Abstract
Background
Although concomitant pulmonary vein isolation (PVI) is used more frequently than the Cox‐Maze procedure, which is currently the gold standard treatment for atrial fibrillation (AF), data on the comparative effectiveness of the two procedures after concomitant mitral valve (MV) surgery are still limited.
Objective
We conducted a systematic review to identify randomized controlled trials (RCTs) and observational studies comparing the mid‐term mortality and recurrence of AF after concomitant Cox‐Maze and PVI in patients with AF undergoing MV surgery based on 12‐month follow‐up.
Methods
Medline, EMBASE databases, and the Cochrane Library were searched from 1987 up to March 2022 for studies comparing concomitant Cox‐Maze and PVI. Additionally, a meta‐analysis of RCTs was performed to compare the mid‐term clinical outcomes between these two surgical ablation techniques.
Results
Three RCTs and three observational studies meeting the inclusion criteria were included in this systematic review with 790 patients in total (532 concomitant Cox‐Maze and 258 PVI during MV surgery). Most studies reported that the concomitant Cox‐Maze procedure was associated with higher freedom from AF at 12‐month follow‐up than PVI. Regarding AF recurrence, estimates pooled across the three RCTs indicated large heterogeneity and high uncertainty. In the largest and highest quality RCT, 12‐month AF recurrence was higher in the PVI arm (risk ratio = 1.58, 95% CI: 0.91–2.73). In two out of three higher‐quality observational studies, 12‐month AF recurrence was higher in PVI than in the Cox‐Maze arm (estimated adjusted probabilities 11% vs. 8% and 35% vs. 17%, respectively). RCTs demonstrated comparable 12‐month mortality between concomitant Cox‐Maze and PVI, while observational studies demonstrated the survival benefit of Cox‐Maze.
Conclusions
Concomitant Cox‐Maze in AF patients undergoing MV surgery is associated with better mid‐term freedom from AF when compared to PVI with comparable mid‐term survival. Large observational studies suggest that there might be a mid‐term survival benefit among patients after concomitant Cox‐Maze. Further large RCTs with longer standardized follow‐up are required to clarify the benefits of concomitant Cox‐Maze in AF patients during MV surgery.
Keywords: ablation surgery, atrial fibrillation, Cox‐Maze, mitral valve surgery, pulmonary vein isolation
1. INTRODUCTION
Up to 50% of patients undergoing mitral valve (MV) surgery experience preoperative atrial fibrillation (AF). 1 Incidence of stroke in patients with AF who are optimally anticoagulated remains between 2% and 5% per year, depending on the individual risk factors. 2 Surgical ablation during MV surgery has now been clearly shown to be associated with reduced 30‐day mortality and stroke and is recommended at the time of concomitant mitral operations to restore sinus rhythm. 1 , 3 , 4 , 5 , 6 While there have been many studies on concomitant surgical ablation in cardiac surgery, analysis of mid‐term or long‐term outcomes of simultaneous Cox‐Maze procedure or pulmonary vein isolation (PVI) during MV surgery is still lacking. 1 , 5 The performance rate of concomitant surgical ablation in patients with AF at the time of MV surgery has risen from 52% to 62% over the last decade. 7
The first maze procedure (Cox‐Maze I) was performed by James Cox on September 25, 1987. 8 The procedure evolved into the Cox‐Maze III and became the gold standard surgical treatment of AF. 3 , 9 In 2002, Damiano and associates have modified the Cox‐Maze III technique and replaced the majority of the incisions with a combination of bipolar radiofrequency and cryothermal ablation lines (AtriCure, Inc., Westchester, OH, USA) in a procedure termed the Cox‐Maze IV, which has shortened and simplified the operation and decreased morbidity and mortality. 10 The Cox‐Maze IV is currently the gold standard surgical treatment for AF, with estimated freedom from AF and from antiarrhythmic drugs at 1 year postoperatively of 93% and 85%, respectively. 11 However, concomitant PVI is still being used more frequently than the Cox‐Maze procedure as it is the simplest and most rapidly completed set of ablation lesions. 12 In addition, PVI can be performed without cardiopulmonary bypass (CPB) in coronary artery bypass graft surgery. 13
In the attempt to evaluate mid‐term efficacy and safety of concomitant Cox‐Maze relative to PVI in patients with AF undergoing MV surgery, we conducted a systematic review to critically evaluate randomized controlled trials (RCTs) and nonrandomized studies directly comparing the two procedures in this setting with additional subgroup meta‐analysis of RCTs.
2. PATIENTS AND METHODS
2.1. Study protocol
This systematic review was conducted in line with PRISMA (Preferred Reporting Items for Systematic Reviews and Meta‐Analyses) and AMSTAR (Assessing the methodological quality of systematic reviews) Guidelines. The study protocol was registered with the PROSPERO register of systematic reviews (PROSPERO ID: CRD42021250576). 14
2.2. Literature search and study selection
An initial scoping search and a systematic literature search were performed by a medical librarian to identify RCTs and observational studies that compared concomitant Cox‐Maze and PVI in patients who underwent MV surgery. Medline, EMBASE databases, and the Cochrane Library were searched from 1987 up to 15th of March 2022 using the following search terms: Maze surgery, Maze procedure, Maze technique, maze or Cox‐Maze versus PVI, ablation surgery, ablation procedure, and surgical ablation. Details of the search strategy are provided in the Supporting Information.
We defined 12‐month mortality and recurrence of AF postoperatively as outcomes of interest. We expected that a variety of outcomes illustrating efficacy/safety would be reported, but they were not reported consistently over the studies.
After deduplication, study eligibility was assessed independently by two investigators. Any discrepancies were resolved by discussion between the three investigators. The studies were selected through the following two levels of screening: in the first step studies were independently screened based on titles and abstracts, and in the second step, full‐text reports were evaluated based on predefined criteria. Studies were eligible if they compared outcomes in patients with AF undergoing MV surgery and concomitant Cox‐Maze procedure or PVI and met the following inclusion criteria:
-
1.
Population: adults or adolescents (12 years or older);
-
2.
Comparator: Cox‐Maze versus PVI;
-
3.
Provided outcomes: death, recurrence of AF at 12‐month follow‐up;
-
4.
Design: RCT or nonrandomized studies of interventions (NRSI) with at least 20 patients per treatment of interest;
-
5.
Published in English language.
Studies were ineligible if they had follow‐up shorter than 12 months and if they were duplicates. For studies reported in more than one publication, or when institutions reported subsequent studies with accumulating numbers of patients or increased lengths of follow‐up, only the most complete reports (in terms of reported outcomes and control of confounding) were included.
2.3. Risk of bias—study quality assessment
Risk of bias in individual studies was assessed independently by two investigators. Discrepancies were resolved through discussion between three investigators. RCTs were assessed using the revised Cochrane Collaboration Risk of Bias tool 2 (RoB2) for RCTs, 15 and NRSI were assessed using Cochrane Collaboration Risk of bias in nonrandomized studies of interventions (ROBINS‐I) tool (see Supporting Information for details). 16
2.4. Data extraction and outcomes
We assessed and extracted data on study characteristics, patients' baseline data and data regarding study outcomes independently by two investigators with verification for accuracy by two other investigators. The investigators looked for information on the sources of funding for individual studies included in the review, but it was not required to be reported. Digitizing software was used to recover graphically presented data. For RCTs, we extracted n/N data. For NRSI, we intended to extract adjusted effect measures; however, out of the three identified studies, one provided no relevant quantitative data, while the other two reported adjusted proportions (estimated probabilities from multivariate models). Where necessary, study authors were contacted to obtain additional information.
The primary outcomes were recurrence of AF and mortality after a 12‐month follow‐up. The secondary outcomes included aortic cross‐clamp time (XCT), CPB time, rate of MV repair, and duration of preoperative AF.
2.5. Subgroup meta‐analysis
We intended to generate random‐effects pooled effect estimates, separately for RCTs and NRSIs, but only three RCTs and two NRSIs with numerical data on primary outcomes were identified. Therefore: (i) we generated frequentist (Mantel–Haenszel relative risk, Paul‐Mandel for τ 2, Hartung‐Knapp adjustment) and Bayesian (vaguely informative prior for ln(RR) [mean = 0, SD = 4, half‐normal for τ]) random‐effects pooled estimates and prediction intervals specifically to illustrate uncertainty (CI width) and heterogeneity of the RCT outcomes (width of prediction intervals); (ii) adjusted proportions retrieved from two NRSIs were used to calculate individual study risk ratios (PVI/Cox‐Maze) by the Miettinen‐Nurminen method for more intuitive presentation. 17 We used package meta in R 18 for the frequentist and package bayesmeta for Bayesian meta‐analysis in R. 19 , 20
3. RESULTS
3.1. Identification of studies
A total of 409 studies were identified from the literature search, 7 of which reporting on 3 RCTs and 3 NRSIs 21 , 22 , 23 , 24 , 25 , 26 that met the eligibility criteria and were included in the present systematic review. The full PRISMA flow diagram outlining the study selection process is available in Figure 1.
Figure 1.
PRISMA (Preferred Reporting Items for Systematic Reviews and Meta‐analyses) flow diagram. RCT, randomized controlled trial.
3.2. Study characteristics
Details of individual studies and patients' baseline characteristics including the history of AF, intraoperative data and mid‐term postoperative outcomes are summarized in Table 1. Three out of six studies were RCTs. 21 , 22 , 23 The studies included a total of 790 patients (532 undergoing concomitant Cox‐Maze and 258 undergoing PVI during MV surgery). The proportion of patients who had preoperative longstanding persistent AF was reported in five out of six studies, while Gillinov et al. 26 reported the duration of preoperative AF. The proportion of longstanding persistent AF was similar in both compared subgroups in most studies. The performance rate of concomitant left atrial appendage occlusion was reported in five/six studies 21 , 22 , 24 , 25 , 26 and was 100% in all subgroups apart from the PVI subgroup in one study with the rate of 70%. 24 The rate of MV repair was reported in five/six studies and was comparable between the subgroups. 24 All included studies reported concomitant Cox‐Maze III or IV procedures performed in a standardized fashion. XCT and CPB time across the studies are demonstrated in Table 1.
Table 1.
Details of studies included in a systematic review
| Characteristics | Blackstone et al. 21 | Albrecht et al. 22 | Srivastava et al. 23 | Stulak et al. 24 | Onorati et al. 25 | Gillinov et al. 26 |
|---|---|---|---|---|---|---|
| Year | 2019 | 2009 | 2008 | 2014 | 2011 | 2006 |
| Country of origin | USA | Brazil | India | USA | Italy | USA |
| Type of study | RCT | RCT | RCT | NRSI | NRSI | NRSI |
| Study period | 2010–2013 | 1999–2004 | 2000–2005 | 1993–2011 | 2003–2008 | 1993–2004 |
| Follow‐up (median) (months) | 12 | 36 | 44 | 38 | 30 | 13.5 |
| No. of patients | ||||||
| Cox‐Maze | 66 | 20 | 40 | 256 | 109 | 41 |
| PVI | 67 | 20 | 40 | 68 | 32 | 31 |
| Age (mean± SD) (years) | ||||||
| Cox‐Maze | 68.2 ± 10.4 | 51.7 ± 12.4 | 37.1 ± 11.1 | 62 | 64 ± 9 | 64 ± 9.3 |
| PVI | 71.3 ± 10.3 | 55.1 ± 9.2 | 40.9 ± 11.4 | 68 | 65 ± 8 | 66 ± 12 |
| Female gender (%) | ||||||
| Cox‐Maze | 42.4 | 75 | 52.5 | NR | 28 | 51 |
| PVI | 43.3 | 70 | 50 | NR | 44 | 48 |
| Longstanding persistent AF (%) | ||||||
| Cox‐Maze | 54.6 | 100 | 100 | 55 | 100 | 54 ma |
| PVI | 50.8 | 100 | 100 | 19 | 100 | 3 ma |
| XCT (mean ± SD) (min) | ||||||
| Cox‐Maze | 107.4 ± 44 | 78.5 ± 15.9 | NR | 59 ± 5 | 74 ± 6 | NR |
| PVI | 98.4 ± 38.7 | 74.8 ± 19.2 | NR | 65 ± 6 | 73 ± 7 | NR |
| CPB time (mean ± SD) (min) | ||||||
| Cox‐Maze | 152.4 ± 60.8 | 123 ± 21 | NR | 103 ± 7 | 112 | NR |
| PVI | 143.3 ± 65.9 | 99.9 ± 23.8 | NR | 90 ± 8 | 110 | NR |
| Rate of MV repair (%) | ||||||
| Cox‐Maze | 56.1 | 55 | 2.5 | NR | 64 | 76 |
| PVI | 62.7 | 60 | 7.5 | NR | 72 | 68 |
| LAAO (%) | ||||||
| Cox‐Maze | 100 | 100 | NR | 100 | 100 | 100 |
| PVI | 100 | 100 | NR | 70 | 100 | 100 |
| Recurrence of AF at 12 m (%) | ||||||
| Cox‐Maze | 23 | 15 | 20 | 13 | 17 | 8 |
| PVI | 36 | 10 | 12.5 | 30 | 35 | 11 |
| Mortality at 12 m (%) | ||||||
| Cox‐Maze | 6.1 | 15 | 10 | 2 | 1.4 | 2 |
| PVI | 7.5 | 5 | 10 | 10 | 6.0 | 2 |
| Stroke at 12 m (%) | ||||||
| Cox‐Maze | 1.5 | NR | NR | 1 | NR | 2 |
| PVI | 4.5 | NR | NR | 1 | NR | 2 |
| Follow‐up monitoring protocol | Transtelephonic monitoring | ECG | ECG | ECG | 24 h Holter ECG | ECG |
Note: Data are mean ± SD, count (%).
Abbreviations: AF, atrial fibrillation; CPB, cardiopulmonary bypass; ECG, electrocardiogram; LAAO, left atrial appendage occlusion; m, month; MV, mitral valve; NR, not reported; NRSI, nonrandomized study; PVI, pulmonary vein isolation; RCT, randomized controlled trial; XCT, cross‐clamp time.
Median.
3.3. Risk of bias
Bias assessment of RCTs is demonstrated in Table 2. There were some concerns about the risk of bias in the two single‐center trials 22 , 23 (allocation concealment, postoperative use of antiarrhythmics and anticoagulants), while the CTSN trial had a low risk of bias (Table 2). 13 , 21 With respect to baseline and intraoperative characteristics, PVI (total N = 127) and Cox‐Maze patients (total N = 126) appeared reasonably balanced within the individual trial. Bias assessment of NRSIs is demonstrated in Table 3. Out of three NRSIs, one had a serious risk of bias (Table 3). 24
Table 2.
Randomized controlled trials: Main characteristics and risk of bias assessment
| Srivastava et al., 23 India | Albrecht et al., 22 Brazil | CTSN 2015/2019, USA 13 , 21 | |
|---|---|---|---|
| General design | Single‐center, open‐label, enrollment 2000–2005, 5‐year follow‐up with 3‐month intervals; four treatment arms including Cox‐Maze IV and PVI. | Single‐center, open‐label, enrollment 1999–2004, 5‐year follow‐up with 6‐month intervals; three treatment arms including Cox‐Maze III and PVI. | Multicentric, assessor‐blind, enrollment 2010–2013, 12‐month follow‐up with 3‐month intervals; three treatment arms including Cox‐Maze IV and PVI. |
| Patients | Age 12–60 years, undergoing MV surgery, no active CAD, no antiarrhythmics, LA ≤ 6 cm. | Age 18–79, LVEF > 20%, undergoing MV surgery, no previous surgeries. | Age ≥ 18 years, undergoing MV surgery, no previous ablations, no thyroid disease, life expectancy > 1 year. |
| Postoperatively | If NSR not achieved: DC or amiodarone. | If NSR not achieved: DC or amiodarone; anticoagulation as per indication. | AAD first 3 months after surgery. Further ablation when needed. All patients anticoagulated over a 12‐month period. |
| Declared endpoints | Conversion to NSR, AF free survival at 1‐year follow‐up. | Not explicitly stated. | Primary: AF after 6 and 12 months (3‐day Holter ECG). Secondary: composite of death, stroke, HF hospitalization, NYHA class worsening, and mitral valve reintervention; mortality; need for rhythm‐related interventions. |
| Patient evaluation | Clinical symptoms, ECG, echo. | Clinical symptoms, ECG, echo, treadmill. | Transtelephonic monitoring. |
| Risk of bias | |||
| Randomization | Some concern. | Some concern. | Low risk. |
| Treatment deviations | Some concern. | Some concern. | Low risk. |
| Missing outcome data | Low risk. | Low risk. | Low risk. |
| Other RoB2 biasesa | Low risk. | Low risk. | Low risk. |
| Overall risk of bias | Some concerns. | Some concerns. | Low risk. |
Abbreviations: AAD, antiarrhythmic drugs; AF, atrial fibrillation; CAD, coronary artery disease; DC, direct cardioversion; ECG, electrocardiography; HF, heart failure; LA, left atrium; LVEF, left ventricular ejection fraction; NSR, normal sinus rhythm; NYHA, New Your Heart Association; OR, operating room.
Other RoB2 biases—measurement of outcomes; selection of reported results.
Table 3.
Observational (nonrandomized controlled) studies: Main characteristics and risk of bias assessment
| Gillinov et al., 26 USA | Onorati et al., 25 Italy | Stulak et al., 24 USA | |
|---|---|---|---|
| Declared objective | Efficacy of treatment for paroxysmal AF with concomitant MV surgery: Cox‐Maze III, PVI and PVI with connecting lesions. | Outcomes of “limited” (PVI) and “extensive” (Cox‐Maze IV) treatment for persistent AF with concomitant MV surgery. | Outcomes in patients with surgically treated persistent/paroxysmal AF undergoing MV surgery. |
| Outcomes | Prevalence of AF at 1, 3, 6, 12 months and yearly follow‐up. | Prevalence of AF at 3, 6, 12, 18, 24 months and mid‐term mortality/hospitalizations. | Freedom from AF at 1, 1–5, and >5‐year follow‐up. |
| General design | Retrospective analysis of a single‐center registry 1993–2004. Cox‐Maze III (n = 41), PVI (n = 31)/PVI + left atrial connection lesions (n = 80). Standardized follow‐up. | Prospective. Consecutive patients (2003–2008), concomitant PVI (n = 32) and Cox‐Maze IV (n = 105) during MV surgery. Standardized follow‐up, 24‐h ECG Holter. | Retrospective analysis of a single‐center registry 1993–2011. Cox‐Maze III (n = 256) and PVI (n = 68) with MV surgery as a subset of a larger cohort. |
| Risk of bias | |||
| Confounding | Low‐moderate risk. Adjustment: demographic characteristics, NYHA class, MV disease, AF duration, LV function, other cardiac comorbidity, concomitant procedures. Chrono bias and post‐baseline confounding (AAD/anticoagulant use) possible. | Low‐moderate risk. PS baseline covariates: demographic characteristics, AF duration, LV function, atrial size, habits, diabetes and other comorbidity, previous surgery. Sextiles of PS stratification + adjustment for AF at discharge. Chrono bias and post‐baseline confounding (AAD/anticoagulant use) possible. | Serious risk. Declared use of a multivariable model, but no data on adjustments at all, including the subset of interest: surgical treatment of AF + MV surgery. Chrono bias and post‐baseline confounding (AAD/anticoagulant use) possible. |
| Selection bias | Low risk. | Low risk. | Moderate risk. |
| Missing data bias | Low‐moderate risk. | Low risk. | Moderate risk. |
| Other biasesa | Low risk. | Low risk. | Moderate risk. |
| Overall risk of bias | Moderate risk. | Moderate risk. | Serious risk. |
Abbreviations: AAD, antiarrhythmic drugs; AF, atrial fibrillation; ECG, electrocardiography; LV, left ventricle; MV, mitral valve; NYHA, New York Heart Association; PS, propensity score; PVI, pulmonary vein isolation; RCT, randomized controlled trial.
Other ROBINS‐I biases: misclassification, deviation from intended treatments, measurement of outcomes, selection of reported results.
3.4. Mortality after 12‐month follow‐up
A lower 12‐month mortality was observed in several studies, although the difference was not statistically significant. Onorati et al. 25 reported higher adjusted 12‐month mortality among patients who underwent PVI (6.0%) as compared to Cox‐Maze (1.4%) (risk ratio = 4.29, 95% CI: 0.67–27.0).
Because of the lack of RCTs and a limited number of enrolled patients, a meta‐analysis of RCTs comparing patients who underwent concomitant Cox‐Maze and PVI during MV surgery resulted in a wide CI/CrI (two–threefold lower to two–threefold higher risk with PVI) (Figure 2A). Considering very wide prediction intervals, there was high uncertainty when comparing the 12‐month mortality between the patients who underwent concomitant Cox‐Maze and PVI.
Figure 2.
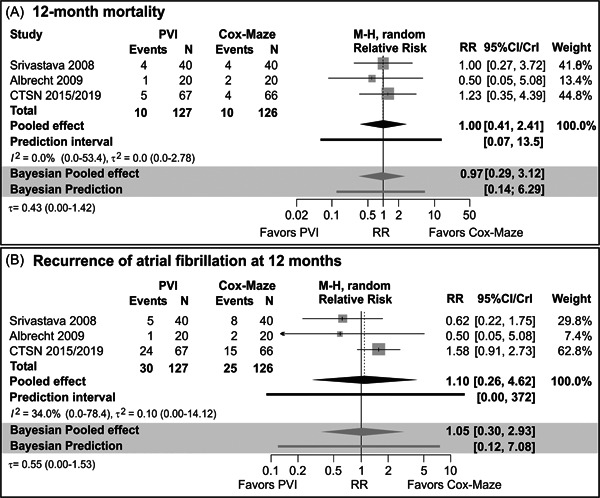
Forest plot showing the effect of PVI and Cox‐Maze procedure for AF during the mitral valve surgery on 12‐month mortality and recurrence of AF in randomized controlled trials. AF, atrial fibrillation; CI, confidence interval; M‐H, Mantel–Haenszel statistic; PVI, pulmonary vein isolation.
3.5. Recurrence of AF after 12‐month follow‐up
Two single‐center RCTs 22 , 23 indicated comparable results in AF recurrence during 12‐month follow‐up after the concomitant Cox‐Maze and PVI, while the CTSN trial clearly demonstrated a higher risk of AF recurrence after the concomitant PVI procedure during MV surgery (Table 1). 21 However, CI/CrI (from three to fourfold lower to three to fourfold higher risk after PVI) and prediction intervals around the pooled estimates were very wide (Figure 2B) illustrating uncertainty when comparing 12‐month recurrence of AF among patients who underwent Cox‐Maze and PVI. However, Srivastava et al. 23 excluded patients with enlarged left atrium (>6 cm) or if they received antiarrhythmic drugs preoperatively from their RCT, which are actually subsets of patients that would benefit most from the Cox‐Maze procedure.
Adjusted recurrence of AF at 12 months appeared comparable in a study by Gillinov et al. 18 (PVI 11% vs. Cox‐Maze 8%), although patients who underwent PVI had a significantly shorter duration of preoperative AF (Table 1). AF recurrence was significantly higher among patients who underwent PVI in the study by Onorati et al. 25 (adjusted estimates PVI 35% vs. Cox‐Maze 17%, risk ratio=2.13, 95%CI 1.10‐3.94) (Table 1). Among additional reported outcomes, the authors also demonstrated a higher adjusted recurrence of AF at 6 months, a higher adjusted proportion of antiarrhythmics at 12 months, and a higher adjusted risk of death or hospitalization among patients who underwent PVI. 25 Furthermore, in the study of 324 patients who underwent isolated MV surgery, Stulak et al. 24 reported a significantly higher recurrence of AF after a 12‐month follow‐up among patients who underwent concomitant PVI (30% vs. 13% Cox‐Maze).
4. DISCUSSION
The outcomes of concomitant Cox‐Maze and PVI during MV surgery have been extensively evaluated, but comparative data on the mortality and freedom from AF are still limited. In this systematic review, we investigated the comparison of mid‐term clinical outcomes between these two surgical ablation techniques. Across most studies with included AF patients undergoing MV surgery, the concomitant Cox‐Maze procedure was associated with higher freedom from AF at 12‐month follow‐up when compared with concomitant PVI. Second, our systematic review suggested that RCTs have demonstrated similar 12‐month mortality between concomitant Cox‐Maze and PVI, while observational studies have shown survival benefit of Cox‐Maze at 12‐month follow‐up.
Importantly, several long‐term studies demonstrated a survival benefit of AF ablation surgery. 27 Even more, risk‐adjusted analysis confirmed the safety of concomitant ablation surgery and found that the additional procedure is not associated with increased Society of Thoracic Surgeons morbidity or mortality. 1 , 28 Interestingly, Mehaffey et al. 29 reported recently that surgeons perform concomitant Cox‐Maze IV surgery among 27%–78% of patients depending on whether they reported barriers to implementation of the evidence‐based recommendation. However, a clinical practice guideline recommended that surgical ablation for symptomatic AF in the setting of left atrial enlargement (>4.5 cm) and more than moderate mitral regurgitation by PVI alone is not recommended (Class III no benefit, Level C expert opinion). 3
Although the association of concomitant Cox‐Maze and freedom from AF is evident, our results indicated that more RCTs with longer standardized follow‐up (at least 2 years) are required to clarify the benefits of concomitant Cox‐Maze in AF patients undergoing MV surgery. In addition, institutional experience is of paramount importance due to the fact that a center might have higher morbidity or early postoperative mortality while introducing the Cox‐Maze technique. Therefore, a clear advantage of this technique remains valid for centers with substantial experience in antiarrhythmic surgery. Furthermore, the reviewed data strongly suggest that both XCT and CPB time did not seem to be prolonged with concomitant Cox‐Maze procedure, 22 , 24 , 25 although they were not consistently reported across all the studies.
The benefit of concomitant Cox‐Maze in mitral patients with AF is in line with the results of other studies that have not only shown both freedom from AF and mortality benefit but have also demonstrated improvement in quality of life. 30 , 31 , 32 , 33 The reason for differences in comparison with PVI is that only full biatrial Cox‐Maze III or IV lesions can provide optimal effectiveness and superior outcomes with greater than 90% freedom from AF, particularly in patient with persistent AF. 24 , 26 Still, procedures with an incomplete set of lesions are in some reports incorrectly referred to as a Cox‐Maze procedure and may provide inconsistent results while this variability can induce relative confusion among surgical teams. However, our subgroup analysis with meta‐analysis of RCTs did not suggest that there is a significant survival benefit of concomitant Cox‐Maze procedure among RCTs. This can be explained by the fact that one of the RCTs was potentially underpowered and biased as many cases were excluded from randomization over the study period. 22 Furthermore, in the same RCT all patients with postoperative AF were intensely treated to restore the sinus rhythm. However, it has also been difficult to demonstrate a survival benefit in mitral patients after the concomitant Cox‐Maze procedure in other reports. 34 This may be related to a few reasons such as limited cohorts and short follow‐up study periods in RCTs. On the other hand, several observational studies with larger data sets have reported significant survival benefits. 24 , 25 However, given that AF has been clearly demonstrated to be an independent predictor of mortality, restoration of sinus rhythm is vital for quality of life and survival. 35
Previous studies have also reported that the Cox‐Maze procedure in AF patients undergoing concomitant cardiac surgery has a potential protective effect from stroke and thromboembolism in the long‐term period. 36 Unfortunately, these clinical outcomes along with other potential outcomes of interest (rehospitalization, permanent pacemaker implantation) were poorly reported in both RCTs and observational studies included in this systematic review. The available data from the studies included in our systematic review has demonstrated that early postoperative morbidity related to the risk of stroke was not increased with the performance of the concomitant Cox‐Maze procedure. 24 , 26 More importantly, would be to see the evidence in future trials whether restoration of sinus rhythm in these patients with concomitant Cox‐Maze procedure can improve long‐term survival and quality of life while reducing the risk of late stroke. However, despite this reported efficacy of Cox‐Maze, the widespread acceptance has been limited due to the technical complexity of the procedure, knowledge of effective lesion application, increased cross‐clamp time and its possible complications such as higher reported incidence of permanent pacemaker implantation. 36 While adding a full Cox‐Maze procedure did not affect the operative mortality of MV procedures with the improvements in ablation sources and shorter operative times, we believe that restricting the set of ablation lesions to PVI alone might be beneficial in some of the high‐risk patients although it requires further investigation.
Interestingly, we found concomitant Cox‐Maze to be associated with somewhat lower rates of MV repair. This can be partially explained by the technical complexity of this procedure and, therefore, the greater likelihood of the decision to proceed with valve replacement instead of repair when considering performing a more complex ablation procedure. However, for optimal outcomes, the surgeons should become more skilled in the Cox‐Maze technique through fellowship training, peer‐to‐peer education, or proctorship. 3
There are certain limitations to this systematic review. Although all the available literature has been examined, the quality of the studies included must be considered. Several eligible studies were retrospective cohort studies and only three RCTs comparing both concomitant ablation procedures were found. Most studies did not report the outcomes of long‐term mortality (>12 months), postoperative stroke or rehospitalization rate due to AF as well as other relevant clinical outcomes. However, sufficient studies were available to evaluate mid‐term outcomes between both subgroups and standardized Cox‐Maze lesion set patterns were used in all the studies. On the other hand, the available data was insufficient to allow a robust meta‐analysis as a primary goal of our study. It is also well established that there is no difference in outcomes between the cut‐and‐sew and a cryoablation/bipolar technique (AtriCure, Inc., Westchester, OH, USA) of the Cox‐Maze procedure. 37 , 38 In addition, some studies did not perform standardized follow‐up screening for outcome assessment, nor did they report antiarrhythmic and anticoagulation protocols, as recommended by current professional organizations. 39 , 40 , 41 In our opinion, each patient should receive a standardized postoperative treatment with amiodarone, if not contraindicated, for at least 6 weeks of duration. 42 , 43 The number of patients lost to follow‐up was not reported in all the studies, therefore the reported outcomes might not reflect the true outcomes within the studies. Future trials can be improved by adhering to this performance and reporting standards to better evaluate the effect of concomitant AF surgery. Therefore, we would like to emphasize that the standardized postoperative follow‐up protocol should include regular outpatient visits during the first 24 months and annually thereafter. 3 A 12‐lead electrocardiogram should be obtained at every follow‐up visit while a 24‐h Holter monitor at every follow‐up visit after 6 months, consistent with established guidelines. 39 , 44 Still, even with the follow‐up Holter monitoring, not all events may be captured. Long‐term outcome assessment may help evaluate whether the type of concomitant AF surgery influences mortality, neurological, or thromboembolic risk, which are the primary goals of AF treatment. Large high‐quality randomized trials evaluating the effect of different AF surgery types and lesion sets and comparing outcomes within different AF subgroups could provide guidance about which intervention has the most favorable efficacy and safety profile.
5. CONCLUSIONS
This systematic review found that concomitant Cox‐Maze in AF patients undergoing MV surgery is associated with a strong tendency of better mid‐term freedom from AF when compared to PVI with comparable mid‐term postoperative survival outcomes. Large observational studies suggested that there might be a mid‐term survival benefit for patients undergoing concomitant Cox‐Maze procedure, although large multicenter RCT are still needed to assess the long‐term survival benefit of the Cox‐Maze procedure. Successful intraoperative ablation of AF improves outcomes and thus the ideal operation would correct both MV dysfunction and AF.
AUTHOR CONTRIBUTIONS
Davorin Sef: Conceptualization; formal analysis; investigation; methodology; validation; resources; writing—original draft; critical revision and approval of the article. Vladimir Trkulja and Shahzad G. Raja: Conceptualization; formal analysis; validation; resources; supervision; writing—reviewing and editing; critical revision and approval of the article. Joanne Hooper: Conceptualization; formal analysis; validation; resources. Marko I. Turina: Validation; supervision; writing—reviewing and editing; critical revision and approval of the article.
CONFLICT OF INTEREST
The authors declare no conflict of interest.
Supporting information
Supporting information.
ACKNOWLEDGMENTS
The authors would like to thank Prof. Dr. Vinay Badhwar (Department of Cardiovascular and Thoracic Surgery, West Virginia University, Morgantown, WV, USA) for providing his expert opinion and constructive feedback in improving the article.
Sef D, Trkulja V, Raja SG, Hooper J, Turina MI. Comparing mid‐term outcomes of Cox‐Maze procedure and pulmonary vein isolation for atrial fibrillation after concomitant mitral valve surgery: a systematic review. J Card Surg. 2022;37:3801‐3810. 10.1111/jocs.16888
REFERENCES
- 1. Badhwar V, Rankin JS, Ad N, et al. Surgical ablation of atrial fibrillation in the United States: trends and propensity matched outcomes. Ann Thorac Surg. 2017;104(2):493‐500. [DOI] [PubMed] [Google Scholar]
- 2. Stroke prevention in atrial fibrillation study. Final results. Circulation. 1991;84(2):527‐539. [DOI] [PubMed] [Google Scholar]
- 3. Badhwar V, Rankin JS, Damiano RJ, Jr. , et al. The Society of Thoracic Surgeons 2017 clinical practice guidelines for the surgical treatment of atrial fibrillation. Ann Thorac Surg. 2017;103(1):329‐341. [DOI] [PubMed] [Google Scholar]
- 4. Vogt PR, Brunner La Rocca HP, Candinas R, et al. Temporary loss of cardiac autonomic innervation after the maze procedure. Eur J Cardiothorac Surg. 1997;12(1):75‐81. [DOI] [PubMed] [Google Scholar]
- 5. Hassanabad AF, Jefferson HL, Shanmugam G, Kent WDT. Atrial fibrillation: current and emerging surgical strategies. J Card Surg. 2019;34(11):1305‐1320. [DOI] [PubMed] [Google Scholar]
- 6. Hornero F, Rodríguez I, Bueno M, et al. Surgical ablation of permanent atrial fibrillation by means of maze radiofrequency: mid‐term results. J Card Surg. 2004;19(5):383‐388. [DOI] [PubMed] [Google Scholar]
- 7. Calkins H, Hindricks G, Cappato R, et al. 2017 HRS/EHRA/ECAS/APHRS/SOLAECE expert consensus statement on catheter and surgical ablation of atrial fibrillation. Heart Rhythm. 2017;14(10):e275‐e444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Cox JL. The first maze procedure. J Thorac Cardiovasc Surg. 2011;141(5):1093‐1097. [DOI] [PubMed] [Google Scholar]
- 9. Yii M, Yap CH, Nixon I, Chao V. Modification of the Cox‐Maze III procedure using bipolar radiofrequency ablation. Heart Lung Circ. 2007;16(1):37‐49. [DOI] [PubMed] [Google Scholar]
- 10. Robertson JO, Saint LL, Leidenfrost JE, Damiano RJ, Jr. Illustrated techniques for performing the Cox‐Maze IV procedure through a right mini‐thoracotomy. Ann Cardiothorac Surg. 2014;3(1):105‐116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Henn MC, Lancaster TS, Miller JR, et al. Late outcomes after the Cox maze IV procedure for atrial fibrillation. J Thorac Cardiovasc Surg. 2015;150(5):1168‐1176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Ad N, Suri RM, Gammie JS, Sheng S, O'Brien SM, Henry L. Surgical ablation of atrial fibrillation trends and outcomes in North America. J Thorac Cardiovasc Surg. 2012;144(5):1051‐1060. [DOI] [PubMed] [Google Scholar]
- 13. Gillinov AM, Gelijns AC, Parides MK, et al. Surgical ablation of atrial fibrillation during mitral‐valve surgery. N Engl J Med. 2015;372(15):1399‐1409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Stewart LA, Clarke M, Rovers M, et al. Preferred reporting items for a systematic review and meta‐analysis of individual participant data: the PRISMA‐IPD statement. JAMA. 2015;313(16):1657‐1665. [DOI] [PubMed] [Google Scholar]
- 15. Sterne JAC, Savović J, Page MJ, et al. RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ. 2019;366:l4898. [DOI] [PubMed] [Google Scholar]
- 16. Sterne JA, Hernán MA, Reeves BC, et al. ROBINS‐I: a tool for assessing risk of bias in non‐randomised studies of interventions. BMJ. 2016;355:i4919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Miettinen O, Nurminen M. Comparative analysis of two rates. Stat Med. 1985;4(2):213‐226. [DOI] [PubMed] [Google Scholar]
- 18. Balduzzi S, Rücker G, Schwarzer G. How to perform a meta‐analysis with R: a practical tutorial. Evid Based Ment Health. 2019;22(4):153‐160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Röver C. Bayesian random‐effects meta‐analysis using the bayesmeta R package. J Stat Softw. 2020;93(6):1‐51. [Google Scholar]
- 20. Schwarzer G. meta: an R package for meta‐analysis. R News. 2007;7(3):40‐45. [Google Scholar]
- 21. Blackstone EH, Chang HL, Rajeswaran J, et al. Biatrial maze procedure versus pulmonary vein isolation for atrial fibrillation during mitral valve surgery: new analytical approaches and end points. J Thorac Cardiovasc Surg. 2019;157(1):234‐243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Albrecht A, Kalil RA, Schuch L, et al. Randomized study of surgical isolation of the pulmonary veins for correction of permanent atrial fibrillation associated with mitral valve disease. J Thorac Cardiovasc Surg. 2009;138(2):454‐459. [DOI] [PubMed] [Google Scholar]
- 23. Srivastava V, Kumar S, Javali S, et al. Efficacy of three different ablative procedures to treat atrial fibrillation in patients with valvular heart disease: a randomised trial. Heart Lung Circ. 2008;17(3):232‐240. [DOI] [PubMed] [Google Scholar]
- 24. Stulak JM, Suri RM, Burkhart HM, et al. Surgical ablation for atrial fibrillation for two decades: are the results of new techniques equivalent to the Cox maze III procedure? J Thorac Cardiovasc Surg. 2014;147(5):1478‐1486. [DOI] [PubMed] [Google Scholar]
- 25. Onorati F, Mariscalco G, Rubino AS, et al. Impact of lesion sets on mid‐term results of surgical ablation procedure for atrial fibrillation. J Am Coll Cardiol. 2011;57(8):931‐940. [DOI] [PubMed] [Google Scholar]
- 26. Gillinov AM, Bakaeen F, McCarthy PM, et al. Surgery for paroxysmal atrial fibrillation in the setting of mitral valve disease: a role for pulmonary vein isolation? Ann Thorac Surg. 2006;81(1):19‐26. [DOI] [PubMed] [Google Scholar]
- 27. Iribarne A, DiScipio AW, McCullough JN, et al. Surgical atrial fibrillation ablation improves long‐term survival: a multicenter analysis. Ann Thorac Surg. 2019;107(1):135‐142. [DOI] [PubMed] [Google Scholar]
- 28. Rankin JS, Grau‐Sepulveda MV, Ad N, et al. Associations between surgical ablation and operative mortality after mitral valve procedures. Ann Thorac Surg. 2018;105(6):1790‐1796. [DOI] [PubMed] [Google Scholar]
- 29. Mehaffey JH, Charles EJ, Berens M, et al. Barriers to atrial fibrillation ablation during mitral valve surgery. J Thorac Cardiovasc Surg . Published online March 17, 2021. 10.1016/j.jtcvs.2021.03.039 [DOI] [PMC free article] [PubMed]
- 30. Ad N, Cheng DCH, Martin J, et al. Surgical ablation for atrial fibrillation in cardiac surgery: a consensus statement of the International Society of Minimally Invasive Cardiothoracic Surgery (ISMICS) 2009. Innovations (Phila). 2010;5(2):74‐83. [DOI] [PubMed] [Google Scholar]
- 31. Joshibayev S, Bolatbekov B. Early and long‐term outcomes and quality of life after concomitant mitral valve surgery, left atrial size reduction, and radiofrequency surgical ablation of atrial fibrillation. Anatol J Cardiol. 2016;16(10):797‐803. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Melo J, Santiago T, Aguiar C, et al. Surgery for atrial fibrillation in patients with mitral valve disease: results at five years from the international registry of atrial fibrillation surgery. J Thorac Cardiovasc Surg. 2008;135(4):863‐869. [DOI] [PubMed] [Google Scholar]
- 33. Lonnerholm S, Blomstrom P, Nilsson L, Oxelbark S, Jideus L, Blomstrom‐Lundqvist C. Effects of the maze operation on health‐related quality of life in patients with atrial fibrillation. Circulation. 2000;101(22):2607‐2611. [DOI] [PubMed] [Google Scholar]
- 34. Saint LL, Damiano RJ, Cuculich PS, et al. Incremental risk of the Cox‐maze IV procedure for patients with atrial fibrillation undergoing mitral valve surgery. J Thorac Cardiovasc Surg. 2013;146(5):1072‐1077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Glotzer TV, Hellkamp AS, Zimmerman J, et al. Atrial high rate episodes detected by pacemaker diagnostics predict death and stroke: report of the atrial diagnostics ancillary study of the MOde Selection Trial (MOST). Circulation. 2003;107(12):1614‐1619. [DOI] [PubMed] [Google Scholar]
- 36. Prasad SM, Maniar HS, Camillo CJ, et al. The Cox maze III procedure for atrial fibrillation: long‐term efficacy in patients undergoing lone versus concomitant procedures. J Thorac Cardiovasc Surg. 2003;126(6):1822‐1828. [DOI] [PubMed] [Google Scholar]
- 37. Aranda‐Michel E, Serna‐Gallegos D, Kilic A, et al. The impact of the Cox‐Maze technique on freedom from atrial fibrillation. Ann Thorac Surg. 2021;112(5):1417‐1423. [DOI] [PubMed] [Google Scholar]
- 38. García‐Villarreal OA. The Cox‐Maze procedure: accuracy and completeness do matter. Ann Thorac Surg. 2022;113(3):1050. [DOI] [PubMed] [Google Scholar]
- 39. Calkins H, Kuck KH, Cappato R, et al. 2012HRS/EHRA/ECAS expert consensus statement on catheter and surgical ablation of atrial fibrillation: recommendations for patient selection, procedural techniques, patient management and follow‐up, definitions, endpoints, and research trial design: a report of the Heart Rhythm Society (HRS) task force on catheter and surgical ablation of atrial fibrillation developed in partnership with the European Heart Rhythm Association (EHRA), a registered branch of the European Society of Cardiology (ESC) and the European Cardiac Arrhythmia Society (ECAS); and in collaboration with the American College of Cardiology (ACC), American Heart Association (AHA), the Asia Pacific Heart Rhythm Society (APHRS), and the Society of Thoracic Surgeons (STS) endorsed by the governing bodies of the American College of Cardiology Foundation, the American Heart Association, the European Cardiac Arrhythmia Society, the European Heart Rhythm Association, the Society of Thoracic Surgeons, the Asia Pacific Heart Rhythm Society, and the Heart Rhythm Society. Heart Rhythm. 2012;9(94):632‐696. [DOI] [PubMed] [Google Scholar]
- 40. Papadimas E, Tan YK, Choong A, Kofidis T, Teoh KLK. Anticoagulation after isolated mitral valve repair: a systematic review and meta‐analysis of clinical outcomes. Heart Lung Circ. 2021;30(2):247‐253. [DOI] [PubMed] [Google Scholar]
- 41. Lauritzen DJ, Vodstrup HJ, Christensen TD, Hald MO, Christensen R, Heiberg J. Discontinuation of anticoagulants after successful surgical ablation of atrial fibrillation. J Card Surg. 2020;35(9):2216‐2223. [DOI] [PubMed] [Google Scholar]
- 42. Babokin V, Trofimov N. Prevention of atrial fibrillation recurrence after the maze IV procedure. Ann Thorac Surg. 2020;109(5):1624‐1625. [DOI] [PubMed] [Google Scholar]
- 43. Lapenna E, De Bonis M, Giambuzzi I, et al. Long‐term outcomes of stand‐alone maze IV for persistent or long‐standing persistent atrial fibrillation. Ann Thorac Surg. 2020;109(1):124‐131. [DOI] [PubMed] [Google Scholar]
- 44. Murashita T, Rankin JS, Wei LM, Roberts HG, Alkhouli MA, Badhwar V. Oral anticoagulation may not be necessary for patients discharged in sinus rhythm after the Cox Maze IV procedure. J Thorac Cardiovasc Surg. 2018;155(3):997‐1006. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supporting information.


