Abstract
The endoplasmic reticulum (ER) is a cytosolic organelle that plays an essential role in the folding and processing of new secretory proteins, including insulin. The pathogenesis of diabetes, a group of metabolic disorders caused by dysfunctional insulin secretion (Type 1 diabetes, T1DM) or insulin sensitivity (Type 2 diabetes, T2DM), is known to involve the excess accumulation of “poorly folded proteins”, namely, the induction of pathogenic ER stress in pancreatic β-cells. ER stress is known to contribute to the dysfunction of the insulin-producing pancreatic β-cells. T1DM and T2DM are multifactorial diseases, especially T2DM; both environmental and genetic factors are involved in their pathogenesis, making it difficult to create experimental disease models. In recent years, however, the development of induced pluripotent stem cells (iPSCs) and other regenerative technologies has greatly expanded research capabilities, leading to the development of new candidate therapies. In this review, we will discuss the mechanism by which dysregulated ER stress responses contribute to T2DM pathogenesis. Moreover, we describe new treatment methods targeting protein folding and ER stress pathways with a particular focus on pivotal studies of Wolfram syndrome, a monogenic form of syndromic diabetes caused by pathogenic variants in the WFS1 gene, which also leads to ER dysfunction.
Keywords: ER stress, endoplasmic reticulum, β-cell dysfunction, Wolfram syndrome, WFS1, type 2 diabetes
1. Introduction
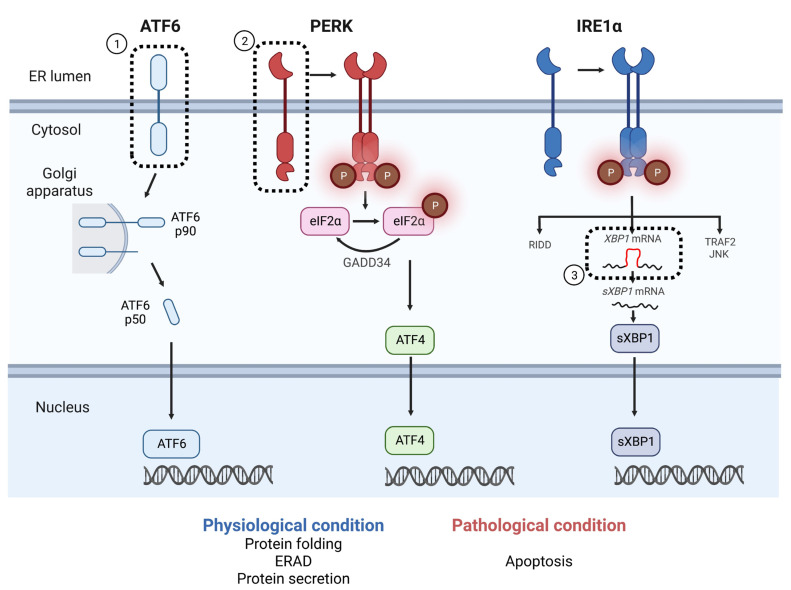
The endoplasmic reticulum (ER) is an intracellular organelle that is known for its role in protein folding, calcium storage, and lipid metabolism. The ER lumen is uniquely equipped to ensure the proper folding and maturation of newly synthesized secretory and transmembrane proteins. Pathological insults that perturb ER homeostasis, such as high protein demand, viral infections, environmental toxins, inflammatory cytokines, decreased ER calcium levels, and mutant protein expression, lead to an accumulation of misfolded and unfolded proteins in the ER lumen, a condition termed ER stress [1]. Cells adaptively counteract ER stress by triggering the unfolded protein response (UPR). The UPR has three major regulators: activating transcription factor 6α (ATF6α), protein kinase R-like ER kinase (PERK), and inositol requiring enzyme 1α (IRE1α) (Figure 1) [2,3]. IRE1α forms oligomers on sensing ER stress, followed by autophosphorylation and acquisition of RNase activity [4]. The activated IRE1α splices the XBP1 intron and induces the expression of the transcription factor, spliced XBP1 (sXBP1) [5,6,7]. Consequently, ER proteins are translocated, folded, and secreted. IRE1α also suppresses protein folding loads by degrading mRNA. This is called regulated IRE1-dependent decay (RIDD) [8]. In addition, IRE1α activates the c-Jun N-terminal kinase (JNK) pathway by binding to TRAF2, which in turn activates the autophagy pathway [9]. PERK can transiently arrest protein synthesis by phosphorylating eukaryotic translation initiation factor 2 subunit α (eIF2α). Phosphorylated eIF2α also induces expression of the transcription factor ATF4 [10], which regulates redox homeostasis, amino acid metabolism, protein synthesis, apoptosis, and autophagy. ATF6α (p90) is transported from the ER to the Golgi apparatus upon sensing ER stress and cleaved [11]. Cleaved ATF6α (p50) translocates to the nucleus and acts as a transcription factor, regulating the transcription of ER chaperones.
Figure 1.
UPR mechanism and the reported SNPs associated with T2DM. The three major regulators (ATF6α, PERK, and IRE1α) are involved in the UPR pathway under pathophysiological conditions. On sensing ER stress, ATF6α (p90) is cleaved at the Golgi apparatus. Cleaved ATF6α (p50) translocates to the nucleus and acts as a transcription factor. PERK arrests protein synthesis by phosphorylating eukaryotic translation initiation factor 2 subunit α (eIF2α). Phosphorylated eIF2α also induces the expression of ATF4, which regulates protein synthesis, apoptosis, and autophagy. IRE1α forms oligomers and autophosphorylates on sensing ER stress, followed by the acquisition of RNase activity. GADD33 dephosphorylates eIF2α and restores protein synthesis. The activated IRE1α splices the XBP1 intron and induces the expression of the transcription factor, spliced XBP1 (sXBP1). IRE1α also suppresses protein folding loads by degrading mRNA, called regulated IRE1-dependent decay (RIDD). In addition, IRE1α activates the TRAF2/JNK pathway followed by the activation of the autophagy pathway. ①–③: The molecules whose SNPs have been reported to contribute to the development of T2DM or insulin resistance include the following. ① ATF6 SNPs, detected in a population of Pima Indians and Dutch Caucasians [12,13]; ② PERK SNPs, detected in Han Chinese populations correlated with increased risk of prediabetes, elevated BMI, and insulin resistance [14]; ③ XBP1 SNPs, reported at high frequency in Han Chinese populations with T2DM and impaired glucose regulation [14,15]—ATF6α (or ATF4), activating transcription factor 6α (or 4); PERK, protein kinase R-like ER kinase; inositol requiring enzyme 1α (IRE1α); ERAD, ER-associated degradation.
The UPR acts as a binary switch. Under physiological conditions, UPR-regulated survival mechanisms outweigh death mechanisms, and proper protein folding in the ER is achieved, including the folding of proinsulin in pancreatic β-cells. Under pathological conditions, the death factors regulated by the UPR pathway such as C/EBP-homologous protein (CHOP) outplay its survival pathways, leading to cell death. This process is called pathological or terminal UPR [16], and sterile inflammation induced by thioredoxin-interacting protein (TXNIP) plays a critical role in this process [17,18]. Given the many vital and complex functions of the ER, ER failure can trigger a range of diseases. Dysregulation of ER homeostasis underlies pancreatic β-cell dysfunction in type 1 diabetes (T1DM) and type 2 diabetes (T2DM) as well as in monogenic forms of diabetes, including Wolfram syndrome.
In this review, we discuss (1) the relationship between ER stress and pancreatic β-cell dysfunction in diabetes, (2) UPR-related gene variants as risk factors for the development of T2DM, (3) the role of the WFS1 gene and its contribution to the development of Wolfram syndrome and T2DM, and (4) the opportunities for clinical application of ER stress reducers and induced pluripotent stem cell (iPSC)-based treatment against T2DM and monogenic forms of diabetes.
2. ER Stress in Diabetes and Pancreatic β-Cell Dysfunction
T1DM is a chronic autoimmune-mediated disease in which pancreatic β-cells are targeted [19,20]. Inflammation localized to pancreatic islets (insulitis) followed by pancreatic β-cell disruption, makes β-cells unable to produce insulin [19], which is necessary to normalize blood sugar levels. In pre-diabetic non-obese diabetic (NOD) mice, an accepted mouse model of T1DM, ER stress markers are augmented in islets [21], suggesting a close relationship between ER stress and the development of T1DM. In addition, immune cells recognize post-translational modifications on β-cell proteins, which are added by enzymes activated under conditions of high cytosolic Ca2+ concentrations and ER stress [22]. Furthermore, in a virus-induced diabetes rat model, IRE1α, one of the main regulators of the UPR pathway, is activated before the initiation of insulitis [23]. Knocking out IRE1α in NOD mice leads to pancreatic β-cell dedifferentiation prior to insulitis, and these dedifferentiated pancreatic β-cells evade autoimmune destruction [24]. As supporting evidence, the expression level of CHOP, the molecule that mediates ER stress-induced apoptosis, is increased in the islets of T1DM patients [25], and the proinsulin to C-peptide ratio, used as a biomarker of ER stress, is elevated in new-onset T1DM children [26]. Altogether, these data point to ER stress as an essential and early driver of autoimmune-mediated T1DM in both rodent models and human patients.
T2DM is characterized by insulin resistance and hyperinsulinemia followed by pancreatic β-cell dysfunction and death. Obesity is a major risk factor for the development of T2DM. Dietary- or genetic-induced obesity in mice is known to cause ER stress, which induces IRE1α-dependent activation of JNK. Activated JNK phosphorylates serine 307 of insulin receptor substrate 1 (IRS-1), a substrate for the insulin receptor tyrosine kinase. In turn, the phosphorylation level of Akt, the distal cascade in insulin signaling, is suppressed, and peripheral insulin resistance develops [27]. Insulin synthesis is thought to be enhanced in pancreatic β-cells in the early stages of T2DM to compensate for insulin resistance. Consequently, proinsulin misfolding occurs also in the early stage of T2DM pancreatic β-cells [28]. It has been reported that serum ER stress markers are high in patients with T2DM [29]. Pancreatic β-cell dysfunction is caused by different mechanisms in T1DM, which is attributable to autoimmunity, and in T2DM, which is known to be induced by insulin resistance. However, the excess ER stress in pancreatic β-cell or peripheral tissue is involved in the pathogenesis of both T1DM and T2DM.
3. Genetic Risk Factors for T2DM: Findings from GWAS and the Involvement of UPR Genes
T2DM is a multifactorial disease caused by both genetic and environmental factors. Therefore, it is difficult to identify the risk of developing T2DM based only on genetic factors. However, examining genetic risk factors and the molecular mechanisms by which such mutations induce pathological changes that contribute to the development of T2DM aids in our understanding of the heterogeneous nature and pathogenesis of T2DM. Genome-wide association studies (GWAS) provide a method to search for genetic predispositions for diseases using genome-wide single nucleotide polymorphism (SNP) arrays. Large-scale GWAS approaches gathered data from multiple populations and identified more than 500 SNPs associated with T2DM development [30,31]. Interestingly, SNPs occurred in genes that transcribe proteins related to the UPR. In this section, we will first discuss how the dysfunction of molecules that compose the UPR is involved in the pathogenesis of diabetes. Next, we will discuss the association between T2DM and SNPs in UPR-related genes identified by GWAS.
3.1. Monogenic and Syndromic Diabetes Derived from UPR Impairment
The variants identified in UPR-related genes known to cause monogenic diabetes are listed in Table 1. Walcott–Rallison syndrome (WRS, OMIM: 226980) is a relatively well-known disease associated with dysfunction of the PERK (EIF2AK3) gene [32,33,34]. In addition to the development of diabetes in early infancy, WRS has a variety of manifestations, such as skeletal phenotypes (multiple epiphyseal dysplasia, multiple epiphyseal-metaphyseal dysplasia), hepatic dysfunction, and impaired renal function [33,35]. There are no reported cases of diabetes due to pathogenic variants in the IRE1 (ERN1) gene; however, IRE1α knockdown in C57BL/6 mice induces diabetes via a mechanism that is independent of autoimmune-mediated β-cell death [36,37]. Mesencephalic astrocyte-derived neurotrophic factor (MANF) encodes a secretory neurotrophic factor, originally discovered in astrocytes, that localizes to the ER and indirectly regulates the UPR. ER calcium depletion and ER stress increase MANF expression and secretion [38,39,40,41]. ER stress is elevated in the pancreatic β cells of Manf-knockout mice, resulting in ER stress-induced cell death and diabetes [42]. Recently, childhood-onset diabetes and a neurodevelopmental delay caused by the MANF gene variant have been reported [43]. Those gene disruptions strongly indicate a relationship between abnormalities in the UPR and dysfunction of pancreatic β-cells. There is no clear explanation for the differences in symptoms caused by functional mutations in UPR-related genes. Differences in the expression levels between tissues or the role of each molecule in pancreatic β-cells are speculated to influence the development of diabetes.
Table 1.
Genetic Variants in UPR Pathways that are Related to Diabetes.
| UPR Pathway | Gene | Protein | Syndrome | Symptoms Other than Diabetes |
|---|---|---|---|---|
| PERK | EIF2AK3 [32,33,34] | PERK | Walcott–Rallison syndrome | Liver dysfunction Skeletal dysplasia |
| EIF2S3 [44,45] | eIF2 γ subunit | Mental retardation, epileptic seizures, hypogonadism, Hypogenitalism, microcephaly, and obesity (MEHMO) syndrome | Mental retardation Epilepsy Hypogonadism/hypogenitalism Microcephaly and obesity |
|
| EIF2B1 [46] | eIF2B | Liver dysfunction | ||
| DNAJC3 [47] | p58IPK | Ataxia, combined cerebellar and peripheral, with hearing loss, and diabetes mellitus (ACPHD) | Combined cerebellar Afferent ataxia Mild upper motor neuron damage Peripheral neuropathy Sensorineural hearing loss |
|
| PPP1R15B [48] | CReP | Short stature Intellectual disability Microcephaly |
||
| IRE1 | ERN1 [36,37] #1 | IRE1α | ||
| Other | WFS1 [49] | Wolframin | Wolfram syndrome, Wolfram syndrome-related disorder | Optic nerve atrophy, hearing loss, diabetes insipidus, neurodegeneration |
| IER3IP1 [50,51] | IER3IP1 | Microcephaly with simplified gyration, epilepsy, and permanent neonatal diabetes syndrome (MEDS) | Microcephaly Epilepsy |
|
| CISD2 [52] | ERIS | Wolfram syndrome (type 2) | Upper gastrointestinal ulceration and bleeding | |
| MANF [42,43] | MANF | Short stature, hearing loss, developmental delay, microcephaly | ||
| CREBRF [53] #2 | CREB3 | |||
| YIPF5 [54] | YIPF5 | Microcephaly Epilepsy |
||
| TANGO1 [55] | TANGO1 | Dentinogenesis imperfecta Short stature Skeletal abnormalities |
||
| SIL1 [56] #1 | SIL1 | Marinesco–Sjögren syndrome | Early-onset cerebellar ataxia Short stature |
|
| ERDJ4 [57] #1 | ERDJ4 |
#1 Diabetic phenotype was reported only in mouse; #2 Decreased the risk of T2DM in Samoans.
3.2. SNPs in UPR-Related Genes and Their Contribution to the Development of T2DM
SNPs in UPR-related genes are thought to increase the risk for T2DM development. The GWAS studies have identified SNPs in ATF6, the gene with no previously reported diabetes-associated pathogenic variants [58,59] (Figure 1 ①). For example, a study in a population of Pima Indians reported that SNPs in ATF6 affect T2DM development and serum insulin levels in the oral glucose tolerance test (OGTT) [12]. Studies in Dutch Caucasian populations also reported that ATF6 SNPs are significantly associated with glucose intolerance and the development of T2DM [13]. On the other hand, studies in Caucasian, African American, and Han Chinese populations have shown that SNPs in ATF6 did not significantly impact the development of T2DM [60,61]. There are studies on Han Chinese populations indicating that PERK SNPs are correlated with an increased risk of prediabetes, elevated BMI, and insulin resistance [14] (Figure 1 ②). A recent GWAS focused on Han Chinese populations reported a high frequency of XBP1 SNPs in the T2DM patient group and impaired glucose regulation indicated by the OGTT [14,15] (Figure 1 ③). However, there are no other reports showing that SNPs in PERK-related genes affect the development of T2DM. The association between UPR dysfunction and the development of diabetes is evident; however, it remains unclear why the risk of developing T2DM differs by SNPs in UPR-related genes and race.
3.3. Wolfram Syndrome and WFS1-Related Disorders: A Prototype of ER-Related Disease
We have focused on the role of ATF6, PERK, and IRE1α as UPR-related genes responsible for monogenic diabetes or disease susceptibility in T2DM. In this section, we will describe the genetic basis for Wolfram syndrome, a well-recognized prototype of ER stress-related disease, and the relationship between its causative gene, WFS1 and T2DM risk.
Wolfram syndrome (OMIM: 222300) is a rare autosomal recessive disorder characterized by insulin-dependent diabetes, optic nerve atrophy, diabetes insipidus, hearing loss, and neurodegeneration [62,63]. Diabetes in Wolfram syndrome presents in patients around six years of age, and optic nerve atrophy manifests around age ten. Diabetes insipidus, neurogenic bladder, obstructive sleep apnea, and deafness may also develop in the next two decades of life, along with brainstem symptoms and cerebellar atrophy, such as dysphagia, ataxia, and central sleep apnea [62,64,65,66,67,68].
Wolfram syndrome is caused by recessive pathogenic variants in the Wolfram syndrome 1 (WFS1) gene [49]. Approximately 200 WFS1 pathogenic variants have been identified in Wolfram syndrome. The WFS1 gene encodes WFS1 (also known as wolframin), a protein localized to the ER membrane that interacts with ATF6 to regulate the UPR during ER stress [69,70]. WFS1 also plays a critical role in maintaining ER and cytosolic Ca2+ homeostasis [1,71], regulating sterile inflammation [72], and insulin granule acidification in pancreatic β-cells [73]. Recently, it has been shown that WFS1 functions as a vesicular cargo protein receptor for exporting secretory proteins from ER [74].
Wolfram syndrome has been recognized as a spectrum disorder since recessive WFS1 variants can vary in clinical severity [75]. In contrast, some dominant de novo WFS1 variants are known to cause severe neonatal-onset diabetes, congenital sensorineural hearing loss, congenital cataracts, and developmental delays, which are known as WFS1-related disorders [75,76]. There are also known WFS1-related disorders characterized by developing only one or few symptoms seen in Wolfram syndrome. For example, a dominant WFS1 p.Trp314Arg variant causes mild diabetes in an autosomal dominant fashion [77]. In all cases, however, pathogenic WFS1 variants cause pathological ER stress in pancreatic β-cells, neurons, retinal ganglion cells, and oligodendrocytes, leading to dysfunction and degeneration of affected tissues [69,70,78,79,80]. Wolfram syndrome and WFS1-related disorder, which result from pathogenic ER stress caused by a single gene abnormality, have been well-recognized as a prototype of human ER-related disease [70,81].
WFS1 variants are typically associated with and discussed as the drivers of Wolfram syndrome; however, WFS1 variants have additionally been identified as risk factors for the development of T2DM. Variants with a reported increased risk for T2DM include rs10010131, rs734312, rs6446482, and the c.1672C > T, p.Arg558Cys missense variant [82,83,84]. The WFS1 p.Arg558Cys missense variant is associated with mild syndromic manifestations of Wolfram syndrome. Our study in collaboration with Dor Yeshorim (https://doryeshorim.org/, accessed on 1 April 2022) revealed a high WFS1 p.Arg558Cys allele frequency (2.32%) in the Ashkenazi Jewish population [85]. Although WFS1 is the responsible gene for the rare Wolfram syndrome, some of its variants may contribute to the common T2DM development.
4. Therapeutics Targeting ER Stress as Treatments for Wolfram Syndrome and T2DM
WFS1 is a locus of broad interest to Wolfram syndrome or Wolfram syndrome-related disorders, including T2DM. Developing a treatment for Wolfram syndrome may provide clues to finding a new therapeutic approach for T2DM. In this section, we describe treatments for Wolfram syndrome, including pharmacological agents targeting the ER and protein folding components as well as regenerative therapies.
4.1. Pharmacological Agents to Treat Wolfram Syndrome and T2DM
The link between UPR-related genes and increased risk for Wolfram syndrome and T2DM advocates for the use of ER stress-targeting drugs as therapies for these metabolic disorders.
Dantrolene sodium, a US Food and Drug Administration (FDA)-approved drug for malignant hyperthermia and muscle spasm, stabilizes ER calcium by targeting ryanodine receptors. Dantrolene sodium treatment can prevent cell death in neural progenitor cells differentiated from Wolfram syndrome patients induced pluripotent stem cell (iPSC) [80]. We recently conducted the dantrolene sodium phase 1b/2 clinical study on Wolfram syndrome patients (National Clinical Trial (NCT) number, NCT02829268). Pancreatic β-cell function improved in a select few Wolfram syndrome patients, and there was a significant correlation between baseline β-cell function and change in β-cell responsiveness [86]. Although its effects were limited, dantrolene sodium was the first therapeutic candidate compound for Wolfram syndrome found by high-throughput screening. Intracellular Ca2+ stabilization is also a central feature of another Wolfram syndrome therapeutic, the phosphodiesterase 4 (PDE4) inhibitor ibudilast. Ibudilast rescues cell viability and cytosolic Ca2+ homeostasis to improve insulin secretion from pancreatic β-cells in a Wolfram syndrome cellular model [87]. Although the efficacy cannot be compared with that of dantrolene as clinical trials have not yet been conducted, it supports the possibility that Ca2+ stabilizers could be candidate drugs for Wolfram syndrome treatment.
Chemical chaperones, such as 4-phenylbutyric acid (4-PBA) and tauroursodeoxycholic acid (TUDCA), which are known to reduce ER stress by stabilizing protein folding, have been used for treatment in Wolfram syndrome and T2DM models. We recently reported the preclinical study data on a combination of 4-PBA and TUDCA treatment, indicating that these drugs effectively stabilized WFS1 protein and improved patient-derived pancreatic β-cell functionality [85]. Based on these findings, we plan a clinical study with a combination of 4-PBA and TUDCA treatment. A chemical chaperone that enhances ER function can also normalize hyperglycemia and systemic insulin sensitivity and alleviate the increased ER stress in leptin-deficient (ob/ob) mice, a T2DM experimental mouse model [88]. Thus, chaperone-mediated restoration of proper protein folding improves β-cell function and glucose homeostasis in Wolfram syndrome and T2DM.
As noted, shared mechanisms lead to the development of Wolfram syndrome, T1DM, and T2DM. A drug commonly used to treat T2DM, glucagon-like peptide (GLP)-1 receptor agonist, also attenuates diabetes and neurodegeneration in Wolfram syndrome [89,90,91,92,93,94,95]. GLP-1 is an incretin hormone that stimulates insulin secretion after an oral glucose administration. Similar to endogenous GLP-1, the GLP-1 receptor agonist promotes insulin secretion from pancreatic β-cells and inhibits glucagon secretion from pancreatic α-cells. GLP-1 receptor agonist has been reported to attenuate ER stress in pancreatic β-cells and improve insulin secretion in Wolfram syndrome [93,94,95]. Furthermore, surprisingly, the GLP-1 receptor agonist improves cognitive, visual, and auditory function in a rat model of Wolfram syndrome [89,90,92]. GLP-1 receptor agonist is an excellent example of translating therapeutics commonly used in T2DM for the treatment of Wolfram syndrome.
Valproic acid is a therapeutic candidate for the treatment of Wolfram syndrome approached from a completely different perspective. Valproic acid is a drug used to treat bipolar disorder, and we have previously shown that valproic acid enhances the gene expression of WFS1 [96]. In addition, Valproate was shown to enhance the dissociation of WFS1 and GPR94, a component of the UPR involved in protein folding and degradation. Based on these findings, a phase 2 randomized, double-blind clinical trial is ongoing to evaluate the efficacy of sodium valproate as a treatment for Wolfram syndrome patients’ vision loss and neurological symptoms (NCT number, NCT03717909).
Overall, these data indicate that pharmacologic manipulation of UPR may provide a novel therapeutic target not only for Wolfram syndrome but also for T2DM.
4.2. Regenerative Therapy
In recent years, the clinical application of cell replacement therapy in diabetes has been under development. Attempts have been made to restore insulin capacity by regenerating pancreatic β-cells in diabetic patients with impaired function. iPSCs were generated from various diabetic patients, and it became possible to differentiate iPSCs lines into stem cell-derived pancreatic β-cells (SC-β-cells). As a result, several SC-β-cells have been generated from T1DM patients, and studies for clinical application have been underway. The first phase 1/2 study of SC-β-cells transplantation in T1DM patients is ongoing (NCT number, NCT04786262).
SC-β-cells or β-like cells differentiated from patient-derived iPSCs are also available for replacement therapy in Wolfram syndrome [97,98]. We have previously shown that Wolfram syndrome-patient iPSCs have a low differentiation efficiency into SC-β-cells and reduced insulin secretion, whereas corrected SC-β-cells, in which the pathogenic variant of WFS1 was repaired by CRISPR/Cas9, showed marked insulin secretion recovery [97]. In addition, corrected SC-β-cell transplantation recovered normal glucose homeostasis in streptozotocin-induced diabetic mice. Although there are still many challenges to overcome, such as developing transplantation methods, the current availability of genetically engineered SC-β-cells could be a therapeutic breakthrough for monogenic forms of diabetes, such as Wolfram syndrome. Multiple iPSCs have been generated from patients with monogenic diabetes to elucidate the function of genes that, when mutated, increase the risk for the development of T2DM [99]. Although there are no reports describing the generation of SC-β-cells from T2DM patient-derived iPSCs, there are several reports of differentiating iPSCs into β-like cells [100,101,102,103]. With further progress in identifying protective or risk genes for T2DM [100,101,102], we can expect increased utilization of regenerative therapy methods, including transplantation of gene-edited SC-β-cells to T2DM patients.
5. Conclusions and Future Perspectives
Excess ER stress in pancreatic β-cells is involved in the pathogenesis of T2DM, and dysfunction of multiple UPR-related genes, including WFS1, are candidate genetic risk factors for T2DM. Creating an experimental disease model is essential for studying the genetic risk factors for T2DM development; however, the heterogeneity of T2DM makes this task difficult. Since Wolfram syndrome is a monogenic form of diabetes caused by mutations in WFS1, Wolfram syndrome is a prototype for ER stress-related diseases such as T2DM. To understand the genetic background of T2DM, we can utilize the Wolfram syndrome experimental models that have already been established, including SC-β-cells.
In the future, we expect to develop individualized medical care for Wolfram syndrome patients, prescribing effective drugs selected based on their personal WFS1 variant. Moreover, regenerative medicine using SC-β-cells and gene editing technology would make it possible to transplant the patient’s tissue in a “normal” condition. For T2DM, if more risk genes are identified in the future, it would also become possible to develop regenerative therapies.
Acknowledgments
This work was partly supported by the grants from the National Institutes of Health (NIH)/NIDDK (DK132090, DK020579) and philanthropic supports from the Silberman Fund, the Ellie White Foundation for the Rare Genetic Disorders, the Snow Foundation, the Unravel Wolfram Syndrome Fund, the Stowe Fund, the Feiock Fund, the Cachia Fund, the Gildenhorn Fund, the Eye Hope Foundation, the Gadot Fund, Ontario Wolfram League, Associazione Gentian—Sindrome di Wolfram Italia, Alianza de Familias Afectadas por el Sindrome Wolfram Spain, Wolfram syndrome UK, and Association Syndrome de Wolfram France to F. Urano. The authors thank all the members of the Washington University Wolfram Syndrome Study and Research Clinic for their support (https://wolframsyndrome.wustl.edu, accessed on 10 June 2022) and all the participants in the Wolfram syndrome International Registry and Clinical Study, Research Clinic, and Clinical Trials for their time and efforts. We are grateful for the editing assistance provided by InPrint: A Scientific Communication Network at Washington University in St. Louis. The figures were created with BioRender.com.
Author Contributions
S.M. and F.U. both participated in writing, editing, and reviewing the manuscript. All authors have read and agreed to the published version of the manuscript.
Conflicts of Interest
F. Urano is an inventor of three patents related to the treatment of Wolfram syndrome, SOLUBLE MANF IN PANCREATIC BETA CELL DISORDERS (US 9,891,231) and TREATMENT FOR WOLFRAM SYNDROME AND OTHER ER STRESS DISORDERS (US 10,441,574 and US 10,695,324). F. Urano is a Founder and President of CURE4WOLFRAM, INC and the Head of the Scientific Advisory Board of Opris Biotechnologies.
Funding Statement
This work was partly supported by the grants from the National Institutes of Health (NIH)/NIDDK (DK112921, DK020579). S. Morikawa was supported by Manpei Suzuki Diabetes Foundation and Japan Society for the Promotion of Science (JSPS) Overseas Research Fellowships.
Footnotes
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content.
References
- 1.Hara T., Mahadevan J., Kanekura K., Hara M., Lu S., Urano F. Calcium efflux from the endoplasmic reticulum leads to beta-cell death. Endocrinology. 2014;155:758–768. doi: 10.1210/en.2013-1519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Lin J.H., Li H., Yasumura D., Cohen H.R., Zhang C., Panning B., Shokat K.M., Lavail M.M., Walter P. IRE1 signaling affects cell fate during the unfolded protein response. Science. 2007;318:944–949. doi: 10.1126/science.1146361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Han D., Lerner A.G., Vande Walle L., Upton J.P., Xu W., Hagen A., Backes B.J., Oakes S.A., Papa F.R. RE1alpha kinase activation modes control alternate endoribonuclease outputs to determine divergent cell fates. Cell. 2009;138:562–575. doi: 10.1016/j.cell.2009.07.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Credle J.J., Finer-Moore J.S., Papa F.R., Stroud R.M., Walter P. On the mechanism of sensing unfolded protein in the endoplasmic reticulum. Proc. Natl. Acad. Sci. USA. 2005;102:18773–18784. doi: 10.1073/pnas.0509487102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Shen X., Ellis R.E., Lee K., Liu C.-Y., Yang K., Solomon A., Yoshida H., Morimoto R., Kurnit D.M., Mori K., et al. Complementary Signaling Pathways Regulate the Unfolded Protein Response and Are Required for C. elegans Development. Cell. 2001;107:893–903. doi: 10.1016/S0092-8674(01)00612-2. [DOI] [PubMed] [Google Scholar]
- 6.Calfon M., Zeng H., Urano F., Till J.H., Hubbard S.R., Harding H.P., Clark S.G., Ron D. IRE1 couples endoplasmic reticulum load to secretory capacity by processing the XBP-1 mRNA. Nature. 2002;415:92–96. doi: 10.1038/415092a. [DOI] [PubMed] [Google Scholar]
- 7.Yoshida H., Matsui T., Yamamoto A., Okada T., Mori K. XBP1 mRNA Is Induced by ATF6 and Spliced by IRE1 in Response to ER Stress to Produce a Highly Active Transcription Factor. Cell. 2001;107:881–891. doi: 10.1016/S0092-8674(01)00611-0. [DOI] [PubMed] [Google Scholar]
- 8.Hollien J., Weissman J.S. Decay of endoplasmic reticulum-localized mRNAs during the unfolded protein response. Science. 2006;313:104–107. doi: 10.1126/science.1129631. [DOI] [PubMed] [Google Scholar]
- 9.Castillo K., Rojas-Rivera D., Lisbona F., Caballero B., Nassif M., Court F.A., Schuck S., Ibar C., Walter P., Sierralta J., et al. BAX inhibitor-1 regulates autophagy by controlling the IRE1alpha branch of the unfolded protein response. EMBO J. 2011;30:4465–4478. doi: 10.1038/emboj.2011.318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Vattem K.M., Wek R.C. Reinitiation involving upstream ORFs regulates ATF4 mRNA translation in mammalian cells. Proc. Natl. Acad. Sci. USA. 2004;101:11269–11274. doi: 10.1073/pnas.0400541101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Haze K., Yoshida H., Yanagi H., Yura T., Mori K. Mammalian transcription factor ATF6 is synthesized as a transmembrane protein and activated by proteolysis in response to endoplasmic reticulum stress. Mol. Biol. Cell. 1999;10:3787–3799. doi: 10.1091/mbc.10.11.3787. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Thameem F., Farook V.S., Bogardus C., Prochazka M. Association of amino acid variants in the activating transcription factor 6 gene (ATF6) on 1q21-q23 with type 2 diabetes in Pima Indians. Diabetes. 2006;55:839–842. doi: 10.2337/diabetes.55.03.06.db05-1002. [DOI] [PubMed] [Google Scholar]
- 13.Meex S.J., van Greevenbroek M.M., Ayoubi T.A., Vlietinck R., van Vliet-Ostaptchouk J.V., Hofker M.H., Vermeulen V.M.M.-J., Schalkwijk C.G., Feskens E.J.M., Boer J.M.A., et al. Activating transcription factor 6 polymorphisms and haplotypes are associated with impaired glucose homeostasis and type 2 diabetes in Dutch Caucasians. J. Clin. Endocrinol. Metab. 2007;92:2720–2725. doi: 10.1210/jc.2006-2280. [DOI] [PubMed] [Google Scholar]
- 14.Feng N., Ma X., Wei X., Zhang J., Dong A., Jin M., Zhang H. Common variants in PERK, JNK, BIP and XBP1 genes are associated with the risk of prediabetes or diabetes-related phenotypes in a Chinese population. Chin. Med. J. 2014;127:2438–2444. [PubMed] [Google Scholar]
- 15.Liu S., Ma G., Yao S., Chen Z., Wang C., Zhao B., Li K. Polymorphism—116C/G of the human X box binding protein 1 gene is associated with risk of type 2 diabetes in a Chinese Han population. Gene. 2016;575:71–74. doi: 10.1016/j.gene.2015.08.037. [DOI] [PubMed] [Google Scholar]
- 16.Anthony T.G., Wek R.C. TXNIP switches tracks toward a terminal UPR. Cell Metab. 2012;16:135–137. doi: 10.1016/j.cmet.2012.07.012. [DOI] [PubMed] [Google Scholar]
- 17.Oslowski C.M., Hara T., O’Sullivan-Murphy B., Kanekura K., Lu S., Hara M., Ishigaki S., Zhu L.J., Hayashi E., Hui S.T., et al. Thioredoxin-interacting protein mediates ER stress-induced beta cell death through initiation of the inflammasome. Cell Metab. 2012;16:265–273. doi: 10.1016/j.cmet.2012.07.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Lerner A.G., Upton J.P., Praveen P.V., Ghosh R., Nakagawa Y., Igbaria A., Shen S., Nguyen V., Backes B.J., Heiman M., et al. IRE1alpha induces thioredoxin-interacting protein to activate the NLRP3 inflammasome and promote programmed cell death under irremediable ER stress. Cell Metab. 2012;16:250–264. doi: 10.1016/j.cmet.2012.07.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Gepts W. Pathologic anatomy of the pancreas in juvenile diabetes mellitus. Diabetes. 1965;14:619–633. doi: 10.2337/diab.14.10.619. [DOI] [PubMed] [Google Scholar]
- 20.Bottazzo G.F., Dean B.M., McNally J.M., MacKay E.H., Swift P.G., Gamble D.R. In situ characterization of autoimmune phenomena and expression of HLA molecules in the pancreas in diabetic insulitis. N. Engl. J. Med. 1985;313:353–360. doi: 10.1056/NEJM198508083130604. [DOI] [PubMed] [Google Scholar]
- 21.Tersey S.A., Nishiki Y., Templin A.T., Cabrera S.M., Stull N.D., Colvin S.C., Evans-Molina C., Rickus J.L., Maier B., Mirmira R.G., et al. Islet beta-cell endoplasmic reticulum stress precedes the onset of type 1 diabetes in the nonobese diabetic mouse model. Diabetes. 2012;61:818–827. doi: 10.2337/db11-1293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Marre M.L., Profozich J.L., Coneybeer J.T., Geng X., Bertera S., Ford M.J., Trucco M., Piganelli J.D. Inherent ER stress in pancreatic islet beta cells causes self-recognition by autoreactive T cells in type 1 diabetes. J. Autoimmun. 2016;72:33–46. doi: 10.1016/j.jaut.2016.04.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Yang C., Diiorio P., Jurczyk A., O’Sullivan-Murphy B., Urano F., Bortell R. Pathological endoplasmic reticulum stress mediated by the IRE1 pathway contributes to pre-insulitic beta cell apoptosis in a virus-induced rat model of type 1 diabetes. Diabetologia. 2013;56:2638–2646. doi: 10.1007/s00125-013-3044-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Lee H., Lee Y.S., Harenda Q., Pietrzak S., Oktay H.Z., Schreiber S., Liao Y., Sonthalia S., Ciecko A.E., Chen Y.-G., et al. Beta Cell Dedifferentiation Induced by IRE1alpha Deletion Prevents Type 1 Diabetes. Cell Metab. 2020;31:822–836.e5. doi: 10.1016/j.cmet.2020.03.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Marhfour I., Lopez X.M., Lefkaditis D., Salmon I., Allagnat F., Richardson S.J., Morgan N.G., Eizirik D.L. Expression of endoplasmic reticulum stress markers in the islets of patients with type 1 diabetes. Diabetologia. 2012;55:2417–2420. doi: 10.1007/s00125-012-2604-3. [DOI] [PubMed] [Google Scholar]
- 26.Freese J., Al-Rawi R., Choat H., Martin A., Lunsford A., Tse H., Mick G., McCormick K. Proinsulin to C-Peptide Ratio in the First Year after Diagnosis of Type 1 Diabetes. J. Clin. Endocrinol. Metab. 2021;106:e4318–e4326. doi: 10.1210/clinem/dgab463. [DOI] [PubMed] [Google Scholar]
- 27.Ozcan U., Cao Q., Yilmaz E., Lee A.H., Iwakoshi N.N., Ozdelen E., Tuncman G., Gorgun C., Glimcher L.H., Hotamisligil G.S. Endoplasmic reticulum stress links obesity, insulin action, and type 2 diabetes. Science. 2004;306:457–461. doi: 10.1126/science.1103160. [DOI] [PubMed] [Google Scholar]
- 28.Arunagiri A., Haataja L., Pottekat A., Pamenan F., Kim S., Zeltser L.M., Paton A.W., Paton J.C., Tsai B., Itkin-Ansari P., et al. Proinsulin misfolding is an early event in the progression to type 2 diabetes. Elife. 2019;8:e44532. doi: 10.7554/eLife.44532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Nourbakhsh M., Sharifi R., Heydari N., Nourbakhsh M., Ezzati-Mobasser S., Zarrinnahad H. Circulating TRB3 and GRP78 levels in type 2 diabetes patients: Crosstalk between glucose homeostasis and endoplasmic reticulum stress. J. Endocrinol. Investig. 2022;45:649–655. doi: 10.1007/s40618-021-01683-5. [DOI] [PubMed] [Google Scholar]
- 30.Cho Y.S., Chen C.H., Hu C., Long J., Ong R.T., Sim X., Takeuchi F., Wu Y., Go M.J., Yamauchi T., et al. Meta-analysis of genome-wide association studies identifies eight new loci for type 2 diabetes in east Asians. Nat. Genet. 2011;44:67–72. doi: 10.1038/ng.1019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Vujkovic M., Keaton J.M., Lynch J.A., Miller D.R., Zhou J., Tcheandjieu C., Huffman J.E., Assimes T.L., Lorenz K., Zhu X., et al. Discovery of 318 new risk loci for type 2 diabetes and related vascular outcomes among 1.4 million participants in a multi-ancestry meta-analysis. Nat. Genet. 2020;52:680–691. doi: 10.1038/s41588-020-0637-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Zhang P., McGrath B., Li S., Frank A., Zambito F., Reinert J., Gannon M., Ma K., McNaughton K., Cavener D.R. The PERK eukaryotic initiation factor 2 alpha kinase is required for the development of the skeletal system, postnatal growth, and the function and viability of the pancreas. Mol. Cell Biol. 2002;22:3864–3874. doi: 10.1128/MCB.22.11.3864-3874.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Stoss H., Pesch H.J., Pontz B., Otten A., Spranger J. Wolcott-Rallison syndrome: Diabetes mellitus and spondyloepiphyseal dysplasia. Eur. J. Pediatr. 1982;138:120–129. doi: 10.1007/BF00441137. [DOI] [PubMed] [Google Scholar]
- 34.Delepine M., Nicolino M., Barrett T., Golamaully M., Lathrop G.M., Julier C. EIF2AK3, encoding translation initiation factor 2-alpha kinase 3, is mutated in patients with Wolcott-Rallison syndrome. Nat. Genet. 2000;25:406–409. doi: 10.1038/78085. [DOI] [PubMed] [Google Scholar]
- 35.Wolcott C.D., Rallison M.L. Infancy-onset diabetes mellitus and multiple epiphyseal dysplasia. J. Pediatr. 1972;80:292–297. doi: 10.1016/S0022-3476(72)80596-1. [DOI] [PubMed] [Google Scholar]
- 36.Hassler J.R., Scheuner D.L., Wang S., Han J., Kodali V.K., Li P., Nguyen J., George J.S., Davis C., Wu S.P., et al. The IRE1alpha/XBP1s Pathway Is Essential for the Glucose Response and Protection of beta Cells. PLoS Biol. 2015;13:e1002277. doi: 10.1371/journal.pbio.1002277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Tsuchiya Y., Saito M., Kadokura H., Miyazaki J.I., Tashiro F., Imagawa Y., Iwawaki T., Kohno K. IRE1-XBP1 pathway regulates oxidative proinsulin folding in pancreatic beta cells. J. Cell Biol. 2018;217:1287–1301. doi: 10.1083/jcb.201707143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Henderson M.J., Richie C.T., Airavaara M., Wang Y., Harvey B.K. Mesencephalic astrocyte-derived neurotrophic factor (MANF) secretion and cell surface binding are modulated by KDEL receptors. J. Biol. Chem. 2013;288:4209–4225. doi: 10.1074/jbc.M112.400648. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Glembotski C.C., Thuerauf D.J., Huang C., Vekich J.A., Gottlieb R.A., Doroudgar S. Mesencephalic astrocyte-derived neurotrophic factor protects the heart from ischemic damage and is selectively secreted upon sarco/endoplasmic reticulum calcium depletion. J. Biol. Chem. 2012;287:25893–25904. doi: 10.1074/jbc.M112.356345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Mizobuchi N., Hoseki J., Kubota H., Toyokuni S., Nozaki J., Naitoh M., Koizumi A., Nagata K. ARMET is a soluble ER protein induced by the unfolded protein response via ERSE-II element. Cell Struct Funct. 2007;32:41–50. doi: 10.1247/csf.07001. [DOI] [PubMed] [Google Scholar]
- 41.Petrova P., Raibekas A., Pevsner J., Vigo N., Anafi M., Moore M.K., Peaire A.E., Shridhar V., Smith D.I., Kelly J., et al. MANF: A new mesencephalic, astrocyte-derived neurotrophic factor with selectivity for dopaminergic neurons. J. Mol. Neurosci. 2003;20:173–188. doi: 10.1385/JMN:20:2:173. [DOI] [PubMed] [Google Scholar]
- 42.Lindahl M., Danilova T., Palm E., Lindholm P., Voikar V., Hakonen E., Ustinov J., Andressoo J.-O., Harvey B.K., Otonkoski T., et al. MANF is indispensable for the proliferation and survival of pancreatic beta cells. Cell Rep. 2014;7:366–375. doi: 10.1016/j.celrep.2014.03.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Montaser H., Patel K.A., Balboa D., Ibrahim H., Lithovius V., Naatanen A., Chandra V., Demir K., Acar S., Ben-Omran T., et al. Loss of MANF Causes Childhood-Onset Syndromic Diabetes Due to Increased Endoplasmic Reticulum Stress. Diabetes. 2021;70:1006–1018. doi: 10.2337/db20-1174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Steinmuller R., Steinberger D., Muller U. MEHMO (mental retardation, epileptic seizures, hypogonadism and -genitalism, microcephaly, obesity), a novel syndrome: Assignment of disease locus to Xp21.1-p22.13. Eur. J. Hum. Genet. 1998;6:201–206. doi: 10.1038/sj.ejhg.5200180. [DOI] [PubMed] [Google Scholar]
- 45.Borck G., Shin B.S., Stiller B., Mimouni-Bloch A., Thiele H., Kim J.R., Thakur M., Skinner C., Aschenbach L., Smirin-Yosef P., et al. eIF2gamma mutation that disrupts eIF2 complex integrity links intellectual disability to impaired translation initiation. Mol. Cell. 2012;48:641–646. doi: 10.1016/j.molcel.2012.09.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.De Franco E., Caswell R., Johnson M.B., Wakeling M.N., Zung A., Dung V.C., Ng B., Goonetilleke R., Jury M.V., El-Khateeb M., et al. De Novo Mutations in EIF2B1 Affecting eIF2 Signaling Cause Neonatal/Early-Onset Diabetes and Transient Hepatic Dysfunction. Diabetes. 2020;69:477–483. doi: 10.2337/db19-1029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Synofzik M., Haack T.B., Kopajtich R., Gorza M., Rapaport D., Greiner M., Schönfeld C., Freiberg C., Schorr S., Holl R.W., et al. Absence of BiP co-chaperone DNAJC3 causes diabetes mellitus and multisystemic neurodegeneration. Am. J. Hum. Genet. 2014;95:689–697. doi: 10.1016/j.ajhg.2014.10.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Abdulkarim B., Nicolino M., Igoillo-Esteve M., Daures M., Romero S., Philippi A., Senée V., Lopes M., Cunha D.A., Harding H.P., et al. A Missense Mutation in PPP1R15B Causes a Syndrome Including Diabetes, Short Stature, and Microcephaly. Diabetes. 2015;64:3951–3962. doi: 10.2337/db15-0477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Inoue H., Tanizawa Y., Wasson J., Behn P., Kalidas K., Bernal-Mizrachi E., Mueckler M., Marshall H., Donis-Keller H., Crock P., et al. A gene encoding a transmembrane protein is mutated in patients with diabetes mellitus and optic atrophy (Wolfram syndrome) Nat. Genet. 1998;20:143–148. doi: 10.1038/2441. [DOI] [PubMed] [Google Scholar]
- 50.Abdel-Salam G.M., Schaffer A.E., Zaki M.S., Dixon-Salazar T., Mostafa I.S., Afifi H.H., Gleeson J.G. A homozygous IER3IP1 mutation causes microcephaly with simplified gyral pattern, epilepsy, and permanent neonatal diabetes syndrome (MEDS) Am. J. Med. Genet. Part A. 2012;158A:2788–2796. doi: 10.1002/ajmg.a.35583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Poulton C.J., Schot R., Kia S.K., Jones M., Verheijen F.W., Venselaar H., Wit M.-C.Y., Graaff E., Bertoli-Avella A.M., Mancini G.M.S. Microcephaly with simplified gyration, epilepsy, and infantile diabetes linked to inappropriate apoptosis of neural progenitors. Am. J. Hum. Genet. 2011;89:265–276. doi: 10.1016/j.ajhg.2011.07.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Amr S., Heisey C., Zhang M., Xia X.J., Shows K.H., Ajlouni K., Pandya A., Satin L.S., El-Shanti H., Shiang R. A homozygous mutation in a novel zinc-finger protein, ERIS, is responsible for Wolfram syndrome 2. Am. J. Hum. Genet. 2007;81:673–683. doi: 10.1086/520961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Minster R.L., Hawley N.L., Su C.T., Sun G., Kershaw E.E., Cheng H., Buhule O., Lin J., Reupena S., Viali S., et al. A thrifty variant in CREBRF strongly influences body mass index in Samoans. Nat. Genet. 2016;48:1049–1054. doi: 10.1038/ng.3620. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.De Franco E., Lytrivi M., Ibrahim H., Montaser H., Wakeling M.N., Fantuzzi F., Patel K., Demarez C., Cai Y., Igoillo-Esteve M., et al. YIPF5 mutations cause neonatal diabetes and microcephaly through endoplasmic reticulum stress. J. Clin. Investig. 2020;130:6338–6353. doi: 10.1172/JCI141455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Lekszas C., Foresti O., Raote I., Liedtke D., Konig E.M., Nanda I., Vona B., De Coster P., Cauwels R., Malhotr V., et al. Biallelic TANGO1 mutations cause a novel syndromal disease due to hampered cellular collagen secretion. Elife. 2020;9:e51319. doi: 10.7554/eLife.51319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Ittner A.A., Bertz J., Chan T.Y., van Eersel J., Polly P., Ittner L.M. The nucleotide exchange factor SIL1 is required for glucose-stimulated insulin secretion from mouse pancreatic beta cells in vivo. Diabetologia. 2014;57:1410–1419. doi: 10.1007/s00125-014-3230-z. [DOI] [PubMed] [Google Scholar]
- 57.Fritz J.M., Dong M., Apsley K.S., Martin E.P., Na C.L., Sitaraman S., Weaver T.E. Deficiency of the BiP cochaperone ERdj4 causes constitutive endoplasmic reticulum stress and metabolic defects. Mol. Biol. Cell. 2014;25:431–440. doi: 10.1091/mbc.e13-06-0319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Kohl S., Zobor D., Chiang W.C., Weisschuh N., Staller J., Gonzalez Menendez I., Chang S., Beck S.C., Garrido M.G., Sothilingam V., et al. Mutations in the unfolded protein response regulator ATF6 cause the cone dysfunction disorder achromatopsia. Nat. Genet. 2015;47:757–765. doi: 10.1038/ng.3319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Ansar M., Santos-Cortez R.L., Saqib M.A., Zulfiqar F., Lee K., Ashraf N.M., Ullah E., Wang X., Sajid S., Khan F.S., et al. Mutation of ATF6 causes autosomal recessive achromatopsia. Hum. Genet. 2015;134:941–950. doi: 10.1007/s00439-015-1571-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Chu W.S., Das S.K., Wang H., Chan J.C., Deloukas P., Froguel P., Baier L.J., Jia W., McCarthy M.I., Ng M.C.Y., et al. Activating Transcription Factor 6 (ATF6) Sequence Polymorphisms in Type 2 Diabetes and Pre-Diabetic Traits. Diabetes. 2007;56:856–862. doi: 10.2337/db06-1305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Hu C., Zhang R., Wang C., Ma X., Wang J., Bao Y., Xiang K., Jia W. Lack of association between genetic polymorphisms within DUSP12—ATF6 locus and glucose metabolism related traits in a Chinese population. BMC Med. Genet. 2011;12:3. doi: 10.1186/1471-2350-12-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Barrett T.G., Bundey S.E. Wolfram (DIDMOAD) syndrome. J. Med. Genet. 1997;34:838–841. doi: 10.1136/jmg.34.10.838. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Barrett T.G., Bundey S.E., Macleod A.F. Neurodegeneration and diabetes: UK nationwide study of Wolfram (DIDMOAD) syndrome. Lancet. 1995;346:1458–1463. doi: 10.1016/S0140-6736(95)92473-6. [DOI] [PubMed] [Google Scholar]
- 64.Urano F. Wolfram Syndrome: Diagnosis, Management, and Treatment. Curr. Diab. Rep. 2016;16:6. doi: 10.1007/s11892-015-0702-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Wolfram D.J., Wagener H.P. Diabetes mellitus and simple optic atrophy among siblings: Report of four cases. May Clin Proc. 1938;1:715–718. [Google Scholar]
- 66.Marshall B.A., Permutt M.A., Paciorkowski A.R., Hoekel J., Karzon R., Wasson J., Viehover A., White N., Shimony J., Manwaring L., et al. Phenotypic characteristics of early Wolfram syndrome. Orphanet J. Rare Diseases. 2013;8:64. doi: 10.1186/1750-1172-8-64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Lugar H.M., Koller J.M., Rutlin J., Marshall B.A., Kanekura K., Urano F., Bischoff A.N., Shimony J.S., Hershey T., The Washington University Wolfram Syndrome Research Study Group Neuroimaging evidence of deficient axon myelination in Wolfram syndrome. Sci. Rep. 2016;6:21167. doi: 10.1038/srep21167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Hershey T., Lugar H.M., Shimony J.S., Rutlin J., Koller J.M., Perantie D.C., Paciorkowski A.R., Eisenstein S.A., Permutt M.A., The Washington University Wolfram Study Group Early Brain Vulnerability in Wolfram Syndrome. PLoS ONE. 2012;7:e40604. doi: 10.1371/journal.pone.0040604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Fonseca S.G., Ishigaki S., Oslowski C.M., Lu S., Lipson K.L., Ghosh R., Hayashi E., Ishihara H., Oka Y., Permutt M.A., et al. Wolfram syndrome 1 gene negatively regulates ER stress signaling in rodent and human cells. J. Clin. Investig. 2010;120:744–755. doi: 10.1172/JCI39678. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Fonseca S.G., Fukuma M., Lipson K.L., Nguyen L.X., Allen J.R., Oka Y., Urano F. WFS1 is a novel component of the unfolded protein response and maintains homeostasis of the endoplasmic reticulum in pancreatic beta-cells. J. Biol. Chem. 2005;280:39609–39615. doi: 10.1074/jbc.M507426200. [DOI] [PubMed] [Google Scholar]
- 71.Zatyka M., Da Silva Xavier G., Bellomo E.A., Leadbeater W., Astuti D., Smith J., Michelangeli F., Rutter G.A., Barrett T.G. Sarco(endo)plasmic reticulum ATPase is a molecular partner of Wolfram syndrome 1 protein, which negatively regulates its expression. Hum. Mol. Genet. 2015;24:814–827. doi: 10.1093/hmg/ddu499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Morikawa S., Blacher L., Onwumere C., Urano F. Loss of Function of WFS1 Causes ER Stress-Mediated Inflammation in Pancreatic Beta-Cells. Front. Endocrinol. 2022;13:849204. doi: 10.3389/fendo.2022.849204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Hatanaka M., Tanabe K., Yanai A., Ohta Y., Kondo M., Akiyama M., Shinoda K., Oka Y., Tanizawa Y. Wolfram syndrome 1 gene (WFS1) product localizes to secretory granules and determines granule acidification in pancreatic beta-cells. Hum. Mol. Genet. 2011;20:1274–1284. doi: 10.1093/hmg/ddq568. [DOI] [PubMed] [Google Scholar]
- 74.Wang L., Liu H., Zhang X., Song E., Wang Y., Xu T., Li Z. WFS1 functions in ER export of vesicular cargo proteins in pancreatic beta-cells. Nat. Commun. 2021;12:6996. doi: 10.1038/s41467-021-27344-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.De Franco E., Flanagan S.E., Yagi T., Abreu D., Mahadevan J., Johnson M.B., Jones G., Acosta F., Mulaudzi M., Lek N., et al. Dominant ER Stress-Inducing WFS1 Mutations Underlie a Genetic Syndrome of Neonatal/Infancy-Onset Diabetes, Congenital Sensorineural Deafness, and Congenital Cataracts. Diabetes. 2017;66:2044–2053. doi: 10.2337/db16-1296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Morikawa S., Tajima T., Nakamura A., Ishizu K., Ariga T. A novel heterozygous mutation of the WFS1 gene leading to constitutive endoplasmic reticulum stress is the cause of Wolfram syndrome. Pediatr. Diabetes. 2017;18:934–941. doi: 10.1111/pedi.12513. [DOI] [PubMed] [Google Scholar]
- 77.Bonnycastle L.L., Chines P.S., Hara T., Huyghe J.R., Swift A.J., Heikinheimo P., Mahadevan J., Peltonen S., Huopio H., Nuutila P., et al. Autosomal dominant diabetes arising from a Wolfram syndrome 1 mutation. Diabetes. 2013;62:3943–3950. doi: 10.2337/db13-0571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Cagalinec M., Liiv M., Hodurova Z., Hickey M.A., Vaarmann A., Mandel M., Zeb A., Choubey V., Kuum M., Safiulina D., et al. Role of Mitochondrial Dynamics in Neuronal Development: Mechanism for Wolfram Syndrome. PLoS Biol. 2016;14:e1002511. doi: 10.1371/journal.pbio.1002511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Abreu D., Asada R., Revilla J.M.P., Lavagnino Z., Kries K., Piston D.W., Urano F. Wolfram syndrome 1 gene regulates pathways maintaining beta-cell health and survival. Lab. Investig. A J. Tech. Methods Pathol. 2020;100:849–862. doi: 10.1038/s41374-020-0408-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Lu S., Kanekura K., Hara T., Mahadevan J., Spears L.D., Oslowski C.M., Martinez R., Yamazaki-Inoue M., Toyoda M., Neilson A., et al. A calcium-dependent protease as a potential therapeutic target for Wolfram syndrome. Proc. Natl. Acad. Sci. USA. 2014;111:E5292–E5301. doi: 10.1073/pnas.1421055111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Ishihara H., Takeda S., Tamura A., Takahashi R., Yamaguchi S., Takei D., Yamada T., Inoue H., Soga H., Katagiri H., et al. Disruption of the WFS1 gene in mice causes progressive beta-cell loss and impaired stimulus-secretion coupling in insulin secretion. Hum. Mol. Genet. 2004;13:1159–1170. doi: 10.1093/hmg/ddh125. [DOI] [PubMed] [Google Scholar]
- 82.Alfaifi M. Interaction between rs6446482 polymorphisms in the WFS1 gene in type 2 diabetes patients. J. King Saud Univ. Sci. 2022;34:101721. doi: 10.1016/j.jksus.2021.101721. [DOI] [Google Scholar]
- 83.Cheng S., Wu Y., Wu W., Zhang D. Association of rs734312 and rs10010131 polymorphisms in WFS1 gene with type 2 diabetes mellitus: A meta-analysis. Endocr. J. 2013;60:441–447. doi: 10.1507/endocrj.EJ12-0325. [DOI] [PubMed] [Google Scholar]
- 84.Bansal V., Boehm B.O., Darvasi A. Identification of a missense variant in the WFS1 gene that causes a mild form of Wolfram syndrome and is associated with risk for type 2 diabetes in Ashkenazi Jewish individuals. Diabetologia. 2018;61:2180–2188. doi: 10.1007/s00125-018-4690-3. [DOI] [PubMed] [Google Scholar]
- 85.Kitamura R.A., Maxwell K.G., Ye W., Kries K., Brown C.M., Augsornworawat P., Hirsch Y., Johansson M.M., Weiden T., Ekstein J., et al. Multidimensional analysis and therapeutic development using patient iPSC-derived disease models of Wolfram syndrome. JCI Insight. 2022;7:e156549. doi: 10.1172/jci.insight.156549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Abreu D., Stone S.I., Pearson T.S., Bucelli R.C., Simpson A.N., Hurst S., Brown C.M., Kries K., Onwumere C., Gu H., et al. A phase Ib/IIa clinical trial of dantrolene sodium in patients with Wolfram syndrome. JCI Insight. 2021;6:e145188. doi: 10.1172/jci.insight.145188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Nguyen L.D., Fischer T.T., Abreu D., Arroyo A., Urano F., Ehrlich B.E. Calpain inhibitor and ibudilast rescue beta cell functions in a cellular model of Wolfram syndrome. Proc. Natl. Acad. Sci. USA. 2020;117:17389–17398. doi: 10.1073/pnas.2007136117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Ozcan U., Yilmaz E., Ozcan L., Furuhashi M., Vaillancourt E., Smith R.O., Gorgun C.Z., Hotamisligil G.S. Chemical chaperones reduce ER stress and restore glucose homeostasis in a mouse model of type 2 diabetes. Science. 2006;313:1137–1140. doi: 10.1126/science.1128294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Seppa K., Jagomae T., Kukker K.G., Reimets R., Pastak M., Vasar E., Terasmaa A., Plaas M. Liraglutide, 7,8-DHF and their co-treatment prevents loss of vision and cognitive decline in a Wolfram syndrome rat model. Sci. Rep. 2021;11:2275. doi: 10.1038/s41598-021-81768-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Jagomae T., Seppa K., Reimets R., Pastak M., Plaas M., Hickey M.A., Kukker K.G., Moons L., Groef L.D., Vasar E., et al. Early Intervention and Lifelong Treatment with GLP1 Receptor Agonist Liraglutide in a Wolfram Syndrome Rat Model with an Emphasis on Visual Neurodegeneration, Sensorineural Hearing Loss and Diabetic Phenotype. Cells. 2021;10:3193. doi: 10.3390/cells10113193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Scully K.J., Wolfsdorf J.I. Efficacy of GLP-1 Agonist Therapy in Autosomal Dominant WFS1-Related Disorder: A Case Report. Horm. Res. Paediatr. 2020;93:409–414. doi: 10.1159/000510852. [DOI] [PubMed] [Google Scholar]
- 92.Seppa K., Toots M., Reimets R., Jagomae T., Koppel T., Pallase M., Hasselholt S., Mikkelsen M.K., Nyengaard J.R., Vasar E., et al. GLP-1 receptor agonist liraglutide has a neuroprotective effect on an aged rat model of Wolfram syndrome. Sci. Rep. 2019;9:15742. doi: 10.1038/s41598-019-52295-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Toots M., Seppa K., Jagomäe T., Koppel T., Pallase M., Heinla I., Terasmaa A., Plaas M., Vasar E. Preventive treatment with liraglutide protects against development of glucose intolerance in a rat model of Wolfram syndrome. Sci. Rep. 2018;8:10183. doi: 10.1038/s41598-018-28314-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Kondo M., Tanabe K., Amo-Shiinoki K., Hatanaka M., Morii T., Takahashi H., Seino S., Yamada Y., Tanizawa Y. Activation of GLP-1 receptor signalling alleviates cellular stresses and improves beta cell function in a mouse model of Wolfram syndrome. Diabetologia. 2018;61:2189–2201. doi: 10.1007/s00125-018-4679-y. [DOI] [PubMed] [Google Scholar]
- 95.Sedman T., Runkorg K., Krass M., Luuk H., Plaas M., Vasar E., Volke V. Exenatide Is an Effective Antihyperglycaemic Agent in a Mouse Model of Wolfram Syndrome 1. J. Diabetes Res. 2016;2016:9239530. doi: 10.1155/2016/9239530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Kakiuchi C., Ishigaki S., Oslowski C.M., Fonseca S.G., Kato T., Urano F. Valproate, a mood stabilizer, induces WFS1 expression and modulates its interaction with ER stress protein GRP94. PLoS ONE. 2009;4:e4134. doi: 10.1371/journal.pone.0004134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Maxwell K.G., Augsornworawat P., Velazco-Cruz L., Kim M.H., Asada R., Hogrebe N.J., Morikawa S., Urano F., Millman J.R. Gene-edited human stem cell-derived beta cells from a patient with monogenic diabetes reverse preexisting diabetes in mice. Sci. Transl. Med. 2020;12:9106. doi: 10.1126/scitranslmed.aax9106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Shang L., Hua H., Foo K., Martinez H., Watanabe K., Zimmer M., Kahler D.J., Freeby M., Chung W., LeDuc C., et al. Beta-cell dysfunction due to increased ER stress in a stem cell model of Wolfram syndrome. Diabetes. 2014;63:923–933. doi: 10.2337/db13-0717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Teo A.K., Windmueller R., Johansson B.B., Dirice E., Njolstad P.R., Tjora E., Raeder H., Kulkarni R.N. Derivation of human induced pluripotent stem cells from patients with maturity onset diabetes of the young. J. Biol. Chem. 2013;288:5353–5356. doi: 10.1074/jbc.C112.428979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Dwivedi O.P., Lehtovirta M., Hastoy B., Chandra V., Krentz N.A.J., Kleiner S., Jain D., Richard A.-M., Abaitua F., Beer N.L., et al. Loss of ZnT8 function protects against diabetes by enhanced insulin secretion. Nat. Genet. 2019;51:1596–1606. doi: 10.1038/s41588-019-0513-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Wang X., Sterr M., Ansarullah, Burtscher I., Bottcher A., Beckenbauer J., Siehler J., Meitinger T., Häring H.-U., Staiger H., et al. Point mutations in the PDX1 transactivation domain impair human beta-cell development and function. Mol. Metab. 2019;24:80–97. doi: 10.1016/j.molmet.2019.03.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Amin S., Cook B., Zhou T., Ghazizadeh Z., Lis R., Zhang T., Khalaj M., Crespo M., Perera M., Xiang J.Z., et al. Discovery of a drug candidate for GLIS3-associated diabetes. Nat. Commun. 2018;9:2681. doi: 10.1038/s41467-018-04918-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Cosentino C., Toivonen S., Diaz Villamil E., Atta M., Ravanat J.L., Demine S., Schiavo A.A., Pachera N., Deglasse J.-P., Jonas J.-C., et al. Pancreatic beta-cell tRNA hypomethylation and fragmentation link TRMT10A deficiency with diabetes. Nucleic Acids Res. 2018;46:10302–10318. doi: 10.1093/nar/gky839. [DOI] [PMC free article] [PubMed] [Google Scholar]