Abstract
Acute liver injury is a type of liver diseases, and it has raised concerns worldwide due to the lack of effective therapies. The aim of this study is to investigate the protective effects of nootkatone (NOOT) on carbon tetrachloride (CCl4)-caused acute liver injury in mice. Mice were randomly divided into control, CCl4 model, NOOT, and NOOT (5, 10, and 20 mg/kg/day) plus CCl4 groups, respectively. Mice in the CCl4 plus NOOT groups were orally administrated with NOOT at 5, 10, and 20 mg/kg/days for seven days prior to 0.3% CCl4 injection at 10 mL/kg body weight, respectively. Our results showed that NOOT supplementation significantly ameliorated CCl4-induced increases of serum AST and ALT levels, hepatocyte necrosis, inflammatory response, oxidative stress, and caspases-9 and -3 activities in the livers of mice. Moreover, NOOT supplementation significantly upregulated the expression of Nrf2 and HO-1 mRNAs but downregulated the expression of NF-κB mRNAs and the levels of IL-1β, IL-6, and TNF-α proteins in the liver tissues, compared to those in the CCl4 model group. In conclusion, for the first time, our results reveal that NOOT could offer protective effects against CCl4-caused oxidative stress and inflammatory response via the opposite regulation of Nrf2/HO-1 pathway and NF-κB pathway.
Keywords: nootkatone, oxidative stress, acute liver injury, Nrf2/HO-1 pathway, NF-κB pathway
1. Introduction
In the past 20 years, liver diseases, including acute liver injury, chronic hepatitis, nonalcoholic fatty liver, and liver cancer, have been on the rise. Worldwide, liver diseases have become a leading cause of death [1]. However, so far, it still lacks effective treatment options. Therefore, the development of drugs for treating liver diseases remains an unmet medical need.
Acute liver damage is one class of liver diseases and occurs frequently. It is well known that the induction of oxidative stress, inflammatory response, necrosis, and apoptosis are all considered generally as the basis of acute liver injury caused by drugs, chemicals, or alcohol. These findings also provide the critical targets for clinical treatment and drug development [2]. Carbon tetrachloride (CCl4), a highly toxic chemical substance, has been widely used to build an acute liver damage model in rats and mice to develop and identify new hepatoprotective agents [3,4,5]. In the liver tissues of rodents, CCl4 could be metabolized quickly to trichloromethyl, dichloromethyl, or monochloromethyl radicals via the metabolic enzyme cytochrome 2E1 (CYP2E1), then producing excessive reactive oxygen species (ROS), and finally inducing cell necrosis or apoptosis [6]. Natural products have various biology functions, including antioxidant, anti-aging, immune-regulation, anti-inflammatory, and anti-microbial activities, which have been considered an important resource for the discovery of new drugs against acute liver damage [3,4,5,7,8,9,10].
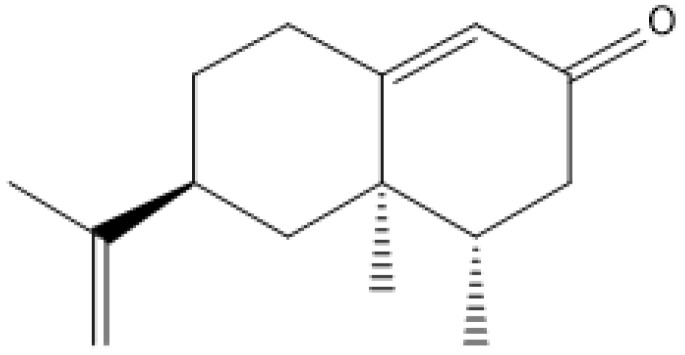
Many studies have reported that inhibition of oxidative stress or blockade of an inflammatory response may partly contribute to explaining the molecular mechanisms of natural products protecting against acute liver injury caused by CCl4 exposure [3,5,11,12]. Nootkatone (NOOT, Figure 1), one of the major active ingredients of the essential oil of Alpiniae oxyphyllae, exhibited multiple biological functions, such as antioxidant, anti-inflammatory, neuroprotection, and anti-microbial activities [13,14]. The previous studies have reported that the ethanol extract of Alpiniae oxyphyllae supplementation could ameliorate CCl4 exposure-caused acute liver injury by upregulating superoxide dismutase (SOD) and catalase (CAT) activities and downregulating malondialdehyde (MDA) levels in vitro [15]. Consistently, a recent study showed that intraperitoneal administration of NOOT at 50 mg/kg per day for four days could effectively inhibit lipopolysaccharide (LPS)-induced oxidative damage and inflammatory response in the brain tissues of mice [14]. NOOT could also directly activate the NF-E2-related factor (Nrf2) signaling pathway, which is a critical endogenous antioxidant signaling pathway in response to oxidative stress and plays a critical role during oxidative damage [9,10,14,15]. In another study, it is also reported that NOOT supplementation could effectively inhibit the expression of NAD(P)H oxidase-4 (NOX4) protein and then inhibit the production of ROS, finally inhibiting cell apoptosis and kidney fibrosis induced by unilateral ureteral obstruction surgical operation in mice [13]. In addition, several studies also reported that NOOT supplementation could directly target the inhibition of the expression of nuclear factor-kappaB (NF-κB), followed by inhibiting the LPS-induced production of pro-inflammatory factors in vitro, such as interleukin-1β (IL-1β), IL-6, TNF-α, and inducible nitric oxide synthase (iNOS) [14].
Figure 1.
The chemical structure of nootkatone (NOOT).
To date, it is not clear whether NOOT supplementation could provide a protective effect against acute liver injury caused by CCl4 exposure. Therefore, in the present study, the protective effects of NOOT supplementation on CCl4 exposure-induced acute liver injury were explored by using a mouse model. Furthermore, the underlying molecular mechanisms involving the regulation of Nrf2 and NF-κB pathways were explored.
2. Materials and Methods
2.1. Chemicals and Reagents
NOOT (CAS number: 4674-50-4 and its purity ≥ 97%) was provided by Aladdin Reagent Company (Shanghai, China). CCl4 was provided by Kaixing Chemical Company (Tianjin, China). BCA™ protein assay kit was obtained from Beyotime company (Haimen, China). Carboxyl methyl cellulose sodium (CMC-Na) was purchased from Sigma-Aldrich (Shanghai, China).
2.2. Animals and Treatments
In the present study, the Institutional Animal Care and Use Committee of China Agricultural University approved the animal experiments at 1 June 2022 (approvement number: CAU20220601-1). C57BL/6 mice aged 8-week-old (male; the body weight was in the range of 20–22 g) were obtained from Vital River Animal Technology Company (Beijing, China). Before the treatments, all mice were given adaptive feeding for one week. Five mice were kept in each cage and all mice had free access to water and food during experiments and in a standard animal house with the room temperature at 22 ± 3 °C and the relative humidity at 55 ± 5%. A 12 h light–dark cycle was performed during experiments.
All mice were randomly divided into six groups: untreated control group, NOOT 20 mg/kg/day group (i.e., NOOT 20 group), CCl4 model group, NOOT 5 mg/kg/day plus CCl4 group (i.e., CCl4+NOOT 5 group), NOOT 10 mg/kg/day plus CCl4 group (i.e., CCl4+NOOT 10 group), and NOOT 20 mg/kg/day plus CCl4 group (i.e., CCl4+NOOT 20 group). Eight mice were in each group. All mice in the CCl4-treated groups were injected intraperitoneally (i.p.) with 0.3% CCl4 (it was dissolved in olive oil) at the dose of 10 mL/kg body weight. NOOT was suspended using 0.5% CMC-Na to the final working solution for standby. Mice in the NOOT-treated groups were treated with the final concentrations of 5, 10, and 20 mg/kg/day for seven days prior to CCl4 treatment. Mice in the NOOT 20 mg/kg/day group were orally treated with NOOT at the dose of 20 mg/kg per day for seven days. In the control group, mice were orally treated with an equal volume of vehicle (i.e., 0.5% CMC-Na) and injected i.p. with an equal volume of olive oil. At 24 h after CCl4 administration, mice were anesthetized for euthanasia using the i.p. injection of pentobarbital sodium at 80 mg/kg body weight. The blood and liver samples were isolated for the corresponding histopathological and biochemical measurements.
2.3. Serum Biochemical Analysis
Blood samples of mice were collected in 1.5 mL Eppendorf tubes, and then the blood samples were centrifuged at 3000× g for 15 min, and the serum samples were collected for the biochemical measurements. The activities of aspartate aminotransferase (AST) and alanine aminotransferase (ALT) in the serum were determined by using a Hitachi 7080 automatic analyzer (Hitachi High-Technologies Corporation Company, Japan) according to the a previously published study [3].
2.4. Histopathological Assessment
The liver tissues from four mice in each group were fixed in 4% neutral formaldehyde for the histopathological analysis. The liver tissues were treated with a series of gradient ethanol dehydration, transparency, embedding, and section. Hematoxylin-eosin staining was performed according to a previous study [3]. Furthermore, the degree of liver injury was scored using a semi-quantitative score (SQS) system, according to the previous study [3]. The detail information of SQS was shown in Supplementary File S1.
2.5. Measurement for Malondialdehyde (MDA) Levels, and Catalase (CAT), and Superoxide Dismutase (SOD) Activities in the Liver Tissues of Mice
Parts of liver tissues (about 50 mg) were isolated and homogenized with 0.5 mL of cold Tris-buffer. Then, the homogenates were collected and centrifuged at 3000× g for 15 min at a temperature of 4 °C. Then, the supernatants were collected for the measurements of MDA levels and CAT and SOD activities, according to the corresponding manufacturer’s instructions provided by the MDA, CAT, and SOD commercial kits (Nanjing Jiancheng Company, Nanjing, China), respectively. A BCA™ protein assay kit was employed to measure the protein concentration of each sample.
2.6. Measurement of Inflammatory Markers IL-1β, TNF-α, and IL-6 Levels in Liver Tissues of Mice
The levels of inflammatory markers, including TNF-α, IL-1β, and IL-6 proteins, were determined using the corresponding TNF-a, IL-1 β and IL-6 ELISA kits (R&D Systems, Minneapolis, MN, USA). A BCA™ protein assay kit was employed to measure the protein concentration of each sample.
2.7. Measurements of Caspases-9 and -3 Activities in the Liver Tissues of Mice
A small part of liver tissues (about 20 mg) was isolated and lysed at 4 °C using 0.5 mL lysis buffer provided by the commercial kits for 15 min. Then, the lysate of each sample was centrifuged at 12,000× g for 15 min. After centrifugation, the supernatants were collected for the measurement of caspases-3 and -9 activities using the commercial caspases-3 and -9 kits (Beyotime Company, Haimen, China). A BCA™ protein assay kit was employed to measure the protein concentration of each sample.
2.8. Quantitative Reverse-Transcription (qRT)-PCR for the Gene Expression
About 20 mg liver tissues were used to isolate the total RNA for the gene expression analysis. Briefly, the total RNA isolations were performed by using a FastPure Cell/Tissue Total RNA Isolation Kit (No. RC112-01, Vazyme Biotech Co., Ltd., Nanjing, China). The quality of RNAs was assessed using a Nanodrop reader (Therma Fisher, Waltham, MA, USA) and the values of the optical density at 260/280 nm among 1.9~2.1. 500 ng of total RNAs were used to synthesize the cDNAs using a Prime Script RT-PCR kit (Takara Company, Beijing, China). All the primer sequences are shown in Supplementary Table S1. β-actin was used as the internal control gene. An AB7500 real-time PCR instrument (Applied Biosystems, Waltham, MA, USA) was used to perform the qRT–PCR. The 2−ΔΔCt method was employed to calculate the relative expression of targeted genes.
2.9. Statistical Analysis
In the present study, all data are analyzed and shown as mean ± standard deviation (S.D.) unless the specifical mention. A one-way analysis of variance (ANOVA) was selected for the statistical analysis. Then, Tukey’s multiple comparisons post hoc test was further performed. A p-value < 0.05 was considered statistically significant.
3. Results
3.1. NOOT Supplementation Ameliorates CCl4-Induced Liver Dysfunction in Mice
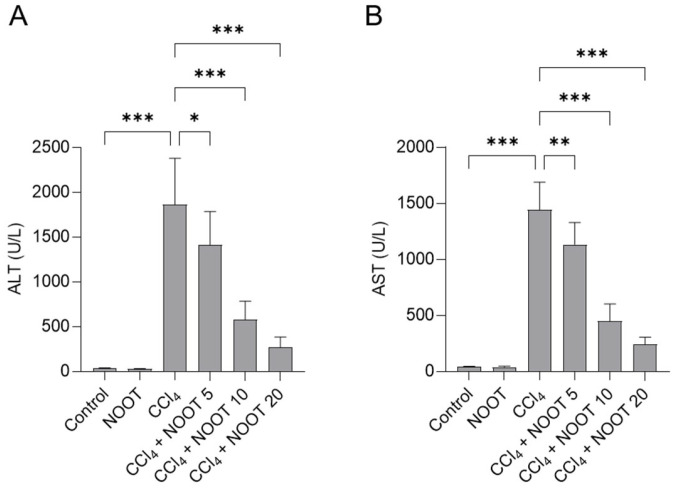
During the course of the experiment, no mice died. The liver functions of mice were further assessed. As shown in Figure 2, in the CCl4-treated mice, the levels of serum ALT and ALT markedly increased to 1863.8 U/L and 1443.0 U/L (both p < 0.001), respectively, compared to those in the control group. NOOT pre-treatment significantly improved CCl4 exposure-induced liver dysfunction. NOOT supplementation at the doses of 10 and 20 mg/kg/day for 7 days significantly decreased the levels of serum ALT to 577.3 U/L and 281.9 U/L (both p < 0.001), respectively, and significantly decreased the levels of serum AST to 451.4 U/L and 242.4 U/L (both p < 0.001), respectively, compared to those in the CCl4 model group. NOOT-alone treatment did not change the levels of serum ALT and AST, compared to those in the control group (Figure 2).
Figure 2.
The changes in serum ALT (A) and AST (B) levels. All results are shown as mean ± S.D. (n = 8). * p < 0.05, ** p < 0.01, and *** p < 0.001, compared between two different groups.
3.2. NOOT Supplementation Ameliorates the Acute Liver Injury Caused by CCl4 Exposure
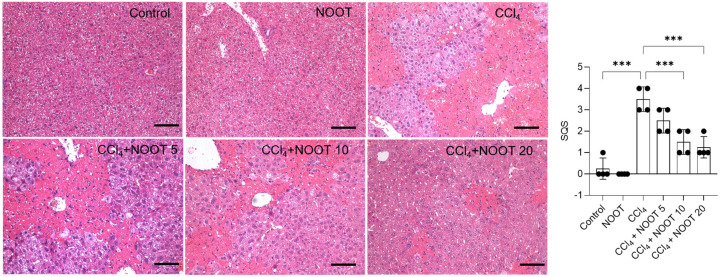
Compared to the control, CCl4 treatment resulted in marked histopathological injury in the liver tissues, which was significantly ameliorated by NOOT pretreatment in a dose-dependent manner. As shown in Figure 3, CCl4 treatment caused cell necrosis, and inflammatory cell infiltrations in the liver tissues and the corresponding SQS increased to 3.5 (p < 0.001), compared to that in the control group. NOOT supplementation could effectively improve CCl4 exposure-caused histopathological injuries. In the CCl4 + NOOT 10, and CCl4 + NOOT 20 groups, the SQS decreased to 1.5, and 1.0 (both p < 0.001), respectively. There was no marked histopathological change in the NOOT-alone treatment group compared to that in the control group.
Figure 3.
NOOT supplementation ameliorates CCl4-induced histopathological changes in the liver tissues of mice. Representative histopathological changes (on the left) and the corresponding the semi-quantitative score (SQS) were shown (on the right). Data are shown as mean ± S.D. (n = 4). *** p < 0.001, compared between two different groups. NOOT, nootkatone. Bar = 50 μm.
3.3. NOOT Supplementation Ameliorates CCl4 Exposure-Caused Oxidative Stress
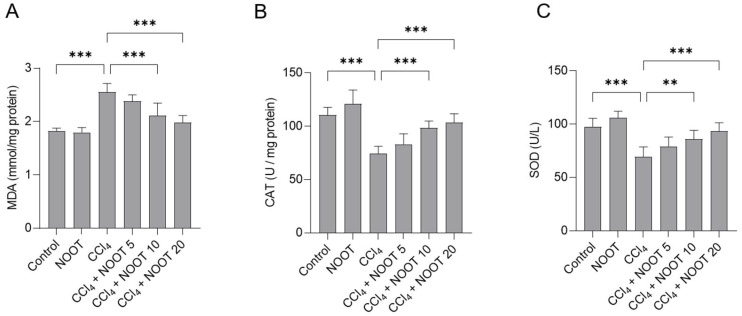
The biomarkers of oxidative stress, including the levels of MDA, and the activities of CAT, and SOD were determined. As shown in Figure 4, CCl4 treatment significantly increased the levels of MDA to 2.56 mmol/mg protein and significantly decreased the activities of CAT and SOD to 74.5 U/mg protein and 69.6 U/mg protein (all p < 0.001), respectively. NOOT supplementation significantly ameliorated CCl4 exposure-induced oxidative stress damage in the liver tissues. Correspondingly, in the CCl4 + NOOT 10, and CCl4 + NOOT 20 groups, MDA levels decreased to 2.11 mmol/mg protein and 1.98 mmol/mg protein, respectively; CAT activities increased to 98.4 U/mg protein and 103.3 U/mg protein, respectively; SOD activities increased to 85.9 U/mg protein and 93.3 U/mg protein, respectively. Compared to the control, mild increases in the levels of CAT and SOD activities and mild decreases in the MDA levels were detected in the NOOT-alone treatment group (Figure 4).
Figure 4.
NOOT supplementation ameliorates CCl4 exposure-caused oxidative stress in the liver tissues of mice. Mice were orally pretreated with NOOT at the doses of 5, 10, and 20 mg/kg/day for 7 days prior to CCl4 exposure, then, the levels of MDA (A), and the activities of CAT (B) and SOD (C) in the liver tissues of mice were measured. Data are presented as mean ± S.D. (n = 8). ** p < 0.01, and *** p < 0.001, compared between two different groups. NOOT, nootkatone.
3.4. NOOT Supplementation Ameliorates CCl4-Induced Inflammtory Response
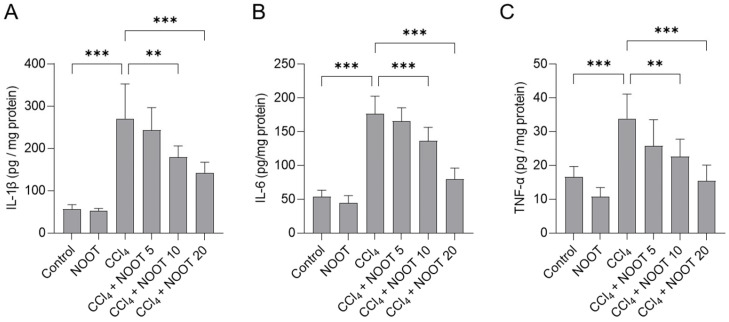
As shown in Figure 5, CCl4 exposure significantly increased the levels of IL-1β, IL-6, and TNF-α proteins in the liver tissues of mice. Compared to the CCl4 model group, NOOT supplementation at 10 and 20 mg/kg/day for 7 days significantly decreased the levels of IL-1β protein from 270.9 pg/mg protein to 179.7 pg/mg protein, and 142.1 pg/mg protein (p < 0.01 or p < 0.001), respectively; significantly decreased the levels of IL-6 protein from 176.5 pg/mg protein to 136.3 pg/mg protein, and 80.1 pg/mg protein (both p < 0.001), respectively; and significantly decreased the levels of TNF-α protein from 33.9 pg/mg protein to 22.7 pg/mg protein, and 15.4 pg/mg protein (p < 0.01 or p < 0.001), respectively. These inflammatory markers had no marked changes in the liver tissues of mice in the NOOT-alone group, compared to that in the control group.
Figure 5.
NOOT supplementation ameliorates CCl4 exposure-caused inflammatory response in the liver tissues of mice. Mice were orally pretreated with NOOT at the doses of 5, 10, and 20 mg/kg/day for 7 days prior to CCl4 exposure; then, the levels of IL-1β (A), IL-6 (B), and TNF-α (C) proteins in the liver tissues of mice were measured. Data are shown as mean ± S.D. (n = 8). ** p < 0.01, and *** p < 0.001, compared between two different groups. NOOT, nootkatone.
3.5. NOOT Supplementation Ameliorates CCl4 Exposure-Caused the Activation of Caspases-9 and -3
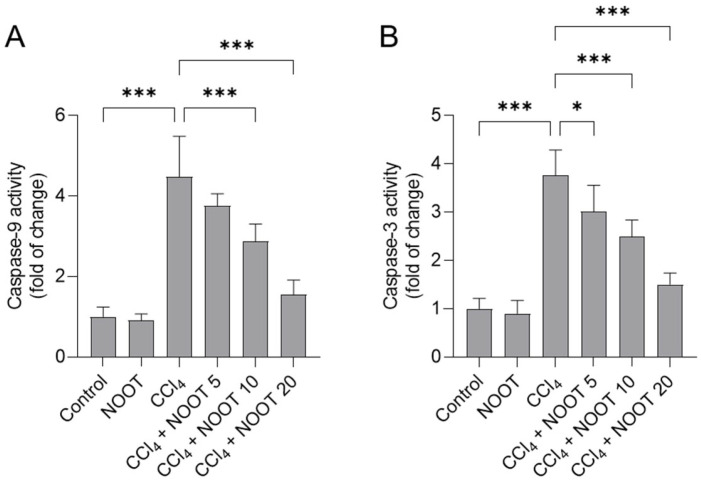
Compared to the control group, CCl4 exposure significantly upregulated the levels of caspases-9 and -3 activities to 4.49- and 3.76-fold (both p < 0.001) in the liver tissues of mice, respectively (Figure 6). Compared to the CCl4 model group, NOOT supplementation at the doses of 10 and 20 mg/kg/day for 7 days significantly decreased caspase-9 activities to 2.87- and 1.55-fold (both p < 0.001), respectively, and significantly decreased caspase-3 activities to 2.49- and 1.50-fold (both p < 0.001), respectively. There was no marked change in the levels of caspases-9 and -3 in the liver tissues of mice in the NOOT-alone treatment group, compared to those in the control group.
Figure 6.
NOOT supplementation ameliorated CCl4 exposure-caused the activation of caspases-9 and -3 in the liver tissues of mice. Mice were orally pretreated with NOOT at the doses of 5, 10, and 20 mg/kg/day for 7 days prior to CCl4 exposure; then, the activities of caspases-9 (A) and -3 (B) in the liver tissues of mice were measured. Data are shown as mean ± S.D. (n = 6). * p < 0.05, and *** p < 0.001, compared between two different groups. NOOT, nootkatone.
3.6. NOOT Supplementation Downregulates the Expression of NF-κB mRNA and Upregulates the Expression of Nrf2 and HO-1 mRNAs
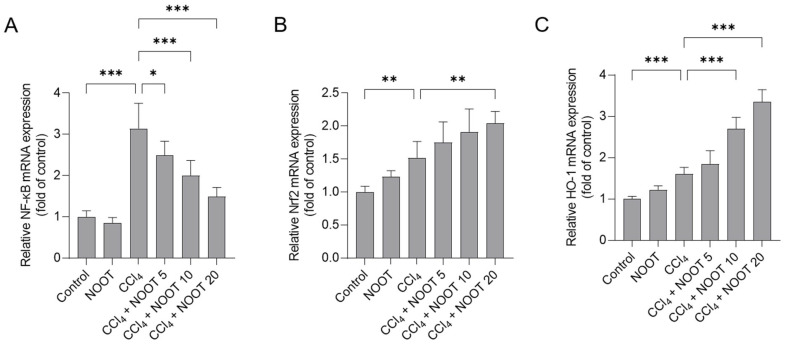
The relative expressions of NF-κB, Nrf2, and HO-1 mRNAs in the liver tissues of mice were examined. Compared to the untreated control group, CCl4 exposure significantly upregulated the expression of NF-κB, Nrf2, and HO-1 mRNAs to 3.14-, 1.54-, and 1.61-fold (p < 0.01 or p < 0.001), respectively (Figure 7). NOOT supplementation could regulate the relative expression of these genes. As shown in Figure 7, in the CCl4+ NOOT 10 and CCl4+ NOOT 20 groups, the relative expression of NF-κB mRNA decreased to 2.01- and 1.49-fold, respectively (both p < 0.001), while the relative expression of Nrf2 mRNA increased to 1.84-, and 2.23-fold (p < 0.01), respectively, and the relative expression of HO-1 mRNA decreased 2.69-, and 3.35-fold (both p < 0.001), respectively, compared to the CCl4 model group.
Figure 7.
The relative expression of NF-κB (A), Nrf2 (B), and HO-1 (C) mRNAs in the liver tissues of mice. Data are shown as mean ± S.D. (n = 6). * p < 0.05, ** p < 0.01, and *** p < 0.001, compared between two different groups. NOOT, nootkatone.
4. Discussion
Chemical liver injury is one class of acute liver injury and liver diseases have become a global public health burden [1]. It has been reported that many hazardous substances, including aflatoxin B1, cadmium, and mercury chloride, could induce acute liver injury, finally resulting in acute hepatitis or liver failure in people [16,17,18]. Currently, effective drugs for treating and preventing acute liver injury are still scarce, and it is essential for the development of effective therapeutic agents against acute liver injury induced by environmental hazardous substances. In this study, our results found that NOOT, a natural product isolated from Alpinia oxyphylla, could effectively ameliorate CCl4 exposure-caused acute liver dysfunction in a mouse model (Figure 2, Figure 3, Figure 4, Figure 5, Figure 6 and Figure 7). The potential molecular mechanisms may involve antioxidant and anti-inflammatory activities through the activation of the Nrf2/HO-1 pathway and the inhibition of the NF-κB pathway (Figure 2, Figure 3, Figure 4, Figure 5, Figure 6 and Figure 7).
CCl4 is a common hepatotoxic compound. It is usually used to establish the acute liver injury model for the development of effective hepatoprotectants in rodents [5,19]. In line with the previous studies [3,4,20], our data showed that CCl4 exposure significantly increased the levels of serum ALT and AST by about 40~50-fold (Figure 2), indicating that acute liver dysfunction model in mice was successfully established. NOOT supplementation at 5, 10, and 20 mg/kg/days for 7 days could dose-dependently decrease the levels of serum ALT and AST (Figure 2), as well as the histopathology damage (Figure 3), indicating the hepaprotective effects of NOOT. Very recently, Yan et al. reported that oral supplementation of NOOT at the doses of 5 or 10 mg/kg/day for 4 weeks could partly abolish d-galactosamine-induced acute liver injury in mice [21]. These data indicated that NOOT supplementation could provide a protective effect against liver dysfunction caused by CCl4 exposure.
It is well known that excessive ROS production is the main basis of CCl4 exposure-induced liver and renal injuries [3,4,20,22]. Excessive ROS production can directly damage DNA, lipids, proteins, and the main organelles of cells, such as the mitochondrion, endoplasmic reticulum, and lysosome [4,10,16,23]. A previous study reported that CCl4 exposure could significantly promote the production of ROS in liver tissues and induce liver dysfunction in mice [24]. In the current study, our data showed that CCl4 treatment significantly upregulates the levels of MDA and downregulates the activities of SOD and CAT in the liver tissues of mice. NOOT supplementation effectively inhibited CCl4-induced increases in MDA levels and rescued the levels of SOD and CAT in the liver tissues of mice (Figure 4). MDA is a biomarker of lipid peroxidation [25]. SOD and CAT are considered two important antioxidant enzymes, which can directly catalyze superoxide anion and hydrogen peroxide (H2O2), respectively, and both finally form non-toxic substances in cells [26]. NOOT was shown to have potent antioxidant activity and NOOT supplementation can effectively inhibit the production of ROS, reduce MDA levels, and upregulate the activities of SOD and CAT in H2O2-treated PC12 cells in vitro [27]. It was also reported that NOOT supplementation could protect against doxorubicin-induced cardiotoxicity and induced testicular toxicity induced by water pipe smoke in mice by inhibiting lipid peroxidation and upregulating the activities of SOD and CAT [28,29]. Taken together, our data indicated that NOOT co-administration could provide a protective effect via the upregulation of endogenous antioxidant enzymes SOD and CAT activities.
In the process of liver dysfunction caused by CCl4 exposure, a concurrent inflammatory reaction was usually induced [30,31,32,33,34,35,36,37,38]. In this study, marked inflammatory responses in the CCl4-treated liver tissues were detected, which were evident due to the increased expression of IL-1β, IL-6, and TNF-α proteins, as well as the inflammatory cell infiltrations in the liver tissues of mice (Figure 5). These findings are consistent with the previous literature [3,4,20,22,30,31,32,33,34,35,36,37,38]. In addition, our data showed that NOOT supplementation could effectively inhibit the expression of IL-1β, IL-6, and TNF-α proteins induced by CCl4 exposure (Figure 5). Several animal and in vitro cell experiments also found that NOOT supplementation could significantly inhibit LPS-, carrageenan-, anterior-cruciate-ligament-transection-, and isoproterenol-induced acute or chronic inflammatory responses [14,39,40,41]. Taken together, these evidences indicate that NOOT supplementation may provide hepaprotective effects by inhibiting the inflammatory response. Moreover, previous studies have demonstrated that the inhibition of NF-κB, adenosine 5‘-monophosphate (AMP)-activated protein kinase (AMPK), or Toll-like receptor 4 (TLR4) pathways could partly contribute to explaining the anti-inflammatory mechanisms of NOOT [14,40,41]. NF-κB is a vital transcriptional factor that could activate the expression of number of pro-inflammatory cytokines, such as TNF-α, IL-1β, and IL-6 [42]. In this study, our results showed that NOOT supplementation can significantly inhibit CCl4 exposure-caused the expression of NF-κB mRNA in the liver tissues (Figure 7). Therefore, these evidences indicate that the anti-inflammatory effect of NOOT may be partly attributed to the inhibition of the NF-κB pathway. Furthermore, a recent study found that NAD(P)H quinone oxidoreductase 1 (NQO1) knockdown could partly attenuate the anti-inflammatory effects of NOOT in LPS-treated cells [14]. Therefore, there is a cross-talk between Nrf2 and NF-κB pathways in the protective effects of NOOT against CCl4 exposure-caused liver dysfunction. Therefore, the precise actional mechanisms need more studies.
Apoptosis is a type of programmed cell death in mammalian cells. It can be activated by many signaling pathways [43]. In the present study, significant upregulation of caspases-9 and -3 activities in CCl4-treated liver tissues of mice were detected, which were partly revised by NOOT supplementation in a dose-dependent manner (Figure 6). It is well known that caspase-3 is a classical biomarker of apoptosis and caspase-9 is a critical biomarker in the process of the mitochondrial apoptotic pathway [43]. Previous studies have shown that CCl4 exposure could cause mitochondrial dysfunction, resulting in inducing the expression of cytochrome C (CytC), caspase-9, and caspase-3, finally inducing apoptotic cell death [3,4,11,44]. Consistently, Nemmar et al. found that NOOT supplementation at 90 mg/kg per day could significantly inhibit caspase-3 activation and DNA damage induced by diesel exhaust particles [45]. In another study, the authors found that Alpiniae oxyphyllae extract (protocatechuic acid and NOOT are suggested as the two main components) supplementation could effectively inhibit mitochondrial dysfunction, and caspase-3 activation, and apoptotic cell death induced by H2O2 in PC12 cells [27]. In short, this evidence indicates that NOOT supplementation can improve liver dysfunction caused by CCl4 exposure via the inhibitory effect on the mitochondrial apoptotic pathway.
Recently, several studies have reported that NOOT is the activator of Nrf2, a vital transcription factor in response to oxidative stress [14,21,41]. Under the normal condition, Nrf2 is located in the cytoplasm via the interaction with Kelch-like ECH-associated protein 1 (Keap1), which results in the ubiquitin-dependent degradation of Nrf2. Under the stress condition, Nrf2 is separated from Nrf2-Keap1 complex and enters the nucleus; then, it transcriptionally upregulates the expression of more than 200 cell protective genes, such as HO-1, SOD, CAT, and NQO1 [46]. In our previous studies, it was shown that the knockdown of Nrf2 or pharmacology inhibition of HO-1 could exacerbate the cytotoxicity in CCl4 -treated HepG2 cells or acute liver injury in mice [3,11]. In the present study, our data showed that NOOT supplementation could significantly upregulate the mRNA expression of Nrf2 and its downstream gene HO-1 (Figure 7). Consistently, Meeran et al.’s study found that NOOT cotreatment at 10 mg/kg/day for 10 days could effectively upregulate the expression of Nrf2 and HO-1 proteins in heart tissues and protect against isoproterenol-induced heart toxicity in rats [41]. Park et al. showed that NOOT treatment could significantly activate the transcriptional expression of Nrf2 and the expression of HO-1 and NQO1 proteins in LPS-treated BV2 cells [14]. Furthermore, the pharmacological inhibition or genic silencing of NQO1, a key downstream target of Nrf2, partly attenuated the inhibitory effects of NOOT on LPS-induced ROS production in BV2 cells [14]. In short, these evidences indicate that the activation of Nrf2/HO-1 may partly contribute to the protective effect of NOOT against liver injury caused by. In addition, the activation of Nrf2 pathway may also partly support the antioxidant activities of NOOT via the transcriptional upregulation of SOD and CAT genes. However, the precise mechanisms still require more investigation.
Alpiniae oxyphyllae, one of the “four famous south medicines” in China, has been widely used as a food and medicine in China [47]. As a traditional Chinese medicine, Alpiniae oxyphyllae has been used for treating diarrhea and neurodegenerative diseases [47]. NOOT is one of the main active ingredients. Like Alpiniae oxyphyllae, the therapeutic effects of NOOT on liver disease, kidney disease, and neurodegenerative disease have been illustrated in vitro and animal studies [47,48]. However, the information on the safety and pharmacokinetic parameters of NOOT is still limited, so it should be further investigated before being applied to humans.
5. Conclusions
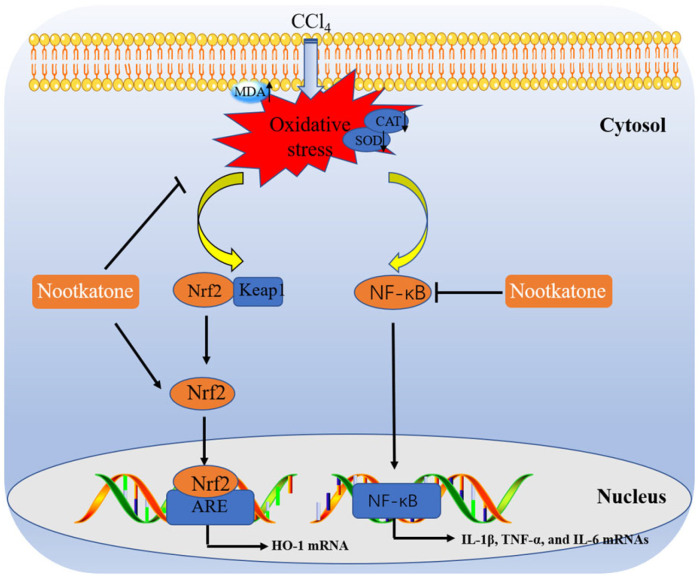
In conclusion, for the first time, our data reveal that NOOT supplementation can effectively improve CCl4 -induced acute liver injury through inhibiting lipid peroxidation, oxidative stress, and inflammatory response, which may be partly attributed to the activation of Nrf2/HO-1 pathway and the inhibition of NF-κB pathway. A proposed work model of NOOT protecting against acute liver injury induced by CCl4 exposure was shown in Figure 8. Our current study shed that NOOT may be a promising candidate for treating acute liver injury.
Figure 8.
A proposed work model of NOOT protecting against acute liver injury caused by CCl4 exposure. MDA, malondialdehyde; CAT, catalase; SOD, superoxide dismutase; Nrf2, NF-E2-related factor; NF-κB, nuclear factor-kappaB; IL-1β, interleukin-1β; IL-6, interleukin-6; TNF-α, tumor necrosis factor-α; HO-1, heme oxygenase-1; ARE, antioxidant response element.
Supplementary Materials
The following supporting information can be downloaded at https://www.mdpi.com/article/10.3390/antiox12010194/s1. Supplementary File S1: Histopathological assessments; Table S1: the primer information for qRT-PCR.
Author Contributions
Conceptualization, C.D. and J.S.; methodology, C.D. and X.Z.; software, J.L.; validation, C.D.; formal analysis, J.L. and C.D.; investigation, C.D. and X.Z.; data curation, C.D.; writing—original draft preparation, C.D.; writing—review and editing, C.D. and J.S.; project administration, C.D.; funding acquisition, C.D., and J.S. All authors have read and agreed to the published version of the manuscript.
Institutional Review Board Statement
The study was conducted according to the guidelines of Institutional Animal Care and Use Committee at the China Agricultural University (Approval number CAU20220601-1).
Informed Consent Statement
Not applicable.
Data Availability Statement
The data are contained within this article.
Conflicts of Interest
The authors declare no conflict of interest.
Funding Statement
This study was supported by Guangdong Major Project of Basic and Applied Basic Research, No. 2020B0301030007. This study was also supported by the National Natural Science Foundation of China (Award number 32102724).
Footnotes
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content.
References
- 1.Xiao J., Wang F., Wong N.K., He J., Zhang R., Sun R., Xu Y., Liu Y., Li W., Koike K., et al. Global liver disease burdens and research trends: Analysis from a Chinese perspective. J. Hepatol. 2019;71:212–221. doi: 10.1016/j.jhep.2019.03.004. [DOI] [PubMed] [Google Scholar]
- 2.Wu Z., Han M., Chen T., Yan W., Ning Q. Acute liver failure: Mechanisms of immune-mediated liver injury. Liver Int. 2010;30:782–794. doi: 10.1111/j.1478-3231.2010.02262.x. [DOI] [PubMed] [Google Scholar]
- 3.Dai C., Li H., Wang Y., Tang S., Velkov T., Shen J. Inhibition of Oxidative Stress and ALOX12 and NF-κB Pathways Contribute to the Protective Effect of Baicalein on Carbon Tetrachloride-Induced Acute Liver Injury. Antioxidants. 2021;10:976. doi: 10.3390/antiox10060976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Dai C., Xiao X., Li D., Tun S., Wang Y., Velkov T., Tang S. Chloroquine ameliorates carbon tetrachloride-induced acute liver injury in mice via the concomitant inhibition of inflammation and induction of apoptosis. Cell Death Dis. 2018;9:1164. doi: 10.1038/s41419-018-1136-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Mitazaki S., Kotajima N., Matsuda S., Ida N., Iide M., Honma S., Suto M., Kato N., Kuroda N., Hiraiwa K., et al. Dimethylthiourea ameliorates carbon tetrachloride-induced acute liver injury in ovariectomized mice. Biomed. Pharmacother. 2018;104:427–436. doi: 10.1016/j.biopha.2018.05.065. [DOI] [PubMed] [Google Scholar]
- 6.Lee J., Giordano S., Zhang J. Autophagy, mitochondria and oxidative stress: Cross-talk and redox signalling. Biochem. J. 2012;441:523–540. doi: 10.1042/BJ20111451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Zou J., Wang S.P., Wang Y.T., Wan J.B. Regulation of the NLRP3 inflammasome with natural products against chemical-induced liver injury. Pharmacol. Res. 2021;164:105388. doi: 10.1016/j.phrs.2020.105388. [DOI] [PubMed] [Google Scholar]
- 8.Meng X., Li Y., Li S., Gan R.Y., Li H.B. Natural Products for Prevention and Treatment of Chemical-Induced Liver Injuries. Compr. Rev. Food Sci. Food Saf. 2018;17:472–495. doi: 10.1111/1541-4337.12335. [DOI] [PubMed] [Google Scholar]
- 9.Dai C., Lin J., Li H., Shen Z., Wang Y., Velkov T., Shen J. The Natural Product Curcumin as an Antibacterial Agent: Current Achievements and Problems. Antioxidants. 2022;11:459. doi: 10.3390/antiox11030459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Dai C., Zhang Q., Shen L., Sharma G., Jiang H., Wang Z., Shen J. Quercetin Attenuates Quinocetone-Induced Cell Apoptosis In Vitro by Activating the P38/Nrf2/HO-1 Pathway and Inhibiting the ROS/Mitochondrial Apoptotic Pathway. Antioxidants. 2022;11:1498. doi: 10.3390/antiox11081498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Peng X., Dai C., Liu Q., Li J., Qiu J. Curcumin Attenuates on Carbon Tetrachloride-Induced Acute Liver Injury in Mice via Modulation of the Nrf2/HO-1 and TGF-β1/Smad3 Pathway. Molecules. 2018;23:215. doi: 10.3390/molecules23010215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Zhang X., Kuang G., Wan J., Jiang R., Ma L., Gong X., Liu X. Salidroside protects mice against CCl4-induced acute liver injury via down-regulating CYP2E1 expression and inhibiting NLRP3 inflammasome activation. Int. Immunopharmacol. 2020;85:106662. doi: 10.1016/j.intimp.2020.106662. [DOI] [PubMed] [Google Scholar]
- 13.Chen C.M., Lin C.Y., Chung Y.P., Liu C.H., Huang K.T., Guan S.S., Wu C.T., Liu S.H. Protective Effects of Nootkatone on Renal Inflammation, Apoptosis, and Fibrosis in a Unilateral Ureteral Obstructive Mouse Model. Nutrients. 2021;13:3921. doi: 10.3390/nu13113921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Park J.E., Park J.S., Leem Y.H., Kim D.Y., Kim H.S. NQO1 mediates the anti-inflammatory effects of nootkatone in lipopolysaccharide-induced neuroinflammation by modulating the AMPK signaling pathway. Free Radic. Biol. Med. 2021;164:354–368. doi: 10.1016/j.freeradbiomed.2021.01.015. [DOI] [PubMed] [Google Scholar]
- 15.Zhang Q., Hu X., Hui F., Song Q., Cui C., Wang C., Zhao Q. Ethanol extract and its dichloromethane fraction of Alpinia oxyphylla Miquel exhibited hepatoprotective effects against CCl(4)-induced oxidative damage in vitro and in vivo with the involvement of Nrf2. Biomed. Pharmacother. 2017;91:812–822. doi: 10.1016/j.biopha.2017.04.131. [DOI] [PubMed] [Google Scholar]
- 16.Dai C., Tian E., Hao Z., Tang S., Wang Z., Sharma G., Jiang H., Shen J. Aflatoxin B1 Toxicity and Protective Effects of Curcumin: Molecular Mechanisms and Clinical Implications. Antioxidants. 2022;11:2031. doi: 10.3390/antiox11102031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Cao Z., Fang Y., Lu Y., Tan D., Du C., Li Y., Ma Q., Yu J., Chen M., Zhou C., et al. Melatonin alleviates cadmium-induced liver injury by inhibiting the TXNIP-NLRP3 inflammasome. J. Pineal Res. 2017;62:e12389. doi: 10.1111/jpi.12389. [DOI] [PubMed] [Google Scholar]
- 18.Reus I.S., Bando I., Andrés D., Cascales M. Relationship between expression of HSP70 and metallothionein and oxidative stress during mercury chloride induced acute liver injury in rats. J. Biochem. Mol. Toxicol. 2003;17:161–168. doi: 10.1002/jbt.10074. [DOI] [PubMed] [Google Scholar]
- 19.Chen M., Huang W., Wang C., Nie H., Li G., Sun T., Yang F., Zhang Y., Shu K., Wang C., et al. High-mobility group box 1 exacerbates CCl₄-induced acute liver injury in mice. Clin. Immunol. (Orlando Fla.) 2014;153:56–63. doi: 10.1016/j.clim.2014.03.021. [DOI] [PubMed] [Google Scholar]
- 20.Venugopal S.K., Wu J., Catana A.M., Eisenbud L., He S.Q., Duan Y.Y., Follenzi A., Zern M.A. Lentivirus-mediated superoxide dismutase1 gene delivery protects against oxidative stress-induced liver injury in mice. Liver Int. 2007;27:1311–1322. doi: 10.1111/j.1478-3231.2007.01612.x. [DOI] [PubMed] [Google Scholar]
- 21.Yan T., Li F., Xiong W., Wu B., Xiao F., He B., Jia Y. Nootkatone improves anxiety- and depression-like behavior by targeting hyperammonemia-induced oxidative stress in D-galactosamine model of liver injury. Environ. Toxicol. 2021;36:694–706. doi: 10.1002/tox.23073. [DOI] [PubMed] [Google Scholar]
- 22.Zhan J., Cao H., Hu T., Shen J., Wang W., Wu P., Yang G., Ho C.T., Li S. Efficient Preparation of Black Tea Extract (BTE) with the High Content of Theaflavin Mono- and Digallates and the Protective Effects of BTE on CCl(4)-Induced Rat Liver and Renal Injury. J. Agric. Food Chem. 2021;69:5938–5947. doi: 10.1021/acs.jafc.1c01851. [DOI] [PubMed] [Google Scholar]
- 23.Wang M., Niu J., Ou L., Deng B., Wang Y., Li S. Zerumbone Protects against Carbon Tetrachloride (CCl4)-Induced Acute Liver Injury in Mice via Inhibiting Oxidative Stress and the Inflammatory Response: Involving the TLR4/NF-kappaB/COX-2 Pathway. Molecules. 2019;24:1964. doi: 10.3390/molecules24101964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Ma J.Q., Ding J., Zhang L., Liu C.M. Hepatoprotective properties of sesamin against CCl4 induced oxidative stress-mediated apoptosis in mice via JNK pathway. Food Chem. Toxicol. 2014;64:41–48. doi: 10.1016/j.fct.2013.11.017. [DOI] [PubMed] [Google Scholar]
- 25.Dai C., Li B., Zhou Y., Li D., Zhang S., Li H., Xiao X., Tang S. Curcumin attenuates quinocetone induced apoptosis and inflammation via the opposite modulation of Nrf2/HO-1 and NF-kB pathway in human hepatocyte L02 cells. Food Chem. Toxicol. 2016;95:52–63. doi: 10.1016/j.fct.2016.06.025. [DOI] [PubMed] [Google Scholar]
- 26.Zhao X.C., Zhang L., Yu H.X., Sun Z., Lin X.F., Tan C., Lu R.R. Curcumin protects mouse neuroblastoma Neuro-2A cells against hydrogen-peroxide-induced oxidative stress. Food Chem. 2011;129:387–394. doi: 10.1016/j.foodchem.2011.04.089. [DOI] [PubMed] [Google Scholar]
- 27.Li R., Wang L., Zhang Q., Duan H., Qian D., Yang F., Xia J. Alpiniae oxyphyllae fructus possesses neuroprotective effects on H(2)O(2) stimulated PC12 cells via regulation of the PI3K/Akt signaling Pathway. Front. Pharmacol. 2022;13:966348. doi: 10.3389/fphar.2022.966348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Al-Salam S., Kandhan K., Sudhadevi M., Tariq S. Nootkatone Ameliorates Doxorubicin Induced Myocardial Injury through Modulation of NF-κB Signals and Oxidative Stress. Cell Physiol. Biochem. 2022;56:401–417. doi: 10.33594/000000559. [DOI] [PubMed] [Google Scholar]
- 29.Ali B.H., Al-Salam S., Adham S.A., Al Balushi K., Al Za’abi M., Beegam S., Yuvaraju P., Manoj P., Nemmar A. Testicular Toxicity of Water Pipe Smoke Exposure in Mice and the Effect of Treatment with Nootkatone Thereon. Oxidative Med. Cell. Longev. 2019;2019:2416935. doi: 10.1155/2019/2416935. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Zhang S., Lu B., Han X., Xu L., Qi Y., Yin L., Xu Y., Zhao Y., Liu K., Peng J. Protection of the flavonoid fraction from Rosa laevigata Michx fruit against carbon tetrachloride-induced acute liver injury in mice. Food Chem. Toxicol. 2013;55:60–69. doi: 10.1016/j.fct.2012.12.041. [DOI] [PubMed] [Google Scholar]
- 31.Zou Y., Xiong J.B., Ma K., Wang A.Z., Qian K.J. Rac2 deficiency attenuates CCl4-induced liver injury through suppressing inflammation and oxidative stress. Biomed. Pharmacother. 2017;94:140–149. doi: 10.1016/j.biopha.2017.07.074. [DOI] [PubMed] [Google Scholar]
- 32.Zhang D.G., Zhang C., Wang J.X., Wang B.W., Wang H., Zhang Z.H., Chen Y.H., Lu Y., Tao L., Wang J.Q., et al. Obeticholic acid protects against carbon tetrachloride-induced acute liver injury and inflammation. Toxicol. Appl. Pharmacol. 2017;314:39–47. doi: 10.1016/j.taap.2016.11.006. [DOI] [PubMed] [Google Scholar]
- 33.Shi H., Han W., Shi H., Ren F., Chen D., Chen Y., Duan Z. Augmenter of liver regeneration protects against carbon tetrachloride-induced liver injury by promoting autophagy in mice. Oncotarget. 2017;8:12637. doi: 10.18632/oncotarget.14478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Cong M., Zhao W., Liu T., Wang P., Fan X., Zhai Q., Bao X., Zhang D., You H., Kisseleva T., et al. Protective effect of human serum amyloid P on CCl4-induced acute liver injury in mice. Int. J. Mol. Med. 2017;40:454–464. doi: 10.3892/ijmm.2017.3028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Cao M., Wang H., Guo L., Yang S., Liu C., Khor T.O., Yu S., Kong A.N. Dibenzoylmethane Protects Against CCl4-Induced Acute Liver Injury by Activating Nrf2 via JNK, AMPK, and Calcium Signaling. AAPS J. 2017;19:1703–1714. doi: 10.1208/s12248-017-0133-1. [DOI] [PubMed] [Google Scholar]
- 36.Torres L.R., Santana F.C., Torres-Leal F.L., Melo I.L., Yoshime L.T., Matos-Neto E.M., Seelaender M.C., Araujo C.M., Cogliati B., Mancini-Filho J. Pequi (Caryocar brasiliense Camb.) almond oil attenuates carbon tetrachloride-induced acute hepatic injury in rats: Antioxidant and anti-inflammatory effects. Food Chem. Toxicol. 2016;97:205–216. doi: 10.1016/j.fct.2016.09.009. [DOI] [PubMed] [Google Scholar]
- 37.Niu L., Cui X., Qi Y., Xie D., Wu Q., Chen X., Ge J., Liu Z. Involvement of TGF-beta1/Smad3 Signaling in Carbon Tetrachloride-Induced Acute Liver Injury in Mice. PLoS ONE. 2016;11:e0156090. doi: 10.1371/journal.pone.0156090. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Yang B.Y., Zhang X.Y., Guan S.W., Hua Z.C. Protective Effect of Procyanidin B2 against CCl4-Induced Acute Liver Injury in Mice. Molecules. 2015;20:12250–12265. doi: 10.3390/molecules200712250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Bezerra Rodrigues Dantas L., Silva A.L.M., da Silva Júnior C.P., Alcântara I.S., Correia de Oliveira M.R., Oliveira Brito Pereira Bezerra Martins A., Ribeiro-Filho J., Coutinho H.D.M., Rocha Santos Passos F., Quintans-Junior L.J., et al. Nootkatone Inhibits Acute and Chronic Inflammatory Responses in Mice. Molecules. 2020;25:2181. doi: 10.3390/molecules25092181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Xu Y., Zhang M., Yang W., Xia B., Wang W., Pan X. Nootkatone protects cartilage against degeneration in mice by inhibiting NF-κB signaling pathway. Int. Immunopharmacol. 2021;100:108119. doi: 10.1016/j.intimp.2021.108119. [DOI] [PubMed] [Google Scholar]
- 41.Meeran M.F.N., Azimullah S., Adeghate E., Ojha S. Nootkatone attenuates myocardial oxidative damage, inflammation, and apoptosis in isoproterenol-induced myocardial infarction in rats. Phytomed. Int. J. Phytother. Phytopharm. 2021;84:153405. doi: 10.1016/j.phymed.2020.153405. [DOI] [PubMed] [Google Scholar]
- 42.Morgan M.J., Liu Z.G. Crosstalk of reactive oxygen species and NF-kappaB signaling. Cell Res. 2011;21:103–115. doi: 10.1038/cr.2010.178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Dai C., Tang S., Deng S., Zhang S., Zhou Y., Velkov T., Li J., Xiao X. Lycopene attenuates colistin-induced nephrotoxicity in mice via activation of the Nrf2/HO-1 pathway. Antimicrob. Agents Chemother. 2015;59:579–585. doi: 10.1128/AAC.03925-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Jang S., Yu L.R., Abdelmegeed M.A., Gao Y., Banerjee A., Song B.J. Critical role of c-jun N-terminal protein kinase in promoting mitochondrial dysfunction and acute liver injury. Redox Biol. 2015;6:552–564. doi: 10.1016/j.redox.2015.09.040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Nemmar A., Al-Salam S., Beegam S., Yuvaraju P., Hamadi N., Ali B.H. In Vivo Protective Effects of Nootkatone against Particles-Induced Lung Injury Caused by Diesel Exhaust Is Mediated via the NF-κB Pathway. Nutrients. 2018;10:263. doi: 10.3390/nu10030263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Buendia I., Michalska P., Navarro E., Gameiro I., Egea J., León R. Nrf2-ARE pathway: An emerging target against oxidative stress and neuroinflammation in neurodegenerative diseases. Pharmacol. Ther. 2016;157:84–104. doi: 10.1016/j.pharmthera.2015.11.003. [DOI] [PubMed] [Google Scholar]
- 47.Zhang Q., Zheng Y., Hu X., Hu X., Lv W., Lv D., Chen J., Wu M., Song Q., Shentu J. Ethnopharmacological uses, phytochemistry, biological activities, and therapeutic applications of Alpinia oxyphylla Miquel: A review. J. Ethnopharmacol. 2018;224:149–168. doi: 10.1016/j.jep.2018.05.002. [DOI] [PubMed] [Google Scholar]
- 48.Jha A.K., Gairola S., Kundu S., Doye P., Syed A.M., Ram C., Kulhari U., Kumar N., Murty U.S., Sahu B.D. Biological Activities, Pharmacokinetics and Toxicity of Nootkatone: A Review. Mini Rev. Med. Chem. 2022;22:2244–2259. doi: 10.2174/1389557522666220214092005. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The data are contained within this article.