Abstract
Background
A growing proportion of patients with early rectal cancer is treated by local excision only. The aim of this study was to evaluate long-term oncological outcomes and the impact of local recurrence on overall survival for surgical local excision in pT1 rectal cancer.
Methods
Patients who only underwent local excision for pT1 rectal cancer between 1997 and 2014 in two Dutch tertiary referral hospitals were included in this retrospective cohort study. The primary outcome was the local recurrence rate. Secondary outcomes were distant recurrence, overall survival and the impact of local recurrence on overall survival.
Results
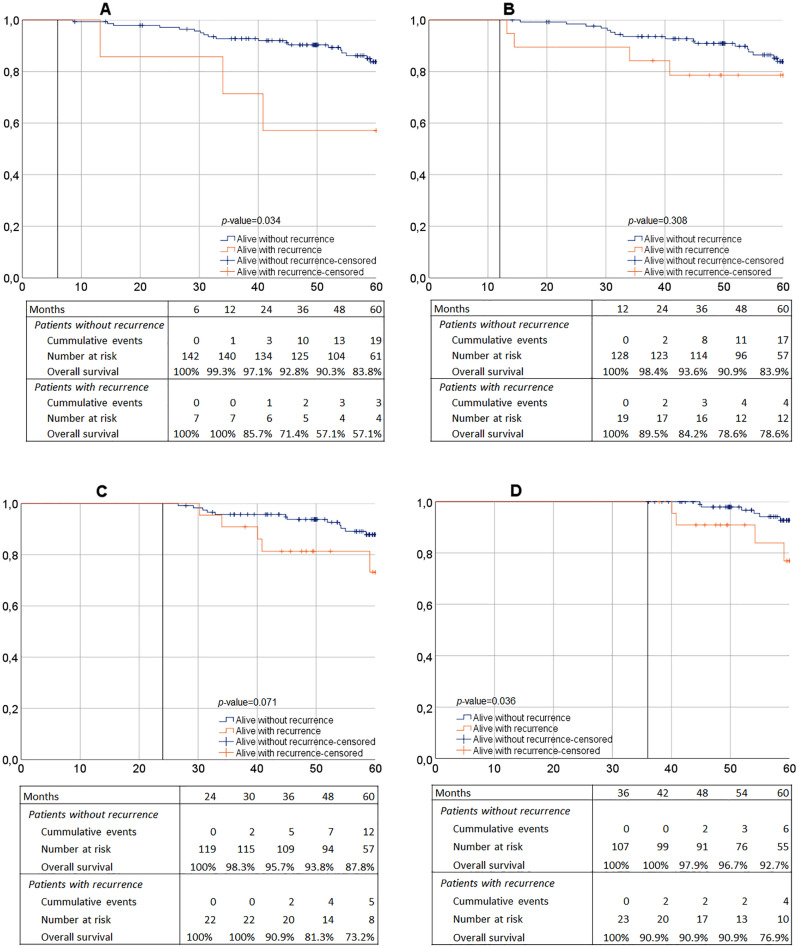
A total of 150 patients (mean age 68.5 ± 10.7 years, 57.3% males) were included in the study. Median length of follow-up was 58.9 months (range 6–176 months). Local recurrence occurred in 22.7% (n = 34) of the patients, with a median time to local recurrence of 11.1 months (range 2.3–82.6 months). The vast majority of local recurrences were located in the lumen. Five-year overall survival was 82.0%, and landmark analyses showed that local recurrence significantly impacted overall survival at 6 and 36 months of follow-up (6 months, p = 0.034, 36 months, p = 0.036).
Conclusions
Local recurrence rates after local excision of early rectal cancer can be substantial and may impact overall survival. Therefore, clinical decision-making should be based on patient- and tumour characteristics and should incorporate patient preferences.
Supplementary Information
The online version contains supplementary material available at 10.1007/s10151-022-02661-6.
Keywords: Rectal cancer, Early cancer, Surgical local excision, Oncological outcome
Introduction
Currently, the standard treatment for rectal cancer is total mesorectal excision (TME). TME surgery may be combined with (chemo)radiotherapy according to the stage of disease [1]. This approach provides a good oncological outcome, but leads to morbidity, affects functional outcome, and influences quality of life [2–4]. To avoid the negative consequences of TME surgery there has been a rising interest in local treatment regimens, especially in early rectal cancer.
In the earliest stage of rectal cancer, local excision by surgical or endoscopic techniques has become the treatment of choice [5]. If the histopathological evaluation of the excised specimen shows risk factors for nodal disease and/or recurrence, completion TME is recommended by guidelines [5]. Nevertheless, this advice is frequently waived by surgeons and/or patients [6]. During the clinical- and shared decision-making process, morbidity and expected functional outcome are weighed against the chance of local recurrence. The risk of local recurrence reported in the literature is highly variable and randomised trials are lacking. A recent meta-analysis of available cohort studies with at least 36 months of follow-up, reported an overall local recurrence rate of 8.1% after local excision of T1 rectal cancer [7]. However, local recurrence rates of the included studies varied from 0 to 40% [7].
Since the clinical consequences of treatment strategies (i.e. organ preservation or radical surgery) are substantial, shared decision-making should be based upon reliable data. In addition, the feasibility of salvage surgery and the impact of local recurrence on overall survival (OS) should also be considered. Therefore, the aim of this study was to evaluate the long-term oncological outcomes of surgical local excision techniques for pT1 rectal cancer. The primary outcome was local recurrence rate. Secondary outcomes were distant recurrence rate, overall survival (OS) rate, and the impact of local recurrence on survival.
Materials and methods
Patient selection
Between 1997 and 2014 all patients who only underwent surgical local excision of pT1 rectal cancer, in two Dutch tertiary referral hospitals for local excision (Laurentius Hospital and IJsselland Hospital) were included. Local excision was performed by either transanal endoscopic microsurgery (TEM) or transanal minimally invasive surgery (TAMIS), and consisted of a full thickness excision. Surgical local excision was performed in patients with lesions < 4 cm or if the invasive part of the lesion was estimated to be < 3 cm based on endoscopy. Local excision was also performed in patients who were staged beyond cT1, because of the known inaccuracy of imaging in early staged rectal cancer [8]. Nevertheless, all patients had histopathologically proven pT1 rectal cancer, defined as a tumour within 15 cm from the anal verge. Patients were excluded in case of: conversion to TME; suspected lymph node involvement at clinical staging; or distant metastases at the time of diagnosis. Clinical staging was performed in all patients and was based on imaging by endorectal ultrasound (ERUS), abdominopelvic computed tomography (CT) scan, and chest X-ray or CT scan. Pelvic magnetic resonance imaging (MRI) was implemented as well during the study period. Several reasons for local excision as a sole treatment were applicable. The included patients either did not require completion TME according to the national guidelines, were clinically unfit to undergo completion surgery, or waived additional treatment by TME surgery during a shared decision-making process [9]. Patients who underwent completion TME were excluded. The study was approved by the Institutional Review Boards of both participating centres.
Surgical procedures and histopathological examination
Excisions were performed according to the TEM or TAMIS technique, as described by Buess et al. and Atallah et al. [10, 11]. A full thickness resection was performed by experienced surgeons. After resection, the specimen was oriented, pinned on cork, fixed in formaldehyde, and sent for histopathological examination. Histopathological assessment included lesion size (i.e. polyp size including tumour), pT-stage, submucosal invasion depth (i.e. Kikuchi classification for sessile lesions), differentiation grade, lymphovascular invasion, and residual tumour classification (R-classification). Tumour budding was not included in the histopathological evaluation, since this was not incorporated in the standardised histopathology reports.
Data collection and follow-up
Patient- and tumour characteristics, as well as perioperative data, were collected. Postoperative complications were scored according to the Clavien-Dindo classification [12]. Based on histopathological tumour characteristics patients were divided into low-risk pT1 tumours and high-risk pT1 tumours. In high-risk tumours at least one of the following characteristics had to be present: poor differentiation, lymphovascular invasion, involved resection margin ≤ 1 mm (residual tumour classification R1), or either unclear or not evaluable resection margins (Rx). In low-risk tumours all these factors had to be absent. Guidelines are conflicting with regard to deep submucosal invasion as a risk factor. Moreover, a recent meta-analysis reported that deep submucosal invasion (i.e. ≥ 1000 µm or Kikuchi level sm2-3) is not an independent risk factor for lymph node metastasis [13]. Therefore, deep submucosal invasion was not included as a high-risk factor.
Follow-up included visits to the outpatient clinic, physical examinations, abdominal ultrasound, chest X-rays and measurements of carcinoembryonic antigen (CEA) levels every 6 months for 3 years, and annually thereafter until 5 years of follow-up. Twelve months after local excision a colonoscopy was performed. The majority of patients received additional periodical surveillance, which included MRI and/or sigmoidoscopies with or without ERUS, every 3 months for the first year after local excision in low-risk pT1 tumours, and during the first 2 years in high-risk pT1 tumours. These patients were defined as the close surveillance group.
The primary study objective was local recurrence, which was defined as pelvic recurrence, and could either be endoluminal located at the local excision scar; or locoregional lymph node recurrence. Preferably, local recurrence was confirmed by a biopsy, if a biopsy could not be obtained, the diagnosis was based on imaging (i.e. MRI/CT/ERUS). Secondary outcomes were distant recurrence, eligibility for salvage surgery in case of local recurrence, local recurrence free survival, OS, and the impact of local recurrence on OS.
Statistical analysis
Patient characteristics, perioperative data and clinical outcomes were evaluated using descriptive statistics. Categorical data were presented by frequencies and percentages. Continuous data were differentiated into variables with a normal distribution and a non-normal distribution based on Q-Q plots. Normally distributed variables were reported as mean with standard deviation and non-normally distributed data by median and ranges. Kaplan–Meier curves and Log Rank tests were performed to investigate associations between categorical variables and time to local recurrence. For continuous variables Cox regression analyses were performed after testing the proportional hazards assumption by Schoenfeld residuals. A similar method was used to assess the influence of non-time dependent variables on local recurrence free survival and OS. Kaplan–Meier curves were used to estimate local recurrence free survival rates and OS rates. Since local recurrence is a time-dependent variable, the influence of local recurrence on OS was analysed in depth using landmark analyses [14]. In a landmark analysis the influence of local recurrence on OS is estimated at a certain time point, analysing only the patients who have survived until this time point [15]. During the analysis OS rates are estimated for patients with- or without recurrence at the set landmark time. If patients are simply categorised by the presence of recurrence during the entire length of follow-up, the timing of recurrence and length of survival itself will create a bias. Therefore, landmark analyses provide a less biased estimation of the time-to-event outcome local recurrence [16, 17]. Landmarks were set on 6, 12, 24, and 36 months of follow-up. P-values of < 0.05 were considered statistically significant. Statistical analyses were carried out using SPSS version 26 (IBM Corp., Armonk, NY, USA).
Results
Patient characteristics and clinical outcomes
From January 1997 to September 2014 a total of 150 patients underwent solely surgical local excision for pT1 rectal cancer. Their mean age was 68.5 ± 10.7 years, and 57.3% were men (Table 1). Table 1 shows peri- and postoperative outcomes. The majority of patients were clinically staged as benign or cT1. One patient staged as cT3, had severe comorbidities and was deemed unfit for TME surgery. Four cT2 patients opted for local excision to have a chance of organ preservation. Median duration of the surgical procedure was 50.0 min (range 10–245 min). Intra-operative complications occurred in 2.0% (n = 3) of the patients and consisted of arterial bleeding (n = 2) or perforation with peritoneal breach (n = 1). Perforation occurred in a patient who underwent a polypectomy prior to the local excision. The specimen did not show residual tumour. In total 4.6% (n = 7) of the patients had a postoperative complication and 2.6% (n = 4) scored grade III or higher according to the Clavien-Dindo classification. No procedure related deaths were recorded. Seventy-eight percent of the patients (n = 117) underwent close surveillance, others underwent regular follow-up.
Table 1.
Demographic, clinical and tumour characteristics (n = 150)
| n (%) | |
|---|---|
| Sex; Male | 86 (57.3) |
| Age in years at time of surgery, mean ± SD | 68.5 ± 10.7 |
| Follow-up in months, median (range) | 58.9 (6–176) |
| BMI in kg/m2, mean ± SD | 26.2 ± 3.8 |
| ASA classification | |
| I-II | 129 (86.0) |
| III | 18 (12.0) |
| Missing | 3 (2.0) |
| Distance in cm from anal verge | |
| 0–5 | 51 (34.0) |
| 6–10 | 69 (46.0) |
| 11–15 | 25 (16.7) |
| Missing | 5 (3.3) |
| Location of tumour | |
| Anterior | 43 (28.7) |
| Posterior | 46 (30.7) |
| Lateral right | 17 (11.3) |
| Lateral left | 28 (18.7) |
| Missing | 16 (10.7) |
| Preoperative staging | |
| Benign | 34 (22.7) |
| cT1 | 58 (38.7) |
| cT2 | 4 (2.7) |
| cT3 | 1 (0.7) |
| Missing | 53 (35.3) |
| Duration of surgical procedure in minutes, median (range) | 50 (10–245) |
| Type of procedure | |
| TEM | 127 (84.7) |
| TAMIS | 18 (12.0) |
| Missing | 5 (3.3) |
| Closure of defect | |
| No | 4 (2.7) |
| Yes | 130 (86.7) |
| Missing | 16 (10.7) |
| Peroperative complication | 3 (2.0) |
| Length of stay in days, median (range) | 3 (1–24) |
| Complications according to Clavien-Dindo | |
| No complications | 143 (95.3) |
| Grade I–II | 3 (2.0) |
| Grade IIIb | 2 (1.3) |
| Grade IVb | 2 (1.3) |
| Radicality of resection | |
| R0 | 141 (94.0) |
| R1/Rx | 9 (6.0) |
| Pathological size of lesion in cm | |
| Scar | 25 (16.7) |
| < 3 | 49 (32.7) |
| ≥ 3 | 56 (37.3) |
| Missing | 20 (13.3) |
| Grade of differentation | |
| Well-to-moderate | 126 (84.0) |
| Poor | 7 (4.7) |
| Missing | 17 (11.3) |
| Lymphovascular invasion | |
| No | 106 (70.7) |
| Yes | 14 (9.3) |
| Missing | 30 (20.0) |
| Kikuchi classifcation | |
| sm1 | 50 (33.3) |
| sm2 | 6 (4.0) |
| sm3 | 17 (11.3) |
| Missing | 77 (51.3) |
| Risk of tumoura | |
| Low-risk | 93 (62.0) |
| High-risk | 29 (19.3) |
| Unknown | 28 (18.7) |
BMI body mass index, ASA American Society of Anesthesiologists, TEM transanal endoscopic microsurgery, TAMIS transanal minimally invasive surgery
aHigh-risk defined as poor differentiation, lymphovascular invasion, distance to resection plane ≤ 1 mm or unevaluable
Histological examination
Ninety-four percent (n = 141) of the patients had clear resection margins (R0) after surgical local excision. No association between resection margins and local recurrence was observed. Tumours were poorly differentiated in 4.7% (n = 7) of the patients. Lymphovascular invasion was present in 9.3% (n = 14) of the patients. Table 1 provides an overview of histopathological risk factors. Combining histopathological risk factors resulted in 19.2% (n = 29) of the patients with a high-risk pT1 tumour. In 1 case, analysis of the specimen revealed a lymph node with tumour invasion.
Oncological outcomes
Median follow-up of all patients was 58.9 months (range 6–175 months). Local recurrence occurred in 22.7% (n = 34) of the 150 patients, with a median time to local recurrence of 11.1 months (range 2.3–82.6 months) (Table 2). Of these local recurrences, preoperative assessment by endoscopy and imaging revealed that 85.3% (n = 29) of the recurrences were located endoluminally at the scar of the local excision, and 14.7% (n = 5) patients had suspected mesorectal lymph node recurrences. Distant recurrence was reported in 7.3% (n = 11) of the patients. Of the 11 patients with distant recurrence, 81.8% (n = 9) had both local and distant recurrences. No significant associations between patient- and tumour characteristics, such as lesion size, and local recurrence rate were found, except for the presence of lymphovascular invasion (p = 0.017, Table 3). Local recurrence was associated with the occurrence of distant metastases (Fisher’s exact test, p < 0.001). Table 2 shows the characteristics of recurrences.
Table 2.
Recurrences of pT1 rectal cancer patients after TEM/TAMIS (n = 36)
| n (%) | |
|---|---|
| Tumour risk | |
| Low-risk | 24 (66.7) |
| High-risk | 9 (25.0) |
| Missing | 3 (8.3) |
| Location recurrence | |
| Local | 34 (94.4) |
| Distant | 11 (30.6) |
| Local and distant | 9 (25.0) |
| Median time to local recurrence in months | 11.1 (2.3–82.6) |
| Median time from TEM/TAMIS to salvage surgery in months | 12.5 (4.4–58.4) |
| Location of local recurrence (pre-operative assessment) | |
| Intraluminal | 29 (85.3) |
| Nodal recurrence | 5 (14.7) |
| Treatment of local recurrence | |
| No surgery | 7 (20.6) |
| Salvage surgery | 27 (79.4) |
| APR | 13 (48.1) |
| LAR | 14 (51.9) |
| Radiotherapy | |
| None/not applicable | 9 (26.5) |
| Short-course radiotherapy | 13 (38.2) |
| Chemoradiation | 11 (32.4) |
| Long course radiotherapy | 1 (2.9) |
| Pathological T-stage local recurrence | |
| pT0 | 2 (7.4) |
| pT1 | 5 (18.5) |
| pT2 | 2 (7.4) |
| pT3 | 12 (44.4) |
| pT4 | 2 (7.4) |
| Missing | 4 (14.8) |
| Pathological N-stage local recurrence | |
| pN0 | 12 (44.4) |
| pN1 | 9 (33.3) |
| pN2 | 2 (7.4) |
| Missing | 4 (14.8) |
| Local recurrence diagnosed based on | |
| MRI, endoscopy, ERUS | 14 (41.2) |
| MRI, endoscopy | 5 (14.7) |
| MRI, ERUS | 1 (2.9) |
| Endoscopy, ERUS | 1 (2.9) |
| MRI | 7 (20.6) |
| Endoscopy | 5 (14.7) |
| Missing | 1 (2.9) |
TEM transanal endoscopic microsurgery, TAMIS transanal minimally invasive surgery, APR abdominal perineal resection, LAR low anterior resection, MRI magnetic resonance imaging, ERUS endorectal ultrasound
Table 3.
Factors associated with 5-year local recurrence by Kaplan Meier estimates
| No local recurrence n = 117 (%) | Local recurrence n = 33 (%) | p value | |
|---|---|---|---|
| Age in years, mean ± SDa | 68.8 ± 11.0 | 67.6 ± 9.7 | 0.75 |
| Sex | 0.21 | ||
| Male | 70 (81.4) | 16 (18.6) | |
| ASA classification | 0.86 | ||
| ASA I-II | 101 (78.3) | 28 (21.7) | |
| ASA III | 14 (77.8) | 4 (22.2) | |
| Distance from anal verge in cm | 0.98 | ||
| 0–5 | 39 (76.5) | 12 (23.5) | |
| 6–10 | 53 (76.8) | 16 (23.2) | |
| 11–15 | 20 (80.0) | 5 (20.0) | |
| Location of tumour | 0.43 | ||
| Anterior | 35 (81.4) | 8 (18.6) | |
| Posterior | 33 (71.7) | 13 (28.3) | |
| Lateral right | 15 (88.2) | 2 (11.8) | |
| Lateral left | 21 (75.0) | 7 (25.0) | |
| Type of procedure | 0.96 | ||
| TEM | 100 (78.7) | 27 (21.3) | |
| TAMIS | 14 (77.8) | 4 (22.2) | |
| Duration of surgical procedure in minutes | 0.057 | ||
| 0–59 | 71 (84.5) | 13 (15.5) | |
| 60–89 | 28 (71.8) | 11 (28.2) | |
| ≥ 90 | 10 (62.5) | 6 (37.5) | |
| Peroperative complication | 0.37 | ||
| Yes | 3 (10) | 0 (0) | |
| Postoperative complications according to Clavien- Dindo | 0.58 | ||
| Grade I–II | 3 (100) | 0 (0) | |
| Grade IIIb | 2 (100) | 0 (0) | |
| Grade IVb | 1 (50) | 1 (50) | |
| Radicality of resection | 0.46 | ||
| R1/Rx | 8 (88.9) | 1 (11.1) | |
| Pathological size of lesion in cm | 0.53 | ||
| Scar | 21 (84.0) | 4 (16.0) | |
| < 3 | 38 (77.6) | 11 (22.4) | |
| ≥ 3 | 40 (71.4) | 16 (28.6) | |
| Grade of differentiation | 0.58 | ||
| Well-to-moderate | 96 (76.2) | 30 (23.8) | |
| Poor | 6 (85.7) | 1 (14.3) | |
| Lymphovascular invasion | 0.017 | ||
| Yes | 7 (50) | 7 (50) | |
| Risk of tumourb | 0.39 | ||
| Low-risk | 71 (76.3) | 22 (23.7) | |
| High-risk | 20 (69.0) | 9 (31.0) | |
| Year of procedure, median (range) | 2007 (1997–2014) | 2007 (1999–2013) |
ASA American Society of Anesthesiologists, TEM transanal endoscopic microsurgery, TAMIS transanal minimally invasive surgery
aCox regression analysis
bHigh-risk defined as poor differentiation, lymphovascular invasion, distance to resection plane ≤ 1 mm or unevaluable
Salvage surgery was performed in 79.4% (n = 27) of the patients with local recurrence, and consisted of a low anterior resection in 51.9% (n = 14), and an abdominoperineal resection in 48.1% (n = 13) of the patients. Postoperative complications occurred in 33.3% (n = 9) of the patients. According to the Clavien-Dindo classification 5 patients had grade II and 4 patients had grade III complications. Prior to the salvage procedure 73.5% (n = 20) received neoadjuvant (chemo) radiotherapy. Outcomes of the histopathological evaluation of the salvage specimen could be retrieved in 85.2% (n = 23) of the patients, and revealed that 51.9% (n = 14) of the patients had a pT3–4 tumour, and 40.7% (n = 11) had tumour positive lymph nodes (i.e. pN1-2) (Table 2). In 1 patient pathological assessment of the specimen showed complete response after neoadjuvant treatment. Median length of follow-up after salvage surgery was 30.5 months (range 7–111 months). Of the 27 patients that underwent salvage surgery, 14.8% (n = 4) eventually died due to disease progression. Close surveillance was not associated with a higher probability of salvage surgery (Fisher’s exact test, p = 0.28).
Local recurrence free survival
Local recurrence free survival was 81.0% at 3 years and 77.1% at 5 years of follow-up (Table 4). The Kaplan–Meier curve for local recurrence free survival is provided in Fig. 1. No differences were observed in local recurrence free survival between low-risk tumours and high-risk tumours (low-risk 75.2% and high-risk 67.8%, HR = 1.41, 95% CI 0.65–3.06, p = 0.39) (Table 4).
Table 4.
Survival outcomes
| 3 year % | 5 year % | 5 year HR | 95% CI | p value | |
|---|---|---|---|---|---|
| Local recurrence free survival | |||||
| Overall | 81.0 | 77.1 | |||
| Low-risk pT1 | 76.7 | 75.2 | Ref | Ref | Ref |
| High-risk pT1 | 71.5 | 67.8 | 1.41 | 0.65–3.06 | 0.39 |
| Overall survival | |||||
| Overall | 91.1 | 82.0 | |||
| Low-risk pT1 | 93.4 | 82.7 | Ref | Ref | Ref |
| High-risk pT1 | 89.4 | 75.0 | 1.51 | 0.58–3.99 | 0.40 |
| Patients without local recurrence | 93.1 | 85.1 | Ref | Ref | Ref |
| Patients with local recurrence | 84.1 | 67.7 | 2.41 | 1.05–5.50 | 0.037 |
| Recurrences | |||||
| Normal surveillance with recurrence | 80.0 | 80.0 | Ref | Ref | Ref |
| Close surveillance with recurrence | 69.4 | 63.1 | 1.94 | 0.24–15.58 | 0.53 |
Fig. 1.
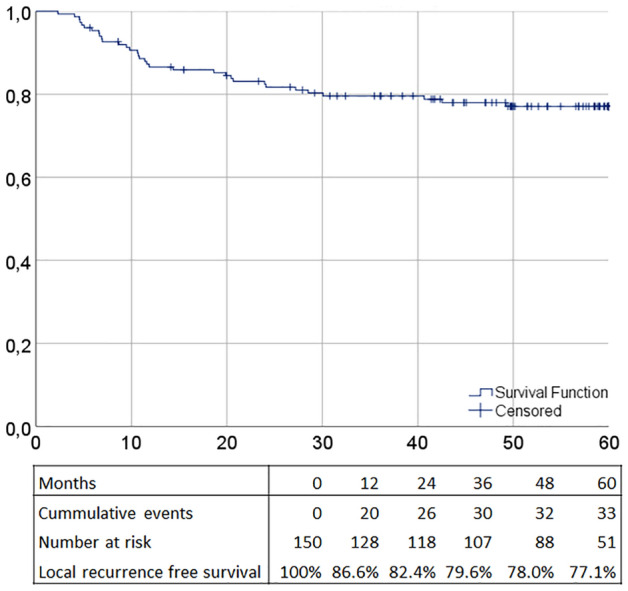
Kaplan–Meier curve of 60-month local recurrence free survival
OS
OS was 91.1 and 82.0% at 3 and 5 years of follow-up, respectively (Table 4, Supplementary Fig. 1). No significant associations between patient- and tumour characteristics and OS were found except for age (HR = 1.10 95% CI 1.05–1.15, p < 0.001). No differences in OS rates were observed between low- and high-risk pT1 tumours (Table 4). Moreover, in patients with local recurrence no differences in OS rates could be detected for normal follow-up or close surveillance (Table 4). Kaplan–Meier estimates suggested that patients with local recurrence had a worse 5-year OS compared to patients without local recurrence (67.6 vs. 86.0% respectively, p = 0.023, Supplementary Fig. 2). In depth landmark analyses showed that for patients with local recurrence at 6 and 36 months, OS was significantly lower (6 months: p = 0.034, 36 months: p = 0.036, Fig. 2). Even though the 24 months landmark showed a trend towards lower OS as well, similar outcomes could not be determined for the landmark of 12 months of follow-up (12 months: p = 0.31, 24 months: p = 0.071, Fig. 2).
Fig. 2.
Landmark analyses. A 6-month landmark. B 12-month landmark. C 24-month landmark. D 36-month landmark. In depth analyses of overall survival based on patients alive at the set landmark. The curves provide estimates for patients with- or without recurrence at the set landmark time
Discussion
In this cohort study of 150 patients with only locally excised pT1 rectal cancers there was a high local recurrence rate of 22.7%, with a median time to local recurrence of approximately 11 months. The majority of the local recurrences were detected endoluminally, local recurrence was associated with the occurrence of distant metastases, and approximately 80% of recurrences were eligible for salvage surgery. In addition, our results suggest that local recurrences may negatively impact OS.
After local excision of high-risk pT1 tumours, radical surgery is recommended to reduce the risk of local recurrence [1, 18]. A recent meta-analysis showed that overall local recurrence rates after local excision of high-risk pT1 tumours can be estimated to be 14% [7]. This conclusion is hampered by a lack of large cohort studies or clinically controlled trials and selection- and/or publication bias of the included cohort studies. It has been suggested, that the local recurrence rate may be slightly higher after surgical local excision compared with endoscopic, however, other studies do not support this outcome and high-quality evidence is lacking [19–21]. Nevertheless, the overall local recurrence rate of 22.7% in the current study is substantial, in particular since the majority of investigated tumours were low-risk tumours and surgery was performed by surgeons experienced in local excision. There may be several explanations for the local recurrence rate. First, due to incomplete reports, an underestimation of histopathological risk factors might be present in this cohort, which may be emphasized by the fact that we were unable to identify an association between high-risk tumours and local recurrence. Moreover, previous studies have shown that interobserver variability of histopathological risk factors is high, and over the past years the increasing evidence about the importance of identification of risk factors in early cancers has led to more detailed histopathology reports [22–24]. Another possible explanation might be the quality of the resected specimens. Nonetheless, 94.0% of the resections were complete (R0), and R-classification was not associated with local recurrence. In addition to involved or unevaluable resection margins, alternative hypotheses for the development of local recurrence, such as the influence of tissue handling, have been proposed [25]. The importance of avoiding fragmented resections seems evident, however, the role of tumour seeding might be underestimated [25, 26]. Tumour seeding theories propose that inadequate handling of tissue and instruments cause implantation of viable tumour cells into the damaged mucosa, which leads to local recurrence at the site of the excision [27]. These hypotheses do not agree with the general idea that recurrences after local excision mainly consist of unidentified lymph node involvement. The reported trend towards an association between the duration of the procedure and local recurrence might suggest a link between difficult resections and therefore tissue handling, and local recurrence. Moreover, the high proportion of endoluminal recurrences supports this theory. Although short-term outcomes of local excision techniques have been studied thoroughly, there seems to be room for further investigation into the influence of procedure related factors on local recurrence. Next to specimen quality and histopathological characteristics, surgical quality of the procedure including potential tumour spill might be an important risk factor for local recurrence. Future research is needed to address these factors and potentially improve oncological outcomes.
Approximately 80% of the patients with local recurrence were eligible for salvage surgery. In a small number of studies the percentage of patients eligible for salvage surgery in case of local recurrence was investigated and varied from 73 to 92%, which corresponds to our findings [28–32]. Few studies have investigated eligibility and outcomes of salvage surgery in recurrences after local excision. To establish accurate outcomes of patients with local recurrence after local excision of early rectal cancer, more robust data is necessary. Based on the limited evidence available, cancer specific and overall survival seems poor. Two smaller cohort studies reported 3- and 5- year cancer free survival of 58% and 53%, respectively. A systematic review by Jones and colleagues, described an overall survival rate of 50% after salvage treatment, conceivably due to the occurrence of distant metastases [33]. In adverse outcomes of early tumours, there may be an important role for tumour biology. However, it remains hard to identify and target these aggressive early tumours appropriately.
Even though this study reported a relatively high recurrence rate, the majority of patients with pT1 tumours will not get a recurrence, and local excision techniques are sufficient treatment for these patients. For this reason, the potential value of rectal preservation in early rectal cancer should be kept in mind. Treatment related morbidity and functional outcomes are of great importance to patients with rectal cancer [34, 35]. In addition, the trade-off between the risk of local recurrence and good functional outcomes may differ for each patient. These trade-offs cause patients and physicians to disregard recommendations of completion surgery. To provide patient tailored treatment, local excision followed by an assessment of risk factors seems appealing. In high-risk tumours additional treatment by completion surgery remains the standard. In particular given the reported high recurrence rate and the potential impact on survival in a patient population predominantly fit for additional treatment. A promising future perspective might be adjuvant chemoradiotherapy, which may provide better functional outcomes and less impact on quality of life compared with completion surgery, while maintaining an acceptable low risk of recurrence. Recently, a meta-analysis reported similar recurrence rates for adjuvant (chemo)radiotherapy and completion surgery in high-risk pT1 tumours. However, the current evidence on this treatment strategy is scarce and hampered by selection bias, therefore more long-term outcome data are necessary prior to implementation of this treatment strategy [7, 36].
One of the limitations of this study is caused by the condition of the included patients, which may have influenced overall survival. Although the majority of patients did not require completion surgery according to the guidelines, the patients with high-risk tumours either waived radical surgery or were clinically unfit to undergo it. However, at the time of the local excision, only 12 patients had a severe systemic disease, i.e. were American Society of Anesthesiologists (ASA) class III. Furthermore, due to the retrospective nature of the study some information could not be retrieved from the patient records. For example, in almost 20% of the cases we were unable to categorize the patient as having a low- or high-risk pT1 tumour, because risk factors were not reported in the histopathological report. Over time we observed a decrease in incomplete reports. Nonetheless, due to incomplete histopathological reports and known interobserver variability the true percentage of high-risk tumours may have been higher [22, 24]. In spite of the relatively high recurrence rate, only 34 patients had a local recurrence. The relatively small number of local recurrences may have caused some of the analyses to not reach statistical significance. Nevertheless, the current cohort is one of the largest cohorts that reports long-term outcomes of surgical local excision in pT1 tumours and counterbalances the optimistic outcomes of other studies [7].
Conclusions
After solely surgical local excision for pT1 rectal cancer, local recurrences were observed in 1 out of 5 patients. Salvage surgery was possible in 80% of these patients, but local recurrence may impact survival negatively. Therefore, this study does not support the optimistic outcomes of earlier studies and shows that close surveillance is necessary to diagnose and treat local recurrence in an early stage. However, for the majority of patients with early rectal cancer local excision will be sufficient, consequently this strategy may be an option in selected patients. Selection of these patients should be based on patient- and tumour characteristics and during the shared decision-making process patients should be informed of the risks associated with local recurrence.
Supplementary Information
Below is the link to the electronic supplementary material.
Supplementary figure 1 Kaplan-Meier curve of 60-month overall survival Supplementary file2 (TIF 5012 kb)
Supplementary figure 2 Kaplan-Meier curve, the influence of local recurrence on 60-month overall survival Supplementary file3 (TIF 5987 kb)
Acknowledgements
We would like to thank B.I. Lissenberg-Witte for her contribution and advice regarding the statistical analyses.
Funding
No funding was received for this study.
Declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval and informed consent
This research study was conducted retrospectively from data obtained for clinical purposes. The Human Investigation Committee (IRB) affiliated with the Laurentius Hospital and IJsselland Hospital approved this study. Since this retrospective study was performed under the Dutch Healthcare Quality, Complaints and Disputes Act informed consent was not required.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
J. W. A. Leijtens and L. J. H. Smits contributed equally.
References
- 1.Glynne-Jones R, Wyrwicz L, Tiret E, Brown G, Rodel C, Cervantes A, Arnold D, on behalf of the ESMO Guidelines Committee Rectal cancer: ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2017;28:22–40. doi: 10.1093/annonc/mdx224. [DOI] [PubMed] [Google Scholar]
- 2.Lezoche E, Paganini AM, Fabiani B, Balla A, Vestri A, Pescatori L, Scoglio D, D'Ambrosio G, Lezoche G. Quality-of-life impairment after endoluminal locoregional resection and laparoscopic total mesorectal excision. Surg Endosc. 2014;28:227–234. doi: 10.1007/s00464-013-3166-2. [DOI] [PubMed] [Google Scholar]
- 3.Sier MF, van Gelder L, Ubbink DT, Bemelman WA, Oostenbroek RJ. Factors affecting timing of closure and non-reversal of temporary ileostomies. Int J Colorectal Dis. 2015;30:1185–1192. doi: 10.1007/s00384-015-2253-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Traa MJ, Orsini RG, Den Oudsten BL, De Vries J, Roukema JA, Bosman SJ, Dudink RL, Rutten HJT. Measuring the health-related quality of life and sexual functioning of patients with rectal cancer: does type of treatment matter? Int J Cancer. 2014;134:979–987. doi: 10.1002/ijc.28430. [DOI] [PubMed] [Google Scholar]
- 5.Borstlap WAA, van Oostendorp SE, Klaver CEL, Hahnloser D, Cunningham C, Rullier E, Bemelman WA, Tuynman JB, Tanis PJ, on behalf of the Research Committee of the European Society of Coloproctology Organ preservation in rectal cancer: a synopsis of current guidelines. Colorectal Dis. 2017 doi: 10.1111/codi.13960. [DOI] [PubMed] [Google Scholar]
- 6.van Groningen JT, van Hagen P, Tollenaar RAEM, Tuynman JB, de Mheen PJ, Doornebosch PG, Tanis PJ, de Graaf EJR, on behalf of the Dutch Colorectal Audit Evaluation of a completion total mesorectal excision in patients after local excision of rectal cancer: a word of caution. J Natl Compr Canc Netw. 2018;16:822–828. doi: 10.6004/jnccn.2018.7026. [DOI] [PubMed] [Google Scholar]
- 7.van Oostendorp SE, Smits LJH, Vroom Y, Detering R, Heymans MW, Moons LMG, Tanis PJ, de Graaf EJR, Cunningham C, Denost Q, Kusters M, Tuynman JB. Local recurrence after local excision of early rectal cancer: a meta-analysis of completion TME, adjuvant (chemo)radiation, or no additional treatment. Br J Surg. 2020;107:1719–1730. doi: 10.1002/bjs.12040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Detering R, van Oostendorp SE, Meyer VM, van Dieren S, Bos ACRK, Dekker JWT, Reerink O, van Waesberghe JHTM, Marijnen CAM, Moons LMG, Beets-Tan RGH, Hompes R, van Westreenen HL, Tanis PJ, Tuynman JB, Dutch ColoRectal Audit Group MRI cT1-2 rectal cancer staging accuracy: a population-based study. Br J Surg. 2020;107:1372–1382. doi: 10.1002/bjs.11590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Dutch guideline colorectal carcinoma. https://richtlijnendatabase.nl/richtlijn/colorectaal_carcinoom_crc/startpagina_-_crc.html. Accessed 13 May 2022.
- 10.Buess G, Hutterer F, Theiss J, Böbel M, Isselhard W, Pichlmaier H. A system for a transanal endoscopic rectum operation. Chirurg. 1984;55:677–680. [PubMed] [Google Scholar]
- 11.Atallah S, Albert M, Larach S. Transanal minimally invasive surgery: a giant leap forward. Surg Endosc. 2010;24:2200–2205. doi: 10.1007/s00464-010-0927-z. [DOI] [PubMed] [Google Scholar]
- 12.Dindo D, Demartines N, Clavien PA. Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg. 2004;240:205–213. doi: 10.1097/01.sla.0000133083.54934.ae. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Zwager LW, Bastiaansen BAJ, Montazeri NSM, Hompes R, Barresi V, Ichimasa K, Kawachi H, Machado I, Masaki T, Sheng W, Tanaka S, Togashi K, Yasue C, Fockens P, Moons LMG, Dekker E. Deep submucosal invasion is not an independent risk factor for lymph node metastasis in T1 colorectal cancer: a meta-analysis. Gastroenterology. 2022 doi: 10.1053/j.gastro.2022.04.010. [DOI] [PubMed] [Google Scholar]
- 14.Van Houwelingen HC. Dynamic prediction by landmarking in event history analysis. Scand J Stat. 2007;34:70–85. doi: 10.1111/j.1467-9469.2006.00529.x. [DOI] [Google Scholar]
- 15.Morgan CJ. Landmark analysis: a primer. J Nucl Cardiol. 2019;26:391–393. doi: 10.1007/s12350-019-01624-z. [DOI] [PubMed] [Google Scholar]
- 16.Dafni U. Landmark analysis at the 25-year landmark point. Circ Cardiovasc Qual Outcomes. 2011;4:363–371. doi: 10.1161/CIRCOUTCOMES.110.957951. [DOI] [PubMed] [Google Scholar]
- 17.Anderson JR, Cain KC, Gelber RD. Analysis of survival by tumor response. J Clin Oncol. 1983;1:710–719. doi: 10.1200/jco.1983.1.11.710. [DOI] [PubMed] [Google Scholar]
- 18.Morino M, Risio M, Bach S, Beets-Tan R, Bujko K, Panis Y, Quirke P, Rembacken B, Rullier E, Saito Y, Young-Fadok T, Allaix ME. Early rectal cancer: the European association for endoscopic surgery (EAES) clinical consensus conference. Surg Endosc. 2015;29:755–773. doi: 10.1007/s00464-015-4067-3. [DOI] [PubMed] [Google Scholar]
- 19.Wang S, Gao S, Yang W, Guo S, Li Y. Endoscopic submucosal dissection versus local excision for early rectal cancer: a systematic review and meta-analysis. Tech Coloproctol. 2016;20:1–9. doi: 10.1007/s10151-015-1383-5. [DOI] [PubMed] [Google Scholar]
- 20.McCarty TR, Bazarbashi AN, Hathorn KE, Thompson CC, Aihara H. Endoscopic submucosal dissection (ESD) versus transanal endoscopic microsurgery (TEM) for treatment of rectal tumors: a comparative systematic review and meta-analysis. Surg Endosc. 2020;34:1688–1695. doi: 10.1007/s00464-019-06945-1. [DOI] [PubMed] [Google Scholar]
- 21.Sagae VMT, Ribeiro IB, de Moura DTH, Brunaldi VO, Logiudice FP, Funari MP, Baba ER, Bernardo WM, de Moura EGH. Endoscopic submucosal dissection versus transanal endoscopic surgery for the treatment of early rectal tumor: a systematic review and meta-analysis. Surg Endosc. 2020;34:1025–1034. doi: 10.1007/s00464-019-07271-2. [DOI] [PubMed] [Google Scholar]
- 22.Davenport A, Morris J, Pritchard SA, Salmo E, Scott M, Haboubi NY. Interobserver variability amongst gastrointestinal pathologists in assessing prognostic parameters of malignant colorectal polyps: a cause for concern. Tech Coloproctol. 2016;20:647–652. doi: 10.1007/s10151-016-1513-8. [DOI] [PubMed] [Google Scholar]
- 23.Ueno H, Hase K, Hashiguchi Y, Shimazaki H, Yoshii S, Kudo SE, Tanaka M, Akagi Y, Suto T, Nagata S, Matsuda K, Komori K, Yoshimatsu K, Tomita Y, Yokoyama S, Shinto E, Nakamura T, Sugihara K. Novel risk factors for lymph node metastasis in early invasive colorectal cancer: a multi-institution pathology review. J Gastroenterol. 2014;49:1314–1323. doi: 10.1007/s00535-013-0881-3. [DOI] [PubMed] [Google Scholar]
- 24.Smits LJH, Vink-Borger E, van Lijnschoten G, Focke-Snieders I, van der Post RS, Tuynman JB, van Grieken NCT, Nagtegaal ID. Diagnostic variability in the histopathological assessment of advanced colorectal adenomas and early colorectal cancer in a screening population. Histopathology. 2022;80:790–798. doi: 10.1111/his.14601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Albert MR, Kaminsky P. Surgical technique for local excision of rectal neoplasia. In: Atallah S, editor. Transanal minimally invasive surgery (TAMIS) and transanal total mesorectal excision (taTME) AG, Cham, Switzerland: Springer; 2019. pp. 89–95. [Google Scholar]
- 26.Junginger T, Goenner U, Hitzler M, Trinh TT, Heintz A, Roth W, Blettner M, Wollschlaeger D. Analysis of local recurrences after transanal endoscopic microsurgery for low risk rectal carcinoma. Int J Colorectal Dis. 2017;32:265–271. doi: 10.1007/s00384-016-2715-2. [DOI] [PubMed] [Google Scholar]
- 27.Long RTL, Edwards RH. Implantation metastasis as a cause of local recurrence of colorectal carcinoma. Am J Surg. 1989;157:194–201. doi: 10.1016/0002-9610(89)90527-8. [DOI] [PubMed] [Google Scholar]
- 28.Doornebosch PG, Ferenschild FTJ, de Wilt JHW, Dawson I, Tetteroo GW, de Graaf EJR. Treatment of recurrence after transanal endoscopic microsurgery (TEM) for T1 rectal cancer. Dis Colon Rectum. 2010;53:1234–1239. doi: 10.1007/DCR.0b013e3181e73f33. [DOI] [PubMed] [Google Scholar]
- 29.Vaid S, Park JS, Sinott RJ. Outcomes of recurrent rectal cancer after transanal excision. Am Surg. 2016;82:152–155. doi: 10.1177/000313481608200219. [DOI] [PubMed] [Google Scholar]
- 30.Stipa F, Giaccaglia V, Burza A. Management and outcome of local recurrence following transanal endoscopic microsurgery for rectal cancer. Dis Colon Rectum. 2012;55:262–269. doi: 10.1097/DCR.0b013e318241ef22. [DOI] [PubMed] [Google Scholar]
- 31.Christoforidis D, Cho HM, Dixon MR, Mellgren AF, Madoff RD, Finne CO. Transanal endoscopic microsurgery versus conventional transanal excision for patients with early rectal cancer. Ann Surg. 2009;249:776–782. doi: 10.1097/SLA.0b013e3181a3e54b. [DOI] [PubMed] [Google Scholar]
- 32.Bikhchandani J, Ong GK, Dozois EJ, Mathis KL. Outcomes of salvage surgery for cure in patients with locally recurrent disease after local excision of rectal cancer. Dis Colon Rectum. 2015;58:283–287. doi: 10.1097/DCR.0000000000000283. [DOI] [PubMed] [Google Scholar]
- 33.Jones HJS, Cunningham C, Nicholson GA, Hompes R. Outcomes following completion and salvage surgery for early rectal cancer: a systematic review. Eur J Surg Oncol. 2018;44:15–23. doi: 10.1016/j.ejso.2017.10.212. [DOI] [PubMed] [Google Scholar]
- 34.Kennedy ED, Borowiec AM, Schmocker S, Cho C, Brierley J, Li S, Victor JC, Baxter NN. Patient and physician preferences for nonoperative management for low rectal cancer: is it a reasonable treatment option? Dis Colon Rectum. 2018;61:1281–1289. doi: 10.1097/DCR.0000000000001166. [DOI] [PubMed] [Google Scholar]
- 35.van der Valk MJM, Hilling DE, Bastiaannet E, Meershoek-Klein Kranenbarg E, Beets GL, Figueiredo NL, Habr-Gama A, Perez RO, Renehan AG, van de Velde CJH, the IWWD Consortium Long-term outcomes of clinical complete responders after neoadjuvant treatment for rectal cancer in the international watch & wait database (IWWD): an international multicentre registry study. The Lancet. 2018;391:2537–2545. doi: 10.1016/s0140-6736(18)31078-x. [DOI] [PubMed] [Google Scholar]
- 36.Borstlap WA, Tanis PJ, Koedam TWA, Marijnen CAM, Cunningham C, Dekker E, van Leerdam ME, Meijer G, van Grieken N, Nagtegaal ID, Punt CJA, Dijkgraaf MGW, De Wilt JH, Beets G, de Graaf EJ, van Geloven AA, Gerhards MF, van Westreenen HL, van de Ven AWH, van Duijvendijk P, de Hingh IHJT, Leijtens JWA, Sietses C, Spillenaar-Bilgen EJ, Vuylsteke RJCLM, Hoff C, Burger JWA, van Grevenstein WMU, Pronk A, Bosker RJI, Prins H, Smits AB, Bruin S, Zimmerman DD, Stassen LPS, Dunker MS, Westerterp M, Coene PP, Stoot J, Bemelman WA, Tuynman JB. A multi-centred randomised trial of radical surgery versus adjuvant chemoradiotherapy after local excision for early rectal cancer. BMC Cancer. 2016;16:513. doi: 10.1186/s12885-016-2557-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary figure 1 Kaplan-Meier curve of 60-month overall survival Supplementary file2 (TIF 5012 kb)
Supplementary figure 2 Kaplan-Meier curve, the influence of local recurrence on 60-month overall survival Supplementary file3 (TIF 5987 kb)