In neuroscience research, neuronal models are crucial tools for elucidating the molecular and cellular processes involved in the disorders of the nervous system. Facilitating easily and reproducibly executable studies, in vitro models, such as the SH-SY5Y cell line culture, help us explore the pathophysiological mechanisms of neurodegenerative diseases; they are also essential for the efficient screening of drugs for treating the diseases of the nervous system (Peng et al., 2021; Strother et al., 2021).
Originating from the SK-N-SH cell subline (established from a bone marrow biopsy of a 4-year-old female child), the neuroblastoma cell line SH-SY5Y (Figure 1A) is a largely immortalized cell line used in in vitro models of neurological disorders, including Alzheimer’s disease (AD), neurotoxicity, ischemia, and amyotrophic lateral sclerosis (Xicoy et al., 2017; Peng et al., 2021). As they are human-derived, they express several human-specific proteins and protein isoforms that are not inherently present in primary rodent cell cultures (Kovalevich and Langford, 2013). The strain shows moderate dopamine-β-hydroxylase activity and insignificant levels of choline acetyltransferase, acetylcholinesterase, butyryl-cholinesterase, basal norepinephrine and tyrosine hydroxylase activity. Tyrosine hydroxylase, the rate-limiting enzyme in the catecholamine synthesis pathway, converts tyrosine to L-DOPA, the precursor of dopamine, which is converted to norepinephrine by dopamine-β-hydroxylase. Therefore, the SH-SY5Y cell line may exhibit a catecholaminergic phenotype because it can synthesize both dopamine and norepinephrine (Xicoy et al., 2017).
Figure 1.
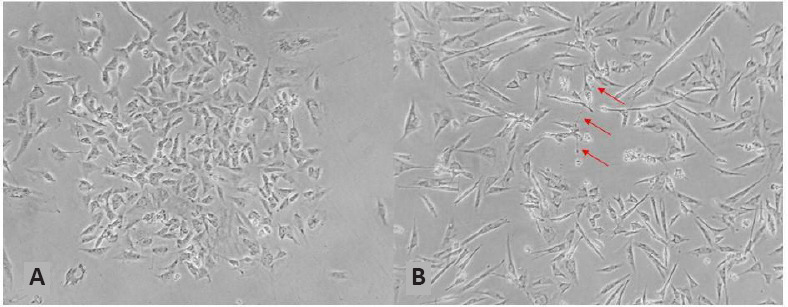
Neuroblastoma cell line SH-SY5Y.
(A) Undifferentiated; (B) cell in the process of differentiation (day 6). Arrows indicate examples of neurites. Unpublished data.
To mimic the effects of drug treatment and/or genetic approaches on the progression of Parkinson’s disease (PD), the expression of candidate genes (obtained from genetic studies in families with PD) may be studied using the cell line. For example, 1-methyl-4-phenylpyridinium (MPP+), 6-hydroxydopamine, and rotenone can alter multiple cellular pathways, affecting mitochondrial dysfunction and oxidative stress (Xicoy et al., 2017).
Moreover, SH-SY5Y cells can be induced to differentiate (Figure 1B) into a more mature, neuron-like phenotype by manipulating the culture medium, allowing large-scale expansion even before differentiation, presenting a low-cost option compared to primary neurons, and obviating the ethical concerns associated with primary human neuronal culture. However, SH-SY5Y cells also have limitations, such as their limited relevance in modeling actual neurons. As it is derived from neuroblastoma, this cell line has cancer-like properties that influence its differentiation fate, viability, growth performance, metabolic properties, and genomic stability. Therefore, in the case of PD, for example, SH-SY5Y cells are not purely ADergic, presenting characteristics that are quite different from normal ADergic neuronal characteristics (Xicoy et al., 2017). The cell line can be compared to embryonic stem cell- or induced pluripotent stem cell-derived human neurons. Unlike primary neurons, induced pluripotent stem cell-derived neurons are easy to obtain and propagate, similar to SH-SY5Y. Unlike SH-SY5Y, their differentiation is more costly and more time-consuming—however, as a tradeoff, they do show a more relevant neuron-like phenotype and will therefore make a better model for many applications, such as the modeling of neurological disorders with a genetic background, general toxicity studies, mimicking axonal degeneration, and electrophysiological studies (Yamanaka, 2020).
When determining whether undifferentiated or differentiated cells should be used for a specific experiment, the following properties must be considered: the formation and extension of neuritic processes; increased electrical excitability of the plasma membrane; formation of synaptophysin-positive functional synapses; and induction of neurotransmitter enzymes, neurotransmitters, and specific neurotransmitter receptors, which are present in differentiated neurons. Although retinoic acid is the most used differentiation agent, there are different methods for cell differentiation, such as phorbol esters, dibutyryl cyclic AMP, and staurosporine, a PKC inhibitor that triggers neuritogenesis and cell cycle arrest in SH-SY5Y cells. These methods, unlike cell differentiation with retinoic acid, can increase vulnerability to toxic insults and lead to decreased levels of Bcl-2 (Kovalevich and Langford, 2013).
Widely cited recent papers using SH-SY5Y cells have demonstrated their important applications, as described below. Regarding PD and the use of undifferentiated SH-SY5Y cells as a disease model, a study published by Ramalingam et al. (2019) demonstrated that a complex mechanism is involved in DAergic neurodegeneration in response to rotenone, a natural insecticide, pesticide, and piscicide extracted from the roots of plants of the genera Lonchocarpus and Derris. This mechanism involves mitochondrial dysfunction, altered mTOR nutrient-sensing pathways, ER stress, and disrupted expression of tethering proteins mediating mitochondria-associated ER contacts protein dynamics. In addition, using undifferentiated SH-SY5Y cells, Martinez et al. (2020) investigated whether oxidative stress, altered neurodevelopment, and cell death pathways contribute to the response to the widely used herbicide glyphosate and its metabolite aminomethylphosphonic acid. According to the authors, the SH-SY5Y cell line can be considered as an in vitro system for pesticide screening. The results demonstrated that glyphosate and aminomethylphosphonic acid induced cytotoxic effects on neuronal development, oxidative stress, and cell death via apoptotic, autophagic, and necrotic pathways, and they confirmed that environmental exposure to glyphosate is a health concern. The results found by Liu et al. (2019) indicated that ginsenoside Re, one of the most important active components among ginsenosides (bioactive compounds found in the medicinal herb P. ginseng), may be a potential therapeutic agent for the treatment of AD. In summary, the authors demonstrated that ginsenoside Re exhibited strong neuroprotective activity against Aβ25–35-induced neurotoxicity in undifferentiated SH-SY5Y cells, inhibiting the apoptosis of ROS-dependent ASK-1/JNK/BAX cells and activating Nrf2/HO-1 antioxidant pathways.
Based on the hypothesis that increased expression level of the TRMP2 gene results in the greater susceptibility of SH-SY5Y cells to ROS-induced cell death, An et al. (2019) generated a stable cell line that overexpressed the human TRPM2 channel and used a propidium iodide staining assay to directly examine H2O2-induced death in wild-type cells and in cells overexpressing TRPM2. This study provides consistent evidence that exposure to H2O2 induces SH-SY5Y cell death, regardless of TRPM2 expression levels—TRPM2 expression level increases the susceptibility to ROS-induced cell death.
Rakotoarisoa et al. (2019a) focused on the characterization of self-assembled liquid crystalline nanostructures of catalase and curcumin, targeting a combination therapy for neurodegenerative diseases. The results showed that nanoparticles loaded with catalase and curcumin effectively delivered therapeutic molecules into SH-SY5Y cells, as evidenced by increased antioxidant enzyme activity. Cubosomes preserve catalase, which ensures cell defense against ROS. Furthermore, double-charged cubosomes, which contained a combination of catalase and curcumin, also exhibited enhanced catalase activity in differentiated SH-SY5Y cells. In another study, Rakotoarisoa et al. (2019b) investigated the use of liquid crystalline nanocarriers, which co-encapsulate curcumin and fish oil, as potential neuroprotective agents for the combined treatment of neurodegenerative diseases. In SH-SY5Y cells differentiated with retinoic acid, morphological changes were observed in cells subjected to ROS damage after exposure to hydrogen peroxide. The effects of curcumin and fish oil co-encapsulated by nanocarriers were quantified using flow cytometry. In addition, intracellular ROS levels and apoptosis levels were measured to compare their neuroprotective potential. These results indicate that dual-drug-loaded cubosomes may be suitable for combination treatments against neurodegenerative diseases. Omar et al. (2018) used SH-SY5Y cells as a model for AD, where they induced the formation and aggregation of Aβ fibrils with or without copper and L-DOPA as toxicity inducers; they evaluated the therapeutic potential of both isolated olive biophenols and olive extract. Exposure of SH-SY5Y cells to Aβ42, copper, and L-DOPA resulted in decreased cell viability and morphological changes owing to severe increase in the levels of reactive oxygen and nitrogen species. After 24 hours of pretreatment with olive biophenols, attenuation of cell death caused by Aβ-induced toxicity was observed, inhibiting overproduction and facilitating the elimination of excess reactive oxygen and nitrogen species, thus reducing apoptosis by increasing and prolonging the upregulation of survival pathways, including phosphatidylinositol-3-OH kinase/AKT and JNK (c-Jun N-terminal kinase) pathways. Thus, the authors showed that olive biophenols reduce toxicity and protect SH-SY5Y cells and may be promising neuroprotective compounds against neurodegenerative diseases, such as Alzheimer’s and Parkinson’s disease.
Lead (Pb), a heavy metal, is recognized as an environmental pollutant that threatens human health, particularly because of its negative effect on the development of the nervous system. Therefore, Yang et al. (2020) used the SH-SY5Y model to evaluate the effect of metformin (2 mM) on mitochondrial dynamics in the presence of Pb (25 μM). SH-SY5Y cells exposed to Pb show an increase in mitochondrial fragmentation and morphology. Mitochondrial fragmentation is dependent on oxidative stress, and the latter results in decreased energy availability and cell death. Metformin treatment ameliorated Pb-induced neurotoxicity by inhibiting mitochondrial fragmentation. The protective effect of metformin is mediated by activation of the Nrf2/ARE antioxidant pathway in an AMPK-dependent manner. Thus, this study shows that metformin can abolish Pb-induced mitochondrial fragmentation and impair cellular homeostasis, depending on the activation of the AMPK/Nrf2 pathway in SH-SY5Y cells. Zhao et al. (2020) evaluated, in SH-SY5Y cells, the involvement of the long non-coding RNA small molecule RNA host gene 1 (SNHG1) in the pathology of PD. Increased SNHG1 expression level was found in SH-SY5Y cells treated with 1-methyl-4-phenylpyridinium (MPP+), which reduced viability and increased apoptosis in SH-SY5Y cells. In addition, MPP+-induced cytotoxicity decreased after the upregulation of SNHG1 by miR-153-3p-mediated suppression. In addition, phosphate and tension homology deleted on chromosome ten (PTEN) was identified as a direct target of miR-153-3p, and forced expression of PTEN exhibited functions similar to SNHG1 overexpression in regulating viability and apoptosis in SH-SY5Y cells. Finally, the study showed that SNHG1 activated the PTEN/AKT/mTOR signaling pathway in SH-SY5Y cells by targeting miR-153-3p, thereby demonstrating the potential of SNHG1 as a promising therapeutic target for PD.
Abdul et al. (2019) conducted another study using SH-SY5Y cells to model AD. They evaluated the effects of four compounds, curcumin, piperine, bacoside A, and chebulinic acid. After in silico selection, two compounds, curcumin and piperine, were selected, and a cell viability of up to 85% was observed, indicating that the compounds protect against Aβ-induced neuronal damage. They also showed that curcumin and piperine achieved a synergistic effect, indicating that the combination of these two compounds at lower concentrations may provide better results than their use alone for Aβ proteins. The combination of curcumin and piperine inhibited the aggregation and disaggregation of fibrillar Aβ42 and reversed Aβ-induced upregulation of neuronal oxidative stress. In conclusion, curcumin and piperine have promising neuroprotective effects that could potentially be therapeutic and curative for AD.
Several studies have been conducted to generate, in addition to publications, patent filings from acquired knowledge. Based on the information contained in the WIPO Patentscope, 97 patents containing the use of SH-SY5Y cells as a cell model were deposited from 2019 to the present, many of which focused on the search for new molecules for the treatment of diseases associated with the nervous system. Justin et al. (2022) established a methodology for the synthesis and neuroprotective activity of two glitazones for the treatment of neurodegenerative disorders, targeting the activation of central PGC-1 signaling through the stimulation of the PPARγ receptor, because the functional interaction between PGC-1 and PPARR-y is considered a key interaction in the normal physiology of the neuroprotective mechanism. The results indicated that pretreatment with glitazones increased cell viability, protected cell morphology, and decreased the release of pro-inflammatory cytokines IL-1β, tumor necrosis factor alpha, lipid peroxide, and nitric oxide in SH-SY5Y human neuroblastoma cells (patent application number IN202141039298).
Finally, both differentiated and undifferentiated SH-SY5Y cells have been widely used as neuronal cell models to study PD. However, a variety of methods to differentiate SH-SY5Y cells are also being studied, although they have some limitations, as some differentiation agents have more targeted applications for neurotoxicity. Briefly, the SH-SY5Y cell line is no longer the best option available for many applications; however, it is not obsolete. Each model has limitations, and the best model for an application is the simplest one that answers the research question. In some cases, SH-SY5Y may indeed be the best fit for the research goal.
This work was supported in part by Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (Capes-Brazil), No. 001 (to MIG).
Footnotes
C-Editors: Zhao M, Liu WJ, Qiu Y; T-Editor: Jia Y
References
- 1.Abdul Manap AS, Wei Tan AC, Leong WH, Yin Chia AY, Vijayabalan S, Arya A, Wong EH, Rizwan F, Bindal U, Koshy S, Madhavan P. Synergistic effects of curcumin and piperine as potent acetylcholine and amyloidogenic inhibitors with significant neuroprotective activity in SH-SY5Y cells via computational molecular modeling and in vitro assay. Front Aging Neurosci. 2019;11:206. doi: 10.3389/fnagi.2019.00206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.An X, Fu Z, Mai C, Wang W, Wei L, Li D, Li C, Jiang LH. Increasing the TRPM2 channel expression in human neuroblastoma SH-SY5Y Cells augments the susceptibility to ROS-induced cell death. Cells. 2019;8:28. doi: 10.3390/cells8010028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Justin A, Dhanabal SP, Ashwini P, Kumar BRP, Mandal S. Methodology for the syntheses and neuroprotective activity of two glitazones to treat neurodegenerative disorders. Depositor: JSS Academy of Higher Education and Research, Mysuru. IN202141039298. Deposit:Aug. 31 2021 Grant:May 20, 2022. 2022. Available at:https://patentscope.wipo.int/search/en/detail.jsf?docId=IN362988629&_cid=P11-LA1FOZ-20868-1 .
- 4.Kovalevich J, Santerre M, Langford D. Considerations for the use of SH-SY5Y neuroblastoma cells in neurobiology. Methods Mol Biol. 2021;2311:9–23. doi: 10.1007/978-1-0716-1437-2_2. [DOI] [PubMed] [Google Scholar]
- 5.Liu M, Bai X, Yu S, Zhao W, Qiao J, Liu Y, Zhao D, Wang J, Wang S. Ginsenoside Re inhibits ROS/ASK-1 dependent mitochondrial apoptosis pathway and activation of Nrf2-antioxidant response in beta-amyloid-challenged SH-SY5Y cells. Molecules. 2019;24:2687. doi: 10.3390/molecules24152687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Martinez MA, Rodriguez JL, Lopez-Torres B, Martinez M, Martinez-Larranaga MR, Maximiliano JE, Anadon A, Ares I. Use of human neuroblastoma SH-SY5Y cells to evaluate glyphosate-induced effects on oxidative stress, neuronal development and cell death signaling pathways. Environ Int. 2020;135:105414. doi: 10.1016/j.envint.2019.105414. [DOI] [PubMed] [Google Scholar]
- 7.Omar SH, Scott CJ, Hamlin AS, Obied HK. Olive biophenols reduces Alzheimer's pathology in SH-SY5Y cells and APPswe mice. Int J Mol Sci. 2018;20:125. doi: 10.3390/ijms20010125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Peng Y, Chu S, Yang Y, Zhang Z, Pang Z, Chen N. Neuroinflammatory in vitro cell culture models and the potential applications for neurological disorders. Front Pharmacol. 2021;12:671734. doi: 10.3389/fphar.2021.671734. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Rakotoarisoa M, Angelov B, Espinoza S, Khakurel K, Bizien T, Angelova A. Cubic liquid crystalline nanostructures involving catalase and curcumin:BioSAXS study and catalase peroxidatic function after cubosomal nanoparticle treatment of differentiated SH-SY5Y cells. Molecules. 2019a;24:3058. doi: 10.3390/molecules24173058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Rakotoarisoa M, Angelov B, Garamus MV, Angelova A. Curcumin- and fish oil-loaded spongosome and cubosome nanoparticles with neuroprotective potential against H2O2-induced oxidative stress in differentiated human SH-SY5Y cells. ACS Omega. 2019b doi:10.1021/acsomega.8B03101. [Google Scholar]
- 11.Ramalingam M, Huh YJ, Lee YI. The impairments of alpha-synuclein and mechanistic target of rapamycin in rotenone-induced SH-SY5Y cells and mice model of Parkinson's disease. Front Neurosci. 2019;13:1028. doi: 10.3389/fnins.2019.01028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Strother L, Miles GB, Holiday AR, Cheng Y, Doherty GH. Long-term culture of SH-SY5Y neuroblastoma cells in the absence of neurotrophins:A novel model of neuronal ageing. J Neurosci Methods. 2021;362:109301. doi: 10.1016/j.jneumeth.2021.109301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Xicoy H, Wieringa B, Martens GJ. The SH-SY5Y cell line in Parkinson's disease research:a systematic review. Mol Neurodegener. 2017;12:10. doi: 10.1186/s13024-017-0149-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Yamanaka S. Pluripotent stem cell-based cell therapy-promise and challenges. Cell Stem Cell. 2020;27:523–531. doi: 10.1016/j.stem.2020.09.014. [DOI] [PubMed] [Google Scholar]
- 15.Yang L, Li X, Jiang A, Li X, Chang W, Chen J, Ye F. Metformin alleviates lead-induced mitochondrial fragmentation via AMPK/Nrf2 activation in SH-SY5Y cells. Redox Biol. 2020;36:101626. doi: 10.1016/j.redox.2020.101626. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Zhao J, Geng L, Chen Y, Wu C. SNHG1 promotes MPP(+)-induced cytotoxicity by regulating PTEN/AKT/mTOR signaling pathway in SH-SY5Y cells via sponging miR-153-3p. Biol Res. 2020;53:1. doi: 10.1186/s40659-019-0267-y. [DOI] [PMC free article] [PubMed] [Google Scholar]


