Abstract
Eye health is crucial, and the onset of diseases can reduce vision and affect the quality of life of patients. The main causes of progressive and irreversible vision loss include various pathologies, such as cataracts, ocular atrophy, corneal opacity, age-related macular degeneration, uncorrected refractive error, posterior capsular opacification, uveitis, glaucoma, diabetic retinopathy, retinal detachment, undetermined disease and other disorders involving oxidative stress and inflammation. The eyes are constantly exposed to the external environment and, for this reason, must be protected from damage from the outside. Many drugs, including cortisonics and antinflammatory drugs have widely been used to counteract eye disorders. However, recent advances have been obtained via supplementation with natural antioxidants and nutraceuticals for patients. In particular, evidence has accumulated that polyphenols (mostly deriving from Citrus Bergamia) represent a reliable source of antioxidants able to counteract oxidative stress accompanying early stages of eye diseases. Luteolin in particular has been found to protect photoreceptors, thereby improving vision in many disease states. Moreover, a consistent anti-inflammatory response was found to occur when curcumin is used alone or in combination with other nutraceuticals. Additionally, Coenzyme Q10 has been demonstrated to produce a consistent effect in reducing ocular pressure, thereby leading to protection in patients undergoing glaucoma. Finally, both grape seed extract, rich in anthocyanosides, and polynsatured fatty acids seem to contribute to the prevention of retinal disorders. Thus, a combination of nutraceuticals and antioxidants may represent the right solution for a multi-action activity in eye protection, in association with current drug therapies, and this will be of potential interest in early stages of eye disorders.
Keywords: eyes diseases, oxidative damage, antioxidant compounds, vitamin A, vitamin C, lutein, trehalose, astaxanthin, curcumin, quercetin, Coenzyme Q10, PUFAs, BPF, grape seed
1. Introduction
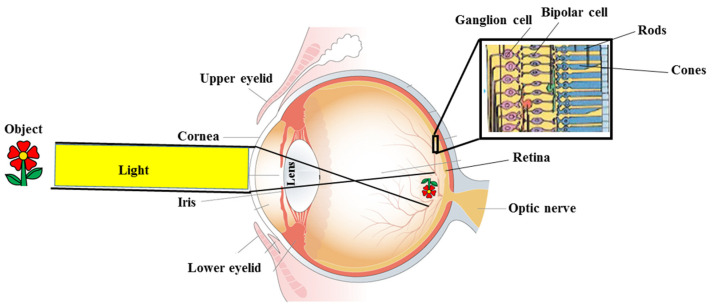
To date, it is known that improving eye health is one of the objectives of the UN Summit on Sustainable Development, and this can be achieved by including promotion, prevention, care, and rehabilitation strategies. Improving eye health includes not only best vision, but also reducing disability and increasing wellbeing [1]. Therefore, it is possible to improve eye health and solve world problems associated with it; for example, the reduction of hunger reduces eye diseases related to malnutrition, but also, better eye health reduces poverty and thus reduces hunger [2]. The normal human eye measures approximately 22 to 27 mm in the anteroposterior diameter and possesses a circumference from 69 to 85 mm. The human eyeball consists of three primary sections: (1) the outer layer of support of the eye, which includes clear cornea, opaque sclera, and their interdigitation, designated as limbo; (2) the central uveal layer of the eye comprising the iris, ciliary body, and choroid; (3) the inner layer of the eye, commonly referred to as the retina [3,4]. The visual process begins with the crossing of light through the clear cornea, the pupillary opening, the crystalline lens, and the retina. Finally, the visual impulse is transmitted to the brain through the optic nerve [5,6]. Light is a form of electromagnetic energy that enters our eyes through the pupil; light converges through the cornea and the lens upon the receptors of the retina that are located on the back wall of the eye. The pupil is surrounded by a pigmented iris that can expand or contract, making the pupil larger or smaller as the incident light level changes. Retinal receptors detect light energy and, through a transduction process, generate the action potentials that then travel along the optic nerve [7,8]. In Figure 1, eye structure and the Table 1 mechanism of vision are shown.
Figure 1.
Eye structure and mechanism of vision.
The eye, regardless of its complex structure, is connected structurally and functionally with vascular, nervous, endocrine, and immune tissue. Nevertheless, it is constantly exposed to the external environment, and for this reason, it must be protected from damage from the outside. A very important protection to the eye is guaranteed by the production of a tear film, consisting of aqueous components, electrolytes, lipids, and mucus. In this way, the lubrication is maintained, and any factor that disturbs its composition can alter the stability, osmolarity, tissue structure, and mechanical and inflammatory mechanisms of the eye [9,10,11]. Eye diseases are due to aging, the occurrence of local and specific diseases, or concomitant pathologies. Impaired vision negatively affects quality of life and daily activities, increases the risk of disability and depression in older age, and may lead to loss of independence [12,13,14]. Common eye diseases are ocular atrophy, corneal opacity, cataracts, uncorrected refractive error, posterior capsular opacification, uveitis, glaucoma, age-related macular degeneration, diabetic retinopathy, retinal detachment, and undetermined disease [15,16]. The drug therapy adopted for these pathologies can be supported by the intake of particular foods or food supplements, which have been shown to address the pathophysiologic mechanisms directly involved. Numerous micronutrients and nutraceuticals products, in fact, can affect some eye components through the involvement of different metabolic pathways [17,18,19]. Since many eye diseases have oxidative stress as a common denominator, it is essential to study the main natural antioxidant compounds in order to justify their use in clinical practice. The bibliography used to write this review was downloaded from PubMed using terms “ocular diseases”; “antioxiant and ocular diseases”; and “pharmacology of ocular hepatologies”. This review can be divided into two sections: in the first part, the close correlation between the main pathologies affecting the eye and oxidative damage is described; in the second part, the beneficial effects of some antioxidant compounds on eye health are developed.
2. Reactive Oxygen Species and Eye Health
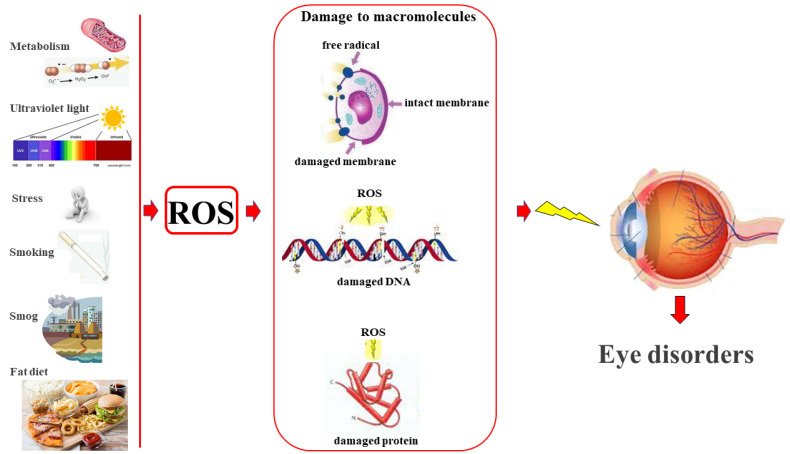
Oxygen (O2) is essential for life as it is crucial for aerobic breathing of cells and tissues, as well as for ensuring the energy cycle of life. Under normal conditions, aerobic metabolism predicts that O2 undergoes a reduction reaction, giving rise to water (H2O). On the contrary, in some situations, there is an incomplete reduction of O2, leading to the formation of very unstable and reactive species, known as reactive oxygen species (ROS), and their accumulation [20]. In metabolic reactions, the first ROS to form is the superoxide anion (O2−), which is the most abundant of ROS and possesses a half-life of milliseconds. Because of its very short half-life, this radical ion is not able to attack biological macromolecules, but may stimulate chain reactions resulting in the formation of high concentrations of ROS. The O2− is also used by the immune system to kill pathogenic microorganisms; phagocytes produce O2− in large quantities through the enzyme NADPH oxidase and use it to eliminate pathogens through an oxygen-dependent mechanism. Due to the toxicity of O2−, all organisms have developed superoxide antagonist enzyme isoforms; superoxide dismutase (SOD) is capable of catalyzing superoxide neutralization, producing hydrogen peroxide (H2O2) [21]. H2O2 is the second most-present ROS; it is a small molecule and a non-radical oxygen species, which spreads easily in biological membranes, propagating its effects even at a distance. In addition, H2O2 can generate other ROS and decompose, converting to water and oxygen through an exothermic reaction. Since the oxidation state of oxygen in H2O2 is −1 (intermediate between states 0 and −2), H2O2 can function as both and oxidizing and reducing agent. Reactions in which H2O2 is cycled as an oxidant is favored in an acidic environment, while reactions in which it behaves as a reducing agent are favored in a basic environment [22]. The hydroxyl radical (OH•) is the most powerful and harmful of all oxygen species, since, despite having a very short half-life, it shows a high reactivity. Therefore, it is a very dangerous compound for the body because cannot be eliminated by an enzymatic reaction and because it reacts with every oxidizable available compound. OH• can damage all kinds of biological macromolecules, and the only means to protect cellular structures is the use of antioxidants or another efficient repair system [23]. Biological macromolecules (lipids, proteins, and nucleic acids) can be damaged by ROS, although lipids are more susceptible to oxidation. There are defensive mechanisms, both exogenous and exogenous, against oxidative attack; if the production of ROS and the ability of antioxidant biological systems to counteract the effects of ROS metabolites are unbalanced, oxidative stress occurs, which produces varying degrees of damage and abnormality in cells [24]. Exogenous antioxidant enzymes include superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GSH-Px) [25]. Oxidative stress involves the onset of many pathologies, including cancer, neurodegenerative diseases, atherosclerosis, cardio-circulatory pathologies, asthma, infertility, aging, dermatitis, hypertension, diabetes, and rheumatoid arthritis, among others [26,27,28,29,30]. Oxidative stress is also involved in eye diseases, such as dry-eye disorder, cataracts, glaucoma, eye surface disorders, retinitis pigmentosa, diabetic retinopathy, uveitis, age-related macular degeneration and toxic neuropathies [31,32]. The human eye is constantly exposed to sunlight, artificial light, and high metabolic activity; in addition, exposure to ionizing radiation, environmental toxins, and chemotherapy contribute to oxidative damage in eye tissues, making the eye particularly susceptible to oxidative damage. Prolonged exposure to ROS, therefore, constitutes a considerable risk for the health of the eye, whose cells, following the damage of their macromolecules, highlight the impairment of the metabolism, resulting in necrotic or apoptotic death [33]. Eye dryness disorder is defined by the dysfunction of the eye surface, which consists in a deterioration of the tear film and which leads to dryness of the eyes. This disorder involves several portions of the eye, including the eyelids, tear glands, and various tissues of the eye surface [34]. A significant increase in oxidative activity, associated with a decrease in antioxidant defenses in the fluids and tissues of the eye, can damage the eye surface, causing serious alteration to the tissues of the cornea and conjunctiva, evolving in the development of eye dryness and visual damage. In fact, in patients suffering from dry eye, in addition to an imbalance in the state of the tear film, the overexpression of the production of ROS on the eye surface was detected [35,36]. Retinal eye diseases are numerous and particularly linked to oxidative stress for three important reasons: 1) the retina is constantly subjected to the action of visible light that generates photooxidation; 2) the work carried out by the retina is considerable and requires a large consumption of oxygen which, during mitochondrial respiration, could favour the accumulation of ROS; 3) PUFAs are particularly present in the retina which is, consequently, susceptible to lipid peroxidation [37]. Diabetic retinopathy is considered, to date, the main cause of blindness in developed countries. Impaired blood glucose levels cause significant damage to vision, which is aggravated in the presence of ROS accumulation. Free radicals damage cells by acting on biological macromolecules; they disaggregate amino acids by altering the protein structure, fragment nucleic acids by damaging DNA, and modify lipids and membrane structure [38]. These alterations lead, over time, to cell death by necrosis. Finally, ROS accumulation damage in diabetic patients causes systemic alterations caused by vascular dysfunction [39,40]. Glaucoma includes a group of disorders in which selective retinal ganglion cell (RGC) loss occurs; RGCs are located on the inner surface of the retina and connect its axons to the optic nerve. This disease is the second cause of blindness in the world and is a progressive optic neuropathy caused, in most cases, by elevated intraocular pressure [41]. Glaucoma is closely related to oxidative stress, and evidence of this correlation has been provided in both animal and human experiments [42]. In the models used, glaucoma was induced by increased intraocular pressure, optic nerve compression, axotomy, reduced blood supply to the optic nerve, and autoimmune damage. In all these induced situations, an increase in ROS, a reduction in antioxidants, and an increase in retinal lipid peroxidation have consistently been found [43]. The mechanism of action involves ROS and biological macromolecules, proteins, and nucleic acids [44,45]. Cataracts consists in the opacification of the lens, which is located inside the eye and has the purpose of filtering the light that penetrates through the pupil, directing it towards the retina [46]. The triggering causes of cataract onset are multiple; including aging, genetic inheritance, metabolic, and environmental or nutritional insults; sometimes, it can be a consequence of other eye diseases (retinal degenerative, uveitis, glaucoma) or systemic diseases such as diabetes [47]. Nowadays, it is increasingly believed that cataracts are caused by the presence of high-molecular-weight protein aggregates, or by the breakdown of the microarchitecture of the lens. This hypothesis involves post-translational modifications of lens proteins that alter their conformation, leading to destabilization and eventual aggregation [48]. Membrane, luminal, or secretory proteins are synthesized in the rough endoplasmic reticulum (ER) and transported into its lumen [49,50,51]. When internal or external factors intervene, the proteins, inside ER, conform incorrectly, becoming misfolded proteins. Poorly folded proteins in the ER trigger cataract formation processes [48]. In cells, misfolded proteins are eliminated or corrected in suitable conformations by specific pathways, including the unfolded-protein response (UPR). However, chronic UPR further amplifies the degradation, modification, and aggregation of the proteins of the lens in a downstream cascade. UPR culminates in calcium imbalance, protein degradation, oxidative insults, redox state disturbances, and loss of antioxidant defense mechanisms [52]. Recent studies have shown that, in patients with cataracts, ROS induced a reduction in DNA methylation in the Keap1 promoter gene, activating the expression of Keap1 protein. In physiological conditions, Keap1 can increase the nuclear factor and erythroid 2-related factor 2 (Nrf2) proteasomal degradation. Since Nrf2 controls the basal and induced expression of a number of antioxidant-response genes, it is evident that the reduction of Keap1-induced ROS increases the degradation of Nrf2 and, as a result, reduces a cell’s antioxidant control system [53,54,55]. The consequence is that oxidative damage increases exponentially, along with the development of cataracts of the lens. Finally, an excessive production of free radicals alters the redox state, modulating an inflammatory response, which leads to the exacerbation of oxidative damage favoring numerous pathological states [56]. The main negative function of ROS occurs on biological macromolecules, generating lipid peroxidation, DNA oxidation, and protein alteration, which contributes to the damage of cell structure and function. A prolonged condition of ROS-induced cell damage results in the onset of inflammation and pathologies affecting different body districts. Figure 2 shows how the accumulation of ROS can generate eye pathologies.
Figure 2.
Prolonged damage to biological macromolecules ROS-induced favors the onset of eye diseases.
3. Role of Antioxidants and Nutraceuticals in Maintaining Eye Health
As oxidation leads to the onset of damage and in several districts, the role of antioxidants has become of massive interest for doctors and patients in the treatment and prevention of diseases. An antioxidant compound can be defined as a substance that can delay or prevent oxidation [57]. The body uses different strategies against the production and accumulation of ROS: firstly, antioxidant enzymes are used, as already reported, such as CAT, SOD, GSH-px. It is important to remember that some antioxidant enzymes need micronutrients to function properly, such as zinc, selenium, copper and manganese [58]. Secondly, ROS can be reduced or neutralized by the intake of antioxidant nutrients, such as vitamin E (a-tocopherol), beta-carotene, and vitamin C, among others [59]. An insufficient intake of foods with antioxidant function or an unbalanced diet can alter the body’s natural antioxidant system and facilitate the damage induced by ROS. Additional defense mechanisms include antioxidant compounds, such as metallothionein, melanin, and glutathione [60,61]. In the following sections, we will investigate some natural compounds with marked antioxidant activity. Among them, those that are most used in clinical practice and that, to date, are part of the antioxidant mixtures used in clinical trials, have been selected.
3.1. Vitamin A and Lutein
Among dietary factors, vitamin intake has become increasingly positive in reducing oxidative stress and, as a result, improving the outcome of many eye pathologies. Vitamin A is a fat-soluble life-essential group of compounds characterized by an unsaturated isoprenoid chain structure. Famously, vitamin A performs important functions including cell proliferation, reproduction, foetal growth and development, vision in darkness, corneal and conjunctiva development, immune system functioning, and central nervous system formation [62]. Unlike water-soluble vitamins, vitamin A easily accumulates in the body, especially in the liver and adipose tissue. This characteristic determines the advantage of resisting the development of clinical deficiency symptoms, but at the same time, it has the disadvantage of being able to provide accumulation toxicity. Vitamin A can be supplied in the diet both from products of animal origin, such as retinol, and vegetable carotenoids, such as provitamin A [63]. As already discussed, the retina is responsible for visual perception, mediated through specific structures known as cones and rods that are fundamental for vision in light and dark conditions, respectively. Active vitamin A is associated with a protein receptor coupled to protein G, the complex known as rhodopsin, generating a chain of reactions whose last consequence is the transmission of optical perceptions to the brain via the optic nerve [62]. Vitamin A is responsible for the maintenance of homeostasis reduction–oxidation. In fact, retinol binds to different proteins, acting as a redox reagent. In addition, carotenoids, such as β-carotene, α-carotene, lutein, lycopene, and cryptoxanthin, are well-known antioxidants [64,65]. An excessive intake of vitamin A is potentially toxic; toxicity has been associated with 100,000 RE per day (1 RE = 1 μg retinol) in adults and 10,000 RE per day in children. In women in the first trimester of pregnancy, a dose of 3000–9000 RE per day created teratogenic effects. The Group of Experts on Vitamins and Minerals have not been able to set a safe limit for Vitamin A; therefore, it is recommended that a maximum intake per day is 700 lg for men and 600 lg for women [66]. Among the approximately 850 types of naturally occurring carotenoids, very few of them are present in human tissues. Among these it is important to mention lutein and its stereoisomers, zeaxanthin and meso-zeaxanthin, present in the human retina [67]. It is a class of a carotenoid, named xanthophyll, that cannot be synthesized de novo in the human body, and are absorbed only through diet. The most xanthophyll-rich foods, including lutein and zeaxanthin, are leafy green vegetables, such as cabbage, broccoli, peas, spinach, lettuce, and also egg yolk [68]. These compounds are hydrophobic; however, due to the presence of the hydroxyl group, lutein and zeaxanthin are relatively polar compounds. After their intake, food carotenoids are dispersed in gastric juice and incorporated into lipid droplets, transferred to micelles with food lipids, and finally into the bloodstream. Therefore, fat-rich diets generally facilitate the absorption of dietary carotenoids [69]. Lutein is particularly concentrated in the central portion of the retina, where the photoreceptor cells, responsible for visual acuity and central vision, are located. In the macula of the retina are found: zeaxanthin, in the middle-peripheral region; meso-zeaxanthin, in the epicentre; and lutein, concentrated in the periphery. The absence of these three cartenoids is often used to predict the risk of developing macular diseases [69]. Lutein is also found in the lens, protecting it from age-related eye diseases, such as cataracts [70]. Lutein is retained in the human retina for a prolonged period of time and it has been shown that, even after three months from the interruption of lutein supplementation, the optical density of the macular pigment remains high despite its low serum concentration [71,72]. Lutein has been shown to exert an extremely powerful antioxidant action with several mechanisms of action: (1) render the role of the oxygen singlet poorly active; (2) reduce or eliminate free radicals; (3) filter blue light, thereby reducing phototoxic damage to photoreceptor cells; (4) reduce the expression of inducible nitric oxide synthase (iNOS) [73,74,75]. In addition, lutein is able to turn off the inflammatory process, inhibiting the pro-inflammatory cytokine cascade, the expression of nuclear-kB transcription factor (NF-kB), and the activation of the complement system [76,77,78,79]. Many clinical studies have attributed lutein to antioxidant and anti-inflammatory properties in the eye, justifying a benefit in some diseases such as age-related macular degeneration, diabetic retinopathy, cataracts, retinitis pigmentosa, and myopia [80,81]. Lutein supplementation showed a relatively high safety profile and was classified as “GRAS” by the US Food and Drug Administration (FDA) [82]. Although German, Canadian and American studies have reported daily intakes of lutein of 1.9, 1.4, and 2 mg, respectively [83,84], these results have been shown to be undervalued, and the randomized clinical trial “Age-Related Eye Disease Study 2” (AREDS 2) showed a lutein intake of 10 mg per day over 5 years in more than 4000 patients. Subsequently, the Council for Responsible Nutrition (CRN) stated that lutein intake is safe up to 20 mg/day [85].
3.2. Vitamin C and Coenzyme Q10
Vitamin C (ascorbic acid) is, chemically, a low-molecular-weight carbohydrate capable of donating electrons to free radicals from both the second and third carbon, quenching their reactivity and acting as a reducing agent. While most vertebrates can synthesize this compound, humans, together with guinea pigs, some fish, birds, and insects, rely exclusively on dietary intake to maintain the body’s levels of vitamin C. This is a hydrophilic vitamin, and despite its small size, it does not cross the plasma membrane by passive diffusion [86]. During the detoxification reactions of ROS, vitamin C oxidizes to dehydroascorbate, but this oxidized form can subsequently be reduced to generate vitamin C again by glutathione-dependent enzymes. However, if the oxidative damage is continuous, dehydroascorbate undergoes an irreversible degradation; in the event in which vitamin C is present in excessive doses, it can act as a pro-oxidant, contributing to the formation of hydroxyl radicals and increasing oxidative damage. This means that vitamin C can pass from being an antioxidant in physiological conditions to a pro-oxidant under pathological conditions [87]. Vitamin C is particularly present in the eye in the aqueous humor (saline fluid that is located between the cornea and the crystalline) and vitreous humor (connective tissue of gelatinous consistency occupying the eyeball cavity between the posterior surface of the lens and the retina). Particularly in these districts, its concentration exceeds plasma concentrations by 20 to 70 times [88]. In the eye, vitamin C absorbs UV light, preventing the penetration of UV rays and subsequent photoinduced oxidative damage in the tissues, behaving as a physiological “sunscreen” [89]. In addition, vitamin C can scavenge or quench the superoxide anion radical, hydrogen peroxide, hydroxyl radical, singlet oxygen, and reactive nitrogen oxide [90], protecting the cornea, the lens and other ocular tissues against oxidative damage. Finally, vitamin C has been shown to play a role in the prevention of lipid peroxidation of membranes [91]. Since it has been shown that individuals with vitamin C deficiency developed cataracts more easily than others, and that there was a close correlation between vitamin C and the health of the crystalline, numerous studies have been conducted on the relationship between vitamin C and the risk of cataracts [92,93]. The recommended daily allowance (RDA) based on the intake of vitamin C, is 75 and 90 mg/day for women and men, respectively, established by the U.S. Institute of Medicine (IOM) in 2000 [94]. Recent data suggest that the current RDA for vitamin C set by the IOM for men and women may be too low. On the basis of a comprehensive review of the scientific evidence, it was concluded that 200 mg/d is the optimum intake of vitamin C for the majority of the adult population, to the advantage of the health benefits for the eye [95]. Coenzyme Q10 (coQ10) possesses a quinone structure and, for this reason, is also known as ubiquinone. The chemical structure of coQ10 is very similar to that of vitamin K; nevertheless, this cofactor is not considered a vitamin because it is the only fat-soluble antioxidant that animal cells synthesize de novo in the body [96]. It is found in all cell membranes and its main function is to be a cofactor of the mitochondrial enzymes that cooperate in the formation of ATP, an energy source needed to perform cellular biochemical functions. In particular, this liposoluble compound works to transport the electrons in mitochondria during aerobic cellular respiration, from complex I (NAHD ubiquinone oxioreductase) and complex II (succinate ubiquinone reductase) to complex III (ubiquinone cytochrome c reductase). Another function of coQ10 is to participate in the creation of a proton gradient in the intermembrane space [97]. This compound also possesses direct and indirect antioxidant properties in its reduced form (CoQ10H2). The direct antioxidant property is achieved by reducing the accumulation of ROS, while the indirect action occurs with the regeneration of a form of vitamin E (α-tocopherol) [98]. CoQ10 collaborate in lowering the lysosomal pH, transporting H+ ions inside, in order to facilitate an acidic environment necessary to degrade cellular debris [99]. Finally, it has been recognized that coQ10 participates in gene expression, and this could explain its effects on overall tissue metabolism [100,101]. Since the quantity of coQ10 present in the body is determined by two sources, biosynthesis [102,103] and dietary supplementation, its deficiency may occur for the following reasons: (1) reduced dietary intake; (2) impaired biosynthesis; (3) increased usage by the body [104]. A shortage of coQ10 is mainly manifested by reduced energy metabolism, impaired protection from free radicals, and deacidification of lysosomes [105,106,107]. Since the retina is the most metabolically active tissue of the body, with the highest consumption of energy (tissue/size ratio), patients with coQ10 deficiency may develop retinopathies, suggesting that coQ10 can play an important role in pathogenesis of retinal conditions [108,109]. In addition, a study by Que et al. showed higher coQ10 concentrations in young people (30 years) compared to older human retinas (80 years), highlighting how the oxidative stress plays a key role in the pathogenesis of many age-related diseases, such as atherosclerosis, cataracts, and Alzheimer’s disease [110,111,112]. In this way, the accumulation of ROS in aging results in increased cell damage that mediates the apoptotic mechanisms of cell death [113]. Age-related macular degeneration (AMD) causes loss of central vision, which has a significant impact on quality of life. Plasma coQ10 levels are substantially reduced in patients with AMD, compared with control patients, and this suggests an association between coQ10 and AMD pathogenesis [114]. Since the retina and the ocular macula are exposed to light more than any other organ or tissue in the body, these districts will be particularly sensitive to oxidative stress and lipid peroxidation. The result of this oxidative damage leads to apoptotic cell death [115]. Glaucoma is characterized by the loss of RGC, which are fundamental in the transmission of the signal from the photoreceptors to the optic nerve. As the prevalence of glaucoma increases with age, there may be a possible correlation between RGC and coQ10 deficiency in old age [116,117]. Experimental studies have shown that intravitreal administration of coQ10 minimizes apoptosis in RGC. This supported the neuroprotective role of coQ10 [118,119]. The normal concentration range of coQ10 in human plasma is 0.8–1.2 mg/L, and in cases of deficiency, supplementation typically given to adults at 1.2–3 g/day [120].
3.3. Astaxanthin
Carotenoids are a class of compounds with over 600 natural fat-soluble pigments that play a crucial role in the photosynthetic process, as well as protective activity against damage caused by excessive exposure of light and oxygen. They are taken through the diet, constituting a robust nutritional role as a source of vitamin A [121]. Their oxygenated derivatives are known as xanthophylls; both classes of pigments share a structural scheme consisting of alternating single and double bonds, responsible for the absorption of the excess energy contained in other molecules and carrying out, accordingly, an antioxidant role [122]. Astaxanthin, a xanthophyll carotenoid, is a naturally occurring red pigment in numerous living organisms (bacteria, microalgae and yeasts), present as a secondary metabolite. This compound is biosynthesized by phytoplankton and microalgae (such as Haematococcus pluvialis, Chlorella zofingiensis and Xanthophyllomyces dendrorhous), which are the basis for the feeding of zooplankton and krill, the ideal food of animal species which store this pigment in the skin and adipose tissue. Finally, all the superior aquatic species that feed on these foods become rings of the trophic chain containing astaxanthin. This carotenoid is responsible for the colouring of some sea creatures, including salmonids, tuna, shrimps, crustaceans, lobsters, and crayfish [123]. The chemical structure of astaxanthin provides a long chain structure and two terminal polar groups; this particular conformation provides the compound with both lipophilic and hydrophilic properties. Precisely because of this feature, astaxanthin extends through the entire double-layer membrane, carrying out its protective activity both inside and outside the cell membrane [124,125]. Astaxanthin has been shown to have numerous beneficial activities on human health, including a protective effect on the skin, the cardiovascular and nervous system, and antioxidant, anti-inflammatory, anti-cancer, and antidiabetic properties, among others [126,127]. In the last two decades, the potential role of astaxanthin in protecting eye health has been highlighted, showing a significant improvement in macular degeneration, cataracts, diabetic retinopathy, and glaucoma [128]. To date, astaxanthin is considered to be the most beneficial antioxidant carotenoid provided in nature; in fact, it is more potent than most known antioxidants, according to the following ratios: 6000 times more powerful than vitamin C; 550 times stronger than vitamin E (alpha-tocopherol); and 40 times more powerful than beta-carotene [129]. The reason for this exceptional oxidative protection is explained by its chemical structure: the terminal polar groups quench free radicals and the double bonds, its intermediate segment, removes high-energy electrons [130]. The peculiarity of astaxanthin is to be able to neutralize single oxygens and radicals in both the non-polar (hydrophobic) and polar (hydrophilic) zones [131]. Several studies have confirmed the antioxidant efficacy of astaxanthin and have identified the reduction of oxidative marker levels, such as malondialdehyde (MDA), and the increase in antioxidant agents including SOD, CAT and GPX1 [132,133,134]. Therefore, we can conclude that astaxanthin exerts antioxidant activities not only through direct scavenging of radicals, but also by activating the cellular antioxidant defense system [135,136]. Finally, astaxanthin exerts a robust anti-inflammatory [137,138] and anti-apoptotic activity [139,140]. In the last two decades, it has been shown that treatment with astaxanthin improves many eye conditions. For example, Otzuka et al., showed that treatment with axanthin 100 mg/kg could inhibit retinal dysfunction caused by light [141]. At the same time, Parisi et al. pointed out that patients with nonadvanced AMD could improve their clinical condition. In particular, a study compared a group of 15 patients who were treated with oral supplementation of astaxanthin (4 mg), zeaxanthin (1 mg), lutein (10 mg), vitamin C (180 mg), vitamin E (30 mg), zinc (22.5 mg), and copper (1 mg) daily for 12 months with a control group (12 patients). The results showed that patients treated with supplementation showed selective improvement of retinal function, compared with the control group [142]. The administration of astaxanthin/lutein/zeaxanthin over a two-year period has been able to improve visual acuity, contrast sensitivity, and vision-related functions [143]. Diabetic retinopathy, the main complication of diabetes, is considered to be the result of chronic oxidative stress and inflammation. The bioactive compound astaxanthin has been shown to exert neuroprotective effects in experimental models of diabetic retinopathy, reducing oxidative stress, inhibiting the activity of NF-κB transcription factor, and reducing the expression of inflammation mediators [144,145]. To confirm this, Yeh et al. demonstrated in a model of rats with streptozotocin-induced diabetes that treatment with astaxanthin resulted in a reduction in histological lesions typical of diabetic retinopathy, oxidative stress, and inflammation, an increase in antioxidant enzymes and a reduction in the expression of NF-κB [146]. The use of astaxanthin has been shown to reduce lens opacification in cataracts induced by prolonged steroid treatment or hyperglycemia [147,148]. Growing evidence suggests that astaxanthin has numerous beneficial effects in several eye diseases affecting both the anterior and posterior poles. Finally, this compound showed an optimal safety profile with no adverse events in any clinical study [149].
3.4. Trehalose
Trehalose, also referred to as α,α-Trehalose or α-D-Glucopyranosyl-α-D-glucopyranooside, is a non-reducing disaccharide consisting of two portions of glucose linked through an α,α1,1-glucosidic bond [150]. This component is usually found in the environment in different species of plants, fungi, algae, bacteria, yeasts, insects, and other lower invertebrates, but never in mammals or other vertebrates. Once ingested with food, trehalose is hydrolyzed in the small intestine in two molecules of D-glucose through the trehalase enzyme. It is also known to be used as a sweetener; in industry as, a stabiliser or packaging material; in the pharmaceutical industry, as an excipient; and in the textile industry, as a texturing agent 2 [151]. The chemical structure of trehalose justifies the impossibility of crossing cell membranes; to date, it has been established that this compound can penetrate the cytosol through vesicular internalization of endocytosis [152,153]. Trehalose has also recently been used for its therapeutic effects in the treatment of cardiometabolic disease and degeneration in animal and human models [154]. Takahashi et al. [155] found that trehalose reduced neuronal damage in a model of ischemia in rabbits. In the case of neurodegeneration or traumatic brain injury, oral administration of 2% trehalose has been shown to improve the cognitive characteristics involved in injured brain areas [156]. In addition, this compound induced the increase of zinc and iron in the brain, essential for the maintenance of brain functions [157,158]. Finally, trehalose increased the expression of proteins involved in synaptic activity, neuroprotection, and neurogenesis [159]. Trehalose has become known for its pro-autophagic action; it is able to eliminate and recycle damaged macromolecules in response to cellular stress. For this reason, it was considered protective against the aggregation of the β-amyloid protein in neuronal cell lines [160] and metabolic disorders. More generally, it is possible to affirm that the action of this compound guarantees homeostatic paths that depend on the type of tissue involved [161]. Trehalose has been found to be a stress-reactive factor; in particular, it is a response factor when the cells of the organisms that produce it are exposed to environmental stress, such as heat, cold, dessication, and oxidation. When stress conditions become excessive, these organisms synthesize trehalose, which helps them maintain cellular integrity. With this strategy, these organisms can prevent the denaturation of proteins, facilitate their stabilization, and inhibit protein aggregation [162,163]. Another important function of trehalose is to properly moisturize the dried tissues of the organisms that synthesize this compound. This strategy, known as the “water hypothesis”, is based on the ability of trehalose to form hydrogen bonds between its polar groups and lipids or membrane proteins [164]. In this way, trehalose can maintain the integrity of phospholipids guaranteeing the functional properties of biological membranes [165]. There is a large volume of evidence that testifies to a specific role of trehalose in performing antioxidant and anti-inflammatory effects and, in recent years, has shown beneficial effects in ophthalmology [166]. For example, cell exposure to H2O2 causes severe oxidative damage to the amino acids of cellular proteins; nevertheless, trehalose is able to reduce such damage, in this way protecting both proteins and lipids of the membrane [167]. The work produced by Cejková et al. showed a protective effect of trehalose on cornea damaged by ultraviolet radiation B (UVB). In addition, ocular tissues, exposed to photodamage, react more easily by suppressing oxidative, inflammatory, and apoptotic pathways when exposed to this compound [168]. Trehalose has an established ability to maintain the degree of hydration; this compound has been used to protect the cells of the anterior ocular surface in dry-eye disease [169]. In addition, the effect of trehalose was enhanced when it was added to a single formulation with hyaluronate, an anionic glycosaminoglycan polysaccharide with lubricative and water-retaining properties [170]. The trehalose highlighted the ability to reduce photodamage caused by UVB radiation in the epithelium of the cornea [171]. Following UVB irradiation, trehalose was found to reduce the resulting corneal disorders, speeding up healing, suppressing neovascularization, restoring corneal transparency, and restoring proper immunohistochemistry [172]. Chen and Haddad (2004) also highlighted the effectiveness of trehalose against hypoxic or anoxic lesions. The cornea is not only affected by damage caused by ROS, but also by insufficient oxygen supply. In these cases, apoptotic death occurs; trehalose has been shown to effectively suppress these disorders [173].
3.5. Curcumin and Quercetin
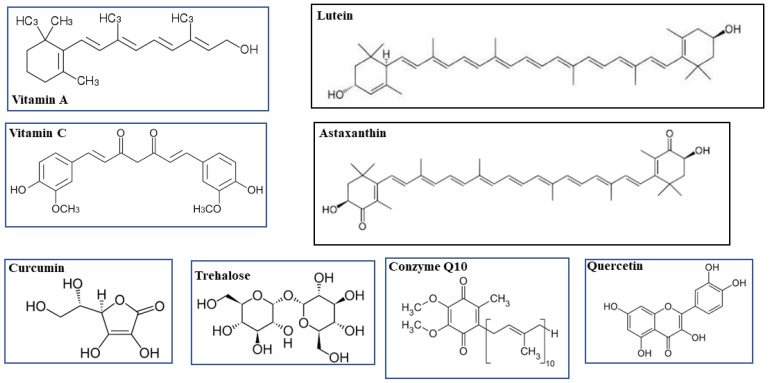
Curcumin is a pigment insoluble in water extracted from the rhizome of Curcuma longa, a species that belongs to the Zingiberaceae family. The obtained powder contains 2–5% curcumin. Curcumin is a biologically active phytochemical compound with health benefits, and its extract also contains β-carotene, lycopene, epigallocatechin gallate, and quercetin [174,175]. In recent years, several studies have confirmed the use of curcumin for the prevention and treatment of many diseases, especially inflammatory diseases and cancer [176,177]. Curcumin inhibits the production of free radicals, and therefore shows antioxidant properties [178]. The effect of curcumin on oxidative damage is based on its ability to scavenge ROS and reactive nitrogen species (RNS) [179]. It can modulate the activity of active enzymes in the neutralization of free radicals, GSH-Px, CAT, and SOD [180]. In addition, it can inhibit enzymes that generate ROS, such as lipoxygenase/cyclooxygenase and xanthine hydrogenase/oxidase Finally, being lipophilic, curcumin is able to eliminate peroxylic radicals, behaving like vitmin E. Antioxidant properties of curcumin cause the inhibition of oxidative stress and this reduces the risk for many lifestyle diseases [181]. In addition, curcumin exerts an anti-inflammatory effect; it lowers the expression of the gene IκBα, gene cyclooxygenase-2 (COX-2), prostaglandin E-2 (PGE-2), interleukin 1-6-8 (IL-1, IL-6, IL-8), and tumor necrosis factor α (TNF-α). The anti-inflammatory effect is also exerted by the ability of curcumin to activate the proliferator-activated peroxisome receptor γ (PPAR-γ), a nuclear receptor protein that binds to the peroxisome proliferator response element (PPRE) and regulates gene transcription [182]. Recently it has been highlighted that ROS are fundamental regulators of angiogenesis, the process that allows new blood vessels form within the vascular system, and that vascular function critically depends on the amount of ROS present. In fact, while high doses of ROS induce oxidative stress and subsequent cell death, both conditions inhibit angiogenesis; low doses of ROS promote it through some sublethal damage to the cell membrane, subsequently releasing the growth factor of fibroblasts FGF-2, which are directly involved in angiogenesis [183,184]. Given the properties of curcumin, this compound could be used in the treatment of diseases related to angiogenesis, including eye diseases [185,186]. Most retinal diseases, as already mentioned, imply an abundance of ROS and reduced levels of scavenger antioxidants. In particular, RGC and photoreceptors are extremely sensitive to oxidative stress damage, and it is known that the accumulation of ROS is often involved in several diseases of the retina, such as uveitis, age-related macular degeneration, diabetic retinopathy, retinal tumors, and proliferative vitreoretinopathy (PVR) [187]. In order to know the effect of curcumin after an oxidative stress insult, Munia et al., have shown that this nutraceutical compound was capable of protecting human retinal epithelial cells from death [188]. Dry-eye disease is characterized not only by reduced secretion of tears, but also rapid tear evaporation, responsible for the damage to the eye surface [189]. This disease includes an inflammatory process involving IL-6, IL-8, IL-1β [190]. It has been shown that curcumin could exert a protective effect through its anti-inflammatory activity, inhibiting the expression of pro-inflammatory cytokines in conjunctiva [191]. Uveitis is an inflammation of the eye that includes the iris and adjacent tissue. Lal et al. reported an improvement in patients with chronic uveitis who have been given oral capsules of curcumin (75 mg/capsule) [192]. A decrease in aqueous flare and keratic precipitates was observed after treatment. Its beneficial effects can be derived from their antioxidant, anti-inflammatory, and antifibrinolytic properties [189]. Because curcumin has low oral solubility and bioavailability, its biomedical potential cannot be exploited in animals and humans [193]. The exogenous curcumin administered in humans for a period of about 8 weeks has been shown to be able to adequately perform all the functions of this natural compound [194]. Quercetin is a member of the subclass of flavonols and is abundant in the human diet. It has received considerable attention from the scientific community in recent years thanks to numerous effects on human health, including antioxidant [195], anti-inflammatory [196], anti-cancer [197,198], anti-aging [199], and anti-autoimmune effects, [200] and effects upon metabolic pathologies [201]. The eye surface serves as a protective and functional barrier for the rest of the eye. Diseases of the eye surface can affect the structure of the cornea or conjunctiva, leading to corneal thinning, inflammation, and visual deficits [202]. Prolonged inflammation in these districts can lead to a partial or even complete loss of vision, affecting quality of life. Unfortunately, to date, we do not possess a non-invasive treatment that can preserve corneal function; surgery and corneal transplantation remain the only solution [203]. Given the problems and side effects involved in a corneal transplant, scientists have sought an alternative. The effectiveness of the administration of ocular drugs depends on many factors, including drug absorption, bioavailability, and retention on the front surface. Lipophilic drugs, in general, are associated with increased corneal epithelial permeability, and the solubilization of these compounds in water eye drops was tested. Topical application of quercetin or other flavonoids may be more effective in treating conditions affecting the eye surface [204]. Studies have shown that a protective role is carried out by quercetin when administered at an average daily consumption of about 16–23 mg/day in human populations [205,206,207]. In Figure 3, the chemical structures of the examined compounds are represented.
Figure 3.
Chemical structures of the examined compounds.
3.6. PUFAs
Polyunsaturated fatty acids (PUFAs) are lipids whose hydrocarbon chain has a polar hydrophilic end with a carboxyl group (-COOH) and another end with a non-polar hydrophobic methyl group (-CH3). The n-3 and the n-6 represent two classes of PUFAs defined as “essential”, as they must be taken with the diet because humans do not have the desaturases Δ12 and Δ15, enzymes that catalyze the formation of double bonds along the hydrocarbon chain [208]. Specifically, linoleic acid (LA, 18:2) is the n-6 PUFA from which γ-linolenic and arachidonic acid derive, while from α-linolenic acid (ALA, 18:3), one derives n-3 PUFAs, such as eicosapentaenoic acid (EPA, 20:5 n-3) and docosahexaenoic acid (DHA, 22:6 n-3). LA is present in safflower, soy, and corn oils, while the vegetables that contain ALA are flaxseed, beans, nuts, and the leaves of some green plants. In the liver, the amount of EPA and DHA obtained from the metabolism of ALA is very small, therefore they must be taken with the diet. Both lean and fatty marine fish, fish oil, and algal-derived supplements are particularly rich in EPA and DHA, although they are present in small quantities in many foods of animal origin. For this reason, they must be present in the daily diet [209]. In particular, EPA and DHA can be present in cellular phospholipid membranes and have powerful antioxidant and anti-inflammatory effects [210]. The positive effects of taking n-3 PUFAs are well documented; in fact, the long-term effects against certain pathologies, such as cardiovascular diseases, neurodegenerative diseases, and osteoarthritis, are known [211]. The n-3 PUFAs are also implicated in diseases affecting the eye. For example, in the case of neovascular eye diseases, such as retinopathy of prematurity, diabetic retinopathy, and age-related macular degeneration, current therapies have significant side effects. Clinical and experimental investigations have shown that such treatments could be accompanied by a higher intake of n-3 PUFAs [212]. Despite conflicting results between fundamental and clinical research, it appears that PUFAs can act positively on the damage that determines the pathogenesis of glaucoma [213]. Indeed, the effect of PUFAs can be both on intraocular pressure (IOP) and on survival of RGCs. In the first case, endogenous prostaglandins (PGs), obtained from the metabolism of PUFAs, by activating the EP4 and FP receptors, reduce IOP. In the second case, thanks to their anti-inflammatory and antioxidant effects, n-3 PUFAs can reduce the inflammation and oxidative stress responsible for the RGC dysfunction or death [214]. In particular, patients with pseudoexfoliative (PEX) glaucoma have benefited from the administration of a high, rich DHA nutraceutical formulation that reduced oxidative stress and inflammation. The fact that PUFAs may have beneficial effects for glaucoma is linked to their positive action on endothelial dysfunction and atherosclerosis [215]. Regarding cataracts, a study was conducted on male Wistar rats, in which it was found that the antioxidant and anti-inflammatory activity of lutein increased in the presence of EPA + DHA [216]. In particular, micellar lutein with EPA + DHA has been shown to positively regulate α-crystalline chaperone function [217]. Chang et al. demonstrated a reduction in free fatty acid levels in patients with senile cataracts compared with normal controls. The levels of DHA were particularly low [168]. Studies on the molecular mechanisms responsible for aging in the eye have allowed us to define the involvement of the ELOVL2 (elongation of very-long-chain fatty acids-like 2) enzyme in the regulation of molecular aging in the retina [218]. The ELOVL2 enzyme catalyzes an elongation reaction of n-3 and n-6 PUFAs, which are essential for retinal function. DHA is the main PUFA in the retina and is involved in the photoreceptor function in retinal development and has an antioxidant role [219]. The involvement of PUFAs in AMD, which is the leading cause of blindness in the elderly, has been demonstrated. In particular, the analysis of the eyes of subjects with AMD showed a reduction in PUFAs, and a direct correlation was also reported between the reduced dietary intake of n-3 PUFAs and the increased risk of AMD. Given the function of the ELOVL2 enzyme, it appears that this is directly involved in the onset of AMD along with PUFAs [220]. Furthermore, it appears that DHA may reduce the risk of AMD occurrence by stimulating the synthesis of endogenous antioxidants and the selective autophagy of misfolded proteins [221]. In a study conducted on the eyes of patients with AMD, the analysis of the lipid profile of the retina reported a low ratio between n-3 and n-6 PUFAs, demonstrating the protective role of dietary n-3 PUFAs against AMD [222]. The efficacy of PUFA treatment has also been studied in patients suffering from a multifactorial inflammatory disease, such as dry-eye disease (DED) [223]. The improvement, over time, of parameters such as tear breakup time (TBUT), ocular surface disease index (OSDI), osmolarity and Schirmer’s test, led to the conclusion that PUFAs have a positive effect on nonspecific typical DED when administered briefly and not in combination with other eye medications. Other authors acknowledge the efficacy of n-3 PUFA supplementation in managing DED, even though they consider the evidence to be uncertain and inconsistent [224]. Numerous studies indicate that PUFAs derived specialized-pro-resolving mediators (SPM) capable of maintaining ocular surface health and immune homeostasis, thanks to the fact that SPM pathways and receptors are highly expressed on the ocular surface [225]. Here, the SPMs, produced endogenously, regulate wound healing, innate immunity, and nerve regeneration. SPMs are involved in the protection of the cornea; they are present in significant quantities in healthy human tears; and they guarantee an anti-inflammatory state by increasing the speed of healing of corneal wounds. SPMs reduce the risk factors for corneal transplant rejection by improving graft viability and inhibiting the initiation of alloimmunity [226]. Furthermore, endogenous SPMs have been shown to improve nerve regeneration in the stroma and cornea, representing potential topical therapies in the case of corneal diabetic neuropathy. Some SPM appear to improve symptoms of allergic conjunctivitis, such as reduced mucin secretion and total conjunctival immune cell count. They can also be used effectively as topical treatments for immune-driven DED, as well as for the reduction of ocular surface damage due to viral or bacterial infections, also caused by the use of contact lenses [227]. Furthermore, variations in the levels of very long-chain (VLC)-PUFAs and in the n-3/n-6 ratio were studied in two different experimental models, such as the spontaneously diabetic Nile rat and the Akita mouse, which represent genetic models of diabetes. The evaluations were made in diabetic conditions and following the integration of n-3 PUFAs in the diet. VLC-PUFAs represent a special class of retinal lipids deriving from PUFAs. The levels of VLC-PUFAs and the n-3/n-6 ratio were also measured in human retinal punches from diabetic and non-diabetic donors [228]. In retinal punches of diabetic and retinopathic patients, the levels of VLC-PUFAs were lower than in healthy subjects of the same age. Dietary supplementation with n-3 PUFAs increased the ratio of n-3/n-6 VLC-PUFAs in both experimental models. Therefore, the authors Gorusupudi et al., concluded that the enrichment of the diet with n-3 PUFAs reduces the risk of diabetes onset and of the retinopathy caused by it [226]. In addition, the effect of dietary supplementation of n-3 PUFAs on myopia, a condition that is increasing worldwide, was investigated in experiments conducted on mice modelling lens-induced myopia (LIM). The results obtained showed that EPA and its metabolites are capable of inhibiting choroidal thinning and myopia progression [229].
3.7. Grape Seed Extract and Bergamot Polyphenolic Fraction
Grape seed extract has shown beneficial effects in many diseases, thanks to its composition as a flavonoid polyphenolic compound; its main components are: +catechin, −epicatechin gallate, gallate and −epigallocatechin [230]. Beneficial properties of grape seed include prevention and treatment of diabetes and its complications [231], prevention of obesity and inflammatory reaction [232,233], the ability to modify early cerebrovascular injury caused by hypertension [234], alleviation of exercise fatigue [235], protection of the myocardium from injury [236], lowering blood lipids, regulating the metabolism, and improving the intestinal flora [237,238]. Most of these activities are carried out thanks to the antioxidant effect of grape seed extract, which makes it more powerful and effective than other plant polyphenolic compounds [239,240]. Among the active ingredients present in grape seed extract, proanthocyanidins are responsible for the biological, therapeutic and pharmacological properties [241]. Many studies have looked at the correlation between grape seed and eye disease: for example, Mani Satyam et al. showed in an in vivo study in rats that the administration of grape seed extract one day before treatment with sodium selenite, capable of inducing cataract formation in animals, was able to significantly reduce the disease and its damage [242]. Another recent study showed that treatment with grape seed extracts was able to reduce the damage of retinal degeneration caused by aging, by attenuating the expression of some pro-inflammatory cytokines [interleukin 6 (IL-6), IL-12 and IL-1β)] or the formation of their messenger RNAs [243]. The protective effects of grape seed proanthocyanidins on retinal ganglion cells have also been confirmed by an important study by Li et al., demonstrating this result in several neurodegenerative disorders [244].
Bergamot belongs to the Rutaceae family of dicotyledonous angiosperm plants that includes about 1600 species and are characterized by the presence of oleiferous glands producing ethereal aromatic oils. This plant, which belongs to the citrus genus, prefers areas with tropical and subtropical climates, and it prefers a rocky-calcareous soil [245]. The most important citrus fruits are lemon (Citrus limonum), sweet orange (Citrus sinensis), lime (Citrus aurantifolia), mandarin (Citrus reticulata), grapefruit (Citrus vitis) and bergamot (Citrus bergamia). Bergamot is a plant endemic to Calabria, Italy, and its fruit is considered a subspecies of bitter orange or a hybrid derived from bitter orange and lemon [246]. Bergamot pulp and juice are a rich source of polyphenols, including flavonoids, and are used for innumerable human health benefits, such as anti-inflammatory, antiviral [247], antitumor [248,249], hypolipidaemic [250,251], antioxidant, cardioprotective [252], antiplatelet [253] and metabolic protective effects [254]. In addition, bergamot is rich in vitamins, dietary fibre, minerals, and secondary metabolites, including polyphenols, flavonoids, carotenoids, essential oils, sugars, ascorbic acid, and some trace elements [255,256]. Among flavonoids, the main ones are neohesperidin, naringin, neodiosmin, eriodictyol, and neoerythocithrin [257,258]. This composition makes the bergamot’s flavonoid profile unique, justifying its use in many human pathologies [259,260]. Although specific data on the protective effects for the eyes of bergamot polyphenolic fraction (BPF) are not available in the literature, it is hypothetical that the high polyphenolic content and the strong antioxidant properties can also bring benefits to the ocular region. It would be appropriate to carry out relevant trials in order to confirm or deny the potential involvement of BPF on the ocular region.
4. Antioxidant Mixtures to Protect/Reduce Eye Diseases
As can be seen from the chemical structure of the compounds examined, the presence of OH groups or the catecholic group explain and justify their antioxidant properties. In fact, data available in the literature indicate that electron donors, particularly the hydroxyl group, are an essential component for performing antioxidant activity [261,262,263,264]. In the last decades, compounds described in this review are not used individually; rather, it is preferred in clinical practice to use mixtures where they are used together. Since their use has not demonstrated adverse effects in a range of concentrations, it is preferable to exploit their synergistic effects. With this strategy, each individual component of the mixture can use its own mechanism of action, providing its support to the common purpose of reducing the severity of eye diseases. Obviously, this type of treatment must be subjected to preliminary scientific studies, which clearly demonstrate that among the components used there is no interference, onset of accumulation effects, or competitive reactions [265,266,267,268,269]. For most eye diseases, the use of a mixture of antioxidants was evaluated, and patients were treated with a specific combination of high doses of zinc and antioxidant vitamins [500 mg of vitamin C, 400 units (about 270 mg) of vitamin E, 15 mg of β-carotene, 80 mg of zinc and 2 mg of copper]. In most cases, this treatment has proved to be efficient, while in healthy patients, the use of the mixture has proved ineffective in preventing the onset of disease [270,271]. Clinical studies have shown the doses of the compounds that should not be exceeded in the different mixtures used. Table 1, generated from literature data, shows the appropriate concentrations for each individual component.
Table 1.
Concentration of components to be administered in eye pathology.
The shown concentration of each component is slightly lower than the recommended daily intake value, so as to avoid potential antagonism or accumulation reactions [272]. The clinical use of antioxidant mixtures is recommended in conjunction with eye disorders in order to avoid exacerbation of the disease [273]. To date, it is common opinion in the clinical field to also administer these mixtures to healthy subjects; this decision should not be made, as already mentioned, in order to prevent the occurrence of eye diseases, but to avoid the accumulation of ROS and the consequent oxidative damage. For this very reason, it would be advisable to use these protective mixtures from the age of 40 years onwards. This can be explained by the need to not already have accumulated excessive amounts of ROS related to aging, to avoid undergoing demanding pharmacological treatments that may affect oxidative stress and have not been subjected to an excess of adverse environmental factors [274,275].
5. Conclusions
The eye is an organ continuously exposed to ionizing radiation, industrial smoke, pollutants, and engine exhausts, which makes the eyes extremely susceptible to oxidative attack. In addition, it is highly exposed to both light and robust metabolic activity. These two conditions expose the eyes to a continuous phototoxic damage and to an increase/accumulation of oxidative damage, responsible of the principal pathologies that affect their health [56,276,277]. It has been widely shown that sunlight, as well as incandescent lamps, can represent a risk factor for the health of the eye, especially for the retina. To date, the replacement of incandescent light with compact fluorescent lamps (CFLs) or light-emitting diodes (LEDs), is particularly diffuse, and these low-energy devices have increased the possibility of phototoxic eye damage [278,279,280]. On the other hand, oxidative stress, which occurs in the case of an overproduction of ROS or a failure of cellular buffering mechanisms, is able to alter the health of the eye by oxidation of biological macromolecules and an imbalance at the molecular and cellular level [281,282,283]. The main ophthalmic processes in which oxidative stress is involved are: eye surface disorders, glaucoma, diabetic retinopathy, cataracts, toxic neuropathies, uveitis, retinitis pigmentosa, and age-related macular degeneration [284,285,286,287,288]. The human body has some systems of defense against oxidative stress, which are located in the cytoplasm, in the cell membrane, and in the extracellular space: (1) enzyme systems, located in intracellular space, the best known are SOD, CAT and GSH-Px; these enzymes solve the formed ROS. SODs are metalloproteins that accelerate the dismutation of superoxide into hydrogen peroxide. There are two molecular types of SOD in humans: cytosolic (CuZnSOD), which contains copper and zinc, and mitochondrial (MnSOD), which contains manganese [289,290,291,292,293]. CAT is found in peroxisome and mitochondria and its function is to catalyze the dismutation of hydrogen peroxide in water and molecular oxygen [294,295,296,297]. GSH-Px is found in the cytosol and mitochondria. This works by eliminating hydroperoxides, transforming them into water. This reaction is associated with the transformation of reduced glutathione into oxidized GSH [298,299,300]. (2) Free radical scavengers, slow oxidation reactions, or “capture” free radicals transform them into less aggressive compounds. They can be water-soluble, like glutathione and vitamin C, or fat-soluble, like vitamin E and carotenoids. (3) Chelating agents of transition metals are molecules that bind iron and copper, avoiding that these metals act in the reactions of Fenton and Haber-Weiss [301,302,303]. The protective effects of exogenous antioxidants, taken through diet, are added to those generated by endogenous antioxidants, as described. The integration of exogenous antioxidants could take place with the administration of vitamins A, C and E and Coenzyme Q10 [304,305,306]. In this review, the protective roles of some exogenous antioxidants (vitamin A, vitamin C, Coenzyme Q10, lutein, quercetin, PUFAs and curcumin), against the main eye pathologies, in which oxidative damage is directly involved, have been investigated.
Acknowledgments
This work was supported by PON-MIUR 03PE000_78_1, PON-MIUR 03PE000_78_2 and PONa3 00359.
Abbreviations
| O2 | Oxygen; |
| H2O | water; |
| ROS | Reactive Oxygen Species; |
| O2− | Superoxide anion; |
| SOD | Superoxide Dismutase; |
| H2O2 | Hydrogen Peroxide; |
| OH• | Hydroxyl Radical; |
| SOD | Superoxide Dismutase; |
| CAT | Catalase; |
| GSH-Px | Glutathione Peroxidase; |
| RGC | Retinal Ganglion Cell; |
| ER | Endoplasmic Reticulum; |
| UPR | Unfolded Protein Response; |
| Nrf2 | 2-related factor 2; |
| iNOS | Nitric Oxide Synthase inducible; |
| NF-kB | Nuclear-kB transcription Factor; |
| FDA | Food and Drug Administration; |
| RDA | Recommended Daily Allowance; |
| IOM | Institute of Medicine; |
| coQ10 | Coenzyme Q10; |
| AMD | Age-related Macular Degeneration; |
| RNS | Reactive Nitrogen Species; |
| COX-2 | Cyclooxygenase-2; |
| PGE-2 | Prostaglandin E-2; |
| IL-1 | Interleukin 1; |
| TNF-α | Tumor Necrosis Factor α; |
| PPAR-γ | Proliferator-Activated Peroxisome Receptor γ; |
| PPRE | Peroxisome Proliferator Response Element; |
| PVR | Proliferative Vitreoretinopathy; |
| PUFAs | Polynsatured Fatty Acids; |
| LA | Linoleic Acid; |
| ALA | α-Linolenic Acid; |
| EPA | Eicosapentaenoic Acid; |
| DHA | Docosahexaenoic Acid; |
| DED | Dry eye disease; |
| BPF | Bergamot polyphenolic Fraction |
Author Contributions
J.M., V.S. and V.M. (Vincenzo Mollace) conceptualized and designed the manuscript; A.C. (Adriano Carnevali), R.M.B., F.O., M.G., V.M. (Vincenzo Musolino), C.C., F.S., S.N., L.G., S.R., A.C. (Antonio Cardamone), A.C. (Annarita Coppoletta), M.N., R.C., F.B. and R.M., wrote the manuscript. All authors have read and agreed to the published version of the manuscript.
Conflicts of Interest
The authors declare no conflict of interest.
Funding Statement
The work was supported by public resources from the Italian Ministry of Research.
Footnotes
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content.
References
- 1.Zhang J.H., Ramke J., Mwangi N., Furtado J., Yasmin S., Bascaran C., Ogundo C., Jan C., Gordon I., Congdon N., et al. Global eye health and the sustainable development goals: Protocol for a scoping review. BMJ Open. 2020;10:e035789. doi: 10.1136/bmjopen-2019-035789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Burton M.J., Faal H.B., Ramke J., Ravilla T., Holland P., Wang N., West S.K., Congdon N.G., Foster A. Announcing the Lancet global health Commission on global eye health. Lancet Glob Health. 2019;7:e1612–e1613. doi: 10.1016/S2214-109X(19)30450-4. [DOI] [PubMed] [Google Scholar]
- 3.Kels B.D., Grzybowski A., Grant-Kels J.M. Human ocular anatomy. Clin. Dermatol. 2015;33:140–146. doi: 10.1016/j.clindermatol.2014.10.006. [DOI] [PubMed] [Google Scholar]
- 4.Gupta B., Mishra V., Gharat S., Momin M., Omri A. Cellulosic Polymers for Enhancing Drug Bioavailability in Ocular Drug Delivery Systems. Pharmaceuticals. 2021;14:1201. doi: 10.3390/ph14111201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Matlen B.J., Gentner D., Franconeri S.L. Spatial alignment facilitates visual comparison. J. Exp. Psychol. Hum. Percept. Perform. 2020;46:443–457. doi: 10.1037/xhp0000726. [DOI] [PubMed] [Google Scholar]
- 6.Parravano M., Petri D., Maurutto E., Lucenteforte E., Menchini F., Varano M., van Nispen R.M.A., Virgili G. Association Between Visual Impairment and Depression in Patients Attending Eye Clinics: A Meta-analysis. JAMA Ophthalmol. 2021;139:753–761. doi: 10.1001/jamaophthalmol.2021.1557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Vlasits A., Baden T. Motion Vision: A New Mechanism in the Mammalian Retina. Curr. Biol. 2019;29:R933–R935. doi: 10.1016/j.cub.2019.08.018. [DOI] [PubMed] [Google Scholar]
- 8.Morais F.B. Vision and the Nobel Prize. Arq. Bras. Oftalmol. 2018;81:161–165. doi: 10.5935/0004-2749.20180035. [DOI] [PubMed] [Google Scholar]
- 9.Gipson I.K. The ocular surface: The challenge to enable and protect vision: The Friedenwald lecture. Investig. Opthalmol. Vis. Sci. 2007;48:4391–4398. doi: 10.1167/iovs.07-0770. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Aragona P., Rolando M. Towards a dynamic customised therapy for ocular surface dysfunctions. Br. J. Ophthalmol. 2013;97:955–960. doi: 10.1136/bjophthalmol-2012-302568. [DOI] [PubMed] [Google Scholar]
- 11.Pellegrini M., Senni C., Bernabei F., Cicero A.F.G., Vagge A., Maestri A., Scorcia V., Giannaccare G. The Role of Nutrition and Nutritional Supplements in Ocular Surface Diseases. Nutrients. 2020;12:952. doi: 10.3390/nu12040952. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.VanNasdale D.A., Jones-Jordan L.A., Hurley M.S., Shelton E.R., Robich M.L., Crews J.E. Association between Vision Impairment and Physical Quality of Life Assessed Using National Surveillance Data. Optom. Vis. Sci. 2021;98:1063–1069. doi: 10.1097/OPX.0000000000001773. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Terheyden J.H., Finger R.P. Vision-related Quality of Life with Low Vision-Assessment and Instruments] Klin. Monbl. Augenheilkd. 2019;236:261–268. doi: 10.1055/a-0838-5810. [DOI] [PubMed] [Google Scholar]
- 14.Crews J.E., Chou C.F., Zack M.M., Zhang X., Bullard K.M., Morse A.R., Saaddine J.B. The Association of Health-Related Quality of Life with Severity of Visual Impairment among People Aged 40-64 Years: Findings from the 2006–2010 Behavioral Risk Factor Surveillance System. Ophthalmic Epidemiol. 2016;23:145–153. doi: 10.3109/09286586.2016.1168851. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Marmamula S., Barrenakala N.R., Challa R., Reddy Kumbham T., Brahmanandam Modepalli S., Yellapragada R., Bhakki M., Khanna R.C., Friedman D.S. Prevalence and risk factors for visual impairment among elderly residents in ‘homes for the aged’ in India: The Hyderabad Ocular Morbidity in Elderly Study (HOMES) Br. J. Ophthalmol. 2021;105:32–36. doi: 10.1136/bjophthalmol-2019-315678. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ohno-Matsui K., Kawasaki R., Jonas J.B., Reddy Kumbham T., Brahmanandam Modepalli S., Yellapragada R., Bhakki M., Khanna R.C., Friedman D.S., Ohno-Matsui K., et al. International photographic classification and grading system for myopic maculopathy. Am. J. Ophthalmol. 2015;159:877–883. doi: 10.1016/j.ajo.2015.01.022. [DOI] [PubMed] [Google Scholar]
- 17.Lawrenson J.G., Downie L.E. Nutrition and Eye Health. Nutrients. 2019;11:2123. doi: 10.3390/nu11092123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Zhang A.C., Singh S., Craig J.P., Downie L.E. Omega-3 Fatty Acids and Eye Health: Opinions and Self-Reported Practice Behaviors of Optometrists in Australia and New Zealand. Nutrients. 2020;12:1179. doi: 10.3390/nu12041179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ibuki M., Lee D., Shinojima A., Miwa Y., Tsubota K., Kurihara T. Rice Bran and Vitamin B6 Suppress Pathological Neovascularization in a Murine Model of Age-Related Macular Degeneration as Novel HIF Inhibitors. Int. J. Mol. Sci. 2020;21:8940. doi: 10.3390/ijms21238940. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Mittler R. ROS Are Good. Trends Plant Sci. 2017;22:11–19. doi: 10.1016/j.tplants.2016.08.002. [DOI] [PubMed] [Google Scholar]
- 21.Jie Z., Liu J., Shu M., Ying Y., Yang H. Detection strategies for superoxide anion: A review. Talanta. 2022;236:122892. doi: 10.1016/j.talanta.2021.122892. [DOI] [PubMed] [Google Scholar]
- 22.Akuji M.A., Chambers D.J. Hydrogen peroxide: More harm than good? Br. J. Anaesth. 2017;118:958–959. doi: 10.1093/bja/aex151. [DOI] [PubMed] [Google Scholar]
- 23.Fleming A.M., Burrows C.J. On the irrelevancy of hydroxyl radical to DNA damage from oxidative stress and implications for epigenetics. Chem. Soc. Rev. 2020;49:6524–6528. doi: 10.1039/D0CS00579G. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Klaunig J.E. Oxidative Stress and Cancer. Curr. Pharm. Des. 2018;24:4771–4778. doi: 10.2174/1381612825666190215121712. [DOI] [PubMed] [Google Scholar]
- 25.Halliwell B. Free radicals and antioxidants: Updating a personal view. Nutr. Rev. 2012;70:257–265. doi: 10.1111/j.1753-4887.2012.00476.x. [DOI] [PubMed] [Google Scholar]
- 26.Singh A., Kukreti R., Saso L., Kukreti S. Oxidative Stress: A Key Modulator in Neurodegenerative Diseases. Molecules. 2019;24:1583. doi: 10.3390/molecules24081583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Chainy G.B.N., Sahoo D.K. Hormones and oxidative stress: An overview. Free Radic. Res. 2020;54:1–26. doi: 10.1080/10715762.2019.1702656. [DOI] [PubMed] [Google Scholar]
- 28.Senoner T., Dichtl W. Oxidative Stress in Cardiovascular Diseases: Still a Therapeutic Target? Nutrients. 2019;11:2090. doi: 10.3390/nu11092090. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Münzel T., Camici G.G., Maack C., Bonetti N.R., Fuster V., Kovacic J.C. Impact of Oxidative Stress on the Heart and Vasculature: Part 2 of a 3-Part Series. J. Am. Coll. Cardiol. 2017;70:212–229. doi: 10.1016/j.jacc.2017.05.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Pyo I.S., Yun S., Yoon Y.E., Choi J.W., Lee S.J. Mechanisms of Aging and the Preventive Effects of Resveratrol on Age-Related Diseases. Molecules. 2020;25:4649. doi: 10.3390/molecules25204649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Chunchha B., Kubo E., Singh D.P. Switching of Redox Signaling by Prdx6 Expression Decides Cellular Fate by Hormetic Phenomena Involving Nrf2 and Reactive Oxygen Species. Cells. 2022;11:1266. doi: 10.3390/cells11081266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Li H., Weng Y., Lai L., Lei H., Xu S., Zhang Y., Li L. KLF9 regulates PRDX6 expression in hyperglycemia-aggravated bupivacaine neurotoxicity. Mol. Cell Biochem. 2021;476:2125–2134. doi: 10.1007/s11010-021-04059-8. [DOI] [PubMed] [Google Scholar]
- 33.McLaughlin T., Medina A., Perkins J., Year M., Wang J.J., Zhang S.X. Switching of Redox Signaling by Prdx6 Expression Decides Cellular Fate by Hormetic Phenomena Involving Nrf2 and Reactive Oxygen Species. Mol. Neurodegener. 2022;17:25. doi: 10.1186/s13024-022-00528-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Santos F.M., Mesquita J., Castro-de-Sousa J.P., Ciordia S., Paradela A., Tomaz C.T. Vitreous Humor Proteome: Targeting Oxidative Stress, Inflammation, and Neurodegeneration in Vitreoretinal Diseases. Antioxidants. 2022;11:505. doi: 10.3390/antiox11030505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Rouen P.A., White M.L. Dry Eye Disease: Prevalence, Assessment, and Management. Home Healthc. Now. 2018;36:74–83. doi: 10.1097/NHH.0000000000000652. [DOI] [PubMed] [Google Scholar]
- 36.Seen S., Tong L. Dry eye disease and oxidative stress. Acta Ophthalmol. 2018;96:e412–e420. doi: 10.1111/aos.13526. [DOI] [PubMed] [Google Scholar]
- 37.Álvarez-Barrios A., Álvarez L., García M., Artime E., Pereiro R., González-Iglesias H. Antioxidant Defenses in the Human Eye: A Focus on Metallothioneins. Antioxidants. 2021;10:89. doi: 10.3390/antiox10010089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Bazan N.G. Neurotrophins induce neuroprotective signaling in the retinal pigment epithelial cell by activating the synthesis of the anti-inflammatory and anti-apoptotic neuroprotectin D1. Adv. Exp. Med. Biol. 2008;613:39–44. doi: 10.1007/978-0-387-74904-4_3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Pinazo-Durán M.D., Gallego-Pinazo R., García-Medina J.J., Zanón-Moreno V., Nucci C., Dolz-Marco R., Martínez-Castillo S., Galbis-Estrada C., Marco-Ramírez C., López-Gálvez M.I., et al. Oxidative stress and its downstream signaling in aging eyes. Clin. Interv. Aging. 2014;9:637–652. doi: 10.2147/CIA.S52662. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Maiuolo J., Carresi C., Gliozzi M., Musolino V., Scarano F., Coppoletta A.R., Guarnieri L., Nucera S., Scicchitano M., Bosco F., et al. Effects of Bergamot Polyphenols on Mitochondrial Dysfunction and Sarcoplasmic Reticulum Stress in Diabetic Cardiomyopathy. Nutrients. 2021;13:2476. doi: 10.3390/nu13072476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Maiuolo J., Gliozzi M., Musolino V., Carresi C., Nucera S., Macrì R., Scicchitano M., Bosco F., Scarano F., Ruga S., et al. The Role of Endothelial Dysfunction in Peripheral Blood Nerve Barrier: Molecular Mechanisms and Pathophysiological Implications. Int. J. Mol. Sci. 2019;20:3022. doi: 10.3390/ijms20123022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Kang J.M., Tanna A.P. Glaucoma. Med. Clin. N. Am. 2021;105:493–510. doi: 10.1016/j.mcna.2021.01.004. [DOI] [PubMed] [Google Scholar]
- 43.Ferreira S.M., Lerner S.F., Brunzini R., Reides C.G., Evelson P.A., Llesuy S.F. Time course changes of oxidative stress markers in a rat experimental glaucoma model. Investig. Ophthalmol. Vis. Sci. 2010;51:4635–4640. doi: 10.1167/iovs.09-5044. [DOI] [PubMed] [Google Scholar]
- 44.Kong Y.X., Crowston J.G., Vingrys A.J., Trounce I.A., Bui B.V. Functional changes in the retina during and after acute intraocular pressure elevation in mice. Investig. Ophthalmol. Vis. Sci. 2009;50:5732–5740. doi: 10.1167/iovs.09-3814. [DOI] [PubMed] [Google Scholar]
- 45.Tezel G., Yang X., Cai J. Proteomic identification of oxidatively modified retinal proteins in a chronic pressure-induced rat model of glaucoma. Investig Ophthalmol. Vis. Sci. 2005;46:3177–3187. doi: 10.1167/iovs.05-0208. [DOI] [PubMed] [Google Scholar]
- 46.Giannaccare G., Pellegrini M., Bernabei F., Senni C., Aloi M., Scalzo G.C., Ceravolo D., Iovino C., Scorcia V. Comparative analysis of ocular redness score evaluated automatically in glaucoma patients under different topical medications. Eur. J. Ophthalmol. 2021;31:2405–2411. doi: 10.1177/1120672120969612. [DOI] [PubMed] [Google Scholar]
- 47.Ahmad A., Ahsan H. Biomarkers of inflammation and oxidative stress in ophthalmic disorders. J. Immunoassay Immunochem. 2020;41:257–271. doi: 10.1080/15321819.2020.1726774. [DOI] [PubMed] [Google Scholar]
- 48.Shiels A., Hejtmancik J.F. Biology of Inherited Cataracts and Opportunities for Treatment. Annu. Rev. Vis. Sci. 2019;5:123–149. doi: 10.1146/annurev-vision-091517-034346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Periyasamy P., Shinohara T. Age-Related Cataracts: Role of unfolded protein response, Ca2+ mobilization, epigenetic DNA modifications, and loss of Nrf2/Keap1 dependent cytoprotection. Prog. Retin. Eye Res. 2017;60:1–19. doi: 10.1016/j.preteyeres.2017.08.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Maiuolo J., Bulotta S., Verderio C., Benfante R., Borgese N. Selective activation of the transcription factor ATF6 mediates endoplasmic reticulum proliferation triggered by a membrane protein. Proc. Natl. Acad. Sci. USA. 2011;108:7832–7837. doi: 10.1073/pnas.1101379108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Maiuolo J., Gliozzi M., Musolino V., Carresi C., Nucera S., Scicchitano M., Scarano F., Bosco F., Oppedisano F., Macrì R., et al. Environmental and Nutritional “Stressors” and Oligodendrocyte Dysfunction: Role of Mitochondrial and Endoplasmatic Reticulum Impairment. Biomedicines. 2020;8:553. doi: 10.3390/biomedicines8120553. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Lama A., Pirozzi C., Mollica M.P., Trinchese G., Di Guida F., Cavaliere G., Calignano A., Mattace Raso G., Berni Canani R., Meli R. Polyphenol-rich virgin olive oil reduces insulin resistance and liver inflammation and improves mitochondrial dysfunction in high-fat diet fed rats. Mol Nutr Food Res. 2017;61:1600418. doi: 10.1002/mnfr.201600418. [DOI] [PubMed] [Google Scholar]
- 53.Rowan S., Jiang S., Francisco S.G., Pomatto L.C.D., Ma Z., Jiao X., Campos M.M., Aryal S., Patel S.D., Mahaling B., et al. Aged Nrf2-Null Mice Develop All Major Types of Age-Related Cataracts. Investig. Ophthalmol. Vis. Sci. 2021;62:10. doi: 10.1167/iovs.62.15.10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Palsamy P., Bidasee K.R., Ayaki M., Augusteyn R.C., Chan J.Y., Shinohara T. Methylglyoxal induces endoplasmic reticulum stress and DNA demethylation in the Keap1 promoter of human lens epithelial cells and age-related cataracts. Free Radic. Biol. Med. 2014;72:134–148. doi: 10.1016/j.freeradbiomed.2014.04.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Palsamy P., Bidasee K.R., Shinohara T. Selenite cataracts: Activation of endoplasmic reticulum stress and loss of Nrf2/Keap1-dependent stress protection. Biochim. Biophys. Acta. 2014;1842:1794–1805. doi: 10.1016/j.bbadis.2014.06.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Palsamy P., Bidasee K.R., Shinohara T. Valproic acid suppresses Nrf2/Keap1 dependent antioxidant protection through induction of endoplasmic reticulum stress and Keap1 promoter DNA demethylation in human lens epithelial cells. Exp. Eye Res. 2014;121:26–34. doi: 10.1016/j.exer.2014.01.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Perez-Garmendia R., Lopez de Eguileta Rodriguez A., Ramos-Martinez I., Martínez Zuñiga N., Gonzalez-Salinas R., Quiroz-Mercado H., Zenteno E., Ramírez Hernández E., Hernández-Zimbrón L.F. Interplay between Oxidative Stress, Inflammation, and Amyloidosis in the Anterior Segment of the Eye; Its Pathological Implications. Oxid. Med. Cell Longev. 2020;2020:6286105. doi: 10.1155/2020/6286105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Gulcin İ. Antioxidants and antioxidant methods: An updated overview. Arch. Toxicol. 2020;94:651–715. doi: 10.1007/s00204-020-02689-3. [DOI] [PubMed] [Google Scholar]
- 59.Wołonciej M., Milewska E., Roszkowska-Jakimiec W. Trace elements as an activator of antioxidant enzymes. Postepy Hig. Med. Dosw. (Online) 2016;70:1483–1498. doi: 10.5604/17322693.1229074. [DOI] [PubMed] [Google Scholar]
- 60.Milisav I., Ribarič S., Poljsak B. Antioxidant Vitamins and Ageing. Subcell Biochem. 2018;90:1–23. doi: 10.1007/978-981-13-2835-0_1. [DOI] [PubMed] [Google Scholar]
- 61.Adeoye O., Olawumi J., Opeyemi A., Christiania O. Review on the role of glutathione on oxidative stress and infertility. JBRA Assist. Reprod. 2018;22:61–66. doi: 10.5935/1518-0557.20180003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Carazo A., Macáková K., Matoušová K., Krčmová L.K., Protti M., Mladěnka P. Vitamin A Update: Forms, Sources, Kinetics, Detection, Function, Deficiency, Therapeutic Use and Toxicity. Nutrients. 2021;13:1703. doi: 10.3390/nu13051703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Saari J.C. Vitamin A and Vision. Subcell Biochem. 2016;81:231–259. doi: 10.1007/978-94-024-0945-1_9. [DOI] [PubMed] [Google Scholar]
- 64.Koekkoek W.A., van Zanten A.R. Antioxidant Vitamins and Trace Elements in Critical Illness. Nutr. Clin. Pract. 2016;31:457–474. doi: 10.1177/0884533616653832. [DOI] [PubMed] [Google Scholar]
- 65.Blaner W.S., Shmarakov I.O., Traber M.G. Vitamin A and Vitamin E: Will the Real Antioxidant Please Stand Up? Annu. Rev. Nutr. 2021;41:105–131. doi: 10.1146/annurev-nutr-082018-124228. [DOI] [PubMed] [Google Scholar]
- 66.Bartlett H., Eperjesi F. An ideal ocular nutritional supplement? Ophthal. Physiol. Opt. 2004;24:339–349. doi: 10.1111/j.1475-1313.2004.00218.x. [DOI] [PubMed] [Google Scholar]
- 67.Martini D., Negrini L., Marino M., Riso P., Del Bo C., Porrini M. What Is the Current Direction of the Research on Carotenoids and Human Health? An Overview of Registered Clinical Trials. Nutrients. 2022;14:1191. doi: 10.3390/nu14061191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Perry A., Rasmussen H., Johnson E. Xanthophyll (lutein, zeaxanthin) content in fruits, vegetables and corn and egg products. J. Food Compos. Anal. 2009;22:9–15. doi: 10.1016/j.jfca.2008.07.006. [DOI] [Google Scholar]
- 69.Bernstein P.S., Li B., Vachali P.P., Gorusupudi A., Shyam R., Henriksen B.S., Nolan J.M. Lutein, zeaxanthin, and meso-zeaxanthin: The basic and clinical science underlying carotenoid-based nutritional interventions against ocular disease. Prog. Retin. Eye Res. 2016;50:34–66. doi: 10.1016/j.preteyeres.2015.10.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Li B., George E.W., Rognon G.T., Gorusupudi A., Ranganathan A., Chang F.Y., Shi L., Frederick J.M., Bernstein P.S. Imaging lutein and zeaxanthin in the human retina with confocal resonance Raman microscopy. Proc. Natl. Acad. Sci. USA. 2020;117:12352–12358. doi: 10.1073/pnas.1922793117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Manayi A., Abdollahi M., Raman T., Nabavi S.F., Habtemariam S., Daglia M., Nabavi S.M. Lutein and cataract: From bench to bedside. Crit. Rev. Biotechnol. 2016;36:829–839. doi: 10.3109/07388551.2015.1049510. [DOI] [PubMed] [Google Scholar]
- 72.Yoshizako H., Hara K., Takai Y., Kaidzu S., Obana A., Ohira A. Comparison of macular pigment and serum lutein concentration changes between free lutein and lutein esters supplements in Japanese subjects. Acta Ophthalmol. 2016;94:e411–e416. doi: 10.1111/aos.13106. [DOI] [PubMed] [Google Scholar]
- 73.Kijlstra A., Tian Y., Kelly E.R., Berendschot T.T. Lutein: More than just a filter for blue light. Prog. Retin. Eye Res. 2012;31:303–315. doi: 10.1016/j.preteyeres.2012.03.002. [DOI] [PubMed] [Google Scholar]
- 74.Junghans A., Sies H., Stahl W. Macular pigments lutein and zeaxanthin as blue light filters studied in liposomes. Arch. Biochem. Biophys. 2001;391:160–164. doi: 10.1006/abbi.2001.2411. [DOI] [PubMed] [Google Scholar]
- 75.Rafi M.M., Shafaie Y. Dietary lutein modulates inducible nitric oxide synthase (iNOS) gene and protein expression in mouse macrophage cells (RAW 264.7) Mol. Nutr. Food Res. 2007;51:333–340. doi: 10.1002/mnfr.200600170. [DOI] [PubMed] [Google Scholar]
- 76.Chung R.W.S., Leanderson P., Lundberg A.K., Jonasson L. Lutein exerts anti-inflammatory effects in patients with coronary artery disease. Atherosclerosis. 2017;262:87–93. doi: 10.1016/j.atherosclerosis.2017.05.008. [DOI] [PubMed] [Google Scholar]
- 77.Liu T., Liu W.H., Zhao J.S., Meng F.Z., Wang H. Lutein protects against β-amyloid peptide-induced oxidative stress in cerebrovascular endothelial cells through modulation of Nrf-2 and NFkB. Cell. Biol. Toxicol. 2017;33:57–67. doi: 10.1007/s10565-016-9360-y. [DOI] [PubMed] [Google Scholar]
- 78.Chang J., Zhang Y., Li Y., Lu K., Shen Y., Guo Y., Qi Q., Wang M., Zhang S. NrF2/ARE and NF-kB pathway regulation may be the mechanism for lutein inhibition of human breast cancer cell. Future Oncol. 2018;14:719–726. doi: 10.2217/fon-2017-0584. [DOI] [PubMed] [Google Scholar]
- 79.Tian Y., Kijlstra A., van der Veen R.L., Makridaki M., Murray I.J., Berendschot T.T. Lutein supplementation leads to decreased soluble complement membrane attack complex sC5b-9 plasma levels. Acta Ophthalmol. 2015;93:141–145. doi: 10.1111/aos.12535. [DOI] [PubMed] [Google Scholar]
- 80.Li L.H., Lee J.C.-Y., Leung H.H., Lam W.C., Fu Z., Lo A.C.Y. Lutein Supplementation for Eye Diseases. Nutrients. 2020;12:1721. doi: 10.3390/nu12061721. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Buscemi S., Corleo D., Di Pace F., Petroni M.L., Satriano A., Marchesini G. The Effect of Lutein on Eye and Extra-Eye Health. Nutrients. 2018;10:1321. doi: 10.3390/nu10091321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Ranard K.M., Jeon S., Mohn E.S., Griffiths J.C., Johnson E.J., Erdman J.W., Jr. Dietary guidance for lutein: Consideration for intake recommendations is scientifically supported. Eur. J. Nutr. 2017;56((Suppl. 3)):37–42. doi: 10.1007/s00394-017-1580-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Stahl W. Macular carotenoids: Lutein and zeaxanthin. Dev. Ophthalmol. 2005;38:70–88. doi: 10.1159/000082768. [DOI] [PubMed] [Google Scholar]
- 84.Food and Nutrition Board Staff, Panel on Dietary Antioxidants. Institute of Medicine Staff . Dietary Reference Intakes for Vitamin C, Vitamin E, Selenium, and Carotenoids. National Academy of Sciences; Washington, DC, USA: 2000. [Google Scholar]
- 85.Shao A., Hathcock J.N. Risk assessment for the carotenoids lutein and lycopene. Regul. Toxicol. Pharm. 2006;45:289–298. doi: 10.1016/j.yrtph.2006.05.007. [DOI] [PubMed] [Google Scholar]
- 86.Pehlivan F.E. Vitamin C-an antioxidant agent. In: Hamza A.H., editor. Vitamin C. IntechOpen; London, UK: 2017. [Google Scholar]
- 87.Koppenol W.H., Hider R.H. Iron and Redox Cycling. Do’s and Don’ts. Free Radic. Biol. Med. 2019;133:3–10. doi: 10.1016/j.freeradbiomed.2018.09.022. [DOI] [PubMed] [Google Scholar]
- 88.Shui Y.B., Holekamp N.M., Kramer B.C., Crowley J.R., Wilkins M.A., Chu F., Malone P.E., Mangers S.J., Hou J.H., Siegfried C.J., et al. The gel state of the vitreous and ascorbate-dependent oxygen consumption: Relationship to the etiology of nuclear cataracts. Arch. Ophthalmol. 2009;127:475–482. doi: 10.1001/archophthalmol.2008.621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Barros A.I.R.N.A., Nunes F.M., Gonçalves B., Bennett R.N., Silva A.P. Effect of cooking on total vitamin C contents and antioxidant activity of sweet chestnuts. Food Chem. 2011;128:165–172. doi: 10.1016/j.foodchem.2011.03.013. [DOI] [PubMed] [Google Scholar]
- 90.Brubaker R.F., Bourne W.M., Bachman L.A., McLaren J.W. Ascorbic acid content of human corneal epithelium. Investig. Opthalmol. Vis. Sci. 2000;41:1681–1683. [PubMed] [Google Scholar]
- 91.Talluri R.S., Katragadda S., Pal D., Mitra A.K. Mechanism of Lascorbic acid uptake by rabbit corneal epithelial cells: Evidence for the involvement of sodium-dependent vitamin C transporter 2. Curr. Eye Res. 2006;31:481–489. doi: 10.1080/02713680600693629. [DOI] [PubMed] [Google Scholar]
- 92.Liu F., Xiong J., Hu J., Ran Z., Wang J., Li Z., Chen M., Wang Y. Vitamin C and risk of age-related cataracts: A systematic review and meta-analysis. Int. J. Clin. Exp. Med. 2018;11:8929–8940. [Google Scholar]
- 93.Wei L., Liang G., Cai C., Lv J. Association of vitamin C with the risk of age-related cataract: A meta-analysis. Acta Ophthalmol. 2016;94:e170–e176. doi: 10.1111/aos.12688. [DOI] [PubMed] [Google Scholar]
- 94.Institute of Medicine . Dietary Reference Intakes: The Essential Guide to Nutrient Requirements. National Academy of Sciences Press; Washington, DC, USA: 2006. [Google Scholar]
- 95.Frei B., Birlouez I., Lykkesfeldt J. What is the optimum intake of vitamin C in humans? Crit. Rev. Food Sci. Nutr. 2012;52:815–829. doi: 10.1080/10408398.2011.649149. [DOI] [PubMed] [Google Scholar]
- 96.Bhagavan H.N., Chopra R.K. Coenzyme Q10: Absorption, tissue uptake, metabolism and pharmacokinetics. Free Radic. Res. 2006;40:445–453. doi: 10.1080/10715760600617843. [DOI] [PubMed] [Google Scholar]
- 97.Acosta M.J., Fonseca L.V., Desbats M.A., Cerqua C., Zordan R., Trevisson E., Salviati L. Coenzyme Q biosynthesis in health and disease. Biochim. Biophy. Acta (BBA)-Bioenerg. 2016;1857:1079–1085. doi: 10.1016/j.bbabio.2016.03.036. [DOI] [PubMed] [Google Scholar]
- 98.Manzar H., Abdulhussein D., Yap T.E., Cordeiro M.F. Cellular Consequences of Coenzyme Q10 Deficiency in Neurodegeneration of the Retina and Brain. Int. J. Mol. Sci. 2020;21:9299. doi: 10.3390/ijms21239299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Hargreaves I., Heaton R.A., Mantle D. Disorders of Human Coenzyme Q10 Metabolism: An Overview. Int. J. Mol. Sci. 2020;21:6695. doi: 10.3390/ijms21186695. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Groneberg D.A., Kindermann B., Althammer M., Klapper M., Vormann J., Littarru J., Doring F. Coenzyme Q10 affects expression of genes involved in cell signalling, metabolism and transport in human CaCo-2 cells. Int. J. Biochem. Cell Biol. 2005;37:1208–1218. doi: 10.1016/j.biocel.2004.11.017. [DOI] [PubMed] [Google Scholar]
- 101.Tsui H.S., Pham N.V.B., Amer B.R., Bradley M.C., Gosschalk J.E., Gallagher-Jones M., Ibarra H., Clubb R.T., Blaby-Haas C.E., Clarke C.F. Human COQ10A and COQ10B are distinct lipid-binding START domain proteins required for coenzyme Q function. J. Lipid Res. 2019;60:1293–1310. doi: 10.1194/jlr.M093534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Arenas-Jal M., Suñé-Negre J.M., García-Montoya E. Coenzyme Q10 supplementation: Efficacy, safety, and formulation challenges. Compr. Rev. Food Sci. Food Saf. 2020;19:574–594. doi: 10.1111/1541-4337.12539. [DOI] [PubMed] [Google Scholar]
- 103.Awad A.M., Bradley M.C., Fernández-Del-Río L., Nag A., Tsui H.S., Clarke C.F. Coenzyme Q10 deficiencies: Pathways in yeast and humans. Essays Biochem. 2018;62:361–376. doi: 10.1042/EBC20170106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Quinzii C.M., López L.C., Gilkerson R.W., Dorado B., Coku J., Naini A.B., Lagier-Tourenne C., Schuelke M., Salviati L., Carrozzo R., et al. Reactive oxygen species, oxidative stress, and cell death correlate with level of CoQ10 deficiency. FASEB J. 2010;24:3733–3743. doi: 10.1096/fj.09-152728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Quinzii C.M., Luna-Sanchez M., Ziosi M., Hidalgo-Gutierrez A., Kleiner G., Lopez L.C. The role of sulfide oxidation impairment in the pathogenesis of primary CoQ deficiency. Front. Physiol. 2017;8:525. doi: 10.3389/fphys.2017.00525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Heaton R.A., Heales S., Rahman K., Sexton D.W., Hargreaves I. The Effect of Cellular Coenzyme Q10 Deficiency on Lysosomal Acidification. J. Clin. Med. 2020;9:1923. doi: 10.3390/jcm9061923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Rötig A., Appelkvist E.L., Geromel V., Chretien D., Kadhom N., Edery P., Lebideau M., Dallner G., Munnich A., Ernster L., et al. Quinone-responsive multiple respiratory-chain dysfunction due to widespread coenzyme Q10 deficiency. Lancet. 2000;356:391–395. doi: 10.1016/S0140-6736(00)02531-9. [DOI] [PubMed] [Google Scholar]
- 108.Mollet J., Giurgea I., Schlemmer D., Dallner G., Chretien D., Delahodde A., Bacq D., de Lonlay P., Munnich A., Rötig A. Prenyldiphosphate synthase, subunit 1 (PDSS1) and OH-benzoate polyprenyltransferase (COQ2) mutations in ubiquinone deficiency and oxidative phosphorylation disorders. J. Clin. Investig. 2007;117:765–772. doi: 10.1172/JCI29089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Qu J., Kaufman Y., Washington I. Coenzyme Q10 in the human retina. Investig. Ophthalmol. Vis. Sci. 2009;50:1814–1818. doi: 10.1167/iovs.08-2656. [DOI] [PubMed] [Google Scholar]
- 110.Dusting G.J., Triggle C. Are we over oxidized? Oxidative stress, cardiovascular disease, and the future of intervention studies with antioxidants. Vasc. Health Risk Manag. 2005;1:93–97. doi: 10.2147/vhrm.1.2.93.64080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Nunomura A., Moreira P.I., Lee H.G., Zhu X., Castellani R.J., Smith M.A., Perry G. Neuronal Death and Survival Under Oxidative Stress in Alzheimer and Parkinson Diseases. CNS Neurol. Disord. Drug Targets. 2008;6:411–423. doi: 10.2174/187152707783399201. [DOI] [PubMed] [Google Scholar]
- 112.Lenaz G. Role of mitochondria in oxidative stress and ageing. Biochim. Biophys. Acta Bioenerg. 1998;1366:53–67. doi: 10.1016/S0005-2728(98)00120-0. [DOI] [PubMed] [Google Scholar]
- 113.Sas K., Szabó E., Vécsei L. Mitochondria, Oxidative Stress and the Kynurenine System, with a Focus on Ageing and Neuroprotection. Molecules. 2018;23:191. doi: 10.3390/molecules23010191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Bilbao-Malavé V., González-Zamora J., de la Puente M., Recalde S., Fernandez-Robredo P., Hernandez M., Layana A.G., Saenz de Viteri M. Mitochondrial Dysfunction and Endoplasmic Reticulum Stress in Age Related Macular Degeneration, Role in Pathophysiology, and Possible New Therapeutic Strategies. Antioxidants. 2021;10:1170. doi: 10.3390/antiox10081170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Schniertshauer D., Gebhard D., Bergemann J. Age-Dependent Loss of Mitochondrial Function in Epithelial Tissue Can Be Reversed by Coenzyme Q10. J Aging Res. 2018;2018:6354680. doi: 10.1155/2018/6354680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Lee D., Shim M.S., Kim K.Y., Noh Y.H., Kim H., Kim S.Y., Weinreb R.N., Ju W.K. Coenzyme Q10 inhibits glutamate excitotoxicity and oxidative stress–mediated mitochondrial alteration in a mouse model of glaucoma. Investig. Ophthalmol. Vis. Sci. 2014;55:993–1005. doi: 10.1167/iovs.13-12564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Miles M.V. The uptake and distribution of coenzyme Q10. Mitochondrion. 2007;7:S72–S77. doi: 10.1016/j.mito.2007.02.012. [DOI] [PubMed] [Google Scholar]
- 118.Nucci C., Tartaglione R., Cerulli A., Mancino R., Spano A., Cavaliere F., Rombola L., Bagetta G., Corasaniti M.T., Morrone L.A. Retinal damage caused by high intraocular pressure–induced transient ischemia is prevented by coenzyme Q10 in rat. Int. Rev. Neurobiol. 2007;82:397–406. doi: 10.1016/S0074-7742(07)82022-8. [DOI] [PubMed] [Google Scholar]
- 119.Russo R., Cavaliere F., Rombolà L., Gliozzi M., Cerulli A., Nucci C., Fazzi E., Bagetta G., Corasaniti M.T., Morrone L.A. Rational basis for the development of coenzyme Q10 as a neurotherapeutic agent for retinal protection. Prog. Brain Res. 2008;173:575–582. doi: 10.1016/S0079-6123(08)01139-4. [DOI] [PubMed] [Google Scholar]
- 120.Mancini A., Festa R., Raimondo S., Pontecorvi A., Littarru G.P. Hormonal influence on coenzyme Q10 levels in blood plasma. Int. J. Mol. Sci. 2011;12:9216–9225. doi: 10.3390/ijms12129216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Shishodia S. Molecular mechanisms of curcumin action: Gene expression. Biofactors. 2013;39:37–55. doi: 10.1002/biof.1041. [DOI] [PubMed] [Google Scholar]
- 122.Mordi R.C., Ademosun O.T., Ajanaku C.O., Olanrewaju I.O., Walton J.C. Free Radical Mediated Oxidative Degradation of Carotenes and Xanthophylls. Molecules. 2020;25:1038. doi: 10.3390/molecules25051038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Moukarzel A.A., Bejjani R.A., Fares F.N. Xanthophylls and eye health of infants and adults. J. Med. Liban. 2009;57:261–267. [PubMed] [Google Scholar]
- 124.Yuan J.P., Peng J., Yin K., Wang J.H. Potential health-promoting effects of astaxanthin: A high-value carotenoid mostly from microalgae. Mol. Nutr. Food Res. 2011;55:150–165. doi: 10.1002/mnfr.201000414. [DOI] [PubMed] [Google Scholar]
- 125.Kidd P. Astaxanthin, cell membrane nutrient with diverse clinical benefits and anti-aging potential. Altern. Med. Rev. 2011;16:355–364. [PubMed] [Google Scholar]
- 126.Fakhri S., Abbaszadeh F., Dargahi L., Jorjani M. Astaxanthin: A mechanistic review on its biological activities and health benefits. Pharmacol. Res. 2018;136:1–20. doi: 10.1016/j.phrs.2018.08.012. [DOI] [PubMed] [Google Scholar]
- 127.Ambati R.R., Phang S.M., Ravi S., Aswathanarayana R.G. Astaxanthin: Sources, extraction, stability, biological activities and its commercial applications—A review. Mar. Drugs. 2014;12:128–152. doi: 10.3390/md12010128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Davinelli S., Nielsen M.E., Scapagnini G. Astaxanthin in Skin Health, Repair, and Disease: A Comprehensive Review. Nutrients. 2018;10:522. doi: 10.3390/nu10040522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Giannaccare G., Pellegrini M., Senni C., Bernabei F., Scorcia V., Cicero A.F.G. Clinical Applications of Astaxanthin in the Treatment of Ocular Diseases: Emerging Insights. Mar. Drugs. 2020;18:239. doi: 10.3390/md18050239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Nishida Y., Nawaz A., Hecht K., Tobe K. Astaxanthin as a Novel Mitochondrial Regulator: A New Aspect of Carotenoids, beyond Antioxidants. Nutrients. 2021;14:107. doi: 10.3390/nu14010107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Higuera-Ciapara I., Félix-Valenzuela L., Goycoolea F.M. Astaxanthin: A review of its chemistry and applications. Crit. Rev. Food Sci. Nutr. 2006;46:185–196. doi: 10.1080/10408690590957188. [DOI] [PubMed] [Google Scholar]
- 132.McNulty H.P., Byun J., Lockwood S.F., Jacob R.F., Mason R.P. Differential effects of carotenoids on lipid peroxidation due to membrane interactions: X-ray diffraction analysis. Biochim. Biophys. Acta. 2007;1768:167–174. doi: 10.1016/j.bbamem.2006.09.010. [DOI] [PubMed] [Google Scholar]
- 133.Choi H.D., Kim J.H., Chang M.J., Kyu-Youn Y., Shin W.G. Effects of astaxanthin on oxidative stress in overweight and obese adults. Phytother. Res. 2011;25:1813–1818. doi: 10.1002/ptr.3494. [DOI] [PubMed] [Google Scholar]
- 134.Grattagliano I., Palmieri V.O., Portincasa P., Moschetta A., Palasciano G. Oxidative stress-induced risk factors associated with the metabolic syndrome: A unifying hypothesis. J. Nutr. Biochem. 2008;19:491–504. doi: 10.1016/j.jnutbio.2007.06.011. [DOI] [PubMed] [Google Scholar]
- 135.Kim J.H., Chang M.J., Choi H.D. Protective effects of Haematococcus astaxanthin on oxidative stress in healthy smokers. J. Med. Food. 2011;14:1469–1475. doi: 10.1089/jmf.2011.1626. [DOI] [PubMed] [Google Scholar]
- 136.Xue X.L., Han X.D., Li Y., Chu X.F., Miao W.M., Zhang J.L., Fan S.J. Astaxanthin attenuates total body irradiation-induced hematopoietic system injury in mice via inhibition of oxidative stress and apoptosis. Stem Cell Res. Ther. 2017;8:7. doi: 10.1186/s13287-016-0464-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Fang Q., Guo S., Zhou H., Han R., Wu P., Han C. Astaxanthin protects against early burn-wound progression in rats by attenuating oxidative stress-induced inflammation and mitochondria-related apoptosis. Sci. Rep. 2017;7:41440. doi: 10.1038/srep41440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Macedo R.C., Bolin A.P., Marin D.P., Otton R. Astaxanthin addition improves human neutrophils function: In vitro study. Eur. J. Nutr. 2010;49:447–457. doi: 10.1007/s00394-010-0103-1. [DOI] [PubMed] [Google Scholar]
- 139.De la Fuente M. Effects of antioxidants on immune system ageing. Eur. J. Clin. Nutr. 2002;56:S5–S8. doi: 10.1038/sj.ejcn.1601476. [DOI] [PubMed] [Google Scholar]
- 140.Yamagishi R., Aihara M. Neuroprotective effect of astaxanthin against rat retinal ganglion cell death under various stresses that induce apoptosis and necrosis. Mol. Vis. 2014;20:1796–1805. [PMC free article] [PubMed] [Google Scholar]
- 141.Otsuka T., Shimazawa M., Nakanishi T., Ohno Y., Inoue Y., Tsuruma K., Ishibashi T., Hara H. Protective effects of a dietary carotenoid, astaxanthin, against light-induced retinal damage. J. Pharmacol. Sci. 2013;123:209–218. doi: 10.1254/jphs.13066FP. [DOI] [PubMed] [Google Scholar]
- 142.Parisi V., Tedeschi M., Gallinaro G., Varano M., Saviano S., Piermarocchi S., CARMIS Study Group Carotenoids and antioxidants in age-related maculopathy italian study: Multifocal electroretinogram modifications after 1 year. Ophthalmology. 2008;115:324–333. doi: 10.1016/j.ophtha.2007.05.029. [DOI] [PubMed] [Google Scholar]
- 143.Piermarocchi S., Saviano S., Parisi V., Tedeschi M., Panozzo G., Scarpa G., Boschi G., Lo Giudice G., Carmis Study Group Carotenoids in Age-related Maculopathy Italian Study (CARMIS): Two-year results of a randomized study. Eur. J. Ophthalmol. 2012;22:216–225. doi: 10.5301/ejo.5000069. [DOI] [PubMed] [Google Scholar]
- 144.Benlarbi-Ben Khedher M., Hajri K., Dellaa A., Baccouche B., Hammoum I., Boudhrioua-Mihoubi N., Dhifi W., Ben Chaouacha-Chekir R. Astaxanthin inhibits aldose reductase activity in Psammomys obesus, a model of type 2 diabetes and diabetic retinopathy. Food Sci. Nutr. 2019;7:3979–3985. doi: 10.1002/fsn3.1259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Mahajan N., Arora P., Sandhir R. Perturbed Biochemical Pathways and Associated Oxidative Stress Lead to Vascular Dysfunctions in Diabetic Retinopathy. Oxid. Med. Cell Longev. 2019;2019:8458472. doi: 10.1155/2019/8458472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Yeh P.T., Huang H.W., Yang C.M., Yang W.S., Yang C.H. Astaxanthin Inhibits Expression of Retinal Oxidative Stress and Inflammatory Mediators in Streptozotocin-Induced Diabetic Rats. PLoS ONE. 2016;11:e0146438. doi: 10.1371/journal.pone.0146438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Ishikawa S., Hashizume K., Nishigori H., Tezuka Y., Sanbe A., Kurosaka D. Effect of astaxanthin on cataract formation induced by glucocorticoids in the chick embryo. Curr. Eye Res. 2015;40:535–540. doi: 10.3109/02713683.2014.935445. [DOI] [PubMed] [Google Scholar]
- 148.Yang M., Chen Y., Zhao T., Wang Z. Effect of astaxanthin on metabolic cataract in rats with type 1 diabetes mellitus. Exp. Mol. Pathol. 2020;113:104372. doi: 10.1016/j.yexmp.2020.104372. [DOI] [PubMed] [Google Scholar]
- 149.Ito N., Seki S., Ueda F. The Protective Role of Astaxanthin for UV-Induced Skin Deterioration in Healthy People-A Randomized, Double-Blind, Placebo-Controlled Trial. Nutrients. 2018;10:817. doi: 10.3390/nu10070817. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Libkind D., Moliné M., Colabella F. Isolation and Selection of New Astaxanthin-Producing Strains of Phaffia rhodozyma. Methods Mol Biol. 2018;1852:297–310. doi: 10.1007/978-1-4939-8742-9_18. [DOI] [PubMed] [Google Scholar]
- 151.O’Neill M.K., Piligian B.F., Olson C.D., Woodruff P.J., Swarts B.M. Tailoring Trehalose for Biomedical and Biotechnological Applications. Pure Appl. Chem. 2017;89:1223–1249. doi: 10.1515/pac-2016-1025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Hosseinpour-Moghaddam K., Caraglia M., Sahebkar A. Autophagy induction by trehalose: Molecular mechanisms and therapeutic impacts. J. Cell. Physiol. 2018;233:6524–6543. doi: 10.1002/jcp.26583. [DOI] [PubMed] [Google Scholar]
- 153.Stewart S., He X. Intracellular Delivery of Trehalose for Cell Banking. Langmuir. 2018;35:7414–7422. doi: 10.1021/acs.langmuir.8b02015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Zhang M., Oldenhof H., Sieme H., Wolkers W.F. Combining endocytic and freezing-induced trehalose uptake for cryopreservation of mammalian cells. Biotechnol. Prog. 2017;33:229–235. doi: 10.1002/btpr.2399. [DOI] [PubMed] [Google Scholar]
- 155.Menzies F.M., Fleming A., Caricasole A., Bento C.F., Andrews S.P., Ashkenazi A., Füllgrabe J., Jackson A., Jimenez Sanchez M., Karabiyik C., et al. Autophagy and Neurodegeneration: Pathogenic Mechanisms and Therapeutic Opportunities. Neuron. 2017;93:1015–1034. doi: 10.1016/j.neuron.2017.01.022. [DOI] [PubMed] [Google Scholar]
- 156.Takahashi S., Isaka M., Hamaishi M., Imai K., Orihashi K., Sueda T. Trehalose protects against spinal cord ischemia in rabbits. J. Vasc. Surg. 2014;60:490–496. doi: 10.1016/j.jvs.2013.06.078. [DOI] [PubMed] [Google Scholar]
- 157.Portbury S.D., Hare D.J., Finkelstein D.I., Adlard P.A. Trehalose improves traumatic brain injury-induced cognitive impairment. PLoS ONE. 2017;12:e0183683. doi: 10.1371/journal.pone.0183683. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Maiuolo J., Macrì R., Bava I., Gliozzi M., Musolino V., Nucera S., Carresi C., Scicchitano M., Bosco F., Scarano F., et al. Myelin Disturbances Produced by Sub-Toxic Concentration of Heavy Metals: The Role of Oligodendrocyte Dysfunction. Int. J. Mol. Sci. 2019;20:4554. doi: 10.3390/ijms20184554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Meyer N., Rinholm J.E. Mitochondria in Myelinating Oligodendrocytes: Slow and Out of Breath. Metabolites. 2021;11:359. doi: 10.3390/metabo11060359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Ugarte M., Osborne N.N. Recent advances in the understanding of the role of zinc in ocular tissues. Metallomics. 2014;6:189–200. doi: 10.1039/C3MT00291H. [DOI] [PubMed] [Google Scholar]
- 161.Sarkar S., Davies J.E., Huang Z., Tunnacliffe A., Rubinsztein D.C. Trehalose, a novel mTOR-independent autophagy enhancer, accelerates the clearance of mutant huntingtin and alpha-synuclein. J. Biol. Chem. 2007;282:5641–5652. doi: 10.1074/jbc.M609532200. [DOI] [PubMed] [Google Scholar]
- 162.Zhang Y., DeBosch B.J. Using trehalose to prevent and treat metabolic function: Effectiveness and mechanisms. Curr. Opin. Clin. Nutr. Metab. Care. 2019;22:303–310. doi: 10.1097/MCO.0000000000000568. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Kaushik J.K., Bhat R. Why trehalose an exceptional protein stabilizer? An analysis of the thermal stability of proteins in the presence of the compatible osmolyte trehalose. J. Biol. Chem. 2003;278:26458–26465. doi: 10.1074/jbc.M300815200. [DOI] [PubMed] [Google Scholar]
- 164.Jain N.K., Roy I. Trehalose and protein stability. Curr. Protoc. Protein Sci. 2010;59:4–9. doi: 10.1002/0471140864.ps0409s59. [DOI] [PubMed] [Google Scholar]
- 165.Hill-Bator A., Misiuk-Hojto M., Marycz K., Grzesiak J. Trehalose-based eye drops preserve viability and functionality of cultured human corneal epithelial cells during desiccation. Biomed. Res. Int. 2014;2014:292139. doi: 10.1155/2014/292139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Elbein A.D., Pan Y.T., Pastuszak I., Carroli D. New insights on trehalose: A multifunctional molecule. Glycobiology. 2003;13:17R–27R. doi: 10.1093/glycob/cwg047. [DOI] [PubMed] [Google Scholar]
- 167.Laihia J., Kaarniranta K. Trehalose for Ocular Surface Health. Biomolecules. 2020;10:809. doi: 10.3390/biom10050809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Da Costa Morato Nery D., da Silva C.G., Mariani D., Fernandes P.N., Pereira M.D., Panek A.D., Eleutherio E.C. The role of trehalose and its transporter in protection against reactive oxygen species. Biochim. Biophys. Acta. 2008;1780:1408–1411. doi: 10.1016/j.bbagen.2008.05.011. [DOI] [PubMed] [Google Scholar]
- 169.Lee H.J., Yoon Y.S., Lee S.J. Mechanism of neuroprotection by trehalose: Controversy surrounding autophagy induction. Cell Death Dis. 2018;9:712. doi: 10.1038/s41419-018-0749-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Chiambaretta F., Doan S., Labetoulle M., Rocher N., Fekin L.E., Messaoud R., Khairallah M., Baudouin C. A randomized, controlled study of the efficacy and safety of new eyedrop formulation for moderate to severe dry eye. Eur. J. Ophthalmol. 2017;27:1–9. doi: 10.5301/ejo.5000836. [DOI] [PubMed] [Google Scholar]
- 171.Lievens C., Berdy G., Douglass D., Montaquila S., Lin H., Simmons P., Carlisle-Wilcox C., Vehige J., Haque S. Evaluation of an enhanced viscosity artificial tear for moderate to severe dry eye disease: A multicenter, double-masked, randomized 30-day study. Cont Lens Anterior Eye. 2019;42:443–449. doi: 10.1016/j.clae.2018.12.003. [DOI] [PubMed] [Google Scholar]
- 172.Pinto-Bonilla J.C., Del Olmo-Jimeno A., Llovet-Osuna F., Hernander-Gallilea E. A randomized crossover study comparing trehalose/hyaluronate eyedrops and standard treatment:patient satisfaction in the treatment of dry eye syndrome. Ther. Clin. Risk Manag. 2015;11:595–603. doi: 10.2147/TCRM.S77091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Čejková J., Stipek S., Crkovska J., Ardan T., Platenik J., Cejka C., Midelfart A. UV Rays, the prooxidant/antioxidant imbalance in the cornea and oxidative damage. Physiol. Res. 2004;53:1–10. doi: 10.33549/physiolres.930398. [DOI] [PubMed] [Google Scholar]
- 174.Čejková J., Cejka C., Luyckx J. Trehalose treatment accelerates the healing of UVB-irradiated corneas. Comparative immunohistochemical studies on corneal cryostat sections and corneal impression cytology. Histol. Histopathol. 2012;27:1029–1040. doi: 10.14670/HH-27.1029. [DOI] [PubMed] [Google Scholar]
- 175.Talero E., Ávila-Roman J., Motilva V. Chemoprevention with phytonutrients and microalgae products in chronic inflammation and colon cancer. Curr. Pharm Des. 2012;18:3939–3965. doi: 10.2174/138161212802083725. [DOI] [PubMed] [Google Scholar]
- 176.Taylor R.A., Leonard M.C. Curcumin for inflammatory bowel disease: A review of human studies. Altern. Med. Rev. 2011;16:152–156. [PubMed] [Google Scholar]
- 177.Carmona-Ramírez I., Santamaría A., Tobón-Velasco J.C., Orozco-Ibarra M., González-Herrera I.G., Pedraza-Chaverrí J., Maldonado P.D. Curcumin restores Nrf2 levels and prevents quinolinic acid-induced neurotoxicity. J. Nutr. Biochem. 2013;24:14–24. doi: 10.1016/j.jnutbio.2011.12.010. [DOI] [PubMed] [Google Scholar]
- 178.Aggarwal S., Ichikawa H., Takada Y., Sandur S.K., Shishodia S., Aggarwal B.B. Curcumin (diferuloylmethane) down-regulates expression of cell proliferation and antiapoptotic and metastatic gene products through suppression of IκBα Kinase and akt activation. Mol. Pharmacol. 2006;69:195–206. doi: 10.1124/mol.105.017400. [DOI] [PubMed] [Google Scholar]
- 179.Prudʼhomme G.J. Cancer stem cells and novel targets for antitumor strategies. Curr. Pharm. Des. 2012;18:2838–2849. doi: 10.2174/138161212800626120. [DOI] [PubMed] [Google Scholar]
- 180.Radomska-Leśniewska D.M., Osiecka-Iwan A., Hyc A., Góźdź A., Dąbrowska A.M., Skopiński P. Therapeutic potential of curcumin in eye diseases. Cent. Eur. J. Immunol. 2019;44:181–189. doi: 10.5114/ceji.2019.87070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.Menon V.P., Sudheer A.R. Antioxidant and anti-inflammatory properties of curcumin. Adv. Exp. Med. Biol. 2007;595:105–125. doi: 10.1007/978-0-387-46401-5_3. [DOI] [PubMed] [Google Scholar]
- 182.Lin Y.G., Kunnumakkara A.B., Nair A., Merritt W.M., Han L.Y., Armaiz-Pena G.N., Kamat A.A., Spannuth W.A., Gershenson D.M., Lutgendorf S.K., et al. Curcumin inhibits tumor growth and angiogenesis in ovarian carcinoma by targeting the nuclear factor-κB pathway. Clin. Cancer Res. 2007;13:3423–3430. doi: 10.1158/1078-0432.CCR-06-3072. [DOI] [PubMed] [Google Scholar]
- 183.Marchiani A., Rozzo C., Fadda A., Delogu G., Ruzza P. Curcumin and curcumin-like molecules: From spice to drugs. Curr. Med. Chem. 2014;21:204–222. doi: 10.2174/092986732102131206115810. [DOI] [PubMed] [Google Scholar]
- 184.Ushio-Fukai M., Nakamura Y. Reactive oxygen species and angiogenesis: NADPH oxidase as target for cancer therapy. Cancer Lett. 2008;266:37–52. doi: 10.1016/j.canlet.2008.02.044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.Ushio-Fukai M. Redox signaling in angiogenesis: Role of NADPH oxidase. Cardiovasc Res. 2006;71:226–235. doi: 10.1016/j.cardiores.2006.04.015. [DOI] [PubMed] [Google Scholar]
- 186.Radomska-Leśniewska D.M., Skopiński P., Bałan J.B., Białoszewska A., Jóźwiak J., Rokicki D., Skopińska-Różewska E., Borecka A., Hevelke A. Angiomodulatory properties of Rhodiola spp. And other natural antioxidants. Cent. Eur. J. Immunol. 2015;40:249–262. doi: 10.5114/ceji.2015.52839. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Radomska-Leśniewska D.M., Bałan J.B., Skopiński P. Angiogenesis modulation by exogenous antioxidants. Cent. Eur. J. Immunol. 2017;42:370–376. doi: 10.5114/ceji.2017.72804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Nebbioso M., Franzone F., Greco A., Gharbiya M., Bonofiglio V., Polimeni A. Recent Advances and Disputes About Curcumin in Retinal Diseases. Clin. Ophthalmol. 2021;15:2553–2571. doi: 10.2147/OPTH.S306706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189.Munia I., Gafray L., Bringer M.A., Goldschmidt P., Proukhnitzky L., Jacquemot N., Cercy C., Ramchani Ben Otman K., Errera M.H., Ranchon-Cole I. Cytoprotective effects of natural highly bio-available vegetable derivatives on human-derived retinal cells. Nutrients. 2020;12:879. doi: 10.3390/nu12030879. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Niederkorn J.Y., Stern M.E., Pflugfelder S.C., De Paiva C.S., Corrales R.M., Gao J., Siemasko K. Desiccating stress induces T cell-mediated Sjogren’s Syndrome-like lacrimal keratoconjunctivitis. J. Immunol. 2006;176:3950–3957. doi: 10.4049/jimmunol.176.7.3950. [DOI] [PubMed] [Google Scholar]
- 191.Li D.Q., Luo L., Chen Z., Li D.-Q., Kim H.-S., Song X.J., Pflugfelder S.C. JNK and ERK MAP kinases mediate induction of IL-1beta, TNF-alpha and IL-8 following hyperosmolar stress in human limbal epithelial cells. Exp. Eye Res. 2006;82:588–596. doi: 10.1016/j.exer.2005.08.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Chung S.H., Choi S.H., Choi J.A., Chuck R.S., Joo C.K. Curcumin suppresses ovalbumin-induced allergic conjunctivitis. Mol. Vis. 2012;18:1966–1972. [PMC free article] [PubMed] [Google Scholar]
- 193.Gupta S.K., Agarwal R., Srivastava S., Agarwal P., Agrawal S.U., Saxena R., Galpalli N. The anti-inflammatory effects of Curcuma longa and Berberis aristata in endotoxin-induced uveitis in rabbits. Investig. Ophthalmol. Vis. Sci. 2008;4:4036–4040. doi: 10.1167/iovs.07-1186. [DOI] [PubMed] [Google Scholar]
- 194.Agarwal R., Gupta S.K., Agarwal P., Srivastava S. Topically applied standardized aqueous extract of Curcuma longa Linn. suppresses endotoxin-induced uveal inflammation in rats. Indian J. Exp. Biol. 2013;51:797–803. [PubMed] [Google Scholar]
- 195.Michalik L., Auwerx J., Berger J.P., Chatterjee V.K., Glass C.K., Gonzalez F.J., Grimaldi P.A., Kadowaki T., Lazar M.A., O’Rahilly S., et al. International Union of Pharmacology. LXI. Peroxisome proliferator-activated receptors. Pharmacol. Rev. 2006;58:726–741. doi: 10.1124/pr.58.4.5. [DOI] [PubMed] [Google Scholar]
- 196.Salehi M., Mashhadi N.S., Esfahani P.S., Feizi A., Hadi A., Askari G. The Effects of Curcumin Supplementation on Muscle Damage, Oxidative Stress, and Inflammatory Markers in Healthy Females with Moderate Physical Activity: A Randomized, Double-Blind, Placebo-Controlled Clinical Trial. Int. J. Prev. Med. 2021;12:94. doi: 10.4103/ijpvm.IJPVM_138_20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Xu D., Hu M.J., Wang Y.Q., Cui Y.L. Antioxidant Activities of Quercetin and Its Complexes for Medicinal Application. Molecules. 2019;24:1123. doi: 10.3390/molecules24061123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.Tang S.M., Deng X.T., Zhou J., Li Q.P., Ge X.X., Miao L. Pharmacological basis and new insights of quercetin action in respect to its anti-cancer effects. Biomed. Pharmacother. 2020;121:109604. doi: 10.1016/j.biopha.2019.109604. [DOI] [PubMed] [Google Scholar]
- 199.Reyes-Farias M., Carrasco-Pozo C. The Anti-Cancer Effect of Quercetin: Molecular Implications in Cancer Metabolism. Int. J. Mol. Sci. 2019;20:3177. doi: 10.3390/ijms20133177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 200.Capriglione F., Maiuolo J., Celano M., Damante G., Russo D., Bulotta S., Maggisano V. Quercetin Protects Human Thyroid Cells against Cadmium Toxicity. Int. J. Mol. Sci. 2021;22:6849. doi: 10.3390/ijms22136849. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Shen C.Y., Jiang J.G., Yan G.L., Wang D.W., Zhu W. Anti-ageing active ingredients from herbs and nutraceuticals used in traditional Chinese medicine: Pharmacological mechanisms and implications for drug discovery. Br. J. Pharmacol. 2017;174:1395–1425. doi: 10.1111/bph.13631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 202.Shen P., Lin W., Deng X., Ba X., Han L., Chen Z., Qin K., Huang Y., Tu S. Potential Implications of Quercetin in Autoimmune Diseases. Front. Immunol. 2021;12:689044. doi: 10.3389/fimmu.2021.689044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Yi H., Peng H., Wu X., Xu X., Kuang T., Zhang J., Fan G. The Therapeutic Effects and Mechanisms of Quercetin on Metabolic Diseases: Pharmacological Data and Clinical Evidence. Oxid. Med. Cell Longev. 2021;2021:6678662. doi: 10.1155/2021/6678662. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204.McKay T.B., Karamichos D. Quercetin and the ocular surface: What we know and where we are going. Exp. Biol. Med. 2017;242:565–572. doi: 10.1177/1535370216685187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 205.Liu Y., Gan L., Carlsson D.J., Fagerholm P., Lagali N., Watsky M.A., Munger R., Hodge W.G., Priest D., Griffith M. A simple, cross-linked collagen tissue substitute for corneal implantation. Investig. Ophthalmol. Vis. Sci. 2006;47:1869–1875. doi: 10.1167/iovs.05-1339. [DOI] [PubMed] [Google Scholar]
- 206.Davies N.M. Biopharmaceutical considerations in topical ocular drug delivery. Clin. Exp. Pharmacol. Physiol. 2000;27:558–562. doi: 10.1046/j.1440-1681.2000.03288.x. [DOI] [PubMed] [Google Scholar]
- 207.Nishimuro H., Ohnishi H., Sato M., Ohnishi-Kameyama M., Matsunaga I., Naito S., Ippoushi K., Akasaka H., Saitoh S., Shimamoto K., et al. Estimated daily intake and seasonal food sources of quercetin in Japan. Nutrients. 2015;7:2345–2358. doi: 10.3390/nu7042345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 208.Neamtu A.A., Maghiar T.A., Alaya A., Olah N.K., Turcus V., Neamtu C., Maghiar A.M., Mathe E. A Comprehensive View on the Quercetin Impact on Colorectal Cancer. Molecules. 2022;27:1873. doi: 10.3390/molecules27061873. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 209.Oppedisano F., Bulotta R.M., Maiuolo J., Gliozzi M., Musolino V., Carresi C., Ilari S., Serra M., Muscoli C., Gratteri S., et al. The Role of Nutraceuticals in Osteoarthritis Prevention and Treatment: Focus on n-3 PUFAs. Oxid. Med. Cell Longev. 2021;2021:4878562. doi: 10.1155/2021/4878562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210.Mollace V., Gliozzi M., Carresi C., Musolino V., Oppedisano F. Re-assessing the mechanism of action of n-3 PUFAs. Int. J. Cardiol. 2013;170((Suppl. 1)):S8–S11. doi: 10.1016/j.ijcard.2013.06.038. [DOI] [PubMed] [Google Scholar]
- 211.Oppedisano F., Macrì R., Gliozzi M., Musolino V., Carresi C., Maiuolo J., Bosco F., Nucera S., Zito M.C., Guarnieri L., et al. The Anti-Inflammatory and Antioxidant Properties of n-3 PUFAs: Their Role in Cardiovascular Protection. Biomedicines. 2020;8:306. doi: 10.3390/biomedicines8090306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 212.Oppedisano F., Mollace R., Tavernese A., Gliozzi M., Musolino V., Macrì R., Carresi C., Maiuolo J., Serra M., Cardamone A., et al. PUFA Supplementation and Heart Failure: Effects on Fibrosis and Cardiac Remodeling. Nutrients. 2021;13:2965. doi: 10.3390/nu13092965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 213.Oppedisano F., Maiuolo J., Gliozzi M., Musolino V., Carresi C., Nucera S., Scicchitano M., Scarano F., Bosco F., Macrì R., et al. The Potential for Natural Antioxidant Supplementation in the Early Stages of Neurodegenerative Disorders. Int. J. Mol. Sci. 2020;21:2618. doi: 10.3390/ijms21072618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 214.Gong Y., Fu Z., Liegl R., Chen J., Hellström A., Smith L.E. ω-3 and ω-6 long-chain PUFAs and their enzymatic metabolites in neovascular eye diseases. Am. J. Clin. Nutr. 2017;106:16–26. doi: 10.3945/ajcn.117.153825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 215.Kalogerou M., Kolovos P., Prokopiou E., Papagregoriou G., Deltas C., Malas S., Georgiou T. Omega-3 fatty acids protect retinal neurons in the DBA/2J hereditary glaucoma mouse model. Exp. Eye Res. 2018;167:128–139. doi: 10.1016/j.exer.2017.12.005. [DOI] [PubMed] [Google Scholar]
- 216.Bao J., Yang Z., Zheng S., Li J., Shentu X. Circulating fatty acids and risk of primary open-angle glaucoma: A mendelian randomization study. Gene. 2022;811:146078. doi: 10.1016/j.gene.2021.146078. [DOI] [PubMed] [Google Scholar]
- 217.Romeo Villadóniga S., Rodríguez García E., Sagastagoia Epelde O., Álvarez Díaz M.D., Domingo Pedrol J.C. Effects of Oral Supplementation with Docosahexaenoic Acid (DHA) plus Antioxidants in Pseudoexfoliative Glaucoma: A 6-Month Open-Label Randomized Trial. J. Ophthalmol. 2018;2018:8259371. doi: 10.1155/2018/8259371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 218.Saccà S.C., Cutolo C.A., Ferrari D., Corazza P., Traverso C.E. The Eye, Oxidative Damage and Polyunsaturated Fatty Acids. Nutrients. 2018;10:668. doi: 10.3390/nu10060668. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 219.Padmanabha S., Vallikannan B. Fatty acids modulate the efficacy of lutein in cataract prevention: Assessment of oxidative and inflammatory parameters in rats. Biochem. Biophys. Res. Commun. 2018;500:435–442. doi: 10.1016/j.bbrc.2018.04.098. [DOI] [PubMed] [Google Scholar]
- 220.Padmanabha S., Vallikannan B. Fatty acids influence the efficacy of lutein in the modulation of α-crystallin chaperone function: Evidence from selenite induced cataract rat model. Biochem. Biophys. Res. Commun. 2020;529:425–431. doi: 10.1016/j.bbrc.2020.06.021. [DOI] [PubMed] [Google Scholar]
- 221.Chang D., Rong S., Zhang Y., Sha Q., Liang M., Zhang X., Li M., Pan H. Serum free fatty acids level in senile cataract. J. Am. Coll Nutr. 2014;33:406–411. doi: 10.1080/07315724.2013.875420. [DOI] [PubMed] [Google Scholar]
- 222.Chen D., Chao D.L., Rocha L., Kolar M., Nguyen Huu V.A., Krawczyk M., Dasyani M., Wang T., Jafari M., Jabari M., et al. The lipid elongation enzyme ELOVL2 is a molecular regulator of aging in the retina. Aging Cell. 2020;19:e13100. doi: 10.1111/acel.13100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 223.Johansson I., Monsen V.T., Pettersen K., Mildenberger J., Misund K., Kaarniranta K., Schønberg S., Bjørkøy G. The marine n-3 PUFA DHA evokes cytoprotection against oxidative stress and protein misfolding by inducing autophagy and NFE2L2 in human retinal pigment epithelial cells. Autophagy. 2015;11:1636–1651. doi: 10.1080/15548627.2015.1061170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 224.Gorusupudi A., Liu A., Hageman G.S., Bernstein P.S. Associations of human retinal very long-chain polyunsaturated fatty acids with dietary lipid biomarkers. J. Lipid Res. 2016;57:499–508. doi: 10.1194/jlr.P065540. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 225.Chi S.C., Tuan H.I., Kang Y.N. Effects of Polyunsaturated Fatty Acids on Nonspecific Typical Dry Eye Disease: A Systematic Review and Meta-Analysis of Randomized Clinical Trials. Nutrients. 2019;11:942. doi: 10.3390/nu11050942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 226.Downie L.E., Ng S.M., Lindsley K.B., Akpek E.K. Omega-3 and omega-6 polyunsaturated fatty acids for dry eye disease. Cochrane Database Syst. Rev. 2019;12:CD011016. doi: 10.1002/14651858.CD011016.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 227.Flitter B.A., Fang X., Matthay M.A., Gronert K. The potential of lipid mediator networks as ocular surface therapeutics and biomarkers. Ocul. Surf. 2021;19:104–114. doi: 10.1016/j.jtos.2020.04.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 228.Gorusupudi A., Chang F.Y., Nelson K., Hageman G.S., Bernstein P.S. n-3 PUFA Supplementation Alters Retinal Very-Long-Chain-PUFA Levels and Ratios in Diabetic Animal Models. Mol. Nutr. Food Res. 2019;63:e1801058. doi: 10.1002/mnfr.201801058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 229.Mori K., Kuroha S., Hou J., Jeong H., Ogawa M., Ikeda S.I., Kang J.X., Negishi K., Torii H., Arita M., et al. Lipidomic analysis revealed n-3 polyunsaturated fatty acids suppressed choroidal thinning and myopia progression in mice. FASEB J. 2022;36:e22312. doi: 10.1096/fj.202101947R. [DOI] [PubMed] [Google Scholar]
- 230.Monagas M., Quintanilla-López J.E., Gómez-Cordovés C., Bartolomé B., Lebrón-Aguilar R. MALDI-TOF MS analysis of plant proanthocyanidins. J. Pharm Biomed. Anal. 2010;51:358–372. doi: 10.1016/j.jpba.2009.03.035. [DOI] [PubMed] [Google Scholar]
- 231.Prasain J.K., Peng N., Dai Y., Moore R., Arabshahi A., Wilson L. Liquid chromatography tandem mass spectrometry identification of proanthocyanidins in rat plasma after oral administration of grape seed extract. Phytomedicine. 2009;16:233–243. doi: 10.1016/j.phymed.2008.08.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 232.Gao Z., Liu G., Hu Z., Shi W., Chen B., Zou P. Grape seed proanthocyanidins protect against streptozotocin-induced diabetic nephropathy by attenuating endoplasmic reticulum stress-induced apoptosis. Mol. Med. Rep. 2018;18:1447–1454. doi: 10.3892/mmr.2018.9140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 233.Décordé K., Teissèdre P.L., Sutra T., Ventura E., Cristol J.P., Rouanet J.M. Chardonnay grape seed procyanidin extract supplementation prevents high-fat diet-induced obesity in hamsters by improving adipokine imbalance and oxidative stress markers. Mol. Nutr. Food Res. 2009;53:659–666. doi: 10.1002/mnfr.200800165. [DOI] [PubMed] [Google Scholar]
- 234.Chacón M.R., Ceperuelo-Mallafré V., Maymó-Masip E., Mateo-Sanz J.M., Arola L., Guitiérrez C. Grape-seed procyanidins modulate inflammation on human differentiated adipocytes in vitro. Cytokine. 2009;47:137–142. doi: 10.1016/j.cyto.2009.06.001. [DOI] [PubMed] [Google Scholar]
- 235.Hao J.P., Shi H., Zhang J., Zhang C.M., Feng Y.M., Qie L.Y. Role of GSPE in improving early cerebral vascular damage by inhibition of Profilin-1 expression in a ouabain-induced hypertension model. Eur. Rev. Med. Pharmacol. Sci. 2018;22:6999–7012. doi: 10.26355/eurrev_201810_16171. [DOI] [PubMed] [Google Scholar]
- 236.Xianchu L., Ming L., Xiangbin L., Lan Z. Grape seed proanthocyanidin extract supplementation affects exhaustive exercise-induced fatigue in mice. Food Nutr. Res. 2018;62:62. doi: 10.29219/fnr.v62.1421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 237.Yang D., Li S., Gao L., Lv Z., Bing Q., Lv Q. Dietary grape seed procyanidin extract protects against leadinduced heart injury in rats involving endoplasmic reticulum stress inhibition and AKT activation. J. Nutr. Biochem. 2018;62:43–49. doi: 10.1016/j.jnutbio.2018.07.013. [DOI] [PubMed] [Google Scholar]
- 238.Pons Z., Margalef M., Bravo F.I., Arola-Arnal A., Muguerza B. Chronic administration of grape-seed polyphenols attenuates the development of hypertension and improves other cardiometabolic risk factors associated with the metabolic syndrome in cafeteria diet-fed rats. Br. J. Nutr. 2017;117:200–208. doi: 10.1017/S0007114516004426. [DOI] [PubMed] [Google Scholar]
- 239.Pinent M., Bladé C., Salvadó M.J., Blay M., Pujadas G., Fernández-Larrea J. Procyanidin effects on adipocyterelated pathologies. Crit. Rev. Food Sci. Nutr. 2006;46:543–550. doi: 10.1080/10408390500354537. [DOI] [PubMed] [Google Scholar]
- 240.Jhun J.Y., Moon S.J., Yoon B.Y., Byun J.K., Kim E.K., Yang E.J. Grape seed proanthocyanidin extract-mediated regulation of STAT3 proteins contributes to Treg differentiation and attenuates inflammation in a murine model of obesity-associated arthritis. PLoS ONE. 2013;8:e78843. doi: 10.1371/journal.pone.0078843. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 241.Sherif A.A., Abdelhalim S.Z., Salim E.I. Immunohistochemical and biochemical alterations following administration of proanthocyanidin extract in rats hepatocellular carcinoma. Biomed. Pharmacother. 2017;93:1310–1319. doi: 10.1016/j.biopha.2017.07.039. [DOI] [PubMed] [Google Scholar]
- 242.Bagchi D., Bagchi M., Stohs S.J., Das D.K., Ray S.D., Kuszynski C.A. Free radicals and grape seed proanthocyanidin extract: Importance in human health and disease prevention. Toxicology. 2000;148:187–197. doi: 10.1016/S0300-483X(00)00210-9. [DOI] [PubMed] [Google Scholar]
- 243.Mollace V., Rosano G.M.C., Anker S.D., Coats A.J.S., Seferovic P., Mollace R., Tavernese A., Gliozzi M., Musolino V., Carresi C., et al. Pathophysiological Basis for Nutraceutical Supplementation in Heart Failure: A Comprehensive Review. Nutrients. 2021;13:257. doi: 10.3390/nu13010257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 244.Rodríguez-Pérez C., García-Villanova B., Guerra-Hernández E., Verardo V. Grape Seeds proanthocyanidins: An overview of in vivo bioactivity in animal models. Nutrients. 2019;11:2435. doi: 10.3390/nu11102435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 245.Miller E.J., Gemensky-Metzler A.J., Wilkie D.A., Wynne R.M., Curto E.M., Chandler H.L. Effects of grape seed extract, lutein, and fish oil on responses of canine lens epithelial cells in vitro. Am. J. Vet. Res. 2018;79:770–778. doi: 10.2460/ajvr.79.7.770. [DOI] [PubMed] [Google Scholar]
- 246.Li L., Geng X., Tian L., Wang D., Wang Q. Grape seed proanthocyanidins protect retinal ganglion cells by inhibiting oxidative stress and mitochondrial alteration. Arch. Pharm. Res. 2020;43:1056–1066. doi: 10.1007/s12272-020-01272-9. [DOI] [PubMed] [Google Scholar]
- 247.Xu J.J., Wu X., Li M.M., Li G.Q., Yang Y.T., Luo H.J., Huang W.H., Chung H.Y., Ye W.C., Wang G.C., et al. Antiviral Activity of Polymethoxylated Flavones from “Guangchenpi”, the Edible and Medicinal Pericarps of Citrus reticulata ‘Chachi’. J. Agric. Food Chem. 2014;62:2182–2189. doi: 10.1021/jf404310y. [DOI] [PubMed] [Google Scholar]
- 248.Tripoli E., La Guardia M., Giammanco S., Di Majo D., Giammanco M. Citrus flavonoids: Molecular structure, biological activity and nutritional properties: A review. Food Chem. 2007;104:466–479. doi: 10.1016/j.foodchem.2006.11.054. [DOI] [Google Scholar]
- 249.Carresi C., Musolino V., Gliozzi M., Maiuolo J., Mollace R., Nucera S., Maretta A., Sergi D., Muscoli C., Gratteri S., et al. Anti-oxidant effect of bergamot polyphenolic fraction counteracts doxorubicin-induced cardiomyopathy: Role of autophagy and c-kitposCD45negCD31neg cardiac stem cell activation. J. Mol. Cell. Cardiol. 2018;119:10–18. doi: 10.1016/j.yjmcc.2018.04.007. [DOI] [PubMed] [Google Scholar]
- 250.Musolino V., Gliozzi M., Nucera S., Carresi C., Maiuolo J., Mollace R., Paone S., Bosco F., Scarano F., Scicchitano M., et al. The effect of bergamot polyphenolic fraction on lipid transfer protein system and vascular oxidative stress in a rat model of hyperlipemia. Lipids Health Dis. 2019;18:115. doi: 10.1186/s12944-019-1061-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 251.Musolino V., Gliozzi M., Carresi C., Maiuolo J., Mollace R., Bosco F., Scarano F., Scicchitano M., Maretta A., Palma E., et al. Lipid-lowering effect of bergamot polyphenolic fraction: Role of pancreatic cholesterol ester hydrolase. J. Biol. Regul. Homeost Agents. 2017;31:1087–1093. [PubMed] [Google Scholar]
- 252.Mollace V., Scicchitano M., Paone S., Casale F., Calandruccio C., Gliozzi M., Musolino V., Carresi C., Maiuolo J., Nucera S., et al. Hypoglycemic and Hypolipemic Effects of a New Lecithin Formulation of Bergamot Polyphenolic Fraction: A Double Blind, Randomized, Placebo- Controlled Study. Endocr. Metab. Immune Disord Drug Targets. 2019;19:136–143. doi: 10.2174/1871530319666181203151513. [DOI] [PubMed] [Google Scholar]
- 253.Benavente-García O., Castillo J. Update on uses and properties of Citrus flavonoids: New findings in anticancer, cardiovascular, and anti-inflammatory activity. J. Agric. Food Chem. 2008;56:6185–6205. doi: 10.1021/jf8006568. [DOI] [PubMed] [Google Scholar]
- 254.Mahato N., Sharma K., Sinha M., Cho M.H. Citrus waste derived nutra-pharmaceuticals for health benefits: Current trends and future perspectives. J. Funct. Foods. 2018;40:307–316. doi: 10.1016/j.jff.2017.11.015. [DOI] [Google Scholar]
- 255.Taghizadeh-Alisaraei A., Hosseini S.H., Ghobadian B., Motevali A. Biofuel production from citrus wastes: A feasibility study in Iran. Renew. Sustain. Energy Rev. 2017;69:1100–1112. doi: 10.1016/j.rser.2016.09.102. [DOI] [Google Scholar]
- 256.Sharma K., Mahato N., Cho M.H., Lee Y.R. Converting citrus wastes into value-added products: Economic and environmently friendly approaches. Nutrition. 2017;34:29–46. doi: 10.1016/j.nut.2016.09.006. [DOI] [PubMed] [Google Scholar]
- 257.Cautela D., Vella F.M., Laratta B. The effect of processing methods on phyto- chemical composition in bergamot juice. Foods. 2019;8:474. doi: 10.3390/foods8100474. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 258.Di Donna L., De Luca G., Mazzotti F., Napoli A., Salerno R., Taverna D. Stat- in-like principles of bergamot fruit (Citrus bergamia): Isolation of 3-hydrox- ymethylglutaryl flavonoid glycosides. J. Nat. Prod. 2009;72:1352–1354. doi: 10.1021/np900096w. [DOI] [PubMed] [Google Scholar]
- 259.Firrman J., Liu L., Argoty G.A., Zhang L., Tomasula P., Wang M. Analysis of temporal changes in growth and gene expression for commensal gut microbes in response to the polyphenol naringenin. Microbiol. Insights. 2018;11:1–12. doi: 10.1177/1178636118775100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 260.Lima A.C.D., Cecatti C., Fidélix M.P., Adorno M.A.T., Sakamoto I.K., Cesar T.B. Effect of daily consumption of orange juice on the levels of blood glucose, lipids, and gut microbiota metabolites: Controlled clinical trials. J. Med. Food. 2019;22:202–210. doi: 10.1089/jmf.2018.0080. [DOI] [PubMed] [Google Scholar]
- 261.Mare R., Mazza E., Ferro Y., Gliozzi M., Nucera S., Paone S., Aversa I., Pujia R., Marafioti G., Musolino V., et al. A new breakfast brioche containing bergamot fiber prevents insulin and glucose increase in healthy volunteers: A pilot study. Minerva Endocrinol (Torino) 2021;46:214–225. doi: 10.23736/S2724-6507.20.03243-5. [DOI] [PubMed] [Google Scholar]
- 262.Zheng Q.T., Yang Z.H., Yu L.Y., Ren Y.Y., Huang Q.X., Liu Q., Ma X.Y., Wang Z.B., Zheng X. Synthesis and antioxidant activity of curcumin analogs. J. Asian Nat. Prod. Res. 2017;19:489–503. doi: 10.1080/10286020.2016.1235562. [DOI] [PubMed] [Google Scholar]
- 263.Frankel R.A., Michels K.A., Kim K., Kuhr D.L., Omosigho U.R., Wactawski-Wende J., Levine L., Perkins N.J., Mumford S.L. Serum antioxidant vitamin concentrations and oxidative stress markers associated with symptoms and severity of premenstrual syndrome: A prospective cohort study. BMC Womens Health. 2021;21:49. doi: 10.1186/s12905-021-01187-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 264.Ahmadi S.M., Farhoosh R., Sharif A., Rezaie M. Structure-Antioxidant Activity Relationships of Luteolin and Catechin. J. Food Sci. 2020;85:298–305. doi: 10.1111/1750-3841.14994. [DOI] [PubMed] [Google Scholar]
- 265.Harej A., Macan A.M., Stepanić V., Klobučar M., Pavelić S.K., Raić-Malić S. The Antioxidant and Antiproliferative Activities of 1,2,3-Triazolyl-L-Ascorbic Acid Derivatives. Int. J. Mol. Sci. 2019;20:4735. doi: 10.3390/ijms20194735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 266.Slavova-Kazakova A., Janiak M.A., Sulewska K., Kancheva V.D., Karamać M. Synergistic, additive, and antagonistic antioxidant effects in the mixtures of curcumin with (-)-epicatechin and with a green tea fraction containing (-)-epicatechin. Food Chem. 2021;360:129994. doi: 10.1016/j.foodchem.2021.129994. [DOI] [PubMed] [Google Scholar]
- 267.Sentkowska A., Pyrzyńska K. Investigation of antioxidant activity of selenium compounds and their mixtures with tea polyphenols. Mol. Biol. Rep. 2019;46:3019–3024. doi: 10.1007/s11033-019-04738-2. [DOI] [PubMed] [Google Scholar]
- 268.Aliaga C., López de Arbina A., Pastenes C., Rezende M.C. Antioxidant-spotting in micelles and emulsions. Food Chem. 2018;245:240–245. doi: 10.1016/j.foodchem.2017.10.094. [DOI] [PubMed] [Google Scholar]
- 269.Dawidowicz A.L., Olszowy-Tomczyk M., Typek R. Synergistic and antagonistic antioxidant effects in the binary cannabinoids mixtures. Fitoterapia. 2021;153:104992. doi: 10.1016/j.fitote.2021.104992. [DOI] [PubMed] [Google Scholar]
- 270.Akuffo K., Nolan J., Howard A., Moran R., Stack J., Klein R. Sustained supplementation and monitored response with diering carotenoid formulations in early age-related macular degeneration. Eye. 2015;29:902–912. doi: 10.1038/eye.2015.64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 271.Stringham J.M., Stringham N.T. Serum and retinal responses to three different doses of macular carotenoids over 12 weeks of supplementation. Exp. Eye Res. 2016;151:1–8. doi: 10.1016/j.exer.2016.07.005. [DOI] [PubMed] [Google Scholar]
- 272.Age-Related Eye Disease Study Research Group A randomized, placebo-controlled, clinical trial of high-dose supplementation with vitamins C and E, beta carotene, and zinc for age-related macular degeneration and vision loss: AREDS report no. 8. Arch. Ophthalmol. 2001;119:1417–1436. doi: 10.1001/archopht.119.10.1417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 273.Camelo S., Latil M., Veillet S., Dilda P.J., Lafont R. Beyond AREDS Formulations, What Is Next for Intermediate Age-Related Macular Degeneration (iAMD) Treatment? Potential Benefits of Antioxidant and Anti-inflammatory Apocarotenoids as Neuroprotectors. Oxid. Med. Cell Longev. 2020;2020:4984927. doi: 10.1155/2020/4984927. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 274.Rayapudi S., Schwartz S.G., Wang X., Chavis P. Vitamin A and fish oils for retinitis pigmentosa. Cochrane Database Syst. Rev. 2013;2013:CD008428. doi: 10.1002/14651858.CD008428.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 275.Tsao Y.T., Wu W.C., Chen K.J., Liu C.F., Hsueh Y.J., Cheng C.M., Chen H.C. An Assessment of Cataract Severity Based on Antioxidant Status and Ascorbic Acid Levels in Aqueous Humor. Antioxidants. 2022;11:397. doi: 10.3390/antiox11020397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 276.Harris A., Siesky B., Huang A., Do T., Mathew S., Frantz R., Gross J., Januleviciene I., Verticchio Vercellin A.C. Lutein Complex Supplementation Increases Ocular Blood Flow Biomarkers in Healthy Subjects. Int. J. Vitam Nutr. Res. 2019;89:5–12. doi: 10.1024/0300-9831/a000576. [DOI] [PubMed] [Google Scholar]
- 277.Kunwar A., Barik A., Sandur S.K., Indira Priyadarsini K. Differential antioxidant/pro-oxidant activity of dimethoxycurcumin, a synthetic analogue of curcumin. Free Radic. Res. 2011;45:959–965. doi: 10.3109/10715762.2011.571681. [DOI] [PubMed] [Google Scholar]
- 278.Thys-Jacobs S. Micronutrients and the premenstrual syndrome: The case for calcium. J. Am. Coll. Nutr. 2000;19:220–227. doi: 10.1080/07315724.2000.10718920. [DOI] [PubMed] [Google Scholar]
- 279.Scalia S., Marchetti N., Bianchi A. Comparative evaluation of different co-antioxidants on the photochemical- and functional-stability of epigallocatechin-3-gallate in topical creams exposed to simulated sunlight. Molecules. 2013;18:574–587. doi: 10.3390/molecules18010574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 280.Meščić Macan A., Gazivoda Kraljević T., Raić-Malić S. Therapeutic Perspective of Vitamin C and Its Derivatives. Antioxidants. 2019;8:247. doi: 10.3390/antiox8080247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 281.Suganuma M., Saha A., Fujiki H. New cancer treatment strategy using combination of green tea catechins and anticancer drugs. Cancer Sci. 2011;102:317–323. doi: 10.1111/j.1349-7006.2010.01805.x. [DOI] [PubMed] [Google Scholar]
- 282.Ozates S., Elgin K.U., Yilmaz N.S., Demirel O.O., Sen E., Yilmazbas P. Evaluation of oxidative stress in pseudo-exfoliative glaucoma patients treated with and without topical coenzyme Q10 and vitamin E. Eur. J. Ophthalmol. 2019;29:196–201. doi: 10.1177/1120672118779486. [DOI] [PubMed] [Google Scholar]
- 283.Gilbert R., Peto T., Lengyel I., Emri E. Zinc Nutrition and Inflammation in the Aging Retina. Mol. Nutr. Food Res. 2019;63:e1801049. doi: 10.1002/mnfr.201801049. [DOI] [PubMed] [Google Scholar]
- 284.O’Hagan J.B., Khazova M., Price L.L.A. Low-energy light bulbs, computers, tablets and the blue light hazard. Eye. 2016;30:230–233. doi: 10.1038/eye.2015.261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 285.Chamorro E., Carralero S.F., Bonnin-Arias C., Pérez-Carrasco M.J., de Luna J.M. Photoprotective effects of blue light absorbing filter against LED light exposure on human retinal pigment epithelial cells in vitro. J Carcinog Mutagen. 2013;S6:008. doi: 10.4172/2157-2518. [DOI] [Google Scholar]
- 286.Schick T., Ersoy L., Lechanteur Y.T., Saksens N.T., Hoyng C.B., den Hollander A.I., Kirchhof B., Fauser S. History of sunlight exposure is a risk factor for age-related macular degeneration. Retina. 2016;36:787–790. doi: 10.1097/IAE.0000000000000756. [DOI] [PubMed] [Google Scholar]
- 287.Babizhayev M.A., Yegorov Y.E. Reactive oxygen species and the aging eye: Specific role of metabolically active mitochondria in maintaining lens function and in the initiation of the oxidation-induced maturity onset cataract–A novel platform of mitochondria-targeted antioxidants with broad therapeutic potential for redox regulation and detoxification of oxidants in eye diseases. Am. J. Ther. 2016;23:e98–e117. doi: 10.1097/MJT.0b013e3181ea31ff. [DOI] [PubMed] [Google Scholar]
- 288.Cruzat A., Qazi Y., Hamrah P. In vivo confocal microscopy of corneal nerves in health and disease. Ocul. Surf. 2017;15:15–47. doi: 10.1016/j.jtos.2016.09.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 289.Williams D.L. Oxidative stress and the eye. Vet. Clin. N. Am. Small Anim. Pract. 2008;38:179–192. doi: 10.1016/j.cvsm.2007.10.006. [DOI] [PubMed] [Google Scholar]
- 290.Wong P., Markey M., Rapp C.M., Darrow R.M., Ziesel A., Organisciak D.T. Enhancing the efficacy of AREDS antioxidants in light-induced retinal degeneration. Mol. Vis. 2017;23:718–739. [PMC free article] [PubMed] [Google Scholar]
- 291.Li L., Jin R., Li Y., Yoon H.S., Yoon H.J., Yoon K.C. Effects of eye drops containing a mixture of 3% diquafosol sodium and tocopherol acetate (vitamin E) on the ocular surface of murine dry eye. Cutan. Ocul. Toxicol. 2021;40:350–358. doi: 10.1080/15569527.2021.1973022. [DOI] [PubMed] [Google Scholar]
- 292.McBee W.L., Lindblad A.S., Ferris F.L., 3rd Who should receive oral supplement treatment for age-related macular degeneration? Curr. Opin. Ophthalmol. 2003;14:159–162. doi: 10.1097/00055735-200306000-00009. [DOI] [PubMed] [Google Scholar]
- 293.Olivares-González L., Salom D., González-García E., Hervás D., Mejía-Chiqui N., Melero M., Velasco S., Muresan B.T., Campillo I., Vila-Clérigues N., et al. NUTRARET: Effect of 2-Year Nutraceutical Supplementation on Redox Status and Visual Function of Patients With Retinitis Pigmentosa: A Randomized, Double-Blind, Placebo-Controlled Trial. Front Nutr. 2022;9:847910. doi: 10.3389/fnut.2022.847910. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 294.Ivanov I.V., Mappes T., Schaupp P., Lappe C., Wahl S. Ultraviolet radiation oxidative stress affects eye health. J. Biophotonics. 2018;11:e201700377. doi: 10.1002/jbio.201700377. [DOI] [PubMed] [Google Scholar]
- 295.Hsueh Y.J., Chen Y.N., Tsao Y.T., Cheng C.M., Wu W.C., Chen H.C. The Pathomechanism, Antioxidant Biomarkers, and Treatment of Oxidative Stress-Related Eye Diseases. Int. J. Mol. Sci. 2022;23:1255. doi: 10.3390/ijms23031255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 296.Shen J., Tower J. Effects of light on aging and longevity. Ageing Res. Rev. 2019;53:100913. doi: 10.1016/j.arr.2019.100913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 297.Ozawa Y. Oxidative stress in the light-exposed retina and its implication in age-related macular degeneration. Redox Biol. 2020;37:101779. doi: 10.1016/j.redox.2020.101779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 298.Robinett N.G., Peterson R.L., Culotta V.C. Eukaryotic copper-only superoxide dismutases (SODs): A new class of SOD enzymes and SOD-like protein domains. J. Biol. Chem. 2018;293:4636–4643. doi: 10.1074/jbc.TM117.000182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 299.Habashy W.S., Milfort M.C., Rekaya R., Aggrey S.E. Cellular antioxidant enzyme activity and biomarkers for oxidative stress are affected by heat stress. Int. J. Biometeorol. 2019;63:1569–1584. doi: 10.1007/s00484-019-01769-z. [DOI] [PubMed] [Google Scholar]
- 300.Yang X.H., Li L., Xue Y.B., Zhou X.X., Tang J.H. Flavonoids from Epimedium pubescens: Extraction and mechanism, antioxidant capacity and effects on CAT and GSH-Px of Drosophila melanogaster. PeerJ. 2020;8:e8361. doi: 10.7717/peerj.8361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 301.Birben E., Sahiner U.M., Sackesen C., Erzurum S., Kalayci O. Oxidative stress and antioxidant defense. World Allergy Organ J. 2012;5:9–19. doi: 10.1097/WOX.0b013e3182439613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 302.Koppula S., Akther M., Haque M.E., Kopalli S.R. Potential Nutrients from Natural and Synthetic Sources Targeting Inflammaging-A Review of Literature, Clinical Data and Patents. Nutrients. 2021;13:4058. doi: 10.3390/nu13114058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 303.Bonacci S., Paonessa R., Costanzo P., Salerno R., Maiuolo J., Nardi M., Procopio A., Oliverio M. Peracetylation as a strategy to improve oleuropein stability and its affinity to fatty foods. Food Funct. 2018;9:5759–5767. doi: 10.1039/C8FO01874J. [DOI] [PubMed] [Google Scholar]
- 304.Bulotta S., Corradino R., Celano M., Maiuolo J., D’Agostino M., Oliverio M., Procopio A., Filetti S., Russo D. Antioxidant and antigrowth action of peracetylated oleuropein in thyroid cancer cells. J. Mol. Endocrinol. 2013;51:181–189. doi: 10.1530/JME-12-0241. [DOI] [PubMed] [Google Scholar]
- 305.Costanzo P., Oliverio M., Maiuolo J., Bonacci S., De Luca G., Masullo M., Arcone R., Procopio A. Novel Hydroxytyrosol-Donepezil Hybrids as Potential Antioxidant and Neuroprotective Agents. Front. Chem. 2021;9:741444. doi: 10.3389/fchem.2021.741444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 306.Cosco D., Paolino D., Maiuolo J., Marzio L.D., Carafa M., Ventura C.A., Fresta M. Ultradeformable liposomes as multidrug carrier of resveratrol and 5-fluorouracil for their topical delivery. Int. J. Pharm. 2015;489:1–10. doi: 10.1016/j.ijpharm.2015.04.056. [DOI] [PubMed] [Google Scholar]