Abstract
Rationale
At present, clinicians aiming to support patients through the challenges after critical care have limited evidence to base interventions.
Objectives
Evaluate a multicentre integrated health and social care intervention for critical care survivors. A process evaluation assessed factors influencing the programme implementation.
Methods
This study evaluated the impact of the Intensive Care Syndrome: Promoting Independence and Return to Employment (InS:PIRE) programme. We compared patients who attended this programme with a usual care cohort from the same time period across nine hospital sites in Scotland. The primary outcome was health-related quality of life (HRQoL) measured via the EuroQol 5-dimension 5-level instrument, at 12 months post hospital discharge. Secondary outcome measures included self-efficacy, depression, anxiety and pain.
Results
137 patients who received the InS:PIRE intervention completed outcome measures at 12 months. In the usual care cohort, 115 patients completed the measures. The two cohorts had similar baseline demographics. After adjustment, there was a significant absolute increase in HRQoL in the intervention cohort in relation to the usual care cohort (0.12, 95% CI 0.04 to 0.20, p=0.01). Patients in the InS:PIRE cohort also reported self-efficacy scores that were 7.7% higher (2.32 points higher, 95% CI 0.32 to 4.31, p=0.02), fewer symptoms of depression (OR 0.38, 95% CI 0.19 to 0.76, p=0.01) and similar symptoms of anxiety (OR 0.58, 95% CI 0.30 to 1.13, p=0.11). There was no significant difference in overall pain experience. Key facilitators for implementation were: integration with inpatient care, organisational engagement, flexibility to service inclusion; key barriers were: funding, staff availability and venue availability.
Conclusions
This multicentre evaluation of a health and social care programme designed for survivors of critical illness appears to show benefit at 12 months following hospital discharge.
Keywords: critical care, ARDS, pulmonary rehabilitation
Key messages.
What is already known on this topic?
Post intensive care syndrome is well studied and describes the problems faced by survivors of critical illness. At present there is limited evidence describing effective treatments which may help mitigate the issues faced by survivors of critical illness.
What this study adds?
The implementation of an integrated model of health and social care, which supports survivors of critical illness, is feasible. Those who attended the Intensive Care Syndrome: Promoting Independence and Return to Employment programme, appeared to have improved health-related quality of life in comparison to critical care survivors who had not received the intervention.
How this study might affect research, practice or policy and what are the implications of this study?
Future research for critical illness survivors should focus on complex approaches, which combine health and social care.
Introduction
Survivors of critical illness can face significant challenges following discharge. These challenges are multifaceted and include new or worsening emotional, physical, cognitive and social problems.1–5 Collectively known as post intensive care syndrome (PICS), these problems can lead to significant costs for the individual, the healthcare system and society.6
As a result of the COVID-19 pandemic, there has been an energised focus by critical care clinicians to create follow-up services.7 Many of these services have been modelled on established programmes of work which include peer support and multidisciplinary teams (MDTs).8 9 However, there is limited evidence for this type of intervention, with minimal data demonstrating the effectiveness of any care model, following critical care discharge.10 As such, there is an urgent need for evaluation studies in this area.
This multicentre study aimed to evaluate whether an integrated health and social care intervention following critical illness had a measurable effect on Health-Related Quality of Life (HRQoL) 1 year after hospital discharge. Specifically, using a contemporary control, we report the effects of this programme on HRQoL, emotional health and pain. An embedded process evaluation assessed factors influencing the programme implementation.
Methods
Participants gave written informed consent to participate in the study before taking part.
Study setting
The study involved the expansion of the Intensive care Syndrome: Promoting Independence and Return to Employment (InS:PIRE) programme from a single intensive care unit (ICU), to four other ICUs throughout Scotland between 2016 and 2020.
Study design
We used a multicentre prospective cohort study design, with the aim of understanding the impact of the InS:PIRE programme on HRQoL for ICU survivors. We compared this intervention cohort with patients who had been admitted to ICU and had not received the intervention. We report a cohort study, as per the Strengthening the Reporting of Observational Studies in Epidemiology guidelines.
A process evaluation was undertaken via inperson learning sessions which took place twice a year across the implementation and evaluation period (2.5 years). All sites were represented by members of the MDT at each learning session. Specifically details about barriers and facilitators to successful implementation were captured from a staffing and service perspective. These details were presented by individual teams at each learning session. TQ and JM analysed and interpreted these findings.
Intervention
The InS:PIRE programme is a complex intervention which has been described previously.11 Briefly, all patients receive individual reviews with: (1) ICU doctor and nurse; (2) Pharmacist; and (3) Physiotherapist. These reviews offer a debrief of the ICU stay, an assessment of ongoing problems, goal setting and patient-directed recovery plans. Patients were considered to have completed the intervention if they received these three ‘core’ reviews. Clinical neuropsychology input is available at every site via group sessions and individual reviews as required. Peer support is embedded in the programme through the use of shared waiting areas, group sessions, and the presence of patient and caregiver volunteers further along the recovery trajectory.12 Vocational (occupational) rehabilitation and support for caregivers is also integrated. Patients attend InS:PIRE longitudinally, initially attending for 5 weeks, with return appointments at 3 months and 12 months.
The intervention involved local community organisations, determined by local clinical teams. Specifically, financial and social care advice was available to patients, including advice on welfare benefits and housing.13 These sessions were delivered through a combination of individual appointments, drop-in sessions or group discussions.
During the expansion of InS:PIRE, each new site conducted focus groups involving local ICU patients and caregivers. These groups ensured that the model was feasible and responsive to local care needs. The groups also helped establish and refine the outcome measures used.
Participants are invited between 4 weeks and 12 weeks after hospital discharge. Inclusion criteria were: patients receiving level 3 care (multiple organ support and/or invasive respiratory support) or more than 7 days of level 2 care (single organ support or postoperative care).14 In contrast to the feasibility work from InS:PIRE, there was no upper age limit for inclusion. Exclusion criteria were any patient who was terminally ill, had suffered a traumatic brain injury or was an inpatient under psychiatric services. We provide further information and a conceptual overview in online supplemental material S1.
thoraxjnl-2021-218428supp001.pdf (758.2KB, pdf)
Intervention cohort
Five sites implemented the InS:PIRE programme as part of a quality improvement collaborative (intervention cohort) over 2 years. The intervention cohort were consecutively recruited to this study during the initial InS:PIRE programme attendance. Intervention cohort recruitment occurred between May 2016 to October 2018 (follow-up completed December 2019). Participants completed outcome measures at a preplanned 12-month follow-up. Participants were given the opportunity to complete questionnaires inperson or via telephone.
Usual care cohort
The usual care cohort were recruited by postal survey between 10 months and 16 months post hospital discharge, from eight hospitals in Scotland. These sites have the same patient case mix as the intervention cohort. Only sites which did not have any ICU follow-up services, at the time of recruitment, were included with the same inclusion/exclusion criteria as the intervention cohort. Patients from four of the control sites were included in the control group, before the site implemented the intervention. These patients received no follow-up care and were not invited to attend the intervention. Questionnaire packs and prepaid envelopes were sent to eligible patients. Reminder packs were sent for non-responders after 1 month.
Usual care cohort questionnaires were sent between June 2017 and March 2020. Although ethical approval was in place to continue beyond March 2020, the impact of the COVID-19 pandemic was unknown, and this study was closed to minimise any confounding effect.
Outcome measures
Health-related quality of life
The primary outcome of HRQoL at 1 year measured by EuroQol 5-Dimension 5-level dimension 5-level (EQ-5D-5L) was decided a priori.15 16 This survey generates two summary measures of HRQoL. First, the health utility score summarises five domains (mobility, self-care, usual activities, pain/discomfort and anxiety/depression) into a single number with 1.0 representing the best possible health, 0.0 representing a health state equivalent to death and negative values representing a state worse than death. The minimal clinically important difference (MCID) for this score is 0.08.17 18 Second, the EuroQol Visual Analogue Scale records participants’ self-rated health on the day of testing by marking on a continuous scale from 0 (worst health) to 100 (best health) with an MCID of 8%.17 18
Self-efficacy
Self-efficacy was measured to understand the mechanisms behind any changes in HRQoL. The General Self-Efficacy (GSE) Scale, which is a 10-item questionnaire generating a score with 31 levels (minimum 10 to maximum 40) was used to quantify these effects.19 20
Mental health outcomes
The Hospital Anxiety and Depression Scale (HADS) was used to evaluate mental health. HADS generates two separate seven-item scores; one for anxiety and one for depression.21 The cut-offs for these scores are as shown in online supplemental material S2. For the purposes of this analysis, we defined anxiety and depression as a score of 8 or greater (≥8/21) in the respective subscores.2 21 Both HADS and EQ-5D-5L have been recommended as core outcome measures in acute respiratory failure research.22 Appropriate licensing requirements were in place.
Pain outcomes
The Brief Pain Inventory (BPI) short form was used to measure pain.23 The BPI is summarised in online supplemental material S2. In this study, pain scores of intervention and usual care cohorts were compared, including: (average and worst scores), mean or summary pain interference (pain effects on life), pain interference on work and pain interference in enjoyment of life. The authors of BPI recommend that all scores are calculated as an average.
Baseline demographics
Comorbidity and in-hospital data were obtained via electronic medical records. The Scottish Index of Multiple Deprivation (SIMD) was used to define deprivation for each participant. SIMD is a relative measure of deprivation across geographically defined data zones in Scotland.24 This is either given in deciles or quintiles; for the purpose of this analysis we chose quintiles to use fewer degrees of freedom in the modelling process thus allowing the inclusion of important clinical variables.
Statistical analysis
Wilcoxon rank-sum, Pearson χ2 and Fisher’s exact tests were used to analyse differences in baseline demographics between the cohorts. To evaluate the effect of the intervention in relation to usual care, multivariable regression was used. Linear regression was used to evaluate continuous variables (EQ-5D, GSE, BPI) and logistic regression for categorical outcomes (HADS-anxiety and HADS-depression). The effect of the intervention against the usual care cohort is reported as either an absolute change (continuous variables) or an OR for anxiety or depression. Relative increases are calculated as the estimated difference over the full range of the outcome measure.
Missing values were imputed with multivariate imputation by chained equations using 5 imputations and 30 iterations.25 Analyses were carried out using R V.4.0.4.26 A significance value of p<0.05 was used.
Models were created using domain knowledge, outputs from a recent expert consensus conference and previous evidence.2 5 27–30 Covariates for adjustment were chosen before data analysis. All models were adjusted for: surgery at admission or in the first week of ICU; time from hospital discharge to follow-up; age; gender; ICU length of stay; Acute Physiology and Chronic Health Evaluation II (APACHE ll) Score; deprivation; history of harmful alcohol or drug use; pre-existing psychiatric diagnoses; and a pre-ICU history of chronic pain.
The final modelled effect estimates and standardised errors which were created with imputed data, were pooled using standard Rubin’s rules. All estimated effect errors were generated in a robust manner using a sandwich estimator.
Sensitivity analyses
We undertook a sensitivity analysis using a propensity score matched cohort approach. Specific details alongside full results of this approach are presented in online supplemental material S3. The decision to match, and the matching approach was planned a priori. We propensity matched the intervention cohort with the usual care cohort, using nearest neighbour matching (calliper=0.1). Covariate balance was reviewed between the cohorts using Pearson’s χ2 test and the Mann‐Whitney U Test. Covariates were iteratively included in the match until balance in the two cohorts was achieved. This process was completed before considering any outcome variables. The following covariates were included in the propensity score: surgery at admission or in the first week of ICU; time from hospital discharge to follow-up; age; hospital length of stay; advanced respiratory support; ICU length of stay; history of harmful alcohol or drug use; pre-existing psychiatric diagnoses. Once matching was complete the outcome measures underwent the same adjustment strategies used in the primary analysis of the unmatched cohorts.
A further sensitivity analysis using a mixed effects analysis, aimed to account for any clustering effects due to hospital site variation for the main outcome measure (HRQoL). This analysis measured the variability between hospital type (ie, large tertiary referral hospital or medium general acute hospital) (online supplemental material S4).
Results
Baseline characteristics
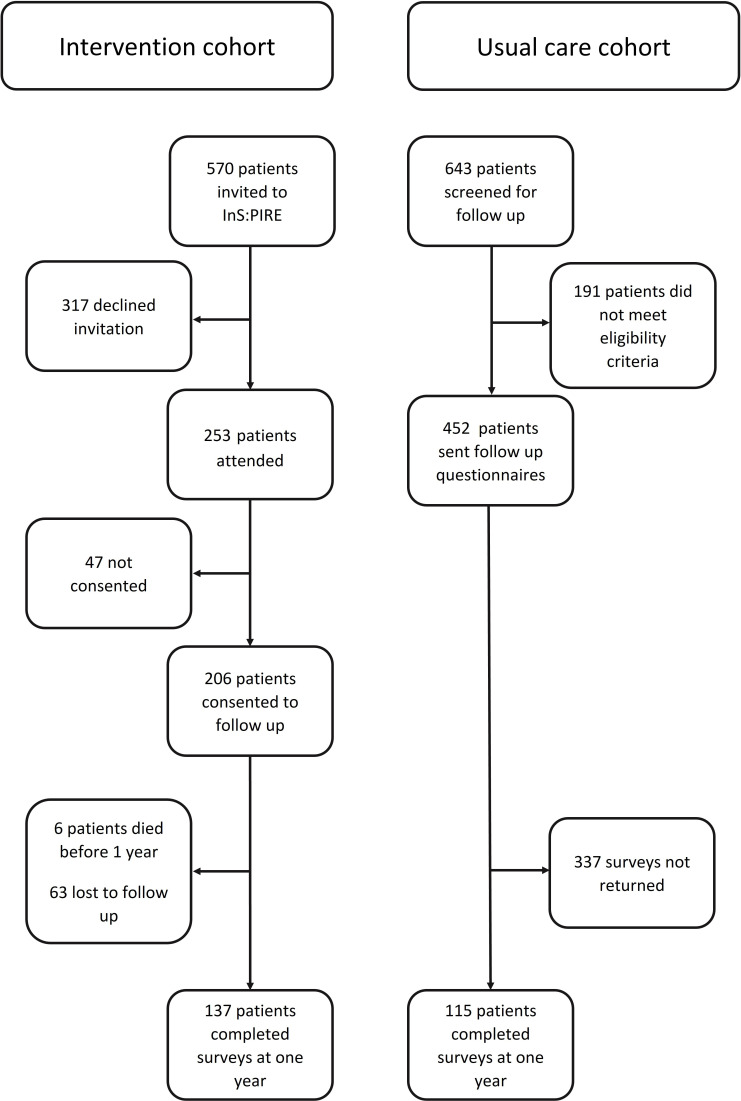
Five hundred and seventy patients were invited to attend InS:PIRE; 253 attended and 206 patients consented to participate in the research study. Six patients died before 1-year follow-up and 63 were lost to follow-up. Thus, 137/200 (68.5%) patients who received the intervention completed outcome measures at 1 year (figure 1).
Figure 1.
Recruitment flow chart. Patient flow and recruitment, intervention cohort and usual care cohort. InS:PIRE, Intensive Care Syndrome: Promoting Independence and Return to Employment.
In the usual care cohort, 643 patients were screened; 191 were ineligible. As such, 452 were sent questionnaire packs, of which 115 (25.4%) were returned (figure 1). Details of responders and non-responders of the postal survey are described in online supplemental material S5.
The cohorts had a similar age (58.7 (IQR: 50.8–67.6) years vs 63.5 (IQR: 49.5–71.5) years), severity of illness (APACHE II: 20.0 (IQR: 15.0–25.3) vs 19.0 (IQR: 14.2–25.0)), time to follow-up (15.2 (IQR:13.2–16.5) months vs 15.9 (14.8–17.3) months), and there was a similar spread in both cohorts across the socioeconomic gradient. There was a difference in admission specialty profile as well as hospital and ICU length of stay across cohorts (table 1). To account for these imbalances, baseline demographics were adjusted for as outlined in the methods section. Modelling strategy details and outputs can be found in online supplemental material S6. A breakdown of missing variables is shown in online supplemental material S7.
Table 1.
Baseline characteristics for intervention and usual care cohorts (unmatched and unadjusted)
| Demographic | Usual care cohort (n=115) |
Intervention cohort (n=137) |
P value |
| Age, years, median (IQR) | 63.5 (49.5–71.5) | 58.7 (50.8–67.6) | 0.06 |
| Gender, male (%) | 67 (58.3) | 73 (53.3) | 0.43 |
| Admitting specialty (%): | 0.03 | ||
| Medical | 52 (44.8) | 83 (60.6) | |
| Surgery | 60 (52.2) | 54 (39.4) | |
| ICU length of stay, median (IQR) | 4.95 (2.5–9.5) | 10.5 (6.9–17.3) | <0.01 |
| Hospital length of stay, median (IQR) | 18.0 (11.4–35.0) | 30.5 (17.0–49.6) | <0.01 |
| APACHE II Score, median (IQR) | 19 (14.2–25.0) | 20 (15.0–25.3) | 0.28 |
| Advanced respiratory support (%) | 100 (87.0) | 121 (88.3) | 0.81 |
| Complex cardiovascular support requiring multiple vasoactive drugs (%) | 21 (18.3) | 30 (21.9) | 0.54 |
| Renal replacement therapy (%) | 19 (16.5) | 32 (23.4) | 0.21 |
| Two or greater comorbidities (%) | 54 (47.0) | 60 (43.8) | 0.41 |
| Charlson Comorbidity Index (CCI) Score, median (IQR) | 3 (1–4) | 3 (1–4) | 0.53 |
| Pre-existing psychiatric diagnosis (%) | 28 (24.3) | 39 (28.5) | 0.60 |
| History of harmful alcohol or drug use (%) | 15 (13.0) | 25 (18.2) | 0.33 |
| Premorbid history of chronic pain (%) | 15 (13.0) | 18 (13.1) | 0.91 |
| Deprivation index, SIMD 2016 (%): | 0.31 | ||
| Quintile 1 (most deprived) | 34 (29.6) | 50 (36.5) | |
| Quintile 2 | 27 (23.5) | 36 (26.3) | |
| Quintile 3 | 12 (10.4) | 20 (14.6) | |
| Quintile 4 | 18 (15.7) | 14 (10.2) | |
| Quintile 5 (least deprived) | 21 (18.3) | 17 (12.4) | |
| Time to follow-up, median months (IQR) | 15.2 (13.2–16.5) | 15.9 (14.8–17.3) | <0.01 |
Time to follow-up, months, from hospital discharge. For missing data, see online supplemental material S7.
ICU, intensive care unit; APACHE II, Acute Physiology and Chronic Health Evaluation II; SIMD, Scottish Index of Multiple Deprivation.
Outcomes
Health-related quality of life
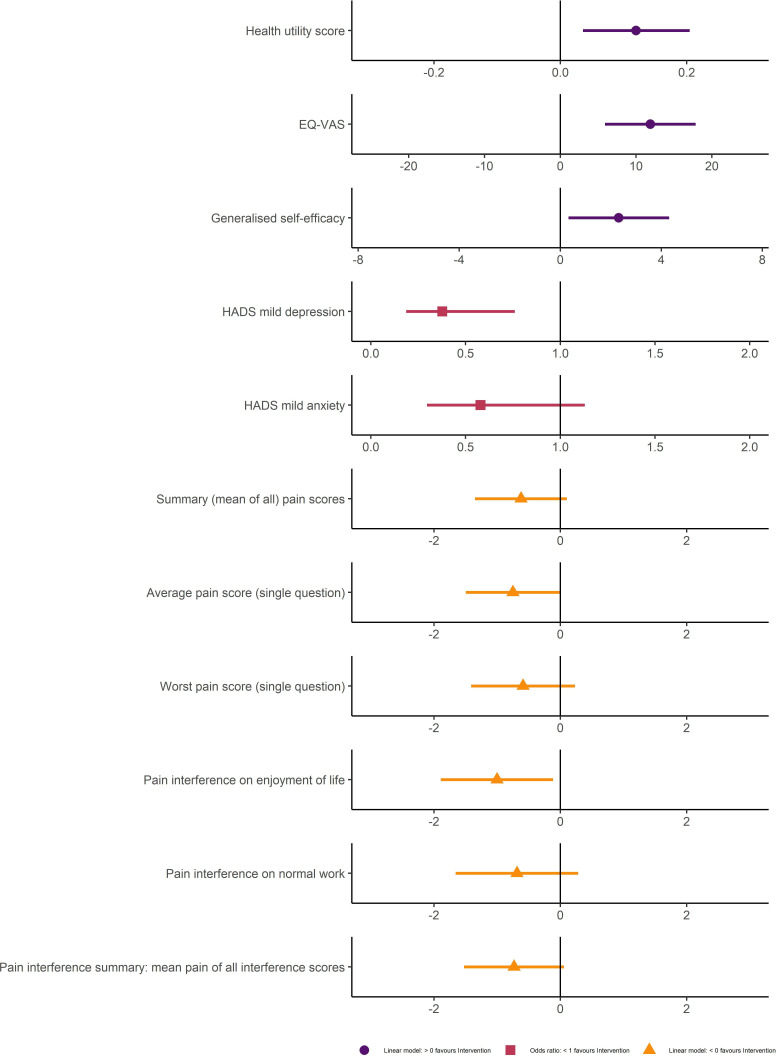
The intervention cohort demonstrated a 0.12 (95% CI 0.04 to 0.20, p=0.01) adjusted absolute increase (7.5% relative increase) in EQ-5D health utility scores at 1 year, in comparison to the usual care cohort (table 2). Patients in the intervention cohort also experienced an adjusted absolute increase in EQ-VAS of 11.88% (95% CI 5.91 to 17.86, p<0.001). The adjusted effects of the intervention compared with usual care on both EQ-5D summary scores as well as all other outcomes are summarised in figure 2.
Table 2.
Effect of intervention at 1-year follow-up (unmatched)
| Outcome measure | Adjusted estimate | P value | 95% CI | Relative difference with intervention |
| EQ-5D summary scores | ||||
| Health utility score | 0.12 | 0.01 | 0.04 to 0.20 | 7.5 % |
| EQ-5D VAS | 11.88 | <0.001 | 5.91 to 17.86 | 11.9 % |
| Generalised self-efficacy | 2.32 | 0.02 | 0.32 to 4.31 | 7.7 % |
| Brief Pain Inventory Scores | ||||
| Summary (mean) pain score (across BPI) | −0.62 | 0.09 | −1.35 to 0.11 | 6.2 % |
| Average pain score (single question) | −0.75 | 0.05 | −1.50 to 0.00 | 7.5 % |
| Worst pain score (single question) | −0.59 | 0.16 | −1.41 to 0.23 | 5.9% |
| Pain interference with enjoyment of life (single question) | −1.00 | 0.03 | −1.89 to −0.11 | 10.0 % |
| Pain interference on normal work (single question) | −0.69 | 0.16 | −1.66 to 0.28 | 6.9% |
| Mean pain interference summary | −0.73 | 0.07 | −1.52 to 0.06 | 7.3% |
| Hospital anxiety and depression (HADS) ORs | ||||
| HADS depression | 0.38 | 0.01 | 0.19 to 0.76 | 62 % |
| HADS anxiety | 0.58 | 0.11 | 0.30 to 1.13 | 43 % |
Effect of the intervention on quality-of-life outcome measures compared with usual care at 1-year follow-up. Linear regression models with absolute effects and scaled relative effects for: (1) EQ-5D, EuroQol five-dimension health state; (2) EQ-VAS: EuroQol Visual Analogue Scale; (3) Generalised self efficacy (GSE); (4) Summary (mean of all) pain scores: mean of four scores from brief pain inventory; (5) ‘average pain’; (6) ‘worst pain’; (7) ‘least pain’; (8) ‘pain right now’. All pain scores ranges=0–10. Logistic regression, with ORs for risk of screening for depression (HADS-depression ≥8/21) and anxiety (HADS-anxiety ≥8/21).
BPI, Brief Pain Inventory; HADS, Hospital Anxiety and Depression Scale; VAS, Visual Analogue Scale.
Figure 2.
Forest plot (adjusted unmatched). Effect of the intervention on measured outcomes representing the absolute difference in scores (linear models) or risk of screening positive for the condition (ORs), 1 year after intensive care compared with usual care. Point estimate values (circle, square, triangle) and 95% CI. InS:PIRE, Intensive care Syndrome: Promoting Independence and Return to Employment; EuroQol Health Utility Score, absolute difference, taken from EuroQol 5-Dimension 5-level (EQ-5D-5L) ‘crosswalk UK scores’, range - 0.594 to 1.0; EuroQol Visual Analogue Scale (EQ-VAS), absolute difference, range 0 to 100; Generalised Self Efficacy: absolute difference, range 10 to 40; HADS: Hospital Anxiety and Depression Scale with individual component scores for anxiety and depression, odds risk ratios of screening positive for anxiety or depression; Brief pain inventory (BPI), absolute difference in pain scores, all scores range from 0 to 10, average (single component score), worst pain (single component score), and summary score (composite / mean score from four pain scores: ‘average’, ‘worst’, ‘least’ and ‘pain right now’); Pain interference scores from BPI, absolute difference, scores range from 0 to 10, enjoyment in life (single component), work (single component), and summary pain interference (composite / mean score from seven interference components).
Self-efficacy
The intervention cohort had an adjusted absolute increase in self-efficacy of 2.32 points (95% CI 0.32 to 4.31, p=0.02) resulting in a relative increase of 7.7% (table 2) at 12 months, in comparison to the usual care cohort.
Mental health outcomes
Defining anxiety or depression as a score of 8 or greater in HADS, the intervention cohort had a 62% adjusted odds reduction of screening for depression compared with the usual care cohort (OR 0.38, 95% CI 0.19 to 0.76, p=0.01) at 12 months (table 2). Odds of screening for anxiety at 1 year in intervention vs usual care was not significantly different (OR 0.58, 95% CI 0.30 to 1.13, p=0.11) in this analysis.
Pain outcomes
The number of patients reporting having had pain ‘other than everyday kinds of pain’ was 149/252 (59.1%) across both cohorts. Adjusted linear regression models demonstrated those in the intervention cohort had a 7.5% reduction in average pain score (single question) at 1 year compared with usual care (95% CI −1.50 to 0.00, p=0.05). No statistically significant differences were observed in the worst pain score, or the summary pain score. There was a 10% reduction in the interference of pain on enjoyment in life (95% CI:−1.89 to −0.11, p=0.03). All adjusted effects of the intervention on pain are described in table 2.
Sensitivity analysis: propensity score matching
We were able to successfully match almost two-thirds (65.2%) of the usual care cohort to the intervention cohort. The unadjusted outcome measure differences between the matched intervention and usual care cohort are presented in online supplemental material S8. The comparison of the matched demographics, alongside the adjusted effect of the intervention on the various outcome measures is shown in figure 3. The adjusted matched and unmatched analyses also demonstrated increased HRQoL and self-efficacy scores, and reduced rates of depression. A full description of propensity score matching, and results are shown in online supplemental material S3. Variance between hospital sites (cluster sensitivity analyses) were minimal for all models (online supplemental material S4). The effects across the different outcomes are summarised in online supplemental material S9.
Figure 3.
Propensity score matching panel. Demographic table (A): representative dataset of baseline characteristics after propensity score matching. ICU, intensive care unit; APACHE II, Acute Physiology and Chronic Health Evaluation II; SIMD, Scottish index of multiple deprivation; time to follow-up, months, from hospital discharge. Outcome measures table with splines (B): effects of intervention on all outcome measures alongside coefficient graph of effect size. Effect of intervention: absolute change in scores (linear models) and odds risk ratio of screening for the condition (anxiety or depression) 1 year after intensive care compared with usual care. Point estimate values (circle, square, triangle) and 95% CI. InS:PIRE, Intensive Care Syndrome: Promoting Independence and Return to Employment; EuroQol Health Utility Score, absolute change, taken from EuroQol 5-Dimension 5-level (EQ-5D-5L) ‘crosswalk UK scores’, range - 0.594 to 1.0; EuroQol Visual Analogue Scale (EQ-VAS), absolute change, range 0 to 100; Generalised Self-efficacy: absolute change, range 10 to 40; HADS: Hospital Anxiety and Depression Scale with individual component scores for anxiety and depression, odds risk ratios of screening positive for anxiety or depression; Brief pain inventory (BPI), absolute change in pain scores, all scores range from 0 to 10, average (single component score), worst pain (single component score), and summary score (composite/mean score from four pain scores: ‘average’, ‘worst’, ‘least’ and ‘pain right now’); pain interference scores from BPI, absolute change, scores range from 0 to 10, enjoyment in life (single component), work (single component), and summary pain interference (composite/mean score from seven interference components).
Process evaluation
The process evaluation identified a number of facilitators and barriers to the implementation of this intervention (figure 4). Facilitators included a flexible approach to inclusion of key services. Every site included social and economic support but this could be provided by different services (ie, statutory community organisations, in-hospital financial services or local charities). This flexibility ensured that patients received the correct intervention in a manageable and sustainable way. Other facilitators included the introduction of the programme during the patient’s inpatient stay and the inclusion of relatives. This ensured that the invitation to participate following hospital discharge did not come as a surprise. The use of volunteering coordinators within the hospital setting (available across many UK hospital trusts) also helped support the statutory processes required for volunteer inclusion.
Figure 4.
Key themes generated from the process evaluation, barriers and facilitators for intervention implementation.
Barriers were also described. First, finding an appropriate venue to host the programme was a challenge across most (80%) sites. However, teams adapted to this by using teaching hubs in hospitals, and community venues. A fundamental issue related to sustainability was the need for ongoing funding, following the cessation of research income. Engaging early with the National health Service (NHS) management structure and working to achieve broad NHS aims within the delivery of the programme helped achieve sustainability. For example, focussing on the use of health and social care integration and the person-centred care approach, helped highlight the wider benefits of the programme. Of note, all five sites involved in this evaluation have received ongoing funding for the InS:PIRE programme within their local health boards.
Discussion
This multicentre study, evaluating a critical care recovery programme, has demonstrated a significant and clinically important difference in HRQoL for survivors of critical illness. To our knowledge, this is the first study internationally to report any enduring benefit from an outpatient intervention designed for ICU survivors.
Previous interventions targeting PICS have demonstrated isolated improvements, such as a reduction in post-traumatic stress disorder. However, benefits in overall HRQoL have remained elusive.31–33 Existing strategies have largely focused on specific interventions, often with a single healthcare professional group or small MDT.10 Few studies have targeted complex approaches combining the healthcare MDT with a recognition of the significant financial and social drivers of reduced HRQoL after critical illness. InS:PIRE differs from previous studies by bringing these concepts together. The MDT involved is not limited to the specific dimensions of PICS or new problems, but instead is directed by what is important to the patient.34 In this way interventions are targeted to both the problems having the greatest impact on each patient’s life and the solutions that patients are most motivated to pursue. Signposting to existing community organisations also allows patients to take an active role in improving their health, and with the guidance of the MDT, patients can improve their knowledge of the healthcare system and overall health literacy.
Peer support, a core component of the intervention, is important and valued by ICU survivors.35 Peer support in InS:PIRE differs from other programmes, with support embedded in the wider intervention, as opposed to stand-alone ‘self-help’ groups. This ensures peer support reaches patients who may not have access to stand-alone interventions. All peer support programmes are likely to benefit from the normalisation of the shared lived experience. Recent evidence also hypothesised that peer support could have an impact on anxiety.35 Interestingly, although anxiety was lower, there was not a significant difference in this symptom in the intervention cohort. More work is required to understand anxiety in survivors of critical illness, including optimal pathways for support.
The lasting benefits from InS:PIRE are likely to come from the improvement in patient self-determination alongside an enhancement of the skillset required to navigate the health and social care systems. The InS:PIRE programme’s longitudinal nature, offers patients the opportunity to incrementally increase independence and consolidate self-management skills. This is in direct contrast to previously tested interventions in this field which often offer a single appointment.32
The involvement of primary caregivers in the programme is also likely to have contributed to the lasting effects from InS:PIRE. Perhaps more significantly, there will have been direct effects on caregivers themselves which may benefit the entire family or care unit. Work which aims to assess the impact on caregivers and family members is ongoing.
Almost 60% of patients across the cohorts described pain at the time of assessment. InS:PIRE had a positive impact on some elements of pain (eg, interference with enjoyment of life); these improvements may be due to the targeted pharmacy management approaches which addressed pain management issues alongside the integrated physical rehabilitation services.36 37 However, InS:PIRE did not consistently reduce pain across all domains of BPI. Further research is required into the underlying mechanisms alongside any potential mediators of pain.
A goal of this study was to understand whether a complex intervention, known to be successful at a single site, could be scaled up to other sites and clinical teams. Our process evaluation identified key barriers and facilitators to the implementation of InS:PIRE. The findings of this evaluation are consistent with previous evidence demonstrating funding, staff provision and organisational buy-in are key to the successful implementation of post-ICU care.38 This evaluation has shown that complex interventions such as InS:PIRE are safe and feasible in the post-ICU discharge period and could be scaled up for future randomised controlled trials.
Limitations
The limitations of this study are notable. This study was not designed as a randomised controlled trial, therefore causality cannot be inferred. While there was substantial overlap in baseline characteristics between the intervention and usual care cohorts, as demonstrated by the propensity score matching analysis, patients were not randomly enrolled to either intervention or usual care. We have assessed the impact of this limitation by undertaking multiple sensitivity analyses.
This study was designed to assess the impact and effectiveness of the intervention as a whole. This has resulted in a unique study of a complex intervention for intensive care survivors but this approach lacks deeper understanding of the individual component effects. Further work is underway to gain a better understanding of the impacts of the individual programme components and their interactions.
The low return rates in the postal survey may have contributed to selection bias that was not fully addressed by the imputation, modelling or propensity score matching processes. Further, those who attended the InS:PIRE programme could have been more engaged with their health, thus we would have expected ‘better’ outcomes in this cohort. However, the spread across the socioeconomic gradient and the high proportion of patients with a history of alcohol excess and drug use who attended, would suggest that ‘difficult to access’ patients were included in the intervention. Additionally, like other studies in this field, over 30% of the intervention cohort were lost to follow-up, which may have influenced the results.32 Data were not available on why patients declined to attend InS:PIRE which limits the ability to address modifiable factors to improve the uptake in this group. We opted to use imputation to account for the low rate of missing data in this analysis.
The multiple imputation approach utilised, attempted to quantify uncontrolled confounding variables. Despite this, there may have been unmeasured confounding factors not accounted for in this analysis, which could have influenced the reported results. Moreover, although the propensity matched analysis replicated the findings of the primary analysis (covariate adjustment), due to the lower propensity matched cohort size, there is a possibility that some differences may have not been detected. There are alternative approaches such as inverse probability weighting regression and propensity score regression, however, each approach has similar individual limitations.
We have not included a cognition outcome measure. This study was conceptualised and approved before the publication of the core outcome measure set for acute respiratory failure research. Cognitive outcome measures were also not prioritised by our patient and family groups involved in the codesign of this study. Future work should address the impact, if any, of this intervention on cognition.22
Conclusion
This multicentre evaluation of a health and social care programme designed for survivors of critical illness, appears to show benefit for those that attend at 12 months following hospital discharge.
Acknowledgments
The authors thank the patients who took the time to participate in this study.
Footnotes
Twitter: @jomcpeake22
Contributors: JM and TQ conceived the idea for the paper. JM, PH and MS conducted the analysis. JM, PH and MS contributed to the interpretation of the findings. All authors critically revised the paper for intellectual content and approved the final version of the manuscript. JM, PH and MS take full responsibility for the content of this paper. PH is the guarantor of this work.
Funding: The project was funded by an award from the Health Foundation (173544) and a THIS.Institute (University of Cambridge) Fellowship (307748/PD-2019-02-16).
Competing interests: None declared.
Provenance and peer review: Not commissioned; externally peer reviewed.
Supplemental material: This content has been supplied by the author(s). It has not been vetted by BMJ Publishing Group Limited (BMJ) and may not have been peer-reviewed. Any opinions or recommendations discussed are solely those of the author(s) and are not endorsed by BMJ. BMJ disclaims all liability and responsibility arising from any reliance placed on the content. Where the content includes any translated material, BMJ does not warrant the accuracy and reliability of the translations (including but not limited to local regulations, clinical guidelines, terminology, drug names and drug dosages), and is not responsible for any error and/or omissions arising from translation and adaptation or otherwise.
Data availability statement
Data are available upon reasonable request.
Ethics statements
Patient consent for publication
Not applicable.
Ethics approval
This study involves human participants and was approved by Liverpool Central Research Ethics Committee (reference: 17/NM/0199).
References
- 1. Needham DM, Davidson J, Cohen H, et al. Improving long-term outcomes after discharge from intensive care unit: report from a stakeholders' conference. Crit Care Med 2012;40:502–9. 10.1097/CCM.0b013e318232da75 [DOI] [PubMed] [Google Scholar]
- 2. Mikkelsen ME, Still M, Anderson BJ, et al. Society of critical care medicine's international consensus conference on prediction and identification of long-term impairments after critical illness. Crit Care Med 2020;48:1670–9. 10.1097/CCM.0000000000004586 [DOI] [PubMed] [Google Scholar]
- 3. Pandharipande PP, Girard TD, Jackson JC. Delirium is an independent predictor of long-term cognitive impairment: results from the brain ICU (bringing to light the risk factors and incidence of neuropsychological dysfunction in ICU survivors) study. D16 randomized and observational studies in critical care. American thoracic Society, 2013: A5237–A. [Google Scholar]
- 4. Herridge MS, Tansey CM, Matté A, et al. Functional disability 5 years after acute respiratory distress syndrome. N Engl J Med 2011;364:1293–304. 10.1056/NEJMoa1011802 [DOI] [PubMed] [Google Scholar]
- 5. Hatch R, Young D, Barber V, et al. Anxiety, depression and post traumatic stress disorder after critical illness: a UK-wide prospective cohort study. Crit Care 2018;22:310. 10.1186/s13054-018-2223-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Lone NI, Seretny M, Wild SH, et al. Surviving intensive care: a systematic review of healthcare resource use after Hospital discharge*. Crit Care Med 2013;41:1832–43. 10.1097/CCM.0b013e31828a409c [DOI] [PubMed] [Google Scholar]
- 7. D'Cruz RF, Perrin F, Birring SS, et al. Provision of holistic care after severe COVID-19 pneumonia: anticipating clinical need and managing resources. Lancet Respir Med 2020;8:1175–6. 10.1016/S2213-2600(20)30529-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Sevin CM, Bloom SL, Jackson JC, et al. Comprehensive care of ICU survivors: development and implementation of an ICU recovery center. J Crit Care 2018;46:141–8. 10.1016/j.jcrc.2018.02.011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. McPeake J, Shaw M, Iwashyna TJ, et al. Intensive care syndrome: promoting Independence and Return to Employment (InS:PIRE). Early evaluation of a complex intervention. PLoS One 2017;12:e0188028. 10.1371/journal.pone.0188028 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Schofield‐Robinson OJ, Lewis SR, Smith AF. Follow‐up services for improving long‐term outcomes in intensive care unit (ICU) survivors. Cochrane Database Syst Rev 2018;11. 10.1002/14651858.CD012701.pub2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. McPeake J, Iwashyna TJ, Devine H, et al. Peer support to improve recovery following critical care discharge: a case-based discussion. Thorax 2017;72:856–8. 10.1136/thoraxjnl-2016-209661 [DOI] [PubMed] [Google Scholar]
- 12. Robinson C, Hibbert E, Bastin AJ, et al. An international study exploring the experience of survivors of critical illness as volunteers within ICU recovery services. Crit Care Explor 2020;2:e0273. 10.1097/CCE.0000000000000273 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. McPeake JM, Henderson P, Darroch G, et al. Social and economic problems of ICU survivors identified by a structured social welfare consultation. Crit Care 2019;23:153. 10.1186/s13054-019-2442-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Medicine FoIC . Guidelines for the provision of intensive care services version 2. FICM London, 2019. [Google Scholar]
- 15. Herdman M, Gudex C, Lloyd A, et al. Development and preliminary testing of the new five-level version of EQ-5D (EQ-5D-5L). Qual Life Res 2011;20:1727–36. 10.1007/s11136-011-9903-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Janssen MF, Pickard AS, Golicki D, et al. Measurement properties of the EQ-5D-5L compared to the EQ-5D-3L across eight patient groups: a multi-country study. Qual Life Res 2013;22:1717–27. 10.1007/s11136-012-0322-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Pickard AS, Neary MP, Cella D. Estimation of minimally important differences in EQ-5D utility and vas scores in cancer. Health Qual Life Outcomes 2007;5:70. 10.1186/1477-7525-5-70 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Vainiola T, Pettilä V, Roine RP, et al. Comparison of two utility instruments, the EQ-5D and the 15D, in the critical care setting. Intensive Care Med 2010;36:2090–3. 10.1007/s00134-010-1979-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Schwarzer R, Jerusalem M. Generalized self-efficacy scale. Measures in health psychology: A user’s portfolio Causal and control beliefs 1995;1:35–7 http://userpage.fu-berlin.de/~health/engscal.htm [Google Scholar]
- 20. Vincent E, Sewell L, Wagg K, et al. Measuring a change in self-efficacy following pulmonary rehabilitation: an evaluation of the praise tool. Chest 2011;140:1534–9. 10.1378/chest.10-2649 [DOI] [PubMed] [Google Scholar]
- 21. Zigmond AS, Snaith RP. The hospital anxiety and depression scale. Acta Psychiatr Scand 1983;67:361–70. 10.1111/j.1600-0447.1983.tb09716.x [DOI] [PubMed] [Google Scholar]
- 22. Needham DM, Sepulveda KA, Dinglas VD, et al. Core outcome measures for clinical research in acute respiratory failure survivors. An international modified Delphi consensus study. Am J Respir Crit Care Med 2017;196:1122–30. 10.1164/rccm.201702-0372OC [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Cleeland CS. The brief pain inventory user guide. Houston, TX: The University of Texas MD Anderson Cancer Center, 2009: 1–11. [Google Scholar]
- 24. Government S . Scottish index of multiple deprivation. Scottish government Edinburgh, 2016. [Google Scholar]
- 25. Sv B, Groothuis-Oudshoorn K. Mice: multivariate imputation by chained equations in R. J Stat Softw 2010:1–68. 10.18637/jss.v045.i03 [DOI] [Google Scholar]
- 26. R_Core_Team . R: a language and environment for statistical computing, 2019. Available: https://www.gbif.org/tool/81287/r-a-language-and-environment-for-statistical-computing
- 27. Haines KJ, Hibbert E, McPeake J, et al. Prediction models for physical, cognitive, and mental health impairments after critical illness: a systematic review and critical appraisal. Crit Care Med 2020;48:1871. 10.1097/CCM.0000000000004659 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Marra A, Pandharipande PP, Girard TD, et al. Co-Occurrence of post-intensive care syndrome problems among 406 survivors of critical illness. Crit Care Med 2018;46:1393. 10.1097/CCM.0000000000003218 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Battle CE, James K, Bromfield T, et al. Predictors of post-traumatic stress disorder following critical illness: a mixed methods study. J Intensive Care Soc 2017;18:289–93. 10.1177/1751143717713853 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Milton A, Schandl A, Soliman IW, et al. Development of an ICU discharge instrument predicting psychological morbidity: a multinational study. Intensive Care Med 2018;44:2038–47. 10.1007/s00134-018-5467-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Samuelson KAM, Corrigan I. A nurse-led intensive care after-care programme - development, experiences and preliminary evaluation. Nurs Crit Care 2009;14:254–63. 10.1111/j.1478-5153.2009.00336.x [DOI] [PubMed] [Google Scholar]
- 32. Cuthbertson BH, Rattray J, Campbell MK, et al. The practical study of nurse led, intensive care follow-up programmes for improving long term outcomes from critical illness: a pragmatic randomised controlled trial. BMJ 2009;339:b3723. 10.1136/bmj.b3723 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Walsh TS, Salisbury LG, Merriweather JL, et al. Increased hospital-based physical rehabilitation and information provision after intensive care unit discharge: the recover randomized clinical trial. JAMA Intern Med 2015;175:901–10. 10.1001/jamainternmed.2015.0822 [DOI] [PubMed] [Google Scholar]
- 34. McPeake JM, Harhay MO, Devine H, et al. Exploring patients' goals within the intensive care unit rehabilitation setting. Am J Crit Care 2019;28:393–400. 10.4037/ajcc2019436 [DOI] [PubMed] [Google Scholar]
- 35. McPeake J, Hirshberg EL, Christie LM, et al. Models of peer support to remediate Post-Intensive care syndrome: a report developed by the Society of critical care medicine thrive international peer support collaborative. Crit Care Med 2019;47:e21–7. 10.1097/CCM.0000000000003497 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Devine H, MacTavish P, Quasim T. Musculoskeletal problems in intensive care unit patients post discharge. Critical Care 2016;20:165. 10.1186/s13054-016-1208-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. MacTavish P, Quasim T, Purdie C, et al. Medication-Related problems in intensive care unit survivors: learning from a multicenter program. Ann Am Thorac Soc 2020;17:1326–9. 10.1513/AnnalsATS.202005-444RL [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Haines KJ, McPeake J, Hibbert E, et al. Enablers and barriers to implementing ICU follow-up clinics and peer support groups following critical illness: the thrive collaboratives. Crit Care Med 2019;47:1194. 10.1097/CCM.0000000000003818 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
thoraxjnl-2021-218428supp001.pdf (758.2KB, pdf)
Data Availability Statement
Data are available upon reasonable request.