Abstract
Background
Multisystem Inflammatory Syndrome in Children (MIS-C), which occurs 2–6 weeks after initial exposure to SARS-CoV-2, was first identified in early 2020 when patients presented with fever and significant inflammation, often requiring management in the intensive care unit. To date, there has been no clinical trial to determine the most effective treatment. This study compares anti-inflammatory treatments that were selected based on current treatments for Kawasaki disease, a coronary artery vasculitis that shares many clinical features with MIS-C.
Methods
This randomized, comparative effectiveness trial of children with MIS-C uses the small N Sequential Multiple Assignment Randomized Trial (snSMART) design for rare diseases to compare multiple therapies within an individual. Study participants were treated first with intravenous immunoglobulin (IVIG), and if needed, subjects were then randomized to one of three additional treatments (steroids, anakinra, or infliximab). Participants were re-randomized to remaining treatments if they did not demonstrate clinical improvement.
Conclusion
This trial continues to enroll eligible participants to determine the most effective therapies in addition to IVIG and best order in which to use them to treat MIS-C.
Trial Registration
Keywords: Multisystem inflammatory syndrome in children (MIS-C), Infliximab, Anakinra, Steroids, snSMART, Randomized clinical trial
Abbreviations: IVIG, Intravenous immunoglobulin; snSMART, small N Sequential Multiple Assignment Randomized Trial
1. Introduction
Multisystem Inflammatory Syndrome in Children (MIS-C), a post-inflammatory syndrome in children following infection with SARS-CoV-2, was first described in April 2020, at the onset of COVID-19 pandemic [1]. The clinical presentation of MIS-C shares many features with Kawasaki disease (KD), a coronary artery vasculitis that presents with fever, rash, conjunctival injection, mucocutaneous erythema, and cervical lymphadenopathy. Given these clinical similarities, physicians treated the majority of MIS-C patients with intravenous immunoglobulin (IVIG), the standard of care for KD patients [2,3]. Some MIS-C patients did not improve with IVIG alone and were treated with steroids, anakinra (IL-1 blockade), or infliximab (TNFα blockade), all anti-inflammatory therapies that have been successfully used in the treatment of children with KD [[4], [5], [6], [7], [8], [9]]. There has been no randomized clinical trial to determine the effectiveness of these therapies. Therefore, we designed a randomized, comparative effectiveness trial to determine the best combination of anti-inflammatory therapies and the order in which they should be given in the treatment of MIS-C.
2. Rationale for therapeutic choices in this study
There are several lines of evidence that support the use of IVIG, steroids, anakinra and infliximab in KD, and thus these were used as the rationale for using these same therapies to treat MIS-C patients. Over the last two years of the pandemic, observational studies have demonstrated the effectiveness of one or more of these therapies, thus providing further support for their use in the clinical care of these patients [[10], [11], [12], [13]].
In KD, IVIG boosts the host's anti-inflammatory response by stimulating tolerogenic myeloid dendritic cells to secrete the immunosuppressive cytokine IL-10, which inhibits pro-inflammatory cytokine release from most immune cells [14]. Recently, Zhu et al. have shown that IVIG is a potent activator of neutrophil cell death in KD and MIS-C patients via PI3K and NADPH oxidase [15]. The observation that MIS-C is mediated by a robust innate immune response with activated neutrophils secreting IL-1b provided a strong rationale for use of IVIG as first-line therapy.
Steroids have been extensively used as an anti-inflammatory therapy in several vasculitides in childhood and are used in conjunction with IVIG by many centers for KD patients with coronary artery abnormalities (CAA) or persistent fever [4]. Coronary artery dilation can be a feature of MIS-C and thus it was logical to choose steroids as a therapy given that steroids in combination with IVIG have been shown to significantly reduce the incidence of CAA in KD patients when compared to treatment with IVIG alone [4].
Anakinra, a recombinant IL-1 receptor antagonist, competitively inhibits IL-1 binding to the IL-1 type 1 receptor. Genes in the IL-1 pathway are upregulated in the peripheral blood of KD patients during the acute phase of illness [16]. Anakinra blocks arterial inflammation in a murine model of KD [17]. A Phase I/IIa study of anakinra in KD patients with CAA demonstrated the safety and sustained serum concentration of IV anakinra in patients treated with doses up to 11 mg/kg/day [18]. An additional clinical trial of anakinra for persistent fever in KD patients also demonstrated the safety and tolerability of this biologic [6]. Given the familiarity with IL-1 blockade in KD and the demonstration that IL-1b played a major role in the inflammation in MIS-C, anakinra was used in the treatment of MIS-C patients early in the pandemic [11].
Infliximab is a monoclonal antibody that binds to and reduces levels of circulating tumor necrosis factor alpha (TNFα). Given that TNFα is increased in acute KD and highest in children with coronary artery aneurysms, infliximab has been used to treat KD [19,20]. A Phase III randomized trial of infliximab in children with IVIG-resistant KD demonstrated the safety of infliximab. Infliximab reduced inflammation more rapidly than a second dose of IVIG [5]. A recent randomized, multicenter, comparative effectiveness trial of infliximab vs. 2nd IVIG in IVIG-resistant KD in the US (KIDCARE) further demonstrated the safety and efficacy of infliximab and revealed the increased risk of hemolytic anemia with increasing doses of IVIG [21]. Infliximab was recently approved for the treatment of refractory KD in Japan and a multicenter, prospective, open-label study in Japan further demonstrated the safety of infliximab in KD patients [8]. Thus, due to the use of infliximab in refractory KD patients, physicians started treating complicated MIS-C patients with infliximab [12].
Given the similarities between KD and MIS-C, therapies that were safe and effective for treatment of KD were also used for children with MIS-C. In the absence of clinical trial data for this new condition, centers around the globe reached for some combination of these anti-inflammatory to treat MIS-C patients [22]. However, a clinical trial was urgently needed to evaluate which anti-inflammatory regimen was most effective. Thus, this trial was designed and launched to fill this knowledge gap.
3. Objectives
The main goal of this study is to determine the anti-inflammatory therapy from first randomization with the lowest rate of second randomization in the treatment of MIS-C. This study will also evaluate the best order in which the therapies should be given to achieve the greatest therapeutic effect.
4. Study design
This study is a randomized comparative effectiveness trial of children with MIS-C using the patient-centered “small N Sequential Multiple Assignment Randomized Trial (snSMART) designed for rare disease trials as it efficiently evaluates multiple treatments within an individual [[23], [24], [25]].
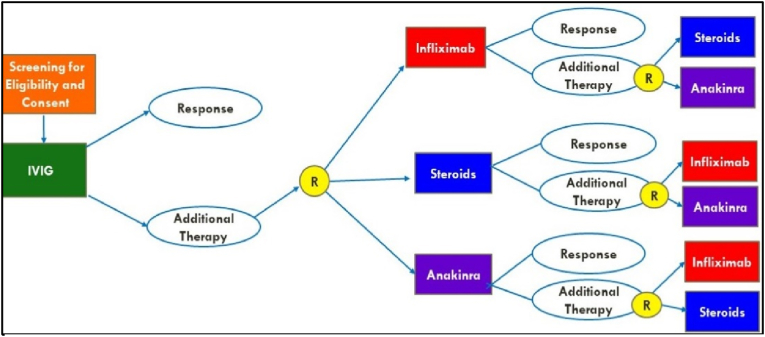
All subjects will be treated with IVIG at the standard dose of 2 g/kg over 12 h (max 100g; for obese patients, dose based on ideal body weight). Subjects will subsequently be randomized to receive additional anti-inflammatory therapy of steroids (2 mg/kg/day methylprednisolone IV divided q12 h; max 60 mg/day), anakinra (up to 10 mg/kg/day IV with max 200 mg q6h) or infliximab (10 mg/kg IV once over 2 h) if needed. This study allows for re-randomization to one of the two remaining arms if clinically warranted (See Fig. 1)
Fig. 1.
snSMART study overview for MIS-C patients. Study drugs administered as follows: Infliximab: 10 mg/kg IV dose once; Steroids: 2 mg/kg IV divided BID (max 60 mg) with steroid taper for 2–3 weeks post-discharge; Anakinra: Up to 10 mg/kg/day (max 200 mg every 6 h) with taper and discontinuation prior to discharge. Color to be used. (For interpretation of the references to color in this figure legend, the reader is referred to the Web version of this article.)
Randomization to additional therapy is at the discretion of the treating physician (but not required) for patients who meet one or more of the criteria listed in Table 1, including fever after completion of IVIG, persistent or worsening inflammation, and/or persistent or worsening cardiac dysfunction. The schedule of all tests and laboratory studies is shown in Table 2.
Table 1.
Criteria for randomization to additional therapy after completion of IVIG infusion.
|
Table 2.
Schedule of study data and sample collection The schedule of all tests and laboratory studies is depicted in the following table.
| Time point |
Baseline (prior to any therapy) |
12 h after IVIG completed (prior to additional tx) |
12 h after first randomized therapy completed (prior to add'l therapy) |
12 h after second randomized therapy (or just prior to discharge if no further randomization) |
2 weeks after discharge |
|---|---|---|---|---|---|
| Visit Window | +1 day | +1 day | +1 day | ±10 days | |
| Informed Consent/Assent | X | ||||
| Inclusion/Exclusion Criteria | X | ||||
| Weight and Height Measurements | X | X | |||
| Medical history | X | ||||
| Demographics | X | ||||
| Concomitant Meds | X | X | X | X | X |
| Physical Exam | X | X | X | X | X |
| Urine Pregnancy Testa | X | ||||
| Clinical Laboratory tests (e.g. CBC, CRP)b | X | X | X | X | X |
| Research Study Samplesc | X | X | X | X | X |
| Echocardiogramb | X | X | X | X | X |
| Adverse events assessment | X | X | X | X | X |
For females of child-bearing potential.
Most closely associated with study lab draw if available.
Over a 30-day period, the total volume of research samples will not exceed 2 ml/kg. Research samples volumes will be adjusted as needed depending on patient's weight. Blood samples are limited to 8.5 ml of blood distributed between red top tubes for serum, purple top tubes for plasma, and PAX-gene tubes for whole blood RNA.
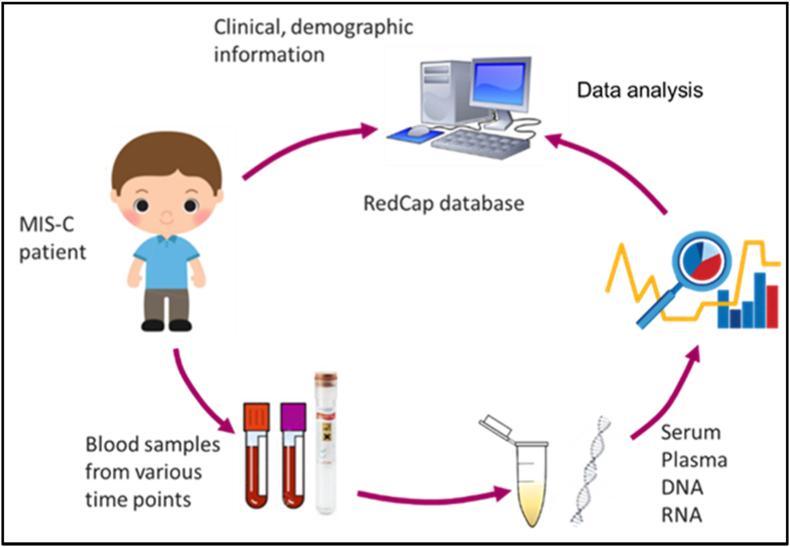
Clinical, laboratory, and echocardiographic data will be obtained as part of the routine care of the patient (e.g. hematology and chemistry results, medications) and stored in a HIPPAA-secure REDCap Database. Plasma, serum, whole blood RNA and DNA samples will be collected at various time points for future analysis (See Fig. 2).
Fig. 2.
Sample collection processColor to be used. (For interpretation of the references to color in this figure legend, the reader is referred to the Web version of this article.)
Echocardiography will include measurements of (1) the coronary arteries: internal lumen of the proximal right and proximal left anterior descending coronary arteries, normalized to body surface area and expressed as z-scores and American Heart Association classification schema for coronary artery aneurysms and (2) ventricular function, valves and pericardial effusion, including quantitative assessment of left ventricular (LV) size and systolic function (LV end-diastolic volume, ejection fraction, and longitudinal strain), presence and degree of mitral and aortic regurgitation by standard color flow mapping and pulsed Doppler techniques, and parameters of LV diastolic function with tissue Doppler imaging and MV inflow. All quantitative measures will be measured on three consecutive beats and values will be averaged.
5. Inclusion and exclusion criteria
Patients will be eligible to participate if they meet the CDC criteria for MIS-C. This includes fever, at least two body systems involved, predominantly cardiovascular, mucocutaneous, or gastrointestinal, and have no other leading diagnosis. In addition, proof of exposure to SARS-CoV-2, usually by positive IgG to SARS-CoV-2 nucleocapsid, will be needed. Parent or legal guardian must be able and willing to provide informed consent and subject willing and able to provide assent when appropriate.
Patients will be excluded if they have an immunodeficiency, a medical condition that prevents them from receiving one or more of the study drugs, or have received any of the study drugs prior to enrollment.
6. Statistical methods
6.1. Statistical analysis plan
Since this is a superiority, snSMART study design, an intent-to-treat (ITT) approach will be used to analyze the data regarding patient outcomes. A per protocol analysis of patients completing the protocol will be performed as a sensitivity analysis analogous to the ITT analyses. Results will be reported as point estimates (odds ratios or mean differences across groups, as appropriate) and interval estimates (95% confidence intervals). All tests of significance for the secondary outcomes will be 2-sided and Hochberg adjustments will be made for multiple comparisons. A p-value of 0.05 will be considered statistically significant unless otherwise specified. Statistical analyses will be conducted using the statistical software R 3.6.3 (www.rproject.org). and rjags. Demographic and baseline characteristics will be compared among the study arms using Fisher's exact test for categorical variables, and ANOVA/pairwise two-sample t-tests for continuous variables. Appropriate non-parametric alternatives will be considered if parametric assumptions fail.
6.2. Sample size and power
We project enrollment of 180 children with MIS-C. We anticipate that 50 (27%) children will improve with IVIG alone, while 130 (73%) will require additional treatment post-IVIG resulting in randomization across the three interventions over the study period. With a worst-case attrition rate of 17%, we will achieve 108 evaluable subjects for the snSMART design. All sample size calculations were performed using the snSMART Sample Size App [https://umich-biostatistics.shinyapps.io/snsmart _sample_ size_app/]. This two-stage design uses a Bayesian joint stage model for estimating the response rates of each individual treatment in a three-arm snSMART design. The approach distinguishes the best treatment from the second-best treatment using the Bayesian joint stage model. We require approximately 36 subjects per arm (108 in total for three agents) at the initial randomization. The probability of successfully identifying the best treatment is 0.85, when the difference of response rates between the best and second-best treatment is at least 0.25 and the response rate of the best treatment is 0.75. We conducted sensitivity analyses for the power using the joint-stage regression model (JSRM) method of Chao et al. (2020) [25] that used Dunnett's approach under generalized estimating equations (GEE) and obtained similar results for approximately 85% power and alpha = 0.05. Therefore, an anticipated sample size of approximately 108 evaluable subjects should provide reasonable evidence for a treatment effect in this rare disease setting. Note that if attrition is slightly worse at 25%, then we would require an overall N of 144, in which case we would still be well-powered with this sample size.
6.3. Analysis of the primary outcome for aim 1
Comparisons between the three treatment arms with respect to the primary endpoint of best clinical response rate will be conducted using a Fisher's exact test for proportions. Differences in the rates between the multiple arms, along with the odds ratio (OR) and their 95% confidence intervals will be reported. A Bayesian Joint Stage Model will be considered, which uses all data from both randomized and re-randomized subjects in estimation and inference, using “linkage parameters” to connect the data from each randomization. This approach provides an unbiased and efficient estimation of the treatment effects and dynamic treatment regimens. Also, as a sensitivity analysis, a multivariable joint-stage GEE regression model will be performed to study the association between clinical, biomarker and demographic factors and intervention arm, adjusting for baseline demographic, stratification factors, and clinical characteristics. Variables significantly associated with both treatment group and outcome (p < 0.10) will be included in a multivariable model as covariates.
6.4. Comparison to IVIG alone
The baseline characteristics and outcomes of subjects treated only with IVIG will be compared to those randomized to receive infliximab, anakinra or steroids.
7. Conclusion
This study continues to enroll eligible patients to determine the most effective treatment regimen in addition to IVIG and the best order of therapies for MIS-C. Recently, this study has expanded from its initial enrollment center at Rady Children's Hospital San Diego, to also include enrollment at Children's Hospital Michigan. Additional sites will be considered if MIS-C persists in sufficient numbers.
Author contributions
Sonia Jain: Methodology, Formal Analysis, Writing-original draft preparation.
Feng He: Formal Analysis.
Kiana Brown: Data curation, Writing-original draft preparation, review and editing.
Jane C. Burns: Conception, Writing-review and editing, Supervision.
Adriana H. Tremoulet: Conception, Writing-review and editing, Supervision.
Declaration of competing interests
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgements
We thank Joan Pancheri RN, BSN for research assistance and Emelia Bainto, Samantha Roberts and Nipha Sivilay for data management. We also thank our study participants and their parents/guardians.
Footnotes
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Contributor Information
Sonia Jain, Email: sojain@health.ucsd.edu.
Feng He, Email: fhe@health.ucsd.edu.
Kiana Brown, Email: K2brown@health.ucsd.edu.
Jane C. Burns, Email: jcburns@health.ucsd.edu.
Adriana H. Tremoulet, Email: atremoulet@health.ucsd.edu.
References
- 1.Control, Centers for Disease Control and Prevention . 2020. Multisystem Inflammatory Syndrome in Children (MIS-C) Associated with Coronavirus Disease 2019 (COVID-19) 2020 May 14. [last accessed Sept 2022. [Google Scholar]
- 2.Elias M.D., McCrindle B.W., Larios G., Choueiter N.F., Dahdah N., Harahsheh A.S., Jain S., Manlhiot C., Portman M.A., Raghuveer G., Giglia T.M., Dionne A., of the International Kawasaki Disease Registry Management of multisystem inflammatory syndrome in children associated with COVID-19: a survey from the international Kawasaki disease registry. CJC Open. 2020 Nov;2(6):632–640. doi: 10.1016/j.cjco.2020.09.004. Epub 2020 Sep 11. PMID: 32935083; PMCID: PMC7484693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Feldstein L.R., Rose E.B., Horwitz S.M., Collins J.P., Newhams M.M., Son M.B.F., Newburger J.W., Kleinman L.C., Heidemann S.M., Martin A.A., Singh A.R., Li S., Tarquinio K.M., Jaggi P., Oster M.E., Zackai S.P., Gillen J., Ratner A.J., Walsh R.F., Fitzgerald J.C., Keenaghan M.A., Alharash H., Doymaz S., Clouser K.N., Giuliano J.S., Jr., Gupta A., Parker R.M., Maddux A.B., Havalad V., Ramsingh S., Bukulmez H., Bradford T.T., Smith L.S., Tenforde M.W., Carroll C.L., Riggs B.J., Gertz S.J., Daube A., Lansell A., Coronado Munoz A., Hobbs C.V., Marohn K.L., Halasa N.B., Patel M.M., Randolph A.G. Overcoming COVID-19 investigators; CDC COVID-19 response team. Multisystem inflammatory syndrome in U.S. Children and adolescents. N. Engl. J. Med. 2020 Jul 23;383(4):334–346. doi: 10.1056/NEJMoa2021680. Epub 2020 Jun 29. PMID: 32598831; PMCID: PMC7346765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Kobayashi T., Saji T., Otani T., Takeuchi K., Nakamura T., Arakawa H., Kato T., Hara T., Hamaoka K., Ogawa S., Miura M., Nomura Y., Fuse S., Ichida F., Seki M., Fukazawa R., Ogawa C., Furuno K., Tokunaga H., Takatsuki S., Hara S., Morikawa A. RAISE study group investigators. Efficacy of immunoglobulin plus prednisolone for prevention of coronary artery abnormalities in severe Kawasaki disease (RAISE study): a randomised, open-label, blinded-endpoints trial. Lancet. 2012 Apr 28;379(9826):1613–1620. doi: 10.1016/S0140-6736(11)61930-2. Epub 2012 Mar 8. PMID: 22405251. [DOI] [PubMed] [Google Scholar]
- 5.Tremoulet A.H., Jain S., Jaggi P., Jimenez-Fernandez S., Pancheri J.M., Sun X., Kanegaye J.T., Kovalchin J.P., Printz B.F., Ramilo O., Burns J.C. Infliximab for intensification of primary therapy for Kawasaki disease: a phase 3 randomised, double-blind, placebo-controlled trial. Lancet. 2014 May 17;383(9930):1731–1738. doi: 10.1016/S0140-6736(13)62298-9. Epub 2014 Feb 24. PMID: 24572997. [DOI] [PubMed] [Google Scholar]
- 6.Koné-Paut I., Tellier S., Belot A., Brochard K., Guitton C., Marie I., Meinzer U., Cherqaoui B., Galeotti C., Boukhedouni N., Agostini H., Arditi M., Lambert V., Piedvache C. Phase II open label study of anakinra in intravenous immunoglobulin-resistant Kawasaki disease. Arthritis Rheumatol. 2021 Jan;73(1):151–161. doi: 10.1002/art.41481. Epub 2020 Nov 17. PMID: 32779863. [DOI] [PubMed] [Google Scholar]
- 7.Yang J., Jain S., Capparelli E.V., Best B.M., Son M.B., Baker A., Newburger J.W., Franco A., Printz B.F., He F., Shimizu C., Hoshino S., Bainto E., Moreno E., Pancheri J., Burns J.C., Tremoulet A.H. Anakinra treatment in patients with acute Kawasaki disease with coronary artery aneurysms: a phase I/IIa trial. J. Pediatr. 2022 Apr;243:173–180.e8. doi: 10.1016/j.jpeds.2021.12.035. Epub 2021 Dec 23. PMID: 34953816. [DOI] [PubMed] [Google Scholar]
- 8.Miura M., Kobayashi T., Igarashi T., Hamada H., Iwata N., Sasaki Y., Matsukawa M., Sato N., Kubo H., Takei S. Real-world safety and effectiveness of infliximab in pediatric patients with acute Kawasaki disease: a postmarketing surveillance in Japan (sakura study) Pediatr. Infect. Dis. J. 2020 Jan;39(1):41–47. doi: 10.1097/INF.0000000000002503. PMID: 31815838. [DOI] [PubMed] [Google Scholar]
- 9.Dionne A., Burns J.C., Dahdah N., Tremoulet A.H., Gauvreau K., de Ferranti S.D., Baker A.L., Son M.B., Gould P., Fournier A., Newburger J.W., Friedman K.G. Treatment intensification in patients with Kawasaki disease and coronary aneurysm at diagnosis. Pediatrics. 2019 Jun;143(6) doi: 10.1542/peds.2018-3341. Epub 2019 May 2. PMID: 31048414. [DOI] [PubMed] [Google Scholar]
- 10.Kaushik S., Aydin S.I., Derespina K.R., Bansal P.B., Kowalsky S., Trachtman R., Gillen J.K., Perez M.M., Soshnick S.H., Conway E.E., Jr., Bercow A., Seiden H.S., Pass R.H., Ushay H.M., Ofori-Amanfo G., Medar S.S. Multisystem inflammatory syndrome in children associated with severe acute respiratory syndrome coronavirus 2 infection (mis-C): a multi-institutional study from New York city. J. Pediatr. 2020 Sep;224:24–29. doi: 10.1016/j.jpeds.2020.06.045. Epub 2020 Jun 14. PMID: 32553861; PMCID: PMC7293760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.DeBiasi R.L., Harahsheh A.S., Srinivasalu H., Krishnan A., Sharron M.P., Parikh K., Smith K., Bell M., Michael D., Delaney M., Campos J., Vilain E., LoTempio J., Kline J.N., Ronis T., Majumdar S., Sadler E., Conway S.R., Berul C.I., Sule S., Lahoz R., Ansusinha E., Pershad J., Bundy V., Wells E., Bost J.E., Wessel D. Children's national hospital MIS-C taskforce. Multisystem inflammatory syndrome of children: subphenotypes, risk factors, biomarkers, cytokine profiles, and viral sequencing. J. Pediatr. 2021 Oct;237:125–135.e18. doi: 10.1016/j.jpeds.2021.06.002. Epub 2021 Jun 25. PMID: 34181987. [DOI] [PubMed] [Google Scholar]
- 12.Abdel-Haq N., Asmar B.I., Deza Leon M.P., McGrath E.J., Arora H.S., Cashen K., Tilford B., Charaf Eddine A., Sethuraman U., Ang J.Y. SARS-CoV-2-associated multisystem inflammatory syndrome in children: clinical manifestations and the role of infliximab treatment. Eur. J. Pediatr. 2021 May;180(5):1581–1591. doi: 10.1007/s00431-021-03935-1. Epub 2021 Jan 16. PMID: 33452570; PMCID: PMC7810600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.McArdle A.J., Vito O., Patel H., Seaby E.G., Shah P., Wilson C., Broderick C., Nijman R., Tremoulet A.H., Munblit D., Ulloa-Gutierrez R., Carter M.J., De T., Hoggart C., Whittaker E., Herberg J.A., Kaforou M., Cunnington A.J., Levin M., Bats Consortium. Treatment of multisystem inflammatory syndrome in children. N. Engl. J. Med. 2021 Jul 1;385(1):11–22. doi: 10.1056/NEJMoa2102968. Epub 2021 Jun 16. PMID: 34133854; PMCID: PMC8220965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Franco A., Kumar J., Lin G., Behnamfar N., Hsieh L.E., Shimizu C., Tremoulet A.H., Burns J.C., Linden J. Pediatric tolerogenic DCs expressing CD4 and immunoglobulin-like transcript receptor (ILT)-4 secrete IL-10 in response to Fc and adenosine. Eur. J. Immunol. 2018 Mar;48(3):482–491. doi: 10.1002/eji.201747139. Epub 2018 Jan 15. PMID: 29244203. [DOI] [PubMed] [Google Scholar]
- 15.Zhu Y.P., Shamie I., Lee J.C., Nowell C.J., Peng W., Angulo S., Le L.N., Liu Y., Miao H., Xiong H., Pena C.J., Moreno E., Griffis E., Labou S.G., Franco A., Broderick L., Hoffman H.M., Shimizu C., Lewis N.E., Kanegaye J.T., Tremoulet A.H., Burns J.C., Croker B.A. Pediatric emergency medicine Kawasaki disease research group consortium. Immune response to intravenous immunoglobulin in patients with Kawasaki disease and MIS-C. J. Clin. Invest. 2021 Oct 15;131(20) doi: 10.1172/JCI147076. PMID: 34464357; PMCID: PMC8516453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Hoang L.T., Shimizu C., Ling L., Naim A.N., Khor C.C., Tremoulet A.H., Wright V., Levin M., Hibberd M.L., Burns J.C. Global gene expression profiling identifies new therapeutic targets in acute Kawasaki disease. Genome Med. 2014 Nov 20;6(11):541. doi: 10.1186/s13073-014-0102-6. PMID: 25614765; PMCID: PMC4279699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Lee Y., Schulte D.J., Shimada K., Chen S., Crother T.R., Chiba N., Fishbein M.C., Lehman T.J., Arditi M. Interleukin-1β is crucial for the induction of coronary artery inflammation in a mouse model of Kawasaki disease. Circulation. 2012 Mar 27;125(12):1542–1550. doi: 10.1161/CIRCULATIONAHA.111.072769. Epub 2012 Feb 23. PMID: 22361326; PMCID: PMC3337219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Tremoulet A.H., Jain S., Kim S., Newburger J., Arditi M., Franco A., Best B., Burns J.C. Rationale and study design for a phase I/IIa trial of anakinra in children with Kawasaki disease and early coronary artery abnormalities (the ANAKID trial) Contemp. Clin. Trials. 2016 May;48:70–75. doi: 10.1016/j.cct.2016.04.002. Epub 2016 Apr 11. PMID: 27080929; PMCID: PMC5221485. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Furukawa S., Matsubara T., Jujoh K., Yone K., Sugawara T., Sasai K., Kato H., Yabuta K. Peripheral blood monocyte/macrophages and serum tumor necrosis factor in Kawasaki disease. Clin. Immunol. Immunopathol. 1988 Aug;48(2):247–251. doi: 10.1016/0090-1229(88)90088-8. PMID: 3390972. [DOI] [PubMed] [Google Scholar]
- 20.Matsubara T. [Serum gamma interferon levels in relation to tumor necrosis factor and interleukin 2 receptor in patients with Kawasaki disease involving coronary-artery lesions] Arerugi. 1990 Feb;39(2 Pt 1):118–123. Japanese. PMID: 2112909. [PubMed] [Google Scholar]
- 21.Burns J.C., Roberts S.C., Tremoulet A.H., He F., Printz B.F., Ashouri N., Jain S.S., Michalik D.E., Sharma K., Truong D.T., Wood J.B., Kim K.K., Jain S., KIDCARE Multicenter Study Group Infliximab versus second intravenous immunoglobulin for treatment of resistant Kawasaki disease in the USA (KIDCARE): a randomised, multicentre comparative effectiveness trial. Lancet Child Adolesc Health. 2021 Dec;5(12):852–861. doi: 10.1016/S2352-4642(21)00270-4. Epub 2021 Oct 27. Erratum in: Lancet Child Adolesc Health. 2022 Feb;6(2):e5. PMID: 34715057. [DOI] [PubMed] [Google Scholar]
- 22.Dove M.L., Jaggi P., Kelleman M., Abuali M., Ang J.Y., Ballan W., Basu S.K., Campbell M.J., Chikkabyrappa S.M., Choueiter N.F., Clouser K.N., Corwin D., Edwards A., Gertz S.J., Ghassemzadeh R., Jarrah R.J., Katz S.E., Knutson S.M., Kuebler J.D., Lighter J., Mikesell C., Mongkolrattanothai K., Morton T., Nakra N.A., Olivero R., Osborne C.M., Panesar L.E., Parsons S., Patel R.M., Schuette J., Thacker D., Tremoulet A.H., Vidwan N.K., Oster M.E. Multisystem inflammatory syndrome in children: survey of protocols for early hospital evaluation and management. J. Pediatr. 2021 Feb;229:33–40. doi: 10.1016/j.jpeds.2020.10.026. Epub 2020 Oct 16. PMID: 33075369; PMCID: PMC7566788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Tamura R.N., Krischer J.P., Pagnoux C., Micheletti R., Grayson P.C., Chen Y.F., Merkel P.A. A small n sequential multiple assignment randomized trial design for use in rare disease research. Contemp. Clin. Trials. 2016 Jan;46:48–51. doi: 10.1016/j.cct.2015.11.010. Epub 2015 Nov 14. PMID: 26586608; PMCID: PMC4695231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Wei B., Braun T.M., Tamura R.N., Kidwell K.M. A Bayesian analysis of small n sequential multiple assignment randomized trials (snSMARTs) Stat. Med. 2018 Nov 20;37(26):3723–3732. doi: 10.1002/sim.7900. Epub 2018 Jul 16. PMID: 30010207. [DOI] [PubMed] [Google Scholar]
- 25.Chao Y.C., Trachtman H., Gipson D.S., Spino C., Braun T.M., Kidwell K.M. Dynamic treatment regimens in small n, sequential, multiple assignment, randomized trials: an application in focal segmental glomerulosclerosis. Contemp. Clin. Trials. 2020 May;92 doi: 10.1016/j.cct.2020.105989. Epub 2020 Mar 19. PMID: 32200006; PMCID: PMC8173713. [DOI] [PMC free article] [PubMed] [Google Scholar]