Abstract
Background
P450 oxidoreductase (POR) deficiency (PORD) is characterized by congenital adrenal hyperplasia (CAH) and disorders of sex development (DSD) in both sexes. PORD can also associate with skeletal defects. However, the prevalence of these phenotypes is unknown.
Aim
To evaluate the prevalence of CAH, DSD, and infertility of patients with POR gene pathogenic variants by a systematic review of the literature.
Methods
The literature search was performed through PubMed, MEDLINE, Cochrane, Academic One Files, Google Scholar, and Scopus databases. All studies reporting information on CAH, DSD, testicular adrenal rest tumor (TARTs), and fertility in patients with POR gene pathogenic variants were included. Finally, the prevalence of abnormal phenotypes was calculated.
Results
Of the 246 articles initially retrieved, only 48 were included for a total of 119 (46 males and 73 females) patients with PORD. We also included the case of a male patient who consulted us for CAH and TARTs but without DSD. This patient, found to be a carrier of combined heterozygous POR mutation, reached fatherhood spontaneously. All the patients found had CAH. The presence of DSD was found in 65.2%, 82.1%, and 82.1% of patients with compound heterozygosity, homozygosity, or monoallelic heterozygous variants, respectively. The prevalence was significantly higher in females than in males. The prevalence of TARTs in patients with PORD is 2.7%. Only 5 women with PORD became pregnant after assisted reproductive techniques and delivered a healthy baby. Except for the recently reported proband, no other studies focused on male infertility in patients with POR gene variants.
Conclusion
This systematic review of the literature reports the prevalence of CAH, DSD, and TARTs in patients with PORD. The unknown prevalence of POR gene pathogenetic variants and the paucity of studies investigating fertility do not allow us to establish whether PORD is associated with infertility. Further studies on both women and men are needed to clarify this relationship.
Keywords: Congenital adrenal hyperplasia, CAH, Testicular adrenal rest tumor, TART, POR, Sperm parameters, Fertility, Disorders of sex development, DSD
Introduction
Congenital adrenal hyperplasia (CAH) describes a group of autosomal recessive disorders of cortisol biosynthesis with varying levels of severity [1]. Indeed, the clinical phenotype is typically classified as a classic form (the most severe one), and non-classic that is mild or late onset. Disorders of sex development (DSD) depend on the affected gene that cause CAH but also on sex proband. The presence of salt-wasting, postnatal virilization, sex steroid deficiency, hypertension, and other features, such as skeletal defects, are caused by the specific gene that is mutated [1]. Among the long-term complications of CAH, infertility is frequent in both female and male patients [2–8]. Male patients may experience the growth of testicular adrenal rest tumors (TARTs), which can cause intra-testicular compression of efferent seminiferous tubules in turn affecting spermatogenesis [9].
CAHs have a specific hormonal pattern based on the enzymatic dysfunction that causes it. The CYP21A2 genotype is the main, but not the only, determinant of the phenotype in patients with 21α-hydroxylase deficiency (21α-OHD) [10]. In some patients suspected of having a 21α-OHD, no pathogenic variants were found in one or both alleles even after complete sequencing of the CYP21A2 gene [11]. Other patients have pathogenetic variants of the CYP21A2 gene, but the genotype does not fully match the phenotype. These apparent discrepancies between genotype and phenotype suggest the presence of other genetic factors including modifier genes that can modulate the clinical expression of some aspects of 21α-OHD [10].
In humans, P450 oxidoreductase (POR) deficiency (PORD) causes an unusual and rare form of CAH, whose exact incidence is not known [12]. POR is an 82-kDa membrane-bound protein containing 680 residues encoded by a 32-kb gene containing 15 exons mapping on chromosome 7q11.2 [13]. It is necessary for the metabolic activity of P450 cytochrome enzymes including CYP17A1, CYP21A2, CYP19A1, and CYP51A1 [14]. Consequently, PORD can affect the function of these enzymes with different phenotype based on the residual enzymatic activity. As an example, studies on the CYP17A1, the steroidogenic enzyme that catalyzes both 17α-hydroxylase and 17,20 lyase activities [15], show that the levels of some P450 activities are determined, at least in part, by the stereochemistry of the interaction of POR with the cytochrome P450 [13]. In the case of CYP17A1, the 17,20 lyase reaction, but not the 17α-hydroxylase reaction, is very sensitive to this stereochemistry, as shown by three lines of evidence [13]. First, variants of basic residues in the redox-partner binding site of CYP17A1 selectively reduce the 17,20 lyase activity [16]. Second, cytochrome b5 acts as an allosteric factor to promote the interaction of P450c17 with POR, selectively increasing 17,20 lyase activity [17–19]. Third, phosphorylation of CYP17A1 selectively increases the 17,20 lyase activity of CYP17A1 [19–21].
PORD phenotype is characterized by DSD in both sexes and is often associated with skeletal defects [13, 22]. The latter, known as Antley–Bixler syndrome (ABS), is characterized by craniosynostosis, brachycephaly, radio-ulnar or radio-humeral synostosis, bowed femora, arachnodactyly, midface hypoplasia, proptosis, and choanal stenosis. ABS is transmitted with an autosomal recessive mechanism by POR pathogenic variants and with an autosomal dominant mechanism by gain-of-function variants of the fibroblast growth factor receptor 2 (FGFR2) gene [23]. No definitive data are available on TARTs and fertility in male patients with recessive PORD.
Therefore, this study aimed to systematically review the literature to gather all the available information on gender and genotype-related prevalence of CAH, DSD, TARTs, and fertility in patients with heterozygous or homozygous POR gene variants. In the resulting database, we added also the case of a male patient with CAH and TARTs but without DSD. He resulted in being a carrier of a combined heterozygous POR pathogenic variant and achieved fatherhood spontaneously.
Systematic review of the literature
Methods
Sources
Data for the systematic review were independently extracted by C.G. and R.C. A systematic search was performed through PubMed, MEDLINE, Cochrane, Academic One Files, Google Scholar, and Scopus databases, from the beginning of each database through May 22, 2021. The search strategy was based on the combination of the following Medical Subjects Headings (MeSH) terms and keywords, using “AND” between each MeSH search term: “P450 oxidoreductase” AND “congenital adrenal hyperplasia”, “P450 oxidoreductase” AND “DSD”, “P450 oxidoreductase” AND “homozygosity”, “P450 oxidoreductase” AND “heterozygosity”, “P450 oxidoreductase” AND “pregnancy”. Additional manual searches were made using the reference lists of relevant studies. Only articles available in English full-text have been included.
Study selection
All studies that reported the clinical phenotype of patients with POR gene variants were included. In particular, we focused on the presence of CAH, DSD (defined as virilization in female patients, under-masculinization in male patients, and abnormalities of the reproductive system in both sexes), TARTs, and fertility. Review articles and studies on experimental animals were excluded.
Description of the proband
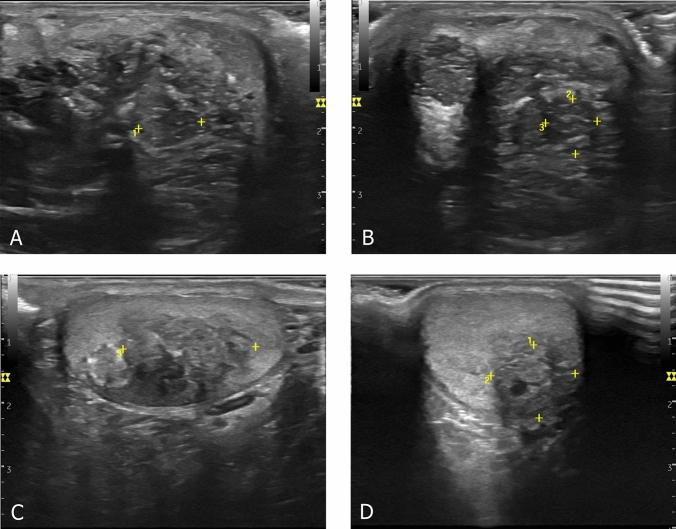
We added to our database the case of a patient not previously described in the literature who resulted in a carrier of compound heterozygous c.1891G > A, p. (Val631Ile) and c.516G > A variant. He had classical salt-wasting CAH, TARTs (Fig. 1), infertility, abnormal sperm parameters (oligoasthenoteratozoospermia, OAT), and extremely elevated ACTH serum levels. He achieved spontaneous paternity after adding dexamethasone 0.25 mg/day to his daily cortisol replacement therapy. This therapeutic arrangement led to the normalization of ACTH serum levels.
Fig. 1.
Ultrasound pictures of the testicular adrenal rest tumors. Upper panels: longitudinal (A) and transversal (B) scans of the right testis. Lower panels: longitudinal (C) and transversal (D) scans of the left testis
Results
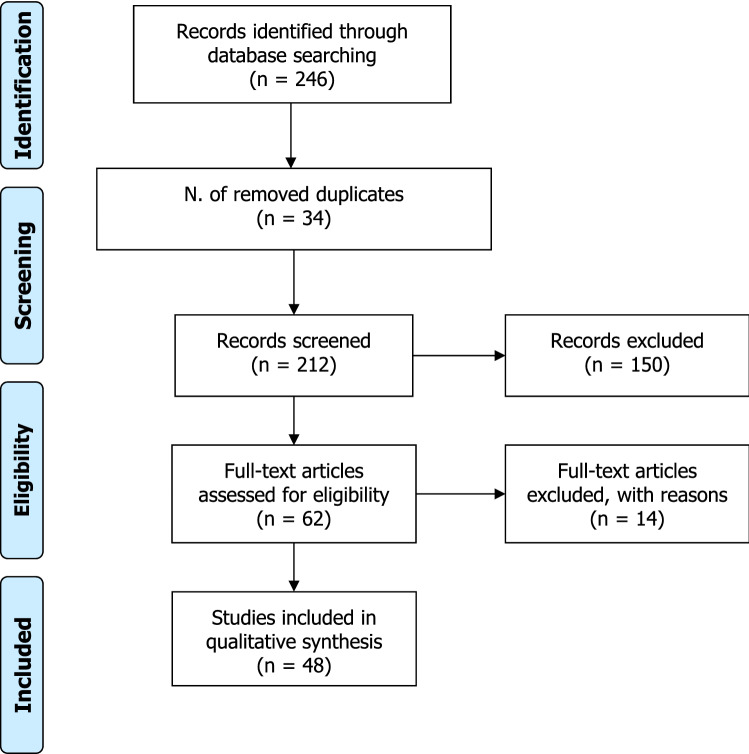
Using the above-mentioned search terms, we found 246 articles. After the exclusion of 34 duplicated records, 212 articles were screened. Thirty-one articles were excluded because the English full-text was not available. Of the remaining articles, 119 were excluded after having read their title and abstract, since they did not satisfy the inclusion criteria. In particular, 10 studies were excluded because performed in vitro and/or on animals, and 24 were excluded because they were reviews. The remaining 62 full-texts were carefully read. Finally, 48 articles matched the inclusion criteria [14, 22, 24–69]. These studies included 119 patients (46 males and 73 females) with PORD (Fig. 2). They had homozygous, combined, or heterozygous POR gene variants in 58.0% (69/119), 32.8% (39/119), and 9.2% (11/119), respectively. These patients are worldwide distributed. The main features of the included studies are summarized in Table 1.
Fig. 2.
Flowchart of the included studies
Table 1.
Summary of the studies included in this systematic review
| Authors | Study design | Sample size (M/F) | Age of patients (years) | POR genotype | CAH | DSD | TARTs | Fertility |
|---|---|---|---|---|---|---|---|---|
| Arlt et al. [24] | Case series and in-vitro study | 1/2 | 2–14 |
Compound Het. (the authors did not indicate patient-specific mutations): c.531 T > G/p.Tyr178Asp c.849G > C/p.Ala284Pro c.1360G > A/p.Arg454His c.1696G > A/Cys566Tyr |
Y (XX) Y (XY) Y (XX) |
Y N Y |
NR N NR |
Unsearched NR NR |
| Fluck et al. [25] | Case series and in-vitro study |
2/2 XX XY XY XX |
5 4.5 1.9 23 |
Het: c.1370G > A(p.R457H)/731 + 1G > A Het: c.1475 T > A(p.V492E) Homo: c.859G > C (p.A287P) Het: c.1706G > A(p.C569Y)/c.1822G > T(V608F) |
Y Y Y Y |
Y Y Y N |
NR N N NR |
Unsearched (all) |
| Adachi et al. [26] | Case series |
1/1 XY XX |
14 11 |
Het: 1329insC/R457H Het:1698insC/R454H |
Y Y |
N Y |
N NR |
Unsearched (all) |
| Wudy et al. [27] | Case report | 1/0 | 18 | Homo: exon 8, GCT > CCT /p.Ala284Pro | Y | N | N | Unsearched |
| Shackleton et al. [28] | Case series |
1/1 XX XY |
At birth At birth |
Het: p.Y178D/p.C566Y Het: p.Y178D/p.C566Y |
Y Y |
Y N |
NR N |
Unsearched (all) |
| Huang et al. [22] | Cohort study (18/19 excluded for lack of data) and in-vitro |
0/1 XX (patient 16) |
NR | Het: c.859G > C (p.A287P)/- | Y | Y | NR | Unsearched |
| Fukami et al. [29] | Case series |
4/6 XY XY XX XY XY XX XX XX XX XX |
26 29 10.9 17.9 17.5 2 6.7 9.5 13.2 15 |
Het: c.1370G > A(p.R457H)/c.1835-1858del(p.L612_W620delinsR) Het: c.1370G > A(p.R457H)/c.1835-1858del(p.L612_W620delinsR) Het: c.1329_1330insC(I444fsX449)/c.1733A > G(p.Y578C) Het: c.1329_1330insC(I444fsX449)/c.1733A > G(p.Y578C) Het: c.15A > G(p.G5G)/c.1370G > A(p.R457H) Homo: c.1370G > A(p.R457H)/ c.1370G > A(p.R457H) Het: c.1370G > A(p.R457H)/- Homo: c.1370G > A(p.R457H)/c.1370G > A(p.R457H) Het: c.1329_1330insC(p.I444fsX449)/c.1370G > A(p.R457H) Homo: c.1370G > A(p.R457H)/c.1370G > A(p.R457H) |
Y Y Y Y Y Y Y Y Y Y |
Y Y Y N N Y Y Y Y Y |
N N NR N N NR NR NR NR NR |
Unsearched (all) |
| Fukami et al. [30] | Case series |
1/2 XY XX XX |
10 5 1 |
Homozygous: R457H/R457H Homozygous: R457H/R457H Homozygous: R457H/R457H |
Y Y Y |
N Y Y |
N NR NR |
Unsearched (all) |
| Homma et al. [31] | Case–control |
5/2 XY XY XY XY XY XX XX |
22 23 15 16 0.3 0.7 12 |
Homo: R457H/R457H Homo: R457H/R457H Het: R457H/Q201X Het: R457H/Q201X Het: R457H/A462_S463insIA Het: R457H/E580Q Het: R457H/Q201X |
Y all |
N N N N Y Y N |
N N N N N NR NR |
|
| Williamson et al. [32] | Case report | 0/1 | NR | Het: p.A278P/p.H604P | Y | Y | NR | Unsearched |
| Hershkovitz et al. [33] | Case series |
4/0 XY XY XY XY |
21 14.6 16.5 At birth |
Homo: c.1697G > A(p.G539R) Homo: c.1697G > A(p.G539R) Homo: c.1697G > A(p.G539R) Homo: c.1697G > A(p.G539R) |
Y Y Y Y |
Y Y Y Y |
N N N N |
Unsearched (all) |
| Nakamura et al. [34] | Case series (2/3 duplicated) | 0/1 | 8 | Het: p.T228I | Y | Y | NR | Unsearched |
| Ko et al. [35] | Case report | 0/1 | 0.58 |
Het: c.1329_1330insC(I444fsX449)/ c.1370G > A (R457H) |
Y | Y | NR | Unsearched |
| Sahakitrungruang et al. [36] | Case series |
1/3 XY XX XX XX |
3 24 21 3.5 |
Het: delGGA651-653(delE217)/859G > C(A287P) Het: 555T > A(N185K)/1730T > G(L577R) Het: 1615G > A(G539R)/1363delC Het: 1615G > A(G539R)/697-698insGAAC |
Y Y Y Y |
Y N Y Y |
N NR NR NR |
Unsearched Infertile Unsearched Unsearched |
| Fukami et al. [37] | Cohort study (23/35 duplicated, 11/12 lack of data) | 1/0 | 13.1 | Het: c.1370G > A(p.R457H)/(-)(DeltaExons 2–13) | Y | Y | N | Unsearched |
| Iijima et al. [38] | Case report | 0/1 | 2.5 | Het: 348delV/R457H | Y | N | NR | Unsearched |
| Idkowiak et al. [39] | Case report and in-vitro study | 1/0 | 9 |
Het:c.32062delG(p.E601fsX12)/ c.32171A > G(p.Y607C) |
Y | Y | N | Unsearched |
| Tomalik-Scharte et al. [40] | Case report | 0/1 | 48 | Homo: c.852G > C | Y | Y | NR | Unsearched |
| But et al. [41] | Case report | 0/1 | 10 | Homo:c.1370G > A/p.R457H | Y | Y | NR | Unsearched |
| Idkowiak et al. [42] | Case series |
2/4 XX XX XX XX XY XY |
12 23 19 16 16 13.5 |
Homo: A287P/A287P Homo: A287P/A287P Het: T142A/Y376LfsZ74 Het: A287P/R223X Het: R457H/Y576X Het: A287P/IVS7-dupT |
Y Y Y Y Y Y |
Y Y N Y N N |
NR NR NR N N |
Unsearched (all) |
| Fluck et al. [43] | Case series |
0/2 XX XX |
At birth At birth |
Homo: c.1196_1204delCCTCGGAGC (p.Pro399_Glu401del) |
Y Y |
Y Y |
NA (all) | Unsearched (all) |
| Herkert et al. [44] | Case report | 0/1 | 19 |
Het:c.2640A > G, p.T142A c.30843dupC, p.Y376LfsX74 |
Y | N | NR | Unsearched |
| Krone et al. [45] | Cohort study (6/30 duplicated, 2/24 no POR mutation) |
9/13 XX XX XY XY XY XX XX XX XX XY XY XX XY XX XX XY XX XX XY XY XX XX |
At birth At birth At birth At birth At birth 12 At birth At birth At birth At birth At birth At birth At birth 24 At birth At birth At birth 18 At birth 12 16 31 |
Het: p.A287/- Het: A287P/ IVS6-2A > T Het: A287P/ IVS6-2A > T Het: A287P/V472AfsX102 Het: Q455RfsX544/IVS7 + 2dupT Homo: A287P/ A287P Het: A287P/DelexU1-1 Het: A287P/IVS8 + 1G > A Homo: A287P/ A287P Homo: A287P/ A287P Het: A287P/G188_V191dup Homo: A287P/ A287P Het: A287P/I444HfsX6 Homo: A287P/ A287P Het: Y87X/ - Het: R457H/Y576X Homo: R498P/R498P Het: Y376LfsX74/T142A Het: A287P/R616X Het: A287P/IVS7 + 2dupT Het: A287P/R223X Het: A287P/Dup ex 2–5 |
Y (all) |
Y N Y Y Y Y N Y Y N Y Y Y Y Y Y Y N N N Y Y |
Unsearched (all) | |
| Boia et al. [46] | Case report | 1/0 | 3 | Homo: c.859G > C | Y | Y | N | Unsearched |
| Guaragna-Filho et al. [47] | Case report | 0/1 | 9 | Het: p.Arg223*/p.Met408Lys | Y | Y | NR | Unsearched |
| Sànchez-Garvìn et al. [48] | Case report | 0/1 | 7 |
Het:c.1615G > A (p.Gly539Arg) p.Gly80Arg |
Y | Y | NR | Unsearched |
| Nakanishi et al. [49] | Case report | 0/1 | At birth | Homo:p.R457H/ p.R457H | Y | Y | NR | Unsearched |
| Oldani et al. [50] | Case report | 0/1 | IUD | Het: c.859G > C(p.Ala287Pro)/c.732A > T | Y | Y | NR | NA |
| Koika et al. [51] | Case report | 1/0 | 36 |
Het.:c.1591_1593delGTC(p.del531Val)/G858C,A259G rs1057868(C/TA503V) rs1057870 (G/AS572S) |
Y | Y | NR | Unsearched |
| Parween et al. [52] | Case report and in-vitro study | 0/1 | At birth | Het:p.L374H/c.5 + 4A > G | Y | Y | NR | Unsearched |
| Bonamichi et al. [53] | Case series | 0/1 | 15 | Homo:p.A287P/p.A287P | Y | Y | NR | Unsearched |
| Tzetis et al. [54] | Case series (2 samples excluded for lack of data) | 0/1 | Fetus | Homo:c.859G > C | Y | Y | NR | Unsearched |
| Woo et al. [55] | Case report | 1/0 | 7 | Het: p.Arg457His/p.Gln555Profs19 | Y | N | N | Unsearched |
| Bai et al. [56] | Case report | 0/1 | 27 | Homo: c.1370G > A(p.R457H)/c.1370G > A(p.R457H) | Y | Y | NR | Unsearched |
| Song et al. [57] | Case report | 0/1 | 28 | Het: c.976 T > G(p.Y326D) | Y | Y | NR | Y (ART) |
| Khadilkar et al. [58] | Case report | 0/1 | 17 | Het: c.430G > A(p.G144S)/c.1265G > A(p.W422X) | Y | Y | NR | Unsearched |
| Oh et al. [59] | Case report | 0/1 | 21 | Het:c.1370G > A (p.Arg457His); c.529G > C (p.Gly177Arg) |
Y (mild phenotype) |
Y (hypoplastic uterus) |
NR | Unsearched |
| Fan et al. [60] | Case series |
5/3 XX XY XY XY XY XX XY XX |
4.2 2.2 1 0.5 3.5 17.8 9.8 12.5 |
Het: c.1370G > A(p.R457H)/c.744C > G(p.Y248X) Het:c.1370G > A(p.R457H)/c.744C > G(p.Y248X) Het:c.1370G > A(p.R457H)/c.1660C > T(p.R554X) Het:c.1370G > T(p.R457L)/c.1820A > G(p.Y607C) Het:c.1370G > A(p.R457H)/c.629A > G(p.D210G) Het:c.1370G > A(p.R457H)/c.517-19_517-10delGGCCCCTGTGinsC Het:c.1370G > A(p.R457H)/c.517-19_517-10delGGCCCCTGTGinsC Homo: c.1370G > A(p.R457H)/c.1370G > A(p.R457H) |
Y Y Y Y Y Y Y Y |
Y Y Y Y Y Y Y Y |
NR N N N N NR N NR |
Unsearched (all) |
| Lee et al. [61] | Case series (1/4 duplicated) |
2/1 XX XY XY |
5.3 3.3 2.6 |
Homo: c.1370G > A(p.R457H)/c.1370G > A(p.R457H) Homo: c.1370G > A(p.R457H)/c.1370G > A(p.R457H) Het:c.1329_1330insC(p.I444Hfs*6)/c.1370G > A(p.R457H) |
Y Y Y |
Y Y Y |
NR N N |
Unsearched (all) |
| Papadakis et al. [62] | Case series |
0/5 XX XX XX XX XX |
30 36 33 38 19 |
Het: c.1249-IG > C(p.?)/c.1324C > T(p.Pro442Ser) Het: c.1825C > T(p.Gln609*)/c.1859G > C(p.Trp620Ser) Het: c.1825C > T(p.Gln609*)/c.1859G > C(p.Trp620Ser) Het: c.1648C > T(p.Arg550Trp)/- Homo: c.859G > C(p.Ala287Pro)/c.859G > C(p.Ala287Pro) |
Y Y Y Y Y |
N N Y N N |
NR (all) |
Y (ART) Y (ART) Unsearched Unsearched Unsearched |
| Parween et al. [14] | Case report |
0/1 XX |
11 | Het:c.73_74delCT(p.L25Ffs*93)/c.1648C > T(p.R550W) | Y | Y | NR | Unsearched |
| Zhang et al. [63] | Case report |
0/1 XX |
35 |
Het:c.1195_1203delCCTCGGAGC(p.399_401delPSE)/ IVS14-1G > G/C |
Y | Y | NR | Y (ART) |
| Aljabri et al. [64] | Case report |
1/0 XY |
2.5 |
Compound: Homo: nonsense Het c.317A > G(p.His106Arg) |
Y | N | N | Unsearched |
| Song et al. [65] | Case report |
0/1 XX |
10.5 |
Compound: Homo c.1370G > A(p.R457H) Het c.2978T > A(p.I993N) |
Y | Y | NR | Unsearched |
| Unal et al. [66] | Case series |
1/1 XX XY |
8.5 1.25 |
Homo:c.929_937delTCTCGGACT(p.Ile310-Ser313delinsThr Homo:c.929_937delTCTCGGACT(p.Ile310-Ser313delinsThr |
Y Y |
Y N |
NR N |
Unsearched (all) |
| Wang et al. [67] | Case series |
0/1 XX |
31 | Het: p.Arg223Ter/p.Tyr67Cys | Y | N | NR | Infertility |
| Rakover et al. [68] | Case series |
0/1 XX |
11.8 | Homo: c.1615G > A(p.G539R) | Y | Y | NR | Unsearched |
| Pan et al. (69) | Case report |
0/1 XX |
29 | Het: c.1370G > A(p.Arg457His)/c.1196_1204del(p.Pro399_Glu401del) | Y | N | NR | Y (ART) |
| Patient from the present study | Case report and literature systematic review | 1/0 | 17 | Het: c.1891G > A, p. (Val631Ile); c.516G > A | Y | N | Y | Y (spontaneous) |
ART assisted reproductive technique, CAH congenital adrenal hyperplasia, DSD disorders of sex development, F female, Het heterozygous, Homo homozygous, M male, N no, NR not reported, TARTs testicular adrenal-rest tumors, Y yes, IUD intrauterine death
Congenital adrenal hyperplasia
The results showed that all the 119 patients with PORD had CAH. All of them showed increased serum 17α-hydroxyprogesterone levels at baseline or after the ACTH-stimulation test. Hence, no difference in the prevalence of CAH among homozygous, combined, or heterozygous POR gene pathogenetic variant was found (Table 1).
Disorder of sex development
DSD was described in 85 out of the 119 patients (71.4%) but there was a different genotype (homozygous or heterozygous) and gender-related distribution. DSD was found in 65.2% (45/69) of patients with compound heterozygous variants, 82.1% (32/39) of patients with homozygous variants, and 72.7% (8/11) of those with monoallelic heterozygous variants. Moreover, DSD occurred more frequently in females affecting 79.5% (58/73) of them and 58.7% (27/46) male patients (p < 0.05).
Testicular adrenal rest tumors
Out of 119 patients retrieved in the literature, ultrasound evaluation was performed only in 15 male probands without evidence of TARTs. Considering the case reported here, the prevalence of TARTs in PORD is, therefore, 6.6% (1/15), taking into account only patients who have undergone testicular ultrasound screening.
Fertility
Considering the higher prevalence of pediatric probands found in the literature, we found an important limitation in the assessment of fertility in patients with PORD. Of these, only 27 probands were over the age of 18 and, therefore, eligible for fertility research. No studies had focused on fertility in patients with PORD until 2017. To date, only five studies of female patients with PORD who have successfully delivered after ART-induced pregnancy have been published [57, 62, 63, 69].
Discussion
By reviewing data from 48 articles including 119 patients from around the world, the present study aims to show the gender- and genotype-related prevalence of CAH, DSD, TARTs, and infertility in patients with heterozygous or homozygous POR gene variants. Genotype–phenotype correlation is sometimes a complex and attempted association and is still a matter of research, considering the most recent studies that have focused on mutations of the CYP21A2 gene [70, 71]. This review allows a better understanding of the PORD phenotype. Our results show that CAH can be caused by both homozygous and heterozygous POR gene variants. Among 119 patients DSD was found in 85 (71.4%), respectively, in 65.2% (45/69) of patients with compound heterozygous variants, 82.1% (32/39) of patients with homozygous variants (82.1%), and 72.7% (8/11) of those with monoallelic heterozygous variants. Furthermore, DSD had a higher frequency in females affecting 79.5% (58/73) and 58.7% (27/46) male patients (p < 0.05), indicating that DSD affects both homozygous and heterozygous carrier patients, although with a higher prevalence in the former.
Specifically analyzing the individual variants, the most frequent are c.859G > C (p.A287P), typically with a higher prevalence of DSD when the heterozygous mutation is found in association with another monoallelic variant (DSD present in 81% of probands with compound heterozygous variants); c.1370G > A(p.R457H), that shows a higher prevalence of DSD when the homozygous variants are found (79%); c.1697G > A(p.G539R), with no difference between homozygous or heterozygous pathogenic variants (DSD present in 100% of probands) (Table 2). All the other detected variants, including the one found in our proband, were found in 1–2 patients (for each variant), so statistical analysis could not be applied (Table 3). According to a recent publication on POR polymorphisms, variants of this gene can influence CAH phenotypical expression acting as a genetic modifier of CYP21A2 defects [72]. Although the literature has so far focused on different allelic mutations on a single gene, we should now consider the coexistence of POR variants and polymorphisms in CAH patients carrying CYP21A2, CYP11B1, CYP17A1, HSD3B2, StAR, or CYP11A1 variants.
Table 2.
Prevalence of disorders of sex development (DSD) in homozygous and heterozygous pathogenetic variants
| Mutation | Homozygous mutations | DSD in homozygous (%) | Heterozygous mutations | DSD in heterozygous (%) | Compound heterozygous mutations | DSD in compound heterozygous mutations (%) |
|---|---|---|---|---|---|---|
| c.859G > C (p.A287P) | 12 | 75 | 3 | 66 | 16 | 81 |
| c.1370G > A(p.R457H) | 14 | 79 | 4 | 25 | 23 | 74 |
| c.1697G > A(p.G539R) | 5 | 100 | 2 | 100 |
Table 3.
Single pathogenetic variant associations of P450 oxidoreductase-associated congenital adrenal hyperplasia
| Mutation | Homozygous mutations | Heterozygous mutations | Composed heterozygous mutations |
|---|---|---|---|
| c.1706G > A(p.C569Y)/c.1822G > T(V608F) | 1 | ||
| GCT > CCT (p.Ala284Pro) | 1 | ||
| p.Y178D/p.C566Y | 2 | ||
| c.1733A > G(p.Y578C) | 2 | ||
| p.T228I | 1 | ||
| 555T > A(N185K)/1730T > G(L577R) | 1 | ||
|
c.32062delG(p.E601fsX12)/ c.32171A > G(p.Y607C) |
1 | ||
| c.852G > C | 1 | ||
| T142A/Y376LfsZ74 | 1 | ||
| p.Pro399_Glu401del | 2 | ||
| c.2640A > G, p.T142A | 2 | ||
| Q455RfsX544/IVS7 + 2dupT | 1 | ||
| Y87X | 1 | ||
| R498P | 1 | ||
| p.Arg223Ter | 2 | ||
| c.1615G > A (p.Gly539Arg) | 1 | ||
| p.del531Val/G858C,A259G rs1057868(C/TA503V) rs1057870 (G/AS572S) | 1 | ||
| p.L374H/c.5 + 4A > G | 1 | ||
| c.976T > G(p.Y326D) | 1 | ||
| c.430G > A(p.G144S)/c.1265G > A(p.W422X) | 1 | ||
| c.1249-IG > C(p.?)/c.1324C > T(p.Pro442Ser) | 1 | ||
| c.1825C > T(p.Gln609*)/c.1859G > C(p.Trp620Ser) | 2 | ||
| c.1648C > T(p.Arg550Trp) | 1 | 1 | |
| c.1195_1203delCCTCGGAGC(p.399_401delPSE)/IVS14-1G > G/C | 1 | ||
| c.317A > G(p.His106Arg) | 1 | ||
| c.929_937delTCTCGGACT(p.Ile310-Ser313delinsThr | 2 | ||
| p. (Val631Ile); c.516G > A | 1 |
This is the first article reporting the presence of TARTs in a patient with CAH due to PORD. So far, TARTs have been described in patients with 21α-OHD and 11ß-hydroxylase deficiency [73, 74]. It is thought that poor hormonal control, leading to high blood levels of ACTH, is an important factor in the pathogenesis of TARTs by inducing hypertrophy and hyperplasia of these adrenal-like cells within the testis [75, 76]. Accordingly, the case herein reported showed that TARTs tend to grow when ACTH levels are elevated. However, TARTs are also found in properly treated patients, whereas some poorly controlled CAH patients never develop TARTs despite they are chronically exposed to elevated ACTH levels [77, 78]. The most plausible explanation for this observation is that in the embryological period aberrant adrenal cells do not nestle in the testes in all males, so the presence of these aberrant adrenal cells within the testis is a prerequisite for the development of TARTs [73]. This ectopic migration does not seem to be related to a specific genotype as so far described. However, the presence of TARTs in the patients described in this article allows us to speculate that also POR gene variants, can cause TART development. Articles on PORD patients do not report the presence of TARTs in any of the patients described.
The present study shows, for the first time, spontaneous fatherhood in a patient with CAH and TARTs due to variants of the POR gene, although paternity must to be genetically proven once the child is born. Female patients, on the other side, may have infertility due to increased androgen secretion or impaired sex steroid production [79], but few cases of successful births by ART-induced pregnancy have been reported [57, 62, 63, 69]. This observation suggests that women with PORD must undergo ART programs to achieve pregnancy.
In conclusion, the unknown incidence of POR gene variants and the poorness of fertility-investigating reports enlighten that it is still unclear whether PORD is associated with human infertility since most of the cases reported so far did not focus on patients’ fertility. Further studies exploring the relationship between POR genotype and fertility are needed. Evidence from the male proband herein reported suggests that spontaneous fatherhood can occur in male patients with PORD but proper management of CAH is needed to reach spontaneous fertility without the need to undergo ART.
Funding
Open access funding provided by Università degli Studi di Catania within the CRUI-CARE Agreement. This research did not receive any specific grant from funding agencies in the public, commercial, or not-profit sectors.
Declarations
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Research involving human participants and/or animals
This study was conducted at the Division of Endocrinology, Metabolic Diseases and Nutrition and Pediatric Endocrinology of the University-Teaching Hospital Policlinico “G. Rodolico”, University of Catania (Catania, Italy). The protocol was approved by the internal Institutional Review Board. The study has been conducted according to the principles expressed in the Declaration of Helsinki.
Informed consent
Informed consent was obtained from the patient after a full explanation of the purpose and nature of all procedures used.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
C. Gusmano and R. Cannarella have contributed equally to this study.
References
- 1.Claahsen-van der Grinten HL, Speiser PW, Ahmed SF, Arlt W, Auchus RJ, Falhammar H, Flück CE, Guasti L, Huebner A, Kortmann B, et al. Congenital adrenal hyperplasia-current insights in pathophysiology, diagnostics, and management. Endocrine Rev. 2022;43(1):91–159. doi: 10.1210/endrev/bnab016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Jääskeläinen J, Kiekara O, Hippeläinen M, Voutilainen R. Pituitary gonadal axis and child rate in males with classical 21-hydroxylase deficiency. J Endocrinol Invest. 2000;23(1):23–27. doi: 10.1007/BF03343671. [DOI] [PubMed] [Google Scholar]
- 3.Stikkelbroeck NM, Otten BJ, Pasic A, Jager GJ, Sweep CG, Noordam K, Hermus AR. High prevalence of testicular adrenal rest tumors, impaired spermatogenesis, and Leydig cell failure in adolescent and adult males with congenital adrenal hyperplasia. J Clin Endocrinol Metab. 2001;86(12):5721–5728. doi: 10.1210/jcem.86.12.8090. [DOI] [PubMed] [Google Scholar]
- 4.Reisch N, Flade L, Scherr M, Rottenkolber M, Pedrosa Gil F, Bidlingmaier M, Wolff H, Schwarz HP, Quinkler M, Beuschlein F, Reincke M. High prevalence of reduced fecundity in men with congenital adrenal hyperplasia. J Clin Endocrinol Metab. 2009;94(5):1665–1670. doi: 10.1210/jc.2008-1414. [DOI] [PubMed] [Google Scholar]
- 5.Arlt W, Willis DS, Wild SH, Krone N, Doherty EJ, Hahner S, Han TS, Carroll PV, Conway GS, Rees DA, et al. United Kingdom Congenital Adrenal Hyperplasia Adult Study Executive (CaHASE). Health status of adults with congenital adrenal hyperplasia: a cohort study of 203 patients. J Clin Endocrinol Metab. 2010;95(11):5110–5121. doi: 10.1210/jc.2010-0917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Falhammar H, Nyström HF, Ekström U, Granberg S, Wedell A, Thorén M. Fertility, sexuality and testicular adrenal rest tumors in adult males with congenital adrenal hyperplasia. Eur J Endocrinol. 2012;166(3):441–449. doi: 10.1530/EJE-11-0828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bouvattier C, Esterle L, Renoult-Pierre P, de la Perrière AB, Illouz F, Kerlan V, Pascal-Vigneron V, Drui D, Christin-Maitre S, et al. Clinical outcome, hormonal status, gonadotropic axis, and testicular function in 219 adult men born with classic 21-hydroxylase deficiency. A French National Survey. J Clin Endocrinol Metab. 2015;100(6):2303–2313. doi: 10.1210/jc.2014-4124. [DOI] [PubMed] [Google Scholar]
- 8.King TF, Lee MC, Williamson EE, Conway GS. Experience in optimizing fertility outcomes in men with congenital adrenal hyperplasia due to 21 hydroxylase deficiency. Clin Endocrinol (Oxf) 2016;84(6):830–836. doi: 10.1111/cen.13001. [DOI] [PubMed] [Google Scholar]
- 9.Rohayem J, Bäumer LM, Zitzmann M, Fricke-Otto S, Mohnike K, Gohlke B, Reschke F, Jourdan C, Müller HL, Dunstheimer D, et al. Semen quality and testicular adrenal rest tumour development in 46, XY congenital adrenal hyperplasia: the importance of optimal hormonal replacement. Eur J Endocrinol. 2021;184(4):487–501. doi: 10.1530/EJE-20-1154. [DOI] [PubMed] [Google Scholar]
- 10.Baronio F, Ortolano R, Menabò S, Cassio A, Baldazzi L, Di Natale V, Tonti G, Vestrucci B, Balsamo A. 46, XX DSD due to androgen excess in monogenic disorders of steroidogenesis: genetic, biochemical, and clinical features. Int J Mol Sci. 2019;20(18):4605. doi: 10.3390/ijms20184605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Reardon W, Smith A, Honour JW, Hindmarsh P, Das D, Rumsby G, Nelson I, Malcolm S, Adès L, Sillence D, et al. Evidence for digenic inheritance in some cases of Antley-Bixler syndrome? J Med Genet. 2000;37(1):26–32. doi: 10.1136/jmg.37.1.26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Baranowski ES, Arlt W, Idkowiak J. Monogenic disorders of adrenal steroidogenesis. Horm Res Paediatr. 2018;89(5):292–310. doi: 10.1159/000488034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Miller WL, Huang N, Pandey AV, Flück CE, Agrawal V. P450 oxidoreductase deficiency: a new disorder of steroidogenesis. Ann NY Acad Sci. 2005;1061:100–108. doi: 10.1196/annals.1336.012. [DOI] [PubMed] [Google Scholar]
- 14.Parween S, Fernández-Cancio M, Benito-Sanz S, Camats N, Rojas Velazquez MN, López-Siguero JP, Udhane SS, Kagawa N, Flück CE, Audí L, et al. Molecular basis of CYP19A1 deficiency in a 46,XX patient with R550W mutation in POR: expanding the PORD phenotype. J Clin Endocrinol Metab. 2020;105(4):dgaa076. doi: 10.1210/clinem/dgaa076. [DOI] [PubMed] [Google Scholar]
- 15.Miller WL, Auchus RJ, Geller DH. The regulation of 17,20 lyase activity. Steroids. 1997;62(1):133–142. doi: 10.1016/s0039-128x(96)00172-9. [DOI] [PubMed] [Google Scholar]
- 16.Geller DH, Auchus RJ, Mendonça BB, Miller WL. The genetic and functional basis of isolated 17,20-lyase deficiency. Nat Genet. 1997;17(2):201–205. doi: 10.1038/ng1097-201. [DOI] [PubMed] [Google Scholar]
- 17.Auchus RJ, Lee TC, Miller WL. Cytochrome b5 augments the 17,20-lyase activity of human P450c17 without direct electron transfer. J Biol Chem. 1998;273(6):3158–3165. doi: 10.1074/jbc.273.6.3158. [DOI] [PubMed] [Google Scholar]
- 18.Geller DH, Auchus RJ, Miller WL. P450c17 mutations R347H and R358Q selectively disrupt 17,20-lyase activity by disrupting interactions with P450 oxidoreductase and cytochrome b5. Mol Endocrinol. 1999;13(1):167–175. doi: 10.1210/mend.13.1.0219. [DOI] [PubMed] [Google Scholar]
- 19.Pandey AV, Miller WL. Regulation of 17,20 lyase activity by cytochrome b5 and by serine phosphorylation of P450c17. J Biol Chem. 2005;280(14):13265–13271. doi: 10.1074/jbc.M414673200. [DOI] [PubMed] [Google Scholar]
- 20.Zhang LH, Rodriguez H, Ohno S, Miller WL. Serine phosphorylation of human P450c17 increases 17,20-lyase activity: implications for adrenarche and the polycystic ovary syndrome. Proc Natl Acad Sci USA. 1995;92(23):10619–10623. doi: 10.1073/pnas.92.23.10619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Pandey AV, Mellon SH, Miller WL. Protein phosphatase 2A and phosphoprotein SET regulate androgen production by P450c17. J Biol Chem. 2003;278(5):2837–2844. doi: 10.1074/jbc.M209527200. [DOI] [PubMed] [Google Scholar]
- 22.Huang N, Pandey AV, Agrawal V, Reardon W, Lapunzina PD, Mowat D, Jabs EW, Van Vliet G, Sack J, Flück CE, et al. Diversity and function of mutations in p450 oxidoreductase in patients with Antley-Bixler syndrome and disordered steroidogenesis. Am J Hum Genet. 2005;76(5):729–749. doi: 10.1086/429417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Miller WL. Mechanisms in endocrinology: rare defects in adrenal steroidogenesis. Eur J Endocrinol. 2018;179(3):R125–R141. doi: 10.1530/EJE-18-0279. [DOI] [PubMed] [Google Scholar]
- 24.Arlt W, Walker EA, Draper N, Ivison HE, Ride JP, Hammer F, Chalder SM, Borucka Mankiewicz M, Hauffa BP, Malunowicz EM, et al. Congenital adrenal hyperplasia caused by mutant P450 oxidoreductase and human androgen synthesis: analytical study. Lancet. 2004;363(9427):2128–2135. doi: 10.1016/S0140-6736(04)16503-3. [DOI] [PubMed] [Google Scholar]
- 25.Fluck CE, Tajima T, Pandey AV, Arlt W, Okuhara K, Verge CF, Jabs EW, Mendonca BB, Fujieda K, Miller WL. Mutant P450 oxidoreductase causes disordered steroidogenesis with and without Antley-Bixler syndrome. Nat Genet. 2004;36(3):228–230. doi: 10.1038/ng1300. [DOI] [PubMed] [Google Scholar]
- 26.Adachi M, Tachibana K, Asakura Y, Yamamoto T, Hanaki K, Oka A. Compound heterozygous mutations of cytochrome P450 oxidoreductase gene (POR) in two patients with Antley-Bixler syndrome. Am J Med Genet A. 2004;128A(4):333–339. doi: 10.1002/ajmg.a.30169. [DOI] [PubMed] [Google Scholar]
- 27.Wudy SA, Hartmann MF, Draper N, Stewart PM, Arlt W. A male twin infant with skull deformity and elevated neonatal 17-hydroxyprogesterone: a prismatic case of P450 oxidoreductase deficiency. Endocr Res. 2004;30(4):957–964. doi: 10.1081/erc-200044174. [DOI] [PubMed] [Google Scholar]
- 28.Shackleton C, Marcos J, Malunowicz EM, Szarras-Czapnik M, Jira P, Taylor NF, Murphy N, Crushell E, Gottschalk M, Hauffa B, et al. Biochemical diagnosis of Antley-Bixler syndrome by steroid analysis. Am J Med Genet A. 2004;128A(3):223–231. doi: 10.1002/ajmg.a.30104. [DOI] [PubMed] [Google Scholar]
- 29.Fukami M, Horikawa R, Nagai T, Tanaka T, Naiki Y, Sato N, Okuyama T, Nakai H, Soneda S, Tachibana K, et al. Cytochrome P450 oxidoreductase gene mutations and Antley-Bixler syndrome with abnormal genitalia and/or impaired steroidogenesis: molecular and clinical studies in 10 patients. J Clin Endocrinol Metab. 2005;90(1):414–426. doi: 10.1210/jc.2004-0810. [DOI] [PubMed] [Google Scholar]
- 30.Fukami M, Hasegawa T, Horikawa R, Ohashi T, Nishimura G, Homma K, Ogata T. Cytochrome P450 oxidoreductase deficiency in three patients initially regarded as having 21-hydroxylase deficiency and/or aromatase deficiency: diagnostic value of urine steroid hormone analysis. Pediatr Res. 2006;59(2):276–280. doi: 10.1203/01.pdr.0000195825.31504.28. [DOI] [PubMed] [Google Scholar]
- 31.Homma K, Hasegawa T, Nagai T, Adachi M, Horikawa R, Fujiwara I, Tajima T, Takeda R, Fukami M, Ogata T. Urine steroid hormone profile analysis in cytochrome P450 oxidoreductase deficiency: implication for the backdoor pathway to dihydrotestosterone. J Clin Endocrinol Metab. 2006;91(7):2643–2649. doi: 10.1210/jc.2005-2460. [DOI] [PubMed] [Google Scholar]
- 32.Williamson L, Arlt W, Shackleton C, Kelley RI, Braddock SR. Linking Antley-Bixler syndrome and congenital adrenal hyperplasia: a novel case of P450 oxidoreductase deficiency. Am J Med Genet A. 2006;140A(17):1797–1803. doi: 10.1002/ajmg.a.31385. [DOI] [PubMed] [Google Scholar]
- 33.Hershkovitz E, Parvari R, Wudy SA, Hartmann MF, Gomes LG, Loewental N, Miller WL. Homozygous mutation G539R in the gene for P450 oxidoreductase in a family previously diagnosed as having 17,20-lyase deficiency. J Clin Endocrinol Metab. 2008;93(9):3584–3588. doi: 10.1210/jc.2008-0051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Nakamura N, Adachi M, Machida J, Okuzumi S. Foot anomalies in Antley-Bixler syndrome: three case reports. J Pediatr Orthop B. 2008;17(5):241–245. doi: 10.1097/BPB.0b013e32830cc35c. [DOI] [PubMed] [Google Scholar]
- 35.Ko JM, Cheon CK, Kim GH, Yoo HW. A case of Antley-Bixler syndrome caused by compound heterozygous mutations of the cytochrome P450 oxidoreductase gene. Eur J Pediatr. 2009;168(7):877–880. doi: 10.1007/s00431-008-0849-0. [DOI] [PubMed] [Google Scholar]
- 36.Sahakitrungruang T, Huang N, Tee MK, Agrawal V, Russell WE, Crock P, Murphy N, Migeon CJ, Miller WL. Clinical, genetic, and enzymatic characterization of P450 oxidoreductase deficiency in four patients. J Clin Endocrinol Metab. 2009;94(12):4992–5000. doi: 10.1210/jc.2009-1460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Fukami M, Nishimura G, Homma K, Nagai T, Hanaki K, Uematsu A, Ishii T, Numakura C, Sawada H, Nakacho M, et al. Cytochrome P450 oxidoreductase deficiency: identification and characterization of biallelic mutations and genotype-phenotype correlations in 35 Japanese patients. J Clin Endocrinol Metab. 2009;94(5):1723–1731. doi: 10.1210/jc.2008-2816. [DOI] [PubMed] [Google Scholar]
- 38.Iijima S, Ohishi A, Ohzeki T. Cytochrome P450 oxidoreductase deficiency with Antley–Bixler syndrome: steroidogenic capacities. J Pediatr Endocrinol Metab. 2009;22(5):469–475. doi: 10.1515/jpem.2009.22.5.469. [DOI] [PubMed] [Google Scholar]
- 39.Idkowiak J, Malunowicz EM, Dhir V, Reisch N, Szarras-Czapnik M, Holmes DM, Shackleton CH, Davies JD, Hughes IA, Krone N, et al. Concomitant mutations in the P450 oxidoreductase and androgen receptor genes presenting with 46, XY disordered sex development and androgenization at adrenarche. J Clin Endocrinol Metab. 2010;95(7):3418–3427. doi: 10.1210/jc.2010-0058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Tomalik-Scharte D, Maiter D, Kirchheiner J, Ivison HE, Fuhr U, Arlt W. Impaired hepatic drug and steroid metabolism in congenital adrenal hyperplasia due to P450 oxidoreductase deficiency. Eur J Endocrinol. 2010;163(6):919–924. doi: 10.1530/EJE-10-0764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.But WM, Lo IF, Shek CC, Tse WY, Lam ST. Ambiguous genitalia, impaired steroidogenesis, and Antley-Bixler syndrome in a patient with P450 oxidoreductase deficiency. Hong Kong Med J. 2010;16(1):59–62. [PubMed] [Google Scholar]
- 42.Idkowiak J, O'Riordan S, Reisch N, Malunowicz EM, Collins F, Kerstens MN, Köhler B, Graul-Neumann LM, Szarras-Czapnik M, Dattani M, Silink M, et al. Pubertal presentation in seven patients with congenital adrenal hyperplasia due to P450 oxidoreductase deficiency. J Clin Endocrinol Metab. 2011;96(3):E453–E462. doi: 10.1210/jc.2010-1607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Fluck CE, Mallet D, Hofer G, Samara-Boustani D, Leger J, Polak M, Morel Y, Pandey AV. Deletion of P399_E401 in NADPH cytochrome P450 oxidoreductase results in partial mixed oxidase deficiency. Biochem Biophys Res Commun. 2011;412(4):572–577. doi: 10.1016/j.bbrc.2011.08.001. [DOI] [PubMed] [Google Scholar]
- 44.Herkert JC, Blaauwwiekel EE, Hoek A, Veenstra-Knol HE, Kema IP, Arlt W, Kerstens MN. A rare cause of congenital adrenal hyperplasia: Antley-Bixler syndrome due to POR deficiency. Neth J Med. 2011;69(6):281–283. [PubMed] [Google Scholar]
- 45.Krone N, Reisch N, Idkowiak J, Dhir V, Ivison HE, Hughes BA, Rose IT, O'Neil DM, Vijzelaar R, Smith MJ, et al. Genotype-phenotype analysis in congenital adrenal hyperplasia due to P450 oxidoreductase deficiency. J Clin Endocrinol Metab. 2012;97(2):E257–267. doi: 10.1210/jc.2011-0640. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Boia ES, Popoiu MC, Puiu M, Stanciulescu CM, David VL. Antley-Bixler syndrome: surgical management of ambiguous genitalia—a case report. Med Princ Pract. 2014;23(4):384–386. doi: 10.1159/000356857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Guaragna-Filho G, Castro CC, Carvalho RR, Coeli FB, Ferraz LF, Petroli RJ, Mello MP, Sewaybricker LE, Lemos-Marini SH, D’Souza-Li LF, et al. 46, XX DSD and Antley-Bixler syndrome due to novel mutations in the cytochrome P450 oxidoreductase gene. Arq Bras Endocrinol Metabol. 2012;56(8):578–585. doi: 10.1590/s0004-27302012000800020. [DOI] [PubMed] [Google Scholar]
- 48.Sánchez-Garvín D, Albaladejo S, Ezquieta B, Corripio R. Disorder of sex development as a diagnostic clue in the first Spanish known newborn with P450 oxidoreductase deficiency. BMJ Case Rep. 2013;2013:bcr2013010251. doi: 10.1136/bcr-2013-010251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Nakanishi K, Yamashita A, Miyamoto T, Takeguchi R, Furuya A, Matsuo K, Tanahashi Y, Kawamura M, Sengoku K. P450 oxidoreductase deficiency with maternal virilization during pregnancy. Clin Exp Obstet Gynecol. 2016;43(6):902–904. [PubMed] [Google Scholar]
- 50.Oldani E, Garel C, Bucourt M, Carbillon L. Prenatal diagnosis of Antley-Bixler syndrome and POR deficiency. Am J Case Reports. 2015;16:882–885. doi: 10.12659/AJCR.895526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Koika V, Armeni AK, Georgopoulos NA. Delayed diagnosis of disorder of sex development (DSD) due to P450 oxidoreductase (POR) deficiency. Hormones (Athens) 2016;15(2):277–282. doi: 10.14310/horm.2002.1679. [DOI] [PubMed] [Google Scholar]
- 52.Parween S, Roucher-Boulez F, Fluck CE, Lienhardt-Roussie A, Mallet D, Morel Y, Pandey AV. P450 oxidoreductase deficiency: loss of activity caused by protein instability from a novel L374H mutation. J Clin Endocrinol Metab. 2016;101(12):4789–4798. doi: 10.1210/jc.2016-1928. [DOI] [PubMed] [Google Scholar]
- 53.Bonamichi BD, Santiago SL, Bertola DR, Kim CA, Alonso N, Mendonca BB, Bachega TA, Gomes LG. Long-term follow-up of a female with congenital adrenal hyperplasia due to P450-oxidoreductase deficiency. Arch Endocrinol Metab. 2016;60(5):500–504. doi: 10.1590/2359-3997000000213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Tzetis M, Konstantinidou A, Sofocleous C, Kosma K, Mitrakos A, Tzannatos C, KitsiouTzeli S. Compound heterozygosity of a paternal submicroscopic deletion and a maternal missense mutation in POR gene: Antley-Bixler syndrome phenotype in three sibling fetuses. Birth Defects Res A Clin Mol Teratol. 2016;106(7):536–541. doi: 10.1002/bdra.23492. [DOI] [PubMed] [Google Scholar]
- 55.Woo H, Ko JM, Shin CH, Yang SW. Two cases of Antley-Bixler syndrome caused by mutations in different genes, FGFR2 and POR. J Genet Med. 2016;13(1):31–35. [Google Scholar]
- 56.Bai Y, Li J, Wang X. Cytochrome P450 oxidoreductase deficiency caused by R457H mutation in POR gene in Chinese: case report and literature review. J Ovarian Res. 2017;10(1):16. doi: 10.1186/s13048-017-0312-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Song T, Wang B, Chen H, Zhu J, Sun H. In vitro fertilization-frozen embryo transfer in a patient with cytochrome P450 oxidoreductase deficiency: a case report. Gynecol Endocrinol. 2017 doi: 10.1080/09513590.2017.1393663. [DOI] [PubMed] [Google Scholar]
- 58.Khadilkar KS, Jagtap V, Lila A, Bandgar T, Shah NS. Cytochrome P450 oxidoreductase deficiency: novel cause of ambiguity with primary amenorrhea. Indian J Endocrinol Metab. 2017;21(2):360–362. doi: 10.4103/2230-8210.202031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Oh J, Song JS, Park JE, Jang SY, Ki CS, Kim DK. A case of Antley-Bixler syndrome with a novel likely pathogenic variant (c.529G>C) in the POR gene. Ann Lab Med. 2017;37(6):559–562. doi: 10.3343/alm.2017.37.6.559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Fan L, Ren X, Song Y, Su C, Fu J, Gong C. Novel phenotypes and genotypes in Antley-Bixler syndrome caused by cytochrome P450 oxidoreductase deficiency: based on the first cohort of Chinese children. Orphanet J Rare Dis. 2019;14(1):299. doi: 10.1186/s13023-019-1283-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Lee Y, Choi JH, Oh A, Kim GH, Park SH, Moon JE, Ko CW, Cheon CK, Yoo HW. Clinical, endocrinological, and molecular features of four Korean cases of cytochrome P450 oxidoreductase deficiency. Ann Pediatr Endocrinol Metab. 2020;25(2):97–103. doi: 10.6065/apem.1938152.076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Papadakis GE, Dumont A, Bouligand J, Chasseloup F, Raggi A, Catteau-Jonard S, Boute-Benejean O, Pitteloud N, Young J, Dewailly D. Non-classic cytochrome P450 oxidoreductase deficiency strongly linked with menstrual cycle disorders and female infertility as primary manifestations. Hum Reprod. 2020;35(4):939–949. doi: 10.1093/humrep/deaa020. [DOI] [PubMed] [Google Scholar]
- 63.Zhang T, Li Z, Ren X, Huang B, Zhu G, Yang W, Jin L. Clinical and genetic analysis of cytochrome P450 oxidoreductase (POR) deficiency in a female and the analysis of a novel POR intron mutation causing alternative mRNA splicing : Overall analysis of a female with POR deficiency. J Assist Reprod Genet. 2020;37(10):2503–2511. doi: 10.1007/s10815-020-01899-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Aljabri A, Alnaim F, Alsaleh Y. Combined homozygous 21 hydroxylase with heterozygous P450 oxidoreductase mutation in a Saudi boy presented with hypertension. BMJ Case Rep. 2020;13(9):e233942. doi: 10.1136/bcr-2019-233942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Song F, Feng S, Shen X, Du M, Yin H, Liu R, Chen X. Next-generation sequencing revealed disease-causing variants in two genes in a patient with combined features of spherocytosis and Antley-Bixler syndrome with genital anomalies and disordered steroidogenesis. Front Genet. 2020;21(11):976. doi: 10.3389/fgene.2020.00976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Unal E, Demiral M, Yıldırım R, Taş FF, Ceylaner S, Özbek MN. Cytochrome P450 oxidoreductase deficiency caused by a novel mutation in the POR gene in two siblings: case report and literature review. Hormones (Athens) 2021;20(2):293–298. doi: 10.1007/s42000-020-00249-z. [DOI] [PubMed] [Google Scholar]
- 67.Wang W, Han R, Yang Z, Zheng S, Li H, Wan Z, Qi Y, Sun S, Ye L, Ning G. Targeted gene panel sequencing for molecular diagnosis of congenital adrenal hyperplasia. J Steroid Biochem Mol Biol. 2021;14(211):105899. doi: 10.1016/j.jsbmb.2021.105899. [DOI] [PubMed] [Google Scholar]
- 68.Rakover YT, Admoni O, Elias-Assad G, London S, Barhoum MN, Ludar H, Almagor T, Zehavi Y, Sultan C, Bertalan R et al (2021) The evolving role of whole-exome sequencing in the management of disorders of sex development. Endocr Connect EC-21-0019.R2 [DOI] [PMC free article] [PubMed]
- 69.Pan P, Zheng L, Chen X, Huang J, Yang D, Li Y. Successful live birth in a Chinese woman with P450 oxidoreductase deficiency through frozen-thawed embryo transfer: a case report with review of the literature. J Ovarian Res. 2021;14(1):22. doi: 10.1186/s13048-021-00778-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Karaoğlan M, Nacarkahya G, Aytaç EH, Keskin M. Challenges of CYP21A2 genotyping in children with 21-hydroxylase deficiency: determination of genotype-phenotype correlation using next generation sequencing in Southeastern Anatolia. J Endocrinol Invest. 2021;44(11):2395–2405. doi: 10.1007/s40618-021-01546-z. [DOI] [PubMed] [Google Scholar]
- 71.Mahmoud RAA, Amr NH, Toaima NN, Kamal TM, Elsedfy HH. Genotypic spectrum of 21-hydroxylase deficiency in an endogamous population. J Endocrinol Invest. 2022;45(2):347–359. doi: 10.1007/s40618-021-01648-8. [DOI] [PubMed] [Google Scholar]
- 72.Pecori Giraldi F, Einaudi S, Sesta A, Verna F, Messina M, Manieri C, Menegatti E, Ghizzoni L. POR polymorphisms are associated with 21 hydroxylase deficiency. J Endocrinol Invest. 2021;44(10):2219–2226. doi: 10.1007/s40618-021-01527-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Dumic M, Duspara V, Grubic Z, Oguic SK, Skrabic V, Kusec V. Testicular adrenal rest tumors in congenital adrenal hyperplasia-cross-sectional study of 51 Croatian male patients. Eur J Pediatr. 2017;176(10):1393–1404. doi: 10.1007/s00431-017-3008-7. [DOI] [PubMed] [Google Scholar]
- 74.El-Maouche D, Arlt W, Merke DP. Congenital adrenal hyperplasia. Lancet. 2017;390(10108):2194–2210. doi: 10.1016/S0140-6736(17)31431-9. [DOI] [PubMed] [Google Scholar]
- 75.Bonaccorsi AC, Adler I, Figueiredo JG. Male infertility due to congenital adrenal hyperplasia: testicular biopsy findings, hormonal evaluation, and therapeutic results in three patients. Fertil Steril. 1987;47:664–670. doi: 10.1016/s0015-0282(16)59119-5. [DOI] [PubMed] [Google Scholar]
- 76.Clark RV, Albertson BD, Munabi A, et al. Steroidogenic enzyme activities, morphology and receptor studies of a testicular adrenal rest in a patient with congenital adrenal hyperplasia. J Clin Endocrinol Metab. 1990;70:1408–1413. doi: 10.1210/jcem-70-5-1408. [DOI] [PubMed] [Google Scholar]
- 77.Cabrera MS, Vogiatzi MG, New MI. Long-term outcome in adult males with classic congenital adrenal hyperplasia. J Clin Endocrinol Metab. 2001;86:3070–3078. doi: 10.1210/jcem.86.7.7668. [DOI] [PubMed] [Google Scholar]
- 78.Stikkelbroeck NMML, Otten BJ, Pasic A, et al. High prevalence of testicular adrenal rest tumours, impaired spermatogenesis, and Leydig cell failure in adolescent and adult males with congenital adrenal hyperplasia. J Clin Endocrinol Metab. 2001;86:5721–5728. doi: 10.1210/jcem.86.12.8090. [DOI] [PubMed] [Google Scholar]
- 79.Gomes LG, Bachega TASS, Mendonca BB. Classic congenital adrenal hyperplasia and its impact on reproduction. Fertil Steril. 2019;111(1):7–12. doi: 10.1016/j.fertnstert.2018.11.037. [DOI] [PubMed] [Google Scholar]