Key Points
Question
Is the oral JAK2/IRAK1 inhibitor pacritinib superior to placebo in patients hospitalized with severe COVID-19?
Findings
In this phase 2 randomized clinical trial of 200 patients, the rate of progression to invasive mechanical ventilation, extracorporeal membrane oxygenation, or death by day 28 was 17.2% with pacritinib vs 22.8% with placebo. Among patients with elevated interleukin 6, the rate was 17.5% vs 30.4%.
Meaning
Pacritinib did not demonstrate a significant benefit over placebo in patients with severe COVID-19.
This randomized clinical trial evaluates the efficacy and safety of pacritinib vs placebo in the treatment of adults with severe COVID-19.
Abstract
Importance
The morbidity and mortality associated with COVID-19 remain high despite advances in standard of care therapy, and the role of anti-inflammatory agents that inhibit the interleukin 6/JAK2 pathway is still being elucidated.
Objective
To evaluate the efficacy and safety of the oral JAK2/IRAK1 inhibitor pacritinib vs placebo in the treatment of adults with severe COVID-19.
Design, Setting, and Participants
This phase 2, double-blind, placebo-controlled, randomized clinical trial enrolled hospitalized adult patients with severe COVID-19 at 21 centers across the US between June 2020 and February 2021, with approximately 1.5 months of safety follow-up per patient. Data analysis was performed from September 2021 to July 2022.
Interventions
Patients were randomized 1:1 to standard of care plus pacritinib (400 mg per os on day 1 followed by 200 mg twice daily on days 2-14) vs placebo, for 14 days.
Main Outcomes and Measures
The primary end point was death or need for invasive mechanical ventilation (IMV) or extracorporeal membrane oxygenation (ECMO) by day 28. All-cause mortality and safety were also assessed.
Results
A total of 200 patients were randomized to pacritinib (99 patients; 56 men [56.6%]; median [range] age, 60 [19-87] years) or placebo (101 patients; 64 men [63.4%]; median [range] age 59 [28-94] years). The percentage requiring supplementary oxygen was 99.0% (98 patients) in the pacritinib group vs 98.0% (99 patients) in the placebo group. The percentage who progressed to IMV, ECMO, or death was 17.2% (17 patients) in the pacritinib group vs 22.8% (23 patients) in the placebo group (odds ratio, 0.62; 95% CI, 0.28-1.35; P = .23). Among patients with elevated interleukin 6, the rate was 17.5% (11 of 63 patients) in the pacritinib group vs 30.4% (21 of 96 patients) in the placebo group. The adverse event rate was similar for pacritinib vs placebo (78.1% [75 patients] vs 80.2% [81 patients]), with no excess in infection (14.6% [14 patients] vs 19.8% [20 patients]), bleeding (8.3% [8 patients] vs 10.9% [11 patients]), or thrombosis (8.3% [8 patients] vs 7.9% [8 patients]). Rates of grade 3 or higher adverse events were lower with pacritinib than placebo (29.2% [28 patients] vs 40.6% [41 patients]).
Conclusions and Relevance
The study did not meet its primary end point in patients with severe COVID-19. Subgroup analyses may indicate specific populations with hyperinflammation that could benefit from pacritinib, although further clinical trials would be needed to confirm these effects.
Trial Registration
ClinicalTrials.gov Identifier: NCT04404361
Introduction
COVID-19, a respiratory disease caused by the novel coronavirus SARS-CoV-2, has led to substantial morbidity and mortality worldwide.1,2 COVID-19 can lead to the development of acute respiratory distress syndrome, a hyperinflammatory state that can lead to multiorgan dysfunction.3 Acute respiratory distress syndrome is mediated by inflammatory cell infiltration in the lung, thought to be associated with interleukin (IL)–6-induced differentiation of T helper 17 cells.4,5,6,7 When the PRE-VENT study (Phase 3 Randomized, Double-Blind, Placebo-Controlled, Multicenter Study of Pacritinib Plus Standard of Care Versus Placebo and Standard of Care in Hospitalized Patients With Severe COVID-19 With or Without Cancer) was initiated, there were no studies showing benefit to any anti-inflammatory therapy in COVID-19. Despite advances in treatment, including immunomodulatory strategies, preventing disease progression in hospitalized patients with COVID-19 remains an unmet need.
Pacritinib, a selective JAK2/IRAK1/CSF1R inhibitor,8 is approved by the US Food and Drug Administration for the treatment of patients with myelofibrosis who have a platelet count less than 50 ×109/L.9,10,11 Myelofibrosis is a clonal hematopoietic stem cell malignant neoplasm, with pathophysiology caused by the production of inflammatory cytokines.12 As a JAK2 and IRAK1 inhibitor, pacritinib decreases levels of soluble IL-17A and soluble IL-6 in human primary cell systems13 but does not have inhibitory activity to JAK1.8 This lack of JAK1 effect is potentially beneficial in the setting of both malignancy and viral disease, because JAK1 is responsible for differentiation and activity of natural killer cells,14 which contribute to innate antitumoral and antiviral responses.15
Since the initiation of the PRE-VENT study in May 2020, immunomodulatory therapy has been shown to improve outcomes in patients with severe COVID-19, including treatment with glucocorticoids,16,17 the IL-6 receptor inhibitor tocilizumab,18,19,20 and the JAK1/2 inhibitor baricitinib.19 Data for the JAK inhibitor class have been mixed. Baricitinib received Emergency Use Authorization (EUA) in the US on the basis of 2 large, placebo-controlled trials showing benefit in the percentage of hospitalized patients progressing to mechanical ventilation21 or death.22 Similarly, the JAK1/3 inhibitor tofacitinib reduced the composite end point of respiratory failure or death compared with placebo.23 By contrast, the JAK1/2 inhibitor ruxolitinib was not associated with clinical benefit in patients with severe COVID-19.24 The PRE-VENT study was designed to evaluate the efficacy and safety of pacritinib in combination with standard of care therapy, including corticosteroids, for the treatment of adults hospitalized with severe COVID-19.
Methods
Study Design and Participants
PRE-VENT is a phase 2, randomized, double-blind, placebo-controlled clinical trial that included 21 centers from the US. Eligible participants were adults with severe COVID-19, defined as confirmed disease in patients hospitalized with at least 1 of the following criteria but not requiring invasive mechanical ventilation (IMV): hypoxemia (oxygen saturation ≤93% on ambient air), respiratory rate greater than 30 breaths per minute, or ratio of arterial oxygen partial pressure to fraction of inspired oxygen less than 300. The full list of inclusion and exclusion criteria is found in the study protocol (Supplement 1).
PRE-VENT was performed in accordance with ethical principles of the Declaration of Helsinki25 and Good Clinical Practice Guidelines. Institutional review board approval was obtained at all sites, and all participants or legally authorized representatives provided written informed consent. This study follows the Consolidated Standards of Reporting Trials (CONSORT) reporting guideline.
Randomization and Blinding
Participants were centrally randomized 1:1 to receive pacritinib plus standard of care or placebo plus standard of care according to a permuted block design stratified by age (<60 vs ≥60 years) and baseline Ordinal Scale Score (OSS; 3 or 4 vs 5). The OSS is a 7-point assessment of clinical status, with scoring as follows: 1, not hospitalized with resumption of normal activities; 2, not hospitalized but unable to resume normal activities; 3, hospitalized but not requiring supplemental oxygen; 4, hospitalized requiring supplemental oxygen but not meeting criteria for categories 5 or 6; 5, hospitalized on noninvasive positive pressure ventilation or high-flow nasal cannula; 6, hospitalized requiring IMV and/or extracorporeal membrane oxygenation (ECMO); and 7, death.26
Participants, study staff, investigators, and study sponsor were blinded to treatment assignment. An independent data monitoring committee evaluated safety and efficacy data blinded to treatment group during the trial. Information on participant sex, race (American Indian or Alaska Native, Asian, Black or African American, Native Hawaiian or Pacific Islander, White, multiracial, not reported, or unknown), and ethnicity (Hispanic or Latino) was obtained through self-report captured in the electronic medical record in accordance with US Food and Drug Administration guidance.27 Race and ethnicity data were included given the disparities in presentation and outcome of COVID-19.28
Procedures
Patients receiving both pacritinib and placebo also received standard of care therapy, including corticosteroids. Use of full-dose anticoagulation or antiplatelet agents was prohibited before randomization, although these therapies were permitted during the study as needed; prophylactic-dose or intermediate-dose anticoagulation or antiplatelet agents were permitted throughout the study. The use of IL-6 inhibitors was permitted but required study drug discontinuation. Concomitant use of strong CYP3A4 inhibitors and strong CYP450 inducers was prohibited, as was use of cancer-directed cytotoxic or immunomodulatory therapy.
Patients in the pacritinib group received a single dose of 400 mg of pacritinib on study day 1 followed by 200 mg twice daily on days 2 through 14; patients in the placebo group received placebo capsules in a similar fashion. Assigned treatment could be continued for an additional 7 days according to physician discretion. Patients continued study drug regardless of whether they remained hospitalized through day 14. Safety assessments included physical examinations, laboratory studies, and electrocardiogram monitoring. The schedule of assessments is outlined in eFigure 1 in Supplement 2. After hospital discharge, monitoring included weekly phone calls to assess OSS and safety.
Outcomes
The primary end point was the proportion of participants who progressed to IMV, ECMO, or death by day 28 in each treatment group. Secondary end points included 15-day and 28-day mortality, ventilator-free days, time to improvement by at least 2 points on the OSS, and use of immunomodulatory agents. Biomarkers were analyzed as prespecified tertiary end points. All assessments that were not prespecified are reported as exploratory. Adverse events (AEs) were documented from randomization through 30 days following the last dose of study drug.
Statistical Analysis
Data analysis was performed from September 2021 to July 2022. The original version of the protocol specified a sample size of 364 patients for this phase 3 study with a planned interim analysis after 200 patients had enrolled and reached the 28-day end point. Because of a decrease in COVID-19 cases at PRE-VENT study sites, before unblinding, the study design was amended to a phase 2 study with a sample size based on a 1-sided α level of .10, absolute difference in effect size of 13%, and 80% power with 200 patients. Efficacy data were analyzed in the intention-to-treat population (all randomized patients). Safety data were analyzed in the safety population (all treated patients). Statistical analysis was done using SAS statistical software version 9.4 (SAS Institute).
The analysis for the primary end point was performed using a last observation carried forward imputation approach for handling missing data. Sensitivity analysis was performed using multiple imputation method, wherein missing values were imputed using a missing-at-random method with 20 replications, including the randomization stratification factors, baseline and day-14 clinical status assessment.
The Cochran-Mantel-Haenszel test stratified by baseline age (<60 vs ≥60 years) and OSS (3 or 4 vs 5) was used to compare the proportion of patients with progression to IMV and/or ECMO or death and the mortality rates between treatment groups. Odds ratios (ORs) and associated 95% CIs were estimated via stratified Cochran-Mantel-Haenszel test. Kaplan-Meier methods were used to estimate median time to clinical improvement. The hazard ratio of the time to IMV, ECMO, or death by treatment group was estimated using a Cox proportional hazards model stratified by randomization factors (age and OSS). The number of days requiring mechanical ventilation was compared by treatment group using the Wilcoxon rank sum test. Subgroup analyses were performed using similar methods as the primary analysis. Statistical significance was set at 2-sided P < .05.
The statistical analysis plan for this study indicated that patients who discontinued study follow-up while inpatients and before day 26 would be considered to have met the primary end point. Results of this planned primary end point analysis are reported in eTable 1 in Supplement 2.
Results
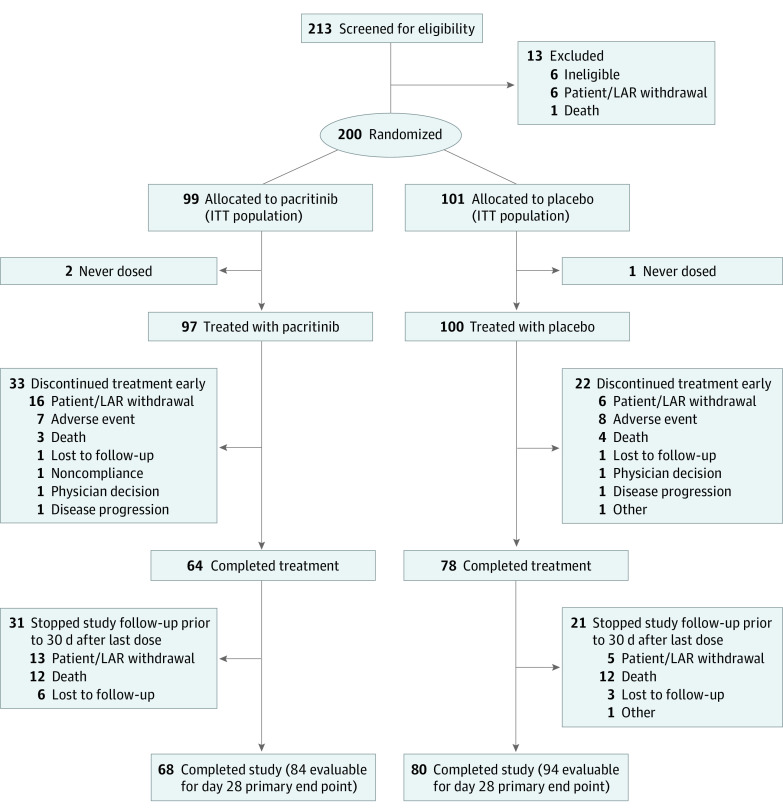
Among 200 patients randomized (99 to pacritinib and 101 to placebo) between June 2020 and February 2021, baseline characteristics were similar between treatment groups (Table 1), including median (range) age (60 [19-87] years for pacritinib and 59 [28-94] years for placebo), sex (56 men [56.6%] in the pacritinib group and 64 men [63.4%] in the placebo group), and percentage of patients with OSS of 5 (50.5% [50 patients] in the pacritinib group and 46.5% [47 patients] in the placebo group). The percentage requiring supplementary oxygen was 99.0% (98 patients) in the pacritinib group vs 98.0% (99 patients) in the placebo group. A total of 197 patients were treated with the study drug (Figure 1), with a median (IQR) duration of treatment of 14 (9-14) days in the pacritinib group and 14 (14-14) days in the placebo group. The most common medical comorbidities among randomized patients were hypertension (114 patients [57.0%]) and diabetes (80 patients [40.0%]). At baseline, most randomized patients had elevated levels of inflammatory markers, including IL-6, ferritin, C-reactive protein, and D-dimer.
Table 1. Baseline Patient Characteristics.
| Characteristic | Patients, No. (%) | |
|---|---|---|
| Pacritinib (n = 99) | Placebo (n = 101) | |
| Age, median (range), y | 60 (19-87) | 59 (28-94) |
| Sex | ||
| Male | 56 (56.6) | 64 (63.4) |
| Female | 43 (43.4) | 37 (36.6) |
| Racea | ||
| American Indian or Alaska Native | 0 | 2 (2.0) |
| Asian | 2 (2.0) | 2 (2.0) |
| Black or African American | 17 (17.2) | 19 (18.8) |
| Native Hawaiian or Pacific Islander | 1 (1.0) | 0 |
| White | 74 (74.7) | 72 (71.3) |
| Other | 1 (1.0) | 3 (3.0) |
| Not reported or unknown | 4 (4.0) | 3 (3.0) |
| Ethnicitya | ||
| Hispanic or Latino | 13 (13.1) | 14 (13.9) |
| Not Hispanic or Latino | 79 (79.8) | 81 (80.2) |
| Not reported or unknown | 7 (7.1) | 6 (6.0) |
| Comorbidities | ||
| Chronic lung disease | 20 (20.2) | 21 (20.8) |
| Diabetes | 41 (41.4) | 39 (38.6) |
| Cardiovascular disease | 9 (9.1) | 20 (19.8) |
| Hypertension | 53 (53.5) | 61 (60.4) |
| Chronic renal disease | 7 (7.1) | 6 (5.9) |
| Chronic liver disease | 4 (4.0) | 2 (2.0) |
| Immunocompromised state | 4 (4.0) | 2 (2.0) |
| Cancer | 11 (11.1) | 12 (11.9) |
| Neurologic disorder or stroke | 4 (4.0) | 7 (6.9) |
| Body mass index, median (IQR)b | 32.2 (28.6-37.6) | 32.7 (28.4-36.5) |
| Ordinal Scale Score | ||
| 3 | 1 (1.0) | 2 (2.0) |
| 4 | 48 (48.5) | 51 (50.5) |
| 5 | 50 (50.5) | 47 (46.5) |
| 6 | 0 | 1 (1.0)c |
| Inflammatory markers, median (IQR) | ||
| C-reactive protein, mg/dL | 7.8 (4.6-11.9) | 7.8 (4.8-16.0) |
| D-dimer, μg/mL | 0.8 (0.5-1.4) | 0.8 (0.5-1.7) |
| Ferritin, ng/mL | 690 (379-1386) | 901 (447-1527) |
| Interleukin 6, pg/mL | 8.5 (3.2-21.0) | 7.0 (3.8-18.0) |
| Lactate dehydrogenase, IU/L | 438 (323-570) | 504 (371-763) |
| Procalcitonin, ng/mL | 0.10 (0.07-0.23) | 0.11 (0.06-0.19) |
| Troponin I, ng/mL | 0.02 (0.01-0.04) | 0.01 (0.01-0.02) |
SI conversion factors: To convert C-reactive protein to milligrams per liter, multiply by 10; D-dimer to nanomoles per liter, multiply by 5.476; ferritin to micrograms per liter, multiply by 1; lactate dehydrogenase to nanomoles per liter, multiply by 0.111; troponin I to micrograms per liter, multiply by 1.
Data on race and ethnicity were captured according to patient self-reported data captured in the electronic medical record. All categories of race and ethnicity are shown in the table. The other category may include individuals where race is described as multiracial. Race and ethnicity data were included given disparities in presentation and outcome of COVID-19.28
Body mass index is calculated as weight in kilograms divided by height in meters squared.
One patient was intubated between randomization and first dose.
Figure 1. Study Flow Diagram.
Diagram shows the flow of patients from screening through study completion, defined as 30 days after the last dose of study drug. The number of patients evaluable for the primary end point (with a day 28 Ordinal Scale Score) are also shown. There were 15 patients randomized to pacritinib and 8 randomized to placebo who did not have an Ordinal Scale Score documented at day 28. The safety population included 197 patients (by actual treatment group, distribution was 96 in the pacritinib group and 101 in the placebo group since 1 patient randomized to pacritinib received placebo). ITT indicates intention to treat; LAR, legal authorized representative.
Although the percentage of patients who discontinued study treatment prematurely because of AEs, death, or physician decision was similar in the pacritinib and placebo groups, a greater percentage of patients randomized to pacritinib stopped study treatment prematurely because of patient or legal representative withdrawal of consent (16 patients [16.2%] vs 6 patients [5.9%]). Similarly, a greater percentage of patients in the pacritinib group discontinued all participation in the study, including safety follow-up through 30 days after the last dose, compared with the placebo group (19 patients [19.2%] vs 9 patients [8.9%]), resulting in an imbalance between the pacritinib and placebo groups in patients with an evaluable day 28 OSS (84.8% [84 of 99 patients] vs 93.1% [94 of 101 patients]) (Figure 1). The imbalance in study follow-up was largely associated with withdrawal owing to patient decision (13 patients [13.1%] vs 5 patients [5.0%]) and loss to follow-up (6 patients [6.1%] vs 3 patients [3.0%]). No patient who withdrew consent for follow-up had worsening OSS at the time of withdrawal, few had symptomatic AEs, and none had high-grade or serious AEs before withdrawal. Most AEs reported in patients taking pacritinib who withdrew from follow-up were asymptomatic, grade 1 laboratory abnormalities. Only 4 of the 15 patients taking pacritinib who withdrew early had potentially symptomatic AEs. These included 1 patient with blurry vision (grade 1); 1 with palpitations (grade 1); 1 with diarrhea, dizziness, and nausea (all grade 1); and 1 with anxiety (grade 2). A total of 6 patients reported no treatment-emergent AEs. Patients who withdrew from the study in the pacritinib group largely did so while they were still hospital inpatients (9 of 15 patients), and the median time of withdrawal was early in the study (day 5). By contrast, patients who withdrew from the study in the placebo group largely did so after having been discharged from the hospital (6 of 8 patients).
Primary Efficacy End Point
The percentage of patients who required IMV or ECMO, or who died by day 28 was 17.2% (17 patients) in the pacritinib group vs 22.8% (23 patients) in the placebo group (OR, 0.62; 95% CI, 0.28 to 1.35; P = .23). The absolute risk reduction between pacritinib and placebo was 5.6% (95% CI, −5.6% to 16.5%). A sensitivity analysis based on multiple imputation yielded similar results (17.3% vs 23.0%; OR, 0.62; 95% CI, 0.28 to 1.35; P = .23). Preplanned exploratory subgroup analysis showed a benefit associated with pacritinib among patients with elevated baseline IL-6 levels, based on progression to the primary end point for pacritinib (17.5% [11 of 63 patients]) vs placebo (30.4% [21 of 69 patients]) (OR, 0.36; 95% CI, 0.14 to 0.94; P = .04). Similarly, response rates favored the pacritinib group among patients with baseline elevations in C-reactive protein, D-dimer, and lactate dehydrogenase (Figure 2).
Figure 2. Subgroup Analyses of Primary End Point.
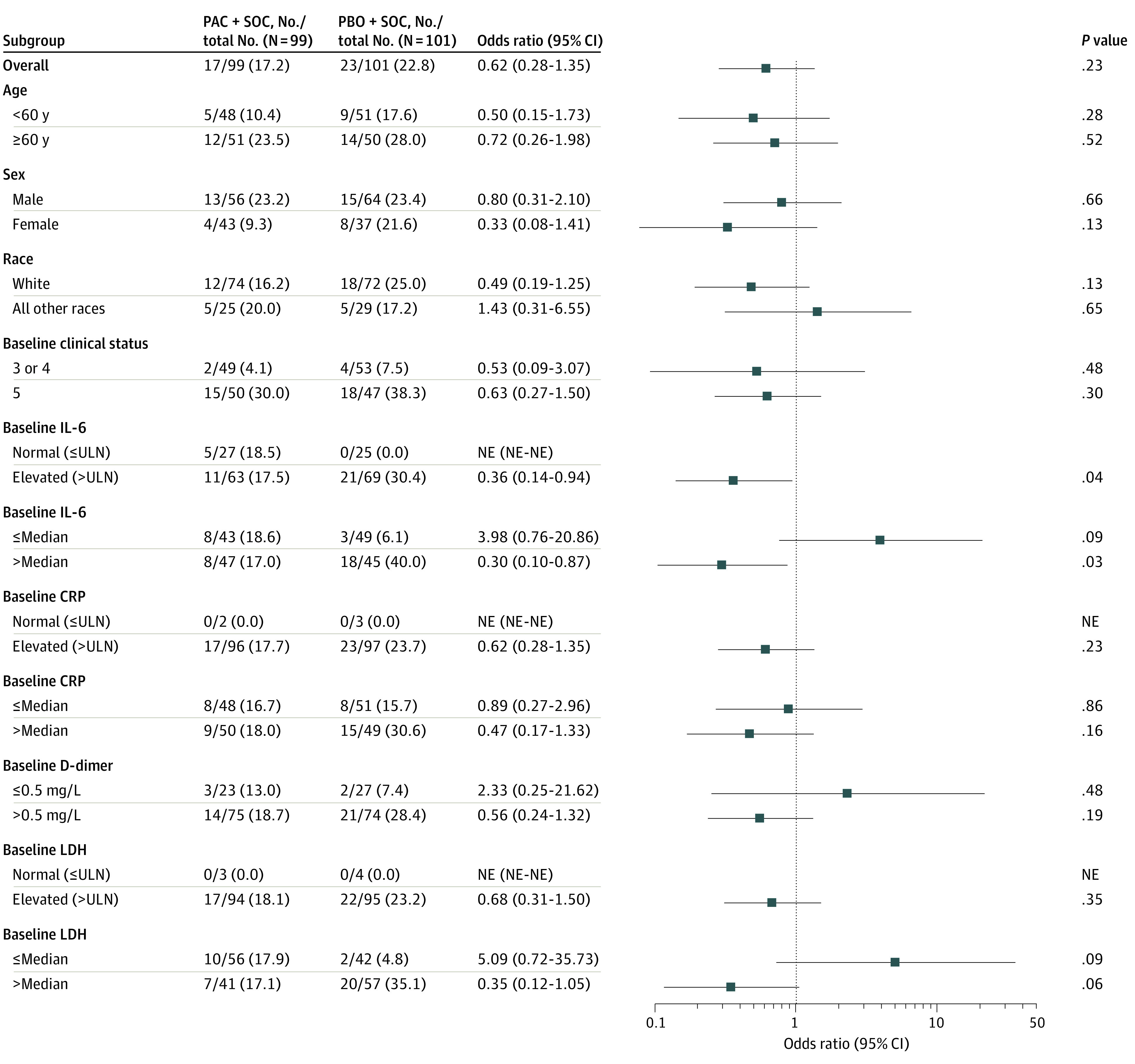
Odds ratios for invasive mechanical ventilation, extracorporeal membrane oxygenation, or death by day 28 in the intention-to-treat population are shown. Odds ratios are the Cochran-Mantel Haenszel odds ratios of the end point in the pacritinib (PAC) compared with the placebo (PBO) group. Ordinal Scale Score is represented as clinical status. CRP indicates C-reactive protein; IL-6, interleukin 6; LDH, lactate dehydrogenase; NE, not evaluable; SOC, standard of care; ULN, upper limit of normal.
Secondary and Tertiary Efficacy End Points
There was no difference in the percentage of patients who died by day 28 in the pacritinib vs placebo groups (10.1% [10 patients] vs 7.9% [8 patients]; OR, 1.29; 95% CI, 0.46-3.58; P = .63). The mean (SD) number of days requiring mechanical ventilation was 3.6 (8.3) in the pacritinib group and 4.5 (8.8) in the placebo group (median [IQR], 0.0 [0.0-0.0] days for both groups). The percentage of patients who experienced clinical improvement at any time during study (decrease in OSS of at least 2 points) was 68.7% (68 patients) in the pacritinib group and 73.3% (74 patients) in the placebo group. The median time to clinical improvement was 9.0 days (95% CI, 8.0-10.0 days) in the pacritinib group and 9.0 days (95% CI, 7.0-11.0 days) in the placebo group (Table 2). The use of immunomodulatory agents was high in both groups (97 patients [98.0%] in the pacritinib group vs 96 patients [95.0%] in the placebo group) (eTable 2 in Supplement 2), because the standard of care had shifted to include corticosteroids shortly after study initiation. There were no significant differences in inflammatory biomarker trends over time (eFigure 2 and eFigure 3 in Supplement 2). Among patients who remained hospitalized, median levels of IL-6 decreased by 29% in the pacritinib group and by 0% in the placebo group at day 3, although this difference was not statistically significant. By the next assessment on day 8, most patients had either been discharged from the hospital or were no longer enrolled in the study.
Table 2. Key Secondary Outcomes (Intention-to-Treat Population).
| Outcome | Patients, No. (%) | Comparison, OR (95% CI) | P value | |
|---|---|---|---|---|
| Pacritinib (n = 99) | Placebo (n = 101) | |||
| Death by day 28 | 10 (10.1) | 8 (7.9) | 1.29 (0.46-3.58) | .63 |
| Death by day 15 | 5 (5.1) | 4 (4.0) | 1.25 (0.31-4.96) | .75 |
| Ventilator-free days through day 28, mean (SD)a | 22.10 (10.20) | 22.60 (9.35) | NA | .75 |
| Time on a ventilator, db | ||||
| Mean (SD) | 3.6 (8.4) | 4.5 (8.8) | NA | .39 |
| Median (IQR) | 0.0 (0.0-0.0) | 0.0 (0.0-0.0) | NA | |
| Improvement by at least 2 points relative to baseline | 68 (68.7) | 74 (73.3) | NA | .51 |
| Time to improvement by at least 2 points, median (95% CI), d | 9.0 (8.0-10.0) | 9.0 (7.0-11.0) | 1.09 (0.79-1.53)c | .57 |
| Use of immunomodulatory agentsd | 97 (98.0) | 96 (95.0) | NA | .27 |
Abbreviations: NA, not applicable; OR, odds ratio.
Refers to number of days patient has Ordinal Scale Score less than 6. Days with missing data are not counted toward ventilator-free days.
Refers to number of days patient has Ordinal Scale Score 6; in case of death, patient carried forward as being on ventilator through day 28.
Data are hazard ratio (95% CI).
Includes corticosteroids and interleukin 6 inhibitors.
Safety
Most patients in both groups experienced at least 1 AE during treatment (pacritinib vs placebo, 78.1% [75 patients] vs 80.2% [81 patients]), although the rate of serious and high-grade AEs was lower in the pacritinib group compared with the placebo group (29.2% [28 patients] vs 40.6% [41 patients]) (Table 3). The rates of cardiac events (33.3% [32 patients] vs 32.7% [33 patients]), bleeding (8.3% [8 patients] vs 10.9% [11 patients]), gastrointestinal events (31.3% [30 patients] vs 29.7% [30 patients]), and infection (14.6% [14 patients] vs 19.8% [20 patients]) were similar in both groups. The rate of diarrhea was higher in the pacritinib group compared with the placebo group (12.5% [12 patients] vs 4.0% [4 patients]). Diarrhea in the pacritinib group was exclusively grade 1 to 2, with a median onset of 2 days. Diarrhea resolved in 83.3% of patients (10 of 12 patients), with a median duration of 8 days. Infection events occurred at similar rates on pacritinib and placebo; the most common across both groups were septic shock (3.1% [3 patients] vs 5.9% [6 patients]) and pneumonia (3.1% [3 patients] vs 3.0% [3 patients]). Thromboses occurred at similar rates in both groups (8.3% [8 patients] vs 7.9% [8 patients]) and consisted mostly of venous thromboses and thromboembolism; only 1 case of arterial thrombosis was noted in the pacritinib group. A list of overall and high-grade AEs is provided in eTable 3 in Supplement 2.
Table 3. AE Overview in the Safety Population.
| Event | Patients, No. (%) | |
|---|---|---|
| Pacritinib (n = 96) | Placebo (n = 101) | |
| Treatment-emergent AEs | 75 (78.1) | 81 (80.2) |
| Cardiac AEsa | 32 (33.3) | 33 (32.7) |
| Hemorrhage AEsa | 8 (8.3) | 11 (10.9) |
| Gastrointestinal disordersb | 30 (31.3) | 30 (29.7) |
| Infection AEsb | 14 (14.6) | 20 (19.8) |
| Thrombosisc | 8 (8.3) | 8 (7.9) |
| Major cardiac AEsd | 3 (3.1) | 3 (3.0) |
| AEs related to study treatment | 26 (27.1) | 29 (28.7) |
| Serious AEs | 20 (20.8) | 33 (32.7) |
| Grade ≥3 AEs | 28 (29.5) | 41 (40.6) |
| Fatal AEs | 12 (12.5) | 12 (11.9) |
| AEs leading to treatment discontinuation | 9 (9.4) | 12 (11.9) |
| AEs leading to treatment interruption | 8 (8.3) | 5 (5.0) |
| AEs leading to dose reduction | 2 (2.1) | 1 (1.0) |
Abbreviation: AE, adverse event.
Determined by Standardised Medical Dictionary for Regulatory Activities Query.
Determined by System Organ Class, Medical Dictionary for Regulatory Activities version 23.1.
Includes venous and arterial thrombosis as well as embolic disease, including pulmonary and cerebral embolism.
Major adverse cardiac events include death due to a cardiac event, ischemic stroke, and myocardial infarction.
Fatal AEs occurred in 12 patients in each study group. Among pacritinib-treated patients, 9 deaths were attributed to respiratory failure, 2 to cardiopulmonary arrest, and 1 to embolic stroke. Among placebo-treated patients, 10 deaths were attributed to respiratory failure and 2 to cardiac arrest. No deaths were considered to be related to study drug.
Discussion
PRE-VENT was a phase 2 randomized clinical trial evaluating the safety and efficacy of pacritinib plus standard of care compared with placebo plus standard of care in patients with severe COVID-19. Although the study did not meet its primary objective, a strong signal for benefit was noted in the prespecified exploratory subgroups of patients with increased levels of inflammatory markers at baseline, particularly IL-6. The safety profile of pacritinib in patients with severe COVID-19 was acceptable, as there was no increased risk noted for specific toxic effects associated with the JAK inhibitor class, including infection, bleeding, thrombotic events, or major cardiac AEs.
The effect size of pacritinib compared with placebo in prevention of IMV, ECMO, or death in the overall population (OR, 0.62) was comparable to the effect size for baricitinib plus remdesivir in prevention of IMV or ECMO in the ACTT-2 (A Multicenter, Adaptive, Randomized Blinded Controlled Trial of the Safety and Efficacy of Investigational Therapeutics for the Treatment of COVID-19 in Hospitalized Adults) study (OR, 0.61)21 or baricitinib in prevention of high-flow oxygen, noninvasive positive pressure ventilation, IMV, ECMO, or death in the COV-BARRIER (A Randomized, Double-Blind, Placebo-Controlled, Parallel-Group Phase 3 Study of Baricitinib in Patients With COVID-19 Infection) study (OR, 0.85).22 Both ACTT-2 and COV-BARRIER were adequately powered phase 3 confirmatory trials, with a total sample size of more than 1000 patients. PRE-VENT, by contrast, was designed as a phase 2 study, and although the results are encouraging, the study was not powered to detect the observed effect size.
Since April 2020, when PRE-VENT was designed, standard of care therapy for severe COVID-19 has evolved rapidly. Almost all patients enrolled in the PRE-VENT trial received corticosteroids and prophylactic-dose or intermediate-dose anticoagulation as part of standard therapy. In November 2020, approximately 4 months before study closure, the JAK1/2 inhibitor baricitinib was approved, in combination with remdesivir, under EUA in the US. In June 2021, approximately 4 months after study closure, the IL-6 inhibitor tocilizumab received an EUA as well. The success of these agents, both of which target the IL-6 signaling pathway, support the PRE-VENT finding of an observed effect of pacritinib in the preplanned exploratory subgroup of patients with baseline IL-6 elevation.
After the completion of PRE-VENT, pacritinib was approved in the US for treatment of patients with myelofibrosis who have severe thrombocytopenia. Patients with hematologic malignant neoplasms, particularly those with myeloid neoplasms such as myelofibrosis, are at increased risk of severe complications of COVID-19.29
Limitations
Limitations of this study should be noted. First, because of a reduction in the number of cases at PRE-VENT study sites between the initial waves and the Delta wave of COVID, the study was amended from a larger phase 3 study to a 200-patient phase 2 study, limiting the study’s ability to detect significant differences between pacritinib and placebo. Second, the study was heavily impacted by challenges in patient retention. Pacritinib has primarily been studied in the outpatient oncology setting, where patient retention is reinforced by long-term relationships with medical practitioners and, often, by shared decision-making with the support of loved ones. In the acute COVID-19 setting, by contrast, the physician-patient relationship was negatively impacted by the patient’s acuity of illness and physical isolation, and decisions about trial enrollment had to be made in the context of policies that restricted family visitation. The investigators also note that this study was conducted early during the pandemic, at a time when the scientific community’s understanding of the disease was nascent and when misinformation about disease severity and treatment was rampant. All of these factors, combined with the uncertainties and unprecedented challenges posed by the pandemic, may have contributed to higher-than-expected attrition and missing data, which impact assessments of both efficacy and safety.
Conclusions
In this study, pacritinib did not yield a significant benefit over placebo in patients with severe COVID-19 in the PRE-VENT phase 2 randomized clinical trial. Like most interventional COVID-19 studies initiated early in the pandemic, PRE-VENT was designed in the absence of early phase data. It is possible that a study selecting for patients with pathological activation of the IL-6/JAK/STAT pathway would have shown a larger effect, and future studies would be needed to confirm the efficacy signal for pacritinib in patients with severe COVID-19 who have elevated IL-6 levels. The PRE-VENT data set provides important safety information on the use of pacritinib in patients with severe COVID-19.
Trial Protocol
eFigure 1. Study Schema
eTable 1. Endpoint Analysis Using Prespecified Conservative Approach
eTable 2. Use of Immunomodulatory and Antiviral Therapies at Any Point on Study (Intention-to-Treat Population)
eFigure 2. Time to Improvement in Clinical Status (Intention-to-Treat Population)
eFigure 3. Median Percent Change in Biomarkers on Study
eTable 3. Adverse Events (AEs) by Treatment Group in the Safety Population
Data Sharing Statement
References
- 1.Piroth L, Cottenet J, Mariet AS, et al. Comparison of the characteristics, morbidity, and mortality of COVID-19 and seasonal influenza: a nationwide, population-based retrospective cohort study. Lancet Respir Med. 2021;9(3):251-259. doi: 10.1016/S2213-2600(20)30527-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Reif J, Heun-Johnson H, Tysinger B, Lakdawalla D. Measuring the COVID-19 mortality burden in the United States: a microsimulation study. Ann Intern Med. 2021;174(12):1700-1709. doi: 10.7326/M21-2239 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Ranjeva S, Pinciroli R, Hodell E, et al. Identifying clinical and biochemical phenotypes in acute respiratory distress syndrome secondary to coronavirus disease-2019. EClinicalMedicine. 2021;34:100829. doi: 10.1016/j.eclinm.2021.100829 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Yu ZX, Ji MS, Yan J, et al. The ratio of Th17/Treg cells as a risk indicator in early acute respiratory distress syndrome. Crit Care. 2015;19:82. doi: 10.1186/s13054-015-0811-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Mikacenic C, Hansen EE, Radella F, Gharib SA, Stapleton RD, Wurfel MM. Interleukin-17A is associated with alveolar inflammation and poor outcomes in acute respiratory distress syndrome. Crit Care Med. 2016;44(3):496-502. doi: 10.1097/CCM.0000000000001409 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Li JT, Melton AC, Su G, et al. Unexpected role for adaptive αβTh17 cells in acute respiratory distress syndrome. J Immunol. 2015;195(1):87-95. doi: 10.4049/jimmunol.1500054 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Xu Z, Shi L, Wang Y, et al. Pathological findings of COVID-19 associated with acute respiratory distress syndrome. Lancet Respir Med. 2020;8(4):420-422. doi: 10.1016/S2213-2600(20)30076-X [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Singer JW, Al-Fayoumi S, Ma H, Komrokji RS, Mesa R, Verstovsek S. Comprehensive kinase profile of pacritinib, a nonmyelosuppressive Janus kinase 2 inhibitor. J Exp Pharmacol. 2016;8:11-19. doi: 10.2147/JEP.S110702 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Mesa RA, Vannucchi AM, Mead A, et al. Pacritinib versus best available therapy for the treatment of myelofibrosis irrespective of baseline cytopenias (PERSIST-1): an international, randomised, phase 3 trial. Lancet Haematol. 2017;4(5):e225-e236. doi: 10.1016/S2352-3026(17)30027-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Mascarenhas J, Hoffman R, Talpaz M, et al. Pacritinib vs best available therapy, including ruxolitinib, in patients with myelofibrosis: a randomized clinical trial. JAMA Oncol. 2018;4(5):652-659. doi: 10.1001/jamaoncol.2017.5818 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Gerds AT, Savona MR, Scott BL, et al. Determining the recommended dose of pacritinib: results from the PAC203 dose-finding trial in advanced myelofibrosis. Blood Adv. 2020;4(22):5825-5835. doi: 10.1182/bloodadvances.2020003314 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Fisher DAC, Fowles JS, Zhou A, Oh ST. Inflammatory pathophysiology as a contributor to myeloproliferative neoplasms. Front Immunol. 2021;12:683401. doi: 10.3389/fimmu.2021.683401 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Singer JW, Al-Fayoumi S, Taylor J, Velichko S, O’Mahony A. Comparative phenotypic profiling of the JAK2 inhibitors ruxolitinib, fedratinib, momelotinib, and pacritinib reveals distinct mechanistic signatures. PLoS One. 2019;14(9):e0222944. doi: 10.1371/journal.pone.0222944 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Witalisz-Siepracka A, Klein K, Prinz D, et al. Loss of JAK1 drives innate immune deficiency. Front Immunol. 2019;9:3108. doi: 10.3389/fimmu.2018.03108 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Fleming SB. Viral inhibition of the IFN-induced JAK/STAT signalling pathway: development of live attenuated vaccines by mutation of viral-encoded IFN-antagonists. Vaccines (Basel). 2016;4(3):E23. doi: 10.3390/vaccines4030023 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Sterne JAC, Murthy S, Diaz JV, et al. ; WHO Rapid Evidence Appraisal for COVID-19 Therapies (REACT) Working Group . Association between administration of systemic corticosteroids and mortality among critically ill patients with COVID-19: a meta-analysis. JAMA. 2020;324(13):1330-1341. doi: 10.1001/jama.2020.17023 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Horby P, Lim WS, Emberson JR, et al. ; RECOVERY Collaborative Group . Dexamethasone in hospitalized patients with Covid-19. N Engl J Med. 2021;384(8):693-704. doi: 10.1056/NEJMoa2021436 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.RECOVERY Collaborative Group . Tocilizumab in patients admitted to hospital with COVID-19 (RECOVERY): a randomised, controlled, open-label, platform trial. Lancet. 2021;397(10285):1637-1645. doi: 10.1016/S0140-6736(21)00676-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Alunno A, Najm A, Mariette X, et al. Immunomodulatory therapies for the treatment of SARS-CoV-2 infection: an update of the systematic literature review to inform EULAR points to consider. RMD Open. 2021;7(3):e001899. doi: 10.1136/rmdopen-2021-001899 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Gordon AC, Mouncey PR, Al-Beidh F, et al. ; REMAP-CAP Investigators . Interleukin-6 receptor antagonists in critically ill patients with Covid-19. N Engl J Med. 2021;384(16):1491-1502. doi: 10.1056/NEJMoa2100433 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Kalil AC, Patterson TF, Mehta AK, et al. ; ACTT-2 Study Group Members . Baricitinib plus remdesivir for hospitalized adults with Covid-19. N Engl J Med. 2021;384(9):795-807. doi: 10.1056/NEJMoa2031994 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Marconi VC, Ramanan AV, de Bono S, et al. ; COV-BARRIER Study Group . Efficacy and safety of baricitinib for the treatment of hospitalised adults with COVID-19 (COV-BARRIER): a randomised, double-blind, parallel-group, placebo-controlled phase 3 trial. Lancet Respir Med. 2021;9(12):1407-1418. doi: 10.1016/S2213-2600(21)00331-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Guimarães PO, Quirk D, Furtado RH, et al. ; STOP-COVID Trial Investigators . Tofacitinib in patients hospitalized with Covid-19 pneumonia. N Engl J Med. 2021;385(5):406-415. doi: 10.1056/NEJMoa2101643 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Han MK, Antila M, Ficker JH, et al. Ruxolitinib in addition to standard of care for the treatment of patients admitted to hospital with COVID-19 (RUXCOVID): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Rheumatol. 2022;4(5):e351-e361. doi: 10.1016/S2665-9913(22)00044-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.World Medical Association . World Medical Association Declaration of Helsinki: ethical principles for medical research involving human subjects. JAMA. 2013;310(20):2191-2194. doi: 10.1001/jama.2013.281053 [DOI] [PubMed] [Google Scholar]
- 26.Cao B, Wang Y, Wen D, et al. A trial of lopinavir-ritonavir in adults hospitalized with severe Covid-19. N Engl J Med. 2020;382(19):1787-1799. doi: 10.1056/NEJMoa2001282 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.US Food and Drug Administration Office of Minority Health . Collection of race and ethnicity data in clinical trials: guidance for industry and Food and Drug Administration staff. October 26, 2016. Accessed October 12, 2022. https://www.fda.gov/media/75453/download
- 28.Magesh S, John D, Li WT, et al. Disparities in COVID-19 outcomes by race, ethnicity, and socioeconomic status: a systematic-review and meta-analysis. JAMA Netw Open. 2021;4(11):e2134147. doi: 10.1001/jamanetworkopen.2021.34147 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Pagano L, Salmanton-García J, Marchesi F, et al. ; EPICOVIDEHA Working Group . COVID-19 infection in adult patients with hematological malignancies: a European Hematology Association Survey (EPICOVIDEHA). J Hematol Oncol. 2021;14(1):168. doi: 10.1186/s13045-021-01177-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Trial Protocol
eFigure 1. Study Schema
eTable 1. Endpoint Analysis Using Prespecified Conservative Approach
eTable 2. Use of Immunomodulatory and Antiviral Therapies at Any Point on Study (Intention-to-Treat Population)
eFigure 2. Time to Improvement in Clinical Status (Intention-to-Treat Population)
eFigure 3. Median Percent Change in Biomarkers on Study
eTable 3. Adverse Events (AEs) by Treatment Group in the Safety Population
Data Sharing Statement