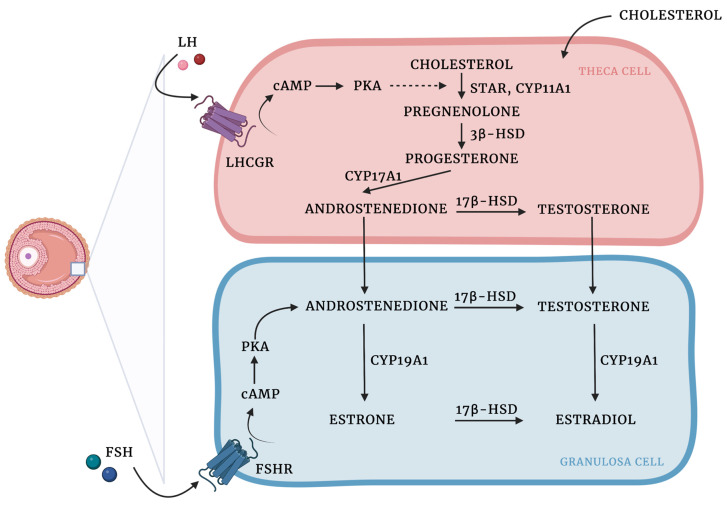
Figure 1.
Ovarian steroidogenesis: two cell, two-gonadotropin theory. Ovarian steroids are synthesized from cholesterol, which diffuses from the circulation into theca cells and is mobilized into mitochondria by steroidogenic acute regulatory protein (STAR) activity [102]. LH binds to LHCGR on the cell surface, which results in the increased expression of steroidogenic enzymes involved in androgen production. Cholesterol is then converted into pregnenolone by the cholesterol sidechain cleavage enzyme (CYP11A1). In the smooth endoplasmic reticulum, pregnenolone is transformed into progesterone due to the activity of 3β-hydroxysteroid dehydrogenase (3β-HSD). Then, due to the activity of CYP17A1 progesterone is converted to androstenedione, which in turn might be transformed into testosterone by 17β-hydroxysteroid dehydrogenase (17β-HSD) or translocated into the GCs, where aromatase (CYP450arom; CYP19A1) converts androstenedione to estrone and testosterone to estradiol. 17β-HSD might also produce estradiol using estrone as a substrate [103,104,105,106]. Created with BioRender.com.