Abstract
The genome of the obligate intracellular bacterium Chlamydia pneumoniae CWL029 encodes a family of 21 proteins with predicted outer membrane localization. These polymorphic membrane proteins (Pmps) are heterogeneous in both amino acid sequence and predicted size but are unified by the conserved amino acid motifs GGAI and FXXN repeated in the N-terminal half of each protein. Reverse transcriptase PCR analysis showed that all pmp genes are transcribed. To determine whether all proteins are expressed, specific antisera were generated by immunization with mutually exclusive synthetic peptides representing each of the 21 predicted Pmps. Each antiserum reacted with, and was typically immunospecific for, the corresponding peptide immunogen by enzyme-linked immunosorbent assay. Western blot analyses of purified elementary bodies showed that 11 of the 21 Pmps were detectable. Attempts to demonstrate by Sarykosyl fractionation that the Pmps were localized to the outer membrane revealed that several of the Pmps were unstable and readily degraded. Analyses of additional C. pneumoniae strains showed that although some Pmps are conserved, others vary between strains, in both molecular weight and level of expression.
Chlamydia pneumoniae has been recognized as an important cause of acute respiratory tract infections, including pneumonia, pharyngitis, and bronchitis (6). It is estimated that 50 to 70% of adults are seropositive, with primary infection occurring in teenage years and prevalence rising with age (3). Recent attention has focused on the association between C. pneumoniae and atherosclerosis (5, 12). The biology of Chlamydia is characterized by its obligate intracellular growth and by a unique biphasic developmental cycle, alternating between an infectious extracellular form, the elementary body (EB), and the metabolically active intracellular form, the reticulate body.
Proteins exposed on the surface of infectious organisms are primary mediators in pathogen infection and transmission. The recent completion of the C. pneumoniae CWL029 genome sequence has provided complete information on the coding capacity of this organism, including a comprehensive group of genes that are predicted to encode surface proteins (9). The C. pneumoniae genome encodes a family of 21 polymorphic membrane proteins (Pmps), which are predicted to be localized to the bacterial outer membrane (9). These proteins are polymorphic in amino acid sequence and predicted size but are related by the conserved amino acid motifs GGAI and FXXN, which are repeated numerous times in the amino-terminal half of each protein (8, 9). Phylogenetic analysis groups these proteins into six related families, implying at least six specific roles for the proteins in chlamydial biology (8).
Variation in amino acid sequence or expression of surface proteins of some infectious organisms plays a vital role in establishing an infection and ensuring pathogen survival. This variation can be seen in phase, functional, and antigenic diversity (1, 4). The function of the Pmps in Chlamydia growth and development remains unknown. Indeed, it is unknown whether the variation evidenced by the genome sequence is utilized by the bacterium during its developmental cycle, for interaction with the host cell, or between strains. The aim of this study was to use existing genome sequence data and, through functional genomics, to examine Pmp expression at both the transcriptional and translational levels.
MATERIALS AND METHODS
Cell lines and bacterial strains.
HEp-2 cells were routinely grown in Iscoves's modified Dulbecco's medium (University of California San Francisco Cell Culture Facility) supplemented with 10% fetal calf serum, 0.1 mg of vancomycin per ml, and 0.1 mg of streptomycin per ml. C. pneumoniae strains CWL029 (kindly provided by C. Black, Centers for Disease Control and Prevention), TW-183, and AR-29 (both kindly provided by C.-C. Kuo, University of Washington) were centrifuged onto HEp-2 cells and incubated for 72 h in the presence of 1 μg of cycloheximide per ml as previously described (7). EB were purified on discontinuous Renografin (E. R. Squibb & Sons, Princeton, N.Y.) density gradients as previously described (11) and frozen at −80°C until use.
RT-PCR analysis.
Oligonucleotide primers were designed to amplify a 300 to 400-bp fragment of each pmp gene (Table 1). Total RNA was extracted from uninfected HEp-2 cells and C. pneumoniae CWL029-infected HEp-2 cells at 72 h postinfection. Trizol (Life Technologies, Rockville, Md.) was added directly to the cell monolayers, and RNA was extracted and purified as specified by the manufacturer. RNase-free DNase (Life Technologies) was used to ensure complete removal of contaminating DNA. cDNA was transcribed from 2 μg of each RNA, using 1 μM of the reverse oligonucleotide primer and murine leukemia virus reverse transcriptase (RT) (Promega, Madison, Wis.). The DNA was then amplified by PCR using 1 μM of each oligonucleotide primer and Taq polymerase (Perkin-Elmer, Foster City, Calif.). To ensure chlamydial specificity, reactions were performed for each gene using uninfected HEp-2 host cell RNA. In addition, matched reactions in mixtures lacking RT were performed for each gene to ensure complete removal of contaminating DNA.
TABLE 1.
Oligonucleotide primers used for RT-PCR analysis of pmp genes
| Gene | Sequence (5′ to 3′) of:
|
|
|---|---|---|
| Forward primer | Reverse primer | |
| pmpG1 | GGGAGTTTAACATCGATTTCTGTACC | GGCAATGGAAACCTTCTGAAAACG |
| pmpG2 | CGTGATTTTAGTGATTCG | GAGAAATCGCTTAAATTTGATTTTGCTACC |
| pmpG3 | CCACGTTTATATGGGTTTTCTTCG | CTTGAGGAGGTATTTTCAG |
| pmpG4 | CGAGGGTGTCATATGAGATCG | CCCAGAAGCTGCTGTATTGCTAAC |
| pmpG5 | GGATCAACCAGCCTGTCACT | GTCTTGCAAGGTATGGTTCTC |
| pmpG6 | CCACGTATCTTTGACGCTAGC | CGGAGAATCTTCGAAAGAATAACGAGTTCC |
| pmpG7 | CCTCTTCTTGAACTTACCACC | TCGCAATTCCTGAAAGCC |
| pmpG8 | CCCAAGCATCTTTCTATGAAAATACCC | GAATGTTAGGGCTTTATCAGC |
| pmpG9 | TACACTCCTCCAGCCCAAAC | TCGACTGAATAGAACAAGCTCC |
| pmpG10 | GGGTTTATGAAATCGCAATTTTCC | CGCTAAGAAAGTAAGACTCGAAAATCC |
| pmpG11 | TCTTTCCAACCCACATCCTC | CTACCCCTCAGTAAAAATGCC |
| pmpG12 | GAGGAAACCATGACCATACTTCGAAATTTTCTTACC | CTGAGGTGAACGCTAAGTAAG |
| pmpG13 | CATCTCTACGTGCAAATGAAAACG | CTGACTAAATCCTGAAAAACTCAAGGG |
| pmpH | GGAGATCTGCAACGTAGC | AAATGTGGATTTTAGTCCTGTAGACACTCG |
| pmpE/F1 | CTTTGAGAAGAATGTCTGTCCC | CCCACTATAAATCCCTCCTCC |
| pmpE/F2 | CTACTTTGTCAACAATTCCGTC | TAAATCGCCCCTCCTGAAC |
| pmpE/F3 | CTACTTTGTCAACAATTCCGTC | TAAATCGCCCCTCCTGAAC |
| pmpE/F4 | ATCCAGAACACCTCGGAAC | AATCCATAGCTTGGGAGCAAC |
| pmpA | GGCTATAACTTATTAGGACTC | GGTCACATTGGATTCCGAGAAGAGCCC |
| pmpB/C | GTGAATATATCTGATAGG | GGAGTGTAGGTTTGAGGATCG |
| pmpD | CGTTTTGAGAATGTCGCC | CCCTGTCACATCGAACTCGAAAATAATACG |
Selection and conjugation of synthetic peptides.
Table 2 lists the 20-mer peptides selected for synthesis. Peptides were selected from the C. pneumoniae CWL029 genome sequence as being specific for each of the Pmps based on the following criteria: they (i) were less than 50% identical to any other 20-mer stretch of amino acids in the entire genome, (ii) did not contain any long stretches of internal hydrophilic residues, and (iii) did not contain any internal cysteine residues. A second peptide was chosen for a Pmp when the first failed to generate an antibody that recognized EB protein by immunoblot analysis. When the Pmp contained predicted frameshifts, the second peptide was chosen from the opposite side of the frameshift from the first peptide. All peptides had a C-terminal cysteine added to the native sequence to facilitate conjugation. Peptides were synthesized using solid-phase techniques by Genemed Synthesis Inc. (South San Francisco, Calif.) and conjugated to keyhole limpet hemocyanin (KLH) using Sulfo-SMCC cross-linker (Pierce, Rockford, Ill.).
TABLE 2.
Synthetic peptides used for production of Pmp-specific sera
| Pmp paralog | Peptide |
|---|---|
| CpnG1 | 385-SEADGSTQLNINGDPKNKEY |
| CpnG1.1 | 112-QDPQATARFSGFSTLSFIQS |
| CpnG2 | 296-VTGSTSVADALNINSPDTGD |
| CpnG3 | 391-TGSSTTVTDVLKVNETPADS |
| CpnG3.1 | 96-QTVDAGTVAGAAVNSSVVDK |
| CpnG4 | 711-LGESKTLAKILSGNSDSLPF |
| CpnG4.1 | 260-NKAGANPDPILTLSGNESLH |
| CpnG5 | 731-EDTLEFRSPQGFYTDSSSEA |
| CpnG5.1 | 287-STGTISITGSDSINVIGNTS |
| CpnG6 | 839-MEAPASGGTIEELVINPVVK |
| CpnG7 | 721-FDIANFLWGKATRAPWVLSE |
| CpnG8 | 717-RAFLGGLPMPSFGSITDMLK |
| CpnG8.1 | 497-ALEGNKSVSIETAGANKTIT |
| CpnG9 | 389-TSITAALSDALNLNGPDLAG |
| CpnG10 | 725-LLDKLPGSWSHKPLVLEGQL |
| CpnG10.1 | 251-NSVTATAGNGGALSGDADVT |
| CpnG11 | 380-SEGTSSDVLKINNGSAGALN |
| CpnG12 | 295-HSESHKITDLVINAPEGKET |
| CpnG12.1 | 161-SGAAIYTPYLLGSKASRPSV |
| CpnG13 | 756-GHGHSNVYFATVYSNITKSL |
| CpnG13.1 | 171-YNRSAGFGAAIRGTSITIEN |
| CpnH | 424-HSLPTTSPSNKDITINANGA |
| CpnE/F1 | 386-HELPSSFPILFNFETGHTGT |
| CpnE/F1.1 | 751-VSLAYMFTSEHTHTMYQGLL |
| CpnE/F2 | 422-HQHPTTNPLIFNPNANHQGT |
| CpnE/F2.1 | 117-TITKNRGPLIFFSNRGLNNA |
| CpnE/F3 | 422-HDQTTTDPIVFNYEPHHLGT |
| CpnE/F3.1 | 672-NALLGIRILPPQNLKEHDLE |
| CpnE/F4 | 345-LQRYTIQNSIQKFNPNPEHL |
| CpnA | 417-SATENYNSLYINHQRLLEAG |
| CpnA.1 | 679-VFGGPVYQIMEQNPKQSSNN |
| CpnB/C | 1186-VSTKETNAQELKLNEKATST |
| CpnB/C.1 | 448-YAVGQVTLEDIANLKMTNNT |
| CpnD | 1026-YEENTIRGLPDKDVSPLSAP |
| CpnD.1 | 461-AGTVDLNETISEVAFKQNTA |
Immunizations.
Six-week-old BALB/c mice were injected intraperitoneally with KLH-conjugated peptide suspended in complete Freund's adjuvant (Sigma, St. Louis, Mo.). Five mice were immunized per group. Control mice received unconjugated KLH. The mice were boosted subcutaneously on days 14 and 28 with conjugated peptide suspended in incomplete Freund's adjuvant. The animals were exsanguinated on day 38, and their serum was frozen at −20°C.
Indirect enzyme-linked immunosorbent assay to determine the antipeptide antibody titer.
Polystyrene 96-well plates (Immulon 2; Dynex Technologies, Chantilly, Va.) were coated for 3 h at 37°C with unconjugated peptide or KLH (100 ng/well) in phosphate-buffered saline (PBS) (pH 7.4). The wells were then blocked with 5% dried nonfat milk in PBS–0.1% Tween 20 (PBS-Tween) overnight at 4°C. Subsequently, the plates were incubated for 1 h with dilutions of sera in PBS-Tween and then for 1 h with a 1:10,000 dilution of horseradish peroxidase-conjugated goat anti-mouse immunoglobulin G antibodies (Promega). All incubations were carried out at 37°C; washes were done with PBS-Tween. Bound antibody was detected using 3,3,′5,5′-tetramethylbenzidine (Sigma). Absorbance values were read at 450 nm.
Preparation of the chlamydial outer membrane complex (COMC).
Purified C. pneumoniae EB were suspended in PBS containing 2% Sarkosyl (Sigma) and 5 mM EDTA and incubated for 30 min at 37°C. Detergent-insoluble complexes were pelleted by centrifugation (10,000 × g). The pellet was suspended in 2% Sarkosyl with 5 mM MgCl2 and digested with 50 μg of DNase per ml and 50 μg of RNase per ml for 30 min at 37°C. Insoluble material was pelleted by centrifugation (10,000 × g), washed once with PBS, and suspended in PBS.
Pmp protein stability.
To determine Pmp protein stablity, purified C. pneumoniae EB were suspended in PBS containing 2% Sarkosyl and 5 mM EDTA. Protease inhibitors were added to aliquots, and samples were removed at various times and analyzed by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and immunoblotting (see below). The following protease inhibitors (Boehringer-Mannheim, Indianapolis, Ind.) were evaluated: antipain dihydrochloride (50 μg/ml), bestatin (40 μg/ml), chymostatin (60 μg/ml), E-64 (10 μg/ml), leupeptin (5 μg/ml), pepstatin (0.7 μg/ml), phosphoramidon (300 μg/ml), Pefabloc (1 mg/ml), and aprotinin (2 μg/ml).
SDS-PAGE and immunoblot analysis.
Proteins of purified chlamydial EB and COMC were solubilized in SDS sample buffer containing 50 mM dithiothreitol, heated to 100°C for 5 min, and separated by SDS-PAGE (8% polyacrylamide gels). Proteins were visualized using Coomassie blue stain or electrotransferred to nitrocellulose membranes. The membranes were blocked overnight at 4°C in PBS containing 5% bovine serum albumin. They were then incubated for 1 h at room temperature in primary antibody diluted 1:1,000 in PBS–3% bovine serum albumin. After they were washed in PBS-Tween to remove unbound antibody, bound antibody was detected using a 1:5,000 dilution of anti-mouse alkaline phosphatase secondary antibody (Promega). A stabilized alkaline phosphatase substrate solution (Promega) was used for colorimetric detection.
RESULTS
All pmp genes are transcribed during infection.
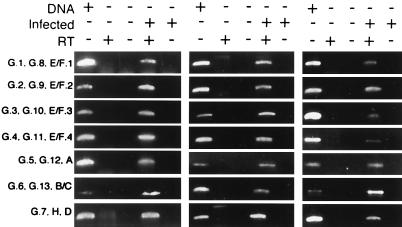
To determine pmp transcription during chlamydial growth, total RNA from C. pneumoniae-infected HEp-2 cells was isolated and analyzed by RT-PCR using primers specific for each of the 21 genes. As a control, analyses were also carried out for each gene using uninfected HEp-2 cells. Transcripts were detected for each gene, demonstrating that all pmp genes are transcribed during infection (Fig. 1). This included six pmp genes that were predicted to be truncated because of frameshifts; therefore, even genes that are apparently not capable of producing a functional protein are transcribed.
FIG. 1.
Analysis of pmp gene transcription. Each pmp gene was amplified by RT-PCR using primer pairs listed in Table 1 and with C. pneumoniae DNA as a positive control. RNA obtained from uninfected or infected cells was used as the template with or without RT in the assay mixture.
Generation of antisera specific for each Pmp.
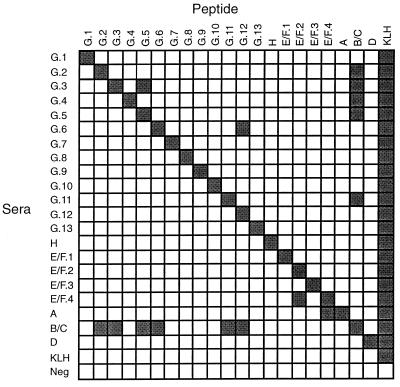
To determine whether pmp genes are translated during infection, 21 groups of mice were immunized with synthetic peptides specific for each of the Pmp proteins. By ELISA, each antiserum reacted with its corresponding peptide immunogen to very high titer, with the weakest reactivity providing 50% of the maximum titer of 1:16,000 (data not shown). Although each peptide immunogen was selected for lack of amino acid sequence identity, the 21 antisera were tested for reactivity to each of the 21 peptides by ELISA. Mutually exclusive reactivity for the homologous serum-peptide pairs was shown for each with a few exceptions (Fig. 2). Antisera for PmpB/C showed detectable cross-reactivity to peptides representing PmpG2, PmpG3, PmpG5, PmpG6, PmpG11, and PmpG12. Likewise, the antisera from some of these G family-based peptides also cross-reacted to the B/C peptide, thus having a two-way cross-reaction, with the exception of PmpG4, PmpG6, and PmpG12. Several antisera displayed nonreciprocal cross-reactivity, including PmpG3-reactive serum to PmpG5 peptide, PmpG6 serum to PmpG12, PmpE/F4 serum to PmpE/F2, and PmpA serum to PmpE/F4.
FIG. 2.
Specificity of Pmp peptide-elicited sera. Strong reactivity, represented by filled squares, was found for each serum to each peptide used for immunization by ELISA following adsorption of peptide to wells of microtiter plates. If the reactivity of a peptide had a twofold or greater optical density with respect to that of the homologous reaction, it is shown as a filled square.
Pmp expression during infection.
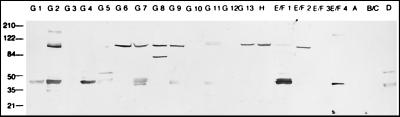
Since each of the pmp genes was transcribed, protein expression was evaluated by immunoblotting of C. pneumoniae CWL029 EB lysates using each of the peptide-specific antisera. Of the 21 sera, 8 (anti-PmpG2, anti-PmpG6 through anti-Pmp9, anti-PmpG13, anti-PmpH, and anti-PmpE/F2) detected strong immunospecific antigens and 2 sera (anti-Pmp-G11 and anti-Pmp-E/F1) reacted weakly to antigens with molecular weights of approximately 90,000 to 100,000 (Fig. 3). A weak band for anti-PmpE/F4 was revealed in some experiments but not others. No Pmp-specific reactivity was detected for the remaining sera by immunoblot analysis regardless of high antipeptide reactivity. Despite the cross-reactivity observed for some of the Pmp sera by peptide ELISA, such as PmpB/C sera with PmpG-family proteins, these sera did not react to C. pneumoniae proteins by immunoblot analysis. Therefore, at least half of the transcribed pmp genes are translated into stable proteins; however, the presence of weakly reactive bands should be interpreted with caution since we cannot unequivocally rule out the possibility that these are weak because they are due to cross-reactivity with other Pmps. Two immunoblot-detected proteins did not show a molecular weight similar to that predicted by the coding sequence. PmpG6 migrated at approximately 100,000 (Fig. 3), although it is predicted to encode a 144-kDa protein. PmpG11 was consistently detected as a set of doublet antigens with molecular weights of approximately 105,000 and 110,000 (Fig. 3). PmpG8 revealed an additional component of ∼80,000 (Fig. 3).
FIG. 3.
Detection of Pmps in EB lysates of CWL029 using a peptide-derived serum in immunoblots. This is a composite figure following analysis of each of the 21 Pmp sera. Prominent immunoreactive bands are present at molecular weights of approximately 90,000. pmpG3, pmpG4, pmpG5, pmpG10, pmpG12, and pmpE/F3 are not predicted to produce proteins because of gene sequence frameshifts or truncation. The sizes of molecular weight markers are shown on the left in thousands.
Because some of the pmp genes in CWL029 have predicted frameshifts (8, 9) (PmpG3, PmpG4, PmpG5, PmpG10, PmpE/F3) or are truncated (PmpG12), it was not surprising that sera specific for these proteins did not detect antigens in EB lysates. For the frameshifted genes, peptides were synthesized and antisera were produced to each of the polypeptides flanking the frameshift, but none of these paired sera reacted by immunoblot analysis (data not shown). However, the pmpG1, pmpA, pmpB/C, and pmpD genes are complete (8, 9) and were transcribed, but no specific detectable bands were observed by immunoblot analysis, suggesting that these are either not stable or not translated.
Variation in protein stability among different Pmps.
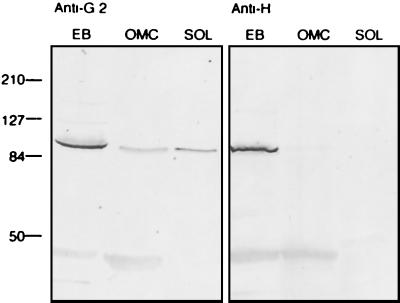
The COMC is structurally unique among bacteria and is operationally defined as consisting of the proteins that remain in a macromolecular complex following extraction with Sarkosyl and are not found in the soluble fraction without the addition of a reducing reagent such as dithiothreitol or 2-mercaptoethanol (2). At least two of the Pmp proteins for C. psittaci and C. pneumoniae are expressed and localized to the surface of EB (10, 14). Initial testing of Pmp sera by immunoblotting COMC preparations gave inconsistent results since Pmp sera that detected antigens using EB lysates either revealed bands in both the insoluble and soluble fractions or, paradoxically, were not detected in either fraction. For example, PmpG2 was detected in the COMC-insoluble fraction and a weaker antigen was also seen in the soluble fraction (Fig. 4). For PmpH, although strongly present in EB lysates, an immunoreactive band was not detected in either the COMC-insoluble or -soluble fractions (Fig. 4). Among the Pmps detected in EB lysates, PmpE/F2, PmpE/F4, PmpG8, PmpG13, and PmpH were notably absent following Sarkosyl extraction whereas PmpG2, PmpG6, PmpG7, PmpG9, and PmpG11 were detected in COMC preparations (data not shown).
FIG. 4.
Instability of Pmps following Sarkosyl lysis of EB. Immunoblots of EB in SDS sample buffer (EB) and the insoluble (OMC) and soluble (SOL) fractions following extraction in Sarkosyl are shown. The immunoblots were probed with PmpG2- or PmpH-specific sera. The sizes of molecular weight markers are shown on the left in thousands.
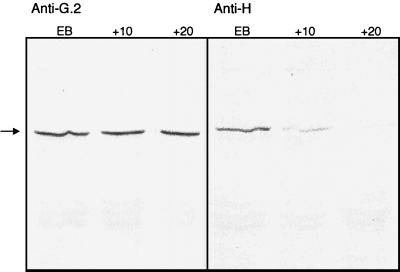
The finding that some of the Pmps were present in EB lysates but not detectable in either the COMC-insoluble or -soluble fractions suggested that these Pmps were not stable during the solubilization procedure. To test this hypothesis, samples were withdrawn at different times following the addition of Sarkosyl, immunoblotted, and probed with anti-PmpG2 and anti-PmpH. A progressive loss of protein was observed over the time of incubation for PmpH but not for PmpG2 (Fig. 5). To test whether these were lost to proteolysis, a variety of protease inhibitors were added with the Sarkosyl solubilization buffer and probed with anti-PmpH. Antipain dihydrochloride and, especially, chymostatin significantly prevented the loss of immunoreactivity detected by immunoblot analysis (Fig. 6). These data show that PmpH is susceptible to proteolysis, probably by an endogenous chlamydial protease released following lysis by Sarkosyl.
FIG. 5.
Degradation of PmpH following Sarkosyl extraction of EB. Aliquots were collected at 10 and 20 min following the addition of 2% Sarkosyl and probed by immunoblot analysis using either anti-PmpG2 or anti-PmpH sera.
FIG. 6.
Inhibition of PmpH degradation by addition of protease inhibitors. EB were either not extracted (lane 1) or extracted with 2% Sarkosyl in the presence of the protease inhibitor antipain-dihydrochloride (lane 2), bestatin (lane 3), chymostatin (lane 4), E-64 (lane 5), leupeptin (lane 6), pepstatin (lane 7), phosphoramidon (lane 8), Pefabloc (lane 9), or aprotinin (lane 10) or in the absence of protease inhibitor (lane 11).
Evidence for strain variation among different C. pneumoniae strains.
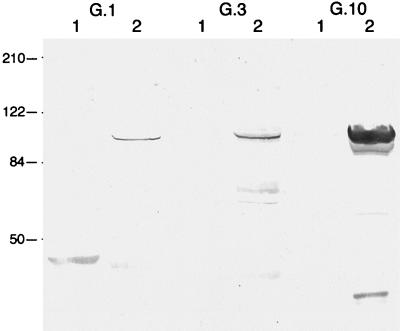
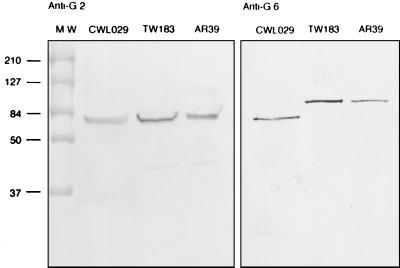
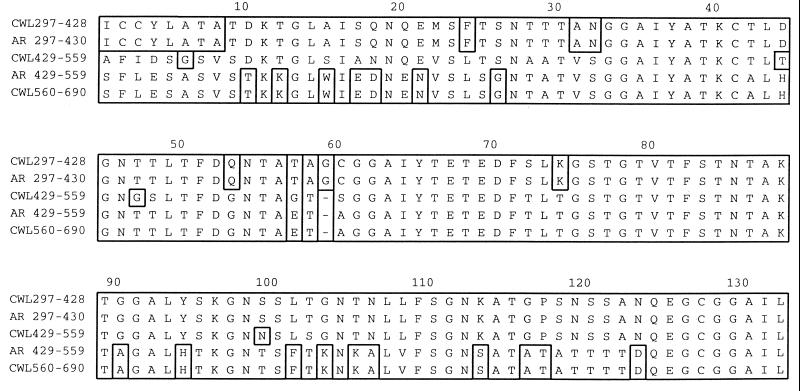
The foregoing experiments were all conducted with the CWL029 strain of C. pneumoniae used in the original genome sequence (9). Given the variation of Pmp expression, it is pertinent to consider whether there is stability of Pmp expression phenotype among strains of C. pneumoniae. Thus, each of the Pmp sera were tested using EB lysates of TW183 (7) and several Pmp sera were also tested with AR39 (16). Similar antigens were detected by immunoblot analysis for each of the strains (data not shown), including the doublet antigens for PmpG11, with several exceptions. Anti-PmpG1, anti-PmpG3, and anti-PmpG10, which did not detect antigens for their respective gene products in CWL029, detected antigens in TW183 (Fig. 7). This was surprising since pmpG3 and pmpG10 are predicted to contain frameshifted open reading frames for CWL029. The sequences flanking the frameshift for pmpG3 and pmpG10 were amplified from TW183 and sequenced. The frameshift for pmpG10 was resolved in TW183 from the loss of two guanine bases and was also resolved in AR39 by the addition of a guanine residue. However, the sequence for pmpG3 appeared to conserve the frameshift. Interestingly the immunoreactive band to PmpG6 for CWL029 had a molecular weight of 100,000, although the protein is predicted to be 144,000 Da. Moreover, a 130,000-molecular-weight antigen was detected for both TW183 and AR39 (Fig. 8). PmpG6 for AR39 is predicted to have a molecular weight of 131,000 because its gene contains 393 fewer nucleotides (16) compared to the CWL029 homolog. The pmpG6 gene encodes a 131-amino-acid sequence that is repeated in CWL029 three times (8) but is only repeated in AR39 twice (Fig. 9). Based on sequence homology of the repeats between C. pneumoniae strains, the additional repeat in CWL029 is the second in the series (amino acids 429 to 559). Its origin is probably from the first repeat since its sequence is most similar to the first of the repeats.
FIG. 7.
Detection of PmpG1, PmpG3, and PmpG10 in TW183 (lane 2) but not in CWL029 (lane 1). The sizes of molecular weight markers are shown on the left in thousands.
FIG. 8.
Variation in PmpG6 molecular weight among C. pneumoniae strains. Immnoblots of EB lysates obtained from C. pneumoniae CWL029, TW183, and AR39 probed with anti-PmpG2 and anti-PmpG6 are shown. The sizes of molecular weight markers are shown on the left in thousands.
FIG. 9.
Alignment of the PmpG6 three internal tandem repeat sequences for CWL029 (9) and two for AR39 (16).
DISCUSSION
One of the many surprising findings following the completion of the C. pneumoniae genome sequence was the presence of a large family of 21 related pmp genes predicted to encode outer membrane proteins (9). The diversity of these genes is remarkable since there is little nucleotide or amino acid sequence identity (<50%), but they are strongly related by the presence of conserved amino acid GGA(I,L,V) and FXXN repeats in the aminoterminal half of each protein. The pmp family represents >5% of the total coding capacity of the genome and 22% of the increased coding capacity of C. pneumoniae versus C. trachomatis (8). Thus, considerable commitment has been made to maintaining and diversifying this family in C. pneumoniae. Phylogenetic analysis suggests the presence of six related families, each with at least one C. trachomatis and one C. pneumoniae ortholog (8). The G family in C. pneumoniae contains 13 protein paralogs, but 4 of these are predicted to encode frameshifts (9). Comparison of pmp genes based on those determined for CWL029 (9) and AR39 (16) shows that the sequences are nearly identical between these two C. pneumoniae strains, with only three Pmps containing one or two conserved amino acid changes (PmpE/F4, PmpG10, and PmpG8), including genes with predicted frameshifts.
The pmpG10 gene contains a string of 13 guanine residues for CWL029 that results in a shift out of reading frame; however, AR39 contains 14 guanine residues in this string and is not frameshifted. TW-183 pmpG10 contains 11 guanine residues and is not frameshifted. Consistent with these sequence predictions, anti-PmpG10 serum reacted with antigens in both AR39 and TW183 but not in CWL029. It also appears significant that the CWL029 strain with a different passage history contains 14 guanine residues and produces a protein product (10). These data suggest that a slip-strand mechanism altering the number of guanine residues within and between strains of C. pneumoniae accounts for variation in the expression of PmpG10. This conclusion is supported by the recent report describing clonal variation in the expression of PmpG10 for another strain of C. pneumoniae (A. S. Pedersen, S. Birkelund, and G. Christiansen, Proc. Fourth Meet. Eur. Soc. Chlamydia Res., p. 44, 2000). The C. trachomatis pmpG gene (CT871) contains 9 guanine residues (the longest string of guanine residues in the genome), suggesting that phase variation is a common phenotype for PmpG. However, poly(G) tracts are not a characteristic of other pmp genes. Another difference between AR39 and CWL029 is pmpG6, that in CWL029 encodes 131 additional amino acids due to an apparent tandem duplication of 393 nucleotides. The amplification of the pmp gene family and the confidently predicted frameshifts in CWL029 and AR39 suggest a specific molecular mechanism to promote functional or antigenic diversity.
To begin assessment of the role and mechanisms of Pmp diversity, we evaluated the expression of each of the pmp genes. RT-PCR analysis revealed that each of the pmp genes in C. pneumoniae, including those with predicted frameshifts, is transcribed. This is consistent with the previous finding that each of the nine pmp genes in C. trachomatis is transcribed (13). Thus, chlamydiae expend a considerable metabolic cost in pmp transcription despite the potential lack of production of a functional protein.
We next assessed whether the transcribed pmp genes are translated. To produce a reagent specific to each Pmp, 20-mer polypeptide sequences were selected that were unique to each of the 21 predicted coding sequences and the synthetic peptides were used to produce monospecific antisera that could be used as probes for the mutually exclusive detection of each Pmp. High-titer antiserum was elicited to each polypeptide and, with a few exceptions, was found to be highly specific for the respective polypeptide immunogen and thereby suitable for probing the expression of each Pmp paralog. The cross-reactivity observed between B/C- and G-family peptides and sera, although potentially problematic, was mitigated by the finding that the anti-B/C serum did not react to EB proteins. Similar findings for the other antisera with cross-reactive peptides suggest that those antisera reactive to proteins in EB lysates were suitable for the detection of specific Pmp paralogs.
Using each of the Pmp-specific sera to probe immunoblots of EB lysates enabled the detection of 11 different Pmps. No reactivity was observed for the remaining 10 Pmp, which included those encoded by the five frameshifted genes and one truncated gene. Production of antisera to the amino-terminal open reading frame upstream of the predicted frameshift also did not reveal a protein product. Based on the nucleotide sequence and complete coding sequence, it was expected that PmpA, PmpB/C, and PmpD should be expressed; however, no immunoreactive bands were detected in CWL029 or TW183. In contrast anti-PmpG1, whose respective gene also appears to encode a complete protein, was not reactive to lysates of CWL029 but detected an antigen in TW183. Testing of TW183 EB lysates revealed immunoreactive bands for PmpG3 and PmpG10, both with sequence frameshifts in CWL029 and whose proteins were not detected in lysates of CWL029 EB. These findings demonstrate that there is variation in Pmp expression among strains of C. pneumoniae.
Although the relative insensitivity of immunoblot analysis precludes an unequivocal conclusion that PmpA, PmpB/C, and PmpD are not expressed, if present they are minor constituents of the C. pneumoniae outer membrane. The genes encoding these proteins have nevertheless been maintained and the reading frames have been preserved in both CWL029 and AR29. Given our findings of the endogenous protease sensitivity of at least some of the Pmps, one possibility is that these three genes are expressed but processed or degraded by proteolytic digestion, rendering them undetectable by immunoblot analysis. The presence of two immunoreactive high-molecular-weight antigens for PmpG8 and PmpG11 is consistent with the possibility of proteolytic modification. The finding of an endogenous protease leads to the hypothesis that Pmps are subject to posttranslational processing. Of the eight strongly positive full-length Pmps by immunoblot analysis, six are members of the PmpG family and the other two are PmpH and PmpE/F2. It has recently been shown that for C. trachomatis Pmps, only PmpG and PmpH are highly expressed (15).
Variation in the size of Pmp products was found for PmpG6, whose molecular weight differed by nearly 13,000 among strains. Comparison of the pmp gene family for CWL029 and AR39 reveals that only the pmpG6 gene differs significantly among the 21 orthologs. For the CWL029 strain, the pmpG6 product contains three tandem repeats of 131 amino acids whereas in AR39 it contains two repeats. Thus, interstrain variation in Pmp expression may be expected among strains of chlamydiae. Overall, the molecular bases for Pmp variation is consistent with the notion that slip-strand base pairing and posttranscriptional mechanisms of regulation play important roles in Pmp expression.
The function of chlamydial Pmps remains unknown, although based on sequence prediction (9, 17) and experimental testing (10, 14), these are surface proteins and consequently likely to be critical to chlamydial virulence. Variation, both within and between strains, is the major theme of the Pmp protein family, evidenced by diversity in sequence, frameshifts, expression, size, and protease sensitivity. These data suggest that variation is a central requisite for the function of these proteins in the biology of chlamydiae.
ACKNOWLEDGMENTS
This work was supported by National Institutes of Health grants AI40250, AI39258, AI42156, and EY07757. J.G. was supported by a fellowship from the Francis I. Proctor Foundation for Research in Ophthalmology.
We thank C. Fenner for review and for editing the manuscript.
REFERENCES
- 1.Belland R J, Morrison S G, Carlson J H, Hogan D M. Promoter strength influences phase variation of neisserial opa genes. Mol Microbiol. 1997;23:123–135. doi: 10.1046/j.1365-2958.1997.1971556.x. [DOI] [PubMed] [Google Scholar]
- 2.Caldwell H D, Kromhout J, Schachter J. Purification and partial characterization of the major outer membrane protein of Chlamydia trachomatis. Infect Immun. 1981;31:1161–1176. doi: 10.1128/iai.31.3.1161-1176.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Campbell L A, Kuo C C, Grayston J T. Chlamydia pneumoniae and cardiovascular disease. Emerg Infect Dis. 1998;4:571–579. doi: 10.3201/eid0404.980407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Carrington M, Boothroyd J. Implications of conserved structural motifs in disparate trypanosome surface proteins. Mol Biochem Parasitol. 1996;81:119–126. doi: 10.1016/0166-6851(96)02706-5. [DOI] [PubMed] [Google Scholar]
- 5.Grayston J T. Does Chlamydia pneumoniae cause atherosclerosis? Arch Surg. 1999;134:930–934. doi: 10.1001/archsurg.134.9.930. [DOI] [PubMed] [Google Scholar]
- 6.Grayston J T, Aldous M B, Easton A, Wang S P, Kuo C C, Campbell L A, Altman J. Evidence that Chlamydia pneumoniae causes pneumonia and bronchitis. J Infect Dis. 1993;168:1231–1235. doi: 10.1093/infdis/168.5.1231. [DOI] [PubMed] [Google Scholar]
- 7.Grayston J T, Kuo C C, Campbell L A, Wang S P. Chlamydia pneumoniae sp. nov. for Chlamydia strain TWAR. Int J Syst Bacteriol. 1989;39:88. [Google Scholar]
- 8.Grimwood J, Mitchell W, Stephens R S. Phylogenetic analysis of a multigene family conserved between Chlamydia trachomatis and Chlmaydia pneumoniae. In: Stephens R S, Byrne G I, Christiansen G, Clarke I N, Grayston J T, Rank R G, Ridgway G L, Saikku P, Schachter J, Stamm W E, editors. Chlamydial infections. Proceedings of the Ninth International Symposium on Human Chlamydial Infection. San Francisco, Calif: International Chlamydia Symposium; 1998. pp. 263–266. [Google Scholar]
- 9.Kalman S, Mitchell W, Marathe R, Lammel C, Fan J, Hyman R W, Olinger L, Grimwood J, Davis R W, Stephens R S. Comparative genomes of Chlamydia pneumoniae and C. trachomatis. Nat Genet. 1999;21:385–389. doi: 10.1038/7716. [DOI] [PubMed] [Google Scholar]
- 10.Knudsen K, Madsen A S, Mygind P, Christiansen G, Birkelund S. Identification of two novel genes encoding 97- to 99-kilodalton outer membrane proteins of Chlamydia pneumoniae. Infect Immun. 1999;67:375–383. doi: 10.1128/iai.67.1.375-383.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Koehler J E, Burgess R R, Thompson N E, Stephens R S. Chlamydia trachomatis RNA polymerase major ς subunit. Sequence and structural comparison of conserved and unique regions with Escherichia coli ς70 and Bacillus subtilis ς43. J Biol Chem. 1990;265:13206–13214. [PubMed] [Google Scholar]
- 12.Lindholt J S, Fasting H, Henneberg E W, Ostergaard L. A review of Chlamydia pneumoniae and atherosclerosis. Eur J Vasc Endovasc Surg. 1999;17:283–289. doi: 10.1053/ejvs.1998.0757. [DOI] [PubMed] [Google Scholar]
- 13.Lindquist E A, Stephens R S. Transcriptional activity of a sequence variable protein family in Chlamydia trachomatis. In: Stephens R S, Byrne G I, Christiansen G, Clarke I N, Grayston J T, Rank R G, Ridgway G L, Saikku P, Schachter J, Stamm W E, editors. Chlamydial infections. Proceedings of the Ninth International Symposium on Human Chlamydial Infection. San Francisco, Calif: International Chlamydia Symposium; 1998. pp. 259–262. [Google Scholar]
- 14.Longbottom D, Findlay J, Vretou E, Dunbar S M. Immunoelectron microscopic localisation of the OMP90 family on the outer membrane surface of Chlamydia psittaci. FEMS Microbiol Lett. 1998;164:111–117. doi: 10.1111/j.1574-6968.1998.tb13075.x. [DOI] [PubMed] [Google Scholar]
- 15.Mygind P H, Christiansen G, Roepstorff P, Birkelund S. Membrane proteins PmpG and PmpH are major constituents of Chlamydia trachomatis L2 outer membrane complex. FEMS Microbiol Lett. 2000;186:163–169. doi: 10.1111/j.1574-6968.2000.tb09098.x. [DOI] [PubMed] [Google Scholar]
- 16.Read T D, Brunham R C, Shen C, Gill S R, Heidelberg J F, White O, Hickey E K, Peterson J, Utterback T, Berry K, Bass S, Linher K, Weidman J, Khouri H, Craven B, Bowman C, Dodson R, Gwinn M, Nelson W, DeBoy R, Kolonay J, McClarty G, Salzberg S L, Eisen J, Fraser C M. Genome sequences of Chlamydia trachomatis MoPn and Chlamydia pneumoniae AR39. Nucleic Acids Res. 2000;28:1397–1406. doi: 10.1093/nar/28.6.1397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Stephens R S, Kalman S, Lammel C, Fan J, Marathe R, Aravind L, Mitchell W, Olinger L, Tatusov R L, Zhao Q, Koonin E V, Davis R W. Genome sequence of an obligate intracellular pathogen of humans: Chlamydia trachomatis. Science. 1998;282:754–759. doi: 10.1126/science.282.5389.754. [DOI] [PubMed] [Google Scholar]