Abstract
Introduction
Respiratory syncytial virus (RSV) is a leading cause of lower respiratory tract infection (LRTI)-related hospitalizations in older adults. Without RSV-specific treatment for adults, testing is uncommon, leading to potential underestimation of RSV incidence in real-world data studies. This study aimed to quantify the frequency of RSV testing during LRTI-related hospitalizations of older adults to inform interpretation of incidence estimates.
Methods
Administrative and billing data for hospitalizations of adults aged ≥ 65 years with a primary or secondary diagnosis of LRTI during the 2016–2019 RSV seasons (October–April) were extracted from the US all-payer Premier Healthcare Database (PHD). Billing codes identified RSV tests administered during eligible hospitalizations. The proportion of LRTI-related hospitalizations with a billed RSV test was calculated for each hospital in PHD, and summarized descriptively by hospital bed size, teaching status, and population served.
Results
Most of the 937 study hospitals performed RSV testing infrequently during LRTI hospitalization; median percentage of LRTI hospitalizations with RSV testing was 4.3%, and 78.4% of hospitals performed RSV testing in less than 25% of LRTI-related hospitalizations. RSV testing varied extensively by hospital type. Median percentage tested was significantly higher for hospitals with ≥ 200 beds (9.1%) versus < 200 beds (1.6%), for teaching (11.0%) versus non-teaching (2.5%) hospitals, and in urban (7.4%) versus rural (0.7%) settings. The median percentage of RSV testing increased over time, from 0.8% to 6.3% between the 2016/17 and 2018/19 seasons.
Conclusion
A small proportion of older adults hospitalized with LRTI are tested for RSV in US hospitals. Large variability occurs across hospital types. Consequently, retrospective database analyses likely result in a substantial underestimation of the true RSV-related hospitalization incidence. RSV incidence studies using real-world data need to assess for RSV testing frequency and adjust their results for under ascertainment associated with limited testing.
Supplementary Information
The online version contains supplementary material available at 10.1007/s40121-023-00758-5.
Keywords: Lower respiratory tract infection (LRTI), Respiratory syncytial virus (RSV), RSV testing
Key Summary Points
| Accurate estimation of respiratory syncytial virus (RSV) incidence in lower respiratory tract infections (LRTI)-related hospitalizations is crucial for understanding the clinical and economic burden of the disease, particularly among older adults (≥65 years). |
| We used the comprehensive all-payer Premier Healthcare Database (PHD) to quantify the frequency of RSV testing in US hospitals and to inform the interpretation of incidence estimates. |
| Among 937 study hospitals, 78.4% performed RSV testing in less than 25% of LRTI-related hospitalizations, and the median percentage of LRTI hospitalizations with RSV testing was 4.3%. |
| Our findings imply that relying on retrospective database analyses can result in a substantial underestimation of the true RSV incidence as a consequence of limited testing. RSV incidence studies using such data should adjust their interpretations accordingly. |
Introduction
Respiratory syncytial virus (RSV) is a single negative-stranded ribonucleic acid virus with two major antigenic subtypes, A and B, often exhibiting an alternating predominance across different epidemic seasons [1–3]. While RSV infection is common and often self-limited in children [4], RSV can lead to more severe illness among older adults, and is recognized as an important contributor to lower respiratory tract infections (LRTI) [5]. Symptoms of RSV infection in older adults can range from mild to life-threatening, potentially leading to respiratory failure (8–13%) or even death (2–5%) [6]. In the US, 1,360,322 outpatient visits, 119,391 emergency department visits, and 159,247 hospitalizations annually are estimated to be RSV-related, with ~ 13,000 deaths predicted based on a death rate of 8% for hospitalized patients with RSV infection [7]. Since the risk of hospitalization generally increases with age [8], the burden of disease is expected to increase with the aging population and longer life expectancy, further adding to unmet clinical needs.
Currently, there are no approved treatments for RSV in older adults, and supportive care is the standard-of-care. Despite the development of rapid, low-complexity molecular testing options for RSV [9, 10], diagnostic testing may not be prioritized for patients with LRTI, as the results have minimal impact on treatment decision-making. One consequence of low RSV testing frequency is a substantial underestimation of adult RSV-disease incidence rates if derived from real-world data analyses. Prospective epidemiology studies document a high RSV incidence among older adults of 3–7% annually [5]. A systematic literature review collated data on the incidence rate of medically-attended RSV in US adults, and reported a low annual rate of RSV-associated hospitalization in older adults from real-word data ICD code-based analyses (1–5 per 100,000), as compared to a pooled estimate of 188 per 100,000 adults based on prospective studies [7]. Together, these data point to limited standard-of-care testing as a substantial source of RSV incidence underestimation (up to > 100-fold) in laboratory surveillance studies and unadjusted retrospective analyses.
An accurate estimation of the incidence rate of RSV infection in the elderly population is important for understanding the clinical and economic burden of the disease, and to identify populations which may be at greatest need for treatment or prevention of RSV. The accuracy of real-world studies in estimating RSV incidence depends on the amount of RSV testing supporting the diagnosis codes that are reported through real-world data. Therefore, we conducted a descriptive retrospective analysis of US hospital data from adults aged ≥ 65 years who were hospitalized for LRTI, focusing on the three RSV seasons prior to the pandemic (October 1–April 30 for 2016/17, 2017/18, and 2018/19). The primary objective was to quantify the level of RSV testing in this cohort to inform the interpretation of incidence estimates. A secondary objective was to describe associations between levels of testing and hospital characteristics.
Methods
Study Design and Data Source
This retrospective, hospital-based cohort study analyzed all-payer electronic healthcare data from a nationwide network of hospitals captured in the Premier Healthcare Database (PHD). The database includes over 127 million inpatient admissions and more than 11 million annualized admissions since 2012, from 1113 participating hospitals, representing ~ 25% of annual inpatient admissions in the US [11]. Variables on hospital and visit characteristics were available. Further, patient data from standard hospital discharge billing files were available for demographics and disease states, admission and discharge diagnoses, information on billed services including costs, and patient disposition and discharge health status. This study did not require an institutional review board review due to the de-identified nature of patient health information.
Inclusion Criteria and Study Measures
In this study, data on each LRTI-related visit from all hospitals that contributed data to the PHD were extracted for adult inpatients, aged ≥ 65 years, with any International Classification of Diseases, Tenth Revision (ICD-10) diagnosis code (including admitting, primary discharge, and secondary discharge) of LRTI (see Table S1 in the electronic supplementary material) with an admission date during three consecutive RSV seasons (October 1–April 30) between 2016 and 2019. This timeframe represented the years after the ICD-10 crossover (the ICD-10 coding system took effect on October 1, 2015) and prior to the start of the COVID-19 pandemic in late 2019, as COVID-19 would have confounded the RSV testing and hospitalization frequencies. An exploratory analysis was performed during the post-COVID period (2019/20 and 2020/21) and are described in the electronic supplementary material (Appendix B).
This analysis did not account for readmissions. In other words, each inpatient admission was considered to be an independent event, regardless of whether the patient had been readmitted. However, demographic information, such as age, sex, and race, was determined using patient-level data from the patient’s first eligible LRTI-related hospitalization admission (index visit) during each season.
Data on clinical diagnoses (ICD-10 diagnosis codes), standardized billing for RSV testing, and hospital characteristics [i.e., hospital size (bed size < 200 and ≥ 200), teaching status (teaching and non-teaching), and population served (urban and rural)], as well as the patients’ demographics, were collected. Due to the non-independent nature of the hospital-level data (i.e., hospitals often contribute data across multiple timepoints), visit-level data (i.e., the total number of visits and the associated RSV testing, regardless of which hospital provided care) were used for the analyses comparing RSV testing frequencies over time.
Billing codes were used to identify RSV tests administered during eligible hospitalizations. Billing codes were extracted from each hospital’s chargemaster—a comprehensive table of items billable to a hospital patient or a patient's health insurance provider—and mapped to Premier’s standardized language (Table S2 in the electronic supplementary material). We relied on information from the standardized chargemaster data rather than the hospitals’ laboratory data for the following reasons: first, the PHD dataset has complete billing data, while only around 40% of hospitals have available laboratory data, suggesting greater generalizability of study outcomes when using the entire PHD dataset; second, the PHD dataset includes standardized language for the data across all hospitals, allowing for more convenient and reliable comparison across the hospitals; and, finally, since the primary aim of this study was to assess the prevalence of RSV testing, laboratory test results were not highly relevant.
Statistical Analyses
In this descriptive study, all data were analyzed using SAS software v9.4 to generate general descriptive statistics, such as mean, median, standard deviation (SD), and interquartile range (IQR). Statistical pairwise comparisons between subgroups defined by hospital characteristics were performed using Mann–Whitney U tests and the Kruskal–Wallis test.
Results
Characteristics of LRTI-Related hospitalizations
A total of 937 hospitals had LRTI-related hospitalizations that met the inclusion criteria for the analysis (i.e., any hospital that contributed data to the PHD) and, altogether, treated 1,450,670 older adults with 2,018,434 eligible LRTI-related inpatient visits during the study period. The mean (SD) age at the index visits was 77.2 (7.6) years and 47.1% (683,370/1,450,670) were male. Most patients were Caucasian (1,195,117; 82.4%).
Among 937 hospitals, there were 442 hospitals (47.2%) classified as larger bed sized (≥ 200 beds) hospitals. There were more non-teaching hospitals (663; 70.6%) and hospitals serving urban populations (665; 71.0%) reporting billing data in the PHD database.
Hospital-Level RSV Testing Frequencies
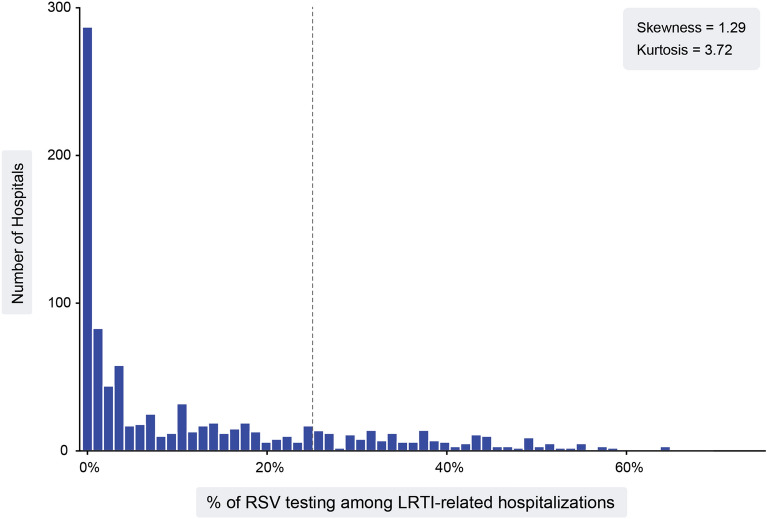
The frequency of RSV testing varied substantially across hospitals. Nearly 300 hospitals did not bill for, and thus presumably did not perform, RSV testing at any of their LRTI-related hospitalizations. In total, 78.4% (735/937) of the hospitals tested less than 25% of their LRTI patients. Across all hospitals, the mean (SD) and the median (IQR) proportions of LRTI-related hospitalizations that were billed for RSV testing were 12.4% (± 15.8) and 4.3% (0.2–20.7), respectively (Fig. 1). The distribution of RSV testing across hospitals was positively skewed with a skewness value of 1.29 and leptokurtic with a kurtosis value of 3.72.
Fig. 1.
Number of hospitals by RSV testing frequency. The number of hospitals (y-axis) associated with their level of RSV testing among LRTI adult (ages 65 +) inpatients, represented as a percentage (x-axis), are shown, with 78.4% of the distribution of hospitals that lie to the left of the dotted vertical line at 25% indicating that 78.4% of hospitals test ≤ 25% of LRTI inpatients for RSV. Skewness is a measure of symmetry, with a value of 0 for normal distributions, while values greater than 1 or less than − 1 indicate a highly skewed distribution. Kurtosis measures the heaviness of the distribution tails, or how many outliers are present in the distribution. Kurtosis values > 1 are considered “peaked” and < − 1 are considered “flat.” LRTI lower respiratory tract infection, RSV respiratory syncytial virus
Visit-Level RSV Testing Frequencies by Time Period
Over the pre-pandemic period (2016–2019), the median percentage of RSV testing across all inpatient visits in the consecutive RSV seasons increased over time from 0.8% in 2016/17 to 2.8% and 6.3% in 2017/18 and 2018/19, respectively (Table 1). The mean visit-level RSV testing frequencies across all hospitals between 2016 and 2019 was 15.2% (306,618/2,018,434; LRTI visits with RSV billing).
Table 1.
Proportion of LRTI-related hospitalizations with a billed RSV test
| Number of hospitals | Mean (± SD) | Median (range) | P value | ||
|---|---|---|---|---|---|
| Total |
Total pre-pandemic (2016–2019) |
937 | 12.4 (± 15.8) | 4.3 (0.0–69.0) | N/A |
| RSV season | 10/1/2016–4/30/2017 | 855 | 8.7 (± 13.7) | 0.8 (0.0–62.4) | < 0.0001 |
| 10/1/2017–4/30/2018 | 819 | 12.6 (± 16.9) | 2.8 (0.0–72.6) | ||
| 10/1/2018–4/30/2019 | 847 | 15.8 (± 18.5) | 6.3 (0.0–73.3) | ||
| Bed size | ≥ 200 | 442 | 15.3 (± 16.2) | 9.1 (0.0–60.1) | < 0.0001 |
| < 200 | 495 | 9.9 (± 15.0) | 1.6 (0.0–69.0) | ||
| Teaching status | Non-teaching | 663 | 10.7 (± 14.9) | 2.5 (0.0–69.0) | < 0.0001 |
| Teaching | 274 | 16.5 (± 17.0) | 11.0 (0.0–67.8) | ||
| Population served | Rural | 272 | 8.0 (± 14.0) | 0.7 (0.0–69.0) | < 0.0001 |
| Urban | 665 | 14.2 (± 16.1) | 7.4 (0.0–67.8) |
The chi-square test was performed at the individual patient’s visit-level, whereas the rest of the statistical tests were performed at the hospital-level. This is due to the same hospitals contributing to each year, resulting in non-independence
LRTI lower respiratory tract infection, RSV respiratory syncytial virus, SD standard deviation
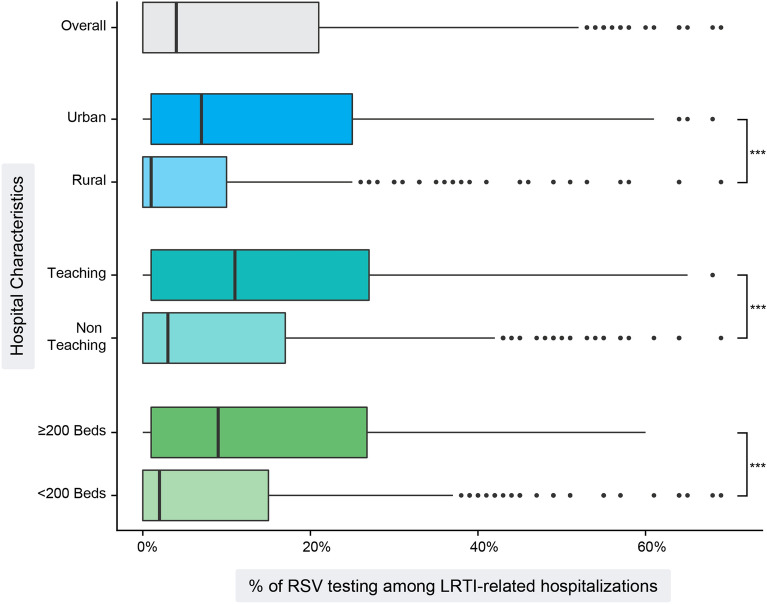
Hospital-Level RSV Testing Frequencies by Hospital Characteristics
Low frequencies of RSV testing were observed across all stratifications of the data by hospital characteristic (Fig. 2). The upper quartile values for all box-and-whisker plots did not exceed 30%, with median testing frequency below 10% in all subgroups except for the teaching hospitals. In pairwise comparisons, RSV testing frequency was significantly higher in the hospitals serving urban populations compared to rural populations (7.4% vs. 0.7%, p < 0.0001), teaching hospitals compared to non-teaching hospitals (11.0% vs. 2.5%, p < 0.0001), and larger hospitals (9.1% for ≥ 200-bed hospitals vs. 1.6% for < 200-bed hospitals, p < 0.0001) (Fig. 2; Table 1).
Fig. 2.
Distributions of hospital RSV testing frequencies among LRTI-related hospitalizations by hospital characteristics. Boxplots show distributions, with the IQR (the 25th through the 75th percentiles) represented as the main body of the box, and the median (50th percentile) drawn as a vertical line. The lines extending from the box are the representative ± 1.5 × IQR, and the individual points beyond the lines are potential outliers of the distribution. ***P value < 0.0001. IQR interquartile range, LRTI lower respiratory tract infection, RSV respiratory syncytial virus
Discussion
In this study, we document low levels of RSV testing performed in US hospitals. RSV infection in elderly patients can lead to severe LRTI, hospitalization, and potential progression to critical conditions such as respiratory failure or death [4, 12]. Despite potential threats, RSV testing in elderly inpatients is infrequent, likely due to current treatment options being supportive only, with no RSV-specific treatments in use. Low testing rates can result in underestimation of RSV incidence, particularly in real-world data studies relying on pathogen-specific ICD codes, which will not be assigned without a standard-of-care RSV test. RSV incidence studies using real-world data need to assess for RSV testing frequency at the source healthcare facilities and adjust their results for under ascertainment associated with limited testing.
A number of studies have reported potential underestimation of RSV infection due to several reasons, including the low sensitivity and underuse of RSV-specific ICD-10 codes [13, 14]. Zhang and colleagues have similarly suggested that hospitalizations caused by RSV in the US setting are under-recorded, especially in the older adult population [15]. Belongia et al. documented this under ascertainment by testing previously collected influenza study specimens for RSV, and found that, among 243 RSV infections (29 involving hospitalization) identified, only one had been diagnosed by standard-of-care testing [16]. Although the frequency of RSV testing has been increasing numerically over the past pre-pandemic years, the absolute frequency remains low (median: 6.3% in 2018/19). The median frequency was lower than the mean frequency across the US hospitals. Positive skewness and leptokurtic distribution (a narrow and peaked distribution exceeding that of normal distribution) (Fig. 1) suggest that a small number of hospitals with relatively high testing frequencies are elevating the mean value, while the majority of hospitals are rarely testing LRTI patients for RSV infection. More than one-third of the hospitals from the PHD database had RSV testing frequency close to zero percent, further supporting the implication that any reported RSV incidence rate would likely be an underestimated value [17].
To explore whether there were associations between RSV testing frequency and hospital characteristics, we performed subgroup analyses based on population served (urban vs. rural), teaching hospital status, and hospital size. Rural hospitals and smaller hospitals exhibited significantly lower frequencies of RSV testing compared to urban hospitals and larger hospitals (≥ 200 beds). These findings are intuitive, as smaller hospitals serving rural populations may be less likely to have staff, time, and monetary resources to support non-essential activities, potentially resulting in deprioritizing RSV testing. Likewise, academic teaching hospitals, which are often located in larger cities and offer specialized care, had significantly higher testing frequency compared to non-teaching community hospitals, presumably due to a greater interest in the research aspect of RSV and LRTI-associated conditions. It is also possible that the patients visiting academic hospitals may exhibit more serious conditions and undergo more diagnostic testing overall, thereby driving the significant differences. However, in spite of the significant differences between the groups, the general trend of low RSV testing frequencies persisted across all subgroups.
Accurate estimation of RSV epidemiology and disease burden is important to identify at-risk populations. In a CDC meeting of experts for the purpose of identifying gaps in the epidemiology of RSV, they noted a need to document potential underestimation of the disease burden due to testing behaviors [18]. Epidemiology studies that aim to estimate the burden of RSV using real-world data should consider the limitations of clinical RSV testing practices at present, and correct for undertesting to create more accurate estimates of RSV disease burden. Model-based incidence studies use a time series design to estimate the proportion of potentially related events due to a specific pathogen. These are an alternative to traditional retrospective claims studies that rely exclusively on ICD-code data, and they produce estimates that are closer to prospective incidence estimates, which are considered the gold standard [7].
The results of this study should be interpreted in the context of some limitations. First, this retrospective analysis of an administrative database relied on coding accuracy, which may have impacted the estimated testing frequency. However, since the chargemaster data are used for estimation in our study, the impact should be minimal as hospitals are unlikely to omit billing of the performed tests. Furthermore, we did not rely on RSV-specific codes which are documented to have low sensitivity, and may be underused due to the low level of testing [19]. Second, patients admitted to the hospital could have potentially been tested in the outpatient setting. While not formally documented, RSV testing in outpatients is understood to be less frequent than inpatient RSV testing, and thus the impact of missed outpatient testing is expected to be small. A strength of our analysis is the size and comprehensiveness of the PHD database, which lends high generalizability to our study findings. Further, the results from these analyses were straightforward and transparent, with a clear indication of low RSV testing frequency, which can lead to an underestimated RSV incidence.
Conclusions
We have demonstrated that the majority of US hospitals have very low levels of RSV testing for LRTI inpatients, with large variability in the percentage tested by different hospitals. These findings imply that relying on retrospective real-world database analyses to determine the RSV-related hospitalization incidence can result in a substantial underestimation of the true RSV incidence. Current incidence estimates based on real-world administrative data should be adjusted upwards to account for this underestimation.
Supplementary Information
Below is the link to the electronic supplementary material.
Acknowledgements
Funding
Pfizer Inc. funded this study and the journal’s Rapid Service fee.
Medical Writing, Editorial, and Other Assistance
Editorial and medical writing support was provided by Donghyun David Lee at EVERSANA and was funded by Pfizer Inc. Programming was conducted by Jingying Zhou at Pfizer. Additional support on data analyses was provided by Tamuno Alfred at Pfizer.
Authorship
All authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article. All authors had full access to the data in this study and take complete responsibility for the integrity of the data and accuracy of the data analysis.
Author Contributions
Mark H. Rozenbaum designed the study. Jen Judy conducted the analyses. All named authors contributed to interpretation of the data, critically reviewed the manuscript for intellectual content and approved the final version of the manuscript.
Disclosures
Mark H. Rozenbaum, Jen Judy, Kari Yacisin, Samantha K. Kurosky, and Elizabeth Begier are employees of Pfizer and hold Pfizer stock and/or stock options. Diana Tran is an employee of EVERSANA, which was a paid consultant to Pfizer in connection with the development of this manuscript.
Prior Presentation
This manuscript is based on work that was previously presented at the 12th International RSV Symposium, hosted by the International Society for Influenza and other Respiratory Virus Diseases (ISIRV) in Belfast, North Ireland, UK from 29 September to 2 October 2022.
Compliance with Ethics Guidelines
All database records were de-identified in compliance with the US patient confidentiality requirements, including the Health Insurance Portability and Accountability Act of 1996. This study did not require an institutional review board (IRB) review due to the deidentified nature of patient health information.
Data Availability
The datasets generated during and/or analyzed during the current study are not publicly available due the terms of our data use agreements with Premier Healthcare Database.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Chatterjee A, Mavunda K, Krilov LR. Current state of respiratory syncytial virus disease and management. Infect Dis Ther. 2021;10(Suppl 1):5–16. doi: 10.1007/s40121-020-00387-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Ciarlitto C, Vittucci AC, Antilici L, et al. Respiratory Syncityal Virus A and B: three bronchiolitis seasons in a third level hospital in Italy. Ital J Pediatr. 2019;45(1):115. doi: 10.1186/s13052-019-0704-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Collins PL, Fearns R, Graham BS. Respiratory syncytial virus: virology, reverse genetics, and pathogenesis of disease. Curr Top Microbiol Immunol. 2013;372:3–38. doi: 10.1007/978-3-642-38919-1_1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Coultas JA, Smyth R, Openshaw PJ. Respiratory syncytial virus (RSV): a scourge from infancy to old age. Thorax. 2019;74(10):986–993. doi: 10.1136/thoraxjnl-2018-212212. [DOI] [PubMed] [Google Scholar]
- 5.Falsey AR, Hennessey PA, Formica MA, Cox C, Walsh EE. Respiratory syncytial virus infection in elderly and high-risk adults. N Engl J Med. 2005;352(17):1749–1759. doi: 10.1056/NEJMoa043951. [DOI] [PubMed] [Google Scholar]
- 6.Branche AR, Falsey AR. Respiratory syncytial virus infection in older adults: an under-recognized problem. Drugs Aging. 2015;32(4):261–269. doi: 10.1007/s40266-015-0258-9. [DOI] [PubMed] [Google Scholar]
- 7.McLaughlin JM, Khan F, Begier E, Swerdlow DL, Jodar L, Falsey AR. Rates of medically attended RSV among US adults: a systematic review and meta-analysis. Open Forum Infect Dis. 2022;9(7):ofac300. doi: 10.1093/ofid/ofac300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Widmer K, Griffin MR, Zhu Y, Williams JV, Talbot HK. Respiratory syncytial virus- and human metapneumovirus-associated emergency department and hospital burden in adults. Influenza Other Respir Viruses. 2014;8(3):347–352. doi: 10.1111/irv.12234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Hogan CA, Caya C, Papenburg J. Rapid and simple molecular tests for the detection of respiratory syncytial virus: a review. Expert Rev Mol Diagn. 2018;18(7):617–629. doi: 10.1080/14737159.2018.1487293. [DOI] [PubMed] [Google Scholar]
- 10.Haber N. Respiratory syncytial virus infection in elderly adults. Med Mal Infect. 2018;48(6):377–382. doi: 10.1016/j.medmal.2018.01.008. [DOI] [PubMed] [Google Scholar]
- 11.Premier Healthcare Database White Paper: Data that informs and performs S, 2020. Premier Applied Sciences®, Premier Inc. https://learn.premierinc.com/white-papers/premier-healthcare-database-whitepaper.
- 12.Schubert L, Steininger J, Lotsch F, et al. Surveillance of respiratory syncytial virus infections in adults, Austria, 2017 to 2019. Sci Rep. 2021;11(1):8939. doi: 10.1038/s41598-021-88537-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Cai W, Tolksdorf K, Hirve S, et al. Evaluation of using ICD-10 code data for respiratory syncytial virus surveillance. Influenza Other Respir Viruses. 2020;14(6):630–637. doi: 10.1111/irv.12665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Makari D, Staat MA, Henrickson KJ, Wu X, Ambrose CS. The underrecognized burden of respiratory syncytial virus among infants presenting to US Emergency Departments. Clin Pediatr (Phila) 2015;54(6):594–597. doi: 10.1177/0009922814546040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Zheng Z, Warren JL, Shapiro ED, Pitzer VE, Weinberger DM. Estimated Incidence of Respiratory Hospitalizations Attributable to RSV Infections across Age and Socioeconomic Groups. 2022. [DOI] [PMC free article] [PubMed]
- 16.Belongia EA, King JP, Kieke BA, et al. Clinical features, severity, and incidence of RSV illness during 12 consecutive seasons in a community cohort of adults >/=60 years old. Open Forum Infect Dis. 2018;5(12):ofy316. doi: 10.1093/ofid/ofy316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Sim JY, Chen YC, Hsu WY, et al. Circulating pediatric respiratory pathogens in Taiwan during 2020: dynamic change under low COVID-19 incidence. J Microbiol Immunol Infect. 2022 doi: 10.1016/j.jmii.2022.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kim L, Rha B, Abramson JS, et al. Identifying gaps in respiratory syncytial virus disease epidemiology in the United States prior to the introduction of vaccines. Clin Infect Dis. 2017;65(6):1020–1025. doi: 10.1093/cid/cix432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.McLaughlin JM, Khan F, Schmitt HJ, et al. Respiratory syncytial virus-associated hospitalization rates among US infants: a systematic review and meta-analysis. J Infect Dis. 2022;225(6):1100–1111. doi: 10.1093/infdis/jiaa752. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The datasets generated during and/or analyzed during the current study are not publicly available due the terms of our data use agreements with Premier Healthcare Database.