Abstract
Objectives
Immunoglobulin-G4-related disease (IgG4-RD) is a distinct systemic autoimmune-mediated disease manifesting as chronic inflammation and tissue fibrosis. Since the role of DNA methylation in the pathogenesis of IgG4-RD is still unclear, we conduct this study to investigate epigenetic modifications in IgG4-RD.
Methods
A genome-wide DNA methylation study was conducted with B cells, CD4+ T cells, and salivary gland tissues from IgG4-RD patients and matched controls by using the Illumina HumanMethylation 850K BeadChip. We further performed pyrosequencing and immunohistochemistry assays to validate the methylation status of some targets of interest.
Results
We identified differentially methylated CpG sites including 44 hypomethylated and 166 hypermethylated differentially methylated probes (DMPs) in B cells and 260 hypomethylated and 112 hypermethylated DMPs in CD4+ T cells from 10 IgG4-RD patients compared with 10 healthy controls. We also identified 36945 hypomethylated and 78380 hypermethylated DMPs in salivary gland tissues of 4 IgG4-RD patients compared with 4 controls. DPM2 (cg21181453), IQCK (cg10266221), and ABCC13 (cg05699681, cg04985582) were hypermethylated and MBP (cg18455083) was hypomethylated in B cells, CD4+ T cells, and salivary gland tissues of IgG4-RD patients. We also observed the hypomethylated HLA-DQB2 in CD4+ T cells from IgG4-RD patients. Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway analysis of DMPs in salivary gland tissues of IgG4-RD patients revealed enrichment of pathways involved in the regulation of immune cell responses and fibrosis.
Conclusion
This is the first DNA methylation study in peripheral B cells, CD4+ T cells, and salivary gland tissues from IgG4-RD patients. Our findings highlighted the role of epigenetic modification of DNA methylation and identified several genes and pathways possibly involved in IgG4-RD pathogenesis.
Supplementary Information
The online version contains supplementary material available at 10.1186/s13075-022-02978-5.
Keywords: IgG4-RD, DNA methylation, B cells, CD4+ T cells, Salivary gland tissues
Introduction
Immunoglobulin-G4-related disease (IgG4-RD) is a distinct systemic autoimmune-mediated disease characterized by the infiltration of IgG4+ plasma cells into one or multiple organs and the development of tumor-like masses [1]. Chronically activated B cells, T cells, and macrophages accumulate in fibrotic lesions of IgG4-RD [2]. Moreover, IgG4-RD has an estimated incidence of 0.28–1.08 per person, with 336-1,300 newly diagnosed patients per year [3]. Systemic corticosteroid administration and B cell depletion are the main therapeutic strategies, and both could significantly ameliorate IgG4-RD. However, because of glucocorticoid-induced toxicity and high relapse rate after rituximab treatment [4], IgG4-RD pathogenesis needs to be further explored.
Epigenetic mechanisms are found to be related to the risk of various autoimmune diseases [5–7]. DNA methylation is a well-studied epigenetic modification involved in gene expression regulation [8]. So far, numerous studies have provided evidence of aberrant DNA methylation profiles in rheumatoid arthritis (RA), systemic lupus erythematosus (SLE), and primary Sjögren's syndrome (pSS) [9–18]. However, changes in DNA methylation of IgG4-RD patients are poorly understood.
In this study, B cells and CD4+ T cells were purified from 10 IgG4-RD patients and 10 age- and sex-matched healthy controls (HCs). DNA methylation was examined using a genome-wide Illumina 850K CpG promoter. We also performed DNA methylation analysis of salivary gland tissues biopsied from 4 IgG4-RD patients and 4 HCs. Our findings demonstrated the pattern of DNA methylation in peripheral B cells, CD4+ T cells, and salivary gland tissues involving key genes and pathways associated with IgG4-RD pathogenesis.
Materials and methods
Study cohort and ethics
A total of 20 IgG4-RD patients were enrolled at Peking Union Medical College Hospital between 2020 and 2022 in our study. The diagnosis of IgG4-RD was established according to the 2019 American College of Rheumatology/European League Against Rheumatism classification criteria and the 2011 comprehensive diagnostic criteria for IgG4-RD [19, 20]. Peripheral blood samples were obtained from IgG4-RD patients and age-, sex-, and ethnicity-matched healthy blood donors. Salivary gland tissues were obtained from individuals examined for a possible IgG4-RD diagnosis. The biopsy specimens that showed no inflammation and negative serology were considered as case controls. Demographic and clinical characteristics of the study participants are presented in Supplementary Table 1. The diagnostic score was calculated according to the 2019 American College of Rheumatology/European League Against Rheumatism classification criteria for IgG4-RD. Disease activity was assessed on the basis of the IgG4-RD responder index [21].
This study was approved by the Ethics Board of Peking Union Medical College Hospital (No. JS-3389). All participants signed a written informed consent.
Isolation of B cells and CD4+ T cells
Peripheral blood mononuclear cells (PBMCs) were isolated from fresh peripheral blood samples through density gradient centrifugation (Ficoll-Paque, Cat. No. 7111011, DAKEWE, China). CD4+ T cells and B cells were separated from PBMCs using CD4 MicroBeads, human (Cat. No. 130-045-101, Miltenyi Biotec, Cambridge, MA) and B Cell Isolation Kit II (Cat. No. 130-091-151, Miltenyi Biotec, Cambridge, MA), respectively, according to the manufacturer’s protocol. The purity of isolated CD4+ T cells and B cells was confirmed to be >95% by flow cytometry.
DNA methylation profiling
DNA was extracted from cells or tissues using DNeasy Blood & Tissue Kit (Cat. No. 69506, Qiagen, Germany). The purity and concentration of DNA was measured using Nanodrop 2000 (ThermoScietific). Approximately, 500 ng of genomic DNA from each sample was used for sodium bisulfite conversion using the EZ DNA Methylation-Gold Kit (Cat. No. D5006, Zymo Research, USA). Genome-wide DNA methylation was assessed with the Illumina Infinium Human Methylation 850K BeadChip (Illumina Inc, USA) according to the manufacturer’s instructions. These steps were performed by Novogene Company (Beijing, China).
Analysis of DNA methylation data
DNA methylation files were processed in the R environment using the Minfi statistical analysis package (V1.30.0) for background signal correction and normalization of each channel to control probes included in each array. For each sample, (1) CpG sites with a p value of >0.01 and (2) probes with single nucleotide polymorphisms or their single base extension and X and Y chromosomes at the CpG site were excluded from the analysis [22–24].
The methylated and unmethylated probe values were used to calculate the β value ratio according to the following formula: β value = (methylated)/(methylated + unmethylated + 100). The mean difference β value (Δβ) and the adjusted p value between HCs and IgG4-RD patients for each CpG site were calculated using CHAMP packages (V2.14.0) [22]. For B cells or CD4+ T cells isolated from PBMCs, the adjusted p value ≤ 0.05 or Δβ > 0.05 was considered statistically significant. For tissue samples, the adjusted p value < 0.05 and Δβ ≥ 0.2 was considered statistically significant [25–29]. Differential methylation regions (DMRs) were analyzed using the DMRcate package (V1.20.0), with FDR-corrected p values < 0.05 being considered statistically significant.
Pathway analysis
Statistically significant gene lists were used to perform the Kyoto Encyclopedia of Genes and Genomes (KEGG) and Gene Ontology (GO) analyses. The ClusterProfiler package (V3.12.0) was used for pathway enrichment analysis [30].
Pyrosequencing
DNA of purified cells and tissues was extracted using a QIAamp DNA Mini Kit (Cat.No. 51304, Qiagen, Germany) and then bisulfite-modified with the Qiagen EpiTect Bisulfite Kit (Cat.No. 59104, Qiagen, Germany). Primers were designed using PyroMark Assay Design 2.0 (Qiagen, Germany) and presented in Supplementary Table 9. PCR amplification of the target region was performed using the KAPA2G Robust HotStart PCR kit (Cat. No. KK5525, Roche) on the ABI 9700 PCR System (Applied Biosystems, USA) to produce biotinylated PCR amplicons required for the pyrosequencing reaction. After bisulfite treatment and PCR, the substrate mixture, enzyme mixture, and four deoxynucleoside triphosphate (dNTP) (Cat.No. 201900, Qiagen, Germany) were successively added for the reaction on the pyrosequencing detector (PyroMark Q96 ID, Qiagen, Germany). The methylation status of each site was automatically analyzed using the Pyro Q—CpG software (Qiagen, Germany).
Immunohistochemistry
Deparaffinated sections of salivary glands from IgG4-RD patients and controls were immersed in Tris-EDTA antigen retrieval solution, pH 9.0 (Cat.No. G1203, Servicebio, Wuhan, China) and microwaved for antigen retrieval. Then sections were incubated in 3% hydrogen peroxide for 25 min to quench endogenous peroxidase activity, followed by tissues uniformly covered with 3% BSA (Cat.No. GC305010, Servicebio) at room temperature for 30 min. Diluted primary antibody anti-Myelin Basic Protein antibody (dilution 1:400, ab7349, Abcam) was added to sections and incubated overnight at 4°C. After incubating with the HRP-labeled secondary antibody (dilution 1:200, Cat.No. GB23302, Servicebio) at room temperature for 50 min, sections were reacted with DAB (3,3-N-Diaminobenzidine) solution (Cat.No. G1212, Servicebio) for 5 min, and counterstained with hematoxylin (Cat.No. G1004, Servicebio) for 3 min.
Results
Global DNA methylation patterns in peripheral blood and salivary gland tissues from IgG4-RD patients and controls
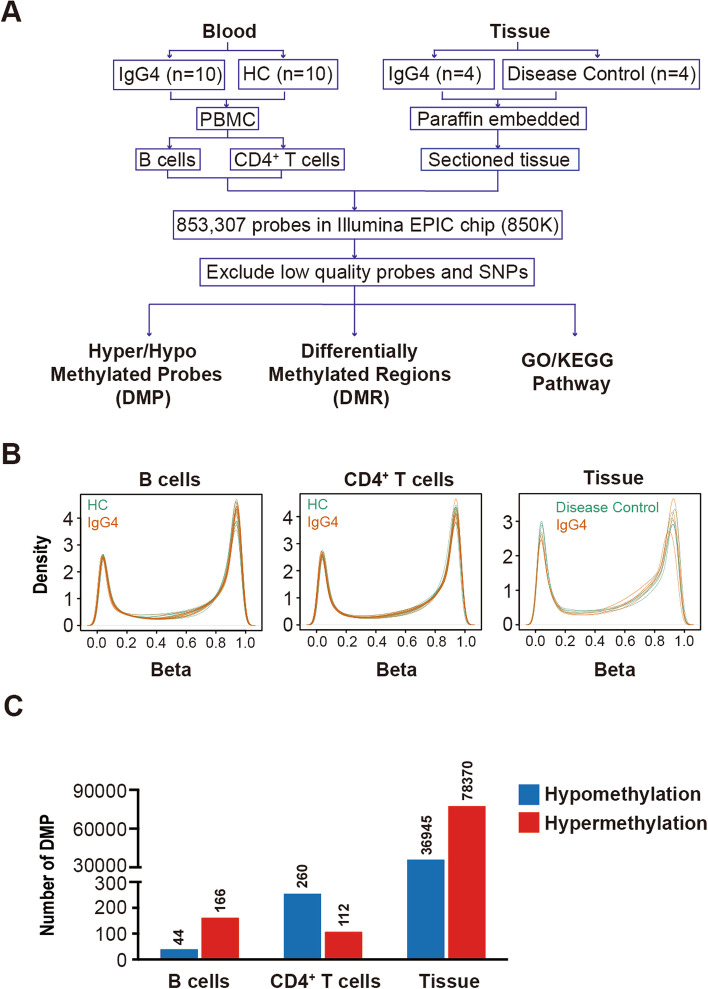
The DNA methylation profile of B cells and CD4+ T cells from 10 HCs and 10 IgG4-RD patients was analyzed. The DNA methylation profile of salivary gland tissues from 4 IgG4-RD patients and 4 controls was also assayed. The workflow is presented in Fig. 1A. The overall DNA methylation beta value displayed similar distribution patterns between the two groups (Fig. 1B). 44 and 166 sites were hypomethylated and hypermethylated, respectively, in B cells (Fig. 1C). Moreover, 260 and 112 sites were hypomethylated and hypermethylated, respectively, in CD4+ T cells from IgG4-RD patients compared with HCs (Fig. 1C). In total, 36,945 sites were hypomethylated and 78,370 sites were hypermethylated in salivary gland tissues of IgG4-RD patients compared with HCs (Fig. 1C).
Fig. 1.
Genome-wide DNA methylation patterns in IgG4-RD patients. A Graphical overview of the study design. B Distribution of methylation beta values in B cells, CD4+ T cells, and salivary gland tissues of IgG4-RD patients and controls. C Number of hypomethylation and hypermethylation probes in B cells, CD4+ T cells, and salivary gland tissues of IgG4-RD patients compared with controls
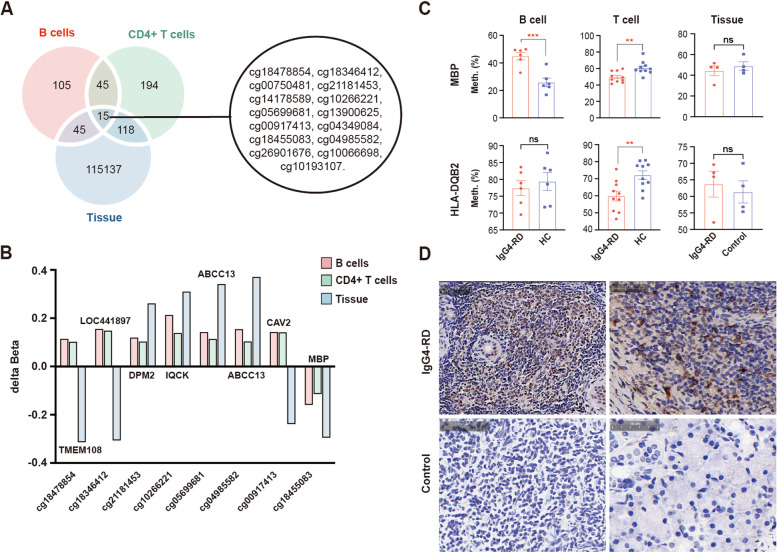
Next, genes and differentially methylated probes (DMPs) of salivary gland tissues were obtained and compared with those of B cells and CD4+ T cells in IgG4-RD patients. As presented in Fig. 2A, 15 DMPs were common to all three cell types. To further identify the methylation status of common genes in the three cell types, the delta beta value was calculated. DPM2 (cg21181453), IQCK (cg10266221), and ABCC13 (cg05699681, cg04985582) were hypermethylated and MBP (cg18455083) was hypomethylated in B cells, CD4+ T cells, and salivary gland tissues of IgG4-RD patients compared with HCs (Fig. 2B). Although TMEM108 (cg18478854), LOC441897 (cg18346412) and CAV2 (cg00917413) were hypermethylated in both B cells and CD4+ T cells, they were hypomethylated in salivary gland tissues of IgG4-RD patients compared with HCs (Fig. 2B).
Fig. 2.
Overlapped DNA methylation profiles in B cells, CD4+ T cells, and salivary gland tissues of IgG4-RD patients. A Venn diagram showing overlapped differentially methylated CpG sites in B cells, CD4+ T cells, and salivary gland tissues of IgG4-RD patients. B Delta beta values of overlapped differentially methylated genes and CpG sites in B cells, CD4+ T cells, and salivary gland tissues of IgG4-RD patients. C The methylation rate of MBP and HLA-DQB2 in B cells, CD4+ T cells, and salivary gland tissues of IgG4-RD patients compared with controls through pyrosequencing. D Immunohistochemistry staining of MBP in salivary gland tissues of two IgG4-RD patients and two controls (scale: 50μm)
We further performed pyrosequencing in B cells, CD4+ T cells, and salivary gland tissues from another cohort consisted of 10 IgG4-RD patients and 10 age-, sex-, and ethnicity-matched healthy blood donors, to validate the methylation state of MBP and HLA-DQB2. In line with the results obtained by DNA methylation chip, the methylation rate of MBP and HLA-DQB2 in CD4+ T cells was significantly lower in IgG4-RD patients than in HCs (Fig. 2C). Although a tendency of lower DNA methylation rate of MBP was observed in salivary gland tissues of IgG4-RD patients compared with controls, no statistical difference was found (Fig. 2C). Immunohistochemical staining showed a high expression of MBP in salivary gland tissues of IgG4-RD patients, which further supported the hypomethylated status of MBP (Fig. 2D).
DNA methylation status of B cells from IgG4-RD patients
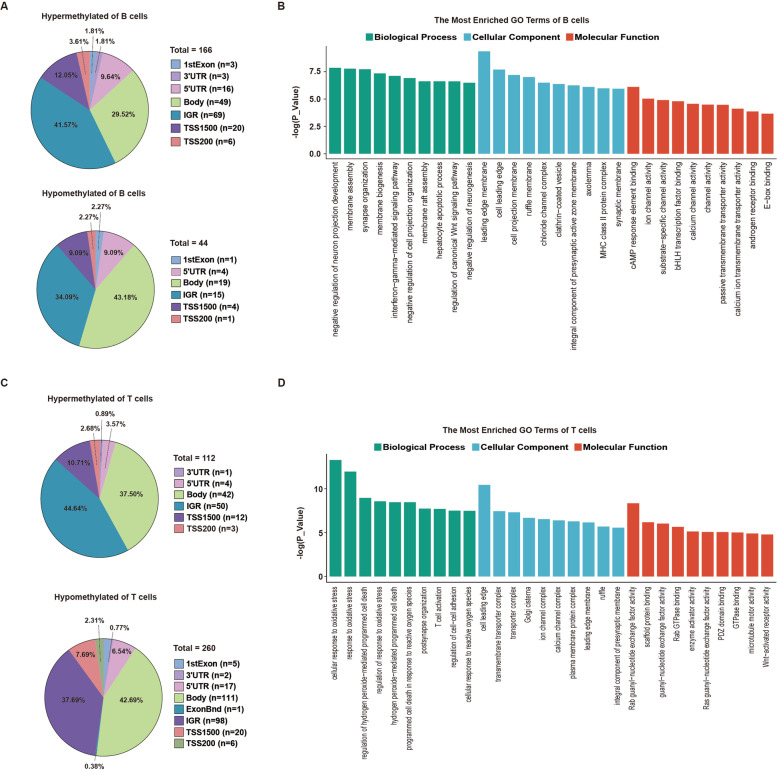
Next, we analyzed B cells from 10 IgG4-RD patients and 10 HCs and identified 166 hypermethylated and 44 hypomethylated probes (Fig. 3A). Among them, most DMPs were present within the intergenic regions (IGRs) or the gene body (Fig. 3A). Table S2 and Table S3 present the top associated hypermethylated and hypomethylated probes with annotated genes, respectively. IQCK, UMODL1, AHDC1, LARS2, RASA3, USP16, BTBD11, CLECL1, LOC441897, and ABCC13 were hypermethylated while TMEM9B, CLIC6, MIR492, ARNTL, TNN, MBP, CDK15, PIGL, HLA-DRB1, and MYO1D were hypomethylated at CpG sites in B cells from IgG4-RD patients.
Fig. 3.
DNA methylation status in B cells and CD4+ T cells of IgG4-RD patients. A Distribution of DMPs in B cells of IgG4-RD patients. B Significant GO categories in B cells of IgG4-RD patients. C Distribution of DMPs in CD4+ T cells of IgG4-RD patients. D Significant GO categories in CD4+ T cells of IgG4-RD patients
GO analysis of the DMPs revealed enrichment for multiple categories. Among them, the interferon-gamma-mediated signaling pathway (related genes: TRIM68, HLA-DRB1, HLA-DQB2, and IFNGR2) and regulation of the canonical Wnt signaling pathway (related genes: STK4, BICC1, KANK1, ARNTL, TNN, and PSMD5) were found to be enriched (Fig. 3B).
DNA methylation status of CD4+ T cells from IgG4-RD patients
Furthermore, we examined CD4+ T cells from 10 IgG4-RD patients and 10 HCs and identified 112 hypermethylated and 260 hypomethylated probes (Fig. 3C). Similar to B cells, most DMPs of CD4+ T cells were present within the IGRs or the gene body (Fig. 3C). Table S4 and Table S5 present the top associated hypermethylated and hypomethylated probes with annotated genes. MAST4, AHDC1, BICC1, USP16, IQCK, LOC727677, MYO1D, LOC441897, CAV2, and TGFBR2 exhibited hypermethylation while TMEM9B, CLIC6, CCS, PAWR, FIP1L1, ARHGAP15, ARHGAP26, PKD1L1, HLA-DRB1, and HLA-DQB2 exhibited hypomethylation at CpG sites in CD4+ T cells from IgG4-RD patients. Pyrosequencing validated the hypomethylated status of MBP and HLA-DQB2 in CD4+ T cells of IgG4-RD patients compared with HCs (Fig. 2C).
GO analysis of the DMPs revealed enrichment for multiple categories. Among them, cellular response to oxidative stress (related genes: FUT8, ARNTL, CFLAR, PTPRK, ETS1, TRAP1, FOXO1, OXR1, PAWR, PRR5L, CCS, PYROXD1, and MIR21), T cell activation (related genes: CLECL1, TGFBR2, ZMIZ1, EOMES, CD3E, TIGIT, CLEC7A, PAWR, KIF13B, CD300A, FZD7, and MIR21), and Wnt-activated receptor activity (related genes: LRP5 and FZD7) were found to be enriched (Fig. 3D).
Identified DMPs and DMRs in salivary gland tissues of IgG4-RD patients
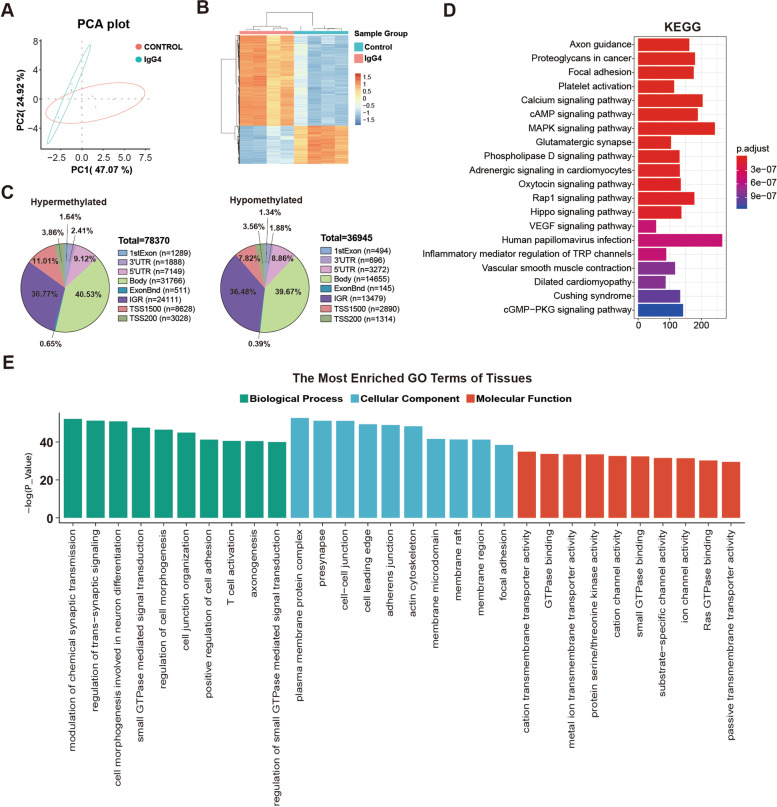
Furthermore, we investigated the DNA methylation status in the affected organ by using the salivary gland tissues of IgG4-RD patients. In total, 78370 hypermethylated and 36945 hypomethylated probes were identified in the salivary gland tissues of IgG4-RD patients compared with controls (Fig. 4A–C). Among them, most DMPs were also present within the IGRs or the gene body (Fig. 4C). Table S6 and Table S7 present the top 25 associated hypermethylated and hypomethylated probes with annotated genes. LOH12CR1, PFKP, ARSB, SNX29, TBCD, STARD13, GALNT10, EIF4E3, VGLL4, TGFBR3, GLG1, RBM34, EPAS1, ITPKB, TBS22D1, SLC44A2, TRIOBP, ACAD11, KCNAB2, PDE4D, RERE, ARID1B, UAP1, LGALS3, and LOC102724933 genes exhibited hypermethylation. CD69, RUNX1, TMEM229B, JAZF1, SLC7A6, FAM69A, EVL, CYTIP, WIPF1, TNFSF8, LOC102724, NCKAP1L, SETBP1, KIAA0748, PPP1R16B, GRAP2, CASP8, ARSG, ITGB2-AS1, MGAT1, CD37, MAP2K2, TNFAIP8L2, and RCSD1 exhibited hypomethylation at CpG sites in the salivary gland tissues of IgG4-RD patients.
Fig. 4.
DNA methylation status in salivary gland tissues of IgG4-RD patients. A Principal components analysis of DMPs. Ellipses show the 95% confidence interval for the distribution of each group. Each dot represents an individual patient. B Supervised hierarchical clustering of DNA methylation in salivary gland tissues of IgG4-RD patients. C Distribution of DMPs in salivary gland tissues of IgG4-RD patients. D KEGG pathway analysis of DMPs in salivary gland tissues of IgG4-RD patients. E Significant GO categories in salivary gland tissues of IgG4-RD patients
The KEGG pathway analysis identified the pathways whose genes from the salivary gland tissues of IgG4-RD patients were differentially methylated: “calcium signaling pathway,” “cAMP signaling pathway,” “MAPK signaling pathway,” “oxytocin signaling pathway,” “Rap1 signaling pathway,” “Hippo signaling pathway,” “VEGF signaling pathway,” and “cGMP-PKG signaling pathway” (Fig. 4D). The GO analysis of the DMPs revealed enrichment for the positive regulation of cell adhesion and T cell activation (Fig. 4E).
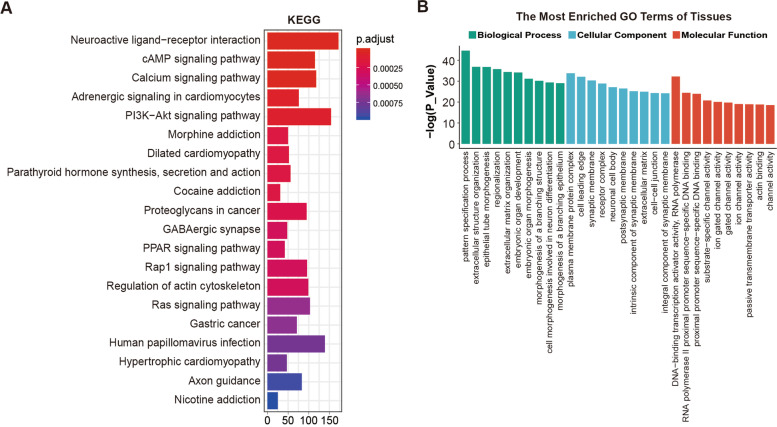
Finally, we characterized the most significant DMRs in the salivary gland tissues of IgG4-RD patients (Table S8). For the better characterization of genes detected in the DMRs, GO and KEGG pathway analyses were performed. The KEGG analyses identified genes involved in the cAMP signaling pathway, calcium signaling pathway, PI3K-Akt signaling pathway, PPAR signaling pathway, Rap1 signaling pathway, and Ras signaling pathway (Fig. 5A). The GO analyses revealed genes most enriched in extracellular matrix organization and ion channer activity (Fig. 5B).
Fig. 5.
Enrichment of DMR-related genes in salivary gland tissues of IgG4-RD patients. A KEGG pathway analysis of DMR-related genes in salivary gland tissues of IgG4-RD patients. B Significant GO categories of DMR-related genes in salivary gland tissues of IgG4-RD patients
Discussion
Currently, epigenetic modifications in IgG4-RD are poorly understood. We here performed a genome-wide DNA methylation analysis of peripheral B cells and CD4+ T cells, as well as salivary gland tissues from IgG4-RD patients. A set of DMPs, DMRs, and involved pathways were identified in IgG4-RD patients compared with HCs. Our observations related to DNA methylation patterns provided new insights into epigenetic modifications of peripheral immune cells and tissue-specific differences possibly involved in IgG4-RD pathogenesis.
MBP (cg18455083) was hypomethylated in CD4+ T cells and salivary gland tissues of IgG4-RD patients. MBP encodes for myelin basic protein, which is the major target of T cells in lesions of multiple sclerosis (MS) patients and the animal model of experimental autoimmune encephalomyelitis [31]. Numerous studies have demonstrated the role of myelin reactivity of T cells in MS [32–34]. A recent study also found that myelin-specific CD8+ T cells exacerbated brain inflammation in central nervous system autoimmunity [35]. Currently, literature about whether myelin has a role in IgG4-RD pathogenesis is lacking. Since CD4+ cytotoxic T lymphocytes have been demonstrated to be clonally expanded in the peripheral blood of IgG4-RD patients and implicated in IgG4-RD pathogenesis [36, 37], exploring whether myelin-specific T cells contribute to IgG4-RD development in our future study will be interesting.
A study by a Japanese group suggested the involvement of the HLA class in IgG4-related pancreatitis [38]. Another study by another Japanese group found six HLA class-associated variants, namely rs1143146 (HLA-A), rs1050716 (HLA-C), c.759_763delCCCCCinsTCCCG (HLA-C), rs1050451 (HLA-C), rs4154112 (HLA-DQB1), and rs1049069 (HLA-DQB1), to be associated with the relapse of IgG4-related pancreatitis [39]. In our study, we also found the hypomethylated status of HLA-DQB2 in CD4+ T cells from IgG4-RD patients.
In the present study, several differentially methylated genes with crucial roles in the immune cell regulation or fibrosis were identified in the salivary gland tissues of IgG4-RD patients. GALNT10 encodes for polypeptide N-acetylgalactosaminyltransferase 10, which is associated with the increased regulatory CD4+ T cell infiltration and decreased granzyme B expression in CD8+ T cells in tumor tissues [40]. TGFBR3 encodes for the TGF-beta type III receptor (TGFBR3), an essential constituent of TGF-beta signaling, that regulates both immune responses and fibrosis [41, 42]. ITPKB encodes for inositol-trisphosphate 3-kinase B, which is important for B cell survival, development, and function as well as T cell maturation in the thymus [43, 44]. LGALS3 encodes for galectin-3, which has been implicated in various autoimmune diseases through modulation of T cell functions [45, 46]. CD69 encodes for CD69, an activated marker for B and T cells that is pivotal in autoimmunity [47]. RUNX1 encodes for Runt-related transcription factor 1 (RUNX1), a transcription factor that regulates fibrosis [48]. CD37 encodes for CD37, a B-cell surface antigen widely expressed on mature B cells [49]. The tetraspanin CD37 was recently found to orchestrate the α4β1 integrin-Akt signaling axis and support longer plasma cell survival [50].
The KEGG pathway analysis revealed enrichment of the “calcium signaling pathway,” “MAPK signaling pathway,” and “Hippo signaling pathway” in the salivary gland tissues of IgG4-RD patients. Chronic calcium signaling in IgE+ B cells restricts plasma cell differentiation and survival [51]. A recent study found that intraglandular injection of IL-4 into mouse submandibular glands could promote fibrogenesis in IgG4-related sialadenitis through ROS-p38 MAPK-p16INK4A [52]. Although literature about the role of the Hippo signaling pathway in IgG4-RD development is lacking, the yes-associated protein and transcriptional coactivator with PDZ-binding motif, which are transcriptional effectors of the Hippo pathway, have been identified as key matrix stiffness-regulated coordinators of fibroblast activation and matrix synthesis [53].
A previous study identified 55 genes in CD4+ T cells and 1628 genes in B cells with differentially methylated CpGs from the peripheral blood of SLE patients. They displayed marked hypomethylation in interferon-regulated genes. Especially, differentially methylated CpGs in B cells were predominantly hypermethylated, and the essential upstream regulators included TNF and EP300 [54]. An integrative analysis of CD4+ and CD8+ T cells from the multiple-autoimmune disease methylation, including Graves’ disease (GD), RA, SLE, and systemic sclerosis (SSc), demonstrated that hypomethylation of IFN-related genes is a common feature of autoimmune diseases in CD4+ T cells [55]. Only 119 probes across 74 genes in purified peripheral CD4+ T cells were detected to be differentially methylated in pSS patients compared with controls, while in B lymphocytes, a total of 6707 differentially methylated probes across 3619 genes were identified. Moreover, methylation alteration in B cells was more frequent in interferon-regulated genes in patients who were autoantibody positive [56]. Hypomethylation of interferon (IFN)-regulated genes in B cells and minor salivary gland biopsies of pSS patients were also identified in another study [18]. In our study, both B cells and CD4+ T cells did not display marked differentially methylated CpGs from the peripheral blood of IgG4-RD patients. The predominant DNA methylation changes were observed in affected tissues of IgG4-RD patients with enriched “MAPK signaling pathway,” “Hippo signaling pathway,” and “VEGF signaling pathway.” Moreover, hypomethylation of IFN-related genes or enriched interferon pathways were not found in peripheral blood or salivary gland tissues of IgG4-RD patients.
The main limitation of this study is a limited sample size, especially tissue specimens, since IgG4-RD is a rare disease. Therefore, longitudinal studies are required to enlarge the sample size, and additional functional assays should be performed to fully elucidate the role of DNA methylation in IgG4-RD. Besides the current analysis of B cells, CD4+ T cells in IgG4-RD is a little general. It would be interesting to analyze the DNA methylation changes in some important subsets of B cells and CD4+ T cells, such as plasma B cells, regulatory B cells (Bregs), CD4+ cytotoxic T cells, Th2, follicular helper T cells (Tfh), and regulatory T cells (Tregs) in IgG4-RD patients in our future study.
Conclusion
In conclusion, this is the first study to identify DNA methylation changes in peripheral B cells and CD4+ T cells, as well as salivary gland tissues from IgG4-RD patients. Our study not only demonstrated aberrant DNA methylation modifications but also emphasized the potential role of DNA methylation changes of MBP and HLA class, as well as other key genes and pathways in IgG4-RD pathogenesis.
Supplementary Information
Additional file 1: Supplementary Table 1. Demographic and clinical characteristics of IgG4-RD patients for genome-wide DNA methylation study.
Additional file 2: Supplementary Table 2. The top 10 hypermethylated CpG sites in B cells of IgG4-RD patients.
Additional file 3: Supplementary Table 3. The top 10 hypomethylated CpG sites in B cells of IgG4-RD patients.
Additional file 4: Supplementary Table 4. The top 10 hypermethylated CpG sites in CD4+ T cells of IgG4-RD patients.
Additional file 5: Supplementary Table 5. The top 10 hypomethylated CpG sites in CD4+ T cells of IgG4-RD patients.
Additional file 6: Supplementary Table 6. The top 25 hypermethylated CpG sites in salivary gland tissues of IgG4-RD patients.
Additional file 7: Supplementary Table 7. The top 25 hypomethylated CpG sites in salivary gland tissues of IgG4-RD patients.
Additional file 8: Supplementary Table 8. The DMRs in salivary gland tissues of IgG4-RD patients.
Additional file 9: Supplementary Table 9. The primers for pyrosequencing.
Acknowledgements
None.
Patient and public involvement
Not applicable.
Abbreviations
- IgG4-RD
Immunoglobulin-G4-related disease
- KEGG
Kyoto Encyclopedia of Genes and Genomes
- PBMCs
Peripheral blood mononuclear cells
- DMRs
Differential methylation regions
- GO
Gene Ontology
- DMPs
Differential methylation probes
- MBP
Myelin basic protein
- MS
Multiple sclerosis
- TGFBR3
TGF-beta type III receptor
- ITPKB
Inositol-trisphosphate 3-kinase B
- RUNX1
Runt-related transcription factor 1
Authors’ contributions
WZ and YF conceived and designed the study. MW, JZ, LP, YZ, XZ, YF, and WZ recruited participants. AW performed experiments with help from YP, YC, JL, ZL, and HL. XW performed data analysis. XW, AW, YF, and WZ wrote the manuscript with input from all authors. WZ acts as the guarantor responsible for the overall content of the present work. All authors approved the final version of the manuscript.
Funding
This work was supported by the National Natural Science Foundation of China (81971545, 81971544, 82071839), National High Level Hospital Clinical Research Funding [2022-PUMCH-A-005], and CAMS Innovation Fund for Medical Sciences (CIFMS) (2021-1-I2M-003).
Availability of data and materials
Data are available upon reasonable request. Not applicable.
Declarations
Ethics approval and consent to participate
This study was approved by the ethics board of Peking Union Medical College Hospital (No.JS-3389). All participants have signed a written informed consent.
Competing interests
The authors declare that they have no competing interests.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Xunyao Wu and Anqi Wang contributed equally.
Yunyun Fei and Wen Zhang contributed equally to this paper.
Contributor Information
Yunyun Fei, Email: feiyunyun@pumch.cn.
Wen Zhang, Email: zhangwen91@sina.com.
References
- 1.Katerji R, Smoller BR. Immunoglobulin-G4-related skin disease. Clin Dermatol. 2021;39(2):283–290. doi: 10.1016/j.clindermatol.2020.10.009. [DOI] [PubMed] [Google Scholar]
- 2.Perugino CA, Stone JH. IgG4-related disease: an update on pathophysiology and implications for clinical care. Nat Rev Rheumatol. 2020;16(12):702–714. doi: 10.1038/s41584-020-0500-7. [DOI] [PubMed] [Google Scholar]
- 3.Umehara H, Okazaki K, Masaki Y, Kawano M, Yamamoto M, Saeki T, et al. A novel clinical entity, IgG4-related disease (IgG4RD): general concept and details. Mod Rheumatol. 2012;22(1):1–14. doi: 10.1007/s10165-011-0508-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Maritati F, Peyronel F, Vaglio A. IgG4-related disease: a clinical perspective. Rheumatology (Oxford) 2020;59(Suppl 3):iii123–iii131. doi: 10.1093/rheumatology/kez667. [DOI] [PubMed] [Google Scholar]
- 5.Deng Q, Luo Y, Chang C, Wu H, Ding Y, Xiao R. The Emerging Epigenetic Role of CD8+T Cells in Autoimmune Diseases: A Systematic Review. Front Immunol. 2019;10:856. doi: 10.3389/fimmu.2019.00856. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Surace AEA, Hedrich CM. The Role of Epigenetics in Autoimmune/Inflammatory Disease. Front Immunol. 2019;10:1525. doi: 10.3389/fimmu.2019.01525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Shu Y, Hu Q, Long H, Chang C, Lu Q, Xiao R. Epigenetic Variability of CD4+CD25+ Tregs Contributes to the Pathogenesis of Autoimmune Diseases. Clin Rev Allergy Immunol. 2017;52(2):260–272. doi: 10.1007/s12016-016-8590-3. [DOI] [PubMed] [Google Scholar]
- 8.Jones PA. Functions of DNA methylation: islands, start sites, gene bodies and beyond. Nat Rev Genet. 2012;13(7):484–492. doi: 10.1038/nrg3230. [DOI] [PubMed] [Google Scholar]
- 9.Liu Y, Aryee MJ, Padyukov L, Fallin MD, Hesselberg E, Runarsson A, et al. Epigenome-wide association data implicate DNA methylation as an intermediary of genetic risk in rheumatoid arthritis. Nat Biotechnol. 2013;31(2):142–147. doi: 10.1038/nbt.2487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Nygaard G, Firestein GS. Restoring synovial homeostasis in rheumatoid arthritis by targeting fibroblast-like synoviocytes. Nat Rev Rheumatol. 2020;16(6):316–333. doi: 10.1038/s41584-020-0413-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Zhu H, Wu LF, Mo XB, Lu X, Tang H, Zhu XW, et al. Rheumatoid arthritis-associated DNA methylation sites in peripheral blood mononuclear cells. Ann Rheum Dis. 2019;78(1):36–42. doi: 10.1136/annrheumdis-2018-213970. [DOI] [PubMed] [Google Scholar]
- 12.Scharer CD, Blalock EL, Mi T, Barwick BG, Jenks SA, Deguchi T, et al. Epigenetic programming underpins B cell dysfunction in human SLE. Nat Immunol. 2019;20(8):1071–1082. doi: 10.1038/s41590-019-0419-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Zhao M, Liu S, Luo S, Wu H, Tang M, Cheng W, et al. DNA methylation and mRNA and microRNA expression of SLE CD4+ T cells correlate with disease phenotype. J Autoimmun. 2014;54:127–136. doi: 10.1016/j.jaut.2014.07.002. [DOI] [PubMed] [Google Scholar]
- 14.Imgenberg-Kreuz J, Carlsson Almlöf J, Leonard D, Alexsson A, Nordmark G, Eloranta ML, et al. DNA methylation mapping identifies gene regulatory effects in patients with systemic lupus erythematosus. Ann Rheum Dis. 2018;77(5):736–743. doi: 10.1136/annrheumdis-2017-212379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Imgenberg-Kreuz J, Sandling JK, Norheim KB, Johnsen SJA, Omdal R, Syvänen AC, et al. DNA Methylation-Based Interferon Scores Associate With Sub-Phenotypes in Primary Sjögren's Syndrome. Front Immunol. 2021;12:702037. doi: 10.3389/fimmu.2021.702037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Konsta OD, Le Dantec C, Charras A, Cornec D, Kapsogeorgou EK, Tzioufas AG, et al. Defective DNA methylation in salivary gland epithelial acini from patients with Sjögren's syndrome is associated with SSB gene expression, anti-SSB/LA detection, and lymphocyte infiltration. J Autoimmun. 2016;68:30–38. doi: 10.1016/j.jaut.2015.12.002. [DOI] [PubMed] [Google Scholar]
- 17.Brække Norheim K, Imgenberg-Kreuz J, Jonsdottir K, Janssen EA, Syvänen AC, Sandling JK, et al. Epigenome-wide DNA methylation patterns associated with fatigue in primary Sjögren's syndrome. Rheumatology (Oxford) 2016;55(6):1074–1082. doi: 10.1093/rheumatology/kew008. [DOI] [PubMed] [Google Scholar]
- 18.Imgenberg-Kreuz J, Sandling JK, Almlöf JC, Nordlund J, Signér L, Norheim KB, et al. Genome-wide DNA methylation analysis in multiple tissues in primary Sjögren's syndrome reveals regulatory effects at interferon-induced genes. Ann Rheum Dis. 2016;75(11):2029–2036. doi: 10.1136/annrheumdis-2015-208659. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Wallace ZS, Naden RP, Chari S, Choi HK, Della-Torre E, Dicaire JF, et al. The 2019 American College of Rheumatology/European League Against Rheumatism classification criteria for IgG4-related disease. Ann Rheum Dis. 2020;79(1):77–87. doi: 10.1136/annrheumdis-2019-216561. [DOI] [PubMed] [Google Scholar]
- 20.Umehara H, Okazaki K, Masaki Y, Kawano M, Yamamoto M, Saeki T, et al. Comprehensive diagnostic criteria for IgG4-related disease (IgG4-RD), 2011. Mod Rheumatol. 2012;22(1):21–30. doi: 10.1007/s10165-011-0571-z. [DOI] [PubMed] [Google Scholar]
- 21.Wallace ZS, Khosroshahi A, Carruthers MD, Perugino CA, Choi H, Campochiaro C, et al. An International Multispecialty Validation Study of the IgG4-Related Disease Responder Index. Arthritis Care Res. 2018;70(11):1671–1678. doi: 10.1002/acr.23543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Morris TJ, Butcher LM, Feber A, Teschendorff AE, Chakravarthy AR, Wojdacz TK, et al. ChAMP: 450k Chip Analysis Methylation Pipeline. Bioinformatics. 2014;30(3):428–430. doi: 10.1093/bioinformatics/btt684. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Paul DS, Teschendorff AE, Dang MA, Lowe R, Hawa MI, Ecker S, et al. Increased DNA methylation variability in type 1 diabetes across three immune effector cell types. Nat Commun. 2016;7:13555. doi: 10.1038/ncomms13555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Northcott PA, Buchhalter I, Morrissy AS, Hovestadt V, Weischenfeldt J, Ehrenberger T, et al. The whole-genome landscape of medulloblastoma subtypes. Nature. 2017;547(7663):311–317. doi: 10.1038/nature22973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Souren NY, Gerdes LA, Lutsik P, Gasparoni G, Beltrán E, Salhab A, et al. DNA methylation signatures of monozygotic twins clinically discordant for multiple sclerosis. Nat Commun. 2019;10(1):2094. doi: 10.1038/s41467-019-09984-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Martino D, Neeland M, Dang T, Cobb J, Ellis J, Barnett A, et al. Epigenetic dysregulation of naive CD4+ T-cell activation genes in childhood food allergy. Nat Commun. 2018;9(1):3308. doi: 10.1038/s41467-018-05608-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Verschoor CP, McEwen LM, Kohli V, Wolfson C, Bowdish DM, Raina P, et al. The relation between DNA methylation patterns and serum cytokine levels in community-dwelling adults: a preliminary study. BMC Genet. 2017;18(1):57. doi: 10.1186/s12863-017-0525-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Lipka DB, Witte T, Toth R, Yang J, Wiesenfarth M, Nöllke P, et al. RAS-pathway mutation patterns define epigenetic subclasses in juvenile myelomonocytic leukemia. Nat Commun. 2017;8(1):2126. doi: 10.1038/s41467-017-02177-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Ren X, Yang X, Cheng B, Chen X, Zhang T, He Q, et al. HOPX hypermethylation promotes metastasis via activating SNAIL transcription in nasopharyngeal carcinoma. Nat Commun. 2017;8:14053. doi: 10.1038/ncomms14053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Wu T, Hu E, Xu S, Chen M, Guo P, Dai Z, et al. clusterProfiler 4.0: A universal enrichment tool for interpreting omics data. Innovation (New York, NY) 2021;2(3):100141. doi: 10.1016/j.xinn.2021.100141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Trager NNM, Butler JT, Harmon J, Mount J, Podbielska M, Haque A, et al. A Novel Aza-MBP Altered Peptide Ligand for the Treatment of Experimental Autoimmune Encephalomyelitis. Mol Neurobiol. 2018;55(1):267–275. doi: 10.1007/s12035-017-0739-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.von Essen MR, Ammitzbøll C, Börnsen L, Sellebjerg F. Assessment of commonly used methods to determine myelin-reactivity of T cells in multiple sclerosis. Clin Immunol. 2021;230:108817. doi: 10.1016/j.clim.2021.108817. [DOI] [PubMed] [Google Scholar]
- 33.Zhao D, Feng F, Zhao C, Wu F, Ma C, Bai Y, et al. Role of perforin secretion from CD8+ T-cells in neuronal cytotoxicity in multiple sclerosis. Neurol Res. 2018;40(1):62–67. doi: 10.1080/01616412.2017.1398371. [DOI] [PubMed] [Google Scholar]
- 34.Cheng W, Ma Y, Gong F, Hu C, Qian L, Huang Q, et al. Cross-reactivity of autoreactive T cells with MBP and viral antigens in patients with MS. Front Biosci (Landmark Ed) 2012;17(5):1648–1658. doi: 10.2741/4010. [DOI] [PubMed] [Google Scholar]
- 35.Wagner CA, Roqué PJ, Mileur TR, Liggitt D, Goverman JM. Myelin-specific CD8+ T cells exacerbate brain inflammation in CNS autoimmunity. J Clin Invest. 2020;130(1):203–213. doi: 10.1172/JCI132531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Mattoo H, Stone JH, Pillai S. Clonally expanded cytotoxic CD4(+) T cells and the pathogenesis of IgG4-related disease. Autoimmunity. 2017;50(1):19–24. doi: 10.1080/08916934.2017.1280029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Mattoo H, Mahajan VS, Maehara T, Deshpande V, Della-Torre E, Wallace ZS, et al. Clonal expansion of CD4(+) cytotoxic T lymphocytes in patients with IgG4-related disease. J Allergy Clin Immunol. 2016;138(3):825–838. doi: 10.1016/j.jaci.2015.12.1330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Oguchi T, Ota M, Ito T, Hamano H, Arakura N, Katsuyama Y, et al. Investigation of susceptibility genes triggering lachrymal/salivary gland lesion complications in Japanese patients with type 1 autoimmune pancreatitis. PLoS One. 2015;10(5):e0127078. doi: 10.1371/journal.pone.0127078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Fujibayashi S, Sasajima J, Goto T, Tanaka H, Kawabata H, Fujii T, et al. A high-throughput sequence analysis of Japanese patients revealed 11 candidate genes associated with type 1 autoimmune pancreatitis susceptibility. Biochem Biophys Rep. 2016;6:76–81. doi: 10.1016/j.bbrep.2016.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Zhang G, Lu J, Yang M, Wang Y, Liu H, Xu C. Elevated GALNT10 expression identifies immunosuppressive microenvironment and dismal prognosis of patients with high grade serous ovarian cancer. Cancer Immunol Immunother. 2020;69(2):175–187. doi: 10.1007/s00262-019-02454-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Yang Z, Xiao Z, Guo H, Fang X, Liang J, Zhu J, et al. Novel role of the clustered miR-23b-3p and miR-27b-3p in enhanced expression of fibrosis-associated genes by targeting TGFBR3 in atrial fibroblasts. J Cell Mol Med. 2019;23(5):3246–3256. doi: 10.1111/jcmm.14211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Schwartze JT, Becker S, Sakkas E, Wujak ŁA, Niess G, Usemann J, et al. Glucocorticoids recruit Tgfbr3 and Smad1 to shift transforming growth factor-β signaling from the Tgfbr1/Smad2/3 axis to the Acvrl1/Smad1 axis in lung fibroblasts. J Biol Chem. 2014;289(6):3262–3275. doi: 10.1074/jbc.M113.541052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Schurmans S, Pouillon V, Maréchal Y. Regulation of B cell survival, development and function by inositol 1,4,5-trisphosphate 3-kinase B (Itpkb) Adv Enzym Regul. 2011;51(1):66–73. doi: 10.1016/j.advenzreg.2010.08.001. [DOI] [PubMed] [Google Scholar]
- 44.Westernberg L, Conche C, Huang YH, Rigaud S, Deng Y, Siegemund S, et al. Non-canonical antagonism of PI3K by the kinase Itpkb delays thymocyte β-selection and renders it Notch-dependent. Elife. 2016;5:e10786. [DOI] [PMC free article] [PubMed]
- 45.de Oliveira FL, Gatto M, Bassi N, Luisetto R, Ghirardello A, Punzi L, et al. Galectin-3 in autoimmunity and autoimmune diseases. Exp Biol Med (Maywood) 2015;240(8):1019–1028. doi: 10.1177/1535370215593826. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Beccaria CG, Amezcua Vesely MC, Fiocca Vernengo F, Gehrau RC, Ramello MC, Tosello Boari J, et al. Galectin-3 deficiency drives lupus-like disease by promoting spontaneous germinal centers formation via IFN-γ. Nat Commun. 2018;9(1):1628. doi: 10.1038/s41467-018-04063-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Gorabi AM, Hajighasemi S, Kiaie N, Gheibi Hayat SM, Jamialahmadi T, Johnston TP, et al. The pivotal role of CD69 in autoimmunity. J Autoimmun. 2020;111:102453. doi: 10.1016/j.jaut.2020.102453. [DOI] [PubMed] [Google Scholar]
- 48.Huang J, Li Y, Xu D, Zhang X, Zhou X. RUNX1 regulates SMAD1 by transcriptionally activating the expression of USP9X, regulating the activation of hepatic stellate cells and liver fibrosis. Eur J Pharmacol. 2021;903:174137. doi: 10.1016/j.ejphar.2021.174137. [DOI] [PubMed] [Google Scholar]
- 49.Xu-Monette ZY, Li L, Byrd JC, Jabbar KJ, Manyam GC, Maria de Winde C, et al. Assessment of CD37 B-cell antigen and cell of origin significantly improves risk prediction in diffuse large B-cell lymphoma. Blood. 2016;128(26):3083–3100. doi: 10.1182/blood-2016-05-715094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.van Spriel AB, de Keijzer S, van der Schaaf A, Gartlan KH, Sofi M, Light A, et al. The tetraspanin CD37 orchestrates the α(4)β(1) integrin-Akt signaling axis and supports long-lived plasma cell survival. Sci Signal. 2012;5(250):ra82. doi: 10.1126/scisignal.2003113. [DOI] [PubMed] [Google Scholar]
- 51.Newman R, Tolar P. Chronic calcium signaling in IgE(+) B cells limits plasma cell differentiation and survival. Immunity. 2021;54(12):2756–2771.e2710. doi: 10.1016/j.immuni.2021.11.006. [DOI] [PubMed] [Google Scholar]
- 52.Min SN, Zhu MQ, Mao XD, Li W, Wei T, Mei M, et al. Contribution of Interleukin-4-Induced Epithelial Cell Senescence to Glandular Fibrosis in IgG4-Related Sialadenitis. Arthritis Rheum. 2022;74(6):1070–1082. doi: 10.1002/art.42052. [DOI] [PubMed] [Google Scholar]
- 53.Liu F, Lagares D, Choi KM, Stopfer L, Marinković A, Vrbanac V, et al. Mechanosignaling through YAP and TAZ drives fibroblast activation and fibrosis. Am J Phys Lung Cell Mol Phys. 2015;308(4):L344–L357. doi: 10.1152/ajplung.00300.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Ulff-Møller CJ, Asmar F, Liu Y, Svendsen AJ, Busato F, Grønbaek K, et al. Twin DNA Methylation Profiling Reveals Flare-Dependent Interferon Signature and B Cell Promoter Hypermethylation in Systemic Lupus Erythematosus. Arthritis Rheum. 2018;70(6):878–890. doi: 10.1002/art.40422. [DOI] [PubMed] [Google Scholar]
- 55.Chen S, Pu W, Guo S, Jin L, He D, Wang J. Genome-Wide DNA Methylation Profiles Reveal Common Epigenetic Patterns of Interferon-Related Genes in Multiple Autoimmune Diseases. Front Genet. 2019;10:223. doi: 10.3389/fgene.2019.00223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Miceli-Richard C, Wang-Renault SF, Boudaoud S, Busato F, Lallemand C, Bethune K, et al. Overlap between differentially methylated DNA regions in blood B lymphocytes and genetic at-risk loci in primary Sjögren's syndrome. Ann Rheum Dis. 2016;75(5):933–940. doi: 10.1136/annrheumdis-2014-206998. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional file 1: Supplementary Table 1. Demographic and clinical characteristics of IgG4-RD patients for genome-wide DNA methylation study.
Additional file 2: Supplementary Table 2. The top 10 hypermethylated CpG sites in B cells of IgG4-RD patients.
Additional file 3: Supplementary Table 3. The top 10 hypomethylated CpG sites in B cells of IgG4-RD patients.
Additional file 4: Supplementary Table 4. The top 10 hypermethylated CpG sites in CD4+ T cells of IgG4-RD patients.
Additional file 5: Supplementary Table 5. The top 10 hypomethylated CpG sites in CD4+ T cells of IgG4-RD patients.
Additional file 6: Supplementary Table 6. The top 25 hypermethylated CpG sites in salivary gland tissues of IgG4-RD patients.
Additional file 7: Supplementary Table 7. The top 25 hypomethylated CpG sites in salivary gland tissues of IgG4-RD patients.
Additional file 8: Supplementary Table 8. The DMRs in salivary gland tissues of IgG4-RD patients.
Additional file 9: Supplementary Table 9. The primers for pyrosequencing.
Data Availability Statement
Data are available upon reasonable request. Not applicable.