Abstract
Research question
What is the effect of mRNA severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) vaccination in young oocyte donors in terms of ovarian response to stimulation, fertilization rate, embryo development and clinical outcomes in recipients?
Design
This retrospective, multicentre cohort study evaluated 115 oocyte donors who had undergone at least two ovarian stimulation protocols (before and after complete SARS-CoV-2 vaccination) between November 2021 and February 2022. Comparisons were made of the primary outcomes of days of stimulation, total dose of gonadotrophins and laboratory performance in ovarian stimulation in oocyte donors before and after vaccination. A total of 136 cycles in matched recipients were analysed as secondary outcomes and, from those, 110 women received a fresh single-embryo transfer, with analysis of biochemical β-human chorionic gonadotrophin concentrations and rates of clinical pregnancy with heartbeat.
Results
Longer stimulation was required in the post-vaccination than pre-vaccination group (10.31 ± 1.5 versus 9.51 ± 1.5 days; P < 0.001) along with higher gonadotrophin consumption (2453.5 ± 740 versus 2235.5 ± 615 IU; P < 0.001) with a similar starting dose of gonadotrophins in both groups. More oocytes were retrieved in the post-vaccination group (16.62 ± 7.1 versus 15.38 ± 7.0; P = 0.02). However, the number of metaphase II (MII) oocytes was similar between groups (pre-vaccination 12.61 ± 5.9 versus post-vaccination 13.01 ± 6.6; P = 0.39) and the ratio of MII/retrieved oocytes favoured the pre-vaccination group (0.83 ± 0.1 versus 0.77 ± 0.2 post-vaccination; P = 0.019). In recipients with a similar number of provided oocytes, the fertilization rate, total number of obtained blastocysts, number of top-quality blastocysts, and rates of biochemical pregnancy and clinical pregnancy with heartbeat were not significantly different between groups.
Conclusions
This study shows no adverse influence of mRNA SARS-CoV-2 vaccination on ovarian response in a young population.
KEY WORDS: Coronavirus, COVID-19, Infertility, mRNA vaccine, Oocyte donation, SARS-CoV-2
INTRODUCTION
The severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection compelled scientists to develop safe and effective vaccines. During the ongoing coronavirus disease 2019 (COVID-19) pandemic, the scientific community promoted vaccination programmes to reduce morbidity and mortality. A two-dose regimen of mRNA SARS-CoV-2 vaccine has been shown to confer 95% protection against COVID-19 in people 16 or more years of age (Polack et al., 2020). Nonetheless, in women of reproductive age, the rapid vaccine development raised mistrust and reluctance related to future fertility outcomes and vaccine safety (Murewanhema, 2021; Chen et al., 2021; Jing et al., 2020; Schaler and Wingfield, 2021), particularly for the novel mRNA-based formulations. There is an important need to review the data to improve our understanding of the effects of COVID-19 and vaccines on the human reproductive system and pregnancy (Nasab and Abhari, 2022).
A recent publication suggested that there was no measurable detrimental effect on the function of the ovarian follicle after mRNA SARS-CoV-2 vaccination (Bentov et al., 2021). Moreover, anti-Müllerian hormone concentrations do not seem to be affected following mRNA SARS-CoV-2 vaccination (Mohr-Sasson et al., 2022). However, the impact of COVID-19 vaccination on IVF laboratory outcomes warrant further investigation. The available medical evidence seems encouraging. Orvieto and colleagues (Orvieto et al., 2021) showed no detrimental effects in patients undergoing IVF in terms of stimulation characteristics, oocyte yield, fertilization or top-quality embryo rate, after receiving mRNA SARS-CoV-2 vaccine compared with their IVF cycles prior to vaccination. Furthermore, a recent retrospective cohort study including 200 vaccinated women and 200 age-matched unvaccinated women undergoing IVF showed that the mRNA SARS-CoV-2 vaccine did not affect the mean number of oocytes retrieved and the rate of clinical pregnancy with heartbeat in vaccinated versus unvaccinated patients (Avraham et al., 2022). Of note, the mean age of the participants in the aforementioned trials was over 36 years, so there is a paucity of medical evidence about the impact of the COVID-19 mRNA vaccine in younger populations undergoing ovarian stimulation.
The aim of the current observational study was to investigate, in ovarian stimulation cycles of oocyte donors before and after vaccination, the influence of mRNA SARS-CoV-2 vaccination on cycle characteristics and laboratory outcomes. Primary end-points were the number of cumulus–oocyte complexes (COC) retrieved, mean number of metaphase II (MII) oocytes, mature/total oocyte ratio, length of stimulation (days) and dose of gonadotrophins (IU). Secondary end-points were the fertilization rate, blastocyst formation rate, high-quality blastocyst rate, and rates of biochemical pregnancy and clinical pregnancy with heartbeat in matched recipients.
MATERIALS AND METHODS
This study was approved by the Ethics Institutional Committee of Instituto Bernabeu on 1 October 2021 (reference MR38).
Trial design
This retrospective multicentre cohort study enrolled oocyte donor participants who had received two doses of the mRNA vaccine at least 7 days before starting the ovarian stimulation cycle (the post-vaccination group). This study group was matched to previous stimulations in the same oocyte donors (the previous stimulations making up the pre-vaccination group). The ovarian stimulations after vaccination used the same types of stimulation protocol as those before the vaccination schedule, and both stimulations were performed within 1 year. Donors were included only once in the data analysis.
Participants
The trial was conducted between November 2021 and February 2022 at Accuna Medical Center and the Instituto Bernabeu Medical Center (Alicante, Spain). Eligible oocyte donors were included according to the Spanish Fertility Legislation, in brief, with an age 18–33 years, a negative family history of genetically transmitted diseases, a normal karyotype, negative screening for genetic diseases, negative results for sexually transmitted diseases (HIV, hepatitis B and C, and syphilis), a normal reproductive system, good physical and mental health, a previous history of fertility and/or an adequate response to treatment with ovarian stimulation, and an adequate body mass index (BMI).
Ovarian stimulation in oocyte donors
Oocyte donors started stimulation in a random protocol during the follicular phase with an initial dose of 100–300 IU/day of recombinant FSH (Bemfola; Gedeon Richter, Spain) according to the antral follicle count (AFC) and BMI. AFC was taken to include follicles with a mean diameter ranging from 2 to 10 mm measured using a transvaginal ultrasound probe with a frequency of ≥7 MHz. In addition, to prevent a premature LH peak, donors received 200 mg of natural micronized progesterone each night (Utrogestan; SEID, Spain) orally from the first day of stimulation until the day before triggering (Castillo et al., 2006; Castillo et al., 2019). Donors were monitored from day 5–6 of stimulation by transvaginal ultrasound scans every 2–3 days. A 0.2 mg bolus of gonadotrophin-releasing hormone (GnRH) agonist (triptorelin [Decapeptyl]; Ipsen Pharma, Spain]) was used to induce final oocyte maturation when at least three follicles were 18 mm or more in diameter. Oocyte aspiration was performed 36 h later by transvaginal ultrasound-guided needle-aspiration.
IVF laboratory and clinical outcomes
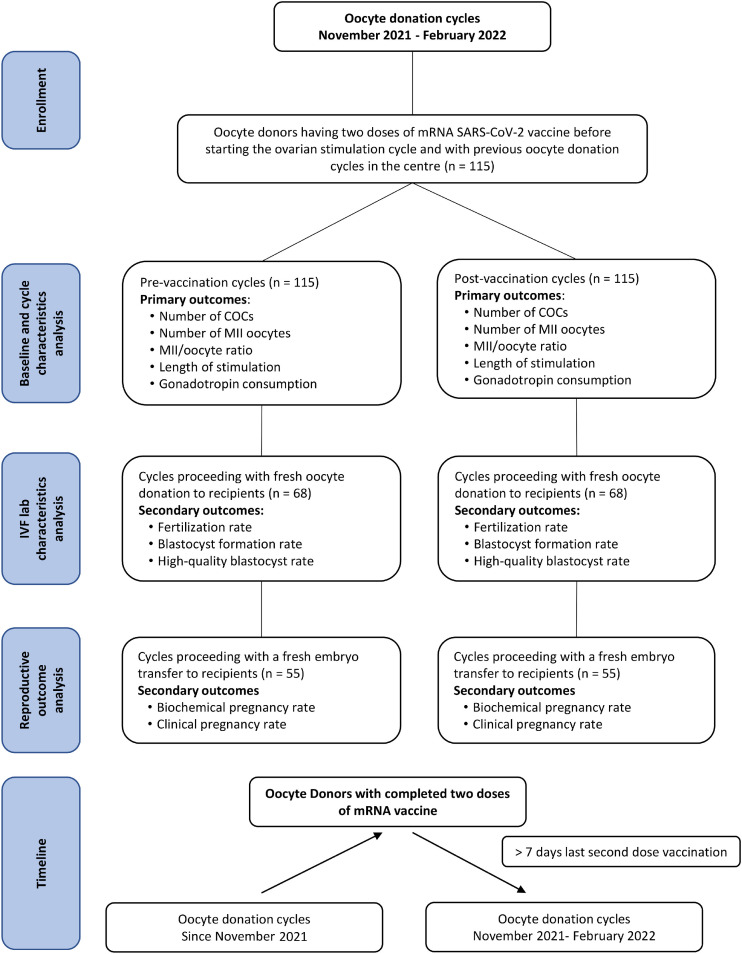
The authors’ oocyte donation programme guarantees a minimum of eight MII oocytes to be provided for recipients; hence the COC were removed and all the eggs were fertilized by intracytoplasmic sperm injection (ICSI). The oocyte donor cycles without recipients in both stimulations, pre- and post-vaccination, were excluded from the IVF laboratory analysis. This produced 68 recipients for the 115 pre-vaccination donor stimulations, with a different group of women representing the 68 recipients for the 115 post-vaccination stimulations, in order to avoid statistical bias (Figure 1 ). Additionally, each recipient contributed one cycle to the study. In the recipient laboratory cycles, the fertilization and blastocyst formation rates were compared between the groups using the blastocyst quality grading (ASEBIR, 2015).
Figure 1.
Flow chart showing the oocyte donor and recipient cycles included in the study, the cycle characteristics and reproductive outcomes analysed.
All embryo transfer procedures were performed at the blastocyst stage. Rates of biochemical pregnancy and clinical pregnancy with heartbeat were additionally compared between recipient groups receiving only a fresh single-embryo transfer.
Recipients and endometrial preparation
The recipients were infertile patients undergoing their first or second oocyte donation cycle. Eligible participants were aged less than 50 years, with a BMI below 30 kg/m2, and had no systemic diseases. Individuals with concomitant severe male factor infertility (<1 × 106 spermatozoa/ml), uterine diseases (e.g. fibroids, polyps, Asherman syndrome or previously diagnosed Müllerian duct abnormalities) or hydrosalpinx were excluded.
In patients with regular ovarian function a GnRH analogue (Gonapeptyl 3.75 mg; Ipsen-Pharma, Spain) was administered in the mid-luteal phase of the immediately preceding cycle for pituitary desensitization; this step was omitted in those with inactive ovaries. Subsequently, for endometrial preparation these women received standard substitutive hormonal therapy with transdermal oestrogen (Evopad 50 μg); Janssen Pharmaceutica, Belgium) or oral oestradiol valerate (Progynova; Delpharm, France) at increasing doses for at least 12 days. An endometrial thickness of 7 mm or more and a trilaminar appearance on ultrasonography were confirmed before embryo transfer. Micronized progesterone supplementation started with intravaginal capsules 400 mg every 12 h (Cyclogest; Gedeon Richter Ibérica, Spain). In pregnant patients, the hormonal treatment was sustained for 12 weeks.
Vaccination status in recipients
Most of the cycles included Foreign patients; thus, inconsistent and heterogeneous information was recorded about the vaccination status in the recipients and male partners, mainly due to variations in the vaccination programmes among different countries (Supplementary Table).
Outcomes
Primary outcomes were the number of COC retrieved, mean number of MII oocytes, oocyte maturity ratio (calculated as the ratio of MII oocytes to total oocytes retrieved), length of stimulation (days) and dose of gonadotrophins (IU). Secondary end-points were the fertilization rate, blastocyst formation rate and high-quality blastocyst rate. Other outcomes included biochemical pregnancy (serum concentrations of β-human chorionic gonadotrophin of over 5 IU/ml 10 days after embryo transfer) and rate of clinical pregnancy with heartbeat (diagnosed by ultrasonographic visualization of a gestational sac) in matched recipients (Zegers-Hochschild et al., 2017).
Statistical analysis
Statistical analysis was performed using R Statistical Software, version 4.1.2 (The R Foundation, Austria) and the Statistical Product and Service Solutions software, version 20.0 (SPSS, USA). For categorical variables, descriptive analysis was performed using the frequency and percentage. Numerical variables were presented as the number of cases, mean ± standard deviation and 95% confidence interval (CI). The Shapiro–Wilk test was performed to evaluate normal distributions. Depending on whether the variable had a normal distribution, the comparison between means was carried out using either a paired Student's t-test or Wilcoxon signed-rank test. McNemar's test was used for the statistical analysis of qualitative variables. Values of P < 0.05 were considered statistically significant.
RESULTS
A total of 115 egg donation cycles with ovarian stimulation before vaccination and 115 after complete vaccination were included in the analysis (Figure 1). Overall, the mean age of the oocyte donors was 24.32 years (95% CI 21–25), the BMI was 23.19 kg/m2 (95% CI 23–24) and the AFC was 17.93 (95% CI 17–19).
The baseline characteristics of the oocyte donors in the two groups are presented in Table 1 . The mean age was similar (pre-vaccination 25.82 years, 95% CI 25–27, versus post-vaccination 26.35 years, 95% CI 26–27; P = 0.3]. The BMI was identical in the two groups (23.19 kg/m2, 95% CI 23–24, for both groups). However, the baseline AFC differed significantly between the groups (pre-vaccination 15.85, 95% CI 15–17 versus post-vaccination 20.02, 95% CI 18.22; P < 0.001). There were no statistically significant differences for the starting day of ovarian stimulation between the groups: the mean starting day of stimulation for the pre-vaccination group was 6.54 ± 2.58 (95% CI 2–12) versus for the post-vaccination group 6.87 ± 2.21 (95% CI 1,12) (P = 0.18). The starting dose of gonadotrophins was similar for the two groups (post-vaccination 244.35 ± 55.61 IU, 95% CI 100–300 versus pre-vaccination 240.87 ± 53.02 IU, 95% CI 100–300; mean difference 3.48 ± 29.60 IU, 95% CI –100 to 300; P = 0.254). In addition, the starting dose was changed in 24 of 115 egg donors in the post-vaccination group (20.9%). The starting dose was increased in 15 egg donors (13.0%), resulting in a mean increase in the starting dose of 60.00 IU (95% CI 42.8–77.20) in these women. Conversely, the starting dose was decreased in 9 egg donors (7.8%), resulting in a mean decrease in the starting dose of 55.56 IU (95% CI 34.55–76.56).
Table 1.
Baseline characteristics of the oocyte donors
| Parameter | Pre-vaccination (n = 115)a | 95% CI | Post-vaccination (n = 115)a | 95% CI | P-valueb |
|---|---|---|---|---|---|
| Age (years) | 25.82 | 25–27 | 26.35 | 26–27 | 0.3 |
| Body mass index (kg/m2) | 23.19 | 23–24 | 23.19 | 23–24 | – |
| Antral follicle count | 15.85 | 15–17 | 20.02 | 18–22 | <0.001 |
The same women were included in the pre- and post-vaccination groups.
Data are means.
Welch's two-sample t-test.
Primary outcomes measure
With regards to oocyte yield, a higher number of oocytes was retrieved in the post-vaccination group (pre-vaccination 15.38 ± 7.0, 95% CI 14–17 versus post-vaccination 16.62 ± 7.1, 95% CI 15–18; mean difference –1.24 ± 6.0, 95% CI –2.34 to 0.14; P = 0.028) (Table 2 ). Nonetheless, the number of MII oocytes was similar in the two groups (pre-vaccination 12.61 ± 5.9, 95% CI 12–14 versus post-vaccination 13.01 ± 6.6, 95% CI 12–14; mean difference –0.40 ± 4.9, 95% CI –1.32 to 0.52; P = 0.39]. Finally, the ratio of MII to retrieved oocytes favoured the pre-vaccination group (pre-vaccination 0.83 ± 0.1, 95% CI 0.80–0.85 versus post-vaccination 0.77 ± 0.2, 95% CI 0.74–0.80; mean difference 0.05 ± 0.2, 95% CI 0.02–0.09; P = 0.019).
Table 2.
Ovarian stimulation and oocyte yield parameters
| Parameter | Pre-vaccination (n = 115)a | 95% CI | Post-vaccination (n = 115)a | 95% CI | Difference | 95% CIb | P-value | |
|---|---|---|---|---|---|---|---|---|
| Duration of stimulation (days) | 9.51 | 9.2–9.8 | 10.31 | 10–11 | –0.80 | –1.13 to –0.48 | <0.001c | |
| Dose of gonadotrophins (IU) | 2235.54 | 2122–2349 | 2453.48 | 2,317–,590 | –218 | –305.27 to –130.60 | <0.001b | |
| Oocytes retrieved (n) | 15.38 | 14–17 | 16.62 | 15–18 | –1.24 | –2.34 to –0.14 | 0.028b | |
| Metaphase II oocytes (n) | 12.61 | 12–14 | 13.01 | 12–14 | –0.40 | –1.32 to 0.52 | 0.39b | |
| Mature/total oocyte ratio | 0.83 | 0.80–0.85 | 0.77 | 0.74–0.80 | 0.05 | 0.02 to 0.09 | 0.019c | |
The same women were included in the pre- and post-vaccination groups.
Data are means.
Paired t-test.
Wilcoxon signed-rank test.
Cycle parameters in oocyte donors
Longer stimulation was required in the post-vaccination group (pre-vaccination 9.51 ± 1.5 days, 95% CI 9.2–9.8 versus post-vaccination 10.31 ± 1.5 days, 95% CI 10–11; mean difference –0.80 ± 1.74, 95% CI –1.13 to –0.48, P < 0.001) (Table 2). There was also a concomitant higher gonadotrophin consumption (pre-vaccination 2235.54 ± 615 IU, 95% CI 2122–2349 versus post-vaccination 2453.48 ± 740 IU, 95% CI 2317–2590; mean difference –218 ± 472 IU, 95% CI –305.27 to –130.60; P < 0.001).
Secondary outcomes
Laboratory outcomes
A total of 136 cycles involving donated oocytes were included in this analysis, comparing 68 cycles pre-vaccination with 68 post-vaccination (Table 3 ). With a similar number of provided oocytes (pre-vaccination 9.46, 95% CI 9.07–9.85 versus post-vaccination 9.38, 95% CI 8.98–9.78; P = 0.79), the fertilization rate was similar between the groups (pre-vaccination 82.69%, 95% CI 79.23–86.15 versus post-vaccination 78.84%, 95% CI 75.21–82.47; P = 0.17). Additionally, the total number of obtained blastocysts was similar between groups (pre-vaccination 4.51, 95% CI 4.00–5.02 versus post-vaccination 4.34, 95% CI 3.82–4.86; P = 0.58) with day 5/6 grade A (pre-vaccination 2.29, 95% CI 1.85–2.73 versus post-vaccination 2.32, 95% CI 21.96–2.68; P = 0.90) and grade B (pre-vaccination 2.19, 95% CI 1.82–2.56 versus post-vaccination 1.90, 95% CI 1.56–2.24; P = 0.30).
Table 3.
Laboratory outcomes of the oocyte donation cycles
| Parameter | Pre-vaccination (n = 68)a | 95% CI | Post-vaccination (n = 68)a | 95% CI | Difference | 95% CIb | P-valueb |
|---|---|---|---|---|---|---|---|
| Metaphase oocytes assigned (n) | 9.46 | 9.07–9.85 | 9.38 | 8.98–9.78 | 0.74 | –0.49 to 0.64 | 0.793 |
| Fertilized oocytes (n) | 7.82 | 7.34–8.30 | 7.32 | 6.91–7.73 | 0.50 | –0.15 to 1.15 | 0.133 |
| Fertilization rate (%) | 82.69 | 79.23–86.15 | 78.84 | 75.21–82.47 | 3.86 | –1.74 to 9.45 | 0.173 |
| Obtained blastocysts (n) | 4.51 | 4.00–5.02 | 4.34 | 3.82–4.86 | 0.18 | –0.46 to 0.82 | 0.583 |
| Grade A | 2.29 | 1.85–2.73 | 2.32 | 1.96–2.68 | -0.03 | –0.52 to 0.46 | 0.903 |
| Grade B | 2.19 | 1.82–2.56 | 1.90 | 1.56–2.24 | 0.294 | –0.27 to 0.86 | 0.303 |
The women included in pre- and post-vaccination groups were different.
Data are means.
Paired t-test.
Outcomes in recipients
Overall, the recipients were women aged under 50 years (range 29–48 years) with a mean BMI of 23.38 kg/m2. The baseline characteristics were similar between groups in terms of age (pre-vaccination oocyte group 42.51 years, 95% CI 42–43 versus post-vaccination 41.69 years, 95% CI 41–43, P = 0.252) (Table 4 ). The mean BMI was also similar (pre-vaccination oocyte group 23.55 kg/m2, 95% CI 23–25 versus post-vaccination 23.22 kg/m2, 95% CI 22–24; P = 0.758).
Table 4.
Baseline characteristics of the recipient population
| Parameter | Pre-vaccination (n = 68) | 95% CI | Post-vaccination (n = 68) | 95% CI | P-valuea |
|---|---|---|---|---|---|
| Age (years) | 42.51 | 42–43 | 41.69 | 41–43 | 0.252 |
| BMI (kg/m2)b | 23.55 | 23–25 | 23.22 | 22–24 | 0.758 |
| Infertility cause | 0.446 | ||||
| Premature ovarian failure | 6/68 (8.8%) | 3.6–19% | 8/68 (12%) | 5.6–22% | |
| Advanced maternal age | 55/68 (81%) | 69–89% | 57/68 (84%) | 72–91% | |
| Othersc | 7/68 (10%) | 4.6–21% | 3/68 (4.4%) | 1.1–13% |
The women included in pre- and post- vaccination groups are different.
Values are reported as means and 95% CI or n/N (%).
Paired t-test; McNemar test.
Of a total of 136 recipients, 8 had missing BMI values (2 pre-vaccination, 6 post-vaccination).
Others: recurrent miscarriage, unknown cause infertility.
BMI, body mass index.
Table 5 shows the clinical outcomes in a total of 110 recipients who underwent a fresh single blastocyst-stage embryo transfer. Of those, 55 cycles involved oocytes transferred from the pre-vaccination group and 55 oocytes from the post-vaccination group. The biochemical pregnancy rates (pre-vaccination 73%, 95% CI 59–83% versus post-vaccination 58%, 95% CI 44–71%; P = 0.136) and rates of clinical pregnancy with heartbeat (pre-vaccination 56%, 95% CI 42–69 versus post-vaccination 45%, 95% CI 32–59; P = 0.361) were not significantly different between the groups.
Table 5.
Clinical outcomes in recipients undergoing a fresh single-embryo transfer
| Parameter | Pre-vaccination (n = 55)a | 95% CI | Post-vaccination (n = 55)a | 95% CI | P-valueb |
|---|---|---|---|---|---|
| Biochemical pregnancy rate | 40/55 (73%) | 59–83 | 32/55 (58%) | 44–71 | 0.136 |
| Clinical pregnancy rate | 31/55 (56%) | 42–69 | 25/55 (45%) | 32–59 | 0.361 |
The women included in the pre- and post- vaccination groups were different.
Data are n/N (%)
McNemar test.
DISCUSSION
The present retrospective cohort study exploring cycle outcomes in young oocyte donors who underwent ovarian stimulation before and after receiving the COVID-19 mRNA vaccine confirmed the findings of previous studies in own egg cycles that showed similar outcomes in terms of fertilization rates in women who underwent ovarian stimulation after vaccination in comparison with controls (Avraham et al., 2022; Bentov et al., 2021) or their prior treatment (Orvieto et al., 2021). Nonetheless, these earlier studies included only a small number of participants (Bentov et al., 2021) or focused on women over 35 years of age (Avraham et al., 2022; Orvieto et al., 2021). To the best of the authors’ knowledge, the current study is the first to contribute additional (reassuring) information with regard to younger vaccinated populations undergoing ovarian stimulation and shows that mRNA vaccine had no detrimental effect on IVF outcomes.
The current results show that the fertilization, embryo development and pregnancy rates obtained in matched oocyte recipients were not jeopardized. These findings positively contribute to the growing body of evidence regarding the safety of the mRNA SARS-CoV-2 vaccines (Odeh-Natour et al., 2022) and, in concordance with the aforementioned studies, does not sustain theoretical concerns that the vaccine may induce an immune response that could affect the fertilization process (Kloc et al., 2021).
Interestingly, a longer stimulation (together with a concomitant higher total dose of gonadotrophins required) was seen and, of note, a higher total number of oocytes was collected in cycles after receiving the COVID-19 mRNA vaccine; nonetheless, the final number of MII oocytes remained similar. Medical evidence on the topic is limited, but a previous study with a similar methodology in own egg ovarian stimulation for IVF showed no influence of the mRNA SARS-CoV-2 vaccine on performance in terms of length of ovarian stimulation, total gonadotrophin dose and total number of oocytes collected (Orvieto et al., 2021). Thus, these additional findings should be interpretated with caution (particularly the higher number of collected eggs after vaccination) as they could be due to the individual participants’ intercycle variability in ovarian response between repeated cycles even after using the same protocol (Rombauts et al., 2015).
To the best of the authors’ knowledge, there is no biologically plausible explanation for a better performance in terms of oocyte pool or ovarian response associated with mRNA SARS-CoV-2 vaccines. Nonetheless, a recent large international cohort study found a small (and likely to be temporary) change in menstrual cycle length after COVID-19 vaccination, which suggests an impact on the function of the ovarian/uterine axis and underscores the importance of collecting menstrual cycle data during the development of future vaccines (Edelman et al., 2022). Future larger controlled trials should address this effect and other (potential) long-term effects on ovarian function as countries continue to move forward with their vaccination campaigns.
The two-centre setting could also be considered as a study strength, as it allows for generalizability of the results. As in any cohort study, data were prospectively registered.
Following oocyte assignation to matched recipients, the total number of fertilized oocytes and the fertilization rate after ICSI were similar among pre-vaccination and post-vaccination cycles. Moreover, the number of top-quality embryos was optimal and not significantly different between the groups. Finally, in a subset of recipients receiving a single fresh embryo transfer, similar rates of biochemical pregnancy and clinical pregnancy with heartbeat were found. These encouraging results reflect a lack of detrimental effects of the vaccine on embryo development or implantation performance, with a comparable reproductive outcome in recipients, and further supports the results of previous studies showing normal pregnancy rates in vaccinated women undergoing IVF cycles (Aizer et al., 2022; Avraham et al., 2022; Orvieto et al., 2021).
In terms of the ovarian stimulation cycle performance, this study is limited by the small sample size and aspects inherent to its retrospective observational nature, in which the influence of residual confounders cannot be completely excluded. Conversely, this is the first study to examine the effect of SARS-Cov-2 vaccination on the ovarian response on a young population with the major strength of exploring IVF cycles (before and following vaccination) in the same cohort of patients; this helps to mitigate multiple confounding factors or biases and to attribute the study results to the (absence of) effects of the vaccination. The findings in the oocyte donor population might be well extrapolated to young couples with infertility due to severe male factor or tubal factor cases or women undergoing fertility preservation for social reasons. Nonetheless, generalizability to the general infertile population should be made with caution.
For the outcomes of biochemical and clinical pregnancy in recipients, the findings should be interpreted with caution, because, again, only a limited number of transfer cycles were included in a retrospective data analysis; an additional drawback is the limited information about the vaccination or past infection status of the recipients and the male partners. However, in view of the efficacious vaccination campaign, it is reasonable to assume a balanced proportion of vaccinated recipients and male partners between the groups, thus only strengthening the conclusion that the vaccine had no detrimental effects on fertility (Aizer et al., 2022; Avraham et al., 2022; Huang et al., 2022; Jacobs et al., 2022).
The study design does not permit a verification of vaccine status or dates but this information is readily available for most individuals. Finally, although a rigorous study design and analytical method were implemented, the possibility of residual confounding and bias exists.
CONCLUSION
In conclusion, the present study found no detrimental influence of mRNA SARS-CoV-2 vaccines on donor oocyte cycles, reflecting no adverse effects on the outcomes of assisted reproduction. The safety of SARS-CoV-2 vaccination in relation to IVF cycles is encouraging for the medical community and the health of our patients.
Acknowledgments
ACKNOWLEDGEMENT
The authors would like to thank the staff at Accuna and the Instituto Bernabeu in Alicante, Spain who kindly used their time to make this study possible.
AUTHOR CONTRIBUTIONS
B.A., A.S. and J.C.: collection of data, study conception and design, analysis and interpretation of the data, writing the article and critical review of the article. J.T., J.G. and J.A.O.: analysis and interpretation of the data and critical review of the article. A.B. and R.B.: critical review of the article. The authors agree in considering B.A. and A.S. as co-first authors of the publication.
Biography

Aránzazu Bosch Villegas is a biologist with a Master's degree in the Biotechnology of Assisted Human Reproduction and Clinical Genetics and Genetic Counselling. With international experience as a Clinical Embryologist, she currently supervises the Assisted Reproduction Laboratory of the Instituto Bernabeu in Cartagena, Spain.
Key message.
This study shows that mRNA SARS-CoV-2 vaccination does not adversely influence the ovarian response in young oocyte donors. The safety of SARS-CoV-2 vaccination in relation to IVF cycles is encouraging for the medical community and the health of our patients.
Alt-text: Unlabelled box
Declaration: The authors report no financial or commercial conflicts of interest.
Footnotes
Supplementary material associated with this article can be found, in the online version, at doi:10.1016/j.rbmo.2023.01.002.
Appendix. Supplementary materials
Data Availability
Data will be made available on request.
REFERENCES
- Aizer A, Noach-Hirsh M, Dratviman-Storobinsky O, Nahum R, Machtinger R, Yung Y, Haas J, Orvieto R. The effect of coronavirus disease 2019 immunity on frozen-thawed embryo transfer cycles outcome. Fertil Steril. 2022;117(5):974–979. doi: 10.1016/j.fertnstert.2022.01.009. MayEpub 2022 Jan 10PMID: 35216833PMCID: PMC8743570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- ASEBIR . 3rd edition. Gobalo; Madrid: 2015. Cuadernos de embriología clínica III. Criterios de valoración morfológica de oocitos, embriones tempranos y blastocistos humanos. [Google Scholar]
- Avraham S, Kedem A, Zur H, Youngster M, Yaakov O, Yerushalmi GM, Gat I, Gidoni Y, Hochberg A, Baum M, Hourvitz A, Maman E. Coronavirus disease 2019 vaccination and infertility treatment outcomes. Fertil Steril. 2022;117(6):1291–1299. doi: 10.1016/j.fertnstert.2022.02.025. JunPMID: 35437147PMCID: PMC8872833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bentov Y, Beharier O, Moav-Zafrir A, Kabessa M, Godin M, Greenfield CS, Ketzinel-Gilad M, Ash Broder E, Holzer HEG, Wolf D, Oiknine-Djian E, Barghouti I, Goldman-Wohl D, Yagel S, Walfisch A, Hersko Klement A. Ovarian follicular function is not altered by SARS-CoV-2 infection or BNT162b2 mRNA COVID-19 vaccination. Hum Reprod. 2021;36(9):2506–2513. doi: 10.1093/humrep/deab182. Aug 18PMID: 34364311PMCID: PMC8385874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Castillo JC, Guerrero J, Delgado R, Moliner B, Luque L, Ten J, Fuentes A, Bernabeu A, Llácer J, Bernabeu R. O-124 Natural micronized progesterone versus a GnRH antagonist in egg-donation cycles. An extended experience. Session 38 – ART—Ovarian stimulation. Human Reproduction. 2006;21(suppl_1):i54–i56. doi: 10.1093/oxfordjournals.humrep.a002546. 1 June. [DOI] [Google Scholar]
- Castillo JC, Guerrero J, Delgado R, Moliner B, Luque L, Ten J, Fuentes A, Herencia A, Bodri D, Cirillo P, Bernabeu A, Llácer J, Bernabeu R. Initiation of ovarian stimulation independent of the menstrual cycle (random-start) in an egg donor program: a one-year single center experience. Abstracts of the 35th Annual Meeting of the European Society of Human Reproduction and Embryology, Human Reproduction. 2019;34(Supplement_1):i450–i451. July. [Google Scholar]
- Chen F, Zhu S, Dai Z, Hao L, Luan C, Guo Q, Meng C, Zhang Y. Effects of COVID-19 and mRNA vaccines on human fertility. Hum Reprod. 2021;37(1):5–13. doi: 10.1093/humrep/deab238. Dec 27PMID: 34734259PMCID: PMC8689912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Edelman A, Boniface ER, Benhar E, Han L, Matteson KA, Favaro C, Pearson JT, Darney BG. Association Between Menstrual Cycle Length and Coronavirus Disease 2019 (COVID-19) Vaccination: A U.S. Cohort. Obstet Gynecol. 2022;139(4):481–489. doi: 10.1097/AOG.0000000000004695. Apr 1Epub 2022 Jan 5PMID: 34991109PMCID: PMC8936155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang J, Xia L, Lin J, Liu B, Zhao Y, Xin C, Ai X, Cao W, Zhang X, Tian L, Wu Q. No Effect of Inactivated SARS-CoV-2 Vaccination on in vitro Fertilization Outcomes: A Propensity Score-Matched Study. J Inflamm Res. 2022;15:839–849. doi: 10.2147/JIR.S347729. Feb 9PMID: 35177919PMCID: PMC8843422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jacobs E, Summers K, Sparks A, Mejia R. Fresh Embryo Transfer Cycle Characteristics and Outcomes Following In Vitro Fertilization via Intracytoplasmic Sperm Injection Among Patients With and Without COVID-19 Vaccination. JAMA Netw Open. 2022;5(4) doi: 10.1001/jamanetworkopen.2022.8625. Apr 1PMID: 35452110PMCID: PMC9034396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jing Y, Run-Qian L, Hao-Ran W, Hao-Ran C, Ya-Bin L, Yang G, Fei C. Potential influence of COVID-19/ACE2 on the female reproductive system. Mol Hum Reprod. 2020;26(6):367–373. doi: 10.1093/molehr/gaaa030. Jun 1PMID: 32365180PMCID: PMC7239105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kloc M, Uosef A, Kubiak JZ, Ghobrial RM. Exaptation of Retroviral Syncytin for Development of Syncytialized Placenta, Its Limited Homology to the SARS-CoV-2 Spike Protein and Arguments against Disturbing Narrative in the Context of COVID-19 Vaccination. Biology (Basel) 2021;10(3):238. doi: 10.3390/biology10030238. Mar 19PMID: 33808658PMCID: PMC8003504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mohr-Sasson A, Haas J, Abuhasira S, Sivan M, Doitch Amdurski H, Dadon T, Blumenfeld S, Derazne E, Hemi R, Orvieto R, Afek A, Rabinovici J. The effect of Covid-19 mRNA vaccine on serum anti-Müllerian hormone levels. Hum Reprod. 2022;37(3):534–541. doi: 10.1093/humrep/deab282. Mar 1PMID: 34935913. [DOI] [PubMed] [Google Scholar]
- Murewanhema G. Vaccination hesitancy among women of reproductive age in resource-challenged settings: a cause for public health concern. Pan Afr Med J. 2021;38:336. doi: 10.11604/pamj.2021.38.336.28953. Apr 7PMID: 34285758PMCID: PMC8265245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nasab S, Abhari S. Coronavirus disease 2019 vaccine and in vitro fertilization outcomes: myths vs. facts. Fertil Steril. 2022;117(6):1300. doi: 10.1016/j.fertnstert.2022.04.005. JunEpub 2022 Apr 8PMID: 35525817PMCID: PMC8990686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Odeh-Natour R, Shapira M, Estrada D, Freimann S, Tal Y, Atzmon Y, Bilgory A, Aslih N, Abu-Raya YS, Shalom-Paz E. Does mRNA SARS-CoV-2 vaccine in the follicular fluid impact follicle and oocyte performance in IVF treatments? Am J Reprod Immunol. 2022;87(5):e13530. doi: 10.1111/aji.13530. MayEpub 2022 Mar 14PMID: 35220640PMCID: PMC9111235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Orvieto R, Noach-Hirsh M, Segev-Zahav A, Haas J, Nahum R, Aizer A. Does mRNA SARS-CoV-2 vaccine influence patients' performance during IVF-ET cycle? Reprod Biol Endocrinol. 2021;19(1):69. doi: 10.1186/s12958-021-00757-6. May 13PMID: 33985514PMCID: PMC8116639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Orvieto R, Segev-Zahav A, Aizer A. Does COVID-19 infection influence patients' performance during IVF-ET cycle?: an observational study. Gynecol Endocrinol. 2021;37(10):895–897. doi: 10.1080/09513590.2021.1918080. OctEpub 2021 May 11PMID: 33974475. [DOI] [PubMed] [Google Scholar]
- Polack FP, Thomas SJ, Kitchin N, Absalon J, Gurtman A, Lockhart S, Perez JL, Pérez Marc G, Moreira ED, Zerbini C, Bailey R, Swanson KA, Roychoudhury S, Koury K, Li P, Kalina WV, Cooper D, Frenck RW, Jr, Hammitt LL, Türeci Ö, Nell H, Schaefer A, Ünal S, Tresnan DB, Mather S, Dormitzer PR, Şahin U, Jansen KU, Gruber WC, C4591001 Clinical Trial Group Safety and Efficacy of the BNT162b2 mRNA Covid-19 Vaccine. N Engl J Med. 2020;383(27):2603–2615. doi: 10.1056/NEJMoa2034577. Dec 31Epub 2020 Dec 10PMID: 33301246PMCID: PMC7745181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rombauts L, Lambalk CB, Schultze-Mosgau A, van Kuijk J, Verweij P, Gates D, Gordon K, Griesinger G. Intercycle variability of the ovarian response in patients undergoing repeated stimulation with corifollitropin alfa in a gonadotropin-releasing hormone antagonist protocol. Fertil Steril. 2015;104(4):884–890. doi: 10.1016/j.fertnstert.2015.06.027. Octe2Epub 2015 Jul 15PMID: 26187300. [DOI] [PubMed] [Google Scholar]
- Schaler L, Wingfield M. COVID-19 vaccine - can it affect fertility? Ir J Med Sci. 2021:1–3. doi: 10.1007/s11845-021-02807-9. Oct 15Epub ahead of printPMID: 34651258PMCID: PMC8516490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zegers-Hochschild F, Adamson GD, Dyer S, Racowsky C, de Mouzon J, Sokol R, Rienzi L, Sunde A, Schmidt L, Cooke ID, Simpson JL, van der Poel S. The International Glossary on Infertility and Fertility Care, 2017. Hum Reprod. 2017;32(9):1786–1801. doi: 10.1093/humrep/dex234. Sep 1PMID: 29117321 PMCID: PMC5850297. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Data will be made available on request.