Abstract
In aging societies, the number of older people with cognitive decline and dementia is increasing. Proper nutrition is a key factor that can help in the prevention of these age‐related impairments. This narrative review of longitudinal studies in Japan outlines the relationship between nutrition and the brain and focuses on Japanese nutritional epidemiologic studies. The results of Japanese cohort studies suggest that rather than a single food or nutrient, a nutritionally balanced diet that integrates a variety of foods and nutrients can contribute to the maintenance of brain function and be effective in preventing dementia. In addition, this review considers a nutritional approach to reducing the severity of cognitive decline in individuals with mild cognitive impairment and dementia, and suggests lifelong strategies for the prevention of cognitive decline. It is important that we consider the practice of eating not only as a way to obtain nutrients, but also as an important lifestyle habit to maintain our physical and mental health. Geriatr Gerontol Int 2022; 22: 825–831.
Keywords: cognitive dysfunction, dementia, diet, Japanese, lifestyle
Introduction
Daily eating is one of the most basic activities and is carried out several times a day throughout one's life. Daily eating not only provides the nutrients necessary for brain function but also stimulates the senses (such as sight, smell, and taste), provides physical and mental satisfaction, and enables us to communicate with others through food, for example by enjoying conversation and interaction with others. Regardless of age, ethnicity, or cognitive function, even if a person is unable to consume food orally and uses a gastric tube for feeding, they can still see and smell the food and connect with the surrounding world.
A variety of nutritional factors are thought to be involved in cognitive function. In Western studies, a number of dietary patterns such as the Mediterranean diet, the Dietary approaches to stop hypertention diet (DASH diet) and the Mediterranean‐DASH Intervention for Neurodegenerative Delay diet (MIND diet), have been shown to have dementia‐preventive effects. 1 , 2 However, the Japanese diet is unique among the world's diets, and a review of the evidence among the Japanese population is needed.
This narrative review of longitudinal studies in Japan discusses the relationship between nutrition and the brain, evidence from nutritional epidemiological studies in the Japanese population, and best diet for the lifelong maintenance of cognitive function.
Nutrition for the brain
Nutrition and the brain have a deep relationship. Our brain weighs approximately 1400 g. Although the brain accounts for only approximately 2% of our body weight, it consumes a large amount of glucose, equivalent to 20%–25% of our total daily energy intake (Figure 1). Nutrition not only is an important energy source for the brain, but also plays a role from metabolic and structural perspectives.
Figure 1.
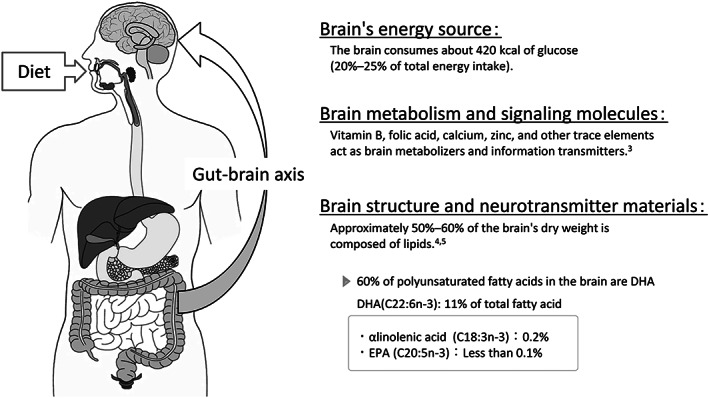
Nutrition for the brain. Nutrition and the brain have a deep relationship. Nutrition not only is an important energy source for the brain, but also plays a role from metabolic and structural perspectives.
For example, vitamin B, folic acid, calcium, zinc, and other trace elements are important for metabolism in the brain, such as in the regulation of neurotransmitters. 3 In terms of the brain structure, fatty acids, phospholipids and amino acids are important elements, and they act as neurotransmitters. 4 Approximately half of the dry weight of the brain is composed of lipids, 5 including large amounts of phospholipids, glycolipids and cholesterol, because the neurons and glial cells that constitute the brain have long and complex protrusions, and cell membrane components account for a large proportion of the brain's composition. Among the various fatty acids that constitute phospholipids, the brain contains large amounts of polyunsaturated fatty acids such as docosahexaenoic acid (DHA, C22:6n‐3) and arachidonic acid (ARA, C20:4n‐6). Approximately 60% of the polyunsaturated fatty acids within the brain are DHA, and the percentage of DHA is higher than that of other n‐3 fatty acids, such as alpha‐linolenic acid (0.2%) and eicosapentaenoic acid (EPA, C20:5n‐3) (less than 0.1%). The hippocampus, which is involved in memory, is reported to contain considerably more DHA and ARA than white matter. 6 DHA and ARA, which are also known to be essential for nerve development, are present in large amounts in breast milk and are added to infant formulas. Infants who suffer from a lack of DHA and ARA subsequently show signs of immature neural development compared with infants who do not. Reports have shown that infants who lack DHA and ARA have reduced information‐processing capability. 7 In fact, patients with Alzheimer's disease reportedly have lower concentrations of DHA in the brain than healthy individuals. 6
The blood–brain barrier regulates the entry of substances into the brain by restricting the entry of some substances and allowing the entry of others. Although a limited number of small molecules, such as sugars, amino acids and vitamins, can pass through the blood–brain barrier, large biomolecules, such as lipoproteins and peptides, can be transported into the brain only selectively by receptor‐mediated transport. Receptors for various nutrients are present in the blood–brain barrier, and their regulatory mechanisms are being studied. 8
In addition, the gut–brain axis is the relationship between the gut and the brain mediated by the immune system, endocrine system and enteric nervous system after the digestion and absorption of food. 4 Diet significantly modifies the composition and function of the host's gut microbiota. At the same time, however, the gut microbiota determines what the host can extract from their diet, including nutrients contributing to bioactive signaling molecules such as neurometabolites, vitamins and short‐chain fatty acids. 9 Many of these molecules, such as serotonin and gamma‐aminobutyric acid, have neuroactive functions because they can regulate nerve signals in the enteric nervous system, thereby affecting brain function and behavior. 10 Large macronutrient alterations, which can result from following Western‐style diets, have been shown to induce microbial dysbiosis, which has been linked to impaired cognition. 11
Gómez‐Pinilla conducted a review of various nutrients such as omega‐3 fatty acid, saturated fatty acid, curcumin, flavonoids, vitamins B, D and E, choline, calcium, zinc, selenium, copper and iron, which are thought to affect cognitive function. 4 Hence, it is thought that our daily diet is closely related to the composition and function of the brain.
Findings of overseas nutritional epidemiological studies
Dementia onset has been linked to several factors, including genetic and lifestyle habits and factors such as decreased physical activity, depression, diabetes and obesity in middle age. 12 Moreover, the maintenance of good lifestyle habits may reduce the risk of cognitive decline even when genetic risk is high. 13 Thus, the maintenance of a healthy diet and good exercise habits is required for the prevention of dementia.
A pro‐oxidant, pro‐inflammatory state is characteristic of aging and age‐related degenerative diseases, and it results in harmful damage to cellular components. 14 Therefore, foods and nutrients that have an anti‐inflammatory effect or antioxidants may be effective in the prevention of dementia onset. This possibility is based on the fact that patients with Alzheimer's disease show increased levels of oxidative modification products in their brains and cerebrospinal fluid, and patients with central nervous system disease show neuroinflammation in the brain. 15 , 16 However, randomized comparative trials in which vitamins E 17 and C were administered, 18 as well as a meta‐analysis that examined the neuropsychological benefit of n‐3 polyunsaturated fatty acids in randomized double‐blind placebo‐controlled studies, found that these substances did not significantly improve cognitive function. 19 Thus, there is no strong evidence to support the use of nutritional supplements or the implementation of dietary interventions to improve cognitive function.
The main reasons why interventional studies have failed to produce clear evidence are as follows. (i) Our daily diet fluctuates greatly. As a result, there is a great variation in nutrient intake, even for the same person, which makes it difficult to identify the effect of any specific nutrient on cognitive function. (ii) While the implementation of nutritional supplements and dietary interventions may be effective for undernourished individuals, the intervention effect may be small in well‐nourished individuals because they already have sufficient nutrients to maintain cognitive function. (iii) Because cognitive function gradually declines over time, it is difficult to assess changes in cognitive function that are attributable to a long‐term nutritional intervention. (iv) Considering that there are a wide variety of confounding factors in the relationship between diet and cognitive function, it is impossible to control all these factors in a study design. Therefore, when using a study design that requires human intervention, it is difficult to observe the causal relationship between diet and cognitive function change or the causal relationship with respect to the onset of dementia. However, this does not mean that dietary interventions are ineffective.
In previous longitudinal observational studies, nutrients and dietary factors considered effective in the prevention of dementia included fish‐derived n‐3 polyunsaturated fatty acids, DHA, eicosatetraenoic acid, folic acid, vitamin E, and antioxidants such as polyphenols and carotenoids. 20 These nutrients are found in large quantities in fish and shellfish, green and yellow vegetables, and fruit. Several studies conducted outside of Japan have reported that the so‐called “Mediterranean diet,” which contains an abundance of these foods, is effective in preventing cognitive decline. 21 In addition to the Mediterranean diet, the DASH diet, 22 developed to prevent hypertension, and the MIND diet, 23 developed based on the Mediterranean and DASH diets, have also been shown to prevent dementia (Table 1). The MIND diet was developed primarily for dementia prevention and is considered to have strong dementia‐preventive effects. 1
Table 1.
The three representative dietary patterns for dementia prevention overseas
| Dietary patterns | DASH diet (Dietary approaches to stop hypertension diet) 22 | Mediterranean diet 21 | MIND diet (Mediterranean‐DASH intervention for neurodegenerative delay diet) 23 |
|---|---|---|---|
| Development | Developed by the National Institutes of Health (NIH) to prevent hypertension | Traditional food eaten by people in Mediterranean countries | Based on the Mediterranean diet and the DASH diet, developed to prevent dementia |
| Characteristics |
Recommended: Low‐fat dairy products Restricted: Salt and alcohol |
Recommended: Olive oil and red wine, whole grains and other unrefined grains, and fish Restricted: Dairy products and meat |
Recommended: Whole grains, vegetables, berries, and nuts Restricted: Red meat, butter, cheese, and confectionery |
Findings of Japanese nutritional epidemiological studies
Compared with the diets of people worldwide, the Japanese diet contains large amounts of fish and shellfish. Although the amount of fat that Japanese people consume has increased in recent years, the amount of fat in the Japanese diet remains low compared with that in other diets worldwide. 24 , 25 The Japanese diet has a unique pattern, with rice as the staple food, miso‐soup and fish or meat as the main dish, and vegetables as the main side dish. Healthy diets include Mediterranean, DASH, and MIND diets, which are considered to be more effective in preventing dementia than the traditional “Western diet” (consisting of large amounts of red meat and fats and small amounts of vegetables and fish). 1 Whether the research findings obtained in a non‐Japanese population or in a non‐Japanese context are applicable to Japanese people remains unknown. The preferred diet for the prevention of dementia in Japan remains unclear.
Therefore, it is necessary to obtain evidence from nutritional epidemiological research in the Japanese population and to confirm the reproducibility of these findings to elucidate the nutritional protective factors that prevent cognitive decline in Japan.
To discuss the nutritional epidemiological findings on diet and cognitive function among Japanese population, this narrative review was conducted using the following search terms in PubMed (National Center for Biotechnology Information [NCBI] at the US National Library of Medicine [NLM]).
Search terms: ((Japanese) AND (((dementia[Title/Abstract]) OR (cognitive function[Title/Abstract])) AND (((dietary pattern[Title/Abstract]) OR (diet[Title/Abstract])) OR (nutrition[Title/Abstract])))) NOT (cross sectional) Filters: Full text, Published until February 19, 2022.
The original database search yielded 100 records. After selecting Japanese cohort studies, and excluding pilot or cross‐sectional studies and studies that focused on a single nutrient or food, the four articles shown in Table 2 met the full inclusion criteria. 26 , 27 , 28 , 29 Three cohort studies were selected because two of the four studies were from the same cohort study.
Table 2.
Representative Japanese longitudinal epidemiological studies on the association between dietary patterns and cognitive decline/dementia
| Cohort | Reference | Subjects for the analyses | Dietary assessment | Cognitive assessment | Main statistical analyses | Main results |
|---|---|---|---|---|---|---|
| Baseline age Number Follow‐up years | Method Dietary pattern | Criteria | Covariates | |||
| Hisayama Study | Ozawa M, et al.(2013) 27 |
60–79 years (mean age 68 years) n = 1006 15 years (median) |
70‐item semi‐quantitative FFQ Seven dietary patterns |
Diagnoses of vascular dementia (VaD), Alzheimer's disease (AD) | Age, sex, education, diabetes, hypertension, total cholesterol, history of stroke, BMI, smoking habits, exercise, and energy intake. | One of the seven dietary patterns with high intakes of soybeans and soybean products, vegetables, algae, and milk and dairy products and a low intake of rice is associated with reduced risk of dementia (HRs (95% CIs) for quartile 1 [ref.] vs. quartile 4 of all‐cause dementia, AD, and VaD: 0.66 (0.46, 0.95), 0.65 (0.40, 1.06), and 0.45 (0.22, 0.91), respectively. |
| Ohsaki Cohort Study | Tomata Y, et al. (2016) 28 |
≥65 years (73.8 ± 5.9 years [mean ± SD]) n = 14 402 4.9 ± 1.5 years (mean ± SD) |
39‐item FFQ Three dietary patterns |
Incident dementia based on Long‐term Care Insurance (LTCI) information | Age, sex, history of stroke, myocardial infarction, hypertension, arthritis, osteoporosis, and fracture, education level, smoking, alcohol drinking, BMI, psychological distress score, time spent walking, motor function score, cognitive function score, number of remaining teeth, and energy intake and protein intake. | One of the three dietary patterns with high intakes of fish, vegetables, mushrooms, potato, seaweeds, pickles, soybean, and fruits was inversely associated with the incident risk of dementia (HRs [95% CIs] for quartile 1 [ref.] vs. quartile 4: 0.80 [0.66–0.97], p‐trend = 0.016). |
| Lu Y, et al. (2021) 29 |
≥65 years (74.0 ± 5.4 years [mean ± SD]) n = 3146 5.0 ± 1.4 years (mean ± SD) |
39‐item FFQ Changes in the 8‐item Japanese Diet Index (JDI8) score (from 1994 to 2006) (JDI8: high intakes of rice, miso soup, seaweeds, pickles, green and yellow vegetables, fish, and green tea; low intakes of beef and pork) |
Incident dementia based on Long‐term Care Insurance (LTCI) information | Sex, age, education level, BMI, smoking status, alcohol drinking status, time spent walking, daily energy intake, history of stroke, hypertension, myocardial infarction, and diabetes, the JDI8 score, and psychological distress. | Compared with no changes in the JDI8 score, the multivariable‐adjusted HRs (95% CIs) were 1.72 (1.13, 2.62) for great decrease, 1.10 (0.73, 1.66) for moderate decrease, 0.82 (0.54, 1.25) for moderate increase, and 0.62 (0.38, 1.02) for great increase (p‐trend <0.0001). | |
| National Institute for Longevity Sciences ‐ Longitudinal Study of Aging (NILS‐LSA) | Otsuka R, et al. (2016) 26 |
60–81 years old n = 570 8.1 ± 2.9 years (mean ± SD) |
3‐day dietary record with photographs Dietary diversity score |
Mini‐Mental State Examination (MMSE) ≤ 27 | Sex, age, follow‐up time, MMSE score at baseline, education, BMI, household annual income, smoking, energy intake, and history of heart diseases | Multivariate adjusted OR for a decline in MMSE score was 0.79 (95% CI 0.70–0.89; p < 0.001) with a 1 SD increase in dietary diversity score. |
BMI, body mass index; CI, confidence interval; FFQ, food frequency questionnaire; HR, hazard ratio; JDI8, 8‐item Japanese diet index; LCTI, long‐term care insurance; MMSE, mini‐mental state examination; OR, odds ratio; SD, standard deviation.
Three representative Japanese longitudinal epidemiological studies revealed that older Japanese people “(i) who consumed a wide variety of foods (diverse food intake: National Institute for Longevity Sciences‐Longitudinal Study of Aging [NILS‐LSA] cohort) 26 ; (ii) who had high adherence to a dietary pattern that included high intakes of beans, soybean products, vegetables, seaweed, milk and dairy products and low intakes of rice (the Hisayama cohort) 27 ; and (iii) who followed a ‘Japanese’ dietary pattern consisting of high intakes of fish, vegetables, mushrooms, seaweed, pickles, soybean products, and green tea (the Ohsaki cohort) 28 , 29 ” would have a lower risk of cognitive decline or incident dementia. A recent study that conducted brain magnetic resonance imaging in individuals in the NILS‐LSA cohort also observed that high dietary diversity suppressed hippocampal volume loss over 2 years. 30
The results of these studies suggest that rather than a single food or nutrient, a nutritionally balanced diet that integrates a variety of foods and nutrients contributes to the maintenance of brain function and is effective in preventing dementia. A variety of nutrients are required for healthy brain development and brain function maintenance, yet it is almost impossible to determine to what degree each nutrient is required. However, as indicated in the Dietary Reference Intakes for Japanese and dietary balance guidelines, 31 from the current point of view, the consumption of a balanced diet that contains a variety of desirable nutrients is believed to be the best strategy to avoid the risk of undernutrition.
Prevention with a lifelong strategy
In young adulthood, it is rare for individuals to make dietary choices with future dementia prevention in mind. However, binge drinking and unbalanced nutrition in youth increase the risk of dementia in the future by increasing the risk of lifestyle‐related diseases such as diabetes, obesity and hypertension. Overnutrition, including obesity, is also known as a risk factor for dementia. 32
From middle age to old age, adherence to a well‐balanced diet that includes a variety of desirable nutrients can help in the prevention of cognitive decline. The “low‐sodium Japanese diet” 33 is considered a good diet from the perspective of the prevention of lifestyle‐related diseases, and a highly diverse diet with a moderate salt content will not only contribute to the prevention of lifestyle‐related diseases in young and middle age but also help in the prevention of dementia in old age. In old age, excessive salt intake should be avoided for the prevention of hypertension and stroke. However, excessive salt reduction may cause a decrease in appetite, which may lead to an increased risk of undernutrition owing to reduced food intake, and thus flexible salt reduction guidance is needed in old age. 31
From early to late old age, it is necessary to consciously prevent undernutrition. In particular, during the period when cognitive functions decline or mild cognitive impairment (MCI) is identified, food intake may decrease owing to the decline in physical functions and activities, and the probability of suffering from undernutrition may increase.
No exact cure for MCI exists, although it is believed that MCI treatment requires healthy lifestyle choices that take into account diet, physical activity, and sleep. 34 From a medical point of view, the recommended diet of patients with MCI and dementia should focus on improving undernutrition and nutritional deficiencies, but from a social and psychological perspective, the diet can be seen as a valuable opportunity to bring enjoyment and social interaction to people with cognitive decline by improving their mental wellbeing. 35
More older adults with MCI and early‐stage Alzheimer's disease than cognitively healthy participants were at risk for malnutrition, and nutritional problems may be associated with individual behavioral psychiatric symptoms of dementia. 36 It is well known that nutritional status can deteriorate as a result of many factors, including but not limited to dysphagia, apraxia and spatial cognition impairment, a bad eating posture and concentration problems, preference changes, depression, and medication side effects. Older people with MCI also reportedly have higher rates of undernutrition and experience weight loss and dietary changes before the onset of dementia. 37 , 38 Therefore, the prevention of undernutrition is a top priority. Further, medical professionals and caregivers should realize that diet is also a way to build social connections, provide nutritional support for patients with MCI, and thereby improve their quality of life. 39 As patients' cognitive function declines, their daily life rhythm may easily be disrupted. Eating meals at a fixed time and in a familiar environment (at a dining table and with tableware and people) as much as possible can help patients with MCI to feel safe and is useful for helping them obtain a daily rhythm.
Eating disturbances appear early in the onset of dementia, 39 and as dementia progresses, eating behavior itself often becomes difficult. Changes in food preferences and eating habits differ by dementia type, 40 with semantic dementia causing changes in food preferences, followed by increased appetite and eating habits, other oral behaviors, and finally dysphagia. In frontal variant frontotemporal dementia, changes in eating habits and increased appetite are the first symptoms. In Alzheimer's disease, dysphagia develops relatively early, but the pattern is unclear. 41 The prevalence of dysphagia in older ages ranges from 7% to 22%, with a reported dramatic increase to 40% to 50% in older residents in long‐term care facilities. 42 Dehydration, malnutrition, weight loss, and aspiration pneumonia can occur as a result of dysphagia in older adults, neurologically ill patients, and patients with dementia. Caregivers could help their patients in many ways throughout the dementia disease process to optimize food intake, increase safety during meals, minimize the risk of aspiration, and enhance mealtimes. 43 , 44 , 45 Because dementia is mostly a progressive disease, the focus should extend from cure to care, and should include assistance and support, rather than emphasizing training and functional recovery. It is necessary to correct the posture to make it easier for the patient to eat, minimize distractions, and ensure that the patient can eat safely, depending on an individual patient's condition. In addition, during this period, the burden on caregivers is also expected to increase. Therefore, it is necessary to provide dietary support that considers not only the patient's condition and living environment but also the caregiver's own eating habits and living environment.
Summary: Daily diet for cognitive function
During the coronavirus disease 2019 pandemic, people with cognitive decline, those who lived alone, those who were frail, and especially older people who had two or more of the above conditions, were greatly affected, particularly in terms of dietary habits, and their nutritional status may have deteriorated. 46
Our lives are maintained through the nourishment obtained from the food in our daily diets. Although the level of our cognitive and physical functions may vary, it is important that we consider the practice of eating not only as a way to obtain nutrients, but also as an important lifestyle habit to maintain our physical and mental health. Even if it is difficult to pay attention to every meal, a good strategy to prevent cognitive decline and dementia is to take small steps to improve and support people and ourselves in our daily situations.
Disclosure Statement
The author declares no conflict of interest.
Acknowledgements
The author is grateful to Shu Zhang, PhD, National Center for Geriatrics and Gerontology, for reviewing the manuscript. This work was supported in part by the Japanese Ministry of Education, Culture, Sports, Science and Technology (grant number 22H00497) and by Research Funding for Longevity Sciences from the National Center for Geriatrics and Gerontology, Japan (grant number 21‐18), a Health Science and Labor Research Grant, Japan (grant number 21GB0301), and the Japan Agency for Medical Research and Development (AMED) (grant number 22de0107002h0004).
Otsuka R. Diet, nutrition, and cognitive function: A narrative review of Japanese longitudinal studies. Geriatr. Gerontol. Int. 2022;22:825–831. 10.1111/ggi.14463
Data Availability Statement
This review does not deal with data, the papers reviewed are all published, and all readers could access there original papers.
References
- 1. Scarmeas N, Anastasiou CA, Yannakoulia M. Nutrition and prevention of cognitive impairment. Lancet Neurol 2018; 17: 1006–1015. [DOI] [PubMed] [Google Scholar]
- 2. Gardener SL, Rainey‐Smith SR. The role of nutrition in cognitive function and brain ageing in the elderly. Curr Nutr Rep 2018; 7: 139–149. [DOI] [PubMed] [Google Scholar]
- 3. Bourre JM. Effects of nutrients (in food) on the structure and function of the nervous system: update on dietary requirements for brain. Part 1: micronutrients. J Nutr Health Aging 2006; 10: 377–385. [PubMed] [Google Scholar]
- 4. Gómez‐Pinilla F. Brain foods: the effects of nutrients on brain function. Nat Rev Neurosci 2008; 9: 568–578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Hayashi K. The lipids of brain and nerve. J Jpn Oil Chem Soc 1971; 20: 745–754. [Google Scholar]
- 6. Söderberg M, Edlund C, Kristensson K, Dallner G. Fatty acid composition of brain phospholipids in aging and in Alzheimer's disease. Lipids 1991; 26: 421–425. [DOI] [PubMed] [Google Scholar]
- 7. Carlson SE, Colombo J. Docosahexaenoic acid and arachidonic acid nutrition in early development. Adv Pediatr 2016; 63: 453–471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Campos‐Bedolla P, Walter FR, Veszelka S, Deli MA. Role of the blood–brain barrier in the nutrition of the central nervous system. Arch Med Res 2014; 45: 610–638. [DOI] [PubMed] [Google Scholar]
- 9. Tan H, O'Toole PW. Impact of diet on the human intestinal microbiota. Curr Opin Food Sci 2015; 2: 71–77. [Google Scholar]
- 10. Wall R, Cryan JF, Ross RP, Fitzgerald GF, Dinan TG, Stanton C. Bacterial neuroactive compounds produced by psychobiotics. Adv Exp Med Biol 2014; 817: 221–239. [DOI] [PubMed] [Google Scholar]
- 11. Oriach CS, Robertson RC, Stanton C, Cryan JF, Dinan TG. Food for thought: the role of nutrition in the microbiota‐gut–brain axis. Clin Nutr Exp 2016; 6: 25–38. [Google Scholar]
- 12. Livingston G, Huntley J, Sommerlad A et al. Dementia prevention, intervention, and care: 2020 report of the lancet commission. Lancet 2020; 396: 413–446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Lourida I, Hannon E, Littlejohns TJ et al. Association of lifestyle and genetic risk with incidence of dementia. JAMA 2019; 322: 430–437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Dominguez LJ, Barbagallo M. Nutritional prevention of cognitive decline and dementia. Acta Biomed 2018; 89: 276–290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Szczechowiak K, Diniz BS, Leszek J. Diet and Alzheimer's dementia – nutritional approach to modulate inflammation. Pharmacol Biochem Behav 2019; 184: 172743. [DOI] [PubMed] [Google Scholar]
- 16. Ionescu‐Tucker A, Cotman CW. Emerging roles of oxidative stress in brain aging and Alzheimer's disease. Neurobiol Aging 2021; 107: 86–95. [DOI] [PubMed] [Google Scholar]
- 17. Farina N, Llewellyn D, Isaac MG, Tabet N. Vitamin E for Alzheimer's dementia and mild cognitive impairment. Cochrane Database Syst Rev 2017; 4: CD002854. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Galasko DR, Peskind E, Clark CM et al. Antioxidants for Alzheimer disease: a randomized clinical trial with cerebrospinal fluid biomarker measures. Arch Neurol 2012; 69: 836–841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Mazereeuw G, Lanctôt KL, Chau SA, Swardfager W, Herrmann N. Effects of omega‐3 fatty acids on cognitive performance: a meta‐analysis. Neurobiol Aging 2012; 33: e17–e29. [DOI] [PubMed] [Google Scholar]
- 20. Otaegui‐Arrazola A, Amiano P, Elbusto A, Urdaneta E, Martínez‐Lage P. Diet, cognition, and Alzheimer's disease: food for thought. Eur J Nutr 2014; 53: 1–23. [DOI] [PubMed] [Google Scholar]
- 21. Singh B, Parsaik AK, Mielke MM et al. Association of mediterranean diet with mild cognitive impairment and Alzheimer's disease: a systematic review and meta‐analysis. J Alzheimers Dis 2014; 39: 271–282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. National Heart, Lung, and Blood Institute . DASH eating plan. [Cited 13 April 2022]. Available from URL: https://www.nhlbi.nih.gov/education/dash-eating-plan.
- 23. Morris MC, Tangney CC, Wang Y et al. MIND diet slows cognitive decline with aging. Alzheimers Dement 2015; 11: 1015–1022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Tsugane S. Why has Japan become the world's most long‐lived country: insights from a food and nutrition perspective. Eur J Clin Nutr 2021; 75: 921–928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Zhou B, Stamler J, Dennis B et al. Nutrient intakes of middle‐aged men and women in China, Japan, United Kingdom, and United States in the late 1990s: the INTERMAP study. J Hum Hypertens 2003; 17: 623–630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Otsuka R, Nishita Y, Tange C et al. Dietary diversity decreases the risk of cognitive decline among Japanese older adults. Geriatr Gerontol Int 2017; 17: 937–944. [DOI] [PubMed] [Google Scholar]
- 27. Ozawa M, Ninomiya T, Ohara T et al. Dietary patterns and risk of dementia in an elderly Japanese population: the Hisayama study. Am J Clin Nutr 2013; 97: 1076–1082. [DOI] [PubMed] [Google Scholar]
- 28. Tomata Y, Sugiyama K, Kaiho Y et al. Dietary patterns and incident dementia in elderly Japanese: the Ohsaki cohort 2006 study. J Gerontol A Biol Sci Med Sci 2016; 71: 1322–1328. [DOI] [PubMed] [Google Scholar]
- 29. Lu Y, Sanae Matsuyama S, Sugawara Y, Sone T, Tsuji I. Changes in a specific dietary pattern and incident dementia: a prospective cohort study. Clin Nutr 2021; 40: 3495–3502. [DOI] [PubMed] [Google Scholar]
- 30. Otsuka R, Nishita Y, Nakamura A et al. Dietary diversity is associated with longitudinal changes in hippocampal volume among Japanese community dwellers. Eur J Clin Nutr 2021; 75: 946–953. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Japanese Ministry of Health, Labour and Welfare . Dietary Reference Intakes 2020. Tokyo (JP). [Cited 13 April 2022]. Available from URL: https://www.mhlw.go.jp/content/10900000/000862500.pdf (Simplified version in English), https://www.mhlw.go.jp/content/10904750/000586553.pdf (Full version in Japanese).
- 32. Dye L, Boyle NB, Champ C, Lawton C. The relationship between obesity and cognitive health and decline. Proc Nutr Soc 2017; 76: 443–454. [DOI] [PubMed] [Google Scholar]
- 33. Nakamura Y, Ueshima H, Okamura T et al. A Japanese diet and 19‐year mortality: national integrated project for prospective observation of non‐communicable diseases and its trends in the aged, 1980. Br J Nutr 2009; 101: 1696–1705. [DOI] [PubMed] [Google Scholar]
- 34. Eshkoor SA, Mun CY, Ng CK, Hamid TA. Mild cognitive impairment and its management in older people. Clin Interv Aging 2015; 10: 687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Manthorpe J, Watson R. Poorly served? Eating and dementia. J Adv Nurs 2003; 41: 162–169. [DOI] [PubMed] [Google Scholar]
- 36. Kimura A, Sugimoto T, Kitamori K et al. Malnutrition is associated with behavioral and psychiatric symptoms of dementia in older women with mild cognitive impairment and early‐stage Alzheimer's disease. Nutrients 2019; 11: 1951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Soto ME, Secher M, Gillette‐Guyonnet S et al. Weight loss and rapid cognitive decline in community‐dwelling patients with Alzheimer's disease. J Alzheimers Dis 2012; 28: 647–654. [DOI] [PubMed] [Google Scholar]
- 38. Fostinelli S, De Amicis R, Leone A et al. Eating behavior in aging and dementia: the need for a comprehensive assessment. Front Nutr 2020; 7: 604488. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Faraday J, Abley C, Beyer F, Exley C, Moynihan P, Patterson JM. How do we provide good mealtime care for people with dementia living in care homes? A systematic review of carer–resident interactions. Dementia 2021; 20: 3006–3031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Brodaty H, Connors MH, Xu J, Woodward M, Ames D, PRIME study group . The course of neuropsychiatric symptoms in dementia: a 3‐year longitudinal study. J Am Med Dir Assoc 2015; 16: 380–387. [DOI] [PubMed] [Google Scholar]
- 41. Kai K, Hashimoto M, Amano K, Tanaka H, Fukuhara R, Ikeda M. Relationship between eating disturbance and dementia severity in patients with Alzheimer's disease. PLoS One 2015; 10: e0133666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Ikeda M, Brown J, Holland AJ, Fukuhara R, Hodges JR. Changes in appetite, food preference, and eating habits in frontotemporal dementia and Alzheimer's disease. J Neurol Neurosurg Psychiatr 2002; 73: 371–376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Easterling CS, Robbins E. Dementia and dysphagia. Geriatr Nurs 2008; 29: 275–285. [DOI] [PubMed] [Google Scholar]
- 44. Affoo, R.H. Dysphagia care for individuals with dementia. 2014 [Cited 13 April 2022]. Available from https://dysphagiacafe.com/2014/10/01/dysphagia-care-for-individuals-with-dementia-oral-care-cognition/.
- 45. Egan A, Andrews C, Lowit A. Dysphagia and mealtime difficulties in dementia: speech and language therapists' practices and perspectives. Int J Lang Commun Disord 2020; 55: 777–792. [DOI] [PubMed] [Google Scholar]
- 46. Kinoshita K, Satake S, Arai H. Impact of frailty on dietary habits among community‐dwelling older persons during the COVID‐19 pandemic in Japan. J Frailty Aging 2022; 11: 109–114. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
This review does not deal with data, the papers reviewed are all published, and all readers could access there original papers.


