Abstract
Introduction
Antibody formation in transfusion-dependent thalassemia is associated with chronic hemolysis and repeated transfusions. Hemolysis produces heme, which mediates B-cell differentiation into plasma cells and produces antibodies influenced by interleukin-21 (IL-21).
Objective
This study aimed to compare IL-21 levels, plasma cell percentage, and red blood cell antibodies between positive and negative allo-autoantibody thalassemia before and after hemin administration.
Materials and Methods
This research employed a quasi-experimental nonequivalent control group pre-test and post-test design performed from April to November 2021 at Soetomo Academic Hospital in Surabaya, Indonesia. Heparinized blood samples of 5 mL and 4 mL and EDTA blood samples of 3 mL were taken from positive (29 patients) and negative (28 patients) allo-autoantibody thalassemia participants. Hemin 20 µM was added to 5 mL of heparinized blood, incubated for 2 hours, prepared into peripheral blood mononuclear cells (PBMCs), and cultured for 3 days. The percentage of plasma cells (CD38+CD184+) of cultured and uncultured PBMCs was measured by BD FACSCalibur Flow Cytometer. IL-21 levels of plasma and supernatants were measured with Sandwich Enzyme-Linked Immunosorbent Assay by Elabscience. Red blood cell antibodies were detected by QWALYS 3 E.M. Technology. Autoantibodies were determined by the Grifols gel tube method.
Results
IL-21 levels were significantly different in the positive and negative allo-autoantibody thalassemia groups after hemin administration. The percentage of plasma cells in the positive allo-autoantibody group increased significantly after the administration of hemin. The percentage of plasma cells between thalassemia groups was not significantly different before the hemin administration but increased significantly after it. Red blood cell antibodies after the administration of hemin were significantly different in the negative allo-autoantibody group but not significantly different in the positive allo-autoantibody group.
Conclusion
Hemin administration affected IL-21 levels, plasma cell percentage, and antibody formation in positive and negative allo-auto antibody thalassemia.
Keywords: thalassemia disease, IL-21 levels, plasma cells, red blood cells antibody
Introduction
Thalassemia is one of the most common inherited disorders in Indonesia, with diverse mutations ranging from mild to severe. The Eijkman National Molecular Institute reports that the frequency of genes carrying alpha thalassemia in Indonesia is 6–11%, and that of genes carrying beta thalassemia is 3–10%. Approximately 1.5–33% of cases of hemoglobinopathy E are also found in Indonesia.1
Although several nucleic acid–based therapeutic approaches have been investigated in thalassemia, limitations exist to their implementation. Therefore, transfusion remains the recommended treatment option in Indonesia.2 The Thalassemia International Federation classifies thalassemia by transfusion need into transfusion-independent thalassemia and transfusion-dependent thalassemia.3
Repeated transfusions can cause various complications. One of the most common complications is iron overload, which can interfere with the synthesis of vitamin D in the liver.4 Other complications include impaired glucose metabolism5 and changes in cortisol levels that can result in mild depression.6 Other frequent consequences of repeated transfusions are alloimmunization and autoimmunization. The formation of antibodies from alloimmunization and autoimmunization processes can cause acute or delayed hemolytic transfusion reactions. Antibodies that are formed also lead to difficulties in obtaining compatible blood, resulting in delayed transfusion. The requirement of additional pre-transfusion tests and higher costs for tests and patient management comprise further impacts.7
Alloimmunization rates have been reported at 5–30% in thalassemia major in Mediterranean and Southeast Asian countries and 17.5% in Thailand in patients with transfusion-dependent thalassemia.8 Studies on the homozygous beta-thalassemia population who received repeated transfusions found that 87 of 385 patients (22.6%) had autoimmunization.9 Autoimmunization rates in patients with thalassemia have been reported in some countries: 28.8% in Egypt,10 23% in Hong Kong,11 22.8% in Albania,12 and 6.5% in the United States.13 However, the rate of alloimmunization or autoimmunization in Indonesia has still not been recorded.
The high incidence of alloimmunization and autoimmunization in repeated transfusions has prompted various studies to seek solutions to prevent this process. Studies in patients with autoimmune hemolytic anemia (AIHA) found an increase in cytokines including IL-21.14 IL-21 levels were also increased in patients with autoimmune Sjogren’s syndrome compared to patients with non-specific chronic sialadenitis (NSCS) and healthy individuals.15 A twofold increase of IL-21 levels was reported in AIHA-modeled mice compared to the control.16 Osteoarthritis patients had higher IL-21 levels compared to healthy individuals.17 Studies in patients with primary Sjogren’s syndrome, systemic lupus erythematosus, and rheumatoid arthritis showed higher mean and standard deviation (SD) of IL-21 levels compared to healthy individuals (n = 20).18 Elevated IL-21 levels in various autoimmune diseases indicate the role of IL-21 in antibody formation.
Studies in sickle cell disease (SCD) patients with alloantibodies have shown that IL-21 produced by follicular helper (Tfh) cells triggers the differentiation of B-lymphocytes into plasma cells and subsequently produces antibodies.19 Plasma cells express CD38, a type II transmembrane glycoprotein that goes on to form a complex with the B-cell receptor, thus regulating plasma cell homing.20 B-cells serve as antigen-presenting cells (APCs) in the secondary or chronic immune response, which triggers the activation of helper T-cells. Antigen presentation by memory B-cells during the chronic immune reaction phase also occurs in autoimmune diseases.21 Mature B-cells form short- or long-lived plasma cells after stimulation by foreign antigens and self-antigens.22 Long-lived plasma cells (plasmablasts) are found in peripheral blood.23 Plasmablasts may derive from all types of activated B-cells.24 Plasmablast cells have been shown to play a role in antibody formation.25
Hemolysis occurs continuously in thalassemia patients. Heme is produced in large quantities from cells undergoing hemolysis. Macrophages take up hemoglobin, secrete heme into the lymphoid tissue microenvironment, and mediate B-cell activation.26 Studies have shown that heme regulates the immune system through the activity of heme oxygenase-1 (HO-1).27 Heme inhibits the differentiation of B-cells into plasma cells by modulating the enzymatic activity of HO −1 in SCD patients.28 It is not known whether heme affects IL-21 levels in thalassemia patients as in SCD patients, indicating that IL-21 plays a role in the differentiation of B-cells into plasma cells. Hemin was used for experimental studies as a heme preparation that is soluble in dimethyl sulfoxide and then diluted in a culture medium with a physiologically relevant pH.29
This study aimed to compare IL-21 levels, plasma cell percentage, and anti-red blood cell antibodies between allo-autoantibody positive and allo-autoantibody negative thalassemia patients before and after hemin administration.
Materials and Methods
This study took a quasi-experimental approach with a nonequivalent control group pre-test and post-test design. It was carried out from April to November 2021 at the Dr. Soetomo General Academic Hospital in Surabaya, Indonesia. The study was approved by the Local Institutional Human Research Ethics Committee at the Soetomo Academic Hospital (number 0527/LOE/301.4.2/VII/2021) in accordance with the standards laid down in the 1964 Declaration of Helsinki.
Patients underwent standard set blood sample drawing after obtaining informed consent. Two tubes of heparin-blood (5 mL and 4 mL) and 1 tube of EDTA-blood (3 mL) were taken from each participant with prior approval, as evidenced by their signing of informed consent. Informed consent was obtained from parents or legal guardians for research participants under 18 years old. The study participants consisted of 29 patients with positive allo-autoantibody transfusion-dependent thalassemia and 28 negative allo-autoantibody patients.
A total of 5 mL of heparin-blood was prepared for culture, and 4 mL was prepared without culture. A 20M hemin solution was added to the samples for culture, which were then incubated for 2 hours and prepared for the manufacture of peripheral blood mononuclear cells (PBMCs). The samples were added to complete RPMI-1640 medium until the volume reached 10 mL and subsequently divided into 2 falcon tubes of equivalent volume. To each tube was added 5 mL of RPMI medium. Ficoll-Hypaque of d = 1.077 g/mL was slowly applied to the wall of the falcon tube with a Pasteur pipette until 2 layers were formed. Ficoll-Hypaque blood samples that had been mixed with RPMI at a ratio of 1:1 were then centrifuged at 1800 rpm for 30 minutes. Centrifugation formed 5 layers: plasma, PBMC, Ficoll-Hypaque, granulocytes, and red blood cells. The PBMC ring that formed was taken slowly using a Pasteur pipette and inserted into a new falcon tube. The PBMC solution was then washed with 2 mL of PBS by centrifugation at 1500 rpm for 10 minutes twice. PBMC pellets that formed at the bottom of the tube were then resuspended in complete RPMI 1640 medium. Cells were counted using a hematology analyzer.
PBMC cells were set at a density of 106 cells/mL, divided into 3 tubes of 1 mL. Fetal bovine serum, streptomycin, and penicillin were added to each tube, and they were then placed in a 5% CO2 incubator for 3 days.30 PBMC cultures were harvested after 3 days and centrifuged at 1500 rpm for 10 minutes to separate PBMC cells from the supernatant fluid. PBMC cells were washed twice and prepared for a flow cytometry test to determine the plasma cell percentage after the hemin administration.
The 4-mL heparin-blood samples were combined with 4 mL of PBS, then mixed and divided into 2 tubes. The sample was prepared to obtain a PBMC like a 5-mL tube. PBMC suspensions were prepared for the determination of the plasma cell percentage before the hemin administration.
The percentage of plasma cells was determined with flow cytometry, using the BD FACSCalibur Flow Cytometer (BD Biosciences-United States). Fluorochrome labeling was performed on cell suspensions containing 100 L of PBMC from cultured or uncultured PBMC tubes. Each tube was combined with 5 L of PE-labeled anti-CD184 and 5 L of APC-labeled anti-CD38 and then incubated for 30 minutes in the dark. Measurement of the percentage of plasma cells was carried out after incubation.
EDTA-blood samples were centrifuged, and plasma was used for the measurement of IL-21 levels and to screen for red blood cell antibodies. The culture supernatants were tested for IL-21 levels and screened for red blood cell antibodies. Plasma was used as a sample before the administration of hemin, and the culture supernatant was used as specimen after the administration of hemin.
IL-21 levels before and after the administration of hemin were measured using the Sandwich Enzyme-Linked Immunosorbent Assay method (Elabscience-United States). Red blood cell antibody screening was carried out using the QWALYS 3 E.M. Technology (Diagast-France) automatic analyzer with the red blood cell magnetization method. The red blood cell antibody screening test using the Hema-Screen Kit contains 3 panels of cells used to detect irregular antibodies. These consist of the Rh-system (D, C, c, E, e, Cw), the Kell system (K, k, Kpa, Kpb), the Duffy system (Fya, Fyb), the Kidd system (Jka/JK1, Jkb/JK2), the Lewis system (Lea, Leb), the MNS system (M, N, S, s), the P1PK system (P1), and the Lutheran system (Lua, Lub). The presence of autoantibodies was detected by mixing the patient’s red blood cells from the EDTA-blood sample with the patient’s plasma using the gel tube method from the Grifols (DG Gel cards-Spain).
Statistical Analysis
Differences in IL-21 levels, plasma cells, and antibodies formed in each positive and negative allo-autoantibody thalassemia group before and after the hemin administration were tested by paired t-test, if normally distributed, or by the Wilcoxon signed-rank test, if not normally distributed, with a significance level of p < 0.05. An independent t-test and the Mann–Whitney test were used to analyze the difference between the positive allo-autoantibody and negative allo-autoantibody thalassemia groups, with a significance level of p < 0.05. All results analyzed using IBM SPSS Statistics Version 21.
Results
The participant characteristics in this study were the variables of gender, age, blood group, age at first blood transfusion, and frequency of transfusions received. These characteristics did not vary between the negative allo-autoantibody thalassemia group and the positive allo-autoantibody thalassemia group. The negative allo-autoantibody thalassemia group was dominated by blood type O (50%), whereas the positive allo-autoantibody thalassemia group was dominated by blood type B (44.8%). All patients were Rh-positive (Table 1).
Table 1.
Characteristics of Research Participants
| Variable | Negative Allo-Autoantibody Thalassemia | Positive Allo-Autoantibody Thalassemia |
|---|---|---|
| Gender | ||
| Male | 14 (50.0%) | 8 (27.6%) |
| Female | 14 (50.0%) | 21 (72.4%) |
| Age (years) | ||
| 1–5 | 3 (10.7%) | 2 (6.9%) |
| 6–10 | 8 (28.6%) | 4 (13.8%) |
| 11–17 | 9 (32.1%) | 10 (34.5%) |
| Adult (18–39) | 8 (28.6%) | 13 (44.8%) |
| Blood type | ||
| A | 7 (25.0%) | 7 (24.1%) |
| B | 5 (17.9%) | 13 (44.8%) |
| O | 14 (50.0%) | 8 (27.6%) |
| AB | 2 (7.1%) | 1 (3.4%) |
| Rh-positive | 28 (100.0%) | 29 (100.0%) |
| Age at first blood transfusion (years) | ||
| 0–1 | 6 (21.4%) | 2 (6.9%) |
| >1–5 | 7 (25.0%) | 10 (34.5%) |
| >5–10 | 10 (35.7%) | 8 (27.6%) |
| >10–17 | 5 (17.9%) | 5 (17.2%) |
| >17 | 0 (0.0%) | 4 (13.8%) |
| Frequency of blood transfusion | ||
| >20–50 | 10 (35.7%) | 9 (31.0%) |
| >50–100 | 8 (28.6%) | 9 (31.0%) |
| >100–200 | 9 (32.1%) | 7 (24.1%) |
| >200 | 1 (3.6%) | 4 (13.8%) |
Red cell alloantibody and autoantibody test results from 29 samples of the positive allo-autoantibody thalassemia group showed 13 (44.8%) autoimmunized, 4 (13.8%) alloimmunized, and 12 (41.4%) alloimmunized and autoimmunized thalassemia patients.
IL-21 Levels
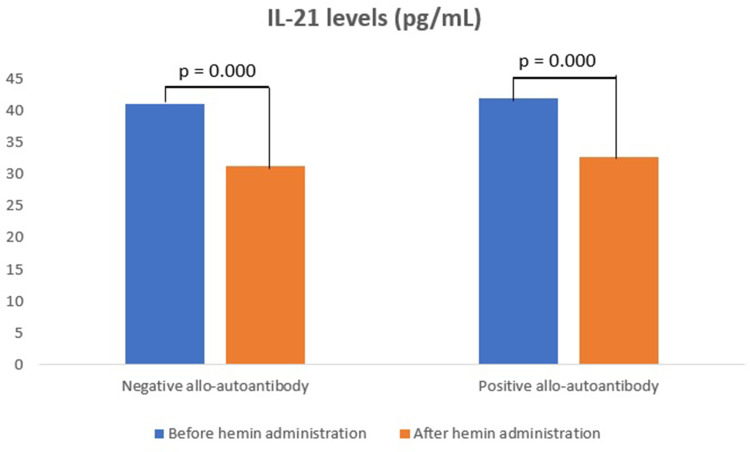
IL-21 levels in the negative allo-autoantibody thalassemia group decreased significantly after the hemin administration, as well as in the positive allo-autoantibody thalassemia group (p = 0.000; p = 0.000). However, changes in IL-21 levels between both groups were not significantly different (p = 0.643). IL-21 levels in the negative allo-autoantibody thalassemia group were not significantly different from the positive allo-autoantibody thalassemia group either before or after the hemin administration (p = 0.756; p = 0.063) (Figure 1).
Figure 1.
IL-21 levels.
Percentage of Plasma Cells
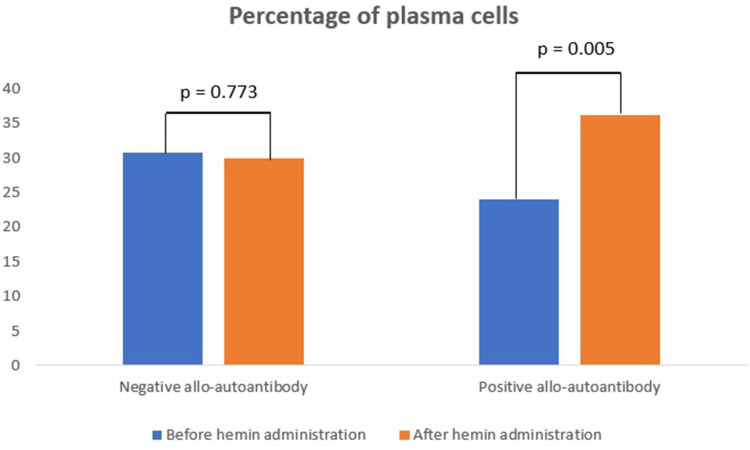
The percentage of plasma cells in negative allo-autoantibody thalassemia was not significantly different after the hemin administration, compared to levels before the hemin administration (p = 0.773). In contrast, the percentage of plasma cells was significantly increased in the positive allo-autoantibody thalassemia group (p = 0.005). Changes in the percentage of plasma cells between both groups varied significantly (p = 0.014).
The percentage of plasma cells in the negative allo-autoantibody thalassemia group was not significantly different from the positive allo-autoantibody thalassemia group before the administration of hemin (p = 0.058) but increased significantly after the administration of hemin (p = 0.200) (Figure 2).
Figure 2.
Percentage of plasma cells.
Red Blood Cell Antibodies
Red blood cell antibodies in previously negative allo-autoantibody thalassemia became positive in some patients after the administration of hemin, resulting in a significant difference (p = 0.000). In addition, red blood cell antibodies were reported positive in the positive allo-autoantibody thalassemia group, although the degree of positivity was not significantly different before and after the hemin administration (p = 0.481). Changes in the formation of red blood cell antibodies between both groups varied significantly (p = 0.002) (Table 2).
Table 2.
Formation of Red Blood Cell Antibodies
| Group | N | Red Blood Cell Antibodies | p (Pre–Post) | Change in Red Blood Cell Antibodies | |
|---|---|---|---|---|---|
| Before Hemin Administration | After Hemin Administration | ||||
| Negative allo-autoantibody thalassemia | 28 | 0.00±0. 00 (0–0) | 1.00±0.00 (0–2) | 0.000** | 1.00±0.00 (0–2) |
| Positive allo-autoantibody thalassemia | 29 | 1.00±1.50 (0–4) | 1.00±0.00 (0–3) | 0.481 | 0.00±1.50 (−4–2) |
| p (between groups) | 0.000* | 0.964 | 0.002* | ||
Notes: Data presented as mean ± SD and median (minimum–maximum). *Significant at p=0.05 (Mann–Whitney test). **Significant at p=0.05 (Wilcoxon signed-rank test).
Discussion
Our study shows that the participants’ age, age at first transfusion, and frequency of transfusion did not vary between the positive allo-autoantibody group and the negative allo-autoantibody group. Another study reported that alloimmunization had a significant relationship with the duration of transfusion and frequency of transfusion. Autoimmunization was also found to develop according to the patient’s age and correlated with the duration and total number of transfusions.31 Blood component, donor, and recipient are several factors that can influence alloimmunization and autoimmunization.32 Another study found that more alloantibodies were detected in patients after receiving transfusions using ≥12 units of red blood cell components.33 Studies in sickle cell disease (SCD) patients also reported that the risk of alloimmunization increased with the number of blood bags used for transfusion.34 A retrospective study analyzing the effect of the number of red blood cell components used for transfusion showed that the frequency of alloimmunization was lower in patients who received transfusions of fewer than 20 blood bags compared to patients who received more than 20.35 The different results obtained in this study might be due to the limited number of participants or other factors that may influence the incidence of alloimmunization or autoimmunization, which were not investigated.
Our study found that IL-21 levels in the positive allo-autoantibody thalassemia group did not differ significantly compared to the negative allo-autoantibody group, either before (p = 0.756) or after the administration of hemin (p = 0.063). Similar results were obtained in a study of beta-thalassemia major patients in Iran, which showed that serum IL-21 levels were not significantly different compared to the control group (healthy individuals).36 No significant difference in IL-21 levels between the thalassemia groups might be due to varying immune responses in each participant, affecting IL-21 levels and resulting in a wide range of IL-21 levels. Activated T-cells induced by IL-6 produce IL-21. IL-21 deficiency interferes with the formation of Th17 cells, which protect against autoimmune diseases.36 IL-21 levels in both thalassemia groups in this study were much lower than those reported by Baharlou et al. Lower IL-21 levels in this study might relate to a protective effect in an attempt to reduce antibody formation. However, this was not further investigated in this study.
IL-21 levels in the negative allo-autoantibody thalassemia group and positive allo-autoantibody thalassemia group decreased significantly after the administration of hemin compared to the levels before (p = 0.000; p = 0.000). The effect of hemin on IL-21 levels has not been widely studied. Animal studies using mouse models susceptible to Toxoplasma gondii infection have shown that CD4 T-cells play an important role in the maintenance of the effector response of CD8 T-cells. Dysfunction in CD8 T-cells is associated with exhausted CD4 T-cells, which causes decreased serum IL-21 levels.37 Research in mice with a respiratory syncytial virus (RSV) infection model showed that RSV infection resulted in follicular helper (Tfh) T-cells unable to produce IL-21 and decreased regulation of IL-21 receptor (IL-21R) expression. Inhibition of IL-21R expression in Tfh cells occurs due to the blockade of programmed cell death ligand-1 (PD-L1) in dendritic cells.38 Through its effects on dendritic cells, hemin can function as an anti-inflammatory (higher T regulator (Treg)/lower T helper −1 (Th1)) in SCD without alloimmunization. However, this effect was not found in the alloimmunized group.39 The administration of exogenous hemin induces Treg cell polarization in purified T-cell/monocyte coculture through the mechanism of action of the monocyte anti-inflammatory heme-degrading enzyme heme oxygenase-1.39 Hemin affects the polarization of the CD4+ T-cell subset toward Tregs and inhibits their differentiation to Th1 and Th17 in non-alloimmunized patients.27 This mechanism causes decreased production of IL-21 by T-cells because Tregs produce more IL-10. The IL-21 cytokines are mostly produced by Tfh and Th17 cells.40 The observed decrease in IL-21 levels after the administration of hemin in our study illustrates that the heme formed from the hemolysis of red blood cells stimulates an immune response to block the formation of antibodies by inhibiting the production of IL-21.
The percentage of plasma cells in the negative allo-auto antibody thalassemia group was not significantly different compared to the positive allo-auto antibody thalassemia group before and after hemin administration. The effect of heme-inhibiting B-cell activation may minimize the risk of alloimmunization in hemolytic disease, such as in SCD patients. Studies on SCD patients with repeated transfusions showed that plasma heme levels between patients with and without alloimmunization were not significantly different in either group before and after transfusion. This indicates no relationship between plasma heme levels and the status of SCD patients with or without alloimmunization either before or after transfusion.28 This further suggests that a transfusion carried out only once does not significantly change the heme level; whether repeated transfusions at a high frequency would affect the heme level and thereby affect the patient’s alloimmunization status is not certain.
The percentage of plasma cells in the negative allo-autoantibody thalassemia group was not significantly different after hemin administration, in contrast to the positive allo-autoantibody thalassemia group, which showed a significant increase after hemin administration. The changes in activated plasma cells between both groups were found to be significantly different. The differentiation of naïve B-cells into plasmablasts between SCD patients without alloimmunization compared to patients with alloimmunization was not significantly different in conditions before heme administration. The differentiation of naïve B-cells into plasmablasts of SCD patients without alloimmunization was less than that of patients with alloimmunization after heme administration. These data indicate that the B-cells of alloimmunized patients are sensitive to the effects of heme and that plasma cell differentiation is increased in the hemolytic state. The inhibitory effect of heme can reduce the risk of alloimmunization of red blood cells, as found in the SCD group without alloimmunization, but not in patients with alloimmunization. Heme inhibits plasma B-cell differentiation by blocking the DOCK8/STAT3 signaling pathway and modulating the enzymatic activity of HO-1.28 Our study found changes in plasma cells in the positive allo-autoantibody thalassemia group, indicating that the positive allo-autoantibody thalassemia group was more sensitive to hemin, in accordance with studies of SCD patients.
Several studies have shown that heme regulates the immune system through the activity of HO-1, an enzyme that degrades heme and inhibits dendritic cell maturation. Studies examining the effect of heme on antibody production in vivo by administering it intraperitoneally to mice have confirmed that it induces HO-1, resulting in reduced IgM production against specific antigens. One study suggests that heme can affect the initiation process of B-cell response activation to antigens, possibly through the inhibition of B-cell proliferation.26 The pattern of HO-1 expression in B-cells and plasma cells is expressed in 2 forms. First, the pattern suggests increased intracellular heme levels in the terminal differentiation of B-cells to plasma cells. Second, heme and HO-1 provide negative feedback via Bach2 and Bach1. Heme and HO-1 modulate humoral immunity by inactivating Bach2 and Bach1, thereby inhibiting Blimp-1 activation. Based on these results, the authors stated that heme modulates B-cell differentiation by binding to Bach2.26
The effect of heme on B-cell differentiation in various lymphoid organs was most likely why no differences in the subset or number of circulating B-cells were detected between alloimmunized and non-alloimmunized SCD patients.41 New strategies are needed to define specific targets of the effector function of B-cells and, if possible, promote regulatory function without altering the function of B-cell-dependent immune surveillance.42 A higher incidence of alloimmunization was found in thalassemic patients than in transfusions in the general population. Patients who received transfusions in infancy or early childhood also had a lower incidence of alloimmunization.43
This study found that red blood cell antibodies in negative allo-autoantibodies thalassemia were negative before hemin administration and became positive after hemin administration in some patients, which resulted in significant changes. Another study, which used experimental rats given hemin, found a significant increase in serum levels of IgG and IgM anti-BSA on the 15th day after intraperitoneal hemin administration.44 This supports the finding that hemin can induce the formation of antibodies in certain concentrations. A study by Li et al reported the increased production of anti-BSA IgG and IgM in immunization in mice and rats after intraperitoneal injection with a formulation containing bovine serum albumin and heme with heme concentrations below 1M. However, heme concentrations between 1M and 5M had no effect, whereas injections of more than 5M showed a suppressive effect. Further research is needed to determine whether this phenomenon also applies to humans. This study used a concentration of 20M, which was directly added to 5 mL of whole blood.44
The positive allo-autoantibody thalassemia group showed positive red blood cell antibody results, but the degree of positivity was not significantly different before and after hemin administration. The changes in the formation of red blood cell antibodies between both groups varied significantly. Research using experimental animal models suggests the possibility of hemin activity as an anti-inflammatory through HO-1 activation, which has the potential to exert an immunoregulatory effect on both innate and adaptive immune responses.45 The underlying immunological process for the loss or persistence of alloantibodies themselves is still poorly understood, and why some alloantibodies to red blood cells last longer are not clear. Alloantibodies that were still detected reflected a strong immune response to foreign antigens.43 Immunological factors were thought to be more influential than the administration of hemin, causing no significant difference between conditions before and after the administration of hemin in the positive allo-autoantibody thalassemia group.
The mechanism of immune suppression by HO-1 occurs through the effect of signals generated by HO-1, which increase the secretion of anti-inflammatory cytokines. The apoptosis of antigen-presenting cells leads to decreased antigen presentation to helper T-cells and other effector T-cells, decreased T-cell differentiation, and increased regulators and anti-inflammatory M2 macrophages. The anti-inflammatory cytokine IL-10 and free heme induce the production of HO-1 to further strengthen the suppression mechanism.44 Another study has suggested that the administration of a heme–albumin complex that specifically carries heme to macrophages in the liver will produce an anti-inflammatory effect. Heme signal as an anti-inflammatory, induced by an increase in intracellular porphyrin concentration in hemolysis, occurred independently.46 The group of patients with negative allo-auto antibody thalassemia after the administration of hemin gave positive results for red blood cell antibodies and decreased IL-21 levels but no difference in the percentage of plasma cells. This discrepancy in the results indicates that another mechanism might be causing the formation of red blood cell antibodies after hemin administration in the negative allo-autoantibody thalassemia group. Hemin can stimulate plasma cells to form alloantibodies, although the difference is not significant. Suppression of IL-21 formation alone is not strong enough to block antibody formation.
We recognize that this study has limitations, requiring further research. The IL-21 levels, plasma cell percentage, and red blood cell antibody screening test were not sufficient to explain the mechanism related to the role of the immune response in forming antibodies in chronic hemolytic conditions in thalassemia patients undergoing repeated transfusions.
Conclusion
IL-21 levels in the negative allo-autoantibody and positive allo-autoantibody thalassemia groups decreased after hemin administration. The percentage of plasma cells in the positive allo-autoantibody thalassemia group was higher after hemin administration than before. An antibody change occurred from negative to positive after the administration of hemin in some patients with negative allo-autoantibody thalassemia, which gave significantly different results. The positive allo-autoantibody thalassemia group did not show an increase in the degree of positivity of red blood cell antibodies, despite the higher percentage of plasma cells. Changes in IL-21 levels, plasma cell percentage, and the formation of red blood cell antibodies after the administration of hemin were found in both thalassemia groups. This indicates that hemolysis affects IL-21 levels, the percentage of plasma cells, and the formation of red blood cell antibodies.
Acknowledgments
We acknowledge all participants for supporting and cooperating in this study.
Funding Statement
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Data Sharing Statement
The statistical data used to support the findings of this study are available from the corresponding author upon request.
Ethical Approval
The study was approved by the Health Research Ethics Committee of Dr. Soetomo General Academic Hospital (no. 0527/LOE/301.4.2/VII/2021).
Author Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis, and interpretation, or in all these areas; took part in drafting, revising, or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Disclosure
The authors declare no competing financial interests or personal relationships that could have influenced the work reported in this article.
References
- 1.Wahidiyat PA, Sari TT, Rahmartani LD, et al. An insight into Indonesian current thalassaemia care and challenges. ISBT Sci Ser. 2020;2020:1–8. [Google Scholar]
- 2.D’Arqom A. Nucleic acid therapy for β-thalassemia. Biologics. 2020;14:95–105. doi: 10.2147/BTT.S265767 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Viprakasit V, Ekwattanakit S. Clinical classification, screening and diagnosis for thalassemia. Hematol Oncol Clin North Am. 2018;32:193–211. doi: 10.1016/j.hoc.2017.11.006 [DOI] [PubMed] [Google Scholar]
- 4.Jazuli MI, Bintoro SUY, Mudjanarko SW. The association between serum ferritin levels and 25 (OH) D levels in adult patients with transfusion dependent thalassemia. J Med Chem Sci. 2022;5:35–41. [Google Scholar]
- 5.Fonda T, Novida H. Management of gestational diabetes mellitus in a beta major thalassemia patient. Curr Intern Med Res Pract Surabaya J. 2021;2:45. doi: 10.20473/cimrj.v2i2.26255 [DOI] [Google Scholar]
- 6.Suryawan A, Ningtiar HW, Irwanto I, et al. Determinant factors of depression in beta major thalassemia children. Folia Med Indones. 2021;57:46. doi: 10.20473/fmi.v57i1.13664 [DOI] [Google Scholar]
- 7.Meulenbroek EM, Wouters D, Zeerleder SS. Lyse or not to lyse: clinical significance of red blood cell autoantibodies. Blood Rev. 2015;29:369–376. doi: 10.1016/j.blre.2015.05.001 [DOI] [PubMed] [Google Scholar]
- 8.Tangvarasittichai S. Impact of alloimmunization on transfusion-dependent patient. Ann Adv Chem. 2017;1:70–82. doi: 10.29328/journal.aac.1001009 [DOI] [Google Scholar]
- 9.Khaled MB, Ouederni M, Sahli N, et al. Predictors of autoimmune hemolytic anemia in beta-thalassemia patients with underlying red blood cells autoantibodies. Blood Cells Mol Dis. 2019;79. doi: 10.1016/j.bcmd.2019.102342 [DOI] [PubMed] [Google Scholar]
- 10.Al-Riyami AZ, Al-Muqbali A, Al-Sudiri S, et al. Risks of red blood cell alloimmunization in transfusion-dependent β-thalassemia in Oman: a 25-year experience of a university tertiary care reference center and a literature review. Transfusion. 2018;58:871–878. doi: 10.1111/trf.14508 [DOI] [PubMed] [Google Scholar]
- 11.Chou ST, Liem RI, Thompson AA. Challenges of alloimmunization in patients with haemoglobinopathies. Br J Haematol. 2012;159:394–404. doi: 10.1111/bjh.12061 [DOI] [PubMed] [Google Scholar]
- 12.Seferi I, Xhetani M, Face M, et al. Frequency and specificity of red cell antibodies in thalassemia patients in Albania. Int J Lab Hematol. 2015;37:569–574. doi: 10.1111/ijlh.12362 [DOI] [PubMed] [Google Scholar]
- 13.Zumberg MS, Procter JL, Lottenberg R, Kitchens CS, Klein HG. Autoantibody formation in the alloimmunized red blood cell recipient. Arch Intern Med. 2015;161:285–290. doi: 10.1001/archinte.161.2.285 [DOI] [PubMed] [Google Scholar]
- 14.Zaninoni A, Fermo E, Vercellati C, et al. Congenital hemolytic anemias: is there a role for the immune system? Front Immunol. 2020;11:1–12. doi: 10.3389/fimmu.2020.01309 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Pontarini E, Murray-Brown WJ, Croia C, et al. Unique expansion of IL-21+ Tfh and Tph cells under control of ICOS identifies Sjögren’s syndrome with ectopic germinal centres and MALT lymphoma. Ann Rheum Dis. 2020;79:1588–1599. doi: 10.1136/annrheumdis-2020-217646 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Gao Y, Jin H, Nan D, et al. The role of T follicular helper cells and T follicular regulatory cells in the pathogenesis of autoimmune hemolytic anemia. Sci Rep. 2019;9:1–11. doi: 10.1038/s41598-019-56365-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Shan Y, Qi C, Liu Y, et al. Increased frequency of peripheral blood follicular helper T cells and elevated serum IL-21 levels in patients with knee osteoarthritis. Mol Med Rep. 2017;15:1095–1102. doi: 10.3892/mmr.2017.6132 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kang KY, Kim HO, Kwok SK, et al. Impact of interleukin-21 in the pathogenesis of primary Sjögren’s syndrome: increased serum levels of interleukin-21 and its expression in the labial salivary glands. Arthritis Res Ther. 2011;13:R179. doi: 10.1186/ar3504 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Godefroy E, Zhong H, Pham P, et al. TIGIT-positive circulating follicular helper T cells display robust B-cell help functions: potential role in sickle cell alloimmunization. Haematologica. 2015;100:1415–1425. doi: 10.3324/haematol.2015.132738 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Khodadadi L, Cheng Q, Radbruch A, et al. The maintenance of memory plasma cells. Front Immunol. 2019;10. doi: 10.3389/fimmu.2019.00721 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Hofmann K, Clauder AK, Manz RA. Targeting B cells and plasma cells in autoimmune diseases. Front Immunol. 2018;9. doi: 10.3389/fimmu.2018.00835 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Dufaud CR, McHeyzer-Williams LJ, McHeyzer-Williams MG. Deconstructing the germinal center, one cell at a time. Curr Opin Immunol. 2017;45:112–118. doi: 10.1016/j.coi.2017.03.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Odendahl M, Mei H, Hoyer BF, et al. Generation of migratory antigen-specific plasma blasts and mobilization of resident plasma cells in a secondary immune response. Blood. 2005;105:1614–1621. doi: 10.1182/blood-2004-07-2507 [DOI] [PubMed] [Google Scholar]
- 24.Chang HD, Tokoyoda K, Hoyer B, et al. Pathogenic memory plasma cells in autoimmunity. Curr Opin Immunol. 2019;61:86–91. doi: 10.1016/j.coi.2019.09.005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Hoyer BF, Moser K, Hauser AE, et al. Short-lived plasmablasts and long-lived plasma cells contribute to chronic humoral autoimmunity in NZB/W mice. J Exp Med. 2004;199:1577–1584. doi: 10.1084/jem.20040168 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Watanabe-Matsui M, Muto A, Matsui T, et al. Heme regulates B-cell differentiation, antibody class switch, and heme oxygenase-1 expression in B cells as a ligand of Bach2. Blood. 2011;117:5438–5448. doi: 10.1182/blood-2010-07-296483 [DOI] [PubMed] [Google Scholar]
- 27.Zhong H, Bao W, Friedman D, et al. Hemin controls T cell polarization in sickle cell alloimmunization. J Immunol. 2014;193:102–110. doi: 10.4049/jimmunol.1400105 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Pal M, Bao W, Wang R, et al. Hemolysis inhibits humoral B-cell responses and modulates alloimmunization risk in patients with sickle cell disease. Blood. 2021;137:269–280. doi: 10.1182/blood.2020008511 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Roumenina LT, Rayes J, Lacroix-Desmazes S, et al. Heme: modulator of plasma systems in hemolytic diseases. Trends Mol Med. 2016;22:200–213. doi: 10.1016/j.molmed.2016.01.004 [DOI] [PubMed] [Google Scholar]
- 30.Godoy-Ramirez K, Franck K, Mahdavifar S, et al. Optimum culture conditions for specific and nonspecific activation of whole blood and PBMC for intracellular cytokine assessment by flow cytometry. J Immunol Methods. 2004;292:1–15. doi: 10.1016/j.jim.2004.04.028 [DOI] [PubMed] [Google Scholar]
- 31.El-Beshlawy A, Salama AA, El-Masry MR, et al. A study of red blood cell alloimmunization and autoimmunization among 200 multitransfused Egyptian β thalassemia patients. Sci Rep. 2020;10. doi: 10.1038/s41598-020-78333-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Wang LY, Liang DC, Liu HC, et al. Alloimmunization among patients with transfusion-dependent thalassemia in Taiwan. Transfus Med. 2006;16:200–203. doi: 10.1111/j.1365-3148.2006.00656.x [DOI] [PubMed] [Google Scholar]
- 33.Dogra A, Sidhu M, Kapoor R, et al. Study of red blood cell alloimmunization in multitransfused thalassemic children of Jammu region. Asian J Transfus Sci. 2015;9:78–81. doi: 10.4103/0973-6247.150958 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Samarah F, Srour MA, Yaseen D, et al. Frequency of red blood cell alloimmunization in patients with sickle cell disease in Palestine. Adv Hematol. 2018;2018:1–7. doi: 10.1155/2018/5356245 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Romphruk AV, Simtong P, Butryojantho C, et al. The prevalence, alloimmunization risk factors, antigenic exposure, and evaluation of antigen-matched red blood cells for thalassemia transfusions: a 10-year experience at a tertiary care hospital. Transfusion. 2019;59:177–184. doi: 10.1111/trf.15002 [DOI] [PubMed] [Google Scholar]
- 36.Baharlou R, Davami MH, Ahmadi Vasmehjani A, et al. Increased IL-17 and TGF-β serum levels in peripheral blood of patients with β-thalassemia major: implication for continual transfusions role in T helper 17-mediated proinflammatory responses. Turkish J Med Sci. 2016;46:749–755. doi: 10.3906/sag-1409-57 [DOI] [PubMed] [Google Scholar]
- 37.Moretto MM, Hwang SJ, Khan IA. Downregulated IL-21 response and T follicular helper cell exhaustion correlate with compromised CD8 T cell immunity during chronic toxoplasmosis. Front Immunol. 2017;8. doi: 10.3389/fimmu.2017.01436 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Gassen RB, Fazolo T, Nascimento de Freitas D, et al. IL-21 treatment recovers follicular helper T cells and neutralizing antibody production in respiratory syncytial virus infection. Immunol Cell Biol. 2021;99:309–322. doi: 10.1111/imcb.12418 [DOI] [PubMed] [Google Scholar]
- 39.Yazdanbakhsh K. Immunoregulatory networks in sickle cell alloimmunization. Hematology. 2016;2016:457–461. doi: 10.1182/asheducation-2016.1.457 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Patente TA, Pinho MP, Oliveira AA, et al. Human dendritic cells: their heterogeneity and clinical application potential in cancer immunotherapy. Front Immunol. 2019;10:1–18. doi: 10.3389/fimmu.2019.00001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Bao W, Zhong H, Manwani D, et al. Regulatory B-cell compartment in transfused alloimmunized and non-alloimmunized patients with sickle cell disease. Am J Hematol. 2013;88:736–740. doi: 10.1002/ajh.23488 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Musette P, Bouaziz JD. B cell modulation strategies in autoimmune diseases: new concepts. Front Immunol. 2018;9:1–5. doi: 10.3389/fimmu.2018.00622 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Tormey CA, Hendrickson JE. Transfusion-related red blood cell alloantibodies: induction and consequences. Blood. 2019;133:1821–1830. doi: 10.1182/blood-2018-08-833962 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Li G, Xue H, Fan Z, et al. Impact of heme on specific antibody production in mice: promotive, inhibitive or null outcome is determined by its concentration. Heliyon. 2017;3:e00303. doi: 10.1016/j.heliyon.2017.e00303 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Martins R, Knapp S. Heme and hemolysis in innate immunity: adding insult to injury. Curr Opin Immunol. 2018;50:14–20. doi: 10.1016/j.coi.2017.10.005 [DOI] [PubMed] [Google Scholar]
- 46.Pfefferlé M, Ingoglia G, Schaer CA, et al. Acute hemolysis and heme suppress anti-CD40 antibody-induced necro-inflammatory liver disease. Front Immunol. 2021;12:1–12. doi: 10.3389/fimmu.2021.680855 [DOI] [PMC free article] [PubMed] [Google Scholar]