Abstract
This study investigated the impact of residential radon exposure on human cancers (i.e., lung cancer and childhood leukemia) through a systematic review and meta-analysis of case–control studies. A total of 9724 articles obtained from electronic databases were assessed; however, only 55 case–control studies were eligible after manually screening and eliminating unnecessary studies. The causal associations were addressed by determining the meta-analysis’s estimated size effects (i.e., ORs/RRs) of the meta-analysis. Residential radon was revealed to significantly increase the incidence of lung cancer and childhood leukemia with pooled ORs of 1.38 [1.19; 1.60] (I2 = 90%; p < 0.00001) and 1.43 [1.19; 1.72] (I2 = 0% and p = 0.51), respectively. In addition, subgroup analyses were performed to reduce the heterogeneity of the initial meta-analyses. The results provided strong evidence that inhaling radon in the indoor environments is closely associated with the development of lung cancer and childhood leukemia in patients living in Europe and areas with high radon levels (≥100 Bq/m3).
Keywords: radon risk, lung cancer, childhood leukemia, case–control studies
1. Introduction
Radon is well known as a naturally occurring radioactive gas that may be found in high concentrations in indoor environments such as homes, schools, and workplaces. The primary sources of residential radon are soils and rocks around the foundation, fuels, building materials, and domestic water [1]. The indoor radon concentration can vary greatly from 10 Bq/m3 to more than 10,000 Bq/m3 (from 0.27 pCi/L to 270 pCi/L) [1]. Indoor radon escapes from the ground into the air, where it decays and produces other radioactive particles [1,2]. When we breathe, these particles are deposited on the cells lining the airways, where they can damage DNA and potentially cause cancers [1,2].
Lung cancer is the most commonly diagnosed cancer worldwide and the leading cause of cancer death [3]. According to Global Cancer Observatory (2020), lung cancer accounted for 11.4% (2.2 million) of total cancer cases and 18% (1.79 million) of cancer deaths [3]. Among the risk factors of lung cancer (e.g., radon, air pollution, arsenic, silica, diesel, chromium, cadmium, and beryllium), radon is well known as one of the 19 environmental carcinogens recognized by the World Health Organization (WHO). The WHO reports that radon causes up to 3–14% of all lung cancers in a country, depending on the country’s average radon level and smoking prevalence [1]. Moreover, radon accounts for 2% (21,000 deaths) of European cancer [3]. The risk of lung cancer increases by approximately 16% per 100 Bq/m3 increase in long-time average radon concentration [1]. Studies on residential radon as a risk factor for lung cancer have attracted attention and provided substantial epidemiological evidence. For instance, Darby et al. (1998) conducted a meta-analysis analysis from 13 studies in Europe and demonstrated a statistical increase of 16% in lung cancer risk per 100 Bq/m3 of residential radon [4]. Lorenzo-Gonzalez et al. (2019) found an increased odds ratio (OR) for lung cancer in 523 individuals exposed to radon ≥200 Bq/m3 compared with those exposed to ≤100 Bq/m3 [5]. It has been reported that the role of radon in the development of lung cancer is related to the emission of alpha particles from radon with a high potential for damage to the respiratory epithelium [6]. First, alpha particles can impact the respiratory epithelium and produce various cytotoxic and genotoxic effects, which favor carcinogenesis [6]. These genotoxic effects result in large-scale molecular changes, including DNA double-strand breaks, translocations and deletions, substitutions, and chromosomal rearrangements, thereby inducing the dysregulation of cytokines and the increased production of proteins associated with carcinogenesis [6]. Secondly, alpha radiation impacts the immune system in the tumor microenvironment [6]. The overproduction of reactive oxygen species (ROS) in the lungs caused by repeated exposure to radon can cause oxidative stress, leading to pulmonary inflammation. Moreover, radon can enhance tumor immunogenicity by increasing genomic instability and cluster mutations in the tumor cells [6].
Childhood leukemias are a hematological cancer of the leukocytes in children [7]. Diseases that lead to the dramatically excessive proliferation of white blood cells, resulting in an invasion of the bone marrow, the lymph nodes, and the spleen with an excessive accumulation of cells in the blood flow, end with an extension of malignant colonization in various tissues of the body, especially the liver and the central nervous system [7]. Childhood leukemia is commonly induced by genetic risk factors (e.g., genetic syndromes and inherited immune system problems) and environmental factors (ionizing/non-ionizing radiation exposure, chemotherapy exposure, prenatal environment, socioeconomic status, and immune system suppression) [7,8]. Regarding environmental risk factors, especially radon exposure, the exact mechanisms underlying leukemia remain unclear. It is hypothesized that when we breathe, a small amount of radon is transported to the red bone marrow, which then causes specific changes in the DNA, especially the creation of double-strand DNA breaks inside normal bone marrow cells. Once the leukocytes in blood cells undergo an out-of-control mutation, they become leukemia cells. Recently, it has been reported that natural background radon contributes to a 20% increase in childhood leukemia cases [9,10]. A causal relationship has been established between residential radon and childhood leukemia, with side-effects depending on the age at the time of exposure, sex, and exposure duration through many studies [9,11]. For example, Axelson et al. (2002) evaluated the possible risk of lymphocytic leukemia from exposure to residential radon in 312 cases and 1418 controls in Sweden during 1980–1989. The study obtained a conditional OR of 1.4 [1.05; 1.91], which suggested some risk of acute lymphocytic leukemia in children from indoor ionizing radiation [12]. Brauner et al. (2010) reported a risk for childhood leukemia associated with a radon increase of 103 Bq/m3 per year with an OR of 1.77 [1.11; 2.83], proposing a high risk of radon exposure causing childhood leukemia [12].
In this light, numerous recent case–control studies have addressed the hypothesis that adults and children who grow up in homes with elevated radon concentrations have an increased risk of developing lung cancer and childhood leukemia, respectively [11,13,14,15,16]. However, these studies reported different conclusions with different effect sizes. Furthermore, these studies had a heterogeneous study population, design, setting, exposure measurement, and risk calculation method, making it difficult to draw consistent conclusions. Therefore, systematic reviews and meta-analyses have been applied to pool individual estimated size effects and to provide a statistically significant conclusion to the hypothesis.
Many recent systematic reviews and meta-analyses have addressed this topic; however, they include very little eligible data, and therefore, their results may be underestimated [2,16,17]. Moreover, the number of case–control studies has been increasing year by year; therefore, this updated systematic review and meta-analysis were designed to access a more significant, larger sample size and higher-quality data source to provide more accurate results than the previous systematic reviews. This study collected and analyzed estimated size effects, namely, the odds ratio (OR) and risk ratio (RR), from case–control studies of lung cancer and childhood leukemia caused by radon. The results from traditional comparisons and subgroup analyses might contribute to discovering the impact of radon exposure on human cancers.
2. Materials and Methods
2.1. Literature Search Strategy
According to the Preferred Reporting Items for Systematic Reviews and Meta-analysis (PRISMA) protocol 2009 [18], a literature search on the impacts of residential radon on human health was performed in the English-language databases, including PubMed, Cochrane Library, EMBASE, MedRxiv, and Elsevier, to obtain relevant articles published up to 2022. The literature search was performed using the following keywords: residential radon, human health, lung cancer, childhood leukemia, and case–control studies. The protocol has been registered with the PROSPERO International Prospective Register of Systematic Reviews. The registration number is CRD42022379937.
2.2. Study Selection and Data Extraction
2.2.1. Study Selection
All case–control studies that evaluated the adverse effects of residential radon exposure on human health, particularly lung cancer and childhood leukemia, were included in this study. All case–control studies that studied at least one case (lung cancer or childhood leukemia patient) and one matched participant in the control arm were considered as long as there were no differences in the relationship between residential radon exposure and human cancers. Case–control studies with multiple components were considered eligible if they directly compared the case and the control, with the right arms included in the meta-analysis. In the case–control studies, results were reported as correlation coefficients (i.e., ORs/RRs) with their corresponding 95% confidence interval (CI) regardless of whether it was the primary outcome or not. No restrictions were set on the case–control status, patient background, indoor environmental quality, and geographical region. Furthermore, the independent and dependent variables for case–control studies should be measured for the individual radon doses and the incidence of lung cancer and childhood leukemia, respectively. If multiple studies reported different outcome types (e.g., incidence or mortality), the study with incidence outcomes was selected.
Studies of workplace exposures (e.g., studies of miners) and studies of different types (e.g., ecological, cohort, and randomized clinical trials) were excluded. Duplicate articles from all or part of other articles were excluded. Among repeated articles, only the latest one was included. Studies that only investigated the exposure to gamma radiation and radon daughters were excluded. The hypothesis of this study was to confirm a causal relationship between residential radon exposure and the incidence of human cancers. In addition, studies of the relationship between exposure to radon in water and cancers were excluded. It can explain that radon needs to be in gaseous form to have remarkably carcinogenic effects on the lungs. In contrast, radon in drinking water is associated with an increased risk of developing other organ cancers, primarily stomach cancer [12]. In terms of water containing radon being used in the home for cooking, washing dishes, and showering, radon gas escapes from the water, goes into the air, and then traps in the lungs. However, it is reported that a minimal amount of radon is transferred from domestic water to indoor air, with a 37 Bq/m3 increase in radon in indoor air from a 370,000 Bq/m3 increase in dissolved radon in domestic water [12]. Therefore, radon escaping from water is not a massive risk of developing lung cancer. Thus, a separate study should be performed for the risk of water-soluble radon because the dose–response relationship would be significantly different from the threat of radon in indoor air.
2.2.2. Data Extraction
Two independent reviewers screened each included study and recorded appropriate data for this systematic review and meta-analysis. Then, a predefined form was used to extract the following data from each case–control study: author name, year of publication, country of study, period of investigation, the age of participant, sample size (cases and controls), types of cancer (i.e., lung cancer and childhood leukemia), residential radon levels (Bq/m3), estimated effect sizes (i.e., OR and RR), and adjustment criteria for the main confounding factor of interest.
2.3. Meta-Analysis
For all dichotomous outcomes, the estimated size effects (i.e., ORs/RRs) on the lung cancer/childhood leukemia incidence in participants with the highest radon exposure relative to the participants with no/lowest radon exposure were pooled using the random-effects model and expressed as the OR/RR with 95% CI [19]. If the CI for an estimate includes 1, it cannot indicate a statistically significant difference between the groups being compared; in contrast, if it does not include 1, it indicates a statistically significant difference [19]. Initially, all studies were pooled together, and a sensitivity analysis was performed to assess the influences of the methodological concerns by grouping them into subgroups, including geographical region, period of investigation, residential radon level, and smoking status. In addition, the heterogeneity of each study was analyzed using the standard coefficient heterogeneity (I2) test. Heterogeneity was considered negligible, moderate, and large when I2 was less than 25%, 25–50%, and more than 50%, respectively [19]. The value p ≥ 0.10 was considered statistically significant, and it suggested that the studies were homogeneous. All these analyses were conducted using Comprehensive Meta-Analysis Review Manager (version 5.3, Copenhagen, Denmark: The Nordic Cochrane Center, The Cochrane Collaboration, 2014 version V3, Biostat Inc., Tampa, FL, USA).
2.4. Quality Assessment of Included Studies
To explore the validity of eligible case–control studies, the quality of biased evaluations was determined according to the Newcastle–Ottawa scale guideline [20]. A “star system” was developed, in which a study was judged on three broad perspectives: the selection of study groups, comparability of study groups, and ascertainment of radon exposure (Table S1).
3. Results and Discussion
3.1. Characteristics of Included Studies
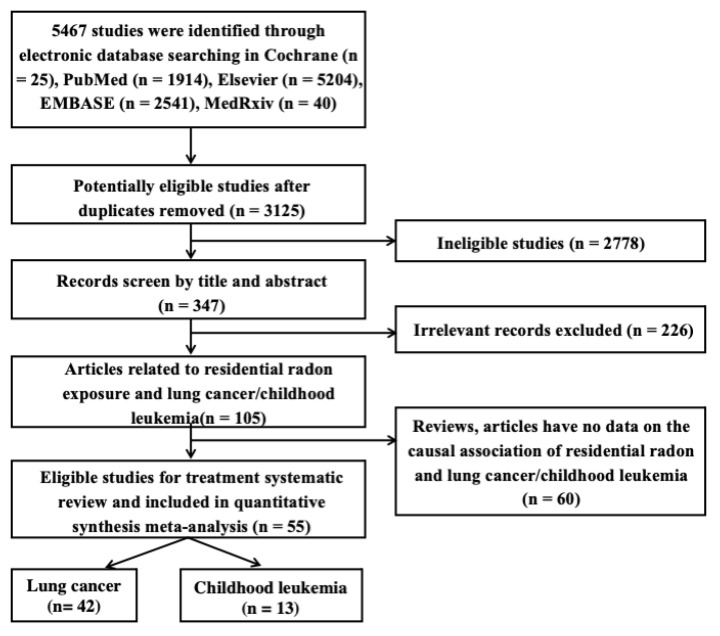
Figure 1 shows the flowchart of the process by which the qualified articles were screened. In this study, 9724 different publications were selected by screening through their titles and abstracts based on the searches from the abovementioned databases. After carefully reviewing these articles on the risk of residential radon exposure for human cancers (i.e., lung cancer and childhood leukemia) owing to residential radon exposure, 55 case–control studies were included for a quantitative meta-analysis.
Figure 1.
Systematic screening stages of literature review.
Description of Included Studies
The main characteristics of the 55 studies are summarized in Table 1. Among the 55 articles, 42 case–control studies reported the incidence of lung cancer in the participants, and the remaining 13 reported a strong relationship between radon exposure and childhood leukemia. These studies covered the period from 1992 to 2022 and included 48,726 cases and 98,691 control-matched participants. They were conducted in different parts of the world, including Africa (n = 1), Asia (n = 8), North America (n = 15), and Europe (n = 31), thereby ensuring good geo-ethnic representation. In addition, they were conducted over different time periods, including <10 years (n = 39) [21,22,23,24], 10–20 years (n = 10) [25,26,27,28], and 20–40 years (n = 6) [11,29,30,31,32,33], thus providing the high statistical significance for the subgroup analysis based on the period of investigations. To evaluate the adverse effects of residential radon exposure on human health, the researchers compared lung cancer/childhood leukemia rates between the cases and controls based on various categories of radon exposure levels, including at <100 Bq/m3 (n = 7), 100–150 Bq/m3 (n = 27), and ≥200 Bq/m3 (n = 21). These comparisons were made based not only on radon exposure levels but also the adjustment criteria, especially the sub-criteria of “sex, age, and smoking status” [22,27,34] and “age, socioeconomic, mom’s age, and parental occupational exposure” [12,16,31] for studies of lung cancer and childhood leukemia, respectively. Further, all included studies revealed the primary outcomes by presenting the estimated size effects (i.e., OR/RR) of the incidence of cancers caused by residential radon inhalation. Overall, most studies reported a high association between residential radon exposure and increased lung cancer/childhood leukemia.
Table 1.
Summary of the characteristics of case–control studies.
| References | Study Location | Period of Investigation | Ages | Human Cancers | Case/Control | Exposure Comparison (Bq/m3) | Types of Size Effects | Adjustment Criteria |
|---|---|---|---|---|---|---|---|---|
| Alavanja et al., 1994 [34] | USA | 1986–1992 | 55–80 | Lung cancer | 538/1183 | >100 vs. <29 | OR | Age, sex, and smoking status |
| Alavanja et al., 1995 [23] | USA | 1986–1992 | 55–75 | Lung cancer | 618/1402 | >200 vs. <50 | OR | Age and sex |
| Alavanja et al., 1999 [21] | USA | 1993–1994 | 65–84 | Lung cancer | 512/3886 | >148 vs. <37 Increase of 100 Bq/m3 |
OR | Age, sex, education, and smoking status |
| Auvinen et al., 1996 [35] | Finland | 1986–1992 | 54–75 | Lung cancer | 1055/1544 | >200 vs. <50 Increase of 100 Bq/m3 |
OR | Cigarette smoking, intensity, duration, and age |
| Barros-Dios et al., 2002 [22] | Spain | 1992–1994 | 35–70 | Lung cancer | 163/241 | 148 vs. <37 | OR | Sex, age, lifetime tobacco use, family history, and habitat |
| Barros-Dios et al., 2012 [36] | Spain | 2004–2008 | 50–70 | Lung cancer | 349/513 | >148 vs. <50 Increase of 100 Bq/m3 |
OR | Age, sex, and tobacco consumption |
| Baysson et al., 2004 [37] | France | 1992–1998 | 40–75 | Lung cancer | 486/984 | >200 vs. <50 | OR | Age, sex, region, smoking status, and occupational exposure |
| Bochicchio et al., 2005 [13] | Sweden | 1993–1996 | 35–90 | Lung cancer | 384/404 | >200 vs. <50 Increase of 100 Bq/m3 |
OR | Age, smoking, and diet |
| Chiu et al., 2010 [14] | Hong Kong | 2002–2004 | 50–70 | Lung cancer | 279/322 | >150 vs. <25 | OR | Age, employment, and years of education |
| Darby et al., 1998 [4] | UK | 1988–1993 | 45–73 | Lung cancer | 982/3185 | 200 vs. <25 | OR | Age, sex, smoking status, country of residence, and social class |
| Field et al., 2000 [24] | USA | 1993–1997 | 40–84 | Lung cancer | 413/614 | 148 vs. <50 | OR | Age, smoking, and education |
| Hystad et al., 2014 [38] | Canada | 1994–1997 | 59–63 | Lung cancer | 2390/3507 | >50 | OR | Age, sex, smoke consumption |
| Kreienbrock et al., 2001 [39] | Germany | 1990–1996 | 40–80 | Lung cancer | 1449/2297 | >140 vs. <50 Increase of 100 Bq/m3 |
OR | Age and sex |
| Kreuzer et al., 2001 [15] | Germany | 1990–1996 | 50–74 | Lung cancer | 58/803 | Increase of 100 Bq/m3 | OR | Age, region, occupational carcinogens, and indoor radon |
| Kreuzer et al., 2003 [40] | Germany | 1990–1997 | 50–75 | Lung cancer | 1192/1640 | >140 vs. <50 | OR | Smoking and asbestos exposure |
| Kudo et al., 2021 [41] | China | 2005–2007 | 39–77 | Lung cancer | 30/39 | >100 vs. < 50 Increase of 100 Bq/m3 |
OR | Age, smoking, and income |
| Lagarde et al., 2000 [26] | Sweden | 1980–1995 | 35–74 | Lung cancer | 258/487 | 140 vs. <50 | OR | Age, sex, and smoking status |
| Lagarde et al., 2002 [27] | Sweden | 1985–1995 | 40–70 | Lung cancer | 110/231 | 140 vs. <50 | RR | Age, sex, and smoking status |
| Letourneau et al., 1994 [42] | Canada | 1983–1990 | 25–76 | Lung cancer | 738/738 | 375 per month | OR | Age and sex |
| Lorenzo-Gonzalez et al., 2019 [5] | Spain | 2002–2017 | 57–78 | Lung cancer | 523/892 | ≥200 vs. ≤100 | OR | Age, sex, and environmental tobacco-smoke exposure |
| Lorenzo-Gonzalez et al., 2020 [43] | Spain | 2004–2019 | 54–71 | Lung cancer | 1842/1862 | >200 vs. <50 | OR | Age, sex, and never smoker |
| Lubin et al., 2003 [44] | USA | 1992–2000 | 35–70 | Lung cancer | 4081/5281 | Increase of 100 Bq/m3 | OR | Age and sex |
| Lubin et al., 2004 [45] | China | 1985–1987 | 40–75 | Lung cancer | 1053/1997 | 200 vs. <100 Increase of 100 Bq/m3 |
OR | Age, sex, and smoking status |
| Nyberg et al., 2000 [29] | Sweden | 1950–1990 | 40–75 | Lung cancer | 1042/2364 | >116 vs. <78 Increase of 100 Bq/m3 |
OR | Tobacco smoking, socioeconomic status, residential radon, and occupational exposures |
| Park et al., 2020 [46] | Korea | 2015–2018 | 57–72 | Lung cancer | 519/519 | ≥100 vs. 25 | OR | Age, sex, and indoor hours, and smoking status |
| Pershagen et al., 1992 [47] | Swedish | 1983–1986 | 35–70 | Lung cancer | 210/209 | >150 vs. <24 | OR | Age, sex, and smoking status |
| Pershagen et al., 1994 [48] | Sweden | 1980–1984 | 35–74 | Lung cancer | 1360/2847 | >400 vs. <50 Increase of 100 Bq/m3 |
OR | Age and sex |
| Pisa et al., 2000 [49] | Italy | 1991–1997 | 30–70 | Lung cancer | 138/291 | >200 vs. <40 | OR | Age and sex |
| Ruano-Ravina et al., 2021 [50] | Spain | 2018–2019 | 51–68 | Lung cancer | 189/747 | >200 vs. <50 | OR | Sex, age, and education |
| Sandler et al., 2007 [51] | USA | 1996–2006 | 40–79 | Lung cancer | 1474/1811 | <100 | RR | Age and sex |
| Schoenberg et al., 1990 [52] | USA | 1980–1989 | 35–60 | Lung cancer | 433/402 | >148 vs. <37 | OR | Age, sex, and smoking status |
| Sobue et al., 2000 [53] | Japan | 1976–1996 | 40–80 | Lung cancer | 28/36 | 100 vs. <25 | OR | Age, sex, occupational exposure, and smoking status |
| Svensson et al., 1989 [54] | Sweden | 1980–1986 | 40–70 | Lung cancer | 210/209 | >200 vs. <50 | RR | Smoking, age, and degree of urbanization |
| Thompson et al., 2011 [55] | USA | 1990–2010 | 40–70 | Lung cancer | 200/397 | ≥250 vs. <25 | OR | Smoking, residency, job exposure, income, and education |
| Torres-Duran et al., 2014 [56] | Spain | 2011–2013 | 61–79 | Lung cancer | 192/329 | 200 vs. <100 | OR | Age and sex |
| Torres-Duran et al., 2015 [57] | Spain | 2000–2012 | 50–70 | Lung cancer | 198/275 | >200 vs. <50 | OR | Age and sex |
| Tse et al., 2011 [58] | China | 2004–2006 | 35–79 | Lung cancer | 1208/1069 | >200 vs. <50 | OR | Age and sex |
| Tse et al., 2022 [59] | Hong Kong | 2004–2006 | 35–79 | Lung cancer | 1069/1208 | Increase of 100 Bq/m3 | OR | Age and sex |
| Wang et al., 2002 [60] | China | 1994–1998 | 30–75 | Lung cancer | 768/1659 | >300 vs. <50 Increase of 100 Bq/m3 |
OR | Age, sex, prefecture, and tobacco use, |
| Wichmanm et al., 2005 [61] | Germany | 1990–1997 | 35–75 | Lung cancer | 2963/4232 | >140 vs. <50 Increase of 100 Bq/m3 |
OR | Sex, age, and smoking status |
| Wilcox et al., 2008 [62] | Canada | 1989–1992 | 50–75 | Lung cancer | 561/740 | 150 vs. <25 Increase of 100 Bq/m3 |
OR | Sex and age |
| William Field et al., 2001 [25] | USA | 1981–2001 | 40–84 | Lung cancer | 413/614 | 55–150 | OR | Age and sex |
| Axelson et al., 2002 [63] | Sweden | 1980–1989 | <20 | Childhood leukemia | 312/1418 | 100 | OR | Age, sex, and county |
| Demoury et al., 2017 [28] | France | 1990–2009 | <15 | Childhood leukemia | 9056/30000 | >89 | OR | Family income and age |
| Kaletsch et al., 1999 [64] | Germany | 1988–1993 | <15 | Childhood leukemia | 164/209 | 70 | RR | Age and sex |
| Kollerud et al., 2014 [30] | Norway | 1967–2009 | <15 | Childhood leukemia | 712/674 | >100 vs. 50 | RR | Socioeconomic |
| Lubin et al., 1998 [65] | USA | 1989–1993 | <5 | Childhood leukemia | 505/443 | ≥148 vs. <37 | RR | Age, income, sex, type of residence, parental smoking habits, and parental occupation |
| Maged et al., 2008 [66] | Egypt | 1996–1998 | 2–14 | Childhood leukemia | 50/110 | >90 vs. <40 | OR | Age and sex |
| McLaughlin et al., 1993 [11] | USA | 1950–1988 | <14 | Childhood leukemia | 112/890 | >50 vs. 0 | OR | Parental occupation exposure |
| Nikkilä et al., 2020 [32] | Finland | 1990–2011 | 2–7 | Childhood leukemia | 1093/3279 | 37 | OR | Age, parental occupation |
| Pedersen et al., 2014 [31] | France | 1968–1991 | <15 | Childhood leukemia | 879/1621 | ≥42 | RR | Socioeconomic status, maternal age, birth order |
| Raaschou-Nielsen et al., 2007 [33] | Denmark | 1968–1994 | <14 | Childhood leukemia | 860/1720 | <260 | RR | Age and sex |
| Steinbuch et al., 1999 [16] | USA | 1989–1993 | <15 | Childhood leukemia | 173/254 | >100 vs. <37 | OR | Sex, age, maternal education, family income, and maternal race |
| Cartwright et al., 2002 [67] | UK | 1992–1996 | <14 | Childhood leukemia | 2226/3773 | >200 vs. <24 | OR | Age and sex |
| Vaclavik Brauner et al., 2010 [12] | Denmark | 1986–1994 | <20 | Childhood leukemia | 985/1969 | >200 vs. <20 | OR | Birth order, mother’s age, and electromagnetic fields |
3.2. Association between Residential Radon Exposure and Human Cancers
3.2.1. Increased Incidence of Lung Cancer
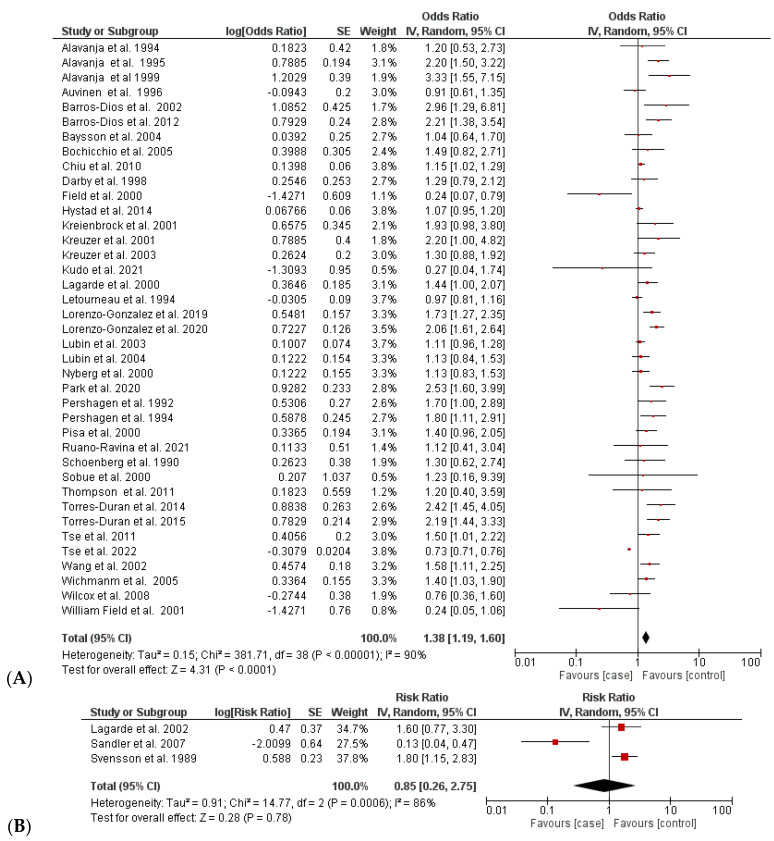
The association between residential radon exposure and increased lung cancer was assessed based on a comparison of the estimated effect sizes (i.e., OR/RR) in 42 case–control studies. The multivariable-adjusted ORs/RRs of lung cancer are shown in Figure 2 and Table 2. A random-effects model was used to calculate the pooled estimated size effects, because substantial heterogeneity was found between these studies (I2 = 90%, p < 0.00001). The pooled OR of 39 studies showed a statistically significant association between the highest residential radon exposure and increased risk of lung cancer (OR = 1.38 (95% CI 1.19; 1.60)). In contrast, the pooled RR of three case–control studies showed no significant difference in the increase in lung cancer between case and control groups with an overall RR of 0.85 [0.26; 2.73] (I2 = 80% and p = 0.0006) [14,44,47].
Figure 2.
Comparison of incidence of radon-caused lung cancer between case and control groups: (A) analysis based on OR values [4,5,13,14,15,21,22,23,24,25,26,29,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,52,53,55,56,57,58,59,60,61,62] and (B) analysis based on RR values [27,51,54]. (B) Red color: OR/RR values of individual studies, Black color: pooled OR/RR of the comparison.
Table 2.
Summary of pooled estimated size effects (95% CI) of case–control studies.
| Human Cancer | Types of Estimate Size Effect | No. of Studies | No. of Cases | No. of Controls | Pooled Estimated Size Effect | I2 | p Value |
|---|---|---|---|---|---|---|---|
| Lung cancer | OR | 39 | 30,884 | 51,759 | 1.38 [1.19; 1.60] | 90 | <0.00001 |
| RR | 3 | 1794 | 2251 | 0.85 [0.26; 2.74] | 86 | 0.0006 | |
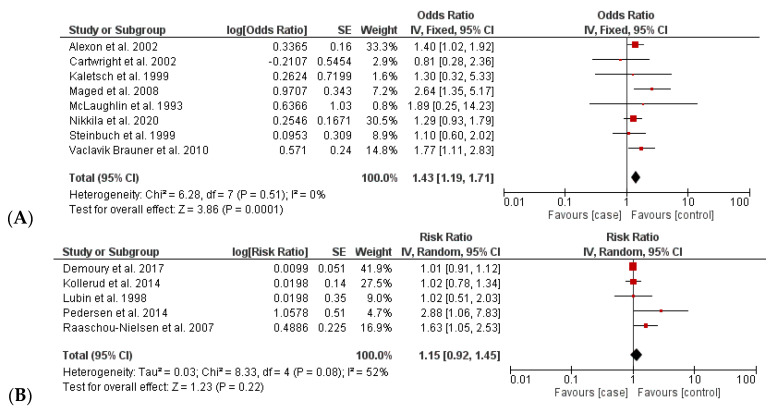
| Childhood leukemia | OR | 8 | 12,053 | 36,804 | 1.43 [1.19; 1.72] | 0.00 | 0.51 |
| RR | 5 | 50,127 | 46,360 | 1.15 [0.92; 1.45] | 52 | 0.08 |
In the sensitivity analysis, removing one study at a time did not significantly affect the pooled OR; it ranged from 1.30 [1.19; 1.62] (I2 = 91.43%, p < 0.0001) to 1.41 [1.27; 1.58] (I2 = 67.30%; p < 0.0001). However, the heterogeneity was primarily high owing to the significant variation in the individual study regions, sample size, and other effect factors (e.g., smoking status and residential radon level).
3.2.2. Increased Incidence of Childhood Leukemia
The pooled estimate of 13 case–control studies for the increase in childhood leukemia induced by residential radon exposure was divided into subcomparisons based on the estimated size effects, namely, OR and RR (Table 2 and Figure 3). The results indicated that the rate of childhood leukemia increased significantly over time with the amount of residential radon exposure with a pooled OR of 1.43 [1.19; 1.72] (I2 = 0% and p = 0.51). In contrast, the pooled RR suggested no association between radon exposure and cancer (RR 1.15 [0.92; 1.45]; I2 = 52%; and p = 0.08). The heterogeneities did not exist in the comparison based on OR values, while the comparison based on RR values exhibited a moderate degree of heterogeneity (I2 = 0% and 52%).
Figure 3.
Comparison of incidence of radon-caused childhood leukemia between case and control groups: (A) analysis based on OR values [11,12,16,32,63,64,66,67] and (B) analysis based on RR values [28,30,31,33,65]. (B) Red color: OR/RR values of individual studies, Black color: pooled OR/RR of the comparison.
3.3. Subgroup Analysis
In this study, a subgroup analysis was designed to assess the impact of residential radon inhalation on human health based on estimated OR values corresponding to four subcriteria (Table S2). First, the subgroup analysis was stratified by the study population, pooling North America, Europe, Asia, and Africa. Second, the included case–control studies were classified based on the study periods for radon measurement, including <10 years, from 10 to 20 years, and >20 years of radon measurement. Third, classifications were performed based on radon exposure levels of ≤100 Bq/m3, 100–150 Bq/m3, and ≥200 Bq/m3. Finally, the included studies were categorized regarding smoking status of patients, including nonsmokers and smokers.
Regarding the relationship between radon exposure and lung cancer, the subgroup analysis was performed using the random-effects model because the comparisons had considerable heterogeneity (I2 > 50%). In the analysis of the study population, a statistically significant increase in lung cancer due to residential radon exposure was observed in the case–control studies conducted in Europe (OR = 1.56 [1.37; 1.78]), but not in those conducted in North America (1.16 [0.95; 1.41], I2 = 69.55%, p < 0.0001) and Asia (1.24 [0.89; 1.71], I2 = 93.78%, p < 0.0001) (Table 3). The results showed that studies conducted for ≤10 years or for 10–20 years presented a strong association between lung cancer and radon exposure with pooled ORs of 1.36 [1.16; 1.59] and 1.84 [1.57; 2.15], respectively. An association was also confirmed when comparing case- and control-exposed lung cancer with the high residential radon levels (100–150 Bq/m3 and ≥200 Bq/m3) (Table 3). Regarding smoking status, residential radon exposure was strongly associated with an increase in lung cancer even among nonsmokers or current smokers with ORs of 1.21 [1.12; 1.31] and 1.38 [1.20; 1.57], respectively. Especially, smokers faced a higher risk of lung cancer than nonsmoker.
Table 3.
Pooled estimated size effects (95% CI) regarding subgroupanalysis of case–control studies.
| Subgroup Analysis | Lung Cancer | Childhood Leukemia | ||||||
|---|---|---|---|---|---|---|---|---|
| No. of Studies | OR (95% CI) | I2 (%) | p Value | No of Studies | OR (95% CI) | I2 (%) | p Value | |
| All studies | 39 | 1.38 [1.19; 1.60] | 90 | <0.00001 | 8 | 1.43 [1.19;1.71] | 0.00 | 0.51 |
| Study population | ||||||||
| North America | 11 | 1.16 [0.95; 1.41] | 69.55 | <0.0001 | 1 | 1.89 [0.25; 14.17] | – | – |
| Europe | 20 | 1.56 [1.37; 1.78] | 42.28 | 0.025 | 6 | 1.36 [1.12; 1.64] | 0.00 | 0.739 |
| Asia | 8 | 1.24 [0.89; 1.71] | 93.78 | <0.0001 | – | – | – | – |
| Africa | – | – | – | – | 1 | 2.64 [1.30; 5.18] | – | – |
| Period of study (years) | ||||||||
| <10 | 32 | 1.36 [1.16; 1.59] | 90.78 | <0.0001 | 6 | 1.49 [1.19; 1.86] | 12.41 | 0.336 |
| 10–20 | 6 | 1.84 [1.57; 2.15] | 0.00 | 0.545 | – | – | – | – |
| >20 | 1 | 1.13 [0.83; 1.55] | – | – | 2 | 1.30 [0.95; 1.79] | 0.00 | 0.714 |
| Level of radon exposure (Bq/m3) | ||||||||
| <100 | – | – | – | – | 4 | 1.44 [0.97; 1.78] | 18.05 | 0.301 |
| 100–150 | 23 | 1.21 [1.13; 1.28] | 63 | <0.0001 | 2 | 1.48 [0.65; 3.35] | 0.00 | 0.831 |
| ≥200 | 16 | 1.37 [1.09; 1.72] | 91 | <0.00001 | 2 | 1.56 [1.02; 2.39] | 42.35 | 0.188 |
| Smoking status | ||||||||
| Nonsmoker | 8 | 1.21 [1.12; 1.31] | 80 | <0.0001 | – | – | – | – |
| Smokers | 31 | 1.38 [1.20; 1.57] | 83 | <0.00001 | – | – | – | – |
The subgroup analyses of studies of childhood leukemia were performed using fixed-effect models because of insignificant heterogeneity (I2 < 50%). Statistically significant associations were observed in European studies and studies conducted for <10 years, with pooled ORs of 1.36 [1.12; 1.64] and 1.49 [1.19; 1.86], respectively. Moreover, the adverse effect of a residential radon level of ≥200 Bq/m3 on childhood leukemia was confirmed. Notably, all subgroup analyses achieved high statistical significance without heterogeneity (I2 = 0%–42.35% and p ≥ 0.1).
3.4. Dose–Response Analyses
In this study, only 15 included studies provided relevant data regarding lung cancer risk due to a 100 Bq/m3 increase in residential radon; therefore, a dose–response analysis was performed based on data extracted from the 15 studies [13,21,29,35,36,39,41,44,45,46,48,59]. Random-effects models were used because of heterogeneity (overall I2 = 93.39% and p < 0.0001). With a 100 Bq/m3 increase in the residential radon level, the risk of lung cancer was not significantly different between the case and control participants (OR = 1.21 [1.01; 1.45], I2 = 93%, p < 0.00001). Therefore, a dose–response relationship was concluded to exist between all lung cancers and residential radon exposure.
3.5. Analysis Bias
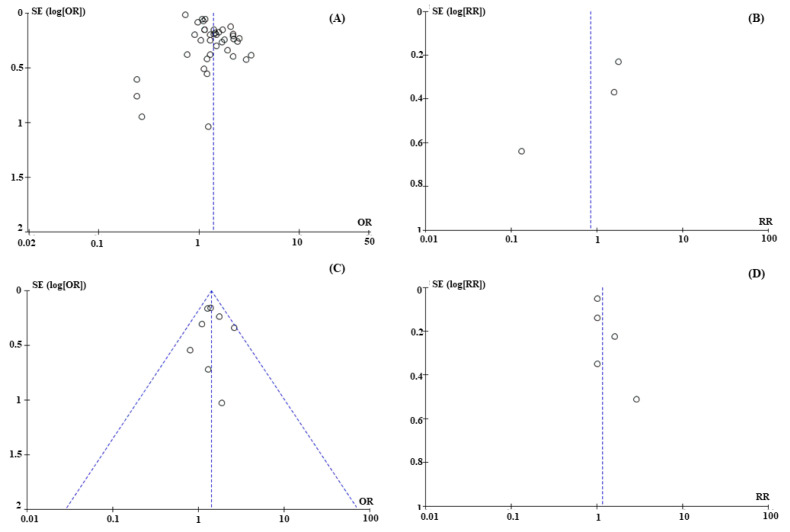
Funnel plots were constructed to detect publication bias. The results indicated that articles with positive OR/RR values have been published, while articles with negative OR/RR values have not been published, leading to an unsymmetrical publication bias (Figure 4A,B). In contrast, although the number of studies of childhood-leukemia-based studies was smaller, the publication was symmetrical and tended to include not only positive outcomes but also several negative ones of each aspect analyzed (Figure 4C,D). Therefore, this meta-analysis should be repeated when large numbers of case–control studies are available in the future to reduce publication bias and analysis heterogeneity.
Figure 4.
Publication bias of case–control studies related to radon-associated lung cancer and childhood leukemia. (A) Lung cancer-based studies with estimated OR values; (B) Lung cancer-based studies with estimated RR values; (C) Childhood leukemia-based studies with estimated OR values; (D) Childhood leukemia-based studies with estimated RR values.
4. Discussion
In this study, the causal associations between residential radon exposure and the increasing incidence of human cancers were assessed through a systematic review and meta-analysis of case–control studies. According to the eligibility criteria, 55 case–control studies with a total of 150,175 participants were enrolled and evaluated, and the results showed that radon exposure was strongly associated with increased lung cancer and childhood leukemia. However, with a 100 Bq/m3 increase in the residential radon level, the lung cancer rates were not significantly different between the case and control patients.
For assessing the relationship between radon exposure and lung cancer, 42 case–control studies that provided estimated size effect values (39 ORs and 3 RRs) were selected to perform a meta-analysis. However, the strong relationship was only confirmed through the meta-analysis based on OR values with a pooled OR of 1.36 [1.18; 1.58], even though the analysis had high heterogeneity. The significant heterogeneity in the statistical analysis can be explained by the fact that these included studies were conducted in various regions of the world and under different radon exposure concentrations with different periods of investigation. To reduce the heterogeneity, a subgroup analysis was then designed to reevaluate the relationship based on subcriteria such as the study population, study period, and residential radon exposure. The results revealed a significant difference in lung cancer rate between the cases and controls in Europe (1.56 [1.37; 1.78]) without heterogeneity (I2 = 42.28% < 50%), but not in North America and Asia. One out of 10 lung cancer cases in Europe is reportedly attributable to indoor radon exposure, and naturally occurring indoor residential radon may have caused around 19,000 lung cancer deaths in Europe in 2019 [68]. The high incidence of radon-associated lung cancer is relevant to the spatial distribution of indoor radon concentrations across Europe, with high indoor concentrations found in granitic zones and areas with certain rock types [69]. Specifically, energy-efficiency-oriented retrofitting, such as replacing old windows with energy-efficient double-glazed ones, insulating walls, and ceilings or replacing old doors with better sealing ones, may reduce ventilation and increase the building’s airtightness, thus increasing indoor concentrations in a radon-prone area [70]. Owing to the small number of studies on lung cancer and radon conducted in Asia and North America (8 and 11, respectively), these meta-analyses over the two regions had low statistical significance. Second, the subgroup analysis based on the period of investigation also confirmed the causal association of lung cancer and radon exposure, especially for participants exposed within 20 years. In particular, patients exposed to radon for 10–20 years had a higher incidence of lung cancer compared with that of patients exposed for less than 10 years, with pooled ORs of 1.84 [1.57; 2.15] and 1.36 [1.16; 1.59], respectively. Third, a comparison based on the residential radon levels indicated that high radon levels (≥100 Bq/m3) could cause significantly adverse effects for the lungs, whereas radon levels ≤100 Bq/m3 could be considered less toxic to the lungs. Notably, the highest risk of lung cancer was confirmed when patients were exposed to radon levels of ≥200 Bq/m3 followed by radon levels of 100–150 Bq/m3 with pooled ORs of 1.37 [1.09; 1.72] and 1.21 [1.13; 1.28], respectively. Finally, the analysis of smoking status indicated a strong association between radon and lung cancer. The results revealed that in the same areas with high radon concentrations, smokers had higher lung cancer incidence than nonsmokers (ORs of 1.38 [1.20; 1.57] > 1.21 [1.12; 1.31], respectively). Thus, it can be said that radon and smoking can be considered to have a synergistic effect on the occurrence and development of lung cancer.
In addition, the impact of residential radon on childhood leukemia was also analyzed based on the estimated ORs and RRs by two discriminant comparisons. However, adverse events were only confirmed by comparing the OR values with pooled ORs of 1.43 [1.19; 1.71] (I2 = 0% and p = 0.51). In contrast, the pooled RR of 1.15 [0.92; 1.45] (I2 = 52% and p = 0.08) from five case–control studies suggested no significant difference between childhood leukemia in the case and control groups. The controversial results may be due to the small size of the analysis, with eight studies reporting OR values versus five studies reporting RR values. Thus, adverse effects must be analyzed when larger sample sizes are obtained in the future. Similar to the lung cancer issues, the impact of radon on childhood leukemia was analyzed based on three subcriteria: study population, period of investigation, and radon level. The results also indicated that European children were more affected by residential radon than North American children, with pooled ORs of 1.36 [1.12; 1.64] and 1.89 [0.25; 14.17], respectively. Investigations with a shorter duration (≤10 years) seemed to yield highly accurate results for predicting adverse effects compared with a longer duration (≥20 years). Notably, the impact of radon exposure was confirmed in participants exposed to high radon levels (≥200 Bq/m3) with OR of 1.56 [1.02; 2.39] (I2 = 42.35% and p = 0.188). Therefore, a high residential radon level over a long period is concluded to be positively associated with childhood leukemia; it mutates cells in the immune system and bone marrow, leading to DNA damage and influencing other biologic mechanisms, and consequently inducing childhood leukemia [71].
Therefore, technical solutions must be found to reduce radon levels in existing buildings. For example, the WHO has recommended several technological solutions, including installing a radon sump system, increasing under-floor ventilation, improving ventilation, preventing radon from passing from the basement into living spaces, and sealing floors and walls [72]. A variety of policy solutions have also been proposed, such as providing information on radon levels and health risks; including radon prevention in building codes; establishing radon concentration reference levels; providing education on subsidies for radon reduction measures; including radon as a risk factor in national strategies related to cancer control, tobacco control, indoor air quality, and energy conservation [72].
Recently, many meta-analyses dealing with this topic have been designed to analyze data extracted from several studies, including case–control, ecological studies, cohort-match control, individual studies, and unpublished studies. For instance, Darby et al. (2004) determined the risk of lung cancer associated with radon exposure through a collaborative analysis of 13 case–control studies in nine European countries [4]. The results showed that lung cancer incidence increased by 0.084 [0.003; 0.158] per 100 Bq/m3 increase in radon. Further, residential radon, particularly for smokers and recent ex-smokers, was concluded to be responsible for approximately 2% of all cancer deaths in Europe [73]. Li et al. (2019) collected OR values from 28 case–control studies (13,748 lung cancer cases and 23,112 controls) to evaluate the risk of residential radon across histological types of lung cancer. The meta-analysis provides evidence for a positive and statistically significant relationship between radon exposure and all histological types of lung cancer such as small-cell lung carcinoma, adenocarcinoma, and squamous cell carcinoma with pooled ORs of 2.03 [1.52; 2.71], 1.58 [1.31; 1.91], and 1.43 [1.18; 1.74], respectively [2]. Further, another meta-analysis from Lu et al. (2020) included 10 studies (eight case–control and two cohort studies) involving 12,231 cases and 16,202 controls relevant to domestic radon exposure and childhood leukemia. A weak association was found in the case–control studies relative to that in the cohort studies, with an OR of 1.22 [1.01;1.42] and hazard ratio (HR) of 0.97 [0.81; 1.15], respectively [73]. Moon et al. (2021) estimated the possible causal association between residential radon exposure and leukemia using three types of studies, including case–control (n = 9), ecological (n = 8), and cohort studies (n = 15) [17]. Pooling of nine case–control studies increased the pooled OR by 1.03 [1.00; 1.06] for each 100 Bq/m3 radon increase for childhood leukemia [17]. All of the most current meta-analyses were performed with only a few case–control studies that were not large enough to accurately estimate the size effect and the significant difference between cases and controls. Moreover, the studies only described meta-analyses with a collaborative approach to collect case–control studies regarding the investigation period and study region. In particular, our study is the first to focus exclusively on the effect of residential radon on two types of human cancers (i.e., lung cancer and childhood leukemia). This study included the most extensive dataset for both emerging cancers (86,688 participants for lung cancer and 63,487 participants for childhood leukemia) and included up-to-date information relevant to the topic, such as study region, smoking status, levels of radon, and two kinds of measurement outcomes (i.e., OR and RR), expecting to provide highly accurate analytical results and improve the quality of evidence for the causal relationship between cancers and residential radon, compared to the published meta-analysis articles. Furthermore, the results from subgroup analyses had negligible heterogeneity, indicating high statistical significance.
5. Limitations of Study
The meta-analysis performed had three primary limitations. First, this study included only case–control studies; these may provide an incomplete overview of the causal association between residential radon and human health. Therefore, this topic should be addressed by analyzing all types of studies, such as cohort studies, ecological studies, and randomized clinical trials in the future. Second, the results may be affected by the classification of radon exposure. Given the different radon concentrations in other regions, the radon exposure classifications designed for each study are also different. Third, because it extracted only the OR/RR values of all lung cancers and all childhood leukemias, the study only investigated the overview of radon-induced cancers. Therefore, detailed conclusions about the incidence of each histological type of lung cancer (e.g., adenocarcinoma, small cell carcinoma, and squamous cell carcinoma) and childhood leukemia (e.g., lymphoid leukemia and myeloid leukemia) could not be drawn.
6. Conclusions
This meta-analysis of 55 case–control studies, including a total of 49,805 case patients and 100,370 control patients, suggested that residential radon was a risk factor for lung cancer and childhood leukemia when using estimated OR values for the analysis versus RR values. Notably, participants who lived in Europe or an area with high radon levels (≥100 Bq/m3) had a higher incidence of cancers compared to those in other regions (North America, Asia, and regions with radon levels <100 Bq/m3). Thus, appropriate technical and policy solutions should be recommended to reduce radon exposure to ensure the health of environmental conditions and residents. Further, although the meta-analysis revealed the high impact of residential radon on human health, the results were limited by their high heterogeneity and unsymmetrical publication bias. Thus, this investigation should be repeated when more relevant articles are available in the future to overcome the current limitations.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/ijerph20010097/s1, Table S1: Quality assessment of included case–control studies based on the Newcastle–Ottawa Scale (NOS) guideline; Table S2: Characteristics of case–control studies for subgroup analysis.
Author Contributions
Y.-C.L. planned the study and contributed the main ideas; L.T.N.N. and D.P. collected the data, and L.T.N.N. was principally responsible for the writing of the manuscript; Y.-C.L. commented on and revised the manuscript. All authors have read and agreed to the published version of the manuscript.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Data is unavailable due to privacy.
Conflicts of Interest
The authors declare no conflict of interest.
Funding Statement
This research was supported by the Basic Science Research Capacity Enhancement Project through a Korea Basic Science Institute (National Research Facilities and equipment Center) grant funded by the Ministry of Education (2019R1A6C1010016), and a grant from the Subway Fine Dust Reduction Technology Development Project of the Ministry of Land Infrastructure and Transport, Republic of Korea (21QPPW-B152306-03).
Footnotes
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content.
References
- 1.WHO Radon and Health. [(accessed on 3 November 2022)]. Available online: https://www.who.int/news-room/fact-sheets/detail/radon-and-health.
- 2.Li C., Wang C., Yu J., Fan Y., Liu D., Zhou W., Shi T. Residential Radon and Histological Types of Lung Cancer: A Meta-Analysis of Case—Control Studies. Int. J. Environ. Res. Public Health. 2020;17:1457. doi: 10.3390/ijerph17041457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Observatory G. Cancer Estimated Number of New Cases and Number of Deaths in 2020, World, Both Sexes, All Ages. [(accessed on 12 December 2022)]. Available online: https://gco.iarc.fr/
- 4.Darby S., Whitley E., Silcocks P., Thakrar B., Green M., Lomas P., Miles J., Reeves G., Fearn T., Doll R. Risk of Lung Cancer Associated with Residential Radon Exposure in South-West England: A Case-Control Study. Br. J. Cancer. 1998;78:394–408. doi: 10.1038/bjc.1998.506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Lorenzo-González M., Ruano-Ravina A., Torres-Durán M., Kelsey K.T., Provencio M., Parente-Lamelas I., Leiro-Fernández V., Vidal-García I., Castro-Añón O., Martínez C., et al. Lung Cancer and Residential Radon in Never-Smokers: A Pooling Study in the Northwest of Spain. Environ. Res. 2019;172:713–718. doi: 10.1016/j.envres.2019.03.011. [DOI] [PubMed] [Google Scholar]
- 6.Riudavets M., Garcia de Herreros M., Besse B., Mezquita L. Radon and Lung Cancer: Current Trends and Future Perspectives. Cancers. 2022;14:3142. doi: 10.3390/cancers14133142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Binger C.M., Ablin A.R., Feuerstein R.C., Kushner J.H., Zoger S., Mikkelsen C. Childhood Leukemia. N. Engl. J. Med. 1969;280:414–418. doi: 10.1056/NEJM196902202800804. [DOI] [PubMed] [Google Scholar]
- 8.Buffler P.A., Kwan M.L., Reynolds P., Urayama K.Y. Environmental and Genetic Risk Factors for Childhood Leukemia: Appraising the Evidence. Cancer Investig. 2005;23:60–75. doi: 10.1081/CNV-46402. [DOI] [PubMed] [Google Scholar]
- 9.Little M.P., Wakeford R., Borrego D., French B., Zablotska L.B., Adams M.J., Allodji R., de Vathaire F., Lee C., Brenner A. V Leukaemia and Myeloid Malignancy among People Exposed to Low Doses (<100 MSv) of Ionising Radiation during Childhood: A Pooled Analysis of Nine Historical Cohort Studies. Lancet Haematol. 2018;5:e346–e358. doi: 10.1016/S2352-3026(18)30092-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Laurent O., Ancelet S., Richardson D.B., Hémon D., Ielsch G., Demoury C., Clavel J., Laurier D. Potential Impacts of Radon, Terrestrial Gamma and Cosmic Rays on Childhood Leukemia in France: A Quantitative Risk Assessment. Radiat. Environ. Biophys. 2013;52:195–209. doi: 10.1007/s00411-013-0464-y. [DOI] [PubMed] [Google Scholar]
- 11.Mclaughlin J.R., King W.D., Anderson T.W., Clarke E.A., Ashmore J.P., King W.D., Clarke E.A., Anderson T.W. Paternal Radiation Exposure and Leukemia in Offspring: The Ontario Case-Control Study. BMJ. 1993;307:959–966. doi: 10.1136/bmj.307.6910.959. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Bräuner E.V., Andersen C.E., Andersen H.P., Gravesen P., Lind M., Ulbak K., Hertel O., Schüz J., Raaschou-Nielsen O. Is There Any Interaction between Domestic Radon Exposure and Air Pollution from Traffic in Relation to Childhood Leukemia Risk? Cancer Causes Control. 2010;21:1961–1964. doi: 10.1007/s10552-010-9608-4. [DOI] [PubMed] [Google Scholar]
- 13.Bochicchio F., Forastiere F., Farchi S., Quarto M., Axelson O. Residential Radon Exposure, Diet and Lung Cancer: A Case-Control Study in a Mediterranean Region. Int. J. Cancer. 2005;114:983–991. doi: 10.1002/ijc.20799. [DOI] [PubMed] [Google Scholar]
- 14.Chiu Y.L., Wang X.R., Qiu H., Yu I.T.S. Risk Factors for Lung Cancer: A Case-Control Study in Hong Kong Women. Cancer Causes Control. 2010;21:777–785. doi: 10.1007/s10552-010-9506-9. [DOI] [PubMed] [Google Scholar]
- 15.Kreuzer M., Gerken M., Kreienbrock L., Wellmann J., Wichmann H.E. Lung Cancer in Lifetime Nonsmoking Men—Results of a Case-Control Study in Germany. Br. J. Cancer. 2001;84:134–140. doi: 10.1054/bjoc.2000.1518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Steinbuch M., Weinberg C.R., Buckley J.D., Robison L.L., Sandler D.P. Indoor Residential Radon Exposure and Risk of Childhood Acute Myeloid Leukaemia. Br. J. Cancer. 1999;81:900–906. doi: 10.1038/sj.bjc.6690784. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Moon J., Yoo H.K. Residential Radon Exposure and Leukemia: A Meta-Analysis and Dose-Response Meta-Analyses for Ecological, Case-Control, and Cohort Studies. Environ. Res. 2021;202:111714. doi: 10.1016/j.envres.2021.111714. [DOI] [PubMed] [Google Scholar]
- 18.Moher D., Liberati A., Tetzlaff J., Altman D.G. Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement. Ann. Intern. Med. 2009;151:264–269. doi: 10.7326/0003-4819-151-4-200908180-00135. [DOI] [PubMed] [Google Scholar]
- 19.Borenstein M., Hedges L.V., Higgins J.P.T., Rothstein H.R. Introduction to Meta-Analysis. John Wiley & Sons; Hoboken, NJ, USA: 2021. [Google Scholar]
- 20.Peterson J., Welch V., Losos M., Tugwell P. The Newcastle-Ottawa Scale (NOS) for Assessing the Quality of Nonrandomised Studies in Meta-Analyses. Ottawa Hospital Research Institute; Ottawa, ON, Canada: 2011. pp. 1–12. [Google Scholar]
- 21.Alavanja M.C.R., Lubin J.H., Mahaffey J.A., Brownson R.C. Residential Radon Exposure and Risk of Lung Cancer in Missouri. Am. J. Public Health. 1999;89:1042–1048. doi: 10.2105/AJPH.89.7.1042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Barros-Dios J.M., Barreiro M.A., Ruano-Ravina A., Figueiras A. Exposure to Residential Radon and Lung Cancer in Spain: A Population-Based: Case-Control Study. Am. J. Epidemiol. 2002;156:548–555. doi: 10.1093/aje/kwf070. [DOI] [PubMed] [Google Scholar]
- 23.Alavanja M.C.R., Brownson R.C., Benichou J., Swanson C., Boice J.D. Attributable Risk of Lung Cancer in Lifetime Nonsmokers and Long-Term Ex-Smokers (Missouri, United States) Cancer Causes Control. 1995;6:209–216. doi: 10.1007/BF00051792. [DOI] [PubMed] [Google Scholar]
- 24.Field R.W., Steck D.J., Smith B.J., Brus C.P., Fisher E.L., Neuberger J.S., Platz C.E., Robinson R.A., Woolson R.F., Lynch C.F. Residential Radon Gas Exposure and Lung Cancer: The Iowa Radon Lung Cancer Study. Am. J. Epidemiol. 2000;151:1091–1102. doi: 10.1093/oxfordjournals.aje.a010153. [DOI] [PubMed] [Google Scholar]
- 25.Field R.W., Steck D.J., Smith B.J., Brus C.P., Fisher E.L., Neuberger J.S., Lynch C.F. The Iowa Radon Lung Cancer Study—Phase I: Residential Radon Gas Exposure and Lung Cancer. Sci. Total Environ. 2001;272:67–72. doi: 10.1016/S0048-9697(01)00666-0. [DOI] [PubMed] [Google Scholar]
- 26.Lagarde F., Axelsson G., Damber L., Mellander H., Nyberg F., Pershagen G. Residential Radon and Lung Cancer among Never-Smokers in Sweden. Epidemiology. 2001;12:396–404. doi: 10.1097/00001648-200107000-00009. [DOI] [PubMed] [Google Scholar]
- 27.Lagarde F., Falk R., Almren K., Nyberg F., Svensson H., Pershagen G. Glass-Based Radon-Exposure Assessment and Lung Cancer Risk. J. Expo. Sci. Environ. Epidemiol. 2002;12:344–354. doi: 10.1038/sj.jea.7500236. [DOI] [PubMed] [Google Scholar]
- 28.Demoury C., Marquant F., Ielsch G., Goujon S., Debayle C., Faure L., Coste A., Laurent O., Guillevic J., Laurier D., et al. Residential Exposure to Natural Background Radiation and Risk of Childhood. Environ. Health Perspect. 2017;125:714–720. doi: 10.1289/EHP296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Nyberg F., Bellander T., Pershagen G., Gustavsson P. Urban Air Pollution and Lung Cancer in Stockholm. Epidemiology. 2001;12:591–592. doi: 10.1097/00001648-200109000-00028. [DOI] [PubMed] [Google Scholar]
- 30.Del Risco Kollerud R., Blaasaas K.G., Claussen B. Risk of Leukaemia or Cancer in the Central Nervous System among Children Living in an Area with High Indoor Radon Concentrations: Results from a Cohort Study in Norway. Br. J. Cancer. 2014;111:1413–1420. doi: 10.1038/bjc.2014.400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Pedersen C., Bräuner E.V., Rod N.H., Albieri V., Andersen C.E., Ulbak K., Hertel O., Johansen C., Schüz J., Raaschou-Nielsen O. Distance to High-Voltage Power Lines and Risk of Childhood Leukemia—An Analysis of Confounding by and Interaction with Other Potential Risk Factors. PLoS ONE. 2014;9:e107096. doi: 10.1371/journal.pone.0107096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Nikkilä A., Arvela H., Mehtonen J., Raitanen J., Heinäniemi M., Lohi O., Auvinen A. Predicting Residential Radon Concentrations in Finland: Model Development, Validation, and Application to Childhood Leukemia. Scand. J. Work Environ. Health. 2020;46:278–292. doi: 10.5271/sjweh.3867. [DOI] [PubMed] [Google Scholar]
- 33.Raaschou-Nielsen O., Andersen C.E., Andersen H.P., Gravesen P., Lind M., Schüz J., Ulbak K. Domestic Radon and Childhood Cancer in Denmark. Epidemiology. 2007;18:S107. doi: 10.1097/01.ede.0000288431.93533.7f. [DOI] [PubMed] [Google Scholar]
- 34.Alavanja M.C.R., Brownson R.C., Lubin J.H., Berger E., Chang J., Boice J.D., Jr. Residential Radon Exposure and Lung Cancer among Nonsmoking Women. JNCI J. Natl. Cancer Inst. 1994;86:1829–1837. doi: 10.1093/jnci/86.24.1829. [DOI] [PubMed] [Google Scholar]
- 35.Auvinen A., Mäkeläinen I., Hakama M., Castrén O., Pukkala E., Reisbacka H., Rytömaa T. Indoor Radon Exposure and Risk of Lung Cancer: A Nested Case—Control Study in Finland. JNCI J. Natl. Cancer Inst. 1996;88:966–972. doi: 10.1093/jnci/88.14.966. [DOI] [PubMed] [Google Scholar]
- 36.Barros-Dios J.M., Ruano-Ravina A., Pérez-Ríos M., Castro-Bernárdez M., Abal-Arca J., Tojo-Castro M. Residential Radon Exposure, Histologic Types, and Lung Cancer Risk. A Case—Control Study in Galicia, Spain. Cancer Epidemiol. Biomark. Prev. 2012;21:951–958. doi: 10.1158/1055-9965.EPI-12-0146-T. [DOI] [PubMed] [Google Scholar]
- 37.Baysson H., Tirmarche M., Tymen G., Gouva S., Caillaud D., Artus J.C., Vergnenegre A., Ducloy F., Laurier D. Indoor Radon and Lung Cancer in France. Epidemiology. 2004;15:709–716. doi: 10.1097/01.ede.0000142150.60556.b8. [DOI] [PubMed] [Google Scholar]
- 38.Hystad P., Brauer M., Demers P.A., Johnson K.C., Setton E., Cervantes-Larios A., Poplawski K., McFarlane A., Whitehead A., Nicol A.M. Geographic Variation in Radon and Associated Lung Cancer Risk in Canada. Can. J. Public Health. 2014;105:e4–e10. doi: 10.17269/cjph.105.4002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Kreienbrock L., Kreuzer M., Gerken M., Dingerkus G., Wellmann J., Keller G., Erich Wichmann H. Case-Control Study on Lung Cancer and Residential Radon in Western Germany. Am. J. Epidemiol. 2001;153:42–52. doi: 10.1093/aje/153.1.42. [DOI] [PubMed] [Google Scholar]
- 40.Kreuzer M., Heinrich J., Wölke G., Rosario A.S., Gerken M., Wellmann J., Keller G., Kreienbrock L., Wichmann H.-E. Residential Radon and Risk of Lung Cancer in Eastern Germany. Epidemiology. 2003;14:559–568. doi: 10.1097/01.ede.0000071410.26053.c4. [DOI] [PubMed] [Google Scholar]
- 41.Kudo H., Yoshinaga S., Li X., Lei S., Zhang S., Sun Q., Koriyama C., Akiba S., Tokonami S. The First Attempt to Reevaluate Radon and Thoron Exposure in Gansu Province Study Using Radon-Thoron Discriminating Measurement Technique. Front Public Health. 2021;9:1–8. doi: 10.3389/fpubh.2021.764201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Letourneau E.G., Krewski D., Choi N.W., Goddard M.J., McGregor R.G., Zielinski J.M., Du J. Case-Control Study of Residential Radon and Lung Cancer in Winnipeg, Manitoba, Canada. Am. J. Epidemiol. 1994;140:310–322. doi: 10.1093/oxfordjournals.aje.a117253. [DOI] [PubMed] [Google Scholar]
- 43.Lorenzo-Gonzalez M., Ruano-Ravina A., Torres-Duran M., Kelsey K.T., Provencio M., Parente-Lamelas I., Piñeiro-Lamas M., Varela-Lema L., Perez-Rios M., Fernandez-Villar A., et al. Lung Cancer Risk and Residential Radon Exposure: A Pooling of Case-Control Studies in Northwestern Spain. Environ. Res. 2020;189:109968. doi: 10.1016/j.envres.2020.109968. [DOI] [PubMed] [Google Scholar]
- 44.Lubin J.H. Studies of Radon and Lung Cancer in North America and China. Radiat. Prot. Dosimetry. 2003;104:315–319. doi: 10.1093/oxfordjournals.rpd.a006194. [DOI] [PubMed] [Google Scholar]
- 45.Lubin J.H., Wang Z.Y., Boice J.D., Xu Z.Y., Blot W.J., De Wang L., Kleinerman R.A. Risk of Lung Cancer and Residential Radon in China: Pooled Results of Two Studies. Int. J. Cancer. 2004;109:132–137. doi: 10.1002/ijc.11683. [DOI] [PubMed] [Google Scholar]
- 46.Park E.J., Lee H., Kim H.C., Sheen S.S., Koh S.B., Park K.S., Cho N.H., Lee C.M., Kang D.R. Residential Radon Exposure and Cigarette Smoking in Association with Lung Cancer: A Matched Case-Control Study in Korea. Int. J. Environ. Res. Public Health. 2020;17:2946. doi: 10.3390/ijerph17082946. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Pershagen G., Liang Z.-H., Hrubec Z., Svensson C., Boice J.D., Jr. Residential Radon Exposure and Lung Cancer in Swedish Women. Health Phys. 1992;63:179–186. doi: 10.1097/00004032-199208000-00005. [DOI] [PubMed] [Google Scholar]
- 48.Pershagen G., Akerblom G., Axelson O., Clavensjo B., Damber L., Desai G., Enflo A., Lagarde F., Mellander H., Svartengren M. Residential Radon Exposure and Lung Cancer in Sweden. N. Engl. J. Med. 1994;330:159–164. doi: 10.1056/NEJM199401203300302. [DOI] [PubMed] [Google Scholar]
- 49.Pisa F.E., Barbone F., Betta A., Bonomi M., Alessandrini B., Bovenzi M. Residential Radon and Risk of Lung Cancer in an Italian Alpine Area. Arch. Environ. Health Int. J. 2001;56:208–215. doi: 10.1080/00039890109604444. [DOI] [PubMed] [Google Scholar]
- 50.Ruano-Ravina A., Cameselle-Lago C., Torres-Durán M., Pando-Sandoval A., Dacal-Quintas R., Valdés-Cuadrado L., Hernández-Hernández J., Consuegra-Vanegas A., Tenes-Mayén J.A., Varela-Lema L., et al. Indoor Radon Exposure and COPD, Synergic Association? A Multicentric, Hospital-Based Case–Control Study in a Radon-Prone Area. Arch. Bronconeumol. 2021;57:630–636. doi: 10.1016/j.arbres.2020.11.015. [DOI] [PubMed] [Google Scholar]
- 51.Sandler D.P., Weinberg C.R., Shore D.L., Archer V.E., Bishop Stone M., Lyon J.L., Rothney-Kozlak L., Shepherd M., Stolwijk J.A.J. Indoor Radon and Lung Cancer Risk in Connecticut and Utah. J. Toxicol. Environ. Health A. 2006;69:633–654. doi: 10.1080/15287390500261117. [DOI] [PubMed] [Google Scholar]
- 52.Schoenberg J.B., Klotz J.B., Wilcox H.B., Nicholls G.P., Gil-del-Real M.T., Stemhagen A., Mason T.J. Case-Control Study of Residential Radon and Lung Cancer among New Jersey Women. Cancer Res. 1990;50:6520–6524. [PubMed] [Google Scholar]
- 53.Sobue T., Lee V.S., Ye W., Tanooka H., Mifune M., Suyama A., Koga T., Morishima H., Kondo S. Residential Radon Exposure and Lung Cancer Risk in Misasa, Japan: A Case-Control Study. J. Radiat. Res. 2000;41:81–92. doi: 10.1269/jrr.41.81. [DOI] [PubMed] [Google Scholar]
- 54.Svensson C., Pershagen G., Klominek J. Lung Cancer in Women and Type of Dwelling in Relation to Radon Exposure. Cancer Res. 1989;49:1861–1865. [PubMed] [Google Scholar]
- 55.Thompson R.E. Epidemiological Evidence for Possible Radiation Hormesis from Radon Exposure: A Case-Control Study Conducted in Worcester, MA. Dose-Response. 2011;9:59–75. doi: 10.2203/dose-response.10-026.Thompson. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Torres-Durán M., Ruano-Ravina A., Parente-Lamelas I., Leiro-Fernández V., Abal-Arca J., Montero-Martínez C., Pena-Álvarez C., González-Barcala F.J., Castro-Añón O., Golpe-Gómez A., et al. Lung Cancer in Never-Smokers: A Case-Control Study in a Radon-Prone Area (Galicia, Spain) Eur. Respir. J. 2014;44:994–1001. doi: 10.1183/09031936.00017114. [DOI] [PubMed] [Google Scholar]
- 57.Torres-Duran M., Ruano-Ravina A., Parente-Lamelas I., Leiro-Fernandez V., Abal-Arca J., Montero-Martinez C., Pena-Alvarez C., Castro-Anon O., Golpe-Gomez A., Martinez C. Residential Radon and Lung Cancer Characteristics in Never Smokers. Int. J. Radiat. Biol. 2015;91:605–610. doi: 10.3109/09553002.2015.1047985. [DOI] [PubMed] [Google Scholar]
- 58.Tse L.A., Yu I.T.S., Qiu H., Au J.S.K., Wang X.R. A Case-Referent Study of Lung Cancer and Incense Smoke, Smoking, and Residential Radon in Chinese Men. Environ. Health Perspect. 2011;119:1641–1646. doi: 10.1289/ehp.1002790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Tse L.A., Wang F., Wong M.C.-S., Au J.S.-K., Yu I.T.-S. Risk Assessment and Prediction for Lung Cancer among Hong Kong Chinese Men. BMC Cancer. 2022;22:585. doi: 10.1186/s12885-022-09678-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Wang Z., Lubin J.H., Wang L., Zhang S., Boice J.D., Cui H., Zhang S., Conrath S., Xia Y., Shang B., et al. Residential Radon and Lung Cancer Risk in a High-Exposure Area of Gansu Province, China. Am. J. Epidemiol. 2002;155:554–564. doi: 10.1093/aje/155.6.554. [DOI] [PubMed] [Google Scholar]
- 61.Wichmann H.E., Rosario A.S., Heid I.M., Kreuzer M., Heinrich J., Kreienbrock L. Increased Lung Cancer Risk Due to Residential Radon in a Pooled and Extended Analysis of Studies in Germany. Health Phys. 2005;88:71–79. doi: 10.1097/01.HP.0000142497.31627.86. [DOI] [PubMed] [Google Scholar]
- 62.Wilcox H.B., Al-Zoughool M., Garner M.J., Jiang H., Klotz J.B., Krewski D., Nicholson W.J., Schoenberg J.B., Villeneuve P.J., Zielinski J.M. Case-Control Study of Radon and Lung Cancer in New Jersey. Radiat. Prot. Dosim. 2008;128:169–179. doi: 10.1093/rpd/ncm330. [DOI] [PubMed] [Google Scholar]
- 63.Axelson O., Fredrikson M., Åkerblom G., Hardell L. Leukemia in Childhood and Adolescence and Exposure to Ionizing Radiation in Homes Built from Uranium-Containing Alum Shale Concrete. Epidemiology. 2002;13:146–150. doi: 10.1097/00001648-200203000-00008. [DOI] [PubMed] [Google Scholar]
- 64.Kaletsch U., Kaatsch P., Meinert R., Schüz J., Czarwinski R., Michaelis J. Childhood Cancer and Residential Radon Exposure—Results of a Population-Based Case-Control Study in Lower Saxony (Germany) Radiat. Environ. Biophys. 1999;38:211–215. doi: 10.1007/s004110050158. [DOI] [PubMed] [Google Scholar]
- 65.Lubin J.H., Linet M.S., Boice J.D., Buckley J., Conrath S.M., Hatch E.E., Kleinerman R.A., Tarone R.E., Wacholder S., Robison L.L. Case-Control Study of Childhood Acute Lymphoblastic Leukemia and Residential Radon Exposure. J. Natl. Cancer Inst. 1998;90:294–300. doi: 10.1093/jnci/90.4.294. [DOI] [PubMed] [Google Scholar]
- 66.Maged A.F., Mokhtar G.M., El-Tobgui M.M., Gabbr A.A., Attia N.I., Shady M.M.A. Domestic Radon Concentration and Childhood Cancer Study in Cairo, Egypt. J. Environ. Sci. Health Part C. 2000;18:153–170. doi: 10.1080/10590500009373519. [DOI] [Google Scholar]
- 67.Cartwright R.A., Law G., Roman E., Gilman E., Eden O.B., Mott M., Muir K., Goodhead D., Kendall G., Doll R., et al. The United Kingdom Childhood Cancer Study of Exposure to Domestic Sources of Ionising Radiation: I: Radon Gas. Br. J. Cancer. 2002;86:1721–1726. doi: 10.1038/sj.bjc.6600276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Murray C.J.L., Aravkin A.Y., Zheng P., Abbafati C., Abbas K.M., Abbasi-Kangevari M., Abd-Allah F., Abdelalim A., Abdollahi M., Abdollahpour I. Global Burden of 87 Risk Factors in 204 Countries and Territories, 1990–2019: A Systematic Analysis for the Global Burden of Disease Study 2019. Lancet. 2020;396:1223–1249. doi: 10.1016/S0140-6736(20)30752-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Kropat G., Bochud F., Jaboyedoff M., Laedermann J.-P., Murith C., Palacios M., Baechler S. Major Influencing Factors of Indoor Radon Concentrations in Switzerland. J. Environ. Radioact. 2014;129:7–22. doi: 10.1016/j.jenvrad.2013.11.010. [DOI] [PubMed] [Google Scholar]
- 70.Symonds P., Rees D., Daraktchieva Z., McColl N., Bradley J., Hamilton I., Davies M. Home Energy Efficiency and Radon: An Observational Study. Indoor Air. 2019;29:854–864. doi: 10.1111/ina.12575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Peckham E.C., Scheurer M.E., Danysh H.E., Lubega J., Langlois P.H., Lupo P.J. Residential Radon Exposure and Incidence of Childhood Lymphoma in Texas, 1995–2011. Int. J. Environ. Res. Public Health. 2015;12:12110–12126. doi: 10.3390/ijerph121012110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Darby S., Hill D., Auvinen A., Barros-Dios J.M., Baysson H., Bochicchio F., Deo H., Falk R., Forastiere F., Hakama M., et al. Radon in Homes and Risk of Lung Cancer: Collaborative Analysis of Individual Data from 13 European Case-Control Studies. Br. Med. J. 2005;330:223–226. doi: 10.1136/bmj.38308.477650.63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Lu Y., Liu L., Chen Q., Wei J., Cao G., Zhang J. Domestic Radon Exposure and Risk of Childhood Leukemia: A Meta-Analysis. J. Buon. 2020;25:1035–1041. [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Data is unavailable due to privacy.