Abstract
Context
Anterior cruciate ligament injury commonly occurs via noncontact motor coordination errors that result in excessive multiplanar loading during athletic movements. Preventing motor coordination errors requires neural sensorimotor integration activity to support knee-joint neuromuscular control, but the underlying neural mechanisms driving injury-risk motor control are not well understood.
Objective
To evaluate brain activity differences for knee sensorimotor control between athletes with high or low injury-risk mechanics.
Design
Case-control study.
Setting
Research laboratory.
Patients or Other Participants
Of 38 female high school soccer players screened, 10 were selected for analysis based on magnetic resonance imaging compliance, injury-risk classification via 3-dimensional biomechanics during a drop vertical jump, and matching criteria to complete neuroimaging during knee motor tasks.
Main Outcome Measure(s)
Peak knee-abduction moment during landing was used for group allocation into the high (≥21.74 newton meters [Nm], n = 9) or low (≤10.6 Nm, n = 11) injury-risk classification (n = 11 uncategorized, n = 7 who were not compliant with magnetic resonance imaging). Ten participants (5 high risk, 5 low risk) with adequate data were matched and compared across 2 neuroimaging paradigms: unilateral knee-joint control and unilateral multijoint leg press against resistance.
Results
Athletes with high injury-risk biomechanics had less neural activity in 1 sensory-motor cluster for isolated knee-joint control (precuneus, peak Z score = 4.14, P ≤ .01, 788 voxels) and greater brain activity for the multijoint leg press in 2 cognitive-motor clusters: the frontal cortex (peak Z score = 4.71, P < .01, 1602 voxels) and posterior cingulate gyrus (peak Z score = 4.43, P < .01, 725 voxels) relative to the low injury-risk group.
Conclusions
The high injury-risk group's lower relative engagement of neural sensory resources controlling the knee joint may elevate demand on cognitive motor resources to control loaded multijoint action. The neural activity profile in the high injury-risk group may manifest as a breakdown in neuromuscular coordination, resulting in elevated knee-abduction moments during landing.
Keywords: functional magnetic resonance imaging, motion capture, injury prevention, neuroimaging, sensorimotor control, landing neuromuscular control
Current recommendations for physical activity encourage young adolescents to engage in at least 60 minutes of moderate to vigorous physical activity per day.1 Although this may lower the risk for cardiac disease, physical activity can increase the incidence for traumatic musculoskeletal injuries, such as anterior cruciate ligament (ACL) rupture. Direct ACL injury costs to the US health care system are approximately $13 billion per year, and the injury contributes to early-onset osteoarthritis and subsequent reduced quality of life.2,3 The most common mechanism of ACL injury is noncontact, theorized to be secondary to sensorimotor coordination errors that result in the failure to ensure safe knee positioning and prevent excessive ligament loading.4
The potential neuromuscular coordination contribution to noncontact injury is further supported by recent evidence5 suggesting a neural connectivity-based predisposition to ACL injury. Specifically, depressed sensorimotor connectivity and somatosensory-cerebellar connectivity was reported in men and women, respectively, who went on to sustain an ACL injury.5 However, these studies, despite being prospective in nature, quantified resting-state brain activation and may have been limited without more direct measures of sensorimotor performance or neuromuscular control. Neuromuscular control is commonly quantified via the peak external knee-abduction moment (pKAM), or medial collapse of the knee, during the landing from a standardized drop vertical jump (DVJ). Knee-abduction landing mechanics can be used as a reliable screening and intervention target, predicting ACL injury in young female athletes with 78% sensitivity and 73% specificity.6 To connect neural activity and injury-risk mechanics, resting-state electrocortical activity differences were investigated in female athletes classified as at high or low injury-risk based on pKAM.7 Athletes at high risk demonstrated greater deterministic, or regular and predictable, neural profiles than those at low risk, indicating a decreased ability to adapt to unanticipated perturbations. Thus, less adaptable neural profiles may have caused breakdowns in the perception-action cycle of neuromuscular control and injury-risk biomechanics. Though informative, this previous research evaluated brain activity at rest, and movement-related neural activity may have further delineated specific central nervous system (CNS) processes for neuromuscular control contributing to injury risk. To our knowledge, no authors have directly linked lower extremity movement-related neural activity and standardized biomechanical injury-risk measures during landing to inform the development of interventions that address the role of the CNS in ACL injury.
This gap in knowledge represents important CNS contributions to injury risk that are unaccounted for and not targeted with current prevention efforts.8 Thus, the purpose of our study was to evaluate neural correlates of isolated knee-joint and multijoint control of the lower extremity relative to injury-risk neuromuscular control during the DVJ. Specifically, we aimed to isolate neural signatures associated with maladaptive biomechanics to support the development of more targeted sensorimotor interventions that promote neuroplasticity for ACL injury-risk reduction. We hypothesized that athletes with high-risk biomechanics would exhibit different neural activity during functional magnetic resonance imaging (fMRI) movement tasks relative to athletes with low-risk biomechanics.
METHODS
Participant and Group Selection
Female high school soccer players between the ages of 13 and 19 years with no history of lower extremity injury were recruited for the study. Participants and a parent or legal guardian if under age 18 completed informed assent and consent, respectively, before any data were collected. The Cinncinnati Children's Hospital Medical Center Institutional Review Board approved all study procedures. Thirty-eight female high school soccer players (age = 16.10 ± 0.87 years, height = 165.10 ± 4.64 cm, weight = 63.43 ± 8.80 kg) completed a standardized DVJ for group determination (described in the next section). Seven girls had orthodontic braces, claustrophobia, or another magnetic resonance imaging (MRI)–related contraindication, were not able to complete the neuroimaging experiments, and were excluded. Of the remaining 31 athletes, 9 were placed in the high injury-risk group (≥21.74 newton-meters [Nm]) and 11 in the low injury-risk group (≤10.6 Nm) based on the established threshold cutoffs).6,9 Eleven girls were between thresholds and were excluded. Head motion during neuroimaging with motor tasks was a critical concern, and due to the small sample size, we minimized confounding factors in the data by conservatively limiting inclusion to <2 mm of absolute motion and <0.30 mm of relative head motion during either motor task. The conservative head-motion threshold applied to this study also included screening for excessive task-correlated head motion, blood oxygenation level–dependent signal model fit with dissociable baseline (ie, rest) and task (ie, move) blocks, and a stable baseline and task activation profile. Of the 20 girls assigned to a risk group, 7 had excessive right-sided head motion during neuroimaging, and 5 had excessive left-sided head motion. As such, the left side was selected for analysis in all participants. Of the 5 girls with excessive head motion, 3 were in the high injury-risk group, resulting in 6 potential matches between injury-risk groups. One athlete could not be matched across groups due to activity level or sport participation status differences. Final analyses thus yielded 5 pairs for neuroimaging assessment (n = 10), based on age (±1 year), sport (all soccer), activity level or sport participation (starter versus reserve), and suitability for MRI (no metal, no claustrophobia, etc).
The injury-risk classification for each participant was evaluated with 3-dimensional (3D) biomechanics during a standardized DVJ. This task involved falling forward from a 31-cm box and then immediately performing a maximum vertical jump while raising both arms and reaching for a target set at 100% of maximum jump height.6 Each participant's bilateral pKAM was computed during landing and averaged across 3 trials to determine the injury-risk classification as high (≥21.74 Nm) or low (≤10.6 Nm). Computations were based on previous research establishing a high pKAM as a potential marker for injury risk and on biomechanical studies that identified thresholds for injury risk relative to knee loading.6,9
Key Points
Athletes with high injury-risk biomechanics exhibited altered brain activity for knee motor control relative to athletes with low injury-risk biomechanics.
The neural activity associated with high injury-risk biomechanics may reduce the ability to cognitively regulate knee position, which reduces neuromuscular capacity and contributes to the injury risk.
Identification of neurologic contributors to injury risk may enable novel intervention development and inform future research to enhance injury-prevention effectiveness.
Biomechanical Risk Quantification and Analysis
Participants were instrumented with 31 markers of 9-mm diameter (B&L Engineering) to create 3D coordinates for kinematic and kinetic analysis of the DVJ. Marker trajectories and ground reaction forces during the DVJ task were quantified using a 39-camera, high-speed, passive optical 3D motion-capture system (Raptor-E; Motion Analysis Corp) sampled at 240 Hz and two 60- × 90-cm force plates (Bertec Corp) sampled at 1200 Hz, respectively, and postprocessed with Cortex software (version 6.2; Motion Analysis Corp). Before participants performed the DVJ, we recorded data from all joints in a neutral position during a standing trial. A kinematic model comprising 12 skeletal segments (upper arm × 2, lower arm × 2, trunk-thorax, pelvis, thigh × 2, shank × 2, and foot × 2) and 36 degrees of freedom was defined using Visual3D (version 5.0.1; C-Motion, Inc). Vertical ground reaction forces (VGRF) and kinematic data were low-pass filtered with a cubic smoothing spline at a 12-Hz cutoff frequency. The VGRF data for each limb were used to normalize the kinetic data to 100% of stance at 1% increments, with initial contact defined as VGRF >10 N. Based on the 3D kinematic and force-plate data, pKAM was computed using inverse-dynamic analysis in Visual3D along a standard joint coordinate system.
Functional Magnetic Resonance Imaging Data Collection
All girls wore standardized athletic shorts and socks without shoes for consistent skin-tactile feedback. They first completed a mock scanner session in which they were familiarized with the knee motor tasks via a video that explained and illustrated the motor tasks. After watching the video, each person practiced the task with experimenter guidance. Participants were then positioned supine on the MRI table with customized padding and straps to minimize head motion. To reduce motion and increase comfort about the head area, the girls were placed in the head coil with ear plugs and headphones. Small foam pads were also placed around the headphones to fill any remaining space between the headphones and coil. Fluidized positioners (models 14010004, 1401007, 1401011; Mölnlycke) were placed underneath the individual's back and head. Straps were used to secure the torso. Handlebars were attached to the MRI table to standardize hand position and minimize accessory motion. Next, participants practiced the tasks with a qualified staff member responsible for the familiarization protocol. This process ensured reliable data collection and quality while enabling neural activity collection independent of task novelty; the task had previously demonstrated intraclass correlation coefficients of 0.62 to 0.92 for neural activity across the primary sensorimotor regions.10
Isolated Knee Extension-Flexion
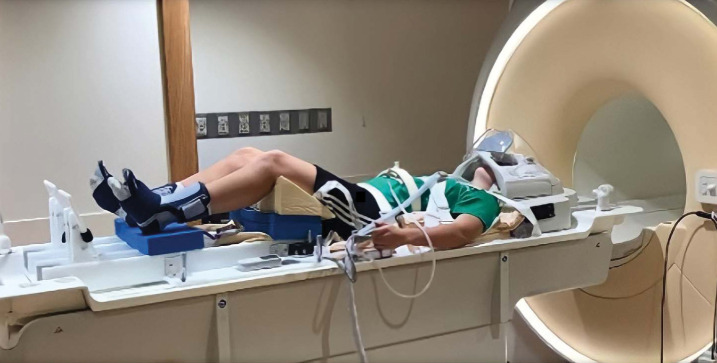
Participants were placed in a custom test apparatus designed to allow knee-extension and -flexion while minimizing head and accessory joint motion (Figure 1). They performed a left unilateral knee extension and flexion coordination task in which they moved the lower leg between terminal knee extension (0°) and 45° of flexion at a rate of 1.2 Hz paced to a metronome. The participants were instructed not to touch their heel on the table or rest during flexion and to avoid locking the knee during extension to prevent jerking movements and minimize external cues for position control. This task was performed repeatedly for 30 seconds over 4 cycles, with 30 seconds of rest between cycles. Each scan session started with a 30-second blank screen, and then the individual saw a countdown of “2, 1, Move Left.” At the end of the movement block, “2, 1, and STOP” was the cue to ease the leg back to the normal position and minimize head motion during transitions.
Figure 1.
Functional magnetic resonance imaging set-up for the isolated knee task.
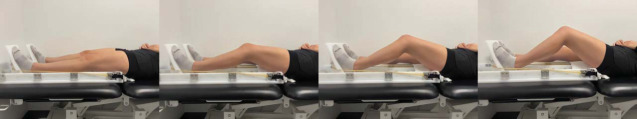
Multijoint Leg Press Against Resistance (Ankle, Knee, and Hip Extension-Flexion)
Participants were then placed in a leg-press unit with 2 foot pedals that ran on tracks (Figure 2). The task involved flexing the left knee to approximately 45° along the sagittal-plane track and then extending to 0° against resistance bands set at approximately 20% of each girl's mass at full extension. Her feet were strapped to the pedals and moved in tracks during the task with the pelvis fixed to the table, and thus, hip and ankle motion occurred to accommodate knee flexion. Rubber resistance bands were placed around the pedals to provide tension when the girls extended the lower extremity. Each scan was paced in the same manner as the knee-positioning task. Participants practiced both tasks with standardized range of motion and goniometer monitoring and were visually monitored during the scan; scans were repeated if individuals did not go through the approximate range of motion or displayed accessory movements.
Figure 2.
Functional magnetic resonance imaging while the participant completes the leg press.
The MRI Data Acquisition and Analyses
Neuroimaging data were acquired on an Ingenia scanner (model 3.0 T; Philips Medical Systems) using a 32-channel phased-array head coil. The fMRI data were acquired with a gradient echo-planar imaging sequence using a periodic block design in which each 30 seconds of motor task was followed by 30 seconds of rest. Twenty frames were acquired per cycle, for a total of 4 cycles, with a 3-second repetition time, 3.75 × 3.75 mm in-plane resolution, and 5-mm slice thickness for 38 axial slices (field of view = 240 × 240 mm, matrix = 64 × 64). Image registration involved the collection of a 3D high-resolution T1-weighted image (repetition time = 8.3 milliseconds, echo time = 3.7 milliseconds, field of view = 256 × 256 mm, matrix = 256 × 256, slice thickness = 1 mm, 176 slices).
Analyses were performed using the software package Functional MRI of the Brain (FMRIB) software library (FSL; version 5.0.10; Oxford Centre).11,12 Functional MRI data were spatially registered to correct for head motion and spatially smoothed to improve sensitivity in quantifying functional activation during the knee-movement tasks.13 Standard preprocessing was applied to individual data, including nonbrain removal, spatial smoothing using a Gaussian kernel of 5 mm full width at half maximum, and standard motion correction.14 Realignment parameters, including 3 rotations and 3 translations, from the motion-correction procedure were included in the design matrix as covariates to account for the confounding effects of head movement.15 High-pass temporal filtering at 120 seconds and time-series statistical analysis were performed using a linear model with local autocorrelation correction.16 Functional images were coregistered with the corresponding high-resolution T1-weighted image and normalized to the standard 2-mm Montreal Neuroimaging Institute 152 template using the FMRIB nonlinear image registration tool.14,15 The individual (task level) and group comparison analyses were completed with whole-brain statistical parametric mapping to identify neural activity specific to the task (movement relative to rest) and then task activity differences between groups (high risk relative to low risk). First-level analysis of lower extremity functional movement relative to rest was carried out using a general linear model analysis, which yielded a β coefficient with a standard error that was used to conduct a t test. The t statistic was converted to a z score and corrected for multiple-comparisons error using random field threshold-cluster corrected z > 2.3 and a significance threshold of P < .05 at the participant level to determine regions active during movement relative to rest conditions.
Statistical Analysis
Neuromuscular control was quantified via pKAM and normalized pKAM, which were checked for outliers, normality, and homogeneity of variances. No outliers were identified via boxplots, and normality was not violated as assessed by the Shapiro-Wilk test for each group and variable (P > .05). Homogeneity of variances was checked with the Levene test and was not violated (P > .05). Therefore, we used parametric testing to determine differences between groups. An independent t test was calculated to compare pKAM and demographics between the high-risk and low-risk groups with α < 0.05. Independent t tests were also conducted to compare brain activity between those with high and low injury-risk biomechanics (P values < .05; random field cluster was corrected for multiple comparisons, z > 2.3; implemented with FMRIB's local analysis of mixed effects 1 + 2).17
RESULTS
Participant demographics are reported in the Table. The high injury-risk group had a higher pKAM for both absolute and mass-normalized moments (33.19 ± 5.32 Nm and 0.49 ± 0.09 Nm/kg, respectively) than the low injury-risk group (2.80 ± 2.54 Nm and 0.05 ± 0.04 Nm/kg, respectively; P < .001; Figure 3).
Table.
Participant Demographics
| Characteristic |
Injury-Risk Group |
P Valuec |
|
| High (n = 5)a |
Low (n = 5)b |
||
| Mean ± SD | |||
| Age, y | 16.2 ± 0.84 | 16.4 ± 0.89 | .72 |
| Height, cm | 166.6 ± 4.39 | 163.4 ± 5.90 | .36 |
| Weight, kg | 68.54 ± 13.52 | 60.08 ± 5.25 | .23 |
| Body mass index, kg/m2 | 24.68 ± 4.56 | 22.5 ± 1.35 | .34 |
Knee-abduction moment ≥21.74 Nm.
Knee-abduction moment ≤10.6 Nm.
P values for t test between groups.
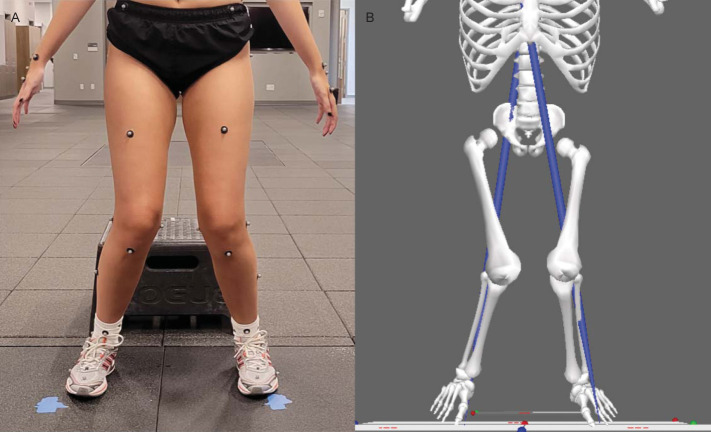
Figure 3.
A, Exemplar of high injury-risk movement associated with the brain activity in Figures 4 and 5. B, High injury-risk movement data prepared in Visual3D (C-Motion, Inc) for determination of peak knee-abduction moment.
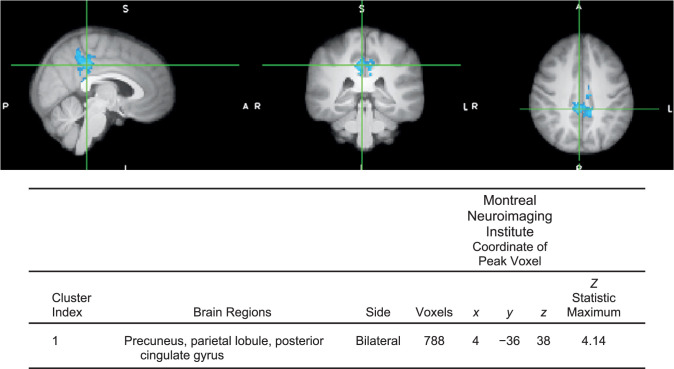
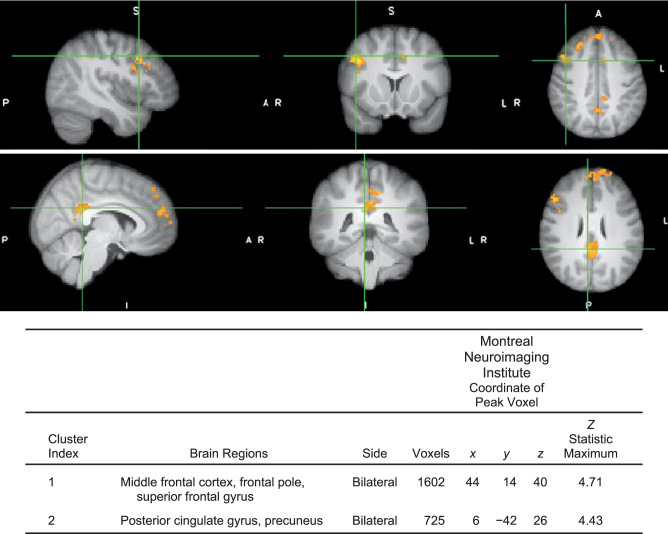
Athletes with high injury-risk biomechanics demonstrated less neural activity in 1 cluster for isolated knee movement (precuneus; peak z score = 4.14, P ≤ .01, 788 voxels; Figure 4) and greater brain activity for the multijoint leg press in 2 clusters: (1) frontal cortex (peak z score = 4.71, P < .01, 1602 voxels; Figure 5) and (2) posterior cingulate gyrus (peak z score = 4.43, P < .01, 725 voxels) compared with athletes in the low injury-risk group. No other differences were present between groups. Head motion during the isolated knee task was 0.26 ± 0.04 mm of absolute motion and 0.07 ± 0.02 mm of relative motion, and during the leg-press task, 0.38 ± 0.17 mm of absolute motion and 0.1 ± 0.03 mm of relative motion.
Figure 4.
Contrast image for isolated knee control between those with high versus low injury-risk knee-abduction moment during landing (P < .05, multiple voxel comparison cluster corrected, mixed-effects analysis z > 2.3). Anatomical regions within each cluster were defined using FSLeyes (version 5.0.10; Oxford Centre for Functional MRI of the Brain and Software Library's atlasquery function based on peak voxel coordinate and probability of anatomical region presence using the Harvard-Oxford cortical structural atlas in the Montreal Neuroimaging Institute space). Crosshair is over the peak voxel coordinate. Blue indicates less neural activity in the high relative to the low injury-risk group (no regions had greater activity in the high injury-risk group relative to the low injury-risk group for this task). Table provides cluster statistics and location data.
Figure 5.
Contrast image for multijoint control between those with high versus low injury-risk knee-abduction moment during landing (P < .05, multiple voxel comparison cluster corrected, mixed-effects analysis z > 2.3). Anatomical regions within each cluster, listed in the Table, were defined using FSLeyes (version 5.0.10; Oxford Centre for Functional MRI of the Brain and Software Library's (FSL's) atlasquery function based on peak voxel coordinate and probability of anatomical region presence using the Harvard-Oxford cortical structural atlas in the Montreal Neuroimaging Institute space. Crosshair is over the peak voxel coordinate. Orange indicates greater neural activity in the high relative to the low injury-risk group (no regions had less activity in the high injury-risk group relative to the low injury-risk group for this task).
DISCUSSION
This study provided insights into brain activity differences for isolated knee and lower extremity multijoint movement between young female soccer athletes with high and low injury-risk biomechanics. Participants with high-risk landings had less sensory-motor brain activity (precuneus) for isolated knee control and more cognitive-motor activity (frontal regions and posterior cingulate) for multijoint movement, supporting our hypothesis that high injury-risk biomechanics are related to differential brain-activation strategies. These preliminary data partially aligned with a previous finding5 of reduced sensory-related neural connectivity related to ACL injury, as both tasks revealed differences in sensory-related neural activity among those with high injury-risk neuromuscular control.
Isolated Knee Extension-Flexion Neural Activity Associated With High ACL Injury-Risk Neuromuscular Control
Participants with high injury-risk biomechanics, as stratified by pKAM, demonstrated less brain activity in the precuneus and surrounding regions during the isolated knee-movement task compared with the low injury-risk group. This isolated knee task required proprioceptive prediction, integration of feedback and spatial awareness with every movement to achieve full knee extension, and approximately 40° of flexion without touching the heel to the table in synchronization with the metronome timing. The precuneus plays a critical role in regulating proprioceptive feedback to refine the prediction of limb spatial location and contributes to critical anticipatory, sensory, and attention connections with the motor cortex to refine actions to external stimuli.18–20 Less relative brain activity in regions responsible for integrating sensorimotor information for isolated knee control may reduce the athlete's ability to maintain knee alignment during landing, contributing to a high pKAM.
Less synaptic activity in the precuneus and parietal cortex could hinder neuromuscular control under situations with intensive spatial-attentional demands or conflicting sensory stimuli, such as on the athletic field, thereby increasing the ACL injury risk.21 For instance, ACL-deficient individuals with poor subjective and functional outcomes had reduced parietal cortex activity versus control participants in an fMRI task similar to the isolated knee-control task.22 Furthermore, parietal cortex activity was inversely related to knee-flexion force match error.23 Thus, depressed sensory integration neural activity may play a role not only in the neuroplasticity associated with the injury but also in the neuromuscular control strategies implicated in primary injury risk.
Multijoint Leg-Press Neural Activity Associated With High ACL Injury-Risk Neuromuscular Control
Although participants with high injury-risk biomechanics had less sensory-motor activity during the isolated knee task, they showed greater cognitive-motor activity in the multijoint loaded leg-press task. Specific to motor tasks, increased activity can be caused by the increased neuronal firing rates that are required to generate higher muscle forces or movement velocity or manage movement complexity.24 Therefore, the added complexity of the multijoint coordination task relative to isolated knee movement may increase the demand on cognitive-motor resources, particularly in those with high injury-risk mechanics. We hypothesize that this effect might be associated with the multijoint task being constrained to sliding the leg in a track with the foot pedal, combined with the added resistance to alter the afferent feedback. The altered afferent feedback from the different task demands may have been the foundation for the increased frontal and posterior cingulate gyrus activity, whereas in the single-joint task, the high-risk group displayed decreased activity. The complexity of coordinating 3 joints as opposed to a single joint may result in a sufficient challenge to require the high-risk group to engage in neural compensation similar to the CRUNCH (Compensation-Related Utilization of Neural Circuits) hypothesis,25 whereby the demand to spread attention across 3 moving joints and associated elevated frontal activity, with high connectivity to the parietal cortex, results in a different sensory-attention neural-activation pattern.26
This neural strategy may sustain fundamental motor performance in isolation, but during sporting activity, increased cognitive demands due to external distractors or unpredictable environments may quickly exhaust the capacity for complex motor coordination and lead to a breakdown in neuromuscular control (and subsequent elevated pKAM). This theory is further supported by less efficient (increased) cognitive-motor integration activity being inversely correlated with cognitive ability and poorer cognitive abilities prospectively associated with an increased risk of injury and injury-risk neuromuscular control.27–29 The reverse has also been found: those with superior visual-spatial or motor capabilities tend to have decreased, or more efficient, sensory-cognitive neural activity.30–32 These studies suggest that cognitive-motor neural efficiency (ie, decreased activity in the frontal regions during basic cognition or motor control) may facilitate an athlete's ability to maintain low injury-risk coordination when challenged with combined cognitive-motor stimuli. Similarly, highly trained athletes displayed comparable brain-activation profiles (ie, reduced frontal activity) while executing motor tasks, indicating that training to control forces across the lower extremity may lead to more efficient neural recruitment and injury-resistant neuromuscular control.30 However, we caution that these speculations are reverse inferenced from the neural data, and future experiments manipulating the environment or adding distractors would be required to determine if this neural-activation pattern indeed results in an elevated breakdown in neuromuscular control.
Clinical Implications
Despite the breakthroughs in injury-prevention neuromuscular-training protocols that reduce biomechanical risk factors, ACL injuries continue to be prevalent.33 Our results preliminarily indicated that neural-activation patterns were related to ACL injury-risk biomechanics and could provide a pathway for clinicians and future researchers to consider new, innovative strategies that promote adaptive sensorimotor brain activity. Such neuroplasticity might be achieved by augmenting traditional approaches with targeted biofeedback,34 optimized motor learning techniques,35 dual-tasking methods, and sensory perturbations.36 An initial report34 specific to ACL injury-prevention training suggested that augmenting neuromuscular training with real-time biofeedback can transfer low injury-risk movement patterns to virtual sport and potentially address the neural activity associated with injury risk.
Limitations
We used a DVJ task to examine the neural correlates of KAM due to its prospective association with injury in young female athletes. Yet the potential limitations of using pKAM as an injury predictor37 have been noted, including the inadequacy of the DVJ for predicting the ACL injury risk. Nonetheless, that cohort of athletes was approximately 5 years older, had task and data-processing differences, and was more likely to have been exposed to injury-prevention programs than our participants. Because this was a pilot study with a small sample size, future investigation will be required to validate these preliminary neural markers of ACL injury-risk biomechanics and examine potential covariates (leg dominance, age, scanner motor performance, etc). Also, due to their increased injury risk, we evaluated only young female soccer players, limiting generalizability of the results to boys or men, older athletes, or athletes in different sports.
CONCLUSIONS
Young female soccer athletes with high injury-risk landing biomechanics engaged in a different sensory and cognitive neural-activation strategy relative to their low injury-risk peers for knee and multijoint lower extremity movement. Specifically, those with high injury-risk landing strategies may have reduced neural engagement for sensory regulation of isolated knee-joint control and elevated demands on cognitive-motor processing to control loaded multijoint action. The high injury-risk neural-activation profile for lower extremity movement may translate to a reduced capability to regulate knee position and maintain low injury-risk mechanics during landing. Future research is required to further evaluate the hypotheses generated from this work with larger sample sizes and more sophisticated assays of neuromuscular control and motor performance during neural recordings.
ACKNOWLEDGMENTS
For their support of and assistance with this study, we recognize the following people. From Seton High School (Cincinnati, OH), we thank Ron Quinn, Lisa Larosa, Holly Laiveling, and the entire soccer coaching staff as well as the Seton administration and athletic director Wendy Smith. From Madeira High School (Madeira, OH), we thank soccer head coach Dan Brady, athletic director Joe Kimling, and principal David Kennedy. We appreciate the soccer parents and players for participating in and enthusiastically supporting the project as well as their patience with the testing, scheduling, and follow-up testing. Special acknowledgment goes to athletic trainers Cindy Busse, ATC (Seton High School), and Glenna Knapp, ATC (Madeira High School). Without their time and commitment and their passion for the health and well-being of their student-athletes, this study would not have been possible.
Funding Statement
Dustin R. Grooms, PhD, ATC, CSCS, has current and ongoing funding support from the National Institutes of Health (NIH)/National Center for Complementary and Integrative Health (Award R21 AT009339-02), NIH/National Institute of Arthritis and Musculoskeletal and Skin Diseases (NIAMS; Awards R01AR076153, R01AR077248), and the US Department of Defense Congressionally Directed Medical Research Program, Peer Reviewed Orthopaedic Research Program research award (81XWH-18-1-0707). Gregory D. Myer, PhD, CSCS, has consulted with commercial entities to support applications to the US Food and Drug Administration but has no financial interest in the commercialization of the products. Dr Myer's institution receives current and ongoing grant funding from NIH/NIAMS Grants (Awards U01AR067997, R01AR070474, R01AR055563, R01AR076153, and R01AR077248) and industry-sponsored research funding related to brain injury prevention and assessment with Q30 Innovations, LLC, and ElMinda, Ltd. Dr Myer receives author royalties from Human Kinetics and Wolters Kluwer. He is an inventor of biofeedback technologies (2017 Nonprovisional patent pending, Augmented and Virtual Reality for Sport Performance and Injury Prevention Application filed 11/10/2016 [62/420,119], Software Copyrighted) designed to enhance rehabilitation and prevent injuries and has the potential for future licensing royalties (not applicable to this report). Industry funding and products are not directly related to this research (we did not include any data related to Q30 innovations or augmented or virtual reality products). This work was directly supported by the NIH and NIAMS (Awards R01AR076153 and R01AR077248). The funders played no role in study design, data collection, analysis, or decision to publish or in manuscript preparation.
Footnotes
Opinions, interpretations, conclusions, and recommendations are those of the author and are not necessarily endorsed by the US Department of Defense. There were no conflicts of interest pertaining to this study.
REFERENCES
- 1.Global recommendations on physical activity for health. World Health Organization; 2010. Accessed December 16 2019. http://www.ncbi.nlm.nih.gov/books/NBK305057/ [PubMed]
- 2.Luc B, Gribble PA, Pietrosimone BG. Osteoarthritis prevalence following anterior cruciate ligament reconstruction: a systematic review and numbers-needed-to-treat analysis. J Athl Train . 2014;49(6):806–819. doi: 10.4085/1062-6050-49.3.35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Mather RC, III, Koenig L, Kocher MS, et al. Societal and economic impact of anterior cruciate ligament tears. J Bone Joint Surg Am . 2013;95(19):1751–1759. doi: 10.2106/JBJS.L.01705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Swanik CB. Brains and sprains: the brain's role in noncontact anterior cruciate ligament injuries. J Athl Train . 2015;50(10):1100–1102. doi: 10.4085/1062-6050-50.10.08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Diekfuss JA, Grooms DR, Yuan W, et al. Does brain functional connectivity contribute to musculoskeletal injury? A preliminary prospective analysis of a neural biomarker of ACL injury risk. J Sci Med Sport . 2019;22(2):169–174. doi: 10.1016/j.jsams.2018.07.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Hewett TE, Myer GD, Ford KR, et al. Biomechanical measures of neuromuscular control and valgus loading of the knee predict anterior cruciate ligament injury risk in female athletes: a prospective study. Am J Sports Med . 2005;33(4):492–501. doi: 10.1177/0363546504269591. [DOI] [PubMed] [Google Scholar]
- 7.Bonnette S, Diekfuss JA, Grooms DR, et al. Electrocortical dynamics differentiate athletes exhibiting low- and high- ACL injury risk biomechanics. Psychophysiology . 2020;57(4):e13530. doi: 10.1111/psyp.13530. https://doi.org/10.1111/psyp.13530 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Powers CM, Fisher B. Mechanisms underlying ACL injury-prevention training: the brain-behavior relationship. J Athl Train . 2010;45(5):513–515. doi: 10.4085/1062-6050-45.5.513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Myer GD, Ford KR, Khoury J, Hewett TE. Three-dimensional motion analysis validation of a clinic-based nomogram designed to identify high ACL injury risk in female athletes. Phys Sportsmed . 2011;39(1):19–28. doi: 10.3810/psm.2011.02.1858. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Grooms DR, Diekfuss JA, Ellis JD, et al. A novel approach to evaluate brain activation for lower extremity motor control. J Neuroimaging . 2019;29(5):580–588. doi: 10.1111/jon.12645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Desikan RS, Ségonne F, Fischl B, et al. An automated labeling system for subdividing the human cerebral cortex on MRI scans into gyral based regions of interest. Neuroimage . 2006;31(3):968–980. doi: 10.1016/j.neuroimage.2006.01.021. [DOI] [PubMed] [Google Scholar]
- 12.Smith SM, Jenkinson M, Woolrich MW, et al. Advances in functional and structural MR image analysis and implementation as FSL. Neuroimage . 2004;23(suppl 1):S208–S219. doi: 10.1016/j.neuroimage.2004.07.051. [DOI] [PubMed] [Google Scholar]
- 13.Smith AJ, Blumenfeld H, Behar KL, Rothman DL, Shulman RG, Hyder F. Cerebral energetics and spiking frequency: the neurophysiological basis of fMRI. Proc Natl Acad Sci U S A . 2002;99(16):10765–10770. doi: 10.1073/pnas.132272199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Jenkinson M, Bannister P, Brady M, Smith S. Improved optimization for the robust and accurate linear registration and motion correction of brain images. Neuroimage . 2002;17(2):825–841. doi: 10.1016/s1053-8119(02)91132-8. [DOI] [PubMed] [Google Scholar]
- 15.Jenkinson M, Smith S. A global optimisation method for robust affine registration of brain images. Med Image Anal . 2001;5(2):143–156. doi: 10.1016/s1361-8415(01)00036-6. [DOI] [PubMed] [Google Scholar]
- 16.Woolrich MW, Ripley BD, Brady M, Smith SM. Temporal autocorrelation in univariate linear modeling of FMRI data. Neuroimage . 2001;14(6):1370–1386. doi: 10.1006/nimg.2001.0931. [DOI] [PubMed] [Google Scholar]
- 17.Worsley KJ. Jezzard P, Matthews PM, Smith SM, editors. Statistical analysis of activation images. Functional Magnetic Resonance Imaging An Introduction to Methods . Oxford University Press. 251–270. 2001.
- 18.Cavanna AE, Trimble MR. The precuneus: a review of its functional anatomy and behavioural correlates. Brain . 2006;129(3):564–583. doi: 10.1093/brain/awl004. [DOI] [PubMed] [Google Scholar]
- 19.Oshio R, Tanaka S, Sadato N, Sokabe M, Hanakawa T, Honda M. Differential effect of double-pulse TMS applied to dorsal premotor cortex and precuneus during internal operation of visuospatial information. Neuroimage . 2010;49(1):1108–1115. doi: 10.1016/j.neuroimage.2009.07.034. [DOI] [PubMed] [Google Scholar]
- 20.Wenderoth N, Debaere F, Sunaert S, Swinnen SP. The role of anterior cingulate cortex and precuneus in the coordination of motor behaviour. Eur J Neurosci . 2005;22(1):235–246. doi: 10.1111/j.1460-9568.2005.04176.x. [DOI] [PubMed] [Google Scholar]
- 21.Grooms DR, Onate JA. Neuroscience application to noncontact anterior cruciate ligament injury prevention. Sports Health . 2016;8(2):149–152. doi: 10.1177/1941738115619164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kapreli E, Athanasopoulos S, Gliatis J, et al. Anterior cruciate ligament deficiency causes brain plasticity: a functional MRI study. Am J Sports Med . 2009;37(12):2419–2426. doi: 10.1177/0363546509343201. [DOI] [PubMed] [Google Scholar]
- 23.Grooms DR, Criss CR, Simon JE, Haggerty AL, Wohl TR. Neural correlates of knee extension and flexion force control: a kinetically-instrumented neuroimaging study. Front Hum Neurosci . 2020;14:622637. doi: 10.3389/fnhum.2020.622637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Mehta JP, Verber MD, Wieser JA, Schmit BD, Schindler-Ivens SM. The effect of movement rate and complexity on functional magnetic resonance signal change during pedaling. Motor Control . 2012;16(2):158–175. doi: 10.1123/mcj.16.2.158. [DOI] [PubMed] [Google Scholar]
- 25.Reuter-Lorenz PA, Cappell KA. Neurocognitive aging and the compensation hypothesis. Curr Dir Psychol Sci . 2008;17(3):177–182. doi: 10.1111/j.1467-8721.2008.00570.x. [DOI] [Google Scholar]
- 26.Anand M, Diekfuss JA, Slutsky-Ganesh AB, et al. Integrated 3D motion analysis with functional magnetic resonance neuroimaging to identify neural correlates of lower extremity movement. J Neurosci Methods . 2021;355:109108. doi: 10.1016/j.jneumeth.2021.109108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Herman DC, Barth JT. Drop-jump landing varies with baseline neurocognition: implications for anterior cruciate ligament injury risk and prevention. Am J Sports Med . 2016;44(9):2347–2353. doi: 10.1177/0363546516657338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Swanik CB, Covassin T, Stearne DJ, Schatz P. The relationship between neurocognitive function and noncontact anterior cruciate ligament injuries. Am J Sports Med . 2007;35(6):943–948. doi: 10.1177/0363546507299532. [DOI] [PubMed] [Google Scholar]
- 29.Dunst B, Benedek M, Jauk E, et al. Neural efficiency as a function of task demands. Intelligence . 2014;42(100):22–30. doi: 10.1016/j.intell.2013.09.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Del Percio C, Babiloni C, Marzano N, et al. “Neural efficiency” of athletes' brain for upright standing: a high-resolution EEG study. Brain Res Bull . 2009;79(3–4):193–200. doi: 10.1016/j.brainresbull.2009.02.001. [DOI] [PubMed] [Google Scholar]
- 31.Costanzo ME, VanMeter JW, Janelle CM, et al. Neural efficiency in expert cognitive-motor performers during affective challenge. J Mot Behav . 2016;48(6):573–588. doi: 10.1080/00222895.2016.1161591. [DOI] [PubMed] [Google Scholar]
- 32.Di Domenico SI, Rodrigo AH, Ayaz H, Fournier MA, Ruocco AC. Decision-making conflict and the neural efficiency hypothesis of intelligence: a functional near-infrared spectroscopy investigation. Neuroimage . 2015;109:307–317. doi: 10.1016/j.neuroimage.2015.01.039. [DOI] [PubMed] [Google Scholar]
- 33.Beck NA, Lawrence JTR, Nordin JD, DeFor TA, Tompkins M. ACL tears in school-aged children and adolescents over 20 years. Pediatrics . 2017;139(3):e20161877. doi: 10.1542/peds.2016-1877. [DOI] [PubMed] [Google Scholar]
- 34.Grooms DR, Kiefer AW, Riley MA, et al. Brain-behavior mechanisms for the transfer of neuromuscular training adaptions to simulated sport: initial findings from the Train the Brain Project. J Sport Rehabil . 2018;27(5):1–5. doi: 10.1123/jsr.2017-0241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Diekfuss JA, Grooms DR, Hogg JA, et al. Targeted application of motor learning theory to leverage youth neuroplasticity for enhanced injury-resistance and exercise performance: OPTIMAL PREP. J Sci Sport Exerc . 2021;3(1):17–36. doi: 10.1007/s42978-020-00085-y. [DOI] [Google Scholar]
- 36.Chmielewski TL, Hurd WJ, Rudolph KS, Axe MJ, Snyder-Mackler L. Perturbation training improves knee kinematics and reduces muscle co-contraction after complete unilateral anterior cruciate ligament rupture. Phys Ther . 2005;85(8):740–749. doi: 10.1093/ptj/85.8.740. [DOI] [PubMed] [Google Scholar]
- 37.Krosshaug T, Steffen K, Kristianslund E, et al. The vertical drop jump is a poor screening test for ACL injuries in female elite soccer and handball players: a prospective cohort study of 710 athletes. Am J Sports Med . 2016;44(4):874–883. doi: 10.1177/0363546515625048. [DOI] [PubMed] [Google Scholar]