Abstract
Objective:
Describe a severe acute respiratory coronavirus virus 2 (SARS-CoV-2) hospital outbreak and the role of serial testing of patients and healthcare personnel (HCP) in interrupting SARS-CoV-2 transmission.
Design:
Outbreak investigation.
Setting:
Medical floor of a tertiary-care center in Minnesota.
Methods:
Serial testing for SARS-CoV-2 and whole-genome sequencing (WGS) of positive specimens from HCP and patients were used. An outbreak-associated case was defined as a positive SARS-CoV-2 molecular test in an HCP who worked on the floor prior to testing positive or in a patient who was hospitalized on the medical floor bewteen October 27 and December 1, 2020. WGS was used to determine potential routes of transmission.
Results:
The outbreak was detected after a patient hospitalized for 12 days tested positive for SARS-CoV-2. Serial testing of patients and HCP was conducted in response. Overall, 247 HCP and 41 patients participated in serial SARS-CoV-2 testing; 52 HCP (21%) and 19 hospitalized patients (46%) tested positive. One additional HCP tested positive outside serial testing. The WGS of specimens from 27 (51%) HCP and 15 (79%) patients identified 3 distinct transmission clusters. WGS and epidemiologic evidence suggested intrafacility transmission. The proportions of asymptomatic and presymptomatic patients who tested positive (63%) and HCP who worked during their infectious period (75%) highlight the need for serial testing of asymptomatic patients and HCP during outbreaks.
Conclusions:
Coupled with preventive measures such as personal protective equipment use and physical distancing, serial testing of HCP and patients could help detect and prevent transmission within healthcare facilities during outbreaks and when nosocomial transmission is suspected.
Since the start of the coronavirus disease 2019 (COVID-19) pandemic, infection with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) has occurred among patients and healthcare personnel (HCP). SARS-CoV-2 outbreaks have been reported in skilled nursing facilities 1,2 and hospitals. 3–6 To prevent infection of vulnerable individuals, the Center for Medicare and Medicaid Services (CMS) has required federally certified nursing homes to conduct daily symptom screening and routine screening tests for staff based on county test-positivity rates. 7,8 Testing a group of people at a single point in time is called a point-prevalence survey. 9 The CMS requires nursing homes that detect even a single SARS-CoV-2 infection in residents or HCP to conduct repeated point-prevalence surveys, sometimes called serial testing, to define the extent of SARS-CoV-2 transmission and to inform efforts to prevent ongoing spread among residents and HCP. 1 The Centers for Disease Control and Prevention (CDC) recommendations have guided implementation of these interventions. 10 Some states have implemented similar requirements for state-regulated assisted living facilities. 11 In the hospital setting, screening of HCP for signs and symptoms of COVID-19 before the work shift has been conducted throughout the pandemic as required by CMS, 12 but serial SARS-CoV-2 testing of asymptomatic HCP has not been widely implemented, in part because of limitations in personnel resources and laboratory capacity. Here, we describe management of a SARS-CoV-2 outbreak on a hospital medical floor (1) to demonstrate the role of serial testing during outbreaks and when nosocomial transmission is suspected and (2) to describe how whole-genome sequencing (WGS) of SARS-CoV-2 specimens can support epidemiologic data to understand transmission dynamics.
Index case
On October 16, 2020, an 81-year-old patient was admitted to a 50-bed medical floor, primarily serving cardiology patients, for exacerbation of congestive heart failure. The patient had a negative molecular COVID-19 test performed at a different facility on this day. On October 21, the patient tested negative for SARS-CoV-2 (Cepheid GeneXpert Xpress, Sunnyvale, CA) prior to a cardiac procedure. On October 27, the patient was tested for SARS-CoV-2 prior to discharge, and this specimen yielded a positive result on October 30. SARS-CoV-2 IgG antibody testing (Abbott, Lake Forest, IL) conducted on October 30 was negative and a repeated SARS-CoV-2 in-house molecular test (Cepheid GeneXpert Xpress, Sunnyvale, CA) was positive. This patient was suspected to have a hospital-acquired case of COVID-19 given the prior 2 negative tests on day 1 and day 6 of hospitalization and that the positive test occurred on day 12.
Hospital procedures in place prior to the detection of this case included testing of asymptomatic patients (1) upon admission from a congregate living setting; (2) prior to an aerosol-generating procedure, cardiology procedure, or surgery, when pregnant; or (3) prior to placement into a skilled nursing facility. A standard process for contact tracing and risk assessment of HCP SARS-CoV-2 exposures was in place to identify occupational and nonoccupational high-risk exposures for postexposure quarantine. A high-risk exposure was defined as close prolonged contact (within 2 m or 6 feet for at least 15 minutes within a 24-hour period) without appropriate personal protective equipment (PPE) with a person with confirmed COVID-19 based on the latest CDC interim guidance for management of healthcare personnel with SARS-CoV-2 infection or exposure to SARS-CoV-2 at that time. 13 When conducting aerosol-generating procedures on a COVID-19–positive patient, appropriate PPE included a respirator, eye protection, gown, and gloves. For all other interactions, a level-1 ASTM medical-grade mask and eye protection were considered appropriate PPE. 13 HCP with high-risk exposures were required to quarantine from work for 14 days from the date of exposure and were tested 5–7 days after exposure. HCP positive for SARS-CoV-2 were required to isolate for at least 10 days. Requirements for HCP use of eye protection (face shields or safety glasses) during all direct patient-care interactions, universal HCP masking, and patient masking while outside their rooms had been in place since April 2020. Patient mask use was not enforced in patient rooms. On October 30, when the index case was identified, 1 HCP working on this medical floor was isolating at home due to COVID-19.
Methods
Outbreak response
To prevent further spread among HCP and patients, the hospital initiated voluntary serial testing on the affected medical floor on November 1, 2020. Weekly SARS-CoV-2 reverse transcription polymerase chain reaction (RT-PCR) testing of HCP anterior nasal swab specimens (Luminex Aries SARS-CoV-2 assay, Luminex Corporation, Austin, TX, or Cepheid GeneXpert SARS-CoV-2) was conducted through December 1, 2020. Minnesota Department of Health (MDH) recommends conducting testing every 3–7 days. 9 At the time of the outbreak, limited test availability, long turnaround times for results, and limited staff resources to conduct testing constrained the ability to test at a shorter interval. Patients were tested if they had any contact with a roommate or HCP who tested positive for SARS-CoV-2 or if they developed signs or symptoms consistent with COVID-19. HCP who worked on the floor or had cared for a COVID-19–positive patient within the previous 10 days were offered testing. HCP were identified through electronic medical record review and through HCP and manager interviews. All symptomatic HCP were excluded from work and were tested. An outbreak-associated case was defined as a positive SARS-CoV-2 RT-PCR test in an HCP who worked on that medical floor, or patients who were hospitalized there between October 27 and December 1. HCP symptom status and onset date were reported retrospectively.
Respiratory specimens positive for SARS-CoV-2 were submitted to the Minnesota Department of Health (MDH) Public Health Laboratory for WGS, which was conducted by using previously described methods. 14 Phylogenetic relationships, including distinct clustering of viral whole-genome sequences, 15 were inferred based on nucleotide differences via IQ-TREE (http://www.iqtree.org/) using general time reversible substitution models 16 as a part of the Nextstrain (https://nextstrain.org/) workflow. 17 This activity was reviewed by the CDC and was conducted consistent with applicable federal law and CDC policy (45 CFR part 46.102(l)(2), 21 CFR part 56; 42 USC sect. 241(d); 5 USC sect. 552a; 44 USC sect. 3501 et seq).
COVID-19 infection prevention and control practices
On November 10, 2020, the hospital implemented universal patient testing upon admission. Most rooms on the medical floor were double occupancy. On November 19, all hospitalized patients were asked to mask in the presence of HCP. In early November, HCP on the hospital floor were given the option to wear N95 respirators for routine care of patients not known to have COVID-19. On November 17, visitation restriction was reinstituted after being rescinded in July 2020. On December 2, the hospital implemented universal eye protection at all times (not just during direct patient care) in response to HCP absence associated with COVID-19 isolation and quarantine due to increasing transmission.
Results
In total, 247 (68%) of 361 HCP working on the outbreak medical floor were tested in association with this outbreak, including 13 HCP with documented high-risk exposures to patients. Overall, 338 specimens from 247 HCP, and 73 specimens from 41 patients were tested. We identified 72 outbreak-associated cases of SARS-CoV-2 infection: 53 cases in HCP, including 1 HCP tested outside serial testing, and 19 cases in patients (Table 1).
Table 1.
Demographic and Clinical Characteristics of Confirmed COVID-19 Cases Associated with an Outbreak on a Medical Unit—St. Louis County, Minnesota, October–November 2020
| Characteristic | HCP (n=53) |
Patients (n=19) |
||
|---|---|---|---|---|
| No. | % | No. | % | |
| Sex | ||||
| Male | 11 | 21 | 13 | 68 |
| Female | 42 | 79 | 6 | 32 |
| Age, median y (range) | 29 (20–63) | 76 (50–87) | ||
| Symptom status at time of testinga | ||||
| Symptomatic | 47 | 89 | 7 | 37 |
| Presymptomatic b | 1 | 2 | 8 | 42 |
| Asymptomatic | 5 | 9 | 4 | 21 |
| Worked during infectious period c | 40 | 75 | … | … |
| Days hospitalized before positive test, median (range) | 8 (1–30) | |||
Note. HCP, healthcare personnel.
Assessed retrospectively.
Presymptomatic persons did not have symptoms at the time of testing but later developed symptoms consistent with COVID-19.
The infectious period was considered to be 2 days before through 10 days after symptom onset date, or specimen collection date for persons with asymptomatic cases 14 .
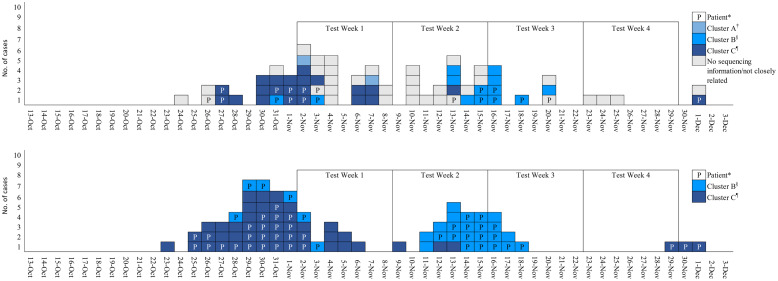
Of 19 patients, 7 (37%) had signs and/or symptoms of COVID-19 and 8 patients (42%) were presymptomatic at the time of testing; 4 patients died. Patients tested positive a median of 8 days after hospitalization (range, 1–30 days). Of 53 HCP who tested positive, 47 (89%) retrospectively reported having symptoms of COVID-19 on the test date, and 1 HCP later developed symptoms although specific symptom information was not collected. Moreover, 40 HCP (75%) worked during their infectious period (2 days before through 10 days after symptom onset or specimen collection for asymptomatic cases). 18 None of the 53 SARS-CoV-2–positive HCP had a documented high-risk exposure at work. Of the 13 HCP with high-risk exposures, none tested positive. The epidemic curve depicting specimen collection date is shown in Figure 1 along with the number of infectious days for patients and days worked during their infectious period for HCP for 2 clusters closely related by WGS. The bottom part of the figure shows that after serial testing was initiated during week 1, the number of infectious persons on the medical floor decreased. Among 312 HCP working on the medical floor who did not previously test positive, HCP participation decreased during the study period, even as the number eligible per week decreased: week 1, 168 (48%) of 353; week 2, 75 (23%) of 331; week 3, 74 (23%) of 316; and week 4, 21(7%).
Fig. 1.
Specimen collection date for COVID-19 patients* (top) and infectious days for patients and infectious days worked for healthcare personnel (HCP) (bottom), St Louis County, Minnesota, October–December 2020.
*Patients are indicated by a P; HCP are indicated by empty boxes.
†Cluster A represents 2 employees who both had symptoms of COVID-19.
§ Cluster B represents 7 patients and 5 employees.
¶Cluster C represents 8 patients and 14 employees. Onset date for 1 patient is unknown.
Whole-genome sequencing
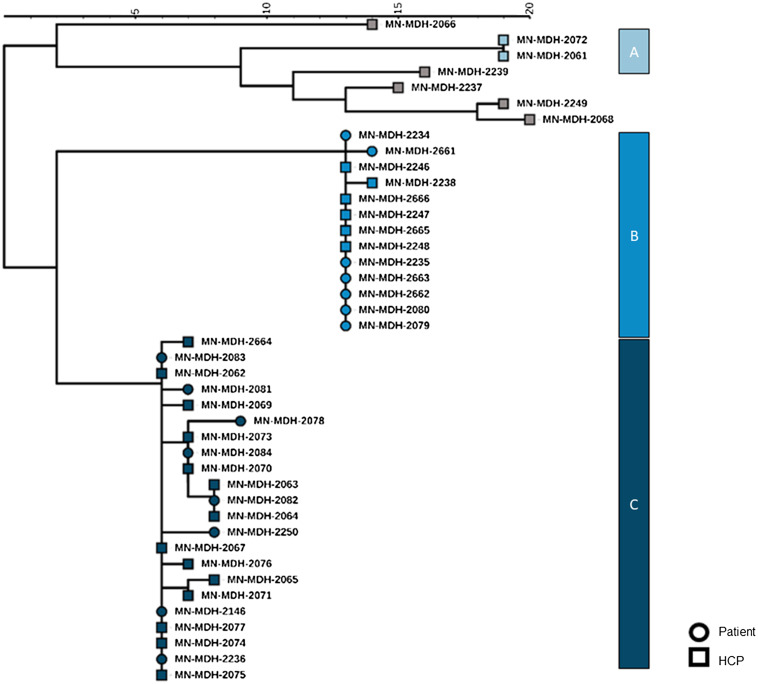
SARS-CoV-2 WGS was completed on specimens from 15 patients and 27 HCP (58% of total 72 positive specimens). WGS results showed 3 distinct clusters, suggesting at least 3 points of virus introduction into the facility (Fig. 2). Cluster A included 2 HCP who tested positive 5 days apart. One HCP was asymptomatic, and there was no known epidemiologic link between these 2 persons except that they worked on the same floor. Cluster B included 7 patients and 6 HCP who provided care to at least 1 of those patients. Cluster C included 8 patients, including the index patient, and 14 HCP. All patients received care from at least 1 HCP in the cluster, and 2 were roommates (the index patient who tested positive while sharing a room with another patient who tested positive 5 days later). For 2 HCP (MN-MDH-2071 and MN-MDH-2065), who lived together and worked on the floor, the specimens showed genomic similarity, but epidemiologic evidence suggested that only 1 infection was acquired at work. Together, epidemiologic data and WGS results suggest that there were multiple virus introductions into the facility as well as within-facility transmission.
Fig. 2.
Phylogenetic tree showing genetic distance between available SARS-CoV-2 specimens collected from healthcare personnel (HCP) and patients, St Louis County, Minnesota, October–December 2020.
A, B, and C refer to the unique clusters identified in the outbreak.
Discussion
Overall, 72 cases were identified during this COVID-19 hospital outbreak, and epidemiologic and molecular evidence suggest that within-facility transmission occurred. Also, 12 SARS-CoV-2–positive patients (63%) were either asymptomatic or presymptomatic at the time of testing. Because the unit where the outbreak occurred primarily served cardiology patients, some symptoms were presumed to be cardiac related and might not have been initially attributed to COVID-19. Some SARS-CoV-2–positive HCP never developed symptoms. Anecdotally, some retrospectively reported that they had symptoms not initially attributed to COVID-19. Throughout the pandemic, mild or barely perceptible symptoms consistent with COVID-19, especially those that overlap with symptoms of seasonal allergies, have challenged HCP and the infection prevention and occupational health teams tasked with COVID-19 symptom screening prior to work shifts.
The findings from this study demonstrate the potential for SARS-CoV-2 to spread within a healthcare facility, despite the use of symptom screening and testing protocols for symptomatic HCP. Repeated testing of patients after admission (eg, on day 3) could help identify patients admitted during the incubation period of SARS-CoV-2 infection. Serial testing of HCP and residents has become a critical part of the SARS-CoV-2 detection and prevention strategy in nursing homes and assisted living facilities 1,19,20 and it has been recommended for other settings, such as psychiatric facilities, where the inability to adhere to preventive measures like masking and physical distancing increases the risk for intrafacility transmission. 21 Early identification of presymptomatic or asymptomatic individuals facilitates the implementation of isolation and quarantine to prevent further transmission. 1,22
In addition to symptom-based testing, the approach to HCP testing in acute care has included testing those with known high-risk exposure. This approach, too, has limitations. No HCP who tested positive during this investigation had an identified high-risk exposure at work. Although transmission can and does occur in healthcare settings, our previous work has shown that HCP are more likely to test positive after household and community exposures. 23 These factors suggest that testing limited to symptomatic HCP, and those with known occupational high-risk exposures might miss asymptomatic, presymptomatic, and mild cases of SARS-CoV-2 infection. In this outbreak, serial testing of all HCP allowed detection of asymptomatic and presymptomatic individuals, regardless of documented exposure risk in or outside the workplace.
Emergence of the SARS-CoV-2 virus variants further underscores the need for rapid identification, isolation, and quarantine of positive and exposed individuals, given that several variants are more transmissible than earlier variants, and people who have been fully vaccinated can be infected and spread the virus to others. 24,25 In Minnesota, local (eg, county) SARS-CoV-2 infection rates influence the number of potential introductions of the SARS-CoV-2 virus into healthcare facilities (unpublished data). One publication has reported a low risk for hospital-acquired SARS-CoV-2 infection among patients, with infection control measures in place that were similar to those used at the hospital in this investigation. 26 However, in that study, routine HCP testing was not conducted and neither community infection rates nor background rates of HCP absence (ie, for isolation or quarantine) were reported. All individuals within, and moving in and out of, a healthcare facility contribute to an ecosystem that supports or hinders SARS-CoV-2 transmission.
The MDH has encouraged the use of serial testing by acute-care and behavioral health facilities, including conducting repeated point-prevalence surveys of all patients and staff in some situations. 9 Point-prevalence survey testing is especially important in situations in which (1) nosocomial transmission is suspected, (2) patients cannot be accommodated in individual rooms, (3) compliance with physical distancing and wearing masks is low, or (4) there has been broad exposure to presymptomatic or asymptomatic staff. 9 In this testing approach, all patients and HCP should be tested every 3–7 days until 14 days have passed since the last positive individual was detected. The interval between repeated testing rounds might be longer or shorter, depending on testing capacity, the ability to divert staff to help with testing, and the ability to control factors that contribute to transmission. The use of a shorter interval (eg, 3 days) early in the testing cycle (eg, in the first 2 weeks) will facilitate identification and isolation of cases more quickly. Given the rapid spread of the o (omicron) variant, the maximum interval between testing rounds might be adjusted (eg, to 3 days) to mitigate SARS-CoV-2 transmission between rounds.
One of the strengths of the investigation was that the proportion (58%) of specimens that underwent WGS was at the higher range than those reported in other published studies (range, 13%– 65%). 6,27 When this outbreak occurred, widespread sequencing surveillance was not being conducted. WGS helps corroborate transmission routes suspected during epidemiologic investigation.
In the hierarchy of controls for controlling exposure to occupational hazards, including SARS-CoV-2, elimination of the hazard, engineering controls to isolate people from the hazard, and administrative controls to change how people work are favored over sole reliance on PPE. 28 Previous investigations have demonstrated transmission within healthcare facilities among HCP, patient to HCP, and HCP to patient, despite the recommended use of PPE supporting the need for higher levels of control. 5,29 Prompt identification of infectious patients and HCP through admission screening and serial testing (examples of administrative controls) can inform changes in PPE use given the situation, including use of N95 respirators for all patient care during facility-based outbreaks to allow for more effective PPE use. 30 As a part of these efforts, however, auditing of PPE compliance is essential. A study of a COVID-19 hospital outbreak in Ireland demonstrated the importance of universal masking throughout the work shift, with WGS-documented spread of SARS-CoV-2 between HCP and patients and among HCP before and after implementation of universal masking requirement, 27 suggesting that additional controls are necessary. This facility had a universal masking requirement for HCP, with exceptions only while eating and on break with >2 m (6 feet) distance from others. The CDC recommendations for HCP PPE use remain unchanged after COVID-19 vaccination. 30
This study had several limitations that might have led to an underestimation of the number of outbreak-associated cases or contributed to unrecognized transmission pathways. First, patients diagnosed shortly after discharge from this unit were not evaluated for nosocomial acquisition. Second, additional chains of transmission with asymptomatic patients, visitors, and HCP might have been missed. Third, HCP participation in voluntary SARS-CoV-2 testing decreased as the investigation progressed. This finding is not unique to this medical facility and suggests that mandatory testing might be needed to get some outbreaks under control. Fourth, HCP could have had unrecognized high-risk exposures during work shifts or outside the hospital that contributed to transmission but cannot be described here. Fifth, HCP might have overstated their PPE adherence during exposure risk assessment leading to misclassification of exposures as low risk. Suboptimal compliance with universal masking policies in tertiary care has been reported previously. 31 There was no formal audit process for PPE compliance, which limited the ability to understand the role of PPE in this outbreak. Lastly, some HCP may not have been identified for testing but spent time on the outbreak medical floor. Notably, this outbreak occurred prior to the authorization of COVID-19 vaccinations.
Increased prevalence of SARS-CoV-2 infection in the community is associated with elevated risk of healthcare-related infections. 2,32 SARS-CoV-2 breakthrough infections can occur in vaccinated HCP and patients. Thus, it remains important to implement protocols that allow for detection of infectious individuals without relying on the presence of symptoms or contact tracing alone. Coupled with preventive measures such as PPE use, serial testing could help detect and prevent transmission within healthcare facilities in outbreak situations or with suspected nosocomial transmission.
Acknowledgments
We thank Ruth Lynfield, MD, Kristen Ehresmann, MPH, Kathryn Como-Sabetti, MPH, and Ashley Fell, MPH for their thoughtful review of the manuscript.
The findings and conclusions in this report are those of the authors and do not necessarily represent the official position of the Centers for Disease Control and Prevention.
Financial support
No financial support was provided relevant to this article.
Conflicts of interest
All authors report no conflicts of interest relevant to this article.
References
- 1. Taylor J, Carter RJ, Lehnertz N, et al. Serial testing for SARS-CoV-2 and virus whole-genome sequencing inform infection risk at two skilled nursing facilities with COVID-19 outbreaks, Minnesota, April–June 2020. Morb Mortal Wkly Rep 2020;69:1288–1295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Cavanaugh AM, Fortier S, Lewis P, et al. COVID-19 Outbreak associated with a SARS-CoV-2 R.1 lineage variant in a skilled nursing facility after vaccination program—Kentucky, March 2021. MMWR Weekly 2021;70:639–643. [DOI] [PMC free article] [PubMed]
- 3. Goldberg L, Levinsky Y, Marcus N, et al. SARS-CoV-2 infection among healthcare workers despite the use of surgical masks and physical distancing—the role of airborne transmission Open Forum Infect Dis 2021;8(3):ofab036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Heinzerling A, Stuckey MJ, Scheuer T, et al. Transmission of COVID-19 to healthcare personnel during exposures to a hospitalized patient—Solano County, California, February 2020. MMWR Weekly 2020;69:472–476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. McGloin JM, Asokaraj N, Feeser B, et al. Coronavirus disease 2019 (COVID-19) outbreak on an inpatient psychiatry unit: mitigation and prevention. Infect Control Hosp Epidemiol 2021. doi: 10.1017/ice.2021.233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Meredith LW, Hamilton WL, Warne B, et al. Rapid implementation of SARS-CoV-2 sequencing to investigate cases of healthcare-associated COVID-19: a prospective genomic surveillance study. Lancet Infect Dis 2020;20:1263–1272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Additional policy and regulatory revisions in response to COVID-19 public health emergency related to long-term care (LTC) facility testing requirements and revised COVID-19 focused survey tool. Centers for Medicare & Medicaid Services website. https://www.cms.gov/medicareprovider-enrollment-and-certificationsurveycertificationgeninfopolicy-and-memos-states-and/interim-final-rule-ifc-cms-3401-ifc-additional-policy-and-regulatory-revisions-response-covid-19. Published 2020. Accessed February 25, 2022.
- 8. COVID-19 testing recommendations for long-term care facilities. Minnesota Department of Health website. https://www.health.state.mn.us/diseases/coronavirus/hcp/ltctestrec.pdf. Published 2021. Accessed June 21, 2021.
- 9. Point-prevalence survey testing in acute care facilities. Minnesota Department of Health website. https://www.health.state.mn.us/diseases/coronavirus/hcp/ppsacute.pdf. Published 2020. Accessed August 19, 2021.
- 10. Testing guidelines for nursing homes. Centers for Disease Control and Prevention website. https://www.cdc.gov/coronavirus/2019-ncov/hcp/nursing-homes-testing.html. Published 2021. Accessed June 21, 2021.
- 11. Long-term care testing: COVID-19. Minnesota Department of Health website. https://www.health.state.mn.us/diseases/coronavirus/hcp/ltctesting.html. Published 2021. Accessed September 15, 2021.
- 12. COVID-19 long-term care facility guidance. Centers for Medicare & Medicaid Services website. https://www.cms.gov/files/document/4220-covid-19-long-term-care-facility-guidance.pdf. Published 2020. Accessed October 1, 2021.
- 13. Interim US guidance for risk assessment and work restrictions for healthcare personnel with potential exposure to SARS-CoV-2. Centers for Disease Control and Prevention website. https://www.cdc.gov/coronavirus/2019-ncov/hcp/guidance-risk-assesment-hcp.html. Published 2021. Accessed June 21, 2021.
- 14. SARS-CoV-2 sequencing protocols. Artic Network website. https://artic.network/ncov-2019. Published 2020. Accessed February 25, 2022.
- 15. Huddleston J, Hadfield J, Sibley TR, et al. Augur: a bioinformatics toolkit for phylogenetic analyses of human pathogens. J Open Source Softw 2021;6:2906. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Nguyen L, Schmidt HA, von Haeseler A, Minh BQ. IQ-TREE: a fast and effective stochastic algorithm for estimating maximum-likelihood phylogenies. Mol Biol Evol 2015;32:268–274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Hadfield J, Megill C, Bell SM, et al. Nextstrain: real-time tracking of pathogen evolution. Bioinformatics 2018;34:4121–4123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Investigating a COVID-19 case. Centers for Disease Control and Prevention website. https://www.cdc.gov/coronavirus/2019-ncov/php/contact-tracing/contact-tracing-plan/investigating-covid-19-case.html. Published 2020. Accessed October 1, 2021.
- 19. Arons MM, Hatfield KM, Reddy SC, et al. Presymptomatic SARS-CoV-2 infections and transmission in a skilled nursing facility. N Engl J Med 2020;382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Blain H, Rolland Y, Tuaillon E, et al. Efficacy of a test-retest strategy in residents and health care personnel of a nursing home facing a COVID-19 outbreak. J Am Med Dir Assoc 2020;21:933–936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Callaghan AW, Chard AN, Arnold P, et al. Screening for SARS-CoV-2 infection within a psychiatric hospital and considerations for limiting transmission within residential psychiatric facilities—Wyoming, 2020. MMWR Weekly 2020;69:825–829. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Wake RM, Morgan M, Choi J, Winn S. Reducing nosocomial transmission of COVID-19: implementation of a COVID-19 triage system. Clin Med 2020;20:e141–e145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Fell A, Beaudoin A, D’Heilly P, et al. SARS-CoV-2 exposure and infection among health care personnel—Minnesota, March 6–July 11, 2020. MMWR Weekly 2020;69:1605–1610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Delta variant: what we know about the science. Centers for Disease Control and Prevention website. https://www.cdc.gov/coronavirus/2019-ncov/variants/delta-variant.html. Published 2021. Accessed August 17, 2021.
- 25. CDC COVID-19 Response Team. SARS-CoV-2 B.1.1.529 (omicron) variant—United States, December 1–8, 2021. MMWR Weekly 2021;70:1731–1734. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Rhee C, Baker M, Vaidya V, et al. Incidence of nosocomial COVID-19 in patients hospitalized at a large US academic medical center. JAMA Netw Open 2020;3(9):e2020498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Lucey M, Macori G, Mullane N, et al. Whole-genome sequencing to track severe acute respiratory syndrome coronavirus 2 (SARS-COV-2) transmission in nosocomial outbreaks. Clin Infect Dis 2021;72:e727–e735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. The National Institute for Occupational Safety and Health (NIOSH). Hierarchy of controls. Centers for Disease Control and Prevention website. https://www.cdc.gov/niosh/topics/hierarchy/default.html. Published 2015. Accessed June 21, 2021.
- 29. Chan ER, Jones LD, Redmond SN, et al. Use of whole-genome sequencing to investigate a cluster of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infections in emergency department personnel. Infect Control Hosp Epidemiol 2021. doi: 10.1017/ice.2021.208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Interim infection prevention and control recommendations for healthcare personnel during the coronavirus disease 2019 (COVID-19) pandemic. Centers for Disease Control and Prevention website. https://www.cdc.gov/coronavirus/2019-ncov/hcp/infection-control-recommendations.html. Published 2021. Accessed February 25, 2022.
- 31. Datta R, Glenn K, Pellegrino A, et al. Increasing face-mask compliance among healthcare personnel during the coronavirus disease 2019 (COVID-19) pandemic. Infect Control Hosp Epidemiol 2021. doi: 10.1017/ice.2021.205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Arnold FW, Bishop S, Oppy L, Scott L, Stevenson G. Surveillance testing reveals a significant proportion of hospitalized patients with SARS-CoV-2 are asymptomatic. Am J Infect Control 2021;49:281–285. [DOI] [PMC free article] [PubMed] [Google Scholar]