Abstract
This cohort study aims to evaluate the predictive validity of multimodal clinical assessment and quantitative measures of in- and off-laboratory mobility for fall-risk estimation in patients with cerebellar ataxia (CA).
Occurrence, severity, and consequences of falling were prospectively assessed for 6 months in 93 patients with hereditary (N = 36) and sporadic or secondary (N = 57) forms of CA and 63 healthy controls. Participants completed a multimodal clinical and functional fall risk assessment, in-laboratory gait examination, and a 2-week inertial sensor-based daily mobility monitoring. Multivariate logistic regression analyses were performed to evaluate the predictive capacity of all clinical and in- and off-laboratory mobility measures with respect to fall (1) status (non-faller vs. faller), (2) frequency (occasional vs. frequent falls), and (3) severity (benign vs. injurious fall) of patients.
64% of patients experienced one or recurrent falls and 65% of these severe fall-related injuries during prospective assessment. Mobility impairments in patients corresponded to a mild-to-moderate ataxic gait disorder. Patients’ fall status and frequency could be reliably predicted (78% and 81% accuracy, respectively), primarily based on their retrospective fall status. Clinical scoring of ataxic symptoms and in- and off-laboratory gait and mobility measures improved classification and provided unique information for the prediction of fall severity (84% accuracy).
These results encourage a stepwise approach for fall risk assessment in patients with CA: fall history-taking readily and reliably informs the clinician about patients’ general fall risk. Clinical scoring and instrument-based mobility measures provide further in-depth information on the risk of recurrent and injurious falling.
Keywords: Cerebellar ataxia, Falls, Fall prediction, Gait analysis, Mobility monitoring
Introduction
Gait disturbances are a common and disabling complication in patients with cerebellar ataxia (CA) with significant implications for patients’ capacity for independent living and quality of life [1, 2]. Walking impairments in CA are linked to a considerable risk of recurrent falling, which frequently results in severe secondary comorbidities and a loss of functional independence. Retrospective and prospective fall surveys in CA indicate that 74–93% of patients experience at least one fall per year [3–5]. Many of these patients suffer from fall-related injuries (74–85%), which potentially (23–31%) result in a fracture or joint dislocation [3, 4]. Since causal therapeutic approaches are currently not available [6, 7], a primary treatment objective in CA is an effective prevention of falling and fall-related injuries.
Fall prevention in CA requires reliable screening and classification procedures to assess and identify individuals at particular risk of falling and severe fall-related complications. Distinct risk factors including symptom severity, the number of non-ataxic symptoms, and the duration and etiology of disease were associated to higher fall rates in CA patients [3–5, 8]. However, the predictive validity of these factors considerably differed between studies. Complementary approaches aim to identify fall risk-related features linked to balance and gait impairments in CA. Accordingly, variability in the movement pattern, a defining feature of gait ataxia [9, 10] that increases with disease severity [11], was shown to be associated with patients’ general fall status and risk of recurrent falling [2, 11, 12]. Subsequent analyses, however, indicated that gait variability markers may be only predictive for a specific subclass of falls (falls resulting from tripping or stumbling) [13]. Furthermore, quantitative gait assessment is currently mostly restricted to in-laboratory settings and thus potentially underestimates the challenges of everyday-life mobility during which patients actually fall.
Recent advances in off-laboratory mobility assessment using body-worn inertial sensors promise a more specific characterization of patients’ balance impairments and fall risk in environmentally valid settings [14]. Inertial sensor-based mobility assessment has been shown to consistently capture central features of ataxic gait impairments under real-life conditions [15]. In contrast to in-laboratory gait assessment approaches that primarily focus on a detailed characterization of spatiotemporal features of walking impairments, body-worn sensors may further provide important complementary insights into patients’ overall mobility status and balance capabilities from a macroscopic perspective [14, 16–19]. This information could supplement and overcome limitations of currently available screening tools for fall risk assessment in CA.
The aim of this study was to evaluate and compare the predictive validity of currently available and recently established screening tools for fall risk identification in patients with CA. To this end, we prospectively assessed the occurrence, frequency, and consequences of falling in a comprehensive cohort of patients with CA of different etiologies. Subsequently, multimodal score-based and clinical fall risk assessment outcomes as well as measures from instrument-based in-laboratory gait examination and long-term off-laboratory mobility monitoring were evaluated with respect to their relevance for forecasting the occurrence, frequency, and severity of falls.
Methods
Participants
Longitudinal cohort study with 93 patients with CA and 63 healthy controls recruited (study ID: DRKS00007762) from 06/2015 to 02/2018 at the University Hospital Munich. Patients were included based on the following criteria: (1) presence of clinical ataxia [20] due to sporadic, secondary, or hereditary forms of CA, (2) age between 18 and 80, and (3) ability to ambulate independently without walking aids. Inclusion criteria for controls were (1) no history of neurological or psychiatric disease, (2) age between 18 and 80, and (3) no other diseases with manifest ambulation problems.
Clinical Work-Up and Fall Assessment at Inclusion
All patients and controls underwent a standardized physical examination, which included a survey of the following information: ambulatory status, functional status, medication, and falls within the preceding 6 months. According to the WHO criteria, a fall was defined as an event which results in a person coming to rest inadvertently on the ground or floor or other lower level [21]. A near-fall was defined as an event that results in a marked postural instability requiring a supporting step and/or balance adjustment. Retrospective fall assessment included information on fall status (faller vs. non-faller), fall frequency (no, occasional, frequent falls (≥ two falls)), and fall severity (1: near-fall; 2: no or mild injury; 3: injury requiring medical attention; 4: injury requiring hospital admission) according to the Hopkins Falls Grading Scale (HFGS) [22]. The subjective level of stability was assessed by the Falls Efficacy Scale-International (FES-I) and the Activities-specific Balance Confidence Scale (ABC-d) [23, 24]. Health-related quality of life was assessed by the Short-Form Health Survey (SF-12) [25]. Cognitive function was screened with the Montreal Cognitive Assessment (MOCA) [26]. Each participant underwent a complete neurological and physical examination including the assessment of functional mobility by the Timed up and Go Test (TUG) and the Functional Gait Assessment Score (FGA) [27, 28]. Severity of ataxia symptoms in patients was rated using the Scale for the Assessment and Rating of Ataxia (SARA) with a maximum of 40 points (indicating most severe form of ataxia) [29].
Prospective Fall Assessment
Each participant was provided with a fall diary covering a 6-month follow-up period. Participants were asked to document near-fall and fall events on a daily basis with information on the time, the environmental circumstances, the fall mechanism (e.g., tripping, vertigo/dizziness, impaired consciousness, others), the duration of the post-fall lying phase, and the related HFGS of each event. Participants were additionally contacted by phone monthly to cross-check and validate the documented information. Based on the prospective fall assessment, participants were categorized with respect to fall status, frequency, and severity.
In-laboratory Gait Examination
In-laboratory gait assessment was performed on a 6.7 m-long pressure-sensitive carpet (GAITRite®, CIR System, Sparta, NJ, USA) at 120 Hz. Participants walked over the carpet at their self-chosen (PWS), slow (SWS), and maximum walking speed (MWS). Each condition was recorded 4 times. Gait assessment was conducted without additional ambulatory aids. For each walking condition, the following spatiotemporal gait parameters were analyzed: gait velocity, base of support, stride length, stride time, swing phase percentage, double support percentage, coefficient of variation (CV) of the base of support, CV of stride time, CV of stride length, gait asymmetry index, and phase synchronization index [30].
Off-laboratory Mobility Assessment
Following the initial visit, monitoring of daily mobility was undertaken for 14 days. Participants wore an inertial sensor-based activity monitor (ActivPAL®, PAL Technologies, Glasgow), which recorded the sequence and period of individual bouts of ambulatory, sedentary, and sleeping behavior at 10 Hz. The inertial sensor was placed on the thigh of the dominant leg approximately 0.1 m cranially and 0.05 m laterally to the patella. Participants were advised to continue their daily activities as usual and not to change their routine. Upon completion of the recording period, participants removed the sensor by themselves and send it back by mail.
The following parameters (expressed as average daily estimates) were computed from the ActivPAL data [31, 32]: daily intensity (amount of daily energy expenditure expressed as the total metabolic equivalents (METS)), daily volume (percentage of ambulatory, sedentary, or sleeping time), daily step count, daily number of sit-to-stance transitions, daily pattern of ambulatory behavior (computed as the exponent alpha that quantifies the distribution of bouts, with lower alpha values indicating a greater contribution of long bouts).
Data Analysis Procedures
Descriptive statistics are reported as mean ± SD. In a first step, analysis of variance (ANOVA) and chi-squared tests were used to test for differences of metric and categorical parameters from clinical assessment, in-laboratory gait examination, off-laboratory mobility assessment, and retro- and prospective fall assessment between patients and controls. The subsequent statistical analyses focused solely on patient data: multivariate backward logistic regression analyses (controlled for age, gender, and leg length) were performed to identify independent predictors associated with the three dependent prospectively assessed fall measures of interest: (I) non-faller vs. faller, (II) occasional vs. frequent faller, and (III) non-severe vs. severe falling (defined as HFGS 3 or 4). For each regression model sensitivity, specificity and correct classification are reported as quality parameter derived by the classification matrix. Initially, all potential predictor parameters from clinical and mobility assessment were subjected to an ANOVA (including post hoc comparison via Sheffé procedures) with respect to the three above-mentioned fall categories. Subsequently, only those parameters that yielded a significance level of their F value ≤ 0.05 were considered in the respective regression model. To avoid collinearity, relationships among parameters were examined using Pearson’s correlations. If parameters were strongly correlated (r > 0.7), only the one most strongly associated with the dependent measure was retained. This statistical approach is based on [33]. Statistical analysis was performed using SPSS (Version 25.0; IBM Corp., Armonk, NY).
Results
Characteristics of the Study Cohort
Demographic information and clinical characteristics of patients and controls are summarized in Table 1. The patient cohort included hereditary degenerative forms of ataxia (N = 36), sporadic degenerative forms of ataxia (sporadic adult-onset ataxia, N = 36), ataxia due to focal vascular or neo-plastic cerebellar lesions (N = 3), and downbeat nystagmus syndrome (N = 18). Patients had a mean SARA score of 10.2 (range: [3;24]), indicating an on average moderate severity of ataxic symptoms. Accordingly, assessment of functional mobility revealed mildly impaired balance and gait capacity of patients compared to controls (FGA: F1,153 = 91.4, p < 0.001) despite normal performance in the TUG. Patients reported increased fear of falling (FES-I: F1,153 = 78.5, p < 0.001), decreased balance confidence (ABC-d: F1,153 = 114.8, p < 0.001), and showed moderately reduced cognitive function (MoCA: F1,153 = 16.3, p < 0.001). Health-related quality of life scores did not differ between patients and controls.
Table 1.
Characteristics of the study cohort
| Healthy subjects | Cerebellar disorders | F1,153 | p | ||||
|---|---|---|---|---|---|---|---|
| Demographical characteristics | |||||||
| N (f/m) | 63 (31/32) | 93 (35/58) | |||||
| Age [y] | 49 ± 14 | 57 ± 18 | 2.4 | n.s | |||
| Diagnoses |
14 SCA 11 FRDA 11 EA2 18 DBN 35 SAOA 1 ARAC 1 ACM 1 post stroke |
||||||
| Clinical performance scales | |||||||
| SARA score [points] | – | 10.2 ± 2.6 | |||||
| FGA [points] | 29 ± 3 | 21 ± 6 | 91.4 | < 0.001 | |||
| TUG [s] | 8.8 ± 3.2 | 11.2 ± 7.1 | 0.7 | n.s | |||
| MOCA [points] | 29 ± 3 | 24 ± 6 | 16.5 | < 0.001 | |||
| Subjective symptome scales | |||||||
| ABC-d [%] | 95 ± 9 | 63 ± 22 | 114.8 | < 0.001 | |||
| FES-I [points] | 17 ± 2 | 31 ± 13 | 78.5 | < 0.001 | |||
| SF-12 [points] | 31 ± 3 | 30 ± 4 | 1.15 | < 0.001 | |||
| df | c2 | p | |||||
| Retrospective falls status | |||||||
| No falls [n, %] | 57, 90 | 32, 34 | 2 | 37.2 | < 0.001 | ||
| Occasional fall [n, %] | 4, 7 | 21, 23 | |||||
| Frequent falls [n, %] | 2, 3 | 40, 43 | |||||
| Retrospective falls severity | |||||||
| Hopkins grade I [%] | 28, 45 | 22, 24 | 3 | 46.8 | < 0.001 | ||
| II [%] | 17, 27 | 46, 49 | |||||
| III [%] | 17, 27 | 14, 15 | |||||
| IV [%] | 0, 0 | 11, 12 | |||||
f, female; m, male; FGA, functional gait assessment; FES-I, falls efficacy scale – international; TUG, timed-up-and-go test; MOCA, montreal cognitive assessment; SF-12, short form 12; BVP, bilateral vestibular failure; UVP, unilateral vestibular failure; SCA, spinocerebellar ataxia; FRDA, friedreich ataxia; EA2, episodic ataxia type 2; DBN, downbeat nystagmus syndrome; SAOA, sporadic adult onset ataxia; ARAC, autosomal recessive cerebellar ataxia; ACM, tumor cerebellar; C2, chi-square test
In- and Off-laboratory Mobility Assessment
Descriptive information on outcomes from in- and off-laboratory mobility assessment is summarized in Table 2. During in-laboratory gait assessment, patients showed walking impairments corresponding to an ataxic gait disorder with reduced walking speed (F1,153 = 39.9, p < 0.001), prolonged double support phases (F1,153 = 22.6, p < 0.001), increased base of support (F1,153 = 65.7, p < 0.001), spatiotemporal variability (stride length CV: F1,153 = 21.3, p < 0.001; stride time CV: F1,153 = 28.1, p < 0.001), and asymmetry within their stride-to-stride pattern (gait asymmetry: F1,153 = 24.5, p < 0.001; phase synchronization: F1,153 = 17.3, p < 0.001). Off-laboratory monitoring of daily life mobility revealed a general reduction of energy expenditure (F1,153 = 48.7, p < 0.001) with a reduced daily amount of time spent during ambulation (F1,153 = 45.2, p < 0.001) and a smaller step count (F1,153 = 45.1, p < 0.001) in patients compared to controls.
Table 2.
In- and off-laboratory gait and mobility assessment
| Healthy subjects | Cerebellar disorders | F1,153 | p | |
|---|---|---|---|---|
| In-laboratory gait measures | ||||
| Gait velocity [m/s] | 1.2 ± 0.2 | 0.9 ± 0.2* | 39.9 | < 0.001 |
| Stride length [m] | 1.3 ± 0.2 | 1.1 ± 0.3 | 34.5 | < 0.001 |
| Stride time [s] | 1.1 ± 0.1 | 1.3 ± 0.1* | 9.0 | 0.003 |
| Swing phase [%] | 38 ± 2 | 37 ± 2 | 63.4 | < 0.001 |
| Double support phase [%] | 24 ± 4 | 30 ± 9* | 22.6 | < 0.001 |
| Base of support [m] | 0.08 ± 0.03 | 0.15 ± 0.06* | 65.7 | < 0.001 |
| Base of support CV [%] | 24 ± 15 | 24 ± 12 | 0.7 | n.s |
| Stride length CV [%] | 2.2 ± 1.1 | 5.6 ± 4.6* | 21.3 | < 0.001 |
| Stride time CV [%] | 2.3 ± 1.9 | 5.2 ± 1.2* | 28.1 | < 0.001 |
| Gait asymmetry index [%] | 2.5 ± 1.5 | 6.8 ± 3.7* | 24.5 | < 0.001 |
| Phase synchronization [%] | 4.1 ± 1.4 | 9.9 ± 5.3* | 17.3 | < 0.001 |
| Mobility | ||||
| Adherence | ||||
| Days recorded [d] | 12.3 ± 1.9 | 12.4 ± 1.2 | 0.2 | n.s |
| Time sensor worn [%] | 96 ± 12 | 99 ± 6 | 0.8 | n.s |
| Volume | ||||
| Median step count ± SD [#] | 10,208 ± 3,299 | 6305 ± 3,302* | 45.1 | < 0.001 |
| Mean ambulation % ± SD [%] | 8.7 ± 2.6 | 5.7 ± 2.5* | 45.2 | < 0.001 |
| Mean sedentary % ± SD [%] | 27.8 ± 7.7 | 34.5 ± 10.2* | 39.2 | < 0.001 |
| Median sleep % ± SD [%] | 40.8 ± 6.7 | 42.3 ± 9.9 | 0.9 | n.s |
| Activity | ||||
| Median ambulation bout # ± SD [#] | 456 ± 134 | 338 ± 139* | 23.8 | < 0.001 |
| Ambulatory bout duration ± SD [s] | 17 ± 5 | 15 ± 4 | 9.8 | .0.02 |
| Mean daily intensity ± SD [MET] | 35 ± 1 | 33 ± 1* | 48.7 | < 0.001 |
| Pattern | ||||
| Mean ambulation alpha ± SD | 1.42 ± 0.04 | 1.43 ± 0.04 | 1.5 | n.s |
| Median sit-walk transitions ± SD | 40 ± 16 | 36 ± 13 | 2.7 | n.s |
*indicates significant difference in the Sheffé posthoc comparison (to healthy subjects)
CV, coefficient of variation; SD, standard deviation
Fall Assessment
Fall epidemiology in patients and controls is summarized in Tables 1 and 3. Both retrospective and prospective assessments revealed a considerably higher incidence of fallers, recurrent fallers, and severe fall-related injuries in patients compared to controls. According to the retrospective fall assessment, 66% of patients had experienced at least one fall within the last 6 months and 66% of those reported recurrent falling. Thirty-six percent of patients that fell reported fall-related injuries that required medical attention.
Table 3.
Outcomes form prospective fall assessment
| Proportion of fallers | Fall events | |||||||
|---|---|---|---|---|---|---|---|---|
|
Healthy subjects N = 56 |
Cerebellar disorders N = 80 |
Healthy subjects N = 9 falls |
Cerebellar disorders N = 313 |
|||||
| N | % | N | % | N | % | N | % | |
| Fall epidemiology | ||||||||
| No falls | 47 | 84 | 28 | 36 | – | – | – | – |
| Occasional fall | 9 | 16 | 18 | 23 | 9 | 100 | 18 | 6 |
| Frequent falls | 0 | 0 | 34 | 41 | 0 | 0 | 295 | 94 |
| Chi2 < 0.001 | Chi2 < 0.001 | |||||||
| Fall severity (HFGS) | N = 18 | N = 925 | ||||||
| 1 | 4 | 31 | 10 | 14 | 9 | 50 | 612 | 66 |
| 2 | 5 | 38 | 44 | 63 | 5 | 20 | 293 | 32 |
| 3 | 4 | 31 | 14 | 18 | 4 | 22 | 16 | 2 |
| 4 | 0 | 0 | 4 | 5 | 0 | 0 | 4 | 0 |
| Chi2 < 0.001 | Chi2 < 0.001 | |||||||
Information on falls epidemiology on considers actual fall events (excluding near-fall events). Information on falls severity considers both fall and near-fall events
HFGS, Hopkins falls grading scale
In the 6-month follow-up period, all participants registered fall information in their falls diary that was counter-checked by structured monthly telephone interviews. Falls diary information from 13 patients and 7 controls was considered invalid and excluded from further analysis due to missing telephone contact data or discrepancies between the falls information documented in the diary and surveyed during monthly phone interviews. The total numbers of near-fall and fall events in patients registered during prospective assessment were 313 and 612, respectively. A majority of falls in patients with CA occurred indoors (75%) in domestic environments (71%), primarily on even surface (71%). Most falls were linked to walking or turning (71%) and 74% occurred either forwards or backwards. The most frequent causes of falling in patients were tripping (33%) and imbalance (23%). The most commonly reported associated symptoms with falling were dizziness and vertigo (20%) (Tables 3 and 4). A total of 5% of falls resulted in injuries which required outpatient medical treatment and 1% necessitated inpatient medical treatment. Overall, outcomes from prospective assessment revealed that 64% of the patients experienced falls within the 6-month follow-up period and 65% of these patients reported recurrent falling. Severe fall-related injuries that required medical attention occurred in 29% of patients that fell (Table 3).
Table 4.
Prospective fall assessment. Fall event details
| fall events | ||||
|---|---|---|---|---|
|
Healthy subjects n = 9 falls |
Cerebellar disorders n = 313 |
|||
| n | % | n | % | |
| Environment | ||||
| At home | 2 | 22 | 207 | 71 |
| Public area | 7 | 78 | 83 | 29 |
| Indoor | 2 | 22 | 218 | 75 |
| Outdoor | 7 | 78 | 72 | 25 |
| xx | ||||
| Even underground | 0 | 0 | 210 | 71 |
| Uneven underground | 1 | 11 | 10 | 3 |
| Stairs | 1 | 11 | 28 | 9 |
| Incline/decline | 2 | 22 | 4 | 1 |
| Slippery underground | 3 | 33 | 22 | 7 |
| Obstacle | 2 | 22 | 21 | 7 |
| Circumstances | ||||
| Morning | 0 | 0 | 84 | 28 |
| Noon/afternoon | 4 | 44 | 144 | 48 |
| Evening | 5 | 56 | 68 | 23 |
| Night | 0 | 0 | 16 | 5 |
| Symptoms | ||||
| Dizziness/ vertigo | 0 | 0 | 60 | 20 |
| Pain | 2 | 22 | 5 | 2 |
| Weakness | 0 | 0 | 6 | 2 |
| Collapse | 0 | 0 | 14 | 4 |
| Not specified | 7 | 78 | 228 | 73 |
| Sequence | ||||
| Anterior | 5 | 56 | 101 | 35 |
| Posterior | 1 | 11 | 114 | 39 |
| Lateral | 3 | 33 | 69 | 24 |
| Vertical | 0 | 0 | 5 | 2 |
| Tripping | 2 | 22 | 104 | 38 |
| Slipping | 3 | 33 | 22 | 8 |
| Weakness | 0 | 0 | 46 | 17 |
| Instability/ imbalance | 0 | 0 | 88 | 32 |
| Inattentive | 0 | 0 | 9 | 3 |
| Contact/ collision | 4 | 44 | 3 | 1 |
| Behavior | ||||
| Locomotion | 4 | 44 | 203 | 66 |
| Transition | 0 | 0 | 42 | 14 |
| Turning | 2 | 22 | 16 | 5 |
| Reaching/leaning | 0 | 0 | 26 | 9 |
| Standing | 0 | 0 | 19 | 6 |
| Sitting | 0 | 0 | 1 | 0 |
| Doing sports/ activities | 3 | 33 | 2 | 0 |
Multivariate Fall Classification Models
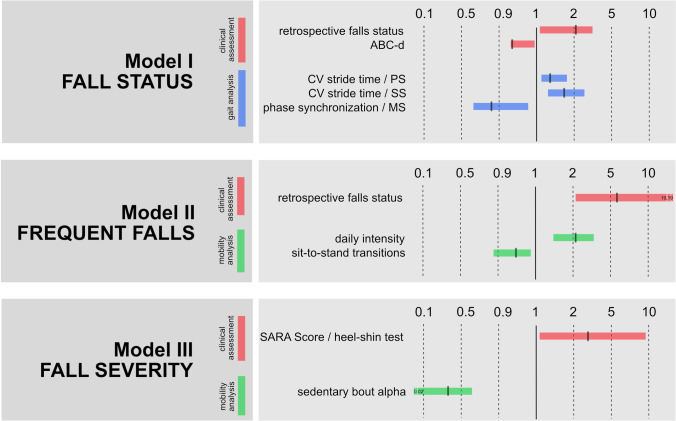
The predictive model for fall status (non-faller vs. faller) was obtained after 10 iteration steps and yielded a correct prediction of 78% (sensitivity: 70%; specificity: 86%). The model included 5 predictive factors from socio-demographic and in-laboratory mobility assessment, with the most important being retrospective fall status and CV of stride time during slow walking (Table 5, Section A). Accordingly, a positive history of falls and impaired dynamic walking stability were the most important risk factors for experiencing falls during follow-up. No parameter of the mobility assessment was significantly represented in this model (Fig. 1).
Table 5.
Multivariate fall classification models
| Model information | Parameter information | |||||||
|---|---|---|---|---|---|---|---|---|
| Correct prediction | Coefficient | SE | df | p | Exp(b) | Low | High | |
| A)Model fall status | 0.78 | |||||||
| Retrospective fall status | 0.73 | 0.32 | 1 | 0.024 | 2.076 | 1.101 | 3.913 | |
| ABC-d | − 0.03 | 0.02 | 1 | 0.037 | 0.936 | 0.940 | 0.998 | |
| Preferred walking—CV of stride time | 0.25 | 0.16 | 1 | 0.045 | 1.289 | 1.102 | 1.761 | |
| Slow walking—CV of stride time | 0.65 | 0.21 | 1 | 0.031 | 1.757 | 1.251 | 2.321 | |
| Maximally fast walking—phase synchronization index | − 0.22 | 0.10 | 1 | 0.022 | 0.806 | 0.670 | 0.971 | |
| B)Model fall frequency | 0.81 | |||||||
| Retrospective fall status | 1.92 | 0.53 | 1 | 0.001 | 6.850 | 2.445 | 19.193 | |
| Daily intensity | 0.83 | 0.12 | 1 | 0.004 | 2.382 | 1.309 | 4.333 | |
| Sit-to-stand transitions | − 0.059 | 0.03 | 1 | 0.033 | 0.943 | 0.893 | 0.995 | |
| C)Model fall severity | 0.84 | |||||||
| SARA subscore heel-shin test | 1.16 | 0.56 | 1 | 0.037 | 3.201 | 1.075 | 9.534 | |
| Sedentary bout alpha | − 1.45 | 0.95 | 1 | 0.044 | 0.467 | 0.002 | 0.621 | |
ABC-d, activity-specific balance confidence scale; CV, coefficient of variation; SARA, scale for the assessment and rating of ataxia
Fig. 1.
Overview of predictive factors for fall events in CA. Figure 1 represents the relevant factors for predicting fall events (model 1-3). Paramters were derived from clinical assessment (red), in-laboratory gait assessment (green), and off-laboratory mobility assessment (blue). Bars represent the limits of Exp(beta) of the regression models
The predictive model for fall frequency (occasional vs. frequent faller) was obtained after 13 iteration steps and achieved a correct prediction of 81% (sensitivity: 77%; specificity: 86%). The model included 3 predictive variables from socio-demographic and off-laboratory mobility assessment, with the most influential being retrospective fall status and intensity of daily mobility (Table 5, Section B). Thus, besides a positive history of falls, high daily activity levels were an independent risk factor for frequent falling during follow-up assessment (Fig. 1).
The predictive model for fall severity (falls that do vs. do not necessitate medical attention) was obtained after 12 iteration steps and yielded a correct prediction of 84% (sensitivity: 75%; specificity: 89%). The model only considered 2 parameters from clinical and off-laboratory mobility assessment, namely the subscore “heel-shin-test” of the SARA and ambulatory bout alpha (Table 5, Section C) (Fig. 1).
Discussion
In this study, we prospectively assessed the occurrence, circumstances, and consequences of falling in patients with CA and evaluated the predictive validity of a comprehensive set of clinical and instrument-based health and mobility screening tools for identifying patients at particular risk of falling. Prospective fall assessment revealed that recurrent and injurious falling is a severe complication already in mild-to-moderate stages of cerebellar disease. Multimodal measures from clinical and in- and off-laboratory assessment not only allowed us to characterize complementary aspects of the health and mobility status in CA, but were differentially associated with distinct aspects of fall events in patients with CA. Consequently, multivariate fall risk classification models built on these measures yielded a high accuracy that outperforms previous models that only considered a limited set of explanatory characteristics. In the following, we will discuss the differential relationships between health status, mobility impairments, and risk of falling in CA and propose a guideline for a multi-level, stepwise approach for fall risk assessment in these patients.
Balance and Mobility Impairments
Patients in our study cohort had different etiologies of CA at rather early stages of disease with mild-to-moderate symptom severity indicated by an average SARA score of 10 points. Clinical scoring of functional mobility in these patients demonstrated moderate impairments of balance and gait regulation. In-laboratory gait assessment showed corresponding walking characteristic of the staggering, broad-based phenotype of ataxic gait disorders [9, 10, 34, 35]. Balance and gait impairments both correlated with disease severity and were linked to reduced subjective balance confidence and increased fear of falling [11].
Off-laboratory measures of daily activity indicated only mildly affected everyday mobility behavior in patients with moderate reductions in the total volume and intensity of ambulatory activity. Patients performed less and shorter periods of walking and spent more time with sedentary behavior. In contrast to gait impairments, alterations of patients’ daily mobility were mainly independent of ataxia severity. Correspondingly, patients reported near-to-normal health-related quality of life scores (SF-12). In contrast, two previous studies on patients with hereditary CA at more advanced disease stages observed considerable impairments of daily activities that correlated with the severity of ataxic symptoms [18, 19]. Our findings thus suggest that despite apparent balance and gait impairments, patients at early stages of CA are more or less able to manage and retain near-to-normal daily life activity. However, impairments of daily mobility become increasingly relevant and disabling for patients at later stages of disease.
Fall Epidemiology and Fall Risk Prediction in Patients with CA
Our 6-month prospective fall assessment in patients with CA revealed a fall incidence of 64%, which lies within the range of previous reports (50% in a 3-month assessment period [3], 84% in a 1-year assessment period [5]). Two third of patients that fell experienced recurrent falling and one third suffered from severe fall-related injuries requiring medical attention or even inpatient treatment. Furthermore, direct comparison of retrospective and prospective fall assessment outcomes revealed a high agreement with respect to fall status, frequency, and severity. Overall, retrospective and prospective fall assessment outcomes both emphasize that recurrent and injurious falling are frequent already in early stages of CA despite the relatively preserved ability for independent ambulation in these patients.
The relative distribution of circumstances and mechanisms of falling reported during prospective assessment largely confirm previous reports [3, 5, 36]. Most falls of patients occurred indoors on even ground and were linked to activities of walking or turning. The predominant causes of falling were tripping and postural instability. This suggests that most falls in CA are intrinsically generated in situations where the center of mass moves outside the base of support and dynamic balance cannot be adequately recovered. Furthermore, the high rate of sensations of vertigo and dizziness associated with falling in patients supports previous reports on the prevalence of persistent or episodic symptoms of vertigo and dizziness in CA (“cerebellar vertigo and dizziness”) [37, 38].
To identify explanatory variables that may predict patients’ fall status, frequency, and severity, we performed multiple regression analyses considering a wide range of sociodemographic characteristics and outcomes from clinical, self-report-based, and in- and off-laboratory gait and mobility assessments. Predictive models for each of the three fall categories yielded good-to-excellent classificatory performance (78–84%) with respect to previously established criteria for fall risk assessment approaches [39, 40]. Accurate prediction of both patients’ general fall status and fall frequency primarily relied on information from retrospective fall assessment. Accordingly, the presence of previous falls was the single most influential predictor for experiencing one or more falls in the 6-month follow-up assessment. Analogous to fall risk assessment guidelines in geriatric populations [41, 42], this finding suggests that patients’ fall history should be routinely surveyed during general medical history taking in order to readily identify those patients with particular risk of falling.
Prediction of patients’ general fall status further relied on parameters from in-laboratory gait assessment. In particular, increased levels of gait variability were identified as an independent risk factor for experiencing falls during the follow-up period. Irregularity of movement is a defining characteristic of cerebellar disease [9, 10] and scales with the severity of symptoms [11]. A close link between increased gait variability and the risk of falling in CA has been previously reported based on retrospective fall information [2, 11]. This relationship has been shown most prominent for slow walking modes. The current findings further complement this hypothesis by giving evidence for a relationship between gait variability at preferred speed walking and prospective fall risk in patients with CA. Gait irregularities have been particularly linked to the risk of falling due to tripping during undisturbed walking [36] — the most prevalent fall mechanism in our cohort. Thus, instrument-based measures of gait instability appear to be a suitable tool to improve the baseline fall risk estimation in patients with CA.
Interestingly, off-laboratory mobility measures did not contribute to basic fall risk estimation, but only became relevant for identifying patients at particular risk of frequent and injurious falling. Accordingly, prolonged periods of sedentary activity were predictive for injurious falling and a higher intensity of daily-life activity for experiencing recurrent falls. At first glance, the latter observation appears to be counterintuitive, since the level of daily physical activity is considered a global health marker [43] and training-induced increases in physical activity were found to not affect the risk of falling in an elderly population [44]. However, previous reports correspondingly observed that higher amounts of physical activity are associated to an increased fall risk in the elderly population [45] and patients with early Parkinson’s disease [17]. This suggests that especially patients with early-stage gait impairments who still maintain near-to-normal levels of daily activity are at particular risk of experiencing recurrent falls during ambulation. It is reasonable that a higher exposure to gait instability during preserved daily activity behavior might influence the risk for frequent falling in a supraordinate way. This risk presumably not decreases before advanced disease stages that are linked to considerably reduced levels of daily activity [17]. However, a clinical advice to reduce ambulatory activity to protect patients from recurrent falling would not be appropriate due to the apparent neuroprotective effects of activity in these patients [46, 47]. Rather, a balance must be found between maintaining activity and applying protective measures that specifically minimize the risk of severe fall-related injuries in these patients.
Whether the severity of ataxic symptoms directly influences fall risk in patients with CA is still controversial. A retrospective study in patients with spinocerebellar ataxias found that fall frequency was associated with the severity of ataxia assessed by the SARA score [4]. However, this association failed to persist in a subsequent prospective follow-up study on the same cohort [5]. A more recent study including patients with sporadic and hereditary forms of CA found no evidence for an association between the SARA score and patients’ fall risk [11]. Our findings support the latter observations but further indicated that the severity of ataxic symptoms becomes relevant for estimating patients’ risk of injurious falling. Accordingly, an increase in the SARA score (subscore for limb ataxia) more than doubled the risk of experiencing fall-related injuries that necessitated medical attention. This association presumably points to a link between the severity of ataxic symptoms (particularly limb dysmetria) and the patients’ capacity for protective postural coping strategies to counterbalance falls. A severe discoordination of limb movements might thus interfere with and hinder the execution of protective postural adjustments while falling.
Taken together, the differential contributions from clinical and instrument-based measures for predicting fall risk in CA encourage and provide guidelines for a multi-level, stepwise fall risk assessment approach in these patients: Accordingly, basic index information that is readily available from medical history taking allows a good estimation of the general fall risk and may promptly inform the clinician which patient would or would not benefit from a more in-depth examination. For those patients at risk of falling, a more elaborate disease severity rating and measures from instrument-based gait and mobility examination provide additional, unique information with respect to the severity of their fall susceptibility and the likelihood for the occurrence of severe fall-related injuries.
Study Limitations
The cross-sectional study design yielded a heterogenous sample of patients including hereditary, sporadic, and secondary forms of CA which comfounds a direct comparison to previous reports that only considered hereditary forms of CA [4, 5, 18]. However, despite this heterogeneity and the preponderance of earlier disease stages in our cohort, we found a good agreement with existing literature regarding the epidemiology, circumstances, and mechanism of falling in CA. Furthermore, we did not analyze effects of medications on fall risk in our patients with CA. Medication status, in particular, the commonly described “fall risk increasing drugs” such as hypnotics, antipsychotics, antidepressants have been shown relevant risk factors for fall occurrence in geriatric populations [48] and should be considered in follow-up studies. Finally, we are aware that the applied technology for in- and off-laboratory assessment of gait and mobility function is elaborate and currently restricted to specialized clinical centers. However, relevant outcome measures of these assessments (e.g., gait variability or intensity of daily activities) have been shown to be reliably transferable to low-cost wearable technology [15, 49] that could in future facilitate a broad application of a multimodal fall risk screening in patients with CA.
Author Contribution
Conceptualization: R.S., M.D., T.B., and M.W.; Methodology: R.S., M.D., T.B., and M.W.; Data acquisition: R.S., A.H., J.D., F.S., and M.W.; Formal analysis and investigation: R.S., A.H., J.D., F.S., and M.W.; Manuscript draft: R.S. and M.W.; Manuscript revision: all authors; Funding acquisition: R.S. and M.W., Resources: R.S., M.W., M.D.
Funding
Open Access funding enabled and organized by Projekt DEAL. This study was supported by the German Federal Ministry for Education and Science (BMBF, IFB 01EO1401) and by the Else-Kroener-Fresenius Foundation (EKFES, 80766157.).
BMBF,IFB 01EO1401, Roman Schniepp, Else Kröner-Fresenius-Stiftung,EKFES, Roman Schniepp, 80766157, Roman Schniepp
Data Availability
Data reported in this article will be shared with any appropriately qualified investigator on request after pseudonymization.
Declarations
Ethics Approval
The study protocol was approved by the local Ethics Committee of the Ludwig-Maximilians University of Munich, (Nr. 421–13) and was conducted according to the Declaration of Helsinki.
Consent to Participate
All participants gave their informed written consent prior to the experiments.
Conflict of Interest
The authors declare no competing interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Buckley E, Mazza C, McNeill A. A systematic review of the gait characteristics associated with cerebellar ataxia. Gait Posture. 2018;60:154–163. doi: 10.1016/j.gaitpost.2017.11.024. [DOI] [PubMed] [Google Scholar]
- 2.Schniepp R, Wuehr M, Schlick C, Huth S, Pradhan C, Dieterich M, Brandt T, Jahn K. Increased gait variability is associated with the history of falls in patients with cerebellar ataxia. J Neurol. 2014;261:213–223. doi: 10.1007/s00415-013-7189-3. [DOI] [PubMed] [Google Scholar]
- 3.van de Warrenburg BP, Steijns JA, Munneke M, Kremer BP, Bloem BR. Falls in degenerative cerebellar ataxias. Mov Disord. 2005;20:497–500. doi: 10.1002/mds.20375. [DOI] [PubMed] [Google Scholar]
- 4.Fonteyn EM, Schmitz-Hubsch T, Verstappen CC, Baliko L, Bloem BR, Boesch S, Bunn L, Charles P, Durr A, Filla A, Giunti P, Globas C, Klockgether T, Melegh B, Pandolfo M, De Rosa A, Schols L, Timmann D, Munneke M, Kremer BP, van de Warrenburg BP. Falls in spinocerebellar ataxias: results of the EuroSCA fall study. Cerebellum. 2010;9:232–239. doi: 10.1007/s12311-010-0155-z. [DOI] [PubMed] [Google Scholar]
- 5.Fonteyn EM, Schmitz-Hubsch T, Verstappen CC, Baliko L, Bloem BR, Boesch S, Bunn L, Giunti P, Globas C, Klockgether T, Melegh B, Pandolfo M, Schols L, Timmann D, van de Warrenburg BP. Prospective analysis of falls in dominant ataxias. Eur Neurol. 2013;69:53–57. doi: 10.1159/000342907. [DOI] [PubMed] [Google Scholar]
- 6.Ilg W, Bastian AJ, Boesch S, Burciu RG, Celnik P, Claaßen J, Feil K, Kalla R, Miyai I, Nachbauer W, Schöls L, Strupp M, Synofzik M, Teufel J, Timmann D. Consensus paper: management of degenerative cerebellar disorders. Cerebellum. 2014;13:248–268. doi: 10.1007/s12311-013-0531-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Klockgether T, Mariotti C, Paulson HL. Spinocerebellar ataxia Nat Rev Dis Primers. 2019;5:24. doi: 10.1038/s41572-019-0074-3. [DOI] [PubMed] [Google Scholar]
- 8.Aizawa CY, Pedroso JL, Braga-Neto P, Callegari MR, Barsottini OG. Patients with autosomal dominant spinocerebellar ataxia have more risk of falls, important balance impairment, and decreased ability to function. Arq Neuropsiquiatr. 2013;71:508–511. doi: 10.1590/0004-282x20130094. [DOI] [PubMed] [Google Scholar]
- 9.Ilg W, Giese M, Gizewski E, Schoch B, Timmann D. The influence of focal cerebellar lesions on the control and adaptation of gait. Brain. 2008;131:2913–2927. doi: 10.1093/brain/awn246. [DOI] [PubMed] [Google Scholar]
- 10.Ilg W, Golla H, Thier P, Giese MA. Specific influences of cerebellar dysfunctions on gait. Brain. 2007;130:786–798. doi: 10.1093/brain/awl376. [DOI] [PubMed] [Google Scholar]
- 11.Schniepp R, Schlick C, Pradhan C, Dieterich M, Brandt T, Jahn K, Wuehr M. The interrelationship between disease severity, dynamic stability, and falls in cerebellar ataxia. J Neurol. 2016;263:1409–1417. doi: 10.1007/s00415-016-8142-z. [DOI] [PubMed] [Google Scholar]
- 12.Chini G, Ranavolo A, Draicchio F, Casali C, Conte C, Martino G, Leonardi L, Padua L, Coppola G, Pierelli F, Serrao M. Local Stability of the trunk in patients with degenerative cerebellar ataxia during walking. The Cerebellum. 2017;16:26–33. doi: 10.1007/s12311-016-0760-6. [DOI] [PubMed] [Google Scholar]
- 13.Schlick C, Rasoul A, Wuehr M, Gerth J, Dieterich M, Brandt T, Jahn K, Schniepp R. Gait variability predicts a subset of falls in cerebellar gait disorders. J Neurol. 2017;264:2322–2324. doi: 10.1007/s00415-017-8634-5. [DOI] [PubMed] [Google Scholar]
- 14.Warmerdam E, Hausdorff JM, Atrsaei A, Zhou Y, Mirelman A, Aminian K, Espay AJ, Hansen C, Evers LJW, Keller A, Lamoth C, Pilotto A, Rochester L, Schmidt G, Bloem BR and Maetzler W. Long-term unsupervised mobility assessment in movement disorders. Lancet Neurol 202010.1016/s1474-4422(19)30397-7 [DOI] [PubMed]
- 15.Ilg W, Seemann J, Giese M, Traschütz A, Schöls L, Timmann D and Synofzik M. Real-life gait assessment in degenerative cerebellar ataxia: towards ecologically valid biomarkers. Neurology 2020:10.1212/WNL.0000000000010176. doi 10.1212/wnl.0000000000010176 [DOI] [PubMed]
- 16.Wuehr M, Huppert A, Schenkel F, Decker J, Jahn K and Schniepp R. Independent domains of daily mobility in patients with neurological gait disorders. Journal of Neurology 202010.1007/s00415-020-09893-2 [DOI] [PMC free article] [PubMed]
- 17.Mactier K, Lord S, Godfrey A, Burn D, Rochester L. The relationship between real world ambulatory activity and falls in incident Parkinson’s disease: influence of classification scheme. Parkinsonism Relat Disord. 2015;21:236–242. doi: 10.1016/j.parkreldis.2014.12.014. [DOI] [PubMed] [Google Scholar]
- 18.Srulijes K, Klenk J, Schwenk M, Schatton C, Schwickert L, Teubner-Liepert K, Meyer M, K CS, Maetzler W, Becker C and Synofzik M. Fall risk in relation to individual physical activity exposure in patients with different neurodegenerative diseases: a pilot study. Cerebellum 2019: 18:340–8. doi 10.1007/s12311-018-1002-x [DOI] [PubMed]
- 19.Subramony SH, Kedar S, Murray E, Protas E, Xu H, Ashizawa T, Tan A. Objective home-based gait assessment in spinocerebellar ataxia. J Neurol Sci. 2012;313:95–98. doi: 10.1016/j.jns.2011.09.016. [DOI] [PubMed] [Google Scholar]
- 20.de Silva R, Greenfield J, Cook A, Bonney H, Vallortigara J, Hunt B, Giunti P. Guidelines on the diagnosis and management of the progressive ataxias. Orphanet J Rare Dis. 2019;14:8. doi: 10.1186/s13023-019-1013-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Zecevic AA, Salmoni AW, Speechley M, Vandervoort AA. Defining a fall and reasons for falling: comparisons among the views of seniors, health care providers, and the research literature. Gerontologist. 2006;46:367–376. doi: 10.1093/geront/46.3.367. [DOI] [PubMed] [Google Scholar]
- 22.Davalos-Bichara M, Lin FR, Carey JP, Walston JD, Fairman JE, Schubert MC, Barron JS, Hughes J, Millar JL, Spar A, Weber KL, Ying HS, Zackowski KM, Zee DS, Agrawal Y. Development and validation of a falls-grading scale. J Geriatr Phys Ther. 2013;36:63–67. doi: 10.1519/JPT.0b013e31825f6777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Greenberg SA. Analysis of measurement tools of fear of falling for high-risk, community-dwelling older adults. Clin Nurs Res. 2012;21:113–130. doi: 10.1177/1054773811433824. [DOI] [PubMed] [Google Scholar]
- 24.Powell LE, Myers AM. The activities-specific balance confidence (ABC) scale. J Gerontol A Biol Sci Med Sci. 1995;50:M28–M34. doi: 10.1093/gerona/50A.1.M28. [DOI] [PubMed] [Google Scholar]
- 25.Fisher KJ, Li F. A community-based walking trial to improve neighborhood quality of life in older adults: a multilevel analysis. Ann Behav Med. 2004;28:186–194. doi: 10.1207/s15324796abm2803_7. [DOI] [PubMed] [Google Scholar]
- 26.Nasreddine ZS, Phillips NA, Bedirian V, Charbonneau S, Whitehead V, Collin I, Cummings JL, Chertkow H. The Montreal Cognitive Assessment, MoCA: a brief screening tool for mild cognitive impairment. J Am Geriatr Soc. 2005;53:695–699. doi: 10.1111/j.1532-5415.2005.53221.x. [DOI] [PubMed] [Google Scholar]
- 27.Podsiadlo D, Richardson S. The timed “Up & Go”: a test of basic functional mobility for frail elderly persons. J Am Geriatr Soc. 1991;39:142–148. doi: 10.1111/j.1532-5415.1991.tb01616.x. [DOI] [PubMed] [Google Scholar]
- 28.Thieme H, Ritschel C, Zange C. Reliability and validity of the functional gait assessment (German version) in subacute stroke patients. Arch Phys Med Rehabil. 2009;90:1565–1570. doi: 10.1016/j.apmr.2009.03.007. [DOI] [PubMed] [Google Scholar]
- 29.Schmitz-Hübsch T, du Montcel ST, Baliko L, Berciano J, Boesch S, Depondt C, Giunti P, Globas C, Infante J, Kang JS, Kremer B, Mariotti C, Melegh B, Pandolfo M, Rakowicz M, Ribai P, Rola R, Schols L, Szymanski S, van de Warrenburg BP, Durr A, Klockgether T, Fancellu R. Scale for the assessment and rating of ataxia: development of a new clinical scale. Neurology. 2006;66:1717–1720. doi: 10.1212/01.wnl.0000219042.60538.92. [DOI] [PubMed] [Google Scholar]
- 30.Plotnik M, Giladi N, Hausdorff JM. A new measure for quantifying the bilateral coordination of human gait: effects of aging and Parkinson’s disease. Exp Brain Res. 2007;181:561–570. doi: 10.1007/s00221-007-0955-7. [DOI] [PubMed] [Google Scholar]
- 31.Rochester L, Chastin SFM, Lord S, Baker K, Burn DJ. Understanding the impact of deep brain stimulation on ambulatory activity in advanced Parkinson’s disease. J Neurol. 2012;259:1081–1086. doi: 10.1007/s00415-011-6301-9. [DOI] [PubMed] [Google Scholar]
- 32.Lord S, Chastin SF, McInnes L, Little L, Briggs P, Rochester L. Exploring patterns of daily physical and sedentary behaviour in community-dwelling older adults. Age Ageing. 2011;40:205–210. doi: 10.1093/ageing/afq166. [DOI] [PubMed] [Google Scholar]
- 33.Galperin I, Hillel I, Del Din S, Bekkers EMJ, Nieuwboer A, Abbruzzese G, Avanzino L, Nieuwhof F, Bloem BR, Rochester L, Della Croce U, Cereatti A, Giladi N, Mirelman A, Hausdorff JM. Associations between daily-living physical activity and laboratory-based assessments of motor severity in patients with falls and Parkinson’s disease. Parkinsonism Relat Disord. 2019;62:85–90. doi: 10.1016/j.parkreldis.2019.01.022. [DOI] [PubMed] [Google Scholar]
- 34.Schniepp R, Möhwald K and Wuehr M. Gait ataxia in humans: vestibular and cerebellar control of dynamic stability. J Neurol 201710.1007/s00415-017-8482-3 [DOI] [PubMed]
- 35.Wuehr M, Schniepp R, Ilmberger J, Brandt T, Jahn K. Speed-dependent temporospatial gait variability and long-range correlations in cerebellar ataxia. Gait Posture. 2013;37:214–218. doi: 10.1016/j.gaitpost.2012.07.003. [DOI] [PubMed] [Google Scholar]
- 36.Schlick C, Rasoul A, Wuehr M, Gerth J, Dieterich M, Brandt T, Jahn K and Schniepp R. Gait variability predicts a subset of falls in cerebellar gait disorders. Journal of Neurology 201710.1007/s00415-017-8634-5 [DOI] [PubMed]
- 37.Feil K, Strobl R, Schindler A, Krafczyk S, Goldschagg N, Frenzel C, Glaser M, Schoberl F, Zwergal A, Strupp M. What is behind cerebellar vertigo and dizziness? Cerebellum. 2019;18:320–332. doi: 10.1007/s12311-018-0992-8. [DOI] [PubMed] [Google Scholar]
- 38.Yu-Wai-Man P, Gorman G, Bateman DE, Leigh RJ, Chinnery PF. Vertigo and vestibular abnormalities in spinocerebellar ataxia type 6. J Neurol. 2009;256:78–82. doi: 10.1007/s00415-009-0068-2. [DOI] [PubMed] [Google Scholar]
- 39.Oliver D, Daly F, Martin FC, McMurdo ME. Risk factors and risk assessment tools for falls in hospital in-patients: a systematic review. Age Ageing. 2004;33:122–130. doi: 10.1093/ageing/afh017. [DOI] [PubMed] [Google Scholar]
- 40.Nunan S, Brown Wilson C, Henwood T, Parker D. Fall risk assessment tools for use among older adults in long-term care settings: a systematic review of the literature. Australas J Ageing. 2018;37:23–33. doi: 10.1111/ajag.12476. [DOI] [PubMed] [Google Scholar]
- 41.Lusardi MM, Fritz S, Middleton A, Allison L, Wingood M, Phillips E, Criss M, Verma S, Osborne J, Chui KK. Determining risk of falls in community dwelling older adults: a systematic review and meta-analysis using posttest probability. J Geriatr Phys Ther. 2017;40:1–36. doi: 10.1519/jpt.0000000000000099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Skelton DA, Todd CJ and ProFa NEG. Prevention of falls network Europe: a thematic network aimed at introducing good practice in effective falls prevention across Europe. Four years on. J Musculoskelet Neuronal Interact 2007: 7:273–8. [PubMed]
- 43.Healy GN, Matthews CE, Dunstan DW, Winkler EA, Owen N. Sedentary time and cardio-metabolic biomarkers in US adults: NHANES 2003–06. Eur Heart J. 2011;32:590–597. doi: 10.1093/eurheartj/ehq451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Zieschang T, Schwenk M, Becker C, Uhlmann L, Oster P, Hauer K. Falls and physical activity in persons with mild to moderate dementia participating in an intensive motor training: randomized controlled trial. Alzheimer Dis Assoc Disord. 2017;31:307–314. doi: 10.1097/WAD.0000000000000201. [DOI] [PubMed] [Google Scholar]
- 45.Chan BK, Marshall LM, Winters KM, Faulkner KA, Schwartz AV, Orwoll ES. Incident fall risk and physical activity and physical performance among older men: the osteoporotic fractures in men study. Am J Epidemiol. 2007;165:696–703. doi: 10.1093/aje/kwk050. [DOI] [PubMed] [Google Scholar]
- 46.Keller JL, Bastian AJ. A home balance exercise program improves walking in people with cerebellar ataxia. Neurorehabil Neural Repair. 2014;28:770–778. doi: 10.1177/1545968314522350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Ahlskog JE. Does vigorous exercise have a neuroprotective effect in Parkinson disease? Neurology. 2011;77:288–294. doi: 10.1212/WNL.0b013e318225ab66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Hart LA, Phelan EA, Yi JY, Marcum ZA, Gray SL. Use of fall risk-increasing drugs around a fall-related injury in older adults: a systematic review. J Am Geriatr Soc. 2020;68:1334–1343. doi: 10.1111/jgs.16369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Arcuria G, Marcotulli C, Amuso R, Dattilo G, Galasso C, Pierelli F, Casali C. Developing a smartphone application, triaxial accelerometer-based, to quantify static and dynamic balance deficits in patients with cerebellar ataxias. J Neurol. 2020;267:625–639. doi: 10.1007/s00415-019-09570-z. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Data reported in this article will be shared with any appropriately qualified investigator on request after pseudonymization.