Abstract
Background:
Alcohol use disorder is associated with difficulties in emotion regulation and cognitive reappraisal. Family history of harmful alcohol use increases risk of substance use disorders, but no studies have examined whether family history is associated with altered neural activation during cognitive reappraisal relative to passive viewing of negative images in a sample of young adults without current substance use disorders.
Methods:
Participants (N=75 with positive [n=31] or negative [n=44] family histories of harmful alcohol use) completed the emotion regulation task during an MRI scan, and the Emotion Regulation Questionnaire to assess use of emotion regulation and suppression strategies. Whole-brain analyses and amygdala region of interest analyses using linear mixed-effects models assessed family history group and cue effects on neural activation during the task.
Results:
The groups did not differ on trait reappraisal, suppression, or negative emotion following reappraisal. In general, group effects in whole-brain and amygdala activation during the cognitive reappraisal contrast indicated small effect sizes (2.2% of voxels had d>.2) that were not significantly different. Participants with positive family histories engaged the right middle and superior frontal gyri to a greater extent than participants with negative family histories during the decrease-negative cue (t=4.14, p=.001).
Conclusions:
For at-risk young adults without current harmful substance use, family history of harmful alcohol use does not appear to be associated with disrupted emotion regulation when instructed to apply cognitive reappraisal. Reappraisal may be a feasible therapeutic target for those who develop a substance use disorder with associated emotion dysregulation.
Keywords: fMRI, emotion regulation, family history of harmful alcohol use, substance use risk
1. Introduction
Developing an alcohol use disorder is approximately 50% heritable (Deak and Johnson, 2021), thus having genetically related family members with a history of harmful alcohol use is a risk factor for developing a substance use disorder. It remains unclear how this genetic predisposition manifests in intermediate behavioral and biological risk factors in late adolescence and young adulthood. One possibility is that genes associated with harmful alcohol use affect development of related behaviors and their underlying neural representations. For example, individuals with substance use disorders (Swendsen et al., 2002) and those with family histories of substance use disorders (Shoal and Giancola, 2001) show greater negative emotionality than those without family histories of substance use disorders, suggesting predisposition to altered emotional reactivity to negative or difficult situations. Coping with difficult emotions is important for psychosocial health (D’Agostino et al., 2017; McRae and Gross, 2020). One strategy commonly used to cope with difficult emotions is cognitive reappraisal, which involves changing thoughts about a stimulus to change feelings (Gross, 2002). Evidence suggests that adults with alcohol use disorder have greater difficulties in emotion regulation (Stellern et al., 2022; Weiss et al., 2022) and employ less reappraisal (Berking et al., 2011) than those without harmful alcohol use. Thus, emotion regulation may be an intermediate process linking genetic risk to development of alcohol use disorder.
Differences in emotion processing and cognitive reappraisal may be apparent in engagement of related neural systems as well. Neuroimaging studies examining family history of alcohol use in first-degree relatives without current harmful substance use patterns show these individuals have less gray matter in cognitive control and reward-related brain regions (Filippi et al., 2019; Henderson et al., 2018) than those without positive family history. Additionally, studies have examined family history of harmful alcohol use and responses to emotional faces: one study has shown increased amygdala activation (Lindsay et al., 2014) and another has shown decreased amygdala activation (Glahn et al., 2007). It seems that amygdala activation is altered during emotional responding and processing, but the directionality and implications remain unclear. These differences in brain structure and function might affect cognitive function related to the management of emotions. Generally, studies examining neural activation during cognitive reappraisal of negative stimuli consistently show recruitment of cognitive control regions across the prefrontal cortex and decreased amygdala activation (Buhle et al., 2014; Morawetz et al., 2017). Individuals with family history of harmful alcohol use demonstrate decreased neural activation in dorsolateral prefrontal cortex during a cognitive control task that involved viewing emotional faces, but participants did not engage specifically in cognitive reappraisal (Cservenka et al., 2014). To our knowledge, no studies have examined whether neural processing of cognitive reappraisal differs by family history of harmful alcohol use in young adults prior to development of an alcohol use disorder.
It may be that young adults with genetic risk, but without current harmful substance use patterns, are less likely to engage in cognitive reappraisal strategies and more likely to exhibit altered neural activation in cognitive control and emotional processing regions of the brain when faced with negative emotional stimuli. Therefore, the present study examined whether family history positive young adults differ from family history negative young adults on neural correlates of cognitive reappraisal and emotional reactivity using an emotion regulation task with negative and neutral imagery (Gianaros et al., 2014; Minkel et al., 2012). We hypothesized that family history positive relative to family history negative participants will: 1) endorse less frequent use of cognitive reappraisal, greater use of emotional suppression, and have higher subjective ratings of negative affect; 2) exhibit less neural activation in cognitive control regions (e.g., prefrontal cortex) during reappraisal and emotional reactivity; and 3) exhibit greater neural activation in the amygdala during cognitive reappraisal and emotional reactivity.
2. Material and Methods
2.1. Procedure
We recruited using advertisements for a study assessing neural factors associated with alcohol use disorder, including whether a parent or sibling had experienced negative consequences due to alcohol use. The study was approved by the Colorado Multiple Institutional Review Board and conducted at the Brain Imaging Center, University of Colorado Anschutz Medical Campus. Participants completed informed consent, MRI safety screeners, alcohol breathalyzer, saliva drug screen, and urine pregnancy tests (females). Data were collected between March 2019 and June 2021. Survey data were collected using REDCap (Harris et al., 2009).
Participants were eligible if they 1) were between 18 and 22 (one participant turned 23 before their laboratory visit), 2) had ever consumed alcohol, 3) were MRI eligible, and 4) scored ≤7 on the Alcohol Use Disorder Identification Test (Saunders et al., 1993). Exclusion criteria were: 1) being treated for a psychiatric disorder, 2) seeking treatment for an alcohol use disorder, 3) regular tobacco use (>20 cigarettes per week), 4) using medications that affect the hemodynamic response, 5) history of head trauma with loss of consciousness >10 minutes, 6) evidence of cannabis misuse (>8 uses of cannabis per month, or >11 on the Cannabis Use Disorder Identification Test-Revised; Adamson et al., 2010), 7) >10 lifetime uses of illicit substances, 8) misused prescription medication in the past year, or 9) pregnancy.
2.2. Measures
2.2.1. Emotion Regulation Task.
Participants completed an emotion regulation task (Gianaros et al., 2014; Minkel et al., 2012) during the MRI scan, assessing both emotional reactivity via passive viewing of images, and regulation capacity via cognitive reappraisal. Participants were shown neutral (e.g., an umbrella) and negative (e.g., injured bodies) images drawn from the International Affective Picture System (Lang et al., 1997). The two image sets used for the look and decrease conditions were matched on normative valence and arousal; valence and arousal means and image identification numbers are presented in Supplemental Table 1. Participants were instructed to “look” (keep their eyes on the image without trying to change their emotions) or “decrease” (use cognitive reappraisal to decrease their negative feelings, finding a way to interpret the image as less negative). Images were displayed for seven seconds. Forty-five images were shown: 15 each for “look-neutral,” “look-negative,” and “decrease-negative.” Following each image, participants rated how negative they felt using a five-button response box attached to their right hand where 1 (thumb) indicates “not at all negative” and 5 (pinkie) indicates “very negative.”
2.2.2. Trait Emotion Regulation.
The Emotion Regulation Questionnaire (Gross and John, 2003) is a 10-item trait measure assessing use of cognitive reappraisal and expressive suppression. The reappraisal subscale had good reliability (α = .88) and the suppression subscale had acceptable reliability (α = .75) in a community sample (Preece et al., 2021). Participants rated how much they agree with each statement; options ranged from 1 (strongly disagree) to 7 (strongly agree). Higher mean scores indicate greater use of the strategy.
2.2.3. Family History of Harmful Alcohol Use.
Participants reported whether family members had experienced social, occupational, or legal consequences due to substance use, and reported which relatives were affected, on the Family History Assessment Module (Rice et al., 1995). We created a dichotomous family history group variable where participants were family history positive if a first-degree relative (parent or sibling) had experienced such consequences, as parental (Acheson et al., 2014; Martz et al., 2018) and sibling (Windle, 2000) substance use are associated with substance use risk. All family history positive participants had a first-degree family member with either harmful alcohol use only (n = 11) or both harmful alcohol and other substance use (n = 20). All others were coded as family history negative. To conduct sensitivity analyses, we calculated a family history density score (see Supplemental Materials).
2.2.4. Lifetime psychiatric diagnoses.
Trained clinical staff administered the Structured Clinical Interview for DSM-5 Disorders Research Version (First et al., 2015), a semistructured interview guide to assess psychiatric disorders. We created separate dichotomous (0 = no, 1 = yes) variables for meeting past criteria and current criteria for each of the following diagnoses: alcohol use disorder, cannabis use disorder, mood disorders, anxiety disorders, and trauma- and stressor-related disorders.
2.2.5. Substance Use.
Participants reported their age at first alcohol use (> one sip), percent of days used alcohol in the past year, times used marijuana in the past year, and lifetime use of e/cigarettes (0 = no, 1 = yes). Participants completed the Alcohol Use Disorder Identification Test (Saunders et al., 1993), a 10-item self-report measure used to screen for harmful alcohol use with a cutoff score ≤ 7 indicating non-hazardous drinking.
2.2.6. Demographic Information.
Participants reported current age at the time of the laboratory visit, sex (0 = male, 1 = female), gender (male, female, trans male/trans man, trans female/trans woman, gender queer/gender non-conforming, different identity), race (American Indian or Alaska Native, Asian, Black/African American, Native Hawaiian or Pacific Islander, Unknown; individuals were allowed to choose all that applied and those endorsing two races were coded as biracial), and ethnicity (Hispanic or Latino/a, not Hispanic or Latino/a). Participants could choose “Prefer not to answer.”
2.4. fMRI Acquisition
We used a Siemens 3.0 Tesla Skyra Magnet with a 20-channel head coil. Functional images were acquired using blood-oxygenated-level-dependent signal across 40 axial slices with TR = 2000-ms, TE = 30-ms, flip angle = 77°, field of view = 220-mm, 40 axial 3-mm thick slices, and multiband slice acceleration factor of 2 to increase spatial resolution while maintaining temporal signal-to-noise ratio. The echoplanar imaging acquisition time for this task was 13 minutes. The acquisition was in oblique orientation. For anatomical reference, high-resolution T1-weighted images were collected.
2.5. fMRI Data Preprocessing
Data were preprocessed with Analysis of Functional NeuroImages version 22.0.21 (AFNI; Cox, 1996). We converted data into AFNI compatible formats and aligned the echoplanar images to the anatomical images. We nonlinearly warped the anatomical data to Montreal Neurological Institute (MNI) standard space using the AFNI @SSwarper command, skull-stripping the brain and deobliquing the data using the MNI152_2009_template_SSW.nii.gz template https://afni.nimh.nih.gov/pub/dist/doc/htmldoc/template_atlas/sswarper_base.html). We deobliqued the task data to match the anatomical data using AFNI’s 3dWarp command. The final voxel resolution was 3-mm isotropic. We censored time points with greater than 0.3-mm Euclidean distance of framewise displacement from analyses. We used an 8-mm kernel for blur, scaled each run to produce a mean intensity for each voxel of 100, and applied a 7-second block model to regression analysis. Regressors of interest were the presentation of each cue (look-neutral, look-negative, decrease-negative). We included six regressors of no interest to account for motion of translation and rotation in the x, y, and z dimensions.
2.6. Data Analytic Plan
We assessed significant differences (p < .05) between the family history positive and negative groups on characteristic and substance use variables, using two-sample t-tests and Wilcoxon rank sum tests for continuous variables, and Pearson’s chi-square tests and Fisher’s Exact Test for categorical data. Linear mixed-effects models and follow-up analysis of variance (ANOVA) with Kenward-Roger approximation assessed the main effects and interactions of family history group and cue on subjective ratings of negative emotions following the emotion regulation task. Data were analyzed in R version 4.1.2.
Whole-brain group-level fMRI analyses using a linear mixed effects model with restricted maximum likelihood were used to identify main effects of group and cue, and group-by-cue interactions on neural activation during the emotion regulation task using AFNI. Using the nlme package in R software implemented in AFNI, we entered the beta weights for family history group, decrease-negative cue, look-negative cue, and look-neutral cue as fixed effects, and participant as the random effect variable. Using a minimum voxel-wise threshold of p ≤ .001 and α = .05, a Monte Carlo simulation using AFNI’s 3dClustSim program with spatial autocorrelation function correction parameters of 0.75, 7.26, and 14.58, produced a minimum cluster size of k ≥ 54 voxels, an adequate cluster size for these analyses. Regions surviving the minimum threshold were defined using the MNI_Glasser_HCP_V1.0: Glasser HCP 2016 Surface-Based Parcellation atlas in AFNI. Next, in R, a linear mixed effects model and analysis of variance (ANOVA) with Kenward-Roger approximation assessed the main effects of group and cue (p < .05), and the group-by-cue interaction (p < .10) within each cluster surviving at the minimum threshold of k ≥ 54 (p ≤ .001). To be sensitive to any possible interaction effects, their impact on the results of main effects, and to account for the decreased power inherent in interactions, we increased our alpha to .10 for follow-up testing of interactions. Post hoc tests using the lsmeans function with Tukey adjusted p-values were used to assess the main effect of cue, and group-by-cue interactions, on reappraisal capacity (decrease-negative relative to look-negative contrast) and emotional reactivity (look-negative relative to look-neutral contrast). As cognitive reappraisal during the task was our primary contrast of interest, we assessed the magnitude of effect of group during this contrast alone, generating an effect size map using Cohen’s d, as recommended in this article (Chen et al., 2021) on transparency and completeness in neuroimaging research.
Post hoc region of interest (ROI) analyses examined activation in the bilateral basolateral and central amygdalae for family history group during the emotion regulation task. We defined two masks for the basolateral and central amygdala subdivisions using the CIT168 Amygdala Atlas v1.0.3 (DOI 10.17605/OSF.IO/R2HVK; Tyszka and Pauli, 2016) using the 3dcalc program in AFNI. We used the 3dresample program in AFNI to resample the 1-mm masks to 3-mm to fit our data, and then extracted values from the masks using the 3dROIstats program in AFNI. Post hoc tests using the lsmeans function with Tukey adjusted p-values were used to assess the contrasts of interest noted above, in these amygdala subdivisions.
We conducted several sensitivity analyses to further investigate effects of family history density, as well as the impact of sex (see Supplemental Materials).
3. Results
3.1. Participant Characteristics and Substance Use
The sample was 75 young adults aged 18–23 (see Table 1 for characteristics by group). The family history positive versus family history negative group had a greater proportion of females (77.42% versus 40.91%), past mood disorders (38.71% versus 13.64%) and past (12.90% versus 0.00%) and current (29.03% versus 2.27%) anxiety disorders. The groups did not differ on the remaining participant characteristic or substance use variables (Table 1).
Table 1.
Participant Characteristics and Substance Use by Family History Group
| FH+ (n = 31) | FH− (n = 44) | |||
|---|---|---|---|---|
| Variable | n (%) | n (%) | Test Statistic | |
| Sexa | Male | 7 (22.58) | 26 (59.09) | χ2(1) = 8.41* |
| Female | 24 (77.42) | 18 (40.91) | ||
| Race | American | 1 (3.23) | 0 (0.00) | |
| Indian/Alaska Native | 1 (3.23) | 4 (9.09) | Fisher’s p = .123 | |
| Asian | 1 (3.23) | 2 (4.55) | ||
| Black/African | 27 (87.10) | 30 (68.18) | ||
| American | 0 (0.00) | 6 (13.64) | ||
| White | 1 (3.23) | 2 (4.54) | ||
| Biracial | ||||
| No | ||||
| Response/“Unknown” | ||||
| Ethnicity | Hispanic | 7 (22.58) | 11 (25.00) | |
| Non-Hispanic | 24 (77.42) | 32 (72.73) | Fisher’s p = 1.000 | |
| No | 0 (0.00) | 1 (2.27) | ||
| Response/“Unknown” | ||||
| Alcohol Use Disorder | Past | 3 (9.68) | 1 (2.27) | Fisher’s p = .300 |
| Current | 1 (3.23) | 0 (0.00) | Fisher’s p = .413 | |
| Cannabis Use Disorder | Past | 1 (3.23) | 1 (2.27) | Fisher’s p = 1.000 |
| Current | 1 (3.23) | 1 (2.27) | Fisher’s p = 1.000 | |
| Mood Disorder | Past | 12 (38.71) | 6 (13.64) | χ2(1) = 4.97* |
| Current | 4 (12.90) | 2 (4.55) | Fisher’s p = .224 | |
| Anxiety Disorder | Past | 4 (12.90) | 0 (0.00) | Fisher’s p = .026* |
| Current | 9 (29.03) | 1 (2.27) | ||
| Fisher’s p = .001* | ||||
| Trauma- and Stressor-Related Disorder | Past | 7 (22.58) | 5 (11.36) | Fisher’s p = .216 |
| Current | 2 (6.45) | 2 (4.55) | Fisher’s p = 1.000 | |
| Have Ever Used e/Cigarettes More Than a Puff | 10 (32.26) | 21 (47.73) | χ2(1) = 121 | |
| High Motion in Scanner | 3 (9.677) | 6 (13.64) | Fisher’s p = .728 | |
| M (SD) | M (SD) | Test Statistic | ||
| Current Age | 21.02 (1.33) | 20.96 (1.37) | t(73) = 0.20 | |
| Age at First Use of Alcohol | 16.29 (1.99) | 16.59 (2.05) | t(73) = 0.63 | |
| AUDIT Score at Study Visit | 4.55 (2.59) | 4.61 (2.28) | t(73) = 0.12 | |
| Median (IQR) | Median (IQR) | Test Statistic | ||
| Times Used Marijuana in Past Yearb | 0.00 (5.00) | 1.00 (3.25) | W = 682.00 | |
| Percent Days Drank Alcohol in Past Yearc | 7 (15.00) | 10 (13.75) | W = 610.50 | |
| Percent of Scan Data Censored (% TRs) d | 2.31 (6.36) | 3.47 (10.33) | W = 622.00 | |
Note. FH+ = family history positive, FH− = family history negative, AUDIT = Alcohol Use Disorder Identification Test, IQR = interquartile range.
Participants reported their sex and gender to be the same (i.e., those who reported sex as female also reported gender as female).
Family history positive participants ranged from 0–365 times, and family history negative ranged from 0–100 times.
Family history positive participants ranged from 1–75%, and family history negative ranged from 0–60%.
Family history positive participants ranged from 0–24.57%, and family history negative ranged from 0–37.57%.
p < .05
3.2. Trait Emotion Regulation and Behavioral Results
Trait cognitive reappraisal (t73= 0.69, p = .494) and expressive suppression (t73= 0.44, p = .665) did not differ by family history group (Table 2). A linear mixed effects model revealed no significant main effect of group (F1,73= 0.78, p = .381) or group-by-cue interaction (F2,146= 0.25, p = .779) on subjective ratings of negative feelings on the emotion regulation task. There was a main effect of cue on subjective negative feelings (F2,146= 588.81, p < .001), where participants reported less negative feelings following the decrease-negative (M = 2.82 ± 0.61) cue relative to the look-negative cue (M = 3.53 ±0.63; t146= 9.85, p < .001), and greater negative feelings following the look-negative cue relative to the look-neutral cue (M = 1.11 ± 0.20; t146= 33.39, p < .001).
Table 2.
Trait Emotion Regulation and Emotion Regulation Task Outcomes by Family History Group
| FH+ (n = 31) | FH− (n = 44) | ||
|---|---|---|---|
| Variable | M (SD) | M (SD) | |
| Emotion Regulation Questionnaire | Cognitive Reappraisal | 5.05 (.94) | 5.20 |
| Expressive Suppression | 3.68 | (99) | |
| (132) | 3.81 | ||
| (132) | |||
| Emotion regulation task | look-neutral | 1.10 (.23) | 1.12 |
| look-negative | 3.48 (.60) | (17) | |
| decrease-negative | 2.75 (.55) | 3.57 | |
| Mean difference of look-negative – decrease-negative | 0.74 (.61) | (.65) | |
| 2.87 | |||
| (.65) | |||
| 0.70 | |||
| (.62) |
Note. Scores on the emotion regulation task indicate the average self-reported negative emotions following the display of neutral and negative images on a scale of 1 (not at all negative) to 5 (very negative). FH+ = family history positive, FH− = family history negative.
3.3. Brain Imaging Results
3.3.1. Whole-Brain Analyses
3.3.1.1. Effect of Cue.
Whole-brain analysis revealed the majority of the brain showed a significant effect of cue on the emotion regulation task. Examination of the cognitive reappraisal (decrease-negative > look-negative) and emotional reactivity (look-negative > look-neutral) contrasts largely reflects what has been previously reported and indicates the task works as expected (see Supplemental Materials and Supplemental Figure 1).
3.3.1.2. Effect of Family History Group.
Whole-brain analysis identified no clusters with a significant main effect of group on neural activation. Whole-brain analysis identified one cluster of 54 voxels (at a threshold of F = 7.11, p = .001) with a significant effect of group-by-cue on neural activation in the right area 9/46d and right anterior and posterior area 9 (Table 3, Figure 1). ANOVA revealed a significant main effect of group (F1,73= 6.73, p = .014) and of cue in this cluster (F2,146 = 18.15, p < .001). Post hoc testing showed that participants demonstrated evidence of cognitive reappraisal with increased neural activation during the decrease-negative cue relative to the look-negative cue (t146= 3.20, p = .005). There was evidence of significant emotional reactivity in this cluster with greater neural activation during the look-negative cue relative to the look-neutral cue (t146= 2.82, p = .015). Post hoc testing revealed that family history positive participants had a relatively greater increase in neural activation than family history negative participants during the decrease-negative cue (t133= 4.14, p = .001; Figure 1). The two groups did not significantly differ in neural activation during the look-negative cue (t(133) = 2.54, p = .121) or look-neutral cue (t133= 0.29, p = .999). Within the groups, for family history negative individuals, activation did not differ across condition (all p > 0.05). However, the family history positive group exhibited greater activation during the decrease-negative cue relative to the look-negative cue (t133= 4.83 p < .001; Figure 1).
Table 3.
Significant Effect of the Interaction of Family History Group and Cue During Whole-Brain Analysis at k ≥ 54 (p ≤ .001)
| n voxels | x | y | z | Peak Coordinate | Additional Regions in Cluster | F statistic |
|---|---|---|---|---|---|---|
| 54 | −28.5 | −58.5 | 31.5 | Right posterior area 9 | Right area 9/46d, right anterior area 9 | 11.59 |
Note. Family history positive participants demonstrated greater activation in this cluster than family history negative participants during the decrease-negative cue only.
Figure 1.

Significant effect of group-by-cue interaction on neural activation at the minimum voxel-wise threshold of k ≥ 54, p < .001. A. Whole-brain analysis revealed a significant effect of group-by-cue on neural activation in the right area 9/46d (middle frontal gyrus) and right anterior and posterior area 9 (superior frontal gyrus) located in the dorsolateral prefrontal cortex. B. Family history positive participants had greater activation than family history negative participants during the decrease-negative cue. The family history positive group demonstrated cognitive reappraisal in this region via greater activation during the decrease-negative cue relative to the look-negative cue. Black = Decrease-negative, red = Look-negative, green = Look-neutral.
Since we expected to find more widespread differences, we assessed the magnitude of the effect of group during the cognitive reappraisal contrast. We generated an effect size map using Cohen’s d (Figure 2). The majority of voxels (97.8%; 64,598 voxels) had values of d < 0.2, considered a small effect size. Only 0.12% of voxels had values of d ≥ .3, and no voxel had values greater than d = 0.45. Using G*Power 3.1.9.7 to calculate an adequately powered sample size, we found we would need a sample of 788 participants to detect a small effect (d = .2) where the groups differed during the decrease-negative relative to look-negative contrast (Figure 2).
Figure 2.
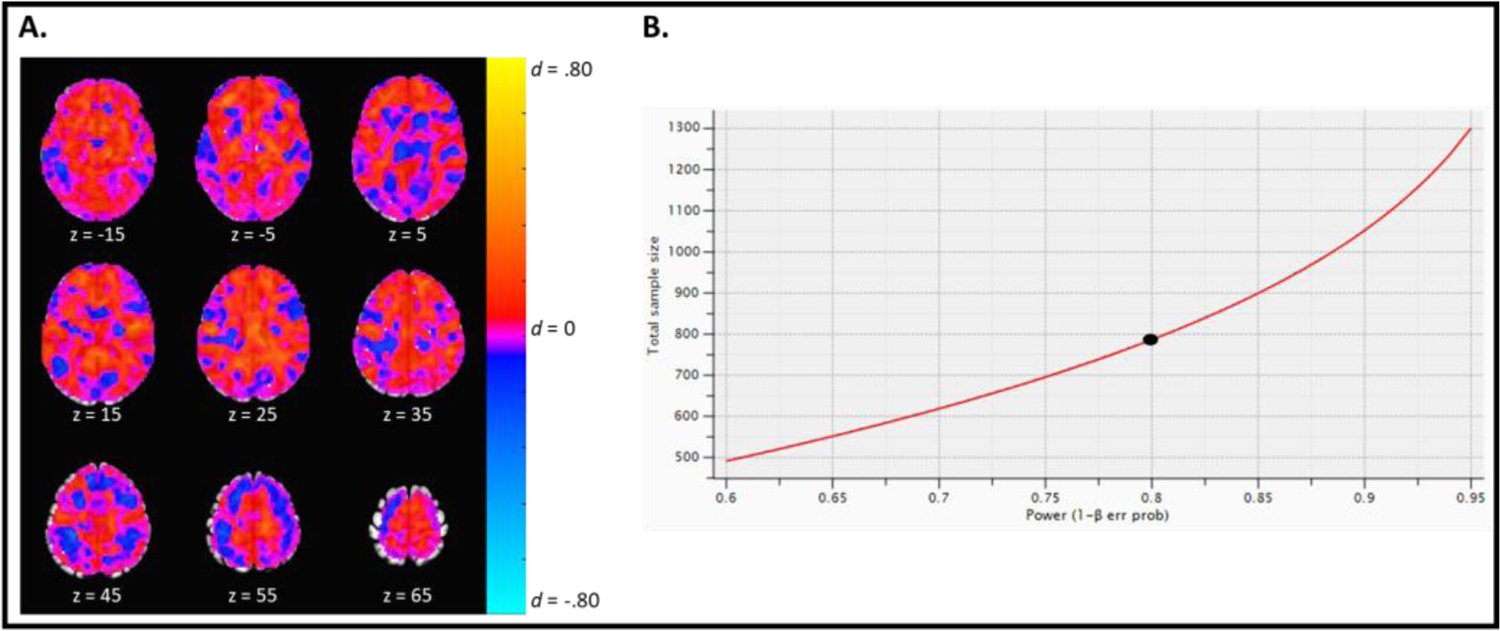
Magnitude of the effect of group on the cognitive reappraisal contrast. A. An unthresholded effect size map depicting the magnitude of the effect of group during the decrease-negative relative to look-negative contrast. Warmer colors (e.g., orange) indicate a greater effect of group during the decrease-negative cue, while cool colors (e.g., blue) indicate a greater effect of group during the look-negative cue. As can be seen, family history group does not appear to have a meaningful effect on cognitive reappraisal capacity as the majority of brain regions have small effect sizes d < .20. B. The graph depicts the sample sizes needed to detect a small effect of d = .20 at various power levels. To be adequately powered (.80) to detect a meaningful yet small effect (d = .20) of group on cognitive reappraisal capacity, we would need a sample of 788 participants (indicated by the black circle).
To confirm that results were not due to confounding factors, we removed participants (n = 29) with any history of a substance use disorder, a current mood, anxiety, or trauma- and stressor-related disorders, or high motion (TRs censored >20%), and included sex as a covariate in the model for the cluster identified above. Sensitivity analyses revealed that results overall were not significantly altered after adjusting the model and removing participants. Specifically, while the main effect of group (F1,43= 3.84, p = .056) dropped below the significance threshold, the main effect of cue (F2,88= 19.50, p < .001) and the group-by-cue interaction (F2,88= 10.24, p < .001) remained significant. The effect of sex (F1,43= 1.16, p = .288) was not significant (see Supplemental Material).
3.3.2. Region of Interest Analyses
Analyses revealed a significant main effect of cue in the right (F2,146= 29.17, p < .001) and left (F2,146= 26.58, p < .001) basolateral amygdalae and right (F2,146= 42.07, p < .001) and left (F2,146= 44.49, p < .001) central amygdalae (Figure 3). In all four clusters, post hoc testing showed evidence of emotional reactivity, with greater activation during the look-negative cue relative to the look-neutral cue (all t146> 5.00, all p < .001). In the left and right basolateral amygdalae, there was no evidence of down-regulation during cognitive reappraisal comparing the decrease-negative relative to the look-negative contrast (t146< 0.5, p > 0.9. The left central amygdala showed evidence of down-regulation during cognitive reappraisal, with less activation during the decrease-negative cue relative to the look-negative cue (t146= 2.50, p = .036). Meanwhile, there was no evidence of down-regulation during cognitive reappraisal in the right central amygdala, comparing the decrease-negative relative to look-negative contrast (t146= 1.08, p = .531).
Figure 3.
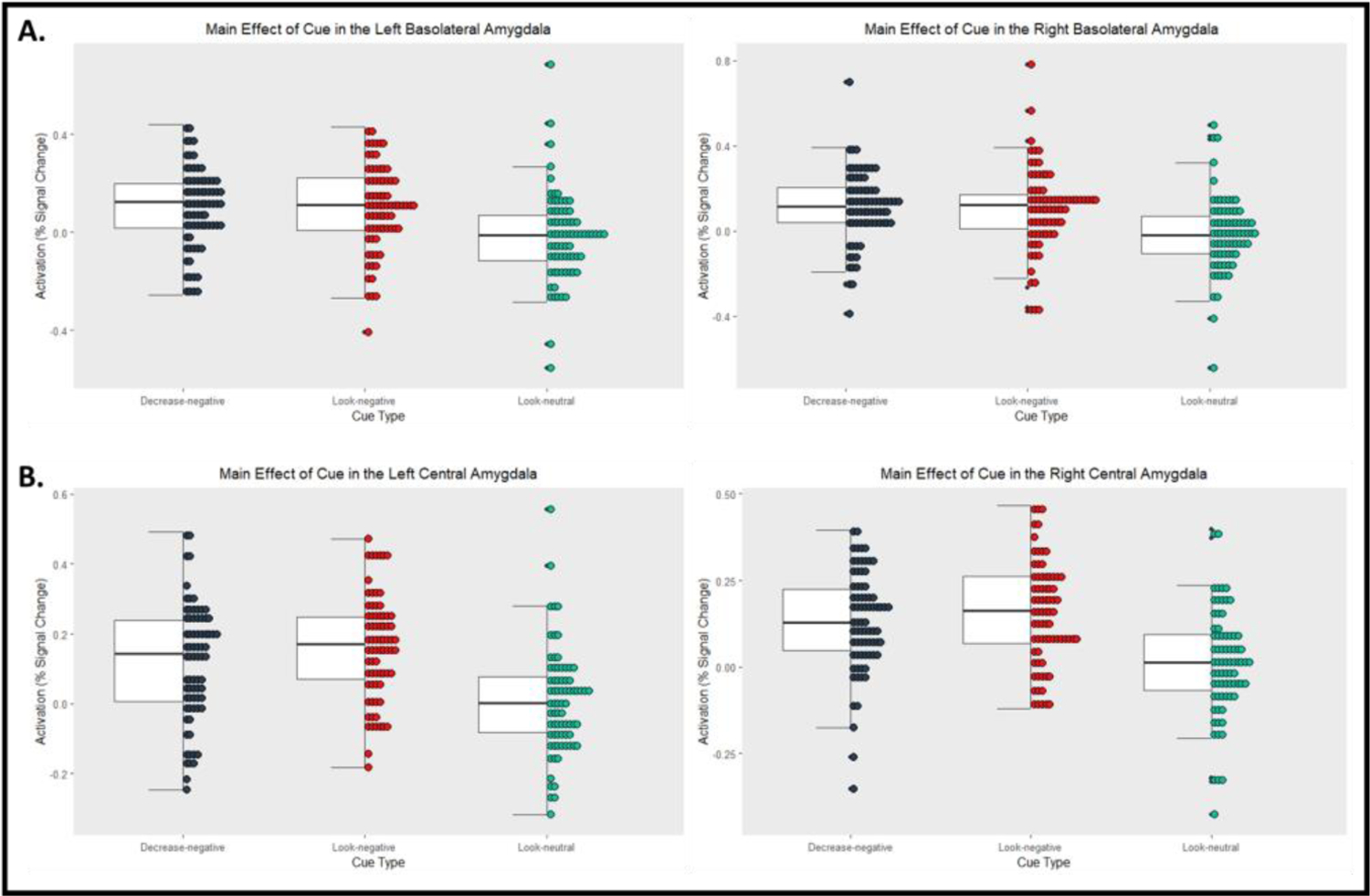
ROI Analyses: Effect of cue on neural activation in the amygdala. Black = Decrease-negative, red = Look-Negative, green = Look-neutral. A. Significant main effect of cue in the left and right basolateral amygdalae with greater activation during look-negative versus look-neutral contrast. B. Significant main effect of cue in the left and right central amygdalae. The left central amygdala demonstrated greater activation during the decrease-negative cue versus look-negative cue, and the look-negative cue versus look-neutral cue. The right central amygdala demonstrated greater activation during the look-negative cue versus look-neutral cue only.
There was no significant main effect of group or group-by-cue interaction in the right or left basolateral or central amygdalae (all F < 1, all p > 0.4, Figure 3).
4. Discussion
We examined whether young adults with and without a family history of harmful alcohol use differ on trait, behavioral, and neural correlates of emotion regulation and emotional reactivity. Overall, we found that young adults with and without a family history, but without current harmful patterns of substance use, engage in cognitive reappraisal at similar rates. The imaging data suggest little evidence for differences in neural activation by family history group at the whole-brain level and in amygdala regions of interest. We did find one cluster located in the right middle frontal gyrus and anterior and posterior superior frontal gyrus of the dorsolateral prefrontal cortex, where the groups differed in activation during the cognitive reappraisal instruction. Sensitivity analyses demonstrated that this group difference was not attributed to motion in the scanner, current psychiatric diagnoses, or lifetime history of a substance use disorder, or to differences in sex.
Contrary to our hypotheses, the groups did not differ on self-reported use of cognitive reappraisal or expressive suppression strategies, nor differed on subjective ratings of negative affect during the emotion regulation task. Consistent with previous research (e.g., McRae et al., 2012), our participants generally reported the lowest level of negative affect following passive viewing of neutral images, then decrease-negative images, and highest ratings after passive viewing of negative images. Our behavioral findings suggest that the “decrease” cue resulted in effective reappraisal for both groups. This may suggest that, prior to development of alcohol use disorder, individuals have unimpaired capacity for cognitive reappraisal.
As a whole, our sample demonstrated reappraisal capacity during the decrease-negative relative to look-negative contrast via greater neural activation in large portions of the brain, including the prefrontal cortex region. We also identified decreased activation in the left central amygdala during decrease-negative relative to the look-negative condition. Further, emotional reactivity was indicated via greater neural activation during the look-negative relative to look-neutral contrast in large portions of the brain, including all four amygdala subdivisions we examined. These results indicate significant task effects in both cognitive control and emotional processing regions.
Yet, when examining the effect of family history of harmful alcohol use, the groups only differed in neural activation during the reappraisal instruction (decrease) in the right middle and superior frontal gyri. Family history positive participants, compared to family history negative participants, demonstrated greater activation in this dorsolateral prefrontal cortex region when instructed to engage in cognitive reappraisal when viewing negative images. This cognitive control region is associated with decreasing negative affect during reappraisal (Buhle et al., 2014). These results suggest family history positive young adults may require greater recruitment of cognitive control regions to downregulate negative affect after witnessing and emotionally reacting to negative stimuli. However, a much larger sample size is needed to detect a meaningful and generalizable effect of family history group. Interestingly, the family history groups did not demonstrate differential activation in the right or left basolateral and central amygdalae, suggesting that, as with self-reported emotion, young adults without current harmful patterns of substance use with and without family history of harmful use may have similar modulation of the amygdala during emotion regulation tasks.
It is possible that the low rates of substance use in our sample may explain our largely null findings, since both groups represent mostly healthy young adult samples. However, individuals with alcohol use disorder can effectively decrease negative emotion when prompted (Jansen et al., 2019; Suzuki et al., 2020), and are similar to healthy controls on neural activation during reappraisal (Jansen et al., 2019). Given our results as a whole, it is unlikely that family history of harmful alcohol use has a meaningful effect on cognitive reappraisal capacity and emotional reactivity in a sample of young adults without current harmful patterns of substance use.
Clinical implications.
Our findings indicate at-risk individuals have the capacity to engage in emotion regulation when provided appropriate instruction and largely do not differ from low-risk individuals on neural activation, similar to previous findings (e.g., Jansen et al., 2019). Given the heritable risk for developing a substance use disorder and possible altered neural activation in cognitive control regions during emotion regulation, at-risk young adults may benefit from tailored interventions that increase cognitive reappraisal skills should they go on to develop a substance use disorder.
Future neuroimaging studies should examine whether there are differences in reappraisal capacity when at-risk young adults view substance use related stimuli versus neutral or negative images (i.e., Suzuki et al., 2020). Results from such studies will help further elucidate neural underpinnings of emotional responses and coping strategies in at-risk populations, which will allow for more precise and timely treatments. Additionally, dysregulation of positive affect is associated with harmful substance use (e.g., Schick et al., 2021). Researchers should examine positive emotions and family history group as risk factors for emotion dysregulation preceding and following the onset of a substance use disorder.
4.1. Limitations
While participants were screened for current harmful substance use prior to enrollment, we did not exclude seven individuals who met criteria for lifetime substance use disorders. However, sensitivity analyses revealed no significant differences in results when these individuals were removed from analyses. It is possible that our sample was particularly resilient (having made it to age 18–22 and not developed a substance use disorder) and not experiencing emotion dysregulation and may not represent an at-risk group. It is also possible that people who are motivated to participate in research about family history of harmful alcohol use are those who are most resilient. Several participants mentioned their desire to help with research about alcohol use because they did not want others to experience a childhood affected by their parent’s alcohol use disorder. It is therefore possible that our sample is not representative of the larger population of young adults at risk for harmful substance use. Finally, some participants had family histories of harmful alcohol use only, and others had history of alcohol and unspecified drug use; it is unknown if there might be different effects of these different types of familial histories. Nonetheless, our findings provide preliminary evidence for the role of emotion regulation capacity as a target for treatment.
4.2. Conclusions
Our findings suggest that young adults with family histories of harmful alcohol use, who do not currently have harmful patterns of use, have intact ability to apply cognitive reappraisal when prompted. There is little evidence that family history groups differ in neural correlates of emotion regulation and reactivity. It is encouraging that genetic risk for substance use disorders does not necessarily result in diminished cognitive reappraisal ability, since this may be a viable therapeutic target in individuals who go on to develop a disorder with associated emotion dysregulation.
Supplementary Material
Highlights.
Examined emotion regulation in young adults with family history of alcohol misuse
Those at risk for SUDs did not show deficits in emotion regulation
Family history of alcohol misuse may affect activation in prefrontal cortex
Family history is not associated with altered amygdala activation
Acknowledgements
Study data were collected and managed using REDCap electronic data capture tools hosted at the University of Colorado. REDCap is a secure, web-based application designed to support data capture for research studies, providing: 1) an intuitive interface for validated data entry; 2) audit trials for tracking data manipulation and export procedures; 3) automated export procedures for seamless data downloads to common statistical packages; and 4) procedures for importing data from external sources.
Role of Funding Source
This study was supported by grant funding from the National Institute on Alcohol Abuse and Alcoholism (R00AA024778 to Joshua Gowin). This project was supported by the NIH/NCATS Colorado CTSA Grant Number UL1TR002535. Its contents are the authors’ sole responsibility and do not necessarily represent official NIH views.
Declarations of interest:
This study was funded by the National Institute of Alcohol Abuse and Alcoholism (R00AA024778). This project was supported by the NIH/NCATS Colorado CTSA Grant Number UL1TR002535; its contents are the authors’ sole responsibility and do not necessarily represent official NIH views. The authors have no conflict of interest to declare.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Conflict of Interest
The authors have no conflicts of interest to declare.
References
- Acheson A, Wijtenburg SA, Rowland LM, Winkler AM, Gaston F, Mathias CW, Fox PT, Lovallo WR, Wright SN, Hong LE, 2014. Assessment of whole brain white matter integrity in youths and young adults with a family history of substance-use disorders. Human brain mapping 35(11), 5401–5413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Adamson SJ, Kay-Lambkin FJ, Baker AL, Lewin TJ, Thornton L, Kelly BJ, Sellman JD, 2010. An improved brief measure of cannabis misuse: the Cannabis Use Disorders Identification Test-Revised (CUDIT-R). Drug and alcohol dependence 110(1–2), 137–143. [DOI] [PubMed] [Google Scholar]
- AFNI, 9.3. @SSwarper base volumes. https://afni.nimh.nih.gov/pub/dist/doc/htmldoc/template_atlas/sswarper_base.html
- Berking M, Margraf M, Ebert D, Wupperman P, Hofmann SG, Junghanns K, 2011. Deficits in emotion-regulation skills predict alcohol use during and after cognitive–behavioral therapy for alcohol dependence. Journal of consulting and clinical psychology 79(3), 307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buhle JT, Silvers JA, Wager TD, Lopez R, Onyemekwu C, Kober H, Weber J, Ochsner KN, 2014. Cognitive reappraisal of emotion: a meta-analysis of human neuroimaging studies. Cerebral cortex 24(11), 2981–2990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen G, Taylor PA, Stoddard J, Cox RW, Bandettini PA, Pessoa L, 2021. Sources of information waste in neuroimaging: mishandling structures, thinking dichotomously, and over-reducing data. BioRxiv. [Google Scholar]
- Cox RW, 1996. AFNI: software for analysis and visualization of functional magnetic resonance neuroimages. Computers and Biomedical research 29(3), 162–173. [DOI] [PubMed] [Google Scholar]
- Cservenka A, Fair DA, Nagel BJ, 2014. Emotional processing and brain activity in youth at high risk for alcoholism. Alcoholism: Clinical and Experimental Research 38(7), 1912–1923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- D’Agostino A, Covanti S, Rossi Monti M, Starcevic V, 2017. Reconsidering emotion dysregulation. Psychiatric Quarterly 88(4), 807–825. [DOI] [PubMed] [Google Scholar]
- Deak JD, Johnson EC, 2021. Genetics of substance use disorders: a review. Psychological medicine, 1–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Filippi I, Hoertel N, Artiges E, Airagnes G, Guérin-Langlois C, Seigneurie A-S, Frère P, Dubol M, Guillon F, Lemaître H, 2019. Family history of alcohol use disorder is associated with brain structural and functional changes in healthy first-degree relatives. European psychiatry 62, 107–115. [DOI] [PubMed] [Google Scholar]
- First MB, Williams JB, Karg RS, Spitzer RL, 2015. Structured clinical interview for DSM-5—Research version (SCID-5 for DSM-5, research version; SCID-5-RV). Arlington, VA: American Psychiatric Association, 1–94. [Google Scholar]
- Gianaros PJ, Marsland AL, Kuan DC-H, Schirda BL, Jennings JR, Sheu LK, Hariri AR, Gross JJ, Manuck SB, 2014. An inflammatory pathway links atherosclerotic cardiovascular disease risk to neural activity evoked by the cognitive regulation of emotion. Biological psychiatry 75(9), 738–745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Glahn DC, Lovallo WR, Fox PT, 2007. Reduced amygdala activation in young adults at high risk of alcoholism: studies from the Oklahoma family health patterns project. Biological psychiatry 61(11), 1306–1309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gross JJ, 2002. Emotion regulation: Affective, cognitive, and social consequences. Psychophysiology 39(3), 281–291. [DOI] [PubMed] [Google Scholar]
- Gross JJ, John OP, 2003. Individual differences in two emotion regulation processes: implications for affect, relationships, and well-being. Journal of personality and social psychology 85(2), 348. [DOI] [PubMed] [Google Scholar]
- Harris PA, Taylor R, Thielke R, Payne J, Gonzalez N, Conde JG, 2009. Research electronic data capture (REDCap)—a metadata-driven methodology and workflow process for providing translational research informatics support. Journal of biomedical informatics 42(2), 377–381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Henderson KE, Vaidya JG, Kramer JR, Kuperman S, Langbehn DR, O’Leary DS, 2018. Cortical thickness in adolescents with a family history of alcohol use disorder. Alcoholism: Clinical and Experimental Research 42(1), 89–99. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jansen JM, van den Heuvel OA, van der Werf YD, De Wit SJ, Veltman DJ, Van Den Brink W, Goudriaan AE, 2019. Emotion processing, reappraisal, and craving in alcohol dependence: A functional magnetic resonance imaging study. Frontiers in psychiatry 10, 227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lang PJ, Bradley MM, Cuthbert BN, 1997. International affective picture system (IAPS): Technical manual and affective ratings. NIMH Center for the Study of Emotion and Attention 1(39–58), 3. [Google Scholar]
- Lindsay DL, Pajtek S, Tarter RE, Long EC, Clark DB, 2014. Amygdala activation and emotional processing in adolescents at risk for substance use disorders. Journal of child & adolescent substance abuse 23(3), 200–204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martz ME, Zucker RA, Schulenberg JE, Heitzeg MM, 2018. Psychosocial and neural indicators of resilience among youth with a family history of substance use disorder. Drug and alcohol dependence 185, 198–206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McRae K, Gross JJ, 2020. Emotion regulation. Emotion 20(1), 1. [DOI] [PubMed] [Google Scholar]
- McRae K, Gross JJ, Weber J, Robertson ER, Sokol-Hessner P, Ray RD, Gabrieli JD, Ochsner KN, 2012. The development of emotion regulation: an fMRI study of cognitive reappraisal in children, adolescents and young adults. Social cognitive and affective neuroscience 7(1), 11–22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Minkel JD, McNealy K, Gianaros PJ, Drabant EM, Gross JJ, Manuck SB, Hariri AR, 2012. Sleep quality and neural circuit function supporting emotion regulation. Biology of mood & anxiety disorders 2(1), 1–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morawetz C, Bode S, Derntl B, Heekeren HR, 2017. The effect of strategies, goals and stimulus material on the neural mechanisms of emotion regulation: A meta-analysis of fMRI studies. Neuroscience & Biobehavioral Reviews 72, 111–128. [DOI] [PubMed] [Google Scholar]
- Preece DA, Becerra R, Hasking P, McEvoy PM, Boyes M, Sauer-Zavala S, Chen W, Gross JJ, 2021. The Emotion Regulation Questionnaire: Psychometric properties and relations with affective symptoms in a United States general community sample. Journal of Affective Disorders 284, 27–30. [DOI] [PubMed] [Google Scholar]
- Rice JP, Reich T, Bucholz KK, Neuman RJ, Fishman R, Rochberg N, Hesselbrock VM, Nurnberger JI, Schuckit MA Jr, Begleiter H, 1995. Comparison of direct interview and family history diagnoses of alcohol dependence. Alcoholism: Clinical and Experimental Research 19(4), 1018–1023. [DOI] [PubMed] [Google Scholar]
- Saunders JB, Aasland OG, Babor TF, De la Fuente JR, Grant M, 1993. Development of the alcohol use disorders identification test (AUDIT): WHO collaborative project on early detection of persons with harmful alcohol consumption-II. Addiction 88(6), 791–804. [DOI] [PubMed] [Google Scholar]
- Schick MR, Weiss NH, Contractor AC, Thomas ED, Spillane NS, 2021. Positive emotional intensity and substance use: the underlying role of positive emotional avoidance in a community sample of military veterans. The American journal of drug and alcohol abuse 47(3), 373–382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shoal GD, Giancola PR, 2001. Cognition, negative affectivity and substance use in adolescent boys with and without a family history of a substance use disorder. J. Stud. Alcohol 62(5), 675–686. [DOI] [PubMed] [Google Scholar]
- Stellern J, Xiao KB, Grennell E, Sanches M, Gowin JL, Sloan ME, 2022. Emotion regulation in substance use disorders: a systematic review and meta-analysis. Addiction. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suzuki S, Mell MM, O’Malley SS, Krystal JH, Anticevic A, Kober H, 2020. Regulation of craving and negative emotion in alcohol use disorder. Biological psychiatry: cognitive neuroscience and neuroimaging 5(2), 239–250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Swendsen JD, Conway KP, Rounsaville BJ, Merikangas KR, 2002. Are personality traits familial risk factors for substance use disorders? Results of a controlled family study. American journal of psychiatry 159(10), 1760–1766. [DOI] [PubMed] [Google Scholar]
- Tyszka JM, Pauli WM, 2016. In vivo delineation of subdivisions of the human amygdaloid complex in a high-resolution group template. Human brain mapping 37(11), 3979–3998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weiss NH, Kiefer R, Goncharenko S, Raudales AM, Forkus SR, Schick MR, Contractor AA, 2022. Emotion regulation and substance use: A meta-analysis. Drug and alcohol dependence 230, 109131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Windle M, 2000. Parental, sibling, and peer influences on adolescent substance use and alcohol problems. Applied Developmental Science 4(2), 98–110. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


