Abstract
The American Diabetes Association (ADA) “Standards of Care in Diabetes” includes the ADA’s current clinical practice recommendations and is intended to provide the components of diabetes care, general treatment goals and guidelines, and tools to evaluate quality of care. Members of the ADA Professional Practice Committee, a multidisciplinary expert committee, are responsible for updating the Standards of Care annually, or more frequently as warranted. For a detailed description of ADA standards, statements, and reports, as well as the evidence-grading system for ADA’s clinical practice recommendations and a full list of Professional Practice Committee members, please refer to Introduction and Methodology. Readers who wish to comment on the Standards of Care are invited to do so at professional.diabetes.org/SOC.
Person-Centered Collaborative Care
Recommendations
4.1 A person-centered communication style that uses person-centered, culturally sensitive, and strength-based language and active listening; elicits individual preferences and beliefs; and assesses literacy, numeracy, and potential barriers to care should be used to optimize health outcomes and health-related quality of life. B
4.2 People with diabetes can benefit from a coordinated multidisciplinary team that may include and is not limited to diabetes care and education specialists, primary care and subspecialty clinicians, nurses, registered dietitian nutritionists, exercise specialists, pharmacists, dentists, podiatrists, and mental health professionals. E
A successful medical evaluation depends on beneficial interactions between the person with diabetes and the care team. The Chronic Care Model (1–3) (see Section 1, “Improving Care and Promoting Health in Populations”) is a person-centered approach to care that requires a close working relationship between the person with diabetes and clinicians involved in treatment planning. People with diabetes should receive health care from a coordinated interdisciplinary team that may include but is not limited to diabetes care and education specialists, primary care and subspecialty clinicians, nurses, registered dietitian nutritionists, exercise specialists, pharmacists, dentists, podiatrists, and mental health professionals. Individuals with diabetes must assume an active role in their care. Based on the preferences of the person with diabetes, the family or support group and health care team together formulate the management plan, which includes lifestyle management (see Section 5, “Facilitating Positive Health Behaviors and Well-being to Improve Health Outcomes”).
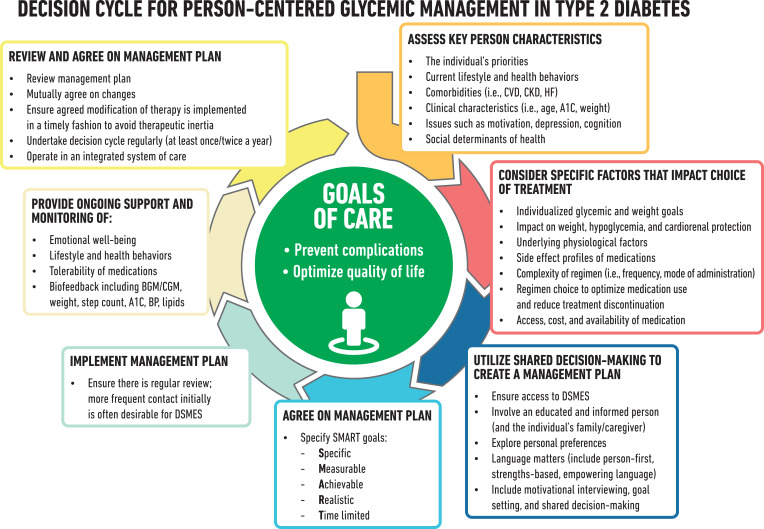
The goals of treatment for diabetes are to prevent or delay complications and optimize quality of life (Fig. 4.1). Treatment goals and plans should be created with people with diabetes based on their individual preferences, values, and goals. This individualized management plan should take into account the person’s age, cognitive abilities, school/work schedule and conditions, health beliefs, support systems, eating patterns, physical activity, social situation, financial concerns, cultural factors, literacy and numeracy (mathematical literacy), diabetes history (duration, complications, current use of medications), comorbidities, disabilities, health priorities, other medical conditions, preferences for care, and life expectancy. Various strategies and techniques should be used to support the person’s self-management efforts, including providing education on problem-solving skills for all aspects of diabetes management.
Figure 4.1.
Decision cycle for person-centered glycemic management in type 2 diabetes. Adapted from Davies et al. (246). BGM, blood glucose monitoring; BP, blood pressure; CGM, continuous glucose monitoring; CKD, chronic kidney disease; CVD, atherosclerotic cardiovascular disease; DSMES, diabetes self-management education and support; HF, heart failure.
Health care professional communication with people with diabetes and families should acknowledge that multiple factors impact glycemic management but also emphasize that collaboratively developed treatment plans and a healthy lifestyle can significantly improve disease outcomes and well-being (4–8). Thus, the goal of communication between health care professionals and people with diabetes is to establish a collaborative relationship and to assess and address self-management barriers without blaming people with diabetes for “noncompliance” or “nonadherence” when the outcomes of self-management are not optimal (9). The familiar terms “noncompliance” and “nonadherence” denote a passive, obedient role for a person with diabetes in “following doctor’s orders” that is at odds with the active role people with diabetes take in directing the day-to-day decision-making, planning, monitoring, evaluation, and problem-solving involved in diabetes self-management. Using a nonjudgmental approach that normalizes periodic lapses in management may help minimize the person’s resistance to reporting problems with self-management. Empathizing and using active listening techniques, such as open-ended questions, reflective statements, and summarizing what the person said, can help facilitate communication. Perceptions of people with diabetes about their own ability, or self-efficacy, to self-manage diabetes constitute one important psychosocial factor related to improved diabetes self-management and treatment outcomes in diabetes (10–12) and should be a target of ongoing assessment, education, and treatment planning.
Language has a strong impact on perceptions and behavior. The use of empowering language in diabetes care and education can help to inform and motivate people, yet language that shames and judges may undermine this effort. The American Diabetes Association (ADA) and the Association of Diabetes Care & Education Specialists (formerly called the American Association of Diabetes Educators) joint consensus report, “The Use of Language in Diabetes Care and Education,” provides the authors’ expert opinion regarding the use of language by health care professionals when speaking or writing about diabetes for people with diabetes or for professional audiences (13). Although further research is needed to address the impact of language on diabetes outcomes, the report includes five key consensus recommendations for language use:
Use language that is neutral, nonjudgmental, and based on facts, actions, or physiology/biology.
Use language free from stigma.
Use language that is strength based, respectful, and inclusive and that imparts hope.
Use language that fosters collaboration between people with diabetes and health care professionals.
Use language that is person centered (e.g., “person with diabetes” is preferred over “diabetic”).
Comprehensive Medical Evaluation
Recommendations
- 4.3 A complete medical evaluation should be performed at the initial visit to:
- Confirm the diagnosis and classify diabetes. A
- Evaluate for diabetes complications, potential comorbid conditions, and overall health status. A
- Review previous treatment and risk factor management in people with established diabetes. A
- Begin engagement with the person with diabetes in the formulation of a care management plan including initial goals of care. A
- Develop a plan for continuing care. A
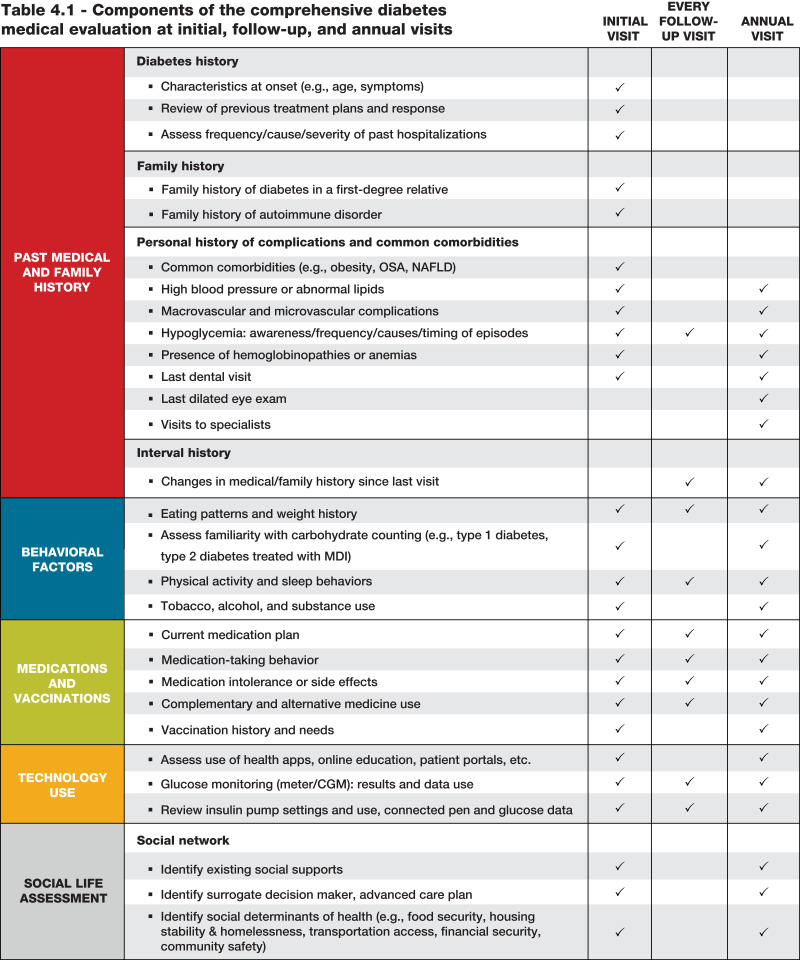
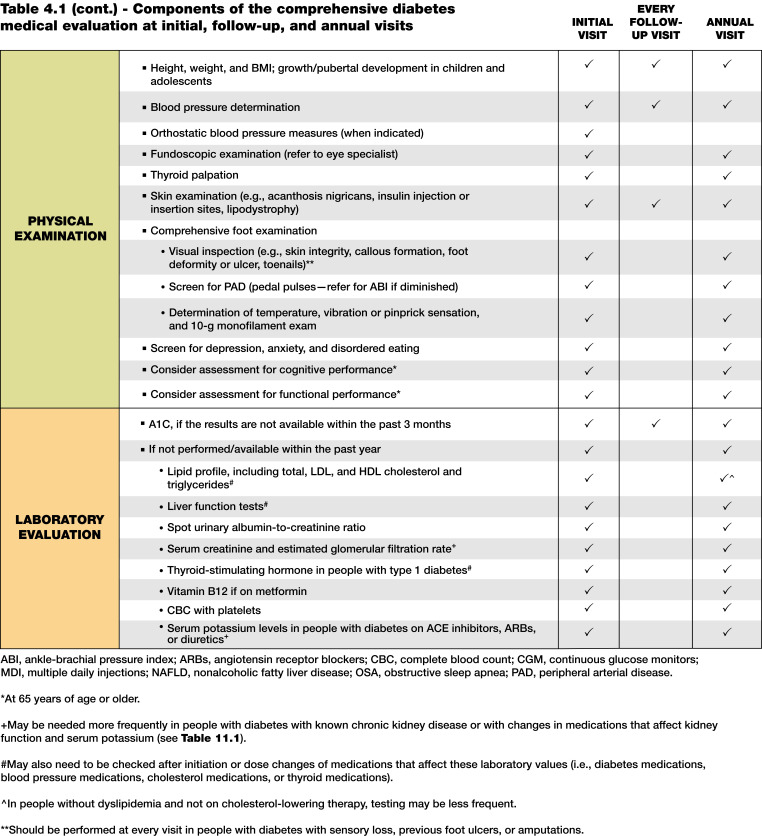
4.4 A follow-up visit should include most components of the initial comprehensive medical evaluation (Table 4.1). A
4.5 Ongoing management should be guided by the assessment of overall health status, diabetes complications, cardiovascular risk, hypoglycemia risk, and shared decision-making to set therapeutic goals. B
Table 4.1.
Components of the comprehensive diabetes medical evaluation at initial, follow-up, and annual visits
The comprehensive medical evaluation includes the initial and follow-up evaluations, assessment of complications, psychosocial assessment, management of comorbid conditions, overall health status, and engagement of the person with diabetes throughout the process. While a comprehensive list is provided in Table 4.1, in clinical practice the health care professional may need to prioritize the components of the medical evaluation given the available resources and time. The goal is to provide the health care team information so it can optimally support people with diabetes. In addition to the medical history, physical examination, and laboratory tests, health care professionals should assess diabetes self-management behaviors, nutrition, social determinants of health, and psychosocial health (see Section 5, “Facilitating Positive Health Behaviors and Well-being to Improve Health Outcomes”) and give guidance on routine immunizations. The assessment of sleep pattern and duration should be considered; a meta-analysis found that poor sleep quality, short sleep, and long sleep were associated with higher A1C in people with type 2 diabetes (14). Interval follow-up visits should occur at least every 3–6 months individualized to the person and then at least annually.
Lifestyle management and psychosocial care are the cornerstones of diabetes management. People with diabetes should be referred for diabetes self-management education and support, medical nutrition therapy, and assessment of psychosocial/emotional health concerns if indicated. People with diabetes should receive recommended preventive care services (e.g., immunizations, cancer screening); smoking cessation counseling; and ophthalmological, dental, and podiatric referrals, as needed.
The assessment of risk of acute and chronic diabetes complications and treatment planning are key components of initial and follow-up visits (Table 4.2). The risk of atherosclerotic cardiovascular disease and heart failure (see Section 10, “Cardiovascular Disease and Risk Management”), chronic kidney disease staging (see Section 11, “Chronic Kidney Disease and Risk Management”), presence of retinopathy (see Section 12, “Retinopathy, Neuropathy, and Foot Care”), and risk of treatment-associated hypoglycemia (Table 4.3) should be used to individualize targets for glycemia (see Section 6, “Glycemic Targets”), blood pressure, and lipids and to select specific glucose-lowering medication (see Section 9, “Pharmacologic Approaches to Glycemic Treatment”), antihypertension medication, and statin treatment intensity.
Table 4.2.
Assessment and treatment plan*
| Assessing risk of diabetes complications |
| • ASCVD and heart failure history |
| • ASCVD risk factors and 10-year ASCVD risk assessment |
| • Staging of chronic kidney disease (see Table 11.1) |
| • Hypoglycemia risk (see Table 4.3) |
| • Assessment for retinopathy |
| • Assessment for neuropathy |
| Goal setting |
| • Set A1C/blood glucose/time-in-range target |
| • If hypertension is present, establish blood pressure target |
| • Diabetes self-management goals |
| Therapeutic treatment plans |
| • Lifestyle management |
| • Pharmacologic therapy: glucose lowering |
| • Pharmacologic therapy: cardiovascular and renal disease risk factors |
| • Use of glucose monitoring and insulin delivery devices |
| • Referral to diabetes education and medical specialists (as needed) |
ASCVD, atherosclerotic cardiovascular disease.
Assessment and treatment planning are essential components of initial and all follow-up visits.
Table 4.3.
Assessment of hypoglycemia risk
| Factors that increase risk of treatment-associated hypoglycemia |
| • Use of insulin or insulin secretagogues (i.e., sulfonylureas, meglitinides) |
| • Impaired kidney or hepatic function |
| • Longer duration of diabetes |
| • Frailty and older age |
| • Cognitive impairment |
| • Impaired counterregulatory response, hypoglycemia unawareness |
| • Physical or intellectual disability that may impair behavioral response to hypoglycemia |
| • Alcohol use |
| • Polypharmacy (especially ACE inhibitors, angiotensin receptor blockers, nonselective β-blockers) |
| • History of severe hypoglycemic event |
| In addition to individual risk factors, consider use of comprehensive risk prediction models (233). |
Additional referrals should be arranged as necessary (Table 4.4). Clinicians should ensure that people with diabetes are appropriately screened for complications and comorbidities. Discussing and implementing an approach to glycemic management with the person is a part, not the sole goal, of the clinical encounter.
Table 4.4.
Referrals for initial care management
| • Eye care professional for annual dilated eye exam |
| • Family planning for individuals of childbearing potential |
| • Registered dietitian nutritionist for medical nutrition therapy |
| • Diabetes self-management education and support |
| • Dentist for comprehensive dental and periodontal examination |
| • Mental health professional, if indicated |
| • Audiology, if indicated |
| • Social worker/community resources, if indicated |
Immunizations
Recommendation
4.6 Provide routinely recommended vaccinations for children and adults with diabetes as indicated by age (see Table 4.5 for highly recommended vaccinations for adults with diabetes). A
Table 4.5.
Highly recommended immunizations for adults with diabetes (Advisory Committee on Immunization Practices, Centers for Disease Control and Prevention)
| Vaccination | Age-group recommendations | Frequency | GRADE evidence type* | Reference |
|---|---|---|---|---|
| Hepatitis B | <60 years of age; ≥60 years of age discuss with health care professionals | Two- or three-dose series | 2 | Centers for Disease Control and Prevention, Use of Hepatitis B Vaccination for Adults With Diabetes Mellitus: Recommendations of the Advisory Committee on Immunization Practices (ACIP) (239) |
| Human papilloma virus (HPV) | ≤26 years of age; 27–45 years of age may also be vaccinated against HPV after a discussion with health care professionals | Three doses over 6 months | 2 for female individuals, 3 for male individuals | Meites et al., Human Papillomavirus Vaccination for Adults: Updated Recommendations of the Advisory Committee on Immunization Practices (240) |
| Influenza | All people with diabetes advised not to receive live attenuated influenza vaccine | Annual | — | Demicheli et al., Vaccines for Preventing Influenza in the Elderly (241) |
| Pneumonia (PPSV23 [Pneumovax]) | 19–64 years of age, vaccinate with Pneumovax | One dose is recommended for those that previously received PCV13. If PCV15 used, follow with PPSV23 ≥1 year later. PPSV23 is not indicated after PCV20. Adults who received only PPSV23 may receive PCV15 or PCV20 ≥1 year after their last dose. | 2 | Centers for Disease Control and Prevention, Updated Recommendations for Prevention of Invasive Pneumococcal Disease Among Adults Using the 23-Valent Pneumococcal Polysaccaride Vaccine (PPSV23) (242) |
| ≥65 years of age | One dose is recommended for those that previously received PCV13. If PCV15 was used, follow with PPSV23 ≥1 year later. PPSV23 is not indicated after PCV20. Adults who received only PPSV23 may receive PCV15 or PCV20 ≥1 year after their last dose. | 2 | Falkenhorst et al., Effectiveness of the 23-Valent Pneumococcal Polysaccharide Vaccine (PPV23) Against Pneumococcal Disease in the Elderly: Systematic Review and Meta-analysis (243) | |
| PCV20 or PCV15 | Adults 19–64 years of age, with an immunocompromising condition (e.g., chronic renal failure), cochlear implant, or cerebrospinal fluid leak | One dose of PCV15 or PCV20 is recommended by the CDC. | 3 | Kobayashi et al., Use of 15-Valent Pneumococcal Conjugate Vaccine and 20-Valent Pneumococcal Conjugate Vaccine Among U.S. Adults: Updated Recommendations of the Advisory Committee on Immunization Practices—United States, 2022 (22) |
| 19–64 years of age, immunocompetent | For those who have never received any pneumococcal vaccine, the CDC recommends one dose of PCV15 or PCV20. | |||
| ≥65 years of age, immunocompetent, have shared decision-making discussion with health care professionals | One dose of PCV15 or PCV20. PCSV23 may be given ≥8 weeks after PCV15. PPSV23 is not indicated after PCV20. | |||
| Tetanus, diphtheria, pertussis (TDAP) | All adults; pregnant individuals should have an extra dose | Booster every 10 years | 2 for effectiveness, 3 for safety | Havers et al., Use of Tetanus Toxoid, Reduced Diphtheria Toxoid, and Acellular Pertussis Vaccines: Updated Recommendations of the Advisory Committee on Immunization Practices—United States, 2019 (244) |
| Zoster | ≥50 years of age | Two-dose Shingrix, even if previously vaccinated | 1 | Dooling et al., Recommendations of the Advisory Committee on Immunization Practices for Use of Herpes Zoster Vaccines (245) |
GRADE, Grading of Recommendations Assessment, Development, and Evaluation; PCV13, 13-valent pneumococcal conjugate vaccine; PCV15, 15-valent pneumococcal conjugate vaccine; PCV 20, 20-valent pneumococcal conjugate vaccine; PPSV23, 23-valent pneumococcal polysaccharide vaccine.
Evidence type: 1, randomized controlled trials (RCTs) or overwhelming evidence from observational studies; 2, RCTs with important limitations or exceptionally strong evidence from observational studies; 3, observational studies or RCTs with notable limitations; 4, clinical experience and observations, observational studies with important limitations, or RCTs with several major limitations. For a comprehensive list, refer to the Centers for Disease Control and Prevention (CDC) at cdc.gov/vaccines/.
The importance of routine vaccinations for people living with diabetes has been elevated by the coronavirus disease 2019 (COVID-19) pandemic. Preventing avoidable infections not only directly prevents morbidity but also reduces hospitalizations, which may additionally reduce risk of acquiring infections such as COVID-19. Children and adults with diabetes should receive vaccinations according to age-appropriate recommendations (15,16). The Centers for Disease Control and Prevention (CDC) provides vaccination schedules specifically for children, adolescents, and adults with diabetes (cdc.gov/vaccines/). The CDC Advisory Committee on Immunization Practices (ACIP) makes recommendations based on its own review and rating of the evidence, provided in Table 4.5 for selected vaccinations. The ACIP evidence review has evolved over time with the adoption of Grading of Recommendations Assessment, Development and Evaluation (GRADE) in 2010 and then the Evidence to Decision or Evidence to Recommendation frameworks in 2018 (17). Here we discuss the particular importance of specific vaccines.
Influenza
Influenza is a common, preventable infectious disease associated with high mortality and morbidity in vulnerable populations, including youth, older adults, and people with chronic diseases. Influenza vaccination in people with diabetes has been found to significantly reduce influenza and diabetes-related hospital admissions (18). In people with diabetes and cardiovascular disease, influenza vaccine has been associated with lower risk of all-cause mortality, cardiovascular mortality, and cardiovascular events (19). Given the benefits of the annual influenza vaccination, it is recommended for all individuals ≥6 months of age who do not have a contraindication. Influenza vaccination is critically important as the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) and influenza viruses will both be active in the U.S. during the 2022–2023 season (20). The live attenuated influenza vaccine, which is delivered by nasal spray, is an option for people who are age 2 years through age 49 years and who are not pregnant, but people with chronic conditions such as diabetes are cautioned against taking the live attenuated influenza vaccine and are instead recommended to receive the inactive or recombinant influenza vaccination. For individuals ≥65 years of age, there may be additional benefit from the high-dose quadrivalent inactivated influenza vaccine (20).
Pneumococcal Pneumonia
Like influenza, pneumococcal pneumonia is a common, preventable disease. People with diabetes are at increased risk for the bacteremic form of pneumococcal infection and have been reported to have a high risk of nosocomial bacteremia, with a mortality rate as high as 50% (21). There are two types of vaccines available in the U.S., pneumococcal conjugate vaccines (PCV13, PCV15, and PCV20) and pneumococcal polysaccharide vaccine (PPSV23), with distinct schedules for children and adults.
It is recommended that all children receive a four-dose series of PCV13 or PCV15 by 15 months of age. For children with diabetes who have incomplete series by ages 2–5 years, the CDC recommends a catch-up schedule to ensure that these children have four doses. Children with diabetes between 6 and 18 years of age are also advised to receive one dose of PPSV23, preferably after receipt of PCV13.
Adults aged ≥65 years whose vaccine status is unknown or who have not received pneumococcal vaccine should receive one dose of PCV15 or PCV20. If PCV15 is used, it should be followed by PPSV23.
Adults aged 19–64 years with certain underlying risk factors or other medical conditions whose vaccine status is unknown or who have not received pneumococcal vaccine should receive one dose of PCV15 or PCV20. As for adults aged ≥65 years, if PCV15 is used, it should be followed by PPSV23.
The recommended interval between PCV15 and PPSV23 is ≥1 year. If PPSV23 is the only dose received, PCV15 or PCV20 may be given ≥1 year later.
For adults with immunocompromising conditions, cochlear implant, or cerebrospinal fluid leak, a minimum interval of 8 weeks can be considered for dosing of PCV15 and PPSV23 when PCV15 has been used.
Adults who received PCV13 should follow the previously recommended PPSV23 series. Adults who received only PPSV23 may receive a PCV15 or PCV20 ≥1 year after their last dose.
Hepatitis B
Compared with the general population, people with type 1 or type 2 diabetes have higher rates of hepatitis B. This may be due to contact with infected blood or through improper equipment use (glucose monitoring devices or infected needles). Because of the higher likelihood of transmission, hepatitis B vaccine is recommended for adults with diabetes aged <60 years. For adults aged ≥60 years, hepatitis B vaccine may be administered at the discretion of the treating clinician based on the person’s likelihood of acquiring hepatitis B infection.
COVID-19
As of September 2022, the COVID-19 vaccines are recommended for all adults and some children, including people with diabetes, under approval from the U.S. Food and Drug Administration (FDA) (24). The bivalent booster protecting against the omicron variant and original strain has now replaced the monovalent booster for many.
For people 6 months to 17 years of age, most can receive the monovalent Moderna vaccine doses 1 and 2 at least 4–8 weeks apart. For those who are moderately or severely immunocompromised, doses 1 and 2 and doses 2 and 3 should be at least 4 weeks apart.
For the Pfizer-BioNTech monovalent vaccine for most people aged 6 months to 4 years, doses 1 and 2 should be at least 3–8 weeks apart and doses 2 and 3 at least 8 weeks apart. For those aged 6 months to 4 years who are moderately or severely compromised, doses 1 and 2 should be at least 4 weeks apart and doses 2 and 3 at least 4 weeks apart. For most people aged 5–11 years, doses 1 and 2 should be at least 3–8 weeks apart and doses 2 and 3 at least 5 months apart. For those who are moderately or severely immunocompromised, doses 1 and 2 should be at least 3 weeks apart and doses 2 and 3 should be at least 8 weeks apart. For most people aged 12–17 years, doses 1 and 2 should be at least 3–8 weeks apart. For those who are moderately to severely immunocompromised, doses 1 and 2 should be at least 3 weeks apart and doses 2 and 3 should be at least 4 weeks apart.
For the Novavax vaccine, for most people over 12 years of age, doses 1 and 2 should be at least 3–8 weeks apart. For those who are moderately to severely immunocompromised, doses 1 and 2 should be at least 3 weeks apart. For most people aged ≥18 years receiving the Moderna vaccine, doses 1 and 2 should be at least 4–8 weeks apart. For those who are moderately or severely compromised, doses 1 and 2 should be at least 4 weeks apart and doses 2 and 3 at least 4 weeks apart. For most people receiving the Pfizer-BioNTech vaccine, doses 1 and 2 should be at least 3–8 weeks apart. For those who are moderately or severely compromised, doses 1 and 2 should be at least 3 weeks apart and doses 2 and 3 at least 4 weeks apart.
For most people aged ≥18 years receiving Novavax vaccine, doses 1 and 2 should be at least 3–8 weeks apart. For those who are moderately to severely compromised, doses 1 and 2 should be at least 3 weeks apart. The Janssen monovalent vaccine is currently authorized for use in certain limited situations due to safety considerations.
For most people 12–17 years of age who received the Moderna vaccine, the Pfizer-BioNTech bivalent booster may be given at least 8 weeks from doses 2 and 3. For those moderately or severely compromised, doses 3 and 4 should be at least 8 weeks apart.
For most people aged 12–17 years who received the Pfizer-BioNTech vaccine, the Pifzer-BioNTech bivalent booster may be given at least 8 weeks from doses 2 and 3. For those moderately or severely compromised, doses 3 and 4 should be at least 8 weeks apart.
For most people aged ≥12 years receiving the Novavax vaccine, the Pfizer-BioNTech bivalent booster may be given as doses 2 and 3 at least 8 weeks apart. For those moderately to severely immunocompromised, doses 2 and 3 should be given at least 8 weeks apart.
Those ≥18 years of age receiving the Moderna vaccine may be given the Moderna bivalent booster 8 weeks after their last dose. Those ≥18 years of age receiving the Pfizer-BioNTech vaccine may receive the Pfizer-BioNTech bivalent booster 8 weeks after their last dose. Those receiving the Janssen vaccine may receive the Moderna or Pfizer-BioNTech bivalent booster 8 weeks after their last dose. Those receiving the Novavax vaccine aged ≥12 years may receive either the Moderna or Pfizer-BioNTech bivalent booster 8 weeks after their last dose.
Assessment of Comorbidities
Besides assessing diabetes-related complications, clinicians and people with diabetes need to be aware of common comorbidities that affect people with diabetes and that may complicate management (25–29). Diabetes comorbidities are conditions that affect people with diabetes more often than age-matched people without diabetes. This section discusses many of the common comorbidities observed in people with diabetes but is not necessarily inclusive of all the conditions that have been reported.
Autoimmune Diseases
Recommendations
4.7 People with type 1 diabetes should be screened for autoimmune thyroid disease soon after diagnosis and periodically thereafter. B
4.8 Adults with type 1 diabetes should be screened for celiac disease in the presence of gastrointestinal symptoms, signs, laboratory manifestations, or clinical suspicion suggestive of celiac disease. B
People with type 1 diabetes are at increased risk for other autoimmune diseases, with thyroid disease, celiac disease, and pernicious anemia (vitamin B12 deficiency) being among the most common (30). Other associated conditions include autoimmune hepatitis, primary adrenal insufficiency (Addison disease), collagen vascular diseases, and myasthenia gravis (31–34). Type 1 diabetes may also occur with other autoimmune diseases in the context of specific genetic disorders or polyglandular autoimmune syndromes (35). Given the high prevalence, nonspecific symptoms, and insidious onset of primary hypothyroidism, routine screening for thyroid dysfunction is recommended for all people with type 1 diabetes. Screening for celiac disease should be considered in adults with diabetes with suggestive symptoms (e.g., diarrhea, malabsorption, abdominal pain) or signs (e.g., osteoporosis, vitamin deficiencies, iron deficiency anemia) (36,37). Measurement of vitamin B12 levels should be considered for people with type 1 diabetes and peripheral neuropathy or unexplained anemia.
Cancer
Diabetes is associated with increased risk of cancers of the liver, pancreas, endometrium, colon/rectum, breast, and bladder (38). The association may result from shared risk factors between type 2 diabetes and cancer (older age, obesity, and physical inactivity) but may also be due to diabetes-related factors (39), such as underlying disease physiology or diabetes treatments, although evidence for these links is scarce. People with diabetes should be encouraged to undergo recommended age- and sex-appropriate cancer screenings and to reduce their modifiable cancer risk factors (obesity, physical inactivity, and smoking). New onset of atypical diabetes (lean body habitus, negative family history) in a middle-aged or older person may precede the diagnosis of pancreatic adenocarcinoma (40). However, in the absence of other symptoms (e.g., weight loss, abdominal pain), routine screening of all such individuals is not currently recommended.
Cognitive Impairment/Dementia
Recommendation
4.9 In the presence of cognitive impairment, diabetes treatment plans should be simplified as much as possible and tailored to minimize the risk of hypoglycemia. B
Diabetes is associated with a significantly increased risk and rate of cognitive decline and an increased risk of dementia (41,42). A recent meta-analysis of prospective observational studies in people with diabetes showed 73% increased risk of all types of dementia, 56% increased risk of Alzheimer dementia, and 127% increased risk of vascular dementia compared with individuals without diabetes (43). The reverse is also true: people with Alzheimer dementia are more likely to develop diabetes than people without Alzheimer dementia. In a 15-year prospective study of community-dwelling people >60 years of age, the presence of diabetes at baseline significantly increased the age- and sex-adjusted incidence of all-cause dementia, Alzheimer dementia, and vascular dementia compared with rates in those with normal glucose tolerance (44). See Section 13, “Older Adults,” for a more detailed discussion regarding screening for cognitive impairment.
Hyperglycemia
In those with type 2 diabetes, the degree and duration of hyperglycemia are related to dementia. More rapid cognitive decline is associated with both increased A1C and longer duration of diabetes (43). The Action to Control Cardiovascular Risk in Diabetes (ACCORD) study found that each 1% higher A1C level was associated with lower cognitive function in individuals with type 2 diabetes (45). However, the ACCORD study found no difference in cognitive outcomes in participants randomly assigned to intensive and standard glycemic management, supporting the recommendation that intensive glucose management should not be advised for the improvement of cognitive function in individuals with type 2 diabetes (46).
Hypoglycemia
In type 2 diabetes, severe hypoglycemia is associated with reduced cognitive function, and those with poor cognitive function have more severe hypoglycemia. In a long-term study of older people with type 2 diabetes, individuals with one or more recorded episodes of severe hypoglycemia had a stepwise increase in risk of dementia (47). Likewise, the ACCORD trial found that as cognitive function decreased, the risk of severe hypoglycemia increased (48). This has also been demonstrated in people with type 1 diabetes. Tailoring glycemic therapy may help to prevent hypoglycemia in individuals with cognitive dysfunction (49). See Section 13, “Older Adults,” for more detailed discussion of hypoglycemia in older people with type 1 and type 2 diabetes.
Nutrition
In one study, following the Mediterranean diet correlated with improved cognitive function (50). However, a Cochrane review found insufficient evidence to recommend any specific dietary change for the prevention or treatment of cognitive dysfunction (51).
Statins
A systematic review has reported that data do not support an adverse effect of statins on cognition (52). The FDA postmarketing surveillance databases have also revealed a low reporting rate for cognitive function–related adverse events, including cognitive dysfunction or dementia, with statin therapy, similar to rates seen with other commonly prescribed cardiovascular medications (52). Therefore, fear of cognitive decline should not be a barrier to statin use in people with diabetes and a high risk for cardiovascular disease.
Hepatitis C Infection
Infection with hepatitis C virus (HCV) is associated with a higher prevalence of type 2 diabetes, which is present in up to one-third of individuals with chronic HCV infection. HCV may impair glucose metabolism by several mechanisms, including directly via viral proteins and indirectly by altering proinflammatory cytokine levels (53). The use of newer direct-acting antiviral drugs produces a sustained virological response (cure) in nearly all cases and has been reported to improve glucose metabolism in individuals with diabetes (54). A meta-analysis of mostly observational studies found a mean reduction in A1C levels of 0.45% (95% CI −0.60 to −0.30) and reduced requirement for glucose-lowering medication use following successful eradication of HCV infection (55).
Pancreatitis
Diabetes is linked to diseases of the exocrine pancreas such as pancreatitis, which may disrupt the global architecture or physiology of the pancreas, often resulting in both exocrine and endocrine dysfunction. Up to half of patients with diabetes may have some degree of impaired exocrine pancreas function (56). People with diabetes are at an approximately twofold higher risk of developing acute pancreatitis (57).
Conversely, prediabetes and/or diabetes has been found to develop in approximately one-third of patients after an episode of acute pancreatitis (58); thus, the relationship is likely bidirectional. Postpancreatitis diabetes may include either new-onset disease or previously unrecognized diabetes (59). Studies of patients treated with incretin-based therapies for diabetes have also reported that pancreatitis may occur more frequently with these medications, but results have been mixed and causality has not been established (60–62).
Islet autotransplantation should be considered for patients requiring total pancreatectomy for medically refractory chronic pancreatitis to prevent postsurgical diabetes. Approximately one-third of patients undergoing total pancreatectomy with islet autotransplantation are insulin free 1 year postoperatively, and observational studies from different centers have demonstrated islet graft function up to a decade after the surgery in some patients (63–67). Both patient and disease factors should be carefully considered when deciding the indications and timing of this surgery. Surgeries should be performed in skilled facilities that have demonstrated expertise in islet autotransplantation.
Fractures
Age-specific hip fracture risk is significantly increased in both people with type 1 diabetes (relative risk 6.3) and those with type 2 diabetes (relative risk 1.7) in both sexes (68). Type 1 diabetes is associated with osteoporosis, but in type 2 diabetes, an increased risk of hip fracture is seen despite higher bone mineral density (BMD) (69). In three large observational studies of older adults, femoral neck BMD T-score and the World Health Organization Fracture Risk Assessment Tool (FRAX) score were associated with hip and nonspine fractures. Fracture risk was higher in participants with diabetes compared with those without diabetes for a given T-score and age or for a given FRAX score (70). Providers should assess fracture history and risk factors in older patients with diabetes and recommend measurement of BMD if appropriate for the patient’s age and sex. Fracture prevention strategies for people with diabetes are the same as for the general population and may include vitamin D supplementation. For patients with type 2 diabetes with fracture risk factors, thiazolidinediones (71) and sodium–glucose cotransporter 2 inhibitors (72) should be used with caution.
Sensory Impairment
Hearing impairment, both in high-frequency and low- to midfrequency ranges, is more common in people with diabetes than in those without, with stronger associations found in studies of younger people (73). Proposed pathophysiologic mechanisms include the combined contributions of hyperglycemia and oxidative stress to cochlear microangiopathy and auditory neuropathy (74). In a National Health and Nutrition Examination Survey (NHANES) analysis, hearing impairment was about twice as prevalent in people with diabetes compared with those without, after adjusting for age and other risk factors for hearing impairment (75). Low HDL cholesterol, coronary heart disease, peripheral neuropathy, and general poor health have been reported as risk factors for hearing impairment for people with diabetes, but an association of hearing loss with blood glucose levels has not been consistently observed (76). In the Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications (DCCT/EDIC) cohort, time-weighted mean A1C was associated with increased risk of hearing impairment when tested after long-term (>20 years) follow-up (77). Impairment in smell, but not taste, has also been reported in individuals with diabetes (78).
Low Testosterone in Men
Recommendation
4.10 In men with diabetes who have symptoms or signs of hypogonadism, such as decreased sexual desire (libido) or activity, or erectile dysfunction, consider screening with a morning serum testosterone level. B
Mean levels of testosterone are lower in men with diabetes compared with age-matched men without diabetes, but obesity is a major confounder (79,80). Testosterone replacement in men with symptomatic hypogonadism may have benefits including improved sexual function, well-being, muscle mass and strength, and bone density (81). In men with diabetes who have symptoms or signs of low testosterone (hypogonadism), a morning total testosterone level should be measured using an accurate and reliable assay (82). In men who have total testosterone levels close to the lower limit, it is reasonable to determine free testosterone concentrations either directly from equilibrium dialysis assays or by calculations that use total testosterone, sex hormone binding globulin, and albumin concentrations (82). Please see the Endocrine Society clinical practice guideline for detailed recommendations (82). Further tests (such as luteinizing hormone and follicle-stimulating hormone levels) may be needed to further evaluate the patient. Testosterone replacement in older men with hypogonadism has been associated with increased coronary artery plaque volume, with no conclusive evidence that testosterone supplementation is associated with increased cardiovascular risk in hypogonadal men (82).
Obstructive Sleep Apnea
Age-adjusted rates of obstructive sleep apnea, a risk factor for cardiovascular disease, are significantly higher (4- to 10-fold) with obesity, especially with central obesity (83). The prevalence of obstructive sleep apnea in the population with type 2 diabetes may be as high as 23%, and the prevalence of any sleep-disordered breathing may be as high as 58% (84,85). In participants with obesity enrolled in the Action for Health in Diabetes (Look AHEAD) trial, it exceeded 80% (86). Individuals with symptoms suggestive of obstructive sleep apnea (e.g., excessive daytime sleepiness, snoring, witnessed apnea) should be considered for screening (87). Sleep apnea treatment (lifestyle modification, continuous positive airway pressure, oral appliances, and surgery) significantly improves quality of life and blood pressure management. The evidence for a treatment effect on glycemic control is mixed (88).
Periodontal Disease
Periodontal disease is more severe, and may be more prevalent, in people with diabetes than in those without and has been associated with higher A1C levels (89–91). Longitudinal studies suggest that people with periodontal disease have higher rates of incident diabetes. Current evidence suggests that periodontal disease adversely affects diabetes outcomes, although evidence for treatment benefits remains controversial (29,92). In an RCT, intensive periodontal treatment was associated with better glycemic outcomes (A1C 8.3% vs. 7.8% in control subjects and the intensive-treatment group, respectively) and reduction in inflammatory markers after 12 months of follow-up (93).
Nonalcoholic Fatty Liver Disease
Screening
Recommendations
4.11a Adults with type 2 diabetes or prediabetes, particularly those with obesity or cardiometabolic risk factors/established cardiovascular disease, should be screened/risk stratified for nonalcoholic fatty liver disease with clinically significant fibrosis (defined as moderate fibrosis to cirrhosis) using a calculated fibrosis-4 index (derived from age, ALT, AST, and platelets [mdcalc.com/calc/2200/fibrosis-4-fib-4-index-liver-fibrosis]), even if they have normal liver enzymes. B
4.11b Adults with diabetes or prediabetes with persistently elevated plasma aminotransferase levels for >6 months and low fibrosis-4 index should be evaluated for other causes of liver disease. B
4.12 Adults with type 2 diabetes or prediabetes with an indeterminate or high fibrosis-4 index should have additional risk stratification by liver stiffness measurement with transient elastography, or the blood biomarker enhanced liver fibrosis. B
4.13 Adults with type 2 diabetes or prediabetes with indeterminate results or at high risk for significant liver fibrosis (i.e., by fibrosis-4 index, liver stiffness measurement, or enhanced liver fibrosis) should be referred to a gastroenterologist or hepatologist for further workup. Multidisciplinary care is recommended for long-term management. B
Nonalcoholic fatty liver disease (NAFLD) is the term used to identify the broad spectrum of disease ranging from nonalcoholic fatty liver with macrovesicular hepatic steatosis (or with mild inflammation) to steatohepatitis (nonalcoholic steatohepatitis [NASH]) to cirrhosis. This is in the absence of ongoing or recent consumption of significant amounts of alcohol (defined as ingestion of >21 standard drinks per week in men and >14 standard drinks per week in women over a 2-year period preceding evaluation) or other secondary causes of fatty liver disease. Diabetes is a major risk factor for developing NASH, disease progression, and worse liver outcomes (94). Recent studies in adults in the U.S. estimate that NAFLD is prevalent in >70% of people with type 2 diabetes (95–97). This is consistent with studies from other countries (98). NASH is defined histologically as having ≥5% hepatic steatosis and associated with inflammation and hepatocyte injury (hepatocyte ballooning), with or without evidence of liver fibrosis (99). Steatohepatitis is estimated to affect more than half of people with type 2 diabetes with NAFLD (100) and appears to be a driver for the development of fibrosis. Fibrosis stages are classified histologically as the following: F0, no fibrosis; F1, mild; F2, moderate (significant); F3, severe (advanced); and F4, cirrhosis. In the U.S., between 12% and 20% of people with type 2 diabetes have clinically significant fibrosis (≥F2) (95,96,100), with similar prevalence worldwide (94,98). NASH is a leading cause of hepatocellular carcinoma (HCC) (101,102) and of liver transplantation in the U.S., with transplant waiting lists being overrepresented by people with type 2 diabetes (103). Clinicians underestimate its prevalence and do not consistently implement appropriate screening strategies, thus missing the diagnosis of the potentially progressive form of NAFLD in high-risk groups, such as those having obesity or type 2 diabetes. This pattern of underdiagnosis is compounded by sparse referral to specialists and inadequate prescription of medications with proven efficacy in NASH (104,105).
The goal of screening for NAFLD is to identify patients at risk for adverse health outcomes associated with NASH such as cirrhosis, HCC, and death from liver disease. This risk is higher in people who have central obesity and cardiometabolic risk factors or insulin resistance, are >50 years of age, and/or have persistently elevated plasma aminotransferases (AST and/or ALT >30 units/L for >6 months) (106,107). Some genetic variants that alter hepatocyte triglyceride metabolism may also increase the risk of NASH progression and cirrhosis (108,109), amplifying the impact of obesity, but the role of genetic testing in clinical practice remains to be established.
Individuals with clinically significant fibrosis (≥F2), especially those with type 2 diabetes, have a greater risk of cirrhosis with liver decompensation, HCC, liver transplantation, and all-cause mortality (110–113). Increased mortality associated with NAFLD is attributable not only to cirrhosis and HCC but also to extrahepatic cancer (102), type 2 diabetes (114), and cardiovascular disease (115,116). The estimated relative impact depends on length of follow-up and population studied, among other factors. Emerging evidence suggests that NAFLD increases the risk of chronic kidney disease, particularly when liver fibrosis is present (117,118), although the association of NAFLD with diabetic retinopathy is less clear (119). Early diagnosis is essential to prevent future cirrhosis and complications.
A recent meta-analysis reported a prevalence of NAFLD of 22% in people with type 1 diabetes (120). This risk may be linked to the fact that about one-third of people with type 1 diabetes in the U.S. have obesity (121). However, there is large variability in NAFLD prevalence across studies, and most measured liver fat by ultrasound. In one of the few studies using the gold-standard MRI technique to quantify liver fat, the prevalence of steatosis in a population with type 1 diabetes with low prevalence of obesity was only 8.8% compared with 68% in people with type 2 diabetes (122). The prevalence of fibrosis was not established in that study. Therefore, screening for fibrosis in people with type 1 diabetes should only be considered in the presence of additional risk factors for NAFLD, such as obesity, incidental hepatic steatosis on imaging, or elevated plasma aminotransferases.
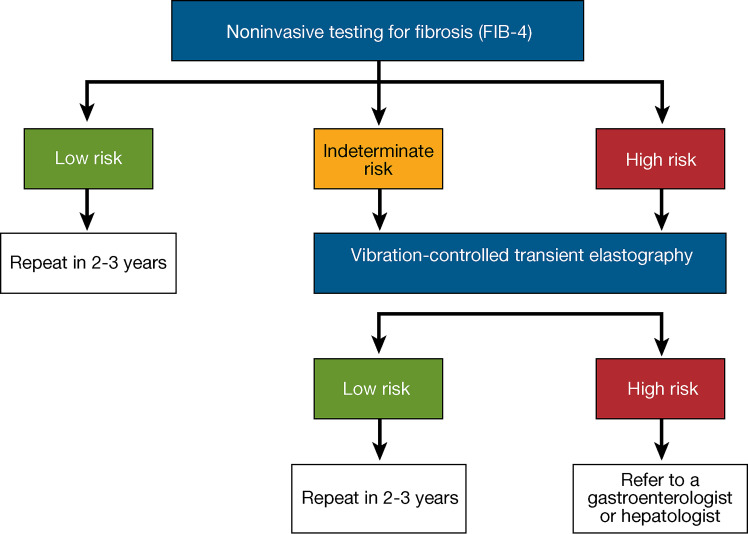
There is consensus that the fibrosis-4 index (FIB-4) is the most cost-effective strategy for the initial screening of people with prediabetes and cardiometabolic risk factors or with type 2 diabetes in primary care and diabetes clinical settings (99,105–107,123–125). See the proposed diagnostic algorithm by an expert group that included ADA representatives in Fig. 4.2 (105). A screening strategy based on elevated plasma aminotransferases >40 units/L would miss most individuals with NASH in these settings, as clinically significant fibrosis (≥F2) is frequently observed with plasma aminotransferases below the commonly used cutoff of 40 units/L (95–97,100,126,127). The American College of Gastroenterology considers the upper limit of normal ALT levels to be 29–33 units/L for male individuals and 19–25 units/L for female individuals (128), as higher levels are associated with increased liver-related mortality, even in the absence of identifiable risk factors. The FIB-4 estimates the risk of hepatic cirrhosis and is calculated from the computation of age, plasma aminotransferases (AST and ALT), and platelet count (mdcalc.com/calc/2200/fibrosis-4-fib-4-index-liver-fibrosis). A value of <1.3 is considered low risk of having advanced fibrosis (F3–F4) and for developing adverse liver outcomes, while >2.67 is considered as having a high probability of advanced fibrosis (F3–F4) and increased risk of adverse liver outcomes. FIB-4 predicts changes over time in hepatic fibrosis (129,130) and allows risk stratification of individuals in terms of future liver-related morbidity and mortality (131,132). FIB-4 has reasonable specificity but low sensitivity, hence a negative result rules out fibrosis while a positive result requires confirmatory testing. (130,133–136). It has a reasonable specificity and negative predictive value to rule out advanced fibrosis but lacks adequate sensitivity and positive predictive value to establish presence of advanced fibrosis in many cases, which is the reason why people with diabetes often fall in the “indeterminate” (or intermediate) risk group for advanced fibrosis and adverse liver outcomes (when FIB-4 is between 1.3 and 2.67). However, its low cost, simplicity, and good specificity make it the initial test of choice (Fig. 4.2). Performance is better in a population with higher prevalence of significant fibrosis (i.e., hepatology clinics) compared with primary care settings. FIB-4 has not been well validated in pediatric populations and does not perform as well in those aged <35 years. In people with diabetes ≥65 years of age, higher cutoffs for FIB-4 have been recommended (1.9–2.0 rather than >1.3) (137,138).
Figure 4.2.
A proposed algorithm for risk stratification in individuals with nonalcoholic fatty liver disease (NAFLD) or nonalcoholic steatohepatitis (NASH). ELF, enhanced liver fibrosis; FIB-4, fibrosis-4 index. Adapted from Kanwal et al. (105).
In people with an indeterminate or high FIB-4, additional risk stratification is required with a liver stiffness measurement (LSM) by transient elastography (Fig. 4.2) or, if unavailable, by commercial blood fibrosis biomarkers such as the enhanced liver fibrosis (ELF) test (139) or others. Use of a second nonproprietary diagnostic panel is not recommended (i.e., NAFLD fibrosis score, others), as they generally do not perform better than FIB-4 (97,133). Transient elastography (LSM) is the best-validated imaging technique for fibrosis risk stratification, and it predicts future cirrhosis and all-cause mortality in NAFLD (99,106,140). An LSM value of <8.0 kPa has a good negative predictive value to exclude advanced fibrosis (≥F3–F4) (141–143) and indicates low risk for clinically significant fibrosis. Such individuals with diabetes can be followed in nonspecialty clinics with repeat surveillance testing every ≥2 years, although the precise time interval remains to be established. If the LSM is >12 kPa, the risk for advanced fibrosis is high and people with diabetes should be referred to the hepatologist (100). FIB-4 followed by LSM helps stratify people with diabetes by risk level and minimize specialty referrals (132,135,140,144,145) (Fig. 4.2).
Specialists may order additional tests for fibrosis risk stratification (105–107,125,140), with magnetic resonance elastography (MRE) having the best overall performance (particularly for early fibrosis stages). However, the accessibility and costs associated with MRE are barriers to its use. While, liver biopsy remains the gold standard for the diagnosis of NASH, its indication is reserved to the discretion of the specialist within a multidisciplinary team approach due to high costs and potential for morbidity associated with this procedure.
In 2020, an expert panel convened by the American Gastroenterological Association that included representatives of the ADA reviewed the published literature on the burden, screening, risk stratification, diagnosis, and management of individuals with NAFLD (105). See Fig. 4.2, which is adapted from this special report (105). A Clinical Care Pathway summarized the diagnosis and management of NAFLD in a subsequent publication (107). Consensus has emerged to start screening with FIB-4 followed by LSM or ELF and patented biomarkers as needed for the noninvasive fibrosis risk stratification of individuals with NAFLD in primary care and diabetes clinics (99,105–107,123–125,146).
After initial risk stratification (i.e., FIB-4, LSM, and/or patented biomarkers), people with diabetes at indeterminate or high risk of fibrosis should be referred, based on practice setting, to a gastroenterologist or hepatologist for further workup within the framework of a multidisciplinary team (105,147,148).
Management
Recommendations
4.14 Adults with type 2 diabetes or prediabetes particularly with overweight or obesity with nonalcoholic fatty liver disease should be recommended lifestyle changes that promote weight loss, ideally within a structured nutrition plan and physical activity program for cardiometabolic benefits B and histological improvement. C
4.15 For adults with type 2 diabetes particularly with overweight or obesity with nonalcoholic fatty liver disease, consider using a glucagon-like peptide 1 receptor agonist with demonstrated benefits in nonalcoholic steatohepatitis as an adjunctive therapy to lifestyle interventions for weight loss. B
4.16 Pioglitazone or glucagon-like peptide 1 receptor agonists are the preferred agents for the treatment of hyperglycemia in adults with type 2 diabetes with biopsy-proven nonalcoholic steatohepatitis, or those at high risk for nonalcoholic fatty liver disease with clinically significant liver fibrosis using noninvasive tests. A
4.17a In adults with type 2 diabetes and nonalcoholic steatohepatitis, use of glucose-lowering therapies other than pioglitazone or glucagon-like peptide 1 receptor agonists may be continued as clinically indicated, but these therapies lack evidence of benefit in nonalcoholic steatohepatitis. B
4.17b Insulin therapy is the preferred agent for the treatment of hyperglycemia in adults with type 2 diabetes with decompensated cirrhosis. C
4.18a Adults with type 2 diabetes and nonalcoholic fatty liver disease are at increased cardiovascular risk; therefore, comprehensive management of cardiovascular risk factors is recommended. B
4.18b Statin therapy is safe in adults with type 2 diabetes and compensated cirrhosis from nonalcoholic fatty liver disease and should be initiated or continued for cardiovascular risk reduction as clinically indicated. B Statin therapy should be used with caution and close monitoring in people with decompensated cirrhosis, given limited safety and efficacy data. B
4.19a Consider metabolic surgery in appropriate candidates as an option to treat nonalcoholic steatohepatitis in adults with type 2 diabetes and nonalcoholic steatohepatitis B and improve cardiovascular outcomes. B
4.19b Metabolic surgery should be used with caution in adults with type 2 diabetes with compensated cirrhosis from nonalcoholic fatty liver disease B and is not recommended in decompensated cirrhosis. B
While steatohepatitis and cirrhosis occur in lean people with diabetes and are believed to be linked to genetic predisposition, insulin resistance, and environmental factors (149–151), there is ample evidence to implicate excess visceral and overall adiposity in people with overweight and obesity in the pathogenesis of the disease (152,153). Obesity in the setting of type 2 diabetes worsens insulin resistance and steatohepatitis, promoting the development of cirrhosis (154). Therefore, clinicians should enact evidence-based interventions (as discussed in Section 5, “Facilitating Positive Health Behaviors and Well-being to Improve Health Outcomes”) to promote healthy lifestyle change and weight loss for people with overweight or obesity and NAFLD. A minimum weight loss goal of 5%, preferably ≥10% (155,156), is needed to improve liver histology, with fibrosis requiring the larger weight reduction to change (156–158). Individualized, structured weight loss and exercise programs offer greater benefit than standard counseling in people with NAFLD (149,159).
Dietary recommendations to induce an energy deficit are not different than those for people with diabetes with obesity without NAFLD and should include a reduction of macronutrient content, limiting saturated fat, starch, and added sugar, with adoption of healthier eating patterns. The Mediterranean diet has the best evidence for improving liver and cardiometabolic health (99,106,123,124,159–163). Both aerobic and resistance training improve NAFLD in proportion to treatment engagement and intensity of the program (164–166).
Obesity pharmacotherapy may assist with weight loss in the context of lifestyle modification if not achieved by lifestyle modification alone (see Section 8, “Obesity and Weight Management for the Prevention and Treatment of Type 2 Diabetes”).
At present, there are no FDA-approved drugs for the treatment of NASH. Therefore, treatment for people with type 2 diabetes and NASH is centered on the dual purpose of treating hyperglycemia and obesity, especially if clinically significant fibrosis (≥F2) is present. The rationale for the treatment of people with type 2 diabetes is based on their high prevalence of NASH with significant fibrosis (10–15% of people with type 2 diabetes) (95,96,98), their higher risk of disease progression and liver-related mortality (94,113,167), and the lack of pharmacological treatments once cirrhosis is established (168). Therefore, early diagnosis and treatment of NAFLD offers the best opportunity for cirrhosis prevention. Pioglitazone and some glucagon-like peptide 1 receptor agonists (GLP-1 RAs) have been shown to be effective to treat steatohepatitis (105,106,169–171) and may slow fibrosis progression (172–174) and decrease cardiovascular disease (106,170), which is the number one cause of death in people with type 2 diabetes and NAFLD (115).
Pioglitazone improves glucose and lipid metabolism and reverses steatohepatitis in people with prediabetes, type 2 diabetes (175,176), or even without diabetes (177–179). Fibrosis also improved in some trials (176,178). A meta-analysis (172) concluded that pioglitazone treatment results in resolution of NASH and may improve fibrosis. Pioglitazone may halt the accelerated pace of fibrosis progression observed in people with type 2 diabetes (173) and is overall cost-effective for the treatment of NASH (180,181). Vitamin E may be beneficial for the treatment of NASH in people without diabetes (177). However, in people with type 2 diabetes, Vitamin E monotherapy was found to be negative in a small randomized controlled trial (RCT) (173), and it did not seem to enhance pioglitazone’s efficacy when used in combination as reported in an earlier trial in this population (176). Pioglitazone causes dose-dependent weight gain (15 mg/day, mean of 1–2%; 45 mg/day, 3–5%), increases fracture risk, may promote heart failure if used in individuals with preexisting congestive heart failure, and may increase the risk of bladder cancer, although this remains controversial (105,106,170,171).
GLP-1 RAs are effective in inducing weight loss and ameliorating elevated plasma aminotransferases and steatosis (169). However, there are only two RCTs of GLP-1 RAs in biopsy-proven individuals with NASH. A small RCT reported that liraglutide improved some features of NASH and, of particular relevance, delayed the progression of fibrosis (182). More recently, once-daily subcutaneous semaglutide in 320 people with biopsy-proven NASH (62% having type 2 diabetes) reported resolution of steatohepatitis in 59% at the higher dose (equivalent to 2.4 mg/week semaglutide) compared with 17% in the placebo group (P < 0.001) (174). Cumulatively, semaglutide did not significantly affect the stage of liver fibrosis in this group of people (70% of whom had F2 or F3 at baseline), but it significantly slowed over 72 weeks the progression of liver fibrosis (4.9% with the GLP-1 RA at the highest dose compared with 18.8% on placebo). Tirzepatide (183), sodium–glucose cotransporter inhibitors (184–186), and insulin (171) reduce hepatic steatosis, but their effects on steatohepatitis remain unknown. The use of glucose-lowering agents other than pioglitazone or GLP-1 RAs may be continued in patients with type 2 diabetes and NAFLD for glycemic control, as clinically indicated. However, they have either failed to improve steatohepatitis in paired-biopsy studies (metformin) or have no RCTs with liver histological end points (i.e., sulfonylureas, glitinides, dipeptidyl peptidase 4 inhibitors, or acarbose).
Insulin is the preferred glucose-lowering agent for the treatment of hyperglycemia in adults with type 2 diabetes with decompensated cirrhosis given the lack of robust evidence about the safety and efficacy of oral agents and noninsulin injectables (i.e., GLP-1 RA, GLP-1/GIP RA) (187), although a recent 48-week study suggested that a GLP-1 RAs may be safe in patients with NASH and compensated cirrhosis (188).
Metabolic surgery improves NASH and cardiometabolic health, altering the natural history of the disease (189). Meta-analyses report that 70–80% of people have improvement in hepatic steatosis, 50–75% in inflammation and hepatocyte ballooning (necrosis), and 30–40% in fibrosis (190,191). It may also reduce the risk of HCC (191). Metabolic surgery should be used with caution in individuals with compensated cirrhosis (i.e., asymptomatic stage of cirrhosis without associated liver complications), but with experienced surgeons the risk of hepatic decompensation is similar to that for those with less advanced liver disease. Because of the paucity of safety and outcome data, metabolic surgery is not recommended in individuals with decompensated cirrhosis (i.e., cirrhosis stage with complications such as variceal hemorrhage, ascites, hepatic encephalopathy, or jaundice), who also have a much higher risk of postoperative development of these liver-related complications (99,106,107).
A number of studies now recognize that adults with type 2 diabetes and NAFLD are at an increased risk of cardiovascular disease and require a comprehensive management of cardiovascular risk factors (99,105–107). Within a multidisciplinary approach, statin therapy should be initiated or continued for cardiovascular risk reduction as clinically indicated. Overall, its use appears to be safe in adults with type 2 diabetes and NASH, including in the presence of compensated cirrhosis (Child-Pugh class A or B cirrhosis) from NAFLD. Some studies even suggest that their use in people with chronic liver disease may reduce episodes of hepatic decompensation and/or overall mortality (192,193). Statin therapy is not recommended in decompensated cirrhosis given limited safety and efficacy data (99,106,107).
Diabetes and Covid-19
Recommendations
4.20 Health care professionals should help people with diabetes aim to achieve individualized targeted glycemic control to reduce the risk of macrovascular and microvascular risk as well as reduce the risk of COVID-19 and its complications. B
4.21 As we move into the recovery phase, diabetes health care services and practitioners should address the impact of the pandemic in higher-risk groups, including ethnic minority, deprived, and older populations. B
4.22 People who have been infected with SARS-CoV-2 should be followed up in the longer term to assess for complications and symptoms of long COVID. E
4.23 People with new-onset diabetes need to be followed up regularly in routine clinical practice to determine if diabetes is transient. B
4.24 Health care professionals need to carefully monitor people with diabetes for diabetic ketoacidosis during the COVID-19 pandemic. C
4.25 People with diabetes and their families/caregivers should be monitored for psychological well-being and offered support or referrals as needed, including mental/behavioral health care, self-management education and support, and resources to address related risk factors. E
4.26 Health care systems need to ensure that the vulnerable populations are not disproportionately disadvantaged by use of technological methods of consultations. E
4.27 There is no clear indication to change prescribing of glucose-lowering therapies in people with diabetes infected by the SARS-CoV-2 virus. B
4.28 People with diabetes should be prioritized and offered SARS-CoV-2 vaccines. B
SARS-CoV-2, the virus that causes the clinical disease COVID-19, was first reported in December 2019 in China and has disproportionately impacted certain groups, including men, older people, ethnic minority populations, and people with certain chronic conditions, including diabetes, cardiovascular disease, kidney disease, and certain respiratory diseases. COVID-19 has now been recognized as a complex multisystem disease including widespread insulin resistance, endothelial dysfunction, hematological disorders, and hyperimmune responses (194). There is now evidence of not only direct but also indirect adverse effects of COVID-19 in people with diabetes. Many people with multiple long-term conditions have diabetes, which has also been associated with worse outcomes in people with COVID-19 (195). The association with BMI and COVID-19 mortality is U-shaped in both type 1 and type 2 diabetes (196).
COVID-19 has disproportionately affected certain groups, such as older people and those from some ethnic populations who are known to have high prevalence of chronic conditions such as diabetes, cardiovascular disease, kidney disease, and certain respiratory diseases (197). People with chronic conditions have experienced some of the worst COVID-19 outcomes, including hospital admission and mortality (198). In people with diabetes, higher blood glucose levels both prior to and during COVID-19 admission have been associated with poor outcomes, including mortality (199). Type 1 diabetes has been associated with higher risk of COVID-19 mortality than type 2 diabetes (200). One whole-population-level study of over 61 million people in England in the first wave of the pandemic reported that after adjustment for age, sex, ethnicity, deprivation, and geographical region, the odds ratios for in-hospital COVID-19–related deaths were 3.51 (95% CI 3.16–3.90) in people with type 1 diabetes and 2.03 (1.97–2.09) in people with type 2 diabetes compared with the general population (201). There were also excess deaths in the first wave by 59.1% in people with type 1 diabetes and 64.3% in people with type 2 diabetes compared with death rates in the same time period for the previous 3 years (196). The largest study of people with diabetes to date, using whole-population data from England with over 3 million people, reported a higher association for mortality in people with type 1 diabetes than type 2 diabetes (196). Male sex, older age, renal impairment, non-White ethnicity, socioeconomic deprivation, and previous stroke and heart failure were associated with increased COVID-19–related mortality in both type 1 and type 2 diabetes (196).
Much of the evidence for recommendations is from a recent systematic review that was commissioned by the World Health Organization on the latest research evidence on the impact of COVID-19 on people with diabetes (200). Data were summarized from 112 systematic reviews that were narratively synthesized. The review reported that there are no appropriate data to determine whether diabetes is a risk factor for acquiring SARS-CoV-2 infection. Diabetes is a risk factor for severe disease and death from COVID-19.
Reasons for the higher rates of COVID-19 and severity in minority ethnic groups are complex and could be due to higher prevalence of comorbid conditions (e.g., diabetes), differences in exposure risk (e.g., overcrowded living conditions, essential worker jobs), and access to treatment (e.g., health insurance status, specialist services, and medications), which all relate to long-standing structural inequities that vary by ethnicity (202).
There is now overwhelming evidence that approximately 30–40% of people who are infected with COVID-19 get persistent and sometimes relapsing and remitting symptoms 4 weeks after infection, which has been termed post-acute sequelae of COVID-19, post-COVID-19 condition, post-acute COVID-19 syndrome, or long COVID (203,204). Currently, data on long COVID specifically in people with diabetes are lacking, and people who have been infected with SARS-CoV-2 should be followed up in the longer term.
There have also been recent reports of development of new-onset diabetes in people who have had COVID-19. There are conflicting reports of new-onset diabetes, with publications from a number of countries. The precise mechanisms for new-onset diabetes in people with COVID-19 are not known but may include previously undiagnosed diabetes presenting early or later in the disease trajectory, stress hyperglycemia, steroid-induced hyperglycemia, and possibly direct or indirect effects of SARS-CoV-2 on the β-cell (205). Whether new-onset diabetes is likely to remain permanent or is more aggressive is not known, and it will be important for health care professionals to monitor these people in the longer term. One large U.S. retrospective study of over 27 million people reported that COVID-19 was associated with significantly increased risk of new-onset type 1 diabetes and a disproportionately higher risk in ethnic minority people (206). Another recent cross-sectional population–based Canadian study observed a slightly higher but nonsignificant increase in diabetes incidence in children during the pandemic, suggesting this resulted from delays in diagnosis early during the pandemic with a catch-up effect (207). Whether COVID-19 leads to new-onset diabetes is not known.
There have been several publications on the risk of diabetic ketoacidosis (DKA) during the pandemic. A German diabetes prospective study using registry data of children and adolescents found an increase in type 1 diabetes in the first 3 months of the first wave, and the frequency of DKA at presentation was significantly higher than those for 2019 (44.7% vs. 24.5%, adjusted risk ratio 1.84) and 2018 (vs. 24.1%, adjusted risk ratio 1.85) as well as the proportion with severe DKA (208). A larger study using national data in England during the first two waves found that rates of DKA were higher than those for preceding years across all pandemic periods studied (209). The study reported lower DKA hospital admissions in people with type 1 diabetes but higher rates of DKA in people with type 1 diabetes and those newly diagnosed with diabetes.
There is also evidence of adverse effects of COVID-19 on mental health (210) and health-promoting lifestyles during the pandemic. Some small studies in people with diabetes have reported longer-term psychological impact of SARS-CoV-2 infection in people with diabetes, including fatigue and risk of suicide (211). Longitudinal follow-up of the Look AHEAD study of older adults with type 2 diabetes reported a 1.6-fold higher prevalence for depressive symptoms and 1.8-fold higher prevalence for loneliness during the pandemic compared with prepandemic levels (212). Furthermore, people with diabetes remain fearful of attending face-to-face contact due to the possible threat from mutant strains of coronavirus (213). Negative emotions due to the pandemic, including lockdowns, have been associated with reduced motivation, physical inactivity, and sedentary behavior (214). Higher levels of pandemic-related distress have been linked to higher A1C (215). Greater pandemic-related life disruptions have been related to higher distress in parents of youth with diabetes, which may have impacted families from racial and ethnic minority groups to a greater degree than non-Hispanic White families (216). On the other hand, for some youth with type 1 diabetes, increased time at home during the early phases of the COVID-19 pandemic provided opportunities for enhanced family support for diabetes self-management and reduced diabetes-related distress (217).
Recurrent lockdowns and other public health measures due to the pandemic have restricted access to routine diabetes care and have affected self-management, care-seeking behavior, and access to medications (218). This has resulted in compromised routine care and management of risk factors (219,220). There have been reductions in diagnosis of type 2 diabetes and reductions in new prescriptions of metformin during the pandemic (221). Due to unemployment or lost income during the pandemic, people living with diabetes have experienced financial hardships that may have reduced their affordability for medications in countries where costs for medications are out of pocket (219). Many individuals with diabetes have avoided or delayed seeking medical attention for routine non-COVID-19–related problems due to fear of infection and/or to reduce strain on health care services (222). Disruptions in care delivery and completion of care processes have been associated with an increased risk of non-COVID-19–related deaths in people with diabetes (223).
Direct contact will still be necessary if blood tests or physical examinations are required. However, it will be important to ensure that disparities are not widened for vulnerable groups such as the elderly and socieconomically challenged and ethnic minority groups due to access to literacy.
As we recover from the pandemic, it is essential that we prioritize the highest-risk groups for their routine review and assessment as well as management of their mental/behavioral health and risk factors. Diabetes professional bodies in some countries have published guidance on risk stratification and who to prioritize for diabetes review (224,225). Factors to consider for prioritization should include demographics, socioeconomical status, education levels, established complications, comorbidities, and modifiable risk factors, which are associated with high risk of progression of diabetes-related complications.
In many countries, health care professionals have reduced face-to-face contact and adapted technological methods of delivering routine diabetes care. One small RCT in adults with type 2 diabetes with follow-up to 16 weeks showed that remote consultations during the pandemic reduced the prevalence of mental health- and diabetes-related emotional distress (226). The number of face-to-face appointments is now increasing, and hybrid models with both virtual and face-to-face consultations are likely to remain (227). Technological interventions such as telehealth in people with diabetes may be a solution to improve care and clinical outcomes (228). However, such technological interventions may further widen disparities in vulnerable populations such as the elderly, ethnic minority groups, frail populations, and those from deprived communities (229).
Several pharmacoepidemiological studies have examined the association between glucose-lowering medications and risk of COVID-19 and have reported conflicting findings, although most studies showed a lower risk of mortality with metformin and a higher risk in people on insulin. However, the absolute differences in the risks have been small, and these findings could be due to confounding by indication (230). The gold standard for assessing the effects of therapies is by RCT, and only one RCT, the Dapagliflozin in Patients with Cardiometabolic Risk Factors Hospitalized with COVID-19 (DARE-19), a double-blind, placebo-controlled RCT in people with and without type 2 diabetes with at least one cardiovascular risk factor, has been reported (231). In this study, dapagliflozin was well tolerated and resulted in fewer events of organ dysfunction, but results were not statistically significant for the dual primary outcome of prevention (time to new or worsening organ dysfunction or death) and the hierarchical composite outcome of recovery by 30 days.
Great progress has been made globally to develop vaccines against SARS-CoV-2, and RCT data and real-world data show that vaccines have led to reduced infections, transmission, hospitalization, and mortality. It is therefore important that people with diabetes have regular SARS-CoV-2 vaccines (see IMMUNIZATIONS, above, for detailed information on COVID-19 vaccines).
It is unclear currently how often people with diabetes will require booster vaccines. Though limited data are available on COVID-19 vaccination attitudes or uptake in people with diabetes in the U.S. (232), diabetes health care professionals may be in a position to address questions and concerns among people with diabetes and encourage vaccination.
Footnotes
Disclosure information for each author is available at https://doi.org/10.2337/dc23-SDIS.
Suggested citation: ElSayed NA, Aleppo G, Aroda VR, et al., American Diabetes Association. 4. Comprehensive medical evaluation and assessment of comorbidities: Standards of Care in Diabetes—2023. Diabetes Care 2023;46(Suppl. 1):S49–S67.2
References
- 1. Stellefson M, Dipnarine K, Stopka C. The chronic care model and diabetes management in US primary care settings: a systematic review. Prev Chronic Dis 2013;10:E26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Coleman K, Austin BT, Brach C, Wagner EH. Evidence on the chronic care model in the new millennium. Health Aff (Millwood) 2009;28:75–85 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Gabbay RA, Bailit MH, Mauger DT, Wagner EH, Siminerio L. Multipayer patient-centered medical home implementation guided by the chronic care model. Jt Comm J Qual Patient Saf 2011;37:265–273 [DOI] [PubMed] [Google Scholar]
- 4. UK Prospective Diabetes Study (UKPDS) Group . Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). Lancet 1998;352:837–853 [PubMed] [Google Scholar]
- 5. Diabetes Control and Complications Trial Research Group; Nathan DM, Genuth S, Lachin J, et al. . The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. N Engl J Med 1993;329:977–986 [DOI] [PubMed] [Google Scholar]
- 6. Lachin JM, Genuth S, Nathan DM, Zinman B; DCCT/EDIC Research Group . Effect of glycemic exposure on the risk of microvascular complications in the diabetes control and complications trial--revisited. Diabetes 2008;57:995–1001 [DOI] [PubMed] [Google Scholar]
- 7. White NH, Cleary PA, Dahms W, Goldstein D, Malone J; Diabetes Control and Complications Trial (DCCT)/Epidemiology of Diabetes Interventions and Complications (EDIC) Research Group . Beneficial effects of intensive therapy of diabetes during adolescence: outcomes after the conclusion of the Diabetes Control and Complications Trial (DCCT). J Pediatr 2001;139:804–812 [DOI] [PubMed] [Google Scholar]
- 8. Rodriguez K, Ryan D, Dickinson JK, Phan V. Improving quality outcomes: the value of diabetes care and education specialists. Clin Diabetes 2022;40:356–365 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Anderson RM, Funnell MM. Compliance and adherence are dysfunctional concepts in diabetes care. Diabetes Educ 2000;26:597–604 [DOI] [PubMed] [Google Scholar]
- 10. Sarkar U, Fisher L, Schillinger D. Is self-efficacy associated with diabetes self-management across race/ethnicity and health literacy? Diabetes Care 2006;29:823–829 [DOI] [PubMed] [Google Scholar]
- 11. King DK, Glasgow RE, Toobert DJ, et al. . Self-efficacy, problem solving, and social-environmental support are associated with diabetes self-management behaviors. Diabetes Care 2010;33:751–753 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Nouwen A, Urquhart Law G, Hussain S, McGovern S, Napier H. Comparison of the role of self-efficacy and illness representations in relation to dietary self-care and diabetes distress in adolescents with type 1 diabetes. Psychol Health 2009;24:1071–1084 [DOI] [PubMed] [Google Scholar]
- 13. Dickinson JK, Guzman SJ, Maryniuk MD, et al. . The use of language in diabetes care and education. Diabetes Care 2017;40:1790–1799 [DOI] [PubMed] [Google Scholar]
- 14. Lee SWH, Ng KY, Chin WK. The impact of sleep amount and sleep quality on glycemic control in type 2 diabetes: a systematic review and meta-analysis. Sleep Med Rev 2017;31:91–101 [DOI] [PubMed] [Google Scholar]
- 15. Robinson CL, Bernstein H, Poehling K, Romero JR, Szilagyi P. Advisory Committee on Immunization Practices recommended immunization schedule for children and adolescents aged 18 years or younger—United States, 2020. MMWR Morb Mortal Wkly Rep 2020;69:130–132 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Freedman MS, Hunter P, Ault K, Kroger A. Advisory Committee on Immunization Practices recommended immunization schedule for adults aged 19 years or older—United States, 2020. MMWR Morb Mortal Wkly Rep 2020;69:133–135 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Lee G, Carr W, ACIP Evidence-Based Recommendations Work Group . Updated framework for development of evidence-based recommendations by the Advisory Committee on Immunization Practices. MMWR Morb Mortal Wkly Rep 2018;67:1271–1272 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Goeijenbier M, van Sloten TT, Slobbe L, et al. . Benefits of flu vaccination for persons with diabetes mellitus: a review. Vaccine 2017;35:5095–5101 [DOI] [PubMed] [Google Scholar]
- 19. Yedlapati SH, Khan SU, Talluri S, et al. . Effects of influenza vaccine on mortality and cardiovascular outcomes in patients with cardiovascular disease: a systematic review and meta-analysis. J Am Heart Assoc 2021;10:e019636. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Grohskopf LA, Alyanak E, Broder KR, Blanton LH, Fry AM, Jernigan DB, et al. . Prevention and control of seasonal influenza with vaccines: recommendations of the Advisory Committee on Immunization Practices—United States, 2020-21 influenza season. MMWR Recomm Rep 2020;69:1–24 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Smith SA, Poland GA. Use of influenza and pneumococcal vaccines in people with diabetes. Diabetes Care 2000;23:95–108 [DOI] [PubMed] [Google Scholar]
- 22. Kobayashi M, Farrar JL, Gierke R, et al. Use of 15-valent pneumococcal conjugate vaccine and 20-valent pneumococcal conjugate vaccine among U.S. adults: updated recommendations of the Advisory Committee on Immunization Practices—United States, 2022. MMWR Morb Mortal Wkly Rep 2022;71:109–117 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Ahmed SS, Pondo T, Xing W, et al. . Early impact of 13-valent pneumococcal conjugate vaccine use on invasive pneumococcal disease among adults with and without underlying medical conditions-United States. Clin Infect Dis 2020;70:2484–2492 [DOI] [PubMed] [Google Scholar]
- 24. Centers for Disease Control and Prevention . Interim clinical considerations for use of COVID-19 vaccines, 2022. Accessed 7 October 2022. Available from https://www.cdc.gov/vaccines/covid-19/clinical-considerations/covid-19-vaccines-us.html
- 25. Selvin E, Coresh J, Brancati FL. The burden and treatment of diabetes in elderly individuals in the U.S. Diabetes Care 2006;29:2415–2419 [DOI] [PubMed] [Google Scholar]
- 26. Grant RW, Ashburner JM, Hong CS, Chang Y, Barry MJ, Atlas SJ. Defining patient complexity from the primary care physician’s perspective: a cohort study. Ann Intern Med 2011;155:797–804 [DOI] [PubMed] [Google Scholar]
- 27. Tinetti ME, Fried TR, Boyd CM. Designing health care for the most common chronic condition—multimorbidity. JAMA 2012;307:2493–2494 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Sudore RL, Karter AJ, Huang ES, et al. . Symptom burden of adults with type 2 diabetes across the disease course: diabetes & aging study. J Gen Intern Med 2012;27:1674–1681 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Borgnakke WS, Ylöstalo PV, Taylor GW, Genco RJ. Effect of periodontal disease on diabetes: systematic review of epidemiologic observational evidence. J Periodontol 2013;84(Suppl.):S135–S152 [DOI] [PubMed] [Google Scholar]
- 30. Nederstigt C, Uitbeijerse BS, Janssen LGM, Corssmit EPM, de Koning EJP, Dekkers OM. Associated auto-immune disease in type 1 diabetes patients: a systematic review and meta-analysis. Eur J Endocrinol 2019;180:135–144 [DOI] [PubMed] [Google Scholar]
- 31. De Block CE, De Leeuw IH, Van Gaal LF. High prevalence of manifestations of gastric autoimmunity in parietal cell antibody-positive type 1 (insulin-dependent) diabetic patients. The Belgian Diabetes Registry. J Clin Endocrinol Metab 1999;84:4062–4067 [DOI] [PubMed] [Google Scholar]
- 32. Triolo TM, Armstrong TK, McFann K, et al. . Additional autoimmune disease found in 33% of patients at type 1 diabetes onset. Diabetes Care 2011;34:1211–1213 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Hughes JW, Riddlesworth TD, DiMeglio LA, Miller KM, Rickels MR, McGill JB. Autoimmune diseases in children and adults with type 1 diabetes from the T1D Exchange clinic registry. J Clin Endocrinol Metab 2016;101:4931–4937 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Kahaly GJ, Hansen MP. Type 1 diabetes associated autoimmunity. Autoimmun Rev 2016;15:644–648 [DOI] [PubMed] [Google Scholar]
- 35. Eisenbarth GS, Gottlieb PA. Autoimmune polyendocrine syndromes. N Engl J Med 2004;350:2068–2079 [DOI] [PubMed] [Google Scholar]
- 36. Rubio-Tapia A, Hill ID, Kelly CP, Calderwood AH; American College of Gastroenterology . ACG clinical guidelines: diagnosis and management of celiac disease. Am J Gastroenterol 2013;108:656–676 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Husby S, Murray JA, Katzka DA. AGA clinical practice update on diagnosis and monitoring of celiac disease-changing utility of serology and histologic measures: expert review. Gastroenterology 2019;156:885–889 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Suh S, Kim KW. Diabetes and cancer: is diabetes causally related to cancer? Diabetes Metab J 2011;35:193–198 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Giovannucci E, Harlan DM, Archer MC, et al. . Diabetes and cancer: a consensus report. CA Cancer J Clin 2010;60:207–221 [DOI] [PubMed] [Google Scholar]
- 40. Aggarwal G, Kamada P, Chari ST. Prevalence of diabetes mellitus in pancreatic cancer compared to common cancers. Pancreas 2013;42:198–201 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Cukierman T, Gerstein HC, Williamson JD. Cognitive decline and dementia in diabetes—systematic overview of prospective observational studies. Diabetologia 2005;48:2460–2469 [DOI] [PubMed] [Google Scholar]
- 42. Biessels GJ, Staekenborg S, Brunner E, Brayne C, Scheltens P. Risk of dementia in diabetes mellitus: a systematic review. Lancet Neurol 2006;5:64–74 [DOI] [PubMed] [Google Scholar]
- 43. Gudala K, Bansal D, Schifano F, Bhansali A. Diabetes mellitus and risk of dementia: a meta-analysis of prospective observational studies. J Diabetes Investig 2013;4:640–650 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Ohara T, Doi Y, Ninomiya T, et al. . Glucose tolerance status and risk of dementia in the community: the Hisayama study. Neurology 2011;77:1126–1134 [DOI] [PubMed] [Google Scholar]
- 45. Cukierman-Yaffe T, Gerstein HC, Williamson JD, et al. ; Action to Control Cardiovascular Risk in Diabetes-Memory in Diabetes (ACCORD-MIND) Investigators . Relationship between baseline glycemic control and cognitive function in individuals with type 2 diabetes and other cardiovascular risk factors: the Action to Control Cardiovascular Risk in Diabetes-Memory in Diabetes (ACCORD-MIND) trial. Diabetes Care 2009;32:221–226 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Launer LJ, Miller ME, Williamson JD, et al. ; ACCORD MIND investigators . Effects of intensive glucose lowering on brain structure and function in people with type 2 diabetes (ACCORD MIND): a randomised open-label substudy. Lancet Neurol 2011;10:969–977 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Whitmer RA, Karter AJ, Yaffe K, Quesenberry CP Jr, Selby JV. Hypoglycemic episodes and risk of dementia in older patients with type 2 diabetes mellitus. JAMA 2009;301:1565–1572 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Punthakee Z, Miller ME, Launer LJ, et al. ; ACCORD Group of Investigators ; ACCORD-MIND Investigators. Poor cognitive function and risk of severe hypoglycemia in type 2 diabetes: post hoc epidemiologic analysis of the ACCORD trial. Diabetes Care 2012;35:787–793 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Lacy ME, Gilsanz P, Eng C, Beeri MS, Karter AJ, Whitmer RA. Severe hypoglycemia and cognitive function in older adults with type 1 diabetes: the Study of Longevity in Diabetes (SOLID). Diabetes Care 2020;43:541–548 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Scarmeas N, Stern Y, Mayeux R, Manly JJ, Schupf N, Luchsinger JA. Mediterranean diet and mild cognitive impairment. Arch Neurol 2009;66:216–225 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Ooi CP, Loke SC, Yassin Z, Hamid TA. Carbohydrates for improving the cognitive performance of independent-living older adults with normal cognition or mild cognitive impairment. Cochrane Database Syst Rev 2011;4:CD007220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Richardson K, Schoen M, French B, et al. . Statins and cognitive function: a systematic review. Ann Intern Med 2013;159:688–697 [DOI] [PubMed] [Google Scholar]
- 53. Lecube A, Hernandez C, Genesca J, Simo R.. Proinflammatory cytokines, insulin resistance, and insulin secretion in chronic hepatitis C patients: a case-control study. Diabetes Care 2006;29:1096–1101 [DOI] [PubMed] [Google Scholar]
- 54. Hum J, Jou JH, Green PK, et al. . Improvement in glycemic control of type 2 diabetes after successful treatment of hepatitis C virus. Diabetes Care 2017;40:1173–1180 [DOI] [PubMed] [Google Scholar]
- 55. Carnovale C, Pozzi M, Dassano A, et al. . The impact of a successful treatment of hepatitis C virus on glyco-metabolic control in diabetic patients: a systematic review and meta-analysis. Acta Diabetol 2019;56:341–354 [DOI] [PubMed] [Google Scholar]
- 56. Piciucchi M, Capurso G, Archibugi L, Delle Fave MM, Capasso M, Delle Fave G.. Exocrine pancreatic insufficiency in diabetic patients: prevalence, mechanisms, and treatment. Int J Endocrinol 2015;2015:595649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Lee Y-K, Huang M-Y, Hsu C-Y, Su Y-C.. Bidirectional relationship between diabetes and acute pancreatitis: a population-based cohort study in Taiwan. Medicine (Baltimore) 2016;95: e2448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Das SLM, Singh PP, Phillips ARJ, Murphy R, Windsor JA, Petrov MS.. Newly diagnosed diabetes mellitus after acute pancreatitis: a systematic review and meta-analysis. Gut 2014;63:818–831 [DOI] [PubMed] [Google Scholar]
- 59. Petrov MS. Diabetes of the exocrine pancreas: American Diabetes Association-compliant lexicon. Pancreatology 2017;17:523–526 [DOI] [PubMed] [Google Scholar]
- 60. Thomsen RW, Pedersen L, Møller N, Kahlert J, Beck-Nielsen H, Sørensen HT.. Incretin-based therapy and risk of acute pancreatitis: a nationwide population-based case-control study. Diabetes Care 2015;38:1089–1098 [DOI] [PubMed] [Google Scholar]
- 61. Tkac I, Raz I.. Combined analysis of three large interventional trials with gliptins indicates increased incidence of acute pancreatitis in patients with type 2 diabetes. Diabetes Care 2017;40:284–286 [DOI] [PubMed] [Google Scholar]
- 62. Egan AG, Blind E, Dunder K, et al. . Pancreatic safety of incretin-based drugs—FDA and EMA assessment. N Engl J Med 2014;370:794–797 [DOI] [PubMed] [Google Scholar]
- 63. Bellin MD, Gelrud A, Arreaza-Rubin G, et al. . Total pancreatectomy with islet autotransplantation: summary of an NIDDK workshop. Ann Surg 2015;261:21–29 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64. Sutherland DER, Radosevich DM, Bellin MD, et al. . Total pancreatectomy and islet autotransplantation for chronic pancreatitis. J Am Coll Surg 2012;214:409–424; discussion 424–426 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65. Quartuccio M, Hall E, Singh V, et al. . Glycemic predictors of insulin independence after total pancreatectomy with islet autotransplantation. J Clin Endocrinol Metab 2017;102:801–809 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66. Webb MA, Illouz SC, Pollard CA, et al. . Islet auto transplantation following total pancreatectomy: a long-term assessment of graft function. Pancreas 2008;37:282–287 [DOI] [PubMed] [Google Scholar]
- 67. Wu Q, Zhang M, Qin Y, et al. . Systematic review and meta-analysis of islet autotransplantation after total pancreatectomy in chronic pancreatitis patients. Endocr J 2015;62:227–234 [DOI] [PubMed] [Google Scholar]
- 68. Janghorbani M, Van Dam RM, Willett WC, Hu FB.. Systematic review of type 1 and type 2 diabetes mellitus and risk of fracture. Am J Epidemiol 2007;166:495–505 [DOI] [PubMed] [Google Scholar]
- 69. Vestergaard P. Discrepancies in bone mineral density and fracture risk in patients with type 1 and type 2 diabetes—a meta-analysis. Osteoporos Int 2007;18:427–444 [DOI] [PubMed] [Google Scholar]
- 70. Schwartz AV, Vittinghoff E, Bauer DC, et al. ; Study of Osteoporotic Fractures (SOF) Research Group; Osteoporotic Fractures in Men (MrOS) Research Group; Health, Aging, and Body Composition (Health ABC) Research Group. Association of BMD and FRAX score with risk of fracture in older adults with type 2 diabetes. JAMA 2011;305:2184–2192 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71. Kahn SE, Zinman B, Lachin JM, et al. ; Diabetes Outcome Progression Trial (ADOPT) Study Group . Rosiglitazone-associated fractures in type 2 diabetes: an analysis from A Diabetes Outcome Progression Trial (ADOPT). Diabetes Care 2008;31:845–851 [DOI] [PubMed] [Google Scholar]
- 72. Taylor SI, Blau JE, Rother KI.. Possible adverse effects of SGLT2 inhibitors on bone. Lancet Diabetes Endocrinol 2015;3:8–10 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73. Baiduc RR, Helzner EP.. Epidemiology of diabetes and hearing loss. Semin Hear 2019;40: 281–291 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74. Helzner EP, Contrera KJ.. Type 2 diabetes and hearing impairment. Curr Diab Rep 2016;16:3. [DOI] [PubMed] [Google Scholar]
- 75. Bainbridge KE, Hoffman HJ, Cowie CC.. Diabetes and hearing impairment in the United States: audiometric evidence from the National Health and Nutrition Examination Survey, 1999 to 2004. Ann Intern Med 2008;149:1–10 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76. Bainbridge KE, Hoffman HJ, Cowie CC.. Risk factors for hearing impairment among U.S. adults with diabetes: National Health and Nutrition Examination Survey 1999–2004. Diabetes Care 2011;34:1540–1545 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77. Schade DS, Lorenzi GM, Braffett BH, et al. ; DCCT/EDIC Research Group . Hearing impairment and type 1 diabetes in the Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications (DCCT/EDIC) cohort. Diabetes Care 2018;41:2495–2501 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78. Rasmussen VF, Vestergaard ET, Hejlesen O, Andersson CUN, Cichosz SL.. Prevalence of taste and smell impairment in adults with diabetes: a cross-sectional analysis of data from the National Health and Nutrition Examination Survey (NHANES). Prim Care Diabetes 2018;12:453–459 [DOI] [PubMed] [Google Scholar]
- 79. Dhindsa S, Miller MG, McWhirter CL, et al. . Testosterone concentrations in diabetic and nondiabetic obese men. Diabetes Care 2010;33: 1186–1192 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80. Grossmann M. Low testosterone in men with type 2 diabetes: significance and treatment. J Clin Endocrinol Metab 2011;96:2341–2353 [DOI] [PubMed] [Google Scholar]
- 81. Bhasin S, Cunningham GR, Hayes FJ, et al. ; Task Force, Endocrine Society . Testosterone therapy in men with androgen deficiency syndromes: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab 2010;95:2536–2559 [DOI] [PubMed] [Google Scholar]
- 82. Bhasin S, Brito JP, Cunningham GR, et al. . Testosterone therapy in men with hypogonadism: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab 2018;103:1715–1744 [DOI] [PubMed] [Google Scholar]
- 83. Li C, Ford ES, Zhao G, Croft JB, Balluz LS, Mokdad AH.. Prevalence of self-reported clinically diagnosed sleep apnea according to obesity status in men and women: National Health and Nutrition Examination Survey, 2005–2006. Prev Med 2010;51:18–23 [DOI] [PubMed] [Google Scholar]
- 84. West SD, Nicoll DJ, Stradling JR.. Prevalence of obstructive sleep apnoea in men with type 2 diabetes. Thorax 2006;61:945–950 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85. Resnick HE, Redline S, Shahar E, et al. ; Sleep Heart Health Study . Diabetes and sleep disturbances: findings from the Sleep Heart Health Study. Diabetes Care 2003;26:702–709 [DOI] [PubMed] [Google Scholar]
- 86. Foster GD, Sanders MH, Millman R, et al. ; Sleep AHEAD Research Group . Obstructive sleep apnea among obese patients with type 2 diabetes. Diabetes Care 2009;32:1017–1019 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87. Bibbins-Domingo K, Grossman DC, Curry SJ, et al. ; US Preventive Services Task Force . Screening for obstructive sleep apnea in adults: US Preventive Services Task Force recommendation statement. JAMA 2017;317:407–414 [DOI] [PubMed] [Google Scholar]
- 88. Shaw JE, Punjabi NM, Wilding JP, Alberti KGMM; International Diabetes Federation Taskforce on Epidemiology and Prevention . Sleep-disordered breathing and type 2 diabetes: a report from the International Diabetes Federation Taskforce on Epidemiology and Prevention. Diabetes Res Clin Pract 2008;81:2–12 [DOI] [PubMed] [Google Scholar]
- 89. Khader YS, Dauod AS, El-Qaderi SS, Alkafajei A, Batayha WQ.. Periodontal status of diabetics compared with nondiabetics: a meta-analysis. J Diabetes Complications 2006;20:59–68 [DOI] [PubMed] [Google Scholar]
- 90. Casanova L, Hughes FJ, Preshaw PM.. Diabetes and periodontal disease: a two-way relationship. Br Dent J 2014;217:433–437 [DOI] [PubMed] [Google Scholar]
- 91. Eke PI, Thornton-Evans GO, Wei L, Borgnakke WS, Dye BA, Genco RJ.. Periodontitis in US Adults: National Health and Nutrition Examination Survey 2009–2014. J Am Dent Assoc 2018;149:576–588.e6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92. Simpson TC, Weldon JC, Worthington HV, et al. . Treatment of periodontal disease for glycaemic control in people with diabetes mellitus. Cochrane Database Syst Rev 2015. (11):CD004714. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93. D’Aiuto F, Gkranias N, Bhowruth D, et al. ; TASTE Group . Systemic effects of periodontitis treatment in patients with type 2 diabetes: a 12 month, single-centre, investigator-masked, randomised trial. Lancet Diabetes Endocrinol 2018;6:954–965 [DOI] [PubMed] [Google Scholar]
- 94. Younossi ZM, Golabi P, de Avila L, et al. . The global epidemiology of NAFLD and NASH in patients with type 2 diabetes: a systematic review and meta-analysis. J Hepatol 2019;71:793–801 [DOI] [PubMed] [Google Scholar]
- 95. Lomonaco R, Godinez Leiva E, Bril F, et al. . Advanced liver fibrosis is common in patients with type 2 diabetes followed in the outpatient setting: the need for systematic screening. Diabetes Care 2021;44:399–406 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96. Ciardullo S, Monti T, Perseghin G. High prevalence of advanced liver fibrosis assessed by transient elastography among U.S. adults with type 2 diabetes. Diabetes Care 2021;44:519–525 [DOI] [PubMed] [Google Scholar]
- 97. Barb D, Repetto EM, Stokes ME, Shankar SS, Cusi K. Type 2 diabetes mellitus increases the risk of hepatic fibrosis in individuals with obesity and nonalcoholic fatty liver disease. Obesity (Silver Spring) 2021;29:1950–1960 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98. Stefan N, Cusi K. A global view of the interplay between non-alcoholic fatty liver disease and diabetes. Lancet Diabetes Endocrinol 2022;10:284–296 [DOI] [PubMed] [Google Scholar]
- 99. Rinella ME, Neuschwander-Tetri BA, Siddiqui MS, et al. . AASLD practice guidance on the clinical assessment and management of nonalcoholic fatty liver disease. Hepatology 2023;77:1797–1835 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100. Harrison SA, Gawrieh S, Roberts K, et al. . Prospective evaluation of the prevalence of non-alcoholic fatty liver disease and steatohepatitis in a large middle-aged US cohort. J Hepatol 2021;75:284–291 [DOI] [PubMed] [Google Scholar]
- 101. Paik JM, Golabi P, Younossi Y, Mishra A, Younossi ZM. Changes in the global burden of chronic liver diseases from 2012 to 2017: the growing impact of NAFLD. Hepatology 2020;72:1605–1616 [DOI] [PubMed] [Google Scholar]
- 102. Simon TG, Roelstraete B, Khalili H, Hagström H, Ludvigsson JF. Mortality in biopsy-confirmed nonalcoholic fatty liver disease: results from a nationwide cohort. Gut 2021;70:1375–1382 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103. Burra P, Becchetti C, Germani G. NAFLD and liver transplantation: disease burden, current management and future challenges. JHEP Rep 2020;2:100192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104. Younossi ZM, Ong JP, Takahashi H, et al. ; Global Nonalcoholic Steatohepatitis Council . A global survey of physicians knowledge about nonalcoholic fatty liver disease. Clin Gastroenterol Hepatol 2022;20:e1456–e1468 [DOI] [PubMed] [Google Scholar]
- 105. Kanwal F, Shubrook JH, Younossi Z, et al. . Preparing for the NASH epidemic: a call to action. Diabetes Care 2021;44:2162–2172 [DOI] [PubMed] [Google Scholar]
- 106. Cusi K, Isaacs S, Barb D, et al. . American Association of Clinical Endocrinology clinical practice guideline for the diagnosis and management of nonalcoholic fatty liver disease in primary care and endocrinology clinical settings: co-sponsored by the American Association for the Study of Liver Diseases (AASLD). Endocr Pract 2022;28:528–562 [DOI] [PubMed] [Google Scholar]
- 107. Kanwal F, Shubrook JH, Adams LA, et al. . Clinical care pathway for the risk stratification and management of patients with nonalcoholic fatty liver disease. Gastroenterology 2021;161:1657–1669 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108. Gellert-Kristensen H, Richardson TG, Davey Smith G, Nordestgaard BG, Tybjaerg-Hansen A, Stender S. Combined effect of PNPLA3, TM6SF2, and HSD17B13 variants on risk of cirrhosis and hepatocellular carcinoma in the general population. Hepatology 2020;72:845–856 [DOI] [PubMed] [Google Scholar]
- 109. Stender S, Kozlitina J, Nordestgaard BG, Tybjærg-Hansen A, Hobbs HH, Cohen JC. Adiposity amplifies the genetic risk of fatty liver disease conferred by multiple loci. Nat Genet 2017;49:842–847 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110. Angulo P, Kleiner DE, Dam-Larsen S, et al. . Liver fibrosis, but no other histologic features, is associated with long-term outcomes of patients with nonalcoholic fatty liver disease. Gastroenterology 2015;149:389–97.e10 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111. Ekstedt M, Hagström H, Nasr P, et al. . Fibrosis stage is the strongest predictor for disease-specific mortality in NAFLD after up to 33 years of follow-up. Hepatology 2015;61:1547–1554 [DOI] [PubMed] [Google Scholar]
- 112. Taylor RS, Taylor RJ, Bayliss S, et al. . Association between fibrosis stage and outcomes of patients with nonalcoholic fatty liver disease: a systematic review and meta-analysis. Gastroenterology 2020;158:1611–1625.e12 [DOI] [PubMed] [Google Scholar]
- 113. Sanyal AJ, Van Natta ML, Clark J, et al. ; NASH Clinical Research Network (CRN) . Prospective study of outcomes in adults with nonalcoholic fatty liver disease. N Engl J Med 2021;385:1559–1569 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114. Mantovani A, Byrne CD, Bonora E, Targher G. Nonalcoholic fatty liver disease and risk of incident type 2 diabetes: a meta-analysis. Diabetes Care 2018;41:372–382 [DOI] [PubMed] [Google Scholar]
- 115. Duell PB, Welty FK, Miller M, et al. ; American Heart Association Council on Arteriosclerosis, Thrombosis and Vascular Biology; Council on Hypertension; Council on the Kidney in Cardiovascular Disease; Council on Lifestyle and Cardiometabolic Health; and Council on Peripheral Vascular Disease . Nonalcoholic fatty liver disease and cardiovascular risk: a scientific statement from the American Heart Association. Arterioscler Thromb Vasc Biol 2022;42:e168–e185 [DOI] [PubMed] [Google Scholar]
- 116. Mantovani A, Csermely A, Petracca G, et al. . Non-alcoholic fatty liver disease and risk of fatal and non-fatal cardiovascular events: an updated systematic review and meta-analysis. Lancet Gastroenterol Hepatol 2021;6:903–913 [DOI] [PubMed] [Google Scholar]
- 117. Ciardullo S, Ballabeni C, Trevisan R, Perseghin G. Liver stiffness, albuminuria and chronic kidney disease in patients with NAFLD: a systematic review and meta-analysis. Biomolecules 2022;12:105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118. Musso G, Gambino R, Tabibian JH, et al. . Association of non-alcoholic fatty liver disease with chronic kidney disease: a systematic review and meta-analysis. PLoS Med 2014;11:e1001680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119. Song D, Li C, Wang Z, Zhao Y, Shen B, Zhao W. Association of non-alcoholic fatty liver disease with diabetic retinopathy in type 2 diabetic patients: a meta-analysis of observational studies. J Diabetes Investig 2021;12:1471–1479 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120. de Vries M, Westerink J, Kaasjager KHAH, de Valk HW. Prevalence of nonalcoholic fatty liver disease (NAFLD) in patients with type 1 diabetes mellitus: a systematic review and meta-analysis. J Clin Endocrinol Metab 2020;105:dgaa575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121. Corbin KD, Driscoll KA, Pratley RE, Smith SR, Maahs DM; Advancing Care for Type 1 Diabetes and Obesity Network (ACT1ON) . Obesity in type 1 diabetes: pathophysiology, clinical impact, and mechanisms. Endocr Rev 2018;39:629–663 [DOI] [PubMed] [Google Scholar]
- 122. Cusi K, Sanyal AJ, Zhang S, et al. . Non-alcoholic fatty liver disease (NAFLD) prevalence and its metabolic associations in patients with type 1 diabetes and type 2 diabetes. Diabetes Obes Metab 2017;19:1630–1634 [DOI] [PubMed] [Google Scholar]
- 123. Arab JP, Dirchwolf M, Álvares-da-Silva MR, et al. . Latin American Association for the Study of the Liver (ALEH) practice guidance for the diagnosis and treatment of non-alcoholic fatty liver disease. Ann Hepatol 2020;19:674–690 [DOI] [PubMed] [Google Scholar]
- 124. Eslam M, Sarin SK, Wong VWS, et al. . The Asian Pacific Association for the Study of the Liver clinical practice guidelines for the diagnosis and management of metabolic associated fatty liver disease. Hepatol Int 2020;14:889–919 [DOI] [PubMed] [Google Scholar]
- 125. European Association for the Study of the Liver . EASL clinical practice guidelines on non-invasive tests for evaluation of liver disease severity and prognosis—2021 update. J Hepatol 2021;75:659–689 [DOI] [PubMed] [Google Scholar]
- 126. Portillo-Sanchez P, Bril F, Maximos M, et al. . High prevalence of nonalcoholic fatty liver disease in patients with type 2 diabetes mellitus and normal plasma aminotransferase levels. J Clin Endocrinol Metab 2015;100:2231–2238 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127. Maximos M, Bril F, Portillo Sanchez, et al. . The role of liver fat and insulin resistance as determinants of plasma aminotransferase elevation in nonalcoholic fatty liver disease. Hepatology 2014;61:153–160 [DOI] [PubMed] [Google Scholar]
- 128. Kwo PY, Cohen SM, Lim JK. ACG clinical guideline: evaluation of abnormal liver chemistries. J Am Coll Gastroenterol 2017;112:18–35 [DOI] [PubMed] [Google Scholar]
- 129. Younossi ZM, Anstee QM, Wai-Sun Wong V, et al. . The association of histologic and noninvasive tests with adverse clinical and patient-reported outcomes in patients with advanced fibrosis due to nonalcoholic steatohepatitis. Gastroenterology 2021;160:1608–1619.e13 [DOI] [PubMed] [Google Scholar]
- 130. Siddiqui MS, Yamada G, Vuppalanchi R, et al. ; NASH Clinical Research Network . Diagnostic accuracy of noninvasive fibrosis models to detect change in fibrosis stage. Clin Gastroenterol Hepatol 2019;17:1877–1885.e5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131. Unalp-Arida A, Ruhl CE. Liver fibrosis scores predict liver disease mortality in the United States population. Hepatology 2017;66:84–95 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132. Lee J, Vali Y, Boursier J, et al. . Prognostic accuracy of FIB-4, NAFLD fibrosis score and APRI for NAFLD-related events: a systematic review. Liver Int 2021;41:261–270 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133. Qadri S, Ahlholm N, Lønsmann I, et al. . Obesity modifies the performance of fibrosis biomarkers in nonalcoholic fatty liver disease. J Clin Endocrinol Metab 2022;107:e2008–e2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134. Bril F, McPhaul MJ, Caulfield MP, et al. . Performance of plasma biomarkers and diagnostic panels for nonalcoholic steatohepatitis and advanced fibrosis in patients with type 2 diabetes. Diabetes Care 2020;43:290–297 [DOI] [PubMed] [Google Scholar]
- 135. Anstee QM, Lawitz EJ, Alkhouri N, et al. . Noninvasive tests accurately identify advanced fibrosis due to NASH: baseline data from the STELLAR trials. Hepatology 2019;70:1521–1530 [DOI] [PubMed] [Google Scholar]
- 136. Singh A, Gosai F, Siddiqui MT, et al. . Accuracy of noninvasive fibrosis scores to detect advanced fibrosis in patients with type-2 diabetes with biopsy-proven nonalcoholic fatty liver disease. J Clin Gastroenterol 2020;54:891–897 [DOI] [PubMed] [Google Scholar]
- 137. McPherson S, Hardy T, Dufour JF, et al. . Age as a confounding factor for the accurate non-invasive diagnosis of advanced NAFLD fibrosis. Am J Gastroenterol 2017;112:740–751 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138. Ishiba H, Sumida Y, Tanaka S, et al. ; Japan Study Group of Non-Alcoholic Fatty Liver Disease (JSG-NAFLD) . The novel cutoff points for the FIB4 index categorized by age increase the diagnostic accuracy in NAFLD: a multi-center study. J Gastroenterol 2018;53:1216–1224 [DOI] [PubMed] [Google Scholar]
- 139. Vali Y, Lee J, Boursier J, et al. ; LITMUS Systematic Review Team . Enhanced liver fibrosis test for the non-invasive diagnosis of fibrosis in patients with NAFLD: a systematic review and meta-analysis. J Hepatol 2020;73:252–262 [DOI] [PubMed] [Google Scholar]
- 140. Castera L, Friedrich-Rust M, Loomba R. Noninvasive assessment of liver disease in patients with nonalcoholic fatty liver disease. Gastroenterology 2019;156:1264–1281.e4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141. Eddowes PJ, Sasso M, Allison M, et al. . Accuracy of FibroScan controlled attenuation parameter and liver stiffness measurement in assessing steatosis and fibrosis in patients with nonalcoholic fatty liver disease. Gastroenterology 2019;156:1717–1730 [DOI] [PubMed] [Google Scholar]
- 142. Mózes FE, Lee JA, Selvaraj EA, et al. ; LITMUS Investigators . Diagnostic accuracy of non-invasive tests for advanced fibrosis in patients with NAFLD: an individual patient data meta-analysis. Gut 2022;71:1006–1019 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143. Elhence A, Anand A, Biswas S, et al. . Compensated advanced chronic liver disease in nonalcoholic fatty liver disease: two-step strategy is better than Baveno criteria. Dig Dis Sci 2022 [DOI] [PubMed] [Google Scholar]
- 144. Chan WK, Treeprasertsuk S, Goh GBB, et al. . Optimizing use of nonalcoholic fatty liver disease fibrosis score, fibrosis-4 score, and liver stiffness measurement to identify patients with advanced fibrosis. Clin Gastroenterol Hepatol 2019;17:2570–2580.e37 [DOI] [PubMed] [Google Scholar]
- 145. Petta S, Wai-Sun Wong V, Bugianesi E, et al. . Impact of obesity and alanine aminotransferase levels on the diagnostic accuracy for advanced liver fibrosis of noninvasive tools in patients with nonalcoholic fatty liver disease. Am J Gastroenterol 2019;114:916–928 [DOI] [PubMed] [Google Scholar]
- 146. Garvey WT, Mechanick JI, Brett EB, et al. ; Reviewers of the AACE/ACE Obesity Clinical Practice Guidelines. American Association of Clinical Endocrinologists and American College Of Endocrinology comprehensive clinical practice guidelines for medical care of patients with obesity. Endocr Pract 2016;22(Suppl. 3):1–203 [DOI] [PubMed] [Google Scholar]
- 147. Lazarus JV, Anstee QM, Hagström H, et al. . Defining comprehensive models of care for NAFLD. Nat Rev Gastroenterol Hepatol 2021;18:717–729 [DOI] [PubMed] [Google Scholar]
- 148. Wong VWS, Zelber-Sagi S, Cusi K, et al. . Management of NAFLD in primary care settings. Liver Int 2022;42:2377–2389 [DOI] [PubMed] [Google Scholar]
- 149. Long MT, Noureddin M, Lim JK. AGA clinical practice update: diagnosis and management of nonalcoholic fatty liver disease in lean individuals: expert review. Gastroenterology 2022;163:764–774.e1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150. Cusi K. Nonalcoholic steatohepatitis in nonobese patients: not so different after all. Hepatology 2017;65:4–7 [DOI] [PubMed] [Google Scholar]
- 151. Younes R, Bugianesi E. NASH in lean individuals. Semin Liver Dis 2019;39:86–95 [DOI] [PubMed] [Google Scholar]
- 152. Loomba R, Friedman SL, Shulman GI. Mechanisms and disease consequences of nonalcoholic fatty liver disease. Cell 2021;184:2537–2564 [DOI] [PubMed] [Google Scholar]
- 153. Cusi K. Role of obesity and lipotoxicity in the development of nonalcoholic steatohepatitis: pathophysiology and clinical implications. Gastroenterology 2012;142:711–725.e6 [DOI] [PubMed] [Google Scholar]
- 154. Schuppan D, Surabattula R, Wang XY. Determinants of fibrosis progression and regression in NASH. J Hepatol 2018;68:238–250 [DOI] [PubMed] [Google Scholar]
- 155. Akbulut UE, Isik IA, Atalay A, et al. . The effect of a Mediterranean diet vs. a low-fat diet on non-alcoholic fatty liver disease in children: a randomized trial. Int J Food Sci Nutr 2022;73:357–366 [DOI] [PubMed] [Google Scholar]
- 156. Koutoukidis DA, Koshiaris C, Henry JA, et al. . The effect of the magnitude of weight loss on non-alcoholic fatty liver disease: a systematic review and meta-analysis. Metabolism 2021;115:154455. [DOI] [PubMed] [Google Scholar]
- 157. Promrat K, Kleiner DE, Niemeier HM, et al. . Randomized controlled trial testing the effects of weight loss on nonalcoholic steatohepatitis. Hepatology 2010;51:121–129 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158. Vilar-Gomez E, Martinez-Perez Y, Calzadilla-Bertot L, et al. . Weight loss through lifestyle modification significantly reduces features of nonalcoholic steatohepatitis. Gastroenterology 2015;149:367–78.e5; quiz e14–e15 [DOI] [PubMed] [Google Scholar]
- 159. Gepner Y, Shelef I, Komy O, et al. . The beneficial effects of Mediterranean diet over low-fat diet may be mediated by decreasing hepatic fat content. J Hepatol 2019;71:379–388 [DOI] [PubMed] [Google Scholar]
- 160. Kawaguchi T, Charlton M, Kawaguchi A, et al. . Effects of Mediterranean diet in patients with nonalcoholic fatty liver disease: a systematic review, meta-analysis, and meta-regression analysis of randomized controlled trials. Semin Liver Dis 2021;41:225–234 [DOI] [PubMed] [Google Scholar]
- 161. Younossi ZM, Corey KE, Lim JK. AGA clinical practice update on lifestyle modification using diet and exercise to achieve weight loss in the management of nonalcoholic fatty liver disease: expert review. Gastroenterology 2021;160:912–918 [DOI] [PubMed] [Google Scholar]
- 162. European Association for the Study of the Liver (EASL); European Association for the Study of Diabetes (EASD) ; European Association for the Study of Obesity (EASO). EASL-EASD-EASO clinical practice guidelines for the management of non-alcoholic fatty liver disease. J Hepatol 2016;64:1388–1402 [DOI] [PubMed] [Google Scholar]
- 163. Plauth M, Bernal W, Dasarathy S, et al. . ESPEN guideline on clinical nutrition in liver disease. Clin Nutr 2019;38:485–521 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164. Orci LA, Gariani K, Oldani G, Delaune V, Morel P, Toso C. Exercise-based interventions for nonalcoholic fatty liver disease: a meta-analysis and meta-regression. Clin Gastroenterol Hepatol 2016;14:1398–1411 [DOI] [PubMed] [Google Scholar]
- 165. Hashida R, Kawaguchi T, Bekki M, et al. . Aerobic vs. resistance exercise in non-alcoholic fatty liver disease: a systematic review. J Hepatol 2017;66:142–152 [DOI] [PubMed] [Google Scholar]
- 166. Sargeant JA, Gray LJ, Bodicoat DH, et al. . The effect of exercise training on intrahepatic triglyceride and hepatic insulin sensitivity: a systematic review and meta-analysis. Obes Rev 2018;19:1446–1459 [DOI] [PubMed] [Google Scholar]
- 167. Kanwal F, Kramer JR, Li L, et al. . Effect of metabolic traits on the risk of cirrhosis and hepatocellular cancer in nonalcoholic fatty liver disease. Hepatology 2020;71:808–819 [DOI] [PubMed] [Google Scholar]
- 168. Younossi Z, Stepanova M, Sanyal AJ, et al. . The conundrum of cryptogenic cirrhosis: Adverse outcomes without treatment options. J Hepatol 2018;69:1365–1370 [DOI] [PubMed] [Google Scholar]
- 169. Patel Chavez C, Cusi K, Kadiyala S. The emerging role of glucagon-like peptide-1 receptor agonists for the management of NAFLD. J Clin Endocrinol Metab 2022;107:29–38 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170. Gastaldelli A, Cusi K. From NASH to diabetes and from diabetes to NASH: mechanisms and treatment options. JHEP Rep 2019;1:312–328 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171. Budd J, Cusi K. Role of agents for the treatment of diabetes in the management of nonalcoholic fatty liver disease. Curr Diab Rep 2020;20:59. [DOI] [PubMed] [Google Scholar]
- 172. Musso G, Cassader M, Paschetta E, Gambino R. Thiazolidinediones and advanced liver fibrosis in nonalcoholic steatohepatitis: a meta-analysis. JAMA Intern Med 2017;177:633–640 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173. Bril F, Kalavalapalli S, Clark VC, et al. . Response to pioglitazone in patients with nonalcoholic steatohepatitis with vs without type 2 diabetes. Clin Gastroenterol Hepatol 2018;16:558–566.e2 [DOI] [PubMed] [Google Scholar]
- 174. Newsome PN, Buchholtz K, Cusi K, et al. ; NN9931-4296 Investigators . A placebo-controlled trial of subcutaneous semaglutide in nonalcoholic steatohepatitis. N Engl J Med 2021;384:1113–1124 [DOI] [PubMed] [Google Scholar]
- 175. Belfort R, Harrison SA, Brown K, et al. . A placebo-controlled trial of pioglitazone in subjects with nonalcoholic steatohepatitis. N Engl J Med 2006;355:2297–2307 [DOI] [PubMed] [Google Scholar]
- 176. Cusi K, Orsak B, Bril F, et al. . Long-term pioglitazone treatment for patients with nonalcoholic steatohepatitis and prediabetes or type 2 diabetes mellitus: a randomized trial. Ann Intern Med 2016;165:305–315 [DOI] [PubMed] [Google Scholar]
- 177. Sanyal AJ, Chalasani N, Kowdley KV, et al. ; NASH CRN . Pioglitazone, vitamin E, or placebo for nonalcoholic steatohepatitis. N Engl J Med 2010;362:1675–1685 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178. Aithal GP, Thomas JA, Kaye PV, et al. . Randomized, placebo-controlled trial of pioglitazone in nondiabetic subjects with nonalcoholic steatohepatitis. Gastroenterology 2008;135:1176–1184 [DOI] [PubMed] [Google Scholar]
- 179. Huang JF, Dai CY, Huang CF, et al. . First-in-Asian double-blind randomized trial to assess the efficacy and safety of insulin sensitizer in nonalcoholic steatohepatitis patients. Hepatol Int 2021;15:1136–1147 [DOI] [PubMed] [Google Scholar]
- 180. Noureddin M, Jones C, Alkhouri N, Gomez EV, Dieterich DT; NASHNET . Screening for nonalcoholic fatty liver disease in persons with type 2 diabetes in the United States is cost-effective: a comprehensive cost-utility analysis. Gastroenterology 2020;159:1985–1987.e4 [DOI] [PubMed] [Google Scholar]
- 181. Mahady SE, Wong G, Craig JC, George J. Pioglitazone and vitamin E for nonalcoholic steatohepatitis: a cost utility analysis. Hepatology 2012;56:2172–2179 [DOI] [PubMed] [Google Scholar]
- 182. Armstrong MJ, Gaunt P, Aithal GP, et al. ; LEAN trial team . Liraglutide safety and efficacy in patients with non-alcoholic steatohepatitis (LEAN): a multicentre, double-blind, randomised, placebo-controlled phase 2 study. Lancet 2016;387:679–690 [DOI] [PubMed] [Google Scholar]
- 183. Gastaldelli A, Cusi K, Fernández Landó L, Bray R, Brouwers B, Rodríguez Á. Effect of tirzepatide versus insulin degludec on liver fat content and abdominal adipose tissue in people with type 2 diabetes (SURPASS-3 MRI): a substudy of the randomised, open-label, parallel-group, phase 3 SURPASS-3 trial. Lancet Diabetes Endocrinol 2022;10:393–406 [DOI] [PubMed] [Google Scholar]
- 184. Cusi K, Bril F, Barb D, et al. . Effect of canagliflozin treatment on hepatic triglyceride content and glucose metabolism in patients with type 2 diabetes. Diabetes Obes Metab 2019;21:812–821 [DOI] [PubMed] [Google Scholar]
- 185. Kahl S, Gancheva S, Straßburger K, et al. . Empagliflozin effectively lowers liver fat content in well-controlled type 2 diabetes: a randomized, double-blind, phase 4, placebo-controlled trial. Diabetes Care 2020;43:298–305 [DOI] [PubMed] [Google Scholar]
- 186. Latva-Rasku A, Honka MJ, Kullberg J, et al. . The SGLT2 inhibitor dapagliflozin reduces liver fat but does not affect tissue insulin sensitivity: a randomized, double-blind, placebo-controlled study with 8-week treatment in type 2 diabetes patients. Diabetes Care 2019;42:931–937 [DOI] [PubMed] [Google Scholar]
- 187. Castera L, Cusi K.. Diabetes and cirrhosis: current concepts on diagnosis and management. Hepatology 2023;77:2128–2146 [DOI] [PubMed] [Google Scholar]
- 188. Loomba R, Abdelmalek MF, Armstrong MJ, et al. ; NN9931-4492 investigators . Semaglutide 2·4 mg once weekly in patients with non-alcoholic steatohepatitis-related cirrhosis: a randomised, placebo-controlled phase 2 trial. Lancet Gastroenterol Hepatol 2023;8:511–522 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189. Aminian A, Al-Kurd A, Wilson R, et al. . Association of bariatric surgery with major adverse liver and cardiovascular outcomes in patients with biopsy-proven nonalcoholic steatohepatitis. JAMA 2021;326:2031–2042 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190. Fakhry TK, Mhaskar R, Schwitalla T, Muradova E, Gonzalvo JP, Murr MM. Bariatric surgery improves nonalcoholic fatty liver disease: a contemporary systematic review and meta-analysis. Surg Obes Relat Dis 2019;15:502–511 [DOI] [PubMed] [Google Scholar]
- 191. Ramai D, Singh J, Lester J, et al. . Systematic review with meta-analysis: bariatric surgery reduces the incidence of hepatocellular carcinoma. Aliment Pharmacol Ther 2021;53:977–984 [DOI] [PubMed] [Google Scholar]
- 192. Kim RG, Loomba R, Prokop LJ, Singh S.. Statin use and risk of cirrhosis and related complications in patients with chronic liver diseases: a systematic review and meta-analysis. Clin Gastroenterol Hepatol 2017;15:1521–1530 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193. Kaplan DE, Serper MA, Mehta R, et al. ; VOCAL Study Group . Effects of hypercholesterolemia and statin exposure on survival in a large national cohort of patients with cirrhosis. Gastroenterology 2019;156:1693–1706.e12 [DOI] [PubMed] [Google Scholar]
- 194. Roberts CM, Levi M, McKee M, Schilling R, Lim WS, Grocott MPW. COVID-19: a complex multisystem disorder. Br J Anaesth 2020;125:238–242 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195. Chudasama YV, Zaccardi F, Gillies CL, et al. . Patterns of multimorbidity and risk of severe SARS-CoV-2 infection: an observational study in the U.K. BMC Infect Dis 2021;21:908. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 196. Holman N, Knighton P, Kar P, et al. . Risk factors for COVID-19-related mortality in people with type 1 and type 2 diabetes in England: a population-based cohort study. Lancet Diabetes Endocrinol 2020;8:823–833 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197. Martin CA, Jenkins DR, Minhas JS, et al. ; Leicester COVID-19 consortium . Socio-demographic heterogeneity in the prevalence of COVID-19 during lockdown is associated with ethnicity and household size: results from an observational cohort study. EClinicalMedicine 2020;25:100466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198. Singh AK, Gillies CL, Singh R, et al. . Prevalence of co-morbidities and their association with mortality in patients with COVID-19: a systematic review and meta-analysis. Diabetes Obes Metab 2020;22:1915–1924 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199. Hartmann-Boyce J, Morris E, Goyder C, et al. . Diabetes and COVID-19: risks, management, and learnings from other national disasters. Diabetes Care 2020;43:1695–1703 [DOI] [PubMed] [Google Scholar]
- 200. Hartmann-Boyce J, Rees K, Perring JC, et al. . Risks of and from SARS-CoV-2 infection and COVID-19 in people with diabetes: a systematic review of reviews. Diabetes Care 2021;44:2790–2811 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201. Barron E, Bakhai C, Kar P, et al. . Associations of type 1 and type 2 diabetes with COVID-19-related mortality in England: a whole-population study. Lancet Diabetes Endocrinol 2020;8:813–822 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 202. Khunti K, Feldman EL, Laiteerapong N, Parker W, Routen A, Peek M. The impact of the COVID-19 pandemic on ethnic minority groups with diabetes Diabetes Care 2023;46:228–236 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203. Chen C, Haupert SR, Zimmermann L, Shi X, Fritsche LG, Mukherjee B. Global prevalence of post COVID-19 condition or long COVID: a meta-analysis and systematic review. J Infect Dis 2022;226:1593–1607 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204. Nalbandian A, Sehgal K, Gupta A, et al. . Post-acute COVID-19 syndrome. Nat Med 2021;27:601–615 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 205. Khunti K, Del Prato S, Mathieu C, Kahn SE, Gabbay RA, Buse JB. COVID-19, hyperglycemia, and new-onset diabetes. Diabetes Care 2021;44:2645–2655 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 206. Qeadan F, Tingey B, Egbert J, et al. . The associations between COVID-19 diagnosis, type 1 diabetes, and the risk of diabetic ketoacidosis: a nationwide cohort from the US using the Cerner Real-World Data. PLoS One 2022;17:e0266809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207. Shulman R, Cohen E, Stukel TA, Diong C, Guttmann A. Examination of trends in diabetes incidence among children during the COVID-19 pandemic in Ontario, Canada, from March 2020 to September 2021. JAMA Netw Open 2022;5:e2223394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 208. Kamrath C, Mönkemöller K, Biester T, et al. . Ketoacidosis in children and adolescents with newly diagnosed type 1 diabetes during the COVID-19 pandemic in Germany. JAMA 2020;324:801–804 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 209. Misra S, Barron E, Vamos E, et al. . Temporal trends in emergency admissions for diabetic ketoacidosis in people with diabetes in England before and during the COVID-19 pandemic: a population-based study. Lancet Diabetes Endocrinol 2021;9:671–680 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210. Violant-Holz V, Gallego-Jiménez MG, González-González CS, et al. . Psychological health and physical activity levels during the COVID-19 pandemic: a systematic review. Int J Environ Res Public Health 2020;17:E9419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 211. Alessi J, Scherer GDLG, Erthal IN, et al. . One in ten patients with diabetes have suicidal thoughts after 1 year of the COVID-19 pandemic: we need to talk about diabetes and mental health not only during Suicide Prevention Awareness Month. Acta Diabetol 2022;59:143–145 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 212. Chao AM, Wadden TA, Clark JM, et al. . Changes in the prevalence of symptoms of depression, loneliness, and insomnia in U.S. older adults with type 2 diabetes during the COVID-19 pandemic: the Look AHEAD Study. Diabetes Care 2022;45:74–82 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 213. Caballero AE, Ceriello A, Misra A, et al. . COVID-19 in people living with diabetes: an international consensus. J Diabetes Complications 2020;34:107671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 214. Stockwell S, Trott M, Tully M, et al. . Changes in physical activity and sedentary behaviours from before to during the COVID-19 pandemic lockdown: a systematic review. BMJ Open Sport Exerc Med 2021;7:e000960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 215. O’Donnell MB, Hilliard ME, Cao VT, et al. . “It just kind of feels like a different world now:” stress and resilience for adolescents with type 1 diabetes in the era of COVID-19. Front Clin Diabetes Healthcare. Accessed 7 October 2022. Available from https://www.frontiersin.org/articles/10.3389/fcdhc.2022.835739 [DOI] [PMC free article] [PubMed]
- 216. Wang CH, Hilliard ME, Carreon SA, et al. . Predictors of mood, diabetes-specific and COVID-19-specific experiences among parents of early school-age children with type 1 diabetes during initial months of the COVID-19 pandemic. Pediatr Diabetes 2021;22:1071–1080 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 217. Ferguson K, Moore H, Kaidbey JH, et al. . Impacts of the COVID-19 pandemic on pediatric type 1 diabetes management: a qualitative study. Sci Diabetes Self Manag Care. 24 September 2022. DOI: 10.1177/26350106221125701 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 218. Mohseni M, Ahmadi S, Azami-Aghdash S, et al. . Challenges of routine diabetes care during COVID-19 era: a systematic search and narrative review. Prim Care Diabetes 2021;15:918–922 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 219. Ratzki-Leewing AA, Ryan BL, Buchenberger JD, Dickens JW, Black JE, Harris SB. COVID-19 hinterland: surveilling the self-reported impacts of the pandemic on diabetes management in the USA (cross-sectional results of the iNPHORM study). BMJ Open 2021;11:e049782. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 220. Seidu S, Hambling C, Holmes P, et al. ; PCDS Research Group . The impact of the COVID pandemic on primary care diabetes services in the UK: a cross-sectional national survey of views of health professionals delivering diabetes care. Prim Care Diabetes 2022;16:257–263 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 221. Carr MJ, Wright AK, Leelarathna L, et al. . Impact of COVID-19 restrictions on diabetes health checks and prescribing for people with type 2 diabetes: a UK-wide cohort study involving 618 161 people in primary care. BMJ Qual Saf 2022;31:503–514 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 222. Vamos EP, Khunti K. Indirect effects of the COVID-19 pandemic on people with type 2 diabetes: time to urgently move into a recovery phase. BMJ Qual Saf 2022;31:483–485 [DOI] [PubMed] [Google Scholar]
- 223. Valabhji J, Barron E, Gorton T, et al. . Associations between reductions in routine care delivery and non-COVID-19-related mortality in people with diabetes in England during the COVID-19 pandemic: a population-based parallel cohort study. Lancet Diabetes Endocrinol 2022;10:561–570 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 224. DiabetesontheNet . How to undertake a remote diabetes review. Accessed 29 August 2022. Available from https://diabetesonthenet.com/diabetes-primary-care/how-undertake-remote-diabetes-review/
- 225. Nagi D, Wilmot E, Owen K, et al. . ABCD position statement on risk stratification of adult patients with diabetes during COVID-19 pandemic. British Journal of Diabetes. 2021;21:123–131 [Google Scholar]
- 226. Alessi J, de Oliveira GB, Franco DW, et al. . Telehealth strategy to mitigate the negative psychological impact of the COVID-19 pandemic on type 2 diabetes: a randomized controlled trial. Acta Diabetol 2021;58:899–909 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 227. Kilvert A, Wilmot EG, Davies M, Fox C. Virtual consultations: are we missing anything? Pract Diabetes 2020;37:143–146 [Google Scholar]
- 228. Phillip M, Bergenstal RM, Close KL, et al. . The digital/virtual diabetes clinic: the future is now-recommendations from an international panel on diabetes digital technologies introduction. Diabetes Technol Ther 2021;23:146–154 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 229. O’Connor S, Hanlon P, O’Donnell CA, Garcia S, Glanville J, Mair FS. Understanding factors affecting patient and public engagement and recruitment to digital health interventions: a systematic review of qualitative studies. BMC Med Inform Decis Mak 2016;16:120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 230. Khunti K, Knighton P, Zaccardi F, et al. . Prescription of glucose-lowering therapies and risk of COVID-19 mortality in people with type 2 diabetes: a nationwide observational study in England. Lancet Diabetes Endocrinol 2021;9:293–303 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 231. Kosiborod MN, Esterline R, Furtado RHM, et al. . Dapagliflozin in patients with cardiometabolic risk factors hospitalised with COVID-19 (DARE-19): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Diabetes Endocrinol 2021;9:586–594 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 232. Czeisler MÉ, Barrett CE, Siegel KR, et al. . Health care access and use among adults with diabetes during the COVID-19 pandemic - United States, February-March 2021. MMWR Morb Mortal Wkly Rep 2021;70:1597–1602 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 233. Karter AJ, Warton EM, Lipska KJ, et al. . Development and validation of a tool to identify patients with type 2 diabetes at high risk of hypoglycemia-related emergency department or hospital use. JAMA Intern Med 2017;177:1461–1470 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 234. Lipska KJ, Ross JS, Wang Y, et al. . National trends in US hospital admissions for hyperglycemia and hypoglycemia among Medicare beneficiaries, 1999 to 2011. JAMA Intern Med 2014;174:1116–1124 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 235. Shorr RI, Ray WA, Daugherty JR, Griffin MR. Incidence and risk factors for serious hypoglycemia in older persons using insulin or sulfonylureas. Arch Intern Med 1997;157:1681–1686 [PubMed] [Google Scholar]
- 236. Abdelhafiz AH, Rodríguez-Mañas L, Morley JE, Sinclair AJ. Hypoglycemia in older people–a less well recognized risk factor for frailty. Aging Dis 2015;6:156–167 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 237. Yun JS, Ko SH, Ko SH, et al. . Presence of macroalbuminuria predicts severe hypoglycemia in patients with type 2 diabetes: a 10-year follow-up study. Diabetes Care 2013;36:1283–1289 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 238. Chelliah A, Burge MR. Hypoglycaemia in elderly patients with diabetes mellitus: causes and strategies for prevention. Drugs Aging 2004;21:511–530 [DOI] [PubMed] [Google Scholar]
- 239. Centers for Disease Control and Prevention (CDC) . Use of hepatitis B vaccination for adults with diabetes mellitus: recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Morb Mortal Wkly Rep 2011;60:1709–1711 [PubMed] [Google Scholar]
- 240. Meites E, Szilagyi PG, Chesson HW, Unger ER, Romero JR, Markowitz LE. Human papillomavirus vaccination for adults: updated recommendations of the Advisory Committee on Immunization Practices. MMWR Morb Mortal Wkly Rep 2019;68:698–702 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 241. Demicheli V, Jefferson T, Di Pietrantonj C, et al. . Vaccines for preventing influenza in the elderly. Cochrane Database Syst Rev 2018;2:CD004876. [DOI] [PubMed] [Google Scholar]
- 242. Centers for Disease Control and Prevention (CDC); Advisory Committee on Immunization Practices . Updated recommendations for prevention of invasive pneumococcal disease among adults using the 23-valent pneumococcal polysaccharide vaccine (PPSV23). MMWR Morb Mortal Wkly Rep 2010;59:1102–1106. [PubMed] [Google Scholar]
- 243. Falkenhorst G, Remschmidt C, Harder T, Hummers-Pradier E, Wichmann O, Bogdan C. Effectiveness of the 23-valent pneumococcal polysaccharide vaccine (PPV23) against pneumococcal disease in the elderly: systematic review and meta-analysis. PLoS One 2017;12:e0169368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 244. Havers FP, Moro PL, Hunter P, Hariri S, Bernstein H. Use of tetanus toxoid, reduced diphtheria toxoid, and acellular pertussis vaccines: updated recommendations of the Advisory Committee on Immunization Practices—United States, 2019. MMWR Morb Mortal Wkly Rep 2020;69:77–83 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 245. Dooling KL, Guo A, Patel M, et al. . Recommendations of the Advisory Committee on Immunization Practices for use of herpes zoster vaccines. MMWR Morb Mortal Wkly Rep 2018;67:103–108 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 246. Davies MJ, Aroda VR, Collins BS, et al. . Management of hyperglycemia in type 2 diabetes, 2022. A consensus report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetes Care 2022;45:2753–2786 [DOI] [PMC free article] [PubMed] [Google Scholar]