Abstract
Rationale:
Early life adversity impacts reward-related behaviors, including reward seeking for drugs of abuse. However, the effects of early stress on natural rewards, such as food and social rewards, which has strong implications for symptoms of psychiatric conditions such as major depressive disorder (MDD), are understudied. To fill this gap, we used the limited bedding and nesting (LBN) procedure to assess the impact of early resource scarcity on motivational drive for both food and social rewards in rats.
Methods:
Male and female Long Evans rats were reared in either an LBN environment, with limited nesting materials and no enrichment, from their postnatal day 2–9 or control environment with ample nesting materials and enrichment. As adults, they were tested for reward seeking behavior on progressive ratio operant tasks: food reward (sucrose) or social reward (access to a same-sex/age conspecific).
Results:
We observed sex differences in the impact of LBN on motivation for natural rewards. In males, LBN increased motivation for both a sucrose and social reward. In females, LBN reduced motivation for sucrose but had no effect on social reward.
Conclusions:
These results suggest that the effects of LBN on motivation for natural rewards are both sex- and reinforcer-dependent, with males and females showing differential motivation for food and social rewards following early scarcity. Our previous data revealed an LBN-driven reduction in motivation for morphine in males and no effect in females, highlighting the reinforcer-dependent impact of early resource scarcity on motivated behavior more widely.
Keywords: early life stress, social motivation, natural reward, food reward, LBN, sex difference
Introduction
Early resource scarcity is associated with increased risk for developing disorders characterized by altered motivated behavior, including substance use disorder (SUD) and major depressive disorder (MDD) (al’Absi et al. 2021; Baskin-Sommers and Foti 2015; LeMoult et al. 2020; Reiss et al. 2019). Low childhood socioeconomic status (SES) increases rates of adult SUD and MDD, even when controlling for current socioeconomic conditions (Angelini et al. 2019; Lee et al. 2015). Resource scarcity impacts mental health, in part, by inducing stress (Evans 2004). Although stress is often conceptualized as “bad”, the body’s stress response is designed to adapt to changes in the environment to promote survival. In some circumstances, stress effects on motivated behavior may be adaptive while being maladaptive in others. In this regard, a stress-induced reduction in motivation that causes withdrawal may initially promote avoidance of dangerous situations, however, when unchecked, this amotivation can contribute to aspects of MDD, particularly avolition (Szczypiński and Gola 2018; Treadway and Zald 2013).
A widely used rodent manipulation for identifying mechanisms by which early life adversity affects offspring is the limited bedding and nesting (LBN) procedure. In LBN, dams experience resource scarcity in the form of reduced access to bedding and nesting materials, typically for a week during the pups’ early postnatal period. This manipulation mimics aspects of scarcity in humans and has been consistently shown to alter maternal behavior, often resulting in fragmented care and hypervigilance in dams (Eck et al. 2019; Gallo et al. 2019; Ivy et al. 2008; Shupe and Clinton 2021; Walker et al. 2017; Wendel et al. 2021). The long-term effects of LBN in offspring include altered reward circuitry and alterations in motivated behavior (Bolton et al. 2018a; Bolton et al. 2018b; Eck et al. 2022; Levis et al. 2019; Ordoñes Sanchez et al. 2021).
Our laboratory employs an LBN manipulation where rats are bred in-house, and dams and offspring are put in a low resource environment during postnatal day (PND) 2–9. Using this procedure, we have found that adult male, but not female, rats have reduced motivation for a low dose of morphine compared to control reared males (Ordoñes Sanchez et al. 2021). These behavioral changes were linked to reduced glutamate transmission and sex-specific transcriptional alterations in the nucleus accumbens (NAc), a key player in reward circuitry (Ordoñes Sanchez et al. 2021). In contrast to reducing motivation for drug reward, we found some evidence that LBN enhances motivation for social reward in males. LBN males acquire reproductive behaviors more rapidly than control males: over the course of three sessions with a primed female, LBN males are quicker to mount, intromit, and ejaculate in early sessions than controls (Eck et al. 2022). Reproduction is driven by motivated and consummatory behaviors. Latency to mount and intromit is thought to reflect motivation (Agmo 1997), so these data suggest that LBN may increase social motivation in males.
In addition to male reproductive behavior, LBN affects other behaviors that have both motivational and hedonic components. For example, other implementations of the LBN model find that LBN reduces consumption of palatable foods and juvenile play behavior in males (Bolton et al. 2018a; Bolton et al. 2018b; Molet et al. 2016). However, procedures that disentangle motivation from pleasure are rarely employed, and no published studies to date have specifically investigated social motivation in LBN rats, or any other model of prenatal adversity. Here, we address this gap by assessing the effect of LBN on sucrose and social self-administration and motivation (measured by progressive ratio). Notably, we perform sucrose self-administration in satiated rats rather than food restricted rats to disentangle motivation driven by caloric need from motivation for a natural reward. Using a recently developed social self-administration procedure (Venniro and Shaham 2020; Venniro et al. 2018), we also tested whether LBN altered self-administration and motivation for access to a sex- and age-matched partner (defined below). Crucially, both sexes were run in parallel and compared to reveal whether there were any sex differences in the effects of LBN on motivation for natural rewards.
Methods
Subjects and limited bedding and nesting (LBN)
Temple University institutional Animal Care and Use Committee approved all animal care and experiments in this study. Long-Evans rats were obtained from Charles River Laboratories (Wilmington, MA) and bred in-house, and their offspring were used for studies. All LBN (n=11) and control (n=14) dams were primiparous. For all experiments, no more than two males and two females (sex defined by external genitalia) were used from each litter and pups were randomly assigned to experiments. At PND2, litters were culled to 10 pups with an even representation of the sexes when possible and litters were put into control or LBN housing. Control housing consisted of our standard housing conditions: 2 cm cob bedding, 2 cotton Nestlets, and one plastic tunnel enrichment. LBN housing consisted of cages fitted with a custom stainless-steel metal platform that prevented access to bedding, one paper towel for nesting material, and no enrichment (Eck et al. 2019). On PND10, all litters were transferred into fresh control housing conditions. Rats were weaned at PND21 and pair-housed with rats of the same sex and condition (i.e., LBN vs. control housing). As adults (PND 60–180), rats were handled daily for at least 5 days prior to use in any experiment. All rats had ad libitum access to water and standard laboratory chow throughout the duration of the experiment. All housing occurred within standard shoebox cages (20×20×40 cm) with vent filter cage tops (on non-ventilated racks). No rats were dropped from these experiments. A different cohort of rats was used for sucrose self-administration and social self-administration.
Sucrose self-administration
Male and female rats performed an oral sucrose self-administration using MedPC Operant Chambers (St. Albans, VT). Rats were housed in a reverse dark/light cycle with lights on from 9:00 P.M. to 9:00 A.M. Behavioral studies occurred during the dark phase. Self-administration experiments took place from 9:00 A.M. to 11:00 A.M. (2-h self-administration sessions in the dark cycle). Rats were first run on an fixed ration (FR)1 schedule where one active lever press resulted in one sucrose pellet reward paired with a 5-s cue light; this lasted 10 days. During this training phase rats were able to receive a maximum of 90 sucrose pellets per session. Following 10 days of FR1, rats were run on one day of sucrose self-administration on a progressive ratio (PR) schedule. Under a PR schedule, the response requirement for each subsequent sucrose pellet delivery increases exponentially until the subject fails to meet the requirement. The response requirement of the ith reinforcement was given by R(i)=[5e0.2i-5] and the session expired when an animal took more than 60 minutes to earn a sucrose pellet (Bari and Pierce 2005; Hopkins et al. 2012; Mietlicki-Baase et al. 2013).
Social self-administration
Rats were trained to self-administer for access to an initially novel same-sex conspecific that was raised under control housing conditions (“target” rat) during daily 120-min sessions on a discrete trial design procedure (60 trials/session, 60 s) in social motivation chambers (Med Associates; ENV-008CT) (Venniro and Shaham 2020). Each rat lever pressed for access to the same target rat over the course of the experiment. Trials began with the illumination of a white house light and 10s later a social-paired active lever released. Rats were given 60s to press the active lever on a FR1 reinforcement schedule before it automatically retracted, and the house light turned off. Pressing the active lever resulted in retraction of the lever, a 20s tone cue, and the lifting of a guillotine-style door. After the door lifted, the resident rat was able to interact with the target rat for 40s. A barrier was present keeping the rats from going into each other’s chambers, but they were able to sniff and touch. After this period the house light turned off, the guillotine door closed, and a new trial shortly began. After 6 days of training, rats were run through this task on a progressive ratio schedule. During the progressive ratio sessions, we increased the ratio of responses per rewards or infusions as detailed (Depoortere et al. 1993). The final completed response ratio represents the ‘breakpoint’ value.
Statistical Analysis
Sample sizes were determined based on prior literature (Venniro et al. 2019; Venniro and Shaham 2020; Venniro et al. 2018). All statistical analyses were performed using R statistical software. Normality of data was assessed using Shapiro-tests. A Fligner-Killeen test was used to assess homogeneity of variance. All significance thresholds were set at p<.05.
Experiment 1: Sucrose self-administration
A repeated measures three-way ANOVA was used to analyze FR1 data, while a two-way ANOVA (sex*stress) was used for PR data. For ANOVA analyses that revealed significant effects, pairwise comparisons with Bonferroni correction were used to detect differences between groups.
Experiment 2: Social self-administration
A repeated measures three-way ANOVA was used to analyze FR1 data, and a two-way ANOVA (sex*stress) was used to analyze PR data. For ANOVA analyses that revealed significant effects, pairwise comparisons with Bonferroni correction were used to detect differences between groups. Unpaired t-tests were used to compare latency to receive a reward.
Results
Effects of early life stress on food motivation
To test the effects of early life stress on motivation for palatable food, we compared LBN vs. control males and females in an operant task in which they must lever press for a sucrose pellet (Fig. 1A). Rats were first trained on a fixed ratio 1 (FR1) schedule for 10 days, where 1 active lever press resulted in 1 sucrose pellet. After 10 days of training, rats were run through 1 session on a progressive ratio (PR) schedule, where the number of lever presses necessary to receive the food reward increased over each trial (Fig. 1A). Using a three-way repeated measures ANOVA, we did not identify a significant day*stress*sex interaction effect (F9,504=1.319, p>0.05), but we did identify significant day*stress (F9,504=2.259, p<0.05; Fig. 1B) and sex*stress (F1,56=6.251, p<0.05; Fig. 1B) interaction effects on rewards received during FR1 training. Pairwise comparisons revealed a significant effect of stress on days 3 and 5, where LBN females received fewer rewards than both control and LBN males on these days FR1 (p<0.05). However, these differences dissipated by day 6 and LBN females earned an equal number of rewards as all groups throughout the rest of training (Fig. 1B). Similar effects were observed on active lever presses. Although there was not a significant day*stress*sex interaction effect (F9,504=1.716, p>0.05; Fig. 1C), significant day*stress (F9,504=4.338, p<0.001; Fig. 1C) and day*sex (F9,504=2.127, p<0.05; Fig. 1C) interaction effects were observed. Pairwise comparisons revealed that LBN females had fewer active lever presses than LBN males on day 2 (p<0.05) and control females on day 3 (p<0.01). By day 4, LBN females show the same rate of active lever presses as all groups.
Figure 1. Effects of LBN on sucrose self-administration.
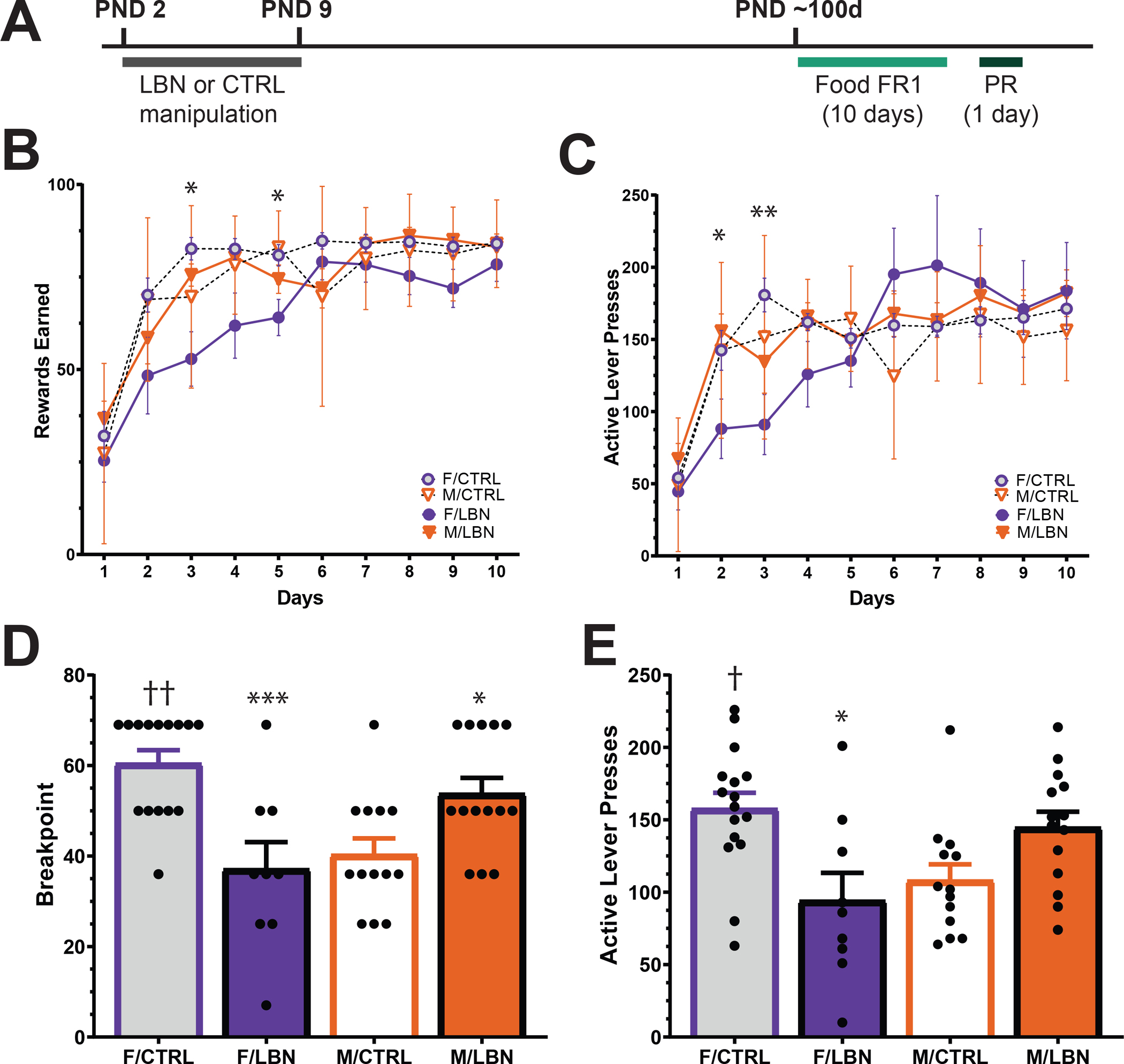
Timeline of experiment: rats undergo LBN during PND 2–9. In adulthood (e.g., PND 60), they begin 10 days of training on an FR1 schedule. Following 10 days of training, they are run through 1 day of PR (A). Number of rewards earned on an FR1 schedule over 10 days (B). Number of active lever presses on an FR1 schedule over 10 days (C). Breakpoint ratio on a PR schedule (D). Number of active lever presses on a PR schedule (E). N=9–15/group. *p<.05, **p<.01, ***p<.001 compared to same-sex control, †=p<0.05, ††=p<0.01 compared to male control. Data are represented as means ± SEMs. CTRL, control housing; F, female; FR1, fixed ratio 1; LBN, limited bedding and nesting housing; M, male; PR, progressive ratio
When rats were run on a PR schedule, we found a significant sex*stress interaction effect (F1,48=18.38, p<0.001; Fig. 1D). Pairwise comparisons revealed baseline sex differences in controls, where control females had a higher breakpoint than control males (p<0.01). We also identified sex-specific effects of LBN, where LBN reduced breakpoint in females (p<0.001) and increased breakpoint in males (p<0.05). Correspondingly, male LBN rats had a higher breakpoint compared to LBN females (p<0.01). When assessing active lever presses during PR, we found a significant effect of stress on number of presses (F1,48=6.576, p<0.05; Fig. 1E). Pairwise comparisons revealed that control females pressed the active lever significantly more than both LBN females (p<0.05) and control males (p<0.05).
Effects of early life stress on social motivation
To test the effects of early life stress on motivation for social rewards, we compared LBN males and females to controls in an operant task in which animals responded for access to a novel same-sex conspecific (Fig. 2A). Control and LBN rats were first trained on a trial-based FR1 schedule for 6 days, where 1 lever press granted them access to a 60-second interaction. After 6 days, rats were run through 1 day of PR, where the number of lever presses necessary to receive the reward increased over each trial (Fig. 2A).
Fig. 2. Effects of LBN on social self-administration.
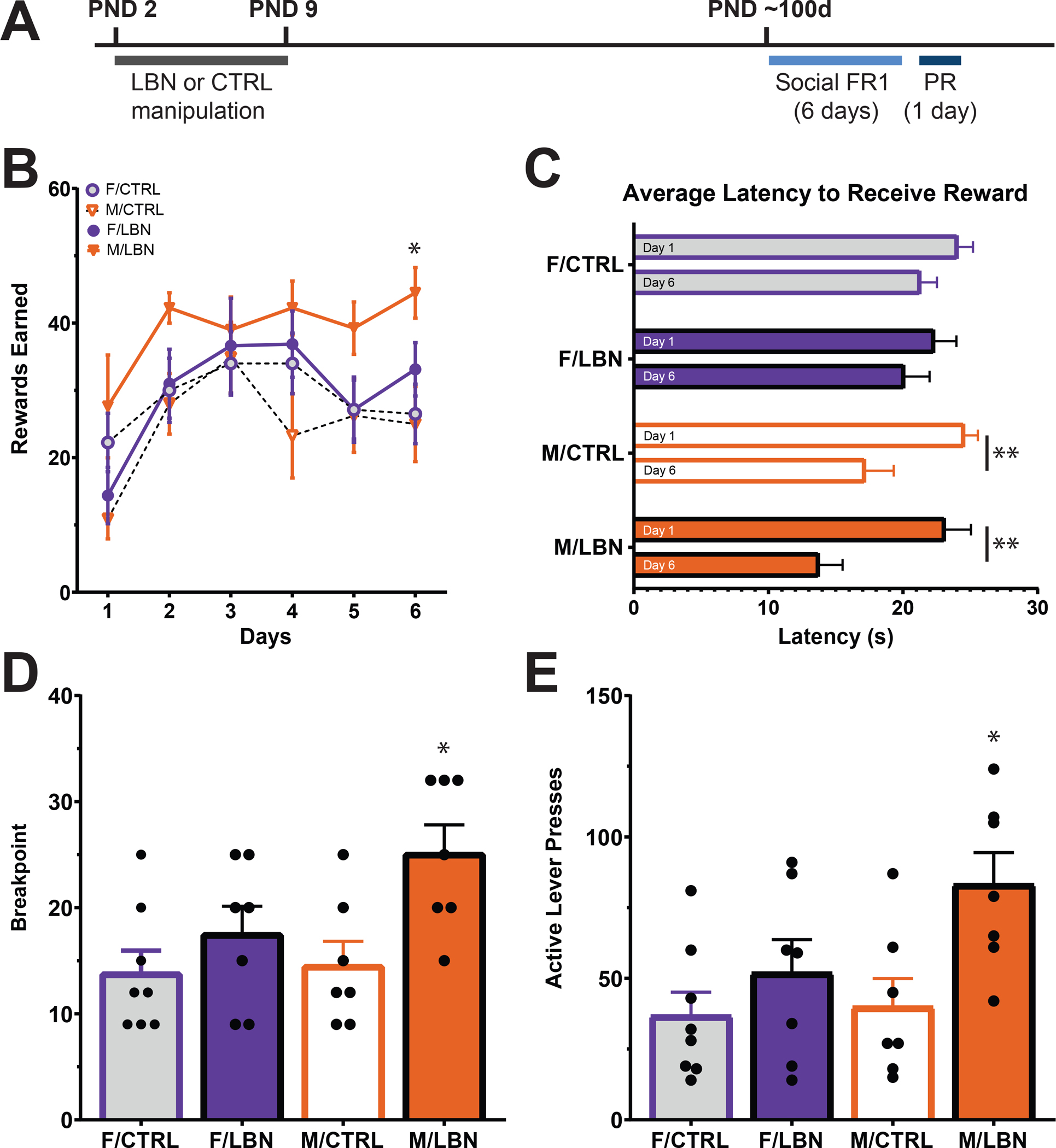
Timeline of experiment: rats undergo LBN during PND 2–9. In adulthood (e.g., PND 60), they begin 6 days of training on a trial based FR1 schedule. Following 6 days of training, they are run through 1 day of PR (A). Rewards earned on trial based FR1 schedule (B). Average latency to receive a reward on day 1 vs. day 6 while on a trial based FR1 schedule (C). Breakpoint ratio on a PR schedule (D). Number of active lever presses on a PR schedule (E). N=7–8/group. *p<.05, **p<.01. Data are represented as means ± SEMs. CTRL, control housing; F, female; FR1, fixed ratio 1; LBN, limited bedding and nesting housing; M, male; PR, progressive ratio
Although there was no significant day*stress*sex interaction effect (F5,140=1.012, p>0.05), a repeated measures three-way ANOVA revealed a significant effect of day (F4,94 = 11.20, p<0.001) and stress (F1,28=4.724, p<0.05) on rewards received during FR1 training (Fig. 2B). Pairwise comparisons identified that on day 6 LBN males lever press significantly more than control males and females (Fig. 2B; p<0.05). Unpaired t-tests revealed that in both LBN (t=3.61, p<0.01) and control males (t=3.15, p<0.01) latency to receive a social reward decreases by day 6 (Fig. 2C); this same effect is not present in females. On a PR schedule, we identified a significant sex*stress interaction effect on breakpoint (two-way ANOVA, F1,26=5.38, p<0.05; Fig. 2D). Pairwise comparisons showed that LBN increases breakpoint in males (p<0.01) but not females. LBN males also have a higher breakpoint than both LBN (p<0.05) and control females (p<0.01). LBN also increased active lever presses in males (two-way ANOVA significant effect of stress: F3,18=4.553, p<0.05; Fig. 2E). Pairwise comparisons revealed this effect to be significant compared to control males and females (both p’s<0.05). Interesting, unlike when testing for responses to palatable food, no baseline sex differences were observed in motivation for access to a social reward (p>0.05).
Discussion
Early resource scarcity can affect reward circuitry and motivated behavior, although many of the aforementioned studies in this space have focused on drugs of abuse. In instances where natural rewards were studied, the paradigms leveraged did not allow for the teasing apart of motivational drive from hedonic processes (e.g., palatable food consumption, male reproductive behavior, social play). Here we address this gap by using operant self-administration procedures to assess the effect of LBN on two distinct natural reinforcers (palatable food and social reward) in both male and female rats. In males, we found that LBN increased motivation for both sucrose and social reinforcers, indictive of an overall enhancement in natural reward processing. This result is consistent with our prior work suggesting an enhanced motivation in LBN male rats for hormonally primed females in the male reproductive behavior assay, but contrasts with previous findings where LBN males were less motivated for morphine and heroin (Eck et al. 2022; Levis et al. 2022; Ordoñes Sanchez et al. 2021). Taken together these data suggest that, in males, the impact of LBN on motivation is reinforcer specific: while motivation for natural rewards is enhanced, motivation for opioids is reduced. In females, LBN reduced motivation for sucrose, which could be indicative of avolition. However, LBN did not alter social motivation in females in this study, nor did it impact morphine motivation in our prior work using two different doses of morphine (Ordoñes Sanchez et al. 2021). Together, these findings highlight that brief early life scarcity has both sex- and reinforcer-specific effects on motivated behavior. These results highlight the importance of female subjects being considered and directly compared to males in these types of studies to promote a more complete understanding how motivated behavior becomes disrupted following early life stress.
Differential effects of LBN on motivated behavior
LBN consistently alters performance on tasks that measure hedonic and motivational aspects of natural reward processing. However, the direction of effects, while consistent within laboratories, can vary even when the duration rats are left in the model is similar (i.e., from PND2–9) (Walker et al. 2017). Work from the Baram laboratory demonstrated that in male rats LBN reduces sucrose preference and the consumption of M&Ms, but in female rats increases consumption of M&Ms and motivation for a palatable food — as evidenced by decreased demand elasticity in a behavioral economics task (Bolton et al. 2018a; Bolton et al. 2018b; Levis et al. 2019; Molet et al. 2016). In contrast, here we find that LBN increases motivation for sucrose in male rats while decreasing it in female rats. Social endpoints are also different. In males, other laboratories find that LBN reduces adolescent peer play in adult rats and reduces social approach in infant and adolescent rats as well as adult C57BL/6J mice (Molet et al. 2016; Rincón-Cortés and Sullivan 2016; Wendel et al. 2021) although, in some cases, no effect of LBN on sociability and preference are observed (Shupe and Clinton 2021). In our laboratory, we have observed that LBN increases motivation for a sexually receptive partner and increases social motivation for a same-sex counterpart in males (Eck et al. 2022). One possible explanation for these discrepancies is differences in the tasks used to evaluate changes in behavior. For example, sucrose preference, M&M consumption, and play behavior are all often characterized as measures of anhedonia instead of avolition. Discrepant findings could also be due to differences in species and strains, particularly given known differences in sociability between strains (Ku et al. 2016). For example, Sprague Dawley rats engage in more juvenile play than Long Evans rats (Ku et al. 2016). Moreover, C57BL/6J mice, unlike rats or CD1 mice, have weak social self-administration and strongly prefer food over social interaction (Ramsey et al. 2022), making it difficult to generalize stress-induced changes in sociability in C57BL/6J mice.
Another important difference between studies is the implementation of the LBN model and control conditions. To reduce bedding, many laboratories use either low bedding or a wire mesh (Ivy et al. 2008; Shupe and Clinton 2021; Walker et al. 2017). However, our laboratory uses a metal grate which increases contact with metal, inducing a different tactile experience compared to other implementations (Eck et al. 2019). Moreover, many laboratories time ship pregnant rat dams, which can be considered an additional stressor (Butler-Struben et al. 2022; Sachs and Lumia 1981), while our lab breeds in house, minimizing prenatal stress. These seemingly minor changes can alter maternal care, which in turn can have long-lasting effects on offspring. For example, when rats are bred in house there is a shift in behavior such that dams spend more time engaged in pup-directed behaviors, such as nursing and licking and grooming the pups (Eck et al. 2019; Shupe and Clinton 2021). This shift comes at the expense of self-care behaviors, such as resting outside the nest and self-grooming. In conditions where rat dams are shipped pregnant, there is evidence of abusive behaviors, such as kicking pups (Gallo et al. 2019; Walker et al. 2017), which we do not observe in our model. However, it is notable that there are different scoring procedures, and our laboratory has not done continuous scoring. In addition to differences in implementation of LBN, control conditions vary between laboratories. Compared to others (Molet et al. 2016; Raineki et al. 2010; Walker et al. 2017), our control conditions contain more environmental enrichment, such as a plastic enrichment tunnel. Notably, after the brief period of resource scarcity, LBN dams and pups are put back in control conditions. Exposure to a highly enriched environment can mitigate some of the effects of LBN (Strzelewicz et al. 2019), so it is possible our laboratories’ slightly enriched environment has some protective effects on offspring.
Circuitry of natural rewards and sociability
Research focused on the impact of stressors on reward processing has historically centered around drugs of abuse or reward-related alterations linked to psychiatric illnesses such as MDD (al’Absi et al. 2021; Baskin-Sommers and Foti 2015; LeMoult et al. 2020; Nestler and Carlezon 2006; Reiss et al. 2019). The majority of this work supports the idea that circuit connectivity within the NAc, such as projections from the ventral tegmental area (VTA), prefrontal cortex, hippocampus, and amygdalar structures are important for reward learning, anticipation, reward-seeking, reinforcement, contextualization, and other goal-directed behaviors (Cooper et al. 2017; Russo and Nestler 2013). Work within this field has consistently shown a cyclical relationship where exposure to drugs of abuse alters activity within this mesolimbic circuit, and that in turn, functional manipulations to this circuit alters motivational drive and reward-related behaviors for drugs of abuse. Research assessing the importance of these regions for natural rewards, as well as how environmental factors such as stress alter their activity, is somewhat limited, albeit some work has been done to begin identifying neural circuits relevant for natural rewards and prosocial behaviors (Nall et al. 2021; Walsh et al. 2022). Given the complexities of these behaviors it is unsurprising that multiple brain regions, circuits, neurotransmitters, and neuromodulators are involved in natural reward processing.
We found that LBN causes reinforcer-specific effects, which might be unexpected given that there are many similar regions and mechanisms involved in mediating drug, food, and social reward. However, our findings indicate that LBN does not simply suppress or enhance reward circuity but has more subtle effects that fine tune the reinforcing properties of distinct rewards. Moreover, these LBN-induced changes are affected by biological sex. We previously found that LBN induces alterations in NAc physiology and gene transcription that differ across sex (Ordoñes Sanchez et al. 2021). In males, LBN reduces NAc presynaptic glutamate transmission and blocks morphine-induced change glutamate plasticity (i.e., AMPA/NMDA ratio) (Ordoñes Sanchez et al. 2021); effects that may reduce male susceptibility to opioid seeking following early scarcity. Additionally in males, LBN increases the expression of genes in the oxytocin signaling pathway in the NAc, which may promote social behavior. These changes in glutamate and oxytocin pathways were not observed in females following LBN (Ordoñes Sanchez et al. 2021). However, LBN causes alterations in genes in the muscarinic acetylcholine receptor signaling pathway in the female NAc (Ordoñes Sanchez et al. 2021), and within the NAc muscarinic receptors can regulate food motivation (Pratt and Kelley 2004). Although the NAc is a hub in reward circuitry, other regions also are key to regulating reward processing. Future studies should determine whether LBN causes sex-specific molecular and physiological changes in other reward regions, as well as explore the possibility that following LBN, males and females have a differential recruitment subcircuits within the reward pathway.
Conclusions
Many psychiatric disorders are characterized by changes in motivation, but simply increasing motivation to treat anhedonia, as an example, may also increase risk for SUD. An ideal treatment would address the disrupted motivation specifically, without broadly changing reward processing. To develop such a treatment, animal models that identify differences in motivation for distinct reinforcers are needed. The findings from this paper combined with our prior work suggest that we have developed such a model. In males, LBN increases motivation for natural rewards, while it reduces motivation for opioids. In females, LBN selectively reduces motivation for food, indicated of an avolition-like phenotype. Now, we and others can leverage these behavior changes to investigate mechanisms that drive motivation for specific reinforcers. This approach may lead to the development of targeted therapeutics that improve motivated behavior and outcomes for people with SUD and MDD.
Acknowledgements:
This work is supported by National Institute of Health grants: DA049837 to D.A.B., DA056534 to D.A.B. and M.E.W., DA046537 to M.E.W, and T32 DA007237 to A.V.W (PI Unterwald), and DA047976 to M.V. This is also funded by a BBRF Young Investigator Grant 28897 to M.V. This work was also supported by a National Science Foundation grant IOS1929829 to D.A.B. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Footnotes
Conflict of Interest: The authors declare that they have no conflict of interest.
References
- Agmo A (1997) Male rat sexual behavior. Brain Res Brain Res Protoc 1: 203–9. [DOI] [PubMed] [Google Scholar]
- al’Absi M, Ginty AT, Lovallo WR (2021) Neurobiological mechanisms of early life adversity, blunted stress reactivity and risk for addiction. Neuropharmacology 188: 108519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Angelini V, Howdon DDH, Mierau JO (2019) Childhood Socioeconomic Status and Late-Adulthood Mental Health: Results From the Survey on Health, Ageing and Retirement in Europe. The Journals of Gerontology: Series B 74: 95–104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bari AA, Pierce RC (2005) D1-like and D2 dopamine receptor antagonists administered into the shell subregion of the rat nucleus accumbens decrease cocaine, but not food, reinforcement. Neuroscience 135: 959–68. [DOI] [PubMed] [Google Scholar]
- Baskin-Sommers AR, Foti D (2015) Abnormal reward functioning across substance use disorders and major depressive disorder: Considering reward as a transdiagnostic mechanism. International Journal of Psychophysiology 98: 227–239. [DOI] [PubMed] [Google Scholar]
- Bolton JL, Molet J, Regev L, Chen Y, Rismanchi N, Haddad E, Yang DZ, Obenaus A, Baram TZ (2018a) Anhedonia Following Early-Life Adversity Involves Aberrant Interaction of Reward and Anxiety Circuits and Is Reversed by Partial Silencing of Amygdala Corticotropin-Releasing Hormone Gene. Biol Psychiatry 83: 137–147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bolton JL, Ruiz CM, Rismanchi N, Sanchez GA, Castillo E, Huang J, Cross C, Baram TZ, Mahler SV (2018b) Early-life adversity facilitates acquisition of cocaine self-administration and induces persistent anhedonia. Neurobiology of Stress 8: 57–67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Butler-Struben HM, Kentner AC, Trainor BC (2022) What’s wrong with my experiment?: The impact of hidden variables on neuropsychopharmacology research. Neuropsychopharmacology 47: 1285–1291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cooper S, Robison AJ, Mazei-Robison MS (2017) Reward Circuitry in Addiction. Neurotherapeutics 14: 687–697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Depoortere RY, Li DH, Lane JD, Emmett-Oglesby MW (1993) Parameters of self-administration of cocaine in rats under a progressive-ratio schedule. Pharmacol Biochem Behav 45: 539–48. [DOI] [PubMed] [Google Scholar]
- Eck SR, Ardekani CS, Salvatore M, Luz S, Kim ED, Rogers CM, Hall A, Lee DE, Famularo ST, Bhatnagar S, Bangasser DA (2019) The effects of early life adversity on growth, maturation, and steroid hormones in male and female rats. Eur J Neurosci. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eck SR, Palmer JL, Bavley CC, Karbalaei R, Ordoñes Sanchez E, Flowers J, Holley A, Wimmer ME, Bangasser DA (2022) Effects of early life adversity on male reproductive behavior and the medial preoptic area transcriptome. Neuropsychopharmacology. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Evans GW (2004) The environment of childhood poverty. American psychologist 59: 77. [DOI] [PubMed] [Google Scholar]
- Gallo M, Shleifer DG, Godoy LD, Ofray D, Olaniyan A, Campbell T, Bath KG (2019) Limited Bedding and Nesting Induces Maternal Behavior Resembling Both Hypervigilance and Abuse. Frontiers in Behavioral Neuroscience 13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hopkins TJ, Rupprecht LE, Hayes MR, Blendy JA, Schmidt HD (2012) Galantamine, an acetylcholinesterase inhibitor and positive allosteric modulator of nicotinic acetylcholine receptors, attenuates nicotine taking and seeking in rats. Neuropsychopharmacology 37: 2310–21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ivy AS, Brunson KL, Sandman C, Baram TZ (2008) Dysfunctional nurturing behavior in rat dams with limited access to nesting material: A clinically relevant model for early-life stress. Neuroscience 154: 1132–1142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ku KM, Weir RK, Silverman JL, Berman RF, Bauman MD (2016) Behavioral Phenotyping of Juvenile Long-Evans and Sprague-Dawley Rats: Implications for Preclinical Models of Autism Spectrum Disorders. PLOS ONE 11: e0158150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee JO, Hill KG, Hartigan LA, Boden JM, Guttmannova K, Kosterman R, Bailey JA, Catalano RF (2015) Unemployment and substance use problems among young adults: Does childhood low socioeconomic status exacerbate the effect? Soc Sci Med 143: 36–44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LeMoult J, Humphreys KL, Tracy A, Hoffmeister J-A, Ip E, Gotlib IH (2020) Meta-analysis: Exposure to Early Life Stress and Risk for Depression in Childhood and Adolescence. Journal of the American Academy of Child & Adolescent Psychiatry 59: 842–855. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levis SC, Bentzley BS, Molet J, Bolton JL, Perrone CR, Baram TZ, Mahler SV (2019) On the early life origins of vulnerability to opioid addiction. Molecular Psychiatry. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levis SC, Birnie MT, Bolton JL, Perrone CR, Montesinos JS, Baram TZ, Mahler SV (2022) Enduring disruption of reward and stress circuit activities by early-life adversity in male rats. Translational Psychiatry 12: 251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mietlicki-Baase EG, Rupprecht LE, Olivos DR, Zimmer DJ, Alter MD, Pierce RC, Schmidt HD, Hayes MR (2013) Amylin receptor signaling in the ventral tegmental area is physiologically relevant for the control of food intake. Neuropsychopharmacology 38: 1685–97. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Molet J, Heins K, Zhuo X, Mei YT, Regev L, Baram TZ, Stern H (2016) Fragmentation and high entropy of neonatal experience predict adolescent emotional outcome. Translational Psychiatry 6: e702–e702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nall RW, Heinsbroek JA, Nentwig TB, Kalivas PW, Bobadilla A-C (2021) Circuit selectivity in drug versus natural reward seeking behaviors. Journal of Neurochemistry 157: 1450–1472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nestler EJ, Carlezon WA Jr. (2006) The mesolimbic dopamine reward circuit in depression. Biol Psychiatry 59: 1151–9. [DOI] [PubMed] [Google Scholar]
- Ordoñes Sanchez E, Bavley CC, Deutschmann AU, Carpenter R, Peterson DR, Karbalaei R, Flowers J, Rogers CM, Langrehr MG, Ardekani CS, Famularo ST, Bongiovanni AR, Knouse MC, Floresco SB, Briand LA, Wimmer ME, Bangasser DA (2021) Early life adversity promotes resilience to opioid addiction-related phenotypes in male rats and sex-specific transcriptional changes. Proceedings of the National Academy of Sciences 118: e2020173118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pratt WE, Kelley AE (2004) Nucleus accumbens acetylcholine regulates appetitive learning and motivation for food via activation of muscarinic receptors. Behavioral neuroscience 118: 730. [DOI] [PubMed] [Google Scholar]
- Raineki C, Moriceau S, Sullivan RM (2010) Developing a Neurobehavioral Animal Model of Infant Attachment to an Abusive Caregiver. Biological Psychiatry 67: 1137–1145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ramsey LA, Holloman FM, Hope BT, Shaham Y, Venniro M (2022) Waving Through the Window: A Model of Volitional Social Interaction in Female Mice. Biol Psychiatry 91: 988–997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reiss F, Meyrose A-K, Otto C, Lampert T, Klasen F, Ravens-Sieberer U (2019) Socioeconomic status, stressful life situations and mental health problems in children and adolescents: Results of the German BELLA cohort-study. PLOS ONE 14: e0213700. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rincón-Cortés M, Sullivan RM (2016) Emergence of social behavior deficit, blunted corticolimbic activity and adult depression-like behavior in a rodent model of maternal maltreatment. Transl Psychiatry 6: e930. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Russo SJ, Nestler EJ (2013) The brain reward circuitry in mood disorders. Nature Reviews Neuroscience 14: 609–625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sachs BD, Lumia AR (1981) Is stress due to shipment of animals a confounding variable in developmental research? Developmental Psychobiology 14: 169–171. [DOI] [PubMed] [Google Scholar]
- Shupe EA, Clinton SM (2021) Neonatal resource scarcity alters maternal care and impacts offspring core temperature and growth in rats. Developmental Psychobiology 63: e22144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Strzelewicz A, Sanchez E, Rondón-Ortiz A, Raneri A, Famularo S, Bangasser D, Kentner A (2019) Access to a high resource environment protects against accelerated maturation following early life stress: A translational animal model of high, medium and low security settings. Hormones and behavior. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Szczypiński JJ, Gola M (2018) Dopamine dysregulation hypothesis: the common basis for motivational anhedonia in major depressive disorder and schizophrenia? Reviews in the Neurosciences 29: 727–744. [DOI] [PubMed] [Google Scholar]
- Treadway MT, Zald DH (2013) Parsing Anhedonia: Translational Models of Reward-Processing Deficits in Psychopathology. Current Directions in Psychological Science 22: 244–249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Venniro M, Russell TI, Zhang M, Shaham Y (2019) Operant Social Reward Decreases Incubation of Heroin Craving in Male and Female Rats. Biol Psychiatry 86: 848–856. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Venniro M, Shaham Y (2020) An operant social self-administration and choice model in rats. Nature Protocols 15: 1542–1559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Venniro M, Zhang M, Caprioli D, Hoots JK, Golden SA, Heins C, Morales M, Epstein DH, Shaham Y (2018) Volitional social interaction prevents drug addiction in rat models. Nature Neuroscience 21: 1520–1529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Walker C-D, Bath KG, Joels M, Korosi A, Larauche M, Lucassen PJ, Morris MJ, Raineki C, Roth TL, Sullivan RM, Taché Y, Baram TZ (2017) Chronic early life stress induced by limited bedding and nesting (LBN) material in rodents: critical considerations of methodology, outcomes and translational potential. Stress 20: 421–448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Walsh JJ, Christoffel DJ, Malenka RC (2022) Neural circuits regulating prosocial behaviors. Neuropsychopharmacology. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wendel KM, Short AK, Noarbe BP, Haddad E, Palma AM, Yassa MA, Baram TZ, Obenaus A (2021) Early life adversity in male mice sculpts reward circuits. Neurobiology of Stress 15: 100409. [DOI] [PMC free article] [PubMed] [Google Scholar]


