Abstract
Although the host immune response to the 60-kDa chlamydial heat shock protein (hsp60) has been implicated in trachoma pathogenesis, no studies have examined mucosal immune responses to hsp60 in populations for which chlamydia is endemic. Tears and sera from Nepali villagers were reacted against hsp60 fusion proteins, whole hsp60, and the major outer membrane protein (MOMP). Tears from villagers without disease were anti-hsp60 immunoglobulin G (IgG) reactive in 6 (38%) of 16 villagers compared with 36 (90%) of 40 with follicular trachoma (TF) (P < 0.001); 47 (89%) of 53 with inflammatory trachoma (TI) (P < 0.001); and 31 (84%) of 37 with conjunctival scarring (TS) (P = 0.002). By multivariate analysis, odds ratios for tear hsp60 IgG immunoreactivity in villagers with TF, TI, and TS were 49.2 (confidence interval [CI], 2.7 to 898), 22.6 (CI, 3 to 170), and 13.6 (CI, 1.4 to 133), respectively. There were no significant differences for tear IgA or secretory IgA (sIgA) reactivity to hsp60 or for tear sIgA and IgG reactivity to MOMP. Serum anti-hsp60 IgG immunoreactivity was associated with TI only. These data suggest that anti-hsp60 IgG immunoreactivity represents largely locally derived antibodies, which may promote disease pathology. In contrast, nonspecific high rates of anti-hsp60 sIgA antibodies suggest chronic or repeat stimulation from an endemic source of organisms.
Trachoma is a chronic follicular conjunctivitis caused by infection of the conjunctival mucosa with the obligate intracellular pathogen Chlamydia trachomatis. This disease represents the leading cause of preventable blindness worldwide (4). While acute chlamydial ocular infections are often self-limiting, persistent or repeat infections can result in conjunctival scarring, eyelid deformity, and blindness.
The host immune response has been implicated in the pathogenesis of chlamydial disease. The chlamydial 60-kDa heat shock protein (hsp60) is thought to be a major target antigen that stimulates a pathogenic inflammatory response (15). hsp60 is a member of a family of stress response proteins that are produced by cells in response to a variety of insults. The heat shock response has been observed in every cell examined to date, and the protein is among the most conserved proteins known, with respect to both structure and function (11). In addition, heat shock proteins have been demonstrated to be important antigens in eliciting a deleterious host immune response in infections with helminthes, protozoa, and bacteria (11).
Chlamydial hsp60 has been associated with a pathogenic immune response in animal models and among patients with chlamydial genital tract infections and trachoma. In the monkey “pocket” model of salpingitis, a delayed hypersensitivity reaction was shown to be mediated by hsp60 (16). Chlamydial hsp60 has also been found to elicit a severe inflammatory response almost identical to that seen in trachoma when inoculated onto the conjunctivae of both previously immunized guinea pigs and monkeys (15, 21). In addition, women with a history of multiple episodes of salpingitis have been found to exhibit lymphocyte proliferation in response to hsp60 more often than healthy women or women with a history of a single episode of salpingitis (26). A strong association between serum antibodies to hsp60 and chlamydia-associated tubal infertility has also been demonstrated which was independent of microimmunofluorescence assay (MIF) evidence of exposure to C. trachomatis (23). Recently in The Gambia, where chlamydial seroprevalence rates were >84% for patients and controls, serum immunoglobulin G (IgG) antibodies to chlamydial hsp60 were significantly associated with scarring trachoma (17).
These data support the notion that the host immune response to chlamydial hsp60 may be important in disease progression. However, to date, studies of hsp60 immunoreactivity have been limited to serum antibody responses in patients with scarring disease only. The objective of this study was to characterize both the mucosal and systemic antibody-mediated responses to hsp60 across all clinical grades of trachoma in patients from an area of Nepal where chlamydia is endemic. In addition, we examined immunoreactivity to the major outer membrane protein (MOMP) and also to five hsp60 fusion proteins (fp) in order to identify immunodominant regions of the protein.
MATERIALS AND METHODS
Study population and specimen collection.
Individuals of ≥1 year of age from nine randomly selected households in a Nepali village where trachoma was endemic were enrolled after informed consent. The bilateral upper tarsal conjunctivae of each study participant were photographed and graded according to the World Health Organization trachoma grading scale (22). Grading was conducted in a blinded fashion by the authors T. Hessel and D. Dean and by T. Lietman. A grade of no trachoma (TO) was used to represent an absence of clinical signs of trachoma, follicular trachoma (TF) to denote ≥5 follicles on the lower two-thirds of the upper tarsus, inflammatory trachoma (TI) to denote >50% of the upper tarsal blood vessels obscured by inflammation, and conjunctival scarring (TS) to represent scarring of the conjunctiva. If the grade for each eye differed for an individual patient, the more advanced grade was recorded. Final grades required consensus of two or more readers. To collect tears, sterile Weck-cel sponges (Edward Weck Inc., Research Triangle Park, N.C.) were applied to the inner canthus of each eye and allowed to swell. Serum samples were obtained. All samples were frozen in liquid nitrogen for transport to the laboratory and were then stored at −80°C until use.
Dot blot analysis.
Dot blots using the Bio-Rad dot blot apparatus were performed according to the manufacturer's instructions (Bio-Rad, Hercules, Calif.). Tears were extracted from eye sponges by thawing sponges on ice, reconstituting with 25 μl of phosphate-buffered saline, and pipetting the liquid off the sponge. Samples collected from both eyes were pooled for each patient. Tears and sera from each patient were diluted 1:100 in phosphate-buffered saline and reacted against whole hsp60 protein, MOMP, and five contiguous fp from C. trachomatis. All purified recombinant proteins used in this research were expressed as fp with glutathione S-transferase, similar to other referenced methods (2, 17). Control dot blots were performed with each batch of samples using a secretory IgA (sIgA) monoclonal antibody (Chemicon International, Temecula, Calif.), anti-chlamydial hsp60 monoclonal antibody IgG1 (monoclonal [mouse] anti-hsp60 IgG1; Affinity Bioreagents, Golden, Colo.), and anti-MOMP monoclonal IgG (monoclonal [mouse] anti-MOMP IgG2, Cortex, San Leandro, Calif.) in a 1:1,000 dilution. The IgG1 antibody for hsp60 was reactive against chlamydial hsp60 and each of the five fp. Sera from highly seropositive and known seronegative individuals were used as positive and negative controls, respectively. The positive control sera reacted to whole hsp60 as well as all five fp and MOMP. Twenty positive control sera were tested at serial dilutions from 1:50 to 1:400. The 1:50 dilution did not improve the detection of antibodies over the 1:100 dilution. Hence, the 1:100 dilution was used for this assay.
Because the C. trachomatis hsp60 sequence has been demonstrated to share 48% homology with human hsp60, 93% with that of Chlamydia psittaci, 60% with that of Escherichia coli, (2), and 80% with that of Chlamydia pneumoniae (12), sera from seronegative individuals were used to ensure that cross-reactivity to other hsp60s was not occurring. Secondary antibodies for the control blots were alkaline phosphatase-conjugated goat anti-mouse IgA, sIgA, and IgG antibodies (Zymed Labs, South San Francisco, Calif.) diluted 1:1,000. Secondary antibodies for patient tears or sera were alkaline phosphatase-conjugated goat anti-human IgA, sIgA, and IgG antibodies (Zymed Labs) diluted 1:1,000. Dot blots were analyzed by densitometry in a Bio-Rad gel documentation system (Bio-Rad), where the mean was taken on background for six negative controls and where 3 standard deviations above the mean standard deviation of these six controls was used to identify an hsp60-reactive sample. There was no cross-reactivity with glutathione S-transferase.
Chlamydial serology by MIF.
Chlamydial antibody titers were determined using MIF according to standard techniques (25). Sera were assayed for IgG antibodies at twofold dilutions from 1:8 to 1:256. Titers of ≥1:8 for C. trachomatis and 1:32 for C. pneumoniae were considered evidence of past infection.
Data analysis.
Multivariate analyses were performed using unconditional logistic regression; family variables (grouping by household) were included in the final model to account for cluster sampling. Analyses were additionally controlled for age and sex. For discrete data, the Pierson chi-square and Fisher's exact tests were used. Measurements of dot blot results were determined by densitometry and were recorded as units after subtracting out the negative control densitometry readings for the respective blot. Immunoreactivity to hsp60 was defined as a positive antibody response to MOMP, whole hsp60, and/or ≥1 fp for densitometry readings above the cutoff. Tear and serum immunoreactivity results for IgA, sIgA, and IgG were compared for patients with and without clinical disease.
RESULTS
This study represented 146 individuals, 1 to 87 years of age. The mean age was 19 years. Sixty-six participants (45%) were ≤10 years old; 76 (52%) were females. One hundred and thirty (89%) individuals had clinical trachoma; 16 (11%) of 146 had TO, 40 (27%) had TF, 53 (36%) had TI, and 37 (26%) had TS. By univariate analysis, children 10 years of age or younger were demonstrated to be statistically significantly likelier to have active disease (TF and TI) than no evidence of disease (TO) (Table 1). In addition, 34 (92%) of 37 patients with scarring disease were >10 years old, compared with 34 (37%) of 93 patients with active disease in the same age group (P < 0.001). There were no statistically significant differences in patient age for TO and TS and no significant differences for gender and trachoma grade (Table 1).
TABLE 1.
Univariate analysis of sIgA tear, IgG tear, and IgG serum immunoreactivity to C. trachomatis hsp60 and of trachoma grade by sex and agea
| Immunoglobulin or trachoma grade | No. of patients
|
P | No. with age (yr) of:
|
P | ||
|---|---|---|---|---|---|---|
| Female | Male | ≤10 | >10 | |||
| With tear IgG antibody | 63 | 57 | NS | 58 | 62 | NS |
| Without tear IgG antibody | 13 | 13 | 8 | 18 | ||
| With tear sIgA antibody | 49 | 54 | NS | 47 | 56 | NS |
| Without tear sIgA antibody | 27 | 16 | 19 | 24 | ||
| With serum IgG antibody | 50 | 45 | NS | 43 | 52 | NS |
| Without serum IgG antibody | 21 | 19 | 16 | 24 | ||
| TO | 6 | 10 | 4 | 12 | ||
| TF | 21 | 19 | NS | 23 | 17 | .027 |
| TI | 28 | 25 | NS | 36 | 17 | .002 |
| TS | 21 | 16 | NS | 3 | 34 | NS |
P values for trachoma grades are in reference to TO. NS, not significant.
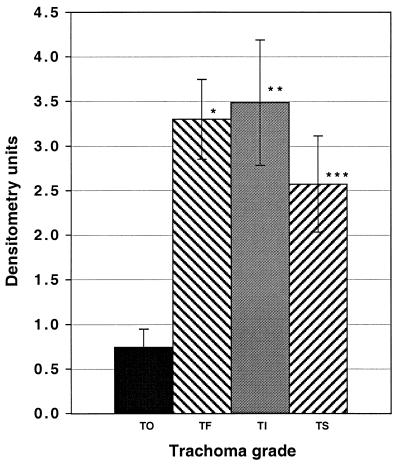
One hundred and forty-five (99%) of 146 patients had tear total IgA anti-hsp60 antibodies; 103 (70%) of the 146 patients had tear sIgA anti-hsp60 antibodies, and 120 (82%) had tear IgG anti-hsp60 antibodies (Fig. 1). Figure 1 graphically represents the mean density with standard error of the mean for each tear sample reacted by dot blot for IgG. Ninety-five (70%) of the 135 patients for whom adequate serum samples were available had sera which were IgG immunoreactive to hsp60. The odds ratio for trachoma grades TF, TI, and TS by sIgA and IgG tear immunoreactivities was determined, controlling for age, sex, and cluster sampling method (Table 2). Tears were found to be IgG reactive to hsp60 in 6 (38%) of 16 patients with TO, compared with 36 (90%) of 40 with TF (P < 0.001), 47 (89%) of 53 with TI (P < 0.001), and 31 (84%) of 37 with TS (P = 0.002). There were no significant differences for sIgA and no association between sIgA and IgG tear immunoreactivities for age or sex (Table 1). However, when we looked at patients of >10 years of age, 28 (82%) of 34 patients with TS had tear anti-hsp60 IgG reactivity, compared with 3 (25%) of 12 patients with TO (P < 0.001).
FIG. 1.
Tear IgG immunoreactivity against C. trachomatis hsp60 by trachoma grade. Measurements of dot blot results were determined by densitometry and are recorded as units. Data are presented as the mean ± standard deviation after subtracting the negative control densitometry readings for each blot. TO, n = 16; TF, n = 40; TI, n = 53; TS, n = 37; ∗, P < 0.001; ∗∗, P < 0.001; ∗∗∗, P = 0.002.
TABLE 2.
Odds ratio for clinical trachoma grades TF, TI, and TS according to IgG tear immunoreactivity to the 60-kDa C. trachomatis hsp by multivariate analysis adjusted for age, sex, and cluster sampling method
| Trachoma grade | Adjusted odds ratio (95% CI) | P |
|---|---|---|
| TF | 49.2 (2.70–897.68) | 0.008 |
| TI | 22.6 (3.02–169.76) | 0.002 |
| TS | 13.6 (1.38–133.11) | 0.025 |
Tears were reacted against MOMP in the same dot blot assay and were compared with anti-hsp60 immunoreactivity and trachoma grade. Unlike for hsp60, there were no statistically significant associations with trachoma grade (Tables 3 and 4). This suggests that the anti-chlamydial hsp60 reactivity was specific for hsp60. Although tear anti-hsp60 immunoreactivity was also determined for each of five fp (constructed to represent the chlamydial hsp60) to identify whether there were immunodominant regions of the protein, none were found (data not shown).
TABLE 3.
Tear IgG immunoreactivity to C. trachomatis hsp60 and MOMP according to clinical grade of trachoma
| Protein used | Immunoreactivity result for:
|
|||
|---|---|---|---|---|
| TO (n = 16) (%) | TF (n = 40) (%) | TI (n = 53) (%) | TS (n = 37) (%) | |
| Whole hsp60 or any fp | 6 (38) | 36 (90) | 47 (89) | 31 (84) |
| Whole hsp60 | 6 (38) | 29 (72) | 37 (70) | 26 (70) |
| MOMP | 6 (38) | 18 (45) | 21 (40) | 15 (41) |
TABLE 4.
Tear sIgA immunoreactivity to C. trachomatis hsp60 and MOMP according to clinical grade of trachoma
| Protein used | Immunoreactivity result for:
|
|||
|---|---|---|---|---|
| TO (n = 16) (%) | TF (n = 40) (%) | TI (n = 53) (%) | TS (n = 37) (%) | |
| Whole hsp60 or any fusion protein | 13 (81) | 30 (75) | 39 (74) | 21 (57) |
| Whole hsp60 | 9 (56) | 23 (58) | 21 (40) | 13 (35) |
| MOMP | 4 (25) | 15 (38) | 21 (40) | 14 (38) |
Sera from 41 (84%) of 49 TI patients were IgG reactive to chlamydial hsp60, compared with 6 (46%) of 13 with TO (P = 0.009). Serum IgG results for TF (55%) and TS (58%) patients were not significant when compared with TO patients. There was no association between serum immunoreactivity and sex or age (Table 1).
All TO patients and 129 (99%) of 130 patients with TF, TI, and TS had evidence of past exposure to C. trachomatis by MIF conducted on patient sera. The median C. trachomatis MIF titer was 1:64 for the total population as well as for each trachoma grade. The median C. trachomatis MIF titer remained 1:64 when patients were grouped according to sex and age. Ninety-four (72%) of 130 patients had evidence of past C. pneumoniae infection by MIF. The mean MIF titer for C. pneumoniae was 1:32 and was unchanged when patients were grouped according to age, sex, or grade. Therefore, there were no statistically significant associations for antibody titers to C. trachomatis or C. pneumoniae by trachoma grade, sex, or age.
DISCUSSION
Our results show that the mucosal antibody response to chlamydial hsp60 is significantly associated with both clinically active and scarring forms of disease. This association was independent of C. trachomatis exposure, given the uniform seropositivity by MIF to C. trachomatis across all trachoma grades. Further, through random selection of households and cluster analysis, we attempted to control for the possible influence of human genetic variation.
Tear immunoreactivity in patients with active disease may be explained by the presence of chlamydiae that produce hsp60 and elicit a local immune response. Tears may contain naturally occurring immunoglobulins stimulated by repeated antigenic challenge of the conjunctivae (18). This response is mediated by antigens presented by macrophages and Langerhans cells in the mucosal epithelium which, in turn, stimulate B and T cells. Immunoglobulins produced in the lacrimal gland, conjunctivae, and accessory glands include predominantly sIgA but also include IgG and IgM (18). Thus, the pattern of locally produced IgG found in tears might be expected to differ from that of serum IgG, especially among trachoma patients who are repeatedly challenged with chlamydial antigens. Indeed, we found that tear IgG reactivity to hsp60 differed appreciably from that of sera, suggesting that tear immunoreactivity represents, to a certain extent, local antibody production. Although the tear sIgA results on the same patient population did not show a predominance of reactivity for any particular trachoma grade, the overall sIgA reactivity of 82% suggests that local antibody production does occur and may be influenced by repeated infections such that there is broad reactivity in the population.
While patients with scarring and active trachoma were significantly likelier to have tear immunoreactivity to hsp60, there were six patients with a trachoma grade of TO in which tear immunoreactivity to hsp60 was demonstrated. By definition, a trachoma grade of active or scarring disease requires clinical evidence of inflammatory changes. However, the TO population likely contains individuals who are in the early stages of chlamydial infection but have not yet manifested clinical disease. This is supported by studies of trachoma patients in The Gambia in which chlamydial infections were detected up to 2 weeks before the development of clinically visible inflammatory changes (3). It is also possible that some of these TO individuals were infected but did not mount an inflammatory response.
The presence of tear anti-hsp60 antibodies in scarring disease may indicate stimulation in response to hsp60 produced by persistent organisms. Previous studies have demonstrated a low prevalence of active infection in patients with scarring disease (4, 13). There is a growing body of literature that shows that C. trachomatis in addition to other species of Chlamydia can persist in the human host (6, 9, 10, 19, 27). Anatomic sites that have been identified include the cervix, synovium, lungs, and arterial vasculature; persistence appeared to have occurred at these sites for many years. In in vitro studies in the presence of gamma interferon, chlamydiae developed into atypical, noninfectious forms which produced near-normal levels of hsp60 (1). In contrast, levels of MOMP and other outer membrane constituents were greatly reduced (1). In a recent in vitro model of apoptosis using a strain commonly found in trachoma populations—serovar A—both active and persistent infections were found to resist apoptosis by preventing release of cytochrome c from the mitochondria (5). This antiapoptotic effect was sustained for the duration of the persistent but not acute infection, with associated sustained production of hsp60 and reduced levels of MOMP. These data suggest an important mechanism for how chlamydiae are able to persist in human host cells. Thus, the high rates of mucosal antibodies to hsp60 and lack of antibodies to MOMP among patients with scarring disease in our study may reflect the presence of persistent organisms that provide a continued source for antigenic stimulation.
Alternately, it is possible that repeated exposure to other bacteria or repeat chlamydial infections may result in a prolonged antibody response and also prime the cellular immune system such that a subsequent ocular challenge may induce a pathogenic cell-mediated response. A hsp60 T-cell epitope has been identified that is conserved among chlamydial biovars (7, 8) and has high homology with E. coli and other bacterial recognition sequences (4, 14). Recent studies in patients with postchlamydial reactive arthritis have shown that while chlamydia-specific CD4+ T-cell clones recognized neither intact human nor E. coli hsp60, a peptide containing a T cell epitope was found to be stimulatory (8). In Yersinia enterocolitica-induced arthritis, hsp60 strongly induced two types of T-cell clones: one clone responded to Y. enterocolitica, C. trachomatis, Borrelia burgdorferi, and human hsp60s with a cytokine profile typical of type 2 T-helper cells; the other clone showed specificity only to bacterial hsp60s with a type 1 T-helper cytokine profile (14). Although the immune response may be directed against bacterium-specific epitopes of hsp60 in trachoma, that response may be primed by other chlamydial infections as well as by exposure to other bacteria. Villagers in regions of chlamydial endemicity may undergo seasonal epidemics of bacterial conjunctivitis as well as repeat C. trachomatis ocular infections (4). Thus, there may be ample opportunity for induction of a deleterious immune response from repeat antigenic challenge throughout a lifetime.
It might therefore be expected that systemic antibodies to chlamydial or bacterial hsp60 would be present in patients with scarring disease. Peeling et al. (17) demonstrated significantly higher rates of serum IgG immunoreactivity to hsp60 among scarred patients than among those without disease. While we found a similar trend, it was significant only for patients with TI. This may reflect the small sample size and, therefore, lack of sufficient power in our study to detect a difference. Yet it is difficult to interpret serum antibodies that may represent cross-reactivity with other chlamydial species prevalent in communities of chlamydial endemicity.
The fact that tear anti-chlamydial hsp60 antibody was significantly associated with inflammation and scarring in our study suggests that there may be, in addition to cell-mediated responses, a component of complement fixing that may promote disease progression. Both IgG and IgA are effective in inducing the release of inflammatory mediators. The four isotypes of IgG induce the release of granule enzymes from neutrophils, but only IgG1, IgG2, and IgG3 activate the complement pathway (20). This pathway promotes acute inflammation through a variety of complex interactions but can also activate macrophages to secrete additional mediators of inflammation. Activated macrophages may serve an additional role of promoting tissue damage through release of acid hydrolases and hydrogen peroxide (H2O2), through tissue reorganization involving fibrogenesis and angiogenesis factors, and through the release of tumor necrosis factor alpha, a potent inducer of fibroblasts which effectuate tissue remodeling (24). Thus, hsp60 antibody may be an important indicator of infection but may also be a risk factor for disease progression.
Additional research is required to map specific epitopes of known or putative C. trachomatis membrane proteins, including hsp60, and to further define the nature of the pathogenic mechanisms underlying conjunctival scarring among trachoma patients.
ACKNOWLEDGMENTS
This research was supported in part by Public Health Service grants EY00310 and EY/AI12219 (to D.D.) from the National Institutes of Health, the Rainer's Fund (to T.H.), and the American Academy of Allergy, Asthma, and Immunology (to R.P.). T.H. was a Howard Hughes Medical Institute Medical Student Research Training Fellow. R.P. has a Fellowship from the Office of the Dean, University of California at San Francisco.
We acknowledge Jocelyn Phegan and Amy Helmer for excellent technical assistance and Mark Pletcher for critical review of the manuscript. We also thank Tom Lietman for trachoma grading of photographs. We are grateful for the invaluable assistance of ophthalmic assistants R. Karki and H. L. Dhami and for the excellent technical support of G. Amar and the rest of the staff of the Lumbini Rana-Ambika Eye Hospital, as well as to the Seva Foundation and villagers of Kapilvastu District, Lumbini Zone, Nepal, without whom this research would not have been possible.
REFERENCES
- 1.Beatty W L, Byrne G I, Morrison R P. Morphologic and antigenic characterization of interferon gamma-mediated persistent Chlamydia trachomatis infection in vitro. Proc Natl Acad Sci USA. 1993;90:3998–4002. doi: 10.1073/pnas.90.9.3998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Cerrone M C, Ma J J, Stephens R S. Cloning and sequence of the gene for heat shock protein 60 from Chlamydia trachomatis and immunological reactivity of the protein. Infect Immun. 1991;59:79–90. doi: 10.1128/iai.59.1.79-90.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Dawson C R. Trachoma and other chlamydial eye diseases. In: Orfilia J, Byrne G I, Chernesky M A, editors. Chlamydial infections. Bologna, Italy: Societa Editrice Esculapio; 1994. pp. 277–287. [Google Scholar]
- 4.Dean D. Trachoma. In: Connor D H, Schwartz D A, Chandler F W, editors. Pathology of infectious diseases. Stamford, Conn: Appleton and Lange Publishers; 1997. pp. 498–507. [Google Scholar]
- 5.Dean D, Powers V C. Persistent Chlamydia trachomatis infections resist apoptotic stimuli. Infect Immun. 2001;69:2442–2447. doi: 10.1128/IAI.69.4.2442-2447.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Dean D, Suchland R, Stamm W. Evidence for long-term cervical persistence of Chlamydia trachomatis by omp1 genotyping. J Infect Dis. 2000;182:909–916. doi: 10.1086/315778. [DOI] [PubMed] [Google Scholar]
- 7.Deane K H, Jecock R M, Pearce J H, Gaston J S. Identification and characterization of a DR4-restricted T cell epitope within chlamydia heat shock protein 60. Clin Exp Immunol. 1997;109:439–445. doi: 10.1046/j.1365-2249.1997.4711371.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Gaston J S H, Deane K H O, Goodall J C, Pearce J H. The pathogenic significance of T cell recognition of chlamydial antigens. In: Jones R, et al., editors. Chlamydial infections. Proceedings of the 9th International Symposium on Human Chlamydial Infection. San Francisco, Calif: International Chlamydia Symposium; 1998. pp. 502–505. [Google Scholar]
- 9.Gerard H C, Branigan P J, Schumacher H R, Hudson A P. Synovial Chlamydia trachomatis in patients with reactive arthritis/Reiter's syndrome are viable but show aberrant gene expression. J Rheumatol. 1998;25:734–742. [PubMed] [Google Scholar]
- 10.Grayston J T. Background and current knowledge of Chlamydia pneumoniae and atherosclerosis. J Infect Dis. 2000;181:S402–S410. doi: 10.1086/315596. [DOI] [PubMed] [Google Scholar]
- 11.Kaufmann S H. Heat shock proteins and the immune response. Immunol Today. 1990;11:129–136. doi: 10.1016/0167-5699(90)90050-j. [DOI] [PubMed] [Google Scholar]
- 12.Kikuta L C, Puolakkainen M, Kuo C C, Campbell L A. Isolation and sequence analysis of the Chlamydia pneumoniae GroE operon. Infect Immun. 1991;59:4665–4669. doi: 10.1128/iai.59.12.4665-4669.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Mabey D C, Bailey R L, Ward M E, Whittle H C. A longitudinal study of trachoma in a Gambian village: implications concerning the pathogenesis of chlamydial infection. Epidemiol Infect. 1992;108:343–351. doi: 10.1017/s0950268800049815. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Mertz A K, Ugrinovic S, Lauster R, Wu P, Grolms M, Bottcher U, Appel H, Yin Z, Schiltz E, Batsford S, Schauer-Petrowski C, Braun J, Distler A, Sieper J. Characterization of the synovial T cell response to various recombinant Yersinia antigens in Yersinia enterocolitica-triggered reactive arthritis. Heat-shock protein 60 drives a major immune response. Arthritis Rheum. 1998;41:315–326. doi: 10.1002/1529-0131(199802)41:2<315::AID-ART16>3.0.CO;2-#. [DOI] [PubMed] [Google Scholar]
- 15.Morrison R P, Lyng K, Caldwell H D. Chlamydial disease pathogenesis. Ocular hypersensitivity elicited by a genus-specific 57-kD protein. J Exp Med. 1989;169:663–675. doi: 10.1084/jem.169.3.663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Patton D L, Sweeney Y T, Kuo C C. Demonstration of delayed hypersensitivity in Chlamydia trachomatis salpingitis in monkeys: a pathogenic mechanism of tubal damage. J Infect Dis. 1994;169:680–683. doi: 10.1093/infdis/169.3.680. [DOI] [PubMed] [Google Scholar]
- 17.Peeling R W, Bailey R L, Conway D J, Holland M J, Campbell A E, Jallow O, Whittle H C, Mabey D C. Antibody response to the 60-kDa chlamydial heat-shock protein is associated with scarring trachoma. J Infect Dis. 1998;177:256–259. doi: 10.1086/517367. [DOI] [PubMed] [Google Scholar]
- 18.Pleyer U, Baatz H. Antibacterial protection of the ocular surface. Ophthalmologica. 1997;211(Suppl. 1):2–8. doi: 10.1159/000310878. [DOI] [PubMed] [Google Scholar]
- 19.Rahman M U, Cheema M A, Schumacher H R, Hudson A P. Molecular evidence for the presence of chlamydia in the synovium of patients with Reiter's syndrome. Arthritis Rheum. 1992;35:521–529. doi: 10.1002/art.1780350506. [DOI] [PubMed] [Google Scholar]
- 20.Roitt I M. Essential immunology. 8th ed. Oxford, United Kingdom: Blackwell Scientific Publications; 1994. [Google Scholar]
- 21.Taylor H R, Maclean I W, Brunham R C, Pal S, Wittum-Hudson J. Chlamydial heat shock proteins and trachoma. Infect Immun. 1990;58:3061–3063. doi: 10.1128/iai.58.9.3061-3063.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Thylefors B, Dawson C R, Jones B R, West S K, Taylor H R. A simple system for the assessment of trachoma and its complications. Bull W H O. 1987;65:477–483. [PMC free article] [PubMed] [Google Scholar]
- 23.Toye B, Laferriere C, Claman P, Jessamine P, Peeling R. Association between antibody to the chlamydial heat-shock protein and tubal infertility. J Infect Dis. 1993;168:1236–1240. doi: 10.1093/infdis/168.5.1236. [DOI] [PubMed] [Google Scholar]
- 24.Vassalli P. The pathophysiology of tumor necrosis factors. Annu Rev Immunol. 1992;10:411–452. doi: 10.1146/annurev.iy.10.040192.002211. [DOI] [PubMed] [Google Scholar]
- 25.Wang S P, Kuo C C, Grayston J T. Formalinized Chlamydia trachomatis organisms as antigen in the micro-immunofluorescence test. J Clin Microbiol. 1979;10:259–261. doi: 10.1128/jcm.10.2.259-261.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Witkin S S, Jeremias J, Toth M, Ledger W J. Proliferative response to conserved epitopes of the Chlamydia trachomatis and human 60-kilodalton heat-shock proteins by lymphocytes from women with salpingitis. Am J Obstet Gynecol. 1994;171:455–460. doi: 10.1016/0002-9378(94)90282-8. [DOI] [PubMed] [Google Scholar]
- 27.Wu L, Skinner S J, Lambie N, Vuletic J C, Blasi F, Black P N. Immunohistochemical staining for Chlamydia pneumoniae is increased in lung tissue from subjects with chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2000;162:1148–1151. doi: 10.1164/ajrccm.162.3.9912134. [DOI] [PubMed] [Google Scholar]