Abstract
Unrestrained transcriptional activity of β-CATENIN and its binding partner TCF7L2 frequently underlies colorectal tumor initiation and is considered an obligatory oncogenic driver throughout intestinal carcinogenesis. Yet, the TCF7L2 gene carries inactivating mutations in about 10% of colorectal tumors and is non-essential in colorectal cancer (CRC) cell lines. To determine whether CRC cells acquire TCF7L2-independence through cancer-specific compensation by other T-cell factor (TCF)/lymphoid enhancer-binding factor (LEF) family members, or rather lose addiction to β-CATENIN/TCF7L2-driven gene expression altogether, we generated multiple CRC cell lines entirely negative for TCF/LEF or β-CATENIN expression. Survival of these cells and the ability to propagate them demonstrate their complete β-CATENIN- and TCF/LEF-independence. Nonetheless, one β-CATENIN-deficient cell line eventually became senescent, and absence of TCF/LEF proteins and β-CATENIN consistently impaired CRC cell proliferation, reminiscent of mitogenic effects of WNT/β-CATENIN signaling in the healthy intestine. Despite this common phenotype, β-CATENIN-deficient cells exhibited highly cell-line-specific gene expression changes with little overlap between β-CATENIN- and TCF7L2-dependent transcriptomes. Apparently, β-CATENIN and TCF7L2 independently control sizeable fractions of their target genes. The observed divergence of β-CATENIN and TCF7L2 transcriptional programs, and the finding that neither β-CATENIN nor TCF/LEF activity is strictly required for CRC cell survival has important implications when evaluating these factors as potential drug targets.
Subject terms: Cancer genetics, Cancer, Gastrointestinal cancer, Colorectal cancer, Cell biology, Cell adhesion, Cell division, Cell signalling, Molecular biology, Cell division, Transcription, Transcriptomics
Introduction
The WNT/β-CATENIN pathway is an evolutionarily conserved metazoan signal transduction cascade with pivotal functions throughout embryogenesis and in post-natal tissue homeostasis. Central to this pathway is the so-called destruction complex, a multifactorial assembly comprising among others the tumor suppressors AXIN and APC, CASEIN KINASE1α (CK1α), and GLYCOGEN SYNTHASE KINASE 3 (GSK3). Within the microcompartment of the destruction complex, CK1α and GSK3 sequentially phosphorylate β-CATENIN and thereby prime it for proteasomal degradation, thus keeping the pathway in the off state1. Binding of WNT growth factors to FRIZZLED and LRP cell surface receptors through mechanisms that are still not fully understood, inhibits the destruction complex. This results in β-CATENIN stabilization and translocation into the nucleus where it functions as cofactor of members of the TCF/LEF family of sequence specific DNA-binding proteins2,3. Complexes formed by β-CATENIN and TCF/LEF proteins together with additional regulatory factors in turn elicit cell-type-specific and ubiquitous transcriptional responses2,3, which represent the main output of the WNT/β-CATENIN pathway.
Beginning in late stage mouse embryonic development, WNT/β-CATENIN signaling is absolutely required for the maintenance of highly proliferative stem and progenitor cell compartments in the intestine, but also for lineage decisions, differentiation and proper positioning of intestinal epithelial cells (IECs)1. In agreement with its potent mitogenic effects, aberrant WNT/β-CATENIN pathway activity is a major oncogenic driver in colorectal tumorigenesis1,4. In fact, more than 90% of human colorectal cancers harbor mutations leading to aberrant activation of WNT/β-CATENIN signaling5. Animal studies further indicated that WNT/β-CATENIN pathway activity not only suffices for tumor initiation but might also be essential for tumor maintenance6, altogether providing strong incentives to interfere with WNT/β-CATENIN signaling for therapeutic purposes4.
In the healthy intestine, WNT/β-CATENIN pathway-induced transcriptional responses are predominantly if not exclusively mediated by TCF7L2, which is the sole TCF/LEF family member capable to maintain stem and progenitor cell proliferation in vivo7–9, and to sustain survival and proliferation of small intestinal and colonic organoids in vitro8–11. Based on the close and essential partnership between β-CATENIN and TCF7L2 in healthy IECs, one would assume that TCF7L2 is indispensable to transmit oncogenic WNT/β-CATENIN pathway activity. However, various findings led to more complex and conflicting views about TCF7L2 and its role in colorectal tumorigenesis. For example, Hrckulak and colleagues, using transgenic mice, reported that in the context of Apc-deficiency, Tcf7l2 was obligatory for tumor initiation in vivo and for the establishment and growth of Apc-deficient small intestinal organoids in vitro8. In contrast, in another study, Tcf7l2 was claimed to be a haploinsufficient tumor suppressor and dispensable for tumor growth, although a genetically different mouse model comprising the ApcMin gene and a different conditional Tcf7l2 allele were employed12. Nonetheless, the observation of frequently occurring TCF7L2 loss-of-function mutations in human CRC genomes likewise pointed to potential tumor suppressor properties of TCF7L25,13,14, and indicated that CRC cells unlike healthy IECs may not depend on TCF7L2 to stay alive. Indeed, earlier RNA interference studies supported the proposed tumor suppressor function of TCF7L2, and demonstrated that TCF7L2 may not be strictly required for human CRC cell line survival15. Acquired TCF7L2-independence could further be confirmed experimentally by CRISPR/Cas9-mediated inactivation of TCF7L2 in CRC cell lines. However, no support for antiproliferative or tumor suppressive activities of TCF7L2 was found8,10. Rather, in these models TCF7L2-deficiency was accompanied by highly cell-line-specific phenotypic alterations ranging from no discernible effects in some cases to reduced proliferation and enhanced migration and invasion in others8,10, the latter being reminiscent of the physiological functions of TCF7L2 in healthy IECs. Altogether, it thus appears that certain aspects of the function of TCF7L2 in healthy IECs might be conserved in CRC cells, but the strict addiction to β-CATENIN/TCF7L2-driven transcription could be lost during cancer progression.
To explain the perplexing TCF7L2-independence of CRC cells it was suggested that other TCF/LEF family members might compensate the loss of TCF7L2 but this has not been proven so far8,10,16. As an alternative, CRC cell survival and proliferation might depend on gene expression programs that are driven not by β-CATENIN and TCF/LEF proteins but instead by β-CATENIN and other transcription factors17. Further, also complete independence of CRC cells from WNT/β-CATENIN pathway activity was reported18,19 which likewise would explain why a subset of human colorectal tumors and CRC cell lines can tolerate the loss of TCF7L2.
To shed more light on the importance of TCF7L2 and its role in colorectal cancer, we initially aimed to test the idea that other TCF/LEF family members rescue TCF7L2-deficiency. However, the ability to create CRC cell lines that do not express any TCF/LEF proteins, demonstrates that TCF/LEF activity is not strictly required for CRC cell survival. This prompted us to extend our analyses to β-CATENIN which similarly turned out to be non-essential in four out of eight CRC cell lines examined. Despite being expendable, β-CATENIN and TCF7L2 still promoted CRC cell proliferation, possibly related to their function in intestinal stem and progenitor cells. Although expression of cognate WNT/β-CATENIN target genes is impaired in β-CATENIN- and TCF/LEF-deficient cells, comprehensive and unbiased analyses of β-CATENIN- and TCF7L2-dependent transcriptomes revealed highly cell-line-specific transcriptional effects and extensive uncoupling of β-CATENIN and TCF7L2 transcriptional activities. Apparently, unlike in the healthy intestinal epithelium, the two factors act largely independently of each other in the context of CRC cells. The pronounced context-dependent and highly distinct, yet non-essential functions of β-CATENIN and TCF/LEF proteins challenge their broad utility as drug targets for CRC therapy.
Results
CRC cells survive and can be propagated in the complete absence of TCF/LEF expression
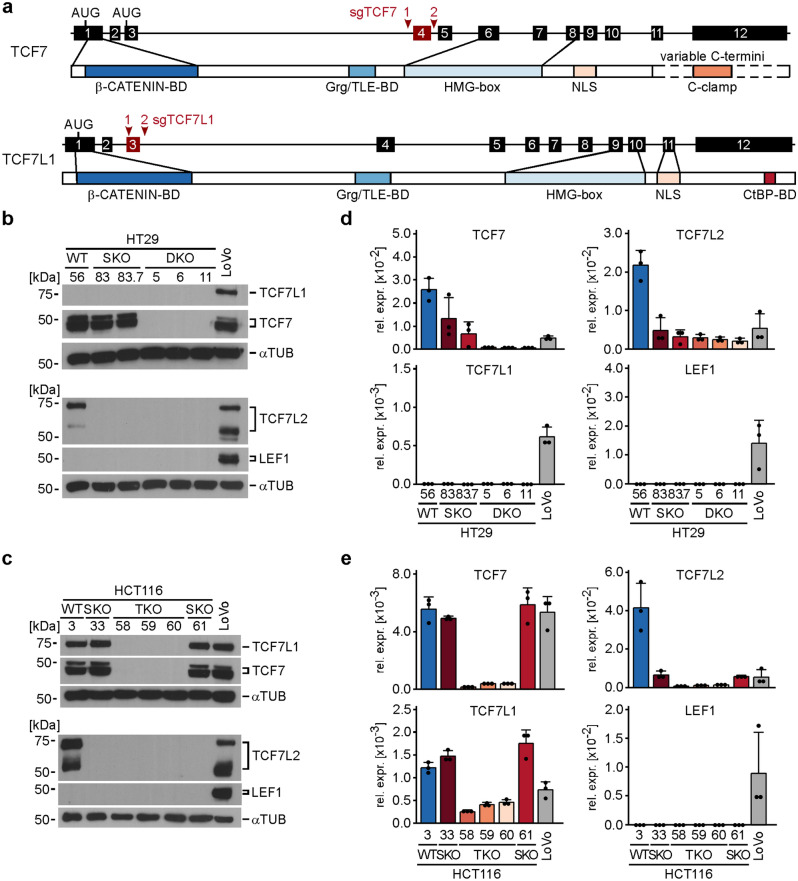
Previously it was proposed that lethality, which results from TCF7L2-deficiency in healthy IECs, might be prevented in CRC cells due to expression of other TCF/LEF family members that could substitute for TCF7L28,10. To test this idea, we attempted to additionally inactivate the TCF7 and TCF7L1 genes in HT29 and HCT116 TCF7L2 single knock-out (SKO) cells which we had generated and extensively characterized in our previous study10. Figure 1a schematically depicts the structure of the two genes and the targeting strategy used for CRISPR/Cas9-mediated exon deletion. TCF7 and TCF7L2 are the only TCF/LEF family members expressed in HT29 cells, while HCT116 cells express TCF7, TCF7L1, and TCF7L2. LEF1 is not expressed in HT29 and HCT116 cells. (Fig. 1b–e). Therefore, if it was possible to generate and propagate viable, i.e. capable of living, HT29 TCF7/TCF7L2 double knock-out (DKO) and HCT116 TCF7/TCF7L1/TCF7L2 triple knock-out (TKO) cells, these would not express any TCF/LEF proteins. Strikingly, by using the CRISPR/Cas9 system and a frame-shift inducing exon-deletion strategy we succeeded in generating multiple clonally-derived HT29 DKO and HCT116 TKO cell lines. Compared to their wild type (WT) counterparts, these cells showed much reduced TCF7, TCF7L1, and TCF7L2 transcript levels, and, importantly, had no detectable TCF/LEF protein expression (Fig. 1b–e; Supplementary Tables S1–S3). From this we conclude that TCF7 and TCF7L1 do not compensate the loss of TCF7L2 to maintain CRC cell viability. Rather, it appears that TCF/LEF expression is entirely dispensable for the survival of HT29 and HCT116 cells.
Figure 1.
HT29 and HCT116 CRC cells survive and can be propagated in the complete absence of TCF/LEF expression. (a) Schematic representation of the TCF7 and TCF7L1 genes and their protein products. Exons are depicted as numbered boxes. Exons chosen for CRISPR/Cas9-induced deletion are shown in red. The locations of sgRNA target sites are indicated by red arrowheads. The position of start codons (AUG) and functionally important domains of the TCF7 and TCF7L1 proteins are marked and connected to the exon(s) by which they are encoded. HMG-box DNA-binding domain, NLS nuclear localization sequence, Grg Groucho-related gene, TLE Transducin-like enhancer of split, CtBP Carboxy-terminal binding protein, BD binding domain. (b,c) Western blot analyses to detect TCF/LEF expression in whole cell lysates from HT29 (b) and HCT116 cells (c) with the genotypes indicated. WT wild type, SKO single knock-out, biallelic inactivation of TCF7L2, DKO double knock-out, HT29 cells with biallelic inactivation of TCF7 and TCF7L2, TKO triple knock-out, HCT116 cells with biallelic inactivation of TCF7, TCF7L1, and TCF7L2. LoVo cells were included as positive control for TCF/LEF detection. α-TUBULIN (αTUB) was used to demonstrate equal loading. Molecular weights are given in kDa. Representative results from one of three independent biological replicates are shown. Full size/uncropped versions of the Western blot images are shown in Supplementary Fig. S7. (d,e) The bar plots summarize and compare RNA expression levels of TCF/LEF family members determined by qRT-PCR in HT29 (d) and HCT116 cells (e) with the genotypes indicated. LoVo cells were included as positive control. TCF/LEF transcript levels were normalized to those of GAPDH, and are displayed as relative expression (rel. expr.). Each dot represents an individual measurement. Error bars indicate SD (n = 3). Statistical significance of gene expression differences was not analyzed for this series of experiments due to different group sizes of WT, SKO, DKO and TKO cell clones (see “Methods”).
Redundant and non-redundant functions of TCF/LEF family members in HT29 and HCT116 CRC cell lines
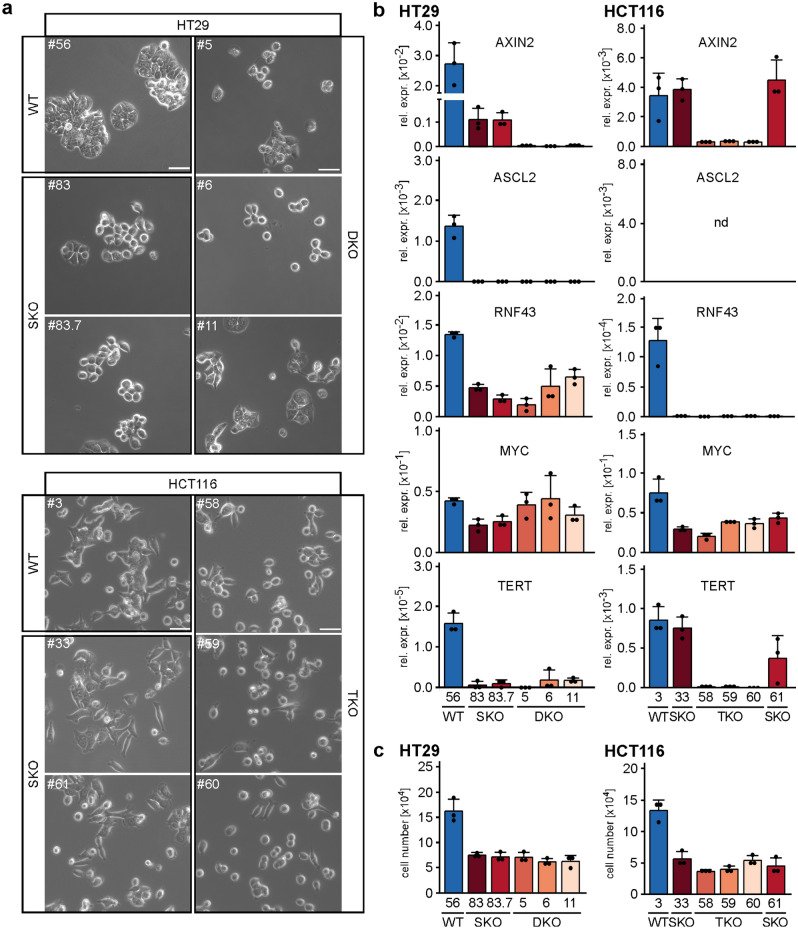
Even though TCF7 and TCF7L1 apparently do not play a role in acquired TCF7L2-independence of CRC cells, we nonetheless asked whether their additional absence had an impact on certain phenotypic changes observed in TCF7L2 SKO cells10. First, we examined cellular morphology. We found that HT29 SKO and HT29 DKO cells both exhibited a loss of the densely clustered and cobblestone-like growth pattern displayed by a representative HT29 WT cell clone. However, HT29 SKO and DKO cells did not noticeably differ from each other (Fig. 2a). In contrast, the elongated, spindle-like shape of HCT116 WT cells clearly changed only in the complete absence of TCF/LEF expression. Compared to HCT116 WT and SKO cells, HCT116 TKO cells grew more disperse and acquired roundish cell shapes, similar to HT29 SKO and DKO cells (Fig. 2a). Apparently, TCF7L2 has a unique impact on HT29 cell morphology whereas in HCT116 cells TCF7, TCF7L1, and TCF7L2 may function interchangeably or in a combinatorial fashion to determine cellular appearance. Next, to examine potential cell-type-specific overlapping as well as distinct functions of TCF/LEF family members at a molecular level, we analyzed expression of WNT/β-CATENIN target genes by qRT-PCR. Even though different group sizes of WT, SKO, DKO and TKO cell clones did not allow for statistical analyses using a linear mixed model which we otherwise applied10, we deem the results of these analyses to be confidently interpretable because expression of the genes under investigation was highly similar in cell clones of the same genotype, yet—where applicable—being distinctly different among cell clones with altered TCF/LEF status. Thus, in HT29 cells, the sole absence of TCF7L2 clearly reduced expression of AXIN2, RNF43, MYC, and TERT. Except for AXIN2, their expression did not further decrease in TCF7/TCF7L2 double-deficient cells (Fig. 2b). In HCT116 cells, however, single loss of TCF7L2 impaired expression of RNF43 and MYC, but not of AXIN2 and TERT, the expression of which was diminished only in HCT116 TKO cells (Fig. 2b). Hence, RNF43 and MYC appear to be under unique control by TCF7L2 while AXIN2 and to some extent TERT are additionally regulated by TCF7 and/or TCF7L1. Finally, we analyzed whether defects in cell proliferation, which we had observed in TCF7L2-deficient cells10, were affected by combined loss of TCF/LEF family members. Yet, cell counting and flow cytometry experiments revealed that impaired population dynamics and G1/S phase progression shown by HT29 and HCT116 SKO cells were not further aggravated in HT29 DKO and HCT116 TKO cells (Fig. 2c, Supplementary Fig. S1). In aggregate, these observations argue for context-dependent, combinatorial and partially redundant functions of TCF/LEF family members, for example in the regulation of cell shape and expression of some target genes, but also for non-redundant roles of TCF7L2 as in cell cycle control which is in line with its unique requirement in IEC proliferation.
Figure 2.
Redundant and non-redundant functions of TCF/LEF family members in HT29 and HCT116 CRC cell lines. (a) Representative micrographs from one of three independent biological replicates showing the morphology of HT29 and HCT116 WT, SKO, DKO, and TKO cells 24 h after seeding the same starting numbers of cells. The scale bars represent 50 µm. (b) Expression of the WNT/β-CATENIN target genes AXIN2, ASCL2, RNF43, MYC, and TERT was analyzed by qRT-PCR in HT29 and HCT116 cell clones with the genotypes indicated. Transcript levels were normalized to those of GAPDH, and are displayed as relative expression (rel. expr.). The bar plots summarize the expression data; error bars indicate SD (n = 3). Each dot represents an individual measurement. nd not detectable. (c) To compare population dynamics of WT and TCF/LEF-deficient cells, 5 × 104 HT29 and HCT116 cells with the genotypes indicated were seeded, incubated for 72 h and counted. The resulting cell counts are displayed as bar plots with each dot representing an individual measurement. Error bars indicate SD (n = 3). (b,c) Statistical significance of differences in gene expression levels and cell numbers was not analyzed for this series of experiments due to different group sizes of WT, SKO, DKO and TKO cell clones (see “Methods”).
CRC cells are viable and can be propagated in the absence of β-CATENIN
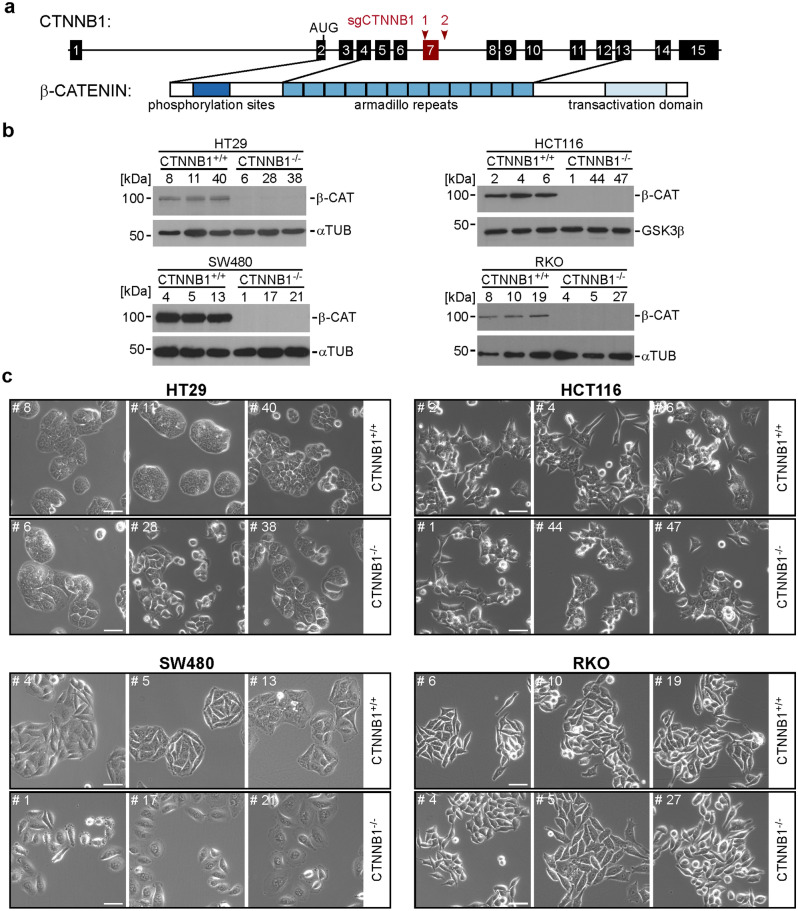
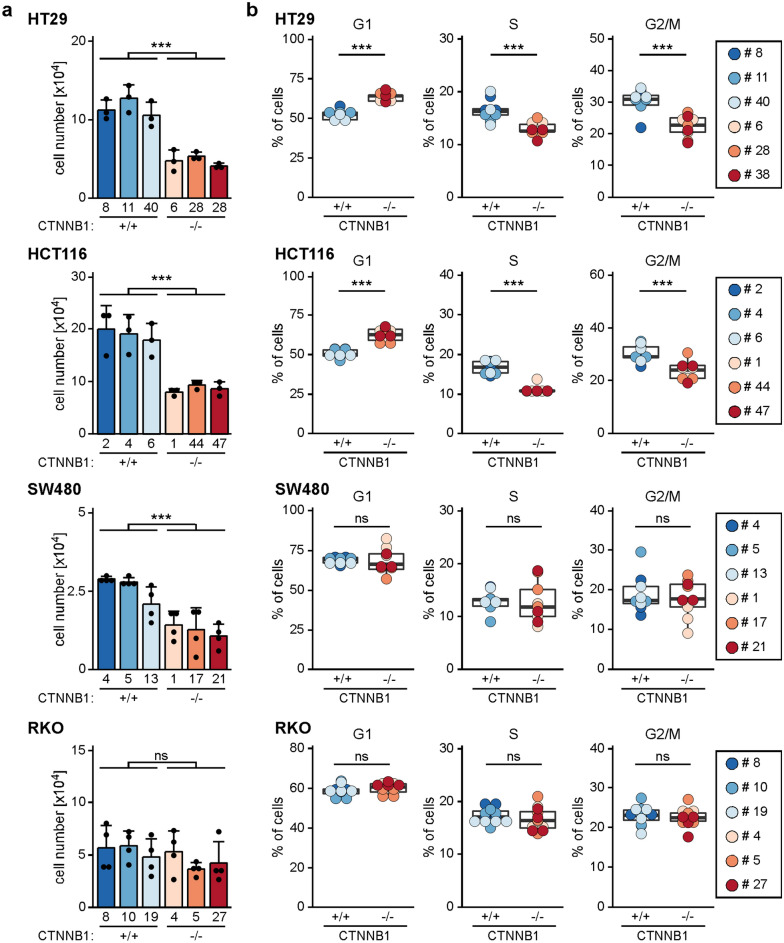
While β-CATENIN can also interact with other DNA-binding proteins to mediate transcriptional responses17, TCF/LEF family members are considered to be the major if not exclusive factors mediating WNT/β-CATENIN pathway-induced transcriptional responses20. The finding that HT29 and HCT116 cells survive in the absence of any TCF/LEF expression therefore raised the question whether CRC cells also exhibited independence from β-CATENIN. To address this, we tested whether viable and proliferating CRC cell line derivatives could be obtained when targeting the CTNNB1 gene (coding for β-CATENIN) using CRISPR/Cas9 technology. The knock-out attempt was made in HT29, HCT116, LoVo, LS174T, LS411, SW403, SW480, and RKO cells. Except for RKO cells, all other cell lines tested have inactivating mutations in APC (HT29, LoVo, LS411, SW403, SW480) or gain-of-function mutations in CTNNB1 (HCT116, LS174T), which lead to growth factor-independent WNT/β-CATENIN pathway activation and transcriptional activity. Additional properties and colorectal cancer driver pathway mutations in these cell lines are listed in Supplementary Table S4. Surprisingly, using the exon deletion strategy outlined in Fig. 3a, we were able to generate several clonal CTNNB1−/− cell lines from HCT116, HT29, SW480, and RKO cells that showed strongly reduced CTNNB1 RNA levels and no β-CATENIN protein expression (Supplementary Fig. S2a–d; Fig. 3b; Supplementary Tables S1, S2, S3). No clones with biallelic inactivation of CTNNB1 were retrieved from LoVo, LS174T, LS411, and SW403 cells (Supplementary Table S3), although it is unclear whether this is because of technical issues or due to essential functions of β-CATENIN in these cells. Irrespective of this, we began to phenotypically analyze those cells that could tolerate CTNNB1 inactivation. For this, we compared CTNNB1−/− cell clones to CTNNB1+/+ cell clones which had been exposed to the CRISPR/Cas9 system and which had undergone the same cloning procedure as the CTNNB1−/− cell clones but had retained the wild type sequences at the CTNNB1 locus. However, when assessing cellular morphology we did not detect changes that could unequivocally be traced to CTNNB1-deficiency. Thus, morphological diversity among HT29 cell clones more likely reflect clonal variability (Fig. 3c). CTNNB1−/− HCT116 and RKO cells showed no apparent differences in cell shape, and the slight change in appearance of SW480 CTNNB1−/− cells could be a secondary effect of altered cell growth and proliferation. For example, we noticed that upon prolonged cultivation, CTNNB1−/− SW480 cell clones became senescent, indicated by proliferation arrest, vastly increased cell size, and pronounced β-GALACTOSIDASE activity (Supplementary Fig. S3)21–23. Yet, senescent CTNNB1−/− SW480 cells seemingly remained alive for several weeks (Supplementary Fig. S3). Importantly, acquisition of a senescent phenotype was not observed with CTNNB1−/− HT29, HCT116, and RKO cells, and did not correlate with WT and mutant states of TP5324. Altogether, we conclude that there is no obligatory requirement for β-CATENIN for viability and proliferation of CRC cell lines.
Figure 3.
Some colorectal cancer cell lines are viable and can be propagated in the absence of β-CATENIN. (a) Schematic representation of the CTNNB1 gene and its protein product β-CATENIN. Exons are depicted as numbered boxes. Exon 7 (red box) was chosen for CRISPR/Cas9-induced deletion using two sgRNAs targeting positions marked by red arrowheads. The location of the start codon (AUG) in exon 2 and functionally important domains of β-CATENIN are indicated and connected to the exons by which they are encoded. (b) Western blot analyses to detect β-CATENIN (β-CAT) expression. Cell lysates were generated from cells with biallelic WT and mutant CTNNB1 genes. α-TUBULIN (αTUB) or GSK3β were used as loading controls. Molecular weights are given in kDa. Representative results from one of three independent biological replicates are shown. Full size/uncropped versions of the Western blot images are shown in Supplementary Fig. S7. (c) Representative micrographs from one of three independent biological replicates showing HCT116, HT29, SW480 and RKO cell clones with biallelic WT and mutant CTNNB1 genes. Images were taken 24 h after seeding the same starting numbers of cells. The scale bars represent 50 µm.
β-CATENIN-deficiency abolishes transcriptional activity of WNT/β-CATENIN signaling
To assess the impact of β-CATENIN-deficiency on transcriptional activity of the WNT/β-CATENIN pathway, we first performed reporter gene assays using the pSuper8xTOPFlash and pSuper8xFOPFlash pair of luciferase expression vectors with WT and mutant TCF/LEF binding sites in their promoter regions, respectively25. The two reporter constructs were transiently transfected into HT29, HCT116, pre-senescent SW480, and RKO cells. Comparison of the luciferase activities produced by the WT and mutant reporter plasmids confirmed the presence of β-CATENIN and TCF/LEF-mediated transcriptional activity in CTNNB1+/+ HT29, HCT116, and SW480 cells, which was completely abolished in CTNNB1−/− cells (Supplementary Fig. S4). In agreement with the WT state of their APC and CTNNB1 genes and the absence of other mutations that could activate the WNT pathway, RKO cells showed no intrinsic β-CATENIN transcriptional activity. Still, upon treatment with the GSK3β inhibitor CT99021, luciferase expression from the WT reporter increased in CTNNB1+/+ RKO cells. As expected, the reporter response was completely blocked in CTNNB1−/− RKO cells (Supplementary Fig. S4).
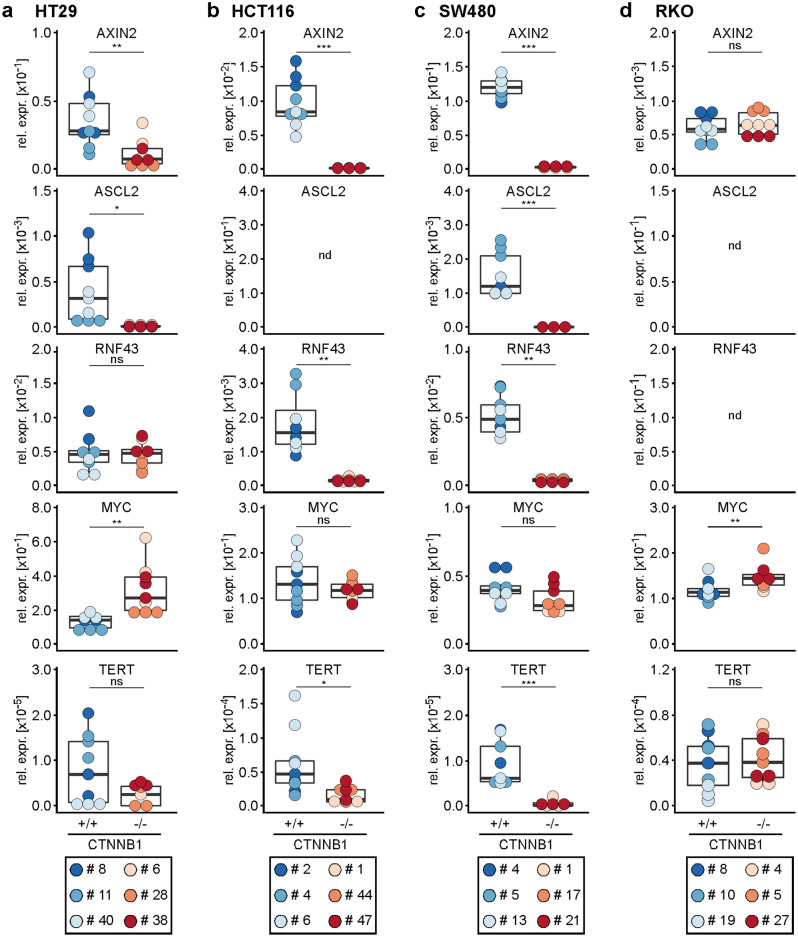
Next, we investigated the impact of β-CATENIN-deficiency on the expression of the cellular target genes AXIN2, ASCL2, RNF43, MYC, and TERT. In cell lines with an active WNT/β-CATENIN pathway (HT29, HCT116, SW480) and provided that the gene under investigation was expressed, we observed that eliminating β-CATENIN was paralleled by reduced expression of AXIN2, ASCL2, and TERT, albeit the reduction in TERT expression did not reach statistical significance in HT29 cells (Fig. 4). Likewise, RNF43 expression was lower in CTNNB1−/− HCT116 and SW480 cells. Surprisingly, however, RNF43 expression was not affected by β-CATENIN-deficiency in HT29 cells even though it had proven to be under control by TCF7L2. Similarly, and also in contrast to the response to TCF/LEF depletion, MYC transcript levels showed only a small, and statistically non-significant decline in β-CATENIN-deficient HCT116 and SW480 cells, and even went up in CTNNB1−/− HT29 cells (Fig. 4a). The latter was also seen in RKO cells (Fig. 4d). On the other hand, AXIN2 and TERT expression was not affected when CTNNB1 was inactivated in RKO cells, which is in line with the resting state of the WNT/β-CATENIN pathway in these cells. Taken together, although the reporter gene assays had demonstrated that β-CATENIN and TCF/LEF-mediated transcriptional activity was effectively and uniformly abolished in β-CATENIN-deficient cells, the identity of endogenous WNT/β-CATENIN target genes varied from cell to cell.
Figure 4.
WNT/β-CATENIN target gene expression is reduced in β-CATENIN-deficient CRC cells. (a–d) Expression of the WNT/β-CATENIN target genes AXIN2, ASCL2, RNF43, MYC, and TERT was analyzed by qRT-PCR in HT29 (a), HCT116 (b), SW480 (c) and RKO (d) cell clones with biallelic WT and mutant CTNNB1 genes. Transcript levels were normalized to GAPDH, and are displayed as relative expression (rel. expr.). The box plots display aggregated expression data from CTNNB1+/+ and CTNNB1−/− cell clones. Each dot represents a separate measurement with dot color identifying individual clones. For statistical analysis, LMM was performed (n = 3). nd not detectable, ns not significant.
β-CATENIN-deficiency affects proliferation and cell cycle progression in some CRC cells
WNT/β-CATENIN signaling is a potent mitogenic pathway in intestinal stem and progenitor cells as well as intestinal tumor cells, and blocking the pathway by inactivating TCF7L2 impairs CRC cell cycle progression (see above and10). Therefore, we were curious to see whether β-CATENIN-deficiency likewise affected proliferation of CRC cells. Indeed, by seeding defined numbers of cells and counting their progeny 72 h later, we found that relative to their WT counterparts β-CATENIN-deficient HT29, HCT116, and SW480 cells displayed reduced population dynamics (Fig. 5) which was not due to increased apoptosis (Supplementary Fig. S5). Rather, analyses of cellular DNA content by flow cytometry suggested a delay in G1/S phase transition in CTNNB1−/− HT29 and HCT116 cells (Fig. 5b), which had also been seen in TCF7L2-deficient cells (see above and10). In contrast, the clearly retarded expansion of SW480 cell populations could not be linked to a defined block in cell cycle progression. Besides, and as expected, proliferation of RKO cells with an inactive WNT/β-CATENIN pathway did not change in the absence of β-CATENIN. In sum, it appears that the mitogenic effects of WNT/β-CATENIN pathway activity found in healthy IECs might be partly retained in HT29, HCT116, and SW480 CRC cells, but the strict dependence on β-CATENIN and TCF7L2 for proliferation and survival appears to be alleviated in the cancer cells.
Figure 5.
Cell-type-specific impairment of population dynamics and cell cycle progression upon loss of β-CATENIN. (a) To compare population dynamics of WT and β-CATENIN-deficient cells, 1 × 104 each of HT29, HCT116, SW480, and RKO cells with the genotypes indicated were seeded, incubated for 72 h, and counted. The resulting cell counts are displayed as bar plots whereby each dot represents an individual measurement. For statistical analysis LMM was performed, the error bars indicate SD (n = 3). ns not significant. (b) To analyze differences in cell cycle distribution, HT29, HCT116, SW480, and RKO cells with the genotypes indicated were stained with propidium iodide and analyzed by flow cytometry. The proportions of cells in different cell cycle phases are depicted by box plots. Each dot represents a separate measurement with dot color identifying individual clones. For statistical analysis LMM was performed (n = 3). ns not significant.
Transcriptome analyses confirm highly cell-line-specific effects of β-CATENIN-deficiency
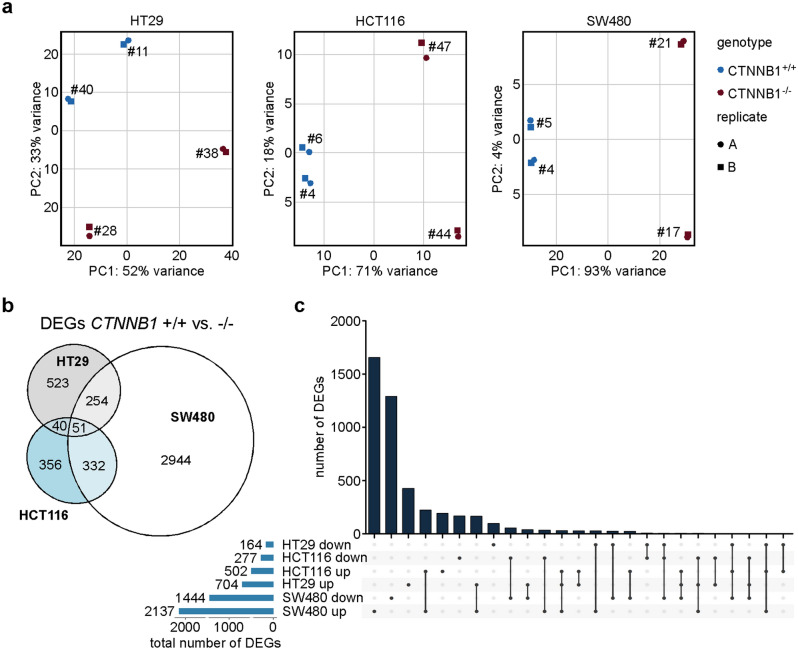
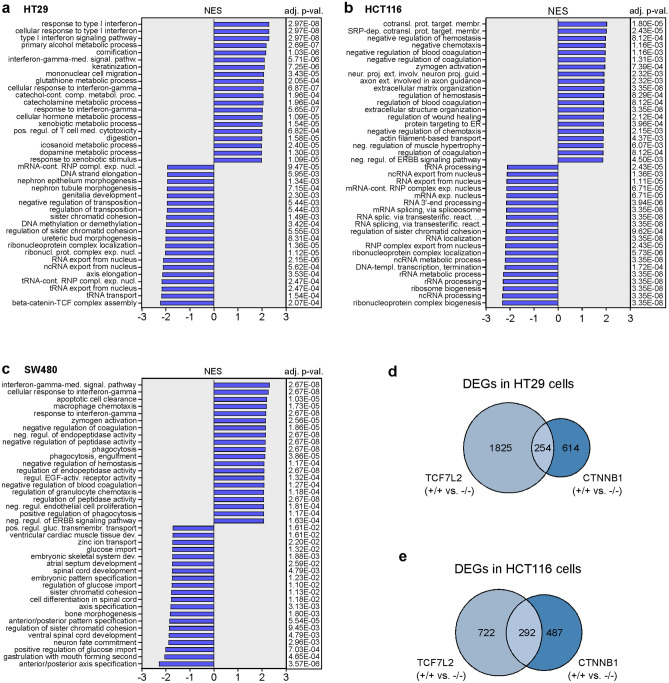
Targeted gene expression analyses had revealed differential, cell-line-specific alterations ensuing from β-CATENIN-deficiency. To capture differences in the transcriptomes of CTNNB1+/+ and CTNNB1−/− cells in an unbiased and comprehensive way, we performed two independent rounds of RNA-seq experiments for two CTNNB1+/+ and CTNNB1−/− clones each derived from the HT29, HCT116, and SW480 cell lines. Principal component analyses (PCA) showed a high degree of similarity between the two replicates for each cell clone (Fig. 6a). Further, for HCT116 and SW480 cells, CTNNB1+/+ and CTNNB1−/− clones were well separated along principal component 1 which covers the largest part of the variance and likely reflects WT and mutant states of CTNNB1. Consistent with the observed clonal variability, PCA indicated considerable differences among HT29 cell clones which cannot be explained solely by their genotypes. Irrespective of this, by comparing gene expression profiles of CTNNB1+/+ and CTNNB1−/− cell clones and by using an absolute log2(fold change) > 1 and an adjusted p-value < 0.05 as thresholds, we identified 868 differentially expressed genes (DEGs) in HT29 cells, 779 DEGs in HCT116 cells, and 3,581 DEGs in SW480 cells (Fig. 6b,c; Supplementary Tables S5–S7). The larger number of β-CATENIN-dependent genes in SW480 cells is in agreement with higher WNT/β-CATENIN pathway activity in this cell line compared to HT29 cells as determined by reporter gene assays (Supplementary Fig. S4 and17). In each cellular background, the majority of DEGs were upregulated in CTNNB1−/− cell clones relative to the CTNNB1+/+ control cells (Fig. 6c; Supplementary Tables S5–S7). Further, comparison of DEGs across cell lines corroborated that β-CATENIN-deficiency produced highly cell-line specific changes in gene expression in HT29, HCT116, and SW480 cells (Fig. 6b,c). This became even more apparent when taking into account the directionality of regulation (Fig. 6c). Altogether, only 51 DEGs were common to HT29, HCT116, and SW480 cells (Fig. 6b, Supplementary Table S8). Notably, aside from CTNNB1 and AXIN2, genes downregulated in all three CTNNB1−/− cell types included NR2F1 (COUP-TFII), ADAM19, and BMP4 which are also known WNT/β-CATENIN target genes26–30. Nevertheless, the majority of the 51 shared DEGs were upregulated in CTNNB1−/− cells and quite often were regulated in opposite directions in the three cell lines. In their sum, the results of the transcriptome analyses therefore revealed an extensive diversification of β-CATENIN-dependent gene expression in CRC cell lines at the level of individual genes. Despite this, the observed cell-line-specific gene expression changes could nonetheless affect similar biological processes. Therefore, to provide further insights into how β-CATENIN-dependent gene expression might shape the phenotypes of HT29, HCT116, and SW480 cells, we performed gene set enrichment analyses based on the GO term collection “Biological process”. This again revealed differences but also commonalities among the three cell lines. (Fig. 7, Supplementary Tables S9–S11). When considering positively enriched GO terms, transcriptomic changes in both HT29 and SW480 CTNNB1−/− cells pointed to altered interferon signaling (Fig. 7). On the other hand, gene expression changes in HCT116 and SW480 CTNNB1−/− cells displayed as common property enrichment for gene sets related to chemotaxis, directional movement, and hemostasis. As seemingly unique characteristic, β-CATENIN-deficient HT29 cells showed positive enrichment of metabolic features. Among the 20 most negatively enriched gene sets, GO terms related to RNA processing and RNA nuclear export were common to HT29 and HCT116 CTNNB1−/− cells, whereas HT29 and SW480 CTNNB1−/− cells shared negative enrichment of GO terms related to developmental processes, morphogenesis, pattern formation, and β-CATENIN-TCF complex assembly. While not among the 20 most negatively enriched GO terms, we noticed that gene sets related to cell cycle control and progression were depleted from the transcriptomes of HT29 and HCT116 CTNNB1−/− cells (Supplementary Tables S9, S10), which is in line with impaired proliferation and G1/S transition of these cells.
Figure 6.
Loss of CTNNB1 results in highly cell-type-specific gene expression changes in CRC cell lines. (a) Principal component analysis (PCA) of transcriptome data derived from HT29, HCT116, and SW480 cell clones with biallelic WT and mutant CTNNB1 genes. For each cell line and genotype, two different cell clones were analyzed by RNA-seq. Two independent biological replicates were performed (n = 2). (b) Venn diagram showing the numbers of differentially expressed genes (DEGs) specific and common to HCT116, HT29, and SW480 cells with biallelic WT and mutant CTNNB1 genes. (c) Upset plot depicting the numbers of DEGs specific and common to HCT116, HT29, and SW480 cells with biallelic WT and mutant CTNNB1 genes taking into account the direction of their regulation. In addition, for each cellular background the total numbers of up- and downregulated DEGs are given. (b,c) As thresholds to call DEGs, absolute values of a log2(fold change) > 1 and an adjusted p-value < 0.05 were used.
Figure 7.
Gene expression changes in β-CATENIN-deficient CRC cell lines affect cell-type-specific and cell-type-overarching biological processes. (a–c) Gene set enrichment analysis of gene sets was performed on the CTNNB1-dependent DEGs in HT29 (a), HCT116 (b) and SW480 cells (c) using Fisher’s exact test. The normalized enrichment scores (NES) and the corresponding adj. p-values for the top 20 positively- and negatively enriched Gene Ontology (GO) terms “Biological process” are shown in the bar plot. (d,e) Comparison of gene expression changes following inactivation of TCF7L2 and CTNNB1 in HT29 (d) and HCT116 cells (e). The Venn diagrams show the numbers of DEGs specific and common to cells with biallelic WT and mutant TCF7L2 or CTNNB1 genes. (d,e) As thresholds to call DEGs, absolute values of a log2(fold change) > 1 and an adjusted p-value < 0.05 were used.
The pronounced cell-line-specificity of transcriptomic changes caused by β-CATENIN-deficiency were reminiscent of similar observations made with TCF7L2-deficient cell lines10. It was therefore of interest to examine whether β-CATENIN and TCF7L2 regulated the same or divergent sets of genes in a given cellular background. For this we made use of RNA-seq data available from our previous study10. Initial PCA for all HT29 and HCT116 TCF7L2 and CTNNB1 WT/KO cell clones segregated the cell clones into four major groups according to cellular background and the two RNA-seq experiments which we had performed (Supplementary Fig. S6). The latter likely is due to differences in NGS library preparation (ribosomal RNA depletion for the TCF7L2 WT/KO cell clones versus oligo-dT capture for the CTNNB1 WT/KO cell clones) and may explain why the WT cell clones did not cluster together. Still, within each of the four major groups, WT and KO cell clones were clearly separated from each other. Since DEGs were determined based on the comparison of the corresponding WT and KO cell clones, we therefore thought that the intersection of TCF7L2- and β-CATENIN-dependent transcriptomes could be informative despite the constraints imposed by the technological differences. According to this analysis, TCF7L2 appeared to control more genes than β-CATENIN in both the HT29 and HCT116 cell backgrounds (Fig. 7d,e), although this has to be taken with caution because differences in NGS library preparation may have led to the detection of generally fewer transcripts in HT29 and HCT116 CTNNB1 WT and KO cells. More importantly, only a comparatively small fraction of the genes regulated by TCF7L2 in HT29 and HCT116 cells, respectively, also seemed to be under control of β-CATENIN (Fig. 7d,e; Supplementary Tables S12, S13). To rule out that we missed genes conjointly controlled by TCF7L2 and β-CATENIN, we asked whether genes that appeared to be selectively affected by CTNNB1 inactivation, were indeed expressed but not differentially regulated in the HT29 and HCT116 TCF7L2 WT and KO cells. This turned out to be the case (Supplementary Tables S14, S15). Thus, we conclude, that β-CATENIN and TCF7L2 control sizeable fractions of their target genes independently from each other, and that HT29 and HCT116 CRC cells do not feature the close partnership between β-CATENIN and TCF7L2 in gene regulation that is a hallmark of healthy IECs.
Discussion
Proliferation and survival of healthy intestinal stem and progenitor cells critically depend on the activity of the WNT/β-CATENIN pathway and its essential effectors β-CATENIN and TCF7L27–9,31,32. Despite the mutual dependence of β-CATENIN and TCF7L2 in healthy IECs and for tumor initiation, high proportions of colorectal cancer genomes carry inactivating mutations in the TCF7L2 gene5,13,14,33. Further, tumor suppressor and growth inhibitory functions were ascribed to TCF7L212,15. These observations suggested that TCF7L2 activity may not be mandatory for survival of colorectal tumor cells, which was experimentally confirmed in several distinct CRC cell lines8,10,16. To explain the differential requirements for TCF7L2 in healthy IECs and in CRC cells it was hypothesized that redundancy among TCF/LEF family members may allow for the loss of TCF7L2 in a cancer context8,10. Here, we tested this idea and report that survival of at least some CRC cell lines does not require any TCF/LEF expression, and that several randomly chosen CRC cell lines even tolerate inactivation of β-CATENIN. Thus, it appears that the vital dependence on β-CATENIN and TCF/LEF transcriptional activity that characterizes normal gut tissue, can be lost during colorectal carcinogenesis and that this may not be an exceptional and rare event. Nonetheless, in the absence of β-CATENIN or TCF/LEF proteins, CRC cell lines suffer from mitotic defects. While this could indicate that CRC cell lines retain to varying degrees some vestiges of the function of β-CATENIN and TCF7L2 in normal tissue, it could also be that these factors are parts of cancer-specific regulatory circuits controlling cell proliferation.
The idea that CRC cells bear the loss of TCF7L2 because other TCF/LEF family members substitute for it, was sparked by reports of functional interchangeability among TCF/LEF family members34,35 and observations that TCF/LEF expression patterns in CRC cell lines and colorectal tumors differ from those in normal IECs8,10,36,37. This includes for instance aberrant upregulation of LEF110,36,37. However, the results of our current study clearly refute this theory and actually demonstrate that none of the TCF/LEF proteins are essential for CRC viability. In retrospect, the finding that it is not expression of other TCF/LEF family members which prevents lethality caused by inactivation of TCF7L2, is not too surprising. TCF7 and TCF7L1 are coexpressed with TCF7L2 in healthy IECs9,10,38, yet fail to complement the TCF7L2 knockout in vivo7–9 and in vitro8–11. Furthermore, several previous studies showed that TCF7L2 and LEF1 have very different transcription regulatory properties and rather antagonize each other39–41 which argues that also LEF1 is unlikely to take over the function of TCF7L2. As a consequence, we rule out that cancer-specific neoexpression and compensation by other TCF/LEF family members account for expendability of TCF7L2 and suggest that this much more likely reflects far-reaching WNT/β-CATENIN pathway independence of CRC cell lines.
In this context it is important, though, to distinguish between a vital requirement for TCF7L2 or any of the other TCF/LEF family members which seemingly can be lost in CRC cell lines, and their impact on gene expression of CRC cells which is clearly detectable. There is no doubt that the cell-line-specific complement of TCF/LEF proteins does influence gene expression profiles of CRC cells and changes in TCF/LEF expression are accompanied by transcriptomic changes37,39,42. This appears to entail unique but also redundant and combinatorial as well as antagonistic activities and seemingly occurs in a manner that is highly dependent on cellular background and the target genes examined as shown here and in other studies8,10,16,37,39,42. As an additional aspect to keep in mind, our current study focused entirely on the importance of TCF/LEF family members and β-CATENIN for WNT target gene expression, survival, and proliferation of CRC cell lines, but did not examine whether additional features of cancer cells were affected. Thus, it is possible that other properties of CRC cells that were reported to be influenced by WNT/β-CATENIN pathway activity, such as stemness, cell fate decisions, epithelial-mesenchymal plasticity, cell adhesion, migration, invasiveness and metastasis1,3,4,43, still depend on TCF/LEF family members and β-CATENIN.
Overall, we and others successfully inactivated TCF7L2 in five different CRC cell lines8,10,16. Further, four out of eight CRC cell lines could cope with β-CATENIN-deficiency. Our lack of success in obtaining CTNNB1−/− derivatives from LS174T, LS411, LoVo, and SW403 cells could have technical reasons such as low transfection efficiency, poor clonogenicity, or insufficient expression of Cas9 and sgRNAs. However, using experimental procedures analogous to those employed here, we could readily generate LEF1−/− LS174T cells39. This demonstrates the ability to disable a WNT/β-CATENIN pathway component by CRISPR/Cas9 technology in this cell line, and hints at a fundamental difference between the functional importance of LEF1 and β-CATENIN in the LS174T cell background. Indeed, completely blocking WNT/β-CATENIN signaling by expression of either dominant-negative TCF7 or TCF7L2 and, alternatively, by shRNA-mediated knockdown of β-CATENIN, resulted in growth arrest of LS174T cells44,45. Accordingly, we do not think that all CRC cell lines—and by extrapolation all colorectal cancers—lose dependency on WNT/β-CATENIN pathway activity mediated by TCF/LEF proteins and β-CATENIN.
The CRISPR/Cas9 system was used before to generate viable and proliferating cells entirely negative for TCF/LEF expression35,46 or β-CATENIN46. However, these earlier studies employed murine embryonic stem cells and HEK293 human embryonic kidney cells which possess no endogenous WNT/β-CATENIN pathway activity and for which no essential functions of WNT/β-CATENIN signaling are known35,46. In contrast, WNT/β-CATENIN signaling is active in CRC cells and the cells are derived from a tissue that exhibits extraordinary dependence on WNT/β-CATENIN pathway activity and its mediators β-CATENIN and TCF7L21,7–10,12,31,32. The ability to generate viable CRC cell lines without any TCF/LEF or β-CATENIN expression, therefore, is remarkable, and raises the question as to the underlying cause for the observed expendability of β-CATENIN and TCF7L2. One possibility could be that in the course of our knock-out experiments we selected for cell clones that had acquired adaptive mutations which enabled the cells to bypass the vital requirement for β-CATENIN and TCF7L2-mediated transcription. However, in view of the rapid and parallel emergence of wild type, heterozygous, and homozygous mutant cell clones in all of our successful knock-out experiments, and given the frequencies and the simplicity of identifying for example CTNNB1−/− cell clones, we think that the genetic and possibly epigenetic changes that allow for TCF/LEF- and β-CATENIN-independent growth and survival existed already in the parental cell lines. Although it is nearly impossible to tell when these mutations might have occurred, we hypothesize they were gained at some point during cancer progression by cells from the tumors-of-origin of certain CRC cell lines. For example, the extreme reliance on mitogenic WNT/β-CATENIN pathway activity displayed by the healthy intestine may to variable degrees be alleviated by oncogenic MAPK or PI3 kinase signaling in colorectal tumors47. Aside from this, it was already shown that CRC cells and oncogenically transformed intestinal organoids can lose WNT/β-CATENIN pathway addiction owing to Hh/GLI signaling18 or nuclear YAP activity, respectively19. Currently, however, it is not known whether all CRC cell lines that allow for inactivation of TCF7L2 or β-CATENIN share as common property high Hh/GLI or YAP activity, or whether other, yet to be identified and possibly context-dependent conditions, can render β-CATENIN and TCF7L2 dispensable. For instance, in keeping with the previously reported function of MYC in CRC cells45, upregulation of this gene, which specifically occurred in TCF/LEF-deficient and CTNNB1−/− HT29 cells, could be a survival mechanism in this cell line.
In healthy IECs, β-CATENIN and TCF7L2 drive common or highly congruent transcriptional programs20. In contrast, the comparative transcriptome analyses conducted with CRC cell lines lacking β-CATENIN or TCF7L2 revealed highly divergent β-CATENIN-dependent gene expression in different CRC cell lines. Further, β-CATENIN and TCF7L2 seemingly control predominantly non-overlapping cohorts of target genes in a given cell line. Thus, CRC cell lines may not only lose their dependence on either β-CATENIN or TCF7L2 but the close cooperation and mutual dependence of the two factors seen in healthy IECs can be broken up. While the mechanistic basis for this remains to be clarified, it is well known that β-CATENIN and TCF/LEF proteins can function separately and control expression of distinct sets of genes independently from each other in various cellular contexts including CRC cells17,35,46,48,49. Examples are TCF/LEF-independent transcriptional activities of β-CATENIN mediated by its interaction with Sox17 and TBX517,48, and β-CATENIN-independent functions of TCF7 and LEF1 protein in the hematopoietic system50,51.
The proven and eminent pathophysiological relevance of the WNT/β-CATENIN pathway in colorectal carcinogenesis make it an attractive drug target. Multiple efforts were undertaken to identify inhibitors for example of β-CATENIN/TCF7L2 transcriptional activity for therapeutic purposes3. However, the possibility that CRC cells quite often may not be strictly dependent on β-CATENIN or TCF7L2 and that the two factors do not necessarily cooperate in gene regulation call into question the broad suitability of the β-CATENIN/TCF7L2 complex as target for treating colorectal cancer. As an additional corollary of the observed context-dependence and divergence of β-CATENIN and TCF7L2 activities, it is virtually impossible to extrapolate from work with a small number of cell lines to colorectal cancer in general, and the significance of results from high throughput inhibitor screens which often are based on a single cell line, is likely to be extremely limited. In this regard, though, our panel of isogenic cell line models that are either wild type or mutant for β-CATENIN and TCF/LEF family members, may serve as versatile tools to verify target specificity of drug candidates and their proposed mechanism of action.
Methods
Cell lines
A list with the parental wild type cell lines and the previously generated TCF7L2−/− cell clones10, as well as their culture conditions can be found in Supplementary Table S1.
Genome editing
CRISPR/Cas9-mediated genome editing was performed as before10 using sgRNAs targeting the sequences presented in Supplementary Table S16. Briefly, 1 × 106 cells were nucleofected with 600 ng of each sgRNA expression construct, and 800 ng of Cas9-turboRFP expression vector. Single RFP+ cells were sorted into 96 well plates 72 h after nucleofection, expanded, and screened for deletion of the target regions by PCR with genotyping primers listed in Supplementary Table S17. For inactivation of the TCF7 gene, HT29 TCF7L2−/− #83.7 cells were used. These cells were derived from HT29 TCF7L2−/− cell clone #8310 by lentiviral transduction with a TCF7 cDNA construct which, however, is not expressed. To obtain HCT116 triple knockout cells, HCT116 TCF7L2−/− cell clone #3310 was cotransfected with the Cas9-turboRFP expression vector and four sgRNA expression vectors targeting TCF7 and TCF7L1. The knock-out attempts of the CTNNB1 gene were made in wild type HT29, HCT116, LoVo, LS411, LS174T, RKO, SW403, and SW480 cells. CTNNB1 was successfully inactivated in HT29, HCT116, SW403, and RKO cells. Details about the targeting strategies and the genotypes of all cell clones generated in this study can be found in Supplementary Table S2. For all genome-editing experiments performed, information about the numbers of clones screened and their genotypes are given Supplementary Table S3.
Western blotting
For Western blotting, cells were lysed in RIPA buffer39. Aliquots of cell lysates with equal protein content were separated by SDS-PAGE and further processed for antigen detection as described39. Information about antibodies and their applications is provided in Supplementary Table S18.
β-GALACTOSIDASE staining
Cells were seeded in 6-well plates and cultivated until they had reached about 50% confluency. β-GALACTOSIDASE staining was carried out following a published protocol with an incubation in staining solution for 16 h52. Pictures of the stained cells were taken using a KEYENCE BZ-9000 microscope.
Measurements of population dynamics and cell cycle analyses
To compare population dynamics of WT and mutant cell lines, the cell numbers given in the corresponding figure legends were plated per well of 6-well plates and further cultivated for 72 h. Thereafter, the cells were detached and converted into single cell suspensions. Cell numbers were determined using a hemocytometer. For cell cycle analyses, 3 × 105 cells/well were seeded in 6-well plates and incubated for 48 h. Cells were then processed for flow cytometry as described39. A CytoFLEX flow cytometer (Beckman Coulter, Krefeld, Germany) in combination with the FlowJo v7.6.5 software (; BD Life Sciences, Heidelberg, Germany) was used for the analyses.
Luciferase assays
To determine WNT/β-CATENIN pathway activity by reporter gene assays, 5 × 104 cells/well were seeded in 24-well plates and transfected with 10 ng pRL-CMV (Promega, Walldorf, Germany) together with either 250 ng pSuper8xTOPFlash or 250 ng pSuper8xFOPFlash25 using the FuGENE 6 reagent (Promega). At 48 h post transfection, cells were lysed and reporter gene activities were determined as described40. Renilla luciferase activity was used for normalization. Where indicated, cells were treated for 40 h with 5 µM CT99021 (Selleckchem, Houston, Texas, USA) to stimulate the WNT/β-CATENIN pathway prior to measurements.
Caspase 3 activity assay
To detect apoptotic activity, 1 × 106 cells/well were seeded in 6-well plates and incubated for 24 h. The cells were lysed using IPN150-buffer with supplements (50 mM Tris/HCl, pH 7.6, 150 mM NaCl, 5 mM MgCl2, 0.1% NP-40 (v/v), 10 mM NaF, 1 mM PMSF, 1 mM DTT, 0.1 mM NaVO3, 1 × cOmplete protease inhibitors [Roche Diagnostics GmbH, Mannheim, Germany]) as previously described39. To measure caspase 3 activity, aliquots of whole cell lysates containing 40 ng of protein were adjusted to 20 µl volume with IPN150 buffer, combined with 80 µl of assay buffer (50 mM HEPES/KOH pH 7.6, 12 mM DTT) and 1 µl of DEVD-AMC substrate (final concentration 60 µm). For each cell lysate, duplicate measurements were carried out (two technical replicates). Substrate conversion was followed over a period of 30 min taking fluorescence readings at 1 min intervals using an Infinite F200 PRO machine (Tecan Trading AG, Männedorf, Switzerland) and excitation and emission wave lengths of 380 nm and 460 nm, respectively. For each kinetic measurement, data points were plotted, and the slope of the linear portion of the resulting curves was calculated. Values from the technical replicates were averaged, and plotted.
Work with RNA and RNA-seq data analysis
RNA isolation, cDNA synthesis, and qRT-PCR were performed as described39, except that a cDNA amount equivalent to 20 ng total RNA was used for qRT-PCR with oligonucleotide primers listed in Supplementary Table S17. For RNA-seq analysis, 1200 ng of total RNA were sent to Macrogen Europe for Sequencing. The library preparation was performed by Macrogen Europe using the TruSeq RNA Sample Prep Kit v2 and the according protocol (TruSeq RNA Sample Preparation v2 Guide, Part # 15026495 Rev. F). Paired-end sequencing was performed on an lllumina NovaSeq 6000 with 2 × 150 bp read length and 50 million read pairs per sample. For bioinformatic analysis, the galaxy.eu platform was used53. All reads were aligned to the human reference genome GRCh37 using RNA STAR Galaxy Version 2.7.5b54. For counting the reads per gene, featureCounts Galaxy Version 2.0.1 was applied55. DESeq2 Galaxy Version 2.11.40.6 + galaxy1 was used for PCA analysis and to identify differentially expressed genes56. For each cell line, CTNNB1−/− clones were compared to CTNNB1+/+ clones. Differentially expressed genes with an absolute value of log2(fold change) > 1 and an adjusted p-value < 0.05 were extracted (Supplementary Tables S5–S7). R was used for further data analysis and visualization of the data. For gene set enrichment analysis (GSEA), the clusterProfiler package was applied57. The Supplementary Tables S9–S11 contain the complete results of the GSEA for each cell line.
Statistical analysis
Where possible, statistical analysis was performed using a linear mixed model (LMM) to account for random and fixed effects. Normal distribution of data was assessed using the car R package58. For LMM analysis, the lme4 R package was applied59. The ggplot2 R package60 was used to generate box plots depicting the median, the lower and upper quartile. Whiskers represent 1.5-times the interquartile range. The ggplot2 R package60 was also used to generate bar plots depicting the mean with error bars representing the standard deviation (SD). The p-values for significant changes are represented as follows: *p < 0.05; **p < 0.01; ***p < 0.001. For each series of experiments, the corresponding number (n) of independent biological replicates is given in the figure legends. Note, however, that LMM was not applicable to the series of experiments involving HT29 and HCT116 TCF/LEF-negative cells due to different group sizes of WT, SKO, DKO, and TKO clones. Despite this we think that the results can be confidently interpreted because gene expression and cell numbers (Figs. 1 and 2; Supplementary Fig. S1) in cell clones of the same genotype are highly similar, while changes of these analytes, which we considered to be attributable to inactivation of TCF/LEF genes, are distinctly different, altogether allowing for meaningful inter-group comparisons.
Supplementary Information
Acknowledgements
The authors are grateful to the team at the Lighthouse Core Facility Freiburg, Germany, for cell sorting assistance, and all members of the Hecht laboratory for critical reading of the manuscript. The results shown here are in part based upon data generated by the TCGA Research Network: https://www.cancer.gov/tcga.
Author contributions
J.F. and A.H. conceived and designed experiments. J.F. and K.R. performed experiments, generated, and analyzed data. J.F. carried out bioinformatic analyses of the RNA-seq data, including identification of differentially expressed genes, and gene set enrichment analyses. J.F. and A.H. created figures and tables, and wrote the manuscript. All authors critically read and commented on the final version of the manuscript.
Funding
Open Access funding enabled and organized by Projekt DEAL. Financial support for this study was received from the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation) (Grant numbers 322977937/GRK2344, DFG HE2004/11-1, and CRC-850 subproject B5 to AH).
Data availability
The datasets generated and/or analyzed during the current study are available in the Gene Expression Omnibus repository (https://www.ncbi.nlm.nih.gov/geo/) under the accession numbers GSE135328 and GSE199835. All other data on which the results and conclusions of this study are based, are available from the corresponding author on reasonable request.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
The online version contains supplementary material available at 10.1038/s41598-022-27261-0.
References
- 1.de Lau W, Barker N, Clevers H. WNT signaling in the normal intestine and colorectal cancer. Front. Biosci. 2007;12:471–491. doi: 10.2741/2076. [DOI] [PubMed] [Google Scholar]
- 2.Anthony CC, Robbins DJ, Ahmed Y, Lee E. Nuclear regulation of Wnt/β-catenin signaling: It's a complex situation. Genes (Basel.) 2020 doi: 10.3390/genes11080886. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Bian J, Dannappel M, Wan C, Firestein R. Transcriptional regulation of Wnt/β-catenin pathway in colorectal cancer. Cells. 2020 doi: 10.3390/cells9092125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Parsons MJ, Tammela T, Dow LE. WNT as a driver and dependency in cancer. Cancer Discov. 2021;11:2413–2429. doi: 10.1158/2159-8290.CD-21-0190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Cancer Genome Atlas Network Comprehensive molecular characterization of human colon and rectal cancer. Nature. 2012;487:330–337. doi: 10.1038/nature11252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Dow LE, et al. Apc restoration promotes cellular differentiation and reestablishes crypt homeostasis in colorectal cancer. Cell. 2015;161:1539–1552. doi: 10.1016/j.cell.2015.05.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Korinek V, et al. Depletion of epithelial stem-cell compartments in the small intestine of mice lacking Tcf-4. Nat. Genet. 1998;19:379–383. doi: 10.1038/1270. [DOI] [PubMed] [Google Scholar]
- 8.Hrckulak D, et al. Wnt effector TCF4 is dispensable for Wnt signaling in human cancer cells. Genes (Basel). 2018 doi: 10.3390/genes9090439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.van Es JH, et al. A critical role for the Wnt effector Tcf4 in adult intestinal homeostatic self-renewal. Mol. Cell. Biol. 2012;32:1918–1927. doi: 10.1128/MCB.06288-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Wenzel J, et al. Loss of the nuclear Wnt pathway effector TCF7L2 promotes migration and invasion of human colorectal cancer cells. Oncogene. 2020;39:3893–3909. doi: 10.1038/s41388-020-1259-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Schuijers J, et al. Ascl2 acts as an R-spondin/Wnt-responsive switch to control stemness in intestinal crypts. Cell Stem Cell. 2015;16:158–170. doi: 10.1016/j.stem.2014.12.006. [DOI] [PubMed] [Google Scholar]
- 12.Angus-Hill ML, Elbert KM, Hidalgo J, Capecchi MR. T-cell factor 4 functions as a tumor suppressor whose disruption modulates colon cell proliferation and tumorigenesis. Proc. Natl. Acad. Sci. U.S.A. 2011;108:4914–4919. doi: 10.1073/pnas.1102300108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Sjöblom T, et al. The consensus coding sequences of human breast and colorectal cancers. Science. 2006;314:268–274. doi: 10.1126/science.1133427. [DOI] [PubMed] [Google Scholar]
- 14.Wood LD, et al. The genomic landscapes of human breast and colorectal cancers. Science. 2007;318:1108–1113. doi: 10.1126/science.1145720. [DOI] [PubMed] [Google Scholar]
- 15.Tang W, Dodge M, Gundapaneni D, Michnoff C, Roth M, Lum L. A genome-wide RNAi screen for Wnt/beta-catenin pathway components identifies unexpected roles for TCF transcription factors in cancer. Proc. Natl. Acad. Sci. U.S.A. 2008;105:9697–9702. doi: 10.1073/pnas.0804709105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Wan C, et al. Genome-scale CRISPR-Cas9 screen of Wnt/β-catenin signaling identifies therapeutic targets for colorectal cancer. Sci. Adv. 2021 doi: 10.1126/sciadv.abf2567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Rosenbluh J, et al. β-catenin driven cancers require a YAP1 transcriptional complex for survival and tumorigenesis. Cell. 2012;151:1457–1473. doi: 10.1016/j.cell.2012.11.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Varnat F, Siegl-Cachedenier I, Malerba M, Gervaz P, Ruiz iAltaba A. Loss of WNT-TCF addiction and enhancement of HH-GLI1 signalling define the metastatic transition of human colon carcinomas. EMBO Mol. Med. 2010;2:440–457. doi: 10.1002/emmm.201000098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Han T, et al. Lineage reversion drives WNT independence in intestinal cancer. Cancer Discov. 2020;10:1590–1609. doi: 10.1158/2159-8290.CD-19-1536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Schuijers J, Mokry M, Hatzis P, Cuppen E, Clevers H. Wnt-induced transcriptional activation is exclusively mediated by TCF/LEF. EMBO J. 2014;33:146–156. doi: 10.1002/embj.201385358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Biran A, et al. Quantitative identification of senescent cells in aging and disease. Aging Cell. 2017;16:661–671. doi: 10.1111/acel.12592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Dimri GP, et al. A biomarker that identifies senescent human cells in culture and in aging skin in vivo. Proc. Natl. Acad. Sci. U.S.A. 1995;92:9363–9367. doi: 10.1073/pnas.92.20.9363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Neurohr GE, et al. Excessive cell growth causes cytoplasm dilution and contributes to senescence. Cell. 2019;176:1083–1097.e18. doi: 10.1016/j.cell.2019.01.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Mouradov D, et al. Colorectal cancer cell lines are representative models of the main molecular subtypes of primary cancer. Cancer Res. 2014;74:3238–3247. doi: 10.1158/0008-5472.CAN-14-0013. [DOI] [PubMed] [Google Scholar]
- 25.Veeman MT, Slusarski DC, Kaykas A, Louie SH, Moon RT. Zebrafish prickle, a modulator of noncanonical Wnt/Fz signaling, regulates gastrulation movements. Curr. Biol. 2003;13:680–685. doi: 10.1016/S0960-9822(03)00240-9. [DOI] [PubMed] [Google Scholar]
- 26.Okamura M, et al. COUP-TFII acts downstream of Wnt/beta-catenin signal to silence PPARgamma gene expression and repress adipogenesis. Proc. Natl. Acad. Sci. U.S.A. 2009;106:5819–5824. doi: 10.1073/pnas.0901676106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Sun L, et al. Epigenetic regulation of a disintegrin and metalloproteinase (ADAM) transcription in colorectal cancer cells: Involvement of β-catenin, BRG1, and KDM4. Front. Cell Dev. Biol. 2020;8:581692. doi: 10.3389/fcell.2020.581692. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Shu W, et al. Wnt/beta-catenin signaling acts upstream of N-myc, BMP4, and FGF signaling to regulate proximal-distal patterning in the lung. Dev. Biol. 2005;283:226–239. doi: 10.1016/j.ydbio.2005.04.014. [DOI] [PubMed] [Google Scholar]
- 29.Kuroda K, Kuang S, Taketo MM, Rudnicki MA. Canonical Wnt signaling induces BMP-4 to specify slow myofibrogenesis of fetal myoblasts. Skelet. Muscle. 2013;3:5. doi: 10.1186/2044-5040-3-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Yokoyama Y, Watanabe T, Tamura Y, Hashizume Y, Miyazono K, Ehata S. Autocrine BMP-4 signaling is a therapeutic target in colorectal cancer. Cancer Res. 2017;77:4026–4038. doi: 10.1158/0008-5472.CAN-17-0112. [DOI] [PubMed] [Google Scholar]
- 31.Ireland H, et al. Inducible Cre-mediated control of gene expression in the murine gastrointestinal tract: Effect of loss of beta-catenin. Gastroenterology. 2004;126:1236–1246. doi: 10.1053/j.gastro.2004.03.020. [DOI] [PubMed] [Google Scholar]
- 32.Fevr T, Robine S, Louvard D, Huelsken J. Wnt/beta-catenin is essential for intestinal homeostasis and maintenance of intestinal stem cells. Mol. Cell. Biol. 2007;27:7551–7559. doi: 10.1128/MCB.01034-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Seshagiri S, et al. Recurrent R-spondin fusions in colon cancer. Nature. 2012;488:660–664. doi: 10.1038/nature11282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Gerner-Mauro KN, Akiyama H, Chen J. Redundant and additive functions of the four Lef/Tcf transcription factors in lung epithelial progenitors. Proc. Natl. Acad. Sci. U.S.A. 2020;117:12182–12191. doi: 10.1073/pnas.2002082117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Moreira S, et al. A single TCF transcription factor, regardless of its activation capacity, is sufficient for effective trilineage differentiation of ESCs. Cell Rep. 2017;20:2424–2438. doi: 10.1016/j.celrep.2017.08.043. [DOI] [PubMed] [Google Scholar]
- 36.Hovanes K, et al. Beta-catenin-sensitive isoforms of lymphoid enhancer factor-1 are selectively expressed in colon cancer. Nat. Genet. 2001;28:53–57. doi: 10.1038/88264. [DOI] [PubMed] [Google Scholar]
- 37.Mayer C-D, La Giclais SMD, Alsehly F, Hoppler S. Diverse LEF/TCF expression in human colorectal cancer correlates with altered Wnt-regulated transcriptome in a meta-analysis of patient biopsies. Genes (Basel). 2020 doi: 10.3390/genes11050538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Kosinski C, et al. Gene expression patterns of human colon tops and basal crypts and BMP antagonists as intestinal stem cell niche factors. Proc. Natl. Acad. Sci. U.S.A. 2007;104:15418–15423. doi: 10.1073/pnas.0707210104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Freihen V, et al. SNAIL1 employs β-Catenin-LEF1 complexes to control colorectal cancer cell invasion and proliferation. Int. J. Cancer. 2020;146:2229–2242. doi: 10.1002/ijc.32644. [DOI] [PubMed] [Google Scholar]
- 40.Hecht A, Stemmler MP. Identification of a promoter-specific transcriptional activation domain at the C terminus of the Wnt effector protein T-cell factor 4. J. Biol. Chem. 2003;278:3776–3785. doi: 10.1074/jbc.M210081200. [DOI] [PubMed] [Google Scholar]
- 41.Schnappauf O, et al. Enhancer decommissioning by Snail1-induced competitive displacement of TCF7L2 and down-regulation of transcriptional activators results in EPHB2 silencing. Biochim. Biophys. Acta. 2016;1859:1353–1367. doi: 10.1016/j.bbagrm.2016.08.002. [DOI] [PubMed] [Google Scholar]
- 42.Murphy M, Chatterjee SS, Jain S, Katari M, DasGupta R. TCF7L1 modulates colorectal cancer growth by inhibiting expression of the tumor-suppressor gene EPHB3. Sci. Rep. 2016;6:28299. doi: 10.1038/srep28299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Zhao H, et al. Wnt signaling in colorectal cancer: Pathogenic role and therapeutic target. Mol. Cancer. 2022;21:144. doi: 10.1186/s12943-022-01616-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.van de Wetering M, et al. Specific inhibition of gene expression using a stably integrated, inducible small-interfering-RNA vector. EMBO Rep. 2003;4:609–615. doi: 10.1038/sj.embor.embor865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.van de Wetering M, et al. The β-catenin/TCF-4 complex imposes a crypt progenitor phenotype on colorectal cancer cells. Cell. 2002;111:241–250. doi: 10.1016/S0092-8674(02)01014-0. [DOI] [PubMed] [Google Scholar]
- 46.Doumpas N, et al. TCF/LEF dependent and independent transcriptional regulation of Wnt/β-catenin target genes. EMBO J. 2019 doi: 10.15252/embj.201798873. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Li J, Ma X, Chakravarti D, Shalapour S, DePinho RA. Genetic and biological hallmarks of colorectal cancer. Genes Dev. 2021;35:787–820. doi: 10.1101/gad.348226.120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Mukherjee S, et al. Sox17 and β-catenin co-occupy Wnt-responsive enhancers to govern the endoderm gene regulatory network. Elife. 2020 doi: 10.7554/eLife.58029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Zhao X, Shan Q, Xue H-H. TCF1 in T cell immunity: A broadened frontier. Nat. Rev. Immunol. 2021 doi: 10.1038/s41577-021-00563-6. [DOI] [PubMed] [Google Scholar]
- 50.Grumolato L, et al. β-Catenin-independent activation of TCF1/LEF1 in human hematopoietic tumor cells through interaction with ATF2 transcription factors. PLoS Genet. 2013;9:e1003603. doi: 10.1371/journal.pgen.1003603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Zhao X, Shao P, Gai K, Li F, Shan Q, Xue H-H. β-catenin and γ-catenin are dispensable for T lymphocytes and AML leukemic stem cells. Elife. 2020 doi: 10.7554/eLife.55360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Itahana K, Itahana Y, Dimri GP. Colorimetric detection of senescence-associated β galactosidase. Methods Mol. Biol. 2013;965:143–156. doi: 10.1007/978-1-62703-239-1_8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Afgan E, et al. The Galaxy platform for accessible, reproducible and collaborative biomedical analyses: 2018 update. Nucleic Acids Res. 2018;46:W537–W544. doi: 10.1093/nar/gky379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Dobin A, et al. STAR: Ultrafast universal RNA-seq aligner. Bioinformatics. 2013;29:15–21. doi: 10.1093/bioinformatics/bts635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Liao Y, Smyth GK, Shi W. featureCounts: An efficient general purpose program for assigning sequence reads to genomic features. Bioinformatics. 2014;30:923–930. doi: 10.1093/bioinformatics/btt656. [DOI] [PubMed] [Google Scholar]
- 56.Love MI, Huber W, Anders S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 2014;15:550. doi: 10.1186/s13059-014-0550-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Yu G, Wang L-G, Han Y, He Q-Y. clusterProfiler: An R package for comparing biological themes among gene clusters. OMICS. 2012;16:284–287. doi: 10.1089/omi.2011.0118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Fox J, Weisberg S. An R Companion to Applied Regression. SAGE; 2019. [Google Scholar]
- 59.Bates D, Mächler M, Bolker B, Walker S. Fitting linear mixed-effects models using lme4. J. Stat. Soft. 2015 doi: 10.18637/jss.v067.i01. [DOI] [Google Scholar]
- 60.Wickham H. ggplot2 Elegant Graphics for Data Analysis. Springer; 2009. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The datasets generated and/or analyzed during the current study are available in the Gene Expression Omnibus repository (https://www.ncbi.nlm.nih.gov/geo/) under the accession numbers GSE135328 and GSE199835. All other data on which the results and conclusions of this study are based, are available from the corresponding author on reasonable request.