Abstract
We investigated if elevated cardiac troponin I (cTnI) serum levels before non-cardiac surgery were predictors of postoperative cardiac events in patients with end stage renal disease (ESRD) undergoing dialysis. In total, 703 consecutive patients with ESRD undergoing dialysis who underwent non-cardiac surgery were enrolled. Preoperative cTnI serum levels were measured at least once in all patients. The primary endpoint was defined as a composite of cardiac death, myocardial infarction (MI), and pulmonary edema during hospitalization or within 30 days after surgery in patients with a hospitalization longer than 30 days after surgery. Postoperative cardiac events occurred in 48 (6.8%) out of 703 patients (cardiac death 1, MI 18, and pulmonary edema 33). Diabetes mellitus (DM), previous ischemic heart disease, and congestive heart failure were more common in patients with postoperative cardiac events. Peak cTnI serum levels were higher in patients with postoperative cardiac event (180 ± 420 ng/L vs. 80 ± 190 ng/L, p = 0.008), and also elevated peak cTnI levels > 45 ng/L were more common in patients with postoperative cardiac events (66.8% vs. 30.5%, p < 0.001). Multivariate logistic regression analysis showed that DM (odds ratio [OR] 2.509, 95% confidence interval [CI] 1.178–5.345, p = 0.017) and serum peak cTnI levels ≥ 45 ng/L (OR 3.167, 95% CI 1.557–6.444, p = 0.001) were independent predictors for the primary outcome of cardiac death/MI/pulmonary edema. Moreover, cTnI levels ≥ 45 ng/L had an incremental prognostic value to the revised cardiac risk index (RCRI) (Chi-square = 23, p < 0.001), and to the combined RCRI and left ventricular ejection fraction (Chi-square = 12, p = 0.001). Elevated preoperative cTnI levels are predictors of postoperative cardiac events including cardiac death, MI, and pulmonary edema in patients with ESRD undergoing non-cardiac surgery.
Keywords: Cardiac troponin I, End stage renal disease, Surgery, Postoperative complications, Myocardial infarction, Risk assessment
Introduction
Patients with chronic kidney disease (CKD) have a higher risk of cardiovascular (CV) disease than non-CKD patients, as CKD has been shown to have a high risk of arteriosclerosis progression [1]. Most patients also have other comorbidities such as hypertension and diabetes mellitus (DM) which are also risk factors for CV disease [1–3]. Elevated serum cardiac troponin I (cTnI) levels have been previously used to diagnose myocardial infarction (MI) and predict the risk of cardiac events [1–4]. However, serum cTnI levels are also increased during myocardial wall distension due to volume and pressure overloading from for example, decompensated heart failure, pulmonary thromboembolism, and extremely high blood pressure, symptoms which also occur in patients with CKD [4–6]. Therefore, when cTnI levels are increased during CKD, especially in dialysis patients, it is often difficult to interpret whether this is due to MI or CKD [7, 8].
CV complications after surgery have a significant impact on patient mortality and morbidity, therefore, the evaluation of postoperative CV events before surgery is vital [9]. Several methods have been proposed to predict postoperative CV events in preoperative patients; the revised cardiac risk index (RCRI) score is one such method [9]. Additionally, monitoring cTnI serum levels is sometimes used for high-risk patients, with some studies reporting elevated preoperative cTnI levels are related to postoperative CV events. However, few studies have reported if elevated preoperative cTnI levels can predict postoperative CV events in dialysis patients [10–13].
Therefore, we investigated if elevated cTnI serum levels before non-cardiac surgery could predict postoperative cardiac events in patients with end stage renal disease (ESRD) undergoing dialysis.
Methods
Study population
From all patients with end stage renal disease (ESRD) undergoing dialysis at the Kyungpook National University Hospital between January 2015 and December 2019, we included 703 consecutive patients who underwent non-cardiac surgery. The following patients were excluded; those diagnosed with myocardial infarction (MI) within 30 days before surgery, and those with sepsis. The study was approved by the Institutional Review Board of Kyungpook National University Hospital (No: 2021-06-004). Informed consent was waived by the board.
Data collection
Patient information was obtained from medical chart. Patient demographics included age, sex, and body mass index (BMI). Blood samples were taken at admission. Cardiovascular (CV) risk factors included hypertension, diabetes mellitus (DM), a previous history of ischemic heart disease (IHD)/congestive haert failure (CHF)/cerebrovascular disease (CVD), and current smoking status. Echocardiographic results were based on examinations performed on the closest date prior to surgery. During echocardiography, the left ventricular ejection fraction (LVEF, %), left ventricular end diastolic dimension (LVEDD, mm), left atrium anteroposterior diameter (mm), and early diastolic mitral inflow velocity/early diastolic mitral annular tissue velocity (E/E') were measured. Electrocardiography (ECG) results were based on examinations at admission. ECG findings included the presence of atrial fibrillation and ST-T changes, and QRS duration (ms). In addition, patient anesthesia type, the risk of surgery, and the revised cardiac risk index (RCRI) scores were included, and the risk of major cardiac adverse events according to the type of surgery was classified into high (> 5%), intermediate (1 ~ 5%), and low risk (< 1%) [14, 15].
Measurement of cardiac troponin I
For all patients, serum cardiac troponin I (cTnI) (ng/L) levels were evaluated at admission or within at least 2 weeks prior to surgery. For patients with abnormal cTnI values, assays were performed repeatedly. Serum cTnI levels were measured using the Dimension Vista system (DIMENSION VISTA® CTNI CARDIAC TROPONIN I FLEX®: Siemens Healthcare Diagnostics Inc., NY, USA). The limit of quantitation was determined at 40 ng/L and corresponded to a coefficient of variation (CV) of 10%. In a study of 199 serum samples from apparently healthy individuals, the upper limit of the 99th percentile for the method was 45 ng/L. The reportable range of the assay was 15–40,000 ng/L.
Clinical outcomes
The primary outcome was defined as cardiac events during hospitalization or within 30 days after surgery in patients with a hospitalization longer than 30 days after surgery. Cardiac events consisted of cardiac death, MI, and pulmonary edema. Secondary outcomes consisted of cardiac death and MI. Acute MI was defined when there is acute myocardial injury with clinical evidence of acute myocardial ischemia and with detection of a rise and/or fall of cTnI values with at least one value above the 99th percentile upper reference limit and at least one of the following: symptoms of myocardial ischemia; new ischemic electrocardiogram changes; development of pathological Q waves; imaging evidence of new loss of viable myocardium or new regional wall motion abnormality in a pattern consistent with an ischemic etiology; identification of a coronary thrombus by angiography or autopsy [11]. A pulmonary edema diagnosis required one or more of the following conditions: development of symptoms or signs of pulmonary edema and evidence of abnormal findings from chest radiography [12].
Statistical analysis
Data were expressed as the mean ± standard deviation, and percentages for continuous and categorical variables. Comparisons between baseline variables were performed using student t tests and Chi-square tests for continuous and categorical variables, respectively. All p-values were two-sided, and a p < 0.05 value was considered statistically significant. Multivariate logistic regression analysis was used to identify independent predictors of cardiac events. Incremental factors added to the model at each step were considered significant when differences in log-likelihoods associated with models corresponded to p < 0.05. Receiver operating characteristic (ROC) curve analyses were performed to determine cut-off values for predicting cardiac events and estimated ROC curves and compared areas under curves (with 95% confidence interval [CI]) in corresponding logistic models. All statistical analyses were performed using IBM SPSS version 20.0 (IBM Corp., Armonk, NY, USA) and MedCalc version 19.8 (MedCalc Software, Ostend, Belgium).
Results
Baseline characteristics of the 703 patients (mean age; 61.4 ± 14.7 years; 425 males [60.5%]) are shown (Table 1). Hypertension (80.9%) and diabetes mellitus (DM) (59%) were common comorbidities. We observed that 115 patients (16.4%) had experienced previous ischemic heart disease (IHD) and 93 (13.2%) and 83 patients (11.8%) had a previous history of congestive heart failure (CHF) and cerebrovascular disease (CVD), respectively. The mean left ventricular ejection fraction (LVEF) was 54.1% ± 10.4%, mean left ventricular end diastolic dimension (LVEDD) was 48.7 ± 7.2 mm, and mean early diastolic mitral inflow velocity/early diastolic mitral annular tissue velocity (E/E') was 15.0 ± 6.7. In terms of laboratory findings, patients with peak cardiac troponin I (cTnI) serum levels ≥ 45 ng/L totaled 233 (33.1%): the mean peak cTnI serum level was 90 ± 200 ng/L. Peak cTnI level positively correlated with the LVEDD (r = 0.079, p = 0.037) and E/E' (r = 0.095, p = 0.011), but there was no correlation between peak cTnI and LVEF (r = −0.054, p = 0.154).
Table 1.
Baseline characteristics according to the presence or absence of cardiac events
| All patients (n = 703) | Cardiac events | P value | |||
|---|---|---|---|---|---|
| Yes (n= 48) | No (n = 655) | ||||
| Demographics | |||||
| Age (year) | 61.4 ± 14.7 | 64.5 ± 14.2 | 61.2 ± 14.7 | 0.132 | |
| Male (%) | 425 (60.5) | 31 (64.6) | 394 (60.2) | 0.545 | |
| Body mass index (kg/m2) | 22.6 ± 3.6 | 22.8 ± 3.9 | 22.5 ± 3.6 | 0.626 | |
| Risk factors and comorbidities | |||||
| Hypertension (%) | 569 (80.9) | 39 (81.2) | 530 (80.9) | 0.955 | |
| Diabetes, all (%) | 415 (59.0) | 37 (77.1) | 378 (57.7) | 0.008 | |
| Diabetes, insulin (%) | 155 (22.0) | 13 (27.1) | 142 (21.7) | 0.383 | |
| Previous IHD (%) | 115 (16.4) | 19 (39.6) | 96 (14.7) | < 0.001 | |
| Previous CHF (%) | 93 (13.2) | 11 (22.9) | 82 (12.5) | 0.040 | |
| Previous CVD (%) | 83 (11.8) | 8 (16.7) | 75 (11.5) | 0.280 | |
| Current smoking (%) | 102 (14.5) | 7 (14.6) | 95 (14.5) | 0.998 | |
| Peak cardiac troponin I (ng/L) | 90 ± 200 | 180 ± 240 | 80 ± 190 | 0.008 | |
| Peak cardiac troponin I ≥ 45 ng/L (%) | 233 (33.1) | 33 (68.8) | 200 (30.5) | < 0.001 | |
| Echocardiography | |||||
| LVEF (%) | 54.1 ± 10.4 | 47.0 ± 15.1 | 54.7 ± 9.8 | 0.001 | |
| LVEDD (mm) | 48.7 ± 7.2 | 51.5 ± 6.7 | 48.4 ± 7.2 | 0.006 | |
| E/E' | 15.0 ± 6.7 | 19.0 ± 9.5 | 14.7 ± 6.4 | 0.007 | |
| Electrocardiography | |||||
| Atrial fibrillation (%) | 33 (4.8) | 3 (6.2) | 30 (4.7) | 0.493 | |
| QRS duration (ms) | 96.3 ± 20.6 | 98.7 ± 23.4 | 96.6 ± 23.8 | 0.551 | |
| ST-T changes (%) | 221 (31.9) | 25 (52.1) | 196 (30.4) | 0.002 | |
| General anesthesia (%) | 255 (37.7) | 11 (61.1) | 254 (37.1) | 0.038 | |
| Surgical risk according to type of surgery | |||||
| High (%) | 71 (10.1) | 4 (8.3) | 67 (10.2) | 0.152 | |
| Intermediate (%) | 302 (43.0) | 16 (33.3) | 286 (43.7) | ||
| Low (%) | 330 (46.9) | 28 (58.3) | 302 (46.1) | ||
| RCRI score | 1.7 ± 0.8 | 2.2 ± 0.9 | 1.7 ± 0.8 | < 0.001 | |
IHD ischemic heart disease, CHF congestive heart failure, CVD cerebrovascular disease, LVEF left ventricular ejection fraction, LVEDD left ventricular end diastolic dimension, E/E' early diastolic mitral inflow velocity/early diastolic mitral annular tissue velocity, RCRI revised cardiac risk index
Cardiac events occurred in 48 (6.8%) patients (cardiac death; 1 patient, myocardial infarction (MI); 18 patients, pulmonary edema; 33 patients). DM, previous IHD, and CHF were more common in patients with postoperative cardiac events. The mean peak cTnI serum level was 9 ± 200 ng/L, and peak cTnI levels were higher in patients with cardiac events (180 ± 240 vs. 80 ± 190, p = 0.008). Also, peak cTnI levels ≥ 45 ng/L were more common in patients with cardiac events (66.8% vs. 30.5%, p < 0.001). Echocardiographic results showed lower LVEF (47.0% ± 15.1% vs. 54.7% ± 9.8%, p = 0.001), higher LVEDD (51.5 ± 6.7 mm vs. 48.4 ± 7.2 mm, p = 0.006), and higher E/E' (19.0 ± 9.5 vs. 14.7 ± 6.4, p = 0.007) in patients with cardiac events. Also, patients with cardiac events showed a greater proportion of ST-T changes during 12-lead electrocardiography (ECG) than those without cardiac events (52.1% vs. 30.4%, p = 0.002). In addition, the general anesthesia rate was higher in patients with cardiac events (61.1% vs. 37.1%, p = 0.038) and the revised cardiac risk index (RCRI) scores were also higher in patients with cardiac events (2.2 ± 0.9 vs. 1.7 ± 0.8, p < 0.001). The groups with high, intermediate, and low risk of cardiac major adverse cardiac event according to the type of surgery were 10.1% (n = 71), 43.0% (n = 302), and 46.9% (n = 330), respectively. However, there was no statistical difference in the incidence of postoperative cardiac events between the three groups. Also we observed no between-group differences regarding age, sex, body mass index (BMI), and comorbidities such as hypertension, previous CVD, and smoking status.
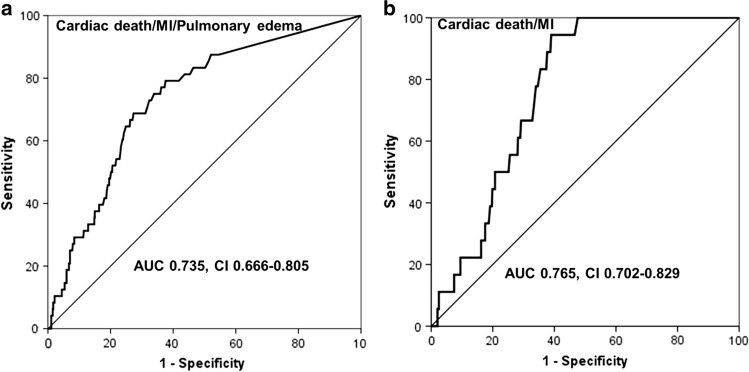
The area under the receiver operating characteristic (ROC) curves for prediction of postoperative cardiac events (cardiac death/MI/pulmonary edema) and cardiac death/MI were 0.735 (95% confidence interval [CI] 0.666–0.805) and 0.765 (95% CI 0.702–0.829) for preoperative cTnI, respectively (Fig. 1). When groups were classified based on cTnI ≥ 45 ng/L levels, the frequency of postoperative MI (p = 0.009) and pulmonary edema (p < 0.001) was higher in patients with peak cTnI ≥ 45 ng/L levels. Also, the composite of cardiac death, MI, and pulmonary edema (p < 0.001) and the composite of cardiac death and MI (p = 0.009) were also high. However, no statistical differences in cardiac death were observed according to the presence or absence of elevated peak cTnI levels (p = 0.543) (Table 2).
Fig. 1.
a The area under the ROC curve for predicting postoperative cardiac events (cardiac death/ myocardial infarction MI/pulmonary edema) for preoperative cTnI. b The area under the ROC curve for predicting postoperative cardiac events (cardiac death/MI) for preoperative cTnI. ROC receiver operating characteristics; MI myocardial infarction; cTnI cardiac troponin I
Table 2.
Cardiac events according to the levels of preoperative peak cardiac troponin I
| cTnI (1–44 ng/L) (n = 470) | cTnI (≥ 45 ng/L) (n = 233) | p value | |
|---|---|---|---|
| Cardiac death/MI (%) | 6 (1.3) | 12 (5.2) | 0.004 |
| Cardiac death (%) | 0 (0) | 1 (0.4) | 0.331 |
| Myocardial infarction (%) | 6 (1.3) | 12 (5.2) | 0.004 |
| Cardiac death/MI/pulmonary edema (%) | 15 (3.2) | 33 (14.2) | < 0.001 |
| Pulmonary edema (%) | 11 (2.3) | 22 (9.4) | < 0.001 |
cTnI cardiac troponin I, MI myocardial infarction
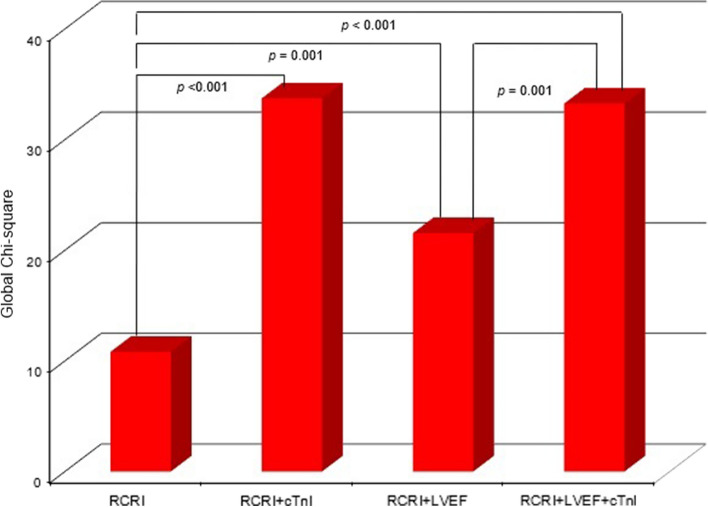
Multivariate logistic regression analyses showed that DM (OR 2.509, 95% CI 1.178–5.345, p = 0.017) and peak cTnI ≥ 45 ng/L levels (OR 3.167, 95% CI 1.557–6.444, p = 0.001) were independent predictors for the primary outcome; cardiac death/MI/pulmonary edema (Table 3). Moreover, cTnI ≥ 45 ng/L levels had an incremental prognostic value for RCRI scores (Chi-square = 23, p < 0.001) and for combined RCRI and LVEF data (Chi-square = 12, p = 0.001) (Fig. 2).
Table 3.
Multivariate logistic regression analysis for predicting cardiac events
| Cardiac events (death/MI/pulmonary edema) | Cardiac events (death/MI) | |||||
|---|---|---|---|---|---|---|
| Odds ratio | 95% CI | p value | Odds ratio | 95% CI | p value | |
| Diabetes | 2.509 | 1.178–5.345 | 0.017 | 5.438 | 1.147–25.767 | 0.033 |
| Previous ischemic heart disease | 1.906 | 0.941–3.861 | 0.073 | 1.374 | 0.423–4.458 | 0.597 |
| Previous congestive heart failure | 0.656 | 0.259–1.660 | 0.373 | 1.180 | 0.305–4.570 | 0.810 |
| Left ventricular ejection fraction | 0.968 | 0.935–1.002 | 0.068 | 0.937 | 0.888–0.990 | 0.020 |
| Left ventricular end diastolic dimension | 1.149 | 0.683–1.934 | 0.601 | 0.882 | 0.368–2.113 | 0.778 |
| ST-T changes on the ECG | 1.391 | 0.710–2.726 | 0.336 | 2.019 | 0.645–6.326 | 0.228 |
| General anesthesia | 1.312 | 0.655–2.627 | 0.443 | 5.575 | 1.822–17.060 | 0.003 |
| Peak cardiac troponin I ≥ 45 ng/L (%) | 3.167 | 1.557–6.444 | 0.001 | 2.175 | 0.651–7.274 | 0.207 |
MI myocardial infarction, CI confidence interval, ECG electrocardiography, cTnI cardiac troponin I
Fig. 2.
Incremental prognostic value of sequential RCRI, cTnI, and LVEF. Global Chi-square on the Y axis indicates the incremental prognostic value of sequential RCRI, cTnI, and LVEF. RCRI revised cardiac risk index; cTnI cardiac troponin I; LVEF left ventricular ejection fraction
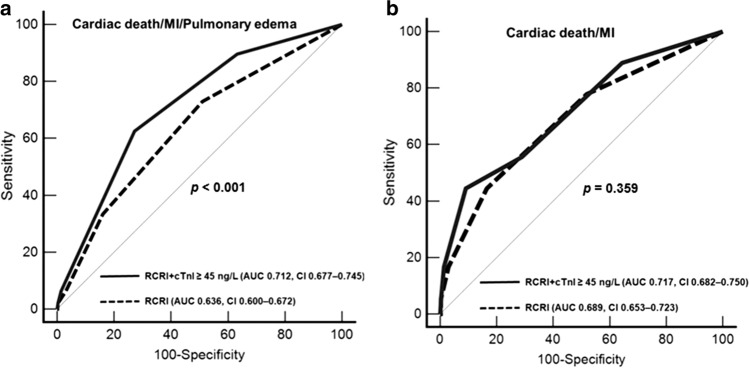
When the RCRI score was reclassified by summing the RCRI and peak cTnI ≥ 45 ng/L, The area under the ROC curve for predicting postoperative cardiac events (cardiac death/MI/pulmonary edema) was 0.636 (95% CI 0.600–0.672) for RCRI. Adding peak cTnI ≥ 45 ng/L to RCRI significantly increased the area under the curve to 0.712 (95% CI 0.677–0.745, p < 0.001) (Fig. 3a). However, the area under the ROC curve for predicting the cardiac death and MI was not significant differences between the RCRI and the combination of RCRI and peak cTnI ≥ 45 ng/L (p = 0.359) (Fig. 3b).
Fig. 3.
a The area under the ROC curve for predicting cardiac death, MI, and pulmonary edema for the RCRI and reclassifying the RCRI with the sum of the RCRI score and cTnI ≥ 45 ng/L as 1 point. b The area under the ROC curve for predicting cardiac death and MI for the RCRI, and the RCRI with the sum of the RCRI score and cTnI ≥ 45 ng/L as 1 point. ROC receiver operating characteristics; MI myocardial infarction; RCRI revised cardiac risk index; cTnI cardiac troponin I
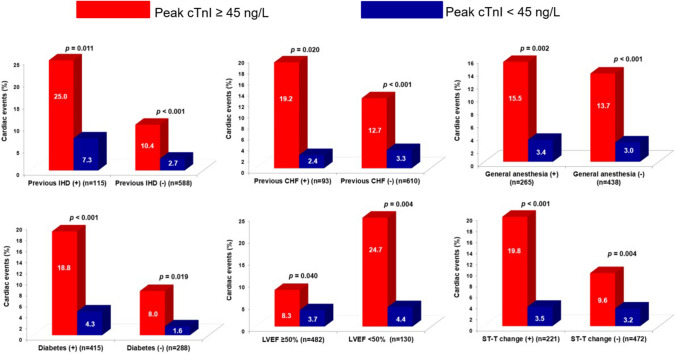
In subgroup analysis, cardiac events were significantly higher in patients with elevated preoperative cTnI levels when compared to those without, regardless of DM, LVEF, ST-T changes by ECG, general anesthesia, and previous IHD and CHF histories (all p for interaction > 0.05) (Fig. 4).
Fig. 4.
Cardiac events in patients with elevated preoperative cardiac troponin I levels compared to those without, regardless of diabetes mellitus, left ventricular ejection fraction, ST-T changes on electrocardiography, general anesthesia, previous ischemic heart disease and congestive heart failure histories
Discussion
Our main findings indicated that elevated preoperative cardiac troponin I (cTnI) levels predicted postoperative cardiac events, including cardiac death, myocardial infarction (MI), and pulmonary edema in patients with end stage renal disease (ESRD) undergoing non-cardiac surgery. In addition, combined the revised cardiac risk index (RCRI) and cTnI scores were more powerful than RCRI scores alone for postoperative cardiac risk stratification.
Methods predicting postoperative cardiac events using appropriate preoperative evaluations, e.g., risk stratification, have been comprehensively studied [9, 14–16]. The RCRI was designed to predict postoperative MI, pulmonary edema, ventricular fibrillation or cardiac arrest, and complete heart block. The RCRI model for cardiac perioperative risk stratification has been recommended by several guidelines, which also suggested preoperative cardiac troponin measurements may be helpful in high-risk patients, such as those with chronic kidney disease (CKD).
Several studies have investigated if preoperative cardiac troponin levels could predict postoperative cardiac complications; Weber et al. observed that high sensitivity cardiac troponin T levels helped predict prognosis after non-cardiac surgery [17–19]. In addition, elevated preoperative cTnI levels were associated with mortality and major adverse cardiac events (MACEs) after non-cardiac surgery [18, 20–24]. Similarly, a meta-analysis also reported preoperative elevated levels of cardiac troponin for the risk assessment of postoperative MACEs and mortality [25]. It was also shown that preoperatively increased cardiac troponin could be used to indicate postoperative MACEs and mortality. However, cTnI may be elevated in patients with several diseases or conditions, without ischemic heart disease (IHD). Most of the studies so far have focused on the general population. The general population and patients with ESRD may have different causes of elevated cTnI. Therefore, there is a limitation in applying the results of the general population study to these patients who are likely to have increased cTnI due to other reasons.
With our aging society, the number of patients with CKD is increasing year on year [26]. These patients experience more cardiovascular (CV) comorbidities than the general population and are at a higher risk of cardiac complication after non-cardiac surgery [27]. Moreover, patients with ESRD on dialysis frequently experience cTnI elevation without cardiac symptoms, eg. chest pain, dyspnea, etc. [28]. Therefore, the relationship between preoperative cTnI levels and postoperative cardiac events in previous general population studies has been proven to some extent, it is not reasonable to apply these results to CKD patients and questionable whether preoperative cTnI elevation is helpful in predicting cardiac event after non-cardiac surgery in patients with ESRD on dialysis. Several previous studies have focused on the relationship between cTnI levels and postoperative mortality in patients with ESRD, however, they were limited to relationships between elevated postoperative cTnI and patient prognoses [5–13]. The role of cTnI as a predictor of postoperative cardiac events in patients with ESRD remains unclear.
In this study, 33% of patients with ESRD had elevated cTnI levels before surgery; this potentially was a useful predictor of postoperative cardiac events. In addition, preoperative cTnI levels had an incremental value for the RCRI, an accepted clinical risk score [15]. Patients with ESRD on dialysis are a high-risk group for CV disease, with a very high risk of cardiac events after surgery. Therefore, preoperative risk assessments for this group are more critical than for the general population. However, sufficient preoperative risk assessments are often difficult to perform because the risk of the preoperative examination, eg. myocardial stress test is high. The cTnI assay is easily facilitated by a relatively simple blood test and is useful for evaluating a patient’s surgical risk without additional patient risk. Therefore, we propose cTnI is useful for predicting high-risk cardiac events after surgery in patients with ESRD.
The result that elevated cTnI did not predict postoperative fatal cardiac events, such as death/MI, except pulmonary edema, suggests that elevation of cTnI may have been caused by volume and/or pressure overload in addition to myocardial ischemia in dialysis patients. However, since cardiac events due to volume and/or pressure overload in dialysis patients can also lead to increased mortality and prolonged hospitalization, The results of our study demonstrating that elevation of preoperative cTnI can predict the occurrence of postoperative cardiac events, including pulmonary edema, are still meaningful.
Our study had limitations. Firstly, as it was retrospective in nature, there may be variables that can affect the results. Secondly, in most patients, cTnI testing was performed on the day of admission for surgery. Therefore, the relationship between the time of blood collection and the time of dialysis differed for each patient. Although cTnI may have increased due to volume overloading before dialysis, cTnI levels were normal in 67% of patients in our study. In addition, considering that most of the patients with elevated cTnI had repeated blood tests, we think that the timing of blood tests has insignificant effect on the results of our study. Thirdly, The cTnI used in our study was not the recently used high-sensitive cTnI. Therefore, there is a limit to applying the results of our study to high-sensitive cTnI. Finally, postoperative cTnI levels were not measured in all patients, so we may have missed a hidden cardiac event or a silent MI. However, postoperative cTnI measurements in all patients were not suitable in clinical practice, and most postoperative cTnI measurements were performed in patients with symptomatic or unstable vital signs. Therefore, it is thought that the effect on the study results was negligible.
In conclusion, elevated preoperative cTnI levels are predictors of postoperative cardiac events, including cardiac death, MI, and pulmonary edema in patients with ESRD undergoing non-cardiac surgery. Thus, the addition of cTnI measurements from relatively easy blood tests could help clinicians assess postoperative cardiac risks events in patients with ESRD.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Gansevoort RT, Correa-Rotter R, Hemmelgarn BR, Jafar TH, Heerspink HJ, Mann JF, Matsushita K, Wen CP. Chronic kidney disease and cardiovascular risk: epidemiology, mechanisms, and prevention. Lancet. 2013;382(9889):339–352. doi: 10.1016/S0140-6736(13)60595-4. [DOI] [PubMed] [Google Scholar]
- 2.Matsushita K, van der Velde M, Astor BC, Woodward M, Levey AS, de Jong PE, Coresh J, Gansevoort RT, Chronic Kidney Disease Prognosis Consortium Association of estimated glomerular filtration rate and albuminuria with all-cause and cardiovascular mortality in general population cohorts: a collaborative meta-analysis. Lancet. 2010;375(9731):2073–2081. doi: 10.1016/S0140-6736(10)60674-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.van der Velde M, Matsushita K, Coresh J, Astor BC, Woodward M, Levey A, de Jong P, Gansevoort RT, El-Nahas M, Eckardt KU, Kasiske BL, Ninomiya T, Chalmers J, Macmahon S, Tonelli M, Hemmelgarn B, Sacks F, Curhan G, Collins AJ, Li S, Chen SC, Hawaii Cohort KP, Lee BJ, Ishani A, Neaton J, Svendsen K, Mann JF, Yusuf S, Teo KK, Gao P, Nelson RG, Knowler WC, Bilo HJ, Joosten H, Kleefstra N, Groenier KH, Auguste P, Veldhuis K, Wang Y, Camarata L, Thomas B, Manley T, Chronic Kidney Disease Prognosis Consortium Lower estimated glomerular filtration rate and higher albuminuria are associated with all-cause and cardiovascular mortality a collaborative meta-analysis of high-risk population cohorts. Kidney Int. 2011;79(12):1341–1352. doi: 10.1038/ki.2010.536. [DOI] [PubMed] [Google Scholar]
- 4.Miller-Hodges E, Anand A, Shah ASV, Chapman AR, Gallacher P, Lee KK, Farrah T, Halbesma N, Blackmur JP, Newby DE, Mills NL, Dhaun N. High-sensitivity cardiac troponin and the risk stratification of patients with renal impairment presenting with suspected acute coronary syndrome. Circulation. 2018;137(5):425–435. doi: 10.1161/CIRCULATIONAHA.117.030320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Chapman AR, Anand A, Boeddinghaus J, Ferry AV, Sandeman D, Adamson PD, Andrews J, Tan S, Cheng SF, D'Souza M, Orme K, Strachan FE, Nestelberger T, Twerenbold R, Badertscher P, Reichlin T, Gray A, Shah ASV, Mueller C, Newby DE, Mills NL. Comparison of the efficacy and safety of early rule-out pathways for acute myocardial infarction. Circulation. 2017;135(17):1586–1596. doi: 10.1161/CIRCULATIONAHA.116.025021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Apple FS, Murakami MM, Pearce LA, Herzog CA. Predictive value of cardiac troponin I and T for subsequent death in end-stage renal disease. Circulation. 2002;106(23):2941–2945. doi: 10.1161/01.CIR.0000041254.30637.34. [DOI] [PubMed] [Google Scholar]
- 7.Donnino MW, Karriem-Norwood V, Rivers EP, Gupta A, Nguyen HB, Jacobsen G, McCord J, Tomlanovich MC. Prevalence of elevated troponin I in end-stage renal disease patients receiving hemodialysis. Acad Emerg Med. 2004;11(9):979–981. doi: 10.1197/j.aem.2004.03.022. [DOI] [PubMed] [Google Scholar]
- 8.deFilippi CR, Herzog CA. Interpreting cardiac biomarkers in the setting of chronic kidney disease. Clin Chem. 2017;63(1):59–65. doi: 10.1373/clinchem.2016.254748. [DOI] [PubMed] [Google Scholar]
- 9.Lee TH, Marcantonio ER, Mangione CM, Thomas EJ, Polanczyk CA, Cook EF, Sugarbaker DJ, Donaldson MC, Poss R, Ho KK, Ludwig LE, Pedan A, Goldman L. Derivation and prospective validation of a simple index for prediction of cardiac risk of major noncardiac surgery. Circulation. 1999;100(10):1043–1049. doi: 10.1161/01.CIR.100.10.1043. [DOI] [PubMed] [Google Scholar]
- 10.Bozbas H, Korkmaz ME, Atar I, Eroglu S, Ozin B, Yildirir A, Muderrisoglu H, Colak T, Karakayali H, Haberal M. Serum levels of cardiac enzymes before and after renal transplantation. Clin Cardiol. 2004;27(10):559–562. doi: 10.1002/clc.4960271007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Thygesen K, Alpert JS, Jaffe AS, Chaitman BR, Bax JJ, Morrow DA, White HD, Executive Group on behalf of the Joint European Society of Cardiology (ESC)/American College of Cardiology (ACC)/American Heart Association (AHA)/World Heart Federation (WHF) Task Force for the Universal Definition of Myocardial Infarction Fourth universal definition of myocardial infarction. J Am Coll Cardiol. 2018;72(18):2231–2264. doi: 10.1016/j.jacc.2018.08.1038. [DOI] [PubMed] [Google Scholar]
- 12.Borges FK, Furtado MV, Rossini AP, Bertoluci C, Gonzalez VL, Bertoldi EG, Pezzali LG, Machado DL, Grutcki DM, Rech LG, Magalhães M, Polanczyk CA. Clinical use of ultrasensitive cardiac troponin I assay in intermediate- and high-risk surgery patients. Dis Markers. 2013;35(6):945–953. doi: 10.1155/2013/169356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.van Waes JA, Peelen LM, Kemperman H, Grobben RB, Nathoe HM, van Klei WA. Kinetics of troponin I in patients with myocardial injury after noncardiac surgery. Clin Chem Lab Med. 2017;55(4):586–594. doi: 10.1515/cclm-2016-0301. [DOI] [PubMed] [Google Scholar]
- 14.Norderud K, Egholm G, Thim T, Olesen KKW, Madsen M, Jensen LO, Bøtker HE, Kristensen SD, Maeng M. Validation of the European society of cardiology and European society of anaesthesiology non-cardiac surgery risk score in patients treated with coronary drug-eluting stent implantation. Eur Heart J Qual Care Clin Outcomes. 2019;5(1):22–27. doi: 10.1093/ehjqcco/qcy032. [DOI] [PubMed] [Google Scholar]
- 15.Goldman L, Caldera DL, Nussbaum SR, Southwick FS, Krogstad D, Murray B, Burke DS, O'Malley TA, Goroll AH, Caplan CH, Nolan J, Carabello B, Slater EE. Multifactorial index of cardiac risk in noncardiac surgical procedures. N Engl J Med. 1977;297(16):845–850. doi: 10.1056/NEJM197710202971601. [DOI] [PubMed] [Google Scholar]
- 16.Detsky AS, Abrams HB, Forbath N, Scott JG, Hilliard JR. Cardiac assessment for patients undergoing noncardiac surgery a multifactorial clinical risk index. Arch Intern Med. 1986;146(11):2131–2134. doi: 10.1001/archinte.1986.00360230047007. [DOI] [PubMed] [Google Scholar]
- 17.Weber M, Luchner A, Seeberger M, Mueller C, Liebetrau C, Schlitt A, Apostolovic S, Jankovic R, Bankovic D, Jovic M, Mitrovic V, Nef H, Mollmann H, Hamm CW. Incremental value of high-sensitive troponin T in addition to the revised cardiac index for peri-operative risk stratification in non-cardiac surgery. Eur Heart J. 2013;34(11):853–862. doi: 10.1093/eurheartj/ehs445. [DOI] [PubMed] [Google Scholar]
- 18.Maile MD, Jewell ES, Engoren MC. Timing of preoperative troponin elevations and postoperative mortality after noncardiac surgery. Anesth Analg. 2016;123(1):135–140. doi: 10.1213/ANE.0000000000001309. [DOI] [PubMed] [Google Scholar]
- 19.Nagele P, Brown F, Gage BF, Gibson DW, Miller JP, Jaffe AS, Apple FS, Scott MG. High-sensitivity cardiac troponin T in prediction and diagnosis of myocardial infarction and long-term mortality after noncardiac surgery. Am Heart J. 2013;166(2):325–332. doi: 10.1016/j.ahj.2013.04.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Puelacher C, Lurati Buse G, Seeberger D, Sazgary L, Marbot S, Lampart A, Espinola J, Kindler C, Hammerer A, Seeberger E, Strebel I, Wildi K, Twerenbold R, de Lavallaz du Fay J, Steiner L, Gurke L, Breidthardt T, Rentsch K, Buser A, Gualandro DM, Osswald S, Mueller C, BASEL-PMI Investigators Perioperative myocardial injury after noncardiac surgery: incidence, mortality, and characterization. Circulation. 2018;137(12):1221–1232. doi: 10.1161/CIRCULATIONAHA.117.030114. [DOI] [PubMed] [Google Scholar]
- 21.Kim BS, Kim TH, Oh JH, Kwon CH, Kim SH, Kim HJ, Hwang HK, Chung SM. Association between preoperative high sensitive troponin I levels and cardiovascular events after hip fracture surgery in the elderly. J Geriatr Cardiol. 2018;15(3):215–221. doi: 10.11909/j.issn.1671-5411.2018.03.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Gillmann HJ, Meinders A, Grohennig A, Larmann J, Bünte C, Calmer S, Sahlmann B, Rustum S, Aper T, Lichtinghagen R, Koch A, Teebken OE, Theilmeier G. Perioperative levels and changes of high-sensitivity troponin T are associated with cardiovascular events in vascular surgery patients. Crit Care Med. 2014;42(6):1498–1506. doi: 10.1097/CCM.0000000000000249. [DOI] [PubMed] [Google Scholar]
- 23.Gualandro DM, Puelacher C, LuratiBuse G, Lampart A, Strunz C, Cardozo FA, Yu PC, Jaffe AS, Barac S, Bock L, Badertscher P, du Lavallaz Fay de J, Marbot S, Sazgary L, Bolliger D, Rentsch K, Twerenbold R, Hammerer-Lercher A, Melo ES, Calderaro D, Duarte AJ, de Luccia N, Caramelli B, Mueller C, TropoVasc and BASEL-PMI Investigators Comparison of high-sensitivity cardiac troponin I and T for the prediction of cardiac complications after non-cardiac surgery. Am Heart J. 2018;203:67–73. doi: 10.1016/j.ahj.2018.06.012. [DOI] [PubMed] [Google Scholar]
- 24.Alcock RF, Kouzios D, Naoum C, Hillis GS, Brieger DB. Perioperative myocardial necrosis in patients at high cardiovascular risk undergoing elective non-cardiac surgery. Heart. 2012;98(10):792–798. doi: 10.1136/heartjnl-2011-301577. [DOI] [PubMed] [Google Scholar]
- 25.Zhao BC, Liu WF, Deng QW, Zhuang PP, Liu J, Li C, Liu KX. Prevention of vascular events after non-cardiac surgery (PREVENGE) investigators, meta-analysis of preoperative high-sensitivity cardiac troponin measurement in non-cardiac surgical patients at risk of cardiovascular complications. Br J Surg. 2020;107(2):e81–e90. doi: 10.1002/bjs.11305. [DOI] [PubMed] [Google Scholar]
- 26.Saran R, Robinson B, Abbott KC, Agodoa LYC, Bhave N, Bragg-Gresham J, Balkrishnan R, Dietrich X, Eckard A, Eggers PW, Gaipov A, Gillen D, Gipson D, Hailpern SM, Hall YN, Han Y, He K, Herman W, Heung M, Hirth RA, Hutton D, Jacobsen SJ, Jin Y, Kalantar-Zadeh K, Kapke A, Kovesdy CP, Lavallee D, Leslie J, McCullough K, Modi Z, Molnar MZ, Montez-Rath M, Moradi H, Morgenstern H, Mukhopadhyay P, Nallamothu B, Nguyen DV, Norris KC, O'Hare AM, Obi Y, Park C, Pearson J, Pisoni R, Potukuchi PK, Rao P, Repeck K, Rhee CM, Schrager J, Schaubel DE, Selewski DT, Shaw SF, Shi JM, Shieu M, Sim JJ, Soohoo M, Steffick D, Streja E, Sumida K, Tamura MK, Tilea A, Tong L, Wang D, Wang M, Woodside KJ, Xin X, Yin M, You AS, Zhou H, Shahinian V. US renal data system 2017 annual data report: epidemiology of kidney disease in the United States. Am J Kidney Dis. 2018;71(3 Suppl 1):A7. doi: 10.1053/j.ajkd.2018.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Brown JH, Hunt LP, Vites NP, Short CD, Gokal R, Mallick NP. Comparative mortality from cardiovascular disease in patients with chronic renal failure. Nephrol Dial Transplant. 1994;9(8):1136–1142. doi: 10.1093/ndt/9.8.1136. [DOI] [PubMed] [Google Scholar]
- 28.Long B, Belcher CN, Koyfman A, Bronner JM. Interpreting troponin in renal disease: a narrative review for emergency clinicians. Am J Emerg Med. 2020;38(5):990–997. doi: 10.1016/j.ajem.2019.11.041. [DOI] [PubMed] [Google Scholar]