Abstract
Hyaluronan (HA) is a linear polysaccharide consisting of disaccharide units which are the d-glucuronic acid and n-acetyl-d-glucosamine. As the largest component of the extracellular matrix in microenvironment, HA polymers with different molecular weights vary in properties to molecular biology function. High molecular weight HA (HMW-HA) is mainly found in normal tissue or physiological condition, and exhibits lubrication and protection properties due to its good water retention and viscoelasticity. On the other hand, an increase in HA catabolism leads to the accumulation of low molecular weight HA (LMW-HA) under pathological circumstances such as inflammation, pre-cancerous and tumor microenvironment. LMW-HA acts as extracellular signals to enhance tumorigenic and metastatic phenotype, such as energy reprogramming, angiogenesis and extracellular matrix (ECM) remodeling. This review discusses the basic properties of this simplest carbohydrate molecule in ECM with enormous potential, and its regulatory role between tumorigenesis and microenvironmental homeostasis. The extensive discoveries of the mechanisms underlying the roles of HA in various physiological and pathological processes would provide more information for future research in the fields of biomimetic materials, pharmaceutical and clinical applications.
Keywords: Hyaluronan, Extracellular matrix, Hyaluronan catabolism, Tumor microenvironment, Extracellular signal
Introduction
Cancer diseases have long been regarded as the most threatening burden of human healthcare. Being the largest populated country, annual cancer incidence in China contributed to nearly 50% of the total global cancer incidence, meanwhile, cancer mortality sustained at the highest level (over 50%) worldwide [1, 2]. Since the GLOBOCAN project was held in the 1980s, cancer incidence and mortality increased with breast, lung, colorectal, gastric and prostate accounting for over 50% of all cancer types [3, 4]. Those cancers normally induce latent symptoms that alarm patients when they undergo a late stage. Terminal stage of cancer mostly developed metastasis which gradually leads to multiple organ failure. Researchers found that the metastasis progression of tumors obeyed the “seed and soil” theory with partial phenotype transformation [5]. In micro-perspective principles of the tumor environment, different tumor cells required specific “soil” for their prosperity [6].
The tumor microenvironment (TME) is the local biological environment in which the tumor cells live. Microenvironment homeostasis depends on a complicated and dynamic compensation mechanism [7]. It is currently known that the tumor microenvironment contains a large amount of extracellular matrix (ECM), the main components of which include collagen, non-collagen glycoproteins, glycosaminoglycans and proteoglycans. Changes in the composition and total content of ECM can strongly affect the biological properties of tumor cells and stromal cells, such as proliferation and motility, and play an important role in tumor metastasis [8]. Hyaluronan (HA) is the main component of glycosaminoglycans in ECM, accounting for about 85% of ECM components [9]. As the largest component of ECM, hyaluronan (HA) acted as a supportive framework in tumor stroma [10, 11]. Normally, homeostasis of HA stayed at the dynamic balance between biosynthesis and hydrolysis every second accompanied by a fluctuant distribution of HA with different molecular weights during either physiological or pathological processes [12].
Under physiological conditions, HMW-HA, synergistically produced by cell membrane HA synthetase (HAS) as tail elongation in a “pendulum” manner, occupies the interstitial space among multiple organs [13]. Pathological conditions such as tumors, inflammation, oxidative stress, and tissue remodeling exacerbate HA degradation, which rapidly degraded into small fragment of different length (molecule weight ranging from 1–1000 kDa) by hyaluronidases (hyaluronidases, HYALs) [14]. Therefore, the fast degradation sometimes is incomplete resulting in the accumulation of low-molecular-weight products of HA in the interstitial space of organs or tissues lesion (arthritis and tumor) [15, 16]. Interestingly, LMW-HA or small HA fragments, but not HMW-HA, are critical for malignant progressions such as tumor invasion and metastasis. Here, we described the turnover and catabolism of HA under different conditions and discussed the causes of tumorigenesis and microenvironment homeostasis regulated by those different types of HA.
HA distribution and molecule weight composition
HA is composed of disaccharide units with β-1,4 or β-1,3 glycosidic bonds and stabilized as linear polymer glycosaminoglycan with molecule weight ranging from 103 to 107 kD [17, 18], depending on the length of disaccharide units. HA can be classified as high or mega-molecule weight HA (HMW-HA or vHMW-HA), with over 1000 kDa; medium molecular weight HA (MMW-HA) with 100 ~ 1000 kDa, low molecular weight HA (LMW-HA) with 20-100 kDa, and HA fragment (fgHA) from oligosaccharide to 20 kDa [19, 20]. In different circumstances, the dominant HA could be very different among microenvironment, since the varied molecular weight HA displays exclusive work from physiological protection to oncogene promotion, depending on the stage of the disease.
In physiological conditions, the highest HA concentration is present in synovial fluid reaching around 1000 μg/mL, followed by that in the vitreous body (around 680-1800 μg/mL), cumulus cell–oocyte complex (100-500 μg/mL), dermis (200 μg/mL), brain (30-120 μg/mL) and the skin (17.5–23.5 μg/mL) [21–25]. HMW-HA has strong water retention and swelling properties, and its volume can be extended up to 1000 times its original size after absorbing water, forming a complex network structure to accommodate the liquid molecules. Therefore, HA is a biological "lubricant" involved in lubricating joints or holding gelatinous connective tissue together [9]. HA fills among soft connective tissues of organs, keeping moisture retention and protecting joint cartilage from mechanical friction. Peri-cellular HA absorbs water from interstitial space, where the mesh scaffolds were formed to expend a moisture living space, tightly surrounding as a hydrogel halo which avoided tissue compression from growth [26].
The catabolism of HA slows down in lymphatic systems, thereby the HA level is stabilized at a certain level. In lymphatic systems, the concentration of HA ranges from 0.5–18 μg/mL, wherein the highest level of HA in thoracic lymph reached 8.5 μg/mL for adults vs 18 μg/mL for children [27]. However, circulated HA in blood stays low in concentration (below 1 μg/mL) [28]. The low concentration of HA might be attributed to the fast uptake by the liver or endothelial cells, yielding a large amount of turnover about 10–100 mg per day and a fast half-time of degradation (2–5 min) in blood [27–29]. According to this physiological property, the increase of serum HA attributes to pathophysiologic deposition of a large amount of HA, leading to circulating accumulation. Serum HA level now is widely recommended as an indicator of liver fibrosis, a vital early sign of liver cirrhosis [30–32]. Compared to the normal control, the mean value of serum HA level has tenfold high in patients with chronic hepatitis, 15-fold higher in patients with liver cirrhosis, but only 5 times that in patients with liver cancer liver [33]. Also, over-produced HA from active HA synthesis in the liver activates the hepatic stellate cells resulting in liver fibrosis [34]. Besides, the decreased concentration of HA in arthrosis is a vital index of patients with rheumatic arthritis (RA) and osteoarthritis (OA) wherein the overloaded inflammation pathogenesis caused hyper-degradation of HA [35].
Property and variety of HA under different microenvironment
In the physiological aqueous solution, polymers of HA self-assemble H-bonds with solvent to generate many randomly hydrophobic zones being regarded as ‘tangle’ of HA cross-link [36]. These hydrophobic pockets endow HA aqueous the property of non-Newtonian flow, bearing elasticity, viscosity and water retention [37]. These structures however are loose and deformed but easily recovered, since the H-bonds only consist of weak non-covalence. Among the microenvironment, various glycoproteins attach to the liner chain of HMW-HA with non-covalent bonds, which stabilizes their location and buffers them in the ECM of organs. Besides, under the catalysis of TSG-6, HMW-HA covalently binds to the heavy chain of IαI which is recruited by tetramer pentraxins to block the amplification of inflammation [38, 39]. On the other hand, this property makes HA a good lubricant in the joint cavities or buffer zones between tissues once the microenvironment shear stress increases and interstitial space decreased [40]. In the tumor microenvironment, LMW-HA takes a high proportion because the balance of biosynthesis and degradation has been interrupted, resulting in incomplete digestion of HMW-HA to LMW-HA or fgHA. The solution of LMW-HA still bears part of viscoelasticity, in a wide range concentration of 10–20,000 μg/mL [36].
In cosmetic use, HA filler is predominant the consumables. During the aging process, a large amount of HMW-HA or vHMW-HA is underwent depolymerization in skin tissues, which attributes to the accumulation of cellular metabolites. The injection of cosmetic HA filler, normally makes of HMW-HA or vHMW-HA, can postpone this cell aging progression in the skin [41]. Evidence in rodent models illustrated that older mice contained lower HA amounts than their younger counterparts [42]. The decreased HA was found in damaged skin from UV-irradiation and photo-damaged conditions. When treating mechanical damage of the cartilago with intra-articular injection of HA filler, symptoms would be released due to recovery of the lubrication within the cartilage [43, 44]. Based on the good swelling property and water retentivity without immune rejection, HA filler or cross-linked HA hydrogels have been widely used as anti-aging and anti-wrinkle products in terms of slowing down HA degradation through HMW-HA overload [45].
Under the physical condition, LMW-HA is rather taken up by phospholipids endocytosis and is hydrolyzed into UDP-oligosaccharide in the lysosome. However, in pathological situations, an increase of LMW-HA attributed to active hyaluronidase or accumulated free radicals would be a marker of disease exacerbation such as inflammatory filtration, tissue injury and metastasis [46, 47]. LMW-HA loses lubrication function since the deficiency of hydrophobic interspaces causes a decrease in viscoelasticity leading to increase tissue rigidity [28]. Besides, high LMW-HA inclines chronic inflammation which induced the initiation of irreversible diseases such as cartilage damage, fibrosis and pre-cancerous condition [48–50].
HA bio-synthesis
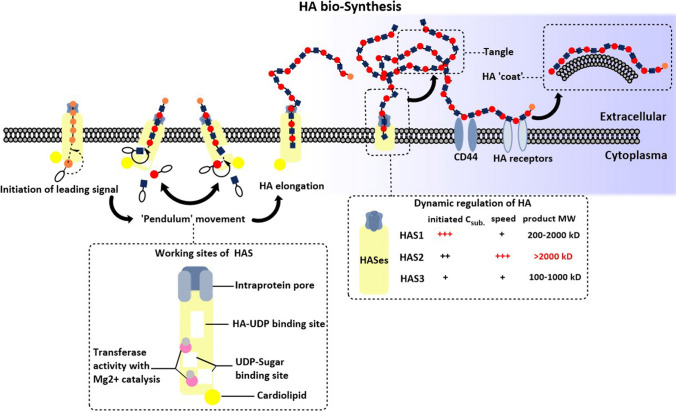
The synthesis of HA could be briefly introduced as HAS recruited and connected UDP-sugars by forming β-glycosidic bonds at the cellular membranes associated with energy consumption. The major site of HA biosynthesis is derived from fibroblasts of tumor stroma rather than epithelium, wherein the HASes are highly expressed [51, 52]. Since HASes family are all anchored in the plasma membrane with an active region located inside the cytoplasm. HA, therefore, was formed intra-membrane [53]. Before the start of synthesis, HAS requires a lipid environment wherein the combination of cardiolipin and active site triggered its activity, while the binding of cholesterol attenuated its activation [54]. Another step would be the recruitment of two UDP-sugar substrates, UDP-GlcNAc and UDP-GlcUA, and two catalytic glycosyltransferases [54]. When biosynthesis is initiated, a chitin primer containing hexosaminidase with 6 constant GlcNAc work as a leading signal [55]. Due to the steric hindrance between UDP-GlcNAc and UDP-GlcUA, the ‘Pendulum Model’ was introduced by P. H. Weigel as the putatively dynamic progression. When the reducing end addition of HA chain elongation occurred, the chemical structure of both GlcUA-β(1,3)-GlcNAc bond and GlcNAc-β(1,4)-GlcUA-bond limited formation of long conjugation system thereby alternatively forming non-planar conformation of the chain [54].
There were two theories explaining HA excretion, one of them was a pressure-regulated molecule pump that recognized intracellular HA and excrete extracellular matrix [53]. The synthesized HA does not remain with HAS for a long time due to the Brownian movement. Along with HA addition, a higher pressure would be gained. Therefore, the outflow of HA was essential for releasing intra-protein pressure as well as lengthening the elongation reaction. The other theory preferred that HA was slowly extruded in situ through the protein pore when the chain reaches ~ 20 kDa [56]. The HA chain outflow might loosely attach to the extra-membrane region of HASes, forming the HAS-HA complex. The accumulation of this complex remains the key regulator for slowing down HA synthesis (Fig. 1).
Fig. 1.
The bio-synthesis of HA. The process of HA synthesis in the ‘Pendulum’ movement pathway starts from the initiation of the leading signal, a chitin primer connecting 6 constant GlcNAc. Two substrates of UDP-sugars, UDP-GlcNAc and UDP-GlcUA were recruited to the membrane, and generated linear polymers connecting with GlcUA-(1,3)-GlcNAc -β(1,4)-GlcUA-bond. The residue addition synthesis was produced inside the HAS, and was delivered by transferase under Mg2+ catalysis. The HA polymers were extruded through intra-protein pore of HAS, hanging out of the extracellular space nearby the membrane or folding as tangle through the intra-molecular and intermolecular H-bonds. Different HA receptors recognized specific HA polymers with different molecule weights. For example, CD44 captured HMW-HA and formed HA ‘coat’ on the external surface of cells. The lower left box with dash line indicates the detail and specific position of HAS how it produces the HA synthesis. The lower right box with the dash line indicates the working requirement and products of HA synthesis when three HAS isoforms, HAS1, HAS2, HAS3, start dynamic regulation
Active and adequate HASes kept HA at the required level. Although the fast turnover of stromal HA causes HA insufficiency, tumor cells may compensate for the expression and activity of HASes at this moment. Tumor cells, normal and cancer-associated fibroblasts could generate HA, though the type and amount of HA vary in specific circumstances [57, 58]. Although the three HAS subtypes had 55% identical sequence to each other (Fig. 2), each of them has the difference in affinity and catalytic efficacy with UDP-sugar substrates, which was determined by the Km value, obeying the Michaelis–Menten equation. The initiation and amount of HA synthesis depend on the intracellular concentration of UDP-substrate and the expression and activity of HAS [59, 60]. HAS1 obtained the highest Michaelis Km value which reflected the lower affinity to substrates and slower rate for HA formation [61]. HAS3 could be activated at very low substrate content and the increase in substrate level affects the elevated synthetic amount of HA. Unlike HAS3, the HA production by HAS1 would only be started when the UDP-sugar doubled compared with the other two isoforms However, HA production generated by HAS1 remains the most sensitive to UDP-sugar changes. HAS2 produced the largest amount of HA, and the initiation threshold of HA generation only required low UDP-sugar. Besides, the synthetase activity of HAS2 is less sensitive to the rise of substrate level [60]. Thus, the microenvironment HA was mainly generated by HAS2, which requires lower activation energy and faster synthetic speed. HAS2 focus on recruiting MMH-HA to produce vHMW-HA while HAS1 and 3 contribute to MMH- to HMW-HA (with molecular weight ranging around 200kD-2000kD and 100-1000kD), respectively [62]. After extracellular emission of HA, long polymer HA tangled to net structure that could let it stably wrap at the cell surface. HA net works as a sponge, whereby providing the lubrication and supporting effort between cell–cell interaction [63]. Therefore, the over-expression of HAS2 is more meaningful among multiple biological progressions.
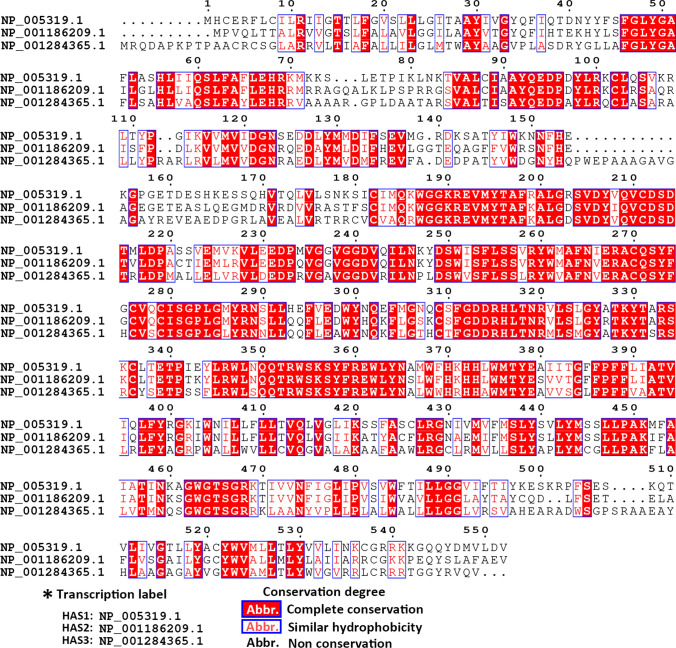
Fig. 2.
The amino acid sequence alignment of three hyaluronan synthases. CLUSTAL X was used for analyzing the multiple alignments of the amino acid sequence of HAS1, HAS2, HAS3. Then the result of alignment was visualized by ESPript 3.0. The transcript label listed on the left was the code from NCBI database of the amino acid sequence of HAS1, HAS2, and HAS3, respectively. The amino acid abbreviation in white color with red background presents the aligned sequences are identical, and words in red color present the aligned sequences have similar hydrophobicity, hydrophilicity or similar side chain. Word in black presents the aligned sequences that are not conservation
Additionally, HAS2 predominates the synthesis of HA in early embryonic development. In the mice model, the absence of HAS1 and HAS3 does not affect the growth or fertility of mice. Importantly, embryonic development is severely damaged in the HAS2 deletion mice model, and the lack of HAS2 reduces cell migration and epithelial-mesenchymal transformation, failing to differentiate mature cells [64]. Aside from generating HA, HAS could act as prognosis indicators in cancer diseases. In the MDA-MB-231-BM cell, high expression of HAS2 promotes invasion, leading to the occurrence of bone metastasis. On the contrary, the transfection of HAS2 siRNA can inhibit the development of breast cancer and the occurrence of metastatic colonization through the EGF-mediated kinase/PI3K/Akt signalling pathway [65]. In contrast, suppression of HAS2 by using antisense-transcript HAS2-AS1 has been shown to attenuate the proliferation and malignant phenotype of glioma, osteosarcoma and breast cancer [66–68]. Furthermore, in the analysis of gene expression profiles of pancreatic cancer cells and fibroblasts, HAS2 was highly expressed as a differential expression gene and was associated with increased invasiveness of pancreatic cancer cells [69]. Similarly, high expression of HAS1 and HAS3 either tumor tissue or tumor stroma predicts poor prognosis or malignant progression in breast cancer, lung cancer, bladder cancer, and also chondrosarcoma [70–73].
HA bio-hydrolysis
Nature balance could not permit infinite HA synthesis. When the threshold of ambient pressure is affected by the increase of HA content, both HAS activity and HA emission will be halted and the switching of HA hydrolysis will be launched. All biological changes require balance, and accumulation of HA in physiological situations accompanied by the acceleration of HA degradation. This process includes the recognition of HA, hyaluronidase involvement and optimal physicochemical property. HYAL 1–5 and PH-20 (coded by SPAM1) were the major enzymes that could degrade HA into fragments [74, 75].
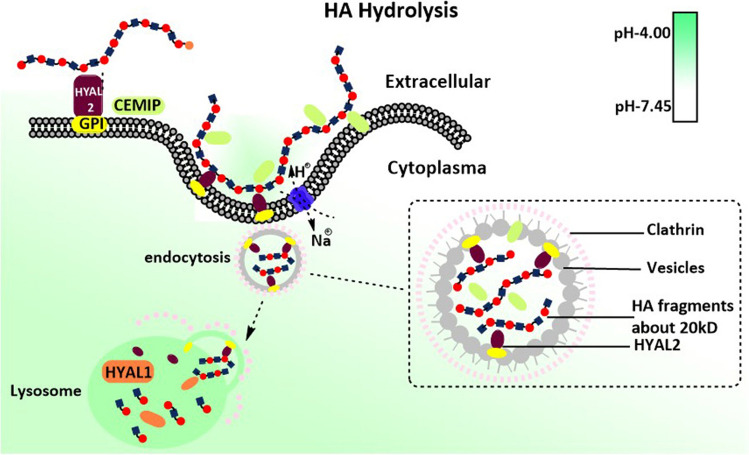
The distribution of HYALs varies in different organs. HYAL1 and HYAL2 are widely detected in many organs except the brain in which HYAL2 has not been detected [76]. HYAL3 and PH-20, on the other hand, are abundant in specific parts [63]. Since HYAL4 shows no degradation activity of HA while HYAL5 did not express in the human body, these two enzymes would not be further discussed in this review. Major functional enzymes HYAL1 and HYAL2 are located at the lysosomes and plasma membrane respectively. To be specific, the 448 glycines of HYAL2 is attached to the glycosylphosphatidyl inositol (GPI) resulting in its membrane location [76]. HA degradation relies on two steps (Fig. 3), the polymers were recognized by HA receptors and HYAL2, covering the outer face of the membrane. And then HA/HYAL2 forms complex which is cupped by the membrane to form caveolae. This invagination process is associated with the activation of Na+-H+ exchange protein that triggered the inflow of H+ [10]. The Na+-H+ exchange and electrochemical flow that come from this ion channel also take responsibility for energy supplement for HA synthesis from the hydrolysis of UDP-sugar bond, instead of directly applying high-energy phosphate bond from ATP or GTP [54]. As a result, the optimal acidic environment is achieved where the pH level is lower than 4, thereby initiating the HA hydrolysis reaction. At this step, the minimum HA fragment remains 20kD regardless of C4 or C3 connective glycosidic linkage, as the HYAL2 endowed both hyaluronan glucuronidase and hyaluronan glucosaminidase activity. When the endocytosis of HA fragments are being carried out with vesicle coating chatinin, this will be delivered to the lysosomes to start the final step of HA degradation [10].
Fig. 3.
The hydrolysis of HA. The two-step process of HA hydrolysis. The first step happens in the ECM, HA is caught by the membrane enzyme HYAL2, which is anchored by GPI and exposed to the external surface. Or like CEMIP, which attached membrane or secreted into ECM. HA and enzymes then form a complex to initiate the endocytosis process. And then, clathrin is recruited at the internal face of invagination at the membrane. During this progression, Na+-H+ ion pump promotes ion exchange for providing energy for endocytosis and adjusting local pH for optimum enzyme activity. HA is digested to LMW-HA (about 20kD) in vesicles, which are encapsulated by clathrin. After being delivered to the lysosome, endocytosis vesicles release content into the lysosome followed by enabling the second step. In the lysosome, HYAL1 take over the final process that cut off LMW-HA into smaller fragments or oligosaccharides. HYAL2 shows no more enzyme activity since the pH in lysosome let it inactivate. These oligosaccharides later may either enter the metabolic pathways or reassemble with UDP and be synthesized to HA again
Lysosomes is the major and final hydrolysis place wherein HYAL1 sustains highly activated Both HYAL1 and HYAL2 require a specific pH environment to gain the best active state. Under the optimum pH, HYAL1 merges endocytosis to digest MMW-HA or intermediates HA to yield HA fragments as small as 6 glycosamine units. In fact, HA with 20 kDa molecular weight does not bind HAYL1 and HYAL2 easily, even under the optimal acidic environment. Besides, HYAL2 has a relatively lower affinity with it, therefore, HYAL1 takes over the rest of the hydrolysis process [35]. Recently, another protein named CEMIP is suggested as a novel hyaluronidase that endowed similar hydrolysis ability as HYAL2, but independently bound to CD44 [10]. CEMIP is encoded by the gene KIAA1199, which is located at chromosome 15q25.1 [77], whereas the other coding gene of HYALs is located at chromosome 3. The location of CEMIP mainly anchors in the rough endoplasmic reticulum, also distributed among nuclear, cytoplasm, membrane and extracellular matrix [77]. The hydrolyzed function of CEMIP depends on the active domain at N-tenimus, where is also the signal domain of cytoplasm secretion, as researchers summarized before [77–79]. No reports provided the direct interaction process between CEMIP and HA, though one of the research groups first proposed that membrane CEMIP could direct connect HA without interacting with CD44 by adding purified recombinant CEMIP into HMW-[3H]HA tracking with 3H label [35]. Many pieces of research indicated the indirect depolymerization of CEMIP on HA by setting up a CEMIP-efficient model in multiple cell lines and CEMIP knock-out transgenic animals [80]. However, the over-expression of CEMIP correlated to HA degradation frequently associated with increased protein amount of HYAL2, this evidence implied the interaction of CEMIP and HYAL2 might be complementary.
Connecting microenvironmental signal to intracellular signalling, HA receptors
As a polysaccharide, HA alone does not modulate cellular biological changes but along with intermediary tools, such as HA receptors to deliver different messages. HA receptors receive various signals and stimulate both inhibitory and oncogenetic signalling depending on HA species. According to the current studies, there are 7 HA receptors, CD44, RHAMM, TLR2, TLR4, LYVE, LAYN and STAB2, which participate in connecting extracellular signal and intracellular signalling in tumor diseases.
HMW-HA/receptors complex system induces tumor suppression effects
For example, in the physiological or precancerous condition, HMW-HA acts as the protective coat surrounding the cell surface, which is recognized by representative HA receptors such as CD44 and RHAMM, forming loose bonds and delivering chemical signals towards hyaluronidase (HYALs) to sustain the stable proliferative rate of normal cells [10, 81]. Even in the pro-metastatic stage, HMW-HA plays major roles in anti-angiogenesis, facilitating anoikis, impeding proliferation, and invasion [20, 40, 82–84]. Sliver et al. reported that HMW-HA inhibited tumor cell uptake of nutrients and angiogenesis through CD44-mediated integrin protein attachment and hydration pathways [85]. The good affinity of HA and CD44 allows them to form a tight HA-CD44 complex with a coat form surrounding membrane. According to this property, remolding intracellular CD44 into soluble CD44 in breast cancer dramatically reduces the binding amount of HMW-HA with CD44 in the cancer cell membrane, blocking HA-mediated intracellular signal transduction, and promoting cancer cell apoptosis, cycle arrest and other phenotypes [86, 87]. In addition, increasing the secretion of soluble CD44 in lung metastases of breast cancer can stimulate the interstitial tissue to produce a large amount of HMW-HA, forming a physical barrier and inhibiting the colonization of metastatic cells [86]. Besides, when HMW-HA binds to LYVE-1, the permeability and integration of lymphatic endothelial cells are broken down in tumor tissues [88, 89].
LMW-HA/receptors complex system facilitates tumor progression
Nevertheless, LMW-HA could also bind to the receptors such as CD44 and LYVE-1 but exhibited the opposite effects [90]. LMW-HA induces the secretion of inflammatory factors, matrix hydrolases and pro-angiogenic factors by activating the specific receptors such as TLR2, TLR4, and LAYN of tumor cells, which dominates in the field of metastasis and colonization [82, 91]. After LMW-HA binds to LAYN, inflammatory infiltration in the microenvironment is attribute to the activation of NF-κB signalling under the over-production of MMP-1 and MMP-13 [92]. The binding of LMW-HA to the receptor LYVE-1 activates the transcriptional activity of the β-catenin protein, destroying the integrity of the lymphatic endothelium and promoting lymphatic metastasis in melanoma [93]. Among many kinds of LMW-HA, LMW-HA with the molecular weight of less than 10 kDa is more likely to bind to TLR2. Scheibner et al. found that in injured lung tissue, when TLR2 recognizes LMW-HA, its intracellular MYD88 and TRAF-6-dependent signalling pathways are activated, resulting in the promotion of the migration of lung cells to finish damaged repairs [94]. In addition, genetic down-regulating TLR4\TLR2 expression inhibits M1 macrophage phenotype induced by LMW-HA and the stimulation of TLR4/MYD88 after LMW-HA activation generated more inflammatory factors PEG2 in macrophage cells [90]. Capturing and directing immune cell migration attributed to the formation of receptors-HA-receptors (CD44 and LYVE-1) sandwiches. Immune cells such as dendritic cells (DCs), whose membrane anchored CD44 with high density, are wrapped by LMW-HA forming a CD44/HA coated surface. When these coated cells travel in vessel system, LYVE-1 in the outer layer of the lymphatic vessel seizes the HA shell which firstly attaches to the membrane of ECs followed by either passing through the gap junction between cells or accessing the lumen of vessels [89, 95].
As for the MMW-HA, this segment was regarded as transient intermediates, they either undergo re-uptake by synthases for chain elongation or be invaginated by membrane HYALs for lysosome hydrolysis [96]. When HYALs decomposed the small HA fragment, the oligosaccharide is drawn into the glycol-metabolism cycle and reassembled to synthetic substrates. Due to the situation of transition and pathologic homeostasis, HA fragment with a wide range of molecular weight co-existed in ECM along with disease progression.
Genetic susceptibility of HA-enzymes
Tumourigenesis attributes to various genetic mutations, one of the important mutation types is the deletion of the coding region in chromosomes which is highly related to the occurrence of tumor [97]. Apart from all types of tumors, there is a major susceptible deletion of hyaluronidases whose coding sequence located in chromosome 3p21.3 across ethnics and countries that contributed to lung cancer [97–102]. Major mutations of various 3p regions found in many cohorts of LC were the deletion of which the sequence of hyaluronidase (HYAL) 1 2 and 3 was located [103–105]. HYAL1 and 2 participate in the separation part of HA degradation, whereas HYAL3 barely exerts hydrolase function but sustains the pluripotency of stem cells [106]. Compared to HYAL3 which contains a highly conserved sequence, large-scale SNP cohorts indicated that mutation happens more often in HYAL1 and HYAL2 [107, 108]. The other mutation is due to the single nucleotide polymorphism which resulted in the change of coding amino acid, leading to loss of hydrolase activity. Other than ‘have or not’ regulation in DNA level, quantity regulation in transcription and protein levels are also crucial. A great deal of research in the past few years has focused on the post-translational modification and epigenetic control of the HASes enzymes. The concentration of HA is related to distortions, while the number and size of HA are related to the alternating expression and activity of HASes and HYALs [109]. HASes deficiency is an autosomal recessive genetic disorder [110].
Pre-metastatic barometer in the tumor microenvironment
Being a prognosis indicator in clinical application
Despite that healthy homeostasis requires enough HMW-HA level, LMW-HA maintained at a high level further enables inflammatory attack and it was also maintained in tumor microenvironment turning up either in peri-tumoral stroma or intra-tumor [111–113]. Evidence showed that enrichment of stroma HA is prevalent among many types of tumors, including bladder, ovarian, breast, prostate, pancreatic, colon and lung cancer, indicating a high possibility of tumor invasion and metastasis [9, 114–118]. This phenomenon mostly accorded to poor survival and advanced tumor stage thereby high HA status becoming a poor prognostic factor for tumors. In the research in the field of ovarian cancer, the stromal HA level positively correlates to advanced progression since the metastatic sites have higher HA filtration [119]. In addition, higher stromal HA indicates a patient with an aggressive stage and a bad outcome in breast cancer. Moreover, high intra-stroma HA levels are also correlated to deep filtration and distant metastasis in colon cancer [120]. Similarly, high HA level could not only be detected in the stroma of prostate cancer, but also the intra-tumor accumulation [121]. This is due to influx uptake or self-generation through tumor cells. High levels of serum LMW-HA are positively correlated with breast cancer lymph node metastasis and the invasive potential of breast cancer cells, whereas reducing LMW-HA production can significantly inhibit the migration and invasion of breast cancer cells[46]. For example, LMW-HA can stimulate pancreatic cancer cell motility [122, 123].
If HA acted as a protective barrier of normal tissues, why is its accumulation surrounding the tumor microenvironment reflected to poor prognosis? This controversy might attribute to the application of specific HA binding antibody which is used as quantitative analysis rather than distinguishing the actual molecule weights of HA. Despite the high amount of HA accumulates in both normal and tumor tissues, the opposite effect might be due to the different molecule weight of HA. The HA recognition mainly depends on the length of N-terminal peptides, thereby the existence of HA with more than 2kD could be linked tidily. However, different molecular-weight-HA have specific roles in tumourigenesis. Once the vHMW-HA is broken, fgHA exerts oncogenetic promotion in aspect of tumor metastasis and metabolism [74]. The fgHA ranging from 20 to 200kD predominately functioned as an angiogenic modulator that stimulated endothelial cells migrated, preparing a supply network for tumor metastasis [124]. HA with 200-400kD molecular weight might be the hydrolysis intermediates, which are associated with tumor cell migration and anoikis resistance, although these intermediates would later be delivered into lysosome digestion. The other feature of fgHA (below 20kD) would be interrupting the interaction of HWM-HA-HA receptor–binding instead of fgHA-HA receptor-binding, followed by triggering cleavage of HA receptors [125]. The accumulation of HA intermediates elevated the average molecule weight of HA in microenvironment, so that interrupting the HA homeostasis by accelerating the synthesis of HMW-HA as well as the rate of HYAL1-degradation. These phenomena might explain why the observation of high HA level in different situated microenvironment displayed both oncogenic and suppressive functions.
Being a regulator in EMT and wound healing progression
Epithelial-mesenchymal transition (EMT) marks one of the metastatic-driven factors and adaptive change. Since detaching from ECM and cell–cell connection triggers the programmed cell death of epithelial cells, the first step of metastatic tumor cells is activating the EMT process [126]. Due to the tight interaction of cells and ECM, HA also participates in the modulation of EMT in tumor cells. Taking breast cancer as an instance, at the primary lesion, pre-metastatic tumor cells undergo interstitial transition by increasing the expression of CD44S isoform, to which HA specifically binds to protect cells from anoikis [127]. HA also acts as an ECM signal to induce the nuclear translocation of CD44, thereby over-expressing lysyl oxidase through activating its transcriptional promoter. The increased expression of lysyl oxidase then facilitates Twist1 to finish the EMT progression [128]. In addition, Excessive production of HA by HAS2 induces over-expression of TGF-β which activates transcriptional factors Snail and Twist to initiate the EMT process in breast cancer cells. This transition progression lets those cancer cells regain stemness and into a higher malignancy [129]. LMW-HA (< 30kD) could increase the mRNA expression of a mesenchymal marker, Snail2 and Vimentin, while the MMW-HA(> 200kD) obviously increased the expression of the epithelial marker, E-cadherin [130].
HA also serves as a wound healing regulator in non-tumor diseases. Treatment of complications in Diabetics with HMW-HA hydrogel effectively reduces inflammation by recruiting M2 macrophages and mobilizing the M2-transition of M1 macrophages to the wound site [131]. Many hydrogels for treating skin wound healing contain HA, which needs to be modified or crosslinked to gelate before medication application [132]. In a German report on the treatment of chronic wounds, wound dressings containing HA exhibit a better cost-effective tendency considering the ratio of cure proportion to cost input. [133]. In summary, the promotion of wound healing in tissues relies on the increased swelling and elasticoviscosity property of crosslinked HA hydrogel, wherein HA works as a structural frame. However, HA plays a signal role to facilitate the EMT progression of tumor cells in the tumor microenvironment, wherein HA binds its receptor first and then activates the downstream triggers.
Being a regulator in fuel recycling and supply
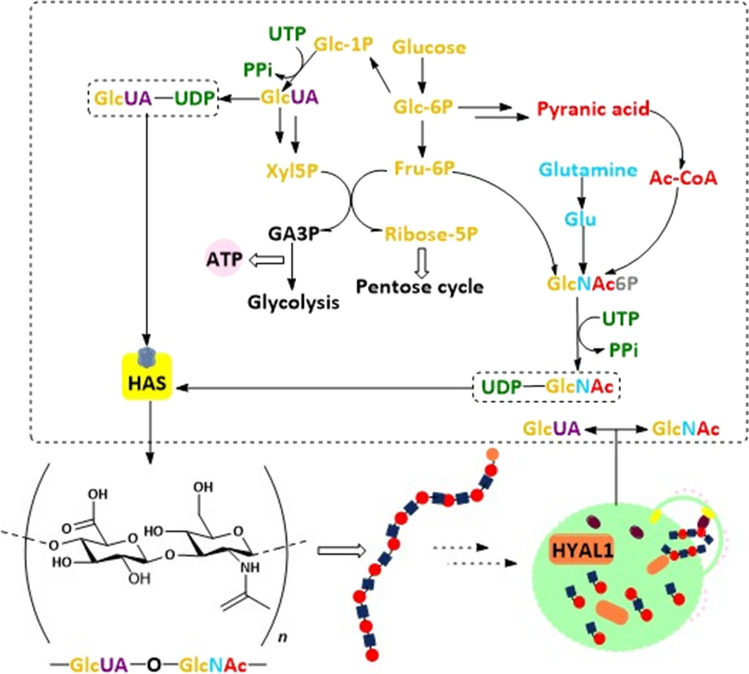
The catabolism of HA reprograms energy supply since it takes part in both consuming and generating energy through multiple metabolism pathways (Fig. 4). In human placenta-derived mesenchymal stem cells, HA at around 1 μg/mL acts as an inducer that switches on mitochondria function to enhance the glycolysis progression with about twice the amount of ATP and lactate production [134]. Radioactive element 14C was used to label HA, oral or intravenous administration in male SD rats. Ninety percent of exogenous intake HA was slowly absorbed from the digestive tract into circulation and then move to organs as part of the energy or took part in constituting to organ tissues. HA with labeling 14C was only found in the skin of the rats, while a large amount of 14C was detected in mice excrement [135].
Fig. 4.
The energy metabolism of HA. The origins and decomposition of 2 substrates, GlcNAc and GlcUA, respectively. The ingredients consist of GlcNAc come glucose, glutamine and pyranic acid, and then UTP provides energy and attaches to GlcNAc. The glycosyl structure in GlcUA also comes from glucose as GlcNAc. In addition, GlcUA could also generate energy by entering glycolysis and the pentose cycle. The residues with different color track their source. The lower left displays a single unit of HA with a disaccharide structure. The lower right briefly flashback the final step of hydrolysis in lysosome where recycles GlcNAc and GlcUA to the cytoplasm
Since the three major materials of GlcNAc are from the products of the hexosamine pathway, which connects carbohydrate, glutamine and nucleotide metabolism together, GlcNAc not only acts as a fundamental substrate of HA, but also a ‘sensor’ of energy availability in tumor cells [136]. Besides, GlcNAc takes over the glycosylation modification of protein, which draws much attention to the regulation of tumor phenotype [137]. Before GlcNAc or GlcUA consumes UTP to UDP-sugar, these two oligosaccharides enter the redox reactions associated with hexokinase and glucuronate reductase, respectively [112, 138]. Despite that tumor cells endow the ‘Warburg effect’, a different metabolism programming, rather than normal cells, degradation of HA is also joined to glycolysis and pentose pathway [138]. Once the energy supply becomes deficient, the “starved” tumor cells activate the secretion of the MMPs family, which cut the subtypes of collagen into pieces, so that they could be swallowed through pinocytosis. Following a similar pathway as collagen, the tumor ‘swallows’ HA or its fragment by endocytosis for energy replenishment [139–141]. The way endocytosis by cells relies on surface receptors of HA such as CD44, LYVE-1, HARE and RHAMM which can trigger HA uptake through self-assembling or recruiting clathrin-coated pits [35, 112]. This phenomenon was also observed in endocardial endothelial cells, wherein HA endocytosis works as high-energy metabolites to fuel cells [142]. Recently, a researcher reported that an increase of GlcNAc from HA cleavage remarkably ensures the energy supply in growth of pancreatic cancer cells [143].
Other evidence indirectly points out that the change of HA content or concentration modulated by alteration of HASes or HYALs also affects the energy supply. Glycolysis activity significantly increases in cells or xenografts when treated with hyaluronidase. It is mainly via ZFP36, the mRNA decay factor, which targets TXNIP transcripts to increase the internalization of the glucose transporter 1 (GLUT1) [144]. In triple-negative breast cancer, the expression of various enzymes involved in the glucose and glutamine metabolism showed obviously changed in the Hs578T subgroup which carries endogenous highHA [145]. Also, the depletion of UDP-sugars dramatically inhibits the EMT progression, probably through reprogramming glycometabolism in breast cancer cells [146]. So far, there are limited studies on whether HA could be taken up by mitochondria as a substrate fuel, but ongoing insight has suggested HA metabolism might be a new bypass energy cycle in tumor disease.
Being a regulator in angiogenesis balance
In a normal microenvironment, HMW-HA takes the major proportion which exerts anti-angiogenesis function. Because of the high hydration, HMW-HA is maintained in the interstitial space between endotheliocyte (ECs) [147]. Crowding these HMW-HA increased the permeation pressure forming physical barriers that restricted tumor invadesome entered circulation [148]. This physical barrier alternatively resisted inflammation infiltration and suppressed immune reaction [83]. Treating HMW-HA in cells causes the decreased expression of COX, and minimizes ROS through self-sacrificing electrons from glucosidic bonds [149]. Recently, a research team finds out that HMW-HA exclusively facilitates the angiogenic action of breast cancer by modeling its immune microenvironment. HMW-HA promotes the migration of ECs by stimulating monocyte/macrophagocyte to secret an increased amount of angiogenic factors, although a similar action is not observed in other cancers [150, 151] The infiltration and migration of monocyte/macrophagocyte are otherwise inhibited by increased concentration of HMW-HA in spinal nerves, maybe because HMW-HA competitive binds to receptors which block the activation of pro-inflammatory LMW-HA/receptor [152]. Based on current evidence, HMW-HA exerts anti-angiogenesis regulation among a majority of tumors, but still exhibits angiogenic action exclusively to specific one.
Contrarily, small HA fragments (~ 20 kDa) can stimulate neo-vascularization and promote tumor cell motility and invasion [153]. LMW-HA and fgHA stimulated angiogenesis in stimulating the growth and migration of ECs under hyper-inflammation [154]. On the surface of ECs, inflammatory signalling receptor TLR was triggered followed by emission of IL-6 and pro-inflammation factors IL-1β thereby boosting vasculum sprout [155]. Moreover, the accumulation of fgHA enhanced stromal VEGF levels, which specifically fostered vessel formation through binding to VEGFR of ECs [156, 157]. Besides, membrane receptor CD44 would transmit signals to NF-κB and TGF-βsignalling pathways [158, 159]. Once the LMW-HA binds to CD44, it would trigger the proliferation of endothelial cells through activating MAPK/SRC cascades [89, 160]. Another vital remodeling factor metalloprotease (MMPs) was also under HA regulation. High fgHA level promoted expression of MMPs such as MMP-13 and MMP9. When fgHA formed covalent bonds to HA receptors, the HMW-HA lost the possibility to compete for this target [161]. In summary, HA stimulated angiogenesis basically relied on indirectly affecting the proliferation and migration pathway of ECs. Different angiogenesis functions might be due to the chemical structure, since fgHA has centralized electron density that binds tightly to a single pocket of the receptor, while HMW-HA with nomadic polymer that formed multiple targeted sites of the receptor, thereby launching an opposite signalling pathway.
Being a regulator in stiffness of cells and ECM
The change of HA either in content or molecular weight affects stiffness in a reciprocal way under the specific condition. On the one hand, accumulation of HA (HMW-HA and fgHA) assists collagen re-polymerization and secretion of integrin to increase ECM stiffness reaching 2–20 fold higher [162, 163]. Under this condition, the HA scaffold becomes denser by the bridge of integrin, and collagen filling enhances the structure. In cancer stroma, an increase of ECM stiffness leads to the formation of the porous net in tumor microenvironment [164]. Kaukonen R et al. reported the growth and invasion of breast tumor cells were inhibited in the normal stroma matrix while the inhibition was relieved under the tumor mimic matrix, which was stiffer than its counterpart [165]. Rising ECM stiffness fostered invasion and migration of breast cancer cells through translocating TWIST1 into nuclear, thereby promoting cellular transcriptional activity [166]. On the other hand, the replacement of glycosaminoglycans by HMW-HA without an additional cross-linker did not change the stiffness of ECM in glioma [167, 168].
Furthermore, the presence of HA in the medium could up-regulate intracellular stiffness, which increases cell movement and proliferation. These biological changes are similar to that of the tumors cultivated in stiffen matrix [167]. Higher HMW-HA density without cross-linker cause cellular stiffness instead of microenvironment stiffness might be due to the encapsulating membrane HA led to over-activate HA receptors. Due to the excellent biocompatibility and reciprocal plasticity, HA recently was utilized for constructing 3D scaffolds with varying stiffness to simulate tumor microenvironment [169]. They followed the principle of augmenting stiffness through increasing HA content and cross-link reagents to adjust suitable porous channels which approached to real microenvironment as much as possible.
Being biomimetic materials in medical application
Recently, the application of HA coating capsules is beneficial for modifying drug delivery systems. In pre-clinical research, HA-coated sh-vector presented a better targeting ability in nude mice growing subcutaneous tumors. Research demonstrated that HMW-HA is a safe material for chemotherapy. A phase I report showed that intravenous injection of HMW-HA remained no harm and did not interfere with pharmacokinetics when combined with 5-fluorouracil and doxorubicin [170]. A phase IIa study illustrated using irinotecan enclosed by HA could extend the mean survival time and improve the tolerance in small cell lung cancer (SCLC) patients in extensive stages who carry CD44 positive [171]. Also, HA-coated irinotecan could prolong mean survival time and slightly extend the treatment response duration in metastatic colorectal cancer patients, compared with traditional 5-fluorouracil treatment [172]. The latest report indicates that based on the high affinity binding property of HA and CD44, the Fe3O4 nanocubes enclosed a small molecule inhibitor LDN193189 can be successfully delivered to HCC stem cells thus attenuating cellular proliferation, migration and EMT process [173]. The reason of HA coating system might be due to the increase in the potential for lymphotropic targeting to inhibit metastasis [174], as well as the high affinity towards HA receptors. The other reason might be good biocompatibility and safety of HA make it an ideal material to develop novel drug delivery systems.
Another prevalent application of HA is building hydrogel as a 3D scaffold to mimic the exact microenvironment. HA hydrogels containing a mixture of hyaluronan, collagen and matrigel could provide a similar ECM stiffness as mimicking the in vivo elasticity modulus of brain condition, which is further applying a better model for evaluating the malignancy of glioma [168]. With additional linkage reagents and other components of ECM, the model could repeat the ideal matrix for cultivating cells [175]. In chitosan-hyaluronan membrane-derived 3D culture models, the stemness phenotype is largely amplified as rapidly growing to tumor spheroids, increasing invasion and interstitialization. This application reproduces the in vivo growth and progression of tumor cells to a great extent [176].
However, one problem would be: no replenishment of HA normally generated by stromal cells to sustain dynamic balance in the model. Except that, the synthesis and hydrolysis of HA seesaw microenvironment, not only chemical components and biological progression had changed, but also the physical properties such as transit pH value, osmotic pressure, stiffness, and viscosity remained constantly fluctuated. Further modification could also concern adding a sponge system obsessing good mechano- or chemo- sensitivity into the HA hydrogel model so that the HA with specific molecule weight could be constantly pumped out when the microenvironment changes. Besides, HA hydrogels with unilateral cell strainer might achieve co-culture tumor cells with stromal cells, and easily re-built functional experiments of both tumor and stromal cells thereafter.
Challenges and perspectives of HA application in the future
Despite the simple structure and components of naïve HA, its metabolism might be one of the most intricate bio-process. HA homeostasis displays the perfect interpretation of natural harmony that survival of the fittest only could help the organism own self-regulation, as many cells contained both HAS and HYALs. Additionally, HA metabolism structured a cycle whereby remodeling ECM matrix and energy supplement. However, more studies are required to understand the detailed proportion of HA polymers with different lengths, which might have great potential for therapeutic intention. If the ratio of HMW-HA to fgHA could be locked in the high value or occlude unresectable tumor vessel with undigestible HA coat, the tumor cells might be limited at in situ position, as well as lose the extracellular signal for invasive and distant migrated competence. Indeed, detecting serum concentration and a precise fragment of HA would provide a more meaningful diagnostic index, therapeutic modification, and prognosis predictor in certain diseases. However, due to the faster dynamic turnover and short half-life of HA in the circulation system, several challenges ought to be solved: how could researchers precisely capture circulated HA? When would be the best cut-off time for evaluating the activity of HA catabolism? And would it be possible to discover a safe tracer that could monitor HA turnover in vivo track?
In summary, due to the different molecular weights of HA components, HA shows dual characteristics in regulating tumor microenvironment. HA in ECM which is composed of HMW-HA, LMW-HA and fgHA, is attributed to the different catabolism yield of HA under certain circumstances. The ratio of the HA mixture does not stay invariant the same. In physiological condition, HMW-HA takes the biggest proportion while the LMW-HA becomes the major component in tumor ECM. HA mixture participates in the dynamic variation of the tumor microenvironment including conversion of the extracellular signal to intracellular signalling cascades, energy generation and consumption and remodeling microenvironment mechanical and biological properties, along with tumor progression. Additionally, the simple structure and good biocompatibility of HA allow for the industrial pipelining process and development of biomimetic materials application although there still are challenges present. From accurately tracing the origin and decomposition of circulated HA in vivo to precisely understanding the metabolism and regulation pathway of HA at a cellular level, cracking these challenges might solve the current dilemma in the knowledge of HA.
Acknowledgements
We express our gratitude to Dr Jocelyn May Fen Ooi, who provide countless help in modification of this paper.
Author contributions
Liu Ying and Li Li wrote the major parts, Wang Li modified and rephrased some parts. Lu Lu and Li Ying collected the researches and modified the paper, Guolin Huang help syntax check, Jinjing Song managed the reference and Li Li designed and supervised the work, and reviewed the manuscript. All authors reviewed the manuscript. All authors read and approved the final mnuscript.
Funding
This work was supported by the Natural Science Foundation Program of Guangxi. Youth Project (No.2022GXNSFBA035500), Independent research project of Guangxi Institute of Chinese Medicine & Pharmaceutical Science (No. GZZY-2021003), Research project of Guangxi Orthopaedics and Traumatology Hospital (No.GSZD2022002) and the Guangxi Traditional Chinese Medicine Key Discipline Construction Project (GZXK-Z-20–75).
Data availability
Not applicable.
Declarations
Ethics approval and consent to participate
Not applicable.
Informed consent
Not applicable.
Competing interests
We declare there are no conflict of interest existed in this paper.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Ying Liu and Li Li are co-authors equally contributed to this paper
References
- 1.Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68:394–424. doi: 10.3322/caac.21492. [DOI] [PubMed] [Google Scholar]
- 2.Chen W, Zheng R, Baade PD, Zhang S, Zeng H, Bray F, Jemal A, Yu XQ, He J. Cancer statistics in China, 2015. CA Cancer J Clin. 2016;66:115–132. doi: 10.3322/caac.21338. [DOI] [PubMed] [Google Scholar]
- 3.Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A. Global cancer statistics, 2012. CA Cancer J Clin. 2015;65:87–108. doi: 10.3322/caac.21262. [DOI] [PubMed] [Google Scholar]
- 4.Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D. Global cancer statistics. CA Cancer J Clin. 2011;61:69–90. doi: 10.3322/caac.20107. [DOI] [PubMed] [Google Scholar]
- 5.Mendoza M, Khanna C. Revisiting the seed and soil in cancer metastasis. Int J Biochem Cell Biol. 2009;41:1452–1462. doi: 10.1016/j.biocel.2009.01.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Paget S. The distribution of secondary growths in cancer of the breast. 1889. Cancer Metastasis Rev. 1989;8:98–101. [PubMed] [Google Scholar]
- 7.Liu Q, Zhang H, Jiang X, Qian C, Liu Z, Luo D. Factors involved in cancer metastasis: a better understanding to "seed and soil" hypothesis. Mol Cancer. 2017;16:176. doi: 10.1186/s12943-017-0742-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Gilkes DM, Semenza GL, Wirtz D. Hypoxia and the extracellular matrix: drivers of tumour metastasis. Nat Rev Cancer. 2014;14:430–439. doi: 10.1038/nrc3726. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Toole BP. Hyaluronan: from extracellular glue to pericellular cue. Nat Rev Cancer. 2004;4:528–539. doi: 10.1038/nrc1391. [DOI] [PubMed] [Google Scholar]
- 10.Chanmee T, Ontong P, Itano N. Hyaluronan: a modulator of the tumor microenvironment. Cancer Lett. 2016;375:20–30. doi: 10.1016/j.canlet.2016.02.031. [DOI] [PubMed] [Google Scholar]
- 11.Becker JC, Andersen MH, Schrama D, Thor SP. Immune-suppressive properties of the tumor microenvironment. Cancer Immunol Immunother. 2013;62:1137–1148. doi: 10.1007/s00262-013-1434-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Garantziotis S, Savani RC. Hyaluronan biology: a complex balancing act of structure, function, location and context. Matrix Biol. 2019;78–79:1–10. doi: 10.1016/j.matbio.2019.02.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Tavianatou AG, Caon I, Franchi M, Piperigkou Z, Galesso D, Karamanos NK. Hyaluronan: molecular size-dependent signaling and biological functions in inflammation and cancer. FEBS J. 2019;286:2883–2908. doi: 10.1111/febs.14777. [DOI] [PubMed] [Google Scholar]
- 14.Saito T, Kawana H, Azuma K, Toyoda A, Fujita H, Kitagawa M, Harigaya K. Fragmented hyaluronan is an autocrine chemokinetic motility factor supported by the HAS2-HYAL2/CD44 system on the plasma membrane. Int J Oncol. 2011;39:1311–1320. doi: 10.3892/ijo.2011.1114. [DOI] [PubMed] [Google Scholar]
- 15.Sugahara KN, Murai T, Nishinakamura H, Kawashima H, Saya H, Miyasaka M. Hyaluronan oligosaccharides induce CD44 cleavage and promote cell migration in CD44-expressing tumor cells. J Biol Chem. 2003;278:32259–32265. doi: 10.1074/jbc.M300347200. [DOI] [PubMed] [Google Scholar]
- 16.Cowman MK. Hyaluronan and hyaluronan fragments. Adv Carbohydr Chem Biochem. 2017;74:1–59. doi: 10.1016/bs.accb.2017.10.001. [DOI] [PubMed] [Google Scholar]
- 17.Abbruzzese F, Basoli F, Costantini M, Giannitelli SM, Gori M, Mozetic P, Rainer A, Trombetta M. Hyaluronan: an overview. J Biol Regul Homeost Agents. 2017;31:9–22. [PubMed] [Google Scholar]
- 18.Joy RA, Vikkath N, Ariyannur PS. Metabolism and mechanisms of action of hyaluronan in human biology. Drug Metab Pers Ther. 2018;33:15–32. doi: 10.1515/dmpt-2017-0031. [DOI] [PubMed] [Google Scholar]
- 19.Monslow J, Govindaraju P, Pure E. Hyaluronan—a functional and structural sweet spot in the tissue microenvironment. Front Immunol. 2015;6:231. doi: 10.3389/fimmu.2015.00231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Schmaus A, Bauer J, Sleeman JP. Sugars in the microenvironment: the sticky problem of HA turnover in tumors. Cancer Metastasis Rev. 2014;33:1059–1079. doi: 10.1007/s10555-014-9532-2. [DOI] [PubMed] [Google Scholar]
- 21.Pitsillides AA, Will RK, Bayliss MT, Edwards JC. Circulating and synovial fluid hyaluronan levels. Effects of intraarticular corticosteroid on the concentration and the rate of turnover. Arthritis Rheum. 1994;37:1030–1038. doi: 10.1002/art.1780370708. [DOI] [PubMed] [Google Scholar]
- 22.Pitsillides AA, Worrall JG, Wilkinson LS, Bayliss MT, Edwards JC. Hyaluronan concentration in non-inflamed and rheumatoid synovium. Br J Rheumatol. 1994;33:5–10. doi: 10.1093/rheumatology/33.1.5. [DOI] [PubMed] [Google Scholar]
- 23.Fraser JR, Laurent TC, Laurent UB. Hyaluronan: its nature, distribution, functions and turnover. J Intern Med. 1997;242:27–33. doi: 10.1046/j.1365-2796.1997.00170.x. [DOI] [PubMed] [Google Scholar]
- 24.Laurent UB, Laurent TC, Hellsing LK, Persson L, Hartman M, Lilja K. Hyaluronan in human cerebrospinal fluid. Acta Neurol Scand. 1996;94:194–206. doi: 10.1111/j.1600-0404.1996.tb07052.x. [DOI] [PubMed] [Google Scholar]
- 25.Meyer LJ, Stern R. Age-dependent changes of hyaluronan in human skin. J Invest Dermatol. 1994;102:385–389. doi: 10.1111/1523-1747.ep12371800. [DOI] [PubMed] [Google Scholar]
- 26.Bullard KM, Kim HR, Wheeler MA, Wilson CM, Neudauer CL, Simpson MA, McCarthy JB. Hyaluronan synthase-3 is upregulated in metastatic colon carcinoma cells and manipulation of expression alters matrix retention and cellular growth. Int J Cancer. 2003;107:739–746. doi: 10.1002/ijc.11475. [DOI] [PubMed] [Google Scholar]
- 27.Tengblad A, Laurent UB, Lilja K, Cahill RN, Engström-Laurent A, Fraser JR, Hansson HE, Laurent TC. Concentration and relative molecular mass of hyaluronate in lymph and blood. Biochem J. 1986;236:521–525. doi: 10.1042/bj2360521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Laurent TC. Biochemistry of hyaluronan. Acta oto-laryngologica Suppl. 1987;442:7–24. doi: 10.3109/00016488709102833. [DOI] [PubMed] [Google Scholar]
- 29.Engström-Laurent A, Laurent UB, Lilja K, Laurent TC. Concentration of sodium hyaluronate in serum. Scand J Clin Lab Invest. 1985;45:497–504. doi: 10.3109/00365518509155249. [DOI] [PubMed] [Google Scholar]
- 30.Engström-Laurent A, Lööf L, Nyberg A, Schröder T. Increased serum levels of hyaluronate in liver disease. Hepatology. 1985;5:638–642. doi: 10.1002/hep.1840050420. [DOI] [PubMed] [Google Scholar]
- 31.Lichtinghagen R, Pietsch D, Bantel H, Manns MP, Brand K, Bahr MJ. The enhanced liver fibrosis (ELF) score: normal values, influence factors and proposed cut-off values. J Hepatol. 2013;59:236–242. doi: 10.1016/j.jhep.2013.03.016. [DOI] [PubMed] [Google Scholar]
- 32.Gudowska M, Cylwik B, Chrostek L. The role of serum hyaluronic acid determination in the diagnosis of liver fibrosis. Acta Biochim Pol. 2017;64:451–457. doi: 10.18388/abp.2016_1443. [DOI] [PubMed] [Google Scholar]
- 33.Tangkijvanich P, Kongtawelert P, Pothacharoen P, Mahachai V, Suwangool P, Poovorawan Y. Serum hyaluronan: a marker of liver fibrosis in patients with chronic liver disease. Asian Pac J Allergy Immunol. 2003;21:115–120. [PubMed] [Google Scholar]
- 34.Yang YM, Noureddin M, Liu C, Ohashi K, Kim SY, Ramnath D, Powell EE, Sweet MJ, Roh YS, Hsin IF, et al. Hyaluronan synthase 2-mediated hyaluronan production mediates notch1 activation and liver fibrosis. Sci Transl Med. 2019;11:9284. doi: 10.1126/scitranslmed.aat9284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Yoshida H, Nagaoka A, Kusaka-Kikushima A, Tobiishi M, Kawabata K, Sayo T, Sakai S, Sugiyama Y, Enomoto H, Okada Y, et al. KIAA1199, a deafness gene of unknown function, is a new hyaluronan binding protein involved in hyaluronan depolymerization. Proc Natl Acad Sci USA. 2013;110:5612–5617. doi: 10.1073/pnas.1215432110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Fusco S, Borzacchiello A, Miccio L, Pesce G, Rusciano G, Sasso A, Netti PA. High frequency viscoelastic behaviour of low molecular weight hyaluronic acid water solutions. Biorheology. 2007;44:403–418. [PubMed] [Google Scholar]
- 37.Fallacara A, Manfredini S, Durini E, Vertuani S. Hyaluronic acid fillers in soft tissue regeneration. Facial Plastic Surg. 2017;33:87–96. doi: 10.1055/s-0036-1597685. [DOI] [PubMed] [Google Scholar]
- 38.Day AJ, Milner CM. TSG-6: A multifunctional protein with anti-inflammatory and tissue-protective properties. Matrix Biol. 2019;78–79:60–83. doi: 10.1016/j.matbio.2018.01.011. [DOI] [PubMed] [Google Scholar]
- 39.Baranova NS, Inforzato A, Briggs DC, Tilakaratna V, Enghild JJ, Thakar D, Milner CM, Day AJ, Richter RP. Incorporation of pentraxin 3 into hyaluronan matrices is tightly regulated and promotes matrix cross-linking. J Biol Chem. 2014;289:30481–30498. doi: 10.1074/jbc.M114.568154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Gorbunova V, Takasugi M, Seluanov A. Hyaluronan goes to great length. Cell stress. 2020;4:227–229. doi: 10.15698/cst2020.09.231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Bukhari SNA, Roswandi NL, Waqas M, Habib H, Hussain F, Khan S, Sohail M, Ramli NA, Thu HE, Hussain Z. Hyaluronic acid, a promising skin rejuvenating biomedicine: a review of recent updates and pre-clinical and clinical investigations on cosmetic and nutricosmetic effects. Int J Biol Macromol. 2018;120:1682–1695. doi: 10.1016/j.ijbiomac.2018.09.188. [DOI] [PubMed] [Google Scholar]
- 42.Miyazaki K, Hanamizu T, Iizuka R, Chiba K. Bifidobacterium-fermented soy milk extract stimulates hyaluronic acid production in human skin cells and hairless mouse skin. Skin Pharmacol Appl Skin Physiol. 2003;16:108–116. doi: 10.1159/000069031. [DOI] [PubMed] [Google Scholar]
- 43.Shinohara T, Izawa T, Mino-Oka A, Mori H, Iwasa A, Inubushi T, Yamaguchi Y, Tanaka E. Hyaluronan metabolism in overloaded temporomandibular joint. J Oral Rehabil. 2016;43:921–928. doi: 10.1111/joor.12443. [DOI] [PubMed] [Google Scholar]
- 44.Kim WK, Choi JH, Shin ME, Kim JW, Kim PY, Kim N, Song JE, Khang G. Evaluation of cartilage regeneration of chondrocyte encapsulated gellan gum-based hyaluronic acid blended hydrogel. Int J Biol Macromol. 2019;141:51–59. doi: 10.1016/j.ijbiomac.2019.08.176. [DOI] [PubMed] [Google Scholar]
- 45.Kavasi RM, Berdiaki A, Spyridaki I, Corsini E, Tsatsakis A, Tzanakakis G, Nikitovic D. HA metabolism in skin homeostasis and inflammatory disease. Food Chem Toxicol. 2017;101:128–138. doi: 10.1016/j.fct.2017.01.012. [DOI] [PubMed] [Google Scholar]
- 46.Wu M, Cao M, He Y, Liu Y, Yang C, Du Y, Wang W, Gao F. A novel role of low molecular weight hyaluronan in breast cancer metastasis. FASEB J. 2015;29:1290–1298. doi: 10.1096/fj.14-259978. [DOI] [PubMed] [Google Scholar]
- 47.Beretta E, Romanò F, Sancini G, Grotberg JB, Nieman GF, Miserocchi G. Pulmonary interstitial matrix and lung fluid balance from normal to the acutely injured lung. Front Physiol. 2021;12:781874. doi: 10.3389/fphys.2021.781874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Nikitovic D, Tzardi M, Berdiaki A, Tsatsakis A, Tzanakakis GN. Cancer microenvironment and inflammation: role of hyaluronan. Front Immunol. 2015;6:169. doi: 10.3389/fimmu.2015.00169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Hamblin MJ, Eberlein M, Black K, Hallowell R, Collins S, Chan-Li Y, Horton MR. Lovastatin inhibits low molecular weight hyaluronan induced chemokine expression via LFA-1 and decreases bleomycin-induced pulmonary fibrosis. Int J Biomed Sci. 2014;10:146–157. [PMC free article] [PubMed] [Google Scholar]
- 50.Siódmiak J, Bełdowski P, Augé WK, Ledziński D, Śmigiel S, Gadomski A. Molecular dynamic analysis of hyaluronic acid and phospholipid interaction in tribological surgical adjuvant design for osteoarthritis. Molecules. 2017;22:1436. doi: 10.3390/molecules22091436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Zhang Z, Tao D, Zhang P, Liu X, Zhang Y, Cheng J, Yuan H, Liu L, Jiang H. Hyaluronan synthase 2 expressed by cancer-associated fibroblasts promotes oral cancer invasion. J Exp Clin Cancer Res. 2016;35:181. doi: 10.1186/s13046-016-0458-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Li Y, Liang J, Yang T, Monterrosa Mena J, Huan C, Xie T, Kurkciyan A, Liu N, Jiang D, Noble PW. Hyaluronan synthase 2 regulates fibroblast senescence in pulmonary fibrosis. Matrix Biol. 2016;55:35–48. doi: 10.1016/j.matbio.2016.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Weigel PH, Hascall VC, Tammi M. Hyaluronan synthases. J Biol Chem. 1997;272:13997–14000. doi: 10.1074/jbc.272.22.13997. [DOI] [PubMed] [Google Scholar]
- 54.Weigel PH. Hyaluronan synthase: the mechanism of initiation at the reducing end and a pendulum model for polysaccharide translocation to the cell exterior. Int J Cell Biol. 2015;2015:367579. doi: 10.1155/2015/367579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Weigel PH, Baggenstoss BA, Washburn JL. Hyaluronan synthase assembles hyaluronan on a [GlcNAc(β1,4)]n-GlcNAc(α1→)UDP primer and hyaluronan retains this residual chitin oligomer as a cap at the nonreducing end. Glycobiology. 2017;27:536–554. doi: 10.1093/glycob/cwx012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Joy RA, Vikkath N, Ariyannur PS. Metabolism and mechanisms of action of hyaluronan in human biology. Drug Metabolism Personalized Ther. 2018;33:15–32. doi: 10.1515/dmpt-2017-0031. [DOI] [PubMed] [Google Scholar]
- 57.Jiang D, Liang J, Noble PW. Hyaluronan in tissue injury and repair. Annu Rev Cell Dev Biol. 2007;23:435–461. doi: 10.1146/annurev.cellbio.23.090506.123337. [DOI] [PubMed] [Google Scholar]
- 58.Herbst RS, Morgensztern D, Boshoff C. The biology and management of non-small cell lung cancer. Nature. 2018;553:446–454. doi: 10.1038/nature25183. [DOI] [PubMed] [Google Scholar]
- 59.Oikari S, Kettunen T, Tiainen S, Häyrinen J, Masarwah A, Sudah M, Sutela A, Vanninen R, Tammi M, Auvinen P. UDP-sugar accumulation drives hyaluronan synthesis in breast cancer. Matrix Biol. 2018;67:63–74. doi: 10.1016/j.matbio.2017.12.015. [DOI] [PubMed] [Google Scholar]
- 60.Rilla K, Oikari S, Jokela TA, Hyttinen JM, Kärnä R, Tammi RH, Tammi MI. Hyaluronan synthase 1 (HAS1) requires higher cellular UDP-GlcNAc concentration than HAS2 and HAS3. J Biol Chem. 2013;288:5973–5983. doi: 10.1074/jbc.M112.443879. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Dicker KT, Gurski LA, Pradhan-Bhatt S, Witt RL, Farach-Carson MC, Jia X. Hyaluronan: a simple polysaccharide with diverse biological functions. Acta Biomater. 2014;10:1558–1570. doi: 10.1016/j.actbio.2013.12.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Itano N, Sawai T, Yoshida M, Lenas P, Yamada Y, Imagawa M, Shinomura T, Hamaguchi M, Yoshida Y, Ohnuki Y, et al. Three isoforms of mammalian hyaluronan synthases have distinct enzymatic properties. J Biol Chem. 1999;274:25085–25092. doi: 10.1074/jbc.274.35.25085. [DOI] [PubMed] [Google Scholar]
- 63.Tammi MI, Oikari S, Pasonen-Seppanen S, Rilla K, Auvinen P, Tammi RH. Activated hyaluronan metabolism in the tumor matrix—causes and consequences. Matrix Biol. 2019;78–79:147–164. doi: 10.1016/j.matbio.2018.04.012. [DOI] [PubMed] [Google Scholar]
- 64.Camenisch TD, Spicer AP, Brehm-Gibson T, Biesterfeldt J, Augustine ML, Calabro A, Jr, Kubalak S, Klewer SE, McDonald JA. Disruption of hyaluronan synthase-2 abrogates normal cardiac morphogenesis and hyaluronan-mediated transformation of epithelium to mesenchyme. J Clin Investig. 2000;106:349–360. doi: 10.1172/JCI10272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Bernert B, Porsch H, Heldin P. Hyaluronan synthase 2 (HAS2) promotes breast cancer cell invasion by suppression of tissue metalloproteinase inhibitor 1 (TIMP-1) J Biol Chem. 2011;286:42349–42359. doi: 10.1074/jbc.M111.278598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Chao H, Spicer AP. Natural antisense mRNAs to hyaluronan synthase 2 inhibit hyaluronan biosynthesis and cell proliferation. J Biol Chem. 2005;280:27513–27522. doi: 10.1074/jbc.M411544200. [DOI] [PubMed] [Google Scholar]
- 67.Zhao Z, Liang T, Feng S. Silencing of HAS2-AS1 mediates PI3K/AKT signaling pathway to inhibit cell proliferation, migration, and invasion in glioma. J Cell Biochem. 2019;120:11510–11516. doi: 10.1002/jcb.28430. [DOI] [PubMed] [Google Scholar]
- 68.Parnigoni A, Caon I, Teo WX, Hua SH, Moretto P, Bartolini B, Viola M, Karousou E, Yip GW, Götte M. The natural antisense transcript HAS2-AS1 regulates breast cancer cells aggressiveness independently from hyaluronan metabolism. Matrix Biol. 2022;109:140–161. doi: 10.1016/j.matbio.2022.03.009. [DOI] [PubMed] [Google Scholar]
- 69.Sato N, Maehara N, Goggins M. Gene expression profiling of tumor-stromal interactions between pancreatic cancer cells and stromal fibroblasts. Can Res. 2004;64:6950–6956. doi: 10.1158/0008-5472.CAN-04-0677. [DOI] [PubMed] [Google Scholar]
- 70.Huang H, Huang J, Yao J, Li N, Yang Z. miR-125a regulates HAS1 and inhibits the proliferation, invasion and metastasis by targeting STAT3 in non-small cell lung cancer cells. J Cell Biochem. 2020;121:3197–3207. doi: 10.1002/jcb.29586. [DOI] [PubMed] [Google Scholar]
- 71.Auvinen P, Rilla K, Tumelius R, Tammi M, Sironen R, Soini Y, Kosma VM, Mannermaa A, Viikari J, Tammi R. Hyaluronan synthases (HAS1-3) in stromal and malignant cells correlate with breast cancer grade and predict patient survival. Breast Cancer Res Treat. 2014;143:277–286. doi: 10.1007/s10549-013-2804-7. [DOI] [PubMed] [Google Scholar]
- 72.Kramer MW, Escudero DO, Lokeshwar SD, Golshani R, Ekwenna OO, Acosta K, Merseburger AS, Soloway M, Lokeshwar VB. Association of hyaluronic acid family members (HAS1, HAS2, and HYAL-1) with bladder cancer diagnosis and prognosis. Cancer. 2011;117:1197–1209. doi: 10.1002/cncr.25565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Hamada S, Nishida Y, Zhuo L, Shinomura T, Ikuta K, Arai E, Koike H, Kimata K, Ushida T, Ishiguro N. Suppression of hyaluronan synthesis attenuates the tumorigenicity of low-grade chondrosarcoma. J Orthop Res. 2018;36:1573–1580. doi: 10.1002/jor.23794. [DOI] [PubMed] [Google Scholar]
- 74.Stern R, Asari AA, Sugahara KN. Hyaluronan fragments: an information-rich system. Eur J Cell Biol. 2006;85:699–715. doi: 10.1016/j.ejcb.2006.05.009. [DOI] [PubMed] [Google Scholar]
- 75.Stern R, Kogan G, Jedrzejas MJ, Soltés L. The many ways to cleave hyaluronan. Biotechnol Adv. 2007;25:537–557. doi: 10.1016/j.biotechadv.2007.07.001. [DOI] [PubMed] [Google Scholar]
- 76.Fiszer-Szafarz B, Litynska A, Zou L. Human hyaluronidases: electrophoretic multiple forms in somatic tissues and body fluids. Evidence for conserved hyaluronidase potential n-glycosylation sites in different mammalian species. Journal Biochem Biophys Methods. 2000;45:103–116. doi: 10.1016/S0165-022X(00)00067-1. [DOI] [PubMed] [Google Scholar]
- 77.Li L, Yan LH, Manoj S, Li Y, Lu L. Central role of CEMIP in tumorigenesis and Its potential as therapeutic target. J Cancer. 2017;8:2238–2246. doi: 10.7150/jca.19295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Guo J, Cheng H, Zhao S, Yu L. GG: a domain involved in phage LTF apparatus and implicated in human MEB and non-syndromic hearing loss diseases. FEBS Lett. 2006;580:581–584. doi: 10.1016/j.febslet.2005.12.076. [DOI] [PubMed] [Google Scholar]
- 79.Yoshida H, Nagaoka A, Nakamura S, Tobiishi M, Sugiyama Y, Inoue S. N-Terminal signal sequence is required for cellular trafficking and hyaluronan-depolymerization of KIAA1199. FEBS Lett. 2014;588:111–116. doi: 10.1016/j.febslet.2013.11.017. [DOI] [PubMed] [Google Scholar]
- 80.Yoshida H, Nagaoka A, Nakamura S, Sugiyama Y, Okada Y, Inoue S. Murine homologue of the human KIAA1199 is implicated in hyaluronan binding and depolymerization. FEBS Open Bio. 2013;3:352–356. doi: 10.1016/j.fob.2013.08.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Liang J, Jiang D, Noble PW. Hyaluronan as a therapeutic target in human diseases. Adv Drug Deliv Rev. 2016;97:186–203. doi: 10.1016/j.addr.2015.10.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Jou IM, Wu TT, Hsu CC, Yang CC, Huang JS, Tu YK, Lee JS, Su FC, Kuo YL. High molecular weight form of hyaluronic acid reduces neuroinflammatory response in injured sciatic nerve via the intracellular domain of CD44. J Biomed Mater Res B Appl Biomater. 2021;109:673–680. doi: 10.1002/jbm.b.34731. [DOI] [PubMed] [Google Scholar]
- 83.Singleton PA. Hyaluronan regulation of endothelial barrier function in cancer. Adv Cancer Res. 2014;123:191–209. doi: 10.1016/B978-0-12-800092-2.00007-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Turley EA, Noble PW, Bourguignon LY. Signaling properties of hyaluronan receptors. J Biol Chem. 2002;277:4589–4592. doi: 10.1074/jbc.R100038200. [DOI] [PubMed] [Google Scholar]
- 85.Slevin M, West D, Kumar P, Rooney P, Kumar S. Hyaluronan, angiogenesis and malignant disease. Int J Cancer. 2004;109:793–794. doi: 10.1002/ijc.20059. [DOI] [PubMed] [Google Scholar]
- 86.Yu Q, Toole BP, Stamenkovic I. Induction of apoptosis of metastatic mammary carcinoma cells in vivo by disruption of tumor cell surface CD44 function. J Exp Med. 1997;186:1985–1996. doi: 10.1084/jem.186.12.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Peterson RM, Yu Q, Stamenkovic I, Toole BP. Perturbation of hyaluronan interactions by soluble CD44 inhibits growth of murine mammary carcinoma cells in ascites. Am J Pathol. 2000;156:2159–2167. doi: 10.1016/S0002-9440(10)65086-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Yu M, He P, Liu Y, He Y, Du Y, Wu M, Zhang G, Yang C, Gao F. Hyaluroan-regulated lymphatic permeability through S1P receptors is crucial for cancer metastasis. Med Oncol. 2015;32:381. doi: 10.1007/s12032-014-0381-1. [DOI] [PubMed] [Google Scholar]
- 89.Johnson LA, Jackson DG. Hyaluronan and its receptors: key mediators of immune cell entry and trafficking in the lymphatic system. Cells. 2021;10:2061. doi: 10.3390/cells10082061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Sokolowska M, Chen LY, Eberlein M, Martinez-Anton A, Liu Y, Alsaaty S, Qi HY, Logun C, Horton M, Shelhamer JH. Low molecular weight hyaluronan activates cytosolic phospholipase A2α and eicosanoid production in monocytes and macrophages. J Biol Chem. 2014;289:4470–4488. doi: 10.1074/jbc.M113.515106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Sapudom J, Nguyen KT, Martin S, Wippold T, Möller S, Schnabelrauch M, Anderegg U, Pompe T. Biomimetic tissue models reveal the role of hyaluronan in melanoma proliferation and invasion. Biomater Sci. 2020;8:1405–1417. doi: 10.1039/C9BM01636H. [DOI] [PubMed] [Google Scholar]
- 92.Murata M, Yudoh K, Shimizu H, Beppu M, Nakamura H, Kato T, Masuko K. Layilin, a talin-binding hyaluronan receptor, is expressed in human articular chondrocytes and synoviocytes and is down-regulated by interleukin-1β. Mod Rheumatol. 2013;23:478–488. doi: 10.3109/s10165-012-0686-x. [DOI] [PubMed] [Google Scholar]
- 93.Du Y, Cao M, Liu Y, He Y, Yang C, Wu M, Zhang G, Gao F. Low-molecular-weight hyaluronan (LMW-HA) accelerates lymph node metastasis of melanoma cells by inducing disruption of lymphatic intercellular adhesion. Oncoimmunology. 2016;5:e1232235. doi: 10.1080/2162402X.2016.1232235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Scheibner KA, Lutz MA, Boodoo S, Fenton MJ, Powell JD, Horton MR. Hyaluronan fragments act as an endogenous danger signal by engaging TLR2. J Immunol. 2006;177:1272–1281. doi: 10.4049/jimmunol.177.2.1272. [DOI] [PubMed] [Google Scholar]
- 95.Johnson LA, Banerji S, Lawrance W, Gileadi U, Prota G, Holder KA, Roshorm YM, Hanke T, Cerundolo V, Gale NW, et al. Dendritic cells enter lymph vessels by hyaluronan-mediated docking to the endothelial receptor LYVE-1. Nat Immunol. 2017;18:762–770. doi: 10.1038/ni.3750. [DOI] [PubMed] [Google Scholar]
- 96.Inoue T, Takemori K, Yamamoto K, Ito H. Characterization of Wistar-Kyoto rats showing hyperadiponectinemia. Life Sci. 2010;86:344–350. doi: 10.1016/j.lfs.2010.01.003. [DOI] [PubMed] [Google Scholar]
- 97.Thorgeirsson TE, Geller F, Sulem P, Rafnar T, Wiste A, Magnusson KP, Manolescu A, Thorleifsson G, Stefansson H, Ingason A, et al. A variant associated with nicotine dependence, lung cancer and peripheral arterial disease. Nature. 2008;452:638–642. doi: 10.1038/nature06846. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Rafnar T, Sulem P, Besenbacher S, Gudbjartsson DF, Zanon C, Gudmundsson J, Stacey SN, Kostic JP, Thorgeirsson TE, Thorleifsson G. Genome-wide significant association between a sequence variant at 15q152 and lung cancer risk. Cancer Res. 2011;71:1356–1361. doi: 10.1158/0008-5472.CAN-10-2852. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Tai AL, Mak W, Ng PK, Chua DT, Ng MY, Fu L, Chu KK, Fang Y, Qiang Song Y, Chen M, et al. High-throughput loss-of-heterozygosity study of chromosome 3p in lung cancer using single-nucleotide polymorphism markers. Can Res. 2006;66:4133–4138. doi: 10.1158/0008-5472.CAN-05-2775. [DOI] [PubMed] [Google Scholar]
- 100.Amos CI, Gorlov IP, Dong Q, Wu X, Zhang H, Lu EY, Scheet P, Greisinger AJ, Mills GB, Spitz MR. Nicotinic acetylcholine receptor region on chromosome 15q25 and lung cancer risk among African Americans: a case-control study. J Natl Cancer Inst. 2010;102:1199–1205. doi: 10.1093/jnci/djq232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Cheng Y, Wang C, Zhu M, Dai J, Wang Y, Geng L, Li Z, Zhang J, Ma H, Jin G. Targeted sequencing of chromosome 15q25 identified novel variants associated with risk of lung cancer and smoking behavior in Chinese. Carcinogenesis. 2017;38:552–558. doi: 10.1093/carcin/bgx025. [DOI] [PubMed] [Google Scholar]
- 102.Rosell R, Wannesson L. A genetic snapshot of small cell lung cancer. Cancer Discov. 2012;2:769–771. doi: 10.1158/2159-8290.CD-12-0346. [DOI] [PubMed] [Google Scholar]
- 103.Csoka AB, Frost GI, Heng HH, Scherer SW, Mohapatra G, Stern R. The hyaluronidase gene HYAL1 maps to chromosome 3p21.2-p21.3 in human and 9F1-F2 in mouse, a conserved candidate tumor suppressor locus. Genomics. 1998;48:63–70. doi: 10.1006/geno.1997.5158. [DOI] [PubMed] [Google Scholar]
- 104.Lerman MI, Minna JD. The 630-kb lung cancer homozygous deletion region on human chromosome 3p21.3: identification and evaluation of the resident candidate tumor suppressor genes. The international lung cancer chromosome 3p21.3 tumor suppressor gene consortium. Cancer Res. 2000;60:6116–6133. [PubMed] [Google Scholar]
- 105.Angeloni D. Molecular analysis of deletions in human chromosome 3p21 and the role of resident cancer genes in disease. Brief Funct Genomic Proteomic. 2007;6:19–39. doi: 10.1093/bfgp/elm007. [DOI] [PubMed] [Google Scholar]
- 106.Csoka AB, Frost GI, Stern R. The six hyaluronidase-like genes in the human and mouse genomes. Matrix Biol. 2001;20:499–508. doi: 10.1016/S0945-053X(01)00172-X. [DOI] [PubMed] [Google Scholar]
- 107.Hung RJ, McKay JD, Gaborieau V, Boffetta P, Hashibe M, Zaridze D, Mukeria A, Szeszenia-Dabrowska N, Lissowska J, Rudnai P. A susceptibility locus for lung cancer maps to nicotinic acetylcholine receptor subunit genes on 15q25. Nature. 2008;452:633–637. doi: 10.1038/nature06885. [DOI] [PubMed] [Google Scholar]
- 108.Braga EA, Kashuba VI, Maliukova AV, Loginov VI, Senchenko VN, Bazov IV, Kiselev LL, Zabarovskii ER. New tumor suppressor genes in hot spots of human chromosome 3: new methods of identification. Mol Biol. 2003;37:194–211. doi: 10.1023/A:1023381218481. [DOI] [PubMed] [Google Scholar]
- 109.Karousou E, Misra S, Ghatak S, Dobra K, Götte M, Vigetti D, Passi A, Karamanos NK, Skandalis SS. Roles and targeting of the HAS/hyaluronan/CD44 molecular system in cancer. Matrix Biol. 2017;59:3–22. doi: 10.1016/j.matbio.2016.10.001. [DOI] [PubMed] [Google Scholar]
- 110.Natowicz MR, Short MP, Wang Y, Dickersin GR, Gebhardt MC, Rosenthal DI, Sims KB, Rosenberg AE. Clinical and biochemical manifestations of hyaluronidase deficiency. N Engl J Med. 1996;335:1029–1033. doi: 10.1056/NEJM199610033351405. [DOI] [PubMed] [Google Scholar]
- 111.Csoka AB, Stern R. Hypotheses on the evolution of hyaluronan: a highly ironic acid. Glycobiology. 2013;23:398–411. doi: 10.1093/glycob/cws218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Vigetti D, Passi A. Hyaluronan synthases posttranslational regulation in cancer. Adv Cancer Res. 2014;123:95–119. doi: 10.1016/B978-0-12-800092-2.00004-6. [DOI] [PubMed] [Google Scholar]
- 113.Wang C, Tammi M, Guo H, Tammi R. Hyaluronan distribution in the normal epithelium of esophagus, stomach, and colon and their cancers. Am J Pathol. 1996;148:1861–1869. [PMC free article] [PubMed] [Google Scholar]
- 114.Picaud L, Thibault B, Mery E, Ouali M, Martinez A, Delord JP, Couderc B, Ferron G. Evaluation of the effects of hyaluronic acid-carboxymethyl cellulose barrier on ovarian tumor progression. J Ovarian Res. 2014;7:40. doi: 10.1186/1757-2215-7-40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Lokeshwar VB, Obek C, Soloway MS, Block NL. Tumor-associated hyaluronic acid: a new sensitive and specific urine marker for bladder cancer. Can Res. 1997;57:773–777. [PubMed] [Google Scholar]
- 116.Lokeshwar VB, Rubinowicz D, Schroeder GL, Forgacs E, Minna JD, Block NL, Nadji M, Lokeshwar BL. Stromal and epithelial expression of tumor markers hyaluronic acid and HYAL1 hyaluronidase in prostate cancer. J Biol Chem. 2001;276:11922–11932. doi: 10.1074/jbc.M008432200. [DOI] [PubMed] [Google Scholar]
- 117.Larson BK, Guan M, Placencio V, Tuli R, Hendifar AE. Stromal hyaluronan accumulation is associated with low tumor grade and nodal metastases in pancreatic ductal adenocarcinoma. Hum Pathol. 2019;90:37–44. doi: 10.1016/j.humpath.2019.05.004. [DOI] [PubMed] [Google Scholar]
- 118.Horai T, Nakamura N, Tateishi R, Hattori S. Glycosaminoglycans in human lung cancer. Cancer. 1981;48:2016–2021. doi: 10.1002/1097-0142(19811101)48:9<2016::AID-CNCR2820480918>3.0.CO;2-A. [DOI] [PubMed] [Google Scholar]
- 119.Anttila MA, Tammi RH, Tammi MI, Syrjanen KJ, Saarikoski SV, Kosma VM. High levels of stromal hyaluronan predict poor disease outcome in epithelial ovarian cancer. Can Res. 2000;60:150–155. [PubMed] [Google Scholar]
- 120.Ropponen K, Tammi M, Parkkinen J, Eskelinen M, Tammi R, Lipponen P, Agren U, Alhava E, Kosma VM. Tumor cell-associated hyaluronan as an unfavorable prognostic factor in colorectal cancer. Can Res. 1998;58:342–347. [PubMed] [Google Scholar]
- 121.Posey JT, Soloway MS, Ekici S, Sofer M, Civantos F, Duncan RC, Lokeshwar VB. Evaluation of the prognostic potential of hyaluronic acid and hyaluronidase (HYAL1) for prostate cancer. Can Res. 2003;63:2638–2644. [PubMed] [Google Scholar]
- 122.Teranishi F, Takahashi N, Gao N, Akamo Y, Takeyama H, Manabe T, Okamoto T. Phosphoinositide 3-kinase inhibitor (wortmannin) inhibits pancreatic cancer cell motility and migration induced by hyaluronan in vitro and peritoneal metastasis in vivo. Cancer Sci. 2009;100:770–777. doi: 10.1111/j.1349-7006.2009.01084.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Cheng XB, Kohi S, Koga A, Hirata K, Sato N. Hyaluronan stimulates pancreatic cancer cell motility. Oncotarget. 2016;7:4829–4840. doi: 10.18632/oncotarget.6617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Rizzardi AE, Vogel RI, Koopmeiners JS, Forster CL, Marston LO, Rosener NK, Akentieva N, Price MA, Metzger GJ, Warlick CA, et al. Elevated hyaluronan and hyaluronan-mediated motility receptor are associated with biochemical failure in patients with intermediate-grade prostate tumors. Cancer. 2014;120:1800–1809. doi: 10.1002/cncr.28646. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Matou-Nasri S, Gaffney J, Kumar S, Slevin M. Oligosaccharides of hyaluronan induce angiogenesis through distinct CD44 and RHAMM-mediated signalling pathways involving Cdc2 and gamma-adducin. Int J Oncol. 2009;35:761–773. doi: 10.3892/ijo_00000389. [DOI] [PubMed] [Google Scholar]
- 126.Chaffer CL, San Juan BP, Lim E, Weinberg RA. EMT, cell plasticity and metastasis. Cancer Metastasis Rev. 2016;35:645–654. doi: 10.1007/s10555-016-9648-7. [DOI] [PubMed] [Google Scholar]
- 127.Cieply B, Koontz C, Frisch SM. CD44S-hyaluronan interactions protect cells resulting from EMT against anoikis. Matrix Biol. 2015;48:55–65. doi: 10.1016/j.matbio.2015.04.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.El-Haibi CP, Bell GW, Zhang J, Collmann AY, Wood D, Scherber CM, Csizmadia E, Mariani O, Zhu C, Campagne A, et al. Critical role for lysyl oxidase in mesenchymal stem cell-driven breast cancer malignancy. Proc Natl Acad Sci USA. 2012;109:17460–17465. doi: 10.1073/pnas.1206653109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Chanmee T, Ontong P, Mochizuki N, Kongtawelert P, Konno K, Itano N. Excessive hyaluronan production promotes acquisition of cancer stem cell signatures through the coordinated regulation of twist and the transforming growth factor β (TGF-β)-snail signaling axis. J Biol Chem. 2014;289:26038–26056. doi: 10.1074/jbc.M114.564120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Tavianatou AG, Piperigkou Z, Barbera C, Beninatto R, Masola V, Caon I, Onisto M, Franchi M, Galesso D, Karamanos NK. Molecular size-dependent specificity of hyaluronan on functional properties, morphology and matrix composition of mammary cancer cells. Matrix Biol Plus. 2019;3:100008. doi: 10.1016/j.mbplus.2019.100008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Liu S, Yu J, Zhang Q, Lu H, Qiu X, Zhou D, Qi Y, Huang Y. Dual cross-linked HHA hydrogel supplies and regulates MΦ2 for synergistic improvement of immunocompromise and impaired angiogenesis to enhance diabetic chronic wound healing. Biomacromol. 2020;21:3795–3806. doi: 10.1021/acs.biomac.0c00891. [DOI] [PubMed] [Google Scholar]
- 132.Mendes C, Thirupathi A, Zaccaron RP, Corrêa M, Bittencourt JVS, Casagrande LR, de Lima ACS, de Oliveira LL, de Andrade TAM, Gu Y. Microcurrent and gold nanoparticles combined with hyaluronic acid accelerates wound healing. Antioxidants. 2022;11:2257. doi: 10.3390/antiox11112257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Blunck D, Schöffski O. Hyaluronic acid treatment versus standard of care in chronic wounds in a German setting: cost-effectiveness analysis. Health Sci Rep. 2023;6:e969. doi: 10.1002/hsr2.969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Solis MA, Wei YH, Chang CH, Yu CH, Huang LLH. Hyaluronan induces a mitochondrial functional switch in fast-proliferating human mesenchymal stem. Int J Stem Cells. 2020;13:151–162. doi: 10.15283/ijsc19004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Oe M, Mitsugi K, Odanaka W, Yoshida H, Matsuoka R, Seino S, Kanemitsu T, Masuda Y. Dietary hyaluronic acid migrates into the skin of rats. Sci World J. 2014;2014:378024. doi: 10.1155/2014/378024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Caon I, Parnigoni A, Viola M, Karousou E, Passi A, Vigetti D. Cell energy metabolism and hyaluronan synthesis. J Histochem Cytochem. 2021;69:35–47. doi: 10.1369/0022155420929772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Lam C, Low JY, Tran PT, Wang H. The hexosamine biosynthetic pathway and cancer: current knowledge and future therapeutic strategies. Cancer Lett. 2021;503:11–18. doi: 10.1016/j.canlet.2021.01.010. [DOI] [PubMed] [Google Scholar]
- 138.Stern R. Hyaluronan catabolism: a new metabolic pathway. Eur J Cell Biol. 2004;83:317–325. doi: 10.1078/0171-9335-00392. [DOI] [PubMed] [Google Scholar]
- 139.Skandalis SS, Karalis T, Heldin P. Intracellular hyaluronan: importance for cellular functions. Semin Cancer Biol. 2020;62:20–30. doi: 10.1016/j.semcancer.2019.07.002. [DOI] [PubMed] [Google Scholar]
- 140.Olivares O, Mayers JR, Gouirand V, Torrence ME, Gicquel T, Borge L, Lac S, Roques J, Lavaut MN, Berthezène P, et al. Collagen-derived proline promotes pancreatic ductal adenocarcinoma cell survival under nutrient limited conditions. Nat Commun. 2017;8:16031. doi: 10.1038/ncomms16031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Qian Y, Wang X, Li Y, Cao Y, Chen X. Extracellular ATP a new player in cancer metabolism: NSCLC cells internalize ATP In vitro and in vivo using multiple endocytic mechanisms. Mol Cancer Res. 2016;14:1087–1096. doi: 10.1158/1541-7786.MCR-16-0118. [DOI] [PubMed] [Google Scholar]
- 142.Seternes T, Øynebråten I, Sørensen K, Smedsrød B. Specific endocytosis and catabolism in the scavenger endothelial cells of cod (Gadus morhua L.) generate high-energy metabolites. J Exp Biol. 2001;204:1537–1546. doi: 10.1242/jeb.204.9.1537. [DOI] [PubMed] [Google Scholar]
- 143.Kim PK, Halbrook CJ, Kerk SA, Radyk M, Wisner S, Kremer DM, Sajjakulnukit P, Andren A, Hou SW, Trivedi A. Hyaluronic acid fuels pancreatic cancer cell growth. eLife. 2021;10:e62645. doi: 10.7554/eLife.62645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Sullivan WJ, Mullen PJ, Schmid EW, Flores A, Momcilovic M, Sharpley MS, Jelinek D, Whiteley AE, Maxwell MB, Wilde BR, et al. Extracellular matrix remodeling regulates glucose metabolism through TXNIP destabilization. Cell. 2018;175:117–132.e121. doi: 10.1016/j.cell.2018.08.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Narvaez CJ, Grebenc D, Balinth S, Welsh JE. Vitamin D regulation of HAS2, hyaluronan synthesis and metabolism in triple negative breast cancer cells. J Steroid Biochem Mol Biol. 2020;201:105688. doi: 10.1016/j.jsbmb.2020.105688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Arnold JM, Gu F, Ambati CR, Rasaily U, Ramirez-Pena E, Joseph R, Manikkam M, San Martin R, Charles C, Pan Y. UDP-glucose 6-dehydrogenase regulates hyaluronic acid production and promotes breast cancer progression. Oncogene. 2020;39:3089–3101. doi: 10.1038/s41388-019-0885-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Huang L, Wang Y, Liu H, Huang J. Local injection of high-molecular hyaluronan promotes wound healing in old rats by increasing angiogenesis. Oncotarget. 2018;9:8241–8252. doi: 10.18632/oncotarget.23246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Almond A. Hyaluronan. Cell Mol Life Sci. 2007;64:1591–1596. doi: 10.1007/s00018-007-7032-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Vasvani S, Kulkarni P, Rawtani D. Hyaluronic acid: a review on its biology, aspects of drug delivery, route of administrations and a special emphasis on its approved marketed products and recent clinical studies. Int J Biol Macromol. 2020;151:1012–1029. doi: 10.1016/j.ijbiomac.2019.11.066. [DOI] [PubMed] [Google Scholar]
- 150.Spinelli FM, Vitale DL, Icardi A, Caon I, Brandone A, Giannoni P, Saturno V, Passi A, García M, Sevic I. Hyaluronan preconditioning of monocytes/macrophages affects their angiogenic behavior and regulation of TSG-6 expression in a tumor type-specific manner. FEBS J. 2019;286:3433–3449. doi: 10.1111/febs.14871. [DOI] [PubMed] [Google Scholar]
- 151.Spinelli FM, Rosales P, Pluda S, Vitale DL, Icardi A, Guarise C, Reszegi A, Kovalszky I, García M, Sevic I, et al. The effects of sulfated hyaluronan in breast, lung and colorectal carcinoma and monocytes/macrophages cells: Its role in angiogenesis and tumor progression. IUBMB Life. 2022;74:927–942. doi: 10.1002/iub.2604. [DOI] [PubMed] [Google Scholar]
- 152.Schimizzi AL, Massie JB, Murphy M, Perry A, Kim CW, Garfin SR, Akeson WH. High-molecular-weight hyaluronan inhibits macrophage proliferation and cytokine release in the early wound of a preclinical postlaminectomy rat model. Spine J. 2006;6:550–556. doi: 10.1016/j.spinee.2005.12.005. [DOI] [PubMed] [Google Scholar]
- 153.West DC, Hampson IN, Arnold F, Kumar S. Angiogenesis induced by degradation products of hyaluronic acid. Science. 1985;228:1324–1326. doi: 10.1126/science.2408340. [DOI] [PubMed] [Google Scholar]
- 154.Noble PW. Hyaluronan and its catabolic products in tissue injury and repair. Matrix Biol. 2002;21:25–29. doi: 10.1016/S0945-053X(01)00184-6. [DOI] [PubMed] [Google Scholar]
- 155.Litwiniuk M, Krejner A, Speyrer MS, Gauto AR, Grzela T. Hyaluronic acid in inflammation and tissue regeneration. Wounds. 2016;28:78–88. [PubMed] [Google Scholar]
- 156.Pardue EL, Ibrahim S, Ramamurthi A. Role of hyaluronan in angiogenesis and its utility to angiogenic tissue engineering. Organogenesis. 2008;4:203–214. doi: 10.4161/org.4.4.6926. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Larcher F, Franco M, Bolontrade M, Rodriguez-Puebla M, Casanova L, Navarro M, Yancopoulos G, Jorcano JL, Conti CJ. Modulation of the angiogenesis response through Ha-ras control, placenta growth factor, and angiopoietin expression in mouse skin carcinogenesis. Mol Carcinog. 2003;37:83–90. doi: 10.1002/mc.10126. [DOI] [PubMed] [Google Scholar]
- 158.Isa IL, Srivastava A, Tiernan D, Owens P, Rooney P, Dockery P, Pandit A. Hyaluronic acid based hydrogels attenuate inflammatory receptors and neurotrophins in interleukin-1β induced inflammation model of nucleus pulposus cells. Biomacromol. 2015;16:1714–1725. doi: 10.1021/acs.biomac.5b00168. [DOI] [PubMed] [Google Scholar]
- 159.Qadri M, Almadani S, Jay GD, Elsaid KA. Role of CD44 in regulating TLR2 activation of human macrophages and downstream expression of proinflammatory cytokines. J Immunol. 2018;200:758–767. doi: 10.4049/jimmunol.1700713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Wang YZ, Cao ML, Liu YW, He YQ, Yang CX, Gao F. CD44 mediates oligosaccharides of hyaluronan-induced proliferation, tube formation and signal transduction in endothelial cells. Exp Biol Med (Maywood) 2011;236:84–90. doi: 10.1258/ebm.2010.010206. [DOI] [PubMed] [Google Scholar]
- 161.Chiou CS, Wu CM, Dubey NK, Lo WC, Tsai FC, Tung TDX, Hung WC, Hsu WC, Chen WH, Deng WP. Mechanistic insight into hyaluronic acid and platelet-rich plasma-mediated anti-inflammatory and anti-apoptotic activities in osteoarthritic mice. Aging. 2018;10:4152–4165. doi: 10.18632/aging.101713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Kai F, Laklai H, Weaver VM. Force matters: biomechanical regulation of cell invasion and migration in disease. Trends Cell Biol. 2016;26:486–497. doi: 10.1016/j.tcb.2016.03.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Kharaishvili G, Simkova D, Bouchalova K, Gachechiladze M, Narsia N, Bouchal J. The role of cancer-associated fibroblasts, solid stress and other microenvironmental factors in tumor progression and therapy resistance. Cancer Cell Int. 2014;14:41. doi: 10.1186/1475-2867-14-41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Ananthanarayanan B, Kim Y, Kumar S. Elucidating the mechanobiology of malignant brain tumors using a brain matrix-mimetic hyaluronic acid hydrogel platform. Biomaterials. 2011;32:7913–7923. doi: 10.1016/j.biomaterials.2011.07.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Kaukonen R, Mai A, Georgiadou M, Saari M, De Franceschi N, Betz T, Sihto H. Normal stroma suppresses cancer cell proliferation via mechanosensitive regulation of JMJD1a-mediated transcription. Nat Commun. 2016;7:12237. doi: 10.1038/ncomms12237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Wei SC, Fattet L, Tsai JH, Guo Y, Pai VH, Majeski HE, Chen AC, Sah RL, Taylor SS, Engler AJ, et al. Matrix stiffness drives epithelial-mesenchymal transition and tumour metastasis through a TWIST1-G3BP2 mechanotransduction pathway. Nat Cell Biol. 2015;17:678–688. doi: 10.1038/ncb3157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Pogoda K, Bucki R, Byfield FJ, Cruz K, Lee T, Marcinkiewicz C, Janmey PA. Soft substrates containing hyaluronan mimic the effects of increased stiffness on morphology, motility, and proliferation of glioma cells. Biomacromol. 2017;18:3040–3051. doi: 10.1021/acs.biomac.7b00324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Wang S, Wang Y, Xiong J, Bao W, Li Y, Qin J, Han G, Hu S, Lei J, Yang Z, et al. Novel brain-stiffness-mimicking matrix gel enables comprehensive invasion analysis of 3D cultured GBM cells. Front Mol Biosci. 2022;9:885806. doi: 10.3389/fmolb.2022.885806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Serban MA, Prestwich GD. Modular extracellular matrices: solutions for the puzzle. Methods. 2008;45:93–98. doi: 10.1016/j.ymeth.2008.01.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Rosenthal MA, Gibbs P, Brown TJ, Wong S, Uren S, Ellis A, Li L, Heldin P, Fox RM. Phase I and pharmacokinetic evaluation of intravenous hyaluronic acid in combination with doxorubicin or 5-fluorouracil. Chemotherapy. 2005;51:132–141. doi: 10.1159/000085621. [DOI] [PubMed] [Google Scholar]
- 171.Alamgeer M, Neil Watkins D, Banakh I, Kumar B, Gough DJ, Markman B, Ganju V. A phase IIa study of HA-irinotecan, formulation of hyaluronic acid and irinotecan targeting CD44 in extensive-stage small cell lung cancer. Invest New Drugs. 2018;36:288–298. doi: 10.1007/s10637-017-0555-8. [DOI] [PubMed] [Google Scholar]
- 172.Gibbs P, Clingan PR, Ganju V, Strickland AH, Wong SS, Tebbutt NC, Underhill CR, Fox RM, Clavant SP, Leung J, et al. Hyaluronan-Irinotecan improves progression-free survival in 5-fluorouracil refractory patients with metastatic colorectal cancer: a randomized phase II trial. Cancer Chemother Pharmacol. 2011;67:153–163. doi: 10.1007/s00280-010-1303-3. [DOI] [PubMed] [Google Scholar]
- 173.Wang Y, Ma S, Liu X, Wei Y, Xu H, Liang Z, Hu Y, Lian X, Huang D. Hyaluronic acid mediated Fe(3)O(4) nanocubes reversing the EMT through targeted cancer stem cell. Colloids Surf B. 2022;222:113071. doi: 10.1016/j.colsurfb.2022.113071. [DOI] [PubMed] [Google Scholar]
- 174.Jhan HJ, Liu JJ, Chen YC, Liu DZ, Sheu MT, Ho HO. Novel injectable thermosensitive hydrogels for delivering hyaluronic acid-doxorubicin nanocomplexes to locally treat tumors. Nanomedicine. 2015;10:1263–1274. doi: 10.2217/nnm.14.211. [DOI] [PubMed] [Google Scholar]
- 175.Wan T, Fan P, Zhang M, Shi K, Chen X, Yang H, Liu X, Xu W, Zhou Y. Multiple crosslinking hyaluronic acid hydrogels with improved strength and 3D printability. ACS Appl Bio Mater. 2022;5:334–343. doi: 10.1021/acsabm.1c01141. [DOI] [PubMed] [Google Scholar]
- 176.Huang YJ, Hsu SH. Acquisition of epithelial-mesenchymal transition and cancer stem-like phenotypes within chitosan-hyaluronan membrane-derived 3D tumor spheroids. Biomaterials. 2014;35:10070–10079. doi: 10.1016/j.biomaterials.2014.09.010. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.