Using homology modelling and molecular docking algorithms, Liu and Li predicted the existence of interactions between SARS‐Cov‐2 and haemoglobin (Hb) and suggested that these interactions may change haemoglobin‐oxygen (Hb‐O2) affinity. 1 Hb‐O2 affinity has been extensively studied in intensive care unit (ICU) patients with COVID‐19 (usually intubated and mechanically ventilated) showing normal to increased affinity. 2 , 3 , 4 Higher Hb‐O2 affinity facilitates Hb oxygenation in the lungs while reducing tissue O2 unloading; a reduced Hb‐O2 affinity has the opposite effect. Only one study has investigated patients with COVID‐19 outside the ICU. 5
In a retrospective, cross‐sectional study, we analysed Hb‐O2 affinity in COVID‐19 patients hospitalised in general wards of our institution. The local ethics committee approved the study. Details on study protocol, methods and additional results are reported in Data S1. All patients had a laboratory‐confirmed diagnosis of COVID‐19 and an arterial blood gas analysis performed on admission, with a Hb‐O2 saturation (SO2) of less than 97%. At the time of analysis, no patient was intubated, mechanically ventilated, or had received a COVID‐19 vaccination. We determined oxygen tension at half‐saturation of Hb (P50), an index of Hb‐O2 affinity, using two equations for the determination of in vivo and standard P50 (Figure S1). 6 In vivo P50 reflects actual arterial blood P50, as determined by Hb structure, red‐cell 2,3‐diphosphoglycerate (DPG), pH and arterial pressure of CO2 (PaCO2). Standard P50 is the P50 value measured at pH 7.4 and PaCO2 40 mm Hg; it is an index of Hb‐O2 affinity in which the influences of pH and PaCO2 have been removed. 7
General characteristics of study patients are shown in Table S1. Systemic inflammation was common (elevated C‐reactive protein and serum ferritin), but only 12% of cases had procalcitonin concentrations greater than 2.00 ng/ml, suggestive of superimposed bacterial infection. Standard P50 was higher than in vivo P50 (p < 0.001), although median values of both parameters fell within the normal range. Table S2 shows correlations of in vivo and standard P50 with clinical and laboratory parameters. Multivariable analysis demonstrated that a compromised respiratory function, expressed by reduced SO2 and a low ratio of arterial O2 pressure to fraction of inspired oxygen (PaO2/FiO2) and high lactate and lactate dehydrogenase, were associated with higher P50 (Table 1). pH had a negative correlation with in vivo P50 and a positive correlation with standard P50. Ninety‐five patients (33%) died within 28 days from hospital admission. Patients who died had more elevated in vivo P50 than those who survived, while no difference was observed for standard P50 (Table S3). However, on multivariable logistic regression, the effect of in vivo P50 disappeared and only advanced age, reduced PaO2/FiO2 ratio and increased lactate dehydrogenase were associated with mortality (Table S4).
TABLE 1.
Multiple regression analysis of factors related to P50
| Variables | In vivo P50 | Standard P50 | ||
|---|---|---|---|---|
| PCor (95% CI) | p | PCor (95% CI) | p | |
| SO2 | −0.581 (−0.659 to −0.490) | <0.001 | −0.563 (−0.645 to −0.469) | <0.001 |
| pH | −0.675 (−0.739 to −0.599) | <0.001 | 0.399 (0.285 to 0.502) | <0.001 |
| Lactate | 0.287 (0.166 to 0.400) | <0.001 | 0.289 (0.167 to 0.402) | <0.001 |
| Multiple regression r | 0.769 (0.711 to 0.816) | <0.001 | 0.679 (0.603 to 0.742) | <0.001 |
| The below analysis was performed excluding SO2 from independent variables; pH and PaCO2 were additionally excluded in standard P50 analysis | ||||
|---|---|---|---|---|
| PaO2/FiO2 | −0.314 (−0.428 to −0.190) | <0.001 | −0.146 (−0.278 to −0.009) | 0.038 |
| pH | −0.603 (−0.681 to −0.512) | <0.001 | — | — |
| Lactate | 0.324 (0.200 to 0.438) | <0.001 | 0.153 (0.016 to 0.284) | 0.030 |
| LDH | — | — | 0.361 (0.235 to 0.475) | <0.001 |
| OxyHb | −0.209 (−0.332 to −0.079) | 0.002 | −0.148 (−0.280 to −0.011) | 0.036 |
| Multiple regression r | 0.501 (0.395 to 0.594) | <0.001 | 0.508 (0.399 to 0.603) | <0.001 |
Abbreviations: LDH, lactate dehydrogenase; OxyHb, arterial oxyhaemoglobin; PaO2/FiO2, PaO2 to fraction of inspired oxygen; PCor, partial correlation coefficient.
Figure 1 shows SO2 values as a function of measured PaO2, or PaO2 adjusted for pH and PaO2; distribution of SO2 was similar to the oxygen dissociation curve (ODC) obtained from Hill's equation as adapted by Doyle (Figure 1), in which normal P50, measured at 37°C, pH 7.40 and PaCO2 40 mm Hg, is assumed to be 26.6 mm Hg, 8 showing an overall agreement between observed and expected SO2. This curve almost perfectly overlapped the standard human Hb ODC reported by Severinghaus 9 (data not shown). Lower levels of SO2 (<85%) were often shifted rightward with respect to the reference SO2 curve expected for a given PaO2, indicating that these patients had reduced Hb‐O2 affinity. Patients with SO2 of less than 85% had a reduced PaO2/FiO2 ratio, high lactate (>1.7 mmol/L) and increased P50 (Table S5). In these patients, standard P50 was significantly higher than 28.0 mm Hg, the upper limit of normal (p < 0.001), and the elevation was not justified by pH or PaCO2 changes. These results show that a severely compromised respiratory function is associated with reduced Hb‐O2 affinity.
FIGURE 1.
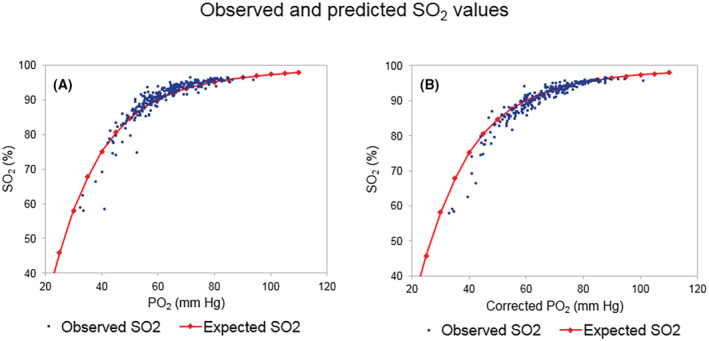
Distribution of observed and predicted SO2 values in COVID‐19 patients. (A) SO2 is reported as a function of observed PaO2. and (B) as a function of PaO2 corrected for arterial blood pH and PaCO2. Red marks represent the expected SO2 value for any given PaO2 according to Doyle equation; blue dots correspond to experimental data.
Our data suggest a preeminent role of patients’ respiratory status and acid‐base balance in the regulation of both in vivo and standard P50. pH was negatively related to in vivo P50, since acidosis reduces Hb‐O2 affinity; 9 in contrast, pH had a positive correlation with standard P50, as described in critically ill patients. 6 PaCO2 behaved in the opposite way. The correlations with standard P50 are likely to represent the indirect effects of pH (and PaCO2), possibly due to the upregulation of 2,3‐DPG production by alkalemia. 10 Our results are partially at variance with previous reports showing increased or normal Hb‐O2 affinity in COVID‐19. 2 , 3 , 4 , 5 , 6 , 11 Most of these studies were conducted in ICUs with determination of standard P50; in these settings a permissive hypercapnia is often allowed when low tidal ventilation is adopted to reduce ventilator‐associated lung injury 12 and may explain the reported reduction in standard P50. Otherwise, reduction of standard P50 could result from 2,3‐DPG downregulation in the presence of respiratory or metabolic acidosis. Compared with our data, ICU studies reported higher PaCO2 and lower pH values, 3 , 4 which can cause standard P50 reduction, but also demonstrated that elevated P50 is usually associated with more severe disease and increased mortality. 4 , 5
Some patients in our study had respiratory distress and were subsequently transferred to the ICU, but at the time of analysis they were spontaneously breathing, and most had normal blood pH with low PaCO2. Median Hb was 129 g/L, confirming that COVID‐19 patients in internal medicine units usually have only mild anaemia. 13 In addition, Hb was similar in patients with SO2 of 85% or more and those with a SO2 below 85%, indicating that anaemia was not involved in the upregulation of P50 associated with worsening respiratory function. Our data in patients with SO2 below 85%, showing increased P50 and lactate concentration, confirm previous observations in acute myocardial infarction, suggesting that P50 elevation represents a compensatory change aimed at preventing tissue hypoxia when oxygen delivery is compromised, 14 whereas high plasma lactate is a marker of the O2 debt associated with decreased oxygen transport and consumption. 15 Although the reported results are preliminary and require confirmation, we document that a severely compromised respiratory function is accompanied by significant elevations of P50 in COVID‐19 that is likely to be an expression of the organism's adaptation to hypoxaemia. This agrees with previous observations showing that a left‐shifted ODC is associated with lower mortality in COVID‐19 patients at hospital admission. 5
AUTHOR CONTRIBUTIONS
Gaetano Bergamaschi designed the study, analysed data, performed statistical analysis and drafted the manuscript. Chiara Barteselli, Virginia Del Rio, Federica Borrelli de Andreis, Ivan Pellegrino, Caterina Mengoli, Emanuela Miceli, Marta Colaneri and Valentina Zuccaro set up the database, collected the data and critically reviewed the manuscript. Michele Di Stefano, Raffaele Bruno and Antonio Di Sabatino designed the study, analysed the data and critically reviewed the manuscript.
CONFLICTS OF INTEREST
The authors declare no competing conflicts of interest.
Supporting information
Figure S1.
Table S1.
Table S2.
Table S3.
Table S4.
Table S5.
Table S6.
Table S7.
Table S8.
Table S9.
Data S1.
ACKNOWLEDGEMENTS
The work was supported by Fondazione IRCCS Policlinico San Matteo, Pavia, Italy. The investigators are grateful to the patients who participated in this study and acknowledge the contribution of the healthcare professionals who faced the COVID‐19 epidemic at the San Matteo Hospital Foundation in Pavia, Italy.
APPENDIX A.
Internal Medicine Covid‐19 Collaborators
Giulia Maria Abruzzese, Giovanna Achilli, Eleonora Alimenti, Giacomo Alunno, Valentina Antoci, Marco Aprile, Alice Argelli, Nicola Aronico, Alessia Ballesio, Giulia Bellini, Alessandra Berbenni, Giampiera Bertolino, Paola Ilaria Bianchi, Silvia Biserni, Alessandra Bonfichi, Tommaso Bosoni, Michele Brattoli, Francesca Calabretta, Marco Callisti, Ginevra Cambiè, Roberta Canta, Pietro Canu, Chiara Cebrelli, Aurora Cecco, Carolina Cicalini, Marina Clemente, Luca Clerici, Silvia Codega, Federico Conca, Luigi Coppola, Filippo Costanzo, Maria Elisa Cremonte, Roberto Cresci, Mariangela Delliponti, Virginia Del Rio, Claudia Delogu, Giuseppe Derosa, Davide Dionisi, Francesco Di Terlizzi, Michele Dota, Francesco Falaschi, Erica Fazzino, Letizia Ferrami, Serena Ferrara, Maria Giovanna Ferrari, Nicola Ferruccio, Giulia Freddi, Carmine Frenna, Chiara Frigerio, Federica Fumoso, Alessandra Fusco, Alice Galeazzo, Vera Gaspari, Antonella Gentile, Alessio Giangreco, Giulia Gori, Giacomo Grandi, Virginia Gregorio, Paolo Grimaldi, Andrea Italia, Francesco Lapia, Mario Andrea Latorre, Marco Vincenzo Lenti, Federica Lepore, Antonio Lobello, Elisabetta Lovati, Pietro Carlo Lucotti, Francesca Lusetti, Stiliano Maimaris, Jacopo Mambella, Alessandra Martignoni, Federica Melazzini, Chiara Mercanti, Stefania Merli, Giusy Cinzia Moltisanti, Maria Elena Monti, Luisa Morbegno, Francesco Mordà, Amedeo Mugellini, Chiara Muggia, Roberta Muscia, Alba Nardone, Lucia Padovini, Ilaria Palumbo, Ignazio Marzio Parisi, Alessandro Pecci, Giulia Paola Peroo, Clarissa Petrucci, Carla Pieresca, Gianluca Pino, Lavinia Pitotti, Silvia Poma, Paola Stefania Preti, Andrea Quadrelli, Alessandro Rascaroli, Giovanni Rigano, Carlo Maria Rossi, Giorgio Rotola, Damiano Ruggeri, Maria Concetta Russo, Umberto Sabatini, Simone Saglio, Giovanni Santacroce, Gabriele Savioli, Jessica Savioli, Simone Salvator Scalia, Davide Scalvini, Yiftach Shoval, Carlo Soffiantini, Simone Soriano, Dario Spadaro, Andrea Staniscia, Debora Stefani, Federico Vai, Matteo Varallo, Marta Vernero
Complete list of Internal Medicine Covid‐19 Collaborators are given in Appendix section.
Contributor Information
Gaetano Bergamaschi, Email: n.bergamaschi@smatteo.pv.it, Email: gaetanobergamaschi@libero.it.
the Internal Medicine Covid‐19 Collaborators:
Giulia Maria Abruzzese, Giovanna Achilli, Eleonora Alimenti, Giacomo Alunno, Valentina Antoci, Marco Aprile, Alice Argelli, Nicola Aronico, Alessia Ballesio, Giulia Bellini, Alessandra Berbenni, Giampiera Bertolino, Paola Ilaria Bianchi, Silvia Biserni, Alessandra Bonfichi, Tommaso Bosoni, Michele Brattoli, Francesca Calabretta, Marco Callisti, Ginevra Cambiè, Roberta Canta, Pietro Canu, Chiara Cebrelli, Aurora Cecco, Carolina Cicalini, Marina Clemente, Luca Clerici, Silvia Codega, Federico Conca, Luigi Coppola, Filippo Costanzo, Maria Elisa Cremonte, Roberto Cresci, Mariangela Delliponti, Virginia Del Rio, Claudia Delogu, Giuseppe Derosa, Davide Dionisi, Francesco Di Terlizzi, Michele Dota, Francesco Falaschi, Erica Fazzino, Letizia Ferrami, Serena Ferrara, Maria Giovanna Ferrari, Nicola Ferruccio, Giulia Freddi, Carmine Frenna, Chiara Frigerio, Federica Fumoso, Alessandra Fusco, Alice Galeazzo, Vera Gaspari, Antonella Gentile, Alessio Giangreco, Giulia Gori, Giacomo Grandi, Virginia Gregorio, Paolo Grimaldi, Andrea Italia, Francesco Lapia, Mario Andrea Latorre, Marco Vincenzo Lenti, Federica Lepore, Antonio Lobello, Elisabetta Lovati, Pietro Carlo Lucotti, Francesca Lusetti, Stiliano Maimaris, Jacopo Mambella, Alessandra Martignoni, Federica Melazzini, Chiara Mercanti, Stefania Merli, Giusy Cinzia Moltisanti, Maria Elena Monti, Luisa Morbegno, Francesco Mordà, Amedeo Mugellini, Chiara Muggia, Roberta Muscia, Alba Nardone, Lucia Padovini, Ilaria Palumbo, Ignazio Marzio Parisi, Alessandro Pecci, Giulia Paola Peroo, Clarissa Petrucci, Carla Pieresca, Gianluca Pino, Lavinia Pitotti, Silvia Poma, Paola Stefania Preti, Andrea Quadrelli, Alessandro Rascaroli, Giovanni Rigano, Carlo Maria Rossi, Giorgio Rotola, Damiano Ruggeri, Maria Concetta Russo, Umberto Sabatini, Simone Saglio, Giovanni Santacroce, Gabriele Savioli, Jessica Savioli, Simone Salvator Scalia, Davide Scalvini, Yiftach Shoval, Carlo Soffiantini, Simone Soriano, Dario Spadaro, Andrea Staniscia, Debora Stefani, Federico Vai, Matteo Varallo, and Marta Vernero
DATA AVAILABILITY STATEMENT
The datasets used in this study are available from the corresponding author upon reasonable request. The data are not publicly available due to privacy or ethical restrictions.
REFERENCES
- 1. Liu W, Li H. COVID‐19: Attacks the 1‐beta chain of hemoglobin and captures the porphyrin to inhibit human heme metabolism. ChemRxiv. 2020. 10.26434/chemrxiv.11938173.v7 [DOI] [Google Scholar]
- 2. Daniel Y, Hunt BJ, Retter A, Henderson K, Wilson S, Sharpe CC, et al. Haemoglobin oxygen affinity in patients with severe COVID‐19 infection. Br J Haematol. 2020;190(3):e126–7. 10.1111/bjh.16888 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Vogel DJ, Formenti F, Retter AJ, Vasques F, Camporota L. A left shift in the oxyhaemoglobin dissociation curve in patients with severe coronavirus disease 2019 (COVID‐19). Br J Haematol. 2020;191(3):390–3. 10.1111/bjh.17128 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Ceruti S, Minotti B, Glotta A, Biggiogero M, Bona G, Marzano M, et al. temporal changes in the oxyhemoglobin dissociation curve of critically Ill COVID‐19 patients. J Clin Med. 2022;11(3):788. 10.3390/jcm11030788 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Valle A, Rodriguez J, Camiña F, Rodriguez‐Segade M, Ortola JB, Rodriguez‐Segade S. The oxyhaemoglobin dissociation curve is generally left‐shifted in COVID‐19 patients at admission to hospital, and this is associated with lower mortality. Br J Haematol. 2022;199(3):332–8. 10.1111/bjh.18431 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Myburgh JA, Webb RK, Worthley LI. The P50 is reduced in critically ill patients. Intensive Care Med. 1991;17(6):355–8. 10.1007/BF01716196 [DOI] [PubMed] [Google Scholar]
- 7. Morgan TJ. The oxyhaemoglobin dissociation curve in critical illness. Crit Care Resusc. 1999;1(1):93–100. [PubMed] [Google Scholar]
- 8. Doyle DJ. A simple method to calculate P50 from a single blood sample. Int J Clin Monit Comput. 1997;14:109–11. 10.1007/BF03356585 [DOI] [PubMed] [Google Scholar]
- 9. Severinghaus JW. Simple, accurate equations for human blood O2 dissociation computations. J Appl Physiol Respir Environ Exerc Physiol. 1979;46(3):599–602. 10.1152/jappl.1979.46.3.599 [DOI] [PubMed] [Google Scholar]
- 10. Böning D, Kuebler WM, Bloch W. The oxygen dissociation curve of blood in COVID‐19. Am J Physiol Lung Cell Mol Physiol. 2021;321(2):L349–57. 10.1152/ajplung.00079.2021 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Gille T, Sesé L, Aubourg E, Fabre EE, Cymbalista F, Ratnam KC, et al. The affinity of hemoglobin for oxygen is not altered during COVID‐19. Front Physiol. 2021;12:578708. 10.3389/fphys.2021.578708 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Amato MB, Barbas CS, Medeiros DM, Magaldi RB, Schettino GP, Lorenzi‐Filho G, et al. Effect of a protective‐ventilation strategy on mortality in the acute respiratory distress syndrome. N Engl J Med. 1998;338(6):347–54. 10.1056/NEJM199802053380602 [DOI] [PubMed] [Google Scholar]
- 13. Bergamaschi G, Borrelli de Andreis F, Aronico N, Lenti MV, Barteselli C, Merli S, et al. Anemia in patients with Covid‐19: pathogenesis and clinical significance. Clin Exp Med. 2021;21(2):239–46. 10.1007/s10238-020-00679-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Yang SC, Puri VK, Raheja R. Oxygen delivery and consumption and P50 in patients with acute myocardial infarction. Circulation. 1986;73(6):1183–5. 10.1161/01.cir.73.6.1183 [DOI] [PubMed] [Google Scholar]
- 15. da Luz PL, Cavanilles JM, Michaels S, Weil MH, Shubin H. Oxygen delivery, anoxic metabolism and hemoglobin‐oxygen affinity (P50) in patients with acute myocardial infarction and shock. Am J Cardiol. 1975;36(2):148–54. 10.1016/0002-9149(75)90518-4 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Figure S1.
Table S1.
Table S2.
Table S3.
Table S4.
Table S5.
Table S6.
Table S7.
Table S8.
Table S9.
Data S1.
Data Availability Statement
The datasets used in this study are available from the corresponding author upon reasonable request. The data are not publicly available due to privacy or ethical restrictions.


