PREFACE
In 2020, the Japan Diabetes Society (JDS) adopted a sweeping decision to release consensus statements on relevant issues in diabetes management that require updating from time to time and launched a ‘JDS Committee on Consensus Statement Development’. These consensus statements are intended to present the committee's take on diabetes management in Japan based on evidence currently available for each of the issues addressed. It is thus hoped that practicing diabetologists will never fail to consult these statements to provide the best available practice in their respective clinical settings. Of note, while as many as one‐third of all people with diabetes in the world are shown to be concentrated in the Asian region, diabetes mellitus varies in its pathology, including the extent of obesity involved in affected patients, between the East and West. Again, given that timely consensus statements to come out of Japan are thus expected to have enormous implications for clinical practice, it is also planned to make each consensus statement available in English.
Following publication in 2020 of the committee's first consensus statement on ‘Medical Nutrition Therapy and Dietary Counseling for People with Diabetes’ (Tonyobyou 2020; 63: 91–109) in Japanese, the committee has taken this opportunity to propose a consensus ‘algorithm for pharmacotherapy in people with diabetes’, in this statement. There are three reasons that prompted the committee to address this issue. First, there are characteristic differences in diabetes pathology between Westerners and the Japanese, where individuals with insulin resistance account primarily for all people with diabetes among the former, while obese and non‐obese individuals account equally for all people with diabetes among the latter, with the severity of insulin insufficiency and insulin resistance varying from one individual to the next. Second, there are differences in treatment strategy adopted for diabetes between the West and Japan. Indeed, up until 2021, metformin was recommended as first‐line therapy for Western patients, with those shown to be effective against relevant comorbidities, e.g., atherosclerotic cardiovascular disease, renal dysfunction, and heart failure (HF), particularly recommended. In contrast, prompted by the results of the Kumamoto study (Diabetes Res Clin Pract. 1995; 28: 103–117) and the J‐DOIT3 study (Lancet Diabetes Endocrinol. 2017; 5: 951–964), multifactorial intervention including glycemic control has been recommended in Japanese people with diabetes to reduce diabetic complications, with the choice of one medication class over the others for each patient remaining an issue to be determined in light of his/her current condition. Third, an analysis of the National Database of Health Insurance Claims and Specific Health Check‐ups in Japan brought a disparity in initial antidiabetic medication prescribing patterns between Western countries and Japan (J Diabetes Investig. 2022; 13: 280–291), with the judicious use of biguanides in elderly patients likely to be widespread as per the JDS warning message, leading to the preferential use of DPP‐4 inhibitors over biguanides in these patients. Again, of note, none of the biguanides were shown to be used as medications of first choice in as many as 38.2% of all non‐JDS‐certified facilities, suggesting the need for the committee to develop a standard algorithm for diabetes pharmacotherapy.
In developing the algorithm for diabetes pharmacotherapy in patients with type 2 diabetes, the working concept was that priority should be given to selecting such medications as would appropriately address the diabetes pathology in each patient, while at the same time weighing the available evidence for these medications and the prescribing patterns in clinical practice in Japan. Specifically, the proposed algorithm would involve choosing medications according to diabetes pathology in each patient in step 1; ensuring their safety in step 2; weighing their additional benefits for comorbidities in step 3; and choosing medications with relevant patient background characteristics of interest in mind in step 4.
It is hoped that the algorithm presented here will not only contribute to improved diabetes management in Japan but continue to evolve into a better one over time, reflecting new evidence as it becomes available.
CHARACTERISTICS OF DIABETES IN THE JAPANESE AND ASIAN POPULATIONS
Type 2 diabetes mellitus is a metabolic disease in which insulin insufficiency or decreased insulin sensitivity (insulin resistance), combined with relative decreased insulin action to varying degrees, accounts for such a lack of insulin action as to cause chronic hyperglycemia 1 . Again, multiple genetic factors responsible for insulin insufficiency or insulin resistance and environmental factors (e.g., overeating or lack of physical activity and resultant obesity) combine to lead to such a lack of insulin action as to cause type 2 diabetes mellitus.
A comparison of the insulin‐secretory capacity and insulin resistance between Westerners and the Japanese as stratified by glucose tolerance shows that the Japanese have less insulin‐secretory capacity than the Westerners, even while their glucose tolerance is shown to be normal and that while Westerners exhibit acutely increased insulin resistance as they move from normal glucose tolerance to diabetes, Japanese tend to exhibit lower insulin‐secretory capacity than that usually associated with increased insulin resistance 2 , 3 . Again, a study comparing insulin sensitivity and initial insulin response between East Asians, Caucasians, and Blacks shows that these races vary in the balance between their insulin‐secretory capacity and insulin resistance and that East Asians and Blacks are more susceptible to diabetes than Caucasians 4 . Again, the pathology of type 2 diabetes mellitus in Japanese is also shown to be characterized as a decreased initial insulin response, regardless of the presence of obesity 5 . On the other hand, a recent study in Hisayama‐cho investigated the correlation between pancreatic β‐cell failure (i.e., low insulinogenic index/HOMA‐IR) or insulin resistance and the onset of type 2 diabetes mellitus and found that while pancreatic β‐cell failure and insulin resistance are both associated with the risk of type 2 diabetes mellitus, they are associated with a markedly increased risk of type 2 diabetes mellitus when they are found together in obese individuals 6 .
In addition, histological studies of the pancreas have shown that, among non‐diabetic Westerners, obese individuals have a significantly greater islet mass than non‐obese individuals and that, among Westerners with type 2 diabetes, both obese and non‐obese individuals have an islet mass about 50% lower than that in non‐diabetic individuals and that no increase in pancreatic β‐cell mass is noted even in obese Japanese 7 , 8 . Research also shows that, among individuals with type 2 diabetes, amyloid deposition is noted in more than 80% of Westerners but only in 30% of Japanese 9 , 10 . Thus, it is suggested that histological findings in the pancreas differ greatly between different races, suggesting that these differences may account in part for differences in their diabetes pathology.
Also of note, advances in genetic analysis of type 2 diabetes mellitus have also led to the identification of numerous type 2 diabetes mellitus susceptibility loci including KCNQ1 11 , 12 , 13 . A meta‐analysis of genome‐wide association studies (GWAS) in type 2 diabetes mellitus has recently shown that many Japanese individuals but very few Westerners, had the R131Q mutation in the GLP‐1 receptor gene (GLP‐1R), which is known to be involved in inducing a 2‐fold increase in insulin secretion. Furthermore, a cross‐racial molecular biological pathway analysis has shown that the pathways involved in the onset of maturity‐onset diabetes of the young (MODY) are most strongly associated with type 2 diabetes mellitus in both races evaluated and that the pathways involved in the regulation of insulin secretion are significantly associated with type 2 diabetes mellitus in Japanese alone 14 .
Thus, taken together, the pathology of type 2 diabetes mellitus clearly differs between Japanese and Westerners, not only functionally but histologically and genetically, and a decreased insulin‐secretory capacity has a greater role to play in the onset of type 2 diabetes mellitus in Japanese than in Westerners.
DIFFERENCES IN TREATMENT STRATEGY FOR JAPANESE AND WESTERN PATIENTS WITH TYPE 2 DIABETES MELLITUS
As detailed above, type 2 diabetes mellitus can be primarily characterized as having, as an underlying core pathology in most Japanese, insulin resistance and insulin insufficiency, whose respective contribution is shown to vary from individual to individual, in contrast to that in Westerners which can be characterized as having obesity and insulin resistance as a core pathology. For its ability to reduce the risk of microangiopathy, macroangiopathy and death, as well as for its beneficial impact on body weight, low hypoglycemia risk, and low cost 15 , 16 , metformin has long been recommended as first‐line therapy in Western countries 17 , 18 . However, the Standards of Medical Care in Diabetes by the American Diabetes Association (ADA) have been extensively revised in 2022 to address compelling issues in diabetes management, such as diabetic comorbidities (e.g., atherosclerotic cardiovascular disease), patient‐related factors in diabetes treatment, and therapeutic needs of affected individuals 19 . In contrast, the treatment strategy for type 2 diabetes mellitus in Japan is characterized as allowing for the choice of medications from all classes to address the diabetes pathology in each affected individual, while taking into account the extent of their metabolic derangement, but also on their age, extent of their obesity, status of their insulin secretion/insulin resistance, severity of their chronic complications, status of their liver/renal function 20 . The rationale for this approach has indeed been provided through the accumulation of relevant evidence, including that from the Kumamoto study 21 and the Japan Diabetes Outcome Interventional Trial 3 (J‐DOIT3) 22 , which corroborated the importance of multifactorial intervention, including glycemic control, in reducing complications in Japanese patients with diabetes.
INITIAL ANTIDIABETIC MEDICATION PRESCRIBING PATTERNS FOR PEOPLE WITH DIABETES IN JAPAN
It is not difficult to imagine how significantly such differences in treatment strategy for type 2 diabetes mellitus might impact on the choice of medications or their prescribing patterns. In this regard, while there are studies on antidiabetic medication prescribing patterns in Japan 23 , 24 , they each suffered from a small sample size and lack of data from elderly patients and a nationwide survey has been awaited to provide a full picture of the prescribing patterns in clinical practice. Thus, the Japan Diabetes Society (JDS) conducted a nationwide survey to clarify the prescribing patterns in clinical practice as a step to developing an algorithm for diabetes pharmacotherapy 25 . The survey demonstrated that, among the more than 1 million people with type 2 diabetes registered with the National Database of Health Insurance Claims and Specific Health Check‐ups from the latter half of the fiscal year 2014 to the fiscal year 2017, the most frequently prescribed of all antidiabetic medications was, unlike those in Western countries 26 , dipeptidyl peptidase‐4 (DPP‐4) inhibitors, followed by biguanides and sodium‐glucose cotransporter 2 (SGLT2) inhibitors, with age shown to the factor most strongly influencing this prescribing pattern; and that the older the patients were, the more likely they were to have been prescribed DPP‐4 inhibitors and the markedly less likely they were to have been prescribed biguanides and SGLT2 inhibitors. An analysis of the initial prescribing pattern by prefecture also showed that the biguanide and DPP‐4 inhibitor prescriptions varied from one prefecture to the next, while an analysis of the initial prescribing pattern by facility (JDS‐certified vs non‐JDS‐certified) showed that no patients receiving initial medication therapy had been initially prescribed biguanides at 38.2% of non‐JDS‐certified facilities and that the DPP‐4 inhibitor prescribing pattern varied greatly between JDS‐ certified and non‐JDS‐certified facilities (i.e., there were not a few non‐JDS‐ certified facilities where almost 100% of patients had been initially prescribed DPP‐4 inhibitors alone). Thus, while the survey results suggested that antidiabetic medications were being chosen to address the characteristics of diabetes in each individual patient and that the JDS recommendations on the use of metformin and SGLT2 inhibitors 27 , 28 were widely adhered to by primary care physicians, the disparity in DPP‐4 inhibitor and biguanide prescribing patterns between regions and facilities, nevertheless, pointed to the need to renew awareness of the JDS‐proposed principle of medication choice for each patient based not only on the extent of their metabolic derangement, but also on their age, extent of their obesity, severity of their chronic complications, status of their liver/renal function, and status of their insulin secretion/insulin resistance, thus a need to formulate an algorithm as a tool to promote the proper use of antidiabetic medications.
WORKING CONCEPT OF AN ALGORITHM FOR PHARMACOTHERAPY IN PATIENTS WITH TYPE 2 DIABETES MELLITUS
Given that type 2 diabetes mellitus differs in pathology between Asians including Japanese and Westerners, the Japan Diabetes Society has long advanced a different treatment strategy for Japanese from that for Westerners (Figure 1). By the same token, it became clear from the survey results that the initial diabetes medication prescribing patterns differ greatly between Japan and Western countries 25 , suggesting that the JDS‐proposed treatment strategy for diabetes has become widespread among diabetologists and general practitioners alike. It is also likely that the initial diabetes medication prescribing patterns reflected the informed use of antidiabetic medications, except imeglimin, on the part of many physicians, based on their glucose‐lowering efficacy and safety profiles that became known after a certain lapse of time since their approval. Furthermore, it became also clear that the disparity in the prescribing patterns of DPP‐4 inhibitors and biguanides between facilities and regions needed to be resolved to ensure the proper use of these medications. Of note, given that evidence has recently been accumulated, mostly overseas, that demonstrates the efficacy of GLP‐1 receptor agonists and SGLT2 inhibitors against diabetic comorbidities (i.e., atherosclerotic cardiovascular disease, heart failure and chronic kidney disease [CKD]), suggesting that these additional benefits (i.e., cardio/reno‐protective and mortality‐reducing effects) are worth considering in medication selection for patients with type 2 diabetes mellitus. Thus, overall, based on the basic concept that (1) medications can be selected to address the diabetes pathology in Japanese and Asians; (2) the medication selection should reflect the prescribing patterns in clinical practice in Japan; and (3) medications can be selected for their additional benefits in patients with comorbidities that call for medical attention, an algorithm for diabetes pharmacotherapy was developed to allow for such choice of medications as to address each patient's pathology/condition, with the priority in medication selection determined, with consideration also given to current prescribing patterns and other relevant factors.
Figure 1.
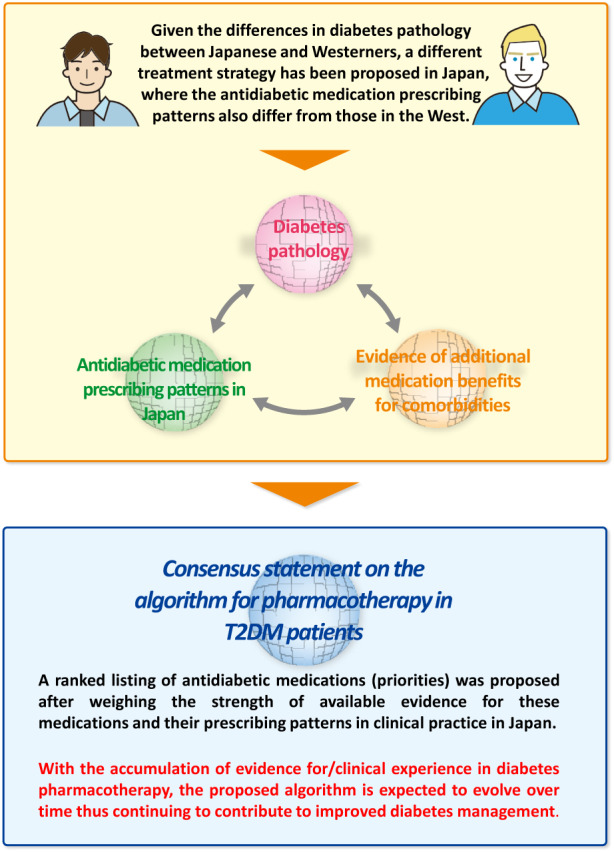
Working concept of an algorithm for pharmacotherapy in type 2 diabetes mellitus.
PROPOSED ALGORITHM ANNOTATED
Assessing the indications for insulin and determining the HbA1c control goal
The overriding premise behind diabetes pharmacotherapy was defined as ensuring safety (Figure 2). Thus, medication selection was first assumed to involve assessing whether there were any absolute or relative indications for insulin therapy in each patient. Then, as individuals 65 years old or older account for more than half of all people with diabetes in Japan, the HbA1c control goal was determined, based on those proposed in the Kumamoto Declaration 2013 and the JDS‐proposed ‘Glycemic targets (HbA1c values) for elderly patients with diabetes’ 20 , 29 . It was also assumed that while the HbA1c control goal of <7% remained valid for prevention of complications in people with diabetes, the HbA1c control goal could also be determined with consideration given to other factors such as their age or comorbidities.
Figure 2.

Proposed algorithm for pharmacotherapy in type 2 diabetes mellitus.
Assessing people with type 2 diabetes for the presence of obesity as a relevant measure (step 1)
While the insulinogenic index (II) or C‐peptide index remain useful as measures of insulin‐secretory capacity, as does Homeostatic Model Assessment – Insulin Resistance (HOMA‐IR) to evaluate insulin resistance in assessing people with diabetes for insulin deficiency or insulin resistance as part of the core pathology, type 2 diabetes mellitus is such a common disease that assessing all affected individuals using these indices is hardly feasible in clinical practice. Given that one of the important aims of the proposed algorithm is to promote the proper use of antidiabetic medications among non‐experts, the presence or absence of obesity was adopted as the single most important clinical measure to allow the core pathology of diabetes to be detected to some extent. Thus, it is recommended that patients are assessed for obesity using the definition of obesity in Japan, body mass index (BMI) 25 kg/m2 or more 30 , in choosing medications for type 2 diabetes mellitus. Given that the extent of obesity (BMI) and insulin resistance are shown to be positively correlated, insulin resistance is assumed to have a greater contribution to type 2 diabetes mellitus in highly obese patients, thus prompting the choice of medications to address the pathology in question. The caveat is that visceral fat accumulation is often noted in Japanese and Asian individuals with a BMI lower than that in obese Westerners and that insulin resistance may be implicated due to visceral fat accumulation in some of these patients, they are usually categorized by BMI as being non‐obese 4 , 31 , 32 . Despite this caveat, however, it is assumed that patients can be accurately assessed for excessive visceral fat accumulation by assessing them for BMI and waist circumference at the same time. Note here that excessive visceral fat accumulation may be suspected in men with a waist circumference of 85 cm or greater as well as in women with a waist circumference of 90 cm or greater. Now, candidate medications for patients with obesity include non‐insulin secretagogues, e.g., biguanides, SGLT2 inhibitors, and thiazolidinediones, as well as insulin secretagogues, e.g., GLP‐1 receptor agonists with potential for weight‐reducing effects and imeglimin, for which obesity/insulin resistance is a good indication, given its insulin‐sensitizing properties.
In most non‐obese individuals with type 2 diabetes mellitus in whom insulin insufficiency is assumed to constitute the core pathology, consideration should be given in medication selection to insulin secretagogues as the mainstay of treatment. Of these, DPP‐4 inhibitors remain the most frequently prescribed for people with type 2 diabetes in Japan, particularly most frequently in elderly patients, probably reflecting the high expectations for their safety in the elderly 25 . DPP‐4 inhibitors are also shown in some reports to exert far greater glucose‐lowering efficacy in Asians than in other races 33 , 34 , suggesting that non‐obese patients with type 2 diabetes likely represent a good indication for this medication class, given its safety and efficacy. Again, while numerous studies conducted to date consistently suggest a low cardiovascular risk with DPP‐4 inhibitors as a class 35 , 36 , 37 , some of these medications are also reported to be associated with an increased risk of heart failure, thus calling for their judicious use in patients at high risk of heart failure 38 . Of the insulin secretagogues, sulfonylureas (SUs) are also of interest, in that they are non‐glucose‐dependent insulin secretagogues and are associated with a high risk of hypoglycemia 39 , while glinides and α‐glucosidase inhibitors also represent good medication candidates for patients exhibiting marked postprandial hyperglycemia. Metformin is shown to exert comparable HbA1c‐lowering efficacy in both non‐obese and obese Japanese patients with type 2 diabetes mellitus and thus represents an option for non‐obese people with type 2 diabetes 40 , 41 . Note here that non‐obese patients include lean patients (BMI <18.5 kg/m2) who are mainly elderly; thus, caution should be exercised in using antidiabetic medications with weight‐reducing properties, i.e., GLP‐1 receptor agonists and SGLT2 inhibitors, in lean patients 16 , as it may be associated with an increased risk of geriatric syndrome, e.g., sarcopenia and frailty.
Giving due consideration to ensuring safety (step 2)
Note that the most desirable attribute required of antidiabetic medications is their ability to ‘lower blood glucose safely’. Thus, the proposed algorithm has included a summary of their glucose‐lowering potency relative to their efficacy and safety and risk of hypoglycemia, as well as precautions (particularly contraindications) for their use in patients with organ derangement (e.g., renal impairment, hepatic disorder [particularly cirrhosis], cardiovascular disorder, and heart failure) in Table 1, with running commentary on areas where caution should be exercised in their use: (1) the use in elderly patients of sulfonylureas and glinides, both of which are associated with a high risk of hypoglycemia; (2) safety precautions in medication selection in patients with renal impairment, a highly common comorbidity in people with type 2 diabetes; and (3) medications contraindicated in patients with heart failure.
Table 1.
A summary of the characteristics of antidiabetic medications for safe glycemic control: a comparison of glucose‐lowering potency, hypoglycemia risk, contraindications, adherence rates and medication costs (a ranked listing of medications initially prescribed by frequency)
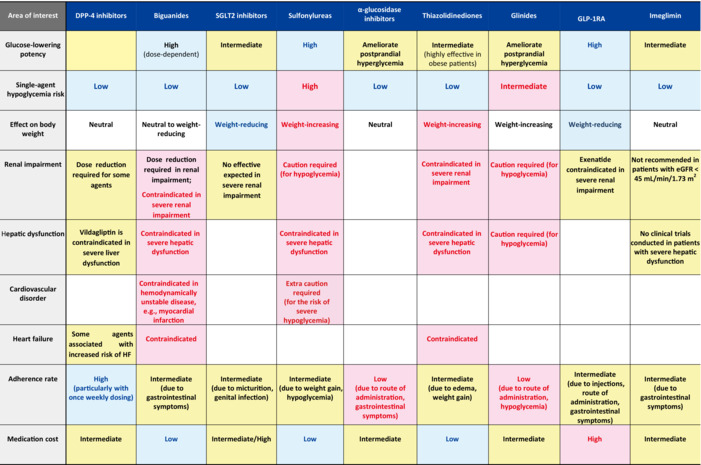
|
According to a network meta‐analysis of the HbA1c‐lowering efficacy of antidiabetic medications, GLP‐1 receptor agonists are shown to be the most potent of all medications in lowering HbA1c, followed by metformin, pioglitazone, and sulfonylureas 42 . It is also shown that metformin lowers glucose dose‐dependently and exerts highly potent glucose‐lowering effects at its high doses and that the thiazolidinedione lowers glucose through its insulin‐sensitizing effects on adipose tissue and skeletal muscle and thus is shown to be more effective in obese patients.
Safety against hypoglycemia risk remains the most relevant of all safety requirements for antidiabetic medications. As single agents, antidiabetic medications other than the non‐glucose dependent sulfonylureas and glinides are generally associated with a low risk of hypoglycemia, while sulfonylureas are among the agents associated with a high risk of hypoglycemia. Indeed, according to a report from the JDS Committee on Survey of Severe Hypoglycemia Associated with Diabetes Treatment, patients treated with sulfonylureas accounted for about 30% of all patients treated with any antidiabetic medications (or about 85% of all patients treated with medications other than insulin therapy) who required emergency transportation for severe hypoglycemia 39 . A finding of particular interest from this survey is that elderly patients accounted for a large proportion of those thus transported for severe hypoglycemia, suggesting that caution should be exercised in the use of sulfonylureas in elderly patients.
The impact of antidiabetic medications on body weight is also particularly relevant to the correction of obesity and the prevention of geriatric syndrome, two major issues referred to above. In this regard, SGLT2 inhibitors are shown to be associated with a weight reduction of 2 kg compared with placebo 16 , suggesting their suitability for use in obese people with type 2 diabetes. GLP‐1 receptor agonists are also shown to have weight‐reducing effects and are thus deemed suitable for use in obese people with type 2 diabetes, with the reduction in body weight reported to be 2 kg on average in patients treated with these medications compared with those treated with placebo 16 . Of these, samaglutide was evaluated for its efficacy in Japanese patients with type 2 diabetes mellitus in a recently reported study, which demonstrated that the medication was associated with a significant reduction in body weight (ranging from 2 to 3 kg) at high doses 43 , 44 . Again, α‐glucosidase inhibitors are shown to be associated with a greater weight reduction in obese Japanese people with type 2 diabetes than miglitol 45 . Conversely, many studies reported a weight gain of about 2 kg in patients treated with sulfonylureas than that in patients treated with placebo, while pioglitazone was shown to be associated with a weight gain of 1–4 kg 46 and likely edema as well.
Caution needs to be exercised in the use of multiple antidiabetic medications in patients with diabetes complicated by renal impairment. Given that most non‐glucose‐dependent insulin secretagogues (e.g., sulfonylureas and glinides) are renally excreted, their use is thought likely to be associated with increased risk of hypoglycemia, and sulfonylureas and nateglinide are both contraindicated for use in those with renal impairment 39 , while, as a glinide with biliary excretion, repaglinide is shown to be relatively safer for use in patients with renal impairment than other glinides. Metformin is shown to be associated with an increased risk of lactic acidosis in patients with renal dysfunction and is thus contraindicated in those with eGFR <30 mL/min/1.73 m2 but is recommended for use at a daily maximum dose of 750 and 1,500 mg in those with eGFR <45 mL/min/1.73 m2 and ≥45 mL/min/1.73 m2, respectively 28 . Pioglitazone is available for use overseas even in renally impaired patients but is contraindicated for use in severely renally impaired patients in Japan. As their glucose‐lowering efficacy is shown to be diminished in renally impaired patients, SGLT2 inhibitors raise concern over their failure to achieve adequate glucose lowering in severely renally impaired patients.
Severe hepatic disorder constitutes a contraindication for biguanides, sulfonylureas, and thiazolidinediones (despite being a relative indication for insulin therapy). Of all patients with cardiovascular disease (CVD), those with hemodynamically unstable disease and heart failure are a contraindication for biguanides, while metformin is no longer contraindicated in patients with heart failure (while still contraindicated in patients with hemodynamically unstable, acute heart failure) in Western countries, following reports of reductions in the need for hospitalization due to heart failure and in mortality risk with metformin 47 , 48 , 49 . While patients with stage C or higher symptomatic heart failure are deemed a contraindication, and those with stage A and B an indication, for thiazolidinediones, consideration should be given in the latter to their dose adjustment, salt restriction, and concomitant use of diuretics for associated fluid retention.
While not mentioned in Table 1, it is important that patients receiving SGLT2 inhibitors be closely monitored for euglycemic diabetic ketoacidosis. SGLT2 inhibitors are expected to increase urinary glucose excretion and to lower blood glucose and insulin, leading to an increased glucagon/insulin ratio and an increased hepatic glycogenesis/lipolysis in adipose tissue, thus resulting in increased usage of lipids for energy metabolism. Therefore, SGLT2 inhibitors are associated with the risk of ketoacidosis even in patients with normoglycemia due to an acute increase in ketone bodies on the basis of insulin insufficiency or during their sick days.
Weighing the additional medication benefits for comorbidities (step 3)
Given that numerous large‐scale clinical trials conducted mainly overseas have shown the efficacy of SGLT2 inhibitors and GLP‐1 receptor agonists against chronic kidney (particularly overt nephropathy), cardiovascular disease, and heart failure, the proposed algorithm included cardiovascular disease, heart failure, and chronic kidney disease (particularly overt nephropathy) as target diseases of secondary interest for which antidiabetic medications may be seen to offer additional benefits. While it should be noted that, unfortunately, the algorithm draws mainly on the evidence available from overseas due to the paucity of data from Japan on this issue, these comorbidities appear to be a valid indication for the use of SGLT2 inhibitors and GLP‐1 receptor agonists, with the caveat that the reduction seen in cardiovascular events in these trials may be accounted for in part by that in HbA1c with these agents 50 , suggesting that their organ‐protective effects may not be completely independent of their glucose‐lowering effects and that further studies are awaited to elucidate the mechanisms involved.
Weighing the additional benefit of antidiabetic medications for cardiovascular disease
Large‐scale clinical trials of SGLT2 inhibitors (i.e., EMPA‐REG, CANVAS, and DECLARE TIMI 58) have been conducted in people with type 2 diabetes having cardiovascular disease or high‐risk patients with type 2 diabetes with major adverse cardiovascular events (MACE) (a composite of cardiovascular death, non‐fatal myocardial infarction (MI), and non‐fatal ischemic stroke) as the primary endpoint and demonstrated a significant reduction in MACE with these agents 51 , 52 , 53 with this finding also confirmed by independent meta‐analyses of these trials 54 , 55 . Likewise, of the GLP‐1 receptor agonists available, those of human origin have been shown in several large‐scale clinical trials (i.e., LEADER, SUSTAIN6, and Harmony) to produce a significant reduction in MACE 56 , 57 , 58 with this outcome also confirmed in a meta‐analysis 54 . Thus, while it should be noted that there is a paucity of data from large‐scale trials in Japanese patients and that Japanese people with diabetes are at lower risk of cardiovascular disease than their Western counterparts, the evidence available overseas is so numerous and of such high quality that SGLT2 inhibitors and GLP‐1 receptor agonists were included as highly recommended options in the order listed for their additional benefit for cardiovascular disease in the proposed algorithm.
Weighing the additional benefit of antidiabetic medications for heart failure
Assessing and treating people with type 2 diabetes mellitus for heart failure is of vital importance, given that even asymptomatic people with type 2 diabetes mellitus are deemed to be in a state called ‘pre‐HF’, i.e., at high risk of heart failure. In this regard, SGLT2 inhibitors (i.e., empagliflozin, canagliflozin, and dapagliflozin) are shown in clinical trials investigating their cardiovascular safety to be useful in preventing heart failure 51 , 52 , 53 , with a similar finding shown in a subset analysis of Asian subjects 59 . Also of interest is a study that compared the real‐world evidence for SGLT2 inhibitors vs DPP‐4 inhibitors in global populations including Japanese and Koreans, which also confirmed the usefulness of SGLT2 inhibitors in preventing heart failure 60 . Furthermore, SGLT2 inhibitors are shown in a clinical trial in heart failure patients with or without type 2 diabetes mellitus exhibiting heart failure with reduced ejection fraction (HFrEF) to produce a significant reduction in heart failure aggravation and cardiovascular death 61 , 62 with this outcome confirmed in a meta‐analysis 63 . SGLT2 inhibitors are also shown in heart failure patients with or without type 2 diabetes mellitus exhibiting heart failure with preserved EF (HFpEF) to produce a significant reduction in heart failure aggravation and cardiovascular death 64 , 65 . Given this evidence, therefore, SGLT2 inhibitors were included as medications of first choice in people with type 2 diabetes having heart failure.
In contrast, GLP‐1 receptor agonists are not consistently shown to be useful in preventing heart failure in clinical studies evaluating their cardiovascular safety but are nevertheless shown to be useful as a class in preventing heart failure in a meta‐analysis 66 . However, their usefulness in a Japanese population remains unclear at the time of this review. Again, the FIGHT trial evaluated the safety of liraglutide in patients with acute heart failure exhibiting dLVEF and demonstrated that, contrary to expectations, treatment with liraglutide led to an increased risk of heart failure in those with type 2 diabetes mellitus 67 , suggesting the need to further investigate the useful of GLP‐1 receptor agonists for each heart failure pathology or stage as well as their respective mechanisms of action.
Weighing the additional benefit of antidiabetic medications for CKD (particularly overt nephropathy)
In sub‐analyses of data from people with type 2 diabetes mellitus at high risk of cardiovascular disease participating in cardiovascular safety trials, SGLT2 inhibitors (i.e., empagliflozin, canagliflozin, and dapagliflozin) are shown to be useful in reducing the composite renal events 51 , 52 , 53 . Of note, reanalysis of the canagliflozin trial data using a rigorous composite endpoint (doubling of serum creatinine, end‐stage renal failure, and renal death) showed a significant reduction in the composite renal endpoint, as well as in renal dysfunction and albuminuria, with the medication 68 . Also of interest are the findings from large‐scale clinical trials evaluating the usefulness of SGLT2 inhibitors (i.e., DAPA‐CKD and CREDENCE) 69 , 70 . While the DAPA‐CKD and CREDENCE trials involved different patient populations (chronic kidney disease patients with or without type 2 diabetes mellitus of whom those without type 2 diabetes mellitus accounted for 32.5% [eGFR, 25–75 mL/min/1.73 m2; urinary albumin/creatinine ratio (ACR), 200–5,000 mg/g] and patients with type 2 diabetes mellitus with chronic kidney disease exhibiting albuminuria [eGFR, ≥30/<90 mL/min/1.73 m2; urinary ACR, >300–5,000 mg/g], respectively), both studies showed a significant reduction in the composite renal endpoint. Interestingly, dapagliflozin was examined for its renoprotective effect by primary cause of chronic kidney disease in an exploratory analysis of the DAPA‐CKD study data, which revealed that despite there being no interaction between its renoprotective effect and any primary cause of chronic kidney disease, the medication offered renoprotection for patients with diabetic nephropathy 71 . Given that most subjects in the DAPA‐CKD and CREDENCE trials were shown to have overt albuminuria (urinary ACR, ≥300 mg/g), at present, it appears that the evidence of renoprotection with SGLT2 inhibitors remains nearly limited to people with type 2 diabetes mellitus having overt nephropathy. In this light, the proposed algorithm noted that SGLT2 inhibitors should be considered as medications of first choice in patients with albuminuria (particularly overt nephropathy), regardless of their glucose‐lowering effects, while not only SGLT2 inhibitors but GLP‐1 receptor agonists, as noted below, should be considered in patients without albuminuria. Again, it should also be noted not only that the renoprotective effect of SGLT2 inhibitors remains unclear in highly renally impaired patients (eGFR, 25 mL/min/1.73 m2) and but that they cannot be expected to be useful as antidiabetic medications in these patients.
Of the GLP‐1 receptor agonists currently available, liraglutide is shown to inhibit the onset of persistent overt albuminuria thereby reducing the occurrence of a composite renal endpoint in a subset analysis of people with type 2 diabetes mellitus at high risk of cardiovascular disease treated with the medication 72 . Again, semaglutide is shown to reduce a composite renal endpoint in people with type 2 diabetes mellitus at high risk of cardiovascular disease 73 , and duraglutide to inhibit the deterioration of eGFR thereby significantly reducing albuminuria in people with type 2 diabetes mellitus exhibiting an eGFR ≥30/<60 mL/min/1.73 m2 74 . As GLP‐1 receptor agonists are also reported to reduce a composite cardiovascular endpoint (cardiovascular death, non‐fatal MI, and non‐fatal cerebral infarction [CI]) in people with type 2 diabetes mellitus having renal dysfunction (eGFR, <60 mL/min/1.73 m2) 75 , GLP‐1 receptor agonists were recommended as medications of second choice in people with type 2 diabetes mellitus having albuminuria but SGLT2 inhibitors or GLP‐1 receptor agonists were recommended as medications worth considering in people with type 2 diabetes mellitus without albuminuria in the proposed algorithm.
Choosing medications with relevant patient background characteristics of interest in mind (step 4)
The proposed algorithm included a summary of relevant patient background characteristics of interest against which to choose medications as well as the medical costs involved (Figure 2, Table 1). Given that medication adherence in people with type 2 diabetes mellitus is shown not only to impact their glycemic control but to be associated with their cardiovascular disease morbidity, mortality, and hospitalization risk, paying attention to maintaining their medication adherence represents an extremely important part of diabetes clinical practice. The medical cost burden should be weighted for each patient, as it includes not only the medication costs but associated healthcare costs.
Individuals with chronic disease, such as diabetes mellitus, must recognize that they need to be on long‐term pharmacotherapy for the disease while adhering to the dosage and usage of the medication(s) prescribed. Indeed, a decline in adherence to these medications not only diminishes their antidiabetic efficacy but increases the risk of hypoglycemia through inappropriate intensification of therapy and contributes to polypharmacy. Unfortunately, however, the adherence rate is shown to be only moderately good at 68.8% in patients newly prescribed antidiabetic medications and at 78.1% in all patients receiving these medications 76 . According to a meta‐analysis of eight observational studies in people with type 2 diabetes mellitus, the relative risks for all‐cause mortality and hospitalization are shown to be 0.72 (95% confidence interval [CI], 0.62–0.82) and 0.90 (0.87–0.94) in those in the high adherence (80% or higher) group compared with the low adherence (<80%) group, suggesting that low adherence is associated with an increased cardiovascular disease risk 77 . Studies overseas also reported that there was a significant correlation between medication adherence and changes in HbA1c in people with type 2 diabetes mellitus, i.e., that a 10% increase in adherence translates into a 0.15% decrease in HbA1c 78 , 79 , and similar results were shown in a questionnaire survey conducted in 1,022 patients with type 2 diabetes mellitus in Japan, i.e., that the more frequent the dosing and the older the patient, the lower the adherence and that there was a significant difference in adherence between those receiving, and those not receiving, their medications as one package 80 . Of note, a systematic review and meta‐analysis of medication adherence in people with type 2 diabetes mellitus has become available 81 . In this review, a comparison of adherence to metformin, sulfonylureas and thiazolidinediones showed that medication adherence is significantly higher with sulfonylureas and thiazolidinediones than with metformin, higher with thiazolidinediones than with sulfonylureas, and higher with DPP‐4 inhibitors than with thiazolidinediones or sulfonylureas. Of note here also is a retrospective study conducted to evaluate the need for intensification of therapy after treatment with biguanides or DPP‐4 inhibitors in medication‐naïve Japanese patients with type 2 diabetes, which suggested that DPP‐4 inhibitors are superior to biguanides 82 . Again, while glinides and α‐glucosidase inhibitors, which need to be taken before meals, are assumed to be associated with lower adherence rates than sulfonylureas, biguanides, or thiazolidinediones 83 , this may be remedied by implementing appropriate measures, e.g., ensuring that all other medications are also taken before meals 80 . Thus, based on the evidence summarized above, the proposed algorithm included a summary of adherence rates for the available antidiabetic medications in a separate table to help in medication selection. In an increasingly aging society such as Japan, it is deemed important that efforts be focused on minimizing dosing frequency as well as on maintaining or improving medication adherence through appropriate measures, e.g., provision of medications in one package or the use of combination medications.
Medical expenditure is ever increasing with the aging of the population 84 . A survey on medical expenditure for lifestyle‐related diseases in 2019 by the National Federation of Health Insurance Societies showed that, of the 10 lifestyle‐related diseases including cerebrovascular disease, ischemic heart disease and dialysis, diabetes mellitus imposes a huge economic burden on affected individuals, accounting for the third largest share in hospitalization costs, the largest share in non‐hospitalization costs, and the second largest share next to dialysis in daily healthcare costs 85 . As high medical expenditure is associated with a decline in adherence to medications 86 , it is also likely to be associated with a decline in their efficacy. Thus, consideration needs to be given to the choice of antidiabetic medication(s), as they vary widely in their prices in Japan and expensive choices impose an increased burden on the patients and the patient's burden is not limited to medication costs. Of note here is a survey conducted using the National Database of Health Insurance Claims and Specific Health Check‐ups to investigate total medical expenditures incurred by Japanese people with type 2 diabetes mellitus in 1 year following their initial antidiabetic prescription (adjusted for age, sex, comorbidities, healthcare institutional attributes, and other relevant factors), which showed that, of all antidiabetic medications, biguanides represented the lowest 1 year expenditure, followed by thiazolidinediones and α‐glucosidase inhibitors, while GLP‐1 receptor agonists represented the highest 25 . An estimation of the economic burden associated with the use of antidiabetic medications is given in a separate table in terms of their prices and associated total medical expenditure. To help reduce each patient's economic burden, therefore, consideration should be given to using generics, switching to biguanides, or switching from multiple single agents to combination medications.
PERIODIC ASSESSMENT OF TREATMENT EFFICACY AND THE NEED FOR ADJUSTMENTS IN PHARMACOTHERAPY
It is proposed in the present algorithm that each medication regimen be reviewed for possible revision every 3 months after its initiation to avoid delays in addressing the needs of patients requiring intensification of therapy and that attention be focused on promoting medical nutrition therapy tailored to address their diabetes pathology and nephropathy, exercise therapy, and lifestyle modification in each patient, while reverting to step 1, as required, to add further medications, increase their medication doses or consider alternative medications.
It should be noted that if left untreated, hyperglycemia increases the subsequent risk of diabetic microangiopathy, macroangiopathy or death in people with diabetes 87 , 88 and that inappropriate glycemic control results at least in part from delays in initiation or intensification of therapy (i.e., clinical inertia) 89 . Indeed, it is reported in a US study that antidiabetic therapy was not appropriately initiated and intensified within 6 months of consultation in 37% and 18% of people with diabetes requiring initiation and intensification of therapy, respectively 90 , and it is also reported in another study that antidiabetic therapy was not intensified within 6 months of consultation in 44% of people with type 2 diabetes mellitus having HbA1c 9% or higher 91 . Thus, while clinical inertia occurs often in clinical practice, pharmacotherapy needs to be immediately adjusted in patients who have failed to achieve their respective HbA1c control goals. In this regard, while it is recommended by the ADA that any medication regimen be reviewed for efficacy as well as for revision every 3–6 months 19 , it is recommended in the current algorithm that any regimen in place be reviewed for revision in a 3 month cycle, reflecting the usual frequency of hospital visits by people with diabetes in Japan, with a focus also on promoting medical nutrition therapy tailored to address the diabetes pathology and nephropathy, as well as exercise therapy and lifestyle modification, in each patient.
DISCLOSURE
Y.O. reports subsidies or donations from Novo Nordisk Pharma Co., Ltd, Kyowa Kirin Co., Ltd, Mitsubishi Tanabe Pharma Co., Ltd, Takeda Pharmaceutical Co., Ltd, Daiichi Sankyo Co., Ltd, Teijin Co., Ltd, Nippon Boehringer Ingelheim Co., Ltd, and Sumitomo Pharma Co., Ltd. D.Y. reports honoraria from Novo Nordisk Pharma Co., Ltd, Ono Pharmaceutical Co., Ltd, research funding from Terumo Co., Ltd, and endowed departments by Novo Nordisk Pharma Co., Ltd, Nippon Boehringer Ingelheim Co., Ltd, Ono Pharmaceutical Co., Ltd, Taisho Pharmaceutical Co., Ltd, and Arkray. R.N. reports honoraria from Sanofi Co., Ltd, Medtronic Co., Ltd, Nippon Boehringer Ingelheim Co., Ltd, Takeda Pharmaceutical Co., Ltd, Kissei Pharmaceutical Co., Ltd, Novartis Pharma Co., Ltd, Eli Lilly Japan Co., Ltd, Novo Nordisk Pharma Co., MSD, Astellas Pharma Inc., and Abbott and subsidies or donations from Taisho Pharmaceutical Co., Ltd, Ono Pharmaceutical Co., Ltd, Takeda Pharmaceutical Co., Ltd, Nippon Boehringer Ingelheim Co., Ltd, and Abbott. N.H. reports research funding from Ono Pharmaceutical Co., Ltd, Mitsubishi Tanabe Pharma Co., Ltd, and Japan Diabetes Foundation. H.K. reports honoraria from Novo Nordisk Pharma Co., Ltd, Sanofi K.K., Sumitomo Pharma Co., Ltd, Nippon Boehringer Ingelheim Co., Ltd, Eli Lilly Japan K.K., Daiichi Sankyo Co., Ltd, Ono Pharmaceutical Co., Ltd, Kissei Pharmaceutical Co., Ltd, Mitsubishi Tanabe Pharma Co., Ltd, Kowa Co., Ltd, Novartis Pharma Co., Ltd, MSD, Sanwa Kagaku Kenkyusho Co., Ltd, research funding from Kissei Pharmaceutical Co., Ltd, Ono Pharmaceutical Co., Ltd, Eli Lilly Japan K.K., subsidies or donations from MSD, Ono Pharmaceutical Co., Ltd, Sumitomo Pharma Co., Ltd, Takeda Pharmaceutical Co., Ltd, Mitsubishi Tanabe Pharma Co., Ltd, Japan Tobacco Inc., Novo Nordisk Pharma Co., Ltd, travel fees from Ono Pharmaceutical Co., Ltd, Sanwa Kagaku Kenkyusho Co., Ltd, Kowa Co., Ltd, Terumo Co., Ltd, and Abott. R.S. reports honoraria from Mitsubishi Tanabe Pharma Co., Ltd, Eli Lilly Japan K.K., Novo Nordisk Pharma Co., Ltd, Ono Pharmaceutical Co., Ltd, Sumitomo Pharma Co., Ltd, Sanofi Co., Ltd, Astellas Pharma Inc., Daiichi Sankyo Co., Ltd, Kowa Co., Ltd, Sanwa Kagaku Kenkyusho Co., Ltd, Takeda Pharmaceutical Co., Ltd, Nippon Boehringer Ingelheim Co., Ltd, Taisho Pharmaceutical Co., Ltd, Novartis Pharma Co., Ltd, research funding from Sumitomo Pharma Co., Ltd, subsidies or donations from Nippon Boehringer Ingelheim Co., Ltd, Mitsubishi Tanabe Pharma Co., Ltd, LifeScan Japan Co., Ltd, and Ono Pharmaceutical Co., Ltd. T.Y. reports honoraria from MSD, Ono Pharmaceutical Co., Ltd, Takeda Pharmaceutical Co., Ltd, Daiichi Sankyo Co., Ltd, Novo Nordisk Pharma Co., Ltd, Sumitomo Pharma Co., Ltd, Teijin Health Care Co., Ltd, Nippon Boehringer Ingelheim Co., Ltd, research funding from Astra Zeneca, Kowa Pharmaceutical Co., Ltd, MSD, Nippon Boehringer Ingelheim Co., Ltd, Aero Switchtherapeutics Inc., Minophagen Pharmaceutical, Co., Ltd, Mitsubishi Corporation Life Sciences Ltd, NIPRO Co., Ltd, subsidies or donations from Novo Nordisk Pharma Co., Ltd, Mitsubishi Tanabe Pharma Co., Ltd, Taisho Pharmaceutical Co., Ltd, Kyowa Kirin Co., Ltd, Takeda Pharmaceutical Co., Ltd, Ono Pharmaceutical Co., Ltd, Astellas Pharma Inc., Sanofi Co., Ltd, Sumitomo Pharma Co., Ltd, Kowa Pharmaceutical Co., Ltd, Takeda Science Foundation, endowed departments by Takeda Pharmaceutical Co., Ltd, Ono Pharmaceutical Co., Ltd, Novo Nordisk Pharma Co., Ltd, Mitsubishi Tanabe Pharma Co., Ltd, MSD, Nippon Boehringer Ingelheim Co., Ltd, Kowa Pharmaceutical Co., Ltd, NTT DOCOMO Inc., and Asahi Mutual Life Insurance Co., Ltd. No other potential conflicts of interest relevant to this article were reported.
Approval of the research protocol: N/A.
Informed consent: N/A.
Registry and the registration no. of the study/trial: N/A.
Animal studies: N/A.
ACKNOWLEDGMENTS
The authors would like to acknowledge that the current consensus statement has been compiled with reference to the opinion and views of executive board members and organizers of the Japan Diabetes Society as well as to public comments provided by the Japanese Circulation Society and the Japanese Society of Nephology. The authors would also like to extend their deep gratitude to all the experts who contributed to the development of the current statement.
REFERENCES
- 1. Survey/Review Committee on the Diagnostic Criteria for Diabetes Mellitus (SRCDCD) . A report from the SCCDCD (globally standardized version). Tonyobyou 2012; 55: 485–504 (Japanese). [Google Scholar]
- 2. Fukushima M, Usami M, Ikeda M, et al. Insulin secretion and insulin sensitivity at different stages of glucose tolerance: a cross‐sectional study of Japanese type 2 diabetes. Metabolism 2004; 53: 831–835. [DOI] [PubMed] [Google Scholar]
- 3. Tripathy D, Carlsson M, Almgren P, et al. Insulin secretion and insulin sensitivity in relation to glucose tolerance: lessons from the Botnia study. Diabetes 2000; 49: 975–980. [DOI] [PubMed] [Google Scholar]
- 4. Kodama K, Tojjar D, Yamada S, et al. Ethnic differences in the relationship between insulin sensitivity and insulin response: a systematic review and meta‐analysis. Diabetes Care 2013; 36: 1789–1796. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Matsumoto K, Miyake S, Yano M, et al. Glucose tolerance, insulin secretion, and insulin sensitivity in nonobese and obese Japanese subjects. Diabetes Care 1997; 20: 1562–1568. [DOI] [PubMed] [Google Scholar]
- 6. Yoshinari M, Hirakawa Y, Hata J, et al. Comparison of the contributions of impaired beta cell function and insulin resistance to the development of type 2 diabetes in a Japanese community: the Hisayama study. Diabetologia 2021; 64: 1775–1784. [DOI] [PubMed] [Google Scholar]
- 7. Butler AE, Janson J, Bonner‐Weir S, et al. Beta‐cell deficit and increased beta‐cell apoptosis in humans with type 2 diabetes. Diabetes 2003; 52: 102–110. [DOI] [PubMed] [Google Scholar]
- 8. Kou K, Saisho Y, Satoh S, et al. Change in β‐cell mass in Japanese nondiabetic obese individuals. J Clin Endocrinol Metab 2013; 98: 3724–3730. [DOI] [PubMed] [Google Scholar]
- 9. Westermark P, Wilander E. The influence of amyloid deposits on the islet volume in maturity onset diabetes mellitus. Diabetologia 1978; 15: 417–421. [DOI] [PubMed] [Google Scholar]
- 10. Kamata K, Mizukami H, Inaba W, et al. Islet amyloid with macrophage migration correlates with augmented β‐cell deficits in type 2 diabetic patients. Amyloid 2014; 21: 191–201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Yasuda K, Miyake K, Horikawa Y, et al. Variants in KCNQ1 are associated with susceptibility to type 2 diabetes mellitus. Nat Genet 2008; 40: 1092–1097. [DOI] [PubMed] [Google Scholar]
- 12. Unoki H, Takahashi A, Kawaguchi T, et al. SNPs in KCNQ1 are associated with susceptibility to type 2 diabetes in east Asian and European populations. Nat Genet 2008; 40: 1098–1102. [DOI] [PubMed] [Google Scholar]
- 13. Yamauchi T, Hara K, Maeda S, et al. A genome‐wide association study in the Japanese population identifies susceptibility loci for type 2 diabetes at UBE2E2 and C2CD4A‐C2CD4B. Nat Genet 2010; 42: 864–868. [DOI] [PubMed] [Google Scholar]
- 14. Suzuki K, Akiyama M, Ishigaki K, et al. Identification of 28 new susceptibility loci for type 2 diabetes in the Japanese population. Nat Genet 2019; 51: 379–386. [DOI] [PubMed] [Google Scholar]
- 15. Holman RR, Paul SK, Bethel MA, et al. 10‐year follow‐up of intensive glucose control in type 2 diabetes. N Engl J Med 2008; 359: 1577–1589. [DOI] [PubMed] [Google Scholar]
- 16. Maruthur NM, Tseng E, Hutfless S, et al. Diabetes medications as monotherapy or metformin‐based combination therapy for type 2 diabetes: a systematic review and meta‐analysis. Ann Intern Med 2016; 164: 740–751. [DOI] [PubMed] [Google Scholar]
- 17. Davies MJ, D'Alessio DA, Fradkin J, et al. A consensus report by the American Diabetes Association (ADA) and the European Association for the Study of diabetes (EASD). Diabetologia 2018; 2018: 2461–2498. [DOI] [PubMed] [Google Scholar]
- 18. Davies MJ, D'Alessio DA, Fradkin J, et al. A consensus report by the American Diabetes Association (ADA) and the European Association for the Study of diabetes (EASD). Diabetes Care 2018; 2018: 2669–2701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. American Diabetes Association Professional Practice Committee . 9. Pharmacologic approaches to glycemic treatment: standards of medical care in diabetes—2022. Diabetes Care 2022; 45(Suppl. 1): S125–S143. [DOI] [PubMed] [Google Scholar]
- 20. Araki E, Goto A, Kondo T, et al. Japanese clinical practice guideline for diabetes 2019. Diabetol Int 2020; 11: 165–223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Ohkubo Y, Kishikawa H, Araki E, et al. Intensive insulin therapy prevents the progression of diabetic microvascular complications in Japanese patients with non‐insulin‐dependent diabetes mellitus: a randomized prospective 6‐year study. Diabetes Res Clin Pract 1995; 28: 103–117. [DOI] [PubMed] [Google Scholar]
- 22. Ueki K, Sasako T, Okazaki Y, et al. Effect of an intensified multifactorial intervention on cardiovascular outcomes and mortality in type 2 diabetes (J‐DOIT3): an open‐label, randomised controlled trial. Lancet Diabetes Endocrinol 2017; 5: 951–964. [DOI] [PubMed] [Google Scholar]
- 23. Kohro T, Yamazaki T, Sato H, et al. Trends in antidiabetic prescription patterns in Japan from 2005 to 2011. Int Heart J 2013; 54: 93–97. [DOI] [PubMed] [Google Scholar]
- 24. Nishimura R, Kato H, Kisanuki K, et al. Treatment patterns, persistence and adherence rates in patients with type 2 diabetes mellitus in Japan: a claims‐based cohort study. BMJ Open 2019; 9: e025806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Bouchi R, Sugiyama T, Goto A, et al. Retrospective nationwide study on the trends in first‐line antidiabetic medication for patients with type 2 diabetes in Japan. J Diabetes Investig 2022; 13: 280–291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Fang M, Wang D, Coresh J, et al. Trends in diabetes treatment and control in U.S. adults, 1999–2018. N Engl J Med 2021; 384: 2219–2228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. The Committee on the Proper Use of SGLT2 Inhibitors . Recommendations on the proper use of SGLT2 inhibitors. Diabetol Int 2020; 11: 1–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Committee on the Proper Use of Biguanides . Recommendations for the proper use of metformin (Japanese). Available from: https://www.nittokyo.or.jp/uploads/files/recommendation_metformin_200318.pdf Accessed October 1, 2022.
- 29. Japan Diabetes Society (JDS)/Japan Geriatrics Society (JGS) Joint Committee on Improving Care for Elderly Patients with Diabetes , Haneda M, Ito H. Glycemic targets for elderly patients with diabetes. Diabetol Int 2016; 7: 331–333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Japan Society for the Study of Obesity (Ed) . Clinical Practice Guidelines for the Management of Obesity 2016. Tokyo: Life Science Publishers Co., Ltd., 2016. (Japanese). [Google Scholar]
- 31. Ma RC, Chan JC. Type 2 diabetes in east Asians: similarities and differences with populations in Europe and the United States. Ann N Y Acad Sci 2013; 1281: 64–91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Hsu WC, Araneta MR, Kanaya AM, et al. BMI cut points to identify at‐risk Asian Americans for type 2 diabetes screening. Diabetes Care 2015; 38: 150–158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Kim YG, Hahn S, Oh TJ, et al. Differences in the glucose‐lowering efficacy of dipeptidyl peptidase‐4 inhibitors between Asians and non‐Asians: a systematic review and meta‐analysis. Diabetologia 2013; 56: 696–708. [DOI] [PubMed] [Google Scholar]
- 34. Seino Y, Kuwata H, Yabe D. Incretin‐based drugs for type 2 diabetes: focus on East Asian perspectives. J Diabetes Investig 2016; 7(Suppl 1): 102–109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. White WB, Cannon CP, Heller SR, et al. Alogliptin after acute coronary syndrome in patients with type 2 diabetes. N Engl J Med 2013; 369: 1327–1335. [DOI] [PubMed] [Google Scholar]
- 36. Green JB, Bethel MA, Armstrong PW, et al. Effect of sitagliptin on cardiovascular outcomes in type 2 diabetes. N Engl J Med 2015; 373: 232–242. [DOI] [PubMed] [Google Scholar]
- 37. Rosenstock J, Perkovic V, Johansen OE, et al. Effect of Linagliptin vs placebo on major cardiovascular events in adults with type 2 diabetes and high cardiovascular and renal risk: the CARMELINA randomized clinical trial. JAMA 2019; 321: 69–79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Scirica BM, Bhatt DL, Braunwald E, et al. Saxagliptin and cardiovascular outcomes in patients with type 2 diabetes mellitus. N Engl J Med 2013; 369: 1317–1326. [DOI] [PubMed] [Google Scholar]
- 39. Namba M, Iwakura T, Nishimura R, et al. Japan diabetes society survey committee on severe hypoglycemia associated with antidiabetic drugs. A report from the survey committee on severe hypoglycemia associated with antidiabetic drugs. Tonyoubyo 2017; 60: 826–842 (Japanese). [Google Scholar]
- 40. Kaku K, Tajima N, Kawamori R. An observational study of metformin use in type 2 diabetes in clinical practice (MORE study). Tonyobyou 2006; 49: 325–331 (Japanese). [Google Scholar]
- 41. Odawara M, Kawamori R, Tajima N, et al. Long‐term treatment study of global standard dose metformin in Japanese patients with type 2 diabetes mellitus. Diabetol Int 2017; 8: 286–295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Tsapas A, Avgerinos I, Karagiannis T, et al. Comparative effectiveness of glucose‐lowering drugs for type 2 diabetes: a systematic review and network meta‐analysis. Ann Intern Med 2020; 173: 278–286. [DOI] [PubMed] [Google Scholar]
- 43. Yamada Y, Katagiri H, Hamamoto Y, et al. Dose‐response, efficacy, and safety of oral semaglutide monotherapy in Japanese patients with type 2 diabetes (PIONEER 9): a 52‐week, phase 2/3a, randomised, controlled trial. Lancet Diabetes Endocrinol 2020; 8: 377–391. [DOI] [PubMed] [Google Scholar]
- 44. Yabe D, Nakamura J, Kaneto H, et al. Safety and efficacy of oral semaglutide versus dulaglutide in Japanese patients with type 2 diabetes (PIONEER 10): an open‐label, randomised, active‐controlled, phase 3a trial. Lancet Diabetes Endocrinol 2020; 8: 392–406. [DOI] [PubMed] [Google Scholar]
- 45. Sugihara H, Nagao M, Harada T, et al. Comparison of three α‐glucosidase inhibitors for glycemic control and bodyweight reduction in Japanese patients with obese type 2 diabetes. J Diabetes Investig 2014; 5: 206–212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Yki‐Järvinen H. Thiazolidinediones. N Engl J Med 2004; 351: 1106–1118. [DOI] [PubMed] [Google Scholar]
- 47. Eurich DT, Majumdar SR, McAlister FA, et al. Improved clinical outcomes associated with metformin in patients with diabetes and heart failure. Diabetes Care 2005; 28: 2345–2351. [DOI] [PubMed] [Google Scholar]
- 48. Andersson C, Olesen JB, Hansen PR, et al. Metformin treatment is associated with a low risk of mortality in diabetic patients with heart failure: a retrospective nationwide cohort study. Diabetologia 2010; 53: 2546–2553. [DOI] [PubMed] [Google Scholar]
- 49. Eurich DT, Weir DL, Majumdar SR, et al. Comparative safety and effectiveness of metformin in patients with diabetes mellitus and heart failure: systematic review of observational studies involving 34,000 patients. Circ Heart Fail 2013; 6: 395–402. [DOI] [PubMed] [Google Scholar]
- 50. Maiorino MI, Longo M, Scappaticcio L, et al. Improvement of glycemic control and reduction of major cardiovascular events in 18 cardiovascular outcome trials: an updated meta‐regression. Cardiovasc Diabetol 2021; 20: 210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Zinman B, Wanner C, Lachin JM, et al. Empagliflozin, cardiovascular outcomes, and mortality in type 2 diabetes. N Engl J Med 2015; 373: 2117–2128. [DOI] [PubMed] [Google Scholar]
- 52. Neal B, Perkovic V, Mahaffey KW, et al. Canagliflozin and cardiovascular and renal events in type 2 diabetes. N Engl J Med 2017; 377: 644–657. [DOI] [PubMed] [Google Scholar]
- 53. Wiviott SD, Raz I, Bonaca MP, et al. Dapagliflozin and cardiovascular outcomes in type 2 diabetes. N Engl J Med 2019; 380: 347–357. [DOI] [PubMed] [Google Scholar]
- 54. Home P. Cardiovascular outcome trials of glucose‐lowering medications: an update. Diabetologia 2019; 62: 357–369. [DOI] [PubMed] [Google Scholar]
- 55. Zelniker TA, Wiviott SD, Raz I, et al. SGLT2 inhibitors for primary and secondary prevention of cardiovascular and renal outcomes in type 2 diabetes: a systematic review and meta‐analysis of cardiovascular outcome trials. Lancet 2019; 393: 31–39. [DOI] [PubMed] [Google Scholar]
- 56. Marso SP, Daniels GH, Brown‐Frandsen K, et al. Liraglutide and cardiovascular outcomes in type 2 diabetes. N Engl J Med 2016; 375: 311–322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Marso SP, Bain SC, Consoli A, et al. Semaglutide and cardiovascular outcomes in patients with type 2 diabetes. N Engl JMed 2016; 375: 1834–1844. [DOI] [PubMed] [Google Scholar]
- 58. Hernandez AF, Green JB, Janmohamed S, et al. Albiglutideand cardiovascular outcomes in patients with type 2 diabetes and cardiovascular disease (harmony outcomes): a double‐blind, randomised placebo‐controlled trial. Lancet 2018; 392: 1519–1529. [DOI] [PubMed] [Google Scholar]
- 59. Kaku K, Lee J, Mattheus M, et al. Empagliflozin and cardiovascular outcomes in Asian patients with type 2 diabetes and established cardiovascular disease‐results from EMPA‐REG OUTCOME((R)). Circ J 2017; 81: 227–234. [DOI] [PubMed] [Google Scholar]
- 60. Kosiborod M, Lam CSP, Kohsaka S, et al. Cardiovascular events associated with SGLT‐2 inhibitors versus other glucose‐lowering drugs: the CVD‐REAL 2 study. J Am Coll Cardiol 2018; 71: 2628–2639. [DOI] [PubMed] [Google Scholar]
- 61. McMurray JJV, Solomon SD, Inzucchi SE, et al. Dapagliflozin in patients with heart failure and reduced ejection fraction. N Engl J Med 2019; 381: 1995–2008. [DOI] [PubMed] [Google Scholar]
- 62. Packer M, Anker SD, Butler J, et al. Cardiovascular and renal outcomes with empagliflozin in heart failure. N Engl J Med 2020; 383: 1413–1424. [DOI] [PubMed] [Google Scholar]
- 63. Zannad F, Ferreira JP, Pocock SJ, et al. SGLT2 inhibitors in patients with heart failure with reduced ejection fraction: a meta‐analysis of the EMPEROR‐reduced and DAPA‐HF trials. Lancet 2020; 396: 819–829. [DOI] [PubMed] [Google Scholar]
- 64. Anker SD, Butler J, Filippatos G, et al. Empagliflozin in heart failure with a preserved ejection fraction. N Engl J Med 2021; 385: 1451–1461. [DOI] [PubMed] [Google Scholar]
- 65. Nassif ME, Windsor SL, Borlaug BA, et al. The SGLT2 inhibitor dapagliflozin in heart failure with preserved ejection fraction: a multicenter randomized trial. Nat Med 2021; 27: 1954–1960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66. Kristensen SL, Rorth R, Jhund PS, et al. Cardiovascular, mortality, and kidney outcomes with GLP‐1 receptor agonists in patients with type 2 diabetes: a systematic review and meta‐analysis of cardiovascular outcome trials. Lancet Diabetes Endocrinol 2019; 7: 776–785. [DOI] [PubMed] [Google Scholar]
- 67. Margulies KB, Hernandez AF, Redfield MM, et al. Effects of Liraglutide on clinical stability among patients with advanced heart failure and reduced ejection fraction: a randomized clinical trial. JAMA 2016; 316: 500–508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68. Perkovic V, de Zeeuw D, Mahaffey KW, et al. Canagliflozin and renal outcomes in type 2 diabetes: results from the CANVAS program randomised clinical trials. Lancet Diabetes Endocrinol 2018; 6: 691–704. [DOI] [PubMed] [Google Scholar]
- 69. Heerspink HJL, Stefánsson BV, Correa‐Rotter R, et al. DAPA‐CKD trial committees and investigators. Dapagliflozin in patients with chronic kidney disease. N Engl J Med 2020; 383: 1436–1446. [DOI] [PubMed] [Google Scholar]
- 70. Perkovic V, Jardine MJ, Neal B, et al. Canagliflozin and renal outcomes in type 2 diabetes and nephropathy. N Engl J Med 2019; 380: 2295–2306. [DOI] [PubMed] [Google Scholar]
- 71. Wheeler DC, Stefánsson BV, Jongs N, et al. DAPA‐CKD trial committees and investigators. Effects of dapagliflozin on major adverse kidney and cardiovascular events in patients with diabetic and non‐diabetic chronic kidney disease: a prespecified analysis from the DAPA‐CKD trial. Lancet Diabetes Endocrinol 2021; 9: 22–31. [DOI] [PubMed] [Google Scholar]
- 72. Mann JFE, Orsted DD, Brown‐Frandsen K, et al. LEADER steering committee and investigators. Liraglutide and renal outcomes in type 2 diabetes. N Engl J Med 2017; 377: 839–848. [DOI] [PubMed] [Google Scholar]
- 73. Marso SP, Bain SC, Consoli A, et al. SUSTAIN‐6 investigators. Semaglutide and cardiovascular outcomes in patients with type 2 diabetes. N Engl J Med 2016; 375: 1834–1844. [DOI] [PubMed] [Google Scholar]
- 74. Tuttle KR, Lakshmanan MC, Rayner B, et al. Dulaglutide versus insulin glargine in patients with type 2 diabetes and moderate‐to‐severe chronic kidney disease (AWARD‐7): a multicentre, open‐label, randomised trial. Lancet Diabetes Endocrinol 2018; 6: 605–617. [DOI] [PubMed] [Google Scholar]
- 75. Mann JFE, Fonseca V, Mosenzon O, et al. Effects of liraglutide versus placebo on cardiovascular events in patients with type 2 diabetes mellitus and chronic kidney disease. Circulation 2018; 138: 2908–2918. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76. Fischer MA, Stedman MR, Lii J, et al. Primary medication non‐adherence: analysis of 195,930 electronic prescriptions. J Gen Intern Med 2010; 25: 284–290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77. Khunti K, Seidu S, Kunutsor S, et al. Association between adherence to pharmacotherapy and outcomes in type 2 diabetes: a meta‐analysis. Diabetes Care 2017; 40: 1588–1596. [DOI] [PubMed] [Google Scholar]
- 78. Schectman JM, Nadkarni MM, Voss JD. The association between diabetes metabolic control and drug adherence in an indigent population. Diabetes Care 2002; 25: 1015–1021. [DOI] [PubMed] [Google Scholar]
- 79. Pladevall M, Williams LK, Potts LA, et al. Clinical outcomes and adherence to medications measured by claims data in patients with diabetes. Diabetes Care 2004; 27: 2800–2805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80. Kurooka N, Eguchi J, Ashida M, et al. Medication adherence in type 2 diabetes: an investigation into the extent and rate of medication adherence required for favorable glycemic control. Tonyobyou 2020; 63: 609–617 (Japanese). [Google Scholar]
- 81. McGovern A, Tippu Z, Hinton W, et al. Comparison of medication adherence and persistence in type 2 diabetes: a systematic review and meta‐analysis. Diabetes Obes Metab 2018; 20: 1040–1043. [DOI] [PubMed] [Google Scholar]
- 82. Horii T, Iwasawa M, Shimizu J, et al. Comparing treatment intensification and clinical outcomes of metformin and dipeptidyl peptidase‐4 inhibitors in treatment‐naïve patients with type 2 diabetes in Japan. J Diabetes Investig 2020; 11: 96–100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83. Boccuzzi SJ, Wogen J, Fox J, et al. Utilization of oral hypoglycemic agents in a drug‐insured U.S. population. Diabetes Care 2001; 24: 1411–1415. [DOI] [PubMed] [Google Scholar]
- 84. Ministry of Health, Labour and Welfare of Japan . Overview of National Health Care Expenditures in Japan in Fiscal Year 2019. Available from: https://www.mhlw.go.jp/toukei/saikin/hw/k‐iryohi/19/dl/kekka.pdf Accessed October 1, 2022. (Japanese).
- 85. National Federation of Health Insurance Societies . Survey on Medical Expenditures for Lifestyle‐Related Diseases in Japan in Fiscal Year 2019. Available from: https://www.kenporen.com/toukei_data/pdf/chosa_r03_06_01.pdf Accessed October 1, 2022. (Japanese).
- 86. Kang H, Lobo JM, Kim S, et al. Cost‐related medication non‐adherence among U.S. adults with diabetes. Diabetes Res Clin Pract 2018; 143: 24–33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87. Paul SK, Klein K, Thorsted BL, et al. Delay in treatment intensification increases the risks of cardiovascular events in patients with type 2 diabetes. Cardiovasc Diabetol 2015; 14: 100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88. Laiteerapong N, Ham SA, Gao Y, et al. The legacy effect in type 2 diabetes: impact of early glycemic control on future complications (the Diabetes & Aging Study). Diabetes Care 2019; 42: 416–426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89. Phillips LS, Branch WT, Cook CB, et al. Clinical inertia. Ann Intern Med 2001; 135: 825–834. [DOI] [PubMed] [Google Scholar]
- 90. Gilstrap LG, Mehrotra A, Bai B, et al. National rates of initiation and intensification of antidiabetic therapy among patients with commercial insurance. Diabetes Care 2018; 41: 1776–1782. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91. Ji X, Kong SX, Milinovich A, et al. Clinical inertia in type 2 diabetes management: evidence from a large. Real‐World Data Set Diabetes Care 2018; 41: e113–e114. [DOI] [PubMed] [Google Scholar]


