Abstract
Shigella sonnei, the main cause of bacillary dysentery in high-income countries, has become increasingly resistant to antibiotics. We monitored the antimicrobial susceptibility of 7121 S. sonnei isolates collected in France between 2005 and 2021. We detected a dramatic increase in the proportion of isolates simultaneously resistant to ciprofloxacin (CIP), third-generation cephalosporins (3GCs) and azithromycin (AZM) from 2015. Our genomic analysis of 164 such extensively drug-resistant (XDR) isolates identified 13 different clusters within CIP-resistant sublineage 3.6.1, which was selected in South Asia ∼15 years ago. AZM resistance was subsequently acquired, principally through IncFII (pKSR100-like) plasmids. The last step in the development of the XDR phenotype involved various extended-spectrum beta-lactamase genes (blaCTX-M-3, blaCTX-M-15, blaCTX-M-27, blaCTX-M-55, and blaCTX-M-134) carried by different plasmids (IncFII, IncI1, IncB/O/K/Z) or even integrated into the chromosome, and encoding resistance to 3GCs. This rapid emergence of XDR S. sonnei, including an international epidemic strain, is alarming, and good laboratory-based surveillance of shigellosis will be crucial for informed decision-making and appropriate public health action.
Subject terms: Bacterial genomics, Policy and public health in microbiology, Clinical microbiology, Epidemiology
There have been increasing reports of extensively drug-resistant (XDR) Shigella sonnei infections in recent years. In this laboratory surveillance study from France, the authors document the rise of XDR isolates from 2005 to 2021 and perform whole genome sequencing to investigate their genomic diversity and evolutionary history.
Introduction
Shigella species (now serogroups) are specialized lineages of Escherichia coli causing invasive intestinal infections in humans, ranging from acute watery diarrhea to dysenteric syndrome. Most people recover spontaneously from shigellosis, but antibiotic therapy is recommended for adults and children with bloody diarrhea, patients at risk of complications or to stop transmission in certain outbreak-prone settings1–3. The drugs currently used are ciprofloxacin (CIP), ceftriaxone (a third-generation cephalosporin, 3GC), and azithromycin (AZM). There are four serogroups of Shigella, with S. sonnei the predominant serogroup circulating in industrialized countries and emerging worldwide, even in countries in which other Shigella serogroups have traditionally predominated4. In 2019, 8848 confirmed cases of shigellosis were reported by 30 countries of the European Union (including the UK) and the European Economic Area; 59.4% were due to S. sonnei5. Many multidrug-resistant (MDR) strains of S. sonnei have been described in recent years. MDR strains were originally linked to travel to Asia or circulation in particular communities or networks, such as gay, bisexual and other men who have sex with men (GBMSM)6–14. However, S. sonnei isolates simultaneously resistant to the three recommended antimicrobial drugs (CIP, 3GCs, and AZM), described as “extensively drug-resistant” (XDR), were reported only exceptionally before 2022 and were often linked to Southeast Asia7,9,11. A study in the context of high-income countries revealed that only 0.8% (36/4222) of the 4222 S. sonnei genomic sequences obtained in the framework of public health surveillance in England, Australia and the US between 2016 and 2019 were inferred to be XDR15. However, no phenotypic antimicrobial susceptibility data were provided to confirm the XDR phenotype. In 2022, the United Kingdom Health Security Agency (UKHSA) and the European Center for Disease Prevention and Control (ECDC) reported an increase in XDR S. sonnei infections in 10 European countries, with France and the UK the most seriously affected5.
In this study, based on a combination of long-term conventional laboratory surveillance data and high-resolution whole-genome analyses, we investigated the timeline of XDR S. sonnei emergence in France, and the genomic diversity and recent evolution of these strains.
Results and discussion
Antimicrobial susceptibility data of S. sonnei in France
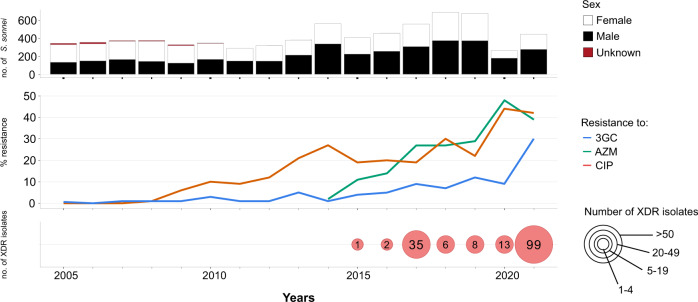
Our review of S. sonnei antimicrobial susceptibility data obtained between 2005 and 2021 (based on 7121 isolates received and confirmed at the French National Reference Center for E. coli, Shigella and Salmonella, FNRC-ESS, Institut Pasteur) revealed a sharp increase in the percentage of isolates resistant to 3GCs, CIP and AZM (P < 0.001) (Fig. 1). A first isolate resistant to 3GCs was identified as early as 2005 (prevalence of 1/342, 0.3%), whereas the first isolates resistant to CIP were identified in 2008 (prevalence of 2/373, 0.5%). Ten isolates resistant to AZM were detected in 2014, the year in which screening for such resistance began (in April) (10/557, 1.8%). In 2021, 29.5% (131/444) of S. sonnei isolates were resistant to 3GCs, 42.3% (188/444) were resistant to CIP and 38.7% (172/444) were resistant to AZM (Fig. 1). The first XDR S. sonnei isolate was identified in 2015, and an additional 163 XDR isolates have since been obtained (Fig. 1 and Supplementary Tables 1 and 2). All but one of these 164 XDR isolates were collected from mainland France. The percentage of XDR S. sonnei isolates increased over the study period (P < 0.001), peaking at 22.3% (99/444) in 2021. All but one of these XDR isolates were also resistant to trimethoprim/sulfamethoxazole and 87.2% (143/164) were resistant to tetracyclines. All these isolates remained susceptible to carbapenems.
Fig. 1. Laboratory surveillance of Shigella sonnei infections in France, 2005–2021.
The upper panel shows the number of isolates, by patient sex, received and analyzed per year. The middle panel shows the percentage of isolates displaying phenotypic resistance to ciprofloxacin (CIP), azithromycin (AZM), or third-generation cephalosporins (3GCs). The lower panel shows the number of extensively drug-resistant (XDR) S. sonnei isolates detected per year. XDR means combined resistance to CIP, AZM, and 3GCs.
One limitation to this study is that shigellosis is not a mandatory notifiable disease in France. Thus, laboratory-based surveillance, despite its official introduction 50 years ago, captures only a portion of the shigellosis burden (see Methods). The COVID-19 pandemic also had a clear impact on our surveillance data, as the number of S. sonnei infections in France decreased sharply in 2020 (as also reported elsewhere in Europe5) and, to a lesser extent, in 2021. This decrease may reflect a true decrease in the number of cases due to travel restrictions (43 and 30% decreases in the proportion of S. sonnei cases reporting international travel in 2020 and 2021, respectively, relative to 2019, in our surveillance data), stricter hygiene measures, lockdowns and school closures. However, it may also reflect poorer access to the healthcare system for patients with mild infections. In addition, it is not possible to rule out better referral to the FNRC-ESS of unusual XDR isolates by the clinical laboratories participating in our network. The high percentage of XDR S. sonnei observed in 2021 might, therefore, constitute an overestimate. However, in 2022, the year in which most public health measures against COVID-19 were lifted in France, XDR S. sonnei still accounted for 21.7% (73/336) of all S. sonnei isolates received from January 1 to August 31 at the FNRC-ESS.
Phylogenomics of the XDR S. sonnei isolates in France
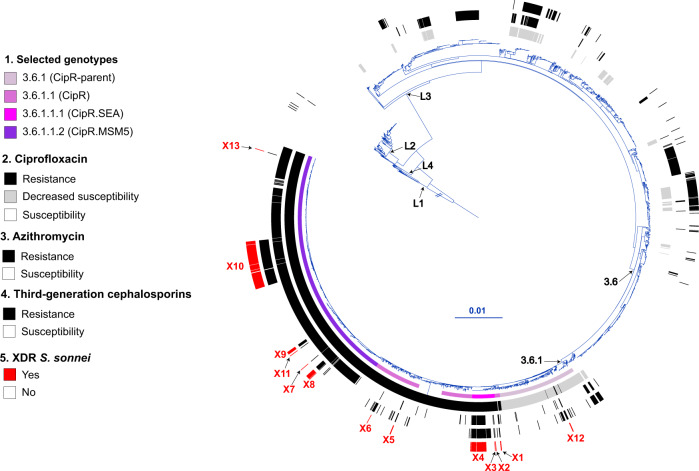
We investigated phylogenetic relationships by sequencing the genomes of the 164 French XDR S. sonnei isolates. We also included 2976 genomes from isolates and historical strains from the FNRC-ESS collected between 1945 and 2021 in the phylogenomic analysis, to provide a phylogenetic context for the XDR S. sonnei isolates. A maximum likelihood (ML) phylogenetic tree was built from an alignment of 59,295 chromosomal recombination-filtered single-nucleotide variants (SNVs) to obtain an accurate view of the evolutionary relationships (vertical evolution) between S. sonnei strains. This ML tree revealed that the 164 XDR S. sonnei isolates were not grouped into a single cluster, instead forming 13 different clusters (X1 to X13), all within lineage L3, the predominant lineage of S. sonnei worldwide over the last two decades (Fig. 2)15,16.
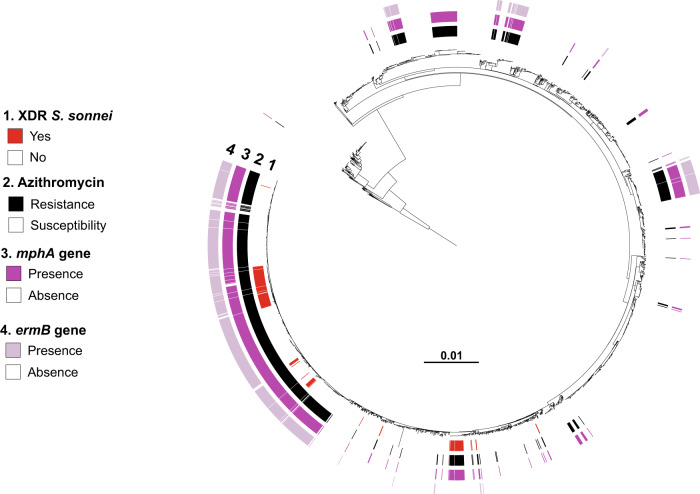
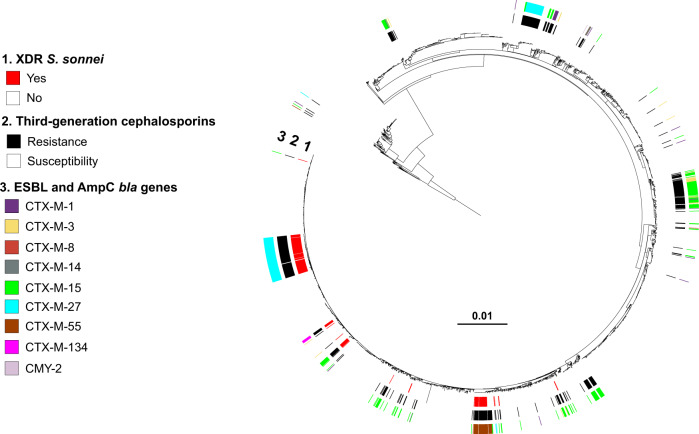
Fig. 2. Maximum-likelihood phylogeny of 3141 S. sonnei genomic sequences.
The S. sonnei reference genome 53G (GenBank accession numbers NC_016822) was also included in addition to the 3140 genomes from the French National Reference Center for E. coli, Shigella, and Salmonella (Supplementary Data 1). The circular phylogenetic tree (in blue) is rooted on S. flexneri 2a strain 2457T and its branch length has been shortened by a factor of 100 (indicated by the double slash) to improve vizualisation. S. sonnei lineages L1 to L4, clade 3.6, and subclade 3.6.1 are indicated. The rings show the associated information (see key) for each isolate, according to its position in the phylogeny, from the innermost to the outermost, in the following order: (1) selected S. sonnei genotypes; (2) antimicrobial susceptibility testing (AST) for ciprofloxacin; (3) AST for azithromycin; (4) AST for third-generation cephalosporins; (5) and the XDR isolates (with the XDR cluster names, X1 to X13, in red). The scale bar indicates the number of substitutions per variable site (SNVs). Due to a deliberate strategy to enrich our genomic dataset with azithromycin-resistant, ciprofloxacin-resistant, and third-generation cephalosporin-resistant isolates collected before 2017, the year in which routine genomic surveillance began in France (see “Methods”), there is an overrepresentation of these resistances in this figure with respect to the global population of 7121 isolates studied.
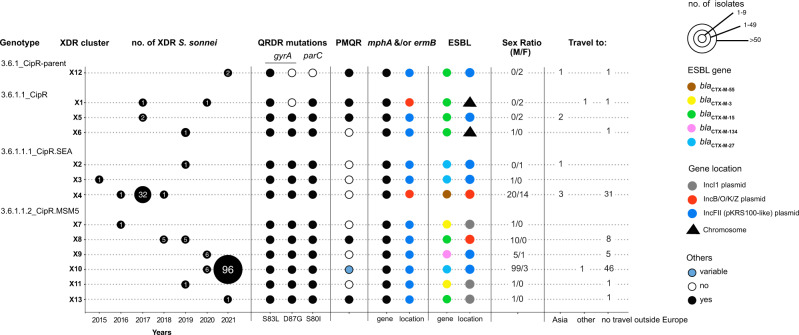
Using a new genomic tool based on a selection of core-genome SNVs developed by Hawkey et al. 15 for categorizing S. sonnei into informative phylogenetic genotypes with a universal nomenclature, we assigned the 164 XDR isolates to four different subtypes of subclade 3.6.1 (Fig. 2). The two clusters with the largest numbers of XDR isolates were X10 (n = 102) belonging to genotype 3.6.1.1.2_CipR.MSM5 (lineage L3, clade 6, subclade 1, subtype 1.2 with alias name CipR.MSM5, as used in previous publications for isolates resistant to CIP in GBMSM10,15), and X4 (n = 34) belonging to genotype 3.6.1.1.1_CipR.SEA (lineage L3, clade 6, subclade 1, subtype 1.1 with alias name CipR.SEA, as used in previous publications for isolates resistant to CIP in Southeast Asia6,15) (Fig. 3). Some of the XDR S. sonnei isolates belonging to genotypes 3.6.1_CipR-parent, 3.6.1.1_CipR and 3.6.1.1.1_CipR.SEA were acquired following travel (mostly to South and Southeast Asia) (Fig. 3). For cluster X4 (genotype 3.6.1.1.1_CipR.SEA), three patients reported travel to Southeast Asia, including one who was the index case of an outbreak at an elementary school (91 cases identified) in 2017, leading to the temporary closure of the school17. The widespread genotype 3.6.1.1.2_CipR.MSM5 was previously reported to be associated with GBMSM in the UK and Australia10,15. In our study, male patients were overrepresented (117 male and four female patients), particularly those aged 13 to 68 years (median age: 34 years; interquartile range: 28–42 years), among the cases caused by this genotype (Fig. 3). Furthermore, all but one of the cases infected with this genotype for whom travel information was available reported no travel outside Europe. Our findings indicate that the 164 XDR isolates were found in different transmission networks, with 31 cases in a school setting and eight cases in travelers returning from outside Europe. The male-to-female case ratio suggested that the majority of the other XDR cases may have been associated with GBMSM networks. However, this study was based on routine laboratory-surveillance data, with no record of the sexual orientation of the patient on the notification form accompanying the bacterial isolates sent to the FNRC-ESS (see “Methods”) and the patients were not interviewed to determine whether they were GBMSM.
Fig. 3. Main characteristics of the 164 XDR S. sonnei isolates.
The number of XDR isolates, categorized by genotype and XDR genomic cluster, is given by year of isolation. For each XDR genomic cluster, the mechanisms of resistance to CIP (mutation in the quinolone resistance-determining (QRDR) region of gyrA and parC, presence of a plasmid-mediated quinolone (PMQR) resistance gene), AZM (presence of mph(A) and/or erm(B) resistance genes) or 3GCs (presence of the extended-spectrum beta-lactamase (ESBL) blaCTX-M genes indicated in the key) are indicated. The chromosomal or plasmid location of the AZM and 3GC resistance genes is indicated. For plasmid-borne genes, the type of plasmid is also indicated. Finally, the sex ratio and information about travel (when known) are also indicated for the cases associated with each XDR genomic cluster. The two cases indicated as “Travel to other” are an individual residing in a French overseas territory in the Indian Ocean (cluster X1) and a traveler returning from Central Africa (X10).
Analysis of the AMR genes and structures in XDR S. sonnei isolates
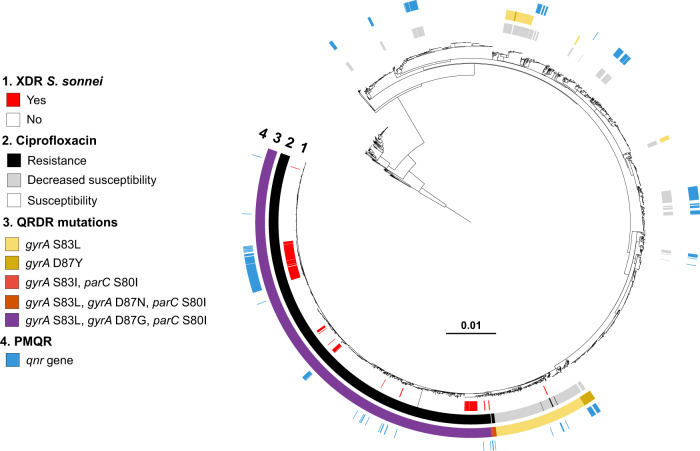
The combined phenotypic and genomic analyses revealed that all but four of the XDR S. sonnei isolates harbored three mutations in the quinolone resistance-determining regions (QRDR) of the gyrA and parC genes, conferring resistance to CIP (MIC ≥ 1 mg/L) (Figs. 2–4). The other four isolates—from the more ancestral genotypes, 3.6.1_CipR-parent and 3.6.1.1_CipR—harbored only one or two QRDR mutations, but also carried a plasmid-borne qnr gene, leading to CIP resistance. Resistance to AZM was conferred by the mphA and/or ermB genes, whereas resistance to 3GCs was conferred by various extended-spectrum beta-lactamase (ESBL) blaCTX-M genes (blaCTX-M-3, blaCTX-M-15, blaCTX-M-27, blaCTX-M-55, and blaCTX-M-134) (Figs. 2, 3, 5, and 6).
Fig. 4. Acquisition of genes encoding resistance to quinolones and fluoroquinolones in our genomic dataset.
Maximum-likelihood phylogeny of 3141 S. sonnei genomic sequences as shown in Fig. 2. The rings show the associated information (see key) for each isolate, according to its position in the phylogeny, from the innermost to the outermost, in the following order: (1) the XDR isolates; (2) antimicrobial susceptibility testing for ciprofloxacin (resistance defined as minimum inhibitory concentration [MIC] ≥ 1 mg/L; susceptibility as MIC ≤ 0.06 mg/L; decreased susceptibility as MIC between 0.12 and 0.5 mg/L); (3) mutations in the quinolone resistance-determining region (QRDR) of gyrA and parC; (4) presence of plasmid-mediated quinolone resistance (PMQR) genes of the qnr family.
Fig. 5. Acquisition of genes encoding resistance to azithromycin in our genomic dataset.
Maximum-likelihood phylogeny of 3141 S. sonnei genomic sequences as shown in Fig. 2. The rings show the associated information (see key) for each isolate, according to its position in the phylogeny, from the innermost to the outermost, in the following order: (1) the XDR isolates; (2) antimicrobial susceptibility testing for azithromycin (resistance defined as minimum inhibitory concentration [MIC] ≥ 32 mg/L; susceptibility as MIC ≤ 16 mg/L); (3) presence of the mph(A) gene; (4) presence of the erm(B) gene.
Fig. 6. Acquisition of selected genes encoding resistance to third-generation cephalosporins in our genomic dataset.
Maximum-likelihood phylogeny of 3141 S. sonnei genomic sequences as shown in Fig. 2. The rings show the associated information (see key) for each isolate, according to its position in the phylogeny, from the innermost to the outermost in the following order: (1) the XDR isolates; (2) antimicrobial susceptibility testing for third-generation cephalosporins (resistance defined as minimum inhibitory concentration [MIC] of ceftriaxone ≥ 4 mg/L; susceptibility as MIC ≤ 1 mg/L); (3) presence of bla genes encoding extended-spectrum beta-lactamases (ESBLs) or cephamycinases.
For better characterization of the mobile (or chromosomally integrated) elements encoding AMR, we also performed long-read sequencing on 16 XDR S. sonnei isolates (one to two isolates per XDR genomic cluster). The XDR S. sonnei isolates contained one to three plasmids carrying AMR genes (Supplementary Table 3). Plasmid sizes ranged from 8379 to 106,936 kb. A small ∼ 8 kb (either non-typable or typed as PTU-E63) plasmid encoding resistance to streptomycin (strA and strB genes), sulfonamides (sul2) and tetracyclines (tet(A)) was found in 12 of 16 XDR isolates from all four genotypes. This plasmid was highly similar to pMHMC-012 (GenBank accession no. CP053763) (Supplementary Fig. 1) from a S. sonnei isolate collected from a GBMSM patient in Boston, USA in 2017 (ref. 18).
Most of the other AMR plasmids belonged to IncFII (PTU-FE) and were related to pKSR100 (GenBank accession no. LN624486)—an epidemic MDR plasmid encoding resistance to AZM with a low fitness cost for its bacterial host — previously described in S. flexneri 3a, 2a, and S. sonnei sublineages associated with GBMSM networks from Europe, North America and Australasia10,15,19 (Fig. 3, Supplementary Table 3). A second, less common, plasmid encoding resistance to AZM was typed as IncB/O/K/Z; plasmids of this type were previously observed in XDR S. sonnei isolates in Southeast Asia6,7 (Fig. 3, Supplementary Table 3).
The ESBL blaCTX-M genes were located on multiple plasmids of different types (IncFII, including the pKSR100-like plasmid encoding resistance to AZM, IncB/O/K/Z, IncI1) or even integrated into the bacterial chromosome (Supplementary Table 3). Highly similar ESBL plasmids (98–99% nucleotide identity) were found in different XDR clusters (Supplementary Figs. 2–5). For example, an IncI1 (PTU-I1) plasmid carrying blaCTX-M-3 was identified in clusters X7 and X11 (same genotype, 3.6.1.1.2_CipR.MSM5) and an IncFII (PTU-FE) plasmid carrying blaCTX-M-27 was identified in clusters X2, X3 and X10 (two different genotypes, 3.6.1.1.1_CipR.SEA and 3.6.1.1.2_CipR.MSM5) (Fig. 3, Supplementary Table 2, and Supplementary Figs. 2 and 3). Some of our ESBL plasmids were also similar to certain plasmids described in previous studies. Hence, the blaCTX-M-27-carrying p202008564-6 plasmid (cluster X10, genotype 3.6.1.1.2_CipR.MSM5) displayed 99.6% nucleotide identity to p893916 (GenBank accession no. NZ_MW396858), from a S. sonnei isolate collected in London, UK, in 2020 (Supplementary Fig. 2)20. The second form of the ESBL plasmid found in cluster X10 isolates—and represented by p202008118-4—probably resulted from an insertion sequence (IS)-driven deletion or acquisition of the azithromycin-resistance gene, erm(B) (Supplementary Fig. 2). The blaCTX-M-134-carrying p202000562-4 plasmid (cluster X9, genotype 3.6.1.1.2_CipR.MSM5) displayed 90.3% nucleotide identity to p3123885 (GenBank accession no. CP049164), from a S. sonnei isolate acquired in Israel in 2019 (Supplementary Fig. 4)9. Finally, the blaCTX-M-3-carrying p201908234-4 plasmid (cluster X11, genotype 3.6.1.1.2_CipR.MSM5) displayed 99.7% nucleotide identity to p711-69 (GenBank accession no. CP049176), from a S. sonnei isolate acquired in Turkey in 2019 (Supplementary Fig. 3)9. In two S. sonnei isolates, 201701093 (cluster X1, genotype 3.6.1.1_CipR) and 201908033 (cluster X6, also 3.6.1.1_CipR), the blaCTX-M-15 ESBL gene was not located on a plasmid but on the bacterial chromosome (Fig. 3, Supplementary Table 3). In isolate 201908033, the ESBL gene was part of a ∼ 42 kb genomic island described in Supplementary Fig. 6, whereas in isolate 201701093, the ESBL gene was integrated into a ∼ 57 kb prophage sequence absent from other XDR genomes (Supplementary Fig. 7).
In the XDR genomic cluster X10, two isolates, 202110142 (accession no. ERR9940949) and 202111148 (ERR9941000) were phenotypically similar to other X10 isolates, except that they were susceptible to 3GCs (and were therefore not classified as XDR). However, they had the same antimicrobial drug resistance gene content (including blaCTX-M-27) as the other X10 XDR isolates. A careful inspection of the assemblies identified an IS (IS26) in both isolates, integrated at the same position of the blaCTX-M-27 promotor and probably leading to a non-functional ESBL gene. Together with the emergence of novel AMR genes, the presence of non-functional AMR genes with unaltered open reading frames (ORFs) constitutes a limitation of AMR prediction based exclusively on genomic data.
Our genomic analysis showed that this XDR phenotype emerged through a complex phenomenon of convergent evolution involving many AMR genes and structures. Studies of a larger number of XDR isolates by long-read sequencing might have revealed even greater diversity. Globally, the diversity of mobile and chromosomal AMR elements involved in XDR S. sonnei is undoubtedly greater. For example, blaCTX-M-14, an ESBL gene not encountered in our study, was identified in seven XDR S. sonnei isolates from Australia, UK, Vietnam and the US, between 2015 and 2019 (ref. 15).
One key element in the generation of the XDR phenotype was the acquisition of resistance to CIP, through fixed chromosomal mutations, in a successful sublineage (3.6.1) in South Asia at some time around 2007 (refs. 6,15). This sublineage subsequently and successively acquired resistance to AZM and 3GCs. These resistances were acquired independently, on multiple occasions (probably depending on the antibiotic selective pressure exerted in the different S. sonnei transmission networks and geographic areas), through the horizontal transfer of various plasmids and AMR genes. In the XDR S. sonnei isolates collected in France, IncFII pKSR100-like plasmids were frequently the vehicles of resistance to AZM, particularly in GBMSM-associated S. sonnei genotypes. Through their acquisition of an additional ESBL gene, these IncFII plasmids were also implicated in the resistance of most of our XDR isolates (clusters X2, X3, X5, X9, X10, and X12), as the vehicles of resistance to C3Gs, ultimately leading to the XDR phenotype. Interestingly, the virulence plasmid (pINV)—a ~210 kb low-copy number non-conjugative plasmid required for the invasion of host epithelial cells by S. sonnei—also belongs to the IncFII incompatibility group ([F27: A–: B–] replicon for pINV and [F35: A–: B–] replicon for pKSR100 according to pMLST 2.0 https://cge.food.dtu.dk/services/pMLST/)21–23. The coexistence of these two plasmids therefore suggests the existence of two types of selection pressure, mediated by type III secretion system (T3SS) expression24 and lipolysaccharide biosynthesis24 for the maintenance of pINV, and antibiotics for the maintenance of pKSR100.The great diversity of ESBL genes (blaCTX-M-3, blaCTX-M-15, blaCTX-M-27, blaCTX-M-55, and blaCTX-M-134) located on different plasmids (IncFII, IncI1, IncB/O/K/Z) or even integrated into the bacterial chromosome suggests that 3GC agents have recently exerted strong selective pressure on S. sonnei. For example, 3GCs are now recommended in place of AZM for the treatment of gonorrhea—a common sexually transmitted infection (STI) in GBMSM—in France and several other European countries25.
An emerging XDR S. sonnei strain
Our genomic analysis revealed that the 164 French XDR S. sonnei isolates were genetically diverse, but with one predominant cluster (X10) accounting for 62.2% (102/164) of XDR isolates. The genomic comparison of these 164 isolates collected in France with 67 previously published international XDR (or inferred to be XDR) S. sonnei isolates (Supplementary Table 4) revealed that 32.8% (22/67) of these international isolates were similar to the French XDR isolates from three clusters (X4, X9, and X10) (Supplementary Fig. 8). Eleven international isolates carrying the ESBL blaCTX-M-55 gene were identified in our X4 cluster. As expected for this cluster, which was associated with Southeast Asia, five isolates were collected in Vietnam in 2016 (01_0932, 01_0953, 01_0975, 01_1077, and 02_2142), three in Australia in 2016–2017 (AUSMDU00005736, AUSMDU00010002, and AUSMDU00006624; the two first being associated with travel to Southeast Asia), two were obtained in the UK in 2015–2016 (187596 and 261400, with no travel information); and one was obtained in the USA in 2017 (PNUSAE008493, with no travel information). Two international isolates carrying the ESBL blaCTX-M-134 gene were found to be similar to our X9 isolates. These two isolates were identified in 2019 in Switzerland (isolate 3123885, from a patient reporting travel to Israel) and in the UK (845732, with no travel information). Our largest XDR cluster, X10—belonging to genotype 3.6.1.1.2_CipR.MSM5 and containing the ESBL blaCTX-M-27 gene on a pKSR100-like plasmid—corresponds to the epidemic strain found in GBMSM in the UK and eight other European countries, with a total of 208 isolates obtained as of February 23, 2022 (ref. 5,26). At the time, France was the country with the largest number of isolates (n = 106) and the earliest isolate, dating from September 2020. All 73 XDR S. sonnei isolates obtained at the FNRC-ESS from January 1, 2022 to August 31, 2022 (a total of 336S. sonnei isolates were received over this time period) belong to genotype 3.6.1.1.2_CipR.MSM5 and contain the ESBL blaCTX-M-27 gene, suggesting that the epidemic X10 strain has continued its intensive circulation in France for a third year. This European epidemic strain was recently identified in the US and Australia25. A descriptive epidemiological study of the UK outbreak (72 cases) revealed that 21 of the 27 cases interviewed (78%) were HIV-negative GBMSM and users of HIV pre-exposure prophylaxis (PrEP)26. These individuals also reported having high-risk sex in England or Europe during the incubation period. Due to the few therapeutic options left, the recommended oral antimicrobial therapy, when required, was either pivmecillinam or fosfomycin (chloramphenicol not being readily accessible in the UK)26. For hospitalized or severe cases, carbapenems (colistin in case of allergy to beta-lactams) were used for three to five days, followed by an oral step-down treatment26. Further studies are required for a more detailed description of the clinical characteristics of these XDR S. sonnei infections (severity, existence of asymptomatic carriage, co-morbid conditions, including co-infections with other pathogens, such as monkeypox virus, etc.) and to confirm the efficacy of this antimicrobial treatment strategy. Post-exposure prophylaxis with doxycycline (Doxy-PEP) for the prevention of STIs in GBMSM is now the subject of heated debate27,28. A careful evaluation of the balance between the public health risks and benefits of this PEP will need to take the recent emergence of XDR S. sonnei into account, as high rates of resistance to tetracyclines (mediated by the two most common genes encoding tetracycline-specific efflux pumps, tet(A) and tet(B)) (Supplementary Tables 1–3) might provide these bacteria with a selective advantage, favoring their spread.
In conclusion, an effective national laboratory-based surveillance of Shigella infections, including antimicrobial susceptibility data, is therefore essential for informed decision-making and appropriate public health action to tackle the spread of these XDR S. sonnei strains currently circulating in different transmission networks. Genomic surveillance based on this new genotyping scheme with a common nomenclature will facilitate the detection and global tracking of these XDR S. sonnei strains of concern.
Methods
Ethics statement
This study was based exclusively on bacterial isolates and associated metadata collected under the mandate for laboratory-based surveillance awarded by the French Ministry of Health to the National Reference Center for Escherichia coli, Shigella and Salmonella (NRC-ESS). As a result, neither informed consent nor approval from an ethics committee was required. Data collection and storage by the NRC-ESS was approved by the French National Commission for Data Protection and Liberties (“Commission Nationale Informatique et Libertés (CNIL)”; approval number: 1474659).
Bacterial isolates
The French national surveillance program for Shigella infections is based on a voluntary laboratory-based network consisting of approximately 1000 clinical laboratories located in mainland France and its overseas territories in South America (French Guiana), the Caribbean (Martinique, Guadeloupe) and the Indian Ocean (La Réunion, Mayotte), which send about 1000–1200 Shigella spp. isolates to the French National Reference Center for E. coli, Shigella and Salmonella (FNRC-ESS) at Institut Pasteur, each year (only 600 in 2020, due to the COVID pandemic). The Shigella isolates are sent to the FNRC-ESS with a notification form that includes basic data: patient name, date of birth, sex, postcode, type of sample (stools or other), isolation date, clinical symptoms and their onset, illness or asymptomatic carriage (if illness, types of symptoms), information about international travel (if yes, date and country), sporadic or outbreak isolates (if clustered, hospital, school, household, nursery, etc.).
We studied all 7121 S. sonnei isolates (one per patient) received by the FNRC-ESS between 2005 and 2021, in the framework of the French national surveillance program for Shigella infections. It has been estimated that this surveillance system detects 50–60% of laboratory-confirmed Shigella infections in France29. The 7121 S. sonnei human isolates studied consisted of 6700 (94.1%) from mainland France and 421 (5.9%) from French overseas territories. From January 2005 to September 2021 (when phenotypic typing was definitively replaced by genomic surveillance), all these isolates were thoroughly characterized with a panel of biochemical tests and serotyped with slide agglutination assays according to standard protocols, as previously described30.
Antimicrobial drug susceptibility testing
Antimicrobial drug susceptibility testing was performed on all S. sonnei isolates, at the time of reception. Isolates were first tested with the disk diffusion (DD) method on Mueller-Hinton agar (Bio-Rad, Marnes-la-Coquette, France) according to the 2005–2015 guidelines of the antibiogram committee of the French Society for Microbiology (CA-SFM), in accordance with the recommendations of the European Committee on Antimicrobial Susceptibility Testing (EUCAST) (https://www.sfm-microbiologie.org/casfm/). The following disks (Bio-Rad, Marnes-La-Coquette, France) were used for the DD method: amoxicillin (AMX, 10 µg) or ampicillin (AMP, 10 µg), cefotaxime (CTX, 5 µg), ceftazidime (CAZ, 30 µg before 2015, 10 µg from 2015), ertapenem (ETP, 10 µg), chloramphenicol (CHL, 30 µg), sulfonamides (SMX, 200 µg), trimethoprim (TMP, 5 µg), trimethoprim-sulfamethoxazole (SXT, 1.25 µg/23.75 µg), streptomycin (STR, 10 µg), amikacin (AKN, 30 µg), gentamicin (GEN, 10 µg), tetracycline (TET, 30 µg), nalidixic acid (NAL, 30 µg), ofloxacin (OFX, 5 µg) or pefloxacin (PEF, 5 µg), ciprofloxacin (CIP, 5 µg), and azithromycin (AZM, 15 µg). Susceptibility to AZM was tested systematically only from April 2014. Resistance to 3GCs was defined as resistance to ceftazidime, cefotaxime, or ceftriaxone. For isolates resistant to NAL, CIP, AZM, or 3GCs, we confirmed the DD data by determining the minimum inhibitory concentrations (MICs) of these drugs with Etest strips (AB Biodisk, Solna, Sweden; bioMérieux, Marcy L’Etoile, France). The Clinical and Laboratory Standards Institute (CLSI) criteria were then used for the final interpretation31. As a means of distinguishing Shigella isolates susceptible to ciprofloxacin (minimum inhibitory concentration [MIC] ≤ 0.25 mg/L) that are wild-type (WT) from those that are non-WT, we defined two categories based on the epidemiological cutoffs used by the CLSI for Salmonella spp.: decreased susceptibility to ciprofloxacin (MIC between 0.12 and 0.5 mg/L) and true susceptibility to ciprofloxacin (MIC ≤ 0.06 mg/L)31.
Whole-genome sequencing
In total, 3109 S. sonnei isolates from the 7121 collected between 2005 and 2021 were sequenced, including all 164 XDR isolates (Supplementary Tables 1 and 2). Between 2017 (the year in which genomic surveillance began in France) and 2021, 2618 clinical isolates were received and sequenced at the FNRC-ESS. We included in this study the 2558 genomes (97.7%) that passed the EnteroBase quality control criteria (https://enterobase.warwick.ac.uk/species/index/ecoli). We also sequenced a selection of 551/4503 (12.2%) of the 2005–2016 isolates. This selection contained, in particular, 96.7% (89/92) of all isolates resistant to 3GCs, 56.3% (294/522) of all isolates resistant to CIP, and 96.7% (116/120) of the isolates resistant to AZM detected from April 2014 to December 2016.
Total DNA was extracted with the MagNA Pure DNA isolation kit (Roche Molecular Systems, Indianapolis, IN, USA), in accordance with the manufacturer’s recommendations. Whole-genome sequencing was performed as part of routine procedures at the FNRC-ESS, and at the Mutualized Platform for Microbiology (P2M) at Institut Pasteur, Paris. The libraries were prepared with the Nextera XT kit (Illumina, San Diego, CA, USA) and sequencing was performed with the NextSeq 500 system (Illumina) generating 150 bp paired-end reads. All reads were filtered with FqCleanER version 21.06 (https://gitlab.pasteur.fr/GIPhy/fqCleanER) with options -q 15 -l 50 to eliminate adaptor sequences and discard low-quality reads with phred scores below 15 and a length of less than 50 bp32.
Assemblies were generated with SPAdes version 3.9.0 (ref. 33) through EnteroBase.
Genotyping
All the genomes studied were genotyped with the hierarchical SNV-based genotyping scheme for S. sonnei described by Hawkey et al.15 and implemented in Mykrobe software version 0.9.0 (https://github.com/katholt/sonneityping)34.
Phylogenomic analysis
We included in the phylogenomic analysis 3140 S. sonnei genomic sequences from isolates and historical strains of the FNRC-ESS (Supplementary Data 1) originating from 3109 S. sonnei isolates collected between 2005 and 2021 and 31 isolated before 2005, to enrich the dataset with rare lineages of S. sonnei (L1, L2, and L4)6,8,10,16.
The paired-end reads were mapped onto the reference genome of S. sonnei 53G (GenBank accession numbers NC_016822)16 with Snippy version 4.6.0/BWA-MEM version 0.7.17 (https://github.com/tseemann/snippy). SNVs were called with Snippy version 4.6.0/Freebayes version 1.3.2 (https://github.com/tseemann/snippy) under the following constraints: mapping quality of 60, a minimum base quality of 13, a minimum read coverage of 4, and a 75% read concordance at a locus for a variant to be reported. An alignment of core genome SNVs was produced in Snippy version 4.6.0 for phylogenetic inference.
Repetitive regions (i.e., insertion sequences, tRNAs) in the alignment were masked (10.26180/5f1a443b19b2f)15. Putative recombinogenic regions were detected and masked with Gubbins version 3.2.0 (ref. 35) (default settings, except -f 32). A maximum likelihood (ML) phylogenetic tree was built from an alignment of 59,295 chromosomal SNVs, with RAxML version 8.2.12, under the GTR model, with 200 bootstrap values36. The final tree was rooted on the S. flexneri 2a strain 2457T genome (GenBank accession no. AE014073) and visualized with iTOL version 6 (https://itol.embl.de)37.
We also used the same phylogenetic approach to compare our 164 XDR S. sonnei isolates with 69 previously published XDR (or inferred to be XDR) S. sonnei isolates (Supplementary Data 1), but with Gubbins used with default settings (in particular -f 25). The final tree, based on 2298 chromosomal SNVs, was rooted on the reference S. sonnei genome 53G (genotype 2.8.2). Two of these international isolates (Vietnamese isolates 02_1181 and 01_1008) were excluded from the analysis due to a proportion of “N” > 25%.
Resistance gene analysis
The presence and type of acquired antibiotic resistance genes (ARGs) were determined with ResFinder version 4.0.1 (https://cge.cbs.dtu.dk/services/ResFinder/)38, Sonneityping/Mykrobe version 0.9.0 (https://github.com/katholt/sonneityping)15,34 on SPAdes assemblies. The presence of mutations in genes encoding resistance to quinolones (gyrA, parC) was also investigated by analyzing the sequences assembled de novo with BLAST version 2.2.26.
Plasmid sequencing
Sixteen XDR S. sonnei isolates (one to two per XDR cluster) were selected (Supplementary Table 3) and sequenced with a Nanopore MinION sequencer (Oxford Nanopore Technologies). Genomic DNA was prepared as follows: the isolates were cultured overnight at 37 °C in alkaline nutrient agar (20 g casein meat peptone E2 from Organotechnie; 5 g sodium chloride from Sigma; 15 g Bacto agar from Difco; distilled water to 1 L; adjusted to pH 8.4; autoclaved at 121 °C for 15 min). A few isolated colonies from the overnight culture were used to inoculate 20 mL of brain-heart infusion (BHI) broth, and were cultured until a final OD600 of 0.8 was reached at 37 °C with shaking (200 rpm—Thermo Fisher Scientific MaxQ 6800). The bacterial cells were harvested by centrifugation and DNA was extracted with one of the two following methods. The first method corresponded to the protocol described by von Mentzer et al.39, except that MaXtract High-Density columns (Qiagen) were used (instead of phase-lock tubes) and the DNA was resuspended in molecular biology-grade water (instead of 10 mM Tris pH 8.0). In the second method, we used Genomic-tip 100/G columns (Qiagen) according to the manufacturer’s protocol. The library was prepared according to the instructions of the “Native barcoding genomic DNA (with EXP-NBD104, EXP-NBD114, and SQK-LSK109)” procedure provided by Oxford Nanopore Technology. Sequencing was then performed on a MinION Mk1C apparatus (Oxford Nanopore Technologies). The genomic sequences of the isolates were assembled from long and short reads, with a hybrid approach and UniCycler version 0.4.8 (ref. 40). A polishing step was performed with Pilon version 1.23 (ref. 41) to generate a high-quality sequence composed of chromosomal and plasmid sequences. The plasmids were then annotated with Prokka version 1.14.5 (https://github.com/tseemann/prokka)42 and corrected manually. Plasmids were aligned and visualized with BRIG version 0.95 (http://sourceforge.net/projects/brig)43.
Plasmid typing
The plasmids were typed with PlasmidFinder version 2.1.1. (https://cge.cbs.dtu.dk/services/PlasmidFinder/)44, pMLST version 1.2 (https://cge.cbs.dtu.dk/services/pMLST/)44, and COPLA version 1.0 (https://castillo.dicom.unican.es/copla/)45 on SPAdes assemblies.
Statistical analysis
Chi-squared tests for trends were used to analyze the proportion of bacterial strains resistant to antimicrobial drugs by year.
Data collection
The data were entered into an Excel (Microsoft) version 15.41 spreadsheet.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Supplementary information
Description of Additional Supplementary Files
Acknowledgements
This study was supported by the Institut Pasteur (F.-X.W. and A.F.), Santé publique France (F.-X.W.), and the French government’s Investissement d’Avenir program, Laboratoire d’Excellence ‘Integrative Biology of Emerging Infectious Diseases’ (grant number ANR-10-LABX-62-IBEID) (F.-X.W.). We thank C. Parsot for his expert advice on the virulence plasmid of Shigella spp. We also thank all the corresponding laboratories of the French National Reference Center for Escherichia coli, Shigella, and Salmonella. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author contributions
S.L. and F.-X.W. conceptualized and designed the study. S.L., E.N., I.Y., and F.-X.W. did the genomic analyses. S.L., E.N., I.Y., L.F., and F.-X.W. contributed to data interpretation and visualization. C.R., I.C., M.L.C., and E.N. performed the laboratory experiments. S.L. and S.F. were involved in sample collection and metadata curation. S. F. and A.F. performed statistical analyses. F.-X.W. supervised the project. F.-X.W. and A.F. were responsible for funding acquisition. F.-X.W. drafted the article. S.L., E.N., S.F., L.F., I.Y., L.F., M.P.G., and A.F. critically reviewed the draft. All the authors read and approved the final manuscript. S.L., E.N., and F.-X.W. accessed and verified the underlying data.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Data availability
The publicly available sequences used in this study are available in GenBank under accession numbers CP053763, NC_016822, AE014073, NZ_MW396858, LN624486, CP049176, CP049164, CP049174, CP049186, CP053751. Short-read sequence data generated in this study were submitted to EnteroBase (https://enterobase.warwick.ac.uk/) and to the European Nucleotide Archive (ENA, https://www.ebi.ac.uk/ena/) under study number PRJEB44801. Whole-genome assemblies have been deposited in FigShare (10.6084/m9.figshare.21594033.v1). All the accession numbers of the short-read sequences produced and used in this study are listed in Supplementary Table 2 and Supplementary Data 1. The plasmid sequences obtained were deposited in GenBank (https://www.ncbi.nlm.nih.gov/genbank/) under accession numbers OP038267-OP038301 and OP038303 (Supplementary Table 3).
Code availability
No custom computer code or custom algorithm was used in this study.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
The online version contains supplementary material available at 10.1038/s41467-023-36222-8.
References
- 1.World Health Organization. Guidelines for the control of shigellosis, including epidemics due to Shigella dysenteriae type 1; https://apps.who.int/iris/handle/10665/43252 (2005).
- 2.World Health Organization. WHO Model Lists of Essential Medicines; http://www.who.int/medicines/publications/essentialmedicines/en/ (2021).
- 3.Centers for Disease Control and Prevention. CDC recommendations for diagnosing and managing Shigella strains with possible reduced susceptibility to ciprofloxacin; https://emergency.cdc.gov/han/han00401.asp (2017).
- 4.Thompson CN, Duy PT, Baker S. The rising dominance of Shigella sonnei: an intercontinental shift in the etiology of Bacillary dysentery. PLoS Negl. Trop. Dis. 2015;9:e0003708. doi: 10.1371/journal.pntd.0003708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.European Centre for Disease Prevention and Control. Increase in extensively-drug resistant Shigella sonnei infections in men who have sex with men in the EU/EEA and the UK; https://www.ecdc.europa.eu/en/publications-data/rapid-risk-assessment-increase-extensively-drug-resistant-shigella-sonnei (2022).
- 6.Chung The H, et al. Dissecting the molecular evolution of fluoroquinolone-resistant Shigella sonnei. Nat. Commun. 2019;10:4828. doi: 10.1038/s41467-019-12823-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Thanh Duy P, et al. Commensal Escherichia coli are a reservoir for the transfer of XDR plasmids into epidemic fluoroquinolone-resistant Shigella sonnei. Nat. Microbiol. 2020;5:256–264. doi: 10.1038/s41564-019-0645-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Baker KS, et al. Travel- and community-based transmission of multidrug-resistant Shigella sonnei lineage among international Orthodox Jewish communities. Emerg. Infect. Dis. 2016;22:1545–1553. doi: 10.3201/eid2209.151953. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Campos-Madueno EI, et al. Rapid increase of CTX-M-producing Shigella sonnei isolates in Switzerland due to spread of common plasmids and international clones. Antimicrob. Agents Chemother. 2020;64:e01057–20. doi: 10.1128/AAC.01057-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Baker KS, et al. Horizontal antimicrobial resistance transfer drives epidemics of multiple Shigella species. Nat. Commun. 2018;9:1462. doi: 10.1038/s41467-018-03949-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Ingle DJ, et al. Co-circulation of multidrug-resistant Shigella among men who have sex with men in Australia. Clin. Infect. Dis. 2019;69:1535–1544. doi: 10.1093/cid/ciz005. [DOI] [PubMed] [Google Scholar]
- 12.Bardsley M, et al. Persistent transmission of Shigellosis in England is associated with a recently emerged multidrug-resistant strain of Shigella sonnei. J. Clin. Microbiol. 2020;58:e01692–19. doi: 10.1128/JCM.01692-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Gaufin T, et al. Antimicrobial-resistant Shigella spp. in San Diego, California, USA, 2017–2020. Emerg. Infect. Dis. 2022;28:1110–1116. doi: 10.3201/eid2806.220131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Williamson D, Ingle D, Howden B. Extensively drug-resistant Shigellosis in Australia among men who have sex with men. N. Engl. J. Med. 2019;381:2477–2479. doi: 10.1056/NEJMc1910648. [DOI] [PubMed] [Google Scholar]
- 15.Hawkey J, et al. Global population structure and genotyping framework for genomic surveillance of the major dysentery pathogen, Shigella sonnei. Nat. Commun. 2021;12:2684. doi: 10.1038/s41467-021-22700-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Holt KE, et al. Shigella sonnei genome sequencing and phylogenetic analysis indicate recent global dissemination from Europe. Nat. Genet. 2012;44:1056–1059. doi: 10.1038/ng.2369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Bernadou, A. et al. First outbreak in France of multi-drug-resistant Shigella sonnei infections in primary school, Southwestern France, March–May 2017. European Scientific Conference on Applied Infectious Disease Epidemiology p. 68 (2017). https://www.escaide.eu/sites/default/files/documents/ESCAIDE_2017%20abstract%20book_final_03.pdf.
- 18.Worley JN, et al. Genomic drivers of multidrug-resistant Shigella affecting vulnerable patient populations in the United States and abroad. mBio. 2021;12:e03188–20. doi: 10.1128/mBio.03188-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Baker KS, et al. Intercontinental dissemination of azithromycin-resistant shigellosis through sexual transmission: a cross-sectional study. Lancet Infect. Dis. 2015;15:913–921. doi: 10.1016/S1473-3099(15)00002-X. [DOI] [PubMed] [Google Scholar]
- 20.Locke RK, Greig DR, Jenkins C, Dallman TJ, Cowley LA. Acquisition and loss of CTX-M plasmids in Shigella species associated with MSM transmission in the UK. Microb. Genom. 2021;7:000644. doi: 10.1099/mgen.0.000644. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Sansonetti PJ, Kopecko DJ, Formal SB. Shigella sonnei plasmids: evidence that a large plasmid is necessary for virulence. Infect. Immun. 1981;34:75–83. doi: 10.1128/iai.34.1.75-83.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Jiang Y, et al. The complete sequence and analysis of the large virulence plasmid pSS of Shigella sonnei. Plasmid. 2005;54:149–159. doi: 10.1016/j.plasmid.2005.03.002. [DOI] [PubMed] [Google Scholar]
- 23.Makino S, Sasakawa C, Yoshikawa M. Genetic relatedness of the basic replicon of the virulence plasmid in shigellae and enteroinvasive Escherichia coli. Micro. Pathog. 1988;5:267–274. doi: 10.1016/0882-4010(88)90099-X. [DOI] [PubMed] [Google Scholar]
- 24.Marteyn B, Gazi A, Sansonetti P. Shigella: a model of virulence regulation in vivo. Gut Microbes. 2012;3:104–120. doi: 10.4161/gmic.19325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Mason, L. et al. The evolution and international spread of extensively drug resistant Shigella sonnei; PREPRINT (Version 1) available at Research Square. 10.21203/rs.3.rs-2057516/v1 (2022).
- 26.Charles H, et al. Outbreak of sexually transmitted, extensively drug-resistant Shigella sonnei in the UK, 2021-22: a descriptive epidemiological study. Lancet Infect. Dis. 2022;22:1503–1510. doi: 10.1016/S1473-3099(22)00370-X. [DOI] [PubMed] [Google Scholar]
- 27.Grant JS, et al. Doxycycline prophylaxis for bacterial sexually transmitted infections. Clin. Infect. Dis. 2020;3:1247–1253. doi: 10.1093/cid/ciz866. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Centers for Disease Control and Prevention. CDC Response to Doxy-PEP data presented at 2022 International AIDS Conference; https://www.cdc.gov/nchhstp/newsroom/2022/Doxy-PEP-clinical-data-presented-at-2022-AIDS-Conference.html (2022).
- 29.Yassine I, et al. Population structure analysis and laboratory monitoring of Shigella by core-genome multilocus sequence typing. Nat. Commun. 2022;13:551. doi: 10.1038/s41467-022-28121-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Langendorf C, et al. Enteric bacterial pathogens in children with diarrhea in Niger: diversity and antimicrobial resistance. PLoS One. 2015;1:e0120275. doi: 10.1371/journal.pone.0120275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.CLSI. Performance Standards for Antimicrobial Susceptibility Testing. 30th edn. Supplement M100 (Clinical and Laboratory Standards Institute, 2020).
- 32.Criscuolo A, Brisse S. AlienTrimmer: a tool to quickly and accurately trim off multiple short contaminant sequences from high-throughput sequencing reads. Genomics. 2013;102:500–506. doi: 10.1016/j.ygeno.2013.07.011. [DOI] [PubMed] [Google Scholar]
- 33.Bankevich A, et al. SPAdes: a new genome assembly algorithm and its applications to single-cell sequencing. J. Comput. Biol. 2012;19:455–477. doi: 10.1089/cmb.2012.0021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Hunt M, et al. Antibiotic resistance prediction for Mycobacterium tuberculosis from genome sequence data with Mykrobe. Wellcome Open Res. 2019;4:191. doi: 10.12688/wellcomeopenres.15603.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Croucher NJ, et al. Rapid phylogenetic analysis of large samples of recombinant bacterial whole genome sequences using Gubbins. Nucleic Acids Res. 2015;43:e15. doi: 10.1093/nar/gku1196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Kozlov AM, Darriba D, Flouri T, Morel B, Stamatakis A. RAxML-NG: a fast, scalable and user-friendly tool for maximum likelihood phylogenetic inference. Bioinformatics. 2019;35:4453–4455. doi: 10.1093/bioinformatics/btz305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Letunic I, Bork P. Interactive Tree Of Life (iTOL) v5: an online tool for phylogenetic tree display and annotation. Nucleic Acids Res. 2021;49:W293–W296. doi: 10.1093/nar/gkab301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Zankari E, et al. Identification of acquired antimicrobial resistance genes. J. Antimicrob. Chemother. 2012;67:2640–2644. doi: 10.1093/jac/dks261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Von Mentzer A, et al. Long-read-sequenced reference genomes of the seven major lineages of enterotoxigenic Escherichia coli (ETEC) circulating in modern time. Sci. Rep. 2021;11:9256. doi: 10.1038/s41598-021-88316-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Wick RR, Judd LM, Gorrie CL, Holt KE. Unicycler: Resolving bacterial genome assemblies from short and long sequencing reads. PLoS Comput. Biol. 2017;13:e1005595. doi: 10.1371/journal.pcbi.1005595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Walker BJ, et al. Pilon: an integrated tool for comprehensive microbial variant detection and genome assembly improvement. PLoS One. 2014;9:e112963. doi: 10.1371/journal.pone.0112963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Seemann T. Prokka: rapid prokaryotic genome annotation. Bioinformatics. 2014;30:2068–2069. doi: 10.1093/bioinformatics/btu153. [DOI] [PubMed] [Google Scholar]
- 43.Alikhan NF, Petty NK, Ben Zakour NL, Beatson SA. BLAST Ring Image Generator (BRIG): simple prokaryote genome comparisons. BMC Genomics. 2011;12:402. doi: 10.1186/1471-2164-12-402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Carattoli A, et al. In silico detection and typing of plasmids using PlasmidFinder and Plasmid Multilocus Sequence Typing. Antimicrob. Agents Chemother. 2014;58:3895–3903. doi: 10.1128/AAC.02412-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Redondo-Salvo S, et al. COPLA, a taxonomic classifier of plasmids. BMC Bioinforma. 2021;22:390. doi: 10.1186/s12859-021-04299-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Description of Additional Supplementary Files
Data Availability Statement
The publicly available sequences used in this study are available in GenBank under accession numbers CP053763, NC_016822, AE014073, NZ_MW396858, LN624486, CP049176, CP049164, CP049174, CP049186, CP053751. Short-read sequence data generated in this study were submitted to EnteroBase (https://enterobase.warwick.ac.uk/) and to the European Nucleotide Archive (ENA, https://www.ebi.ac.uk/ena/) under study number PRJEB44801. Whole-genome assemblies have been deposited in FigShare (10.6084/m9.figshare.21594033.v1). All the accession numbers of the short-read sequences produced and used in this study are listed in Supplementary Table 2 and Supplementary Data 1. The plasmid sequences obtained were deposited in GenBank (https://www.ncbi.nlm.nih.gov/genbank/) under accession numbers OP038267-OP038301 and OP038303 (Supplementary Table 3).
No custom computer code or custom algorithm was used in this study.