Abstract
The pandemic phase of coronavirus disease 2019 (COVID-19) appears to be over in most countries. However, the unexpected behaviour and unstable nature of coronaviruses, including temporary hiatuses, re-emergence, emergence of new variants, and changing outbreak epicentres during the COVID-19 pandemic, have been frequently reported. The mentioned trend shows the fact that in addition to vaccine development, different strategies should be considered to deal effectively with this disease, in long term. In this regard, the role of enzymes in regulating immune responses to Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) has recently attracted much attention. Moreover, several reports confirm the association of short telomeres with sever COVID-19 symptoms. This review highlights the role of several enzymes involved in telomere length (TL) regulation and explains their relevance to SARS-CoV-2 infection. Apparently, inhibition of telomere shortening (TS) through inhibition and/or activation of these enzymes could be a potential target in the treatment of COVID-19, which may also lead to a reduction in disease severity.
Keywords: Coronavirus disease-2019 (COVID-19), Immune responses, SARS-CoV-2 entry blockers, Telomere shortening, Telomerase activation, Telomerase inhibition
Introduction
The Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) causes coronavirus disease 2019 (COVID-19), which shows more severe symptoms and higher risk of mortality among the elderly (Sepe et al. 2022). Since the initial diagnosis of SARS-CoV-2 in February 2020, the virus has tended to evolve continuously via the changes in the genetic code. These changes are the result of genetic selection that normally occurs through either genetic errors/mutations (Papanikolaou et al. 2022) or viral recombination, during the genome replication (V’kovski et al. 2021). In line with this, evidences raise that new variants of the SARS-CoV-2 coronavirus can escape immunity formed by immunizations (e.g. vaccination) and previous infection (Callaway and Ledford 2021). As a consequence, breakthrough infections after vaccination against SARS-CoV-2 were increasingly reported (Bates et al. 2022). Recent studies show that most breakthrough infections were mild or asymptomatic, although persistent symptoms did occur (Bergwerk et al. 2021). Regardless of the efficiency and availability of the developed COVID-19 vaccines conspiracy beliefs, anti-vax and hesitant attitudes make it even more complicated to conduct a fully coordinated vaccination against the disease (Balasuriya et al. 2021; Benoit and Mauldin 2021; Roberts et al. 2022). The individuals with compromised immune systems should be added to the mentioned complexities.
Considering all these facts, comprehensive vaccination programs should be supported by various strategies to increase the effectiveness of biological control of COVID-19 in the long term. The aim of the present review is to support such a concept by focusing on several important telomere-related enzymes to control the progression and severity of COVID-19 symptoms. In addition, regulatory approaches for each enzyme (activation and/or inhibition) are proposed based on the most recent findings (Fig. 1). Furthermore, the importance of telomerase and its association with the disease will be highlighted in the present review remarkably, since the enzyme appear to be a new target for the development of novel therapeutic agents against COVID-19 infection (Valdiani et al. 2022).
Fig. 1.
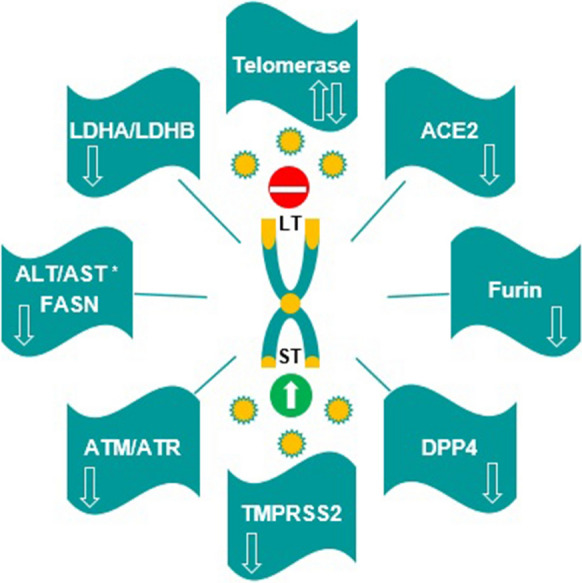
Schematic representation of viral entry in the presence of long and short telomeres as well as the association of telomere-related enzymes with SARS-CoV-2 infection LT Long telomeres, ST Short telomeres, ACE2 Angiotensin converting enzyme-2, DPP4 Dipeptidyl peptidase 4, TMPRSS2 Transmembrane protease serine protease-2, ATM Ataxia-telangiectasia mutated, ATR Ataxia-telangiectasia and Rad3, ALT Alanine aminotransferase, AST Aspartate aminotransferase, FASN Fatty acid synthase, LDHA Lactate dehydrogenase A, LDHB Lactate dehydrogenase B,  : Activation of the enzyme is suggested,
: Activation of the enzyme is suggested,  : Inhibition of the enzyme is suggested,
: Inhibition of the enzyme is suggested,  : Coronavirus,
: Coronavirus,  : Blocking virus entry and mild infection of SARS-CoV-2 in the presence of long telomeres,
: Blocking virus entry and mild infection of SARS-CoV-2 in the presence of long telomeres,  : Easy viral entry and severe infection of SARS-CoV-2 in the presence of short telomeres,
: Easy viral entry and severe infection of SARS-CoV-2 in the presence of short telomeres,  : chromosome and telomeres.*ALT and AST are not directly involved in the regulation of telomere length
: chromosome and telomeres.*ALT and AST are not directly involved in the regulation of telomere length
Principle of the review
Inhibition of telomere shortening via enzymes to control COVID-19 severity
The association between short telomeres and severe COVID-19 symptoms was initially hypothesized by Aviv (2020). Unlike the paradox of telomere length (TL) and cancer, recent investigations have consistently stated the presence of short telomeres in the patients with severe COVID-19 symptoms (Sanchez-Vazquez et al. 2021), especially in elderly patients (Kamal et al. 2020). As evidence grows that short telomeres increase the risk of severe COVID-19 (Froidure et al. 2020; Benetos et al. 2021), several molecular mechanisms have been explained to address the connection between short telomeres and COVID-19. Aviv (2021) suggests that T-cell TL affects the adaptive immune response and the innate immune response to SARS-CoV-2 infection. According to his hypothesis, the individual with long T-cell telomeres, shows strong T-cell response and robust suppression of the innate immune response that is accompanied by ‘calibrated’ (moderate) activity of the innate immune response. In contrast, the individual with short T-cell telomeres, shows weak T-cell response and inadequate suppression of the innate immune response that is accompanied by ‘calibrated’ (strong) activity of the innate immune response, expressed in cytokine storm, lung injury, and severe COVID-19 (Aviv 2021). Despite a consensus on the nexus between short telomeres and COVID-19 (Benetos et al. 2021), it should be noticed that "short telomere" is itself an "effect" but not a "cause"; and, studying the cause has priority over the effect.
Enzymes associated with telomere length
The present review focuses on the 12 important enzymes, including telomerase, ACE2 (Angiotensin Converting Enzyme-2), Furin, TMPRSS2 (Transmembrane Protease Serine Protease-2), DPP4 (Dipeptidyl peptidase 4), LDHA (Lactate dehydrogenase A), LDHB (Lactate dehydrogenase B), ATM (Ataxia-telangiectasia mutated), ATR (Ataxia-telangiectasia and Rad3), ALT (Alanine aminotransferase), AST (Aspartate aminotransferase), and FASN (Fatty acid synthase), which are able to regulate telomere length (TL) and inhibit telomere shortening (TS) directly or indirectly. Table 1 provides an overview of this article as it represents a summary of remarkable findings of the relevant studies to the 12 telomere-associated enzymes, short telomeres, and severity of COVID-19 symptoms.
Table 1.
Summary of recent findings of the relevant studies to the telomere-associated enzymes, short telomeres, and severity of COVID-19 symptoms
| Enzyme | LE/SS | IT | IEI/SMAE | Remarks | References |
|---|---|---|---|---|---|
| Telomerase | H/S | TS | Weak AR/Inhibition* | RS triggers high TA in both B-Cells and T-Cells, and antigen-mediated proliferation (RI), which is insufficient to prevent TS—a hallmark of cancer | (Aviv 2022a; Taheri et al. 2022b; Patrick and Weng 2019)b |
| Telomerase | H–L/S | TS | Weak AR/Inhibition*–Activation* | Weak T-Cell response (AR) initiates strong immune response, insufficient viral clearance, and strong cytokine storm in individuals with short telomeres that may lead to severe COVID-19 symptoms. The opposite situation in individuals with long telomeres may lead to mild COVID-19 symptoms | (Aviv 2020a; 2021a) |
| Telomerase | L/S | TS | Weak AR/Activation* | Age-dependent telomere shortening and genome instability (due to RS) leading to immunosenescence may increase susceptibility to infections. Hence, telomerase activity and long leukocyte telomeres appear to be critical elements for immune homeostasis when encountering infection | (Aviv 2020a;Cunha et al. 2020a; Simões et al. 2020a; Mahmoodpoor et al. 2022) |
| Telomerase | N/A | TS | N/A | Predictive SNP-based markers of LTL do not identify the shorter LTL, as a causal risk factor for COVID-19 susceptibility or severity. Based on the bidirectional MR analysis, aging is not a risk factor for the increased severity of COVID-19 | (Jiang et al. 2022; Huang et al. 2022; Xu et al. 2022) |
| ACE2 | H/S | TS | VEM-CVR/Inhibition | RBD of SARS-CoV-2 spike protein binds to ACE2 receptor | (Sepe et al. 2022) |
| Furin | H/S | TS | VEM-CVR/Inhibition | RBD of SARS-CoV-2 spike protein is cleaved by Furin receptor | (Paulsson-Habegger et al. 2021; Peacock et al. 2021) |
| TMPRSS2 | H/S | TS | VEM-CVR/Inhibition | RBD of SARS-CoV-2 spike protein is primed by TMPRSS2 receptor. RG4 can inhibit TMPRSS2 translation and block SARS-CoV-2 entry | (Hoffmann et al. 2020; Liu et al. 2022) |
| DPP4 | H/S | TS | VEM-CVR/Inhibition | MERS-CoV receptor “DPP4” is a candidate binding target (receptor) of the SARS-CoV-2 spike protein | (Gao et al. 2022; Li et al. 2020) |
| LDHA/LDHB | H/S | TS | VEM-CVR/Inhibition* | LDHB reduces TA and shortens telomeres. Increased levels of LDH is associated with increased odds of severe COVID-19 (~ sixfold), therefore, can be used as a severity marker for the disease | (Henry et al. 2020; Szarpak et al. 2021; McGroder et al. 2021; Wang et al. 2022) |
| ATM/ATR | H/S | TS | VEM-CVR/Inhibition* | Depletion of ATM and ATR can reduce the accumulation of telomerase complex. ATR triggers DNA damage response. Inhibition of ATM/ATR appears to be a potential target in the treatment of COVID-19 | (Tong et al. 2015; Victor et al. 2021; Gulliver et al. 2022) |
| ALT/AST | H/S | TS | Unknown/Inhibition* | Short telomeres are associated with liver disease, which both are considered as risk factors in COVID-19 – studying temporary increase of ALT/AST enzymes in patients diagnosed with COVID-19 may lead to find the link between the level of enzymes and TS | (Shin et al. 2021; Rattan et al. 2022; Liao et al. 2022) |
| FASN | H/S | TS | Virus replication/Inhibition | The cellular lipid synthesis is essential for SARS-CoV-2 replication. Inhibition of FASN can block SARS-CoV-2 replication | (Chu et al. 2021) |
ACE2 Angiotensin converting enzyme-2, ALT Alanine aminotransferase, AR Adaptive response, AST Aspartate aminotransferase, ATM Ataxia-telangiectasia mutated, ATR Ataxia-Telangiectasia and Rad3, CVR Coronavirus receptor, DPP4 Dipeptidyl peptidase 4, FASN Fatty acid synthase, H High, IEI Involvement of enzyme with SARS-CoV-2 infection, ITL Impact on the telomere length, L Low, LDHA Lactate dehydrogenase A, LDHB Lactate dehydrogenase B, LE Level of enzyme before SARS-CoV-2 infection, LTL Leukocyte telomere length, MERS-CoV Middle East Respiratory Syndrome–Coronavirus, MR Mendelian randomization, N/A Not applicable, RBD Receptor-binding domain, RG4 RNA G-quadruplex, RI Replicative immortality, RS Replicative senescence, SMAE Suggested modulation approach to enzyme, SNP Single-nucleotide polymorphism, S Severe, SS Severity of COVID-19 symptoms, TA Telomerase activity, TMPRSS2 Transmembrane protease serine protease-2, TS Telomere shortening, VEM Viral entry mediator
*The suggested modulation against the enzyme, is interpretational, and should be confirmed through further investigation
aThe severity of COVID-19 infection has not been attributed to high/low TA specifically; however, the presence of short telomeres, as a consequence of high TA during cancer (i.e. RI) and/or age-dependent low TA (i.e. RS), has been considered as a risk factor in severe COVID-19 infection
bThe reference explains the association of telomerase activity with cancer and/or aging without linking it to COVID-19
Telomerase
Telomerase is a reverse transcriptase enzyme that regulates the length of telomeres in eukaryote chromosomes. In other words, telomerase is one of the major “causes” paradoxically involved in telomere maintenance/elongation as well as telomere shortening. The enzyme extends human telomeres by adding TTAGGG repeats to the ends of chromosomes (Aviv et al. 2017). Under normal circumstances, activation and inhibition of telomerase lead to the elongation/maintenance and shortening of telomeres, respectively (Akbarizare et al. 2021). In cancer cells, the excessive activity of telomerase enhances the extreme cell division during the replicative immortality process, thus, telomere shortening (Jafri et al. 2016). Despite the paradox of cancer–TL (Aviv et al. 2017), telomere dysfunction can occur irrespective to its length (Victorelli and Passos 2017). Regardless of this contradiction, telomeres and telomerase appear to play a critical role in the function of human immune system (Matthe et al. 2022). The association of telomerase and COVID-19 should be explained, accordingly
Telomerase activation vs. inhibition
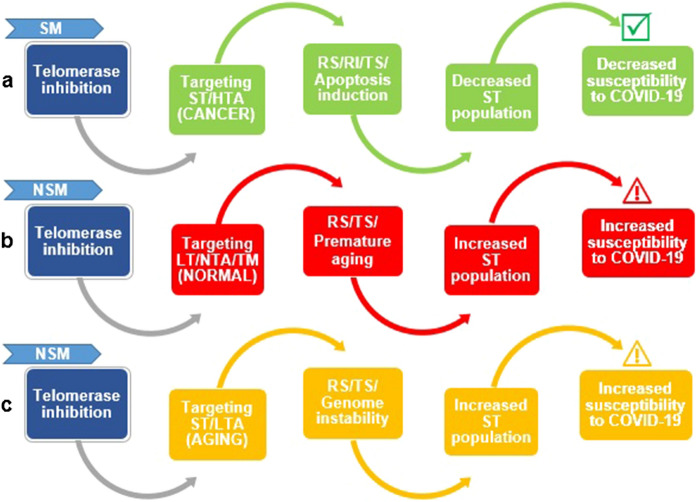
The role of T-cells in protective immunity against COVID-19, and the connotation between immunity based on antibodies and memory T cells, are still unclear (Spellberg et al. 2021). According to a recently developed model that links the aging to COVID-19, T-cell lymphopenia and mortality, an individual with average hematopoietic cell TL at the age of 20 preserves maximal T-cell clonal expansion capacity until the sixth decade of life, when this capacity quickly declines by more than 90% over the next ten years. The sudden decline of the T-cell clonal expansion capacity coincides with the sharp increase in COVID-19 mortality with age (Anderson et al. 2022). Under such circumstances, telomerase, the reverse transcriptase that extends telomeres, is activated during T-cell clonal development and differentiation but the activity of telomerase is "inadequate" to counter telomere shortening with T-cell replication (Aviv et al. 2021). However, the age-dependent T-cell telomere shortening might not be the main problem in the absence of a severe infection (Aviv et al. 2021), because the long half-lives of ~ 5 years of naïve T cells and ~ 5 months of memory T cells in the circulation, impose minimal demand on the clonal development capacity of T cells (Vrisekoop et al. 2008). Despite this, an enormous and quick T-cell clonal development might be required after contracting the SARS-CoV-2 infection (Aviv et al. 2021). This situation is almost similar to replicative immortality process with high telomerase activity – a hallmark of cancer (Taheri et al. 2022). Considering the situation that telomerase reactivation may reversely lead to telomere shortening (due to extensive cell division) during the replicative immortality process versus replicative senescence (Jafri et al. 2016), the “inhibition” of telomerase seems to be a potential target in the treatment of COVID-19, in the future. Apparently, such function is subject to a specific modulation (SM) of immunity, especially in cancer patients or patients prone to cancer, which in turn can lead to reducing the population of existing short telomeres and susceptibility to SARS-CoV-2, as shown in Fig. 2a. Under such circumstances, the natural telomerase inhibitors can be considered as potential agents in the treatment of COVID-19. The ambiguity of using telomerase inhibitors is that their effect on normal telomeres, as well as on the elderly and those prone to premature cellular senescence is still unknown. In the worst-case scenario, telomerase inhibitors might hypothetically lead to an increase in the population of short telomeres, thus, increasing susceptibility to SARS-CoV-2. Such a function is considered as a non-specific modulation (NSM) of immunity via telomerase inhibition (Fig. 2b and c). Probably, because of such concerns, some literatures tend or appear to challenge the idea of "telomerase inhibition" by suggesting the idea of "telomerase activation" as an alternative approach to confront COVID-19 (Aviv 2020; Simões et al. 2020; Mahmoodpoor et al. 2022). On the other hand, predictive SNP-based markers of LTL do not identify the shorter LTL, as a causal risk factor for COVID-19 susceptibility or severity (Jiang et al. 2022; Huang et al. 2022). Similarly, based on the bidirectional Mendelian randomization (MR) analysis, aging is not a risk factor for the increased severity of COVID-19 (Xu et al. 2022).
Fig. 2.
Schematic representation of paradoxical effects of telomerase inhibitors on telomeres and telomerase activity. Reproduced based on Valdiani et al. (2022) SM Specific modulation, NSM Non-specific modulation, ST Short telomeres, HTA High telomerase activity, RS Replicative senescence, RI Replicative immortality, TS Telomere shortening, NT Normal telomeres, LT Long telomeres or normal telomeres, NTA Normal telomerase activity, TM Telomere maintenance/elongation, LTA Low telomerase activity
Therefore, the paradoxical effects of telomerase inhibition as well as selectivity/specificity of telomerase inhibitors should be investigated carefully, prior to use by the patients. Specifically, therapeutic agents targeting the telomere/telomerase system will be beneficial only if the alterations to the LTL are causal or contributory to the disease rather than a side-effect (Xu et al. 2022). Furthermore, it should be reminded that under the normal condition, telomerase activity leads to elongation (or maintenance) of TL while telomerase inhibition results in telomere shortening. From this point of view, telomerase activators can be considered as potential targets in the treatment of COVID-19. More importantly, a recent finding highlights that SARS-CoV-2 infection lowers the expression of Telomeric Repeat-Binding Factor 2 (TRF2) shelterin-protein complex, and consequently, results in telomere shortening in Vero E6 cells (Victor et al. 2021). Therefore, the application of telomerase inhibitors during the SARS-CoV-2 infection should be cautioned. Finally, further investigations should be conducted to understand if telomerase inhibitors and activators are useful in the prevention of SARS-CoV-2 infection and/or deceasing the severity of COVID-19 symptoms.
ACE2
Angiotensin Converting Enzyme-2 is a zinc metalloprotease that plays an important role in the renin–angiotensin–aldosterone system (RAAS) (Zhang et al. 2020). The enzyme is responsible for the regulation of blood pressure in human. A recent study shows that the expression of ACE2, the SARS-CoV-2 cell receptor, which is essential for mediating cell entry of the coronavirus, was increased in the lung cells of aging mice and humans (Sepe et al. 2022). Furthermore, it was observed that the expression of ACE2 was increased upon telomere shortening or dysfunction – common hallmarks of aging – in mammalian cells (in vitro) and mice cells (in vivo). This increase is controlled at the transcriptional level and depends on the ACE2 promoter activity, as a DNA damage response (DDR) elicited by dysfunctional telomeres (Sepe et al. 2022). This study suggests that telomere dysfunction due to telomere shortening or damage, triggers DDR activation during aging. This process eventually leads to the upregulation of ACE2, thus, contributing to make the elderly more susceptible to coronavirus infection. Similar to the case of telomerase inhibition/activation, further research is required to establish whether decreasing ACE2 expression is efficient on the SARS-CoV-2 infection and the severe COVID-19 symptoms. Furthermore, it should be understood how DDR signalling results in an increased level of ACE2 gene expression. Previously, it was confirmed that SARS-CoV-1 spike protein promotes lung injury by down-regulating the expression of ACE2 in the infected lungs (Kuba et al. 2005).
Furin
Furin is a pro-protein convertase, which is a type of enzyme that converts inactive proteins to their biologically active equivalents. SARS-CoV-2 contains a polybasic insertion (PRRAR) at the S1/S2 junction and/or the S2ʹ cleavage sites that can be cleaved by furin. Therefore, furin is known as one of the enzymes to be responsible for the viral entry of SARS-CoV-2 into cells by sequential cleavage of the spike (S) protein of SARS-CoV-2 to mediate membrane fusion (Paulsson-Habegger et al. 2021). A recent study confirmed that furin cleavage site in the SARS-CoV-2 spike protein is required for transmission of the virus in ferrets, while the loss of furin cleavage site attenuates SARS-CoV-2 pathogenesis (Peacock et al. 2021). On the other hand, recent reports are showing that the level of furin is increased in smokers and chronic obstructive pulmonary disease (COPD) cases, significantly (Maremanda et al. 2020). The critical role of the furin cleavage site in infection with SARS-CoV-2 highlights the importance of this site for assessing the neutralization mechanisms of antibodies (Johnson et al. 2021). Therefore, the inhibition of furin seems to be a potential therapeutic target in SARS-CoV-2 treatment.
TMPRSS2
Hoffmann et al. (2020) demonstrated that SARS-CoV-2 uses the SARS-CoV receptor ACE2 for entry and the serine protease TMPRSS2 for spike (S) protein priming, for the first time. The inhibition of TMPRSS2 by serine protease inhibitors has been suggested as an efficient approach to block SARS-CoV-2 entry. Accordingly, a partial blocking of SARS-2-S-driven entry into Caco-2 and Vero-TMPRSS2 cells was initially achieved by using a clinically approved serine protease inhibitor, namely, camostat mesylate (Kawase et al. 2012). However, the full inhibition of this entry was attained when camostat mesylate and E-64d, as the inhibitors of CatB/L, were added to camostat mesylate. Therefore, it was indicated that SARS-2-S protein can use both CatB/L and TMPRSS2 for priming in these cell lines (Hoffmann et al. 2020). A recent study has revealed that the presence of RNA G-quadruplex (RG4) within TMPRSS2 can lead to the inhibition of TMPRSS2 translation and blocking SARS-CoV-2 entry (Liu et al. 2022). Furthermore, G-quadruplex (G4)-specific stabilizers mitigates the SARS-CoV-2 infection in pseudovirus cell systems and mouse models. Consequently, the level of TMPRSS2 protein is increased in lungs of COVID-19 patients. These findings emphasize on an unknown mechanism underlying SARS-CoV-2 infection and recommend RG4 as a potential target for the prevention and treatment of COVID-19 (Liu et al. 2022).
DPP4
Dipeptidyl peptidase 4, also known as the T-cell antigen CD26, is a multi-functional enzyme that acts as a binding protein and a ligand for a variety of extracellular molecules (Deacon 2019). Similar to ACE-2 and TMPRSS2, DPP4 has been introduced as a viral entry mediator for coronavirus. Dipeptidyl peptidase 4 is mainly present on the cell membrane surface, and acts as the functional entry receptor for Middle East Respiratory Syndrome—Coronavirus (MERS-CoV), reportedly (Li et al. 2020; Alves et al. 2022). According to Mongelli et al. (2021), the expression of DPP4 was detected unchanged in post-COVID-19 patients compared to COVID-19-free. Despite this, lung immunoblot analysis of smokers, COPD and idiopathic pulmonary fibrosis (IPF) cases showed that the increased level of proteases and receptor/spike proteins, such as DPP4 is associated with a minor increase in the level of SARS-CoV-2 receptor ACE2 (Maremanda et al. 2020). Nevertheless, no solid evidence has been found to confirm the inhibition of DPP4 as an efficient approach to reduce the clinical symptoms of COVID-19 Maremanda et al. (2020) concludes that altered transcription of the target genes regulating mitochondrial function, cellular senescence, and telomere erosion in the lung of COPD and IPF patients, is associated with alterations in the SARS-CoV-2-ACE2-TMPRSS2-Furin-DPP4 axis. Such interactions could be considered as potential pharmacological targets for COVID-19 (Maremanda et al. 2020). More specifically, COVID-19 may affect DPP4 in patients with diabetes mellitus, leading to high mortality of diabetes mellitus combined with COVID-19. Reportedly, the oral anti-diabetic DPP4 inhibitors, such as sitagliptin, may lead to telomere elongation in type 2 diabetes mellitus (T2DM) patients (Qi et al. 2015). In this setting, DPP4 inhibitors and interleukin 6 (as a pro-inflammatory cytokine and an anti-inflammatory myokine) antagonists can be considered to reduce the effect of COVID-19 infection in patients with diabetes (Gao et al. 2022). Randomized and prospective clinical trials are required to assess this properly (Alves et al. 2022).
LDHA and LDHB
Lactate dehydrogenase is an enzyme that mediates the redox-coupled conversion between lactate and NAD+ with pyruvate and NADH. Lactate dehydrogenase A is a glycolytic enzyme, the main enzyme regulating the Warburg effect, thus, a useful target for novel anti-cancer drug development (Kim et al. 2019). Recently, Wang et al. (2022) have screened metabolic enzymes using telomerase activity assay in pancreatic cancer cells, and discovered that LDHB performed as a telomerase activity regulator. They realised that LDHB shRNA (short RNA) can reduce both telomerase activity and TL in the cells at approximately 20 population doubling levels (PDLs). Additionally, knockout LDHB by sgRNA inhibited cell proliferation. In contrast, stable knockdown of LDHA had no impact on TL. These results indicated that LDHB is required not only to maintain TL but also to prevent cells senescence (Wang et al. 2022). Reportedly, there is a significant association between the higher levels of LDH and shorter telomeres mostly in male COVID-19 patients (McGroder et al. 2021). Furthermore, Henry et al. (2020) showed that high levels of LDH were associated with ~ sixfold increase in the likelihood of severe COVID-19. In accordance with these observations, the results of a Meta data analysis of COVID-19 patients showed that the LDH level can be used as a marker for severe COVID-19 (Szarpak et al. 2021). Very recently, the inhibition of LDH has been proposed as a therapeutic target in the treatment of SARS-CoV-2 infection, since lactate and LDH enzyme appeared to be the major risks in inflammatory diseases, including COVID-19 (Gupta 2022).
PI3K-related kinase/(PIKK) family
ATM and ATR protein kinase
Ataxia-telangiectasia mutated and ATR both belong to the class-IV phosphoinositide 3-kinase (PI3K)-related kinase (PIKK) family (Awasthi et al. 2015). ATM protein kinase is a potent protein kinase, the function of which is lost in patients with the genomic instability syndrome ataxia-telangiectasia (Shiloh and Ziv 2013). The enzyme was known as a factor involved in DNA repair initially, however, there have been conflicting reports on its role in telomere lengthening (Dahl and Arid, 2017). In another attempt, Lee et al. (2015) confirmed that ATM kinase is required for telomere elongation in mouse and human cells. Moreover, it was shown that the depletion of ATM and ATR can reduce assembly of the telomerase complex. Simultaneously, ATM is necessary for telomere elongation in cells expressing a mutant allele of POT1, called POT1ΔOB, which disrupts TL homeostasis (Tong et al. 2015). In fact, POT1 is a single-stranded telomeric DNA-binding protein which is necessary for cis-inhibition of telomerase (Ye et al. 2004). Recently, it was observed that SARS-CoV-2 infection in African green monkey kidney cells (Vero E6) decreased the expression of TRF2 shelterin-protein complex, shortened the relative length of telomeres, and triggered ATR DDR (Victor et al. 2021). More specifically, a significant transcriptional upregulation of the ATR-related protein was detected in the infected Vero E6 cells without detecting any enhanced phosphorylation of the ATR protein. Although, overall reductions in total ATR and checkpoint kinase 1 (CHK1) protein levels were observed after the infection (at 48 h), suggesting that the overall increase in ATR levels corresponding to the increased mRNA levels may have occurred before the test time of 48 h. Furthermore, enhanced phosphorylation of CHK1, a downstream effector molecule of the ATR DDR, and H2AX (H2A histone family member X) were observed. Therefore, confirming the enhanced expression of ATR and CHK1 in a time course experiment post SARS-CoV-2 infection can be a point of focus in future studies (Victor et al. 2021). Considering these observations, DDR inhibitors are suggested as the potent blockers of SARS-CoV-2 replication (Garcia et al. 2021). The inhibition of ATM and ATR can be considered as potential targets in COVID-19 treatment, accordingly. The inhibition of these two enzymes have been alternatively employed for immune stimulation in cancer therapy (Gulliver et al. 2022; Zhang et al. 2019; Sun et al. 2018; Reddy et al. 2015). Paradoxically, early studies have shown that ATM deficiency in A-T human fibroblasts leads to faster telomere shortening, abnormal cell-cycle response to DNA damage, and early senescence (Baross et al. 2004). Fortunately, recent achievements on the effectively and safety of few kinase inhibitors as potential antiviral agents and treatments for COVID-19 have been promising. For instance, the activity of three AXL receptor tyrosine kinase (AXL) inhibitors including gilteritinib, nintedanib, and imatinib against SARS-CoV-2 infection has been successfully tested in vitro (Boytz et al. 2022). Interestingly, knockout of AXL, a target of gilteritinib and nintedanib, reduced SARS-CoV-2 variant infectivity, supporting a role for AXL in SARS-CoV-2 infection. Further mechanistic studies are required to determine underlying differences in virus response (Boytz et al. 2022).
Liver enzymes
ALT and AST
Alanine aminotransferase or SGPT (serum glutamic-pyruvic transaminase), and AST or SGOT (serum glutamic-oxaloacetic transaminase) are intercellular enzymes, mostly found in the liver. Hypothetically, liver function enzymes such as ALT and AST are not directly involved in the regulation of TL. Despite this, recent studies have shown that shorter leucocyte telomere length (LTL) is significantly correlated with advanced hepatic fibrosis and high levels of ALT and AST enzymes in T2DM patients (Dong et al. 2020). The observation also suggests that shorter LTL may be used as a biomarker for advanced fibrosis in T2DM patients (Dong et al. 2020). Furthermore, it was recently confirmed that shorter telomeres were independently identified in elderly diagnosed with advanced liver disease (Rattan et al. 2022). According to another report, TL was shorter in the elevated ALT group of patients than in the normal ALT group, however, the ALT values and TL showed a marginal inverse correlation, which was not statistically significant (Shin et al. 2021). Therefore, it is important to understand the role of telomeres in the progression of liver disease (Rattan et al. 2022). Furthermore, it should be understood whether the ALT and AST play a direct role in the regulation of TL, in the future. Further research is required to establish a better understanding of any connection between short telomeres and temporarily elevated levels of ALT/AST enzymes in patients diagnosed with COVID-19 (Liao et al. 2022). Alternatively, investigating fat metabolism and fatty acid synthase (FASN) will be useful to make a link between the liver enzymes and telomere-associated COVID-19 infection.
FASN
Fatty acid synthase (FASN) is a multi-enzyme protein that catalyzes fatty acid synthesis. Despite it is assumed that liver function enzymes (i.e. ALT and AST) are not regulating the TL directly, but fat metabolism might be associated with TL (Van der Spek et al. 2022). Experimentally, regulation of TL was performed by Fatty Acid Elongase 3 in yeast (Ponnusamy et al. 2008). Moreover, FASN inhibitors are drug candidates for the prevention and treatment of COVID-19 by inhibiting SARS-CoV-2 replication. Nevertheless, the evaluation of the efficacy of fatty acid synthase inhibitors in treating severe COVID-19 in humans is needed (Chu et al. 2021). The link between obesity type 2 diabetes, and fatty liver had been previously established. Firstly, the SARS-CoV-2 virus may directly infect the parenchymal cells of subcutaneous fat. Infection appears to activate the interferon-alpha (IFN-α) pathway and to attract infiltrating leukocytes in lethal COVID-19 cases. Therefore, adipose tissue represents a significant reservoir for SARS-CoV-2 and a source of inflammatory mediators (Basolo et al. 2022). Secondly, elevated serum liver biochemistries seen in COVID-19 patients with the incidence of elevated ALT and AST ranged from 2.5%-50.0% to 2.5%-61.1%, respectively (Garrido et al. 2020). Acute liver injury might be related to the virus infection itself or a consequence of severe inflammatory response, hepatic ischemia, and drug-induced liver injury. The specific virus receptors, such as ACE2, are expressed by liver and bile duct cells (Feng et al. 2020; Hamming et al. 2004). Furthermore, persistent liver function test (LFT) abnormalities are significantly associated with higher prevalence of ultrasound determined fatty liver disease (Liao et al. 2022).
Conclusion and future perspective
The pandemic phase of COVID-19 appears to be over in most countries, unless a significant and severe new variant emerges in the future. Fortunately, some of the side-effects of the disease such as the increased level of ALT/AST enzymes, might be temporary and return to the normal level, approximately after a year (Liao et al. 2022). In contrast, some consequences such as telomere shortening and biological aging induced by COVID-19 infection (Mongelli et al. 2021), may be irreversible. Therefore, telomere shortening not only is a predictor of severity and mortality of COVID-19, but also a disease outcome in the post-COVID-19 cohort. Such complexity requires a better understanding of the association of telomere-associated enzymes with SARS-CoV-2 infection, to perform a multilayer biological defence against the disease. Overall, targeting metabolic pathways and enzymatic approaches appear to be powerful tools for inducing long-term cellular immunity in the post-vaccination period as well as post-COVID-19 recovery period (Primorac et al. 2022). Nevertheless, the paradoxical effects and selectivity of inhibitors as well as activators of telomere-associated enzymes should be carefully investigated before and after COVID-19 infection.
Acknowledgements
Not applicable.
Author contributions
The authors have contributed to this work equally.
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Data availability
Not applicable.
Declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethical approval
Not applicable.
Informed consent
Not applicable.
Consent for publication
All the authors approved the present version for publication purposes and agreed to hold accountability for all aspects of the work.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Alireza Valdiani, Email: alireza.valdiani@gmail.com.
Hamideh Ofoghi, Email: ofoghi@irost.ir.
References
- Akbarizare M, Ofoghi H, Hadizadeh M, Valdiani A. Bilateral telomerase-associated impacts of Spirulina platensis extracts: activation versus inhibition. Eur J Integr Med. 2021;43:101303. doi: 10.1016/j.eujim.2021.101303. [DOI] [Google Scholar]
- Alves MHME, Mahnke LC, Macedo TC, Silva TKDS, Carvalho Junior LB. The enzymes in COVID-19: a review. Biochimie. 2022;197:38–48. doi: 10.1016/j.biochi.2022.01.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anderson JJ, Susser E, Arbeev KG, Yashin AI, Levy D, Verhulst S, Aviv A. Telomere-length dependent T-cell clonal expansion: a model linking ageing to COVID-19 T-cell lymphopenia and mortality. EBioMed. 2022;78:103978. doi: 10.1016/j.ebiom.2022.103978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aviv A. Telomeres and COVID-19. FASEB J. 2020;00:1–6. doi: 10.1096/fj.202001025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aviv A. Short telomeres and severe COVID-19: the connection conundrum. eBioMed. 2021;70:103513. doi: 10.1016/j.ebiom.2021.103513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aviv A. The bullwhip effect, T-cell telomeres, and SARS-CoV-2. Lancet Healthy Longev. 2022;3:e715–e721. doi: 10.1016/S2666-7568(22)00190-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aviv A, Anderson JJ, Shay JW. Mutations, cancer and the telomere length paradox. Trends Cancer. 2017;3(4):253–258. doi: 10.1016/j.trecan.2017.02.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Awasthi P, Foiani M, Kumar A. ATM and ATR signaling at a glance. J Cell Sci. 2015;128(23):4255–4262. doi: 10.1242/jcs.169730. [DOI] [PubMed] [Google Scholar]
- Balasuriya L, Santilli A, Morone J, Ainooson J, Roy B, Njoku A, Mendiola-Iparraguirre A, Duffany KO, Macklin B, Higginbottom J, et al. JAMA. Netw Open. 2021;4(10):e2128575. doi: 10.1001/jamanetworkopen.2021.28575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baross A, Schertzer M, Zuyderduyn SD, Jones SJ, Marra MA, Lansdorp PM. Effect of TERT and ATM on gene expression profiles in human fibroblasts. Genes Chrom Cancer. 2004;39(4):298–310. doi: 10.1002/gcc.20006. [DOI] [PubMed] [Google Scholar]
- Basolo A, Poma AM, Proietti BD, A, Macerola E, Ugolini C, Torregrossa L, Giannini R, Vignali P, Basolo F, , et al. Adipose tissue in COVID-19: detection of SARS-CoV-2 in adipocytes and activation of the interferon-alpha response. J Endocrinol Invest. 2022;45:1021–1029. doi: 10.1007/s40618-022-01742-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bates TA, McBride SK, Winders B, Schoen D, Trautmann L, Curlin ME, Tafesse FG. Antibody response and variant cross-neutralization after SARS-CoV-2 breakthrough infection. JAMA. 2022;327(2):179–181. doi: 10.1001/jama.2021.22898. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Benetos A, Lai TP, Toupance T. The nexus between telomere length and lymphocyte count in seniors hospitalized with COVID-19. J Gerontol A Biol Sci Med Sci. 2021;76(8):e97–e101. doi: 10.1093/gerona/glab026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Benoit SL, Mauldin RF. The “anti-vax” movement: a quantitative report on vaccine beliefs and knowledge across social media. BMC Public Health. 2021;21:2106. doi: 10.1186/s12889-021-12114-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bergwerk M, Gonen T, Lustig Y, Amit S, Lipsitch M, Cohen C, Mandelboim M, Levin EG, Rubin C, Indenbaum V, et al. COVID-19 breakthrough infections in vaccinated health care workers. N Engl J Med. 2021;385:1474–1484. doi: 10.1056/NEJMoa2109072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boytz RM, Słabicki M, Ramaswamy S, Patten JJ, Zou C, Meng C, Hurst BL, Wang J, Nowak RP, Yang PL, et al. Anti-SARS-CoV-2 activity of targeted kinase inhibitors: repurposing clinically available drugs for COVID-19 therapy. J Medic Virol. 2022 doi: 10.1002/jmv.28157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Callaway E, Ledford H. How to redesign COVID vaccines so they protect against variants. Nat. 2021;590:15–16. doi: 10.1038/d41586-021-00241-6. [DOI] [PubMed] [Google Scholar]
- Chu J, Xing C, Du Y, Duan T, Liu S, Zhang P, Cheng C, Henley J, Liu X, Qian C, et al. Pharmacological inhibition of fatty acid synthesis blocks SARS-CoV-2 replication. Nat Metab. 2021;3:1466–1475. doi: 10.1038/s42255-021-00479-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cunha LL, Perazzio SF, Azzi J, Cravedi P, Riella LV. Remodeling of the immune response with aging: Immunosenescence and its potential impact on COVID-19 immune response. Front Immunol. 2020;11:1748. doi: 10.3389/fimmu.2020.01748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dahl ES, Aird KM. Ataxia-telangiectasia mutated modulation of carbon metabolism in cancer. Front Oncol. 2017 doi: 10.3389/fonc.2017.00291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deacon CF. Physiology and pharmacology of DPP4 in glucose homeostasis and the treatment of type 2 diabetes. Front Endocrinol. 2019 doi: 10.3389/fendo.2019.00080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dong K, Zhang Y, Huang JJ, Xia SS, Yang Y. Shorter leucocyte telomere length as a potential biomarker for nonalcoholic fatty liver disease-related advanced fibrosis in T2DM patients. Ann Transl Med. 2020;8(6):308. doi: 10.21037/atm.2020.03.10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feng G, Zheng KI, Yan Q-Q, Rios RS, Targher G, Byrne CD, Poucke SV, Liu W-Y, Zheng M-H. COVID-19 and liver dysfunction: current insights and emergent therapeutic strategies. J Clin Transl Hepatol. 2020;8:18–24. doi: 10.14218/JCTH.2020.00018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Froidure A, Mahieu M, Hoton D, Laterre PF, Yombi JC, Koenig S, Ghaye B, Defour JP, Decottignies A. Short telomeres increase the risk of severe COVID-19. Aging. 2020;12(20):19911–19922. doi: 10.18632/aging.104097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gao Q, Zhang W, Li T, Yang G, Zhu W, Chen N, Jin H. Interrelationship between 2019-nCov receptor DPP4 and diabetes mellitus targets based on protein interaction network. Sci Rep. 2022;12:188. doi: 10.1038/s41598-021-03912-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garcia G, Jr, Sharma A, Ramaiah A, Sen C, Purkayastha A, Kohn DB, Parcells MS, Beck S, Kim H, Bakowski MA, et al. Antiviral drug screen identifies DNA-damage response inhibitor as potent blocker of SARS-CoV-2 replication. Cell Rep. 2021;35(1):108940. doi: 10.1016/j.celrep.2021.108940. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garrido I, Liberal R, Macedo G. COVID-19 and liver disease—what we know on 1st May 2020. Aliment Pharmacol Ther. 2020;52(2):267–275. doi: 10.1111/apt.15813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gulliver C, Hoffmann R, Baillie GS. Ataxia-telangiectasia mutated and ataxia telangiectasia and Rad3-related kinases as therapeutic targets and stratification indicators for prostate cancer. Int J Biochem Cell Biol. 2022;147:106230. doi: 10.1016/j.biocel.2022.106230. [DOI] [PubMed] [Google Scholar]
- Gupta GS. The lactate and the lactate dehydrogenase in inflammatory diseases and major risk factors in COVID-19 patients. Inflammation. 2022;19:1–33. doi: 10.1007/s10753-022-01680-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guterres AN, Villanueva J. Targeting telomerase for cancer therapy. Oncogene. 2020 doi: 10.1038/s41388-020-01405-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hamming I, Timens W, Bulthuis M, Lely AT, Navis GJ, van Goor H. Tissue distribution of ACE2 protein, the functional receptor for SARS coronavirus. A first step in understanding SARS pathogenesis. J Pathol. 2004;203:631–637. doi: 10.1002/path.1570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Henry BM, Aggarwal G, Wong J, Benoit S, Vikse J, Plebani M, Lippi G. Lactate dehydrogenase levels predict coronavirus disease 2019 (COVID-19) severity and mortality: a pooled analysis. Am J Emerg Med. 2020;38(9):1722–1726. doi: 10.1016/j.ajem.2020.05.073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hoffmann M, Kleine-Weber H, Schroeder S, Krüger N, Herrler T, Erichsen S, Schiergens TS, Herrler G, Wu NH, Nitsche A, et al. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell. 2020;181:271–280.e8. doi: 10.1016/j.cell.2020.02.052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang D, Lin S, He J, Wang Q, Zhan Y. Association between COVID-19 and telomere length: A bidirectional Mendelian randomization study. J Medic Virol. 2022;94(11):5345–5353. doi: 10.1002/jmv.28008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jafri MA, Ansari SA, Alqahtani MH, Shay JW. Roles of telomeres and telomerase in cancer, and advances in telomerase targeted therapies. Genome Med. 2016;8:69. doi: 10.1186/s13073-016-0324-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jiang L, Tang B-S, Guo J-F, Li J-C. Telomere length and COVID-19 outcomes: a two-sample bidirectional Mendelian randomization study. Front Genet. 2022;13:805903. doi: 10.3389/fgene.2022.805903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnson BA, Xie X, Bailey AL, Kalveram B, Lokugamage KG, Muruato A, Zou J, Zhang X, Juelich T, Smith JK, et al. Loss of furin cleavage site attenuates SARS-CoV-2 pathogenesis. Nat. 2021;591:293–299. doi: 10.1038/s41586-021-03237-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kamal MA, Alamiry KR, Zaki M. Sex and age differences in telomere length and susceptibility to COVID-19. J Biomed Res Environ Sci. 2020;1(7):303–310. doi: 10.37871/jbres1159. [DOI] [Google Scholar]
- Kawase M, Shirato K, van der Hoek L, Taguchi F, Matsuyama S. Simultaneous treatment of human bronchial epithelial cells with serine and cysteine protease inhibitors prevents severe acute respiratory syndrome coronavirus entry. J Virol. 2012;86:6537–6545. doi: 10.1128/JVI.00094-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim EY, Chung TW, Han CW, Park SY, Park KH, Jang SB, Ha K-T. A novel lactate dehydrogenase inhibitor, 1-(phenylseleno)-4-(trifluoromethyl) benzene, suppresses tumor growth through apoptotic cell death. Sci Rep. 2019;9:3969. doi: 10.1038/s41598-019-40617-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuba K, Imai Y, Rao S, Gao H, Guo F, Guan B, Huan Y, Yang P, Zhang Y, Deng W, et al. A crucial role of angiotensin converting enzyme 2 (ACE2) in SARS coronavirus-induced lung injury. Nat Med. 2005;11:875–879. doi: 10.1038/nm1267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee SS, Bohrson C, Pike AM, Wheelan SJ, Greider CW. ATM kinase is required for telomere elongation in mouse and human cells. Cell Rep. 2015;13(8):1623–1632. doi: 10.1016/j.celrep.2015.10.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li Y, Zhang Z, Yang L, Lian X, Xie Y, Li S, Xin S, Cao P, Lu J. The MERS-CoV receptor DPP4 as a candidate binding target of the SARS-CoV-2 spike. iScience. 2020;23(6):101160. doi: 10.1016/j.isci.2020.101160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liao X, Li D, Ma Z, Zhang L, Zheng B, Li Z, Li G, Liu L, Zhang Z. 12-month post-discharge liver function test abnormalities among patients with COVID-19: a single-center prospective cohort study. Front Cell Infect Microbiol. 2022 doi: 10.3389/fcimb.2022.864933. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu G, Du W, Sang X, Tong Q, Wang Y, Chen G, Yuan Y, Jiang L, Cheng W, Liu D, et al. RNA G-quadruplex in TMPRSS2 reduces SARS-CoV-2 infection. Nat Commun. 2022;13:1444. doi: 10.1038/s41467-022-29135-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mahmoodpoor A, Sanaie S, Roudbari F, Sabzevari T, Sohrabifar N, Kazeminasab S. Understanding the role of telomere attrition and epigenetic signatures in COVID-19 severity. Gene. 2022;811:146069. doi: 10.1016/j.gene.2021.146069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maremanda KP, Sundar IK, Li D, Rahman I. Age-dependent assessment of genes involved in cellular senescence, telomere, and mitochondrial pathways in human lung tissue of smokers, COPD, and IPF: Associations with SARS-CoV-2 COVID-19 ACE2-TMPRSS2-Furin-DPP4 axis. Front Pharmacol. 2020;9(11):584637. doi: 10.3389/fphar.2020.584637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matthe DM, Thoma OM, Sperka T, Neurath MF, Waldner MJ. Telomerase deficiency reflects age-associated changes in CD4+ T cells. Immun Ageing. 2022;19:16. doi: 10.1186/s12979-022-00273-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McGroder CF, Zhang D, Choudhury MA, Salvatore MM, D'Souza BM, Hoffman EA, Wei Y, Baldwin MR, Garcia CK. Pulmonary fibrosis 4 months after COVID-19 is associated with severity of illness and blood leucocyte telomere length. Thorax. 2021;76(12):1242–1245. doi: 10.1136/thoraxjnl-2021-217031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mongelli A, Barbi V, Zamperla GM, Atlante S, Forleo L, Nesta M, Massetti M, Pontecorvi A, Nanni S, Farsetti A, et al. evidence for biological age acceleration and telomere shortening in COVID-19 survivors. Int J Mol Sci. 2021;7(11):6151. doi: 10.3390/ijms22116151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Papanikolaou V, Chrysovergis A, Ragos V, Tsiambas E, Katsinis S, Manoli A, Papouliakos S, Roukas D, Mastronikolis S, Peschos D, Batistatou A, Kyrodimos E, Mastronikolis N. From delta to Omicron: S1-RBD/S2 mutation/deletion equilibrium in SARS-CoV-2 defined variants. Gene. 2022;814:146134. doi: 10.1016/j.gene.2021.146134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Patrick M, Weng NP. Expression and regulation of telomerase in human T cell differentiation, activation, aging and diseases. Cell Immunol. 2019;345:103989. doi: 10.1016/j.cellimm.2019.103989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paulsson-Habegger L, Snabaitis AK, Wren SP. Enzyme inhibition as a potential therapeutic strategy to treat COVID-19 infection. Bioorg Med Chem. 2021;48:116389. doi: 10.1016/j.bmc.2021.116389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peacock TP, Goldhill DH, Zhou J, Baillon L, Frise R, Swann OC, Kugathasan R, Penn R, Brown JC, Sanchez-David RY, et al. The furin cleavage site in the SARS-CoV-2 spike protein is required for transmission in ferrets. Nat Microbiol. 2021;6:899–909. doi: 10.1038/s41564-021-00908-w. [DOI] [PubMed] [Google Scholar]
- Ponnusamy S, Alderson NL, Hama H, Bielawski J, Jiang JC, Bhandari R, Snyder SH, Jazwinski SM, Ogretmen B. Regulation of telomere length by fatty acid elongase 3 in yeast: involvement of inositol phosphate metabolism and Ku70/80 function. J Biol Chem. 2008;283(41):27514–27524. doi: 10.1074/jbc.M802980200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Primorac D, Brlek P, Matišić V, Molnar V, Vrdoljak K, Zadro R, Parčina M. Cellular immunity—the key to long-term protection in individuals recovered from SARS-CoV-2 and after vaccination. Vaccines. 2022;10:442. doi: 10.3390/vaccines10030442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Qi NW, Ling Z, Bing C. The influence of the telomere-telomerase system on diabetes mellitus and its vascular complications. Expert Opin Ther Targets. 2015;19:849–864. doi: 10.1517/14728222.2015.1016500. [DOI] [PubMed] [Google Scholar]
- Rattan P, Penrice DD, Ahn JC, Ferrer A, Patnaik M, Shah VH, Kamath PS, Mangaonkar AA, Simonetto DA. Inverse association of telomere length with liver disease and mortality in the US population. Hepatol Commun. 2022;6(2):399–410. doi: 10.1002/hep4.1803. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reddy V, Wu M, Ciavattone N, McKenty N, Menon M, Barrack ER, Reddy GP, Kim SH. ATM inhibition potentiates death of androgen receptor-inactivated prostate cancer cells with telomere dysfunction. J Biol Chem. 2015;16(42):25522–25533. doi: 10.1074/jbc.M115.671404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roberts HA, Clark DA, Kalina C, Sherman C, Brislin S, Heitzeg MM, Hicks BM. To vax or not to vax: Predictors of anti-vax attitudes and COVID-19 vaccine hesitancy prior to widespread vaccine availability. PLoS ONE. 2022;17(2):e0264019. doi: 10.1371/journal.pone.0264019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sanchez-Vazquez R, Guío-Carrión A, Zapatero-Gaviria A. Shorter telomere lengths in patients with severe COVID-19 disease. Aging. 2021;13(1):1–15. doi: 10.18632/aging.202463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sepe S, Rossiello F, Cancila V, Iannelli F, Matti V, Cicio G, Cabrini M, Marinelli E, Alabi BR, di Lillo A, et al. DNA damage response at telomeres boosts the transcription of SARS-CoV-2 receptor ACE2 during aging. EMBO Rep. 2022;23:e53658. doi: 10.15252/embr.202153658. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shiloh Y, Ziv Y. The ATM protein kinase: regulating the cellular response to genotoxic stress, and more. Nat Rev Mol Cell Biol. 2013;14:197–210. doi: 10.1038/nrm3546. [DOI] [PubMed] [Google Scholar]
- Shin HK, Park JH, Yu JH, Jin Y-J, Suh YJ, Lee J-W, Kim W, et al. Association between telomere length and hepatic fibrosis in non-alcoholic fatty liver disease. Sci Rep. 2021;11:18004. doi: 10.1038/s41598-021-97385-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Simões HG, Rosa TS, Sousa CV. Does longer leukocyte telomere length and higher physical fitness protect master athletes from consequences of coronavirus (SARS-CoV-2) Infection? Front Sports Act Living. 2020;2:87. doi: 10.3389/fspor.2020.00087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spellberg B, Nielsen TB, Casadevall A. Antibodies, immunity, and COVID-19. JAMA Int Med. 2021;181(4):460–462. doi: 10.1001/jamainternmed.2020.7986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sun LL, Yang RY, Li CW, Chen MK, Shao B, Hsu JM, Chan LC, Yang Y, Hsu JL, Lai YJ, et al. Inhibition of ATR downregulates PD-L1 and sensitizes tumor cells to T cell-mediated killing. Am J Cancer Res. 2018;8:1307–1316. [PMC free article] [PubMed] [Google Scholar]
- Szarpak L, Ruetzler K, Safiejko K, Hampel M, Pruc M, Kanczuga-Koda L, Filipiak KJ, Jaguszewski MJ. Lactate dehydrogenase level as a COVID-19 severity marker. Am J Emerg Med. 2021;45:638–639. doi: 10.1016/j.ajem.2020.11.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taheri M, Ghafouri-Fard S, Najafi S, Kallenbach J, Keramatfar E, Atri Roozbahani G, Heidari Horestani M, Hussen BM, Baniahmad A. Hormonal regulation of telomerase activity and hTERT expression in steroid-regulated tissues and cancer. Cancer Cell Int. 2022;22:258. doi: 10.1186/s12935-022-02678-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tong AS, Stern JL, Sfeir A, Kartawinata M, de Lange T, Zhu XD, Bryan TM. ATM and ATR signaling regulate the recruitment of human telomerase to telomeres. Cell Rep. 2015;13(8):1633–1646. doi: 10.1016/j.celrep.2015.10.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- V’kovski P, Kratzel A, Steiner S, Stalder H, Thiel V, Coronavirus biology and replication: implications for SARS-CoV-2. Nat Rev Microbiol. 2021;19:155–170. doi: 10.1038/s41579-020-00468-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Valdiani A, Ofoghi H, Akbarizare M, Talei D. Andrographis paniculata extract as an immunity modulator against cancer via telomerase inhibition. 3 Biotech. 2022 doi: 10.1007/s13205-022-03373-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van der Spek A, Karamujić-Čomić H, Pool R, Bot M, Beekman M, Garmaeva S, Arp PP, Henkelman S, Liu J, Alves AC, et al. Fat metabolism is associated with telomere length in six population-based studies. Human Mol Genet. 2022;31(7):1159–1170. doi: 10.1093/hmg/ddab281. [DOI] [PubMed] [Google Scholar]
- Victor J, Deutsch J, Whitaker A, Lamkin EN, March A, Zhou P, Botten JW, Chatterjee N. SARS-CoV-2 triggers DNA damage response in Vero E6 cells. Biochem Biophys Res Commun. 2021;579:141–145. doi: 10.1016/j.bbrc.2021.09.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Victorelli S, Passos JF. Telomeres and cell senescence - size matters not. Ebio Med. 2017;21:14–20. doi: 10.1016/j.ebiom.2017.03.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vrisekoop N, den Braber I, de Boer AB, Ruiter AF, Ackermans MT, van der Crabben SN, Schrijver EH, Spierenburg G, Sauerwein HP, Hazenberg MD, de Boer RJ, et al. Sparse production but preferential incorporation of recently produced naive T cells in the human peripheral pool. Proc Natl Acad Sci U S A. 2008;22(16):6115–6120. doi: 10.1073/pnas.0709713105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang R, Li J, Zhang C, Guan X, Qin B, Jin R, Qin L, Xu S, Zhang X, Liu R, Ye Q. Lactate dehydrogenase B is required for pancreatic cancer cell immortalization through activation of telomerase activity. Front Oncol. 2022 doi: 10.3389/fonc.2022.821620. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu W, Zhang F, Shi Y, Chen Y, Shi B, Yu G. Causal association of epigenetic aging and COVID-19 severity and susceptibility: a bidirectional Mendelian randomization study. Front Med (lausanne). 2022;23(9):989950. doi: 10.3389/fmed.2022.989950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ye JZ, Hockemeyer D, Krutchinsky AN, Loayza D, Hooper SM, Chait BT, de Lange T. POT1-interacting protein PIP1: a telomere length regulator that recruits POT1 to the TIN2/TRF1 complex. Genes Dev. 2004;18(14):1649–1654. doi: 10.1101/gad.1215404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang Q, Green MD, Lang X, Lazarus J, Parsels JD, Wei S, Parsels LA, Shi J, Ramnath N, Wahl DR, et al. Inhibition of ATM increases interferon signaling and sensitizes pancreatic cancer to immune checkpoint blockade therapy. Cancer Res. 2019;79(15):3940–3951. doi: 10.1158/0008-5472.CAN-19-0761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang H, Penninger JM, Li Y, Zhong N, Slutsky AS. Angiotensin-converting enzyme 2 (ACE2) as a SARS-CoV-2 receptor: molecular mechanisms and potential therapeutic target. Intens Care Med. 2020;46(4):586–590. doi: 10.1007/s00134-020-05985-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.