Abstract
During the first year of the COVID-19 pandemic, facemasks became mandatory, with a great preference for disposable ones. However, the benefits of face masks for health safety are counteracted by the environmental burden related to their improper disposal. An unprecedented influx of disposable face masks entering the environment has been reported in the last two years of the pandemic, along with their implications in natural environments in terms of their biodegradability, released contaminants and ecotoxicological effects. This critical review addresses several aspects of the current literature regarding the (bio)degradation and (eco)toxicity of face masks related contaminants, identifying uncertainties and research needs that should be addressed in future studies. While it is indisputable that face mask contamination contributes to the already alarming plastic pollution, we are still far from determining its real environmental and ecotoxicological contribution to the issue. The paucity of studies on biodegradation and ecotoxicity of face masks and related contaminants, and the uncertainties and uncontrolled variables involved during experimental procedures, are compromising eventual comparison with conventional plastic debris. Studies on the abundance and composition of face mask-released contaminants (microplastics/fibres/ chemical compounds) under pre- and post-pandemic conditions should, therefore, be encouraged, along with (bio)degradation and ecotoxicity tests considering environmentally relevant settings. To achieve this, methodological strategies should be developed to overcome technical difficulties to quantify and characterise the smallest MPs and fibres, adsorbents, and leachates to increase the environmental relevancy of the experimental conditions.
Keywords: Plastic pollution, Microplastics, Protective equipment, Face masks, Biodegradation, Toxicity
Graphical Abstract
1. Introduction
COVID-19 is an acute (and, to some extent, severe) respiratory infectious human disease caused by SARS-CoV-2, a type of coronavirus [1] identified in Wuhan-China in December 2019 [2], [3], [4]. Since its discovery, it has quickly spread across the globe, being classified as a pandemic in early 2020 by the World Health Organization (WHO) [5]. Several recommendations emerged to reduce the chances of contracting the virus, such as physical distance, frequent handwashing, and the use of personal protective equipment (PPE), namely face masks [1], [6]. Face masks act as a barrier to transmitting infectious particles in bodily fluids [6], [7], protecting us from others and vice versa.
Face masks soon became the icon of COVID-19 as a saviour item to suppress contagion. Concomitantly, it also became an icon of COVID-19 pollution. Several studies have been addressing the implications of face masks to the environment, considering their i) production, use, and disposal (including Life Cycle Assessment) and implications on the environmental footprint [8], [9]; ii) presence and distribution in natural ecosystems [10], [11], [12], [13], [14], [15]; iii) presence and release of contaminants that compose them [16], [17], [18], [19], [20]; and iv) the potential toxicity of such released contaminants on aquatic and terrestrial organisms [19], [21]. Recent reviews have also critically discussed their short- and long-term environmental implications, but mostly considering their end-of-life options [18], [22], [23] and how to mitigate/minimize their impact on environmental and human health. However, little is known or discussed considering environmentally relevant scenarios on biodegradation and the potential toxicity of face masks in natural settings and their long-term implications. This paper reviews and critically discusses knowledge gathered within the last two years of the pandemic on the environmental implications of face masks, with particular emphasis on their biodegradability and (eco)toxicity in aquatic and terrestrial ecosystems. It also highlights future research lines to predict the consequences of the massive loads of face masks in the environment.
2. Face masks – types, composition, waste generation and management
Different face masks have been adopted worldwide, being essentially divided into three categories: a) Reusable, b) Surgical, and c) Respirators (commonly used are presented in Table 1). Briefly, reusable face masks usually have two or three fabric layers with at least one layer of cotton [24]. Depending on the fabric combination, the filtration power increases with the number of layers [23], [24], which can be higher than 70% in certified masks [9]. Although the use of reusable face masks decreases the waste volume related to these items [26], it might result in greater water use and energy consumption for their production and washing processes [27]. In addition, reusable face masks also lose their effectiveness when the washing number is exceeded [27].
Table 1.
Types of disposable facemasks, their composition, and main properties.
| Disposable Mask Type | Composition | Main Properties | Reference | |
|---|---|---|---|---|
| Surgical | PP spunbond nonwoven fabric (x2) PP, poly(butylene terephthalate) or poly(tetrafluoroethylene) melt blown filter |
Filters particles > 5 µm | [43], [44] | |
| Respirators | FFP1 | PP or PET spunbond nonwoven fabric (x2) PP melt blown non-woven filter Modacrylic support layer |
Particles inward leak of 25% Filter efficiency > 80% Not adequate for COVID-19 protection |
[44], [45], [46] |
| FFP2 | PP or PET spunbond nonwoven fabric (x2) PP melt blown non-woven filter Modacrylic support layer |
Particles inward leak of 11% Filter efficiency > 94% |
[44], [45] | |
| FFP3 | PP or PET spunbond nonwoven fabric (x2) PP melt blown non-woven filter Modacrylic support layer |
Particles inward leak of 5% Filter efficiency > 99% |
[44], [45] | |
| N95 | PP or PET spunbond nonwoven fabric (x2) PP melt blown non-woven filter Modacrylic nonwoven layer |
Filters particles > 100 nm Filters > 95% of solid and liquid particles > 0.3 µm |
[44], [47] | |
In turn, surgical masks are disposable and mostly made of different plastic nanofibres, being the most common polypropylene (PP) [28], [29] (Table 1). They usually have three to five layers. The middle layers are often made by a nonwoven melt-blown process featuring electrostatic charging and filtration capacities. Such layers are also made by web formation technology, where hot air is used to disperse the meltable polymeric material to produce ultra-fine fibres (2–5 µm thinner than spunbonded nonwoven fabric) [28], [30], [31]. These middle layers also have an electrostatic absorption function, allowing particles, bacteria, and pollutant filtration. Conversely, the inner and outer layers are often non-woven fabrics obtained through spunbonded technology [31], where molten polymers are extruded and stretched to form filaments/fibres, which will then be laid in a web and bonded uniformly by heat throughout the structure to guarantee good hydrophilicity and provide more comfort [33], [34]. The filtration efficiency of such disposable masks is above 80% [35] and the usage period is up to 4 h [24].
Aside from the polymeric material, disposable face masks possess additives and extra components to make them more adjustable, comfortable, and fashionable. For example, certified disposable masks possess a nose wire frame made of metallic compounds (iron constituting 4.58 wt% of the total mass of the face mask, and the trace amount of zinc, calcium, titanium, and manganese) to allow a better adjustment of the mask to the nose area [176]. Bioactive compounds can be incorporated in face mask composition (particularly during the electrospun process) to increase antioxidant activity, thermal resistance, and antibacterial/antiviral properties [177]. The incorporation of pigments is also common to offer a panoply of coloured face masks to the general public.
Respirators, such as filtering facepieces (FFP), N95, KN95, are certified disposable masks [24], [25]. Their filtration efficiency is up to 80 times more effective than cloth masks and up to 15 times more than surgical masks [25]. FFP, in particular, can be divided into FFP1, FFP2, and FFP3 according to their filtration efficiency, which is 80%, 94%, and 99%, respectively [24]. Albeit giving better protection and having a higher period of use (8 h), these are mainly used by health professionals [24], [36].
The filtration efficiency is, however, highly affected by the usage time. For example, it is recommended a usage time of aproximately 3 –10 h for respirators, 3–7 h for medical masks and 4–9 h for handmade masks [37]. If such time is exceeded, their effectiveness is highly affected, with the leakage of microfibres and granular microplastics [38]. In addition, if the recommended number of washes is also exceeded, the quality of the fabrics will deteriorate [39], losing its effectiveness [9]. Notwithstanding, given the reduced cost, lightness, and breathability of the surgical ones, they have become the most used by people in general [32], [40], [41]. In several countries, surgical masks are even mandatory in healthcare facilities (e.g., clinics, hospitals, general healthcare practices).
The clear preference for such masks and (most of all) their incorrect discard are both mirrored in the greater occurrence of this type of mask in different environments [12], [14], [15], [42], which have implied important threats to natural environments. In the first trimester of the pandemic, a monthly need of 89 million disposable masks was estimated to respond to the COVID-19 pandemic. However, this number was greatly exceeded on a large scale in the following months [21], being, at least, billions of face masks were needed and discarded worldwide ( Fig. 1).
Fig. 1.
: Estimated global share of face masks discarded as Covid-19 waste per country.
Source: Benson et al. [175] https://doi.org/10.1016/j.heliyon.2021.e06343. https://creativecommons.org/licenses/by-nc-nd/4.0/.
In the pre-COVID-19 period, face masks were mostly used by healthcare professionals and, being considered medical waste, they were sorted out accordingly to their type into colour-coded bins [48] and then incinerated or autoclaved, followed by safe disposal in landfills [49]. During the pandemic, the use of face masks was extended to all citizens. Thus, according to several governmental guidelines, face masks’ end-of-life would be closed bins or trash bags [6], [23] and then sent to landfills or incineration facilities [9]. Landfilling seems to be the dominant method of municipal waste management and where the vast majority of face masks end up [16], [50], [51]; with predictable impacts on the soil, water and atmosphere due to the formation of leachates and landfill gas [22], [51]. The presence of face masks in landfills and anaerobic digestors seems to affect methane productivity and kinetics negatively [52]. For example, in the digesters amended with face masks, the total cumulative methane production decreased by up to 18%, with a 12–29% decrease in maximum methane production rates than the control digester (without face masks) [52], compromising energy production. Incineration, the second most used method to process PPE waste [16], [50], would be the best option to overcome the waste problem as it eliminates pathogens and the ashes, which are later disposed to a landfill, taking up lesser space [9], [48], [53]. However, given the amount of discarded face masks, it would cause various environmental and health impacts due to releasing many toxins and carbon dioxide (CO2) into the atmosphere [48], [53]. Recycling could also be an option, however, it is not used since face masks are difficult to recycle [54]. Currently, no infrastructure allows the safe disposal of these potentially contaminated items [55]. Even if it did, recycling surgical face masks remain unpractical as it is necessary to sort out the different plastic nanofibres that constitute them and, posteriorly, disinfect them [9], [16].
Apart from traditional waste management methods, several face masks are discarded in the environment [16], [53], resulting from plastic leakage from waste treatment facilities, urban waste bins, or citizens' waste disposal malpractice. Some surveys have already been carried out to assess the disposal made by citizens. According to Tesfaldet et al. [40], out of a total of 605 people, about 70.58% (= 427) claimed to use regular bins to discard face masks, whereas Tan et al. [56] did an online survey in China and found out that out of a total of 10290 surveyed people only 30.1% (= 3097) had the same behaviour. Regarding disposal in the environment, only 0.17% (= 1) of respondents confessed to doing so [41]. Generally, the tendency to deliberately make garbage can come from laziness, lack of sense at the time of the act, peer influence (since the rest of the people also do it, it is acceptable), and lack or wrong location of waste bins [40], [57]. In the case of the pandemic and the disposal of masks, the inexistence of waste bins that do not require touch to be opened also contributes to incorrect disposal [40].
3. Presence and implications of face masks on the environment
Face masks possess a lightweight and, consequently, low air resistance, allowing quick dispersion of these items in natural environments and eventual interaction with biota [18], [29], [40]. Thus, although much of PPE waste (particularly face masks) is generated and discarded in metropolitan areas, it may eventually end up in rivers and oceans through surface runoff [16], [40]. As seen in Table 2, several studies reported the presence of PPE items in the last 2 years, particularly masks, in both urbanized and natural environments.
Table 2.
Occurrence and density of face masks during the COVID-19 pandemic in urbanized and natural environments.
| Country | Sampling Site | Face masks' total density (Face mask/m2) |
Face masks’ mean density (Face mask/m2) |
Reference |
|---|---|---|---|---|
| Iran | Beach | 3.60 × 10-2 | ------- | [14] |
| Hong Kong | Beach | 7 × 10-1 | ------- | [58] |
| Bangladesh | Urban | 5.19 × 10-2 | ------- | [59] |
| Mexico | Urban | 1.88 × 10-2 | ------- | [60] |
| Morocco | Beach | ------- | 1.20 × 10-3 | [12] |
| Iran | Lake | ------- | 9.75 × 10-5 | [61] |
| Philippines | Beach | 7,82 × 10-3 | ------- | [42] |
| Turkey | Urban | 5.46 × 10-4 | ------- | [62] |
| Ethiopia | Lake | ------- | 1.51 × 10-4 | [63] |
| India | Beach | 1.06 × 10-3 | ------- | [13] |
| Canada | Urban | 1.70 × 10-3 | ------- | [40] |
| Peru | Beach | 1.09 × 10-3 | ------- | [64] |
| Bangladesh | Beach | 6.25 × 10-2 | ------- | [65] |
| Argentina | Beach | ------- | 3.35 × 10-4 | [15] |
| Peru | Beach | ------- | 6.23 × 10-4 | [15] |
| Chile | Beach | ------- | 6 × 10-3 | [66] |
| Ghana | Urban | 3.1 × 10-1 | ------- | [67] |
| Canada | Riverside | 1 × 10-4 | ------- | [68] |
| Thailand | Urban | 1.27 × 10-2 | ------- | [41] |
| Morocco | Beach | ------- | 1.09 × 10-5 | [69] |
| India | Urban | 2.99 × 10-2* | ------- | [70] |
Extrapolated value obtained from an estimated sidewalk width of approximately 2 m over an 18 km extension.
Once in open environments, disposable face masks might undergo weathering, fragmentation and (bio)degradation, due to physicochemical and biochemical factors.
3.1. Potential (bio)degradation of disposable face masks
Plastics degradation is defined as changes in polymer properties that are accompanied by a loss of functionality because of chemical, physical, and/or biological processes, resulting in polymer chain bond breakage [71]. It is limited and dependent on plastic’s properties, and biotic and abiotic factors [72], [73]. Biotic degradation results from the activity of living organisms, particularly microorganisms, that break down polymer chains (in the presence of oxygen – aerobically, or in its absence – anaerobically) to CO2 or methane, water, and edible biomass/compost [79]. Abiotic degradation can be divided into photodegradation characterized by the action of light [74], thermal degradation by the action of heat [71], [75], mechanical degradation by the action of external forces [75], [76], and chemical degradation, which can even be divided into hydrolysis through exposure to water (H2O) or oxidative degradation through exposure to oxygen (O2) [76]. Depending on the desired properties and durability, plastics can be photo/oxo-degradable, hydro-degradable, biodegradable/compostable, among others.
As a polymeric-based material, disposable face masks are not different from any plastic waste that end up in natural environments, being, therefore, susceptible to degradation. However, face masks are made mainly from durable polymeric materials such as PP and PE (Table 1) that are characterised by a saturated chemical structure. Such saturated structures are less prone to bulk or molecular changes, including interaction with microorganisms [77]. For example, PP- and PE-based plastics take > 10 years to completely degrade under aerobic or anaerobic conditions, photodegradation and/or thermo-oxidative degradation [78].
Only a few studies addressed face mask degradation ( Table 3). Scanning electron microscopy of disposable face masks collected in urban and marine environments displayed microfibre fragmentation and the formation of cracks, whereas X-ray diffraction patterns showed a notorious decrease in their crystallinity [15], [62]. The inner layer of the face masks seems to be the most resistant to UV- or mechanical weathering, followed by the outermost layer, while the middle layer was found to be the most sensitive one.
Table 3.
Summary of degradation studies in face masks so far.
| Mask Type | Exposure Conditions | Main Results |
Reference | ||||
|---|---|---|---|---|---|---|---|
| Microplastics (MP) quantity | MP Size | Degradation effects/other observations | Other substances released | ||||
| Disposable surgical | Virgin face mask in an aqueous solution and shaken at 300 rpm for 24 h | 4.47 μL/L from the outer layer 1.52 μL/L from the middle layer 0.80 μL/L from the inner layer |
Mostly 10–50 µm and 200–500 µm Outer layer mostly between 15 and 50 µm and 200–500 µm Middle and inner layers mostly between 10 and 200 µm |
Smooth surface | ---- | [80] | |
| Virgin face mask in an aqueous solution with sand and shaken at 300 rpm for 24 h | 14.6 μL/L from the outer layer 6.2 μL/L from the middle layer 7.0 μL/L from the inner layer |
Mostly between 10 and 50 µm | ---- | ---- | [80] | ||
| Virgin face mask UV-light irradiated for 36 h, in an aqueous solution and shaken at 300 rpm for 24 h | 70.73 μL/L from the outer layer 60.36 μL/L from the middle layer 11.44 μL/L from the inner layer |
Mostly 20–500 µm Outer layer mostly between 20 and 100 µm and 100–500 µm Inner layer mostly between 30 and 100 µm and 100–500 µm Middle layer mostly between 50 and 200 µm |
Deformation/damage on the surface Increased roughness Fractured fibres Middle layer suffered more from weathering |
---- | [80] | ||
| Virgin face mask UV-light irradiated for 36 h, in an aqueous solution with sand and shaken at 300 rpm for 24 h | 132.52 μL/L from the outer layer 210.23 μL/L from the middle layer 184.94 μL/L from the inner layer |
Mostly 20–500 µm Outer and inner layers mostly between 20 and 100 µm and 200–500 µm Inner layer mostly between 30 and 100 µm and 100–500 µm Middle layer mostly between 10 and 200 µm |
---- | ---- | [80] | ||
| Disposable surgical | Virgin face mask in 3 L of ultrapure water for 24 h | 3600 MPs/mask | ---- | ---- | ---- | [85] | |
| Virgin face mask in 3 L of ultrapure water with alcohol (75%) for 24 h | 5400 MPs/mask | ---- | ---- | ---- | [85] | ||
| Virgin face mask in 3 L of ultrapure water with detergent (10 mg/L) for 24 h | 4400 MPs/mask | ---- | ---- | ---- | [85] | ||
| Virgin face mask in 3 L of ultrapure water, shaken at 120 rpm for 24 h and dried (x3) | 24,300, 36,400 and 55,900 MPs released in the first, second and third wash cycle, respectively 116,600 MPs/mask in total |
Mostly < 1 mm | Mass loss of 0.47% | ---- | [85] | ||
| Virgin face mask in 3 L of ultrapure water with alcohol (75%) and shaken at 120 rpm for 24 h and dried (x3) | 27,000, 43,200 and 76,800 MPs released in the first, second and third wash cycle, respectively 147,000 MPs/mask in total |
Mostly < 1 mm | Mass loss of 0.85% | ---- | [85] | ||
| Virgin face mask in 3 L of ultrapure water with detergent (10 mg/L) and shaken at 120 rpm for 24 h and dried (x3) | 28,800, 50,000 and 88,000 MPs released in the first, second and third wash cycle, respectively 168,800 MPs/mask in total |
Mostly < 1 mm | Mass loss of 1.14% | ---- | [85] | ||
| Aged face mask (in a building rooftop for 2 months) and shaken at 150 rpm for 24 h (x3) | 24,300–25,400 MPs/24 h before ageing to 6.0 × 108 – 6.4 × 108 MPs/24 h after ageing | ---- | ---- | ---- | [85] | ||
| Disposable surgical | Virgin face mask UV-irradiated (10 h at 65 °C alternating with 2 h at 50 °C (x15)), in an aqueous solution and shaken at 4000 rpm for 24 h | 61 ± 19 MPs/mask 117,400 ± 42,345 MPs/mask |
1–5 mm 25–500 µm |
Mean mass loss of 0.19% | ---- | [79] | |
| Disposable surgical, KF94, FFP1 and KF-AD | Virgin face masks in an aqueous solution and shaken at 150 rpm for 48 h | 202 MPs/surgical mask 161 MPs/KF94 160 MPs/FFP1 74 MPs/KF-AD |
---- | ---- | ---- | [81] | |
| Disposable surgical and N95 | Virgin face masks in an aqueous solution and shaken at 120 rpm for 24 h | 183 ± 78.42 MPs/mask | Mostly 100–500 µm | Fibres predominant over fragments (>70%) | ---- | [82] | |
| Worn face masks (24 h of usage) in an aqueous solution and shaken at 120 rpm for 24 h | 1246.6 ± 403.50 MPs/mask | Mostly 100–500 µm | Fibres predominant over fragments (>70%) | ---- | [82] | ||
| Disposable surgical | Virgin face masks in an aqueous solution and shaken at 200 rpm for 9 days | 2118 fibres/day 800 MPs/day |
Mostly < 10 µm and > 50 µm | Looser structure after immersion Many fibres appeared fractured Many surface cracks and holes |
---- | [83] | |
| Worn and discarded face masks in an aqueous solution and shaken at 200 rpm for 9 days | Mean 25,958 fibres/day Mean 21,441 MPs/day |
---- | Plastic material is brittle and fragmentary Significantly increased fibres’ release |
---- | [83] | ||
| Disposable surgical, common and FFP | Virgin face mask MPs in an aqueous solution and shaken at 60 rpm from 0 to 240 h | 272 ± 12.49 MPs/cm2 surgical face mask 54,400 ± 2498 MPs/common mask 165.7 ± 9.2 MPs/cm2 common mask 187.9 ± 9.45 MPs/cm2 FFP |
Mostly 0.1–1 mm | Middle layer released the greatest number of fibres | ---- | [84] | |
| Virgin face mask MPs in an aqueous solution with sand and shaken at 60 rpm from 0 to 240 h | 68,000 ± 4808 MPs/common mask | Mostly 0.1–1 mm | Middle layer released the greatest number of fibres Increased roughness Deformation/damage on fibre surface |
---- | [84] | ||
| Disposable surgical | Virgin face masks in soil exposed to the environment (sun and rain) for 30 days. New and aged dry masks rubbed 120 times inside a closed chamber | 22,053.84 ± 647.84 MPs/new mask 100,780.17 ± 35,538.62 MPs/aged mask |
New masks: 0.119–2.042 mm Weathered masks: 0.091–2.621 mm |
Aged face masks release higher amount of fibres Middle released higher number of fibres, followed by the outer layer and the inner layer |
---- | [70] | |
| Virgin face masks in soil exposed to the environment (sun and rain) for 30 days. New and aged masks put in water (freshwater and marine) in static immersion for 10 h | 2171.40 MPs/mask (new mask on freshwater) 3679.20 MPs/mask (new mask on seawater) 7921.07 MPs/mask (aged mask on freshwater) 11,785.20 MPs/mask (aged mask on seawater) |
---- | Aged face masks release higher amount of fibres Middle released higher number of fibres, followed by the outer layer and the inner layer |
------- | [70] | ||
| Virgin face masks in soil exposed to the environment (sun and rain) for 30 days. New and aged masks put in water (freshwater and marine) and rubbed continuously for 15 min | 3445.40 MPs/mask (new mask on freshwater) 5623.20 MPs/mask (new mask on seawater) 16,356.00 MPs/mask (aged mask on freshwater) 66,112.50 MPs/mask (aged mask on seawater) |
---- | Aged face masks release higher amount of fibres Middle released higher number of fibres, followed by the outer layer and the inner layer |
------- | [70] | ||
| Disposable surgical and N95 | Virgin face masks in an aqueous solution shaken for 3 min and then leachates were removed (x10) | Mean 2.23 × 103 MPs/mask > 1 µm Mean 2.43 × 109 MPs/mask < 1 µm |
Mostly < 1 µm | Middle layer with higher fibre roughness | ------- | [86] | |
| Disposable surgical | Virgin face mask in an aqueous solution and exposed to shear damage with a kitchen chopper for 1, 15, 30, 60 and 120 s | 2.6 × 103 MPs/mask (1 s) up to 2.8 × 104 MPs/mask (120 s) Mean 2.1 ± 1.4 × 1010 MPs/mask |
Mostly < 0.5 µm | Increased deterioration with time | ------- | [88] | |
| Disposable surgical | Virgin middle layer MPs (<300 µm) in soil with soil invertebrates for 21 and 28 days | ------- | ------- | Increased roughness Surface deterioration Fragmentation of fibres of < 1 µm |
------- | [91] | |
| Disposable surgical | Virgin face mask mixed with wet topsoil (cambisol) for 6 months in the dark at 25 °C | ------- | ------- | Loss of carbon of 10% | ------- | [92] | |
| Virgin face mask in an aqueous solution for 6 months in the dark at 25 °C | ------- | ------- | Low decomposition rate C-release of about 1.8 mg after 1 month |
------- | [92] | ||
| Virgin face mask outer, middle and inner layers (separately) mixed with wet topsoil (cambisol) for 6 months in the dark at 25 °C | ------- | ------- | Additional carbon-release of 12% on soil with outer and inner layer and 20% with middle layer, compared to pure soil Middle layer less resistant to mineralization (MRT of 7 years) Physical deterioration in middle layer Loss of carbon of 6% |
------- | [92] | ||
| Disposable surgical, common and N95 | Virgin face masks in an aqueous solution and shaken at 220 rpm for 24 h | Surgical masks: 1136 ± 87–2343 ± 168 MPs/(piece·d) Common masks: 1034 ± 119–2547 ± 185 MPs/(piece·d) N95 masks: 801 ± 71–2667 ± 97 MPs/(piece·d) |
Mostly < 500 µm | Mass loss from 0.006% to 0.019% | ------- | [93] | |
| Disposable surgical | Virgin face masks in 1,5 L of deionized water and gently stirred every hour for 4 h | ------- | Fibrous particles from 25 µm to 2.5 mm | ------- | Cd, Co, Cu, Pb, Sb, and Ti detected in leachates Sb concentrations ranging from 111 to 393 μg/L Cu concentrations ranging from 0.85 μg/L to 4.17 μg/L Highest concentration of Pb of 6.79 μg/L All face masks released polar organic species (e.g., polyamide-66, polyamide-6 and many oligomers of polyamide) Polyethylene glycol-like derivatives and aromatic amines compounds detected in some masks N-Undecyl-1-undecanamine detected in one face mask |
[94] | |
| Disposable surgical and N95 | Virgin face masks pieces placed in an air sampling system for 24 h at 37 °C | ------- | ------- | ------- | Five phthalate esters (PAEs) detected PAEs mass load ranged from 55 ± 35–1700 ± 140 ng on surgical masks PAEs mass load ranged from 2300 ± 150–5200 ± 800 ng on respirators |
[95] | |
| Disposable surgical and respirators | Virgin face masks in 1 L of deionized water and stirred at 120 rpm at 25 °C in the dark for 5, 10 and 15 days | 76–276 MPs/L | Mostly 40–225 µm | MPs increased with exposure time Surgical masks released more MPs than respirators Fibres > MPs Increased deformation and damage on the surface with time |
Co, Cu, Ni, Sr, Ti and Zn detected in all samples Cd, Cr, Mn and Pb detected only in surgical masks leachates Concentrations of all detected heavy metals higher in surgical masks Acetophenone (AP), 2,4-Di-tert-butylphenol (DTBP) and bis(2-ethylhexyl) phthalate detected in all samples Tributyl acetylcitrate and benzaldehyde, 2,4-dimethyl- detected in some samples AP, benzothiazole, DTBP, bisphenol-A (BPA) and phthalide detected in surgical masks leachates AP, benzothiazole and DTBP detected in respirators leachates |
[96] | |
Surgical face masks aged in water under controlled conditions revealed the breakage of loosely attached microfibres along with rough surfaces related to exposure time (e.g., [79], [80]). For instance, surgical face masks placed in water and subjected to agitation (simulating wave influence), released on average between 50 [81], 400 [79] or even 900 [82] microfibres per day. These numbers depend on the period during which face masks are submerged, i.e., the longer the exposure time, the greater the number of microfibres released [81], [83]. When photo- or mechanical degradation is added to water agitation, the release of fragments/microfibres skyrocket [83], [84], reaching an average of 173 thousand fibres per day [79] or a total of 68 thousand microfibres at the end of 240 h [81]. Here, the UV radiation likely provides sufficient energy for the C—C and C—H bonds in the face masks polymeric material to be broken, generating alkoxy and peroxyl radicals. This, in turn, led to chain scission that, allied with mechanical abrasion, will increase fragments and fibres release. Between fragments and fibre release, the latter is present in a greater proportion [82].
The weathering state and exposure (air, seawater, freshwater) are also major drivers of microplastics and microfibres release. For example, the number of released microfibres by face mask greatly increased 2.5 × 104 times with the ageing process (air-exposure in a building rooftop) compared to pristine ones (from 24300 to 25400 items before ageing to 6.0 × 108 – 6.4 × 108 items after ageing) [85]. Similarly, naturally weathered face masks on terrace topsoil (for 30 days) were further submitted to dry state exposure (simulating atmospheric conditions), to seawater, or freshwater (here simulating two distinct aquatic environments, under static immersion for 10 h or mechanical agitation for 15 min), released 2–8 times more microfibres than the pristine ones. The highest number of released microfibres was revealed in dry state (up to 100,780 microfibres/mask), followed by seawater under mechanical agitation (up to 66,112 microfibres/mask) and freshwater under mechanical agitation (16,356 microfibres/mask) [70]. Thus, microfibres release occurs to a greater extent in the dry state than in the wet state, potentially due to the higher physical abrasion of soil particles that likely increased shedding. In addition, differences between aquatic environments (seawater vs freshwater) are potentially related to water density and alkalinity during static immersion. Seawater had higher density and alkaline pH, which put more hydrostatic pressure on the mask's components than freshwaters, facilitating microfibres release.
In most studies, the size of released microfibres is lower than 10 µm [32], [83], [86], transparent, and come from the middle layer [85], possibly since this is the most susceptible layer, for example, to UV light [87]. The release of even submicron particles from surgical face masks has been reported. Particles of nanometric size (0.1–0.5 µm and 0.1 µm) have been detected by flow cytometry in water containing surgical face masks in mechanical agitation, with values (2.1 ± 1.4 × 1010 items/mask) significantly greater (by ∼ 6 times) than those discovered for microplastics (> 100 µm) by microscopy (1.2 ± 1.07 104 items/mask) [88]. Thus, despite the generally low level of fabric deterioration that might be observed in laboratory experiments (average loss of 1–2% of the initial weight), the high number of micro/nanoplastics that can be swiftly released from a single mask when weathering conditions are simulated under realistic intensity levels of mechanical deterioration raises great concerns. Considering the levels of micro and nanoplastics detected from this previous study and having in mind that approximately more than 10 million masks can reach the oceans monthly [89], 1 × 1011 microfibres and 1 × 1018 micro/nanoplastics are potentially entering the aquatic ecosystem every day [88]. Since wastewater treatment plants or leachate treatment plants do not have filters to remove these microplastics specifically, they end up either washed off as such or discharged into adjacent aquatic environments along with the treated effluent [90]. Such micro(nano)plastics belong to the size fraction easily uptake by aquatic organisms, which imposes a threat to their homeostasis with potential transfer along the food chains (as further discussed in Section 3.2).
The release of micro(nano)sized plastics is part of masks' environmental problem. Disposable masks also release hazardous chemical compounds often added during their manufacturing to enhance face mask quality [16], [97]; which might have bioaccumulative properties when released into the environment. A study conducted by Sullivan et al. [94] even highlights that disposable facemasks in the environment are among the primary sources of these environmental contaminants during and after the Covid-19 pandemic. The release of such additives depends not only on their chemical characteristics but also on the material in which they are contained [98] and the level of fragmentation [99]. Face masks have a three-dimensional porous structure in which the additives are dispersed but not bounded [98], [100], which contributes to their release into the environment. Besides, the larger the polymer pore diameter, the more easily small additives move through the plastic [98]. This results in rapid leaching that can last up to 24 h, particularly in the case of less hydrophobic contaminants [101].
Bussan et al. [102] determined the levels of heavy metals present in 24 surgical and KN95 face masks using ICP-MS. These authors reported that the concentrations of chromium (Cr), nickel (Ni), arsenic (As), cadmium (Cd), mercury (Hg), and titanium (Ti) were below the detection limit. In contrast, detectable concentration levels were observed for copper (Cu), antimony (Sb), and lead (Pb), which implicated a potential for their leaching [102]. When comparing leachates from both types of face masks, concentrations of all detected heavy metals were higher in surgical masks leachates than in N95 respirators [96]. Regarding only surgical face masks, inorganic substances such as Cu, Sb, Cd, and Pb (often associated with the addition of colour) have been detected in leachates obtained from pristine disposable masks agitated in water, with concentrations up to 4.17, 393, 1.92, and 6.79 μg/L, respectively [94]. Other authors detected metals such as Ti, aluminium (Al), silica (Si), magnesium (Mg), strontium (Sr), Cr, manganese (Mn), iron (Fe), cobalt (Co), Ni, and zinc (Zn) in all three layers [19], [96], [103]. However, their variety and concentrations, except for Zn [99], were higher in the outer layer [100], [104], possibly because this is the only coloured layer. In specific cases, like Argentina, where antiviral face masks impregnated with silver (Ag) and Cu nanoparticles were popularized, the environmental threat posed by their incorrect disposal is more pronounced [104]. The main concern is the possible leaching of metal nanoparticles from commercial products (e.g., face masks and antiviral textiles) since they could negatively impact aquatic environments due to their high reactivity [105].
In addition, polar organic species related to polyamide-66 monomer and oligomers (nylon-66 synthesis), polyethylene glycol [94], [106] and phthalate esters plasticizers (most commonly di-methyl phthalate, di-n-butyl phthalate, di-ethyl phthalate, di-isobutyl phthalate, and di(2-ethylhexyl) phthalate) were also detected [95]. The release of such chemical compounds was observed within the first hours of face mask exposure to water (with agitation and/or mechanical stress), as also observed with the release of micro(nano)plastics.
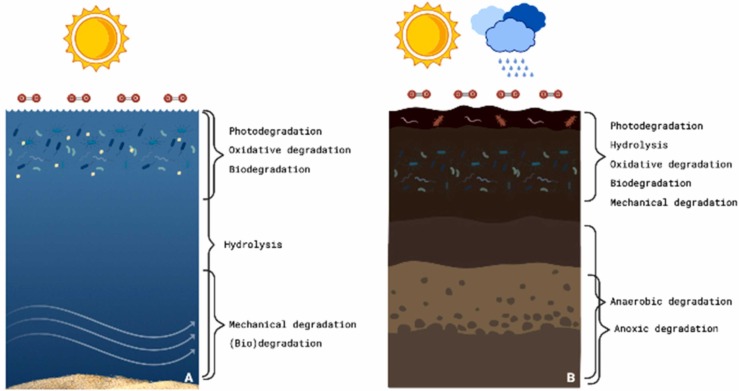
Furthermore, biotic degradation or biodegradation might also occur through the action of microorganisms, usually bacteria or fungi [74], [107]. Beyond being affected by polymer characteristics, it also depends on the organism [73], which, in turn, depends on the environment in which degradation occurs [107]. Since the levels of exposure to each one of these factors vary with the depth in the water column or soil layers, the predominant type of degradation varies throughout it ( Fig. 2).
Fig. 2.
Exemplification of the main degradation type according to the environment. A) water; B) soil.
Knicker & Velasco-Molina [92] conducted a 6-month microcosm experiment at 25 ºC to test the microbial degradability of 10 × 10 mm pieces of single mask layers and a whole mask mixed with topsoil (Cambisol). The slow carbon pool of the centre mask revealed a mean residence time (MRT) of 7 years, whereas the remaining mask material such mean residence time was between 19 and 28 years, which is longer than those determined for the slow fraction of the natural solid organic matter (although not supporting extremely long residence times of mask residues in natural soils). Results showed that disposable face masks are prone to biodegradation, although their nature still had to be revealed in future studies. In addition, the persistence of face masks in soils (for decades) might be sufficiently long to release a great quantity of micro(nano)plastics and hazardous chemical compounds, imposing a threat to soil organisms, as observed for collembolans and earthworms [91].
Face mask biodegradation in aquatic environments remains uncovered. However, as most face masks are PP-based materials, they are likely hardly degradable in the aquatic environment [108], [109], taking possibly up to 450 years for this to occur [109]. Notwithstanding, for biodegradation to start happening, there must first be abiotic degradation since, without it, the mask's surface will be hydrophobic, inhibiting the attachment of microorganisms [107]. After attachment, microorganisms can grow over time using PP as the carbon source [107]. At first, they secrete enzymes that break the main chain into smaller fragments that can pass the semi-permeable outer bacterial membranes [71]. After assimilation, microorganisms use the polymeric backbone as a source of carbon and energy in a process called mineralization [73], [108], resulting in, if in aerobic conditions, an increase in biomass and the formation of CO2 and H2O [107].
3.2. Interaction with the biota
In both terrestrial and aquatic environments, face masks greatly interfere with the biota [110], either on a physiochemical or biological level (as further discussed in Section 3.3).
Physically, several wildlife species driven by curiosity or by biological needs have already been reported to interact with PPE, particularly disposable face masks. For example, cases of entanglement, asphyxia, ingestion, and nest material usage by terrestrial and aquatic organisms have been reported worldwide ( Table 4). These interactions occurred at a macroscale (i.e., with face masks macrodebris). However, in both aquatic or terrestrial environments, face masks face biotic and abiotic factors that affect their physical and chemical integrity, resulting in their fragmentation into smaller pieces of secondary microplastics (< 5 mm) and nanoplastics (< 100 nm) [108], [111].
Table 4.
Reports of interactions between face masks and biota since the beginning of the COVID-19 pandemic.
| Country | Species | Interaction with face masks |
|---|---|---|
| The Netherlands | Fulica atra | Incorporation in the nest |
| Felis catus | Carrying | |
| Eptesicus serotinus | Entanglement | |
| Laridae sp. | Entanglement | |
| Canis lupus familiaris | Ingestion | |
| Anas platyrhynchos | Entanglement | |
| Corvus corone | Ingestion | |
| Corvus corone | Incorporation in the nest | |
| Milvus migrans | Carrying | |
| Coloeus monedula | Incorporation in the nest | |
| Columba livia domestica | Entanglement | |
| Columba palumbus | Entanglement | |
| Larus argentatus | Carrying | |
| Coloeus monedula | Carrying | |
| United Kingdom | Canis lupus familiaris | Ingestion |
| Cygnus olor | Entanglement | |
| Laridae sp. | Entanglement | |
| Falco peregrinus | Entanglement | |
| Vulpes vulpes | Entanglement | |
| Laridae sp. | Carrying | |
| Anas platyrhynchos | Entanglement | |
| Mergus merganser | Entanglement | |
| Larus argentatus | Ingestion | |
| United States of America | Sphoeroides testudineus | Entanglement |
| Canis lupus familiaris | Ingestion | |
| Haemorhous mexicanus | Incorporation in the nest | |
| Buteo jamaicensis | Entanglement | |
| Corvus corone | Incorporation in the nest | |
| Anhinga ahninga | Entanglement | |
| Uria aalge | Entanglement | |
| Australia | Climacteris picumnus | Entanglement |
| Eudyptula minor | Incorporation in the nest | |
| Corvus coronoides | Entanglement | |
| Porphyrio melanotus | Carrying | |
| Chelonia mydas | Ingestion | |
| Eudyptula minor | Incorporation in the nest | |
| Germany | Haliaeetus albicilla | Entanglement |
| Mus musculus | Incorporation in the nest | |
| Columbidae sp. | Entanglement | |
| Ciconia ciconia | Incorporation in the nest | |
| Canada | Turdus migratorius | Entanglement |
| Larus argentatus | Entanglement | |
| France | Octopus vulgaris | Hiding spot |
| Carcinus maenas | Entanglement | |
| Italy | Anas platyrhynchos | Entanglement |
| Larus michahellis | Ingestion | |
| Ireland | Cygnus olor | Entanglement |
| Fraticula arctica | Entanglement | |
| Finland | Phalacrocorax carbo | Incorporation in the nest |
| Brazil | Spheniscus magellanicus | Ingestion |
| Malaysia | Macaca fascicularis | Chewing |
| Philippines | Coral sp. | Entanglement |
| Belgium | Haematopus longirostris | Carrying |
| India | Sciuridae sp. | Incorporation in the nest |
| Scotland | Pica pica | Entanglement |
| Namibia | Arctocephalus pusillus | Entanglement |
| Japan | Chelonia mydas | Ingestion |
| Barbados | Eretmochelys imbricata | Entanglement |
| Tenerife | Delphinus delphis | Ingestion |
Source:Adapted from Hiemstra et al. [128].
If deposited on the soil surface, face masks fragments and microfibres can disperse quickly, altering soil characteristics [91], [110] and affecting their inhabitants. Mészáros et al. studied the effect of fragments obtained from different surgical face masks on the root system of Brassica napus (L.) seedlings and determined that changes depended on fragment size [112]. Besides, ingestion of these fragments by soil invertebrates may occur. Kwak & An and Kokalj et al. exposed some of these invertebrates to soil contaminated with microfibres from face masks and, in both cases, their ingestion and mobilization were detected [91], [103]. Eisenia andrei, Folsomia candida, Porcellio scaber, Tenebrio molitor larvae, and Enchytraeus crypticus are detritivores and bioturbators, thus contributing to the dispersion of fibres over a wider soil area, affecting increasingly greater proportions of soil.
Being small in size, colourful and floatable, microplastics and microfibres released from face masks can also be easily accessible to aquatic organisms that can (un)intentionally ingest them [113]. Once ingested, such items give a false sense of satiety, which will reduce food intake, leading to starvation and, eventually death [18], [57], [114]. Accidental ingestion can also be verified, albeit through distinct processes, by filtering organisms [16], [113] and deposit feeders [115]. Until the death of the organism that consumed it, plastic fragments can penetrate tissues [113] and, consequently, enter the food chain since, when higher trophic level organisms prey on these, microplastics will be transferred. Indeed, microplastics derived from various types of plastics have already been reported in several groups of organisms, for example, bivalves [116], crustaceans [117], [118], [119], [120], cephalopods [121] and fish [122], [123]. Microplastics derived from face masks have also already been detected in rotifers, bivalves, crustaceans and fish [86]. Since these organisms have commercial value and as consumers that we are, there is a high probability that microplastics will end up on our plate, potentially affecting human health [16].
However, microplastics can also enter our system through other routes, for example, inhalation [124]. While face masks contribute to the decreased risk of consumption of microplastics and other particles present in the air [125], [126], they also increase the probability of inhaling fibres from the mask itself [38], [127]. Used masks whose usage period exceeds the 4 h recommendation through continuous use or reuse tend to release a greater amount of fibres than new ones [38], [82] and, as these fibres are contained in the small area between the face and the face mask, it increases the likelihood of inhalation and, thus, their entry into the respiratory system [86], [127].
3.3. Adsorbents and leachates
When exposed to contaminated environments, while leaching intrinsic chemicals, plastics also sorb those contaminants from the environment. Face masks can act as vectors of persistent organic compounds (POPs) [129], viruses [130], pharmaceuticals [131], toxins and heavy metals [16], [132], which likely exacerbates the widespread of those pollutants and their toxicity on biota that interact with or ingest such items.
Persistent organic pollutants (POPs), which may include polychlorinated biphenyls (PCBs), organo-chlorine pesticides, and polycyclic aromatic hydrocarbons (PAHs), are synthetic and chemically stable compounds that do not readily degrade in the environment [132]. In addition to metals being face masks’ additives and, therefore, being considered leachate, they can also be adsorbents. They occur naturally and are essential to the marine environment [133]. However, their levels can exceed acceptable values due to urbanization, industrialization and the broad use of fertilizers and pesticides [134].
As occurs in the leaching of additives, the sorption of these compounds is also dependent on the characteristics of the plastic and the contaminant itself, except with particle size that does not have any relationship with sorption [135]. Indeed, sorption is mainly related to hydrophobicity [135], [136] since most of these contaminants can be defined as being hydrophobic organic contaminants (HOCs), thus, not having much affinity with water [132]. Instead, they tend to sorb to marine plastic debris [102], which contains both crystalline and amorphous regions, the last being where sorption of HOCs generally occurs since there is lower molecule organization [98]. PP, in particular, has three forms of orientation, resulting in a variation between them in the crystal ratio [137]. Isotactic polypropylene (i-PP), the one that face masks are composed of [54], has the higher ratio meaning that it has the most crystalline regions; however, there are also amorphous regions that could justify eventual HOCs sorption [137]. Besides, PP degradation decreases crystalline regions, which could justify higher sorption [15].
By being sorbed, the contaminants are removed from the water column [139], resulting in decreased availability in the water [135], [139]. However, since plastic particles are still accessible for ingestion, both additives and sorbed contaminants remain available to marine biota [138] and, if ingested, will leach on the gastrointestinal tract and eventually, along with smaller microplastics, absorbed by tissues [98], [139] and transported to the circulatory system [140]. Thus, through bioaccumulation and biomagnification, there will be the poisoning of several marine animals [114], [132]. Studies concerning the adsorption of external contaminants on micro/nano)plastics derived from weathered PPE items must be conducted and compared with conventional micro/nano)plastics [141]. Even considering the withdrawal of the mandatory mask usage by the public, the massive loads of such items on earth compartments within these 2.5 years will require attention in the near future.
3.4. (Eco)toxicological effects
Environmental toxicology is a multidisciplinary field of science that studies the adverse effects of chemical, biological, and physical agents on living organisms (including organs, tissues, cell types, organelles, and biochemical reactions) [142]. Ecotoxicology is a branch of environmental toxicology that goes higher in biological organisation, addressing adverse effects on populations, natural communities, and ecosystems [143], [144]. Micro(nano)plastics, effects can be milder to severe [94] depending on the biological organization level in study [127]. Such particles can induce physical stress due to their tridimensionality and physical properties, and/or induce chemical stress when serving as vectors of transport of chemical contaminants and pathogens, or by presenting a cocktail of hazardous chemicals that are added voluntarily during their production as additives to increase polymer properties and prolong their life [145].
Few studies have addressed the potential (eco)toxicological effects of contaminants derived from face masks ( Table 5; schematically represented in Fig. 3). Concerning soil organisms, such consequences can hugely affect the yield and quality of, for example, crops with a potential economic impact. Soil invertebrates, which are bioturbators and indicators of soil quality [146], are soil dependent not only for food but also for respiration as most of them breathe through the skin (except for the larvae of Tenebrio molitor [147]). Either way, they easily ingest microfibres and assimilate the additives or adsorbents present in masks, namely metals and organic contaminants, and bioaccumulate them [146], [147], [148], [149]. Although most results showed no effects on survival, their biological abundance will, in the long term, decrease due to changes in reproduction, growth and immune responses, thus, limiting the ecosystem function and services they provide. Moreover, if all plants and crops have the same reaction as Brassica napus (variation in roots properties, [112]), the uptake of nutrients from the soil and, hence, growth will diminish.
Table 5.
Ecotoxicological effects of exposure to face masks’ microplastics (MPs) and/or leachates.
| ORGANISM | MICROPLASTICS | EXPOSURE TIME | ECOTOXICOLOGICAL EFFECTS | REFERENCE | |
|---|---|---|---|---|---|
| SOIL | Folsomia candida | MPs-fibres Middle Layer MPs < 300 µm 1000 mg/kg |
28 days | Inhibited reproduction Retarded growth of juveniles |
[91] |
| Eisenia Andrei | MPs-fibres Middle layer MPs < 300 µm 1000 mg/kg |
21 days | Decreased intracellular esterase activity Spermatogenesis’ inhibition |
[91] | |
| Porcellio scaber | MPs-fibres All layers MPs < 250 µm 0.06%, 0.5% and 1.5% w/w |
3 weeks | No effect on survival Variation in immune response |
[103] | |
| Tenebrio molitor larvae | MPs-fibres All layers MPs < 250 µm 0.06%, 0.5% and 1.5% w/w |
3 weeks | No effect on survival Change in energy-related biomarkers |
[103] | |
| Enchytraeus crypticus | MPs-fibres All layers MPs < 250 µm 0.02%; 0.06%; 0.17%; 0.5% and 1.5% w/w |
3 weeks | No significant effect on survival or reproduction | [103] | |
| Brassica napus (L.) seedlings | MPs-fibres All layers 0.25, 1, 4, 6.25 cm2 squares 0.5% and 1% (w/v) |
5 and 14 days | Variation in lateral roots number Variation in primary root length |
[112] | |
| AQUATIC | Daphnia magna | MPs-fibres All layers MPs < 250 µm 1 mg/L,10 mg/L and 100 mg/L |
48 h | No effect on mobility or survival | [87] |
| Tigriopus japonicus nauplii | MPs-fibres All layers 10 µm <MPs< 50 µm 0, 1, 10 and 100 MPs/mL |
All life cycle | At 100 MPs/mL: Increased time of maturation Increased birth spacing time Decreased fecundity |
[83] | |
| Danio rerio larvae | UV-degraded fragments and leachates All layers < 2 mm 10 mg/L |
10 days | Down-regulation of reproduction genes Up-regulation of immune response genes |
[19] | |
| Phaeodactylum tricornutum | Face masks leachates and fibres All layers < 0.47 µm |
24, 48 and 72 h | Changes in photosynthetic apparatus Changes in intrinsic properties |
[99] | |
| CELL LINE | MC 3T3-E1 | Face masks leachates All layers 40 µm <MPs< 225 µm |
24 h | Inhibited growth Decreased viability |
[96] |
Fig. 3.
: Potential implications of face masks and their contaminants on aquatic and terrestrial environments, and on human health.
Regarding aquatic organisms, all mentioned organisms are considered key elements in the food chain. As a primary producer, this reaction from Phaeodactylum tricornutum can jeopardize the following trophic levels, resulting in a cascade effect that ends up compromising the entire food web [19], [83],
As seen through the examples, ecotoxicological responses depend on a wide variety of factors, especially the species, type of contamination, concentrations tested and time of exposure and, despite that, most of them include oxidative stress leading to reproductive failure, reduced growth and feeding and, eventually, to higher mortality [9].
These results are in line with those verified for other types of plastics. In oysters, for example, when adults Crassostrea gigas are exposed to microplastics, they exhibit no interferences on growth, but it impacts oxidative stress and lipid peroxidation, having also been registered feeding modifications and lower fertilization success which increased their mortality [150], [151]. Oysters’ larvae are also affected, derived or not from parental exposure [150], [152]. The descendants of Crassostrea gigas exposed parents can suffer from developmental abnormalities, which results in lower mobility and lower growth rates [150], [151], and those from Pinctada margaritifera have increased mortality [152]. On Scrobicularia plana, polystyrene microplastics induced DNA damage, neurotoxicity and oxidative damage [140].
Regarding mussels, it also induced oxidative stress and an increase in antioxidant enzyme activities on Perna viridis [153] and changes in immunological responses and gene expression as well as neurotoxic and genotoxicity effects on Mytilus galloprovincialis [154]. On the other hand, exposed crabs (Carcinus maenas) exhibited reduced food consumption and, thus, reduced energy available for growth [155] and, on shrimps, exposure of Palaemonetes pugio resulted in acute toxicity being fibres the most toxic microplastics [117] although, according to Carreras-Colom et al. there were no adverse effects on Aristeus antennatus [118]. On fish, Dicentrarchus labrax fed with PP microplastics, virgin or contaminated, had an intestinal inflammatory response and an imbalance in gut microbiota, however, with no alterations in their growth [156]. On Trachurus trachurus and Oryzias latipes, it impacted reproduction due to anomalies in the production of vitellogenin [157], [158]. Also, on Oryzias latipes, it was verified gene expression alterations and liver toxicity and pathology [158], [159]. In Apostichopus japonicus sea cucumbers, although microfibres are the most commonly ingested microplastics, they are eliminated without interfering with growth and faecal production [115]. Still, changes were observed in immunity and oxidative stress indices [115]. The annelid Arenicola marina exposed to PVC had reduced antioxidant capacity, immunity, feeding and ability to build holes which leads to higher mortality [160]. However, on another annelid, the oligochaeta Lumbriculus variegatus, exposure to microplastics did not affect it much as it showed no oxidative damage and growth and reproduction were not affected even in chronic exposures [161].
Exposure to fragments and fibres released from face masks (Table 5) seems not to induce lethal effects in organisms from terrestrial or aquatic environments studied, even at concentrations not yet reported in the field. Notwithstanding, some sub-lethal effects were observed, mainly reflected in physiological and biochemical changes. For example, Tenebrio molitor presented alteration in energy-related biomarkers after 3 weeks of exposure to fibres in concentrations > 0.5% w/w [103]. Ecotoxicological effects of face masks are not only related to the fragments released but also to the chemical additives that are present in their polymer matrix. For instance, it has been reported that the toxic effects of face mask leachates containing endocrine-disrupting degradation products induced a similar effect to face mask fragments alone in Danio rerio [19]. Although many of the leachable chemicals from face masks are recognized for their toxic effects on a wide range of aquatic and terrestrial organisms, they have not always been accounted for in the ecotoxicological studies conducted so far.
Although the number of studies addressing the effects of face masks released contaminants be increasing, there is still limited data on how they transfer through the food chain and their direct or indirect impacts on human health [60], [162]. In general, human exposure to face mask-derived contaminants often happens through dermal contact (skin), inhalation (air), and ingestion (food and water) [163]. For example, face masks contain metals (such as titanium dioxide – TiO2 in quantities ranging from 100 to 2000 mg/kg [164]) that can induce carcinogenic and mutagenic effects [165], [166]. Concomitantly, inorganic and organic contaminants released from face masks can lead to oxidative stress and damage the development of neurodegenerative diseases [165], [167]. For example, phthalates were detected in several disposable face masks with concentrations ranging from 115 ng/g to 37700 ng/g, which results in an estimated daily intake ranging from 3.71 to 639 ng/kg-bw/day that can be aggravated in toddlers (4–5 times higher) [168]. Although non-carcinogenic risks related to the inhalation of phthalates were found to be within safe levels, 89.3% of the face mask samples exhibited potential carcinogenic effects on humans [168]. A considerable amount of volatile organic compounds (VOCs), PAHs, and alkanes were also detected on face masks in a landmark study [169].
In addition to the adverse effects of face masks’ chemicals, inhaled microplastics and microfibres can induce, for instance, lung inflammation. In a 720 h experiment simulating breathing with different types of face masks (including disposable or reusable ones) alert for the inhalation danger of microfibres after a long wearing period (except for N95) [38]. If not retained by airway clearance mechanisms, fibres may cause localized responses such as its additives and sorbed contaminants may result in genotoxicity, reproductive toxicity, carcinogenicity and mutagenicity [127], [170]. Microplastics and microfibres released from face masks may also contribute to the dispersion of pathogens [18] and antibiotic resistance genes (ARG) in the environment [171]. The dispersion of ARG and consequent lateral gene transfer (LGT) between unilateral and/or multicellular microorganisms may increase microorganisms’ resistance to antibiotic treatments [171]. According to Zhou et al. [171], the architecture of face masks (microscopy meshing) can provide a preferable base substrate for microbial communities, including antibiotic-resistant pathogens. Given the visible extent of the impact on nature, more research is needed to understand the environmental implications of incorrect PPE disposal.
The (eco)toxicological effects reported so far lack environmental integration, and the experimental procedures present several uncertainties and uncontrolled variables that compromise the eventual comparison between face masks release contaminants and conventional plastic debris. For example, there is great variability of face masks released fragments (micro-nanoplastics/fibres) in terms of physical characteristics (e.g., size-distribution, behaviour), and most (eco)toxicological studies miss the smallest microplastics fraction (including nanoplastics), which generally pass undetected. On the other hand, tests with environmentally relevant concentrations are a challenge, mainly because it is hard to determine the exact proportion of the face masks released contaminants found in the environment. Up to now, no study has attempted to quantify face mask-released contaminants (microplastics/fibres/ chemical compounds) abundance and composition under pre- and post-pandemic conditions, particularly in areas highly impacted by face mask pollution.
4. Final remarks, research, and policy needs
The COVID-19 pandemic came to exacerbate plastic pollution, bringing increased concerns. Despite face masks' crucial role in controlling pandemic scenarios, their incorrect disposal may disturb several ecosystems, affecting their biota through direct physical interactions or by-products derived from their degradation. Studies assessing their impact are increasing daily but still lack environmental integration. To have a consolidated knowledge of the real implications of face masks on the environment, in the short and long term, for a proper risk assessment and management, it is necessary to quantify the abundance and composition of face mask-released contaminants (microplastics/fibres/ chemical compounds) under pre- and post-pandemic conditions, particularly in areas highly impacted by face masks pollution. In addition, future studies should consider: i) degradation of masks under relevant environmental settings (including in situ experiments), comparing the behaviour of their microplastics to those of more conventional plastics; ii) release of leachates; iii) ecotoxicological effects considering environmentally relevant conditions (e.g., tested concentrations, the ageing process, among others), focusing on the potential for bioaccumulation and how this may eventually affect us as consumers; iv) sorption/desorption of contaminants, and v) face masks as a vector of pathogens and antibiotic resistance genes.
Although the mandatory use of face masks by ordinary citizens in public places has been eased in many countries worldwide, such items will remain present in certain routines (e.g., in healthcare facilities) and always “around” in the case of a new epidemic or pandemic scenario. Measures and strategies have been discussed and proposed to mitigate the problem at hand by, for instance, improving the management of plastic waste during pandemic scenarios (e.g., as reviewed by Tripathi et al. [172]), improving recycling or production (with biobased biodegradable options) of these materials [22], [173], [174]. In parallel, legislation restricting certain plastics, such as single-use plastics, should be straightened, along with the stimuli on research and technology towards the development of sustainable and biodegradable options and new routes of faster degradation and mineralization of such items. Citizen science programs (e.g., collecting plastic waste on beaches) and social media must be on the political radar to increase people’s awareness and knowledge, which are essential requirements for responsible behaviours towards the use of plastics, waste prevention, and environmental conservation.
Ultimate success depends on exploring the synergies between the interested and concerned parties, from researchers, industry, government and non-government agencies, media, and the general public. Such synergism will enable meaningful progress on sustainable plastics with superior quality and efficiency when considering their waste production and management. Such interactivity will also align in terms of methods, regulations, and goals towards plastics production and management to avoid miscommunication that disenfranchises stakeholders and ultimately delays technological progress.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgements
Thanks to CESAM by FCT/MCTES (UIDP/50017/2020 + UIDB/ 50017/2020 + LA/P/0094/2020) through national funds. A.L.P.S. was funded by the Portuguese Science Foundation (FCT) through CEECIND/01366/2018 research contract; under POCH funds, co-financed by the European Social Fund and Portuguese National Funds from MEC.
Editor: Teik Thye Lim
Data availability
No data was used for the research described in the article.
References
- 1.Baloch S., Baloch M.A., Zheng T., Pei X. The coronavirus disease 2019 (COVID-19) pandemic. Tohoku J. Exp. Med. 2020;250:271–278. doi: 10.1620/tjem.250.271. [DOI] [PubMed] [Google Scholar]
- 2.Kim Y.C., Reyes-Sandoval A. In: Biomed. Innov. to Combat COVID-19. Rosales-Mendoza S., Comas-Garcia M., Gonzalez-Ortega O., editors. Academic Press; London, UK: 2022. Chapter 7—Viral-vectored vaccines against SARS-CoV-2; pp. 115–127. [Google Scholar]
- 3.Wu D., Wu T., Liu Q., Yang Z. The SARS-CoV-2 outbreak: what we know. Int. J. Infect. Dis. 2020;94:44–48. doi: 10.1016/j.ijid.2020.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.WHO Coronavirus disease (COVID-19), World Heal. Organ. 2020 https://www.who.int/emergencies/diseases/novel-coronavirus-2019/question-and-answers-hub/q-a-detail/coronavirus-disease-covid-19 (accessed October 22, 2021) [Google Scholar]
- 5.W.H.O., Coronavirus disease 2019 (COVID-19): Situation report – 51, 2020.
- 6.Humphreys J. The importance of wearing masks in curtailing the COVID-19 pandemic. J. Fam. Med. Prim. Care. 2020;9:2606. doi: 10.4103/JFMPC.JFMPC_578_20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Gordon C., Thompson A. Use of personal protective equipment during the COVID-19 pandemic. Br. J. Nurs. 2020;29:748–752. doi: 10.12968/bjon.2020.29.13.748. [DOI] [PubMed] [Google Scholar]
- 8.Mvovo I., Magagula H.B. Prevalence of Covid-19 personal protective equipment in aquatic systems and impact on associated fauna. Environ. Syst. Decis. 2022;1:1–10. doi: 10.1007/S10669-022-09851-5/TABLES/2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Prata J.C., Silva A.L.P., Duarte A.C., Rocha-Santos T. Disposable over reusable face masks: public safety or environmental disaster. Environments. 2021;8:31. doi: 10.3390/environments8040031. [DOI] [Google Scholar]
- 10.Ribeiro V.V., De-la-Torre G.E., Castro Í.B. COVID-19-related personal protective equipment (PPE) contamination in the highly urbanized southeast Brazilian coast. Mar. Pollut. Bull. 2022;177 doi: 10.1016/J.MARPOLBUL.2022.113522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Okuku E., Kiteresi L., Owato G., Otieno K., Mwalugha C., Mbuche M., Gwada B., Nelson A., Chepkemboi P., Achieng Q., Wanjeri V., Ndwiga J., Mulupi L., Omire J. The impacts of COVID-19 pandemic on marine litter pollution along the Kenyan Coast: a synthesis after 100 days following the first reported case in Kenya. Mar. Pollut. Bull. 2021;162 doi: 10.1016/j.marpolbul.2020.111840. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Mghili B., Analla M., Aksissou M. Face masks related to COVID-19 in the beaches of the Moroccan Mediterranean: an emerging source of plastic pollution. Mar. Pollut. Bull. 2022;174 doi: 10.1016/j.marpolbul.2021.113181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Gunasekaran K., Mghili B., Saravanakumar A. Personal protective equipment (PPE) pollution driven by the COVID-19 pandemic in coastal environment, Southeast Coast of India. Mar. Pollut. Bull. 2022;180 doi: 10.1016/j.marpolbul.2022.113769. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Akhbarizadeh R., Dobaradaran S., Nabipour I., Tangestani M., Abedi D., Javanfekr F., Jeddi F., Zendehboodi A. Abandoned Covid-19 personal protective equipment along the Bushehr shores, the Persian Gulf: an emerging source of secondary microplastics in coastlines. Mar. Pollut. Bull. 2021;168 doi: 10.1016/J.MARPOLBUL.2021.112386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.De-la-Torre G.E., Dioses-Salinas D.C., Pizarro-Ortega C.I., Severini M.D.F., López A.D.F., Mansilla R., Ayala F., Castillo L.M.J., Castillo-Paico E., Torres D.A., Mendoza-Castilla L.M., Meza-Chuquizuta C., Vizcarra J.K., Mejía M., De La Gala J.J.V., Ninaja E.A.S., Calisaya D.L.S., Flores-Miranda W.E., Rosillo J.L.E., Espinoza-Morriberón D., Gonzales K.N., Torres F.G., Rimondino G.N., Ben-Haddad M., Dobaradaran S., Aragaw T.A., Santillán L. Binational survey of personal protective equipment (PPE) pollution driven by the COVID-19 pandemic in coastal environments: abundance, distribution, and analytical characterization. J. Hazard. Mater. 2022;426 doi: 10.1016/j.jhazmat.2021.128070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Wang Z., Guy C., Ng K.T.W., An C. A new challenge for the management and disposal of personal protective equipment waste during the COVID-19 pandemic. Sustainability. 2021;13 doi: 10.3390/SU13137034. 7034–7034. [DOI] [Google Scholar]
- 17.Lin L., Yuan B., Zhang B., Li H., Liao R., Hong H., Lu H., Liu J., Yan C. Uncovering the disposable face masks as vectors of metal ions (Pb(Ⅱ), Cd(Ⅱ), Sr(Ⅱ)) during the COVID-19 pandemic. Chem. Eng. J. 2022;439 doi: 10.1016/J.CEJ.2022.135613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Silva A.L.P., Prata J.C., Mouneyrac C., Barcelò D., Duarte A.C., Rocha-Santos T. Risks of Covid-19 face masks to wildlife: present and future research needs. Sci. Total Environ. 2021;792 doi: 10.1016/j.scitotenv.2021.148505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Sendra M., Pereiro P., Yeste M.P., Novoa B., Figueras A. Surgical face masks as a source of emergent pollutants in aquatic systems: analysis of their degradation product effects in Danio rerio through RNA-Seq. J. Hazard. Mater. 2022;428 doi: 10.1016/J.JHAZMAT.2021.128186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Fadare O.O., Okoffo E.D. Covid-19 face masks: a potential source of microplastic fibers in the environment. Sci. Total Environ. 2020;737 doi: 10.1016/J.SCITOTENV.2020.140279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Tang Y., Han Y., Zhang W., Yu Y., Huang L., Zhou W., Shi W., Tian D. Bisphenol A and microplastics weaken the antimicrobial ability of blood clams by disrupting humoral immune responses and suppressing hemocyte chemotactic activity. Environ. Pollut. 2022;307 doi: 10.1016/j.envpol.2022.119497. [DOI] [PubMed] [Google Scholar]
- 22.Silva A.L.P., Prata J.C., Duarte A.C., Barcelò D., Rocha-Santos T. An urgent call to think globally and act locally on landfill disposable plastics under and after covid-19 pandemic: Pollution prevention and technological (Bio)remediation solutions. Chem. Eng. J. 2021;426 doi: 10.1016/j.cej.2021.131201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Prata J.C., Silva A.L.P., Walker T.R., Duarte A.C., Rocha-Santos T. COVID-19 pandemic repercussions on the use and management of plastics. Environ. Sci. Technol. 2020;54:7760–7765. doi: 10.1021/acs.est.0c02178. [DOI] [PubMed] [Google Scholar]
- 24.Do Thi H.T., Mizsey P., Toth A.J. Applicability of membranes in protective face masks and comparison of reusable and disposable face masks with life cycle assessment. Sustainability. 2021;13:12574. doi: 10.3390/su132212574. [DOI] [Google Scholar]
- 25.Duncan S., Bodurtha P., Naqvi S. The protective performance of reusable cloth face masks, disposable procedure masks, KN95 masks and N95 respirators: filtration and total inward leakage. PLoS One. 2021;16 doi: 10.1371/journal.pone.0258191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Allison A.L., Ambrose-Dempster E., Aparsi T.D., Bawn M., Casas Arredondo M., Chau C., Chandler K., Dobrijevic D., Hailes H., Lettieri P., Liu C., Medda F., Michie S., Miodownik M., Purkiss D., Ward J. The environmental dangers of employing single-use face masks as part of a COVID-19 exit strategy. UCL Open Environ. 2020 doi: 10.14324/111.444/000031.v1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Klemeš J.J., Fan Y.V., Jiang P. The energy and environmental footprints of COVID-19 fighting measures – PPE, disinfection, supply chains. Energy. 2020;211 doi: 10.1016/J.ENERGY.2020.118701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Aragaw T.A. Surgical face masks as a potential source for microplastic pollution in the COVID-19 scenario. Mar. Pollut. Bull. 2020;159 doi: 10.1016/J.MARPOLBUL.2020.111517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Lee K.P., Yip J., Kan C.W., Chiou J.C., Yung K.F. Reusable face masks as alternative for disposable medical masks: factors that affect their wear-comfort. J. Environ. Res. Public Heal. 2020;17:6623. doi: 10.3390/ijerph17186623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Goh Y., Tan B.Y.Q., Bhartendu C., Ong J.J.Y., Sharma V.K. The face mask: How a real protection becomes a psychological symbol during Covid-19. Brain. Behav. Immun. 2020;88:1–5. doi: 10.1016/J.BBI.2020.05.060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Kumar J., Katto M.S., Siddiqui A.A., Sahito B., Jamil M., Rasheed N., Ali M. Knowledge, attitude, and practices of healthcare workers regarding the use of face mask to limit the spread of the new coronavirus disease (COVID-19. Cureus. 2020;12 doi: 10.7759/cureus.7737. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Chellamani K.P., Veerasubramanian D., Balaji R.V. Surgical face masks: manufacturing. Methods Classif., J. Acad. Ind. Res. 2013;2:320. [Google Scholar]
- 33.Dutton K.C. Overview and analysis of the meltblown process and parameters, J. Text. Appar., Technol. Manag. 2009;6 https://ojs.cnr.ncsu.edu/index.php/JTATM/article/view/342 (accessed November 27, 2021) [Google Scholar]
- 34.Hegde R.R., Bhat G.S. Nanoparticle effects on the morphology and mechanical properties of polypropylene spunbond webs. J. Appl. Polym. Sci. 2010;118:3141–3155. doi: 10.1002/app.32304. [DOI] [Google Scholar]
- 35.Morais F.G., Sakano V.K., Lima L.N., Franco M.A., Reis D.C., Zanchetta L.M., Jorge F., Landulfo E., Catalani L.H., Barbosa H.M.J., John V.M., Artaxo P. Filtration efficiency of a large set of COVID-19 face masks commonly used in Brazil. Aerosol Sci. Technol. 2021;55:1028–1041. doi: 10.1080/02786826.2021.1915466. [DOI] [Google Scholar]
- 36.Abbasi A.S., Khalil A.B., Arslan M. Extensive use of face masks during COVID-19 pandemic: (micro-)plastic pollution and potential health concerns in the Arabian Peninsula. Saudi J. Biol. Sci. 2020;27:3181–3186. doi: 10.1016/J.SJBS.2020.09.054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Sharma A., Omidvarborna H., Kumar P. Efficacy of facemasks in mitigating respiratory exposure to submicron aerosols. J. Hazard. Mater. 2022;422 doi: 10.1016/J.JHAZMAT.2021.126783. [DOI] [PubMed] [Google Scholar]
- 38.Li L., Zhao X., Li Z., Song K. COVID-19: Performance study of microplastic inhalation risk posed by wearing masks. J. Hazard. Mater. 2021;411 doi: 10.1016/j.jhazmat.2020.124955. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Sankhyan S., Heinselman K.N., Ciesielski P.N., Barnes T., Himmel M.E., Teed H., Patel S., Vance M.E. Filtration performance of layering masks and face coverings and the reusability of cotton masks after repeated washing and drying. Aerosol Air Qual. Res. 2021;21 doi: 10.4209/AAQR.210117. [DOI] [Google Scholar]
- 40.Ammendolia J., Saturno J., Brooks A.L., Jacobs S., Jambeck J.R. An emerging source of plastic pollution: environmental presence of plastic personal protective equipment (PPE) debris related to COVID-19 in a metropolitan city. Environ. Pollut. 2021;269 doi: 10.1016/J.ENVPOL.2020.116160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Tesfaldet Y.T., Ndeh N.T., Budnard J., Treeson P. Assessing face mask littering in urban environments and policy implications: the case of Bangkok. Sci. Total Environ. 2022;806 doi: 10.1016/j.scitotenv.2021.150952. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Sajorne R.E., Cayabo G.D.B., Madarcos J.R.V., Madarcos K.G., Omar D.M., Ardines L.B., Sabtal S.A., Mabuhay-Omar J.A., Cheung V., Creencia L.A., Bacosa H.P. Elsevier BV,; 2022. Occurrence of Covid-19 personal protective equipment (Ppe) Litters Along the Eastern Coast of Palawan Island, Philippines. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Santarsiero A., Ciambelli P., Donsì G., Quadrini F., Briancesco R., D’Alessandro D., Fara G.M. Face masks. Technical, technological and functional characteristics and hygienic-sanitary aspects related to the use of filtering mask in the community, Ann. Di Ig. Med. Prev. e Di Comunita. 2020;32:472–520. doi: 10.7416/ai.2020.2371. [DOI] [PubMed] [Google Scholar]
- 44.Bhattacharjee S., Bahl P., Chughtai A.A., Heslop D., MacIntyre C.R. Face masks and respirators: towards sustainable materials and technologies to overcome the shortcomings and challenges. Nano Sel. 2022 doi: 10.1002/nano.202200101. [DOI] [Google Scholar]
- 45.Cook T.M. Personal protective equipment during the coronavirus disease (COVID) 2019 pandemic - a narrative review. Anaesthesia. 2020;75:920–927. doi: 10.1111/anae.15071. [DOI] [PubMed] [Google Scholar]
- 46.Adanur S., Jayswal A. Filtration mechanisms and manufacturing methods of face masks: an overview. J. Ind. Text. 2022;51:3683–3717. doi: 10.1177/1528083720980169. [DOI] [Google Scholar]
- 47.Garcia R.A., Stevanovic T., Berthier J., Njamen G., Tolnai B., Achim A. Cellulose, nanocellulose, and antimicrobial materials for the manufacture of disposable face masks: a review. BioResources. 2021;16:4321–4353. doi: 10.15376/biores.16.2.garcia. [DOI] [Google Scholar]
- 48.Windfeld E.S., Brooks M.S.L. Medical waste management - A review. J. Environ. Manag. 2015;163:98–108. doi: 10.1016/j.jenvman.2015.08.013. [DOI] [PubMed] [Google Scholar]
- 49.Lee C.C., Huffman G.L., Nalesnik R.P. Medical waste management. Environ. Sci. Technol. 1991;25:360–363. doi: 10.1142/9789814327701_0023. [DOI] [Google Scholar]
- 50.Shams M., Alam I., Mahbub M.S. Plastic pollution during COVID-19: plastic waste directives and its long-term impact on the environment. Env. Adv. 2021;5 doi: 10.1016/J.ENVADV.2021.100119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Vaverková M.D. Landfill impacts on the environment-Review. Geosciences. 2019;9:431. doi: 10.3390/geosciences9100431. [DOI] [Google Scholar]
- 52.Albuquerque F.P., Dhadwal M., Dastyar W., Azizi S.M.M., Karidio I., Zaman H., Dhar B.R. Fate of disposable face masks in high-solids anaerobic digestion: Experimental observations and review of potential environmental implications. Case Stud. Chem. Environ. Eng. 2021;3 doi: 10.1016/J.CSCEE.2021.100082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Khoo K.S., Ho L.Y., Lim H.R., Leong H.Y., Chew K.W. Plastic waste associated with the COVID-19 pandemic: crisis or opportunity? J. Hazard. Mater. 2021;417 doi: 10.1016/j.jhazmat.2021.126108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Hu X., Lin Z. Transforming waste polypropylene face masks into S-doped porous carbon as the cathode electrode for supercapacitors. Ion. (Kiel. ) 2021;27:2169–2179. doi: 10.1007/S11581-021-03949-7/FIGURES/9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Uddin M.A., Afroj S., Hasan T., Carr C., Novoselov K.S., Karim N. Environmental impacts of personal protective clothing used to combat COVID- 19. Adv. Sustain. Syst. 2021 doi: 10.1002/ADSU.202100176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Tan M., Wang Y., Luo L., Hu J. How the public used face masks in China during the coronavirus disease pandemic: a survey study. Int. J. Nurs. Stud. 2021;115 doi: 10.1016/J.IJNURSTU.2020.103853. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Roberts K.P., Phang S.C., Williams J.B., Hutchinson D.J., Kolstoe S.E., Bie J., Williams I.D., Stringfellow A.M. Increased personal protective equipment litter as a result of COVID-19 measures. Nat. Sustain. 2021;5:272–279. doi: 10.1038/s41893-021-00824-1. [DOI] [Google Scholar]
- 58.Bondaroff T.P., Cooke S. Masks Beach.: Impact Covid-19 Mar. Plast. Pollut. 2020 [Google Scholar]
- 59.Abedin M.J., Khandaker M.U., Uddin M.R., Karim M.R., Ahamad M.S.U., Islam M.A., Arif A.M., Sulieman A., Idris A.M. PPE pollution in the terrestrial and aquatic environment of the Chittagong city area associated with the COVID-19 pandemic and concomitant health implications. Environ. Sci. Pollut. Res. 2022;29:27521–27533. doi: 10.1007/S11356-021-17859-8/FIGURES/5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Kutralam-Muniasamy G., Shruti V.C. The case of “public congregation vs. COVID-19 PPE pollution”: Evidence, lessons, and recommendations from the annual pilgrimage to the Catholic Holy Site in Mexico City, Mexico. Sci. Total Environ. 2022;821 doi: 10.1016/J.SCITOTENV.2022.153424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Hatami T., Rakib M.R.J., Madadi R., De-la-Torre G.E., Idris A.M. Personal protective equipment (PPE) pollution in the Caspian Sea, the largest enclosed inland water body in the world. Sci. Total Environ. 2022;824 doi: 10.1016/J.SCITOTENV.2022.153771. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Akarsu C., Madenli Ö., Deveci E.Ü. Characterization of littered face masks in the southeastern part of Turkey. Environ. Sci. Pollut. Res. 2021;28:47517–47527. doi: 10.1007/s11356-021-14099-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Aragaw T.A., De-la-Torre G.E., Teshager A.A. Personal protective equipment (PPE) pollution driven by the COVID-19 pandemic along the shoreline of Lake Tana, Bahir Dar, Ethiopia. Sci. Total Environ. 2022;820 doi: 10.1016/J.SCITOTENV.2022.153261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.De-la-Torre G.E., Rakib M.R.J., Pizarro-Ortega C.I., Dioses-Salinas D.C. Occurrence of personal protective equipment (PPE) associated with the COVID-19 pandemic along the coast of Lima, Peru. Sci. Total Environ. 2021;774 doi: 10.1016/J.SCITOTENV.2021.145774. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Rakib M.R.J., De-la-Torre G.E., Pizarro-Ortega C.I., Dioses-Salinas D.C., Al-Nahian S. Personal protective equipment (PPE) pollution driven by the COVID-19 pandemic in Cox’s Bazar, the longest natural beach in the world. Mar. Pollut. Bull. 2021;169 doi: 10.1016/J.MARPOLBUL.2021.112497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Thiel M., de Veer D., Espinoza-Fuenzalida N.L., Espinoza C., Gallardo C., Hinojosa I.A., Kiessling T., Rojas J., Sanchez A., Sotomayor F., Vasquez N., Villablanca R. COVID lessons from the global south – Face masks invading tourist beaches and recommendations for the outdoor seasons. Sci. Total Environ. 2021;786 doi: 10.1016/J.SCITOTENV.2021.147486. [DOI] [Google Scholar]
- 67.Amuah E.E.Y., Agyemang E.P., Dankwa P., Fei-Baffoe B., Kazapoe R.W., Douti N.B. Are used face masks handled as infectious waste? Novel pollution driven by the COVID-19 pandemic. Resour. Conserv. Recycl. Adv. 2022;13 doi: 10.1016/J.RCRADV.2021.200062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.France R.L. First landscape-scale survey of the background level of COVID-19 face mask litter: Exploring the potential for citizen science data collection during a ‘pollution pilgrimage’ of walking a 250-km roadside transect. Sci. Total Environ. 2022;816 doi: 10.1016/J.SCITOTENV.2021.151569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Haddad M.B., De-la-Torre G.E., Abelouah M.R., Hajji S., Alla A.A. Personal protective equipment (PPE) pollution associated with the COVID-19 pandemic along the coastline of Agadir, Morocco. Sci. Total Environ. 2021;798 doi: 10.1016/J.SCITOTENV.2021.149282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Rathinamoorthy R., Balasaraswathi S.R. Disposable tri-layer masks and microfiber pollution – An experimental analysis on dry and wet state emission. Sci. Total Environ. 2022;816 doi: 10.1016/J.SCITOTENV.2021.151562. [DOI] [PubMed] [Google Scholar]
- 71.Shah A.A., Hasan F., Hameed A., Ahmed S. Biological degradation of plastics: a comprehensive review. Biotechnol. Adv. 2008;26:246–265. doi: 10.1016/J.BIOTECHADV.2007.12.005. [DOI] [PubMed] [Google Scholar]
- 72.Almroth B.C., Eggert H. Marine plastic pollution: sources, impacts, and policy issues. Rev. Environ. Econ. Policy. 2019;13:317–326. doi: 10.1093/reep/rez012. [DOI] [Google Scholar]
- 73.Paço A.M.S. Biodegrad. Micro.: Optim. Scale , Dep. De. Biol., Univ. De. Aveiro, Masters Diss. 2018 [Google Scholar]
- 74.Singh B., Sharma N. Mechanistic implications of plastic degradation. Polym. Degrad. Stab. 2008;93:561–584. doi: 10.1016/J.POLYMDEGRADSTAB.2007.11.008. [DOI] [Google Scholar]
- 75.Zhang K., Hamidian A.H., Tubić A., Zhang Y., Fang H., C. Wu J.K., Lam P.K.S. Understanding plastic degradation and microplastic formation in the environment: A review. Environ. Pollut. 2021;274 doi: 10.1016/J.ENVPOL.2021.116554. [DOI] [PubMed] [Google Scholar]
- 76.Lucas N., Bienaime C., Belloy C., Queneudec M., Silvestre F., Nava-Saucedo J.E. Polymer biodegradation: mechanisms and estimation techniques – A review. Chemosphere. 2008;73:429–442. doi: 10.1016/J.CHEMOSPHERE.2008.06.064. [DOI] [PubMed] [Google Scholar]
- 77.Ammala A., Bateman S., Dean K., Petinakis E., Sangwan P., Wong S., Yuan Q., Yu L., Patrick C., Leong K.H. An overview of degradable and biodegradable polyolefins. Prog. Polym. Sci. 2011;36:1015–1049. doi: 10.1016/J.PROGPOLYMSCI.2010.12.002. [DOI] [Google Scholar]
- 78.Canopoli L., Coulon F., Wagland S.T. Degradation of excavated polyethylene and polypropylene waste from landfill. Sci. Total Environ. 2020;698 doi: 10.1016/J.SCITOTENV.2019.134125. [DOI] [PubMed] [Google Scholar]
- 79.Saliu F., Veronelli M., Raguso C., Barana D., Galli P., Lasagni M. The release process of microfibers: from surgical face masks into the marine environment. Environ. Adv. 2021;4 doi: 10.1016/J.ENVADV.2021.100042. [DOI] [Google Scholar]
- 80.Wang Z., An C., Chen X., Lee K., Zhang B., Feng Q. Disposable masks release microplastics to the aqueous environment with exacerbation by natural weathering. J. Hazard. Mater. 2021;417 doi: 10.1016/J.JHAZMAT.2021.126036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Dissanayake J., Torres-Quiroz C., Mahato J., Park J. Facemasks: a looming microplastic crisis. Int. J. Environ. Res. Public Health. 2021;18:7068. doi: 10.3390/IJERPH18137068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Chen X., Chen X., Liu Q., Zhao Q., Xiong X., Wu C. Used disposable face masks are significant sources of microplastics to environment. Environ. Pollut. J. 2021;285 doi: 10.1016/j.envpol.2021.117485. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Sun J., Yang S., Zhou G.J., Zhang K., Lu Y., Jin Q., Lam P.K.S., Leung K.M.Y., He Y. Release of microplastics from discarded surgical masks and their adverse impacts on the marine copepod Tigriopus japonicus. Environ. Sci. Technol. Lett. 2021;8:1065–1070. doi: 10.1021/ACS.ESTLETT.1C00748/SUPPL_FILE/EZ1C00748_SI_001.PDF. [DOI] [Google Scholar]
- 84.Wu P., Li J., Lu X., Tang Y., Cai Z. Release of tens of thousands of microfibers from discarded face masks under simulated environmental conditions. Sci. Total Environ. 2022;806 doi: 10.1016/J.SCITOTENV.2021.150458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Shen M., Zeng Z., Song B., Yi H., Hu T., Zhang Y., Zeng G., Xiao R. Neglected microplastics pollution in global COVID-19: Disposable surgical masks. Sci. Total Environ. 2021;790 doi: 10.1016/J.SCITOTENV.2021.148130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Ma J., Chen F., Xu H., Jiang H., Liu J., Li P., Chen C.C., Pan K. Face masks as a source of nanoplastics and microplastics in the environment: Quantification, characterization, and potential for bioaccumulation. Environ. Pollut. 2021;288 doi: 10.1016/J.ENVPOL.2021.117748. [DOI] [PubMed] [Google Scholar]
- 87.Kokalj A.J., Dolar A., Drobne D., Marinšek M., Dolenec M., Škrlep L., Strmljan G., Mušič B., Škapin A.S. Environmental hazard of polypropylene microplastics from disposable medical masks: acute toxicity towards Daphnia magna and current knowledge on other polypropylene microplastics, Microplastics Nanoplastics 2021 21. 2. 1–15. 2022 doi: 10.1186/S43591-021-00020-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Morgana S., Casentini B., Amalfitano S. Uncovering the release of micro/nanoplastics from disposable face masks at times of COVID-19. J. Hazard. Mater. 2021;419 doi: 10.1016/J.JHAZMAT.2021.126507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Adyel T.M. Accumulation of plastic waste during COVID-19. Sci. (80-. 2020;). 369:1314–1315. doi: 10.1126/science.abd9925. [DOI] [PubMed] [Google Scholar]
- 90.Costa J.P., Santos P.S.M., Duarte A.C., Rocha-Santos T. Nano)plastics in the environment – Sources, fates and effects. Sci. Total Environ. 566–. 2016;567:15–26. doi: 10.1016/J.SCITOTENV.2016.05.041. [DOI] [PubMed] [Google Scholar]
- 91.Kwak J.I., An Y.J. Post COVID-19 pandemic: Biofragmentation and soil ecotoxicological effects of microplastics derived from face masks. J. Hazard. Mater. 2021;416 doi: 10.1016/J.JHAZMAT.2021.126169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Knicker H., Velasco-Molina M. Biodegradability of disposable surgical face masks littered into soil systems during the covid 19 pandemic - A first approach using microcosms. Soil Syst. 2022;6:39. doi: 10.3390/SOILSYSTEMS6020039. [DOI] [Google Scholar]
- 93.Liang H., Ji Y., Ge W., Wu J., Song N., Yin Z., Chai C. Release kinetics of microplastics from disposable face masks into the aqueous environment. Sci. Total Environ. 2022;816 doi: 10.1016/J.SCITOTENV.2021.151650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Sullivan G.L., Delgado-Gallardo J., Watson T.M., Sarp S. An investigation into the leaching of micro and nano particles and chemical pollutants from disposable face masks - linked to the COVID-19 pandemic. Water Res. 2021;196 doi: 10.1016/j.watres.2021.117033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Wang X., Okoffo E.D., Banks A.P.W., Li Y., Thomas K.V., Rauert C., Aylward L.L., Mueller J.F. Phthalate esters in face masks and associated inhalation exposure risk. J. Hazard. Mater. 2022;423 doi: 10.1016/J.JHAZMAT.2021.127001. [DOI] [PubMed] [Google Scholar]
- 96.Liu Z., Wang J., Yang X., Huang Q., Zhu K., Sun Y., Van Hulle S., Jia H. Generation of environmental persistent free radicals (EPFRs) enhances ecotoxicological effects of the disposable face mask waste with the COVID-19 pandemic. Environ. Pollut. 2022;301 doi: 10.1016/J.ENVPOL.2022.119019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Liu R., Mabury S.A. Single-use face masks as a potential source of synthetic antioxidants to the environment. Environ. Sci. Technol. Lett. 2021;8:651–655. doi: 10.1021/acs.estlett.1c00422. [DOI] [Google Scholar]
- 98.Teuten E.L., Saquing J.M., Knappe D.R.U., Barlaz M.A., Jonsson S., Björn A., Rowland S.J., Thompson R.C., Galloway T.S., Yamashita R., Ochi D., Watanuki Y., Moore C., Viet P.H., Tana T.S., Prudente M., Boonyatumanond R., Zakaria M.P., Akkhavong K., Ogata Y., Hirai H., Iwasa S., Mizukawa K., Hagino Y., Imamura A., Saha M., Takada H. Transport and release of chemicals from plastics to the environment and to wildlife. Philos. Trans. R. Soc. B Biol. Sci. 2009;364:2027–2045. doi: 10.1098/RSTB.2008.0284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Sendra M., Rodriguez-Romero A., Yeste P., Blasco J., Tovar-Sánchez A. Products released from surgical face masks can provoke cytotoxicity in the marine diatom Phaeodactylum tricornutum. Sci. Total Environ. 2022 doi: 10.1016/j.scitotenv.2022.156611. [DOI] [PubMed] [Google Scholar]
- 100.Bridson J.H., Gaugler E.C., Smith D.A., Northcott G.L., Gaw S. Leaching and extraction of additives from plastic pollution to inform environmental risk: A multidisciplinary review of analytical approaches. J. Hazard. Mater. 2021;414 doi: 10.1016/j.jhazmat.2021.125571. [DOI] [PubMed] [Google Scholar]
- 101.León V.M., García I., González E., Samper R., Fernández-González V., Muniategui-Lorenzo S. Potential transfer of organic pollutants from littoral plastics debris to the marine environment. Environ. Pollut. 2018;236:442–453. doi: 10.1016/J.ENVPOL.2018.01.114. [DOI] [PubMed] [Google Scholar]
- 102.Bussan D.D., Snaychuk L., Bartzas G., Douvris C. Quantification of trace elements in surgical and KN95 face masks widely used during the SARS-COVID-19 pandemic. Sci. Total Environ. 2022;814 doi: 10.1016/J.SCITOTENV.2021.151924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Kokalj A.J., Dolar A., Drobne D., Škrlep L., Škapin A.S., Marolt G., Nagode A., van Gestel C.A.M. Effects of microplastics from disposable medical masks on terrestrial invertebrates. J. Hazard. Mater. 2022 doi: 10.1016/j.jhazmat.2022.129440. [DOI] [PubMed] [Google Scholar]
- 104.Ardusso M., Forero-López A.D., Buzzi N.S., Spetter C.V., Fernández-Severini M.D. COVID-19 pandemic repercussions on plastic and antiviral polymeric textile causing pollution on beaches and coasts of South America. Sci. Total Environ. 2021;763 doi: 10.1016/J.SCITOTENV.2020.144365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Forero-López A.D., Fabiani M., Lassalle V.L., Spetter C.V., Severini M.D.F. Critical review of the characteristics, interactions, and toxicity of micro/nanomaterials pollutants in aquatic environments. Mar. Pollut. Bull. 2022;174 doi: 10.1016/J.MARPOLBUL.2021.113276. [DOI] [PubMed] [Google Scholar]
- 106.Fernández-Arribas J., Moreno T., Bartrolí R., Eljarrat E. COVID-19 face masks: a new source of human and environmental exposure to organophosphate esters. Environ. Int. 2021;154 doi: 10.1016/J.ENVINT.2021.106654. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Arutchelvi J., Sudhakar M., Arkatkar A., Doble M., Bhaduri S., Uppara P.V. Biodegradation of polyethylene and polypropylene. Indian J. Biotechnol. 2008;7:9–22. [Google Scholar]
- 108.Khoironi A., Hadiyanto H., Anggoro S., Sudarno S. Evaluation of polypropylene plastic degradation and microplastic identification in sediments at Tambak Lorok coastal area, Semarang, Indonesia. Mar. Pollut. Bull. 2020;151 doi: 10.1016/j.marpolbul.2019.110868. [DOI] [PubMed] [Google Scholar]
- 109.Schleiffenbaum E.M. Petition fordert priorisierung der stoffmasken-produktion. Greenpeace Switz. 2020 〈https://www.greenpeace.ch/de/medienmitteilung/59915/petition-fordert-priorisierung-der-stoffmasken-produktion/〉 accessed July 6, 2022. [Google Scholar]
- 110.Du H., Huang S., Wang J. Environmental risks of polymer materials from disposable face masks linked to the COVID-19 pandemic. Sci. Total Environ. 2022;815 doi: 10.1016/J.SCITOTENV.2022.152980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Hasan N.A., Heal R.D., Bashar A., Haque M.M. Face masks: protecting the wearer but neglecting the aquatic environment? - A perspective from Bangladesh. Environ. Chall. 2021;4 doi: 10.1016/J.ENVC.2021.100126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Mészáros E., Bodor A., Szierer Á., Kovács E., Perei K., Tölgyesi C., Bátori Z., Feigl G. Indirect effects of COVID-19 on the environment: how plastic contamination from disposable surgical masks affect early development of plants. J. Hazard. Mater. 2022;436 doi: 10.1016/j.jhazmat.2022.129255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Dharmaraj S., Ashokkumar V., Hariharan S., Manibharathi A., Show P.L., Chong C.T., Ngamcharussrivichai C. The COVID-19 pandemic face mask waste: A blooming threat to the marine environment. Chemosphere. 2021;272 doi: 10.1016/J.CHEMOSPHERE.2021.129601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Selvaranjan K., Navaratnam S., Rajeev P., Ravintherakumaran N. Environmental challenges induced by extensive use of face masks during COVID-19: A review and potential solutions. Environ. Chall. 2021;3 doi: 10.1016/J.ENVC.2021.100039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Mohsen M., Zhang L., Sun L., Lin C., Wang Q., Liu S., Sun J., Yang H. Effect of chronic exposure to microplastic fibre ingestion in the sea cucumber Apostichopus japonicus. Ecotoxicol. Environ. Saf. 2021;209 doi: 10.1016/J.ECOENV.2020.111794. [DOI] [PubMed] [Google Scholar]
- 116.Wakkaf T., El Zrelli R., Kedzierski M., Balti R., Shaiek M., Mansour L., Tlig-Zouari S., Bruzaud S., Rabaoui L. Microplastics in edible mussels from a southern Mediterranean lagoon: preliminary results on seawater-mussel transfer and implications for environmental protection and seafood safety. Mar. Pollut. Bull. 2020;158 doi: 10.1016/J.MARPOLBUL.2020.111355. [DOI] [PubMed] [Google Scholar]
- 117.Gray A.D., Weinstein J.E. Size- and shape-dependent effects of microplastic particles on adult daggerblade grass shrimp (Palaemonetes pugio) Environ. Toxicol. Chem. 2017;36:3074–3080. doi: 10.1002/ETC.3881. [DOI] [PubMed] [Google Scholar]
- 118.Carreras-Colom E., Constenla M., Soler-Membrives A., Cartes J.E., Baeza M., Padrós F., Carrassón M. Spatial occurrence and effects of microplastic ingestion on the deep-water shrimp Aristeus antennatus. Mar. Pollut. Bull. 2018;133:44–52. doi: 10.1016/J.MARPOLBUL.2018.05.012. [DOI] [PubMed] [Google Scholar]
- 119.Severini M.F., Buzzi N.S., López A.F., Colombo C.V., Sartor G.C., Rimondino G.N., Truchet D.M. Chemical composition and abundance of microplastics in the muscle of commercial shrimp Pleoticus muelleri at an impacted coastal environment (Southwestern Atlantic. Mar. Pollut. Bull. 2020;161 doi: 10.1016/J.MARPOLBUL.2020.111700. [DOI] [PubMed] [Google Scholar]
- 120.Watts A.J.R., Lewis C., Goodhead R.M., Beckett S.J., Moger J., Tyler C.R., Galloway T.S. Uptake and retention of microplastics by the shore crab Carcinus maenas. Environ. Sci. Technol. 2014;48:8823–8830. doi: 10.1021/ES501090E/SUPPL_FILE/ES501090E_SI_001.ZIP. [DOI] [PubMed] [Google Scholar]
- 121.Abidli S., Lahbib Y., Menif N.T.El. Microplastics in commercial molluscs from the lagoon of Bizerte (Northern Tunisia) Mar. Pollut. Bull. 2019;142:243–252. doi: 10.1016/J.MARPOLBUL.2019.03.048. [DOI] [PubMed] [Google Scholar]
- 122.Jovanović B. Ingestion of microplastics by fish and its potential consequences from a physical perspective. Integr. Environ. Assess. Manag. 2017;13:510–515. doi: 10.1002/IEAM.1913. [DOI] [PubMed] [Google Scholar]
- 123.Rochman C.M., Tahir A., Williams S.L., Baxa D.V., Lam R., Miller J.T., Teh F.C., Werorilangi S., Teh S.J. Anthropogenic debris in seafood: plastic debris and fibers from textiles in fish and bivalves sold for human consumption.Sci. Rep. 2015;51(5):1–10. doi: 10.1038/srep14340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Cox K.D., Covernton G.A., Davies H.L., Dower J.F., Juanes F., Dudas S.E. Human consumption of microplastics. Environ. Sci. Technol. 2019;53:7068–7074. doi: 10.1021/acs.est.9b01517. [DOI] [PubMed] [Google Scholar]
- 125.Dris R., Gasperi J., Mirande C., Mandin C., Guerrouache M., Langlois V., Tassin B. A first overview of textile fibers, including microplastics, in indoor and outdoor environments. Environ. Pollut. 2017;221:453–458. doi: 10.1016/j.envpol.2016.12.013. [DOI] [PubMed] [Google Scholar]
- 126.Athey S.N., Erdle L.M. Are we underestimating anthropogenic microfiber pollution? A critical review of occurrence, methods, and reporting. Environ. Toxicol. Chem. 2021;00:1–16. doi: 10.1002/etc.5173. [DOI] [PubMed] [Google Scholar]
- 127.Torres-Agullo A., Karanasiou A., Moreno T., Lacorte S. Overview on the occurrence of microplastics in air and implications from the use of face masks during the COVID-19 pandemic. Sci. Total Environ. 2021;800 doi: 10.1016/j.scitotenv.2021.149555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.A.F. Hiemstra, L. Rambonnet, B. Gravendeel, M. Schilthuizen, Effects of COVID-19 litter on animal life, (2022). https://www.covidlitter.com/ (accessed June 3, 2022).
- 129.Anastopoulos I., Pashalidis I. Single-use surgical face masks, as a potential source of microplastics: do they act as pollutant carriers? J. Mol. Liq. 2021;326 doi: 10.1016/J.MOLLIQ.2020.115247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Mol M.P.G., Caldas S. Can the human coronavirus epidemic also spread through solid waste. Waste Manag. Res. 2020;38:485–486. doi: 10.1177/0734242X20918312. [DOI] [PubMed] [Google Scholar]
- 131.Lin L., Yuan B., Hong H., Li H., He L., Lu H., Liu J., Yan C. Post COVID-19 pandemic: disposable face masks as a potential vector of antibiotics in freshwater and seawater. Sci. Total Environ. 2022;820 doi: 10.1016/J.SCITOTENV.2022.153049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Rios L.M., Moore C., Jones P.R. Persistent organic pollutants carried by synthetic polymers in the ocean environment. Mar. Pollut. Bull. 2007;54:1230–1237. doi: 10.1016/J.MARPOLBUL.2007.03.022. [DOI] [PubMed] [Google Scholar]
- 133.Ansari T.M., Marr I.L., Tariq N. Heavy metals in marine pollution perspective–A mini review. J. Appl. Sci. 2003;4:1–20. doi: 10.3923/jas.2004.1.20. [DOI] [Google Scholar]
- 134.Priyadarshani I., Sahu D., Rath B. Microalgal bioremediation: current practices and perspectives. J. Biochem. Technol. 2012;3:299–304. [Google Scholar]
- 135.Hüffer T., Hofmann T. Sorption of non-polar organic compounds by micro-sized plastic particles in aqueous solution. Environ. Pollut. 2016;214:194–201. doi: 10.1016/j.envpol.2016.04.018. [DOI] [PubMed] [Google Scholar]
- 136.Mei W., Chen G., Bao J., Song M., Li Y., Luo C. Interactions between microplastics and organic compounds in aquatic environments: a mini review. Sci. Total Environ. 2020;736 doi: 10.1016/J.SCITOTENV.2020.139472. [DOI] [PubMed] [Google Scholar]
- 137.A. Siciliano, B. Clifford, D. Cahn, B. Hude-velasco, The recovery of waste polypropylene & synthesis of a sustainable extrudable paste for additive manufacturing, (2020).
- 138.Rios L.M., Jones P.R., Moore C.J. Quantitation of persistent organic pollutants adsorbed on plastic debris from the Northern Pacific Gyre’s “eastern garbage patch. J. Environ. Monit. 2010;12:2189–2312. doi: 10.1039/c0em00239a. [DOI] [PubMed] [Google Scholar]
- 139.Lee H., Shim W.J., Kwon J.H. Sorption capacity of plastic debris for hydrophobic organic chemicals. Sci. Total Environ. 470–. 2014;471:1545–1552. doi: 10.1016/J.SCITOTENV.2013.08.023. [DOI] [PubMed] [Google Scholar]
- 140.Ribeiro F., Garcia A.R., Pereira B.P., Fonseca M., Mestre N.C., Fonseca T.G., Ilharco L.M., Bebianno M.J. Microplastics effects in Scrobicularia plana. Mar. Pollut. Bull. 2017;122:379–391. doi: 10.1016/J.MARPOLBUL.2017.06.078. [DOI] [PubMed] [Google Scholar]
- 141.Torres F.G., De-la-Torre G.E. Historical microplastic records in marine sediments: current progress and methodological evaluation. Reg. Stud. Mar. Sci. 2021;46 doi: 10.1016/J.RSMA.2021.101868. [DOI] [Google Scholar]
- 142.Tolleson W.H. Mechanistic ecotoxicology and environmental toxicology. J. Environ. Sci. Heal. Part C. 2018;36:164–166. doi: 10.1080/10590501.2018.1492201. [DOI] [PubMed] [Google Scholar]
- 143.Stapleton P.A. Toxicological considerations of nano-sized plastics, AIMS. Environ. Sci. 2019;6:367–378. doi: 10.3934/ENVIRONSCI.2019.5.367/FULLTEXT.HTML. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Moriarty F. Ecotoxicology. Hum. Exp. Toxicol. 1988;7:437–441. doi: 10.1177/096032718800700510. [DOI] [PubMed] [Google Scholar]
- 145.Xu J.L., Lin X., Wang J.J., Gowen A.A. A review of potential human health impacts of micro- and nanoplastics exposure. Sci. Total Environ. 2022;851 doi: 10.1016/J.SCITOTENV.2022.158111. [DOI] [PubMed] [Google Scholar]
- 146.Fründ H.C., Graefe U., Tischer S. Biol. Earthworms, Springer Berlin Heidelberg. 2011. Earthworms as bioindicators of soil quality; pp. 261–278. [DOI] [Google Scholar]
- 147.Vijver M., Jager T., Posthuma L., Peijnenburg W. Metal uptake from soils and soil–sediment mixtures by larvae of Tenebrio molitor (L.) (Coleoptera) Ecotoxicol. Environ. Saf. 2003;54:277–289. doi: 10.1016/S0147-6513(02)00027-1. [DOI] [PubMed] [Google Scholar]
- 148.Castro-Ferreira M.P., Roelofs D., van Gestel C.A.M., Verweij R.A., Soares A.M.V.M., Amorim M.J.B. Enchytraeus crypticus as model species in soil ecotoxicology. Chemosphere. 2012;87:1222–1227. doi: 10.1016/J.CHEMOSPHERE.2012.01.021. [DOI] [PubMed] [Google Scholar]
- 149.Menta C., Maggiani A., Vattuone Z. Effects of Cd and Pb on the survival and juvenile production of Sinella coeca and Folsomia candida. Eur. J. Soil Biol. 2006;42:181–189. doi: 10.1016/J.EJSOBI.2006.01.001. [DOI] [Google Scholar]
- 150.Bringer A., Cachot J., Dubillot E., Prunier G., Huet V., Clérandeau C., Evin L., Thomas H. Intergenerational effects of environmentally-aged microplastics on the Crassostrea gigas. Environ. Pollut. 2022;294 doi: 10.1016/J.ENVPOL.2021.118600. [DOI] [PubMed] [Google Scholar]
- 151.Sussarellu R., Suquet M., Thomas Y., Lambert C., Fabioux C., Pernet M.E.J., Goïc N.L., Quillien V., Mingant C., Epelboin Y., Corporeau C., Guyomarch J., Robbens J., Paul-Pont I., Soudant P., Huvet A. Oyster reproduction is affected by exposure to polystyrene microplastics. Proc. Natl. Acad. Sci. U. S. A. 2016;113:2430–2435. doi: 10.1073/PNAS.1519019113/-/DCSUPPLEMENTAL. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Gardon T., Huvet A., Paul-Pont I., Cassone A.L., Koua M.S., Soyez C., Jezequel R., Receveur J. G. Le Moullac, Toxic effects of leachates from plastic pearl-farming gear on embryo-larval development in the pearl oyster Pinctada margaritifera. Water Res. 2020;179 doi: 10.1016/J.WATRES.2020.115890. [DOI] [PubMed] [Google Scholar]
- 153.Vasanthi R.L., Arulvasu C., Kumar P., Srinivasan P. Ingestion of microplastics and its potential for causing structural alterations and oxidative stress in Indian green mussel Perna viridis– A multiple biomarker approach. Chemosphere. 2021;283 doi: 10.1016/J.CHEMOSPHERE.2021.130979. [DOI] [PubMed] [Google Scholar]
- 154.Avio C.G., Gorbi S., Milan M., Benedetti M., Fattorini D., D’Errico G., Pauletto M., Bargelloni L., Regoli F. Pollutants bioavailability and toxicological risk from microplastics to marine mussels. Environ. Pollut. 2015;198:211–222. doi: 10.1016/J.ENVPOL.2014.12.021. [DOI] [PubMed] [Google Scholar]
- 155.Watts A.J.R., Urbina M.A., Corr S., Lewis C., Galloway T.S. Ingestion of plastic microfibers by the crab Carcinus maenas and its effect on food consumption and energy balance. Environ. Sci. Technol. 2015;49:14597–14604. doi: 10.1021/ACS.EST.5B04026. [DOI] [PubMed] [Google Scholar]
- 156.Montero D., Rimoldi S., Torrecillas S., Rapp J., Moroni F., Herrera A., Gómez M., Fernández-Montero A., Terova G. Impact of polypropylene microplastics and chemical pollutants on European sea bass (Dicentrarchus labrax) gut microbiota and health. Sci. Total Environ. 2022;805 doi: 10.1016/J.SCITOTENV.2021.150402. [DOI] [PubMed] [Google Scholar]
- 157.Chenet T., Mancia A., Bono G., Falsone F., Scannella D., Vaccaro C., Baldi A., Catani M., Cavazzini A., Pasti L. Plastic ingestion by Atlantic horse mackerel (Trachurus trachurus) from central Mediterranean Sea: a potential cause for endocrine disruption. Environ. Pollut. 2021;284 doi: 10.1016/J.ENVPOL.2021.117449. [DOI] [PubMed] [Google Scholar]
- 158.Rochman C.M., Kurobe T., Flores I., Teh S.J. Early warning signs of endocrine disruption in adult fish from the ingestion of polyethylene with and without sorbed chemical pollutants from the marine environment. Sci. Total Environ. 2014;493:656–661. doi: 10.1016/J.SCITOTENV.2014.06.051. [DOI] [PubMed] [Google Scholar]
- 159.Rochman C.M., Hoh E., Kurobe T., Teh S.J. Ingested plastic transfers hazardous chemicals to fish and induces hepatic stress. Sci. Rep. 2013;3 doi: 10.1038/SREP03263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Browne M.A., Niven S.J., Galloway T.S., Rowland S.J., Thompson R.C. Microplastic moves pollutants and additives to worms, reducing functions linked to health and biodiversity. Curr. Biol. 2013;23:2388–2392. doi: 10.1016/J.CUB.2013.10.012. [DOI] [PubMed] [Google Scholar]
- 161.Silva C.J.M., Silva A.L.P., Campos D., Soares A.M.V.M., Pestana J.L.T., Gravato C. Lumbriculus variegatus (oligochaeta) exposed to polyethylene microplastics: biochemical, physiological and reproductive responses. Ecotoxicol. Environ. Saf. 2021;207 doi: 10.1016/J.ECOENV.2020.111375. [DOI] [PubMed] [Google Scholar]
- 162.De-la-Torre G.E., Pizarro-Ortega C.I., Dioses-Salinas D.C., Ammendolia J., Okoffo E.D. Investigating the current status of COVID-19 related plastics and their potential impact on human health. Curr. Opin. Toxicol. 2021;27:47–53. doi: 10.1016/J.COTOX.2021.08.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Li M., Hou Z., Meng R., Hao S., Wang B. Unraveling the potential human health risks from used disposable face mask-derived micro/nanoplastics during the COVID-19 pandemic scenario: A critical review. Environ. Int. 2022;170 doi: 10.1016/j.envint.2022.107644. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Verleysen E., Ledecq M., Siciliani L., Cheyns K., Vleminckx C., Blaude M.N., De Vos S., Brassinne F., Van Steen F., Nkenda R., Machiels R., Waegeneers N., Van Loco J., Mast J. Titanium dioxide particles frequently present in face masks intended for general use require regulatory control. Sci. Rep. 2022;12:1–9. doi: 10.1038/s41598-022-06605-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Fu Z., Xi S. The effects of heavy metals on human metabolism. Toxicol. Mech. Methods. 2020;30:167–176. doi: 10.1080/15376516.2019.1701594. [DOI] [PubMed] [Google Scholar]
- 166.Vijayaraj A.S., Mohandass C., Joshi D. Microremediation of tannery wastewater by siderophore producing marine bacteria. Environ. Technol. 2020;41:3619–3632. doi: 10.1080/09593330.2019.1615995. [DOI] [PubMed] [Google Scholar]
- 167.Giacoppo S., Galuppo M., Calabrò R.S., D’Aleo G., Marra A., Sessa E., Bua D.G., Potortì A.G., Dugo G., Bramanti P., Mazzon E. Heavy metals and neurodegenerative diseases: an observational study. Biol. Trace Elem. Res. 2014;161:151–160. doi: 10.1007/s12011-014-0094-5. [DOI] [PubMed] [Google Scholar]
- 168.Xie H., Han W., Xie Q., Xu T., Zhu M., Chen J. Face mask—A potential source of phthalate exposure for human. J. Hazard. Mater. 2022;422 doi: 10.1016/J.JHAZMAT.2021.126848. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Jin L., Griffith S.M., Sun Z., Yu J.Z., Chan W. On the flip side of mask wearing: increased exposure to volatile organic compounds and a risk-reducing solution. Environ. Sci. Technol. 2021;55:14095–14104. doi: 10.1021/acs.est.1c04591. [DOI] [PubMed] [Google Scholar]
- 170.Gasperi J., Wright S.L., Dris R., Collard F., Mandin C., Guerrouache M., Langlois V., Kelly F.J., Tassin B. Microplastics in air: Are we breathing it in? Curr. Opin. Environ. Sci. Heal. 2018;1:1–5. doi: 10.1016/j.coesh.2017.10.002. [DOI] [Google Scholar]
- 171.Zhou S.Y.D., Lin C., Yang K., Yang L.Y., Yang X.R., Huang F.Y., Neilson R., Su J.Q., Zhu Y.G. Discarded masks as hotspots of antibiotic resistance genes during COVID-19 pandemic. J. Hazard. Mater. 2022;425 doi: 10.1016/J.JHAZMAT.2021.127774. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Tripathi A., Tyagi V.K., Vivekanand V., Bose P., Suthar S. Challenges, opportunities and progress in solid waste management during COVID-19 pandemic. Case Stud. Chem. Environ. Eng. 2020;2 doi: 10.1016/J.CSCEE.2020.100060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Silva A.L.P., Prata J.C., Walker T.R., Campos D., Duarte A.C., Soares A.M.V.M., Barcelò D., Rocha-Santos T. Rethinking and optimising plastic waste management under COVID-19 pandemic: Policy solutions based on redesign and reduction of single-use plastics and personal protective equipment. Sci. Total Environ. 2020;742 doi: 10.1016/J.SCITOTENV.2020.140565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Nghiem L.D., Iqbal H.M.N., Zdarta J. The shadow pandemic of single use personal protective equipment plastic waste: A blue print for suppression and eradication. Case Stud. Chem. Environ. Eng. 2021;4 doi: 10.1016/J.CSCEE.2021.100125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Benson N.U., Bassey D.E., Palanisami T. COVID pollution: impact of COVID-19 pandemic on global plastic waste footprint. Heliyon. 2021;7 doi: 10.1016/J.HELIYON.2021.E06343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Aragaw T.A., Mekonnen B.A. Current plastic pollution threat due to COVID-19 and its possible mitigation techniques: a waste-to-energy conversion by pyrolysis. Environ. Syst. Res. 2021;10:8. doi: 10.1186/s40068-020-00217-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Zhenfang Z., Dongxiao J., He H., Ramakrishna S. Electro Ultra Fibres Adv. face masks. Mat. Sci. Eng. 2021;143 doi: 10.1016/j.mser.2020.100594. [DOI] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
No data was used for the research described in the article.