Abstract
Background
Cancer is the leading cause of death in Taiwan. Medical expenditures related to cancer accounted for 44.8% of all major illness insurance claims in Taiwan. Prior research has indicated that the dual presence of cancer and mental disorder in patients led to increased medical burden. Furthermore, patients with cancer and concomitant mental disorder could incur as much as 50% more annual costs than those without. Although previous studies have investigated the utilization of patients with both diseases, the effects of morbidity sequence order on patient costs are, however, uncertain. This study explored medical expenditures linked with the comorbidity of cancer and mental disorder, with a focus on the impact of diagnosis sequence order.
Methods
This population-based retrospective matched cohort study retrieved patients with cancer and mental disorder (aged ≥ 20 years) from the Ministry of Health and Welfare Data Science Center 2005–2015 database. 321,045 patients were divided based on having one or both diseases, as well as on the sequence of mental disorder and cancer diagnosis. Study subjects were paired with comparison counterparts free of both diseases using Propensity Score Matching at a 1:1 ratio. Annual Cost per Patient Linear Model (with a log-link function and gamma distribution) was used to assess the average annual cost, covarying for socio-demographic and clinical factors. Binomial Logistic Regression was used to evaluate factors associated with the risk of high-utilization.
Results
The “Cancer only” group had higher adjusted mean annual costs (NT$126,198), more than 5-times that of the reference group (e^β: 5.45, p < 0.001). However, after exclusion of patients with non-cancer and inclusion of diagnosis sequence order for patients with cancer and concomitant mental disorder, the post-cancer mental disorder group had the highest expenditures at over 13% higher than those diagnosed with only cancer on per capita basis (e^β: 1.13, p < 0.001), whereas patients with cancer and any pre-existing mental disorder incurred lower expenditures than those with only cancer. The diagnosis of post-cancer mental disorder was significantly associated with high-utilization (OR = 1.24; 95% CI: 1.047–1.469). Other covariates associated with high-utilizer status included female sex, middle to old age, and late stage cancer.
Conclusion
Presence of mental disorder prior to cancer had a diminishing effect on medical utilization in patients, possibly indicating low medical compliance or adherence in patients with mental disorder on initial treatments after cancer diagnosis. Patients with post-cancer mental disorder had the highest average annual cost. Similar results were found in the odds of reaching high-utilizer status. The follow-up of cancer treatment for patients with pre-existing mental disorders warrants more emphasis in an attempt to effectively allocate medical resources.
Supplementary Information
The online version contains supplementary material available at 10.1186/s12913-023-09056-9.
Keywords: Cancer, Mental disorder, Diagnosis sequence, Medical expenditures, High-utilization
Introduction
Healthcare expenditures continue to grow at a fast pace around the world. U.S. National Health Expenditures grew5% annually from 2005 to 2017, and stands at 3.3 trillion dollars or 17.9% of GDP [1]. US health care spending increased 4.6 percent to reach $3.8 trillion in 2019, similar to the rate of growth of 4.7 percent in 2018. The share of the economy devoted to health care spending was 17.7 percent in 2019 compared with 17.6 percent in 2018 [2, 3]. Conditions in Taiwan are similar: national healthcare expenditures have increased at an average annual rate of 3.6% [4]. Meanwhile, NHI revenue has not kept pace with the growth in expenditures. In 2017, NHI expenses grew 5.5% while revenue only increased 0.5%, and in 2016 revenue actually fell 8.4% while expenses grew 5.6% [4]. Furthermore, over half of all major illness inpatient expenditures in Taiwan is spent for the treatment of just two diseases: cancer(46%) and mental disorder (MD) (11%) [4].
Cancer is the second leading cause of death, and is estimated to be responsible for 9.8 million deaths in 2018or 16.7% of all deaths worldwide [5]. In Taiwan, it ranks as the number 1 cause of mortality, where annual cancer deaths currently account for 28.2% of all deaths [6].The World Health Organization(WHO) also states “the number of new cases is expected to rise by about 70% over the next 2 decades”. Description of past research that individuals are likely to simultaneously suffer from both malignant neoplasms and mental illness. Psychiatric issues were found present in 20%, up to as much as 50%, of cancer patients [7–10]. These study findings suggest that the prevalence of mental disorder among cancer cohorts is consistently much higher than in the general population. Mental disorders encompass different illnesses that are defined by the WHO as being characterized by “abnormal thoughts, perceptions, emotions, behavior and relationships with others.” The WHO also states that individuals with either depression or schizophrenia have a 40 to 60% higher chance of premature death as compared to the general population [5] caused by somatic illnesses such as cancer. In Taiwan, the prevalence of mental illness has risen from 8.8% just ten years ago to 11% in 2016 and is expected to continue to rise [6–11].
The Organisation for Economic Co-operation and Development estimates that cancer, mental health, along with circulatory disease, account for over 40% of all hospital spending, and “cancer is the second most important disease group accounting for around 13% of hospital spending [12].The total annual economic cost of cancer was estimated to be US$ 1.16 trillion in the WHO’s Annual Cancer Report 2018 [5]. In Taiwan, the medical utilization of cancer occupies nearly 40% of all major illness insurance claims (Outpatient: 33.6%, Inpatient: 45.7%) [6]. The dual presence of both cancer and mental disorder in patients leads to increased more expenditures. Past research has shown a disparity in the monetary burden between cancer patients with poor mental health and those without. The increase can be as large as24% to over 50% [13–15]. These large and adverse effects of comorbidity with mental disease merits further research involving cost analysis.
Cancer patients have been shown to be more likely to develop psychiatric disorders [16–18], and the combined cost of cancer with concomitant mental disorder is not an additive equation, but an exponential one [19]. Due to the heavy weighting credited to cancer and mental illness in the profile of healthcare expenditures [4], it would be worthwhile to investigate the costs associated with these disorders [20–23]. Despite the growing importance, there remains a lack of research looking at the intersection of cancer and psychiatric disorder. There has especially been scarce research dividing the comorbid cohort by sequence of diagnosis and conducting a directional analysis. This study attempts to close the knowledge gap in researching the effects of the temporal order of the onset of these two diseases on patient utilization. It’s also one of the first cost analysis studies involving these two important diseases with propensity score matching to pair the study cohort with a comparison cohort. With the depth of information available in the databases of the Taiwan Ministry of Health and Welfare, insights extracted could prove beneficial in enacting policies for resource reallocation, and potentially work toward reducing the economic burden on already strained national funds. Especially as healthcare delivery administrators try to strike a balance between patients’ prolonged survival, quality of life, and medical expenditures [24].
Materials and methods
Our research is using National Health Insurance Research Database (NHIRD) from the Ministry of Health and Welfare (MOHW) Data Science Center 2005–2015, this retrospective matched-cohort study retrieves patients with cancer and/or mental disorder aged over 20 years. Target patients are divided into 3 groups based on diagnosis of either cancer or MD or both. All patients are then matched with a counterpart free of both cancer and MD, forming the comparison counterpart group. A second framework involving cancer patients with concomitant MD compares different diagnosis sequence order groups with cancer patients without MD.
Data sources
Established in 1995, NHIRD is the most complete electronic health record in Taiwan. The NHIRD covers more than 99.6% of the Taiwanese population, and contains demographic variables, outpatient and inpatient information, prescriptions, diagnosis information, medical personnel information and other detailed clinical information [25]. Our data sources were six types of registration files, Registry for Beneficiaries, Ambulatory Care Expenditures, Inpatient Expenditures by Admissions, Personal Attribute file, Hospital Assessment file and Cancer Registry file. Our study was approved by the Research Ethics Committee of China Medical University Hospital, Taiwan, for protect the patients’ privacy, all personal identification numbers were encrypted by the National Health Research Institutes before the data were released. The Taiwan National Health Research Institutes encrypts patient personal information to protect privacy and provides researchers with anonymous scrambled identification numbers associated with relevant diseases information. Therefore, patient informed consent is not required for authorized researchers to access this research database.
Inclusion and exclusion criteria
Our study retrieves subjects and inclusion with cancer from the “Cancer Registry File” (diagnostic code ICD-9-CM: 140.xx to 208.xx excluding Kaposi sarcoma, and gender specific genital cancers) and with mental disorder using diagnostic code 290.xx to 319.xx. Main exclusion criteria include the following: Events occurring in the first 2 years of the data set (2005–2006), due to the inability to determine first instance of diagnosis; Patients with unconfirmed diagnosis, i.e. only1diagnosis; Minors below the age of 20; Patients with missing data or examining the average annual cost per patient (ACPP), and high-utilizer analysis, patients without a full year of cost data was excluded in order to ensure a uniform observation period. Therefore, an additional exclusion criterion was included: Disease duration less than 1 year (from diagnosis to present date or death). Lastly, propensity score matching (PSM) is used to match subjects from each case cohort with control counterparts (free of both cancer and mental disorder) on a 1:1 ratio. Matching was done based on the following 5 variables: sex, age, comorbidity index, salary-based premium, and level of urbanization. The detailed study population flow chart can be found in Figure S1.
High-utilizers
The dual presence of both diseases often leads to patients being in the upper 10% of medical cost spenders, labeled as “high utilizers” (HU). Robinson et al. (2016) found that this group in the top tenth percentile utilize 40% of total annual costs, and that HUs on average incur 4.4 to 9.7 times the cost of non-HUs. In a Medicare-based study, it was found that high-cost patients were much more likely to have a mental health diagnosis (16.0% vs. 6.4%), and among patients segmented as “chronic”, the top 10% of spenders were likely cancer patients [26, 27].
Charlson comorbidities index
Our research used the Charlson Comorbidity Index (CCI) developed by Charlson in 1984 to evaluate the mortality risk and burden of disease, address the confounding influence of comorbidities, and predict outcomes. We followed the method proposed by Charlson; the CCI consists of 17 comorbidities, weighted from 1 to 6 according to mortality risk and disease severity, and then summed scores to form the total CCI score [28]. However, subjects rarely displayed high CCI scores in our research, so we divided the CCI categories into three groups as follows: 0-1point, 2 point, 3 point and 4 or above points.
Statistical analysis
Our research used descriptive statistics, namely frequencies and percentages, to understand the sample distribution with respect to each variable. Means and standard deviations were used to represent continuous data which are average annual costs per capita and end-of-life costs. All cost data is discounted forward to 2018 dollars at a rate of 2% per annum. 2% was chosen as it most closely resembles Taiwan inflation rates averaged over the period from year 2005 to 2018 [29]. Because cost data is generally non-normal and right-skewed, Generalized Linear Model (GLM) with a log-link function (providing multiplicative instead of additive covariate effects) and gamma distribution is used to conduct multivariate analysis. The model outputs parameter estimates for categorical independent variables, which can be exponentiated to compare and show the ratio of the costs of one variable group to that of another group. i.e.:
where β is the variable coefficient, is the estimated sample average of group 1 and is the estimated average of the reference group. In other words, if the exponent of β1 = 1.5, then the cost of those in the target group is 1.5 times, or 50% higher than, that of the reference group. In addition to cost ratios, the model also provides estimated mean costs for each target cohort. The intercept and the variable coefficient summed and exponentiated to directly arrive at estimated mean expenditure for that group. Statistical significance is set at α = 0.05.
Lastly, binomial logistic regression was used to interpret data between High-Utilizers and non-HUs. Patient factors, such as belonging to which sequence order subgroup and other characteristics, was tested for likelihood of HU status. To detect collinearity, the variance inflation factor (VIF) and tolerance of all predictor variables was calculated. All these analyses were conducted using SPSS 22 software.
Results
The extracted case cohort included 321,045 patients that were diagnosed with either cancer, mental disorder, or both. The case cohort was matched with comparison counterparts for a total of 642,178 patients (Figure S1). Within the case cohort, 301,555 had mental disorder, 11,963 had cancer, and 7,527 had both. Further dividing by sequence order,2,498 patients were diagnosed with pre and post-cancer MD, 2,847 with pre-cancer MD, and 2,182post-cancer MD. Among cancer patients the prevalence of mental disorder was 39% (Table S1). In addition, the majority were female (55.3%) in the study samples. Approximately 24% was aged above 60 years old, and the largest age group was in the 40 to 49 years bracket (23.0%). Most were of married status (79%) and more than half did not attain an education of above high-school level (59%). Majority percentage of patients had premium-based salaries below NT$20,008 per month (63%), and fell within occupation category 1 (44%) living in either the Taipei or Central region (56% combined). Patients within urbanization levels 1 and 2 also combined for over 57% of all patients (Table 1).
Table 1.
Patient sociodemographic characteristics
| Variable | Case Cohort | Comparison Cohort | P | |||
|---|---|---|---|---|---|---|
| no | % | no | % | |||
| Sex | 1 | |||||
| Male | 143,457 | 44.70% | 143,485 | 44.70% | ||
| Female | 177,588 | 55.30% | 177,648 | 55.30% | ||
| Age | 1 | |||||
| 20–29 years | 43,811 | 13.60% | 43,824 | 13.60% | ||
| 30–39 years | 60,678 | 18.90% | 60,683 | 18.90% | ||
| 40–49 years | 73,751 | 23.00% | 73,779 | 23.00% | ||
| 50–59 years | 66,025 | 20.60% | 66,048 | 20.60% | ||
| 60–69 years | 38,831 | 12.10% | 38,844 | 12.10% | ||
| over 70 years | 37,949 | 11.80% | 37,955 | 11.80% | ||
| Marital Status | < 0.01 | |||||
| Single | 64,655 | 20.10% | 66,620 | 20.80% | ||
| Married | 256,390 | 79.90% | 254,425 | 79.20% | ||
| Education Level | < 0.01 | |||||
| Middle school or Below | 133,812 | 41.70% | 130,236 | 40.60% | ||
| Highschool | 102,309 | 31.90% | 99,240 | 30.90% | ||
| College or Above | 84,924 | 26.50% | 91,569 | 28.50% | ||
| Premium-based Salary | 1 | |||||
| $20,008 or below | 204,754 | 63.80% | 204,805 | 63.80% | ||
| $20,009–28,800 | 28,934 | 9.00% | 28,940 | 9.00% | ||
| $28,801–45,800 | 54,994 | 17.10% | 55,016 | 17.10% | ||
| $45,801 or above | 32,363 | 10.10% | 32,372 | 10.10% | ||
| Occupation | < 0.01 | |||||
| Category 1 | 139,703 | 43.50% | 143,596 | 44.70% | ||
| Category 2 | 57,393 | 17.90% | 57,298 | 17.80% | ||
| Category 3 | 53,612 | 16.70% | 55,668 | 17.30% | ||
| Category 4/6 | 67,315 | 21.00% | 62,548 | 19.50% | ||
| Category 5 | 3,022 | 0.90% | 1,935 | 0.60% | ||
| Geographic Region | < 0.01 | |||||
| Taipei | 114,379 | 35.60% | 116,335 | 36.20% | ||
| Northern | 36,674 | 11.40% | 39,157 | 12.20% | ||
| Central | 64,430 | 20.10% | 59,816 | 18.60% | ||
| Southern | 44,567 | 13.90% | 44,368 | 13.80% | ||
| Kaohsiung/Ping Tung | 52,951 | 16.50% | 52,912 | 16.50% | ||
| Eastern | 8,044 | 2.50% | 8,457 | 2.60% | ||
| Level of Urbanization | 1 | |||||
| 1 (highest) | 89,445 | 27.90% | 89,476 | 27.90% | ||
| 2 | 93,825 | 29.20% | 93,857 | 29.20% | ||
| 3 | 54,511 | 17.00% | 54,520 | 17.00% | ||
| 4 | 47,272 | 14.70% | 47,278 | 14.70% | ||
| 5 | 6,661 | 2.10% | 6,662 | 2.10% | ||
| 6 | 14,134 | 4.40% | 14,138 | 4.40% | ||
| 7 (lowest) | 15,197 | 4.70% | 15,202 | 4.70% | ||
| CCI | 1 | |||||
| 0–1 | 227,629 | 70.90% | 227,718 | 70.90% | ||
| 2 | 47,096 | 14.70% | 47,102 | 14.70% | ||
| 3 | 23,679 | 7.40% | 23,677 | 7.40% | ||
| 4 or above | 22,641 | 7.10% | 22,636 | 7.10% | ||
| Hospital Level | < 0.01 | |||||
| Medical Center | 59,128 | 18.40% | 23,267 | 7.20% | ||
| Regional | 75,257 | 23.40% | 23,541 | 7.30% | ||
| District | 35,456 | 11.00% | 18,547 | 5.80% | ||
| Clinic | 151,204 | 47.10% | 255,690 | 79.60% | ||
| Hospital Ownership | < 0.01 | |||||
| Public | 60,078 | 18.70% | 15,733 | 4.90% | ||
| Private | 181,144 | 56.40% | 262,141 | 81.70% | ||
| Consortium | 73,508 | 22.90% | 41,787 | 13.00% | ||
| Association | 6,315 | 2.00% | 1,384 | 0.40% | ||
Average annual cost per patient
We used GLM to analyze cancer, MD patients’ costs (Table 2), found that without incorporating diagnose sequence considerations between cancer and MD, the average annual expenditure of cancer patients without any mental disorder diagnosis is more than five-fold that of the reference group (e^β = 5.45, p-value: < 0.001). Patients with both cancer and MD incur annual expenditures that are 3.5-times higher than reference group patients (e^β = 3.55, p-value: < 0.001), The mental disorder only cohort had slightly higher average annual costs, at 1.59 times the cost of the reference cohort. In addition, females incur annual costs 1.04 times higher than males. Using the lowest age group as reference, those aged 60–69 years and over 70 years incur more than double the cost, while those in the 50–59 age group incur 77% more costs. Married patients have slightly reduced average annual expenditures than single patients (e^β = 0.92, p-value: < 0.001). As for education level, with the lowest level (middle school or below) as reference, the high-school group had a very small significant difference (e^β = 0.99, p-value: 0.049). The average annual expenditure of college or above group is less than five-fold that of the reference group (e^β = 0.92, p-value: < 0.001). The group of patients in the lowest premium-based salary bracket had the lowest costs but differences were very small between brackets (e^β = 1.02 to 1.03). Cohort geographic region, residence level of urbanization and occupation also resulted in very small significant differences (e^β = 0.91 to 1.31). The CCI groups of patients with levels of 2, 3 and 4 or above incurred annual costs at over 1.75, 2.39 and 4.44 times the costs of the reference group (CCI level 0–1). Finally, Medical Center hospitals and private hospitals received patients incurring the highest average cost per year.
Table 2.
Effect of diagnosis and other covariates on expenditures
| Variable | e^β | p-value | est. Mean |
|---|---|---|---|
| Cohort | |||
| Cancer & MD | 3.55 | < .001 | 112,757 |
| Cancer only | 5.45 | < .001 | 173,349 |
| MD only | 1.59 | < .001 | 50,435 |
| Free of both | 1 | –– | 31,802 |
| Sex | |||
| Female | 1.04 | < .001 | 76,193 |
| Male | 1 | –– | 73,486 |
| Age | |||
| 20–29 years | 1 | –– | 46,736 |
| 30–39 years | 1.15 | < .001 | 53,831 |
| 40–49 years | 1.4 | < .001 | 65,332 |
| 50–59 years | 1.77 | < .001 | 82,701 |
| 60–69 years | 2.36 | < .001 | 110,107 |
| over 70 years | 2.51 | < .001 | 117,281 |
| Marital Status | |||
| Single | 1 | –– | 78,108 |
| Married | 0.92 | < .001 | 71,685 |
| Education Level | |||
| Middle school or below | 1 | 75,669 | |
| High school | 0.99 | 0.049 | 75,173 |
| College or above | 0.97 | < .001 | 73,655 |
| Premium-based Salary | |||
| $20,008 or below | 1 | –– | 73,505 |
| $20,009–28,800 | 1.03 | < .001 | 75,953 |
| $28,801–45,800 | 1.02 | < .001 | 74,722 |
| $45,801 or above | 1.02 | < .001 | 75,150 |
| Occupation | |||
| Category 1 | 1 | –– | 70,592 |
| Category 2 | 1.06 | < .001 | 74,978 |
| Category 3 | 0.96 | < .001 | 67,932 |
| Category 4/6 | 1 | 0.94 | 70,611 |
| Category 5 | 1.31 | < .001 | 92,399 |
| Geographic Region | |||
| Taipei | 1 | –– | 72,291 |
| Northern | 0.99 | 0.004 | 71,381 |
| Central | 1.08 | < .001 | 77,745 |
| Southern | 1.05 | < .001 | 76,228 |
| Kaohsiung | 1.05 | < .001 | 75,637 |
| Eastern | 1.05 | < .001 | 75,889 |
| Level of Urbanization | |||
| 1 (highest) | 1 | –– | 78,591 |
| 2 | 0.97 | < .001 | 75,950 |
| 3 | 0.98 | < .001 | 77,320 |
| 4 | 0.95 | < .001 | 74,712 |
| 5 | 0.91 | < .001 | 71,862 |
| 6 | 0.91 | < .001 | 71,503 |
| 7 (lowest) | 0.94 | < .001 | 74,133 |
| Charlson Comorbidity Index | |||
| 0–1 | 1 | –– | 36,030 |
| 2 | 1.75 | < .001 | 63,129 |
| 3 | 2.39 | < .001 | 86,141 |
| 4 or above | 4.44 | < .001 | 160,007 |
| Hospital Level | |||
| Medical Center | 1 | –– | 87,755 |
| Regional | 0.87 | < .001 | 76,309 |
| District | 0.82 | < .001 | 71,806 |
| Clinic | 0.74 | < .001 | 65,197 |
| Hospital Ownership | |||
| Public | 1 | –– | 71,612 |
| Private | 1.05 | < .001 | 75,547 |
| Consortium | 1.01 | 0.234 | 72,044 |
| Association | 1.12 | < .001 | 80,434 |
Results of the effects of cancer & MD diagnosis sequence, after adjusting for cancer type, cancer stage, and other covariates on annual expenditures using GLM (Table 3). The cancer group without MD was the reference group and had the second highest average ACPP. Those with a post-cancer MD diagnosis incurred the highest average annual costs spending over 13% more than the cancer only group (e^β = 1.13, p-value: < 0.001). The two other groups, with pre- & post-cancer MD diagnosis, and pre-cancer MD diagnosis, both incurred lower average annual costs as compared to the reference group (e^β = 0.60, p-value: < 0.001; e^β = 0.46, p-value: < 0.001). The pre-cancer MD group had the lowest costs, at less than half that of the reference group.
Table 3.
Effect of diagnosis and cancer covariates on expenditures
| Variable | e^β | p-value | est. Mean |
|---|---|---|---|
| Cohort | |||
| Pre & Post-Cancer MD | 0.60 | < .001 | 65,752 |
| Pre-Cancer MD | 0.46 | < .001 | 51,163 |
| Post-Cancer MD | 1.13 | < .001 | 124,591 |
| Cancer Only(ref) | 1 | –– | 110,160 |
Cancer cohort high-utilizer
Out of the total 19,490 study subjects with a cancer diagnosis, the upper 10% of cost utilizers accounted for 41% of all expenditures. The group with post-cancer MD had the highest percentage of HU: 13%. The cancer only group had the second highest at 9% with HU status. The other 2 groups, pre & post-cancer MD and pre-cancer MD, had the least patients classified as HU (S3). To detect collinearity, the VIF and tolerance of predictor variables was calculated. Variance inflation factors ranged from 1.01958 to 1.58202, well below the accepted level of 10, while the minimum tolerance was 0.6321. Cancer patients with a concomitant MD diagnosis prior to cancer was significantly associated with decreased odds of HU status under adjusted binomial logistic regression (Table 4). MD diagnosis post-cancer was significantly associated with increased odds of HU status. Compared to the reference group (Cancer Only) those with both pre & post-cancer MD diagnosis and those with pre-cancer MD diagnosis had the lowest association with high-utilization (aOR: 0.191 [95% CI: 0.141—0.258], aOR: 0.082 [95% CI: 0.056—0.120], respectively). The post-cancer MD group had an adjusted odds ratio of 1.240 (95% CI: 1.047—1.469). In addtiion, liver and bile duct cancers, colorectal cancer, and oral cancer were all significantly associated with lowered risk of HU status as compared to other cancers. Lung cancer and breast cancer patients had significantly higher risk of being a HU (aOR: 2.139 [95% CI: 1.808- 2.532]; AOR: 1.431 [95% CI: 1.173–1.746]). Later cancer stage was significantly associated with higher HR risk. Other covariates significantly associated with high-utilizer status include the female sex (aOR: 0.819, 95% CI: 0.712- 0.942), and middle to old age from 30 to 69 years old (aOR: 2.330 to 2.704). CCI level3 was significantly associated with lowered risk of HU status as compared to CCI of 2, although the difference was small (aOR: 0.803, 95% CI: 0.669- 0.965), while CCI of 4 or above did not have significantly different risks of HU.
Table 4.
Predictors of cancer cohort HU status
| Variable | cOR | 95% CI | aOR | 95% CI |
|---|---|---|---|---|
| Cohort | ||||
| Pre & Post-Cancer MD | 0.171** | 0.129—0.227 | 0.191** | 0.141—0.258 |
| Pre-Cancer MD | 0.089** | 0.062—0.127 | 0.082** | 0.056—0.120 |
| Post-Cancer MD | 0.945 | 0.814—1.096 | 1.240* | 1.047—1.469 |
| Cancer Only(ref) | 1 | –– | 1 | –– |
| Sex | ||||
| Female (ref) | 1 | –– | 1 | –– |
| Male | 0.783** | 0.707- 0.867 | 0.819** | 0.712—0.942 |
| Age | ||||
| 20–29 years (ref) | 1 | –– | 1 | –– |
| 30–39 years | 2.480** | 1.241—4.950 | 2.704** | 1.798—5.549 |
| 40–49 years | 2.398** | 1.221—4.712 | 2.563** | 1.872—5.202 |
| 50–59 years | 2.696** | 1.377—5.278 | 2.696** | 1.932—5.481 |
| 60–69 years | 2.416** | 1.231—4.744 | 2.330** | 1.969—4.766 |
| over 70 years | 1.906* | 1.014—3.746 | 1.487 | 0.723—3.059 |
| Marital Status | ||||
| Single (ref) | 1 | –– | 1 | –– |
| Married | 0.772* | 0.656—0.908 | 0.86 | 0.712—1.038 |
| Education Level | ||||
| Middle school or Below | 0.921 | 0.806—1.051 | 1.176 | 0.991—1.395 |
| Highschool | 1.003 | 0.864—1.166 | 1.074 | 0.909—1.270 |
| College or Above | 1 | –- | 1 | –– |
| Premium-based Salary | ||||
| $20,008 or below | 0.914 | 0.934—1.514 | 0.978 | 0.778—1.230 |
| $20,009–28,800 | 1.340* | 0.798—1.425 | 1.266 | 0.977—1.639 |
| $28,801–45,800 | 1.164 | 0.631—1.146 | 1.164 | 0.945—1.435 |
| $45,801 or above (ref) | 1 | –– | 1 | –– |
| Occupation | ||||
| Category 1 (ref) | 1 | –– | 1 | –– |
| Category 2 | 1.21 | 1.021—1.433 | 1.073 | 0.879—1.309 |
| Category 3 | 0.962* | 0.811—1.141 | 1.118 | 0.914—1.369 |
| Category 4/6 | 1.376** | 1.192—1.589 | 1.222* | 1.005—1.485 |
| Category 5 | 1.502 | 0.772—2.924 | 1.561 | 0.753—3.237 |
| Geographic Region | ||||
| Taipei (ref) | 1 | –– | 1 | –– |
| Northern | 0.929 | 0.777—1.110 | 1.069 | 0.871—1.313 |
| Central | 1.195* | 1.042—1.371 | 1.117 | 0.867—1.682 |
| Southern | 0.847* | 0.724—0.995 | 1.173 | 0.967—1.423 |
| Kaohsiung Pingtung | 0.792* | 0.676—0.926 | 0.864 | 0.723—1.033 |
| Eastern | 0.968 | 0.703—1.333 | 1.271 | 0.888—1.818 |
| Level of Urbanization | ||||
| 1 (highest) (ref) | 1 | –– | 1 | –– |
| 2 | 0.837* | 0.732—0.958 | 0.800* | 0.685—0.935 |
| 3 | 0.953 | 0.769—1.109 | 0.879 | 0.733—1.054 |
| 4 | 0.85 | 0.711—1.001 | 0.822 | 0.669—1.010 |
| 5 | 0.571* | 0.870—0.829 | 0.640* | 0.417—0.984 |
| 6 | 0.582* | 0.822—0.785 | 0.584* | 0.584—0.833 |
| 7 (lowest) | 0.99 | 1.093—1.244 | 1.093 | 0.837—1.426 |
| CCI | ||||
| 2 (ref) | 1 | –– | 1 | –– |
| 3 | 0.768* | 0.644—0.916 | 0.803* | 0.669—0.965 |
| 4 or above | 0.790* | 0.686—0.911 | 0.902 | 0.769—1.058 |
| Cancer Type | ||||
| Lung and Bronchus | 2.018** | 1.219—1.821 | 2.139** | 1.808—2.532 |
| Liver and Bile Duct | 0.690** | 0.584—0.887 | 0.812* | 0.665—0.992 |
| Colorectal | 0.531** | 0.695—1.005 | 0.561** | 0.459—0.685 |
| Breast | 1.212* | 0.266—0.455 | 1.431** | 1.173—1.746 |
| Oral Cavity | 0.685** | 1.157—1.717 | 0.669** | 0.548—0.818 |
| Others (ref) | 1 | –– | 1 | –– |
| Cancer Stage | ||||
| Stage 0-I(ref) | 1 | –– | 1 | |
| Stage II | 1.994** | 1.685—2.361 | 2.018** | 1.690—2.408 |
| Stage III | 2.608** | 2.212—3.074 | 3.236** | 2.713—3.859 |
| Stage IV | 3.445** | 2.954—4.017 | 3.529** | 2.979—4.181 |
| Hospital Level | ||||
| Medical Center | 3.020** | 2.513—3.628 | 1.228 | 0.956—1.577 |
| Regional | 2.215** | 1.827—2.686 | 0.906 | 0.702—1.171 |
| District | 1.638** | 1.229—2.185 | 1.067 | 0.776—1.467 |
| Clinic (ref) | 1 | –– | 1 | –– |
| Hospital Ownership | ||||
| Public (ref) | 1 | –– | 1 | –– |
| Private | 0.697** | 0.605—0.805 | 1.103 | 0.925—1.315 |
| Consortium | 1.092 | 0.965—1.235 | 1.14 | 0.998—1.302 |
| Association | 1.074 | 0.809—1.426 | 1.156 | 0.838—1.594 |
*p-value < 0.05
**p-value < 0.001
End of life costs
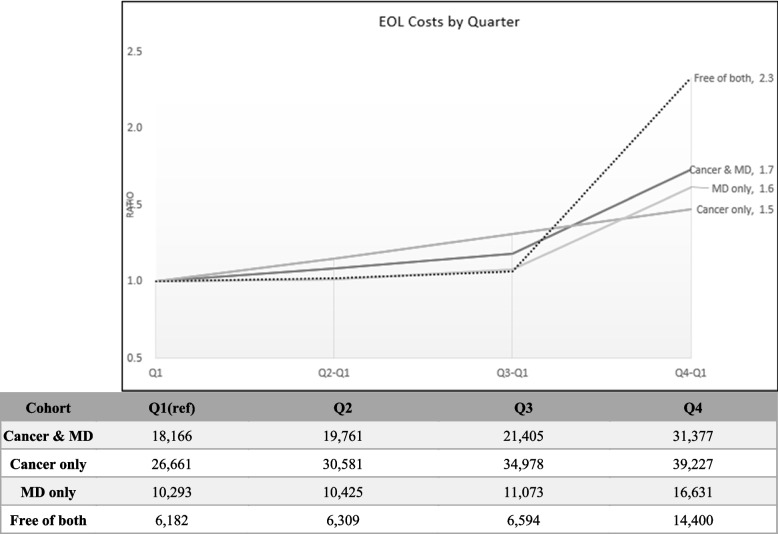
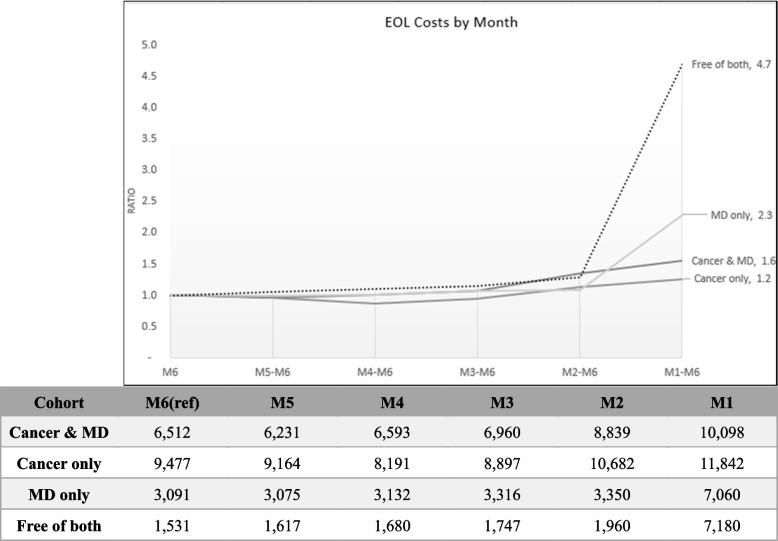
At the end-of-life (EOL), the median quarterly cost of patients free of both disorders increased at the fasted pace among cohorts, where final quarter costs were 2.3 times higher than the first quarter (Fig. 1). Followed by the Cancer & MD groups, and the MD only group, while the cancer only group had the slowest increases in quarterly EOL costs. In addition, the same results were seen for EOL monthly costs during the final half year of life of patients, as shown in Fig. 2. Patient’s free of both disorders increased at the fasted pace among cohorts, where final quarter costs were 4.7 times higher than the first quarter, followed by the MD only group and cancer & MD group. The cancer only group had the slowest increases in quarterly EOL costs, where the final month median cost was only 1.2 times that of the 6th month before death.
Fig. 1.
Median EOL Costs by Quarter
Fig. 2.
Median EOL Costs by Month
Discussion
The presence of MD had a diminishing effect on utilization. Cancer patients with MD had lower costs than those without concomitant MD. This was different from expectations and contrary to results of past research looking at dollar amounts in measuring patient utilization. However, the results of this study are supported by two papers, both reporting inadequate treatment in cancer patients with MD. Davis et al. (2022), in a study examines pancreatic cancer treatment compliance in patients with prodromal depression or anxiety found that patients with prodromal depression or anxiety were significantly less likely to receive chemotherapy [30]. A second paper focused on psychiatric illness patients on colorectal cancer treatments found that individuals with a severe psychiatric illness (SPI) history had significantly less likely to receive guideline recommended treatment than colorectal cancer (CRC) patients with no history of MD. Stage II and III CRC patients with SPI history were 2.15 times less likely (95% CI 1.07–4.33) to receive potentially curative surgical resection and 2.07 times less likely (95% CI 1.72–2.50) to receive adjuvant radiation or chemotherapy [31]. Unfortunately, the reasons behind the inadequacies of treatment of cancer patients with MD cannot be interpreted directly from claims data [32]. Nonetheless, these results could indicate the adverse situation of MD patients in Taiwan in experiencing potential barriers to healthcare (non-adherence, unwillingness to seek and comply with treatment or delays in care), or a dilution effect in the treatment of MD instead of patients’ major chronic disease. In addition, authors speculate the lowered utilization of cancer patients with psychiatric morbidity could also stem from the adverse behavioral effects caused by mental disorder. To have a deeper understanding of how mental disorder negatively impacts adherence, we turn to studies directly assessing correlates of mental disease with patient attitudes toward treatment. Past research revealed 3 major views held by patients associated with MD, which acted as barriers to healthcare; first inadequate information about treatment or misinterpretation of the treatment [33]; secondly, fears about treatment and its side-effects [34]; third, forgetting appointments [35, 36].
Cancer patients with a pre-existing MD had lower expenditures than those without any concomitant MD, while patients with MD onset post-cancer had higher expenditures. This difference in sequence order effect could be caused by the pre-existing mental distress affecting access to treatment in patients during the period immediately after cancer diagnosis, or simply a longer time period with MD in pre-cancer MD patients. Another recent paper on seniors with prostate cancer found that those with prior severe mental illness had lower likelihood of receiving treatment in the first year after diagnosis [37]. For example, the adjusted odds ratio of undergoing surgery was 0.66 (95% CI: 0.49–0.89) and receiving radiation concurrent with hormone therapy was 0.81 (95% CI: 0.67–0.98). Pre-existing mental distress has been found to profoundly affect both delay in treatment and access to treatment, thereby potentially caused the reduced expenditures in this type of patients of this study. Another research disclosed that despite the increased mortality from cancer in people with MD patients, this population receives less cancer screening compared with that of the general population [38]. On the other hand, the set of patients that developed mental disorders post-cancer were found to have higher average annual expenditures per capita than those without MD at any time. [39]The possible reason is that MD will reduce treatment compliance, this result is similar to the previous research, and it is believed that patients with SMI are prone to palliative care; it revealed that SMI patients are prone to palliative care [32]. This also explains why we found that patients in the Post-Cancer MD group were the highest risk to become HUs.
In addition, a research pointed that HU is the group that accounts for the majority of healthcare spending, even disclosed that < 1% of HU in the US account for 22% of US healthcare spending [40]. We found that the costs in "Free of Both" group began to increase after Q3, therefore, we further analyze the time cost trend of EOL. An earlier analysis of Taiwan NHI expenditures comparing EOL costs of various chronic illnesses ranked cancer second in terms of highest final year EOL costs for decedents, but it did not look at disease specific cost trends approaching the final quarters or months [20]. Instead, it analyzed changes in type of medical expense and found that final quarter costs of decedents were dominated by acute inpatient expenses for most chronic diseases. However, other studies focused on cancer patients showed that cancer end-of-life care usually involved palliative or hospice care (ORs ranging 1.79 to 6.88); lower likelihood of intensive care at EOL (ORs ranging 0.26–0.68); lower odds of chemotherapy near death (ORs 0.41, 0.57); lower odds of emergency department use and shorter length of hospital stay [41]. Another research also indicated that individuals with bipolar disorder were more likely to receive palliative care and less likely to receive high-intensity EOL care [42]. Conversely, "Free of both" group had more EOL costs because of progressively complex medical needs [43, 44].
Conclusion
Using a nationally representative database of the population of Taiwan, the study evaluated the expenditures of cancer patients with concomitant mental disorders, with focus on comparing the effects of differences in the sequence of disease onset. Contrary to expectations, the presence of mental disorder could have a diminishing effect on utilization, specifically if the onset of mental disorder was prior to cancer. Patients with post-cancer mental disorders and cancer patients without MD had the highest and second highest costs among groups, respectively. The additional expenditures of cancer patients later diagnosed with concomitant mental disorder may come from more office-visits for mental health care, and excess medication. [45, 46] Patients diagnosed with cancer and pre-existing mental illness had lower expenditures compared to those with only cancer, indicating possible barriers to care. A pre-existing mental disorder reduced expenditures in cancer patients, potentially due to barriers to uptake of treatment caused by psychiatric distress in the period immediately after cancer diagnosis. Those with pre-cancer MD had lower average expenditures, lowered risk of high utilization than patients with post-cancer MD. To the author’s best knowledge, this is the first study to analyze the effects of cancer and mental disorder diagnosis sequence differences on medical expenditure in Taiwan, and the first to distinguish pre & post-cancer, pre-cancer and post-cancer patient group differences in the same study population.
Limitations
Research limitations of this study include; the main source of data is from the NHIRD Database and does not include certain patient variables such as lifestyle factors that potentially influences medical utilization; although lymph node and tumor size, and individually prescribed treatments are available in the database, these variables would not be applicable to all cancers included in the sample; cost data only includes NHIRD reimbursement claims and does not include patient self-pay amounts for additional treatment not covered under the NHIRD; premium-based salary as categorized by the NHIRD database only includes income as reported to the NHIRD Administration for premium calculation and may not reflect true household income; the region reported in the NHIRD database may not be the actual region of utilization as those covered under Taiwan’s national insurance are free to seek treatment nationwide.
Supplementary Information
Additional file 1: Figure S1. Study populations flow chart. Table S1. Number of Patients by Cohort & Cancer Type. Table S2. HU status by cancer cohort.
Acknowledgements
We are grateful to MOHW Health and Welfare Data Science Center at China Medical University for providing support and assistance in administrative, technical and fee discount.
Abbreviations
- ACPP
Annual cost per patient
- CCI
Charlson comorbidity index
- CRC
Colorectal cancer
- EOL
End-of-life
- GLM
Generalized linear model
- HWDC
Welfare data science center
- HU
High-utilizers
- MD
Mental disorder
- MOHW
Ministry of health and welfare
- NHIRD
National health insurance research database
- PSM
Propensity score matching
- SPI
Severe psychiatric illness
- VIF
Variance inflation factor
- WHO
World health organization
Authors’ contributions
K.J.M., S.Y.S., D.N., C.Y.Y. and J.Y.W. designed and conceptualized the study and analyzed the data. K.J.M. drafted the first version of the article. K.J.M., W.S.C. and C.Y.Y. performed the literature search and reviewed the article. All authors contributed substantially to the article and approved the final article for submission. All authors are responsible for the integrity, accuracy, and presentation of the data.
Funding
This study was supported by funding from the Ministry of Science and Technology, Taiwan (Grant no. MOST111-2410-H-039-001-MY2 and MOST110-2410-H-039-001), the China Medical University, Taiwan (Grant no. CMU111-MF-90), and the An-Nan Hospital, Taiwan (Grant no. ANHRF111-09).
Availability of data and materials
The data that support the findings of this study are available from the Health and Welfare Data Science Center, but restrictions apply to the availability of these data, which were used under license for the current study, and so are not publicly available. Data are however available from the authors upon reasonable request and with permission of Health and Welfare Data Science Center.
Declarations
Ethics approval and consent to participate
In this study, all the research database involving humans were conducted ethically and in accordance with the Declaration of Helsinki. Our study protocols were reviewed and approved by the Institutional Review Board of the China Medical University and Hospital (CMUH107-REC3-023). To protect the patients’ privacy, all personal identification numbers were encrypted by the National Health Insurance before the data were analyzed and released. Therefore, patient informed consent is not required for authorized researchers to access this research database. The researchers of this study are not possible to contact any studied patient for obtaining informed consent. No informed consent form is used in this study. Furthermore, the Institutional Review Board of China Medical University (CMUH107-REC3-023) also specifically waived the requirement of informed consent.
Consent for publication
Not applicable.
Competing interests
The authors made no disclosures.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Chi-Yu Yao, Email: d74450@mail.tmanh.org.tw.
Jong-Yi Wang, Email: ericwang@mail.cmu.edu.tw.
References
- 1.Centers for Medicare & Medicaid Services. CMS Office of the Actuary Releases 2017 National Health Expenditures. 2018. Available from: https://www.cms.gov/newsroom/press-releases/cms-office-actuary-releases-2017-national-health-expenditures.
- 2.Centers for Medicare & Medicaid Services. CMS Office of the Actuary Releases 2019 National Health Expenditures. 2020. Available from: https://www.cms.gov/newsroom/press-releases/cms-office-actuary-releases-2019-national-health-expenditures.
- 3.Martin AB, Hartman M, Lassman D, Catlin A. National health care spending in 2019: steady growth for the fourth consecutive year. Health Aff. 2021;40(1):14–24. doi: 10.1377/hlthaff.2020.02022. [DOI] [PubMed] [Google Scholar]
- 4.Ministry of Health and Welfare. National health expenditure data. 2019. Available from: https://dep.mohw.gov.tw/DOS/lp-5071-113.html.
- 5.World Health Organization. Cancer. 2018. Available from: https://www.who.int/health-topics/cancer#tab=tab_1.
- 6.Ministry of Health and Welfare. National health expenditure data. 2018. Available from: https://www.mohw.gov.tw/dl-77019-8cd67219-dfee-4ff0-a8ab-45805bc99fb6.html.
- 7.Zabora J, BrintzenhofeSzoc K, Curbow B, Hooker C, Piantadosi S. The prevalence of psychological distress by cancer site Psycho-oncology. J Psychol Social Behav Dimensions Cancer. 2001;10(1):19–28. doi: 10.1002/1099-1611(200101/02)10:1<19::aid-pon501>3.0.co;2-6. [DOI] [PubMed] [Google Scholar]
- 8.Miovic M, Block S. Psychiatric disorders in advanced cancer. Cancer. 2007;110(8):1665–76. doi: 10.1002/cncr.22980. [DOI] [PubMed] [Google Scholar]
- 9.Derogatis LR, Morrow GR, Fetting J, Penman D, Piasetsky S, Schmale AM, et al. The prevalence of psychiatric disorders among cancer patients. JAMA. 1983;249(6):751–7. doi: 10.1001/jama.1983.03330300035030. [DOI] [PubMed] [Google Scholar]
- 10.Chen ML, Chang HK, Yeh CH. Anxiety and depression in taiwanese cancer patients with and without pain. J Adv Nurs. 2000;32(4):944–51. [PubMed] [Google Scholar]
- 11.Ran M-S, Hall BJ, Su TT, Prawira B, Breth-Petersen M, Li X-H, et al. Stigma of mental illness and cultural factors in Pacific Rim region: a systematic review. BMC Psychiatry. 2021;21(1):1–16. doi: 10.1186/s12888-020-02991-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Organisation for Economic Co-operation and Development. THE ECONOMICS OF PATIENT SAFETY. 2020. Available from: https://www.oecd.org/health/health-systems/Economics-of-Patient-Safety-October-2020.pdf.
- 13.Pan X, Sambamoorthi U. Health care expenditures associated with depression in adults with cancer. J Community Supportive Oncol. 2015;13(7):240. doi: 10.12788/jcso.0150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Shen C, Sambamoorthi U, Rust G. Co-occurring mental illness and health care utilization and expenditures in adults with obesity and chronic physical illness. Dis Manag. 2008;11(3):153–60. doi: 10.1089/dis.2007.0012. [DOI] [PubMed] [Google Scholar]
- 15.Han X, Lin CC, Li C, de Moor JS, Rodriguez JL, Kent EE, et al. Association between serious psychological distress and health care use and expenditures by cancer history. Cancer. 2015;121(4):614–22. doi: 10.1002/cncr.29102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Rashid A, Aqeel M, Malik B, Salim S. The prevalence of psychiatric disorders in breast cancer patients; a cross-sectional study of breast cancer patients experience in Pakistan. Nat Nurt J Psychol. 2021;1(1):1–7. [Google Scholar]
- 17.Park EM, Rosenstein DL. Depression in adolescents and young adults with cancer. Dialogues Clin Neurosci. 2022;17:171. doi: 10.31887/DCNS.2015.17.2/epark. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Walker J, Mulick A, Magill N, Symeonides S, Gourley C, Burke K, et al. Major depression and survival in people with cancer. Psychosom Med. 2021;83(5):410–6. doi: 10.1097/PSY.0000000000000942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Strong V, Waters R, Hibberd C, Murray G, Wall L, Walker J, et al. Management of depression for people with cancer (SMaRT oncology 1): a randomised trial. Lancet. 2008;372(9632):40–8. doi: 10.1016/S0140-6736(08)60991-5. [DOI] [PubMed] [Google Scholar]
- 20.Liu C-N, Yang M-C. National health insurance expenditure for adult beneficiaries in Taiwan in their last year of life. J-Formos Med Assoc. 2002;101(8):552–9. [PubMed] [Google Scholar]
- 21.Lubitz JD, Riley GF. Trends in medicare payments in the last year of life. N Engl J Med. 1993;328(15):1092–6. doi: 10.1056/NEJM199304153281506. [DOI] [PubMed] [Google Scholar]
- 22.Fassbender K, Fainsinger RL, Carson M, Finegan BA. Cost trajectories at the end of life: the canadian experience. J Pain Symptom Manag. 2009;38(1):75–80. doi: 10.1016/j.jpainsymman.2009.04.007. [DOI] [PubMed] [Google Scholar]
- 23.Chastek B, Harley C, Kallich J, Newcomer L, Paoli CJ, Teitelbaum AH. Health care costs for patients with cancer at the end of life. J Oncol Pract. 2012;8(6S):75s–80s. doi: 10.1200/JOP.2011.000469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Eala MAB, Manlongat KD, Dee EC, Baticulon RE, Canal JPA. Mental health care for Filipino patients with cancer. Lancet Oncol. 2021;22(12):e531. doi: 10.1016/S1470-2045(21)00570-2. [DOI] [PubMed] [Google Scholar]
- 25.Lin L-Y, Warren-Gash C, Smeeth L, Chen P-C. Data resource profile: the national health insurance research database (NHIRD) Epidemiol Health. 2018;40:e2018062. doi: 10.4178/epih.e2018062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Joynt KE, Figueroa JF, Beaulieu N, Wild RC, Orav EJ, Jha AK, editors. Segmenting high-cost Medicare patients into potentially actionable cohorts. Healthcare: Elsevier; 2017. [DOI] [PubMed] [Google Scholar]
- 27.Robinson RL, Grabner M, Palli SR, Faries D, Stephenson JJ. Covariates of depression and high utilizers of healthcare: impact on resource use and costs. J Psychosom Res. 2016;85:35–43. doi: 10.1016/j.jpsychores.2016.04.002. [DOI] [PubMed] [Google Scholar]
- 28.Charlson ME, Pompei P, Ales KL, MacKenzie CR. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J chronic Dis. 1987;40(5):373–83. doi: 10.1016/0021-9681(87)90171-8. [DOI] [PubMed] [Google Scholar]
- 29.National Statistics. The Consumer Price Index. 2018. Ministry of Economic Affairs. Available from: https://eng.stat.gov.tw/point.asp?index=2.
- 30.Davis NE, Hue JJ, Kyasaram RK, Elshami M, Graor HJ, Zarei M, et al. Prodromal depression and anxiety are associated with worse treatment compliance and survival among patients with pancreatic cancer. Psycho-Oncology. 2022;31:1390. doi: 10.1002/pon.5945. [DOI] [PubMed] [Google Scholar]
- 31.Mahar AL. The effect of a severe psychiatric illness on colorectal cancer treatment and survival: A population-based retrospective cohort study. PloS one. 2020;15(7):e0235409. doi: 10.1371/journal.pone.0235409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Grassi L. Cancer and severe mental illness: problems and potential solutions. Psycho-oncology (Chichester England) 2020;29(10):1445–51. doi: 10.1002/pon.5534. [DOI] [PubMed] [Google Scholar]
- 33.Ertem MY, Duman Z. The effect of motivational interviews on treatment adherence and insight levels of patients with schizophrenia: a randomized controlled study. Perspect Psychiatr Care. 2019;55(1):75–86. doi: 10.1111/ppc.12301. [DOI] [PubMed] [Google Scholar]
- 34.Achtyes E, Simmons A, Skabeev A, Levy N, Jiang Y, Marcy P, et al. Patient preferences concerning the efficacy and side-effect profile of schizophrenia medications: a survey of patients living with schizophrenia. BMC Psychiatry. 2018;18(1):1–7. doi: 10.1186/s12888-018-1856-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Parsons J, Bryce C, Atherton H. Which patients miss appointments with general practice and the reasons why: a systematic review. Br J Gen Pract. 2021;71(707):e406–e12. doi: 10.3399/BJGP.2020.1017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Eken HN, Dee EC, Powers AR, III, Jordan A. Racial and ethnic differences in perception of provider cultural competence among patients with depression and anxiety symptoms: a retrospective, population-based, cross-sectional analysis. Lancet Psychiatry. 2021;8(11):957–68. doi: 10.1016/S2215-0366(21)00285-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Fried DA, Sadeghi-Nejad H, Gu D, Zhou S, He W, Giordano SH, et al. Impact of serious mental illness on the treatment and mortality of older patients with locoregional high‐grade (nonmetastatic) prostate cancer: retrospective cohort analysis of 49 985 SEER‐Medicare patients diagnosed between 2006 and 2013. Cancer Med. 2019;8(5):2612–22. doi: 10.1002/cam4.2109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Solmi M, Firth J, Miola A, Fornaro M, Frison E, Fusar-Poli P, et al. Disparities in cancer screening in people with mental illness across the world versus the general population: prevalence and comparative meta-analysis including 4 717 839 people. Lancet Psychiatry. 2020;7(1):52–63. doi: 10.1016/S2215-0366(19)30414-6. [DOI] [PubMed] [Google Scholar]
- 39.Chochinov HM, Martens PJ, Prior HJ, Kredentser MS. Comparative health care use patterns of people with schizophrenia near the end of life: a population-based study in Manitoba, Canada. Schizophr Res. 2012;141(2–3):241–6. doi: 10.1016/j.schres.2012.07.028. [DOI] [PubMed] [Google Scholar]
- 40.Fertel BS, Podolsky SR, Mark J, Muir MR, Ladd ME, Smalley CM. Impact of an individual plan of care for frequent and high utilizers in a large healthcare system. Am J Emerg Med. 2019;37(11):2039–42. doi: 10.1016/j.ajem.2019.02.032. [DOI] [PubMed] [Google Scholar]
- 41.Starr LT, Ulrich CM, Corey KL, Meghani SH. Associations among end-of-life discussions, health-care utilization, and costs in persons with advanced cancer: a systematic review. Am J Hospice Palliat Medicine®. 2019;36(10):913–26. doi: 10.1177/1049909119848148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Fond G, Baumstarck K, Auquier P, Pauly V, Bernard C, Orleans V, et al. End-of-life care among patients with bipolar disorder and cancer: a nationwide cohort study. Psychosom Med. 2020;82(7):722–32. doi: 10.1097/PSY.0000000000000839. [DOI] [PubMed] [Google Scholar]
- 43.Kelly EM, James PD, Murthy S, Antonova L, Wong F, Shaw-Stiffel T, et al. Health Care utilization and costs for patients with end-stage liver disease are significantly higher at the end of life compared to those of other decedents. Clin Gastroenterol Hepatol. 2019;17(11):2339–46e1. doi: 10.1016/j.cgh.2019.01.046. [DOI] [PubMed] [Google Scholar]
- 44.Pollock C, James G, Garcia Sanchez JJ, Arnold M, Carrero J-J, Lam CS, et al. Cost of end-of-life inpatient encounters in patients with chronic kidney disease in the United States: a report from the DISCOVER CKD retrospective cohort. Adv therapy. 2022;39(3):1432–45. doi: 10.1007/s12325-021-02010-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Oliva-Moreno J, Trapero-Bertran M, Peña-Longobardo LM, del Pozo-Rubio R. The valuation of informal care in cost-of-illness studies: a systematic review. PharmacoEconomics. 2017;35(3):331–45. doi: 10.1007/s40273-016-0468-y. [DOI] [PubMed] [Google Scholar]
- 46.Tajima-Pozo K, de Castro Oller MJ, Lewczuk A, Montañes-Rada F. Understanding the direct and indirect costs of patients with schizophrenia. F1000Res. 2015;4:182. doi: 10.12688/f1000research.6699.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional file 1: Figure S1. Study populations flow chart. Table S1. Number of Patients by Cohort & Cancer Type. Table S2. HU status by cancer cohort.
Data Availability Statement
The data that support the findings of this study are available from the Health and Welfare Data Science Center, but restrictions apply to the availability of these data, which were used under license for the current study, and so are not publicly available. Data are however available from the authors upon reasonable request and with permission of Health and Welfare Data Science Center.