Abstract
The skin is the main barrier between the body and the environment, protecting it from external oxidative stress induced by ultraviolet rays. It also prevents the entrance of infectious agents such as viruses, external antigens, allergens, and bacteria into our bodies. An overreaction to these agents causes severe skin diseases, including atopic dermatitis, pruritus, psoriasis, skin cancer, and vitiligo. Members of the flavonoid family include apigenin, quercetin, luteolin, and kaempferol. Of these, apigenin has been used as a dietary supplement due to its various biological activities and has been shown to reduce skin inflammation by downregulating various inflammatory markers and molecular targets. In this review, we deal with current knowledge about inflammatory reactions in the skin and the molecular mechanisms by which apigenin reduces skin inflammation.
Keywords: apigenin, skin inflammation, flavonoid, skin, atopic dermatitis, psoriasis, pruritus, skin cancer, vitiligo
1. Introduction
The skin may be divided into several layers according to their main functions, including the epidermis, dermis, and hypodermis [1]. The epidermis is the outermost layer of the skin and acts as a barrier to prevent disruption by external stimuli [2,3]. Moreover, it acts as a defense system to retain immune homeostasis against many pathogens, including bacteria, antigens, and viruses [4]. The epidermis consists largely of keratinocytes and some Langerhans cells (LC), melanocytes, and Merkel cells [5]. Among these, the Langerhans cell is a type of dendritic cell (DC) that can present the necessary antigen for the innate immune response. When LCs are activated by ultraviolet (UV) irradiation, E-cadherin is expressed at low levels in keratinocytes. As a result, migration of LCs into the lymph nodes occurs, and regulatory T cells can be further matured [6,7]. Idoyaga et al. [8] revealed that skin DCs can be targeted for immunomodulatory therapies. In the outer layers of the epidermis, the skin microbiome retains skin acidity to protect against external infections [9,10,11]. Organisms in the microbiome also interact with each other to form a community in the skin, and these interactions ensure immune homeostasis in the skin. On the other hand, dysregulation of the inflammatory equilibrium can induce skin immunological diseases [12,13,14], including atopic dermatitis (AD), pruritus, psoriasis, skin cancer, and vitiligo [15,16,17]. As part of the efforts to attenuate inflammatory symptoms, steroidal and non-steroidal drugs are mostly used as classical treatments [18,19,20,21,22]. However, because of the side effects of anti-inflammatory drugs, studies investigating natural compounds to replace chemical drugs have been more actively performed [23].
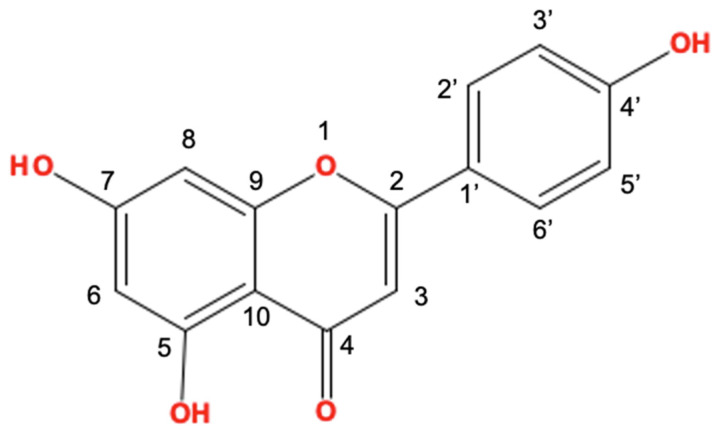
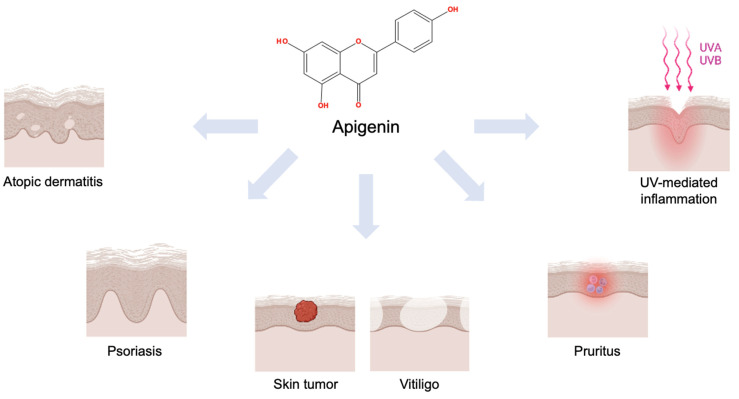
Apigenin (4′,5,7-trihydroxyflavone, Figure 1) is a natural compound that belongs to the subclass of flavonoids [24,25]. In previous studies, Tanacetum, Achillea, Artemisia, and Matricaria genera belonging to the Artemisia family of plants have been reported as the main source plants of apigenin [26,27,28,29]. It has been presented that apigenin has the potential to attenuate skin inflammatory conditions, such as AD, pruritus, and psoriasis, and the tumorigenic response of skin cancers (Figure 2). In addition, its anti-apoptotic [30], anti-inflammatory [31], and anti-hyperglycemic effects [32] have been researched. In this review, we focus on describing the functional properties of apigenin and its potential for pharmacological effects.
Figure 1.
The structure of apigenin (4′,5,7-trihydroxyflavone). This figure was made using JChemPaint software.
Figure 2.
Scheme of the therapeutic efficacies of apigenin on the skin. This figure was created with BioRender.com.
2. Anti-Inflammatory Effects of Apigenin on UV-Irradiated Skin
UV light is the main cause of skin inflammation and can be divided into UVA (320–400 nm), UVB (280–320 nm), and UVC (100–280 nm), according to the wavelength. Especially, UVA and UVB penetrate the skin layers, and can induce skin inflammation and aging in keratinocytes and fibroblasts [33,34]. UVB exposure causes an acute inflammation response in the epidermis by promoting the synthesis and secretion of pro-inflammatory cytokines, such as tumor necrosis factor (TNF)-α and interleukin (IL)-6 from epidermal cells [35]. Moreover, UVA penetrates the dermal layers and indirectly causes DNA damage with degradation of the collagen and elastin fibers [34].
In previous studies, apigenin attenuated skin inflammation by downregulating the expression of cyclooxygenase-2 (COX-2) [36]. Another finding suggests its potential as a novel target for reducing skin inflammation. This compound works in the keratinocyte by targeting the non-receptor tyrosine kinase (e.g., Src) and COX-2 [37]. Apigenin prevented cyclobutene pyrimidine dimers, which are generated by UV exposure. Due to this, apigenin restored the lower level of nucleotide excision repair proteins and affected cell apoptosis [38,39]. Apigenin downregulated the level of metalloproteinase-1 by interfering with the Ca2+ influx-dependent mitogen-activated protein kinase (MAPK) and activator protein-1 (AP-1) pathways in HaCaT or normal human dermal fibroblast cells under UVA-irradiated conditions. Especially, the levels of c-Jun and c-Fos were decreased by apigenin treatment, which suppressed the phosphorylation of extracellular signal-regulated kinase (ERK), c-Jun N-terminal kinase (JNK), and p38 [40,41]. As a result, apigenin could attenuate UV-mediated inflammation by decreasing the transcription of inflammatory cytokines via the downregulation of the AP-1, MAPK, and apoptotic signaling pathways (Figure 3).
Figure 3.
The molecular interactions of apigenin due to UV irradiation.
3. Effect of Apigenin on Attenuating AD
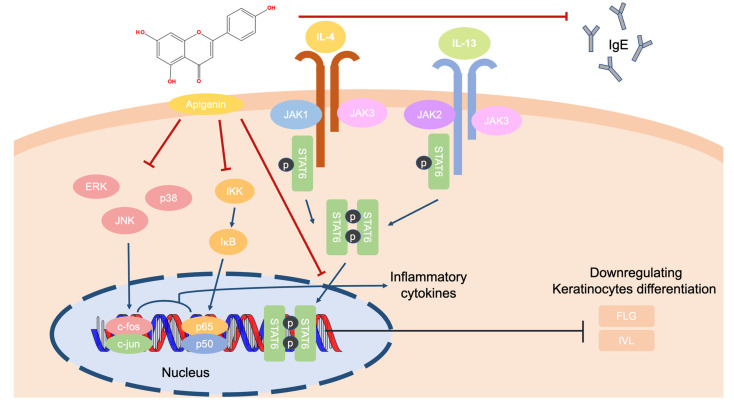
AD is a chronic inflammatory disease that affects 80% of patients in infancy or childhood. Its severe symptoms include itching, dry skin, eczema, and swallowing. The cytokines and chemokines secreted in AD are summarized in Table 1; however, the exact mechanism of the stimulus is still unclear. There are many hypotheses to explain AD pathogenesis, including: (I) disproportion of skin microbiomes [3,42,43,44]; (II) weakness of skin barrier junctions [45,46]; (III) dysregulation between pro- and anti-inflammatory cytokines [47,48]; and (IV) excessive immunoglobulin E (IgE) secretion. Among these explanations, apigenin reduced IgE and interferon (IFN)-γ levels in serum in an NC/Nga mouse model. Moreover, apigenin attenuated damaging skin lesions induced by picrylchloride. Considering the protein levels, apigenin suppressed the phosphorylation of the signal transducer and activator of transcription 6 (STAT6) in IL-4–stimulated mouse spleen cells [49]. In addition, apigenin showed a low expression of IL-31 in messenger RNA in a human mast cell line (HMC-1). In HMC-1 cells, apigenin downregulated nuclear factor-κB (NF-κB) pathway proteins, including the inhibitor of κB kinase, inhibitor of κB, and the p65/NF-κB, and MAPK pathway factors of c-Jun N-terminal kinase, ERK, and p38 [50]. In summary, it is thought that apigenin can ameliorate the symptoms of AD by decreasing the levels of pro-inflammatory cytokines and inflammatory mediators by downregulating the MAPK, NF-κB, and Jak/STAT signaling pathways (Figure 4).
Table 1.
The cytokines and chemokines that stimulate AD.
| Cytokines | Classification | Role | Reference |
|---|---|---|---|
| IL-4 | Th2 cell-derived cytokines |
|
[51,52] |
| IL-13 | |||
| IL-31 | Th2 cell-derived cytokines |
|
[53,54] |
| IL-1α | IL-1 family |
|
[55,56] |
| IL-1β | |||
| IL-33 | IL-1 family |
|
[57,58,59] |
| IFN-γ | Th1 cytokines |
|
[60] |
| TNF-α | |||
| TSLP | IL-7-like cytokines |
|
[61,62,63,64] |
Figure 4.
The role of apigenin in protein regulation in AD-irritated skin cells.
4. Treatment with Apigenin for Alleviating Pruritus
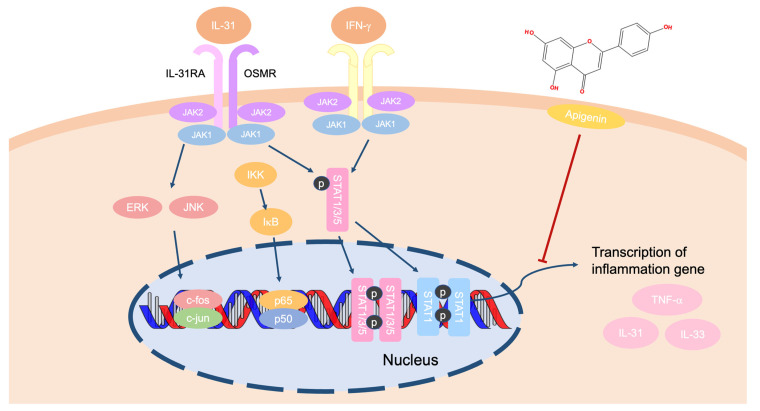
Pruritus or itching negatively affects quality of life [65,66]. Many different factors play a role in this event, but the exact pathogenetic mechanisms are not known [67,68]. Histamine, serotonin, cytokines, peptides, and phospholipid metabolites are included as mediators of pruritus [69]. Among these, cytokines are strong players that manage itching by activating receptors [70]. One example, IL-31, which is derived from the IL-6 family, acts as the therapeutic target of pruritus in the Th2 cell-mediated response. IL-31 has been reported to induce chemokines such as CCL1, CCL17, and CCL22 [71,72]. Additionally, IL-33 has also been considered a pathophysiologically important cytokine that manages innate immune responses [73] and Th2 cell differentiation by promoting the expression of chemokines and pro-inflammatory cytokines, and by activating natural killer cells and dendritic cells [74]. Apigenin weakened the expression of IL-31 in human mast cells and mouse skin through downregulation of MAPK and NF-κB signaling [50]. In an ovalbumin-induced BALB/c mouse model, apigenin regulated the balance of Th1/Th2 cells by downregulating the NF-κB pathway and reducing histamine, IgE, and STAT1 expression. Moreover, apigenin improved the Th1 response by controlling the expression of IFN-γ and T-box protein expressed in T cells [75]. Furthermore, apigenin-treated microglial cells lowered the expression levels of IL-31 and IL-33 without displaying cytotoxicity. This expression was verified by polymerase chain reaction as well as Western blotting via the inhibition of the ERK and JNK pathways [76]. In experiments using astrocytes, apigenin significantly suppressed IL-31 and IL-33 messenger RNA expression. Pre-treatment with apigenin in astrocytes decreased the expression levels of IL-31 and IL-33 at the protein level. In astrocytes, apigenin also inhibited the phosphorylation of MAPK and NF-κB signaling proteins [77]. Taken together, these studies suggest that apigenin can ameliorate pruritus by inhibiting IL-31 and IL-33 secretion through suppression of the NF-κB and MAPK pathways (Figure 5).
Figure 5.
A schema of the efficacy of apigenin against pruritus.
5. The Mechanism of Apigenin for the Amelioration of Psoriasis
Psoriasis is a chronic, immunological skin disease affecting about 125 million patients in America [78]. These patients and others motivate the study of psoriasis to improve pathophysiological knowledge of the condition. Psoriasis is a sustained inflammatory disease caused by the hyperproliferation of keratinocytes and dysfunctional differentiation. In addition, the infiltration of Th17 cells secreting inflammatory cytokines, such as IL-23, into keratinocytes, dominantly occurs in psoriasis [79].
Apigenin showed the greatest effects in a psoriasis model by decreasing cytokine levels. Skin barrier recovery effects were observed in apigenin-treated skin. Apigenin also improved the skin’s condition by increasing the hydration level of the stratum corneum. Meanwhile, apigenin influenced the synthesis of skin structural proteins such as filaggrin, involucrin, and loricrin in mouse models [80]. With co-treatment of apigenin and lipopolysaccharide (LPS) in DCs, this compound significantly inhibited TNF-α messenger RNA expression. Moreover, apigenin suppressed the level of pro-inflammatory cytokines, including IFN-γ, IL-6, IL-1β, IL-23, and IL-10, in both LPS treatment and non-treatment groups. Meanwhile, apigenin affected naïve T cell differentiation by modulating the function of DCs [81]. Overall, apigenin treatment may ameliorate psoriasis symptoms by regulating the transcription of inflammatory cytokines via regulation of the Toll-like receptor 4 pathway (Figure 6).
Figure 6.
The mechanism of apigenin in LPS-treated DCs.
6. The Suppressive Activity of Apigenin on Skin Cancer
The inflammation response promotes cell proliferation to renew damaged cell tissues, so it plays a pivotal role in retaining tissue homeostasis [82]. However, chronic inflammation is also known to induce tumorigenesis. The tumor microenvironment is initiated by the excessive production of inflammatory cytokines. Thus, this phenomenon merits study. Many cytokines and chemokines can be induced in hypoxic conditions in this tumorigenic environment [83]. Skin cancer is a malignant tumor, particularly in Caucasians, with about 1 million cases occurring annually in the United States [84,85,86]. Skin tumors have been named according to their involved cells and clinical behavior. There are three types of skin tumors: basal cell carcinoma; cutaneous malignant melanoma (CM); and non-melanocytic skin cancer (NMSC), which is also known as squamous cell carcinoma [84,87]. Chronic UV exposure most commonly leads to skin cancer among all known risk environments and affects gene mutation, immunosuppression, and oxidative stress [88,89,90]. From various studies, there are several approaches to suppress skin cancer, including through the PI3K/Akt/mTOR, TNF-related apoptosis-inducing ligand, JAK/STAT, and MAPK signaling pathways [91,92,93,94].
6.1. NMSC
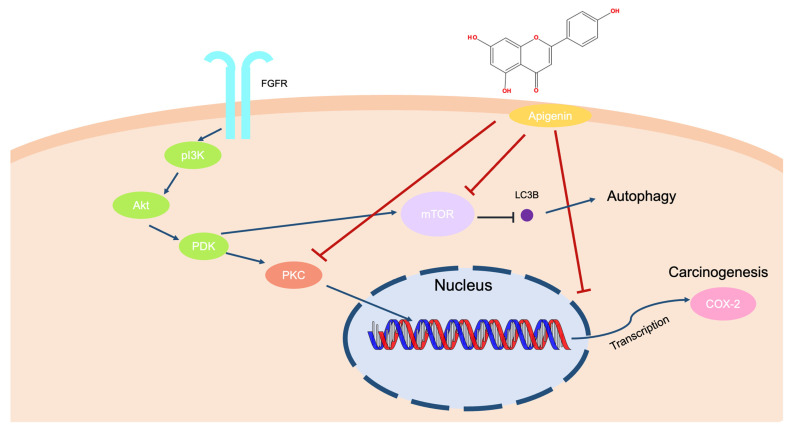
Apigenin downregulates the Akt signaling pathway in UVB-irradiated keratinocytes, blocking the mammalian target of rapamycin (mTOR) activation and suppressing the cell cycle and cell proliferation in mouse skin and keratinocytes. Meanwhile, it promotes autophagy via mTOR inhibition, which inhibits keratinocyte proliferation [95,96]. In primary human epidermal keratinocytes and a skin cutaneous squamous cell carcinoma cell line (COLO-16), treatment with apigenin decreased the conversion of the microtubule-associated protein 1 light chain 3 (LC3) and GFP-LC3 puncta [97]. Apigenin also inhibited skin carcinogenesis by downregulating the COX-2 expression level in UVB-irradiated mouse skins [98]. It is commonly known that 12-O-tetradecanoylphorbol-13-acetate (TPA) can induce a tumor by binding and activating the protein kinase C signaling pathways [99,100]. Apigenin treatment suppressed PKC activity dose-dependently and inhibited TPA-mediated carcinogenesis in mouse skin [101,102]. In summary, it is speculated that apigenin treatment can reduce tumorigenic responses by inducing autophagy and via the inactivation of Akt and PKC in keratinocytes (Figure 7).
Figure 7.
Mechanism of anti-carcinogenesis effects according to apigenin treatment in an NMSC model.
6.2. CMs
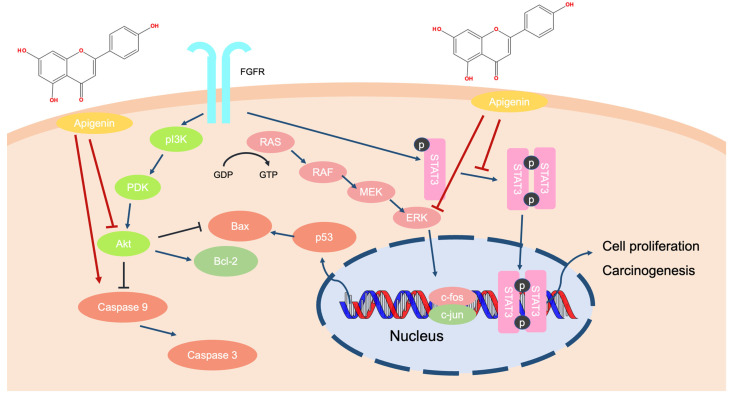
Apigenin inhibited the proliferation of melanoma cell lines by downregulating the AKT signaling pathway, which promotes cell apoptosis [103]. Moreover, apigenin treatment suppressed melanoma metastasis to the lungs in C57BL/6 mice and inhibited the phosphorylation of STAT3 in melanoma cells [104]. The presence of apigenin induced anti-melanoma effects by triggering the apoptosis of A375SM cells. Apoptotic proteins, including the caspases, p53, Bcl-2-associated X protein, and poly ADP-ribose polymerase (PARP), were upregulated by apigenin treatment. The compound also downregulated the levels of Akt, STAT3, and MAPK in melanoma cells [105]. Therefore, it is suggested that apigenin could inhibit carcinogenesis by inducing apoptosis in melanoma cells and downregulating the activities of some important survival factors, such as STAT3, Akt, and MAPK proteins (Figure 8).
Figure 8.
The molecular process of apigenin in melanoma cells.
7. The Therapeutic Effects of Apigenin on Vitiligo
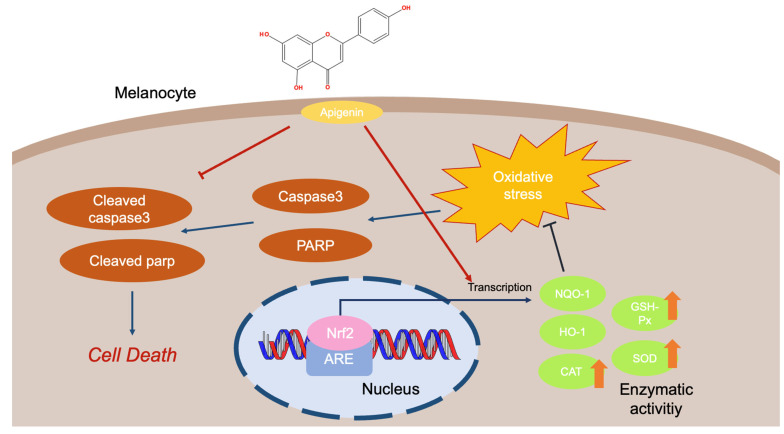
Vitiligo has been reported as a pigmentary disorder impacting about 1% of the world’s population [106]. The symptoms of this disease include inconsistent and various sizes of white spots found on the skin and a change of hair color to white [107,108,109]. The main cause of vitiligo is concerned with the autoimmune chronic destruction of melanocytes. The death of pigment cells expands the white lesions on the skin. Therefore, the therapeutic strategy has focused on preventing the apoptosis of melanocytes from oxidative stress and suppressing the proinflammatory response [110]. Of many different compounds, apigenin was reported to upregulate antioxidant enzyme activities, such as superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GSH-Px). Moreover, apigenin was found to promote the gene expression level that is involved in the antioxidant process the at mRNA and protein levels [111]. Meanwhile, it was revealed that apigenin can affect dopamine (DA)-triggered apoptosis in melanocytes by downregulating cleaved PARP and cleaved caspase 3 levels [112]. Apigenin also protected melanocytes from apoptosis by blocking the phosphorylation of Akt, p38, and JNK, which are induced by DA [112] (Figure 9).
Figure 9.
Antioxidant response of apigenin to protect melanocytes against oxidative stress.
8. Conclusions
This review described the attenuating effects of apigenin on skin inflammatory conditions and cancer as summarized in Table 2. A natural flavonoid, apigenin showed the greatest activity by attenuating the symptoms of skin inflammatory diseases and tumorigenic responses. Apigenin downregulated inflammatory cytokine expression by suppressing the AP-1, MAPK, and NF-κB pathways in keratinocytes. In addition, apigenin induced autophagy by decreasing mTOR activity and inactivating Akt and PKC activities. Moreover, apigenin protects the cell from oxidative stress-induced cell death. Through treatment, apigenin could prevent skin inflammatory responses to retain the proper regulation of inflammatory cells. Furthermore, apigenin could affect the synthesis of skin barrier factors and Ca2+ influx. Based on this review, apigenin could be applied to treat skin inflammatory diseases and cancer.
Table 2.
Mechanism of apigenin against inflammatory skin diseases and cancers.
| The Type of Skin Disease | Mechanisms of Apigenin | Test Model | Dose | References |
|---|---|---|---|---|
| UV-mediated inflammation | Downregulates Src and COX-2 levels. | In vitro | 10, 20, 40, 50 μM | [36,37] |
| Regulates the level of apoptotic proteins and anti-apoptotic proteins. | In vitro | 7, 15 μM | [38,39] | |
| Inhibits MMP-1 expression by suppressing Ca2+ influx. | [40,41] | |||
| Suppresses the MAPK and AP-1 signaling pathways. | In vitro | 1, 5, 10, 20 μM | ||
| Atopic dermatitis | Suppresses phosphorylation of STAT6 in IL-4 stimulated mouse spleen cells. | Ex vivo | 25 μM | [49] |
| Ameliorates damaged skin lesions induced by picrylchloride(piCl). | In vivo | 0.05% feed to mice | ||
| Downregulates the protein levels of the NF-κB, MAPK pathways. | In vitro | 10, 20, 30 μM | [50] | |
| Pruritus | Suppresses IL-31 levels by inhibiting the NF-κB and MAPK signaling pathways. | In vitro | 10, 30 µM | [50] |
| Regulates Th1/Th2 balance by inhibiting the NF-κB pathway, and levels of histamine, IgE, and STAT1 expression. | In vivo | 5, 10, 20 mg/kg of mice | [75] | |
| Enhances the Th1 response by decreasing the expression of IFN-γ, and T-box proteins in T cells. | ||||
| Shows low expression of IL-31, IL-33 in apigenin-treated microglial cells via downregulating ERK and JNK expression. | In vitro | 5, 10, 20, 40, 60, 80, 100 µM | [76] | |
| Inactivates MAPK and NF-κB proteins. | In vitro | 30, 60 µM | [77] | |
| Psoriasis | Promotes the synthesis of skin barrier factors. | In vivo | 60 µL of 0.1% apigenin in 100% ethanol | [80] |
| Downregulates the mRNA expression of inflammatory cytokines in LPS-treated DCs. | In vitro | 8, 20 μM | [81] | |
| Skin cancer | Downregulates mTOR and AKT signaling pathways. | In vitro | 25 μM | [95,96] |
| In vivo | 5 μM in 0.2 mL DMSO/acetone (1:9) vehicle mix of mice | |||
| Induces autophagy by inhibiting mTOR expression and the conversion of LC3. | In vitro | 6, 12, 25, 50 μM | [97] | |
| Decreases carcinogenesis in TPA-mediated mouse skin and PKC activity,. | In vivo | 5, 25 μM to mice 10, 50, 100 μM |
[101,102] | |
| In vitro | ||||
| Attenuates melanoma metastases to the lung by decreasing STAT3 levels. | In vivo | 150 mg/kg of mice | [104] | |
| Promotes the expression of apoptotic proteins in A375SM cells. Inactivates the Akt and MAPK pathway proteins. |
In vitro | 25, 50, 75, 100 µM | [103,105] | |
| In vivo | 25, 50 mg/kg of mice | |||
| Vitiligo | Promotes antioxidant enzyme activity in dose-dependent ways. Increases the expression of antioxidant genes at the mRNA and protein levels. |
In vitro | 1, 5, 10, 20 µM | [111] |
| Protects pigment cells from DA-induced apoptosis by decreasing the level of apoptotic agents. | In vitro | 10 µM | [112] | |
| Inactivates p38, JNK, and Akt levels in the presence of DA. |
9. Perspective
Apigenin is a bioactive compound used as a therapeutic agent for various diseases, such as diabetes, Alzheimer’s disease, cancer, and amnesia [113,114,115,116]. Treatment with apigenin has led to decreased levels of many inflammatory cytokines. According to study results, apigenin could inhibit the inflammatory response in the skin by downregulating transcription factors, such as AP-1, NF-κB, and STAT. These mechanisms not only back up the excellence of apigenin, but also suggest the possibility of using it as a drug for inflammatory skin diseases. There are now many trials assessing active natural compounds as substitutes for chemically synthetic drugs because of severe side effects associated with the latter [117,118]. Previous studies administered apigenin by applying it to damaged skin or cells to attenuate skin inflammation. Nowadays, apigenin products manufactured with chamomile extracts are being sold in markets to attenuate stress hormones, and bad dreams, as well as in the form of commercially available capsules that provide powerful antioxidant supplements to promote healthy aging and skin health [119]. However, flavonoids can be degraded by high temperatures [120], thus studies aimed at stabilizing apigenin therapeutics should be performed. We suggest that apigenin can not only be used as a therapeutic material, but also as a health supplement for skin diseases, based upon the various studies discussed in this review.
Abbreviations
| LC | Langerhans cells |
| UV | Ultraviolet radiation |
| Treg | Regulatory T cells |
| AD | Atopic dermatitis |
| TNF-α | Tumor necrosis factor-α |
| IL-6 | Interleukin-6 |
| COX-2 | Cyclooxygenase-2 |
| Src | Non-receptor tyrosine kinase |
| CPD | Cyclobutene pyrimidine dimers |
| NER | Nucleotide excision repair |
| MMP-1 | Metalloproteinases-1 |
| MAPK | Mitogen-activated protein kinase |
| AP-1 | Activator protein 1 |
| nHDF | Normal Human Dermal Fibroblasts |
| IgE | Immunoglobulin E |
| IFN-γ | Interferon-gamma |
| PiCl | Picrylchloride |
| STAT6 | Signal transducers and activators of transcription 6 |
| IL-4 | Interleukin-4 |
| IL-31 | Interleukin-31 |
| HMC-1 | Human mast cells |
| NF-κB | Nuclear factor kappa-light-chain-enhancer of activated B cells |
| IKK | Inhibitor of NF-κB kinase |
| IκB | Inhibitor of kappaB |
| JNK | c-Jun N-terminal kinase |
| ERK | Extracellular signal-regulated kinase |
| CCL26 | Eosinophil chemokine |
| IL-31RA | Interleukin-31 receptor A |
| ILC2 | Innate lymphoid cells-2 |
| CXCR3 | C-X-C Motif Chemokine Receptor 3 |
| TSLP | Thymic stromal lymphopoietin |
| CCL | C-C motif chemokine ligand |
| NK cells | Natural killer cells |
| OVA | Ovalbumin |
| T-bet | T-box protein expressed in T cells |
| IL-23 | Interleukin-23 |
| LPS | Lipopolysaccharide |
| TLR4 | Toll-like receptor 4 |
| TME | Tumor microenvironments |
| BCCs | Basal cell carcinomas |
| CMs | Cutaneous malignant melanomas |
| NMSC | Nonmelanocytic skin cancer |
| SCCs | Squamous cell carcinomas |
| TRAIL | TNF-related apoptosis-inducing ligand |
| mTOR | Mammalian target of rapamycin |
| HEKs | Human epidermal keratinocytes |
| COLO-16 | Skin cutaneous squamous cell carcinoma cell line |
| LC3 | Light chain 3 |
| TPA | 12-O-tetradecanoylphorbol-13-acetate |
| Bax | Bcl-2-associated X protein |
| PARP | Poly ADP-ribose polymerase |
| SOD | Superoxide dismutase |
| CAT | Catalase |
| GSH-Px | Glutathione peroxidase |
Author Contributions
J.H.Y., M.-Y.K. and J.Y.C. conceptualized this review; J.H.Y. visualized the figure for the paper; J.H.Y., M.-Y.K. and J.Y.C. wrote the manuscript; M.-Y.K. and J.Y.C. administrated the writing and editing. All authors have read and agreed to the published version of the manuscript.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The data are contained within the article.
Conflicts of Interest
The authors declare no conflict of interest.
Funding Statement
This research was supported by grant from the Basic Science Research Program through the National Research Foundation of Korea (NRF), funded by the Korean Ministry of Education (Grant No.: 2017R1A6A1A03015642).
Footnotes
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content.
References
- 1.Hsu Y.-C., Li L., Fuchs E. Emerging interactions between skin stem cells and their niches. Nat. Med. 2014;20:847–856. doi: 10.1038/nm.3643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Belkaid Y., Tamoutounour S. The influence of skin microorganisms on cutaneous immunity. Nat. Rev. Immunol. 2016;16:353–366. doi: 10.1038/nri.2016.48. [DOI] [PubMed] [Google Scholar]
- 3.Byrd A.L., Belkaid Y., Segre J.A. The human skin microbiome. Nat. Rev. Microbiol. 2018;16:143–155. doi: 10.1038/nrmicro.2017.157. [DOI] [PubMed] [Google Scholar]
- 4.Nestle F.O., Di Meglio P., Qin J.-Z., Nickoloff B.J. Skin immune sentinels in health and disease. Nat. Rev. Immunol. 2009;9:679–691. doi: 10.1038/nri2622. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Abdo J.M., Sopko N.A., Milner S.M. The applied anatomy of human skin: A model for regeneration. Wound Med. 2020;28:100179. doi: 10.1016/j.wndm.2020.100179. [DOI] [Google Scholar]
- 6.Tang A., Amagai M., Granger L.G., Stanley J.R., Uddy M.C. Adhesion of epidermal Langerhans cells to keratinocytes mediated by E-cadherin. Nature. 1993;361:82–85. doi: 10.1038/361082a0. [DOI] [PubMed] [Google Scholar]
- 7.Schwarz A., Noordegraaf M., Maeda A., Torii K., Clausen B.E., Schwarz T. Langerhans cells are required for UVR-induced immunosuppression. J. Investig. Dermatol. 2010;130:1419–1427. doi: 10.1038/jid.2009.429. [DOI] [PubMed] [Google Scholar]
- 8.Idoyaga J., Fiorese C., Zbytnuik L., Lubkin A., Miller J., Malissen B., Mucida D., Merad M., Steinman R.M. Specialized role of migratory dendritic cells in peripheral tolerance induction. J. Clin. Investig. 2013;123:844–854. doi: 10.1172/JCI65260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Scharschmidt T.C., Fischbach M.A. What lives on our skin: Ecology, genomics and therapeutic opportunities of the skin microbiome. Drug Discov. Today: Dis. Mech. 2013;10:e83–e89. doi: 10.1016/j.ddmec.2012.12.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Belkaid Y., Segre J.A. Dialogue between skin microbiota and immunity. Science. 2014;346:954–959. doi: 10.1126/science.1260144. [DOI] [PubMed] [Google Scholar]
- 11.Grice E.A. The intersection of microbiome and host at the skin interface: Genomic-and metagenomic-based insights. Genome Res. 2015;25:1514–1520. doi: 10.1101/gr.191320.115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.McLoughlin I.J., Wright E.M., Tagg J.R., Jain R., Hale J.D. Skin microbiome—The next frontier for probiotic intervention. Probiotics Antimicrob. Proteins. 2022;14:630–647. doi: 10.1007/s12602-021-09824-1. [DOI] [PubMed] [Google Scholar]
- 13.Iebba V., Totino V., Gagliardi A., Santangelo F., Cacciotti F., Trancassini M., Mancini C., Cicerone C., Corazziari E., Pantanella F. Eubiosis and dysbiosis: The two sides of the microbiota. New Microbiol. 2016;39:1–12. [PubMed] [Google Scholar]
- 14.Di Meglio P., Perera G.K., Nestle F.O. The multitasking organ: Recent insights into skin immune function. Immunity. 2011;35:857–869. doi: 10.1016/j.immuni.2011.12.003. [DOI] [PubMed] [Google Scholar]
- 15.Robert C., Kupper T.S. Inflammatory Skin Diseases, T Cells, and Immune Surveillance. New Engl. J. Med. 1999;341:1817–1828. doi: 10.1056/NEJM199912093412407. [DOI] [PubMed] [Google Scholar]
- 16.Balato A., Cacciapuoti S., Di Caprio R., Marasca C., Masarà A., Raimondo A., Fabbrocini G. Human Microbiome: Composition and Role in Inflammatory Skin Diseases. Arch. Immunol. Et Ther. Exp. 2019;67:1–18. doi: 10.1007/s00005-018-0528-4. [DOI] [PubMed] [Google Scholar]
- 17.Maru G.B., Gandhi K., Ramchandani A., Kumar G. The role of inflammation in skin cancer. Adv. Exp. Med. Biol. 2014;816:437–469. doi: 10.1007/978-3-0348-0837-8_17. [DOI] [PubMed] [Google Scholar]
- 18.Abdulla A., Adams N., Bone M., Elliott A.M., Gaffin J., Jones D., Knaggs R., Martin D., Sampson L., Schofield P. Guidance on the management of pain in older people. Age Ageing. 2013;42:i1–i57. doi: 10.1093/ageing/afs199. [DOI] [PubMed] [Google Scholar]
- 19.Breivik H., Collett B., Ventafridda V., Cohen R., Gallacher D. Survey of chronic pain in Europe: Prevalence, impact on daily life, and treatment. Eur. J. Pain. 2006;10:287–333. doi: 10.1016/j.ejpain.2005.06.009. [DOI] [PubMed] [Google Scholar]
- 20.Conaghan P.G. A turbulent decade for NSAIDs: Update on current concepts of classification, epidemiology, comparative efficacy, and toxicity. Rheumatol. Int. 2012;32:1491–1502. doi: 10.1007/s00296-011-2263-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Melmed S., Polonsky K.S., Larsen P.R., Kronenberg H.M. Williams Textbook of Endocrinology E-Book. Elsevier Health Sciences; Amsterdam, The Netherlands: 2015. [Google Scholar]
- 22.Emery P. Treatment of rheumatoid arthritis. Bmj. 2006;332:152–155. doi: 10.1136/bmj.332.7534.152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Borquaye L.S., Darko G., Laryea M.K., Roberts V., Boateng R., Gasu E.N. Anti-inflammatory activities of extracts from Oliva sp., Patella rustica, and Littorina littorea collected from Ghana’s coastal shorelines. Cogent Biol. 2017;3:1364063. doi: 10.1080/23312025.2017.1364063. [DOI] [Google Scholar]
- 24.Tang D., Chen K., Huang L., Li J. Pharmacokinetic properties and drug interactions of apigenin, a natural flavone. Expert Opin. Drug Metab. Toxicol. 2017;13:323–330. doi: 10.1080/17425255.2017.1251903. [DOI] [PubMed] [Google Scholar]
- 25.Wang M., Firrman J., Liu L., Yam K. A Review on Flavonoid Apigenin: Dietary Intake, ADME, Antimicrobial Effects, and Interactions with Human Gut Microbiota. BioMed Res. Int. 2019;2019:7010467. doi: 10.1155/2019/7010467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Ornano L., Venditti A., Donno Y., Sanna C., Ballero M., Bianco A. Phytochemical analysis of non-volatile fraction of Artemisia caerulescens subsp. densiflora (Viv.)(Asteraceae), an endemic species of La Maddalena Archipelago (Sardinia–Italy) Nat. Prod. Res. 2016;30:920–925. doi: 10.1080/14786419.2015.1079189. [DOI] [PubMed] [Google Scholar]
- 27.Venditti A., Maggi F., Vittori S., Papa F., Serrilli A.M., Di Cecco M., Ciaschetti G., Mandrone M., Poli F., Bianco A. Antioxidant and α-glucosidase inhibitory activities of Achillea tenorii. Pharm. Biol. 2015;53:1505–1510. doi: 10.3109/13880209.2014.991833. [DOI] [PubMed] [Google Scholar]
- 28.Sharifi-Rad M., Nazaruk J., Polito L., Morais-Braga M.F.B., Rocha J.E., Coutinho H.D.M., Salehi B., Tabanelli G., Montanari C., del Mar Contreras M. Matricaria genus as a source of antimicrobial agents: From farm to pharmacy and food applications. Microbiol. Res. 2018;215:76–88. doi: 10.1016/j.micres.2018.06.010. [DOI] [PubMed] [Google Scholar]
- 29.Venditti A., Frezza C., Sciubba F., Serafini M., Bianco A., Cianfaglione K., Lupidi G., Quassinti L., Bramucci M., Maggi F. Volatile components, polar constituents and biological activity of tansy daisy (Tanacetum macrophyllum (Waldst. et Kit.) Schultz Bip.) Ind. Crops Prod. 2018;118:225–235. doi: 10.1016/j.indcrop.2018.03.056. [DOI] [Google Scholar]
- 30.Zhou Z., Zhang Y., Lin L., Zhou J. Apigenin suppresses the apoptosis of H9C2 rat cardiomyocytes subjected to myocardial ischemia-reperfusion injury via upregulation of the PI3K/Akt pathway. Mol. Med. Rep. 2018;18:1560–1570. doi: 10.3892/mmr.2018.9115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Wang J., Liu Y.-T., Xiao L., Zhu L., Wang Q., Yan T. Anti-Inflammatory Effects of Apigenin in Lipopolysaccharide-Induced Inflammatory in Acute Lung Injury by Suppressing COX-2 and NF-kB Pathway. Inflammation. 2014;37:2085–2090. doi: 10.1007/s10753-014-9942-x. [DOI] [PubMed] [Google Scholar]
- 32.Jung U.J., Cho Y.-Y., Choi M.-S. Apigenin Ameliorates Dyslipidemia, Hepatic Steatosis and Insulin Resistance by Modulating Metabolic and Transcriptional Profiles in the Liver of High-Fat Diet-Induced Obese Mice. Nutrients. 2016;8:305. doi: 10.3390/nu8050305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Mesa-Arango A.C., Flórez-Muñoz S.V., Sanclemente G. Mechanisms of skin aging. Iatreia. 2017;30:160–170. doi: 10.17533/udea.iatreia.v30n2a05. [DOI] [Google Scholar]
- 34.Lephart E.D. Equol’s anti-aging effects protect against environmental assaults by increasing skin antioxidant defense and ECM proteins while decreasing oxidative stress and inflammation. Cosmetics. 2018;5:16. doi: 10.3390/cosmetics5010016. [DOI] [Google Scholar]
- 35.Barker J.N., Griffiths C.E., Nickoloff B.J., Mitra R., Dixit V.M. Keratinocytes as initiators of inflammation. Lancet. 1991;337:211–214. doi: 10.1016/0140-6736(91)92168-2. [DOI] [PubMed] [Google Scholar]
- 36.Tong X., Dross R.T.V., Abu-Yousif A., Morrison A.R., Pelling J.C. Apigenin Prevents UVB-Induced Cyclooxygenase 2 Expression: Coupled mRNA Stabilization and Translational Inhibition. Mol. Cell. Biol. 2007;27:283–296. doi: 10.1128/MCB.01282-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Byun S., Park J., Lee E., Lim S., Yu J.G., Lee S.J., Chen H., Dong Z., Lee K.W., Lee H.J. Src kinase is a direct target of apigenin against UVB-induced skin inflammation. Carcinogenesis. 2012;34:397–405. doi: 10.1093/carcin/bgs358. [DOI] [PubMed] [Google Scholar]
- 38.Britto S.M., Shanthakumari D., Agilan B., Radhiga T., Kanimozhi G., Prasad N.R. Apigenin prevents ultraviolet-B radiation induced cyclobutane pyrimidine dimers formation in human dermal fibroblasts. Mutat. Res. /Genet. Toxicol. Environ. Mutagen. 2017;821:28–35. doi: 10.1016/j.mrgentox.2017.06.002. [DOI] [PubMed] [Google Scholar]
- 39.García Forero A., Villamizar Mantilla D.A., Núñez L.A., Ocazionez R.E., Stashenko E.E., Fuentes J.L. Photoprotective and Antigenotoxic Effects of the Flavonoids Apigenin, Naringenin and Pinocembrin. Photochem. Photobiol. 2019;95:1010–1018. doi: 10.1111/php.13085. [DOI] [PubMed] [Google Scholar]
- 40.Hwang Y.P., Oh K.N., Yun H.J., Jeong H.G. The flavonoids apigenin and luteolin suppress ultraviolet A-induced matrix metalloproteinase-1 expression via MAPKs and AP-1-dependent signaling in HaCaT cells. J Derm. Sci. 2011;61:23–31. doi: 10.1016/j.jdermsci.2010.10.016. [DOI] [PubMed] [Google Scholar]
- 41.Choi S., Youn J., Kim K., Joo da H., Shin S., Lee J., Lee H.K., An I.S., Kwon S., Youn H.J., et al. Apigenin inhibits UVA-induced cytotoxicity in vitro and prevents signs of skin aging in vivo. Int J Mol Med. 2016;38:627–634. doi: 10.3892/ijmm.2016.2626. [DOI] [PubMed] [Google Scholar]
- 42.Meisel J.S., Sfyroera G., Bartow-McKenney C., Gimblet C., Bugayev J., Horwinski J., Kim B., Brestoff J.R., Tyldsley A.S., Zheng Q. Commensal microbiota modulate gene expression in the skin. Microbiome. 2018;6:1–15. doi: 10.1186/s40168-018-0404-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Kong H.H., Oh J., Deming C., Conlan S., Grice E.A., Beatson M.A., Nomicos E., Polley E.C., Komarow H.D., Murray P.R. Temporal shifts in the skin microbiome associated with disease flares and treatment in children with atopic dermatitis. Genome Res. 2012;22:850–859. doi: 10.1101/gr.131029.111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Shi B., Bangayan N.J., Curd E., Taylor P.A., Gallo R.L., Leung D.Y., Li H. The skin microbiome is different in pediatric versus adult atopic dermatitis. J. Allergy Clin. Immunol. 2016;138:1233–1236. doi: 10.1016/j.jaci.2016.04.053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Kim B.E., Leung D.Y. Epidermal barrier in atopic dermatitis. Allergy Asthma Immunol. Res. 2012;4:12–16. doi: 10.4168/aair.2012.4.1.12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Rerknimitr P., Otsuka A., Nakashima C., Kabashima K. The etiopathogenesis of atopic dermatitis: Barrier disruption, immunological derangement, and pruritus. Inflamm. Regen. 2017;37:1–15. doi: 10.1186/s41232-017-0044-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.David Boothe W., Tarbox J.A., Tarbox M.B. Atopic dermatitis: Pathophysiology. Manag. Atopic Dermat. 2017:21–37. doi: 10.1007/978-3-319-64804-0_3. [DOI] [PubMed] [Google Scholar]
- 48.Tsakok T., Woolf R., Smith C., Weidinger S., Flohr C. Atopic dermatitis: The skin barrier and beyond. Br. J. Dermatol. 2019;180:464–474. doi: 10.1111/bjd.16934. [DOI] [PubMed] [Google Scholar]
- 49.Yano S., Umeda D., Yamashita S., Yamada K., Tachibana H. Dietary apigenin attenuates the development of atopic dermatitis-like skin lesions in NC/Nga mice. J. Nutr. Biochem. 2009;20:876–881. doi: 10.1016/j.jnutbio.2008.08.002. [DOI] [PubMed] [Google Scholar]
- 50.Che D.N., Cho B.O., Shin J.Y., Kang H.J., Kim J.-S., Oh H., Kim Y.-S., Jang S.I. Apigenin Inhibits IL-31 Cytokine in Human Mast Cell and Mouse Skin Tissues. Molecules. 2019;24:1290. doi: 10.3390/molecules24071290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Oetjen L.K., Mack M.R., Feng J., Whelan T.M., Niu H., Guo C.J., Chen S., Trier A.M., Xu A.Z., Tripathi S.V. Sensory neurons co-opt classical immune signaling pathways to mediate chronic itch. Cell. 2017;171:217–228. e213. doi: 10.1016/j.cell.2017.08.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Gandhi N.A., Bennett B.L., Graham N.M., Pirozzi G., Stahl N., Yancopoulos G.D. Targeting key proximal drivers of type 2 inflammation in disease. Nat. Rev. Drug Discov. 2016;15:35–50. doi: 10.1038/nrd4624. [DOI] [PubMed] [Google Scholar]
- 53.Cornelissen C., Marquardt Y., Czaja K., Wenzel J., Frank J., Lüscher-Firzlaff J., Lüscher B., Baron J.M. IL-31 regulates differentiation and filaggrin expression in human organotypic skin models. J. Allergy Clin. Immunol. 2012;129:426–433. e428. doi: 10.1016/j.jaci.2011.10.042. [DOI] [PubMed] [Google Scholar]
- 54.Yosipovitch G., Berger T., Fassett M. Neuroimmune interactions in chronic itch of atopic dermatitis. J. Eur. Acad. Dermatol. Venereol. 2020;34:239–250. doi: 10.1111/jdv.15973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Kezic S., O’Regan G.M., Lutter R., Jakasa I., Koster E.S., Saunders S., Caspers P., Kemperman P.M.J.H., Puppels G.J., Sandilands A., et al. Filaggrin loss-of-function mutations are associated with enhanced expression of IL-1 cytokines in the stratum corneum of patients with atopic dermatitis and in a murine model of filaggrin deficiency. J. Allergy Clin. Immunol. 2012;129:1031–1039. e1031. doi: 10.1016/j.jaci.2011.12.989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Archer N.K., Jo J.-H., Lee S.K., Kim D., Smith B., Ortines R.V., Wang Y., Marchitto M.C., Ravipati A., Cai S.S. Injury, dysbiosis, and filaggrin deficiency drive skin inflammation through keratinocyte IL-1α release. J. Allergy Clin. Immunol. 2019;143:1426–1443. e1426. doi: 10.1016/j.jaci.2018.08.042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Roan F., Obata-Ninomiya K., Ziegler S.F. Epithelial cell–derived cytokines: More than just signaling the alarm. J. Clin. Investig. 2019;129:1441–1451. doi: 10.1172/JCI124606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Ryu W.-I., Lee H., Bae H.C., Ryu H.J., Son S.W. IL-33 down-regulates filaggrin expression by inducing STAT3 and ERK phosphorylation in human keratinocytes. J. Dermatol. Sci. 2016;82:131–134. doi: 10.1016/j.jdermsci.2016.01.011. [DOI] [PubMed] [Google Scholar]
- 59.Savinko T., Matikainen S., Saarialho-Kere U., Lehto M., Wang G., Lehtimäki S., Karisola P., Reunala T., Wolff H., Lauerma A. IL-33 and ST2 in atopic dermatitis: Expression profiles and modulation by triggering factors. J. Investig. Dermatol. 2012;132:1392–1400. doi: 10.1038/jid.2011.446. [DOI] [PubMed] [Google Scholar]
- 60.Albanesi C., Scarponi C., Sebastiani S., Cavani A., Federici M., De Pità O., Puddu P., Girolomoni G. IL-4 enhances keratinocyte expression of CXCR3 agonistic chemokines. J. Immunol. 2000;165:1395–1402. doi: 10.4049/jimmunol.165.3.1395. [DOI] [PubMed] [Google Scholar]
- 61.Wang Y.H., Liu Y.J. Thymic stromal lymphopoietin, OX40-ligand, and interleukin-25 in allergic responses. Clin. Exp. Allergy. 2009;39:798–806. doi: 10.1111/j.1365-2222.2009.03241.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Cianferoni A., Spergel J. The importance of TSLP in allergic disease and its role as a potential therapeutic target. Expert Rev. Clin. Immunol. 2014;10:1463–1474. doi: 10.1586/1744666X.2014.967684. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Xie Y., Takai T., Chen X., Okumura K., Ogawa H. Long TSLP transcript expression and release of TSLP induced by TLR ligands and cytokines in human keratinocytes. J. Dermatol. Sci. 2012;66:233–237. doi: 10.1016/j.jdermsci.2012.03.007. [DOI] [PubMed] [Google Scholar]
- 64.Chieosilapatham P., Ogawa H., Niyonsaba F. Current insights into the role of human β-defensins in atopic dermatitis. Clin. Exp. Immunol. 2017;190:155–166. doi: 10.1111/cei.13013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Ständer S., Steinhoff M., Schmelz M., Weisshaar E., Metze D., Luger T. Neurophysiology of pruritus: Cutaneous elicitation of itch. Arch. Dermatol. 2003;139:1463–1470. doi: 10.1001/archderm.139.11.1463. [DOI] [PubMed] [Google Scholar]
- 66.Weisshaar E., Apfelbacher C., Jäger G., Zimmermann E., Bruckner T., Diepgen T., Gollnick H. Pruritus as a leading symptom: Clinical characteristics and quality of life in German and Ugandan patients. Br. J. Dermatol. 2006;155:957–964. doi: 10.1111/j.1365-2133.2006.07430.x. [DOI] [PubMed] [Google Scholar]
- 67.Lyell A. The itching patient: A review of the causes of pruritus. Scott. Med. J. 1972;17:334–347. doi: 10.1177/003693307201701005. [DOI] [PubMed] [Google Scholar]
- 68.Lavery M.J., Kinney M.O., Mochizuki H., Craig J., Yosipovitch G. Pruritus: An overview. What drives people to scratch an itch? Ulst. Med. J. 2016;85:164. [PMC free article] [PubMed] [Google Scholar]
- 69.Schmelz M. Itch—Mediators and mechanisms. J. Dermatol. Sci. 2002;28:91–96. doi: 10.1016/S0923-1811(01)00167-0. [DOI] [PubMed] [Google Scholar]
- 70.Shibuya R., Takimoto-Ito R., Kambe N., Kabashima K. A new era with the development of cytokine-based therapy for pruritus. J. Investig. Dermatol. 2022;142:47–52. doi: 10.1016/j.jid.2021.09.023. [DOI] [PubMed] [Google Scholar]
- 71.Zhang Q., Putheti P., Zhou Q., Liu Q., Gao W. Structures and biological functions of IL-31 and IL-31 receptors. Cytokine Growth Factor Rev. 2008;19:347–356. doi: 10.1016/j.cytogfr.2008.08.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Takamori A., Nambu A., Sato K., Yamaguchi S., Matsuda K., Numata T., Sugawara T., Yoshizaki T., Arae K., Morita H. IL-31 is crucial for induction of pruritus, but not inflammation, in contact hypersensitivity. Sci. Rep. 2018;8:1–11. doi: 10.1038/s41598-018-25094-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Hudson C.A., Christophi G.P., Gruber R.C., Wilmore J.R., Lawrence D.A., Massa P.T. Induction of IL-33 expression and activity in central nervous system glia. J. Leukoc. Biol. 2008;84:631–643. doi: 10.1189/jlb.1207830. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Yasuoka S., Kawanokuchi J., Parajuli B., Jin S., Doi Y., Noda M., Sonobe Y., Takeuchi H., Mizuno T., Suzumura A. Production and functions of IL-33 in the central nervous system. Brain Res. 2011;1385:8–17. doi: 10.1016/j.brainres.2011.02.045. [DOI] [PubMed] [Google Scholar]
- 75.Chen F., He D., Yan B. Apigenin Attenuates Allergic Responses of Ovalbumin-Induced Allergic Rhinitis Through Modulation of Th1/Th2 Responses in Experimental Mice. Dose-Response. 2020;18 doi: 10.1177/1559325820904799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Che D.N., Cho B.O., Kim J.-S., Shin J.Y., Kang H.J., Jang S.I. Effect of Luteolin and Apigenin on the Production of Il-31 and Il-33 in Lipopolysaccharides-Activated Microglia Cells and Their Mechanism of Action. Nutrients. 2020;12:811. doi: 10.3390/nu12030811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Che D.N., Cho B.O., Kim J.-S., Shin J.Y., Kang H.J., Jang S.I. Luteolin and Apigenin Attenuate LPS-Induced Astrocyte Activation and Cytokine Production by Targeting MAPK, STAT3, and NF-κB Signaling Pathways. Inflammation. 2020;43:1716. doi: 10.1007/s10753-020-01245-6. [DOI] [PubMed] [Google Scholar]
- 78.Armstrong A.W., Read C. Pathophysiology, Clinical Presentation, and Treatment of Psoriasis: A Review. JAMA. 2020;323:1945–1960. doi: 10.1001/jama.2020.4006. [DOI] [PubMed] [Google Scholar]
- 79.Rendon A., Schäkel K. Psoriasis Pathogenesis and Treatment. Int J Mol Sci. 2019;20:1475. doi: 10.3390/ijms20061475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Hou M., Sun R., Hupe M., Kim P.L., Park K., Crumrine D., Lin T.-K., Santiago J.L., Mauro T.M., Elias P.M., et al. Topical apigenin improves epidermal permeability barrier homoeostasis in normal murine skin by divergent mechanisms. Exp. Dermatol. 2013;22:210–215. doi: 10.1111/exd.12102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Ginwala R., Bhavsar R., Moore P., Bernui M., Singh N., Bearoff F., Nagarkatti M., Khan Z.K., Jain P. Apigenin Modulates Dendritic Cell Activities and Curbs Inflammation Via RelB Inhibition in the Context of Neuroinflammatory Diseases. J Neuroimmune Pharm. 2021;16:403–424. doi: 10.1007/s11481-020-09933-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Medzhitov R. Origin and physiological roles of inflammation. Nature. 2008;454:428–435. doi: 10.1038/nature07201. [DOI] [PubMed] [Google Scholar]
- 83.Naylor M., Stamp G.W., Foulkes W.D., Eccles D., Balkwill F.R. Tumor necrosis factor and its receptors in human ovarian cancer. Potential role in disease progression. J. Clin. Investig. 1993;91:2194–2206. doi: 10.1172/JCI116446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.D’Orazio J., Jarrett S., Amaro-Ortiz A., Scott T. UV radiation and the skin. Int. J. Mol. Sci. 2013;14:12222–12248. doi: 10.3390/ijms140612222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Rogers H.W., Weinstock M.A., Harris A.R., Hinckley M.R., Feldman S.R., Fleischer A.B., Coldiron B.M. Incidence estimate of nonmelanoma skin cancer in the United States, 2006. Arch. Dermatol. 2010;146:283–287. doi: 10.1001/archdermatol.2010.19. [DOI] [PubMed] [Google Scholar]
- 86.Geller A.C., Annas G.D. Seminars in Oncology Nursing. Elsevier; Amsterdam, The Netherlands: 2003. Epidemiology of Melanoma and Nonmelanoma Skin Cancer; pp. 2–11. [DOI] [PubMed] [Google Scholar]
- 87.Armstrong B.K., Kricker A. The epidemiology of UV induced skin cancer. J. Photochem. Photobiol. B Biol. 2001;63:8–18. doi: 10.1016/S1011-1344(01)00198-1. [DOI] [PubMed] [Google Scholar]
- 88.Saladi R.N., Persaud A.N. The causes of skin cancer: A comprehensive review. Drugs Today. 2005;41:37–54. doi: 10.1358/dot.2005.41.1.875777. [DOI] [PubMed] [Google Scholar]
- 89.Nikolaou V., Stratigos A. Emerging trends in the epidemiology of melanoma. Br. J. Dermatol. 2014;170:11–19. doi: 10.1111/bjd.12492. [DOI] [PubMed] [Google Scholar]
- 90.Gordon R. Seminars in Oncology Nursing: 2013. Elsevier; Amsterdam, The Netherlands: 2003. Skin Cancer: An overview of Epidemiology and Risk Factors; pp. 160–169. [DOI] [PubMed] [Google Scholar]
- 91.Fresno Vara J.A., Casado E., de Castro J., Cejas P., Belda-Iniesta C., González-Barón M. PI3K/Akt signalling pathway and cancer. Cancer Treat. Rev. 2004;30:193–204. doi: 10.1016/j.ctrv.2003.07.007. [DOI] [PubMed] [Google Scholar]
- 92.Johnstone R.W., Frew A.J., Smyth M.J. The TRAIL apoptotic pathway in cancer onset, progression and therapy. Nat. Rev. Cancer. 2008;8:782–798. doi: 10.1038/nrc2465. [DOI] [PubMed] [Google Scholar]
- 93.Brooks A.J., Putoczki T. JAK-STAT Signalling Pathway in Cancer. Cancers. 2020;12:1971. doi: 10.3390/cancers12071971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Guo Y.J., Pan W.W., Liu S.B., Shen Z.F., Xu Y., Hu L.L. ERK/MAPK signalling pathway and tumorigenesis (Review) Exp. Med. 2020;19:1997–2007. doi: 10.3892/etm.2020.8454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Bridgeman B.B., Wang P., Ye B., Pelling J.C., Volpert O.V., Tong X. Inhibition of mTOR by apigenin in UVB-irradiated keratinocytes: A new implication of skin cancer prevention. Cell. Signal. 2016;28:460–468. doi: 10.1016/j.cellsig.2016.02.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Jangdey M.S., Gupta A., Saraf S., Saraf S. Development and optimization of apigenin-loaded transfersomal system for skin cancer delivery: In vitro evaluation. Artif. Cells Nanomed. Biotechnol. 2017;45:1452–1462. doi: 10.1080/21691401.2016.1247850. [DOI] [PubMed] [Google Scholar]
- 97.Gilardini Montani M.S., Cecere N., Granato M., Romeo M.A., Falcinelli L., Ciciarelli U., D’orazi G., Faggioni A., Cirone M. Mutant p53, stabilized by its interplay with HSP90, activates a positive feed-back loop between NRF2 and p62 that induces chemo-resistance to apigenin in pancreatic cancer cells. Cancers. 2019;11:703. doi: 10.3390/cancers11050703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Mirzoeva S., Tong X., Bridgeman B.B., Plebanek M.P., Volpert O.V. Apigenin Inhibits UVB-Induced Skin Carcinogenesis: The Role of Thrombospondin-1 as an Anti-Inflammatory Factor. Neoplasia. 2018;20:930–942. doi: 10.1016/j.neo.2018.07.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Berenblum I. A re-evaluation of the concept of cocarcinogenesis. Carcinog. Carcinog. Test. 1969;11:21–30. doi: 10.1159/000391387. [DOI] [PubMed] [Google Scholar]
- 100.Boutwell R.K. The function and mechanism of promoters of carcinogenesis. CRC Crit. Rev. Toxicol. 1974;2:419–443. doi: 10.3109/10408447309025704. [DOI] [PubMed] [Google Scholar]
- 101.Wei H., Tye L., Bresnick E., Birt D.F. Inhibitory effect of apigenin, a plant flavonoid, on epidermal ornithine decarboxylase and skin tumor promotion in mice. Cancer Res. 1990;50:499–502. [PubMed] [Google Scholar]
- 102.Huang Y.T., Kuo M.L., Liu J.Y., Huang S.Y., Lin J.K. Inhibitions of protein kinase C and proto-oncogene expressions in NIH 3T3 cells by apigenin. Eur. J. Cancer. 1996;32a:146–151. doi: 10.1016/0959-8049(95)00540-4. [DOI] [PubMed] [Google Scholar]
- 103.Zhao G., Han X., Cheng W., Ni J., Zhang Y., Lin J., Song Z. Apigenin inhibits proliferation and invasion, and induces apoptosis and cell cycle arrest in human melanoma cells. Oncol. Rep. 2017;37:2277–2285. doi: 10.3892/or.2017.5450. [DOI] [PubMed] [Google Scholar]
- 104.Cao H.-H., Chu J.-H., Kwan H.-Y., Su T., Yu H., Cheng C.-Y., Fu X.-Q., Guo H., Li T., Tse A.K.-W., et al. Inhibition of the STAT3 signaling pathway contributes to apigenin-mediated anti-metastatic effect in melanoma. Sci. Rep. 2016;6:21731. doi: 10.1038/srep21731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Woo J.S., Choo G.S., Yoo E.S., Kim S.H., Lee J.H., Han S.H., Kim H.J., Jung S.H., Park Y.S., Kim B.S., et al. Apigenin induces apoptosis by regulating Akt and MAPK pathways in human melanoma cell A375SM. Mol Med Rep. 2020;22:4877–4889. doi: 10.3892/mmr.2020.11572. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Whitton M.E., Pinart M., Batchelor J., Leonardi-Bee J., González U., Jiyad Z., Eleftheriadou V., Ezzedine K. Interventions for vitiligo. Cochrane Database Syst. Rev. 2015;2:Cd003263. doi: 10.1002/14651858.CD003263.pub5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Halder R.M., Chappell J.L. Seminars in Cutaneous Medicine and Surgery. Elsevier; Amsterdam, The Netherlands: 2009. Vitiligo Update; pp. 86–92. [DOI] [PubMed] [Google Scholar]
- 108.Mahmoud F., Abul H., Al-Saleh Q., Haines D., Burleson J., Morgan G. Peripheral T-cell activation in non-segmental vitiligo. J. Dermatol. 1998;25:637–640. doi: 10.1111/j.1346-8138.1998.tb02472.x. [DOI] [PubMed] [Google Scholar]
- 109.Mahmoud F., Abul H., Haines D., Al-Saleh C., Khajeji M., Whaley K. Decreased total numbers of peripheral blood lymphocytes with elevated percentages of CD4+ CD45RO+ and CD4+ CD25+ of T-helper cells in non-segmental vitiligo. J. Dermatol. 2002;29:68–73. doi: 10.1111/j.1346-8138.2002.tb00168.x. [DOI] [PubMed] [Google Scholar]
- 110.Taïeb A. Vitiligo as an inflammatory skin disorder: A therapeutic perspective. Pigment Cell Melanoma Res. 2012;25:9–13. doi: 10.1111/j.1755-148X.2011.00939.x. [DOI] [PubMed] [Google Scholar]
- 111.Zhang B., Wang J., Zhao G., Lin M., Lang Y., Zhang D., Feng D., Tu C. Apigenin protects human melanocytes against oxidative damage by activation of the Nrf2 pathway. Cell Stress Chaperones. 2020;25:277–285. doi: 10.1007/s12192-020-01071-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Lin M., Lu S.-s., Wang A.-x., Qi X.-y., Zhao D., Wang Z.-h., Man M.-Q., Tu C.-x. Apigenin attenuates dopamine-induced apoptosis in melanocytes via oxidative stress-related p38, c-Jun NH2-terminal kinase and Akt signaling. J. Dermatol. Sci. 2011;63:10–16. doi: 10.1016/j.jdermsci.2011.03.007. [DOI] [PubMed] [Google Scholar]
- 113.Malik S., Suchal K., Khan S.I., Bhatia J., Kishore K., Dinda A.K., Arya D.S. Apigenin ameliorates streptozotocin-induced diabetic nephropathy in rats via MAPK-NF-κB-TNF-α and TGF-β1-MAPK-fibronectin pathways. Am. J. Physiol. -Ren. Physiol. 2017;313:F414–F422. doi: 10.1152/ajprenal.00393.2016. [DOI] [PubMed] [Google Scholar]
- 114.Torkin R., Lavoie J.-F., Kaplan D.R., Yeger H. Induction of caspase-dependent, p53-mediated apoptosis by apigenin in human neuroblastoma. Mol. Cancer Ther. 2005;4:1–11. doi: 10.1158/1535-7163.1.4.1. [DOI] [PubMed] [Google Scholar]
- 115.Zhao L., Wang J.-L., Liu R., Li X.-X., Li J.-F., Zhang L. Neuroprotective, anti-amyloidogenic and neurotrophic effects of apigenin in an Alzheimer’s disease mouse model. Molecules. 2013;18:9949–9965. doi: 10.3390/molecules18089949. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Popović M., Caballero-Bleda M., Benavente-García O., Castillo J. The flavonoid apigenin delays forgetting of passive avoidance conditioning in rats. J. Psychopharmacol. 2014;28:498–501. doi: 10.1177/0269881113512040. [DOI] [PubMed] [Google Scholar]
- 117.Beg S., Swain S., Hasan H., Barkat M.A., Hussain M.S. Systematic review of herbals as potential anti-inflammatory agents: Recent advances, current clinical status and future perspectives. Pharm. Rev. 2011;5:120–137. doi: 10.4103/0973-7847.91102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Drew A.K., Myers S.P. Safety issues in herbal medicine: Implications for the health professions. Med. J. Aust. 1997;166:538–541. doi: 10.5694/j.1326-5377.1997.tb123246.x. [DOI] [PubMed] [Google Scholar]
- 119.DeRango-Adem E.F., Blay J. Does Oral Apigenin Have Real Potential for a Therapeutic Effect in the Context of Human Gastrointestinal and Other Cancers? Front. Pharmacol. 2021;12:1477. doi: 10.3389/fphar.2021.681477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Barnes J.S., Foss F.W., Jr., Schug K.A. Thermally accelerated oxidative degradation of quercetin using continuous flow kinetic electrospray-ion trap-time of flight mass spectrometry. J. Am. Soc. Mass Spectrom. 2013;24:1513–1522. doi: 10.1007/s13361-013-0698-6. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The data are contained within the article.