐established management regimes for beaches used for tourism and recreation (Davenport & Davenport, 2006). For example, around 45% (>150 km) of sandy beaches are groomed at least seasonally in densely populated southern California, USA (Dugan et al., 2003), while >106,000 m3 of Posidonia wrack are estimated to be removed in 1 year from 44 beaches on the island of Sardinia (de Falco, Simeone & Baroli, 2008). Beach rating systems and ‘ecolabels’ often contain criteria that encourage wrack removal (Zielinski, Botero & Yanes, 2019). The Blue Flag Program is the largest of these ecolabels and includes over 4000 beaches in 47 countries (Boevers, 2008). Klein & Dodds (2018) note that this program emphasises tourism promotion over environmental protection and conservation of beach ecosystems. Indeed, many beaches rated under the Blue Flag scheme are very likely to have management regimes that remove wrack, thereby causing environmental harm (Mir‐Gual et al., 2015; Gilburn, 2012). Thus, the concept of ‘cleaning’ the beach ignores the ecological and conservation value of wrack for the beach ecosystems and broader seascapes/landscapes.
Macroalgal wrack is regularly harvested in some regions of the world, including Chile, North America, Ireland and Australia (Kirkman & Kendrick, 1997; Holden et al., 2018a ). Native macroalgal species are harvested for various uses, including production of alginate and agar, cattle feed, soil fertilizers and conditioners, and feed for abalone hatcheries; seagrasses are harvested for insulation and soil fertilizer (Kirkman & Kendrick, 1997). Methods and equipment used in harvesting macrophytes from beaches vary from artisanal hand picking (a few tonnes y−1) to commercial harvesting using heavy equipment, such as bulldozers (hundreds to thousands of tonnes y−1, Holden et al., 2018a ). Harvesting can remove large proportions of beach wrack. For example, harvests of drift bull kelp (D. potatorum) on Australia's King Island (average harvests of 2500 tonnes year−1; Holden et al., 2018a ) account for approximately 50% of the kelp deposited annually on those beaches (Kirkman & Kendrick, 1997). Non‐native species may also be harvested. For example, quotas of 900–1500 tonnes year−1 for the invasive red alga (Mazzaella japonica) accounts for <16% of the available biomass along a shoreline section of Canada's Vancouver Island (Holden et al., 2018a ). Overall, the harvest methods and their associated impacts, and the relative magnitude of wrack harvest are not well documented (see Kirkman & Kendrick, 1997; Holden et al., 2018a ), suggesting this is a significant gap in the information needed to manage wrack harvest on sandy beaches. A report on impacts of wrack harvest in Ireland recommended using non‐mechanical harvest methods, prohibiting removal of sediment or substrate and minimising disturbance to surrounding environments by commercial wrack harvest (McLaughlin et al., 2006 cited in Holden et al., 2018a ).
(2). Shoreline armouring and coastal development
Many shorelines, including those formed by sandy beaches, have been profoundly altered through the construction of coastal armouring structures and extensive shoreline development (Charlier, Chaineux & Morcos, 2005) (Fig. 1B). Coastal development (e.g. ports, marinas, resorts) can cover, or remove sandy beaches, thereby profoundly impacting the habitat value and function of beach ecosystems (see Dugan et al., 2012; Hubbard et al., 2014; Jaramillo et al., 2021). Shoreline armouring, such as seawalls and revetments, is a common practice around the world to protect coastal development and infrastructure from erosion and coastal hazards (Airoldi et al., 2005; Dugan et al., 2012), and has received the most attention with regard to environmental impacts (Dugan et al., 2008, 2012, 2018). Seawalls have been shown to reduce the overall width of sandy beaches, with the most significant impacts in the upper part of the intertidal zone (Dugan et al., 2008; Jaramillo et al., 2021). The resulting losses in the upper beach zone, suitable for the retention of wrack, disrupt the trophic subsidy provided by donor ecosystems and significantly reduce the diversity and abundance of wrack‐associated invertebrates (e.g. Dugan & Hubbard, 2006; Dugan et al., 2008; Jaramillo, Dugan & Hubbard, 2012; Jaramillo et al., 2021; Dethier et al., 2016). Importantly, these impacts extend up the food web, where armouring can significantly reduce the use of beaches by shorebirds and seabirds (Dugan & Hubbard, 2006; Dugan et al., 2008). Similar impacts may also extend to surf zone fishes, but more research is needed to evaluate this. Other forms of armouring, such as groynes and detached breakwaters, may increase or decrease the standing stock of wrack on beaches and in surf zones, depending on the scale, orientation and design of those structures (e.g. Airoldi et al., 2005; Dugan et al., 2012; Martin et al., 2005).
Although small‐scale beach‐restoration efforts suggest that biodiversity and ecosystem functions of beaches can be restored through the removal of armouring structures (Lee et al., 2018), increased armouring of shorelines is the expected global trend as coastal hazards increase with climate change. As sea level rises, the effects of coastal squeeze exerted by existing armouring structures on beaches are also expected to increase as structures interact more frequently with waves and tides (Dugan et al., 2018). Robust evaluations of the ecosystem services provided by intact dune–beach–surf‐zone systems, and the protocols for quantifying the often dynamic indicators of those services, are generally not available for proposed armouring projects (King et al., 2018). As a result, other than recreation and storm buffering, values of the ecological functions and services of beaches are rarely applied to assess the impacts of these projects. New approaches to evaluating beaches as ecosystems, based on restoration or replacement costs, are urgently needed for mitigating the impacts of shoreline armouring, especially as pressures from sea level rise and coastal squeeze intensify (e.g. King et al., 2018).
(3). Invasive species
Numerous species of non‐native algae have been introduced to coastal waters, either via human activities (e.g. shipping) or as a result of species range shifts due to ocean warming (Hurd et al., 2014). For example, the green alga Codium fragile and the kelp Undaria pinnatifida, both originally from the north Pacific Ocean, now have near‐global distributions (Hurd et al., 2014). The ecological impacts of invasive species on subtidal habitats are equivocal [Suárez‐Jiménez et al. (2017b ) and references therein], but we know far less about the impacts of these invasive species as beach‐cast wrack (see Quijon, Tummon & Duarte, 2017) even though they can be commonly cast ashore (e.g. Piriz, Eyras & Rostagno, 2003; Rodil et al., 2008). Certainly, the composition, quality and perhaps the biomass, of the beach‐cast material could be affected by the displacement of native reef algae with invasive species, thereby altering wrack inputs and food value. However, this effect will depend on the life‐cycle patterns and buoyancy of the invasive species, which alter the ability of algae to be transported to the beach and the timing of the detrital input. For example, U. pinnatifida is now a major component of subtidal reefs in New Zealand, but contributes little to beach wrack, which likely reflects its lack of buoyant structures (e.g. pneumatocysts or buoyant blades) which are present in the native M. pyrifera and D. antarctica that are common in beach wrack in the region (Suárez‐Jiménez et al., 2017a ). However, the invasive brown alga Sargassum horneri is an annual species that can outcompete native kelp species and can seasonally dominate the wrack deposited on some Californian beaches (Marks, Reed & Holbrook, 2020). Beach‐cast of its congener S. muticum appears either to alter or to have no effect on invertebrate composition and densities compared to native macroalgal species (Rodil et al., 2008; Cacabelos et al., 2010). While S. muticum appears to contribute to the food web on some beaches (Olabarria et al., 2009), the magnitude of an invasive species' influence on beach food webs will depend on the ability of native consumers to utilise the new resource and its nutritional quality and palatability. For example, while the invasive U. pinnatifida has similar nutrient characteristics to native species in New Zealand, the consumption of this invasive species by the amphipod B. quoyana appears to be hindered by its physical properties such as toughness (Suárez‐Jiménez et al., 2017b ). With few studies focusing on the impacts of invasive species on the food webs and ecosystem structure of beach systems, it is presently not possible to make generalisations regarding this potential impact.
(4). Climate change impacts to recipient and donor ecosystems
Arguably, the greatest threat to sandy‐beach ecosystems is the effect of climate change, especially via sea‐level rise, more intense and frequent storms, and ocean warming (Fig. 1C). Sea‐level rise and storm frequency are projected to intensify beach loss, through increased erosion rates, inundation, and coastal squeeze (Vitousek et al., 2017). These processes will significantly impact beach ecosystems and their functioning, including the loss of habitat available for wrack deposition and retention and the survival of associated biota (see Schlacher et al., 2008; Myers et al., 2019). The impacts of sea‐level rise on beaches are projected to manifest earliest in the upper zones of beaches where wrack deposits support biodiversity and coastal food webs (Dugan et al., 2012, 2013). These vulnerable zones have already been widely impacted by coastal armouring and development, sediment starvation, and beach management practices (Dugan et al., 2008, 2013; Myers et al., 2019). A case study of several Californian beaches projected that a 0.5 m increase in sea level would result in a 75% loss of the upper beach zone where wrack accumulates and is processed, eliminating around 50% of intertidal biodiversity and numerous vital ecosystem functions (Myers et al., 2019). Furthermore, recent ENSO events that cause a combination of warming, storms and sea‐level rise (i.e. are a proxy for climate change), combined with a multi‐year drought, have caused historically high levels of beach erosion and loss, with beaches retreating beyond previous extremes along the southern portion of the north‐eastern Pacific coast (Barnard et al., 2017). While beach nourishment may be seen as a mitigation measure against this impact (de Schipper et al., 2021), burial of wrack and carrion, and changes to the deposition dynamics of the shoreline are likely to impact ecological processes in sandy‐beach ecosystems.
Ocean warming and related marine heatwave events will also strongly affect key donor ecosystems that supply organic material in the form of drift macrophytes to beaches and surf zones (Fig. 1C). Important donor ecosystems, such as kelp forests, are already being affected by ocean warming worldwide (Wernberg et al., 2019). Heatwave events have led to local extinctions of the kelp E. radiata (Wernberg et al., 2016) and severe reductions in the biomass of seagrass meadows (Arias‐Ortiz et al., 2018) along the west coast of Australia. Climate change and other human impacts have already led to declines in seagrasses and kelp, with declines of 7 and 2% year−1 in recent years, respectively (Waycott et al., 2009; Krumhansl et al., 2016). Furthermore, increasing sea temperatures have either led to, or are predicted to, extend the ranges of tropical macrophytes and consumers into higher latitudes (temperate regions) (Vergés et al., 2014; Hyndes et al., 2016). Ultimately, these climate‐driven effects will limit the supply, or alter the form, of macrophytes and alter the ecosystem functioning of sandy‐beach ecosystems in those regions. For example, ocean warming is projected to reduce the body size and fecundity of intertidal biota such as talitrid amphipods that are key consumers of wrack (Jaramillo et al., 2017), which is likely to alter the rates of wrack processing by detritivores on beaches. Another global impact, ocean acidification, is predicted to alter the nutritional quality of algae, possibly translating to altered trophic dynamics of consumers processing this wrack material (Benítez et al., 2016; Duarte et al., 2016).
To understand these processes, we need studies conducted at larger spatial and temporal scales, involving the dynamics of wrack inputs and connectivity of beaches to source ecosystems, presumably using data from remote sensing and other synoptic resources. For example, wrack on tropical beaches have essentially been ignored, yet they are experiencing increased deposition of Sargassum (Maurer, de Neef & Stapleton, 2015; Schell, Goodwin & Siuda, 2015), and other macroalgal inputs as coral reefs transition to turf macroalgae (Sura et al., 2019). Quantifying the biomass and composition of wrack will play a critical role in determining the shifts in supply over these timescales, and particularly in relation to the impact of the shifting state of donor systems due to climate change and invasive species.
IV. FUTURE DIRECTIONS
Some of the priority research directions emerging from our review include, but are not limited to, five broad themes:
Quantifying the rates of processing of wrack inputs by the entire suite of organisms from microbes to higher order consumers and how their interactions influence wrack persistence and turnover rates.
Developing comprehensive nutrient budgets in beach ecosystems, including the export of nutrients to inland and other coastal systems, which will ultimately develop a predictive understanding of the magnitude of the role of wrack in seascape connectivity.
Quantifying the rates of input and turnover of animal carcasses over a broad range of species and regions that encompass the diversity of vertebrate carnivores/scavengers.
Examining the role of microbes and meiofauna in driving secondary production and food webs in the beach ecosystem.
Determining the magnitude of flows, and their spatial footprint, for nutrients originating from wrack and carrion through coastal food webs over a range of different seascape settings, including the movement of predators (e.g. fish, birds, mammals) and scavengers (e.g. birds, mammals) ultimately linked to marine energy stranded on sandy beaches.
Evaluating the effects of human impacts, including climate change and ocean acidification, through shifts in the quantity and quality of wrack inputs affecting ecosystem functioning of sandy beach systems.
V. CONCLUSIONS
(1) Sandy beaches are iconic features of our coastlines, globally prized for providing valuable ecosystem services such as coastal protection, support of wildlife, fisheries, unique biodiversity, and the creation of tourism and recreation opportunities (Barbier et al., 2011). We show that large quantities of detrital macrophytes can flow into and be processed in this shoreline ecotone worldwide, albeit highly variable in amount and composition. Supply and retention of wrack are influenced by the oceanographic processes that transport it, the geomorphology, orientation and landscape context of beaches, and the condition, life history and morphological characteristics of species that produce the wrack in the marine donor ecosystems.
(2) Wrack deposits on beaches often create hotspots of microbial metabolism, secondary productivity, biodiversity and nutrient remineralisation. Decomposing wrack produces dissolved organic and inorganic nutrients that can return to coastal waters. However, the magnitude and spatial extent of the return of nutrients to other coastal ecosystems is largely unknown. Furthermore, there is a clear need to understand the role of microbes in driving secondary production and biodiversity of macrofauna, and to a lesser extent meiofauna.
(3) Seagrass and kelp are typically the main components of wrack. Many mobile invertebrates of the intertidal zone, particularly amphipods, prefer kelp as a food source, making kelp the most energetically important carbon source in food webs compared to seagrasses.
(4) The invertebrate consumers of wrack and associated microbes, especially amphipods and dipterans, channel energy up the food chain to predatory invertebrates (beetles), fish, birds and occasionally mammals. Large and mobile consumers can become vectors that transport wrack‐derived nutrients across habitat boundaries and more broadly across coastal seascapes. The magnitude and spatial scale of such transfers is currently not quantified.
(5) Increasing wrack biomass in surf zones increases fish diversity and abundance, although diversity declines at very high wrack biomass. Surf‐zone wrack appears to play an important nursery role for some fish species despite its transient nature, but it remains unclear how important this habitat is compared to other potential nursery habitats, and their relative importance to the spawning biomass of those species.
(6) The energetic role of beach‐cast marine carrion in sandy‐beach ecosystems is likely widely underestimated, as it can be removed very rapidly by highly mobile scavengers. These scavengers are predicted to be pivotal biological vectors that transfer marine productivity inland, thereby linking marine and terrestrial ecosystems. However, we currently have very little data on the quantity of carrion on beaches, and the magnitude, frequency, geographic distribution, and spatial scales of these sea‐land couplings performed by scavengers.
(7) Wrack is frequently removed to ‘improve’ the aesthetic value of beaches used intensively for recreation. However, beach grooming also removes the multiple ecological values created by wrack (food, habitat, dune formation). Similarly, shoreline structures such as seawalls impact the supply and retention of wrack and its ecological value. The juxtaposition of conserving wrack whilst clearing wrack or protecting shorelines provides a major management challenge. Consequently, conservation efforts should consider improving societal awareness on the ecological importance of stranded organic matter, which is currently viewed in the same light as anthropogenic debris (rubbish, litter).
(8) Rising ocean temperatures and sea levels due to climate change will alter the supply, deposition and retention of wrack and carrion in sandy‐beach ecosystems and alter the processing of organic material in these systems. Losses of donor material and the ability of beaches to retain material provide the greatest and most widespread threat to the ecosystem functions and services provided by subsidies of wrack and carrion in sandy‐beach ecosystems. This impact will be more pronounced in temperate regions around the globe where kelp subsidies are a prominent component of wrack. To understand the potential level of this impact, we need more data on the quantities and forms of detritus exported from donor systems, the distances that these forms of detritus travel to recipient systems, the retention and fate of detritus in those systems, and the likely loss of ecosystem functions related to reduced detritus in sandy beaches and connected ecosystems.
Supporting information
Table S1. All publications identified in our search on various aspects of wrack or carrion in sandy‐beach ecosystems, and their locations, habitats and research themes.
Table S2. References used to produce Fig. 6.
Table S3. References used to produce Fig. 7, including details of the country (region), habitat sampled and type of material in wrack (mixed = seagrass and macroalgae).
Table S4. References used to produce Fig. 8.
ACKNOWLEDGEMENTS
We thank our various institutes for making the time and effort to compile this review possible, and Lorraine Prentis at Edith Cowan University for help finalising the references. E. L. B. was supported by a Marie Skłodowska‐Curie fellowship 704920‐Adaptive Inversions‐H2020‐MSCA‐IF‐2015. During the writing of this manuscript, C. D. was under the tenure of the Fondo Nacional de Desarrollo Científico y Tecnológico grant No 1200794. J. E. D., K. A. E. and D. M. H. gratefully acknowledge support from the U.S. National Science Foundation (OCE 1458845) and from the Santa Barbara Coastal Long Term Ecological Research program (SBC LTER, OCE 1232779 and 1831937). Any opinions, findings, or recommendations expressed in the paper are those of the authors and do not necessarily reflect the view of the U.S. National Science Foundation.
REFERENCES
References identified with an asterisk (*) are cited in the Supporting Information.
- * Adamson, M. L. & Rigby, M. (1996). Rhabditis (Criistorhabditis) stasileonovi (Belogurov) from beach hoppers (Talitridae; Amphipoda) from the Pacific coast of North America. Fundamental and Applied Nematology 19, 579–584. [Google Scholar]
- * Adin, R. & Riera, P. (2003). Preferential food source utilization among stranded macroalgae by Talitrus saltator (Amphipod, Talitridae): a stable isotopes study in the northern coast of Brittany (France). Estuarine, Coastal and Shelf Science 56, 91–98. [Google Scholar]
- Airoldi, L. , Abbiati, M. , Beck, M. W. , Hawkins, S. J. , Jonsson, P. R. , Martin, D. , Moschella, P. S. , Sundelöf, A. , Thompson, R. C. & Åberg, P. (2005). An ecological perspective on the deployment and design of low‐crested and other hard coastal defence structures. Coastal Engineering 52, 1073–1087. [Google Scholar]
- * Alfaro, A. C. , Jeffs, A. G. & Creese, R. G. (2004). Bottom‐drifting algal/mussel spat associations along a sandy coastal region in northern New Zealand. Aquaculture 241, 269–290. [Google Scholar]
- * Alkemade, R. & Van Rijswijk, P. (1993). Path analyses of the influence of substrate composition on nematode numbers and on decomposition of stranded seaweed at an Antarctic coast. Netherlands Journal of Sea Research 31, 63–70. [Google Scholar]
- Alkemade, R. , Wielemaker, A. & Hemminga, M. A. (1992). Stimulation of decomposition of Spartina anglica leaves by the bacterivorous marine nematode Diplolaimelloides bruciei (Monhysteridae). Journal of Experimental Marine Biology and Ecology 159, 267–278. [Google Scholar]
- * Anderson, R. J. , Levitt, G. J. , Keats, D. W. & Simons, R. H. (1993). The role of herbivores in the collapse of the Gracilaria resource at Saldanha Bay, South Africa. Hydrobiologia 260–261, 285–290. [Google Scholar]
- * Anderson, W. B. & Polis, G. A. (1998). Marine subsidies of Island communities in the Gulf of California: evidence from stable carbon and nitrogen isotopes. Oikos 81, 75–80. [Google Scholar]
- * Andrades, R. , Gomes, M. P. , Pereira‐Filho, G. H. , Souza‐Filho, J. F. , Albuquerque, C. Q. & Martins, A. S. (2014). The influence of allochthonous macroalgae on the fish communities of tropical sandy beaches. Estuarine, Coastal and Shelf Science 144, 75–81. [Google Scholar]
- Arias‐Ortiz, A. , Serrano, O. , Masqué, P. , Lavery, P. S. , Mueller, U. , Kendrick, G. A. , Rozaimi, M. , Esteban, A. , Fourqurean, J. W. , Marbà, N. , Mateo, M. A. , Murray, K. , Rule, M. J. & Duarte, C. M. (2018). A marine heatwave drives massive losses from the world's largest seagrass carbon stocks. Nature Climate Change 1–7, 338–344. [Google Scholar]
- Avery, G. , Avery, D. M. , Braine, S. & Loutit, R. (1987). Prey of coastal black‐backed jackal Canis mesomelas (Mammalia: Canidae) in the Skeleton Coast Park, Namibia. Journal of Zoology 213, 81–94. [Google Scholar]
- * Ávila, E. , Carballo, J. L. , Vega, C. , Camacho, L. , Barrón‐Álvarez, J. J. , Padilla‐Verdín, C. & Yáñez‐Chávez, B. (2011). Deposition of shallow water sponges in response to seasonal changes. Journal of Sea Research 66, 172–180. [Google Scholar]
- * Backlund, H. O. (1945). Wrack Fauna of Sweden and Finland: Ecology and Chorology. Opuscula Entomologica. Supplementum. Lund, Sweden: Entomology Society of Lund. [Google Scholar]
- * Baldanzi, S. , McQuaid, C. D. , Cannicci, S. & Porri, F. (2013). Environmental domains and range‐limiting mechanisms: testing the abundant Centre hypothesis using southern African sandhoppers. PLoS One 8, 1–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- * Balestri, E. , Vallerini, F. & Lardicci, C. (2006). A qualitative and quantitative assessment of the reproductive litter from Posidonia oceanica accumulated on a sand beach following a storm. Estuarine, Coastal and Shelf Science 66, 30–34. [Google Scholar]
- * Balestri, E. , Vallerini, F. & Lardicci, C. (2011). Storm‐generated fragments of the seagrass Posidonia oceanica from beach wrack—a potential source of transplants for restoration. Biological Conservation 144, 1644–1654. [Google Scholar]
- Barbier, E. B. , Hacker, S. D. , Kennedy, C. , Koch, E. W. , Stier, A. C. & Silliman, B. R. (2011). The value of estuarine and coastal ecosystem services. Ecological Monographs 81(2), 169–193. [Google Scholar]
- * Baring, R. J. , Fairweather, P. G. & Lester, R. E. (2014). Storm versus calm: variation in fauna associated with drifting macrophytes in sandy beach surf zones. Journal of Experimental Marine Biology and Ecology 461, 397–406. [Google Scholar]
- * Baring, R. J. , Lester, R. E. & Fairweather, P. G. (2016). Establishing precise estimates of abundance in patchy habitats of the marine nearshore. Marine Environmental Research 120, 68–77. [DOI] [PubMed] [Google Scholar]
- * Baring, R. J. , Fairweather, P. G. & Lester, R. E. (2018. a). Nearshore drift dynamics of natural versus artificial seagrass wrack. Estuarine, Coastal and Shelf Science 202, 164–171. [Google Scholar]
- * Baring, R. J. , Lester, R. E. & Fairweather, P. G. (2018. b). Trophic relationships among animals associated with drifting wrack. Marine and Freshwater Research 69, 1248–1258. [Google Scholar]
- * Baring, R. J. , Lester, R. E. & Fairweather, P. G. (2019). Short‐term accumulation of fauna colonising natural versus artificial seagrass floating near to shore. Marine Biology 166, 1–11. [Google Scholar]
- Barnard, P. L. , Hoover, D. , Hubbard, D. M. , Snyder, A. , Ludka, B. C. , Allan, J. , Kaminsky, G. M. , Ruggiero, P. , Gallien, T. W. , Gabel, L. , McCandless, D. , Weiner, H. M. , Cohn, N. , Anderson, D. L. & Serafin, K. A. (2017). Extreme oceanographic forcing and coastal response due to the 2015–2016 El Niño. Nature Communications 8, 6–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- * Barreiro, F. , Gómez, M. , Lastra, M. , López, J. & de La Huz, R. (2011). Annual cycle of wrack supply to sandy beaches: effect of the physical environment. Marine Ecology Progress Series 433, 65–74. [Google Scholar]
- * Barreiro, F. , Gómez, M. , López, J. , Lastra, M. & de la Huz, R. (2013). Coupling between macroalgal inputs and nutrients outcrop in exposed sandy beaches. Hydrobiologia 700, 73–84. [Google Scholar]
- * Barrett, K. , Anderson, W. B. , Wait, D. A. , Grismer, L. L. , Polis, G. A. & Rose, M. D. (2005). Marine subsidies alter the diet and abundance of insular and coastal lizard populations. Oikos 109, 145–153. [Google Scholar]
- * Becherucci, M. E. & Benavides, H. (2016). Floristic compositions of large arrivals of drift marine algae on beaches of southeastern Buenos Aires province, Argentina [Composición florística de arribazones de algas marinas en playas del sudeste de la provincia de Buenos Aires, Argentina]. Iheringia—Serie Botanica 71, 22–26. [Google Scholar]
- * Behbehani, M. I. & Croker, R. A. (1982). Ecology of beach wrack in northern New England with special reference to Orchestia platensis . Estuarine, Coastal and Shelf Science 15, 611–620. [Google Scholar]
- Behrendorff, L. , Belonje, G. & Allen, B. L. (2018. a). Intraspecific killing behaviour of canids: how dingoes kill dingoes. Ethology Ecology and Evolution 30, 88–98. [Google Scholar]
- Behrendorff, L. , Leung, L. K.‐P. & Allen, B. L. (2018. b). Utilisation of stranded marine fauna washed ashore on K'gari (Fraser Island), Australia, by dingoes. Australian Journal of Zoology 66, 128–138. [Google Scholar]
- Behrendorff, L. , Leung, L. K.‐P. , McKinnon, A. , Hanger, J. , Belonje, G. , Tapply, J. , Jones, D. & Allen, B. L. (2016). Insects for breakfast and whales for dinner: the diet and body condition of dingoes on Fraser Island (K'gari). Scientific Reports 6, 23469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bell, T. W. , Reed, D. C. , Nelson, N. B. & Siegel, D. A. (2018). Regional patterns of physiological condition determine giant kelp net primary production dynamics. Limnology and Oceanography 63, 472–483. [Google Scholar]
- * Beltran, R. , Beca‐Carretero, P. , Marbà, N. , Jiménez, M. A. & Traveset, A. (2020). Spatio‐temporal variation in macrofauna community structure in Mediterranean seagrass wrack. Food Webs 25, e00178. [Google Scholar]
- * Benítez, S. , Duarte, C. , López, J. , Manríquez, P. H. , Navarro, J. M. , Bonta, C. C. , Torres, R. & Quijón, P. A. (2016). Ontogenetic variability in the feeding behavior of a marine amphipod in response to ocean acidification. Marine Pollution Bulletin 112, 375–379. [DOI] [PubMed] [Google Scholar]
- * Bessa, F. , Baeta, A. & Marques, J. C. (2014). Niche segregation amongst sympatric species at exposed sandy shores with contrasting wrack availabilities illustrated by stable isotopic analysis. Ecological Indicators 36, 694–702. [Google Scholar]
- Berdan, E. , Roger, F. , Kinnby, A. , Cervin, G. , Pereyra, R. , Öpel, M. , Wellenreuther, M. , Johannesson, K. , Utlin, R.K. & Andre, C. (2021). A metagenomic analysis of the wrackbed microbiome indicates a phylogeographic break along the North Sea ‐ Baltic Sea transition zone. bioRxiv 2021.11.01.466799. [DOI] [PubMed]
- * Bingham, E. L. , Gilby, B. L. , Olds, A. D. , Weston, M. A. , Connolly, R. M. , Henderson, C. J. , Maslo, B. , Peterson, C. F. , Voss, C. M. & Schlacher, T. A. (2018). Functional plasticity in vertebrate scavenger assemblages in the presence of introduced competitors. Oecologia 188, 583–593. [DOI] [PubMed] [Google Scholar]
- * Blanche, K. R. (1992). Preliminary observations on the distribution and abundance of seaweed flies (Diptera: Coelopidae) on beaches in the Gosford district of New South Wales, Australia. Australian Journal of Ecology 17, 27–34. [Google Scholar]
- * Blandford, M. I. , Katouli, M. , Gilby, B. L. , O'Dea, C. , Olds, A. D. & Schlacher, T. A. (2019). Not all rotten fish stink: microbial changes in decaying carcasses increase cytotoxicity and potential risks to animal scavengers. Estuarine, Coastal and Shelf Science 227, 106350. [Google Scholar]
- * Blomquist, H. L. & Pyron, J. H. (1943). Drifting ‘seaweed’ at Beaufort, North Carolina. American Journal of Botany 30, 28–32. [Google Scholar]
- * Blyth, J. E. & Gilburn, A. S. (2006). Extreme promiscuity in a mating system dominated by sexual conflict. Journal of Insect Behavior 19, 447–455. [Google Scholar]
- * Bodkin, J. L. , Ballachey, B. E. , Dean, T. A. , Fukuyama, A. K. , Jewett, S. C. , McDonald, L. , Monson, D. H. , O'Clair, C. E. & VanBlaricom, G. R. (2002). Sea otter population status and the process of recovery from the 1989 ‘Exxon Valdez’ oil spill. Marine Ecology Progress Series 241, 237–253. [Google Scholar]
- * Bodkin, J. L. , Burdin, A. M. & Ryazanov, D. A. (2000). Age‐ and sex‐specific mortality and population structure in sea otters. Marine Mammal Science 16, 201–219. [Google Scholar]
- * Bodkin, J. L. & Jameson, R. J. (1991). Patterns of seabird and marine mammal carcass deposition along the Central California coast, 1980–1986. Canadian Journal of Zoology 69, 1149–1155. [Google Scholar]
- Boevers, J. (2008). Assessing the utility of beach ecolabels for use by local management. Coastal Management 36, 524–531. [Google Scholar]
- * Bonnet, D. D. (1946). The Portuguese man‐of‐war as a food source for the sand crab (Emerita pacifica). Science 103, 148–149. [DOI] [PubMed] [Google Scholar]
- * Borzone, C. A. & Rosa, L. C. (2009). Impact of oil spill and posterior clean‐up activities on wrack‐living talitrid amphipods on estuarine beaches. Brazilian Journal of Oceanography 57, 315–323. [Google Scholar]
- Bowen, R. E. , Frankic, A. & Davis, M. E. (2006). Human development and resource use in the coastal zone: influences in human health. Oceanography 19, 62–71. [Google Scholar]
- * Boyne, A. W. , Amirault‐Langlais, D. L. & McCue, A. J. (2014). Characteristics of piping plover nesting habitat in the Canadian maritime provinces. Northeastern Naturalist 21, 164–173. [Google Scholar]
- Bridgeford, P. A. (1985). Unusual diet of the lion Panthera leo in the Skeleton Coast Park. Madoqua 1985, 187–188. [Google Scholar]
- Britton, J. C. & Morton, B. (1994). Marine carrion and scavengers. Oceanography and Marine Biology: An Annual Review 32, 369–434. [Google Scholar]
- Brown, A. C. (1961). Physiological‐ecological studies on two sandy‐beach gastropoda from South Africa: Bullia digitalis; meuschen and Bullia laevissima (gmelin). Zoomorphology 49, 629–657. [Google Scholar]
- Brown, A. C. (1971). The ecology of the sandy beaches of the cape peninsula, South Africa. Part 1: introduction. Transactions of the Royal Society of South Africa 39, 247–279. [Google Scholar]
- Brown, A. C. & McLachlan, A. (1990). The Ecology of Sandy Shores. Amsterdam: Elsevier Academic Press. [Google Scholar]
- * Brown, M. B. , Schlacher, T. A. , Schoeman, D. S. , Weston, M. A. , Huijbers, C. M. , Olds, A. D. & Connolly, R. M. (2015). Invasive carnivores alter ecological function and enhance complementarity in scavenger assemblages on ocean beaches. Ecology 96, 2715–2725. [DOI] [PubMed] [Google Scholar]
- Brunet, M. , de Bettignies, F. , Le Duff, N. , Tanguy, G. , Davoult, D. , Leblanc, C. , Gobet, A. & Thomas, F. (2021). Accumulation of detached kelp biomass in a subtidal temperate coastal ecosystem induces succession of epiphytic and sediment bacterial communities. Environmental Microbiology 23, 1638–1655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- * Brunetti, N. & Cuomo, C. (2014). Distribution of the invasive tunicate Styela clava in Long Island sound, New England, USA. BioInvasions Records 3, 13–19. [Google Scholar]
- * Bruno, D. O. , Riccialdelli, L. , Botto, F. & Acha, E. M. (2017). Organic matter sources for fish larvae and juveniles in a marine‐estuarine interface (mar Chiquita lagoon, Argentina). Environmental Biology of Fishes 100, 1609–1622. [Google Scholar]
- * Bucholc, K. , Szymczak‐Zyła, M. , Lubecki, L. , Zamojska, A. , Hapter, P. , Tjernström, E. & Kowalewska, G. (2014). Nutrient content in macrophyta collected from southern Baltic Sea beaches in relation to eutrophication and biogas production. Science of the Total Environment 473–474, 298–307. [DOI] [PubMed] [Google Scholar]
- * Bussolini, L. T. & Waters, J. M. (2015). Genetic analyses of rafted macroalgae reveal regional oceanographic connectivity patterns. Journal of Biogeography 42, 1319–1326. [Google Scholar]
- * Butlin, R. K. , Read, I. L. & Day, T. H. (1982). The effects of a chromosomal inversion on adult size and male mating success in the seaweed fly, Coelopa frigida . Heredity 49, 51–62. [Google Scholar]
- Cacabelos, E. , Olabarria, C. , Incera, M. & Troncoso, J. S. (2010). Do grazers prefer invasive seaweeds? Journal of Experimental Marine Biology and Ecology 393, 182–187. [Google Scholar]
- * Campbell, J. (2018). The importance of beach‐wrack for migratory shorebirds. Stilt 2018, 55. [Google Scholar]
- * Cardona, L. & García, M. (2008). Beach‐cast seagrass material fertilizes the foredune vegetation of Mediterranean coastal dunes. Acta Oecologica 34, 97–103. [Google Scholar]
- Carlton, J. T. & Hodder, J. (2003). Maritime mammals: terrestrial mammals as consumers in marine intertidal communities. Marine Ecology Progress Series 256, 271–286. [Google Scholar]
- * Catenazzi, A. & Donnelly, M. A. (2007. a). The Ulva connection: marine algae subsidize terrestrial predators in coastal Peru. Oikos 116, 75–86. [Google Scholar]
- * Catenazzi, A. & Donnelly, M. A. (2007. b). Role of supratidal invertebrates in the decomposition of beach‐cast green algae Ulva sp. Marine Ecology Progress Series 349, 33–42. [Google Scholar]
- Cavanaugh, K. , Siegel, D. , Reed, D. & Dennison, P. (2011). Environmental controls of giant‐kelp biomass in the Santa Barbara Channel, California. Marine Ecology Progress Series 429, 1–17. [Google Scholar]
- Cavanaugh, K. C. , Reed, D. C. , Bell, T. W. , Castorani, M. C. N. & Beas‐Luna, R. (2019). Spatial variability in the resistance and resilience of giant kelp in southern and Baja California to a multiyear heatwave. Frontiers in Marine Science 6, 413. [Google Scholar]
- Charlier, R. H. , Chaineux, M. C. P. & Morcos, S. (2005). Panorama of the history of coastal protection. Journal of Coastal Research 21(1), 79–111. [Google Scholar]
- * Chávez, V. , Uribe‐Martínez, A. , Cuevas, E. , Rodríguez‐Martínez, R. E. , van Tussenbroek, B. I. , Francisco, V. , Estévez, M. , Celis, L. B. , Monroy‐Velázquez, L. V. , Leal‐Bautista, R. , Álvarez‐Filip, L. , García‐Sánchez, M. , Masia, L. & Silva, R. (2020). Massive influx of pelagic Sargassum spp. on the coasts of the Mexican Caribbean 2014–2020: challenges and opportunities. Water 12(10), 2908. [Google Scholar]
- Choney, G. E. , McMahon, K. , Lavery, P. S. & Collier, N. (2014). Swan grazing on seagrass: abundance but not grazing pressure varies over an annual cycle in a temperate estuary. Marine and Freshwater Research 65, 738–749. [Google Scholar]
- * Chown, S. L. (1996). Kelp degradation by Paractora trichosterna (Thomson) (Diptera: Helcomyzidae) at sub‐Antarctic South Georgia. Polar Biology 16, 171–178. [Google Scholar]
- * Chubarenko, B. , Woelfel, J. , Hofmann, J. , Aldag, S. , Beldowski, J. , Burlakovs, J. , Garrels, T. , Gorbunova, J. , Guizani, S. , Kupczyk, A. , Kotwicki, L. , Domnin, D. , Gajewska, M. , Hogland, W. , Kołecka, K. , et al. (2021). Converting beach wrack into a resource as a challenge for the Baltic Sea (an overview). Ocean & Coastal Management 200, 105413. [Google Scholar]
- * Clark, B. M. , Bennett, B. A. & Lamberth, S. J. (1996. a). Factors affecting spatial variability in seine net catches of fish in the surf zone of False Bay, South Africa. Marine Ecology Progress Series 131, 17–34. [Google Scholar]
- * Clark, B. M. , Bennett, B. A. & Lamberth, S. J. (1996. b). Temporal variations in surf zone fish assemblages from False Bay, South Africa. Marine Ecology Progress Series 131, 35–47. [Google Scholar]
- * Clark, C. D. , De Bruyn, W. J. & Aiona, P. D. (2016). Temporal variation in optical properties of chromophoric dissolved organic matter (CDOM) in Southern California coastal waters with nearshore kelp and seagrass. Limnology and Oceanography 61, 32–46. [Google Scholar]
- Cockcroft, A. C. & McLachlan, A. (1993). Nitrogen budget for a high‐energy ecosystem. Marine Ecology Progress Series 100, 287–299. [Google Scholar]
- * Cocozza, C. , Parente, A. , Zaccone, C. , Mininni, C. , Santamaria, P. & Miano, T. (2011). Chemical, physical and spectroscopic characterization of Posidonia oceanica (L.) Del. residues and their possible recycle. Biomass and Bioenergy 35, 799–807. [Google Scholar]
- Cole, F. R. (1969). The Flies of Western North America, pp. xiv + 694. Berkeley: University of California Press. [Google Scholar]
- * Collins, C. J. , Fraser, C. I. , Ashcroft, A. & Waters, J. M. (2010). Asymmetric dispersal of southern bull‐kelp (Durvillaea Antarctica) adults in coastal New Zealand: testing an oceanographic hypothesis. Molecular Ecology 19, 4572–4580. [DOI] [PubMed] [Google Scholar]
- Colombelli‐Négrel, D. (2019). Benefits, costs and trade‐offs of nesting habitat selection in little penguins. Journal of Ornithology 160, 515–527. [Google Scholar]
- * Colombini, I. , Aloia, A. , Fallaci, M. , Pezzoli, G. & Chelazzi, L. (2000). Temporal and spatial use of stranded wrack by the macrofauna of a tropical sandy beach. Marine Biology 136, 531–541. [Google Scholar]
- * Colombini, I. , Brilli, M. , Fallaci, M. , Gagnarli, E. & Chelazzi, L. (2011). Food webs of a sandy beach macroinvertebrate community using stable isotopes analysis. Acta Oecologica 37, 422–432. [Google Scholar]
- Colombini, I. & Chelazzi, L. (2003). Influence of marine allochthonous input on sandy beach communities. In Oceanography and Marine Biology (Volume 41, eds Gibson R. N. and Atkinson R. J. A.), pp. 115–159. London: CRC Press. [Google Scholar]
- * Colombini, I. , Mateo, M. A. , Serrano, O. , Fallaci, M. , Gagnarli, E. , Serrano, L. & Chelazzi, L. (2009). On the role of Posidonia oceanica beach wrack for macroinvertebrates of a Tyrrhenian sandy shore. Acta Oecologica 35, 32–44. [Google Scholar]
- Conradt, L. (2000). Use of a seaweed habitat by red deer (Cervus elaphus L.). Journal of Zoology 250, 541–549. [Google Scholar]
- * Coupland, G. T. , Duarte, C. M. & Walker, D. I. (2007). High metabolic rates in beach cast communities. Ecosystems 10, 1341–1350. [Google Scholar]
- * Coupland, G. T. & McDonald, J. I. (2008). Extraordinarily high earthworm abundance in deposits of marine macrodetritus along two semi‐arid beaches. Marine Ecology Progress Series 361, 181–189. [Google Scholar]
- * Cowles, A. , Hewitt, J. E. & Taylor, R. B. (2009). Density, biomass and productivity of small mobile invertebrates in a wide range of coastal habitats. Marine Ecology Progress Series 384, 175–185. [Google Scholar]
- * Crafford, J. E. & Scholtz, C. H. (1987). Phenology of stranded kelp degradation by the kelp fly Paractora dreuxi mirabilis (Helcomyzidae) at Marion Island. Polar Biology 7, 289–294. [Google Scholar]
- Craig, P. (1970). The behavior and distribution of the intertidal sand beetle, Thinopinus pictus (Coleoptera: Staphylinidae). Ecology 51(6), 1012–1017. [Google Scholar]
- * Crawley, K. R. & Hyndes, G. A. (2007). The role of different types of detached macrophytes in the food and habitat choice of a surf‐zone inhabiting amphipod. Marine Biology 151, 1433–1443. [Google Scholar]
- * Crawley, K. R. , Hyndes, G. A. & Ayvazian, S. G. (2006). Influence of different volumes and types of detached macrophytes on fish community structure in surf zones of sandy beaches. Marine Ecology Progress Series 307, 233–246. [Google Scholar]
- * Crawley, K. R. , Hyndes, G. A. , Vanderklift, M. A. , Revill, A. T. & Nichols, P. D. (2009). Allochthonous brown algae are the primary food source for consumers in a temperate, coastal environment. Marine Ecology Progress Series 376, 33–44. [Google Scholar]
- Cruz‐Rivera, E. & Hay, M. E. (2000). Can quantity replace quality? Food choice, compensatory feeding, and fitness of marine mesograzers. Ecology 81, 201–219. [Google Scholar]
- Cruz‐Rivera, E. & Hay, M. E. (2003). Prey nutritional quality interacts with chemical defenses to affect consumer feeding and fitness. Ecological Monographs 73, 483–506. [Google Scholar]
- * Cucco, A. , Quattrocchi, G. , Brambilla, W. , Navone, A. , Panzalis, P. & Simeone, S. (2020). The management of the beach‐cast seagrass wracks—a numerical modelling approach. Journal of Marine Science and Engineering 8, 1–27. [Google Scholar]
- * Cullen, S. J. , Young, A. M. & Day, T. H. (1987). Dietary requirements of seaweed flies (Coelopa frigida). Estuarine, Coastal and Shelf Science 24, 701–710. [Google Scholar]
- Cypher, B. L. , Madrid, A. Y. , Van Horn Job, C. L. , Kelly, E. C. , Harrison, S. W. R. & Westall, T. L. (2014). Multi‐population comparison of resource exploitation by island foxes: implications for conservation. Global Ecology & Conservation 2, 255–266. [Google Scholar]
- Davenport, J. & Davenport, J. L. (2006). The impact of tourism and personal leisure transport on coastal environments: a review. Estuarine, Coastal and Shelf Science 67, 280–292. [Google Scholar]
- Davidson, K. H. , Starzomski, B. M. , El‐Sabaawi, R. , Hocking, M. D. , Reynolds, J. D. , Wickham, S. B. & Darimont, C. T. (2021). Marine subsidy promotes spatial and dietary niche variation in an omnivore, the Keen's mouse (Peromyscus keeni). Ecology and Evolution 11, 17700–17722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- * Davis, T. J. & Keppel, G. (2021). Fine‐scale environmental heterogeneity and conservation management: beach‐cast wrack creates microhabitats for thermoregulation in shorebirds. Journal of Applied Ecology 58, 1291–1301. [Google Scholar]
- * Day, T. H. , Dawe, C. , Dobson, T. & Hillier, P. C. (1983). A chromosomal inversion polymorphism in Scandinavian populations of the seaweed fly, Coelopa frigida . Hereditas 99, 135–145. [DOI] [PubMed] [Google Scholar]
- * Day, T. H. , Dobson, T. , Hillier, P. C. , Parkin, D. T. & Clarke, B. (1980). Different rates of development associated with the alcohol dehydrogenase locus in the seaweed fly, Coelopa frigida . Heredity 44, 321–326. [Google Scholar]
- * Day, T. H. , Dobson, T. , Hillier, P. C. , Parkin, D. T. & Clarke, B. (1982). Associations of enzymic and chromosomal polymorphisms in the seaweed fly, Coelopa frigida. Heredity 48, 35–44. [DOI] [PubMed] [Google Scholar]
- * Day, T. H. , Miles, S. , Pilkington, M. D. & Butlin, R. K. (1987). Differential mating success in populations of seaweed flies (Coelopa frigida). Heredity 58, 203–212. [Google Scholar]
- De Bettignies, T. , Wernberg, T. , Lavery, P. S. , Vanderklift, M. A. & Mohring, M. B. (2013). Contrasting mechanisms of dislodgement and erosion contribute to production of kelp detritus. Limnology and Oceanography 58, 1680–1688. [Google Scholar]
- de Carvalho, C. C. C. R. & Caramujo, M. J. (2012). Lipids of prokaryotic origin at the base of marine food webs. Marine Drugs 10, 2698–2714. [DOI] [PMC free article] [PubMed] [Google Scholar]
- * de Falco, G. , Simeone, S. & Baroli, M. (2008). Management of beach‐cast Posidonia oceanica seagrass on the Island of Sardinia (Italy, Western Mediterranean). Journal of Coastal Research 24, 69–75. [Google Scholar]
- de Mesel, I. , Derycke, S. , Swings, J. , Vincx, M. & Moens, T. (2003). Influence of bacterivorous nematodes on the decomposition of cordgrass. Journal of Experimental Marine Biology and Ecology 296, 227–242. [Google Scholar]
- de Schipper, M. A. , Ludka, B. C. , Raubenheimer, B. , Luijendijk, A. P. & Schlacher, T. A. (2021). Beach nourishment has complex implications for the future of sandy shores. Nature Reviews Earth & Environment 2, 70–84. [Google Scholar]
- * Deidun, A. , Saliba, S. & Schembri, P. J. (2009). Considerations on the ecological role of wrack accumulations on sandy beaches in the Maltese islands and recommendations for their conservation management. Journal of Coastal Research SI 56, 410–414. [Google Scholar]
- * Deidun, A. , Saliba, S. & Schembri, P. J. (2011). Quantitative assessment and physical characterisation of Posidonia oceanica wrack beached along the Maltese coastline. Biologia Marina Mediterranea 18, 307–308. [Google Scholar]
- * del Vecchio, S. , Jucker, T. , Carboni, M. & Acosta, A. T. R. (2017). Linking plant communities on land and at sea: the effects of Posidonia oceanica wrack on the structure of dune vegetation. Estuarine, Coastal and Shelf Science 184, 30–36. [Google Scholar]
- * del Vecchio, S. , Marbà, N. , Acosta, A. , Vignolo, C. & Traveset, A. (2013). Effects of Posidonia oceanica beach‐cast on germination, growth and nutrient uptake of coastal dune plants. PLoS One 8, e70607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- * Delingat, J. (2000). Habitat utilization by northern wheatears (Oenanthe oenanthe) stopping over on an offshore Island during migration. Vogelwarte 40, 271–278. [Google Scholar]
- * Dethier, M. N. , Raymond, W. W. , McBride, A. N. , Toft, J. D. , Cordell, J. R. , Ogston, A. S. , Heerhartz, S. M. & Berry, H. D. (2016). Multiscale impacts of armoring on Salish Sea shorelines: evidence for cumulative and threshold effects. Estuarine, Coastal and Shelf Science 175, 106–117. [Google Scholar]
- * Dierschke, V. (1993). Food and feeding ecology of purple sandpipers Calidris maritima on rocky intertidal habitats (Helgoland, German bight). Netherlands Journal of Sea Research 31, 309–317. [Google Scholar]
- * Dierschke, V. (1996). Helgoland only once or forever: site fidelity of waders [Nur einmal oder immer: ortstreue helgoländer watvögel]. Vogelwarte 38, 211–216. [Google Scholar]
- * Dobson, T. (1974. a). Mechanisms controlling species composition in natural populations of the seaweed fly Coelopa . Journal of Natural History 8, 653–673. [Google Scholar]
- * Dobson, T. (1974. b). Studies on the biology of the kelp‐fly Coelopa in Great Britain. Journal of Natural History 8, 155–177. [Google Scholar]
- * Dodson, J. , Eliot, I. , Eliot, M. , Chagué‐Goff, C. & Goff, J. (2014). Wrack line signatures of high‐magnitude water‐level events on the northwest Australian coast. Marine Geology 355, 310–317. [Google Scholar]
- * Duarte, C. , Acuña, K. , Navarro, J. M. & Gómez, I. (2011). Intra‐plant differences in seaweed nutritional quality and chemical defenses: importance for the feeding behavior of the intertidal amphipod Orchestoidea tuberculata . Journal of Sea Research 66, 215–221. [Google Scholar]
- * Duarte, C. , Acuña, K. , Navarro, J. M. , Gómez, I. , Jaramillo, E. & Quijón, P. (2014). Variable feeding behavior in Orchestoidea tuberculata (Nicolet 1849): exploring the relative importance of macroalgal traits. Journal of Sea Research 87, 1–7. [Google Scholar]
- * Duarte, C. , Jaramillo, E. & Contreras, H. (2008). Stranded algal wracks on a sandy beach of south Central Chile: feeding and habitat preferences of juveniles and adults of Orchestoidea tuberculata (Nicolet), (Amphipoda, Talitridae) [Macroalgas varadas sobre la superficie de una playa arenosa del Sur de Chile: preferencias alimentarias y de h bitat de juveniles y adultos de Orchestoidea tuberculata (Nicolet), (Amphipoda, Talitridae)]. Revista Chilena de Historia Natural 81, 69–81. [Google Scholar]
- * Duarte, C. , Jaramillo, E. , Contreras, H. & Acuña, K. (2010. a). Cannibalism and food availability in the talitrid amphipod Orchestoidea tuberculata . Journal of Sea Research 64, 417–421. [Google Scholar]
- * Duarte, C. , López, J. , Benítez, S. , Manríquez, P. H. , Navarro, J. M. , Bonta, C. C. , Torres, R. & Quijón, P. (2016). Ocean acidification induces changes in algal palatability and herbivore feeding behavior and performance. Oecologia 180, 453–462. [DOI] [PubMed] [Google Scholar]
- * Duarte, C. , Navarro, J. M. , Acuña, K. & Gómez, I. (2010. b). Feeding preferences of the sandhopper Orchestoidea tuberculata: the importance of algal traits. Hydrobiologia 651, 291–303. [Google Scholar]
- * Duarte, C. , Quintanilla‐Ahumada, D. , Anguita, C. , Manríquez, P. H. , Widdicombe, S. , Pulgar, J. , Silva‐Rodríguez, E. A. , Miranda, C. , Manríquez, K. & Quijón, P. A. (2019). Artificial light pollution at night (ALAN) disrupts the distribution and circadian rhythm of a sandy beach isopod. Estuarine, Coastal and Shelf Science 248, 565–573. [DOI] [PubMed] [Google Scholar]
- * Due, A. D. & Polis, G. A. (1985). The biology of Vaejovis littoralis Williams, an intertidal scorpion from Baja California, Mexico. Journal of Zoology 207, 563–580. [Google Scholar]
- Duffy, J. E. & Hay, M. E. (1991). Food and shelter as determinants of food choice by an herbivorous marine amphipod. Ecology 72, 1286–1298. [Google Scholar]
- * Dufour, C. , Probert, P. K. & Savage, C. (2012). Macrofaunal colonisation of stranded Durvillaea Antarctica on a southern New Zealand exposed sandy beach. New Zealand Journal of Marine and Freshwater Research 46, 369–383. [Google Scholar]
- Dugan, J. E. , Airoldi, L. , Chapman, M. G. , Walker, S. J. & Schlacher, T. (2012). Estuarine and coastal structures: environmental effects, a focus on shore and nearshore structures. In Treatise on Estuarine and Coastal Science (Volume 8, eds Wolanski E. and McLusky D.), pp. 17–41. London: Academic Press. [Google Scholar]
- Dugan, J. , Defoe, O. , Jaramillo, E. , Jones, A. , Lastra, M. , Nel, R. , Peterson, C. H. , Scapini, F. , Schlacher, T. A. & Schoeman, D. (2010). Give beach ecosystems their day in the sun. Science 329, 1146. [DOI] [PubMed] [Google Scholar]
- Dugan, J. E. , Emery, K. A. , Alber, M. , Alexander, C. R. , Byers, J. E. , Gehman, A. M. , McLenaghan, N. & Sojka, S. E. (2018). Generalizing ecological effects of shoreline armoring across soft sediment environments. Estuaries and Coasts 41, 180–196. [Google Scholar]
- * Dugan, J. E. & Hubbard, D. M. (2006). Ecological responses to coastal armoring on exposed sandy beaches. Shore & Beach 74, 10–16. [Google Scholar]
- * Dugan, J. E. & Hubbard, D. M. (2010). Loss of coastal strand habitat in Southern California: the role of beach grooming. Estuaries and Coasts 33, 67–77. [Google Scholar]
- * Dugan, J. E. , Hubbard, D. M. , Martin, D. L. , Engle, J. M. , Richards, D. M. , Davis, G. E. , Lafferty, K. D. & Ambrose, R. F. (2000). Macrofauna communities of exposed Sandy beaches on the Southern California mainland and Channel Islands. Santa Barbara: Minerals Management Service. [Google Scholar]
- * Dugan, J. E. , Hubbard, D. M. , McCrary, M. D. & Pierson, M. O. (2003). The response of macrofauna communities and shorebirds to macrophyte wrack subsidies on exposed sandy beaches of southern California. Estuarine, Coastal and Shelf Science 58, 25–40. [Google Scholar]
- Dugan, J. E. , Hubbard, D. M. & Page, H. M. (2005). Ecological effects of grooming on exposed sandy beaches in Southern California. In California and the World Ocean ‐ Proceedings of the Conference, pp. 824–826.
- * Dugan, J. E. , Hubbard, D. M. , Page, H. M. & Schimel, J. P. (2011). Marine macrophyte wrack inputs and dissolved nutrients in beach sands. Estuaries and Coasts 34, 839–850. [Google Scholar]
- Dugan, J. E. , Hubbard, D. M. & Quigley, B. J. (2013). Beyond beach width: steps toward identifying and integrating ecological envelopes with geomorphic features and datums for sandy beach ecosystems. Geomorphology 199, 95–105. [Google Scholar]
- Dugan, J. E. , Hubbard, D. M. , Rodil, I. F. , Revell, D. L. & Schroeter, S. (2008). Ecological effects of coastal armoring on sandy beaches. Marine Ecology 29, 160–170. [Google Scholar]
- Duggins, D. O. & Eckman, J. E. (1997). Is kelp detritus a good food for suspension feeders? Effects of kelp species, age and secondary metabolites. Marine Biology 128, 489–495. [Google Scholar]
- Duggins, D. O. , Simenstad, C. A. & Estes, J. A. (1989). Magnification of secondary production by kelp detritus in coastal marine ecosystems. Science 245, 170–173. [DOI] [PubMed] [Google Scholar]
- * Duong, H. L. S. & Fairweather, P. G. (2011). Effects of sandy beach cusps on wrack accumulation, sediment characteristics and macrofaunal assemblages. Austral Ecology 36, 733–744. [Google Scholar]
- * Edward, D. A. , Newton, J. & Gilburn, A. S. (2008). Investigating dietary preferences in two competing dipterans, Coelopa frigida and Coelopa pilipes, using stable isotope ratios of carbon and nitrogen. Entomologia Experimentalis et Applicata 127, 169–175. [Google Scholar]
- * Eereveld, P. , Hübner, L. , Schaefer, G. & Zimmer, M. (2013). Herbivory on macro‐algae affects colonization of beach‐cast algal wrack by detritivores but not its decomposition. Oceanologia 55, 339–358. [Google Scholar]
- Egan, S. , Harder, T. , Burke, C. , Steinberg, P. , Kjelleberg, S. & Thomas, T. (2013). The seaweed holobiont: understanding seaweed‐bacteria interactions. FEMS Microbiology Reviews 37, 462–476. [DOI] [PubMed] [Google Scholar]
- Egglishaw, H. J. (1960). Studies on the family Coelopidae (Diptera). Transactions of the Royal Entomological Society of London 112, 109–140. [Google Scholar]
- * Elias, S. P. , Fraser, J. D. & Buckley, P. A. (2000). Piping plover brood foraging ecology on New York barrier islands. Journal of Wildlife Management 64, 346–354. [Google Scholar]
- Emery, K. A. , Dugan, J. E. , Bailey, R. A. & Miller, R. J. (2021). Species identity drives ecosystem function in a subsidy‐dependent coastal ecosystem. Oikos 196, 1195–1206. [DOI] [PubMed] [Google Scholar]
- Emery, K. A. , Kramer, V. R. , Schooler, N. K. , Michaud, K. M. , Madden, J. R. , Hubbard, D. M. , Miller, R. J. & Dugan, J. E. (2022). Habitat partitioning by mobile intertidal invertebrates of sandy beaches shifts with the tides. Ecosphere 13, e3920. [Google Scholar]
- * Escobar‐Lasso, S. , Gil‐Fernández, M. , Herrera, H. , Fonseca, L. G. , Carrillo‐Jiménez, E. , Sáenz, J. & Wong, G. (2016. a). Scavenging on sea turtle carcasses by multiple jaguars in northwestern Costa Rica. Therya 7, 231–239. [Google Scholar]
- * Escobar‐Lasso, S. , Gil‐Fernández, M. , Sáenz, J. , Carrillo‐Jiménez, E. , Wong, G. & Fonseca, L. G. (2016. b). Inter‐trophic food provisioning between sea and land: the jaguar (Panthera onca) as provider of sea turtle carcasses to terrestrial scavengers. International Journal of Conservation Science 7, 1081–1094. [Google Scholar]
- * Estes, J. A. , Hatfield, B. B. , Ralls, K. & Ames, J. (2003). Causes of mortality in California Sea otters during periods of population growth and decline. Marine Mammal Science 19, 198–216. [Google Scholar]
- Fanini, L. , Cantarino, C. M. & Scapini, F. (2005). Relationships between the dynamics of two Talitrus saltator populations and the impacts of activities linked to tourism. Oceanologia 47, 93–112. [Google Scholar]
- * Fanini, L. , Hughes, L. E. , Springthorpe, R. , Tosetto, L. & Lowry, J. K. (2016). Surface activity patterns of macrofauna on pocket, tidal beaches: insights into the role of wrack and artificial lighting. Regional Studies in Marine Science 7, 63–71. [Google Scholar]
- * Fanini, L. & Lowry, J. (2014). Coastal talitrids and connectivity between beaches: a behavioural test. Journal of Experimental Marine Biology and Ecology 457, 120–127. [Google Scholar]
- Filbee‐Dexter, K. & Wernberg, T. (2018). Rise of turfs: a new battle front of globally declining kelp forests. Bioscience 68, 64–76. [Google Scholar]
- * Fox, C. H. , El‐Sabaawi, R. , Paquet, P. C. & Reimchen, T. E. (2014). Pacific herring Clupea pallasii and wrack macrophytes subsidize semi‐terrestrial detritivores. Marine Ecology Progress Series 495, 49–64. [Google Scholar]
- * Franzén, D. , Infantes, E. & Gröndahl, F. (2019). Beach‐cast as biofertiliser in the Baltic Sea region‐potential limitations due to cadmium‐content. Ocean and Coastal Management 169, 20–26. [Google Scholar]
- Fukuda, R. , Ogawa, H. , Nagata, T. & Koike, I. (1998). Direct determination of carbon and nitrogen contents of natural bacterial assemblages in marine environments. Applied and Environmental Microbiology 64, 3352–3358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- * Garbary, D. J. , Fraser, S. , Ferguson, C. & Lauff, R. F. (2004). Use of eelgrass, Zostera marina, wrack by three species of ladybird beetles (Coleoptera: Coccinellidae) in Prince Edward Island. Canadian Field‐Naturalist 118, 225–228. [Google Scholar]
- * Garbary, D. J. , Miller, A. G. , Williams, J. & Seymour, N. R. (2014). Drastic decline of an extensive eelgrass bed in Nova Scotia due to the activity of the invasive green crab (Carcinus maenas). Marine Biology 161, 3–15. [Google Scholar]
- * Garden, C. J. , Craw, D. , Waters, J. M. & Smith, A. (2011). Rafting rocks reveal marine biological dispersal: a case study using clasts from beach‐cast macroalgal holdfasts. Estuarine, Coastal and Shelf Science 95, 388–394. [Google Scholar]
- * Garden, C. J. & Smith, A. M. (2011). The role of kelp in sediment transport: observations from Southeast New Zealand. Marine Geology 281, 35–42. [Google Scholar]
- * Garrido, J. , Olabarria, C. & Lastra, M. (2008). Colonization of wrack by beetles (insecta, coleoptera) on a sandy beach of the Atlantic coast. Vie et Milieu 58, 243–247. [Google Scholar]
- * Gheskiere, T. , Magda, V. , Greet, P. & Steven, D. (2006). Are strandline meiofaunal assemblages affected by a once‐only mechanical beach cleaning? Experimental findings. Marine Environmental Research 61, 245–264. [DOI] [PubMed] [Google Scholar]
- * Gilburn, A. S. (2012). Mechanical grooming and beach award status are associated with low strandline biodiversity in Scotland. Estuarine, Coastal and Shelf Science 107, 81–88. [Google Scholar]
- * Gilson, A. , Smale, D. , Burrows, M. & O'Connor, N. (2021). Spatio‐temporal variability in the deposition of beach‐cast kelp (wrack) and inter‐specific differences in degradation rates. Marine Ecology Progress Series 674, 89–102. [Google Scholar]
- * Gomes Veloso, V. , Azevedo Sallorenzo, I. , Andrade Barros, W. K. & de Souza, G. (2012). Analysis of feeding behavior and food consumption rate of Atlantorchestoidea brasiliensis (Crustacea: Talitridae) [Análisis del comportamiento alimentario y de la tasa de consumo de alimento de Atlantorchestoidea brasiliensis (Crustacea: Talitridae)]. Ciencias Marinas 38, 653–664. [Google Scholar]
- * Gómez, M. , Barreiro, F. , López, J. & Lastra, M. (2018). Effect of upper beach macrofauna on nutrient cycling of sandy beaches: metabolic rates during wrack decay. Marine Biology 165, 133. [Google Scholar]
- * Gómez, M. , Barreiro, F. , López, J. , Lastra, M. & de la Huz, R. (2013). Deposition patterns of algal wrack species on estuarine beaches. Aquatic Botany 105, 25–33. [Google Scholar]
- * Gómez‐Pujol, L. , Orfila, A. , Álvarez‐Ellacuría, A. , Terrados, J. & Tintoré, J. (2013). Posidonia oceanica beach‐cast litter in Mediterranean beaches: a coastal videomonitoring study. Journal of Coastal Research 65, 1768–1773. [Google Scholar]
- * Gonçalves, S. C. , Anastácio, P. M. , Pardal, M. A. , Cardoso, P. G. , Ferreira, S. M. & Marques, J. C. (2009). Sandy beach macrofaunal communities on the western coast of Portugal ‐ is there a steady structure under similar exposed conditions? Estuarine, Coastal and Shelf Science 81, 555–568. [Google Scholar]
- * Gonçalves, S. C. & Marques, J. C. (2011). The effects of season and wrack subsidy on the community functioning of exposed sandy beaches. Estuarine, Coastal and Shelf Science 95, 165–177. [Google Scholar]
- Goodridge, B. M. & Melack, J. M. (2014). Temporal evolution and variability of dissolved inorganic nitrogen in beach pore water revealed using radon residence times. Environmental Science and Technology 48, 14211–14218. [DOI] [PubMed] [Google Scholar]
- Gosper, D. J. (1983). Birds in the Richmond River district, NSW. Corella 26, 7–13. [Google Scholar]
- Green, E. P. & Short, F. T. (2003). World Atlas of Seagrasses, p. 298. University of California Press, Berkeley. [Google Scholar]
- * Greger, M. , Malm, T. & Kautsky, L. (2007). Heavy metal transfer from composted macroalgae to crops. European Journal of Agronomy 26, 257–265. [Google Scholar]
- * Griffin, C. , Day, N. , Rosenquist, H. , Wellenreuther, M. , Bunnefeld, N. & Gilburn, A. S. (2018). Tidal range and recovery from the impacts mechanical beach grooming. Ocean and Coastal Management 154, 66–71. [Google Scholar]
- Griffiths, C. L. & Griffiths, R. J. (1983). Biology and distribution of the littoral rove beetle Psamathobledius‐Punctatissimus (Le conte) (Coleoptera, Staphylinidae). Hydrobiologia 101, 203–214. [Google Scholar]
- * Griffiths, C. L. & Stenton‐Dozey, J. (1981). The fauna and rate of degradation of stranded kelp. Estuarine, Coastal and Shelf Science 12, 645–653. [Google Scholar]
- * Griffiths, C. L. , Stenton‐Dozey, J. M. E. & Koop, K. (1983). Kelp wrack and the flow of energy through a sandy beach ecosystem. In Sandy Beaches as Ecosystems (eds McLachlan A. and Erasmus T.), pp. 547–556. Springer Netherlands, Dordrecht. [Google Scholar]
- Griggs, G. & Patsch, K. (2018). Natural changes and human impacts on the sand budgets and beach widths of the Zuma and Santa Monica littoral cells, Southern California. Shore & Beach 86, 1–14. [Google Scholar]
- * Grilliot, M. J. , Walker, I. J. & Bauer, B. O. (2019). Aeolian sand transport and deposition patterns within a large woody debris matrix fronting a foredune. Geomorphology 338, 1–15. [Google Scholar]
- * Guala, I. , Simeone, S. , Buia, M. C. , Flagella, S. , Baroli, M. & de Falco, G. (2006). Posidonia oceanica ‘banquette’ removal: environmental impact and management considerations. Biologia Marina Mediterranea 13, 149–153. [Google Scholar]
- Guarino, F. (2001). Diet of a large carnivorous lizard, Varanus varius . Wildlife Research 28, 627–630. [Google Scholar]
- * Guerrero‐Meseguer, L. , Veiga, P. & Rubal, M. (2020). Spatio‐temporal variability of anthropogenic and natural wrack accumulations along the driftline: marine litter overcomes wrack in the northern sandy beaches of Portugal. Journal of Marine Science and Engineering 8(12), 996. [Google Scholar]
- Hamilton, S. L. , Bell, T. W. , Watson, J. R. , Grorud‐Colvert, K. A. & Menge, B. A. (2020). Remote sensing: generation of long‐term kelp bed data sets for evaluation of impacts of climatic variation. Ecology 101, e03031. [DOI] [PubMed] [Google Scholar]
- * Hammann, S. & Zimmer, M. (2014). Wind‐driven dynamics of beach‐cast wrack in a tide‐free system. Open Journal of Marine Science 04, 68–79. [Google Scholar]
- * Harris, L. R. , Harris, K. A. & Nel, R. (2019). Food preferences of two sandy beach scavengers with different foraging strategies. Estuarine, Coastal and Shelf Science 219, 120–127. [Google Scholar]
- * Hawes, N. A. , Taylor, D. I. & Schiel, D. R. (2017). Transport of drifting fucoid algae: nearshore transport and potential for long distance dispersal. Journal of Experimental Marine Biology and Ecology 490, 34–41. [Google Scholar]
- * Hawke, D. J. & Clark, J. M. (2010). Incorporation of the invasive mallow Lavatera arborea into the food web of an active seabird Island. Biological Invasions 12, 1805–1814. [Google Scholar]
- * Hayes, W. B. (1974). Sand‐beach energetics: importance of the isopod Tylos punctatus . Ecology 55, 838–847. [Google Scholar]
- Heck, K. L. Jr. , Carruthers, T. J. B. , Duarte, C. M. , Randall Hughes, A. , Kendrick, G. , Orth, R. J. & Williams, S. W. (2008). Trophic transfers from seagrass meadows subsidize diverse marine and terrestrial consumers. Ecosystems 11, 1198–1210. [Google Scholar]
- * Heerhartz, S. M. , Dethier, M. N. , Toft, J. D. , Cordell, J. R. & Ogston, A. S. (2014). Effects of shoreline armoring on beach wrack subsidies to the nearshore ecotone in an estuarine fjord. Estuaries and Coasts 37, 1256–1268. [Google Scholar]
- * Heerhartz, S. M. , Toft, J. D. , Cordell, J. R. , Dethier, M. N. & Ogston, A. S. (2016). Shoreline armoring in an estuary constrains wrack‐associated invertebrate communities. Estuaries and Coasts 39, 171–188. [Google Scholar]
- * Hemminga, M. A. & Nieuwenhuize, J. (1991). Transport, deposition and in situ decay of seagrasses in a tropical mudflat area (banc D'Arguin, Mauritania). Netherlands Journal of Sea Research 27, 183–190. [Google Scholar]
- * Herkül, K. , Kotta, J. & Kotta, I. (2006). Distribution and population characteristics of the alien talitrid amphipod Orchestia cavimana in relation to environmental conditions in the northeastern Baltic Sea. Helgoland Marine Research 60, 121–126. [Google Scholar]
- Hesp, P. (2002). Foredunes and blowouts: initiation, geomorphology and dynamics. Geomorphology 48, 245–268. [Google Scholar]
- * Hodge, S. & Arthur, W. (1997). Asymmetric interactions between species of seaweed fly. Journal of Animal Ecology 66(5), 743–754. [Google Scholar]
- * Hodge, S. & Early, J. W. (2016). Hymenoptera associated with marine strandlines at Christchurch and banks peninsula. New Zealand Entomologist 39, 117–128. [Google Scholar]
- * Hodge, S. , Marris, J. , Brown, S. D. J. & Emberson, R. (2019). Coleoptera found in marine strandlines on New Zealand beaches: species diversity, seasonal trends and the effect of beach substrate. New Zealand Entomologist 42, 47–66. [Google Scholar]
- * Hodge, S. & Vink, C. (2020). The prevalence and species richness of spiders associated with marine strandlines on different shore types around banks peninsula, New Zealand. New Zealand Journal of Zoology 47, 71–85. [Google Scholar]
- * Holden, J. J. , Dudas, S. E. & Juanes, F. (2016). Is commercial harvesting of beach‐cast seaweed ecologically sustainable? Integrated Environmental Assessment and Management 12, 825–827. [DOI] [PubMed] [Google Scholar]
- * Holden, J. J. , Kingzett, B. C. , MacNeill, S. , Smith, W. , Juanes, F. & Dudas, S. E. (2018. a). Beach‐cast biomass and commercial harvesting of a non‐indigenous seaweed, Mazzaella japonica, on the east coast of Vancouver Island, British Columbia. Journal of Applied Phycology 30, 1175–1184. [Google Scholar]
- * Holden, J. J. , MacNeill, S. K. , Juanes, F. & Dudas, S. E. (2018. b). Beach‐cast deposition and commercial harvesting of a non‐indigenous alga, Mazzaella japonica: implications for macrofauna communities in Baynes sound, British Columbia. Estuarine, Coastal and Shelf Science 210, 162–171. [Google Scholar]
- * Hotten, R. D. (1988). Sand mining on Mission Beach San Diego, California. Shore & Beach 56, 18–21. [Google Scholar]
- Howarth, R. W. & Marino, R. (2006). Nitrogen as the limiting nutrient for eutrophication in coastal marine ecosystems: evolving views over three decades. Limnology & Oceanography 51, 364–376. [Google Scholar]
- Hubbard, D. M. & Dugan, J. E. (2003). Shorebird use of an exposed sandy beach in southern California. Estuarine, Coastal and Shelf Science 58, 41–54. [Google Scholar]
- Hubbard, D. M. , Dugan, J. E. , Schooler, N. K. & Viola, S. M. (2014). Local extirpations and regional declines of endemic upper beach invertebrates in southern California. Estuarine, Coastal and Shelf Science 150, 67–75. [Google Scholar]
- * Huijbers, C. M. , Schlacher, T. A. , McVeigh, R. R. , Schoeman, D. S. , Olds, A. D. , Brown, M. B. , Ekanayake, K. B. , Weston, M. A. & Connolly, R. M. (2016). Functional replacement across species pools of vertebrate scavengers separated at a continental scale maintains an ecosystem function. Functional Ecology 30, 998–1005. [Google Scholar]
- * Huijbers, C. M. , Schlacher, T. A. , Schoeman, D. S. , Olds, A. D. , Weston, M. A. & Connolly, R. M. (2015). Limited functional redundancy in vertebrate scavenger guilds fails to compensate for the loss of raptors from urbanized sandy beaches. Diversity and Distributions 21, 55–63. [Google Scholar]
- * Huijbers, C. M. , Schlacher, T. A. , Schoeman, D. S. , Weston, M. A. & Connolly, R. M. (2013). Urbanisation alters processing of marine carrion on sandy beaches. Landscape and Urban Planning 119, 1–8. [Google Scholar]
- Hurd, C. L. , Harrison, P. J. , Bischof, K. & Lobban, C. S. (2014). Seaweed Ecology and Physiology, 2nd Edition (). Cambridge University Press, Cambridge. [Google Scholar]
- Hyndes, G. , Lavery, P. & Doropoulos, C. (2012). Dual processes for cross‐boundary subsidies: incorporation of nutrients from reef‐derived kelp into a seagrass ecosystem. Marine Ecology Progress Series 445, 97–107. [Google Scholar]
- Hyndes, G. A. , Heck, K. L. , Vergés, A. , Harvey, E. S. , Kendrick, G. A. , Lavery, P. S. , McMahon, K. , Orth, R. J. , Pearce, A. , Vanderklift, M. , Wernberg, T. , Whiting, S. & Wilson, S. (2016). Accelerating tropicalization and the transformation of temperate seagrass meadows. Bioscience 66, 938–945. [DOI] [PMC free article] [PubMed] [Google Scholar]
- * Hyndes, G. A. & Lavery, P. S. (2005). Does transported seagrass provide an important trophic link in unvegetated, nearshore areas? Estuarine, Coastal and Shelf Science 63, 633–643. [Google Scholar]
- Hyndes, G. A. , Nagelkerken, I. , Mcleod, R. J. , Connolly, R. M. , Lavery, P. S. & Vanderklift, M. A. (2014). Mechanisms and ecological role of carbon transfer within coastal seascapes. Biological Reviews 89, 232–254. [DOI] [PubMed] [Google Scholar]
- * Imamura, G. J. , Thompson, R. S. , Boehm, A. B. & Jay, J. A. (2011). Wrack promotes the persistence of fecal indicator bacteria in marine sands and seawater. FEMS Microbiology Ecology 77, 40–49. [DOI] [PubMed] [Google Scholar]
- * Ince, R. , Hyndes, G. A. , Lavery, P. S. & Vanderklift, M. A. (2007). Marine macrophytes directly enhance abundances of sandy beach fauna through provision of food and habitat. Estuarine, Coastal and Shelf Science 74, 77–86. [Google Scholar]
- * Inglis, G. (1989). The colonisation and degradation of stranded Macrocystis pyrifera (L.) C. ag. By the macrofauna of a New Zealand sandy beach. Journal of Experimental Marine Biology and Ecology 125, 203–217. [Google Scholar]
- * Innocenti, G. , Stasolla, G. , Mendelson, M. & Galil, B. S. (2017). Aggressive, omnivorous, invasive: the Erythraean moon crab Matuta victor (Fabricius, 1781) (Crustacea: Decapoda: Matutidae) in the eastern Mediterranean Sea. Journal of Natural History 51, 2133–2142. [Google Scholar]
- * Ishida, T. , Sawahata, T. , Kanaya, G. & Hayasaka, D. (2015). Population dynamics of two sympatric sandhoppers (Trinorchestia species) (Amphipoda, Talitridae) on the pacific coast of northern Tohoku after the 2011 Tohoku‐Oki tsunami. Crustaceana 88, 511–521. [Google Scholar]
- Irmler, U. (2012). Effects of habitat and human activities on species richness and assemblages of Staphylinidae (Coleoptera) in the Baltic Sea coast. Psyche 2012, 879715. [Google Scholar]
- * Jaramillo, E. , de La Huz, R. , Duarte, C. & Contreras, H. (2006). Algal wrack deposits and macroinfaunal arthropods on sandy beaches of the Chilean coast. Revista Chilena de Historia Natural 79, 337–351. [Google Scholar]
- Jaramillo, E. , Dugan, J. E. & Hubbard, D. M. (2012). Ecological implications of extreme events: footprints of the 2010 earthquake along the Chilean coast. PLoS One 7, e35348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jaramillo, E. , Dugan, J. E. , Hubbard, D. M. , Contreras, H. , Duarte, C. , Acuña, E. & Schoeman, D. S. (2017). Macroscale patterns in body size of intertidal crustaceans provide insights on climate change effects. PLoS One 12, e0177116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jaramillo, E. , Dugan, J. , Hubbard, D. , Manzano, M. & Duarte, C. (2021). Ranking the ecological effects of coastal armoring on mobile macroinvertebrates across intertidal zones on sandy beaches. Science of the Total Environment 755, 142573. [DOI] [PubMed] [Google Scholar]
- * Jedrzejczak, M. F. (1999). The degradation of stranded carrion on a Baltic Sea sandy beach. Oceanological Studies 28, 119–141. [Google Scholar]
- * Jȩdrzejczak, M. F. (2002. a). Stranded Zostera marina L. vs wrack fauna community interactions on a Baltic sandy beach (Hel, Poland): a short‐term pilot study. Part I. Driftline effects of fragmented detritivory, leaching and decay rates. Oceanologia 44, 273–286. [Google Scholar]
- * Jȩdrzejczak, M. F. (2002. b). Stranded Zostera marina L. vs wrack fauna community interactions on a Baltic sandy beach (Hel, Poland): a short‐term pilot study. Part II. Driftline effects of succession changes and colonisation of beach fauna. Oceanologia 44, 367–387. [Google Scholar]
- * Jedrzejczak, M. F. (2003). Predrying of stranded wrack material as an aspect of the litterbag techniques in the sandy beach studies. Oceanological and Hydrobiological Studies 32, 59–74. [Google Scholar]
- * Jiménez, M. A. , Beltran, R. , Traveset, A. , Calleja, M. L. , Delgado‐Huertas, A. & Marbà, N. (2017). Aeolian transport of seagrass (Posidonia oceanica) beach‐cast to terrestrial systems. Estuarine, Coastal and Shelf Science 196, 31–44. [Google Scholar]
- * Jiménez, R. S. , Hepburn, C. D. , Hyndes, G. A. , McLeod, R. J. & Hurd, C. L. (2015). Contributions of an annual invasive kelp to native algal assemblages: algal resource allocation and seasonal connectivity across ecotones. Phycologia 54, 530–544. [Google Scholar]
- Jordà, G. , Marbà, N. & Duarte, C. M. (2012). Mediterranean seagrass vulnerable to regional climate warming. Nature Climate Change 2, 821–824. [Google Scholar]
- * Josselyn, M. N. & Mathieson, A. C. (1980). Seasonal influx and decomposition of autochthonous macrophyte litter in a north temperate estuary. Hydrobiologia 71, 197–208. [Google Scholar]
- * Kaspersen, B. S. , Christensen, T. B. , Fredenslund, A. M. , Møller, H. B. , Butts, M. B. , Jensen, N. H. & Kjaer, T. (2016). Linking climate change mitigation and coastal eutrophication management through biogas technology: evidence from a new Danish bioenergy concept. Science of the Total Environment 541, 1124–1131. [DOI] [PubMed] [Google Scholar]
- * Kelly, J. F. (2016). Assessing the spatial compatibility of recreational activities with beach vegetation and wrack in New Jersey: prospects for compromise management. Ocean and Coastal Management 123, 9–17. [Google Scholar]
- * Kemp, P. F. (1986). Direct uptake of detrital carbon by the deposit‐feeding polychaete Euzonus mucronata (Treadwell). Journal of Experimental Marine Biology and Ecology 99, 49–61. [Google Scholar]
- Kensley, B. F. (1974). Aspects of the biology and ecology of the genus Tylos latreille . Annals of the South African Museum 65, 401–471. [Google Scholar]
- * Kersen, P. & Martin, G. (2007). Annual biomass loss of the loose‐lying red algal community via macroalgal beach casts in the Väinameri area, NE Baltic Sea. Proceedings of the Estonian Academy of Sciences: Biology, Ecology 56, 278–289. [Google Scholar]
- King, P. G. , Nelsen, C. , Dugan, J. E. , Hubbard, D. M. , Martin, K. L. & Battalio, R. T. (2018). Valuing beach ecosystems in an age of retreat. Shore & Beach 86(4), 45–59. [Google Scholar]
- * Kirkman, H. & Kendrick, G. A. (1997). Ecological significance and commercial harvesting of drifting and beach‐cast macro‐algae and seagrasses in Australia: a review. Journal of Applied Phycology 9, 311–326. [Google Scholar]
- Klein, L. & Dodds, R. (2018). Blue flag beach certification: an environmental management tool or tourism promotional tool? Tourism Recreation Research 43, 39–51. [Google Scholar]
- Kompfner, H. (1974). Larvae and pupae of some wrack dipterans on a California beach. Pan‐Pacific Entomologist 50, 44–52. [Google Scholar]
- * Koop, K. & Field, J. G. (1980). The influence of food availability on population dynamics of a supralitoral isopod Ligia dilatata Brandt. Journal of Experimental Marine Biology and Ecology 48, 61–72. [Google Scholar]
- * Koop, K. & Griffiths, C. L. (1982). The relative significance of bacteria, meio‐ and macrofauna on an exposed sandy beach. Marine Biology 66, 295–300. [Google Scholar]
- Koop, K. & Lucas, M. I. (1983). Carbon flow and nutrient regeneration from the decomposition of macrophyte debris in a sandy beach microcosm. In Sandy Beaches as Ecosystems (Volume 19, eds McLachlan A. and Erasmus T.), pp. 249–262. Springer, Dordrecht. [Google Scholar]
- * Koop, K. , Newell, R. & Lucas, M. (1982. a). Biodegradation and carbon flow based on kelp (Ecklonia maxima) debris in a sandy beach microcosm. Marine Ecology Progress Series 7, 315–326. [Google Scholar]
- * Koop, K. , Newell, R. C. & Lucas, M. I. (1982. b). Microbial regeneration of nutrients from the decomposition of macrophyte debris on the shore. Marine Ecology Progress Series 9, 91–96. [Google Scholar]
- * Kotwicki, L. , Węsławski, J. M. , Szałtynis, A. , Raczyńska, A. & Kupiec, A. (2005). Deposition of large organic particles (macrodetritus) in a sandy beach system (Puck Bay, Baltic Sea). Oceanologia 47, 181–199. [Google Scholar]
- * Krantz, G. W. (2016). A new species of Halolaelapidae (Acari: Mesostigmata: Rhodacaroidea) from beach wrack in Yaquina Bay, Oregon, USA, with comments on opisthonotal plasticity and cribral development in the family. Journal of Natural History 50, 1797–1812. [Google Scholar]
- Krumhansl, K. A. , Okamoto, D. K. , Rassweiler, A. , Novak, M. , Bolton, J. J. , Cavanaugh, K. C. , Connell, S. D. , Johnson, C. R. , Konar, B. , Ling, S. D. , Micheli, F. , Norderhaug, K. M. , Pérez‐Matus, A. , Sousa‐Pinto, I. , Reed, D. C. , et al. (2016). Global patterns of kelp forest change over the past half‐century. Proceedings of the National Academy of Sciences of the United States of America 113, 13785–13790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krumhansl, K. A. & Scheibling, R. E. (2011). Detrital production in Nova Scotian kelp beds: patterns and processes. Marine Ecology Progress Series 421, 67–82. [Google Scholar]
- Krumhansl, K. A. & Scheibling, R. E. (2012). Production and fate of kelp detritus. Marine Ecology Progress Series 467, 281–302. [Google Scholar]
- * Kuda, T. & Ikemori, T. (2009). Minerals, polysaccharides and antioxidant properties of aqueous solutions obtained from macroalgal beach‐casts in the Noto Peninsula, Ishikawa, Japan. Food Chemistry 112, 575–581. [Google Scholar]
- Kuhn, B. F. , Wiesel, I. & Skinner, J. D. (2008). Diet of brown hyaenas (Parahyaena brunnea) on the Namibian coast. Transactions of the Royal Society of South Africa 63, 150–159. [Google Scholar]
- * Lacerda, C. H. F. , Barletta, M. & Dantas, D. V. (2014). Temporal patterns in the intertidal faunal community at the mouth of a tropical estuary. Journal of Fish Biology 85, 1571–1602. [DOI] [PubMed] [Google Scholar]
- * Lafferty, K. D. , Rodriguez, D. A. & Chapman, A. (2013). Temporal and spatial variation in bird and human use of beaches in southern California. SpringerPlus 2, 1–14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- * Lastra, M. , López, J. & Neves, G. (2015). Algal decay, temperature and body size influencing trophic behaviour of wrack consumers in sandy beaches. Marine Biology 162, 221–233. [Google Scholar]
- * Lastra, M. , López, J. , Troncoso, J. S. , Hubbard, D. M. & Dugan, J. E. (2016). Scavenger and burrowing features of Hippa pacifica (Dana 1852) on a range of tropical sandy beaches. Marine Biology 163(10), 212. [Google Scholar]
- * Lastra, M. , López, J. & Rodil, I. F. (2018). Warming intensify CO2 flux and nutrient release from algal wrack subsidies on sandy beaches. Global Change Biology 24, 3766–3779. [DOI] [PubMed] [Google Scholar]
- * Lastra, M. , López, J. , Troncoso, J. S. & Sampedro, L. (2021). Warming and wrack supply will accelerate CO2 emission and nutrients release on Antarctic sedimentary shores: a case study on a volcanic Island. Ecosystems 24, 855–874. [Google Scholar]
- * Lastra, M. , Page, H. M. , Dugan, J. E. , Hubbard, D. M. & Rodil, I. F. (2008). Processing of allochthonous macrophyte subsidies by sandy beach consumers: estimates of feeding rates and impacts on food resources. Marine Biology 154, 163–174. [Google Scholar]
- * Lastra, M. , Rodil, I. F. , Sánchez‐Mata, A. , García‐Gallego, M. & Mora, J. (2014). Fate and processing of macroalgal wrack subsidies in beaches of Deception Island, Antarctic Peninsula. Journal of Sea Research 88, 1–10. [Google Scholar]
- * Lavery, P. , Bootle, S. & Vanderklift, M. (1999). Ecological effects of macroalgal harvesting on beaches in the Peel‐Harvey Estuary, Western Australia. Estuarine, Coastal and Shelf Science 49, 295–309. [Google Scholar]
- * Lavoie, D. R. (1985). Population dynamics and ecology of beach wrack macroinvertebrates of the Central California coast. Bulletin of Southern California Academy of Sciences 84, 1–22. [Google Scholar]
- * Lee, T. S. , Toft, J. D. , Cordell, J. R. , Dethier, M. N. , Adams, J. W. & Kelly, R. P. (2018). Quantifying the effectiveness of shoreline armoring removal on coastal biota of Puget Sound. PeerJ 2018, e4275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- * Leggett, M. C. , Wilcockson, R. W. , Day, T. H. , Phillips, D. S. & Arthur, W. (1996). The genetic effects of competition in seaweed flies. Biological Journal of the Linnean Society 57, 1–11. [Google Scholar]
- * Lenanton, R. C. J. & Caputi, N. (1989). The roles of food supply and shelter in the relationship between fishes, in particular Cnidoglanis macrocephalus (Valenciennes), and detached macrophytes in the surf zone of sandy beaches. Journal of Experimental Marine Biology and Ecology 128, 165–176. [Google Scholar]
- * Lenanton, R. C. J. , Robertson, A. I. & Hansen, J. A. (1982). Nearshore accumulations of detached macrophytes as nursery areas for fish. Marine Ecology Progress Series 9, 51–57. [Google Scholar]
- * Lewis, T. L. , Mews, M. , Jelinski, D. E. & Zimmer, M. (2007). Detrital subsidy to the supratidal zone provides feeding habitat for intertidal crabs. Estuaries and Coasts 30, 451–458. [Google Scholar]
- Li, F. , Roncevich, L. , Bicknell, C. , Lowry, R. & Ilich, K. (2011). Interannual variability and trends of storminess, Perth, 1994‐2008. Journal of Coastal Research 27, 738–745. [Google Scholar]
- * Liebowitz, D. M. , Nielsen, K. J. , Dugan, J. E. , Morgan, S. G. , Malone, D. P. , Largier, J. L. , Hubbard, D. M. & Carr, M. H. (2016). Ecosystem connectivity and trophic subsidies of sandy beaches. Ecosphere 7, e01503. [Google Scholar]
- Lillebø, A. I. , Flindt, M. R. , Pardal, M. Â. & Marques, J. C. (1999). The effect of macrofauna, meiofauna and microfauna on the degradation of Spartina maritima detritus from a salt marsh area. Acta Oecologica 20, 249–258. [Google Scholar]
- * Lillywhite, H. B. , Sheehy, C. M. III & Zaidan, F. III (2008). Pitviper scavenging at the intertidal zone: an evolutionary scenario for invasion of the sea. BioScience 58, 947–955. [Google Scholar]
- Ling, S. D. , Johnson, C. R. , Frusher, S. D. & Ridgway, K. R. (2009). Overfishing reduces resilience of kelp beds to climate‐driven catastrophic phase shift. Proceedings of the National Academy of Sciences of the United States of America 106, 22341–22345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ling, S. D. , Scheibling, R. E. , Rassweiler, A. , Johnson, C. R. , Shears, N. , Connell, S. D. , Salomon, A. K. , Norderhaug, K. M. , Pérez‐Matus, A. , Hernández, J. C. , Clemente, S. , Blamey, L. K. , Hereu, B. , Ballesteros, E. , Sala, E. , Jarrabou, E. , Cebrian, E. , Zabala, M. , Fujita, D. & Johnson, L. E. (2015). Global regime shift dynamics of catastrophic sea urchin overgrazing. Philosophical Transactions of the Royal Society B: Biological Sciences 370, 20130269. [Google Scholar]
- Ling, S. D. , Kriegisch, N. , Woolley, B. & Reeves, S. E. (2019). Density‐dependent feedbacks, hysteresis, and demography of overgrazing sea urchins. Ecology 100, e02577. [DOI] [PubMed] [Google Scholar]
- * Liu, S. , Trevathan‐Tackett, S. M. , Ewers Lewis, C. J. , Ollivier, Q. R. , Jiang, Z. , Huang, X. & Macreadie, P. I. (2019). Beach‐cast seagrass wrack contributes substantially to global greenhouse gas emissions. Journal of Environmental Management 231, 329–335. [DOI] [PubMed] [Google Scholar]
- * Llewellyn, P. J. & Shackley, S. E. (1996). The effects of mechanical beach‐cleaning on invertebrate populations. British Wildlife 7, 147–155. [Google Scholar]
- Lomstein, B. A. , Guldberg, L. B. , Neubauer, A.‐T. A. , Hansen, J. , Donnelly, A. , Herbert, R. A. , Viaroli, P. , Giordani, G. , Azzoni, R. , de Wit, R. & Finster, K. (2006). Benthic decomposition of Ulva lactuca: a controlled laboratory experiment. Aquatic Botany 85, 271–281. [Google Scholar]
- * López, B. A. , Macaya, E. C. , Jeldres, R. , Valdivia, N. , Bonta, C. C. , Tala, F. & Thiel, M. (2019). Spatio‐temporal variability of strandings of the southern bull kelp Durvillaea Antarctica (Fucales, Phaeophyceae) on beaches along the coast of Chile—linked to local storms. Journal of Applied Phycology 31, 2159–2173. [Google Scholar]
- * López, B. A. , Macaya, E. C. , Rivadeneira, M. M. , Tala, F. , Tellier, F. & Thiel, M. (2018). Epibiont communities on stranded kelp rafts of Durvillaea antarctica (Fucales, Phaeophyceae)—do positive interactions facilitate range extensions? Journal of Biogeography 45, 1833–1845. [Google Scholar]
- * López, B. A. , Macaya, E. C. , Tala, F. , Tellier, F. & Thiel, M. (2017). The variable routes of rafting: stranding dynamics of floating bull kelp Durvillaea antarctica (Fucales, Phaeophyceae) on beaches in the SE Pacific. Journal of Phycology 53, 70–84. [DOI] [PubMed] [Google Scholar]
- Lopez‐Uriarte, E. , Escofet, E. A. , Palacios, E. & Gonzalez, S. (1997). Migrant shorebirds at sandy beaches located between two major wetlands on the Pacific coast of Baja California (Mexico). Natural Areas Journal 17, 212–218. [Google Scholar]
- * Lord, W. D. & Burger, J. F. (1984). Arthropods associated with herring gull (Larus argentatus) and Great Black‐backed gull (Larus marinus) carrion on islands in the Gulf of Maine. Environmental Entomology 13, 1261–1268. [Google Scholar]
- * Losvik, M. H. (1983). Drift–line vegetation on well–drained, medium exposed beaches in the outward region of the fjords of Hordaland, Western Norway. Nordic Journal of Botany 3, 493–508. [Google Scholar]
- Loveless, A. M. & Oldham, C. E. (2010). Natural attenuation of nitrogen in groundwater discharging through a sandy beach. Biogeochemistry 98(1–3), 75–87. [Google Scholar]
- * Lowman, H. E. , Emery, K. A. , Kubler‐Dudgeon, L. , Dugan, J. E. & Melack, J. M. (2019). Contribution of macroalgal wrack consumers to dissolved inorganic nitrogen concentrations in intertidal pore waters of sandy beaches. Estuarine, Coastal and Shelf Science 219, 363–371. [Google Scholar]
- Lowman, H. E. , Emery, K. A. , Dugan, J. E. & Miller, R. J. (2021). Nutritional quality of giant kelp declines due to warming ocean temperatures. Oikos 00, 1–14. [Google Scholar]
- * Luarte, T. , Bonta, C. C. , Silva‐Rodriguez, E. A. , Quijón, P. A. , Miranda, C. , Farias, A. A. & Duarte, C. (2016). Light pollution reduces activity, food consumption and growth rates in a sandy beach invertebrate. Environmental Pollution 218, 1147–1153. [DOI] [PubMed] [Google Scholar]
- Lucrezi, S. & Schlacher, T. A. (2014). The ecology of ghost crabs. Oceanography and Marine Biology: An Annual Review 52, 201–256. [Google Scholar]
- Luijendijk, A. , Hagenaars, G. , Ranasinghe, R. , Baart, F. , Donchyts, G. & Aarninkhof, S. (2018). The state of the world's beaches. Scientific Reports 8, 6641. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lutter, H. , McGrath, M. B. , McGrath, M. A. & Debus, S. J. S. (2006). Observations on nesting Brahminy Kites Haliastur indus in Northern New South Wales. Australian Field Ornithology 23, 177–183. [Google Scholar]
- * Lyons, P. , Thornber, C. , Portnoy, J. & Gwilliam, E. (2009). Dynamics of macroalgal blooms along the Cape Cod National seashore. Northeastern Naturalist 16, 53–66. [Google Scholar]
- * MacMillan, M. R. & Quijón, P. A. (2012). Wrack patches and their influence on upper‐shore macrofaunal abundance in an Atlantic Canada sandy beach system. Journal of Sea Research 72, 28–37. [Google Scholar]
- * MacMillan, M. R. , Tummon Flynn, P. , Duarte, C. & Quijón, P. A. (2016). Near‐edge wrack effects on bare sediments: small scale variation matters in the monitoring of sandy beaches. Marine Environmental Research 122, 196–200. [DOI] [PubMed] [Google Scholar]
- * Macreadie, P. I. , Trevathan‐Tackett, S. M. , Baldock, J. A. & Kelleway, J. J. (2017). Converting beach‐cast seagrass wrack into biochar: a climate‐friendly solution to a coastal problem. Science of the Total Environment 574, 90–94. [DOI] [PubMed] [Google Scholar]
- * Malm, T. , Råberg, S. , Fell, S. & Carlsson, P. (2004). Effects of beach cast cleaning on beach quality, microbial food web, and littoral macrofaunal biodiversity. Estuarine, Coastal and Shelf Science 60, 339–347. [Google Scholar]
- * Marin Jarrin, J. R. & Shanks, A. L. (2011). Spatio‐temporal dynamics of the surf‐zone faunal assemblages at a Southern Oregon sandy beach. Marine Ecology 32, 232–242. [Google Scholar]
- Marks, L. M. , Reed, D. C. & Holbrook, S. J. (2020). Niche complementarity and resistance to grazing promote the invasion success of Sargassum horneri in North America. Diversity 12, 54. [Google Scholar]
- * Marques, J. C. , Gonçalves, S. C. , Pardal, M. A. , Chelazzi, L. , Colombini, I. , Fallaci, M. , Bouslama, M. F. , El Gtari, M. , Charfi‐Cheikhrouha, F. & Scapini, F. (2003). Comparison of Talitrus saltator (Amphipoda, Talitridae) biology, dynamics, and secondary production in Atlantic (Portugal) and Mediterranean (Italy and Tunisia) populations. Estuarine, Coastal and Shelf Science 58, 127–148. [Google Scholar]
- * Marsden, I. D. (1991. a). Kelp‐sandhopper interactions on a sand beach in New Zealand. I. Drift composition and distribution. Journal of Experimental Marine Biology and Ecology 152, 61–74. [Google Scholar]
- * Marsden, I. D. (1991. b). Kelp‐sandhopper interactions on a sand beach in New Zealand. II. Population dynamics of Talorchestia quoyana (Milne‐Edwards). Journal of Experimental Marine Biology and Ecology 152, 75–90. [Google Scholar]
- Martin, D. , Bertasi, F. , Colangelo, M. A. , de Vries, M. , Frost, M. , Hawkins, S. J. , Macpherson, E. , Moschella, P. S. , Satta, M. P. , Thompson, R. C. & Ceccherelli, V. U. (2005). Ecological impact of coastal defence structures on sediments and mobile infauna: evaluating and forecasting consequences of unavoidable modifications of native habitats. Coastal Engineering 52, 1027–1051. [Google Scholar]
- * Martins, M. S. , Massocato, T. F. , Horta, P. A. & Barufi, J. B. (2016). First record of red macroalgae bloom in Southern Atlantic Brazil. Algae 31, 33–39. [Google Scholar]
- * Masri, M. A. , Younes, S. , Haack, M. , Qoura, F. , Mehlmer, N. & Brück, T. (2018). A seagrass‐based biorefinery for generation of single‐cell oils for biofuel and oleochemical production. Energy Technology 6, 1026–1038. [Google Scholar]
- * Mateo, M. A. (2010). Beach‐cast Cymodocea nodosa along the shore of a semienclosed bay: sampling and elements to assess its ecological implications. Journal of Coastal Research 26, 283–291. [Google Scholar]
- * Mateo, M.‐A. , Sánchez‐Lizaso, J.‐L. & Romero, J. (2003). Posidonia oceanica ‘banquettes’: a preliminary assessment of the relevance for meadow carbon and nutrients budget. Estuarine, Coastal and Shelf Science 56, 85–90. [Google Scholar]
- * Matthews, D. C. (1955). Feeding habits of the sand crab Hippa pacifica (Dana). Pacific Science 9, 382–386. [Google Scholar]
- Maurer, A. S. , de Neef, E. & Stapleton, S. (2015). Sargassum accumulation may spell trouble for nesting sea turtles. Frontiers in Ecology and the Environment 13, 394–395. [Google Scholar]
- * McGwynne, L. E. , McLachlan, A. & Furstenburg, J. P. (1988). Wrack break‐down on sandy beaches. its impact on interstitial meiofauna. Marine Environmental Research 25, 213–232. [Google Scholar]
- * McKenzie, P. F. & Bellgrove, A. (2008). Dispersal of Hormosira banksii (Phaeophyceae) via detached fragments: reproductive viability and longevity. Journal of Phycology 44, 1108–1115. [DOI] [PubMed] [Google Scholar]
- * McLachlan, A. (1985). The biomass of macro‐ and interstitial fauna on clean and wrack‐covered beaches in Western Australia. Estuarine, Coastal and Shelf Science 21, 587–599. [Google Scholar]
- McLachlan, A. & Defeo, O. (eds) (2017). The Ecology of Sandy Shores, 3rd Edition (). Elsevier Academic Press, Cambridge. [Google Scholar]
- * McLachlan, A. , Eliot, I. G. & Clarke, D. J. (1985). Water filtration through reflective microtidal beaches and shallow sublittoral sands and its implications for an inshore ecosystem in Western Australia. Estuarine, Coastal and Shelf Science 21, 91–104. [Google Scholar]
- * Mclachlan, A. & McGwynne, L. (1986). Do sandy beaches accumulate nitrogen? Marine Ecology Progress Series 34, 191–195. [Google Scholar]
- McLachlan, A. & Romer, G. (1990). Trophic relationships in a high energy beach and surf zone ecosystem. In Trophic Relationships in the Marine Environment (eds Barnes M. and Gibson R. N.), pp. 356–371. Aberdeen University Press, Aberdeen. [Google Scholar]
- * Mellbrand, K. , Lavery, P. S. , Hyndes, G. & Hambäck, P. A. (2011). Linking land and sea: different pathways for marine subsidies. Ecosystems 14, 732–744. [Google Scholar]
- * Mews, M. , Zimmer, M. & Jelinski, D. E. (2006). Species‐specific decomposition rates of beach‐cast wrack in Barkley Sound, British Columbia, Canada. Marine Ecology Progress Series 328, 155–160. [Google Scholar]
- * Michaud, K. M. , Emery, K. A. , Dugan, J. E. , Hubbard, D. M. & Miller, R. J. (2019). Wrack resource use by intertidal consumers on sandy beaches. Estuarine, Coastal and Shelf Science 221, 66–71. [Google Scholar]
- Miller, R. J. & Page, H. M. (2012). Kelp as a trophic resource for marine suspension feeders: a review of isotope‐based evidence. Marine Biology 159(7), 1391–1402. [Google Scholar]
- Mir‐Gual, M. , Pons, G. X. , Martín‐Prieto, J. A. & Rodríguez‐Perea, A. (2015). A critical view of the Blue Flag beaches in Spain using environmental variables. Ocean and Coastal Management 105, 106–115. [Google Scholar]
- Misson, G. , Mainardis, M. , Incerti, G. , Goi, D. & Peressotti, A. (2020). Preliminary evaluation of potential methane production from anaerobic digestion of beach‐cast seagrass wrack: the case study of high‐adriatic coast. Journal of Cleaner Production 254, 120131. [Google Scholar]
- * Moeller, J. (1965). Ökologische untersuchungen über die terrestrische arthropodenfauna im anwurf mariner algen. Zeitschrift für Morphologie und Ökologie der Tiere 55, 530–586. [Google Scholar]
- * Molloy, F. J. & Bolton, J. J. (1995). Distribution, biomass and production of Gracilaria in Lüderitz Bay, Namibia. Journal of Applied Phycology 7, 381–392. [Google Scholar]
- * Montingelli, M. E. , Benyounis, K. Y. , Quilty, B. , Stokes, J. & Olabi, A. G. (2016). Optimisation of biogas production from the macroalgae Laminaria sp. at different periods of harvesting in Ireland. Applied Energy 177, 671–682. [Google Scholar]
- Moore, P. G. (2002). Mammals in intertidal and maritime ecosystems: interactions, impacts and implications. In Oceanography and Marine Biology: An Annual Review (eds Gibson R. N. and Atkinson R. J. A.), pp. 215–564. Aberdeen University Press/Allen & Unwin, Aberdeen. [Google Scholar]
- * Moore, P. G. & Francis, C. H. (1985). On the water relations and osmoregulation of the Beach‐Hopper Orchestia gammarellus (Pallas) (Crustacea: amphipoda). Journal of Experimental Marine Biology and Ecology 94, 131–150. [Google Scholar]
- Moore, I. & Legner, E. F. (1976). Intertidal rove beetles (Coleoptera: Staphylinidae). In Marine Insects (ed. Cheng L.), pp. 521–552. Elsevier, New York. [Google Scholar]
- * Morrow, K. , Bell, S. S. & Tewfik, A. (2014). Variation in ghost crab trophic links on sandy beaches. Marine Ecology Progress Series 502, 197–206. [Google Scholar]
- * Morton, B. (2003). Observations on the feeding behaviour of Nassarius clarus (Gastropoda: Nassariidae) in Shark Bay, Western Australia. Molluscan Research 23, 239–249. [Google Scholar]
- * Morton, B. & Yuen, W. Y. (2000). The feeding behaviour and competition for carrion between two sympatric scavengers on a sandy shore in Hong Kong: the gastropod, Nassarius festivus (Powys) and the hermit crab, Diogenes edwardsii (De Haan). Journal of Experimental Marine Biology and Ecology 246, 1–29. [DOI] [PubMed] [Google Scholar]
- * Morton, J. K. , Ward, E. J. & de Berg, K. C. (2015). Potential small‐ and large‐scale effects of mechanical beach cleaning on biological assemblages of exposed sandy beaches receiving low inputs of beach‐cast macroalgae. Estuaries and Coasts 38, 2083–2100. [Google Scholar]
- * Mossbauer, M. , Haller, I. , Dahlke, S. & Schernewski, G. (2012). Management of stranded eelgrass and macroalgae along the German Baltic coastline. Ocean and Coastal Management 57, 1–9. [Google Scholar]
- * Munroe, D. , Bushek, D. , Woodruff, P. & Calvo, L. (2017). Intertidal rack‐and‐bag oyster farms have limited interaction with horseshoe crab activity in New Jersey, USA. Aquaculture Environment Interactions 9, 205–211. [Google Scholar]
- Myers, M. R. , Barnard, P. L. , Beighley, E. , Cayan, D. R. , Dugan, J. E. , Feng, D. , Hubbard, D. M. , Iacobellis, S. F. , Melack, J. M. & Page, H. M. (2019). A multidisciplinary coastal vulnerability assessment for local government focused on ecosystems, Santa Barbara area, California. Ocean & Coastal Management 182, 104921. [Google Scholar]
- Neff, J. M. , Page, D. S. & Boehm, P. D. (2011). Exposure of sea otters and harlequin ducks in Prince William Sound, Alaska, USA, to shoreline oil residues 20 years after the Exxon Valdez oil spill. Environmental Toxicology and Chemistry 30, 659–672. [DOI] [PubMed] [Google Scholar]
- * Newton, C. , Bracken, M. E. S. , McConville, M. , Rodrigue, K. & Thornber, C. S. (2013). Invasion of the red seaweed Heterosiphonia japonica spans biogeographic provinces in the Western North Atlantic Ocean. PLoS One 8, e62261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- * Nicastro, A. , Onoda, Y. & Bishop, M. J. (2012). Direct and indirect effects of tidal elevation on eelgrass decomposition. Marine Ecology Progress Series 456, 53–62. [Google Scholar]
- * Nordstrom, K. F. , Jackson, N. L. , Freestone, A. L. , Korotky, K. H. & Puleo, J. A. (2012). Effects of beach raking and sand fences on dune dimensions and morphology. Geomorphology 179, 106–115. [Google Scholar]
- * Nordstrom, K. F. , Jackson, N. L. & Koroty, K. H. (2011. a). Aeolian sediment transport across beach wrack. Journal of Coastal Research 59, 211–217. [Google Scholar]
- * Nordstrom, K. F. , Jackson, N. L. , Korotky, K. H. & Puleo, J. A. (2011. b). Aeolian transport rates across raked and unraked beaches on a developed coast. Earth Surface Processes and Landforms 36, 779–789. [Google Scholar]
- * Nordstrom, K. F. , Jackson, N. L. , Smith, D. R. & Weber, R. G. (2006). Transport of horseshoe crab eggs by waves and swash on an estuarine beach: implications for foraging shorebirds. Estuarine, Coastal and Shelf Science 70, 438–448. [Google Scholar]
- * Ochieng, C. A. & Erftemeijer, P. L. A. (1999). Accumulation of seagrass beach cast along the Kenyan coast: a quantitative assessment. Aquatic Botany 65, 221–238. [Google Scholar]
- * Olabarria, C. , Incera, M. , Garrido, J. , Rodil, I. F. & Rossi, F. (2009). Intraspecific diet shift in Talitrus saltator inhabiting exposed sandy beaches. Estuarine, Coastal and Shelf Science 84, 282–288. [Google Scholar]
- Olabarria, C. , Incera, M. , Garrido, J. & Rossi, F. (2010). The effect of wrack composition and diversity on macrofaunal assemblages in intertidal marine sediments. Journal of Experimental Marine Biology and Ecology 396, 18–26. [Google Scholar]
- * Olabarria, C. , Lastra, M. & Garrido, J. (2007). Succession of macrofauna on macroalgal wrack of an exposed sandy beach: effects of patch size and site. Marine Environmental Research 63, 19–40. [DOI] [PubMed] [Google Scholar]
- * Oldham, C. , McMahon, K. , Brown, E. , Bosserelle, C. & Lavery, P. (2014). A preliminary exploration of the physical properties of seagrass wrack that affect its offshore transport, deposition, and retention on a beach. Limnology and Oceanography: Fluids and Environments 4, 120–135. [Google Scholar]
- Olds, A. D. , Vargas‐Fonseca, E. , Connolly, R. M. , Gilby, B. L. , Huijbers, C. M. , Hyndes, G. A. , Layman, C. A. , Whitfield, A. K. & Schlacher, T. A. (2018). The ecology of fish in the surf zones of ocean beaches: a global review. Fish and Fisheries 19, 78–89. [Google Scholar]
- Oosthuizen, W. H. , Meyer, M. A. , David, J. H. M. , Summers, N. M. , Kotze, G. H. P. , Swanson, S. W. & Shaughnessy, P. O. (1997). Variation in jackal numbers at the Van Reenen Bay seal colony with comment on likely importance of jackals as predators. South African Journal of Wildlife Research 27, 26–29. [Google Scholar]
- Orme, A. R. , Griggs, G. B. , Revell, D. L. , Zoulas, J. G. & Khoo, C. C. (2011). Beach changes along the southern California coast during the 20th century: a comparison of natural and human forcing factors. Shore & Beach 79, 38–50. [Google Scholar]
- * Orr, K. K. , Wilding, T. A. , Horstmeyer, L. , Weigl, S. & Heymans, J. J. (2014). Detached macroalgae: its importance to inshore sandy beach fauna. Estuarine, Coastal and Shelf Science 150, 125–135. [Google Scholar]
- * Orr, M. , Zimmer, M. , Jelinski, D. E. & Mews, M. (2005). Wrack deposition on different beach types: spatial and temporal variation in the pattern of subsidy. Ecology 86, 1496–1507. [Google Scholar]
- * Ortiz, J. F. & Gavio, B. (2010). Notes on the marine algae of the international biosphere reserve seaflower, Caribbean Colombia II: diversity of drift algae in San Andres Island, Caribbean Colombia. Caribbean Journal of Science 46, 313–321. [Google Scholar]
- Ortodossi, N. L. , Gilby, B. L. , Schlacher, T. A. , Connolly, R. M. , Yabsley, N. A. , Henderson, C. J. & Olds, A. D. (2019). Effects of seascape connectivity on reserve performance along exposed coastlines. Conservation Biology 33, 580–589. [DOI] [PubMed] [Google Scholar]
- Osterrieder, S. K. , Salgado Kent, C. & Robinson, R. W. (2017). Responses of Australian sea lions, Neophoca cinerea, to anthropogenic activities in the Perth metropolitan area, Western Australia. Aquatic Conservation: Marine and Freshwater Ecosystems 27, 414–435. [Google Scholar]
- * Paetzold, A. , Lee, M. & Post, D. M. (2008). Marine resource flows to terrestrial arthropod predators on a temperate Island: the role of subsidies between systems of similar productivity. Oecologia 157, 653–659. [DOI] [PubMed] [Google Scholar]
- * Page, H. M. , Schamel, J. , Emery, K. A. , Schooler, N. K. , Dugan, J. E. , Guglielmino, A. , Schroeder, D. M. , Palmstrom, L. , Hubbard, D. M. & Miller, R. J. (2021). Diet of a threatened endemic fox reveals variation in sandy beach resource use on California Channel Islands. PLoS One 16(10), e0258919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- * Pavesi, L. & De Matthaeis, E. (2013). Supralittoral amphipod abundances across habitats on Mediterranean temperate beaches. Journal of Coastal Conservation 17, 841–849. [Google Scholar]
- Pearse, A. S. , Humm, H. J. & Wharton, G. W. (1942). Ecology of sand beaches at Beaufort, NC. Ecological Monographs 12(2), 135–190. [Google Scholar]
- Pedersen, M. F. , Filbee‐Dexter, K. , Norderhaug, K. M. , Fredriksen, S. , Frisk, N. L. , Fagerli, C. W. & Wernberg, T. (2020). Detrital carbon production and export in high latitude kelp forests. Oecologia 192, 227–239. [DOI] [PubMed] [Google Scholar]
- * Pelletier, A. J. D. , Jelinski, D. E. , Treplin, M. & Zimmer, M. (2011). Colonisation of beach‐cast macrophyte wrack patches by talitrid amphipods: a primer. Estuaries and Coasts 34(4), 863–871. [Google Scholar]
- * Pennings, S. C. , Carefoot, T. H. , Zimmer, M. , Danko, J. P. & Ziegler, A. (2000). Feeding preferences of supralittoral isopods and amphipods. Canadian Journal of Zoology 78, 1918–1929. [Google Scholar]
- Percival, S. M. & Evans, P. R. (1997). Brent geese Branta bernicla and Zostera; factors affecting the exploitation of a seasonally declining food resource. Ibis 139, 121–128. [Google Scholar]
- * Petry, M. V. , Da Silva Fonseca, V. S. , Krüger‐Garcia, L. , Da Cruz Piuco, R. & Brummelhaus, J. (2008). Shearwater diet during migration along the coast of Rio Grande do Sul, Brazil. Marine Biology 154, 613–621. [Google Scholar]
- * Phillips, D. S. , Leggett, M. , Wilcockson, R. A. Y. , Day, T. H. & Arthur, W. (1995). Coexistence of competing species of seaweed flies: the role of temperature. Ecological Entomology 20, 65–74. [Google Scholar]
- Pilkey, O. H. , Neal, W. J. , Kelley, J. T. & Cooper, J. A. G. (2011). How beaches work: waves, currents, tides, and wind. In The World's Beaches: A Global Guide to the Science of the Shoreline, 1st Edition (), pp. 81–102. Berkeley: University of California Press. [Google Scholar]
- * Piriz, M. L. , Eyras, M. C. & Rostagno, C. M. (2003). Changes in biomass and botanical composition of beach‐cast seaweeds in a disturbed coastal area from Argentine Patagonia. Journal of Applied Phycology 15, 67–74. [Google Scholar]
- * Polis, G. A. & Hurd, S. D. (1995). Extraordinarily high spider densities on islands: flow of energy from the marine to terrestrial food webs and the absence of predation. Proceedings of the National Academy of Sciences of the United States of America 92, 4382–4386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- * Polis, G. A. & Hurd, S. D. (1996). Linking marine and terrestrial food webs: allochthonous input from the ocean supports high secondary productivity on small islands and coastal land communities. American Naturalist 147, 396–423. [Google Scholar]
- Polis, G. A. , Hurd, S. D. , Jackson, C. T. & Pińero, F. S. (1997). El Nino effects on the dynamics and control of an Island ecosystem in the gulf of California. Ecology 78, 1884–1897. [Google Scholar]
- * Poore, A. G. B. & Gallagher, K. M. (2013). Strong consequences of diet choice in a talitrid amphipod consuming seagrass and algal wrack. Hydrobiologia 701, 117–127. [Google Scholar]
- * Porri, F. , Hill, J. M. & McQuaid, C. D. (2011). Associations in ephemeral systems: the lack of trophic relationships between sandhoppers and beach wrack. Marine Ecology Progress Series 426, 253–262. [Google Scholar]
- * Prasad, M. H. K. , Ganguly, D. , Paneerselvam, A. , Ramesh, R. & Purvaja, R. (2019). Seagrass litter decomposition: an additional nutrient source to shallow coastal waters. Environmental Monitoring and Assessment 191, 5. [DOI] [PubMed] [Google Scholar]
- * Quilliam, R. S. , Jamieson, J. & Oliver, D. (2014). Seaweeds and plastic debris can influence the survival of faecal indicator organisms in beach environments. Marine Pollution Bulletin 84, 201–207. [DOI] [PubMed] [Google Scholar]
- Quijon, P. A. , Tummon, P. & Duarte, C. (2017). Beyond negative perceptions: the role of some marine invasive species as trophic subsidies. Marine Pollution Bulletin 116, 548–539. [DOI] [PubMed] [Google Scholar]
- * Quilter, C. G. (1987). Foraging activity of the sand beach isopod Scyphax ornatus Dana. New Zealand Journal of Zoology 14, 433–439. [Google Scholar]
- * Rae, C. , Hyndes, G. A. & Schlacher, T. A. (2019). Trophic ecology of ghost crabs with diverse tastes: unwilling vegetarians. Estuarine, Coastal and Shelf Science 224, 272–280. [Google Scholar]
- Redondo‐Gómez, D. , Quaggiotto, M.‐M. , Bailey, D. M. , Eguía, S. , Morales‐Reyes, Z. , López‐Pastor, B. N. , Martín‐Vega, D. , Martínez‐Carrasco, C. , Sebastián‐González, E. , Sánchez‐Zapata, J. A. & Moleón, M. (2022). Comparing scavenging in marine and terrestrial ecosystems: a case study with fish and gull carcasses in a small Mediterranean Island. Basic and Applied Ecology 59, 92–104. [Google Scholar]
- Reed, D. C. , Rassweiler, A. , Carr, M. H. , Cavanaugh, K. C. , Malone, D. P. & Siegel, D. A. (2011). Wave disturbance overwhelms top‐down and bottom‐up control of primary production in California kelp forests. Ecology 92, 2108–2116. [DOI] [PubMed] [Google Scholar]
- * Reimer, J. N. , Hacker, S. D. , Menge, B. A. & Ruggiero, P. (2018). Macrophyte wrack on sandy beaches of the US Pacific Northwest is linked to proximity of source habitat, ocean upwelling, and beach morphology. Marine Ecology Progress Series 594, 263–269. [Google Scholar]
- * Revell, D. L. , Dugan, J. E. & Hubbard, D. M. (2011). Physical and ecological responses of sandy beaches to the 1997‐98 El Niño. Journal of Coastal Research 27, 718–730. [Google Scholar]
- * Reynolds, C. E. & Houle, G. (2003). Mantel and partial Mantel tests suggest some factors that may control the local distribution of Aster laurentianus at Îles de la Madeleine, Québec. Plant Ecology 164, 19–27. [Google Scholar]
- * Rhodes, L. L. & Syhre, M. (1995). Okadaic acid production by a New Zealand Prorocentrum lima isolate. New Zealand Journal of Marine and Freshwater Research 29, 367–370. [Google Scholar]
- Ricci, S. , Colombini, I. , Fallaci, M. , Scoccianti, C. & Chelazzi, L. (1998). Arthropods as bioindicators of the red fox foraging activity in a Mediterranean beach‐dune system. Journal of Arid Environments 38, 335–348. [Google Scholar]
- Richards, L. J. (1982). Prey selection by an intertidal beetle: field test of an optimal diet model. Oecologia 55, 325–332. [DOI] [PubMed] [Google Scholar]
- * Rigby, M. C. (1996). The epibionts of beach hoppers (Crustacea: Talitridae) of the North American Pacific coast. Journal of Natural History 30, 1329–1336. [Google Scholar]
- * Robertson, A. I. & Lenanton, R. C. J. (1984). Fish community structure and food chain dynamics in the surf‐zone of sandy beaches: the role of detached macrophyte detritus. Journal of Experimental Marine Biology and Ecology 84, 265–283. [Google Scholar]
- * Robertson, A. I. & Lucas, J. S. (1983). Food choice, feeding rates, and the turnover of macrophyte biomass by a surf‐zone inhabiting amphipod. Journal of Experimental Marine Biology and Ecology 72, 99–124. [Google Scholar]
- * Rodil, I. F. , Fernandes, J. P. & Mucha, A. P. (2015. a). Disentangling the effects of solar radiation, wrack macroalgae and beach macrofauna on associated bacterial assemblages. Marine Environmental Research 112, 104–112. [DOI] [PubMed] [Google Scholar]
- * Rodil, I. F. , Lastra, M. , López, J. , Mucha, A. P. , Fernandes, J. P. , Fernandes, S. V. & Olabarria, C. (2019). Sandy beaches as biogeochemical hotspots: the metabolic role of macroalgal wrack on low‐productive shores. Ecosystems 22, 49–63. [Google Scholar]
- * Rodil, I. F. , Lucena‐Moya, P. , Olabarria, C. & Arenas, F. (2015. b). Alteration of macroalgal subsidies by climate‐associated stressors affects behavior of wrack‐reliant beach consumers. Ecosystems 18, 428–440. [Google Scholar]
- * Rodil, I. F. , Olabarria, C. , Lastra, M. & Arenas, F. (2015. c). Combined effects of wrack identity and solar radiation on associated beach macrofaunal assemblages. Marine Ecology Progress Series 531, 167–178. [Google Scholar]
- * Rodil, I. F. , Olabarria, C. , Lastra, M. & López, J. (2008). Differential effects of native and invasive algal wrack on macrofaunal assemblages inhabiting exposed sandy beaches. Journal of Experimental Marine Biology and Ecology 358, 1–13. [Google Scholar]
- Rodriguez, G. E. , Rassweiler, A. , Reed, D. C. & Holbrook, S. J. (2013). The importance of progressive senescence in the biomass dynamics of giant kelp (Macrocystis pyrifera). Ecology 94, 1848–1858. [DOI] [PubMed] [Google Scholar]
- Rodriguez, G. E. , Reed, D. C. & Holbrook, S. J. (2016). Blade life span, structural investment, and nutrient allocation in giant kelp. Oecologia 182, 397–404. [DOI] [PubMed] [Google Scholar]
- Rogers‐Bennett, L. & Catton, C. A. (2019). Marine heat wave and multiple stressors tip bull kelp forest to sea urchin barrens. Scientific Reports 9, 15050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rose, C. D. , Sharp, W. C. , Kenworthy, W. J. , Hunt, J. H. , Lyons, W. G. , Prager, E. J. , Valentine, J. F. , Hall, M. O. , Whitfield, P. E. & Fourqurean, J. W. (1999). Overgrazing of a large seagrass bed by the sea urchin Lytechinus variegatus in Outer Florida Bay. Marine Ecology Progress Series 190, 211–222. [Google Scholar]
- * Rose, M. D. & Polis, G. A. (1998). The distribution and abundance of coyotes: the effects of allochthonous food subsidies from the sea. Ecology 79, 998–1007. [Google Scholar]
- * Rossi, F. , Olabarria, C. , Incera, M. & Garrido, J. (2010). The trophic significance of the invasive seaweed Sargassum muticum in sandy beaches. Journal of Sea Research 63(1), 52–61. [Google Scholar]
- * Rossi, F. & Underwood, A. J. (2002). Small‐scale disturbance and increased nutrients as influences on intertidal macrobenthic assemblages: experimental burial of wrack in different intertidal environments. Marine Ecology Progress Series 241, 29–39. [Google Scholar]
- Rothäusler, E. , Macaya, E. C. , Molis, M. , Wahl, M. & Thiel, M. (2005). Laboratory experiments examining inducible defense show variable responses of temperate brown and red macroalgae. Revista Chilena de Historia Natural 78, 1–18. [Google Scholar]
- Rothäusler, E. & Thiel, M. (2006). Effect of detachment on the palatability of two kelp species. Journal of Applied Phycology 18, 423–435. [Google Scholar]
- * Rounds, R. A. , Erwin, R. M. & Porter, J. H. (2004). Nest‐site selection and hatching success of waterbirds in coastal Virginia: some results of habitat manipulation. Journal of Field Ornithology 75, 317–329. [Google Scholar]
- Rozen, D. E. , Engelmoer, D. J. P. & Smiseth, P. T. (2008). Antimicrobial strategies in burying beetles breeding on carrion. Proceedings of the National Academy of Sciences of the United States of America 105, 17890–17895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- * Ruiz‐Delgado, M. C. , Reyes‐Martínez, M. J. , Sánchez‐Moyano, J. E. , López‐Pérez, J. & García‐García, F. J. (2015). Distribution patterns of supralittoral arthropods: wrack deposits as a source of food and refuge on exposed sandy beaches (SW Spain). Hydrobiologia 742, 205–219. [Google Scholar]
- * Ruiz‐Delgado, M. C. , Vieira, J. V. , Reyes‐Martínez, M. J. , Borzone, C. A. , Outerelo, R. , Sánchez‐Moyano, J. E. & García‐García, F. J. (2016. a). Colonisation patterns of supralittoral arthropods in naturally stranded wrack debris on Atlantic sandy beaches of Brazil and Spain. Marine and Freshwater Research 67, 1634–1643. [DOI] [PubMed] [Google Scholar]
- * Ruiz‐Delgado, M. C. , Vieira, J. V. , Veloso, V. G. , Reyes‐Martínez, M. J. , Sallorenzo, I. A. , Borzone, C. A. , Sánchez‐Moyano, J. E. & García García, F. J. (2014). The role of wrack deposits for supralittoral arthropods: an example using Atlantic sandy beaches of Brazil and Spain. Estuarine, Coastal and Shelf Science 136, 61–71. [Google Scholar]
- * Ruiz‐Delgado, M. C. , Vierheller Vieira, J. , Reyes‐Martínez, M. J. , Borzone, C. A. , Sánchez‐Moyano, J. E. & García‐García, F. J. (2016. b). Wrack removal as short‐term disturbance for Talitrus saltator density in the supratidal zone of sandy beaches: an experimental approach. Estuaries and Coasts 39, 1113–1121. [Google Scholar]
- * Russell, T. L. , Sassoubre, L. M. , Wang, D. , Masuda, S. , Chen, H. , Soetjipto, C. , Hassaballah, A. & Boehm, A. B. (2013). A coupled modeling and molecular biology approach to microbial source tracking at Cowell Beach, Santa Cruz, CA, United States. Environmental Science and Technology 47(18), 10231–10239. [DOI] [PubMed] [Google Scholar]
- * Russell, T. L. , Sassoubre, L. M. , Zhou, C. , French‐Owen, D. , Hassaballah, A. & Boehm, A. B. (2014). Impacts of beach wrack removal via grooming on surf zone water quality. Environmental Science and Technology 48, 2203–2211. [DOI] [PubMed] [Google Scholar]
- Ruxton, G. D. (2004). Energetic feasibility of an obligate marine scavenger. Marine Ecology Progress Series 266, 59–63. [Google Scholar]
- * Salathé, R. & Riera, P. (2012). The role of Talitrus saltator in the decomposition of seaweed wrack on sandy beaches in northern Brittany: an experimental mesocosm approach. Cahiers de Biologie Marine 53, 517–524. [Google Scholar]
- Santoro, A. E. , Boehm, A. B. & Francis, C. A. (2006). Denitrifier community composition along a nitrate and salinity gradient in a coastal aquifer. Applied and Environmental Microbiology 72, 2102–2109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- * Satoh, A. , Uéda, T. & Hori, M. (2005). The macrofauna associated with stranded wrack on a sandy shore in Japan. Japanese Journal of Ecology 55, 21–27. [Google Scholar]
- Säwström, C. , Hyndes, G. A. , Eyre, B. D. , Huggett, M. J. , Fraser, M. W. , Lavery, P. S. , Thomson, P. G. , Tarquinio, F. , Steinberg, P. D. & Laverock, B. (2016). Coastal connectivity and spatial subsidy from a microbial perspective. Ecology and Evolution 6, 6662–6671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- * Scapini, F. & Dugan, J. (2008). Sun and landscape orientation in adult and juvenile sandhoppers Orchestoidea tuberculata (Amphipoda, Talitridae) from two beaches in south central Chile. Marine Ecology 29, 19–27. [Google Scholar]
- Schell, J. M. , Goodwin, D. S. & Siuda, A. N. S. (2015). Recent Sargassum inundation events in the Caribbean: Shipboard observations reveal dominance of a previously rare form. Oceanography 28, 8–10. [Google Scholar]
- Schiel, D. , Lilley, S. & South, P. (2018). Ecological tipping points for an invasive kelp in rocky reef algal communities. Marine Ecology Progress Series 587, 93–104. [Google Scholar]
- * Schlacher, T. A. , Carracher, L. K. , Porch, N. , Connolly, R. M. , Olds, A. D. , Gilby, B. L. , Ekanayake, K. B. , Maslo, B. & Weston, M. A. (2016). The early shorebird will catch fewer invertebrates on trampled sandy beaches. PLoS One 11, e0161905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- * Schlacher, T. A. , Gilby, B. L. , Olds, A. D. , Henderson, C. J. , Connolly, R. M. , Peterson, C. H. , Voss, C. M. , Maslo, B. , Weston, M. A. , Bishop, M. J. & Rowden, A. (2020). Key ecological function peaks at the land–ocean transition zone when vertebrate scavengers concentrate on ocean beaches. Ecosystems 23, 906–916. [Google Scholar]
- * Schlacher, T. A. , Hutton, B. M. , Gilby, B. L. , Porch, N. , Maguire, G. S. , Maslo, B. , Connolly, R. M. , Olds, A. D. & Weston, M. A. (2017). Algal subsidies enhance invertebrate prey for threatened shorebirds: a novel conservation tool on ocean beaches? Estuarine, Coastal and Shelf Science 191, 28–38. [Google Scholar]
- Schlacher, T. A. , Schoeman, D. S. , Dugan, J. E. , Lastra, M. , Jones, A. , Scapini, F. & Mclachlan, A. (2008). Sandy beach ecosystems: key features, sampling issues, management challenges and climate change impacts. Marine Ecology 29, 70–90. [Google Scholar]
- * Schlacher, T. A. , Strydom, S. & Connolly, R. M. (2013. a). Multiple scavengers respond rapidly to pulsed carrion resources at the land‐ocean interface. Acta Oecologica 48, 7–12. [Google Scholar]
- * Schlacher, T. A. , Strydom, S. , Connolly, R. M. & Schoeman, D. (2013. b). Donor‐control of scavenging food webs at the land‐ocean interface. PLoS One 8, e68221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- * Schooler, N. K. , Dugan, J. E. & Hubbard, D. M. (2019). No lines in the sand: impacts of intense mechanized maintenance regimes on sandy beach ecosystems span the intertidal zone on urban coasts. Ecological Indicators 106, 105457. [Google Scholar]
- * Schooler, N. K. , Dugan, J. E. , Hubbard, D. M. & Straughan, D. (2017). Local scale processes drive long‐term change in biodiversity of sandy beach ecosystems. Ecology and Evolution 7, 4822–4834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- * Schultz Schiro, J. A. , Meyer‐Arendt, K. J. & Schneider, S. K. (2017). Sargassum on Santa Rosa Island, Florida: faunal use and beachgoer perception. Journal of Coastal Conservation 21, 63–83. [Google Scholar]
- * Serrano, R. , Gras, L. , Giménez‐Casalduero, F. , del‐Pilar‐Ruso, Y. , Grindlay, G. & Mora, J. (2019). The role of Cymodocea nodosa on the dynamics of trace elements in different marine environmental compartments at the Mar Menor Lagoon (Spain). Marine Pollution Bulletin 141, 52–60. [DOI] [PubMed] [Google Scholar]
- Seymour, R.J. (2003). The influence of global climate change on extreme wave occurrence on the west coast of the United States. In Proceedings of the Coastal Engineering Conference, pp. 52–60.
- * Shepherd, L. D. & Heenan, P. B. (2017). Origins of beach‐cast Sophora seeds from the Kermadec and Chatham Islands. New Zealand Journal of Botany 55, 241–248. [Google Scholar]
- Sikes, D. S. & Slowik, J. (2010). Terrestrial arthropods of pre‐ and post‐eruption Kasatochi Island, Alaska, 2008–2009: a shift from a plant‐based to a necromass‐based food web. Arctic, Antarctic, and Alpine Research 42, 297–305. [Google Scholar]
- * Simeone, S. & De Falco, G. (2012). Morphology and composition of beach‐cast Posidonia oceanica litter on beaches with different exposures. Geomorphology 151–152, 224–233. [Google Scholar]
- * Singh, C. L. , Huggett, M. J. , Lavery, P. S. , Säwström, C. & Hyndes, G. A. (2021). Kelp‐associated microbes facilitate spatial subsidy in a detrital‐based food web in a shoreline ecosystem. Frontiers in Marine Science 8, 678222. [Google Scholar]
- Skinner, J. D. , van Aarde, R. J. & Goss, R. A. (1995). Space and resource use by brown hyenas Hyaena brunnea in the Namib Desert. Journal of Zoology 237, 123–131. [Google Scholar]
- Smale, D. A. , Wernberg, T. , Oliver, E. C. J. , Thomsen, M. , Harvey, B. P. , Straub, S. C. , Burrows, M. T. , Alexander, L. V. , Benthuysen, J. A. , Donat, M. G. , Feng, M. , Hobday, A. J. , Holbrook, N. J. , Perkins‐Kirkpatrick, S. E. , Scannell, H. A. , et al. (2019). Marine heatwaves threaten global biodiversity and the provision of ecosystem services. Nature Climate Change 9, 306–312. [Google Scholar]
- Small, C. & Nicholls, R. J. (2003). A global analysis of human settlement in coastal zones. Journal of Coastal Research 19, 584–599. [Google Scholar]
- Smith, G. C. (1985). An analysis of prey remnants from Osprey Pandion haliaetus and White‐Bellied Sea‐Eagle Haliaetus leucogaster feeding roosts. Emu—Austral Ornithology 85, 198–200. [Google Scholar]
- Smith, L. A. , Johnston, R. E. & Dell, J. (1978). ‘Part IV Birds’: the islands of the north‐west Kimberley, Western Australia. Wildlife Research Bulletin Western Australia 23, 67–82. [Google Scholar]
- * Soares, A. G. , McLachlan, A. & Schlacher, T. (1996). Disturbance effects of stranded kelp on populations of the sandy beach bivalve Donax serra (Röding). Journal of Experimental Marine Biology and Ecology 205, 165–186. [Google Scholar]
- Soares, A. G. , Schlacher, T. A. & McLachlan, A. (1997). Carbon and nitrogen exchange between sandy beach clams (Donax serra) and kelp beds in the Benguela coastal upwelling region. Marine Biology 127, 657–664. [Google Scholar]
- * Sobocinski, K. L. , Cordell, J. R. & Simenstad, C. A. (2010). Effects of shoreline modifications on supratidal macroinvertebrate fauna on Puget Sound, Washington beaches. Estuaries and Coasts 33, 699–711. [Google Scholar]
- Somers, M. J. (2000). Seasonal variation in the diet of Cape clawless otters (Aonyx copensis) in a marine habitat. African Zoology 35, 261–268. [Google Scholar]
- * Spiller, D. A. , Piovia‐Scott, J. , Wright, A. N. , Yang, L. H. , Takimoto, G. , Schoener, T. W. & Iwata, T. (2010). Marine subsidies have multiple effects on coastal food webs. Ecology 91, 1424–1434. [DOI] [PubMed] [Google Scholar]
- * Stapp, P. & Polis, G. A. (2003). Marine resources subsidize insular rodent populations in the Gulf of California, Mexico. Oecologia 134, 496–504. [DOI] [PubMed] [Google Scholar]
- Staufenberger, T. , Thiel, V. , Wiese, J. & Imhoff, J. F. (2008). Phylogenetic analysis of bacteria associated with Laminaria saccharina . FEMS Microbiology Ecology 64, 65–77. [DOI] [PubMed] [Google Scholar]
- * Stenton‐Dozey, J. & Griffiths, C. L. (1980). Growth, consumption and respiration by larvae of the kelp‐fly Fucellia capensis (Diptera:Anthomyiidae). South African Journal of Zoology 15, 280–283. [Google Scholar]
- * Stenton‐Dozey, J. M. E. & Griffiths, C. L. (1983). The fauna associated with kelp stranded on a sandy beach. In Sandy Beaches as Ecosystems (eds McLachlan A. and Erasmus T.), pp. 557–568. Springer Netherlands, Dordrecht. [Google Scholar]
- * Stocking, K. A. , Duignan, J. P. , Roe, W. D. , Meynier, L. , Alley, M. & Fettermann, T. (2009). Causes of mortality in stranded common dolphin (Delphinus sp.) from New Zealand waters between 1998 and 2008. Pacific Conservation Biology 15, 217–227. [Google Scholar]
- * Strain, E. M. A. , Heath, T. , Steinberg, P. D. & Bishop, M. J. (2018). Eco‐engineering of modified shorelines recovers wrack subsidies. Ecological Engineering 112, 26–33. [Google Scholar]
- Strydom, S. , Murray, K. , Wilson, S. , Huntley, B. , Rule, M. , Heithaus, M. , Bessey, C. , Kendrick, G. A. , Burkholder, D. , Fraser, M. W. & Zdunic, K. (2020). Too hot to handle: unprecedented seagrass death driven by marine heatwave in a world heritage area. Global Change Biology 26, 3525–3538. [DOI] [PubMed] [Google Scholar]
- Stuart, V. , Field, J. & Newell, R. (1982). Evidence for absorption of kelp detritus by the ribbed mussel Aulacomya ater using a new 51Cr‐labelled microsphere technique. Marine Ecology Progress Series 9, 263–271. [Google Scholar]
- Suárez‐Jiménez, R. , Hepburn, C. D. , Hyndes, G. A. , McLeod, R. J. , Taylor, R. B. & Hurd, C. L. (2017. a). Importance of the invasive macroalga Undaria pinnatifida as trophic subsidy for a beach consumer. Marine Biology 164, 113. [Google Scholar]
- Suárez‐Jiménez, R. , Hepburn, C. D. , Hyndes, G. A. , McLeod, R. J. , Taylor, R. B. & Hurd, C. L. (2017. b). The invasive kelp Undaria pinnatifida hosts an epifaunal assemblage similar to native seaweeds with comparable morphologies. Marine Ecology Progress Series 582, 45–55. [Google Scholar]
- Sunagawa, S. , Coelho, L. P. , Chaffron, S. , Kultima, J. R. , Labadie, K. , Salazar, G. , Djahanschiri, B. , Zeller, G. , Mende, D. R. , Alberti, A. , Cornejo‐Castillo, F. M. , Costea, P. I. , Cruaud, C. , D'Ovidio, F. , Engelen, S. , et al. (2015). Structure and function of the global ocean microbiome. Science 348, 1261359. [DOI] [PubMed] [Google Scholar]
- Sura, S. A. , Delgadillo, A. , Franco, N. , Gu, K. , Turba, R. & Fong, P. (2019). Macroalgae and nutrients promote algal turf growth in the absence of herbivores. Coral Reefs 38, 425–429. [Google Scholar]
- * Suursaar, Ü. , Torn, K. , Martin, G. , Herkül, K. & Tiit, K. (2014). Formation and species composition of stormcast beach wrack in the Gulf of Riga, Baltic Sea. Oceanologia 56, 673–695. [Google Scholar]
- * Swinscoe, I. , Oliver, D. M. , Gilburn, A. S. & Quilliam, R. S. (2018). The seaweed fly (Coelopidae) can facilitate environmental survival and transmission of E. coli O157 at sandy beaches. Journal of Environmental Management 223, 275–285. [DOI] [PubMed] [Google Scholar]
- * Talbot, M. M. B. & Bate, G. C. (1988). The relative quantities of live and detrital organic matter in a beach‐surf ecosystem. Journal of Experimental Marine Biology and Ecology 121, 255–264. [Google Scholar]
- Tarquinio, F. , Bourgoure, J. , Koenders, A. , Laverock, B. , Säwström, C. & Hyndes, G. A. (2018). Microorganisms facilitate uptake of dissolved organic nitrogen by seagrass leaves. ISME Journal 12, 2796–2800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tarquinio, F. , Hyndes, G. A. , Laverock, B. , Koenders, A. & Säwström, C. (2019). The seagrass holobiont: understanding seagrass‐bacteria interactions and their role in seagrass ecosystem functioning. FEMS Microbiology Letters 366, 1–15. [DOI] [PubMed] [Google Scholar]
- * Tarr, J. G. & Tarr, P. W. (1987). Seasonal abundance and the distribution of coastal birds on the northern Skeleton Coast, South Africa/Namibia. Madoqua 15, 63–72. [Google Scholar]
- * Terrados, J. , Marín, A. & Celdrán, D. (2013). Use of Posidonia oceanica seedlings from beach‐cast fruits for seagrass planting. Botanica Marina 56, 185–195. [Google Scholar]
- Tershy, B. R. , Breese, D. & Croll, D. A. (1997). Human perturbations and conservation strategies for San Pedro Martir Island, Islas del Golfo de California Reserve, Mexico. Environmental Conservation 24, 261–270. [Google Scholar]
- * Tewfik, A. , Bell, S. S. , McCann, K. S. & Morrow, K. (2016). Predator diet and trophic position modified with altered habitat morphology. PLoS One 11, e0147759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thompson, F. L. , Abreu, P. C. & Cavalli, R. (1999). The use of microorganisms as food source for Penaeus paulensis larvae. Aquaculture 174, 139–153. [Google Scholar]
- Thomsen, M. S. , Mondardini, L. , Alestra, T. , Gerrity, S. , Tait, L. , South, P. M. , Lilley, S. A. & Schiel, D. R. (2019). Local extinction of Bull kelp (Durvillaea spp.) due to a marine heatwave. Frontiers in Marine . Science 6, 84. [Google Scholar]
- * Torn, K. , Martin, G. & Suursaar, Ü. (2016). Beach wrack macrovegetation index for assessing coastal phytobenthic biodiversity [Mereheidise indeks rannikumere põhjataimestiku bioloogilise mitmekesisuse hindamiseks]. Proceedings of the Estonian Academy of Sciences 65, 78–87. [Google Scholar]
- Trevathan‐Tackett, S. M. , Macreadie, P. I. , Sanderman, J. , Baldock, J. , Howes, J. M. & Ralph, P. J. (2017). A global assessment of the chemical recalcitrance of seagrass tissues: implications for long‐term carbon sequestration. Frontiers in Plant Science 8, 925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trias, R. , García‐Lledó, A. , Sánchez, N. , López‐Jurado, J. L. , Hallin, S. & Bañeras, L. (2012). Abundance and composition of epiphytic bacterial and archaeal ammonia oxidizers of marine red and brown macroalgae. Applied and Environmental Microbiology 78, 318–325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- * Urban‐Malinga, B. & Burska, D. (2009). The colonization of macroalgal wrack by the meiofauna in the Arctic intertidal. Estuarine, Coastal and Shelf Science 85, 666–670. [Google Scholar]
- * Urban‐Malinga, B. , Drgas, A. , Ameryk, A. & Tatarek, A. (2009). Meiofaunal (re)colonization of the Arctic intertidal (Hornsund, Spitsbergen) after ice melting: role of wrack deposition. Polar Biology 32, 243–252. [Google Scholar]
- * Urban‐Malinga, B. , Gheskiere, T. , Degraer, S. , Derycke, S. , Opalinski, K. W. & Moens, T. (2008). Gradients in biodiversity and macroalgal wrack decomposition rate across a macrotidal, ultradissipative sandy beach. Marine Biology 155, 79–90. [Google Scholar]
- Urish, D. W. & McKenna, T. E. (2004). Tidal effects on ground water discharge through a sandy marine beach. Ground Water 42, 971–982. [Google Scholar]
- * van der Merwe, D. & McLachlan, A. (1987). Significance of free‐floating macrophytes in the ecology of a sandy beach surf zone. Marine Ecology Progress Series 38, 53–63. [Google Scholar]
- * van Egmond, E. M. , van Bodegom, P. M. , van Hal, J. R. , van Logtestijn, R. S. P. , Broekman, R. A. , Berg, M. P. & Aerts, R. (2019). Growth of pioneer beach plants is strongly driven by buried macroalgal wrack, whereas macroinvertebrates affect plant nutrient dynamics. Journal of Experimental Marine Biology and Ecology 514–515, 87–94. [Google Scholar]
- * van Tussenbroek, B. I. , Hernández Arana, H. A. , Rodríguez‐Martínez, R. E. , Espinoza‐Avalos, J. , Canizales‐Flores, H. M. , González‐Godoy, C. E. , Barba‐Santos, M. G. , Vega‐Zepeda, A. & Collado‐Vides, L. (2017). Severe impacts of brown tides caused by Sargassum spp. on near‐shore Caribbean seagrass communities. Marine Pollution Bulletin 122, 272–281. [DOI] [PubMed] [Google Scholar]
- * Vargas‐Fonseca, E. , Olds, A. D. , Gilby, B. L. , Connolly, R. M. , Schoeman, D. S. , Huijbers, C. M. , Hyndes, G. A. & Schlacher, T. A. (2016). Combined effects of urbanization and connectivity on iconic coastal fishes. Diversity and Distributions 22, 1328–1341. [Google Scholar]
- * Venables, B. J. (1981). Aspects of the population biology of a Venezuelan beach amphipod, Talorchestia margaritae (Talitridae), including estimates of biomass and daily production, and respiration rates. Crustaceana 41, 271–285. [Google Scholar]
- Vergés, A. , Steinberg, P. D. , Hay, M. E. , Poore, A. G. B. , Campbell, A. H. , Ballesteros, E. , Heck, K. L. Jr. , Booth, D. J. , Coleman, M. A. , Feary, D. A. , Figueira, W. , Langlois, T. , Marzinelli, E. M. , Mizerek, T. , Mumby, P. J. , et al. (2014). The tropicalization of temperate marine ecosystems: climate‐mediated changes in herbivory and community phase shifts. Proceedings of the Royal Society B: Biological Sciences 281, 20140846. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Verschut, V. , Strandmark, A. , Esparza‐Salas, R. & Hambäck, P. A. (2019). Seasonally varying marine influences on the coastal ecosystem detected through molecular gut analysis. Molecular Ecology 28, 307–317. [DOI] [PubMed] [Google Scholar]
- * Vieira, J. v. , Ruiz‐Delgado, M. C. , Reyes‐Martínez, M. J. , Borzone, C. A. , Asenjo, A. , Sánchez‐Moyano, J. E. & García‐García, F. J. (2016). Assessment the short‐term effects of wrack removal on supralittoral arthropods using the M‐BACI design on Atlantic sandy beaches of Brazil and Spain. Marine Environmental Research 119, 222–237. [DOI] [PubMed] [Google Scholar]
- * Villares, R. , Fernández‐Lema, E. & López‐Mosquera, M. E. (2016). Evaluation of beach wrack for use as an organic fertilizer: temporal survey in different areas. Thalassas 32, 19–36. [Google Scholar]
- * Virtue, P. & Nichols, P. D. (1994). Lipids from the bull kelp Durvillaea potatorum . Phytochemistry 37, 673–676. [Google Scholar]
- Vitousek, S. , Barnard, P. L. & Limber, P. (2017). Can beaches survive climate change? Journal of Geophysical Research: Earth Surface 122, 1060–1067. [Google Scholar]
- Vousdoukas, M. I. , Ranasinghe, R. , Mentaschi, L. , Plomaritis, T. A. , Athanasiou, P. , Luijendijk, A. & Feyen, L. (2020). Sandy coastlines under threat of erosion. Nature Climate Change 10, 260–263. [Google Scholar]
- Wahl, M. , Goecke, F. , Labes, A. , Dobretsov, S. & Weinberger, F. (2012). The second skin: ecological role of epibiotic biofilms on marine organisms. Frontiers in Microbiology 3, 292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- * Waters, J. M. , King, T. M. , Fraser, C. I. & Craw, D. (2018). Crossing the front: contrasting stormforced dispersal dynamics revealed by biological, geological and genetic analysis of beach‐cast kelp. Journal of the Royal Society Interface 15, 20180046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Waycott, M. , Duarte, C. M. , Carruthers, T. J. B. , Orth, R. J. , Dennison, W. C. , Olyarnik, S. , Calladine, A. , Fourqurean, J. W. , Heck, K. L. Jr. , Hughes, A. R. , Kendrick, G. A. , Kenworthy, W. J. , Short, F. T. & Williams, S. L. (2009). Accelerating loss of seagrasses across the globe threatens coastal ecosystems. Proceedings of the National Academy of Sciences of the United States of America 106, 12377–12381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- * Weber, L. I. , Porto Barbosa, T. , Beraldini, N. G. & da Silva, V. C. (2019). Two‐year study of food availability, demographic structure, and genetic diversity of the most common sandhopper in southeastern Brazil. Regional Studies in Marine Science 32, 100876. [Google Scholar]
- * Wells, F. (2002). Seasonality of beachwrack at Oakajee in the mid‐west region. Records of the Western Australian Museum 21, 269–275. [Google Scholar]
- * Wenner, A. M. (1977). Food supply, feeding habits, and egg production in Pacific mole crabs (Hippa pacifica Dana). Pacific Science 31, 39–47. [Google Scholar]
- * Wenner, A. M. , Ricard, Y. & Dugan, J. (1987). Hippid crab population structure and food availability on Pacific shorelines. Bulletin of Marine Science 41, 221–233. [Google Scholar]
- Wernberg, T. , Bennett, S. , Babcock, R. C. , de Bettignies, T. , Cure, K. , Depczynski, M. , Dufois, F. , Fromont, J. , Fulton, C. J. , Hovey, R. K. , Harvey, E. S. , Holmes, T. H. , Kendrick, G. A. , Radford, B. , Santana‐Garcon, J. , et al. (2016). Climate‐driven regime shift of a temperate marine ecosystem. Science 353, 169–172. [DOI] [PubMed] [Google Scholar]
- Wernberg, T. , Krumhansl, K. , Filbee‐Dexter, K. & Pedersen, M. (2019). Status and trends for the world's kelp forests. In World Seas: An Environmental Evaluation, 2nd Edition (ed. Sheppard C.), pp. 57–78. London: Academic Press. [Google Scholar]
- * Westermeier, R. , Gomez, I. , Rivera, P. J. & Muller, D. (1992). Macroalgas marinas antracticas: distributcion, abundancia y necromasa en isla Rey Jorge, Shetland del Sur, Antarctica. Serie Científica 42, 21–34. [Google Scholar]
- Whelan, C. J. , Wenny, D. G. & Marquis, R. J. (2008). Ecosystem services provided by birds. Annals of the New York Academy of Sciences 1134, 25–60. [DOI] [PubMed] [Google Scholar]
- White, T. C. R. (1993). The Inadequate Environment. Springer‐Verlag, Berlin. [Google Scholar]
- * Whitfield, A. K. (1988). The role of tides in redistributing macrodetrital aggregates within the Swartvlei estuary. Estuaries 11, 152–159. [Google Scholar]
- * Wickham, S. , Shackelford, N. , Darimont, C. , Nijland, W. , Reshitnyk, L. , Reynolds, J. & Starzomski, B. (2020). Sea wrack delivery and accumulation on islands: factors that mediate marine nutrient permeability. Marine Ecology Progress Series 635, 37–54. [Google Scholar]
- * Williams, A. & Feagin, R. (2010). Sargassum as a natural solution to enhance dune plant growth. Environmental Management 46, 738–747. [DOI] [PubMed] [Google Scholar]
- * Wilson, D. S. & Knollenberg, W. G. (1984). Food discrimination and ovarian development in burying beetles (Coleoptera: Silphidae: Nicrophorus). Annals of the Entomological Society of America 77, 165–170. [Google Scholar]
- Wilson, E. E. & Wolkovich, E. M. (2011). Scavenging: how carnivores and carrion structure communities. Trends in Ecology and Evolution 26, 129–135. [DOI] [PubMed] [Google Scholar]
- Witherington, B. , Hirama, S. & Mosier, A. (2011). Sea turtle responses to barriers on their nesting beach. Journal of Experimental Marine Biology and Ecology 401, 1–6. [Google Scholar]
- Wolcott, T. G. (1978). Ecological role of ghost crabs, Ocypode quadrata (Fabricius) on an ocean beach: scavengers or predators? Journal of Experimental Marine Biology and Ecology 31, 67–82. [Google Scholar]
- * Wright, J. M. (1989). Detached chlorophytes as nursery areas for fish in Sulaibikhat Bay, Kuwait. Estuarine, Coastal and Shelf Science 28, 185–193. [Google Scholar]
- * Yamazaki, K. (2012). Seasonal changes in seaweed deposition, seaweed fly abundance, and parasitism at the pupal stage along sandy beaches in Central Japan. Entomological Science 15, 28–34. [Google Scholar]
- Yanenik, J. S. (1980). Beach wrack: phenology of an imported resource and utilization by macroinvertebates of sandy beaches . MA Thesis, University of California, Berkeley.
- Yorke, C. E. , Miller, R. J. , Page, H. M. & Reed, D. C. (2013). Importance of kelp detritus as a component of suspended particulate organic matter in giant kelp Macrocystis pyrifera forests. Marine Ecology Progress Series 493, 113–125. [Google Scholar]
- Zielinski, S. , Botero, C. M. & Yanes, A. (2019). To clean or not to clean? A critical review of beach cleaning methods and impacts. Marine Pollution Bulletin 139, 390–401. [DOI] [PubMed] [Google Scholar]
- Zimmer, M. & Topp, W. (1997). Does leaf litter quality influence population parameters of the common woodlouse, Porcellio scaber (Crustacea: isopoda)? Biology and Fertility of Soils 24, 435–441. [Google Scholar]
- * Zobell, C. E. (1971). Drift seaweeds on the San Diego county beaches. Bieheft Nova Hedwigia 32, 269–314. [Google Scholar]
- Zoulas, J. G. & Orme, A. R. (2007). Multidecadal‐Scale Beach changes in the Zuma Littoral cell, California. Physical Geography 28, 277–300. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Table S1. All publications identified in our search on various aspects of wrack or carrion in sandy‐beach ecosystems, and their locations, habitats and research themes.
Table S2. References used to produce Fig. 6.
Table S3. References used to produce Fig. 7, including details of the country (region), habitat sampled and type of material in wrack (mixed = seagrass and macroalgae).
Table S4. References used to produce Fig. 8.


