Abstract
An 88-year-old male patient on maintenance hemodialysis (HD) therapy experienced gradual losses in appetite and liveliness during the course of 1 month. Physical examinations revealed no abnormalities. However, blood testing indicated non-thyroidal illness syndrome (NTIS) typically observed in patients with severe illness, with serum levels of thyroid stimulating hormone, free triiodothyronine, and free thyroxine of 0.17 μIU/mL, < 1.0 pg/mL, and 0.23 ng/dL, respectively. Brain magnetic resonance imaging to exclude the possibility of central hypothyroidism unexpectedly displayed slight abnormalities inside of the thalami that were characteristic of Wernicke’s encephalopathy. Additional examination disclosed low serum thiamine of 20 ng/mL. Thiamine injections of 100 mg at every HD treatment rapidly restored his appetite, liveliness, and NTIS findings. HD patients are at a particularly high risk of thiamine deficiency (TD) and associated severe symptoms due to losses of thiamine during HD sessions. However, its non-specific initial symptoms, including decreases in appetite and liveliness, as well as undetectability in routine blood tests complicate early detection, resulting in underdiagnosis and more severe outcomes. In the present case, TD manifested only as non-specific symptoms and was ultimately revealed by the presence of NTIS, which was resolved with thiamine supplementation. Thus, NTIS might assist in the early detection of TD as an initial sign in HD patients.
Keywords: End-stage renal disease, Hemodialysis, Non-thyroidal illness syndrome, Thiamine deficiency
Introduction
Thiamine is one of the most important vitamins for regulating basic metabolism [1, 2]. However, hemodialysis (HD) patients exhibit an elevated risk of thiamine deficiency (TD) due to thiamine depletion during HD sessions, appetite loss, and impaired gastrointestinal absorption [3–5]. Prolonged TD leads to severe life-threatening disorders, including Wernicke’s encephalopathy (WE), Wernicke–Korsakoff syndrome, and Beriberi [6]. The prevention and early detection of TD are, therefore, important. Unfortunately, TD and its ensuing life-threatening symptoms are frequently overlooked owing to their inconsistent clinical presentation and undetectability in routine blood tests [7–9]. Furthermore, TD initially presents as non-specific symptoms, such as reduced appetite and liveliness [10], which occur in roughly one-third of HD recipients [11, 12]. A novel approach for detecting early TD in HD patients is strongly needed.
In cases of severe illness, characteristic alterations in serum thyroidal markers, typically a drop in serum triiodothyronine (T3) without concomitant elevation of serum thyroid stimulating hormone (TSH), are widely observed despite the lack of thyroid disease. These thyroidal marker changes are collectively termed non-thyroidal illness syndrome (NTIS), and a clear correlation of NTIS with poor prognosis has been reported for a variety of diseases [13, 14].
We herein describe the clinical outcome of a HD patient with NTIS, which facilitated the early detection of TD presenting only as appetite loss and diminished liveliness.
Case report
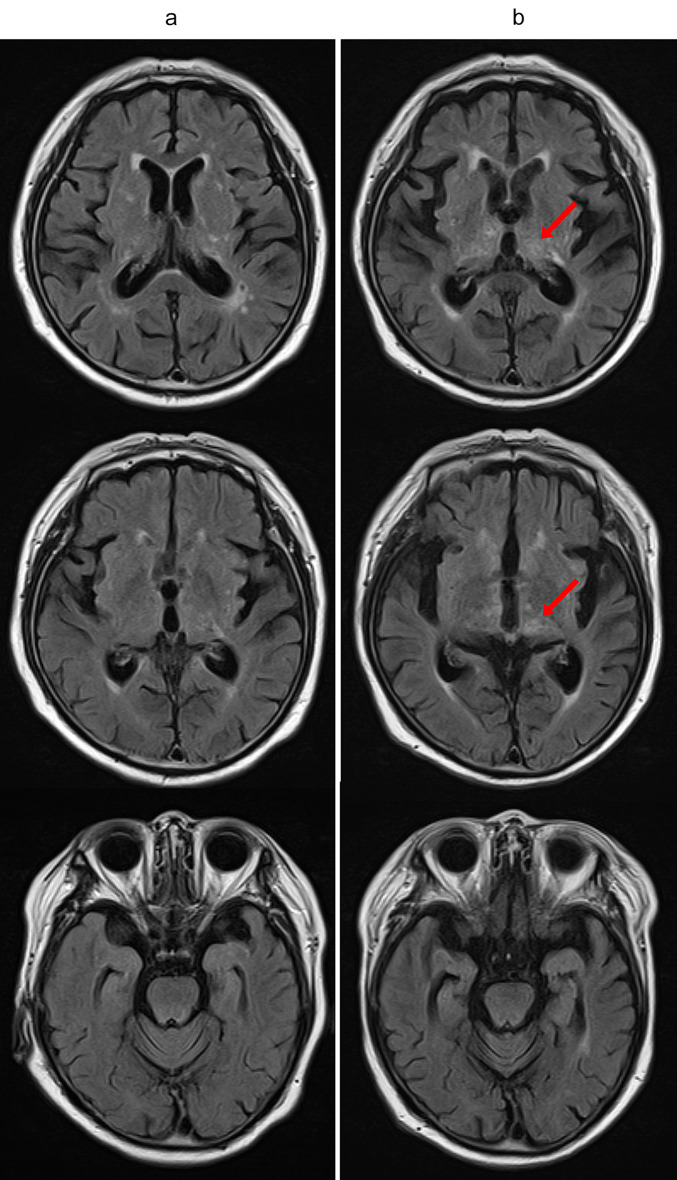
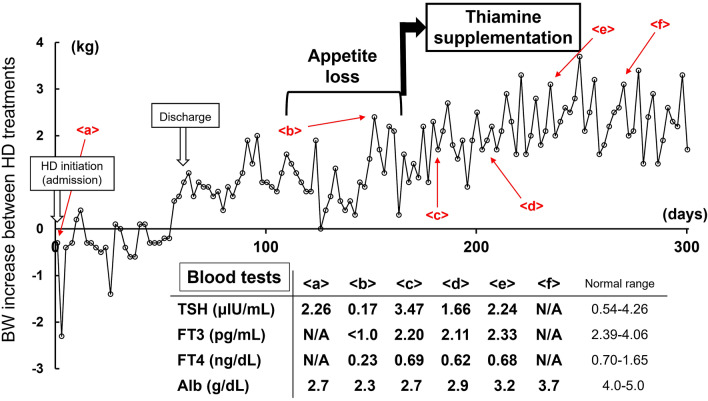
An 88-year-old male patient had been receiving maintenance HD therapy for end-stage kidney disease due to diabetic kidney disease. Four months after commencing HD, he complained of gradual losses in appetite and liveliness. Physical examinations, routine blood tests, and whole-body computed tomography (CT) revealed no obvious abnormalities. Additional testing was performed after his symptoms persisted for over 1 month. Although he was visibly less lively, he was fully conscious and alert. He was served balanced meals by his family and did not drink, smoke, or use any illegal drugs. He was afebrile. His blood pressure and heart rate were approximately 130/70 mmHg and 80 beats/min, respectively. His height was 154 cm and dry body weight was 48.7 kg. An arterio-venous fistula had been placed in his left forearm. He had no leg edema, and his kneecap reflex was normal. Other physical examinations were normal. Maintenance HD therapy was performed 3 times/week for 3 h at the conditions of 200 mL/min blood flow, 500 mL/min dialysate flow, 1.6 mm2 poly-methyl methacrylate membrane (replacing a 1.4 mm2 polysulfone membrane at 3 weeks after HD induction), and 1.2 Kt/V. He was taking 100 mg of aspirin for an old cerebral infarction in addition to 40 mg of nifedipine, 2 mg of pitavastatin, 60 mg of fexofenadine, 15 g of d-sorbitol, and 5 g of Daikenchuto. Epoetin alpha injections of 750 IU were given at every HD treatment for nephrogenic anemia. The results of his early week pre-HD blood sample are presented in Table 1. Serum thyroidal hormone levels assessed due to his diminished liveliness revealed decreased serum TSH of 0.17 μIU/mL, which had been within normal range (2.26 μIU/mL) at the initiation of maintenance HD, as well as extremely low serum free T3 (FT3) and free thyroxine (FT4) of < 1.0 pg/mL and 0.23 ng/dL, respectively. Thyroglobulin antibody, thyroid peroxidase antibody, and thyroid stimulating antibody were negative, and neck ultrasound tomography revealed no abnormal findings in the thyroid gland. Additional examinations were performed under the suspicion of central hypothyroidism or NTIS. His serum levels of cortisol and adrenocorticotropic hormone were within normal range, and abdominal CT did not indicate an adrenal tumor. Brain magnetic resonance imaging (MRI) also disclosed no abnormal findings around the pituitary gland, instead showing a slight abnormal signal inside the thalami (Fig. 1b) that had been absent before (Fig. 1a), suggesting possible WE. The injection of 100 mg fursultiamine at every HD session was started 1 week after the MRI examination. The treatment rapidly improved his appetite and liveliness, with serum thyroid hormone levels being restored by the 14th day of therapy (Fig. 2). His serum thiamine level immediately before the initiation of thiamine treatment was later found to be below normal range at 20 ng/mL and was subsequently improved by the intervention. Based on this clinical course, he was diagnosed as having TD exhibiting NTIS and a loss of appetite and liveliness. At 2 weeks of treatment, the supplementation was changed to oral fursultiamine of 50 mg/day. Aling with an improvement in appetite, weight gain between HD sessions was increased without any self-described decreases in urine frequency or output (Fig. 2). His serum albumin level ultimately increased to 3.7 g/dL, and scores for the Nutritional Risk Index for Japanese Hemodialysis Patients [15] and Geriatric Nutritional Risk Index [16] were improved from 9 (medium risk) to 5 (low risk) and from 73 (high risk) to 95 (low risk), respectively. No further relapses of TD or NTIS were recorded.
Table 1.
Blood test results
| Blood analysis | Normal range | |
|---|---|---|
| Complete blood count | ||
| White blood cells (/mm2) | 6200 | 4500–9000 |
| Hemoglobin (g/dL) | 10.6 | 14.0–18.0 |
| Platelets (× 104/mm2) | 19.1 | 15.8–34.8 |
| Serum chemistry | ||
| Total protein (g/dL) | 6.0 | 6.5–8.0 |
| Albumin (g/dL) | 2.3 | 4.0–5.0 |
| Blood urea nitrogen (mg/dL) | 34.7 | 8.0–21.0 |
| Creatinine (mg/dL) | 3.60 | 0.63–1.05 |
| Uric acid (mg/dL) | 4.5 | < 7.0 |
| Total cholesterol (mg/dL) | 91 | 130–220 |
| Aspartate aminotransferase (IU/L) | 11 | 11–28 |
| Alanine aminotransferase (IU/L) | 5 | 6–30 |
| Lactic dehydrogenase (IU/L) | 174 | 124–222 |
| Sodium (mEq/L) | 137 | 135–147 |
| Potassium (mEq/L) | 3.4 | 3.5–5.1 |
| Chloride (mEq/L) | 106 | 99–108 |
| Calcium (mg/dL) | 7.8 | 8.7–10.3 |
| Phosphate (mg/dL) | 2.7 | 2.5–4.6 |
| Intact parathyroid hormone (pg/mL) | 27 | 35–150 |
| Blood glucose (mg/dL) | 109 | 75–109 |
| β2-Microglobulin (μg/L) | 13.7 | 1.0–1.9 |
| Glycated albumin (%) | 21.8 | 12.4–16.3 |
| C-reactive protein (mg/dL) | 1.38 | < 0.14 |
| Thyroid stimulating hormone (μIU/mL) | 0.17 | 0.54–4.26 |
| Free triiodothyronine (pg/mL) | < 1.0 | 2.39–4.06 |
| Free thyroxine (ng/dL) | 0.23 | 0.70–1.65 |
| Thyroglobulin antibody (IU/mL) | 12 | < 40.0 |
| Thyroid peroxidase antibody (IU/mL) | 9 | < 28.0 |
| Thyroid stimulating antibody (%) | 105 | < 120 |
| Cortisol (μg/dL) | 7.1 | 4.5–21.1 |
| Adrenocorticotropic hormone (pg/mL) | 14.9 | 7.2–63.3 |
| Thiamine (ng/mL) | 20 | 21.3–81.9 |
Fig. 1.
Brain magnetic resonance imaging findings. Head magnetic resonance imaging with T2-weighted fluid-attenuated inversion recovery sequences (a) 6 years before and (b) at the diagnosis of thiamine deficiency. Arrows indicate the slightly high intensity areas inside the thalami, implying possible Wernicke’s encephalopathy
Fig. 2.
Clinical time course of the present case. Time course of body weight changes between hemodialysis treatments from the initiation of maintenance hemodialysis. Letters and inset Table show the alterations in serum thyroidal hormone and albumin levels. Alb albumin, BW body weight, FT3 free triiodothyronine, FT4 free thyroxine, HD hemodialysis, N/A not assessed, TSH thyroid stimulating hormone
Discussion
Typically observed as a drop in serum T3 level without concomitant elevation of serum TSH, NTIS can be found in patients with any type of severe illness [13]. The exact pathophysiological mechanism of this syndrome is not yet fully elucidated, and whether the thyroidal marker changes are adaptive physiological responses or a consequence of the metabolic alterations during stressful circumstances remains a matter of debate [17]. The presence of low serum T3/FT3 or NTIS is widely reported as a strong predictor of poor prognosis, with the recovery of which representing a marker of escape from severe illness and outcomes [18–20]. In the present case, the development of NTIS and subsequent recovery with thiamine supplementation demonstrated the severity of his condition from TD and its rescue with appropriate treatment. Furthermore, he displayed decreases in serum FT4 and TSH levels, which have been noted in severe NTIS and related to a worsened prognosis [21, 22]. Thus, although his apparent symptoms had not appeared severe, our findings implied that he had been in worse condition than expected.
As a water-soluble vitamin, over 40% of serum thiamine is removed by HD treatment [23]. Furthermore, mammals cannot synthesize thiamine and rely on its dietary intake. In the present case, the significant elimination of thiamine by HD treatment along with reduced thiamine intake from appetite loss presumably contributed to the development of TD. However, he had been receiving mild HD treatments of 1.2 Kt/V, and was not under severe starvation conditions given that his body weight change between HD sessions had never been negative (Fig. 2). These findings implicate the presence of additional factors in his TD development, and we presumed that a vicious cycle of appetite loss causing TD, which further exacerbated appetite loss, might have influenced disease formation. Although the initial cause of his appetite loss was unclear, the vicious cycle could have spontaneously occurred without a specific trigger, since HD patients often suffer from decreased appetite without any obvious event [12]. Such hypotheses based on his clinical course emphasize the necessity of vigilance for TD in HD patients.
To the best of our knowledge, no studies have addressed the precise relationship between TD and NTIS, and the exact mechanism of how TD causes NTIS is unknown. However, TD-based appetite loss likely plays a key role considering that starvation is a major risk factor for NTIS development [24]. The fact that this symptom continued for 1 month also supported the notion that decreased calorie intake with TD caused NTIS. However, previous research on NTIS with reduced calorie intake has focused primarily on completely starved patients with severe illness, such as those in the intensive care unit or with anorexia nervosa [25, 26]. Given that starvation is a component of severe illness, it is impossible to dissociate its effects from those of systemic illness. Several studies have assessed the isolated impact of relatively mild fasting on thyroidal metabolism in healthy people or animals; however, the varying results cast doubt on whether or what extent of calorie restriction causes NTIS in patients under non-severe conditions [27–30]. Furthermore, the present case displayed decreases in serum FT4 and TSH levels, both indicators of severe NTIS, despite the lack of severe starvation. These facts suggest the presence of an alternative pathway from TD to NTIS development other than decreased calorie intake. As a possible hypothesis, TD might directly cause NTIS by affecting peripheral thyroidal hormone metabolism. As a key cofactor in the tricarboxylic acid cycle, TD disrupts cellular energy metabolism to produce a severe energy deficiency in tissues [2]. On the other hand, alterations in peripheral thyroidal hormone metabolism play a pivotal role in NTIS development, which are mainly regulated by cellular deiodinases [17, 21]. Since the expression of deiodinases is controlled in response to cellular stress, including energy deficiency [31, 32], TD and ensuing cellular energy deficiency may contribute to NTIS development through peripheral metabolic changes. Further studies are needed to verify this notion.
In addition to the non-specific initial symptoms of TD, its undetectability in routine blood tests complicates early detection. Although decreased serum thiamine is an indicator of the disorder, its measurement requires a shaded and iced whole blood sample obtained independently following suspicion of TD. Furthermore, results may be delayed and not always reflect whole-body thiamine status [9]. Hence, there remains no established method to help clinicians suspect or detect the presence of TD at an early stage, resulting in considerable underdiagnosis. The present patient, however, had exhibited NTIS prior to other obvious symptoms of TD, which ultimately contributed to the diagnosis. Although it is not uncommon for patients with chronic kidney disease or maintenance HD to exhibit NTIS [33], his serum thyroidal marker changes were exacerbated by appetite loss and fully recovered with treatment, indicating that they were affected not by HD, but by TD. Furthermore, serum thyroidal markers are frequently assessed within usual screening tests for reduced liveliness, as in the present case. Larger studies will shed light on the possibility of NTIS as a useful indicator for early stage TD suspicion.
Brain MRI typically displays high signal intensities in FLAIR images around such structures as the aqueduct of the midbrain and thalami [10]. In the present case, however, abnormal MRI findings were detected only inside the thalami and with slightly weak signal intensity. Although WE does not always display abnormal MRI results, this weak and isolated finding may have reflected the mildness of his WE without apparent symptoms. Furthermore, very early stage WE was found to exhibit slight brain MRI findings [34], suggesting the possibility that his WE with NTIS was at a very early stage before the appearance of more pronounced MRI findings. The present case supports the usefulness of NTIS assessment for early WE detection.
In conclusion, the development and detection of NTIS may be an indicator of early TD in HD patients. TD should be suspected in HD patients displaying appetite loss or reduced liveliness, and especially in those exhibiting NTIS.
Acknowledgements
None.
Author contributions
DA drafted the article. YK, MH, KH, and YK revised the article critically for important intellectual content and gave final approval of the submitted version.
Funding
The authors received no specific funding for this work.
Data availability
Data are available upon request.
Declarations
Conflict of interest
The authors have declared that no conflict of interest exists.
Ethical approval
The present case report adhered to the Declaration of Helsinki.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Tylicki A, Łotowski Z, Siemieniuk M, Ratkiewicz A. Thiamine and selected thiamine antivitamins—biological activity and methods of synthesis. Biosci Rep. 2018;38(1):BSR20171148. doi: 10.1042/BSR20171148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Dhir S, Tarasenko M, Napoli E, Giulivi C. Neurological, psychiatric, and biochemical aspects of thiamine deficiency in children and adults. Front Psychiatry. 2019 doi: 10.3389/fpsyt.2019.00207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Hung SC, Hung SH, Tarng DC, Yang WC, Chen TW, Huang TP. Thiamine deficiency and unexplained encephalopathy in hemodialysis and peritoneal dialysis patients. Am J Kidney Dis. 2001;38(5):941–947. doi: 10.1053/ajkd.2001.28578. [DOI] [PubMed] [Google Scholar]
- 4.Bossola M, Di Stasio E, Viola A, Leo A, Carlomagno G, Monteburini T, et al. Dietary intake of trace elements, minerals, and vitamins of patients on chronic hemodialysis. Int Urol Nephrol. 2014;46(4):809–815. doi: 10.1007/s11255-014-0689-y. [DOI] [PubMed] [Google Scholar]
- 5.Bukhari FJ, Moradi H, Gollapudi P, Ju Kim H, Vaziri ND, Said HM. Effect of chronic kidney disease on the expression of thiamin and folic acid transporters. Nephrol Dial Transpl. 2011;26(7):2137–2144. doi: 10.1093/ndt/gfq675. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Vetreno RP, Ramos RL, Anzalone S, Savage LM. Brain and behavioral pathology in an animal model of Wernicke's encephalopathy and Wernicke-Korsakoff Syndrome. Brain Res. 2012;1436:178–192. doi: 10.1016/j.brainres.2011.11.038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Ebels EJ. Underlying illness in Wernicke's encephalopathy. Analysis of possible causes of under-diagnosis. Eur Neurol. 1974;12(4):226–228. doi: 10.1159/000114622. [DOI] [PubMed] [Google Scholar]
- 8.Ihara M, Ito T, Yanagihara C, Nishimura Y. Wernicke's encephalopathy associated with hemodialysis: report of two cases and review of the literature. Clin Neurol Neurosurg. 1999;101(2):118–121. doi: 10.1016/s0303-8467(99)00014-1. [DOI] [PubMed] [Google Scholar]
- 9.Sriram K, Manzanares W, Joseph K. Thiamine in nutrition therapy. Nutr Clin Pract. 2012;27(1):41–50. doi: 10.1177/0884533611426149. [DOI] [PubMed] [Google Scholar]
- 10.Sechi G, Serra A. Wernicke's encephalopathy: new clinical settings and recent advances in diagnosis and management. Lancet Neurol. 2007;6(5):442–455. doi: 10.1016/s1474-4422(07)70104-7. [DOI] [PubMed] [Google Scholar]
- 11.Bossola M, Giungi S, Luciani G, Tazza L. Appetite in chronic hemodialysis patients: a longitudinal study. J Ren Nutr. 2009;19(5):372–379. doi: 10.1053/j.jrn.2009.01.015. [DOI] [PubMed] [Google Scholar]
- 12.Bossola M, Tazza L, Giungi S, Luciani G. Anorexia in hemodialysis patients: an update. Kidney Int. 2006;70(3):417–422. doi: 10.1038/sj.ki.5001572. [DOI] [PubMed] [Google Scholar]
- 13.Kaptein EM, Robinson WJ, Grieb DA, Nicoloff JT. Peripheral serum thyroxine, triiodothyronine and reverse triiodothyronine kinetics in the low thyroxine state of acute nonthyroidal illnesses. A noncompartmental analysis. J Clin Invest. 1982;69(3):526–535. doi: 10.1172/jci110478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Fliers E, Boelen A. An update on non-thyroidal illness syndrome. J Endocrinol Invest. 2021;44(8):1597–1607. doi: 10.1007/s40618-020-01482-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kanda E, Kato A, Masakane I, Kanno Y. A new nutritional risk index for predicting mortality in hemodialysis patients: Nationwide cohort study. PLoS ONE. 2019;14(3):e0214524. doi: 10.1371/journal.pone.0214524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Yamada K, Furuya R, Takita T, Maruyama Y, Yamaguchi Y, Ohkawa S, et al. Simplified nutritional screening tools for patients on maintenance hemodialysis. Am J Clin Nutr. 2008;87(1):106–113. doi: 10.1093/ajcn/87.1.106. [DOI] [PubMed] [Google Scholar]
- 17.Wajner SM, Maia AL. New insights toward the acute non-thyroidal illness syndrome. Front Endocrinol (Lausanne). 2012;3:8. doi: 10.3389/fendo.2012.00008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Fragidis S, Sombolos K, Thodis E, Panagoutsos S, Mourvati E, Pikilidou M, et al. Low T3 syndrome and long-term mortality in chronic hemodialysis patients. World J Nephrol. 2015;4(3):415–422. doi: 10.5527/wjn.v4.i3.415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Van den Berghe G. Dynamic neuroendocrine responses to critical illness. Front Neuroendocrinol. 2002;23(4):370–391. doi: 10.1016/S0091-3022(02)00006-7. [DOI] [PubMed] [Google Scholar]
- 20.Pappa TA, Vagenakis AG, Alevizaki M. The nonthyroidal illness syndrome in the non-critically ill patient. Eur J Clin Invest. 2011;41(2):212–220. doi: 10.1111/j.1365-2362.2010.02395.x. [DOI] [PubMed] [Google Scholar]
- 21.Warner MH, Beckett GJ. Mechanisms behind the non-thyroidal illness syndrome: an update. J Endocrinol. 2010;205(1):1–13. doi: 10.1677/JOE-09-0412. [DOI] [PubMed] [Google Scholar]
- 22.Docter R, Krenning EP, de Jong M, Hennemann G. The sick euthyroid syndrome: changes in thyroid hormone serum parameters and hormone metabolism. Clin Endocrinol (Oxf) 1993;39(5):499–518. doi: 10.1111/j.1365-2265.1993.tb02401.x. [DOI] [PubMed] [Google Scholar]
- 23.Jankowska M, Rudnicki-Velasquez P, Storoniak H, Rutkowski P, Rutkowski B, Krzymiński K, et al. Thiamine diphosphate status and dialysis-related losses in end-stage kidney disease patients treated with hemodialysis. Blood Purif. 2017;44(4):294–300. doi: 10.1159/000480651. [DOI] [PubMed] [Google Scholar]
- 24.Boelen A, Wiersinga WM, Fliers E. Fasting-induced changes in the hypothalamus-pituitary-thyroid axis. Thyroid. 2008;18(2):123–129. doi: 10.1089/thy.2007.0253. [DOI] [PubMed] [Google Scholar]
- 25.Langouche L, Vander Perre S, Marques M, Boelen A, Wouters PJ, Casaer MP, et al. Impact of early nutrient restriction during critical illness on the nonthyroidal illness syndrome and its relation with outcome: a randomized, controlled clinical study. J Clin Endocrinol Metab. 2013;98(3):1006–1013. doi: 10.1210/jc.2012-2809. [DOI] [PubMed] [Google Scholar]
- 26.Croxson MS, Ibbertson HK. Low serum triiodothyronine (T3) and hypothyroidism in anorexia nervosa. J Clin Endocrinol Metab. 1977;44(1):167–174. doi: 10.1210/jcem-44-1-167. [DOI] [PubMed] [Google Scholar]
- 27.Gardner DF, Kaplan MM, Stanley CA, Utiger RD. Effect of tri-iodothyronine replacement on the metabolic and pituitary responses to starvation. N Engl J Med. 1979;300(11):579–584. doi: 10.1056/nejm197903153001102. [DOI] [PubMed] [Google Scholar]
- 28.Sulimani RA. The effects of Ramadan fasting on thyroid functions in healthy male subjects. Nutr Res. 1988;8(5):549–552. doi: 10.1016/S0271-5317(88)80076-9. [DOI] [Google Scholar]
- 29.Ahmadinejad Z, Ziaee V, Rezaee M, Yarmohammadi L, Shaikh H, Bozorgi F. The effect of Ramadan fasting on thyroid hormone profile: a cohort study. Pak J Biol Sci. 2006;9(10):1999–2002. doi: 10.3923/pjbs.2006.1999.2002. [DOI] [Google Scholar]
- 30.de Vries EM, van Beeren HC, Ackermans MT, Kalsbeek A, Fliers E, Boelen A. Differential effects of fasting vs food restriction on liver thyroid hormone metabolism in male rats. J Endocrinol. 2015;224(1):25–35. doi: 10.1530/JOE-14-0533. [DOI] [PubMed] [Google Scholar]
- 31.Boelen A, van Beeren M, Vos X, Surovtseva O, Belegri E, Saaltink DJ, et al. Leptin administration restores the fasting-induced increase of hepatic type 3 deiodinase expression in mice. Thyroid. 2012;22(2):192–199. doi: 10.1089/thy.2011.0289. [DOI] [PubMed] [Google Scholar]
- 32.De Andrade P, Neff L, Strosova M, Arsenijevic D, Patthey-Vuadens O, Scapozza L, et al. Caloric restriction induces energy-sparing alterations in skeletal muscle contraction, fiber composition and local thyroid hormone metabolism that persist during catch-up fat upon refeeding. Front Physiol. 2015 doi: 10.3389/fphys.2015.00254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Kaptein EM. Thyroid hormone metabolism and thyroid diseases in chronic renal failure. Endocr Rev. 1996;17(1):45–63. doi: 10.1210/edrv-17-1-45. [DOI] [PubMed] [Google Scholar]
- 34.Aomura D, Kurasawa Y, Harada M, Hashimoto K, Kamijo Y. Brain MRI detection of early Wernicke's encephalopathy in a hemodialysis patient. 2022;10(3):e05539. 10.1002/ccr3.5539. [DOI] [PMC free article] [PubMed]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Data are available upon request.